Emerging Trends in Pullulan-Based Antimicrobial Systems for Various Applications
Abstract
:1. Introduction
2. Pullulan: Biosynthesis and Production
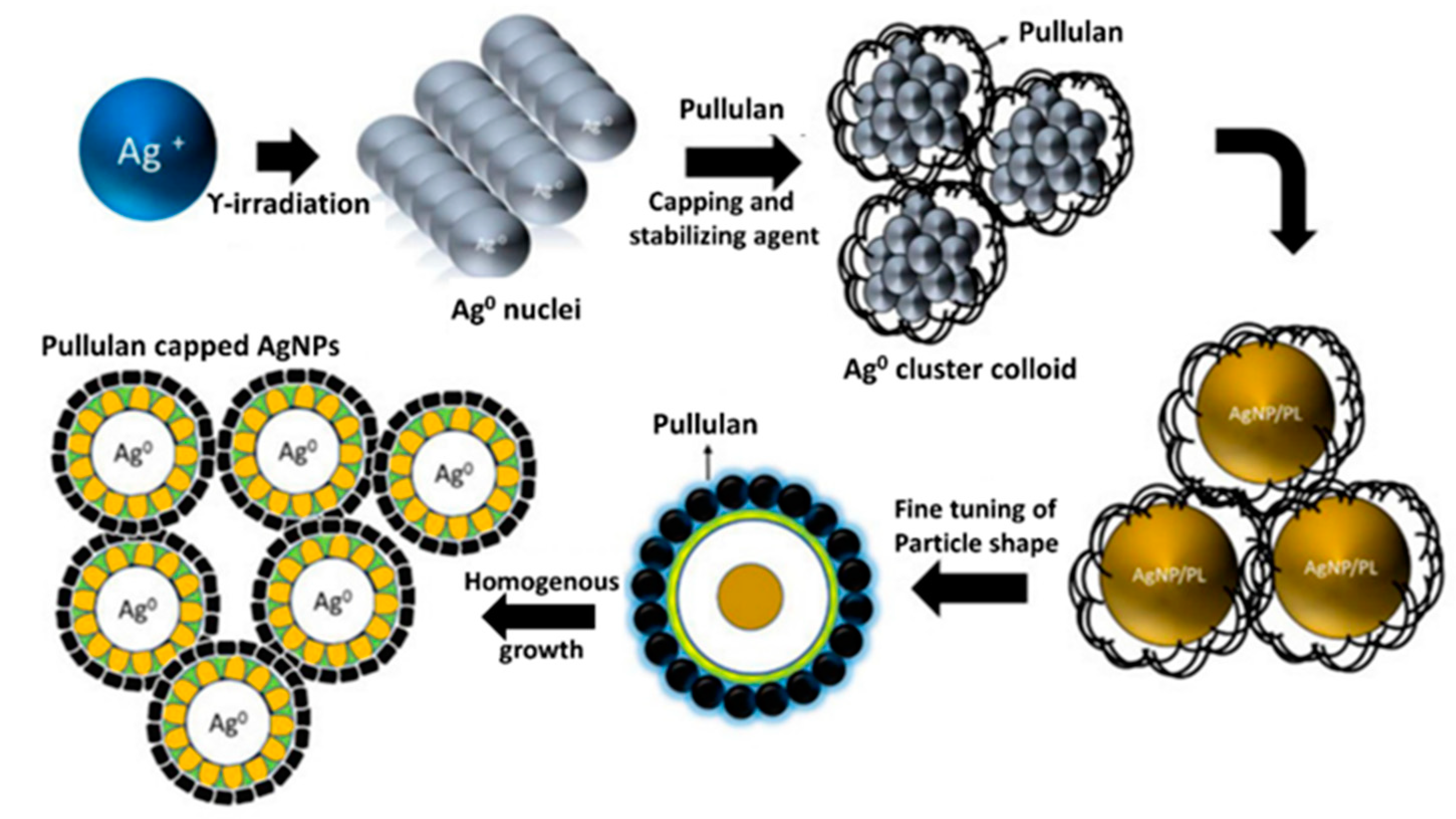
3. Antimicrobial Potential of Pullulan-Mediated Bionanoparticles
4. Pullulan-Based Antimicrobial Nanocomposites
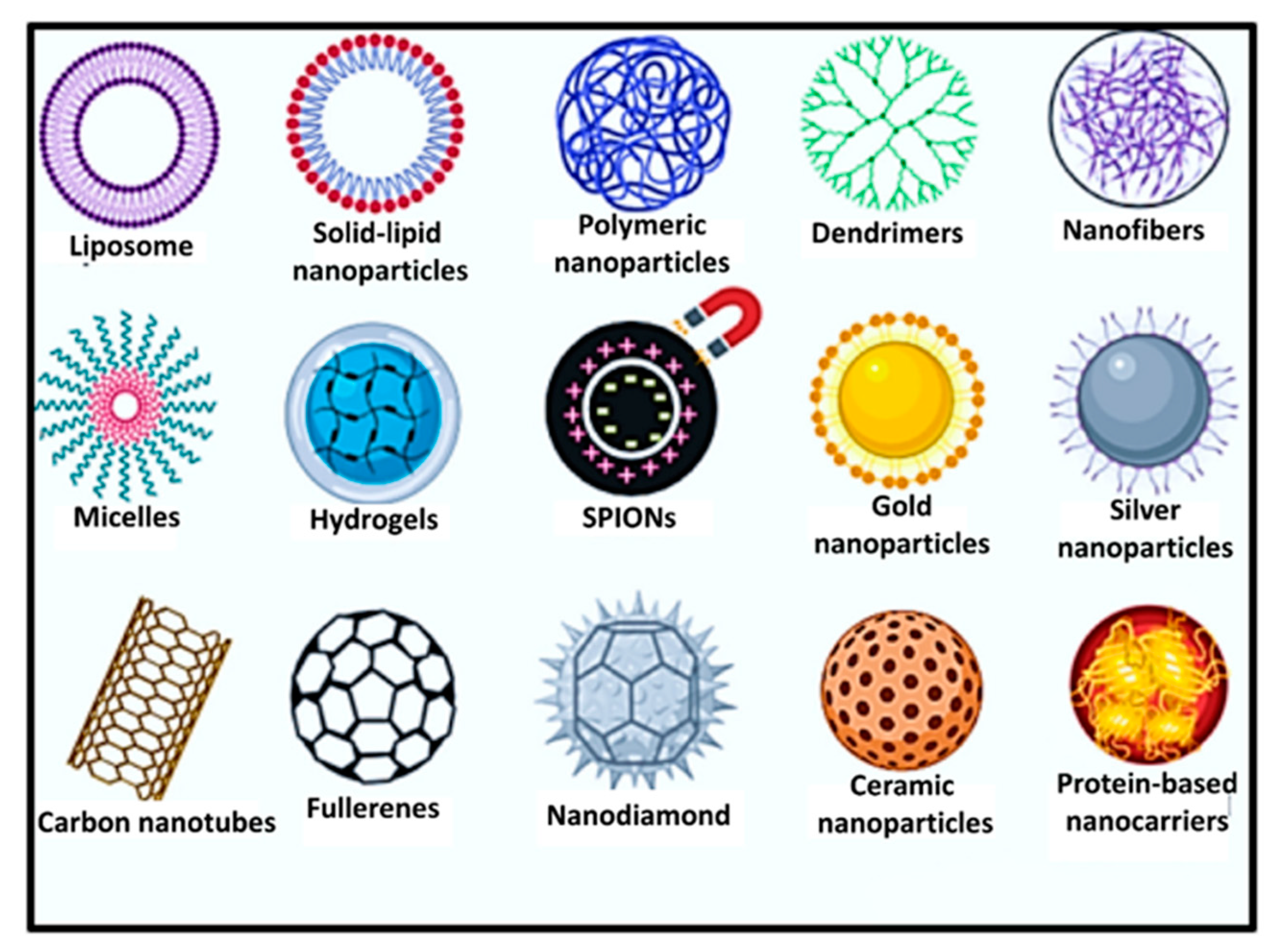
5. Pullulan-Based Nanomaterials in Delivery of Biomolecules/Drugs
6. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Yang, X.; Ye, W.; Qi, Y.; Ying, Y.; Xia, Z. Overcoming Multidrug Resistance in Bacteria Through Antibiotics Delivery in Surface-Engineered Nano-Cargos: Recent Developments for Future Nano-Antibiotics. Front. Bioeng. Biotechnol. 2021, 9, 696514. [Google Scholar] [CrossRef] [PubMed]
- Singh, R.S.; Kaur, N.; Singh, D.; Kennedy, J.F. Investigating aqueous phase separation of pullulan from Aureobasidium pullulans and its characterization. Carbohydr. Polym. 2019, 223, 115103. [Google Scholar] [CrossRef] [PubMed]
- Singh, R.S.; Saini, G.K. Biosynthesis of pullulan and its applications in food and pharmaceutical industry. In Microorganisms in Sustainable Agriculture and Biotechnology; Satyanarayana, T., Johri, B., Prakash, A., Eds.; Springer: Dordrecht, The Netherlands, 2012; pp. 509–553. [Google Scholar] [CrossRef]
- Bauer, R. Physiology of Dematium pullulans de Bary. Zentralbl. Bacteriol. Parasitenkd. Infektionskr. Hyg. Abt. 2 1938, 98, 133–167. [Google Scholar]
- Liu, N.-N.; Chi, Z.; Liu, G.-L.; Chen, T.-J.; Jiang, H.; Hu, Z.; Chi, Z.-M. α-Amylase, glucoamylase and isopullulanase determine molecular weight of pullulan produced by Aureobasidium melanogenum P16. Int. J. Biol. Macromol. 2018, 117, 727–734. [Google Scholar] [CrossRef]
- Luís, Â.; Ramos, A.; Domingues, F. Pullulan Films Containing Rockrose Essential Oil for Potential Food Packaging Applications. Antibiotics 2020, 9, 681. [Google Scholar] [CrossRef] [PubMed]
- Kraśniewska, K.; Pobiega, K.; Gniewosz, M. Pullulan—Biopolymer with Potential for Use as Food Packaging. Int. J. Food Eng. 2019, 15, 20190030. [Google Scholar] [CrossRef]
- Coltelli, M.-B.; Danti, S.; De Clerck, K.; Lazzeri, A.; Morganti, P. Pullulan for Advanced Sustainable Body- and Skin-Contact Applications. J. Funct. Biomater. 2020, 11, 20. [Google Scholar] [CrossRef] [Green Version]
- Luís, Â.; Ramos, A.; Domingues, F. Pullulan–Apple Fiber Biocomposite Films: Optical, Mechanical, Barrier, Antioxidant and Antibacterial Properties. Polymers 2021, 13, 870. [Google Scholar] [CrossRef]
- Emam, H.E.; Ahmed, H.B. Antitumor/antiviral carbon quantum dots based on carrageenan and pullulan. Int. J. Biol. Macromol. 2020, 170, 688–700. [Google Scholar] [CrossRef]
- Soto, K.M.; Hernández-Iturriaga, M.; Loarca-Piña, G.; Luna-Barcenas, G.; Mendoza, S. Antimicrobial effect of nisin electrospun amaranth: Pullulan nanofibers in apple juice and fresh cheese. Int. J. Food Microbiol. 2019, 295, 25–32. [Google Scholar] [CrossRef]
- Li, S.; Yi, J.; Yu, X.; Wang, Z.; Wang, L. Preparation and characterization of pullulan derivative/chitosan composite film for potential antimicrobial applications. Int. J. Biol. Macromol. 2020, 148, 258–264. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.H.; Jeong, D.; Kanmani, P. Study on physical and mechanical properties of the biopolymer/silver based active nanocomposite films with antimicrobial activity. Carbohydr. Polym. 2019, 224, 115159. [Google Scholar] [CrossRef] [PubMed]
- Gniewosz, M.; Kraśniewska, K.; Woreta, M.; Kosakowska, O. Antimicrobial Activity of a Pullulan-Caraway Essential Oil Coating on Reduction of Food Microorganisms and Quality in Fresh Baby Carrot. J. Food Sci. 2013, 78, M1242–M1248. [Google Scholar] [CrossRef]
- Pinto, R.; Almeida, A.; Fernandes, S.C.; Freire, C.; Silvestre, A.; Neto, C.P.; Trindade, T. Antifungal activity of transparent nanocomposite thin films of pullulan and silver against Aspergillus niger. Colloids Surf. B Biointerfaces 2013, 103, 143–148. [Google Scholar] [CrossRef]
- Hassan, A.H.; Cutter, C.N. Development and evaluation of pullulan-based composite antimicrobial films (CAF) incorporated with nisin, thymol and lauric arginate to reduce foodborne pathogens associated with muscle foods. Int. J. Food Microbiol. 2020, 320, 108519. [Google Scholar] [CrossRef]
- Sugumaran, K.R.; Ponnusami, V. Review on production, downstream processing and characterization of microbial pullulan. Carbohydr. Polym. 2017, 173, 573–591. [Google Scholar] [CrossRef]
- Key, S.W.C.; Dailin, D.J.; Selvamani, S.; Malek, R.A.; Sukmawati, D.; Enshasy, H.E. Pullulan production in submerged cultivation: A review. J. Crit. Rev. 2020, 18, 7. [Google Scholar]
- Simon, L.; Caye-Vaugien, C.; Bouchonneau, M. Relation between pullulan production, morphological state and growth conditions in Aureobasidium pullulans: New observations. J. Gen. Microbiol. 1993, 139, 979–985. [Google Scholar] [CrossRef] [Green Version]
- Chen, T.-J.; Chi, Z.; Jiang, H.; Liu, G.-L.; Hu, Z.; Chi, Z.-M. Cell wall integrity is required for pullulan biosynthesis and glycogen accumulation in Aureobasidium melanogenum P16. Biochim. Biophys. Acta (BBA)-Gen. Subj. 2018, 1862, 1516–1526. [Google Scholar] [CrossRef] [PubMed]
- Wei, X.; Liu, G.-L.; Jia, S.-L.; Chi, Z.; Hu, Z.; Chi, Z.-M. Pullulan biosynthesis and its regulation in Aureobasidium spp. Carbohydr. Polym. 2020, 251, 117076. [Google Scholar] [CrossRef]
- Chen, G.; Wang, J.; Su, Y.; Zhu, Y.; Zhang, G.; Zhao, H.; Liu, H.; Yang, Y.; Nian, R.; Zhang, H.; et al. Pullulan production from synthetic medium by a new mutant of Aureobasidium pullulans. Prep. Biochem. Biotechnol. 2017, 47, 963–969. [Google Scholar] [CrossRef]
- Chi, Z.; Wang, F.; Chi, Z.; Yue, L.; Liu, G.; Zhang, T. Bioproducts from Aureobasidium pullulans, a biotechnologically important yeast. Appl. Microbiol. Biotechnol. 2009, 82, 793–804. [Google Scholar] [CrossRef] [PubMed]
- Cheng, K.-C.; Demirci, A.; Catchmark, J.M. Pullulan: Biosynthesis, production, and applications. Appl. Microbiol. Biotechnol. 2011, 92, 29–44. [Google Scholar] [CrossRef] [PubMed]
- Prasongsuk, S.; Lotrakul, P.; Ali, I.; Bankeeree, W.; Punnapayak, H. The current status of Aureobasidium pullulans in biotechnology. Folia Microbiol. 2017, 63, 129–140. [Google Scholar] [CrossRef]
- Hamidi, M.; Kennedy, J.F.; Khodaiyan, F.; Mousavi, Z.; Hosseini, S.S. Production optimization, characterization and gene expression of pullulan from a new strain of Aureobasidium pullulans. Int. J. Biol. Macromol. 2019, 138, 725–735. [Google Scholar] [CrossRef] [PubMed]
- Fraser, C.G.; Jennings, H.J. A Glucan from Tremella mesenterica NRRL-Y6158. Can. J. Chem. 1971, 49, 1804–1807. [Google Scholar] [CrossRef] [Green Version]
- Reis, R.A.; Tischer, C.A.; Gorin, P.A.; Iacomini, M. A new pullulan and a branched (1→3)-, (1→6)-linked β-glucan from the lichenised ascomyceteTeloschistes flavicans. FEMS Microbiol. Lett. 2002, 210, 1–5. [Google Scholar] [CrossRef]
- Forabosco, A.; Bruno, G.; Sparapano, L.; Liut, G.; Marino, D.; Delben, F. Pullulans produced by strains of Cryphonectria parasitica—I. Production and characterisation of the exopolysaccharides. Carbohydr. Polym. 2006, 63, 535–544. [Google Scholar] [CrossRef]
- Waksman, N.; de Lederkremer, R.M.; Cerezo, A.S. The structure of an α-D-glucan from Cyttaria harioti Fischer. Carbohydr. Res. 1977, 59, 505–515. [Google Scholar] [CrossRef]
- Oliva, E.M.; Cirelli, A.F.; de Lederkremer, R.M. Characterization of a pullulan in Cyttaria darwinii. Carbohydr. Res. 1986, 158, 262–267. [Google Scholar] [CrossRef]
- Money, N.P. Fungi and Biotechnology. In Fungi, 3rd ed.; Elsevier: Amsterdam, The Netherlands, 2015; pp. 401–424. [Google Scholar]
- Mina, M.; Tsaltas, D. Contribution of yeast in wine aroma and flavour. In Yeast-Industrial Applications; Morata, A., Loira, I., Eds.; InTech: Rijeka, Croatia, 2017. [Google Scholar] [CrossRef] [Green Version]
- Mishra, B.; Varjani, S. Evaluation of pullulan production by a newly isolated Micrococcus luteus. Indian J. Exp. Biol. 2019, 57, 813–820. [Google Scholar]
- Rühmann, B.; Schmid, J.; Sieber, V. High throughput exopolysaccharide screening platform: From strain cultivation to monosaccharide composition and carbohydrate fingerprinting in one day. Carbohydr. Polym. 2015, 122, 212–220. [Google Scholar] [CrossRef] [Green Version]
- Gniewosz, M.; Sobczak, E. Możliwości wykorzystania Aureobasidium pullulans i pullulanu w biotechnologii żywności. Biotechnologia 1999, 45, 2. [Google Scholar]
- Duan, X.; Chi, Z.; Wang, L.; Wang, X. Influence of different sugars on pullulan production and activities of α-phosphoglucose mutase, UDPG-pyrophosphorylase and glucosyltransferase involved in pullulan synthesis in Aureobasidium pullulans Y68. Carbohydr. Polym. 2008, 73, 587–593. [Google Scholar] [CrossRef] [PubMed]
- Cheng, K.-C.; Demirci, A.; Catchmark, J.M. Evaluation of Medium Composition and Fermentation Parameters on Pullulan Production by Aureobasidium pullulans. Food Sci. Technol. Int. 2011, 17, 99–109. [Google Scholar] [CrossRef] [PubMed]
- Wang, Q.-Q.; Lu, Y.; Ren, Z.-Y.; Chi, Z.; Liu, G.-L.; Chi, Z.-M. CreA is directly involved in pullulan biosynthesis and regulation of Aureobasidium melanogenum P16. Curr. Genet. 2016, 63, 471–485. [Google Scholar] [CrossRef] [PubMed]
- Chi, Z.-M.; Li, J.-F.; Wang, X.-H.; Yao, S.-M. Inositol and Phosphatidylinositol Mediated Glucose Derepression, Gene Expression and Invertase Secretion in Yeasts. Acta Biochim. Biophys. Sin. 2004, 36, 443–449. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rødkaer, S.V.; Faergeman, N.J. Glucose- and nitrogen sensing and regulatory mechanisms inSaccharomyces cerevisiae. FEMS Yeast Res. 2014, 14, 683–696. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Leathers, T.D. Biotechnological production and applications of pullulan. Appl. Microbiol. Biotechnol. 2003, 62, 468–473. [Google Scholar] [CrossRef] [PubMed]
- Youssef, F.; Roukas, T.; Biliaderis, C. Pullulan production by a non-pigmented strain of Aureobasidium pullulans using batch and fed-batch culture. Process Biochem. 1999, 34, 355–366. [Google Scholar] [CrossRef]
- Roukas, T. Pullulan production from brewery wastes by Aureobasidium pullulans. World J. Microbiol. Biotechnol. 1999, 15, 447–450. [Google Scholar] [CrossRef]
- Göksungur, Y.; Dağbağlı, S.; Uçan, A.; Güvenç, U. Optimization of pullulan production from synthetic medium by Aureobasidium pullulans in a stirred tank reactor by response surface methodology. J. Chem. Technol. Biotechnol. 2005, 80, 819–827. [Google Scholar] [CrossRef]
- Wu, S.; Jin, Z.; Tong, Q.; Chen, H. Sweet potato: A novel substrate for pullulan production by Aureobasidium pullulans. Carbohydr. Polym. 2009, 76, 645–649. [Google Scholar] [CrossRef]
- An, C.; Ma, S.-J.; Chang, F.; Xue, W.-J. Efficient production of pullulan by Aureobasidium pullulans grown on mixtures of potato starch hydrolysate and sucrose. Braz. J. Microbiol. 2016, 48, 180–185. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Srikanth, S.; Swathi, M.; Tejaswini, M.; Sharmila, G.; Muthukumaran, C.; Jaganathan, M.; Tamilarasan, K. Statistical optimization of molasses based exopolysaccharide and biomass production by Aureobasidium pullulans MTCC 2195. Biocatal. Agric. Biotechnol. 2013, 3, 7–12. [Google Scholar] [CrossRef]
- Chen, L.; Chi, Z.; Liu, G.-L.; Xue, S.-J.; Wang, Z.; Hu, Z.; Chi, Z.-M. Improved pullulan production by a mutant of Aureobasidium melanogenum TN3-1 from a natural honey and capsule shell preparation. Int. J. Biol. Macromol. 2019, 141, 268–277. [Google Scholar] [CrossRef]
- Li, B.-X.; Zhang, N.; Peng, Q.; Yin, T.; Guan, F.-F.; Wang, G.-L.; Li, Y. Production of pigment-free pullulan by swollen cell in Aureobasidium pullulans NG which cell differentiation was affected by pH and nutrition. Appl. Microbiol. Biotechnol. 2009, 84, 293–300. [Google Scholar] [CrossRef]
- Kachhawa, D.K.; Bhattacharjee, P.; Singhal, R.S. Studies on downstream processing of pullulan. Carbohydr. Polym. 2003, 52, 25–28. [Google Scholar] [CrossRef]
- Chen, X.; Wang, Q.-Q.; Liu, N.-N.; Liu, G.-L.; Chi, Z.; Chi, Z.-M. A glycosyltransferase gene responsible for pullulan biosynthesis in Aureobasidium melanogenum P16. Int. J. Biol. Macromol. 2017, 95, 539–549. [Google Scholar] [CrossRef]
- Shingel, K.I. Current knowledge on biosynthesis, biological activity, and chemical modification of the exopolysaccharide, pullulan. Carbohydr. Res. 2004, 339, 447–460. [Google Scholar] [CrossRef]
- Chen, T.-J.; Liu, G.-L.; Wei, X.; Wang, K.; Hu, Z.; Chi, Z.; Chi, Z.-M. A multidomain α-glucan synthetase 2 (AmAgs2) is the key enzyme for pullulan biosynthesis in Aureobasidium melanogenum P16. Int. J. Biol. Macromol. 2019, 150, 1037–1045. [Google Scholar] [CrossRef]
- Ganduri, V.; Mangamuri, U.; Muvva, V.; Poda, S. Pullulan-Stabilized Silver Nanoparticles -Their Synthesis, Characterization and Application as Bactericidal Agents. J. Appl. Pharm. Sci. 2016, 6, 27–37. [Google Scholar] [CrossRef] [Green Version]
- Wu, J.; Zhong, F.; Li, Y.; Shoemaker, C.; Xia, W. Preparation and characterization of pullulan–chitosan and pullulan–carboxymethyl chitosan blended films. Food Hydrocoll. 2012, 30, 82–91. [Google Scholar] [CrossRef]
- Wu, S.; Chen, J. Using pullulan-based edible coatings to extend shelf-life of fresh-cut ‘Fuji’ apples. Int. J. Biol. Macromol. 2013, 55, 254–257. [Google Scholar] [CrossRef] [PubMed]
- Gupta, M.; Gupta, A.K. In Vitro cytotoxicity studies of hydrogel pullulan nanoparticles prepared by AOT/N-hexane micellar system. J. Pharm. Pharm. Sci. 2004, 7, 38–46. [Google Scholar]
- Nešić, A.; Cabrera-Barjas, G.; Dimitrijević-Branković, S.; Davidović, S.; Radovanović, N.; Delattre, C. Prospect of Polysaccharide-Based Materials as Advanced Food Packaging. Molecules 2020, 25, 135. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yang, Y.; Xie, B.; Liu, Q.; Kong, B.; Wang, H. Fabrication and characterization of a novel polysaccharide based composite nanofiber films with tunable physical properties. Carbohydr. Polym. 2020, 236, 116054. [Google Scholar] [CrossRef]
- Kanmani, P.; Lim, S.T. Synthesis and characterization of pullulan-mediated silver nanoparticles and its antimicrobial activities. Carbohydr. Polym. 2013, 97, 421–428. [Google Scholar] [CrossRef] [PubMed]
- Al-Owasi, Y.; Elshafie, A.; Sivakumar, N.; Al-Bahry, S.N. Synthesis of pullulan-mediated silver nanoparticles (AgNPs) and their antimicrobial activities. SQU J. Sci. 2019, 24, 88–94. [Google Scholar] [CrossRef]
- Slavin, Y.N.; Asnis, J.; Häfeli, U.O.; Bach, H. Metal nanoparticles: Understanding the mechanisms behind antibacterial activity. J. Nanobiotechnol. 2017, 15, 65. [Google Scholar] [CrossRef]
- Priyadarshini, S.; Gopinath, V.; Priyadharsshini, N.M.; MubarakAli, D.; Velusamy, P. Synthesis of anisotropic silver nanoparticles using novel strain, Bacillus flexus and its biomedical application. Colloids Surf. B Biointerfaces 2013, 102, 232–237. [Google Scholar] [CrossRef] [PubMed]
- Raza, M.A.; Kanwal, Z.; Rauf, A.; Sabri, A.N.; Riaz, S.; Naseem, S. Size- and Shape-Dependent Antibacterial Studies of Silver Nanoparticles Synthesized by Wet Chemical Routes. Nanomaterials 2016, 6, 74. [Google Scholar] [CrossRef] [Green Version]
- Alshareef, A.; Laird, K.; Cross, R. Shape-dependent antibacterial activity of silver nanoparticles on Escherichia coli and Enterococcus faecium bacterium. Appl. Surf. Sci. 2017, 424, 310–315. [Google Scholar] [CrossRef]
- Pal, S.; Tak, Y.K.; Song, J.M. Does the Antibacterial Activity of Silver Nanoparticles Depend on the Shape of the Nanoparticle? A Study of the Gram-Negative Bacterium Escherichia coli. Appl. Environ. Microbiol. 2007, 73, 1712–1720. [Google Scholar] [CrossRef] [Green Version]
- Cheon, J.Y.; Kim, S.J.; Rhee, Y.H.; Kwon, O.H.; Park, W.H. Shape-dependent antimicrobial activities of silver nanoparticles. Int. J. Nanomed. 2019, 14, 2773–2780. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Salleh, M.S.N.; Ali, R.R.; Shameli, K.; Hamzah, M.Y.; Kasmani, R.M.; Nasef, M.M. Interaction Insight of Pullulan-Mediated Gamma-Irradiated Silver Nanoparticle Synthesis and Its Antibacterial Activity. Polymers 2021, 13, 3578. [Google Scholar] [CrossRef] [PubMed]
- Zhang, T.; Wang, L.; Chen, Q.; Chen, C. Cytotoxic Potential of Silver Nanoparticles. Yonsei Med. J. 2014, 55, 283–291. [Google Scholar] [CrossRef] [Green Version]
- Guilger-Casagrande, M.; Germano-Costa, T.; Bilesky-José, N.; Pasquoto-Stigliani, T.; Carvalho, L.; Fraceto, L.F.; de Lima, R. Influence of the capping of biogenic silver nanoparticles on their toxicity and mechanism of action towards Sclerotinia sclerotiorum. J. Nanobiotechnol. 2021, 19, 53. [Google Scholar] [CrossRef] [PubMed]
- Khalaf, H.H.; Sharoba, A.M.; El-Tanahi, H.H.; Morsy, M. Stability of antimicrobial activity of pullulan edible films incorporated with nanoparticles and essential oils and their impact on turkey deli meat quality. J. Food Dairy Sci. 2013, 4, 557–573. [Google Scholar] [CrossRef]
- Morsy, M.K.; Sharoba, A.M.; Khalaf, H.H.; El-Tanahy, H.H.; Cutter, C.N. Efficacy of Antimicrobial Pullulan-Based Coating to Improve Internal Quality and Shelf-Life of Chicken Eggs During Storage. J. Food Sci. 2015, 80, M1066–M1074. [Google Scholar] [CrossRef]
- Priyadarshi, R.; Kim, S.-M.; Rhim, J.-W. Pectin/pullulan blend films for food packaging: Effect of blending ratio. Food Chem. 2021, 347, 129022. [Google Scholar] [CrossRef]
- Li, Y.; Yokoyama, W.; Wu, J.; Ma, J.; Zhong, F. Properties of edible films based on pullulan–chitosan blended film-forming solutions at different pH. RSC Adv. 2015, 5, 105844–105850. [Google Scholar] [CrossRef]
- Khan, M.J.; Kumari, S.; Selamat, J.; Shameli, K.; Sazili, A.Q. Reducing Meat Perishability through Pullulan Active Packaging. J. Food Qual. 2020, 2020, 8880977. [Google Scholar] [CrossRef]
- Morsy, M.; Khalaf, H.H.; Sharoba, A.M.; El-Tanahi, H.H.; Cutter, C.N. Incorporation of Essential Oils and Nanoparticles in Pullulan Films to Control Foodborne Pathogens on Meat and Poultry Products. J. Food Sci. 2014, 79, M675–M684. [Google Scholar] [CrossRef] [PubMed]
- Raychaudhuri, R.; Naik, S.; Shreya, A.B.; Kandpal, N.; Pandey, A.; Kalthur, G.; Mutalik, S. Pullulan based stimuli responsive and sub cellular targeted nanoplatforms for biomedical application: Synthesis, nanoformulations and toxicological perspective. Int. J. Biol. Macromol. 2020, 161, 1189–1205. [Google Scholar] [CrossRef] [PubMed]
- Grenha, A.; Rodrigues, S. Pullulan-based nanoparticles: Future therapeutic applications in transmucosal protein delivery. Ther. Deliv. 2013, 4, 1339–1341. [Google Scholar] [CrossRef] [Green Version]
- Dionísio, M.; Cordeiro, C.; Remuñán-López, C.; Seijo, B.; da Costa, A.R.; Grenha, A. Pullulan-based nanoparticles as carriers for transmucosal protein delivery. Eur. J. Pharm. Sci. 2013, 50, 102–113. [Google Scholar] [CrossRef] [Green Version]
- Cevher, E.; Salomon, S.K.; Makrakis, A.; Li, X.W.; Brocchini, S.; Alpar, H.O. Development of chitosan–pullulan composite nanoparticles for nasal delivery of vaccines: Optimisation and cellular studies. J. Microencapsul. 2015, 32, 755–768. [Google Scholar] [CrossRef] [PubMed]



Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rai, M.; Wypij, M.; Ingle, A.P.; Trzcińska-Wencel, J.; Golińska, P. Emerging Trends in Pullulan-Based Antimicrobial Systems for Various Applications. Int. J. Mol. Sci. 2021, 22, 13596. https://doi.org/10.3390/ijms222413596
Rai M, Wypij M, Ingle AP, Trzcińska-Wencel J, Golińska P. Emerging Trends in Pullulan-Based Antimicrobial Systems for Various Applications. International Journal of Molecular Sciences. 2021; 22(24):13596. https://doi.org/10.3390/ijms222413596
Chicago/Turabian StyleRai, Mahendra, Magdalena Wypij, Avinash P. Ingle, Joanna Trzcińska-Wencel, and Patrycja Golińska. 2021. "Emerging Trends in Pullulan-Based Antimicrobial Systems for Various Applications" International Journal of Molecular Sciences 22, no. 24: 13596. https://doi.org/10.3390/ijms222413596
APA StyleRai, M., Wypij, M., Ingle, A. P., Trzcińska-Wencel, J., & Golińska, P. (2021). Emerging Trends in Pullulan-Based Antimicrobial Systems for Various Applications. International Journal of Molecular Sciences, 22(24), 13596. https://doi.org/10.3390/ijms222413596






