Abstract
Elevated atmospheric CO2 concentrations (eCO2) regulate plant architecture and susceptibility to insects. We explored the mechanisms underpinning these responses in wild type (WT) peas and mutants defective in either strigolactone (SL) synthesis or signaling. All genotypes had increased shoot height and branching, dry weights and carbohydrate levels under eCO2, demonstrating that SLs are not required for shoot acclimation to eCO2. Since shoot levels of jasmonic acid (JA) and salicylic acid (SA) tended to be lower in SL signaling mutants than the WT under ambient conditions, we compared pea aphid performance on these lines under both CO2 conditions. Aphid fecundity was increased in the SL mutants compared to the WT under both ambient and eCO2 conditions. Aphid infestation significantly decreased levels of JA, isopentenyladenine, trans-zeatin and gibberellin A4 and increased ethylene precursor ACC, gibberellin A1, gibberellic acid (GA3) and SA accumulation in all lines. However, GA3 levels were increased less in the SL signaling mutants than the WT. These studies provide new insights into phytohormone responses in this specific aphid/host interaction and suggest that SLs and gibberellins are part of the network of phytohormones that participate in host susceptibility.
1. Introduction
Atmospheric carbon dioxide concentrations [CO2] are already 50% higher than before the industrial revolution and they are predicted to double again over the next 60 years [1]. Elevated atmospheric CO2 concentrations (eCO2) suppress photorespiration and increase photosynthetic carbon assimilation in C3 plants leading to substantial increases in carbon gain and crop productivity [2]. High CO2 also reduces stomatal apertures via the stomatal CO2 signal transduction pathways [3]. The CO2-dependent regulation of stomatal conductance restricts plant transpiration and enhances water use efficiency. As a result, the Earth has become greener over the last two decades as plants have produced more leaf area [4]. While these changes can be largely attributed to the regulation of photosynthesis, eCO2 has a strong impact on the physiology of C3 plants that extends far beyond photosynthesis and C metabolism [5]. For example, plants grown under eCO2 have lower concentrations of most mineral nutrients and this effect may diminish crop quality and nutrient cycling in terrestrial agro-ecosystems [6].
Atmospheric CO2 levels also exert a strong influence on the susceptibility of plants to pathogens and herbivores [2]. Insect herbivores are predicted to have an increasingly negative impact on crop production in the coming decades, particularly in African countries [7], and this will have serious implications for food security (Martinelli et al., 2015) [8]. However, current concepts of how eCO2 will influence plant susceptibility to phloem-feeding insects, such as aphids, remains controversial [9], not least because the literature data are variable [10,11]. Populations of the soybean aphid (Aphis glycines) were larger under eCO2, a finding that was related to a higher leaf temperature because of less open stomata [2]. While eCO2 had little impact on aphid performance in oilseed rape [12], pea aphids (Acyrthosiphon pisum) performed better under eCO2 on Medicago sativa [11], but worse on Vicia faba [13].
Aphids are important plant pests that often have a broad host range that significantly reduce the yields of susceptible plants via nutrient depletion, feeding damage to host tissues and the spread of viruses [14]. Aphids are phloem feeders with a probing stylet that wounds plant tissue as the insect searches for the phloem. The stylet secretes saliva that contains elicitors and proteins that modulate plant defensive responses [15]. Aphid feeding triggers the host multi-level immune system that operates to mitigate the adverse effects of pathogens and insects [16]. The host defensive networks that underpin plant responses to aphids have been characterized in a number of plant species [17,18,19,20]. Aphid feeding results in extensive changes to the leaf transcriptome and metabolome signatures [21], revealing a complex interplay between the different hormones regulating plant basal immunity. Changes in the levels of phytohormones such as jasmonic acid (JA), salicylic acid (SA), abscisic acid (ABA) and indole acetic acid (IAA) have been documented [21,22]. However, little attention has been paid to the role of strigolactones (SL) in plant–aphid interactions. SLs fulfil many important roles in the control of plant growth and architecture, seed dormancy and senescence as well as abiotic stress tolerance [23,24,25]. These carotenoid-derived phytohormones are also critical regulators of plant–microbe interactions in the rhizosphere, such as the symbiosis with arbuscular mycorrhizal fungi [26]. They also serve functions in plant responses to biotic stresses as a result of bacterial and fungal pathogens [27,28,29]. Growth under eCO2 increased the resistance of two M. truncatula genotypes to pea aphids by increasing salicylic acid (SA)-dependent defenses and decreasing jasmonic acid (JA) and ethylene-dependent signaling pathways, as well as increasing the density of non-glandular and glandular trichomes [10].
The pea aphid, Acyrthosiphon pisum, can only colonize plant species in the family Fabaceae, but can manipulate host defense signaling networks to increase infestation. Better performance of A pisum on their native hosts was not attributed to variation in ABA levels (which were down-regulated in all aphid clone-plant combinations), but instead modulation of the SA- and JA-defense signaling pathways [30]. However, the importance of SLs in aphid–plant compatibility or in shoot responses to eCO2 have not been characterized. We, therefore, compared shoot architecture and the fecundity of A. pisum in wild type peas and different pea ramosus mutants (rms) [31] under ambient and eCO2 conditions. Specifically, we examined responses to eCO2 and aphids in mutants that are deficient in either SL synthesis (rms1-2 and rms5-3) or SL signaling (rms3-1 and rms4-1). Since SL-deficient mutants have lower levels of the defense-related hormones JA and SA [29], we hypothesized that aphid performance would be enhanced on these genotypes.
2. Results
Wild type plants, the SL synthesis mutants, rms1-2 and rms5-3, and the SL signaling mutants, rms3-1 and rms4-1, were grown in air or eCO2 for up to 32 days after germination. The wild type shoots were significantly taller grown under eCO2 than those grown in air after 14 and 32 days of growth (Figure 1). While eCO2 tended to increase stem height in the SL mutant lines, the effect was only significant in rms3-1 (Figure 1; Day 32).
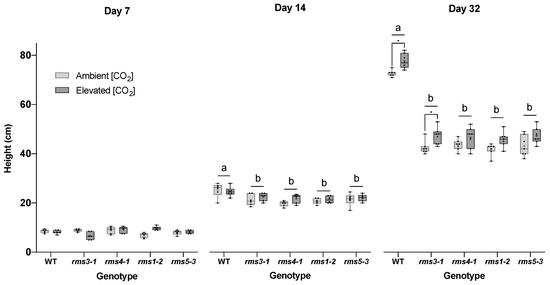
Figure 1.
The effect of high CO2 on plant height in wild type peas and mutants defective either in strigolactone (SL) synthesis (rms1-2 and rms5-3) or signaling (rms3-1 and rms4-1). Plants were grown for up to 32 days either under ambient (420 ppm) or high CO2 (750 ppm). Different letters indicate a statistically significant difference (p < 0.05) between genotype at each time point, while a significant difference between [CO2] within genotype is indicated by an asterisk (p < 0.05). Day 7 and day 14, n = 6; day 32, n = 7. Mean indicated as +.
The SL-defective mutants were significantly more branched than the wild type from day 14 after sowing onwards (Figure 2). Growth under eCO2 significantly increased the branching of the wild type shoots at day 32 (Figure 2). While eCO2 tended to increase shoot branching in all the mutant lines, the effect of eCO2 was only significant in rms1-2 (Figure 2; Day 32).

Figure 2.
The effect of high CO2 on the branching of the shoots of wild type peas and mutants defective either in strigolactone (SL) synthesis (rms1-2 and rms5-3) or signaling (rms3-1 and rms4-1). Plants were grown for up to 32 days either under ambient (420 ppm) or high CO2 (750 ppm). Different letters indicate a statistically significant difference (p ≤ 0.05) between genotype at each time point, while a significant difference between [CO2] within genotype is indicated as: * p < 0.05; *** p < 0.001. Day 7 and day 14, n = 6; day 32, n = 7. Mean indicated as +.
Growth under eCO2 significantly decreased the fresh weight/dry weight ratios of the wild type shoots (Figure 3). This effect was absent from the SL-defective mutants, which had significantly higher fresh weight/dry weight ratios than the wild type 28 days after sowing under both growth conditions (Figure 3A). Growth under eCO2 significantly increased shoot dry weight of all lines (Figure 3B).

Figure 3.
The effect of high CO2 on the fresh weight/dry weight ratios (A) and dry weights (B) of wild type peas and mutants defective either in strigolactone (SL) synthesis (rms1-2 and rms5-3) or signaling (rms3-1 and rms4-1). Plants were grown for 28 days (fresh weight/dry weight) or 32 days (dry weights) either under ambient (420 ppm CO2) or high CO2 (750 ppm). Different letters indicate a statistically significant difference (p < 0.05) between genotype at each time point, while a significant difference between [CO2] within genotype is indicated as: * p < 0.05; ** p < 0.01 (n = 5). Mean indicated as +.
All the lines had similar levels of leaf glucose, fructose, sucrose and starch when plants were grown in air (Figure 4). Growth under eCO2 significantly increased the levels of sucrose and starch in all the lines (Figure 4). While eCO2 also tended to increase the levels of leaf hexoses (glucose and fructose), this effect was only significant in some lines (Figure 4).

Figure 4.
The effect of high CO2 on the levels of glucose, fructose, sucrose and starch in wild type peas and mutants defective either in strigolactone (SL) synthesis (rms1-2 and rms5-3) or signaling (rms3-1 and rms4-1). Plants were grown for 28 days either under ambient (420 ppm CO2; bottom row) or high (750 ppm CO2; top row). Data shown as mean ± SD of three replicates. Significant differences between [CO2] within genotype are indicated as: * p <0.05; ** p < 0.01; *** p < 0.001; **** p < 0.0001.
Aphid fecundity was increased in the SL mutants compared to the wild type plants grown under ambient CO2 conditions (Figure 5). However, growth under eCO2 had no effect on aphid fecundity (Supplemental Figure S1).
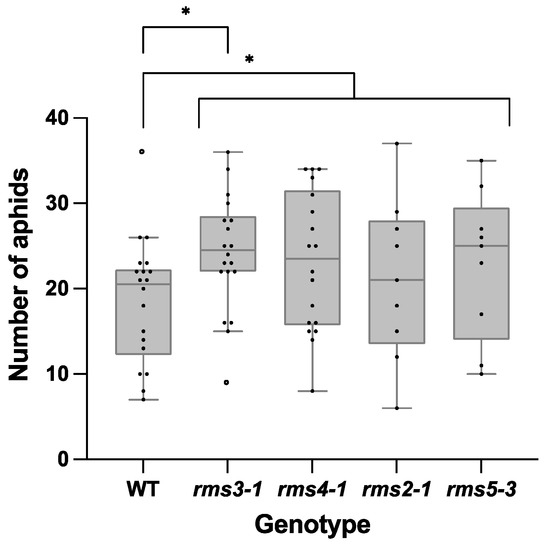
Figure 5.
Aphid fecundity on wild type peas and mutants defective either in strigolactone (SL) synthesis (rms1-2 and rms5-3) or signaling (rms3-1 and rms4-1) grown under ambient CO2 conditions. A single pea aphid nymph was placed on each 5-day-old plant. Plants were then grown in air for 15 days before aphid numbers were counted. Significant differences between genotypes are indicated as: * p < 0.05. Strigolactone mutants as a group were also significantly different from the wildtype. Wildtype (WT) and rms3-1, n = 19; rms4-1, n = 18; rms2-1 and rms5-3, n = 9.
Phytohormone levels were determined in the shoots of plants grown under ambient or eCO2 conditions. Since no significant effects of eCO2 on the levels of measured phytohormones were detected, we compared phytohormone levels in the absence and presence of aphids under eCO2 conditions (Figure 6). Without aphid exposure, the SL mutants had lower levels of JA, SA and gibberellic acid (GA3) than the wild type under eCO2 conditions.
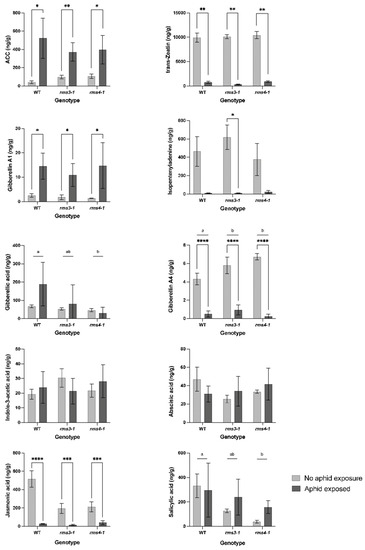
Figure 6.
The effect aphid infestation on the levels of different phytohormones in wild type peas and mutants defective in strigolactone signaling (rms3-1 and rms4-1) under high CO2 growth conditions. Data shown as mean ± SD (n = 3). Different letters indicate a statistically significant difference (p < 0.05) between genotype means (aphid-exposed and non-exposed grouped), while a significant difference between aphid exposure groups within genotype is indicated as: * p < 0.05; ** p < 0.01; *** p < 0.001; **** p < 0.0001.
Aphid infestation changed shoot concentrations of different phytohormones (Figure 6). The presence of aphids had no effect on SA accumulation in the shoots, but the levels of JA, isopentenyladenine, trans-zeatin and gibberellin GA4 were significantly decreased (Figure 6). In contrast, the levels of the ethylene precursor, 1-aminocyclopropane-1-carboxylic acid (ACC) and GA1 were increased after aphid infestation in all lines. Aphid exposure had no statistically significant effect on GA3 levels and there were no significant differences in GA3 accumulation between WT non-exposed and WT aphid-exposed plants. However, there was differential GA3 accumulation between the genotypes as a result of aphid exposure, which explained 18% of the variability in fecundity (Figure 7).
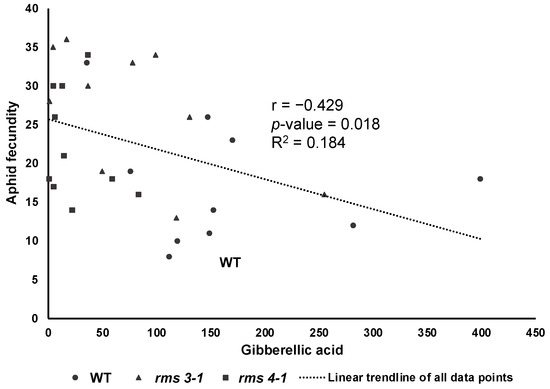
Figure 7.
The number of aphids on each plant was significantly correlated (p = 0.018) with shoot gibberellic acid concentration (ng/g DW) with a single relationship explaining variation across all genotypes (n = 30, n = 10 per genotype).
3. Discussion
Understanding the role of plant immunity in host susceptibility to aphids is essential to generate durable and sustainable aphid control strategies. The infestation of the pea shoots by the specialist pea aphid not only substantially decreased levels of the key defense hormone JA, but also isopentenyladenine and trans-zeatin, which are components of cytokinin (CK) metabolism. CK signaling pathways play a role in plant resistance by regulating SA-dependent defenses, affecting lignification and inducing protective proteins and phytoalexins [32,33]. Taken together, the findings presented here demonstrate that the pea aphids interact with host plants at the molecular level and successfully suppress plant defenses. In these studies, we tested the hypothesis that the performance of the pea aphid, A. pisum, would be enhanced on pea genotypes that were defective in SL signaling. The data presented here show that SL mutants are more susceptible to aphid infestation than the wild type plants. Moreover, the results presented here provide new information concerning how the pea aphid manipulates host defense networks to increase infestation. Aphid infestation was accompanied by large decreases in the levels of JA, isopentenyladenine, trans-zeatin and gibberellin A4 (Figure 6). While modulation of the SA- and JA-defense signaling pathways has previously been reported in this compatible aphid/host interaction [20], effects on gibberellin metabolism and signaling have not been reported previously.
The levels of JA and SA were decreased in the SL-deficient mutants relative to the wild type, suggesting that lack of SL synthesis and signaling impairs defense hormone accumulation. Although lower JA contents have previously been reported in the leaves of the Atmax mutants [34], SLs antagonized the JA pathway in rice roots [35]. The rice SL mutants were also less susceptible to Meloidogyne graminicola infection than the wild type plants [35] In addition, systemic-acquired resistance was increased in Arabidopsis SL mutants, and SA levels were lower in the max2 mutants than wild type [36]. The Arabidopsis SL mutants were more sensitive to infection by the biotrophic pathogen Pseudomonas syringae DC3000 [37]. SL-dependent modulation of AtACC4 expression has previously been reported [38], but no significant changes in ACC levels were observed in the pea SL mutants compared to the wild type. Taken together with the literature evidences, the data presented here demonstrate that SLs interact with defense hormones to regulate immunity and defenses against phloem-feeding insects. The levels of GA3 were lower in the SL mutants than the wild type, significantly so in the rms4 mutant. This finding may be related to the role of gibberellin signaling in the regulation of SL synthesis [39] (Ito et al., 2017). However, decreased GA3 levels have previously been reported in response to aphids [40]. Together, these data suggest that GA3 may thus be important in the defenses of pea plants against aphid infestation (Figure 7).
It is interesting that the presence of aphids also led to a large decrease in the levels of GA4, but increased GA3 and GA1 levels. The pea aphids, therefore, also regulate the synthesis and metabolism of these endogenous plant growth regulators. The finding that gibberellin levels were altered in the SL mutants may be significant in enhancing the ability of the aphids to infest the plants. The rms3-1 mutant is defective in the pea orthologue of the rice D14 SL receptor, which interacts with the GA signaling repressor, SLR1 [41]. Interactions between GA- and SL-mediated control of defense responses are, therefore, likely to be important in the ability of the aphids to infest pea plants. We are currently investigating whether SLs are important in the susceptibility of pea plants to infestation by the generalist (polyphagous) feeder Myzus persicae (green peach aphid), which can infest over 400 plant species. Since M. persicae can infest most dicotyledonous plants, it will be interesting to compare whether the generalist (polyphagous) feeder is also able to decrease the levels of phytohormones involved in pea growth and defense.
SLs regulate growth redistribution and repress shoot branching by rapidly modulating auxin transport [42]. The highly branched SL-deficient (rms1 and rms5) and SL signaling (rms3 and rms4) mutants are indicative of negative feedback control in which auxin up-regulates SL synthesis in an RMS2-dependent manner and SL down-regulates auxin synthesis in an RMS3- and RMS4-dependent manner [43]. The data presented in this study show that growth under eCO2 increased the branching of pea shoots in all genotypes, suggesting that SLs are not required for shoot responses to eCO2. The absence of a marked SL-dependent effect may be due to the large increase in soluble sugars that was found in all plants grown under eCO2 because sugars are known to suppress the auxin-induced SL pathway that promotes bud outgrowth [44]. However, the fresh weight/dry weight ratios were decreased by eCO2 in the wild type, but not in the shoots of the mutants, suggesting that SLs might participate in regulating shoot water content by modulating stomatal conductance. Although SL-deficient tomato and Arabidopsis plants are drought-sensitive because of their constitutively higher stomatal conductance [45], the rms pea mutants maintained similar stomatal conductance to WT plants under optimal conditions [46,47]. While further studies of stomatal response to eCO2 are necessary to understand these genotypic differences in shoot water status, we can conclude that SLs are not involved in growth regulation under eCO2.
In summary, while eCO2 has been predicted to have a major impact on plant susceptibility to herbivores, the data presented here shows that aphid–plant compatibility control of phytohormones is not changed by eCO2. However, we show that strigolactones and gibberellins are part of the network of phytohormones that regulate the susceptibility of peas to infestation by the pea aphid.
4. Materials and Methods
4.1. Biological Resources
Wild type pea (L107, Torsdag cultivar) seeds and mutants deficient in either SL synthesis mutants (rms1-2 and rms5-3) or SL signaling (rms3-1 and rms4-1) were provided by Professor Christine Beveridge. Stocks of the aphid Acyrthosiphon pisum were maintained on wild type pea at a 16-hour day length and 22 ± 3 °C.
4.2. Growth Conditions and Phenotypic Analysis
Seeds of the wild type, rms3-1, rms4-1, rms1-2 and rms5-3 pea mutants were allowed to germinate in vermiculite for 5 days. Thereafter, six seedlings of each genotype were transferred to Levington F2 compost (Evergreen Garden Care Ltd., Camberley, UK) in pots with a top diameter of 14 cm, 9.4-centimeter bottom diameter and a height of 12 cm equating to a pot volume of 1.3 L. These plants were used for experiments 28 days or less in duration. When plants were grown for 32 days, seven seedlings were planted in pots with an 18-centimeter top diameter, 13-centimeter bottom diameter, a height of 16.5 cm and volume of 3.14 L. Plants were grown in Microclima (Snijders Labs, Tilburg, Netherlands) controlled environment chambers at 21 °C, 14 h/10-day/night regime, 70% humidity and either at ambient CO2 (420 ± 20 ppm CO2) or eCO2 (750 ± 50 ppm CO2). Plant height and branching were determined at days 7, 14 and 32 after transplanting. Height was determined by measuring the stem from the soil to the apical shoot. Total branching was calculated as the sum of the height and length of each branch, not including branches shorter than 5 mm. Fresh and dry plant weights were determined at 28 days after transplanting, while dry weight was determined at 32 days.
4.3. Leaf Carbohydrate Measurements
The youngest mature leaves were harvested from 28-day-old plants grown in ambient and eCO2 for 28 days. Leaf carbohydrates were determined spectrophotometrically, as described by Lunn and Hatch (1995) [48]. Briefly, sugars were extracted from 0.1 g of leaf tissue with hot 80% ethanol and centrifuged. Reducing sugars (glucose, fructose and sucrose) were recovered from supernatant and starch was extracted from the pellet. Soluble sugars were measured by the increase in absorbance at 340 nm as NADPH was produced in coupled enzymatic assays using glucose-6-phosphate dehydrogenase, hexokinase, phosphoglucose isomerase and invertase.
4.4. Aphid Fecundity
Wild type (WT), rms3-1 and rms4-1 pea mutants were germinated and transplanted in the same media and growth conditions as described above. In order to obtain A. pisum nymphs of the same age, adult A. pisum were placed on WT pea for 24 h and allowed to produce nymphs. One of these nymphs (foundress) was then placed on a 5-day-old plant from each of the genotypes, directly after transplantation. The aphids were contained on the pea plants using 07ZL flowerpot cages (Insectopia, Austrey, United Kingdom) and allowed to reproduce for 15 days. Ten replicates were performed for each pea genotype at either ambient or elevated CO2. The number of aphids present on each plant was then determined.
4.5. Phytohormone Levels
Entire plant shoots (comprising leaves, stipules and stems) that had been infected with A. pisum were harvested. Metabolism was immediately stopped by immersion in liquid nitrogen. Samples were stored at −80 °C before freeze drying. Five replicates per line (wild type, rms3-1 and rms4-1 mutant pea) were analysed for cytokinins (trans-zeatin, tZ, zeatin riboside, ZR, and isopentenyl adenine, iP), gibberellins (GA1, GA3{gibberellic acid} and GA4), indole-3-acetic acid (IAA), abscisic acid (ABA), salicylic acid (SA), jasmonic acid (JA) and the ethylene precursor 1-aminocyclopropane-1-carboxylic aid (ACC), according to Albacete et al., (2008) [49] with some modifications, as described by Castro-Valdecantos et al., (2021) [50].
4.6. Statistical Analysis
All datasets to be analysed were firstly subjected to a D’Agostino–Pearson normality test as well as Spearman’s test to determine if variances between variables were heteroscedastic (Table S1). A Grubbs test was used to test for outliers within the aphid fecundity dataset. Thereafter, a two-way ANOVA was performed on datasets conforming to a Gaussian distribution and which were homoscedastic (Table S2). If an independent variable ([CO2], genotype or aphid exposure) had a significant effect on the response (p < 0.05), Tukey’s multiple comparison test was used to compare means between genotypes (Table S3), while Sidak’s multiple comparison test was used to compare [CO2] (Table S4) or aphid exposure (Table S5) within genotype. Datasets which were normally distributed, but heteroscedastic, were analysed with a Welch ANOVA (Table S2) test followed by Dunnett’s T3 multiple comparisons test to compare genotype (Table S3) means or Holm–Sidak’s multiple comparisons test to compare [CO2] (Table S4) or aphid exposure (Table S5) within genotype. Log transformations were used [Y = Log(Y)] if the transformation resulted in a normal distribution of the non-normally distributed datasets (Table S1). When transformation did not work, a Kruskal–Wallis test was used to determine if genotype groups had a significant effect on the response measured, while in the case of [CO2] or aphid exposure, a Mann–Whitney test was used (Table S2). Following a significant p-value computed by the Kruskal–Wallis test, multiple comparisons were performed using Dunn’s procedure (Table S3), while a significant effect determined by the Mann–Whitney test was followed by a Holm–Sidak’s multiple comparisons test (Tables S4 and S5). Pearson correlation coefficients (r) and coefficients of determination (R2) were determined between aphid numbers and all the phytohormone concentrations of the plants on which the aphids have fed (Table S6). Graphpad Prism version 9.4.0 was used to perform all statistical analyses apart from the Grubbs test and correlation analysis, which were performed in XLSTAT version 2021.4.1.1185.
Supplementary Materials
The following supporting information can be downloaded at: https://www.mdpi.com/article/10.3390/ijms232012160/s1.
Author Contributions
H.W.S., B.K. and Y.H. undertook all the experimental work. H.W.S. analysed all the data, prepared the figures for the manuscript and assisted with review and editing. I.C.D. oversaw the studies on phytohormones and helped to prepare the manuscript. A.-M.B. co-supervised. H.W.S. discussed the data and helped to prepare the manuscript. C.H.F. had the original concept, designed the studies, discussed the data and wrote the manuscript. All authors have read and agreed to the published version of the manuscript.
Funding
This research was supported by an N8 Agrifood pump-priming award and a Commonwealth Scholarship (ZACN-2018-353) funded by the UK government. The APC was funded by the journal.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
All data are available upon request to the authors.
Acknowledgments
We thank Christine Beveridge and Stacey Cook of the University of Queensland for supplying the seed stocks for the different strigloactone mutant lines. We also thank Alfonso Albacete and María del Puerto Sánchez-Iglesias for their technical support on hormonal analysis, and N8 Agrifood for funds to foster the collaboration between ICD and CHF. HS is a Commonwealth Scholar funded by the UK government.
Conflicts of Interest
There are no conflicts of interest.
References
- Beerling, D.J.; Kantzas, E.P.; Lomas, M.R.; Wade, P.; Eufrasio, R.M.; Renforth, P.; Sarkar, B.; Andrews, M.G.; James, R.H.; Pearce, C.R.; et al. Potential for large-scale CO2 removal via enhanced rock weathering with croplands. Nature 2020, 583, 242–248. [Google Scholar] [CrossRef] [PubMed]
- Ainsworth, E.A.; Long, S. 30 years of free-air carbon dioxide enrichment (FACE): What have we learned about future crop productivity and its potential for adaptation? Glob. Change Biol. 2021, 27, 27–49. [Google Scholar] [CrossRef]
- Zhang, J.; De Oliveira-Cecilato, P.; Takahashi, Y.; Schulze, S.; Dubeaux, G.; Hauser, F.; Azoulay-Shaemwe, T.; Tõldsepp, K.; Kollist, H.; Rappel, W.-J.; et al. Insights into the molecular mechanisms of CO2-mediated regulation of stomatal movements. Curr. Biol. 2018, 28, R1356–R1363. [Google Scholar] [CrossRef] [PubMed]
- Winkler, A.J.; Myneni, R.B.; Hannart, A.; Sitch, S.; Haverd, V.; Lombardozzi, D.; Arora, V.K.; Pongratz, J.; Nabel, J.E.M.S.; Goll, D.S.; et al. Slowdown of the greening trend in natural vegetation with further rise in atmospheric CO2. Biogeosciences 2021, 18, 4985–5010. [Google Scholar] [CrossRef]
- Foyer, C.H.; Noctor, G. Redox homeostasis and signaling in a higher-CO2 world. Annu. Rev. Plant Biol. 2020, 71, 157–182. [Google Scholar] [CrossRef]
- Zhu, C.; Kobayashi, K.; Loladze, I.; Zhu, J.; Jiang, Q.; Xu, X.; Liu, G.; Seneweera, S.; Ebi, K.L.; Ziska, L.H. Carbon dioxide (CO2) levels this century will alter the protein, micronutrients, and vitamin content of rice grains with potential health consequences for the poorest rice-dependent countries. Sci. Adv. 2018, 4, eaaq1012. [Google Scholar] [CrossRef]
- Botha, A.-M.; Kunert, K.J.; Maling’a, J.; Foyer, C.H. Defining biotechnological solutions for insect control in sub-Saharan Africa. Food Energy Secur. 2020, 9, e191. [Google Scholar] [CrossRef]
- Martinelli, F.; Scalenghe, R.; Davino, S.; Panno, S.; Scuderi, G.; Ruisi, P.; Villa, P.; Stroppiana, D.; Boschetti, M.; Goulart, L.R.; et al. Advanced methods of plant disease detection. Agron. Sustain. Dev. 2015, 35, 1–25. [Google Scholar] [CrossRef]
- Sun, Y.; Ge, F. How do aphids respond to elevated CO2? J. Asia-Pac. Entomol. 2011, 14, 217–220. [Google Scholar] [CrossRef]
- Guo, H.J.; Sun, Y.C.; Li, Y.F.; Liu, X.H.; Wang, P.Y.; Zhu-Salzman, K.; Ge, F. Elevated CO2 alters the feeding behaviour of the pea aphid by modifying the physical and chemical resistance of Medicago truncatula. Plant Cell Environ. 2014, 37, 2158–2168. [Google Scholar] [CrossRef]
- Ryalls, J.M.W.; Moore, B.D.; Riegler, M.; Bromfield, L.M.; Hall, A.A.G.; Johnson, S.N. Climate and atmospheric change impacts on sap-feeding herbivores: A mechanistic explanation based on functional groups of primary metabolites. Funct. Ecol. 2017, 31, 161–171. [Google Scholar] [CrossRef]
- Himanen, S.J.; Nissinen, A.; Dong, W.X.; Nerg, A.M.; Stewart, C.N.; Poppy, G.M.; Holopainen, J.K. Interactions of elevated carbon dioxide and temperature with aphid feeding on transgenic oilseed rape: Are Bacillus thuringiensis (Bt) plants more susceptible to non-target herbivores in future climate? Glob. Change Biol. 2008, 14, 1437–1454. [Google Scholar] [CrossRef]
- Mondor, E.B.; Tremblay, M.N.; Awmack, C.S.; Lindroth, R.L. Altered genotypic and phenotypic frequencies of aphid populations under enriched CO2 and O3 atmospheres. Glob. Change Biol. 2005, 11, 1990–1996. [Google Scholar] [CrossRef]
- Smith, C.M.; Chuang, W.P. Plant resistance to aphid feeding: Behavioral, physiological, genetic and molecular cues regulate aphid host selection and feeding. Pest Manag. Sci. 2014, 70, 528–540. [Google Scholar] [CrossRef]
- Kaloshian, I.; Walling, L.L. Hemipteran and dipteran pests: Effectors and plant host immune regulators. J. Integr. Plant Biol. 2016, 58, 350–361. [Google Scholar] [CrossRef] [PubMed]
- Zust, T.; Agrawal, A.A. Mechanisms and evolution of plant resistance to aphids. Nat. Plants 2016, 2, 15206. [Google Scholar] [CrossRef]
- Kiani, M.; Szczepaniec, A. Effects of sugarcane aphid herbivory on transcriptional responses of resistant and susceptible sorghum. BMC Genom. 2018, 19, 774. [Google Scholar] [CrossRef]
- Koch, K.G.; Palmer, N.; Donze, T.; Scully, E.D.; Seravalli, J.; Amundsen, K.; Twigg, P.; Louis, J.; Bradshaw, J.D.; Heng-Moss, T.-M.; et al. Aphid-responsive defense networks in hybrid switchgrass. Front. Plant Sci. 2020, 11, 1145. [Google Scholar] [CrossRef]
- Nguyen, D.; Rieu, I.; Mariani, C.; van Dam, N.M. How plants handle multiple stresses: Hormonal interactions underlying responses to abiotic stress and insect herbivory. Plant Mol. Biol. 2016, 91, 727–740. [Google Scholar] [CrossRef]
- Sanchez-Arcos, C.; Kai, M.; Svatos, A.; Gershenzon, J.; Kunert, G. Untargeted metabolomics approach reveals differences in host plant chemistry before and after infestation with different pea aphid host races. Front. Plant Sci. 2019, 10, 188. [Google Scholar] [CrossRef]
- Foyer, C.H.; Verrall, S.R.; Hancock, R.D. Systematic analysis of phloem-feeding insect induced transcriptional reprogramming in Arabidopsis highlights common features and reveals distinct responses to specialist and generalist insects. J. Exp. Bot. 2015, 66, 495–512. [Google Scholar] [CrossRef]
- Kerchev, P.I.; Karpińska, B.; Morris, J.A.; Hussain, A.; Verrall, S.R.; Hedley, P.E.; Fenton, B.; Foyer, C.H.; Hancock, R.D. Vitamin C and the abscisic acid-insensitive 4 (ABI4) transcription factor are important determinants of aphid resistance in Arabidopsis. Antioxid. Redox Signal. 2013, 18, 2091–2105. [Google Scholar] [CrossRef] [PubMed]
- Al-Babili, S.; Bouwmeester, H.J. Strigolactones, a novel carotenoid-derived plant hormone. Annu. Rev. Plant Biol. 2015, 66, 161–186. [Google Scholar] [CrossRef]
- Machin, D.C.; Bennett, T. Two routes to germinate a seed. Nat. Plants 2020, 6, 602–603. [Google Scholar] [CrossRef]
- Waters, M.T.; Gutjahr, C.; Bennett, T.; Nelson, D.C. Strigolactone signaling and evolution. Annu. Rev. Plant Biol. 2017, 68, 291–322. [Google Scholar] [CrossRef] [PubMed]
- Aliche, E.B.; Screpanti, C.; De Mesmaeker, A.; Munnik, T.; Bouwmeester, H.J. Science and application of strigolactones. New Phytol. 2020, 227, 1001–1011. [Google Scholar] [CrossRef] [PubMed]
- Marzec, M. Strigolactones as part of the plant defense system. Trends Plant Sci. 2016, 16, 30121–30122. [Google Scholar]
- Stes, E.; Depuydt, S.; De Keyser, A.; Matthys, C.; Audenaert, K.; Yoneyama, K. Strigolactones as an auxiliary hormonal defense mechanism against leafy gall syndrome in Arabidopsis thaliana. J. Exp. Bot. 2015, 66, 5123–5134. [Google Scholar] [CrossRef] [PubMed]
- Torres-Vera, R.; García, J.M.; Pozo, M.J.; López-Ráez, J.A. Do strigolactones contribute to plant defense? Mol. Plant Pathol. 2014, 15, 211–216. [Google Scholar] [CrossRef]
- Sanchez-Arcos, C.; Reichelt, M.; Gershenzon, J.; Kunert, G. Modulation of legume defense signaling pathways by native and non-native pea aphid clones. Front. Plant Sci. 2016, 7, 1872. [Google Scholar] [CrossRef]
- Johnson, X.; Brcich, T.; Dun, E.A.; Goussot, M.; Haurogné, K.; Beveridge, C.A.; Rameau, C. Branching genes are conserved across species. Genes controlling a novel signal in pea are coregulated by other long-distance signals. Plant Physiol. 2006, 142, 1014–1026. [Google Scholar] [CrossRef]
- Argueso, C.T.; Ferreira, F.J.; Epple, P.; To, J.P.C.; Hutchison, C.E.; Schaller, G.E.; Dangl, J.L.; Kieber, J.J. Two-component elements mediate interactions between cytokinin and salicylic acid in plant immunity. PLoS Genet. 2012, 8, e1002448. [Google Scholar] [CrossRef] [PubMed]
- Choi, J.; Huh, S.U.; Kojima, M.; Sakakibara, H.; Paek, K.H.; Hwang, I. The Cytokinin-activated transcription factor ARR2 promotes plant immunity via TGA3/NPR1-dependent salicylic acid signaling in Arabidopsis. Dev. Cell 2010, 19, 284–295. [Google Scholar] [CrossRef]
- Haq, B.U.; Ahmad, M.Z.; Ur Rehman, N.; Wang, J.; Li, P.; Li, D.; Zhao, J. Functional characterization of soybean strigolactone biosynthesis and signaling genes in Arabidopsis MAX mutants and GmMAX3 in soybean nodulation. BMC Plant Biol. 2017, 17, 259. [Google Scholar] [CrossRef]
- Lahari, Z.; Ullah, C.; Kyndt, T.; Gershenzon, J.; Gheysen, G. Strigolactones enhance root-knot nematode (Meloidogyne graminicola) infection in rice by antagonizing the jasmonate pathway. New Phytol. 2019, 224, 454–465. [Google Scholar] [CrossRef] [PubMed]
- Kusajima, M.; Fujita, M.; Soudthedlath, K.; Nakamura, H.; Yoneyama, K.; Nomura, T.; Akiyama, K.; Maruyama-Nakashita, A.; Asami, T.; Nakashita, H. Strigolactones modulate salicylic acid-mediated disease resistance in Arabidopsis thaliana. Int. J. Mol. Sci. 2022, 23, 5246. [Google Scholar] [CrossRef]
- Kalliola, M.; Jakobson, L.; Davidsson, P.; Pennanen, V.; Waszczak, C.; Yarmolinsky, D.; Zamora, O.; Palva, E.T.; Kariola, T.; Kollist, H.; et al. Differential role of MAX2 and strigolactones in pathogen, ozone, and stomatal responses. Plants Direct 2020, 4, 1–14. [Google Scholar] [CrossRef]
- Kumar, M.; Kim, I.; Kim, Y.K.; Heo, J.B.; Suh, M.C.; Kim, H.U. Strigolactone signaling genes showing differential expression patterns in Arabidopsis max mutants. Plants 2019, 8, 352. [Google Scholar] [CrossRef] [PubMed]
- Ito, S.; Yamagami, D.; Umehara, M.; Hanada, A.; Yoshida, S.; Sasaki, Y.; Yajima, S.; Kyozuka, J.; Ueguchi-Tanaka, M.; Matsuoka, M.; et al. Regulation of strigolactone biosynthesis by gibberellin signaling. Plant Physiol. 2017, 174, 1250–1259. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.; Liu, J.; Cui, K.; Chen, H.; Yang, Z.; Wu, H.; Shao, S.; King-Jones, K.; Chen, K. Gibberellic acid is selectively downregulated in response to aphid-induced gall formation. Acta Physiol. Plant. 2016, 38, 214. [Google Scholar] [CrossRef]
- Nakamura, H.; Xue, Y.L.; Miyakawa, T.; Hou, F.; Qin, H.M.; Fukui, K.; Shi, X.; Ito, E.; Ito, S.; Park, S.H.; et al. Molecular mechanism of strigolactone perception by DWARF14. Nat. Commun. 2013, 4, 2613. [Google Scholar] [CrossRef]
- Shinohara, N.; Taylor, C.; Leyser, O. Strigolactone can promote or inhibit shoot branching by triggering rapid depletion of the auxin efflux protein PIN1 from the plasma membrane. PLoS Biol. 2013, 11, e1001474. [Google Scholar] [CrossRef]
- Ligerot, Y.; de Saint Germain, A.; Waldie, T.; Troadec, C.; Citerne, S.; Kadakia, N.; Pillot, J.-P.; Prigge, M.; Aubert, G.; Bendahmane, A.; et al. The pea branching RMS2 gene encodes the PsAFB4/5 auxin receptor and is involved in an auxin-strigolactone regulation loop. PLoS Genet. 2017, 13, e1007089. [Google Scholar] [CrossRef]
- Bertheloot, J.; Barbier, F.; Boudon, F.; Perez-Garcia, M.D.; Peron, T.; Citerne, S.; Dun, E.; Beveridge, C.; Godin, C.; Sakr, S. Sugar availability suppresses the auxin-induced strigolactone pathway to promote bud outgrowth. New Phytol. 2020, 225, 866–879. [Google Scholar] [CrossRef] [PubMed]
- Visentin, I.; Vitali, M.; Ferrero, M.; Zhang, Y.; Ruyter-Spira, C.; Novák, O.; Strnad, M.; Lovisolo, C.; Schubert, A.; Cardinale, F. Low levels of strigolactones in roots as a component of the systemic signal of drought stress in tomato. New Phytol. 2016, 212, 954–963. [Google Scholar] [CrossRef] [PubMed]
- Cooper, J.W.; Beyyoudh, L.; Dasgan, H.Y.; Kunert, K.J.; Beveridge, C.; Foyer, C.H. Strigolactones positively regulate chilling tolerance in soybean, pea and Arabidopsis. Plant Cell Environ. 2018, 41, 1298–1310. [Google Scholar] [CrossRef]
- Dodd, I.C.; Ferguson, B.J.; Beveridge, C.A. Apical wilting and petiole xylem vessel diameter of the rms2 branching mutant of pea are shoot controlled and independent of a long-distance signal regulating branching. Plant Cell Physiol. 2008, 49, 791–800. [Google Scholar] [CrossRef] [PubMed]
- Lunn, J.E.; Hatch, M.D. Primary partitioning and storage of photosynthate in sucrose and starch in leaves of C4 plants. Planta 1995, 197, 385–391. [Google Scholar] [CrossRef]
- Albacete, A.; Ghanem, M.E.; Martínez-Andújar, C.; Acosta, M.; Sanchez-Bravo, J.; Martinez, V.; Lutts, S.; Dodd, I.C.; Pérez-Alfocea, F. Hormonal changes in relation to biomass partitioning and shoot growth impairment in salinised tomato (Solanum lycopersicum L.) plants. J. Exp. Bot. 2008, 59, 4119–4131. [Google Scholar] [CrossRef]
- Castro-Valdecantos, P.; Puértolas, J.; Albacete, A.; Dodd, I.C. Girdling changes root and shoot hormonal balance but does not alter drought-induced stomatal closure in soybean. Environ. Exp. Bot. 2021, 192, 104657. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).