PET and SPECT Imaging of the EGFR Family (RTK Class I) in Oncology
Abstract
1. Introduction
1.1. Human Epidermal Growth Factor Receptor Family in Cancer
1.2. Detection of EGFR-Family Expression for Targeted Treatment
1.3. Biopsy Based Methods
1.4. Radionuclide-Based Molecular Imaging
1.5. General Considerations for the Development of Radiotracers for PET and SPECT Imaging
2. This Review
3. HER1 Imaging
3.1. Monoclonal Antibodies for Imaging of HER1 Expression
3.2. Antibody Fragments, scFv and sdAb for Imaging of HER1 Expression
3.3. Affibody Molecules for Imaging of HER1 Expression
3.4. Small Peptides
3.5. HER1 Concluding Remarks
4. HER2 Imaging
4.1. Monoclonal Antibodies
4.2. Antibody Fragments for Imaging of HER2 Expression
4.3. scFv, Diabody, Minibody, sdAb for Imaging of HER2 Expression
4.4. ESPs for Imaging of HER2 Expression
4.5. Aptamers and Peptides
4.6. HER2 Concluding Remarks
5. HER3 Imaging
5.1. Monoclonal Antibodies and Antibody Fragments for Imaging of HER3 Expression
5.2. sdAb, Affibody Molecules and Peptides for Imaging of HER3 Expression
5.3. HER3 Concluding Remarks
6. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Conflicts of Interest
Abbreviations
| BC | Breast cancer |
| ECD | Extra cellular domain |
| EGFR | Epidermal growth factor receptor |
| ESP | Engineered scaffold proteins |
| HER | Human epidermal growth factor |
| ICD | Intra cellular domain |
| IHC | Immunohistochemistry |
| ISH | in situ hybridizations |
| mAb | Monoclonal antibody |
| mBC | Metastatic breast cancer |
| mCRC | Metastatic colorectal cancer |
| NSCLC | Non-small cell lung cancer |
| PC | Prostate cancer |
| PET | Positron emission Tomography |
| pi | post injection |
| RTK | Receptor tyrosine kinase |
| scFv | Single chain variable fragment |
| sdAb | Single domain antibody |
| SPECT | Single photon emission tomography |
References
- Appert-Collin, A.; Hubert, P.; Crémel, G.; Bennasroune, A. Role of ErbB Receptors in Cancer Cell Migration and Invasion. Front. Pharmacol. 2015, 6, 283. [Google Scholar] [CrossRef]
- Yarden, Y.; Sliwkowski, M.X. Untangling the ErbB Signalling Network. Nat. Rev. Mol. Cell. Biol. 2001, 2, 127–137. [Google Scholar] [CrossRef] [PubMed]
- Bouyain, S.; Longo, P.A.; Li, S.; Ferguson, K.M.; Leahy, D.J. The Extracellular Region of ErbB4 Adopts a Tethered Conformation in the Absence of Ligand. Proc. Natl. Acad. Sci. USA 2005, 102, 15024–15029. [Google Scholar] [CrossRef] [PubMed]
- Fuller, S.J.; Sivarajah, K.; Sugden, P.H. ErbB Receptors, Their Ligands, and the Consequences of Their Activation and Inhibition in the Myocardium. J. Mol. Cell. Cardiol. 2008, 44, 831–854. [Google Scholar] [CrossRef] [PubMed]
- Burgess, A.W.; Cho, H.-S.; Eigenbrot, C.; Ferguson, K.M.; Garrett, T.P.J.; Leahy, D.J.; Lemmon, M.A.; Sliwkowski, M.X.; Ward, C.W.; Yokoyama, S. An Open-and-Shut Case? Recent Insights into the Activation of EGF/ErbB Receptors. Mol. Cell 2003, 12, 541–552. [Google Scholar] [CrossRef]
- Jorissen, R.N.; Walker, F.; Pouliot, N.; Garrett, T.P.J.; Ward, C.W.; Burgess, A.W. Epidermal Growth Factor Receptor: Mechanisms of Activation and Signalling. Exp. Cell Res. 2003, 284, 31–53. [Google Scholar] [CrossRef]
- Cho, H.-S.; Leahy, D.J. Structure of the Extracellular Region of HER3 Reveals an Interdomain Tether. Science 2002, 297, 1330–1333. [Google Scholar] [CrossRef]
- Dawson, J.P.; Berger, M.B.; Lin, C.-C.; Schlessinger, J.; Lemmon, M.A.; Ferguson, K.M. Epidermal Growth Factor Receptor Dimerization and Activation Require Ligand-Induced Conformational Changes in the Dimer Interface. Mol. Cell. Biol. 2005, 25, 7734–7742. [Google Scholar] [CrossRef]
- Bennasroune, A.; Gardin, A.; Aunis, D.; Crémel, G.; Hubert, P. Tyrosine Kinase Receptors as Attractive Targets of Cancer Therapy. Crit. Rev. Oncol. Hematol. 2004, 50, 23–38. [Google Scholar] [CrossRef]
- Ullrich, A.; Schlessinger, J. Signal Transduction by Receptors with Tyrosine Kinase Activity. Cell 1990, 61, 203–212. [Google Scholar] [CrossRef]
- Mishra, R.; Patel, H.; Alanazi, S.; Yuan, L.; Garrett, J.T. HER3 Signaling and Targeted Therapy in Cancer. Oncol. Rev. 2018, 12, 355. [Google Scholar] [CrossRef]
- Wang, Z. ErbB Receptors and Cancer. Methods Mol. Biol. 2017, 1652, 3–35. [Google Scholar] [CrossRef] [PubMed]
- Harris, R.C.; Chung, E.; Coffey, R.J. EGF Receptor Ligands. Exp. Cell Res. 2003, 284, 2–13. [Google Scholar] [CrossRef]
- Mujoo, K.; Choi, B.-K.; Huang, Z.; Zhang, N.; An, Z. Regulation of ERBB3/HER3 Signaling in Cancer. Oncotarget 2014, 5, 10222–10236. [Google Scholar] [CrossRef] [PubMed]
- Yarden, Y. The EGFR Family and Its Ligands in Human Cancer. Signalling Mechanisms and Therapeutic Opportunities. Eur. J. Cancer 2001, 37 (Suppl. 4), S3–S8. [Google Scholar] [CrossRef]
- Mota, J.M.; Collier, K.A.; Barros Costa, R.L.; Taxter, T.; Kalyan, A.; Leite, C.A.; Chae, Y.K.; Giles, F.J.; Carneiro, B.A. A Comprehensive Review of Heregulins, HER3, and HER4 as Potential Therapeutic Targets in Cancer. Oncotarget 2017, 8, 89284–89306. [Google Scholar] [CrossRef]
- Kumar, R.; George, B.; Campbell, M.R.; Verma, N.; Paul, A.M.; Melo-Alvim, C.; Ribeiro, L.; Pillai, M.R.; da Costa, L.M.; Moasser, M.M. Chapter Three—HER family in cancer progression: From discovery to 2020 and beyond. In Advances in Cancer Research; Kumar, R., Fisher, P.B., Eds.; Receptor Tyrosine Kinases; Academic Press: Cambridge, MA, USA, 2020; Volume 147, pp. 109–160. [Google Scholar]
- Hynes, N.E.; Horsch, K.; Olayioye, M.A.; Badache, A. The ErbB Receptor Tyrosine Family as Signal Integrators. Endocr. Relat. Cancer 2001, 8, 151–159. [Google Scholar] [CrossRef]
- Yarden, Y.; Pines, G. The ERBB Network: At Last, Cancer Therapy Meets Systems Biology. Nat. Rev. Cancer 2012, 12, 553–563. [Google Scholar] [CrossRef]
- Roskoski, R. The ErbB/HER Family of Protein-Tyrosine Kinases and Cancer. Pharmacol. Res. 2014, 79, 34–74. [Google Scholar] [CrossRef]
- Hendler, F.J.; Ozanne, B.W. Human Squamous Cell Lung Cancers Express Increased Epidermal Growth Factor Receptors. J. Clin. Investig. 1984, 74, 647–651. [Google Scholar] [CrossRef]
- Schechter, A.L.; Stern, D.F.; Vaidyanathan, L.; Decker, S.J.; Drebin, J.A.; Greene, M.I.; Weinberg, R.A. The Neu Oncogene: An Erb-B-Related Gene Encoding a 185,000-Mr Tumour Antigen. Nature 1984, 312, 513–516. [Google Scholar] [CrossRef]
- Slamon, D.J.; Clark, G.M.; Wong, S.G.; Levin, W.J.; Ullrich, A.; McGuire, W.L. Human Breast Cancer: Correlation of Relapse and Survival with Amplification of the HER-2/Neu Oncogene. Science 1987, 235, 177–182. [Google Scholar] [CrossRef]
- Thomas, R.; Weihua, Z. Rethink of EGFR in Cancer with Its Kinase Independent Function on Board. Front. Oncol. 2019, 9, 800. [Google Scholar] [CrossRef]
- Baselga, J. Treatment of HER2-Overexpressing Breast Cancer. Ann. Oncol. 2010, 21, vii36–vii40. [Google Scholar] [CrossRef]
- Li, X.; Zhao, H.; Gu, J.; Zheng, L. Prognostic Role of HER2 Amplification Based on Fluorescence in Situ Hybridization (FISH) in Pancreatic Ductal Adenocarcinoma (PDAC): A Meta-Analysis. World J. Surg. Oncol. 2016, 14, 38. [Google Scholar] [CrossRef][Green Version]
- Mishra, R.; Hanker, A.B.; Garrett, J.T. Genomic Alterations of ERBB Receptors in Cancer: Clinical Implications. Oncotarget 2017, 8, 114371–114392. [Google Scholar] [CrossRef]
- Day, K.C.; Hiles, G.L.; Kozminsky, M.; Dawsey, S.J.; Paul, A.; Broses, L.J.; Shah, R.; Kunja, L.P.; Hall, C.; Palanisamy, N.; et al. HER2 and EGFR Overexpression Support Metastatic Progression of Prostate Cancer to Bone. Cancer Res. 2017, 77, 74–85. [Google Scholar] [CrossRef]
- Kraus, M.H.; Issing, W.; Miki, T.; Popescu, N.C.; Aaronson, S.A. Isolation and Characterization of ERBB3, a Third Member of the ERBB/Epidermal Growth Factor Receptor Family: Evidence for Overexpression in a Subset of Human Mammary Tumors. Proc. Natl. Acad. Sci. USA 1989, 86, 9193–9197. [Google Scholar] [CrossRef] [PubMed]
- Robinson, M.K.; Hodge, K.M.; Horak, E.; Sundberg, Å.L.; Russeva, M.; Shaller, C.C.; von Mehren, M.; Shchaveleva, I.; Simmons, H.H.; Marks, J.D.; et al. Targeting ErbB2 and ErbB3 with a Bispecific Single-Chain Fv Enhances Targeting Selectivity and Induces a Therapeutic Effect in Vitro. Br. J. Cancer 2008, 99, 1415–1425. [Google Scholar] [CrossRef] [PubMed]
- Chung, Y.W.; Kim, S.; Hong, J.H.; Lee, J.K.; Lee, N.W.; Lee, Y.S.; Song, J.Y. Overexpression of HER2/HER3 and Clinical Feature of Ovarian Cancer. J. Gynecol. Oncol. 2019, 30, e75. [Google Scholar] [CrossRef] [PubMed]
- Henry, K.E.; Ulaner, G.A.; Lewis, J.S. Clinical Potential of Human Epidermal Growth Factor Receptor 2 and Human Epidermal Growth Factor Receptor 3 Imaging in Breast Cancer. PET Clin. 2018, 13, 423–435. [Google Scholar] [CrossRef] [PubMed]
- Li, Q.; Zhang, R.; Yan, H.; Zhao, P.; Wu, L.; Wang, H.; Li, T.; Cao, B. Prognostic Significance of HER3 in Patients with Malignant Solid Tumors. Oncotarget 2017, 8, 67140–67151. [Google Scholar] [CrossRef]
- Moghbeli, M.; Makhdoumi, Y.; Soltani Delgosha, M.; Aarabi, A.; Dadkhah, E.; Memar, B.; Abdollahi, A.; Abbaszadegan, M.R. ErbB1 and ErbB3 Co-over Expression as a Prognostic Factor in Gastric Cancer. Biol. Res. 2019, 52, 2. [Google Scholar] [CrossRef] [PubMed]
- Rao, K.; Gaughan, L.; Robson, C.; McCracken, S. The Role of the HER2 and HER3 in Prostate Cancer and Their Potential as Therapeutic Targets. Eur. J. Cancer 2016, 61, S177. [Google Scholar] [CrossRef]
- Lyu, H.; Han, A.; Polsdofer, E.; Liu, S.; Liu, B. Understanding the Biology of HER3 Receptor as a Therapeutic Target in Human Cancer. Acta Pharm. Sin. B 2018, 8, 503–510. [Google Scholar] [CrossRef] [PubMed]
- Dey, N.; Williams, C.; Leyland-Jones, B.; De, P. A Critical Role for HER3 in HER2-Amplified and Non-Amplified Breast Cancers: Function of a Kinase-Dead RTK. Am. J. Transl. Res. 2015, 7, 733–750. [Google Scholar]
- Sergina, N.V.; Rausch, M.; Wang, D.; Blair, J.; Hann, B.; Shokat, K.M.; Moasser, M.M. Escape from HER-Family Tyrosine Kinase Inhibitor Therapy by the Kinase-Inactive HER3. Nature 2007, 445, 437–441. [Google Scholar] [CrossRef]
- Lee, Y.; Ma, J.; Lyu, H.; Huang, J.; Kim, A.; Liu, B. Role of ErbB3 Receptors in Cancer Therapeutic Resistance. Acta Biochim. Biophys. Sin. (Shanghai) 2014, 46, 190–198. [Google Scholar] [CrossRef]
- Segers, V.F.M.; Dugaucquier, L.; Feyen, E.; Shakeri, H.; De Keulenaer, G.W. The Role of ErbB4 in Cancer. Cell Oncol. 2020, 43, 335–352. [Google Scholar] [CrossRef]
- Telesco, S.E.; Vadigepalli, R.; Radhakrishnan, R. Molecular Modeling of ErbB4/HER4 Kinase in the Context of the HER4 Signaling Network Helps Rationalize the Effects of Clinically Identified HER4 Somatic Mutations on the Cell Phenotype. Biotechnol. J. 2013, 8, 1452–1464. [Google Scholar] [CrossRef]
- Jia, X.; Wang, H.; Li, Z.; Yan, J.; Guo, Y.; Zhao, W.; Gao, L.; Wang, B.; Jia, Y. HER4 Promotes the Progression of Colorectal Cancer by Promoting Epithelial-mesenchymal Transition. Mol. Med. Rep. 2020, 21, 1779–1788. [Google Scholar] [CrossRef]
- Williams, C.S.; Bernard, J.K.; Demory Beckler, M.; Almohazey, D.; Washington, M.K.; Smith, J.J.; Frey, M.R. ERBB4 Is Over-Expressed in Human Colon Cancer and Enhances Cellular Transformation. Carcinogenesis 2015, 36, 710–718. [Google Scholar] [CrossRef]
- Portier, B.P.; Minca, E.C.; Wang, Z.; Lanigan, C.; Gruver, A.M.; Downs-Kelly, E.; Budd, G.T.; Tubbs, R.R. HER4 Expression Status Correlates with Improved Outcome in Both Neoadjuvant and Adjuvant Trastuzumab Treated Invasive Breast Carcinoma. Oncotarget 2013, 4, 1662–1672. [Google Scholar] [CrossRef]
- Wang, J.; Yin, J.; Yang, Q.; Ding, F.; Chen, X.; Li, B.; Tian, X. Human Epidermal Growth Factor Receptor 4 (HER4) Is a Favorable Prognostic Marker of Breast Cancer: A Systematic Review and Meta-Analysis. Oncotarget 2016, 7, 76693–76703. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Nafi, S.N.M.; Generali, D.; Kramer-Marek, G.; Gijsen, M.; Strina, C.; Cappelletti, M.; Andreis, D.; Haider, S.; Li, J.-L.; Bridges, E.; et al. Nuclear HER4 Mediates Acquired Resistance to Trastuzumab and Is Associated with Poor Outcome in HER2 Positive Breast Cancer. Oncotarget 2014, 5, 5934–5949. [Google Scholar] [CrossRef] [PubMed]
- Wege, A.K.; Chittka, D.; Buchholz, S.; Klinkhammer-Schalke, M.; Diermeier-Daucher, S.; Zeman, F.; Ortmann, O.; Brockhoff, G. HER4 Expression in Estrogen Receptor-Positive Breast Cancer Is Associated with Decreased Sensitivity to Tamoxifen Treatment and Reduced Overall Survival of Postmenopausal Women. Breast Cancer Res. 2018, 20, 139. [Google Scholar] [CrossRef]
- Akhter, M.H.; Madhav, N.S.; Ahmad, J. Epidermal Growth Factor Receptor Based Active Targeting: A Paradigm Shift towards Advance Tumor Therapy. Artif. Cells Nanomed. Biotechnol. 2018, 46, 1188–1198. [Google Scholar] [CrossRef]
- Ke, X.; Shen, L. Molecular Targeted Therapy of Cancer: The Progress and Future Prospect. Front. Lab. Med. 2017, 1, 69–75. [Google Scholar] [CrossRef]
- Maennling, A.E.; Tur, M.K.; Niebert, M.; Klockenbring, T.; Zeppernick, F.; Gattenlöhner, S.; Meinhold-Heerlein, I.; Hussain, A.F. Molecular Targeting Therapy against EGFR Family in Breast Cancer: Progress and Future Potentials. Cancers 2019, 11, 1826. [Google Scholar] [CrossRef] [PubMed]
- Talwar, V.; Pradeep Babu, K.V.; Raina, S. An Overall Review of Targeted Therapy in Solid Cancers. Curr. Med. Res. Pract. 2017, 7, 99–105. [Google Scholar] [CrossRef]
- Pantaleo, M.A.; Nannini, M.; Maleddu, A.; Fanti, S.; Nanni, C.; Boschi, S.; Lodi, F.; Nicoletti, G.; Landuzzi, L.; Lollini, P.L.; et al. Experimental Results and Related Clinical Implications of PET Detection of Epidermal Growth Factor Receptor (EGFr) in Cancer. Ann. Oncol. 2009, 20, 213–226. [Google Scholar] [CrossRef] [PubMed]
- Niikura, N.; Liu, J.; Hayashi, N.; Mittendorf, E.A.; Gong, Y.; Palla, S.L.; Tokuda, Y.; Gonzalez-Angulo, A.M.; Hortobagyi, G.N.; Ueno, N.T. Loss of Human Epidermal Growth Factor Receptor 2 (HER2) Expression in Metastatic Sites of HER2-Overexpressing Primary Breast Tumors. J. Clin. Oncol. 2012, 30, 593–599. [Google Scholar] [CrossRef]
- Turner, N.H.; Di Leo, A. HER2 Discordance between Primary and Metastatic Breast Cancer: Assessing the Clinical Impact. Cancer Treat. Rev. 2013, 39, 947–957. [Google Scholar] [CrossRef] [PubMed]
- Wei, Q.; Shui, Y.; Zheng, S.; Wester, K.; Nordgren, H.; Nygren, P.; Glimelius, B.; Carlsson, J. EGFR, HER2 and HER3 Expression in Primary Colorectal Carcinomas and Corresponding Metastases: Implications for Targeted Radionuclide Therapy. Oncol. Rep. 2011, 25, 3–11. [Google Scholar] [CrossRef] [PubMed]
- Gebhart, G.; Lamberts, L.E.; Wimana, Z.; Garcia, C.; Emonts, P.; Ameye, L.; Stroobants, S.; Huizing, M.; Aftimos, P.; Tol, J.; et al. Molecular Imaging as a Tool to Investigate Heterogeneity of Advanced HER2-Positive Breast Cancer and to Predict Patient Outcome under Trastuzumab Emtansine (T-DM1): The ZEPHIR Trial. Ann. Oncol. 2016, 27, 619–624. [Google Scholar] [CrossRef] [PubMed]
- Bensch, F.; Brouwers, A.H.; Lub-de Hooge, M.N.; de Jong, J.R.; van der Vegt, B.; Sleijfer, S.; de Vries, E.G.E.; Schröder, C.P. 89Zr-Trastuzumab PET Supports Clinical Decision Making in Breast Cancer Patients, When HER2 Status Cannot Be Determined by Standard Work Up. Eur. J. Nucl. Med. Mol. Imaging 2018, 45, 2300–2306. [Google Scholar] [CrossRef]
- Hicks, R.J.; Hofman, M.S. Is There Still a Role for SPECT–CT in Oncology in the PET–CT Era? Nat. Rev. Clin. Oncol. 2012, 9, 712–720. [Google Scholar] [CrossRef]
- Zanzonico, P. Principles of Nuclear Medicine Imaging: Planar, SPECT, PET, Multi-Modality, and Autoradiography Systems. Radiat. Res. 2012, 177, 349–364. [Google Scholar] [CrossRef] [PubMed]
- Kraeber-Bodéré, F.; Rousseau, C.; Bodet-Milin, C.; Mathieu, C.; Guérard, F.; Frampas, E.; Carlier, T.; Chouin, N.; Haddad, F.; Chatal, J.-F.; et al. Tumor Immunotargeting Using Innovative Radionuclides. Int. J. Mol. Sci. 2015, 16, 3932–3954. [Google Scholar] [CrossRef]
- Bailey, D.L.; Willowson, K.P. An Evidence-Based Review of Quantitative SPECT Imaging and Potential Clinical Applications. J. Nucl. Med. 2013, 54, 83–89. [Google Scholar] [CrossRef]
- Tolmachev, V.; Stone-Elander, S. Radiolabelled Proteins for Positron Emission Tomography: Pros and Cons of Labelling Methods. Biochim. Biophys. Acta 2010, 1800, 487–510. [Google Scholar] [CrossRef]
- Almuhaideb, A.; Papathanasiou, N.; Bomanji, J. 18F-FDG PET/CT Imaging in Oncology. Ann. Saudi Med. 2011, 31, 3–13. [Google Scholar] [CrossRef]
- Ul-Hassan, F.; Cook, G.J. PET/CT in Oncology. Clin. Med. 2012, 12, 368–372. [Google Scholar] [CrossRef]
- Kruse, V.; Belle, S.V.; Cocquyt, V. Imaging Requirements for Personalized Medicine: The Oncologists Point of View. Curr. Pharm. Des. 2014, 20, 2234–2249. [Google Scholar] [CrossRef] [PubMed]
- Weigelt, B.; Peterse, J.L.; van’t Veer, L.J. Breast Cancer Metastasis: Markers and Models. Nat. Rev. Cancer 2005, 5, 591–602. [Google Scholar] [CrossRef] [PubMed]
- Hoeben, B.A.W.; Molkenboer-Kuenen, J.D.M.; Oyen, W.J.G.; Peeters, W.J.M.; Kaanders, J.H.A.M.; Bussink, J.; Boerman, O.C. Radiolabeled Cetuximab: Dose Optimization for Epidermal Growth Factor Receptor Imaging in a Head-and-Neck Squamous Cell Carcinoma Model. Int. J. Cancer 2011, 129, 870–878. [Google Scholar] [CrossRef] [PubMed]
- Eckelman, W.C.; Kilbourn, M.R.; Mathis, C.A. Specific to Nonspecific Binding in Radiopharmaceutical Studies: It’s Not so Simple as It Seems! Nucl. Med. Biol. 2009, 36, 235–237. [Google Scholar] [CrossRef] [PubMed]
- Soret, M.; Bacharach, S.L.; Buvat, I. Partial-Volume Effect in PET Tumor Imaging. J. Nucl. Med. 2007, 48, 932–945. [Google Scholar] [CrossRef] [PubMed]
- Raylman, R.R.; Kison, P.V.; Wahl, R.L. Capabilities of Two- and Three-Dimensional FDG-PET for Detecting Small Lesions and Lymph Nodes in the Upper Torso: A Dynamic Phantom Study. Eur. J. Nucl. Med. 1999, 26, 39–45. [Google Scholar] [CrossRef]
- Krasniqi, A.; D’Huyvetter, M.; Devoogdt, N.; Frejd, F.Y.; Sörensen, J.; Orlova, A.; Keyaerts, M.; Tolmachev, V. Same-Day Imaging Using Small Proteins: Clinical Experience and Translational Prospects in Oncology. J. Nucl. Med. 2018, 59, 885–891. [Google Scholar] [CrossRef]
- Garousi, J.; Orlova, A.; Frejd, F.Y.; Tolmachev, V. Imaging Using Radiolabelled Targeted Proteins: Radioimmunodetection and Beyond. EJNMMI Radiopharm. Chem. 2020, 5, 16. [Google Scholar] [CrossRef]
- Bensch, F.; Lamberts, L.E.; Smeenk, M.M.; Jorritsma-Smit, A.; Lub-de Hooge, M.N.; Terwisscha van Scheltinga, A.G.T.; de Jong, J.R.; Gietema, J.A.; Schröder, C.P.; Thomas, M.; et al. (89)Zr-Lumretuzumab PET Imaging before and during HER3 Antibody Lumretuzumab Treatment in Patients with Solid Tumors. Clin. Cancer Res. 2017, 23, 6128–6137. [Google Scholar] [CrossRef]
- Dijkers, E.C.; Oude Munnink, T.H.; Kosterink, J.G.; Brouwers, A.H.; Jager, P.L.; de Jong, J.R.; van Dongen, G.A.; Schröder, C.P.; Lub-de Hooge, M.N.; de Vries, E.G. Biodistribution of 89Zr-Trastuzumab and PET Imaging of HER2-Positive Lesions in Patients with Metastatic Breast Cancer. Clin. Pharmacol. Ther. 2010, 87, 586–592. [Google Scholar] [CrossRef]
- Menke-van der Houven van Oordt, C.W.; Gootjes, E.C.; Huisman, M.C.; Vugts, D.J.; Roth, C.; Luik, A.M.; Mulder, E.R.; Schuit, R.C.; Boellaard, R.; Hoekstra, O.S.; et al. 89Zr-Cetuximab PET Imaging in Patients with Advanced Colorectal Cancer. Available online: https://pubmed.ncbi.nlm.nih.gov/26309164/ (accessed on 2 December 2020).
- Thomas, G.D. Effect of Dose, Molecular Size, and Binding Affinity on Uptake of Antibodies. In Drug Targeting: Strategies, Principles, and Applications; Francis, G.E., Delgado, C., Eds.; Methods in Molecular MedicineTM; Humana Press: Totowa, NJ, USA, 2000; pp. 115–132. ISBN 978-1-59259-075-9. [Google Scholar]
- Covell, D.G.; Barbet, J.; Holton, O.D.; Black, C.D.; Parker, R.J.; Weinstein, J.N. Pharmacokinetics of Monoclonal Immunoglobulin G1, F(Ab’)2, and Fab’ in Mice. Cancer Res. 1986, 46, 3969–3978. [Google Scholar]
- Sörensen, J.; Velikyan, I.; Sandberg, D.; Wennborg, A.; Feldwisch, J.; Tolmachev, V.; Orlova, A.; Sandström, M.; Lubberink, M.; Olofsson, H.; et al. Measuring HER2-Receptor Expression In Metastatic Breast Cancer Using [68Ga]ABY-025 Affibody PET/CT. Theranostics 2016, 6, 262–271. [Google Scholar] [CrossRef]
- Wahl, R.L.; Parker, C.W.; Philpott, G.W. Improved Radioimaging and Tumor Localization with Monoclonal F(Ab’)2. J. Nucl. Med. 1983, 24, 316–325. [Google Scholar] [PubMed]
- Yokota, T.; Milenic, D.E.; Whitlow, M.; Schlom, J. Rapid Tumor Penetration of a Single-Chain Fv and Comparison with Other Immunoglobulin Forms. Cancer Res. 1992, 52, 3402–3408. [Google Scholar] [PubMed]
- Lindenberg, L.; Adler, S.; Turkbey, I.B.; Mertan, F.; Ton, A.; Do, K.; Kummar, S.; Gonzalez, E.M.; Bhattacharyya, S.; Jacobs, P.M.; et al. Dosimetry and First Human Experience with 89Zr-Panitumumab. Am. J. Nucl. Med. Mol. Imaging 2017, 7, 195–203. [Google Scholar] [PubMed]
- Jia, L.; Zhang, L.; Shao, C.; Song, E.; Sun, W.; Li, M.; Gao, Y. An Attempt to Understand Kidney’s Protein Handling Function by Comparing Plasma and Urine Proteomes. PLoS ONE 2009, 4, e5146. [Google Scholar] [CrossRef] [PubMed]
- Schenk, S.; Schoenhals, G.J.; de Souza, G.; Mann, M. A High Confidence, Manually Validated Human Blood Plasma Protein Reference Set. BMC Med. Genom. 2008, 1, 41. [Google Scholar] [CrossRef] [PubMed]
- Dijkers, E.C.F.; Kosterink, J.G.W.; Rademaker, A.P.; Perk, L.R.; van Dongen, G.A.M.S.; Bart, J.; de Jong, J.R.; de Vries, E.G.E.; Lub-de Hooge, M.N. Development and Characterization of Clinical-Grade 89Zr-Trastuzumab for HER2/Neu ImmunoPET Imaging. J. Nucl. Med. 2009, 50, 974–981. [Google Scholar] [CrossRef] [PubMed]
- Wester, H.-J.; Kessler, H. Molecular Targeting with Peptides or Peptide-Polymer Conjugates: Just a Question of Size? J. Nucl. Med. 2005, 46, 1940–1945. [Google Scholar]
- Deyev, S.; Vorobyeva, A.; Schulga, A.; Proshkina, G.; Güler, R.; Löfblom, J.; Mitran, B.; Garousi, J.; Altai, M.; Buijs, J.; et al. Comparative Evaluation of Two DARPin Variants: Effect of Affinity, Size, and Label on Tumor Targeting Properties. Mol. Pharm. 2019, 16, 995–1008. [Google Scholar] [CrossRef]
- van Dijk, L.K.; Hoeben, B.A.W.; Kaanders, J.H.A.M.; Franssen, G.M.; Boerman, O.C.; Bussink, J. Imaging of Epidermal Growth Factor Receptor Expression in Head and Neck Cancer with SPECT/CT and 111In-Labeled Cetuximab-F(Ab′)2. J. Nucl. Med. 2013, 54, 2118–2124. [Google Scholar] [CrossRef] [PubMed]
- Lindbo, S.; Garousi, J.; Mitran, B.; Vorobyeva, A.; Oroujeni, M.; Orlova, A.; Hober, S.; Tolmachev, V. Optimized Molecular Design of ADAPT-Based HER2-Imaging Probes Labeled with 111In and 68Ga. Mol. Pharm. 2018, 15, 2674–2683. [Google Scholar] [CrossRef] [PubMed]
- Rinne, S.S.; Leitao, C.D.; Mitran, B.; Bass, T.Z.; Andersson, K.G.; Tolmachev, V.; Ståhl, S.; Löfblom, J.; Orlova, A. Optimization of HER3 Expression Imaging Using Affibody Molecules: Influence of Chelator for Labeling with Indium-111. Sci. Rep. 2019, 9, 655. [Google Scholar] [CrossRef]
- Sharifi, J.; Khirehgesh, M.R.; Safari, F.; Akbari, B. EGFR and Anti-EGFR Nanobodies: Review and Update. J. Drug Target. 2020, 1–16. [Google Scholar] [CrossRef] [PubMed]
- Solomon, V.R.; Gonzalez, C.; Alizadeh, E.; Bernhard, W.; Hartimath, S.V.; Barreto, K.; Geyer, C.R.; Fonge, H. 99mTc(CO)3+ Labeled Domain I/II-Specific Anti-EGFR (ScFv)2 Antibody Fragment for Imaging EGFR Expression. Eur. J. Med. Chem. 2018, 157, 437–446. [Google Scholar] [CrossRef]
- Holliger, P.; Hudson, P.J. Engineered Antibody Fragments and the Rise of Single Domains. Nat. Biotechnol. 2005, 23, 1126–1136. [Google Scholar] [CrossRef]
- Löfblom, J.; Feldwisch, J.; Tolmachev, V.; Carlsson, J.; Ståhl, S.; Frejd, F.Y. Affibody Molecules: Engineered Proteins for Therapeutic, Diagnostic and Biotechnological Applications. FEBS Lett. 2010, 584, 2670–2680. [Google Scholar] [CrossRef]
- Garousi, J.; Lindbo, S.; Nilvebrant, J.; Åstrand, M.; Buijs, J.; Sandström, M.; Honarvar, H.; Orlova, A.; Tolmachev, V.; Hober, S. ADAPT, a Novel Scaffold Protein-Based Probe for Radionuclide Imaging of Molecular Targets That Are Expressed in Disseminated Cancers. Cancer Res. 2015, 75, 4364–4371. [Google Scholar] [CrossRef]
- Goldstein, R.; Sosabowski, J.; Livanos, M.; Leyton, J.; Vigor, K.; Bhavsar, G.; Nagy-Davidescu, G.; Rashid, M.; Miranda, E.; Yeung, J.; et al. Development of the Designed Ankyrin Repeat Protein (DARPin) G3 for HER2 Molecular Imaging. Eur. J. Nucl. Med. Mol. Imaging 2015, 42, 288–301. [Google Scholar] [CrossRef]
- De Jong, M.; Breeman, W.A.; Bakker, W.H.; Kooij, P.P.; Bernard, B.F.; Hofland, L.J.; Visser, T.J.; Srinivasan, A.; Schmidt, M.A.; Erion, J.L.; et al. Comparison of (111)In-Labeled Somatostatin Analogues for Tumor Scintigraphy and Radionuclide Therapy. Cancer Res. 1998, 58, 437–441. [Google Scholar]
- Mitran, B.; Varasteh, Z.; Selvaraju, R.K.; Lindeberg, G.; Sörensen, J.; Larhed, M.; Tolmachev, V.; Rosenström, U.; Orlova, A. Selection of Optimal Chelator Improves the Contrast of GRPR Imaging Using Bombesin Analogue RM26. Int. J. Oncol. 2016, 48, 2124–2134. [Google Scholar] [CrossRef] [PubMed]
- Slobbe, P.; Poot, A.J.; Windhorst, A.D.; van Dongen, G.A.M.S. PET Imaging with Small-Molecule Tyrosine Kinase Inhibitors: TKI-PET. Drug Discov. Today 2012, 17, 1175–1187. [Google Scholar] [CrossRef] [PubMed]
- Kostelnik, T.I.; Orvig, C. Radioactive Main Group and Rare Earth Metals for Imaging and Therapy. Chem. Rev. 2019, 119, 902–956. [Google Scholar] [CrossRef]
- Tolmachev, V.; Orlova, A. Influence of Labelling Methods on Biodistribution and Imaging Properties of Radiolabelled Peptides for Visualisation of Molecular Therapeutic Targets. Curr. Med. Chem. 2010, 17, 2636–2655. [Google Scholar] [CrossRef] [PubMed]
- Tolmachev, V.; Orlova, A.; Lundqvist, H. Approaches to Improve Cellular Retention of Radiohalogen Labels Delivered by Internalising Tumour-Targeting Proteins and Peptides. Curr. Med. Chem. 2003, 10, 2447–2460. [Google Scholar] [CrossRef]
- Wiley, H.S.; Herbst, J.J.; Walsh, B.J.; Lauffenburger, D.A.; Rosenfeld, M.G.; Gill, G.N. The Role of Tyrosine Kinase Activity in Endocytosis, Compartmentation, and down-Regulation of the Epidermal Growth Factor Receptor. J. Biol. Chem. 1991, 266, 11083–11094. [Google Scholar] [CrossRef]
- Tolmachev, V.; Tran, T.A.; Rosik, D.; Sjöberg, A.; Abrahmsén, L.; Orlova, A. Tumor Targeting Using Affibody Molecules: Interplay of Affinity, Target Expression Level, and Binding Site Composition. J. Nucl. Med. 2012, 53, 953–960. [Google Scholar] [CrossRef]
- Ahlgren, S.; Tolmachev, V. Radionuclide Molecular Imaging Using Affibody Molecules. Curr. Pharm. Biotechnol. 2010, 11, 581–589. [Google Scholar] [CrossRef]
- Garousi, J.; Andersson, K.G.; Dam, J.H.; Olsen, B.B.; Mitran, B.; Orlova, A.; Buijs, J.; Ståhl, S.; Löfblom, J.; Thisgaard, H.; et al. The Use of Radiocobalt as a Label Improves Imaging of EGFR Using DOTA-Conjugated Affibody Molecule. Sci. Rep. 2017, 7, 5961. [Google Scholar] [CrossRef]
- Mitran, B.; Andersson, K.G.; Lindström, E.; Garousi, J.; Rosestedt, M.; Tolmachev, V.; Ståhl, S.; Orlova, A.; Löfblom, J. Affibody-mediated Imaging of EGFR Expression in Prostate Cancer Using Radiocobalt-labeled DOTA-ZEGFR:2377. Oncol. Rep. 2019, 41, 534–542. [Google Scholar] [CrossRef]
- Rinne, S.S.; Xu, T.; Dahlsson Leitao, C.; Ståhl, S.; Löfblom, J.; Orlova, A.; Tolmachev, V.; Vorobyeva, A. Influence of Residualizing Properties of the Radiolabel on Radionuclide Molecular Imaging of HER3 Using Affibody Molecules. Int. J. Mol. Sci. 2020, 21, 1312. [Google Scholar] [CrossRef] [PubMed]
- Benveniste, R.; Danoff, T.M.; Ilekis, J.; Craig, H.R. Epidermal Growth Factor Receptor Numbers in Male and Female Mouse Primary Hepatocyte Cultures. Cell Biochem. Funct. 1988, 6, 231–235. [Google Scholar] [CrossRef] [PubMed]
- Cuartero-Plaza, A.; Martínez-Miralles, E.; Rosell, R.; Vadell-Nadal, C.; Farré, M.; Real, F.X. Radiolocalization of Squamous Lung Carcinoma with 131I-Labeled Epidermal Growth Factor. Clin. Cancer Res. 1996, 2, 13–20. [Google Scholar] [PubMed]
- Reilly, R.M.; Kiarash, R.; Sandhu, J.; Lee, Y.; Cameron, C.; Hendler, A.; Vallis, K.; Gariépy, J. A Comparison of EGF and MAb 528 Labeled with 111In for Imaging Human Breast Cancer. Available online: https://pubmed.ncbi.nlm.nih.gov/10809207/ (accessed on 2 December 2020).
- Van Loon, J.; Even, A.J.G.; Aerts, H.J.W.L.; Öllers, M.; Hoebers, F.; van Elmpt, W.; Dubois, L.; Dingemans, A.-M.C.; Lalisang, R.I.; Kempers, P.; et al. PET Imaging of Zirconium-89 Labelled Cetuximab: A Phase I Trial in Patients with Head and Neck and Lung Cancer. Radiother. Oncol. 2017, 122, 267–273. [Google Scholar] [CrossRef]
- Even, A.J.G.; Hamming-Vrieze, O.; Elmpt, W.v.; Winnepenninckx, V.J.L.; Heukelom, J.; Tesselaar, M.E.T.; Vogel, W.V.; Hoeben, A.; Zegers, C.M.L.; Vugts, D.J.; et al. Quantitative Assessment of Zirconium-89 Labeled Cetuximab Using PET/CT Imaging in Patients with Advanced Head and Neck Cancer: A Theragnostic Approach. Oncotarget 2017, 8, 3870. [Google Scholar] [CrossRef]
- Dijk, L.K.v.; Boerman, O.C.; Kaanders, J.H.A.M.; Bussink, J. PET Imaging in Head and Neck Cancer Patients to Monitor Treatment Response: A Future Role for EGFR-Targeted Imaging. Clin. Cancer Res. 2015, 21, 3602–3609. [Google Scholar] [CrossRef]
- Niu, G.; Sun, X.; Cao, Q.; Courter, D.; Koong, A.; Le, Q.-T.; Gambhir, S.S.; Chen, X. Cetuximab-Based Immunotherapy and Radioimmunotherapy of Head-Neck Squamous Cell Carcinoma. Clin. Cancer Res. 2010, 16, 2095–2105. [Google Scholar] [CrossRef]
- Ping Li, W.; Meyer, L.A.; Capretto, D.A.; Sherman, C.D.; Anderson, C.J. Receptor-Binding, Biodistribution, and Metabolism Studies of 64Cu-DOTA-Cetuximab, a PET-Imaging Agent for Epidermal Growth-Factor Receptor-Positive Tumors. Cancer Biother. Radiopharm. 2008, 23, 158–171. [Google Scholar] [CrossRef]
- Song, I.H.; Lee, T.S.; Park, Y.S.; Lee, J.S.; Lee, B.C.; Moon, B.S.; An, G.I.; Lee, H.W.; Kim, K.I.; Lee, Y.J.; et al. Immuno-PET Imaging and Radioimmunotherapy of 64Cu-/177Lu-Labeled Anti-EGFR Antibody in Esophageal Squamous Cell Carcinoma Model. J. Nucl. Med. 2016, 57, 1105–1111. [Google Scholar] [CrossRef]
- Nayak, T.K.; Regino, C.A.S.; Wong, K.J.; Milenic, D.E.; Garmestani, K.; Baidoo, K.E.; Szajek, L.P.; Brechbiel, M.W. PET Imaging of HER1-Expressing Xenografts in Mice with 86Y-CHX-A”-DTPA-Cetuximab. Eur. J. Nucl. Med. Mol. Imaging 2010, 37, 1368–1376. [Google Scholar] [CrossRef]
- Nayak, T.K.; Garmestani, K.; Milenic, D.E.; Baidoo, K.E.; Brechbiel, M.W. HER1-Targeted 86Y-Panitumumab Possesses Superior Targeting Characteristics than 86Y-Cetuximab for PET Imaging of Human Malignant Mesothelioma Tumors Xenografts. PLoS ONE 2011, 6, e18198. [Google Scholar] [CrossRef] [PubMed]
- Divgi, C.R.; Welt, S.; Kris, M.; Real, F.X.; Yeh, S.D.; Gralla, R.; Merchant, B.; Schweighart, S.; Unger, M.; Larson, S.M. Phase I and Imaging Trial of Indium 111-Labeled Anti-Epidermal Growth Factor Receptor Monoclonal Antibody 225 in Patients with Squamous Cell Lung Carcinoma. J. Natl. Cancer Inst. 1991, 83, 97–104. [Google Scholar] [CrossRef] [PubMed]
- Tolmachev, V.; Rosik, D.; Wållberg, H.; Sjöberg, A.; Sandström, M.; Hansson, M.; Wennborg, A.; Orlova, A. Imaging of EGFR Expression in Murine Xenografts Using Site-Specifically Labelled Anti-EGFR 111In-DOTA-Z EGFR:2377 Affibody Molecule: Aspect of the Injected Tracer Amount. Eur. J. Nucl. Med. Mol. Imaging 2010, 37, 613–622. [Google Scholar] [CrossRef] [PubMed]
- Bhattacharyya, S.; Kurdziel, K.; Wei, L.; Riffle, L.; Kaur, G.; Hill, G.C.; Jacobs, P.M.; Tatum, J.L.; Doroshow, J.H.; Kalen, J.D. Zirconium-89 Labeled Panitumumab: A Potential Immuno-PET Probe for HER1-Expressing Carcinomas. Nucl. Med. Biol. 2013, 40, 451–457. [Google Scholar] [CrossRef] [PubMed]
- Aghevlian, S.; Lu, Y.; Winnik, M.A.; Hedley, D.W.; Reilly, R.M. Panitumumab Modified with Metal-Chelating Polymers (MCP) Complexed to 111In and 177Lu-An EGFR-Targeted Theranostic for Pancreatic Cancer. Mol. Pharm. 2018, 15, 1150–1159. [Google Scholar] [CrossRef] [PubMed]
- Chekol, R.; Solomon, V.R.; Alizadeh, E.; Bernhard, W.; Fisher, D.; Hill, W.; Barreto, K.; DeCoteau, J.F.; Parada, A.C.; Geyer, C.R.; et al. 89Zr-Nimotuzumab for ImmunoPET Imaging of Epidermal Growth Factor Receptor I. Oncotarget 2018, 9, 17117–17132. [Google Scholar] [CrossRef] [PubMed]
- Solomon, V.R.; Barreto, K.; Bernhard, W.; Alizadeh, E.; Causey, P.; Perron, R.; Gendron, D.; Alam, M.K.; Carr, A.; Geyer, C.R.; et al. Nimotuzumab Site-Specifically Labeled with 89Zr and 225Ac Using SpyTag/SpyCatcher for PET Imaging and Alpha Particle Radioimmunotherapy of Epidermal Growth Factor Receptor Positive Cancers. Cancers 2020, 12, 3449. [Google Scholar] [CrossRef]
- Perk, L.R.; Visser, G.W.M.; Vosjan, M.J.W.D.; Stigter-van Walsum, M.; Tijink, B.M.; Leemans, C.R.; van Dongen, G.A.M.S. (89)Zr as a PET Surrogate Radioisotope for Scouting Biodistribution of the Therapeutic Radiometals (90)Y and (177)Lu in Tumor-Bearing Nude Mice after Coupling to the Internalizing Antibody Cetuximab. J. Nucl. Med. 2005, 46, 1898–1906. [Google Scholar]
- Aerts, H.J.W.L.; Dubois, L.; Perk, L.; Vermaelen, P.; van Dongen, G.A.M.S.; Wouters, B.G.; Lambin, P. Disparity between in Vivo EGFR Expression and 89Zr-Labeled Cetuximab Uptake Assessed with PET. J. Nucl. Med. 2009, 50, 123–131. [Google Scholar] [CrossRef]
- Cai, W.; Chen, K.; He, L.; Cao, Q.; Koong, A.; Chen, X. Quantitative PET of EGFR Expression in Xenograft-Bearing Mice Using 64Cu-Labeled Cetuximab, a Chimeric Anti-EGFR Monoclonal Antibody. Eur J. Nucl. Med. Mol. Imaging 2007, 34, 850–858. [Google Scholar] [CrossRef]
- Niu, G.; Li, Z.; Xie, J.; Le, Q.-T.; Chen, X. PET of EGFR Antibody Distribution in Head and Neck Squamous Cell Carcinoma Models. J. Nucl. Med. 2009, 50, 1116–1123. [Google Scholar] [CrossRef] [PubMed]
- Yamaguchi, A.; Achmad, A.; Hanaoka, H.; Heryanto, Y.D.; Bhattarai, A.; Ratianto, N.; Khongorzul, E.; Shintawati, R.; Kartamihardja, A.A.P.; Kanai, A.; et al. Immuno-PET Imaging for Non-Invasive Assessment of Cetuximab Accumulation in Non-Small Cell Lung Cancer. BMC Cancer 2019, 19, 1000. [Google Scholar] [CrossRef] [PubMed]
- Van Helden, E.J.; Elias, S.G.; Gerritse, S.L.; van Es, S.C.; Boon, E.; Huisman, M.C.; van Grieken, N.C.T.; Dekker, H.; van Dongen, G.a.M.S.; Vugts, D.J.; et al. [89Zr]Zr-Cetuximab PET/CT as Biomarker for Cetuximab Monotherapy in Patients with RAS Wild-Type Advanced Colorectal Cancer. Eur. J. Nucl. Med. Mol. Imaging 2020, 47, 849–859. [Google Scholar] [CrossRef]
- Boyle, A.J.; Cao, P.-J.; Hedley, D.W.; Sidhu, S.S.; Winnik, M.A.; Reilly, R.M. MicroPET/CT Imaging of Patient-Derived Pancreatic Cancer Xenografts Implanted Subcutaneously or Orthotopically in NOD-Scid Mice Using (64)Cu-NOTA-Panitumumab F(Ab’)2 Fragments. Nucl. Med. Biol. 2015, 42, 71–77. [Google Scholar] [CrossRef]
- van Dijk, L.K.; Boerman, O.C.; Franssen, G.M.; Lok, J.; Kaanders, J.H.A.M.; Bussink, J. Early Response Monitoring with 18F-FDG PET and Cetuximab-F(Ab’)2-SPECT after Radiotherapy of Human Head and Neck Squamous Cell Carcinomas in a Mouse Model. J. Nucl. Med. 2014, 55, 1665–1670. [Google Scholar] [CrossRef] [PubMed]
- Wehrenberg-Klee, E.; Turker, N.S.; Heidari, P.; Larimer, B.; Juric, D.; Baselga, J.; Scaltriti, M.; Mahmood, U. Differential Receptor Tyrosine Kinase PET Imaging for Therapeutic Guidance. J. Nucl. Med. 2016, 57, 1413–1419. [Google Scholar] [CrossRef][Green Version]
- Ku, A.; Chan, C.; Aghevlian, S.; Cai, Z.; Cescon, D.; Bratman, S.V.; Ailles, L.; Hedley, D.W.; Reilly, R.M. MicroSPECT/CT Imaging of Cell-Line and Patient-Derived EGFR-Positive Tumor Xenografts in Mice with Panitumumab Fab Modified with Hexahistidine Peptides To Enable Labeling with 99mTc(I) Tricarbonyl Complex. Mol. Pharm. 2019, 16, 3559–3568. [Google Scholar] [CrossRef]
- Krüwel, T.; Nevoltris, D.; Bode, J.; Dullin, C.; Baty, D.; Chames, P.; Alves, F. In Vivo Detection of Small Tumour Lesions by Multi-Pinhole SPECT Applying a (99m)Tc-Labelled Nanobody Targeting the Epidermal Growth Factor Receptor. Sci. Rep. 2016, 6, 21834. [Google Scholar] [CrossRef]
- Piramoon, M.; Hosseinimehr, S.J.; Omidfar, K.; Noaparast, Z.; Abedi, S.M. 99m Tc-Anti-Epidermal Growth Factor Receptor Nanobody for Tumor Imaging. Chem. Biol. Drug Des. 2017, 89, 498–504. [Google Scholar] [CrossRef]
- Vosjan, M.J.W.D.; Perk, L.R.; Roovers, R.C.; Visser, G.W.M.; Stigter-van Walsum, M.; van Bergen En Henegouwen, P.M.P.; van Dongen, G.A.M.S. Facile Labelling of an Anti-Epidermal Growth Factor Receptor Nanobody with 68Ga via a Novel Bifunctional Desferal Chelate for Immuno-PET. Eur. J. Nucl. Med. Mol. Imaging 2011, 38, 753–763. [Google Scholar] [CrossRef]
- Oroujeni, M.; Xu, T.; Gagnon, K.; Rinne, S.S.; Weis, J.; Garousi, J.; Andersson, K.G.; Löfblom, J.; Orlova, A.; Tolmachev, V. The Use of a Non-Conventional Long-Lived Gallium Radioisotope 66Ga Improves Imaging Contrast of EGFR Expression in Malignant Tumours Using DFO-ZEGFR:2377 Affibody Molecule. Pharmaceutics 2021, 13, 292. [Google Scholar] [CrossRef]
- Burley, T.A.; Da Pieve, C.; Martins, C.D.; Ciobota, D.M.; Allott, L.; Oyen, W.J.G.; Harrington, K.J.; Smith, G.; Kramer-Marek, G. Affibody-Based PET Imaging to Guide EGFR-Targeted Cancer Therapy in Head and Neck Squamous Cell Cancer Models. J. Nucl. Med. 2019, 60, 353–361. [Google Scholar] [CrossRef]
- Garousi, J.; Andersson, K.G.; Mitran, B.; Pichl, M.-L.; Ståhl, S.; Orlova, A.; Löfblom, J.; Tolmachev, V. PET Imaging of Epidermal Growth Factor Receptor Expression in Tumours Using 89Zr-Labelled ZEGFR:2377 Affibody Molecules. Int. J. Oncol. 2016, 48, 1325–1332. [Google Scholar] [CrossRef]
- Su, X.; Cheng, K.; Jeon, J.; Shen, B.; Venturin, G.T.; Hu, X.; Rao, J.; Chin, F.T.; Wu, H.; Cheng, Z. Comparison of Two Site-Specifically 18F-Labeled Affibodies for PET Imaging of EGFR Positive Tumors. Mol. Pharm. 2014, 11, 3947–3956. [Google Scholar] [CrossRef] [PubMed]
- Oroujeni, M.; Garousi, J.; Andersson, K.G.; Löfblom, J.; Mitran, B.; Orlova, A.; Tolmachev, V. Preclinical Evaluation of [68Ga]Ga-DFO-ZEGFR:2377: A Promising Affibody-Based Probe for Noninvasive PET Imaging of EGFR Expression in Tumors. Cells 2018, 7, 141. [Google Scholar] [CrossRef] [PubMed]
- Cheng, Q.; Wållberg, H.; Grafström, J.; Lu, L.; Thorell, J.-O.; Hägg Olofsson, M.; Linder, S.; Johansson, K.; Tegnebratt, T.; Arnér, E.S.J.; et al. Preclinical PET Imaging of EGFR Levels: Pairing a Targeting with a Non-Targeting Sel-Tagged Affibody-Based Tracer to Estimate the Specific Uptake. EJNMMI Res. 2016, 6, 58. [Google Scholar] [CrossRef] [PubMed]
- Miao, Z.; Ren, G.; Liu, H.; Qi, S.; Wu, S.; Cheng, Z. PET of EGFR Expression with an 18F-Labeled Affibody Molecule. J. Nucl. Med. 2012, 53, 1110–1118. [Google Scholar] [CrossRef]
- Zhao, P.; Yang, X.; Qi, S.; Liu, H.; Jiang, H.; Hoppmann, S.; Cao, Q.; Chua, M.-S.; So, S.K.; Cheng, Z. Molecular Imaging of Hepatocellular Carcinoma Xenografts with Epidermal Growth Factor Receptor Targeted Affibody Probes. Biomed. Res. Int. 2013, 2013, 759057. [Google Scholar] [CrossRef]
- Oroujeni, M.; Andersson, K.G.; Steinhardt, X.; Altai, M.; Orlova, A.; Mitran, B.; Vorobyeva, A.; Garousi, J.; Tolmachev, V.; Löfblom, J. Influence of Composition of Cysteine-Containing Peptide-Based Chelators on Biodistribution of 99mTc-Labeled Anti-EGFR Affibody Molecules. Amino Acids 2018, 50, 981–994. [Google Scholar] [CrossRef]
- Summer, D.; Garousi, J.; Oroujeni, M.; Mitran, B.; Andersson, K.G.; Vorobyeva, A.; Löfblom, J.; Orlova, A.; Tolmachev, V.; Decristoforo, C. Cyclic versus Noncyclic Chelating Scaffold for 89Zr-Labeled ZEGFR:2377 Affibody Bioconjugates Targeting Epidermal Growth Factor Receptor Overexpression. Mol. Pharm. 2018, 15, 175–185. [Google Scholar] [CrossRef]
- Case, B.A.; Kruziki, M.A.; Stern, L.A.; Hackel, B.J. Evaluation of Affibody Charge Modification Identified by Synthetic Consensus Design in Molecular PET Imaging of Epidermal Growth Factor Receptor. Mol. Syst. Des. Eng. 2018, 3, 171–182. [Google Scholar] [CrossRef] [PubMed]
- Hackel, B.J.; Kimura, R.H.; Gambhir, S.S. Use of 64Cu-Labeled Fibronectin Domain with EGFR-Overexpressing Tumor Xenograft: Molecular Imaging. Radiology 2012, 263, 179–188. [Google Scholar] [CrossRef] [PubMed]
- Miao, Z.; Ren, G.; Liu, H.; Jiang, L.; Cheng, Z. Small-Animal PET Imaging of Human Epidermal Growth Factor Receptor Positive Tumor with a 64Cu Labeled Affibody Protein. Bioconjug. Chem. 2010, 21, 947–954. [Google Scholar] [CrossRef]
- Li, Z.; Zhao, R.; Wu, X.; Sun, Y.; Yao, M.; Li, J.; Xu, Y.; Gu, J. Identification and Characterization of a Novel Peptide Ligand of Epidermal Growth Factor Receptor for Targeted Delivery of Therapeutics. FASEB J. 2005, 19, 1978–1985. [Google Scholar] [CrossRef] [PubMed]
- Rahmanian, N.; Hosseinimehr, S.J.; Khalaj, A.; Noaparast, Z.; Abedi, S.M.; Sabzevari, O. 99mTc-Radiolabeled GE11-Modified Peptide for Ovarian Tumor Targeting. Daru 2017, 25, 13. [Google Scholar] [CrossRef]
- Jiao, H.; Zhao, X.; Han, J.; Zhang, J.; Wang, J. Synthesis of a Novel 99mTc Labeled GE11 Peptide for EGFR SPECT Imaging. Int. J. Radiat. Biol. 2020, 96, 1443–1451. [Google Scholar] [CrossRef]
- Li, X.; Hu, K.; Liu, W.; Wei, Y.; Sha, R.; Long, Y.; Han, Y.; Sun, P.; Wu, H.; Li, G.; et al. Synthesis and Evaluation of [18F]FP-Lys-GE11 as a New Radiolabeled Peptide Probe for Epidermal Growth Factor Receptor (EGFR) Imaging. Nucl. Med. Biol. 2020, 90–91, 84–92. [Google Scholar] [CrossRef] [PubMed]
- Striese, F.; Sihver, W.; Gao, F.; Bergmann, R.; Walther, M.; Pietzsch, J.; Steinbach, J.; Pietzsch, H.-J. Exploring Pitfalls of 64Cu-Labeled EGFR-Targeting Peptide GE11 as a Potential PET Tracer. Amino Acids 2018, 50, 1415–1431. [Google Scholar] [CrossRef]
- Sun, X.; Xiao, Z.; Chen, G.; Han, Z.; Liu, Y.; Zhang, C.; Sun, Y.; Song, Y.; Wang, K.; Fang, F.; et al. A PET Imaging Approach for Determining EGFR Mutation Status for Improved Lung Cancer Patient Management. Sci. Transl. Med. 2018, 10, eaan8840. [Google Scholar] [CrossRef]
- Xiao, Z.; Song, Y.; Kai, W.; Sun, X.; Shen, B. Evaluation of 99mTc-HYNIC-MPG as a Novel SPECT Radiotracer to Detect EGFR-Activating Mutations in NSCLC. Oncotarget 2017, 8, 40732–40740. [Google Scholar] [CrossRef]
- Song, Y.; Xiao, Z.; Wang, K.; Wang, X.; Zhang, C.; Fang, F.; Sun, X.; Shen, B. Development and Evaluation of 18 F-IRS for Molecular Imaging Mutant EGF Receptors in NSCLC. Sci. Rep. 2017, 7, 3121. [Google Scholar] [CrossRef] [PubMed]
- Marcu, L.G. Imaging Biomarkers of Tumour Proliferation and Invasion for Personalised Lung Cancer Therapy. J. Pers. Med. 2020, 10, 222. [Google Scholar] [CrossRef] [PubMed]
- Marcu, L.G.; Moghaddasi, L.; Bezak, E. Imaging of Tumor Characteristics and Molecular Pathways with PET: Developments Over the Last Decade Toward Personalized Cancer Therapy. Int. J. Radiat. Oncol. Biol. Phys. 2018, 102, 1165–1182. [Google Scholar] [CrossRef] [PubMed]
- Turker, N.S.; Heidari, P.; Kucherlapati, R.; Kucherlapati, M.; Mahmood, U. An EGFR Targeted PET Imaging Probe for the Detection of Colonic Adenocarcinomas in the Setting of Colitis. Theranostics 2014, 4, 893–903. [Google Scholar] [CrossRef]
- Van Dijk, L.K.; Boerman, O.C.; Franssen, G.M.; Kaanders, J.H.A.M.; Bussink, J. 111In-Cetuximab-F(Ab’)2 SPECT and 18F-FDG PET for Prediction and Response Monitoring of Combined-Modality Treatment of Human Head and Neck Carcinomas in a Mouse Model. J. Nucl. Med. 2015, 56, 287–292. [Google Scholar] [CrossRef]
- Mitani, S.; Kawakami, H. Emerging Targeted Therapies for HER2 Positive Gastric Cancer That Can Overcome Trastuzumab Resistance. Cancers 2020, 12, 400. [Google Scholar] [CrossRef]
- Pernas, S.; Tolaney, S.M. HER2-Positive Breast Cancer: New Therapeutic Frontiers and Overcoming Resistance. Ther. Adv. Med. Oncol. 2019, 11, 1–16. [Google Scholar] [CrossRef]
- Ross, J.S.; Fletcher, J.A.; Bloom, K.J.; Linette, G.P.; Stec, J.; Symmans, W.F.; Pusztai, L.; Hortobagyi, G.N. Targeted Therapy in Breast Cancer: The HER-2/Neu Gene and Protein. Mol. Cell Proteom. 2004, 3, 379–398. [Google Scholar] [CrossRef]
- Gullick, W.J.; Marsden, J.J.; Whittle, N.; Ward, B.; Bobrow, L.; Waterfield, M.D. Expression of Epidermal Growth Factor Receptors on Human Cervical, Ovarian, and Vulval Carcinomas. Cancer Res. 1986, 46, 285–292. [Google Scholar]
- Yan, H.; Endo, Y.; Shen, Y.; Rotstein, D.; Dokmanovic, M.; Mohan, N.; Mukhopadhyay, P.; Gao, B.; Pacher, P.; Wu, W.J. Ado-Trastuzumab Emtansine Targets Hepatocytes Via Human Epidermal Growth Factor Receptor 2 to Induce Hepatotoxicity. Mol. Cancer Ther. 2016, 15, 480–490. [Google Scholar] [CrossRef]
- Oude Munnink, T.H.; de Vries, E.G.E.; Vedelaar, S.R.; Timmer-Bosscha, H.; Schröder, C.P.; Brouwers, A.H.; Lub-de Hooge, M.N. Lapatinib and 17AAG Reduce 89Zr-Trastuzumab-F(Ab’)2 Uptake in SKBR3 Tumor Xenografts. Mol. Pharm. 2012, 9, 2995–3002. [Google Scholar] [CrossRef] [PubMed]
- Bragina, O.; von Witting, E.; Garousi, J.; Zelchan, R.; Sandström, M.; Medvedeva, A.; Orlova, A.; Doroshenko, A.; Vorobyeva, A.; Lindbo, S.; et al. Phase I Study of 99mTc-ADAPT6, a Scaffold Protein-Based Probe for Visualization of HER2 Expression in Breast Cancer. J. Nucl. Med. 2020, 62, 493–499. [Google Scholar] [CrossRef] [PubMed]
- Guo, X.; Zhou, N.; Chen, Z.; Liu, T.; Xu, X.; Lei, X.; Shen, L.; Gao, J.; Yang, Z.; Zhu, H. Construction of 124I-Trastuzumab for Noninvasive PET Imaging of HER2 Expression: From Patient-Derived Xenograft Models to Gastric Cancer Patients. Gastric Cancer 2020, 23, 614–626. [Google Scholar] [CrossRef]
- Perik, P.J.; Lub-De Hooge, M.N.; Gietema, J.A.; van der Graaf, W.T.A.; de Korte, M.A.; Jonkman, S.; Kosterink, J.G.W.; van Veldhuisen, D.J.; Sleijfer, D.T.; Jager, P.L.; et al. Indium-111-Labeled Trastuzumab Scintigraphy in Patients with Human Epidermal Growth Factor Receptor 2-Positive Metastatic Breast Cancer. J. Clin. Oncol. 2006, 24, 2276–2282. [Google Scholar] [CrossRef]
- Zeglis, B.M.; Mohindra, P.; Weissmann, G.I.; Divilov, V.; Hilderbrand, S.A.; Weissleder, R.; Lewis, J.S. Modular Strategy for the Construction of Radiometalated Antibodies for Positron Emission Tomography Based on Inverse Electron Demand Diels-Alder Click Chemistry. Bioconjug. Chem. 2011, 22, 2048–2059. [Google Scholar] [CrossRef]
- Gaykema, S.B.M.; de Jong, J.R.; Perik, P.J.; Brouwers, A.H.; Schröder, C.P.; Munnink, T.H.O.; Bongaerts, A.H.H.; de Vries, E.G.E.; Hooge, M.N.L. 111In-Trastuzumab Scintigraphy in HER2-Positive Metastatic Breast Cancer Patients Remains Feasible during Trastuzumab Treatment. Mol. Imaging 2014, 13, 1–6. [Google Scholar] [CrossRef]
- Holloway, C.M.B.; Scollard, D.A.; Caldwell, C.B.; Ehrlich, L.; Kahn, H.J.; Reilly, R.M. Phase I Trial of Intraoperative Detection of Tumor Margins in Patients with HER2-Positive Carcinoma of the Breast Following Administration of 111In-DTPA-Trastuzumab Fab Fragments. Nucl. Med. Biol. 2013, 40, 630–637. [Google Scholar] [CrossRef]
- McLarty, K.; Cornelissen, B.; Scollard, D.A.; Done, S.J.; Chun, K.; Reilly, R.M. Associations between the Uptake of 111In-DTPA-Trastuzumab, HER2 Density and Response to Trastuzumab (Herceptin) in Athymic Mice Bearing Subcutaneous Human Tumour Xenografts. Eur. J. Nucl. Med. Mol. Imaging 2009, 36, 81–93. [Google Scholar] [CrossRef] [PubMed]
- Spreckelmeyer, S.; Ramogida, C.F.; Rousseau, J.; Arane, K.; Bratanovic, I.; Colpo, N.; Jermilova, U.; Dias, G.M.; Dude, I.; Jaraquemada-Peláez, M.d.G.; et al. P-NO2-Bn-H4neunpa and H4neunpa-Trastuzumab: Bifunctional Chelator for Radiometalpharmaceuticals and 111In Immuno-Single Photon Emission Computed Tomography Imaging. Bioconjug. Chem. 2017, 28, 2145–2159. [Google Scholar] [CrossRef] [PubMed]
- Kurdziel, K.; Mena, E.; McKinney, Y.; Wong, K.; Adler, S.; Sissung, T.; Lee, J.; Lipkowitz, S.; Lindenberg, L.; Turkbey, B.; et al. First-in-Human Phase 0 Study of 111In-CHX-A”-DTPA Trastuzumab for HER2 Tumor Imaging. J. Transl. Sci. 2019, 5. [Google Scholar] [CrossRef]
- Jauw, Y.W.S.; Bensch, F.; Brouwers, A.H.; Hoekstra, O.S.; Zijlstra, J.M.; Pieplenbosch, S.; Schröder, C.P.; Zweegman, S.; van Dongen, G.A.M.S.; Menke-van der Houven van Oordt, C.W.; et al. Interobserver Reproducibility of Tumor Uptake Quantification with 89Zr-Immuno-PET: A Multicenter Analysis. Eur. J. Nucl. Med. Mol. Imaging 2019, 46, 1840–1849. [Google Scholar] [CrossRef]
- Laforest, R.; Lapi, S.E.; Oyama, R.; Bose, R.; Tabchy, A.; Marquez-Nostra, B.V.; Burkemper, J.; Wright, B.D.; Frye, J.; Frye, S.; et al. [89Zr]Trastuzumab: Evaluation of Radiation Dosimetry, Safety, and Optimal Imaging Parameters in Women with HER2-Positive Breast Cancer. Mol. Imaging Biol. 2016, 18, 952–959. [Google Scholar] [CrossRef] [PubMed]
- O’Donoghue, J.A.; Lewis, J.S.; Pandit-Taskar, N.; Fleming, S.E.; Schöder, H.; Larson, S.M.; Beylergil, V.; Ruan, S.; Lyashchenko, S.K.; Zanzonico, P.B.; et al. Pharmacokinetics, Biodistribution, and Radiation Dosimetry for 89Zr-Trastuzumab in Patients with Esophagogastric Cancer. J. Nucl. Med. 2018, 59, 161–166. [Google Scholar] [CrossRef] [PubMed]
- Ulaner, G.A.; Hyman, D.M.; Lyashchenko, S.K.; Lewis, J.S.; Carrasquillo, J.A. 89Zr-Trastuzumab PET/CT for Detection of Human Epidermal Growth Factor Receptor 2-Positive Metastases in Patients With Human Epidermal Growth Factor Receptor 2-Negative Primary Breast Cancer. Clin. Nucl. Med. 2017, 42, 912–917. [Google Scholar] [CrossRef] [PubMed]
- Ulaner, G.A.; Lyashchenko, S.K.; Riedl, C.; Ruan, S.; Zanzonico, P.B.; Lake, D.; Jhaveri, K.; Zeglis, B.; Lewis, J.S.; O’Donoghue, J.A. First-in-Human Human Epidermal Growth Factor Receptor 2-Targeted Imaging Using 89Zr-Pertuzumab PET/CT: Dosimetry and Clinical Application in Patients with Breast Cancer. J. Nucl. Med. 2018, 59, 900–906. [Google Scholar] [CrossRef]
- Ulaner, G.A.; Hyman, D.M.; Ross, D.S.; Corben, A.; Chandarlapaty, S.; Goldfarb, S.; McArthur, H.; Erinjeri, J.P.; Solomon, S.B.; Kolb, H.; et al. Detection of HER2-Positive Metastases in Patients with HER2-Negative Primary Breast Cancer Using 89Zr-Trastuzumab PET/CT. J. Nucl. Med. 2016, 57, 1523–1528. [Google Scholar] [CrossRef] [PubMed]
- Ulaner, G.A.; Carrasquillo, J.A.; Riedl, C.C.; Yeh, R.; Hatzoglou, V.; Ross, D.S.; Jhaveri, K.; Chandarlapaty, S.; Hyman, D.M.; Zeglis, B.M.; et al. Identification of HER2-Positive Metastases in Patients with HER2-Negative Primary Breast Cancer by Using HER2-Targeted 89Zr-Pertuzumab PET/CT. Radiology 2020, 296, 370–378. [Google Scholar] [CrossRef]
- Tamura, K.; Kurihara, H.; Yonemori, K.; Tsuda, H.; Suzuki, J.; Kono, Y.; Honda, N.; Kodaira, M.; Yamamoto, H.; Yunokawa, M.; et al. 64Cu-DOTA-Trastuzumab PET Imaging in Patients with HER2-Positive Breast Cancer. J. Nucl. Med. 2013, 54, 1869–1875. [Google Scholar] [CrossRef] [PubMed]
- Mortimer, J.E.; Bading, J.R.; Colcher, D.M.; Conti, P.S.; Frankel, P.H.; Carroll, M.I.; Tong, S.; Poku, E.; Miles, J.K.; Shively, J.E.; et al. Functional Imaging of Human Epidermal Growth Factor Receptor 2-Positive Metastatic Breast Cancer Using (64)Cu-DOTA-Trastuzumab PET. J. Nucl. Med. 2014, 55, 23–29. [Google Scholar] [CrossRef]
- Mortimer, J.E.; Bading, J.R.; Park, J.M.; Frankel, P.H.; Carroll, M.I.; Tran, T.T.; Poku, E.K.; Rockne, R.C.; Raubitschek, A.A.; Shively, J.E.; et al. Tumor Uptake of 64Cu-DOTA-Trastuzumab in Patients with Metastatic Breast Cancer. J. Nucl. Med. 2018, 59, 38–43. [Google Scholar] [CrossRef]
- Carrasquillo, J.A.; Morris, P.G.; Humm, J.L.; Smith-Jones, P.M.; Beylergil, V.; Akhurst, T.; O’donoghue, J.A.; Ruan, S.; Modi, S.; Hudis, C.A.; et al. Copper-64 Trastuzumab PET Imaging: A Reproducibility Study. Q. J. Nucl. Med. Mol. Imaging 2019, 63, 191–198. [Google Scholar] [CrossRef]
- Guo, X.; Zhu, H.; Zhou, N.; Chen, Z.; Liu, T.; Liu, F.; Xu, X.; Jin, H.; Shen, L.; Gao, J.; et al. Noninvasive Detection of HER2 Expression in Gastric Cancer by 64Cu-NOTA-Trastuzumab in PDX Mouse Model and in Patients. Mol. Pharm. 2018, 15, 5174–5182. [Google Scholar] [CrossRef] [PubMed]
- Lub-de Hooge, M.N.; Kosterink, J.G.W.; Perik, P.J.; Nijnuis, H.; Tran, L.; Bart, J.; Suurmeijer, A.J.H.; de Jong, S.; Jager, P.L.; de Vries, E.G.E. Preclinical Characterisation of 111In-DTPA-Trastuzumab. Br. J. Pharmacol. 2004, 143, 99–106. [Google Scholar] [CrossRef] [PubMed]
- Orlova, A.; Wållberg, H.; Stone-Elander, S.; Tolmachev, V. On the Selection of a Tracer for PET Imaging of HER2-Expressing Tumors: Direct Comparison of a 124I-Labeled Affibody Molecule and Trastuzumab in a Murine Xenograft Model. J. Nucl. Med. 2009, 50, 417–425. [Google Scholar] [CrossRef] [PubMed]
- Pan, G.; Li, D.; Li, X.; Peng, Y.; Wang, T.; Zuo, C. SPECT/CT Imaging of HER2 Expression in Colon Cancer-Bearing Nude Mice Using 125I-Herceptin. Biochem. Biophys. Res. Commun. 2018, 504, 765–770. [Google Scholar] [CrossRef]
- Massicano, A.; Aweda, T.; Marquez-Nostra, B.; Sayed, R.E.; Beacham, R.; Lapi, S. Imaging HER2 in Response to T-DM1 Therapy in Breast Cancer Xenografts. J. Nucl. Med. 2017, 58, 932. [Google Scholar]
- Jiang, D.; Im, H.-J.; Sun, H.; Valdovinos, H.F.; England, C.G.; Ehlerding, E.B.; Nickles, R.J.; Lee, D.S.; Cho, S.Y.; Huang, P.; et al. Radiolabeled Pertuzumab for Imaging of Human Epidermal Growth Factor Receptor 2 Expression in Ovarian Cancer. Eur. J. Nucl. Med. Mol. Imaging 2017, 44, 1296–1305. [Google Scholar] [CrossRef]
- Dehdashti, F.; Wu, N.; Bose, R.; Naughton, M.J.; Ma, C.X.; Marquez-Nostra, B.V.; Diebolder, P.; Mpoy, C.; Rogers, B.E.; Lapi, S.E.; et al. Evaluation of [89Zr]Trastuzumab-PET/CT in Differentiating HER2-Positive from HER2-Negative Breast Cancer. Breast Cancer Res. Treat. 2018, 169, 523–530. [Google Scholar] [CrossRef]
- Patra, M.; Bauman, A.; Mari, C.; Fischer, C.A.; Blacque, O.; Häussinger, D.; Gasser, G.; Mindt, T.L. An Octadentate Bifunctional Chelating Agent for the Development of Stable Zirconium-89 Based Molecular Imaging Probes. Chem. Commun. 2014, 50, 11523–11525. [Google Scholar] [CrossRef]
- Vugts, D.J.; Klaver, C.; Sewing, C.; Poot, A.J.; Adamzek, K.; Huegli, S.; Mari, C.; Visser, G.W.M.; Valverde, I.E.; Gasser, G.; et al. Comparison of the Octadentate Bifunctional Chelator DFO*-PPhe-NCS and the Clinically Used Hexadentate Bifunctional Chelator DFO-PPhe-NCS for 89Zr-Immuno-PET. Eur. J. Nucl. Med. Mol. Imaging 2017, 44, 286–295. [Google Scholar] [CrossRef]
- Cho, H.; Al-Saden, N.; Lam, H.; Möbus, J.; Reilly, R.M.; Winnik, M.A. A Comparison of DFO and DFO* Conjugated to Trastuzumab-DM1 for Complexing 89Zr—In Vitro Stability and in Vivo MicroPET/CT Imaging Studies in NOD/SCID Mice with HER2-Positive SK-OV-3 Human Ovarian Cancer Xenografts. Nucl. Med. Biol. 2020, 84–85, 11–19. [Google Scholar] [CrossRef] [PubMed]
- Kristensen, L.K.; Christensen, C.; Jensen, M.M.; Agnew, B.J.; Schjöth-Frydendahl, C.; Kjaer, A.; Nielsen, C.H. Site-Specifically Labeled 89Zr-DFO-Trastuzumab Improves Immuno-Reactivity and Tumor Uptake for Immuno-PET in a Subcutaneous HER2-Positive Xenograft Mouse Model. Theranostics 2019, 9, 4409–4420. [Google Scholar] [CrossRef] [PubMed]
- Woo, S.-K.; Jang, S.J.; Seo, M.-J.; Park, J.H.; Kim, B.S.; Kim, E.J.; Lee, Y.J.; Lee, T.S.; An, G.I.; Song, I.H.; et al. Development of 64Cu-NOTA-Trastuzumab for HER2 Targeting: A Radiopharmaceutical with Improved Pharmacokinetics for Human Studies. J. Nucl. Med. 2019, 60, 26–33. [Google Scholar] [CrossRef] [PubMed]
- Paudyal, P.; Paudyal, B.; Hanaoka, H.; Oriuchi, N.; Iida, Y.; Yoshioka, H.; Tominaga, H.; Watanabe, S.; Watanabe, S.; Ishioka, N.S.; et al. Imaging and Biodistribution of Her2/Neu Expression in Non-Small Cell Lung Cancer Xenografts with 64Cu-Labeled Trastuzumab PET. Cancer Sci. 2010, 101, 1045–1050. [Google Scholar] [CrossRef] [PubMed]
- Paterson, B.M.; Buncic, G.; McInnes, L.E.; Roselt, P.; Cullinane, C.; Binns, D.S.; Jeffery, C.M.; Price, R.I.; Hicks, R.J.; Donnelly, P.S. Bifunctional 64Cu-Labelled Macrobicyclic Cage Amine Isothiocyanates for Immuno-Positron Emission Tomography. Dalton Trans. 2015, 44, 4901–4909. [Google Scholar] [CrossRef]
- Tolmachev, V.; Grönroos, T.J.; Yim, C.-B.; Garousi, J.; Yue, Y.; Grimm, S.; Rajander, J.; Perols, A.; Haaparanta-Solin, M.; Solin, O.; et al. Molecular Design of Radiocopper-Labelled Affibody Molecules. Sci. Rep. 2018, 8, 6542. [Google Scholar] [CrossRef]
- Krasniqi, E.; Barchiesi, G.; Pizzuti, L.; Mazzotta, M.; Venuti, A.; Maugeri-Saccà, M.; Sanguineti, G.; Massimiani, G.; Sergi, D.; Carpano, S.; et al. Immunotherapy in HER2-Positive Breast Cancer: State of the Art and Future Perspectives. J. Hematol. Oncol. 2019, 12, 111. [Google Scholar] [CrossRef]
- Lam, K.; Chan, C.; Reilly, R.M. Development and Preclinical Studies of 64Cu-NOTA-Pertuzumab F(Ab’)2 for Imaging Changes in Tumor HER2 Expression Associated with Response to Trastuzumab by PET/CT. MAbs 2017, 9, 154–164. [Google Scholar] [CrossRef]
- Smith-Jones, P.M.; Solit, D.B.; Akhurst, T.; Afroze, F.; Rosen, N.; Larson, S.M. Imaging the Pharmacodynamics of HER2 Degradation in Response to Hsp90 Inhibitors. Nat. Biotechnol. 2004, 22, 701–706. [Google Scholar] [CrossRef]
- Mendler, C.T.; Gehring, T.; Wester, H.-J.; Schwaiger, M.; Skerra, A. 89Zr-Labeled Versus 124I-Labeled ΑHER2 Fab with Optimized Plasma Half-Life for High-Contrast Tumor Imaging In Vivo. J. Nucl. Med. 2015, 56, 1112–1118. [Google Scholar] [CrossRef]
- Richter, A.; Knorr, K.; Schlapschy, M.; Robu, S.; Morath, V.; Mendler, C.; Yen, H.-Y.; Steiger, K.; Kiechle, M.; Weber, W.; et al. First In-Human Medical Imaging with a PASylated 89Zr-Labeled Anti-HER2 Fab-Fragment in a Patient with Metastatic Breast Cancer. Nucl. Med. Mol. Imaging 2020, 54, 114–119. [Google Scholar] [CrossRef]
- Beylergil, V.; Morris, P.G.; Smith-Jones, P.M.; Modi, S.; Solit, D.; Hudis, C.A.; Lu, Y.; O’Donoghue, J.; Lyashchenko, S.K.; Carrasquillo, J.A.; et al. Pilot Study of 68Ga-DOTA-F(Ab’)2-Trastuzumab in Patients with Breast Cancer. Nucl. Med. Commun. 2013, 34, 1157–1165. [Google Scholar] [CrossRef]
- Adams, G.P.; McCartney, J.E.; Tai, M.S.; Oppermann, H.; Huston, J.S.; Stafford, W.F.; Bookman, M.A.; Fand, I.; Houston, L.L.; Weiner, L.M. Highly Specific in vivo Tumor Targeting by Monovalent and Divalent Forms of 741F8 Anti-c-ErbB-2 Single-Chain Fv. Cancer Res. 1993, 53, 4026–4034. [Google Scholar]
- Adams, G.P.; Schier, R.; McCall, A.M.; Crawford, R.S.; Wolf, E.J.; Weiner, L.M.; Marks, J.D. Prolonged in Vivo Tumour Retention of a Human Diabody Targeting the Extracellular Domain of Human HER2/Neu. Br. J. Cancer 1998, 77, 1405–1412. [Google Scholar] [CrossRef]
- Wittrup, K.D.; Thurber, G.M.; Schmidt, M.M.; Rhoden, J.J. Practical Theoretic Guidance for the Design of Tumor-Targeting Agents. Methods Enzymol. 2012, 503, 255–268. [Google Scholar] [CrossRef]
- Adams, G.P.; McCartney, J.E.; Wolf, E.J.; Eisenberg, J.; Huston, J.S.; Bookman, M.A.; Moldofsky, P.; Stafford, W.F.; Houston, L.L.; Weiner, L.M. Enhanced Tumor Specificity of 741F8-1 (SFv’)2, an Anti-c-ErbB-2 Single-Chain Fv Dimer, Mediated by Stable Radioiodine Conjugation. J. Nucl. Med. 1995, 36, 2276–2281. [Google Scholar]
- Adams, G.P.; Shaller, C.C.; Dadachova, E.; Simmons, H.H.; Horak, E.M.; Tesfaye, A.; Klein-Szanto, A.J.P.; Marks, J.D.; Brechbiel, M.W.; Weiner, L.M. A Single Treatment of Yttrium-90-Labeled CHX-A”-C6.5 Diabody Inhibits the Growth of Established Human Tumor Xenografts in Immunodeficient Mice. Cancer Res. 2004, 64, 6200–6206. [Google Scholar] [CrossRef]
- Ueda, M.; Hisada, H.; Temma, T.; Shimizu, Y.; Kimura, H.; Ono, M.; Nakamoto, Y.; Togashi, K.; Saji, H. Gallium-68-Labeled Anti-HER2 Single-Chain Fv Fragment: Development and in Vivo Monitoring of HER2 Expression. Mol. Imaging Biol. 2015, 17, 102–110. [Google Scholar] [CrossRef]
- Olafsen, T.; Kenanova, V.E.; Sundaresan, G.; Anderson, A.-L.; Crow, D.; Yazaki, P.J.; Li, L.; Press, M.F.; Gambhir, S.S.; Williams, L.E.; et al. Optimizing Radiolabeled Engineered Anti-P185HER2 Antibody Fragments for In Vivo Imaging. Cancer Res. 2005, 65, 5907–5916. [Google Scholar] [CrossRef]
- Vaidyanathan, G.; McDougald, D.; Choi, J.; Koumarianou, E.; Weitzel, D.; Osada, T.; Lyerly, H.K.; Zalutsky, M.R. Preclinical Evaluation of 18F-Labeled Anti-HER2 Nanobody Conjugates for Imaging HER2 Receptor Expression by Immuno-PET. J. Nucl. Med. 2016, 57, 967–973. [Google Scholar] [CrossRef]
- Vaneycken, I.; Devoogdt, N.; Van Gassen, N.; Vincke, C.; Xavier, C.; Wernery, U.; Muyldermans, S.; Lahoutte, T.; Caveliers, V. Preclinical Screening of Anti-HER2 Nanobodies for Molecular Imaging of Breast Cancer. FASEB J. 2011, 25, 2433–2446. [Google Scholar] [CrossRef]
- D’Huyvetter, M.; De Vos, J.; Caveliers, V.; Vaneycken, I.; Heemskerk, J.; Duhoux, F.P.; Fontaine, C.; Vanhoeij, M.; Windhorst, A.D.; van der Aa, F.; et al. Phase I Trial of 131I-GMIB-Anti-HER2-VHH1, a New Promising Candidate for HER2-Targeted Radionuclide Therapy in Breast Cancer Patients. J. Nucl. Med. 2020. [Google Scholar] [CrossRef]
- D’Huyvetter, M.; De Vos, J.; Xavier, C.; Pruszynski, M.; Sterckx, Y.G.J.; Massa, S.; Raes, G.; Caveliers, V.; Zalutsky, M.R.; Lahoutte, T.; et al. 131I-Labeled Anti-HER2 Camelid SdAb as a Theranostic Tool in Cancer Treatment. Clin. Cancer Res. 2017, 23, 6616–6628. [Google Scholar] [CrossRef]
- Zhou, Z.; Vaidyanathan, G.; McDougald, D.; Kang, C.M.; Balyasnikova, I.; Devoogdt, N.; Ta, A.N.; McNaughton, B.R.; Zalutsky, M.R. Fluorine-18 Labeling of the HER2-Targeting Single-Domain Antibody 2Rs15d Using a Residualizing Label and Preclinical Evaluation. Mol. Imaging Biol. 2017, 19, 867–877. [Google Scholar] [CrossRef]
- Xavier, C.; Blykers, A.; Vaneycken, I.; D’Huyvetter, M.; Heemskerk, J.; Lahoutte, T.; Devoogdt, N.; Caveliers, V. (18)F-Nanobody for PET Imaging of HER2 Overexpressing Tumors. Nucl. Med. Biol. 2016, 43, 247–252. [Google Scholar] [CrossRef]
- Keyaerts, M.; Xavier, C.; Heemskerk, J.; Devoogdt, N.; Everaert, H.; Ackaert, C.; Vanhoeij, M.; Duhoux, F.P.; Gevaert, T.; Simon, P.; et al. Phase I Study of 68Ga-HER2-Nanobody for PET/CT Assessment of HER2 Expression in Breast Carcinoma. J. Nucl. Med. 2016, 57, 27–33. [Google Scholar] [CrossRef]
- Vorobyeva, A.; Schulga, A.; Rinne, S.S.; Günther, T.; Orlova, A.; Deyev, S.; Tolmachev, V. Indirect Radioiodination of DARPin G3 Using N-Succinimidyl-Para-Iodobenzoate Improves the Contrast of HER2 Molecular Imaging. Int. J. Mol. Sci. 2019, 20, 3047. [Google Scholar] [CrossRef]
- Kim, H.J.; Park, J.Y.; Lee, T.S.; Song, I.H.; Cho, Y.L.; Chae, J.R.; Kang, H.; Lim, J.H.; Lee, J.H.; Kang, W.J. PET Imaging of HER2 Expression with an 18F-Fluoride Labeled Aptamer. PLoS ONE 2019, 14, e0211047. [Google Scholar] [CrossRef]
- Ahlgren, S.; Orlova, A.; Wållberg, H.; Hansson, M.; Sandström, M.; Lewsley, R.; Wennborg, A.; Abrahmsén, L.; Tolmachev, V.; Feldwisch, J. Targeting of HER2-Expressing Tumors Using 111In-ABY-025, a Second-Generation Affibody Molecule with a Fundamentally Reengineered Scaffold. J. Nucl. Med. 2010, 51, 1131–1138. [Google Scholar] [CrossRef]
- Wållberg, H.; Orlova, A. Slow Internalization of Anti-HER2 Synthetic Affibody Monomer 111In-DOTA-ZHER2:342-Pep2: Implications for Development of Labeled Tracers. Cancer Biother. Radiopharm. 2008, 23, 435–442. [Google Scholar] [CrossRef]
- Orlova, A.; Magnusson, M.; Eriksson, T.L.J.; Nilsson, M.; Larsson, B.; Höidén-Guthenberg, I.; Widström, C.; Carlsson, J.; Tolmachev, V.; Ståhl, S.; et al. Tumor Imaging Using a Picomolar Affinity HER2 Binding Affibody Molecule. Cancer Res. 2006, 66, 4339–4348. [Google Scholar] [CrossRef]
- Zahnd, C.; Wyler, E.; Schwenk, J.; Steiner, D.; Mc, M.; McKern, N.; Pecorari, F.; Ward, C.; Joos, T.; Plückthun, A. A Designed Ankyrin Repeat Protein Evolved to Picomolar Affinity to Her2. J. Mol. Biol. 2007, 369, 1015–1028. [Google Scholar] [CrossRef]
- Cheng, Z.; De Jesus, O.P.; Kramer, D.J.; De, A.; Webster, J.M.; Gheysens, O.; Levi, J.; Namavari, M.; Wang, S.; Park, J.M.; et al. 64Cu-Labeled Affibody Molecules for Imaging of HER2 Expressing Tumors. Mol. Imaging Biol. 2010, 12, 316–324. [Google Scholar] [CrossRef]
- Garousi, J.; Lindbo, S.; Borin, J.; von Witting, E.; Vorobyeva, A.; Oroujeni, M.; Mitran, B.; Orlova, A.; Buijs, J.; Tolmachev, V.; et al. Comparative Evaluation of Dimeric and Monomeric Forms of ADAPT Scaffold Protein for Targeting of HER2-Expressing Tumours. Eur. J. Pharm. Biopharm. 2019, 134, 37–48. [Google Scholar] [CrossRef]
- Baum, R.P.; Prasad, V.; Müller, D.; Schuchardt, C.; Orlova, A.; Wennborg, A.; Tolmachev, V.; Feldwisch, J. Molecular Imaging of HER2-Expressing Malignant Tumors in Breast Cancer Patients Using Synthetic 111In- or 68Ga-Labeled Affibody Molecules. J. Nucl. Med. 2010, 51, 892–897. [Google Scholar] [CrossRef]
- Sörensen, J.; Sandberg, D.; Sandström, M.; Wennborg, A.; Feldwisch, J.; Tolmachev, V.; Åström, G.; Lubberink, M.; Garske-Román, U.; Carlsson, J.; et al. First-in-Human Molecular Imaging of HER2 Expression in Breast Cancer Metastases Using the 111In-ABY-025 Affibody Molecule. J. Nucl. Med. 2014, 55, 730–735. [Google Scholar] [CrossRef]
- Xu, Y.; Bai, Z.; Huang, Q.; Pan, Y.; Pan, D.; Wang, L.; Yan, J.; Wang, X.; Yang, R.; Yang, M. PET of HER2 Expression with a Novel 18FAl Labeled Affibody. J. Cancer 2017, 8, 1170–1178. [Google Scholar] [CrossRef]
- Xu, Y.; Wang, L.; Pan, D.; Yu, C.; Mi, B.; Huang, Q.; Sheng, J.; Yan, J.; Wang, X.; Yang, R.; et al. PET Imaging of a 68Ga Labeled Modified HER2 Affibody in Breast Cancers: From Xenografts to Patients. Br. J. Radiol. 2019, 92, 20190425. [Google Scholar] [CrossRef]
- Xu, Y.; Wang, L.; Pan, D.; Yan, J.; Wang, X.; Yang, R.; Li, M.; Liu, Y.; Yang, M. Synthesis of a Novel 89Zr-Labeled HER2 Affibody and Its Application Study in Tumor PET Imaging. EJNMMI Res. 2020, 10, 58. [Google Scholar] [CrossRef]
- Zhou, N.; Liu, C.; Guo, X.; Xu, Y.; Gong, J.; Qi, C.; Zhang, X.; Yang, M.; Zhu, H.; Shen, L.; et al. Impact of 68Ga-NOTA-MAL-MZHER2 PET Imaging in Advanced Gastric Cancer Patients and Therapeutic Response Monitoring. Eur. J. Nucl. Med. Mol. Imaging 2020, 48, 161–175. [Google Scholar] [CrossRef] [PubMed]
- Zahnd, C.; Pecorari, F.; Straumann, N.; Wyler, E.; Plückthun, A. Selection and Characterization of Her2 Binding-Designed Ankyrin Repeat Proteins. J. Biol. Chem. 2006, 281, 35167–35175. [Google Scholar] [CrossRef]
- Vorobyeva, A.; Schulga, A.; Konovalova, E.; Güler, R.; Löfblom, J.; Sandström, M.; Garousi, J.; Chernov, V.; Bragina, O.; Orlova, A.; et al. Optimal Composition and Position of Histidine-Containing Tags Improves Biodistribution of 99m Tc-Labeled DARPin G3. Sci. Rep. 2019, 9, 9405. [Google Scholar] [CrossRef] [PubMed]
- Vorobyeva, A.; Schulga, A.; Konovalova, E.; Güler, R.; Mitran, B.; Garousi, J.; Rinne, S.; Löfblom, J.; Orlova, A.; Deyev, S.; et al. Comparison of Tumor-targeting Properties of Directly and Indirectly Radioiodinated Designed Ankyrin Repeat Protein (DARPin) G3 Variants for Molecular Imaging of HER2. Int. J. Oncol. 2019, 54, 1209–1220. [Google Scholar] [CrossRef] [PubMed]
- Biabani Ardakani, J.; Akhlaghi, M.; Nikkholgh, B.; Hosseinimehr, S.J. Targeting and Imaging of HER2 Overexpression Tumor with a New Peptide-Based 68Ga-PET Radiotracer. Bioorg. Chem. 2021, 106, 104474. [Google Scholar] [CrossRef] [PubMed]
- Guan, S.-S.; Wu, C.-T.; Chiu, C.-Y.; Luo, T.-Y.; Wu, J.-Y.; Liao, T.-Z.; Liu, S.-H. Polyethylene Glycol-Conjugated HER2-Targeted Peptides as a Nuclear Imaging Probe for HER2-Overexpressed Gastric Cancer Detection in vivo. J. Transl. Med. 2018, 16, 168. [Google Scholar] [CrossRef] [PubMed]
- Khodadust, F.; Ahmadpour, S.; Aligholikhamseh, N.; Abedi, S.M.; Hosseinimehr, S.J. An Improved 99mTc-HYNIC-(Ser)3-LTVSPWY Peptide with EDDA/Tricine as Co-Ligands for Targeting and Imaging of HER2 Overexpression Tumor. Eur. J. Med. Chem. 2018, 144, 767–773. [Google Scholar] [CrossRef] [PubMed]
- Li, L.; Wu, Y.; Wang, Z.; Jia, B.; Hu, Z.; Dong, C.; Wang, F. SPECT/CT Imaging of the Novel HER2-Targeted Peptide Probe 99mTc-HYNIC-H6F in Breast Cancer Mouse Models. J. Nucl. Med. 2017, 58, 821–826. [Google Scholar] [CrossRef] [PubMed]
- Sabahnoo, H.; Noaparast, Z.; Abedi, S.M.; Hosseinimehr, S.J. New Small 99mTc-Labeled Peptides for HER2 Receptor Imaging. Eur. J. Med. Chem. 2017, 127, 1012–1024. [Google Scholar] [CrossRef]
- Varmira, K.; Hosseinimehr, S.J.; Noaparast, Z.; Abedi, S.M. A HER2-Targeted RNA Aptamer Molecule Labeled with 99mTc for Single-Photon Imaging in Malignant Tumors. Nucl. Med. Biol. 2013, 40, 980–986. [Google Scholar] [CrossRef]
- Wu, Y.; Li, L.; Wang, Z.; Shi, J.; Hu, Z.; Gao, S.; Miao, W.; Ma, Q.; Dong, C.; Wang, F. Imaging and Monitoring HER2 Expression in Breast Cancer during Trastuzumab Therapy with a Peptide Probe 99mTc-HYNIC-H10F. Eur. J. Nucl. Med. Mol. Imaging 2020, 47, 2613–2623. [Google Scholar] [CrossRef]
- Zhu, G.; Zhang, H.; Jacobson, O.; Wang, Z.; Chen, H.; Yang, X.; Niu, G.; Chen, X. Combinatorial Screening of DNA Aptamers for Molecular Imaging of HER2 in Cancer. Bioconjug. Chem. 2017, 28, 1068–1075. [Google Scholar] [CrossRef]
- Lockhart, A.C.; Liu, Y.; Dehdashti, F.; Laforest, R.; Picus, J.; Frye, J.; Trull, L.; Belanger, S.; Desai, M.; Mahmood, S.; et al. Phase 1 Evaluation of [(64)Cu]DOTA-Patritumab to Assess Dosimetry, Apparent Receptor Occupancy, and Safety in Subjects with Advanced Solid Tumors. Mol. Imaging Biol. 2016, 18, 446–453. [Google Scholar] [CrossRef]
- Sharp, T.L.; Glaus, C.; Fettig, N.; Hewig, A.; Ogbagabriel, S.; Freeman, D.; Beaupre, D.; Hwang, D.-R.; Welch, M.J. Pharmacological Evaluation of 64Cu—DOTA—AMG 888 (U3-1287) in Control and Tumor Bearing Mice Using Biodistribution and MicroPET Imaging. In Proceedings of the World Molecular Imaging Congress 2011, Abstract T206, San Diego, CA, USA, 7–10 September 2011. [Google Scholar]
- Menke-van der Houven van Oordt, C.W.; McGeoch, A.; Bergstrom, M.; McSherry, I.; Smith, D.A.; Cleveland, M.; Al-Azzam, W.; Chen, L.; Verheul, H.; Hoekstra, O.S.; et al. Immuno-PET Imaging to Assess Target Engagement: Experience from 89Zr-Anti-HER3 MAb (GSK2849330) in Patients with Solid Tumors. J. Nucl. Med. 2019, 60, 902–909. [Google Scholar] [CrossRef]
- Alsaid, H.; Skedzielewski, T.; Rambo, M.V.; Hunsinger, K.; Hoang, B.; Fieles, W.; Long, E.R.; Tunstead, J.; Vugts, D.J.; Cleveland, M.; et al. Non Invasive Imaging Assessment of the Biodistribution of GSK2849330, an ADCC and CDC Optimized Anti HER3 MAb, and Its Role in Tumor Macrophage Recruitment in Human Tumor-Bearing Mice. PLoS ONE 2017, 12, e0176075. [Google Scholar] [CrossRef] [PubMed]
- Terwisscha van Scheltinga, A.G.; Lub-de Hooge, M.N.; Abiraj, K.; Schröder, C.P.; Pot, L.; Bossenmaier, B.; Thomas, M.; Hölzlwimmer, G.; Friess, T.; Kosterink, J.G.; et al. ImmunoPET and Biodistribution with Human Epidermal Growth Factor Receptor 3 Targeting Antibody 89Zr-RG7116. MAbs 2014, 6, 1051–1058. [Google Scholar] [CrossRef]
- Yuan, Q.; Furukawa, T.; Tashiro, T.; Okita, K.; Jin, Z.-H.; Aung, W.; Sugyo, A.; Nagatsu, K.; Endo, H.; Tsuji, A.B.; et al. Immuno-PET Imaging of HER3 in a Model in Which HER3 Signaling Plays a Critical Role. PLoS ONE 2015, 10, e0143076. [Google Scholar] [CrossRef] [PubMed]
- Pool, M.; Kol, A.; de Jong, S.; de Vries, E.G.E.; Lub-de Hooge, M.N.; Terwisscha van Scheltinga, A.G.T. (89)Zr-MAb3481 PET for HER3 Tumor Status Assessment during Lapatinib Treatment. MAbs 2017, 9, 1–9. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Rosestedt, M.; Andersson, K.G.; Mitran, B.; Tolmachev, V.; Löfblom, J.; Orlova, A.; Ståhl, S. Affibody-Mediated PET Imaging of HER3 Expression in Malignant Tumours. Sci. Rep. 2015, 5, 15226. [Google Scholar] [CrossRef] [PubMed]
- Dahlsson Leitao, C.; Rinne, S.S.; Altai, M.; Vorontsova, O.; Dunås, F.; Jonasson, P.; Tolmachev, V.; Löfblom, J.; Ståhl, S.; Orlova, A. Evaluating the Therapeutic Efficacy of Mono- and Bivalent Affibody-Based Fusion Proteins Targeting HER3 in a Pancreatic Cancer Xenograft Model. Pharmaceutics 2020, 12, 551. [Google Scholar] [CrossRef]
- Martins, C.D.; Da Pieve, C.; Burley, T.A.; Smith, R.; Ciobota, D.M.; Allott, L.; Harrington, K.J.; Oyen, W.J.G.; Smith, G.; Kramer-Marek, G. HER3-Mediated Resistance to Hsp90 Inhibition Detected in Breast Cancer Xenografts by Affibody-Based PET Imaging. Clin. Cancer Res. 2018, 24, 1853–1865. [Google Scholar] [CrossRef]
- Warnders, F.J.; Terwisscha van Scheltinga, A.G.T.; Knuehl, C.; van Roy, M.; de Vries, E.F.J.; Kosterink, J.G.W.; de Vries, E.G.E.; Lub-de Hooge, M.N. Human Epidermal Growth Factor Receptor 3-Specific Tumor Uptake and Biodistribution of 89Zr-MSB0010853 Visualized by Real-Time and Noninvasive PET Imaging. J. Nucl. Med. 2017, 58, 1210–1215. [Google Scholar] [CrossRef] [PubMed]
- Razumienko, E.J.; Scollard, D.A.; Reilly, R.M. Small-Animal SPECT/CT of HER2 and HER3 Expression in Tumor Xenografts in Athymic Mice Using Trastuzumab Fab-Heregulin Bispecific Radioimmunoconjugates. J. Nucl. Med. 2012, 53, 1943–1950. [Google Scholar] [CrossRef] [PubMed]
- Andersson, K.G.; Rosestedt, M.; Varasteh, Z.; Malm, M.; Sandström, M.; Tolmachev, V.; Löfblom, J.; Ståhl, S.; Orlova, A. Comparative Evaluation of 111In-Labeled NOTA-conjugated Affibody Molecules for Visualization of HER3 Expression in Malignant Tumors. Oncol. Rep. 2015, 34, 1042–1048. [Google Scholar] [CrossRef] [PubMed]
- Orlova, A.; Malm, M.; Rosestedt, M.; Varasteh, Z.; Andersson, K.; Selvaraju, R.K.; Altai, M.; Honarvar, H.; Strand, J.; Ståhl, S.; et al. Imaging of HER3-Expressing Xenografts in Mice Using a (99m)Tc(CO) 3-HEHEHE-Z HER3:08699 Affibody Molecule. Eur. J. Nucl. Med. Mol. Imaging 2014, 41, 1450–1459. [Google Scholar] [CrossRef]
- Rinne, S.S.; Dahlsson Leitao, C.; Gentry, J.; Mitran, B.; Abouzayed, A.; Tolmachev, V.; Ståhl, S.; Löfblom, J.; Orlova, A. Increase in Negative Charge of 68Ga/Chelator Complex Reduces Unspecific Hepatic Uptake but Does Not Improve Imaging Properties of HER3-Targeting Affibody Molecules. Sci. Rep. 2019, 9, 17710. [Google Scholar] [CrossRef]
- Rosestedt, M.; Andersson, K.G.; Mitran, B.; Rinne, S.S.; Tolmachev, V.; Löfblom, J.; Orlova, A.; Ståhl, S. Evaluation of a Radiocobalt-Labelled Affibody Molecule for Imaging of Human Epidermal Growth Factor Receptor 3 Expression. Int. J. Oncol. 2017, 51, 1765–1774. [Google Scholar] [CrossRef]
- Rinne, S.S.; Dahlsson Leitao, C.; Saleh-Nihad, Z.; Mitran, B.; Tolmachev, V.; Ståhl, S.; Löfblom, J.; Orlova, A. Benefit of Later-Time-Point PET Imaging of HER3 Expression Using Optimized Radiocobalt-Labeled Affibody Molecules. Int. J. Mol. Sci. 2020, 21, 1972. [Google Scholar] [CrossRef]
- Da Pieve, C.; Allott, L.; Martins, C.D.; Vardon, A.; Ciobota, D.M.; Kramer-Marek, G.; Smith, G. Efficient [18F]AlF Radiolabeling of ZHER3:8698 Affibody Molecule for Imaging of HER3 Positive Tumors. Bioconjug. Chem. 2016, 27, 1839–1849. [Google Scholar] [CrossRef]
- Rosestedt, M.; Andersson, K.G.; Rinne, S.S.; Leitao, C.D.; Mitran, B.; Vorobyeva, A.; Ståhl, S.; Löfblom, J.; Tolmachev, V.; Orlova, A. Improved Contrast of Affibody-Mediated Imaging of HER3 Expression in Mouse Xenograft Model through Co-Injection of a Trivalent Affibody for in Vivo Blocking of Hepatic Uptake. Sci. Rep. 2019, 9, 6779. [Google Scholar] [CrossRef]
- Dahlsson Leitao, C.; Rinne, S.S.; Mitran, B.; Vorobyeva, A.; Andersson, K.G.; Tolmachev, V.; Ståhl, S.; Löfblom, J.; Orlova, A. Molecular Design of HER3-Targeting Affibody Molecules: Influence of Chelator and Presence of HEHEHE-Tag on Biodistribution of 68Ga-Labeled Tracers. Int. J. Mol. Sci. 2019, 20, 1080. [Google Scholar] [CrossRef]
- Larimer, B.M.; Phelan, N.; Wehrenberg-Klee, E.; Mahmood, U. Phage Display Selection, In Vitro Characterization, and Correlative PET Imaging of a Novel HER3 Peptide. Mol. Imaging Biol. 2017, 20, 300–308. [Google Scholar] [CrossRef] [PubMed]
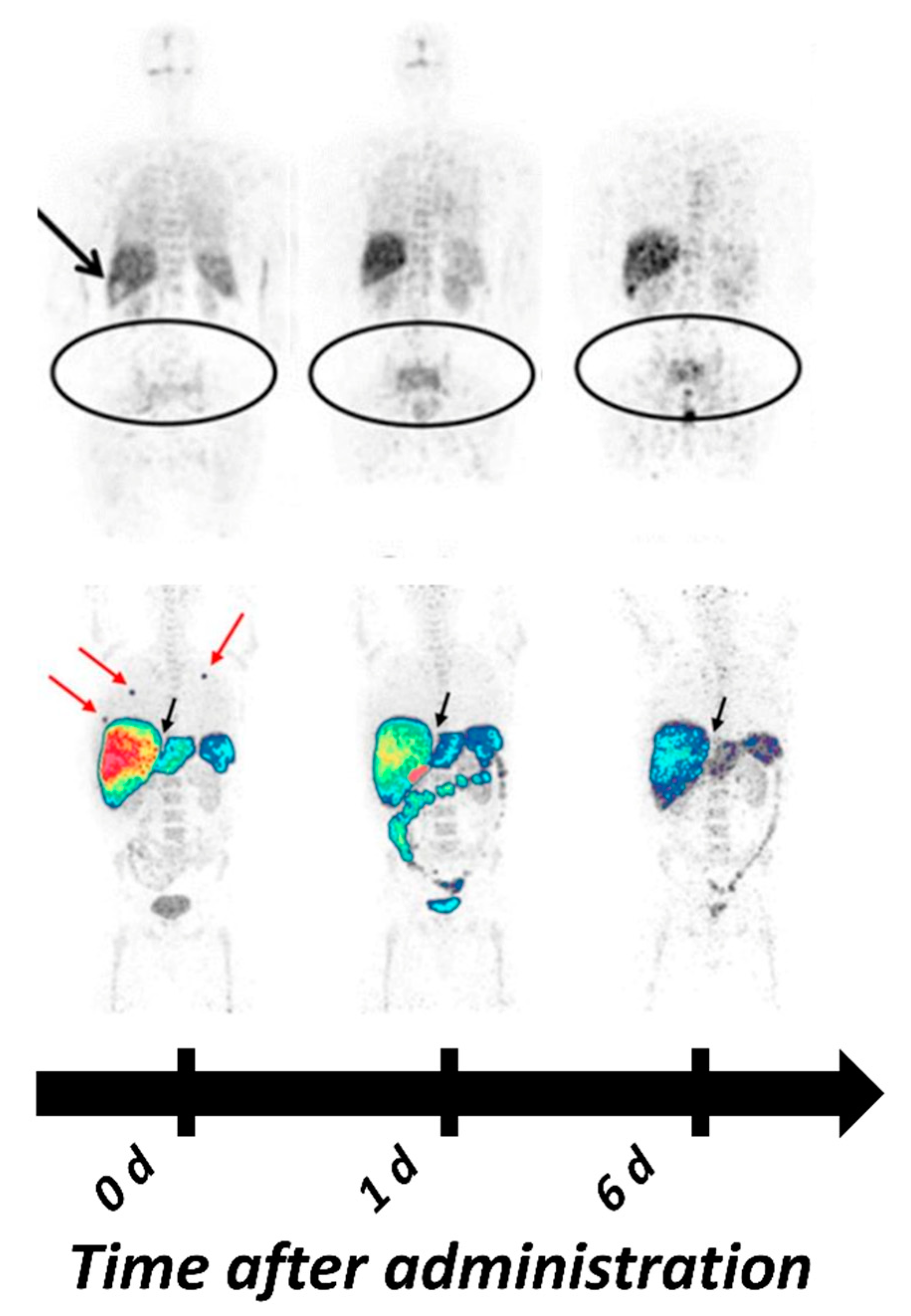

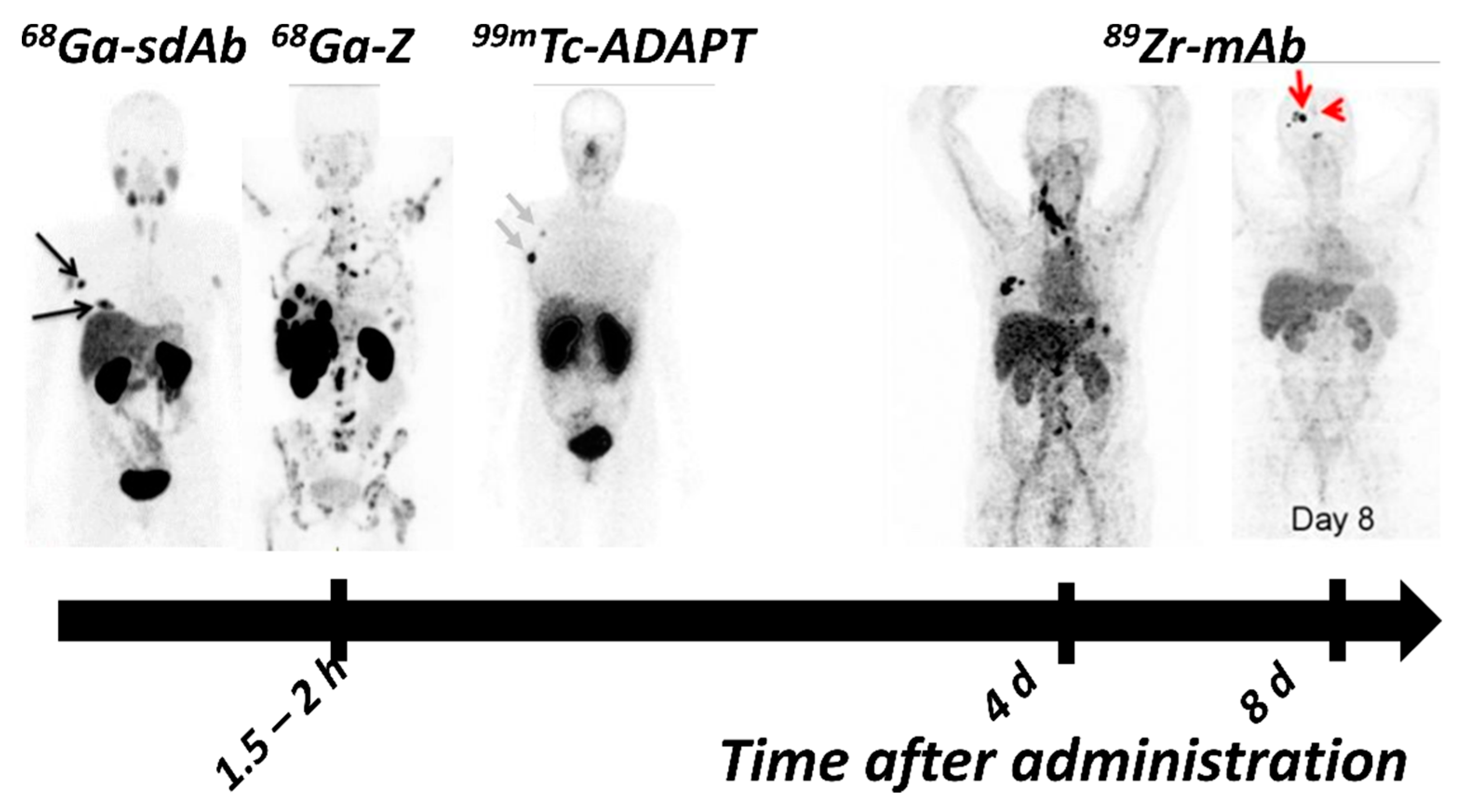

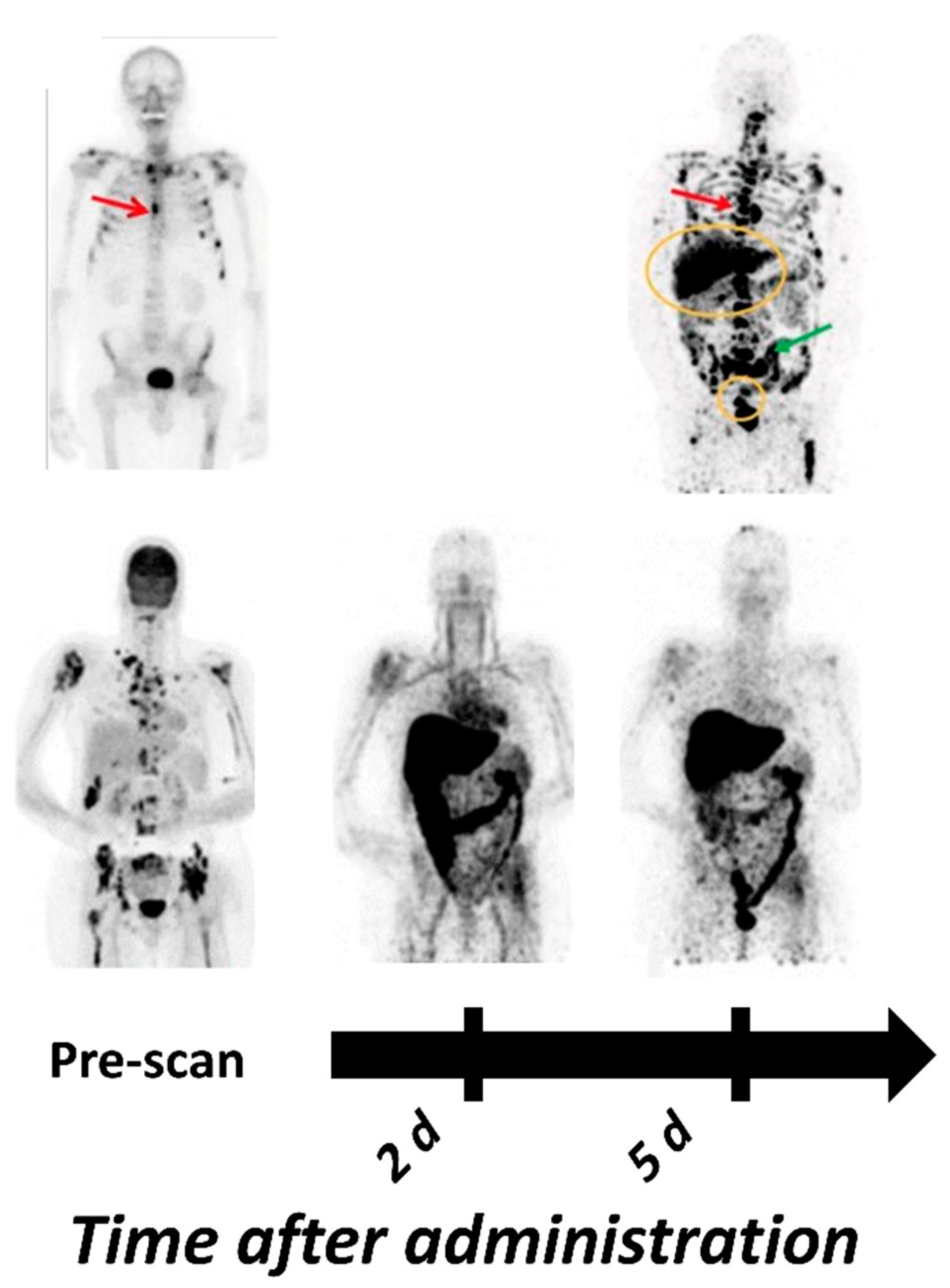
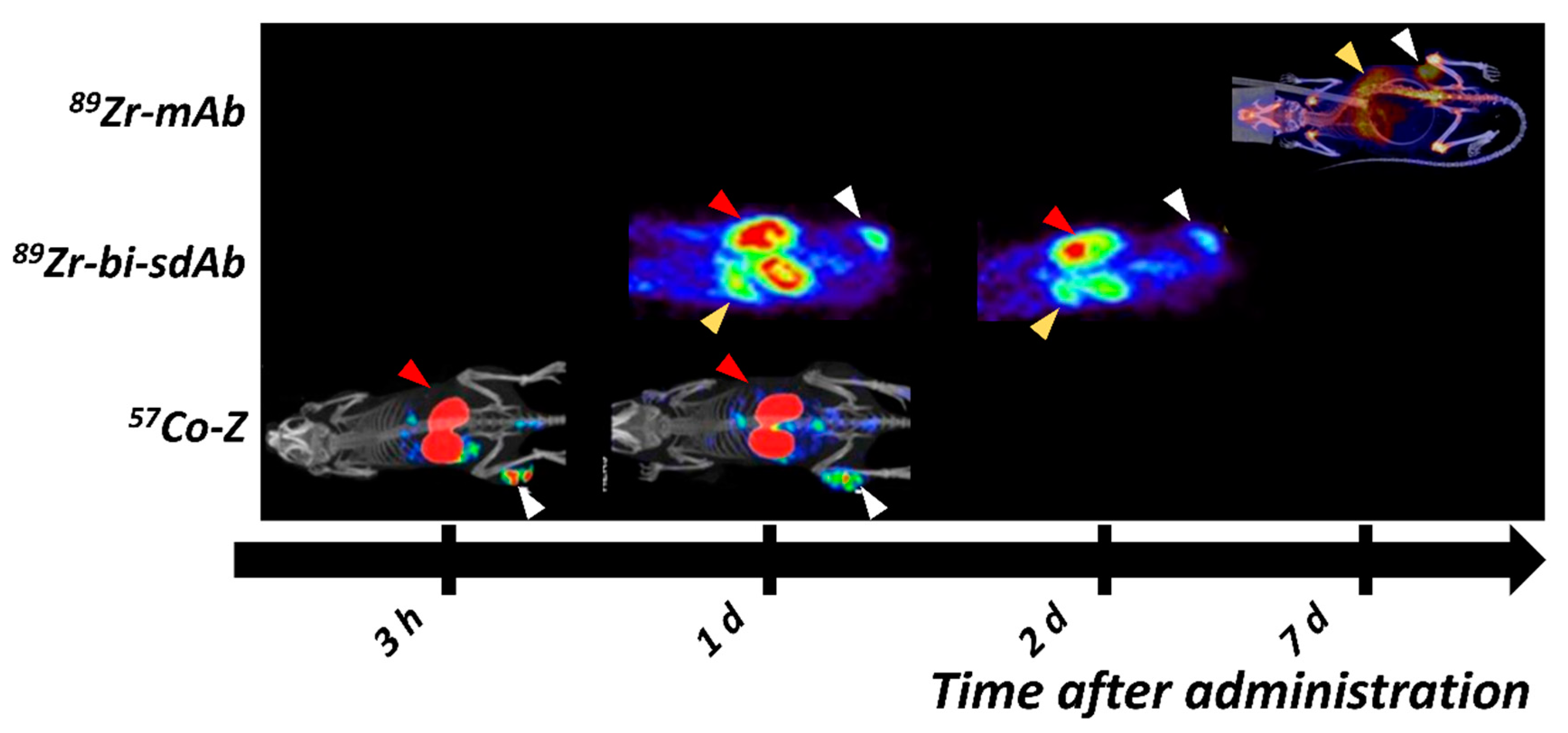
| Tracer | PET/ SPECT | Type of Molecule | Key Points | Reference |
|---|---|---|---|---|
| Clinical Studies | ||||
| [89Zr]Zr-cetuximab | PET | Antibody | Phase II/II Specific tumor uptake in patients Optimal imaging day six | [75,111,130] |
| [89Zr]Zr-panitumumab | PET | Antibody | Tumors could not be visualized, potentially be due to location in areas with high background due metabolic tracer accumulation (liver, abdomen) | [81] |
| [89Zr]Zr-nimotuzumab | PET | Antibody | Currently in Phase I/II studies Estimated absorbed was lower than dose for [89Zr]Zr-DFO-panitumumab | [123], NCT04235114 |
| Preclinical Studies | ||||
| [64Cu]Cu-cetuximab | PET | Antibody | Can visualize HER1 expression 48 h pi xenografts Correlation between uptake, expression and predicative power still unclear | [114,115] |
| [64Cu]Cu-panitumumab | PET | Antibody | HER1 expression in three different HNSCC xenografts could be visualized Inverse relation between tumor uptake and HER1 expression, possibly related to vessel density, vascular permeability and binding site barrier | [128] |
| [111In]In-panitumumab | SPECT | Antibody | [111In]In-CHX”-DTPA- panitumumab has slightly faster blood clearance compared with [89Zr]Zr-DFO-panitumumab Conjugation of an MCP with 13 DOTAs to reduce liver and spleen uptake surprisingly increased hepatic uptake significantly | [121] |
| [64Cu]Cu-NOTA- panitumumab F(ab′)2 | PET | Antibody fragment | Faster clearance compared with full length panitumumab Highest uptake in xenografts 48 h pi Tumor-to-blood ratios of 5–9 at 48 h pi | [131,161] |
| [111In]In-cetuximab F(ab′)2 | SPECT | Antibody fragment | Provides significantly better tumor-to-blood ratios than cetuximab at early time points Highest uptake in xenografts 24 h pi Correlation between uptake and expression in xenografts Therapy response in mice correlated with tracer uptake | [87,132,162] |
| [99mTc]Tc-Pm-Fab-His6 | SPECT | Antibody fragment | Tumor uptake and tumor-to-blood ratios similar to those of [64Cu]Cu-NOTA-panitumumab-F(ab′)2 24 h and 48 h pi Clear visualization of a panel of HER1 expressing xenografts using SPECT | [134] |
| [99mTc]Tc-D10 | SPECT | sdAb | Detection of HER1 expressing lesions 45 min pi Tumor-to-blood ratio comparable to that of F(ab′)2 fragments of cetuximab and panitumumab | [135] |
| [68Ga]Ga/[89Zr]Zr-Df-Bz-NCS-7D12 | PET | sdAb | Higher tumor uptake than uptake of [ 99m Tc]Tc-D10 Good visualization on PET 1h pi | [137] |
| [89Zr]Zr-DFO-ZEGFR:2377 | PET | Affibody | Tumor uptake 3 h pi exceeded uptake of [89 Zr]Zr-DFO-cetuximab at 48 h pi Tumor-to-tissue contrast at 3 h and 24 h was higher than [89 Zr]Zr-DFO-cetuximab except from tumor-to-kidney | [140] |
| [55/57Co]Co-DOTA- ZEGFR:2377 | PET | Affibody | Highest tumor-to-tissue contrast compared with all other studied affibody-based tracers | [105] |
| [68Ga]Ga-DFO-ZEGFR:2377 | PET | Affibody | For imaging at 3 h pi Compared with other affibody molecules against HER1 the imaging contrast was inferior only to the radiocobalt conjugate, but 68Ga is more available than 55Co | [142] |
| [64Cu]Cu-FnEI3.4.3′ | PET | Fibronectin binding domain | Good tumor visualization on PET 1h pi No cross-reactivity with mErbB1 | [149] |
| Tracer | PET/ SPECT | Type of Molecule | Key Points | Reference |
|---|---|---|---|---|
| Clinical Studies | ||||
| [64Cu]Cu-trastuzumab [89Zr]Zr-trastuzumab | PET | Antibody | [89Zr]Zr-trastuzumab was the first clinically studied PET-tracer for imaging of HER2 Successful visualization of HER2 positive lesions in patients with BC and esophagogastric cancer Optimal imaging 4–6 d pi (89Zr) or 48 h pi (64Cu) | [74,179,180,181,185,187] |
| [89Zr]Zr-pertuzumab | PET | Antibody | Successful visualization of HER2 positive primary tumor and metastases in BC patients [89Zr]Zr-pertuzumab appeared to have slightly higher uptake than [89Zr]Zr-trastuzumab in liver, kidney, spleen, and lung | [182,184] |
| [111In]In-CHX-A″-DTPA trastuzumab | SPECT | Antibody | Phase 0 demonstrated safety No data on tumor targeting available | [177] |
| [68Ga]Ga-DOTA-F(ab′)2- trastuzumab | PET | Antibody fragment | HER2-positive lesion were detected in only half the patients with known HER2-positive disease In xenografted mice, the tumor uptake was proportional to HER2 expression | [206,209] |
| [68Ga]Ga-HER2-Nanobody (also 2Rs15d) | PET | sdAb | Well tolerated administration Imaging of HER2 expression 60–90 min pi 90 min pi was suggested as more suitable time point for imaging | [223] |
| [131I]I-GMIB-Anti-HER2- VHH1 (also GMIB-2Rs15d) | SPECT | sdAb | Phase I trial showed uptake in patients with lesions >3 cm Visualization of smaller lesions was difficult, supposedly due to PVE | [219] |
| [68Ga]Ga-DOTA-ABY025 | PET | Affibody | Primary tumors and metastases (even hepatic) could be visualized Correlation between SUV in lesions with confirmed HER2 IHC scores Multicenter Phase II/III study currently underway (NCT03655353) | [78,233] |
| [68Ga]Ga-NOTA-MAL-Cys-MZHER2:342 | PET | Affibody | Optimal imaging contrast was achieved 2 h pi Reporting 100% specificity, 55% sensitivity at an SUV cutoff at 6.6 | [237] |
| [99mTc]Tc-ADAPT6 | SPECT | ADAPT | First ever clinical study with ADAPT Well-tolerated administration An optimal injected protein dose (500 µg) was able to differentiate between positive and negative lesions | [169] |
| [99mTc]Tc-(HE)3-G3 | SPECT | DARPin | First clinical trial with HER2 targeting DARPin to assess safety, distribution, and dosimetry in patients with primary breast cancer is currently ongoing | NCT04277338 |
| Preclinical Studies | ||||
| [64Cu]Cu-NOTA-pertuzumab F(ab′)2 | PET | Antibody fragment | Trastuzumab-induced changes in HER2 could be detected Sensitivity increased from 24 to 48 h pi | [205] |
| [89Zr]Zr-Df-Fab-PAS200 [124I]I-Df-Fab-PAS200 | PET | Antibody fragment | Despite lower tumor uptake of the 124I-labeled variant both tracers showed comparable contrast and good visualization of xenografts 24 h pi Administration of 89Zr-labeled variant in one patient could detect lesions 24 h pi | [207,208] |
| [123/124/125I]I-PIB-G3-(HE)3 | PET/ SPECT | DARPin | Indirect iodination using SPIB and introduction of (HE)3-tag improved tumor-to-tissue ratios Another promising variant for clinical translation among the explored DARPin variants | [86,224,239] |
| [111In]In-DTPA-AHNP-PEG | SPECT | Peptide | Showed higher tumor uptake, retention and tumor-to-tissue ratios than other reported HER2 targeting peptides Good visualization of gastric cancer xenografts up to 48 h pi | [242] |
| Tracer | PET/ SPECT | Type of Molecule | Key Points | Reference |
|---|---|---|---|---|
| Clinical Studies | ||||
| [64Cu]Cu-patritumab (also AMG-888 or U3-1287) | PET | Antibody | Clinical/Phase 1 terminated after 11 patients due to low uptake in HER3 expressing lesions | [249] |
| [89Zr]Zr-GSK 2849330 | PET | Antibody | Tumor uptake was dependent on the injected mass thus indicating good potential for assessment of target occupancy and as a tool for dose selection | [251] |
| [89Zr]Zr-lumretuzumab (also RG-7116) | PET | Antibody | Detected 67.6% of lesions larger than 10 mm No liver metastases could be detected due to high uptake in healthy liver tissue | [73] |
| Preclinical Studies | ||||
| [89Zr]Zr-MSB0010853 | PET | Biparatopic sdAb construct | Maximum tumor uptake 96 h pi was comparable with that of affibody molecules 3–24 h piHigher tumor-to-blood ratios compared with preclinical data on [89Zr]Zr-lumretuzumab and [64Cu]Cu-patritumab | [259] |
| [55/57Co]Co-(HE)3- ZHER3:08698-DOTA | PET/ SPECT | Affibody | Reported best tumor-to-liver contrast among radiometal labeled anti-HER3 affibody molecules | [265] |
| [89Zr]Zr-DFO-ZHER3:08698 | PET | Affibody | Could image changes in HER3 expression induced by HSP90 therapy | [258] |
| [68Ga]Ga-HER3P1 | PET | Peptide | Uptake in xenografts <1 %ID/g due to rapid wash out and low affinity | [269] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rinne, S.S.; Orlova, A.; Tolmachev, V. PET and SPECT Imaging of the EGFR Family (RTK Class I) in Oncology. Int. J. Mol. Sci. 2021, 22, 3663. https://doi.org/10.3390/ijms22073663
Rinne SS, Orlova A, Tolmachev V. PET and SPECT Imaging of the EGFR Family (RTK Class I) in Oncology. International Journal of Molecular Sciences. 2021; 22(7):3663. https://doi.org/10.3390/ijms22073663
Chicago/Turabian StyleRinne, Sara S., Anna Orlova, and Vladimir Tolmachev. 2021. "PET and SPECT Imaging of the EGFR Family (RTK Class I) in Oncology" International Journal of Molecular Sciences 22, no. 7: 3663. https://doi.org/10.3390/ijms22073663
APA StyleRinne, S. S., Orlova, A., & Tolmachev, V. (2021). PET and SPECT Imaging of the EGFR Family (RTK Class I) in Oncology. International Journal of Molecular Sciences, 22(7), 3663. https://doi.org/10.3390/ijms22073663








