Heat Stress Responses and Thermotolerance in Maize
Abstract
1. Introduction
2. Heat Shock and Heat Stress in Lab and Field Conditions
3. Heat Stress Response (HSR), A Cytoplasmic Heat Stress Response
4. Plant UPR in Heat Stress Response
5. Other Transcriptional Regulation Networks Involved in Plant Heat Stress Response
6. Enhancement of Alternative RNA Splicing As a Heat Stress Response
7. Conclusions, Prospects and Maize Breeding
Author Contributions
Funding
Institutional Review Board Statement
Conflicts of Interest
Abbreviations
| HSR | heat stress response |
| HSFs | heat shock transcription factors |
| HSPs | heat shock proteins |
| UPR | unfolded protein response |
| ER | endoplasmic reticulum |
| bZIP | basic leucine zipper |
| ABA | abscisic acid |
| SA | salicylic acid |
| DREB2A | dehydration-responsive element-binding protein 2A |
| ERF1 | ETHYLENE RESPONSE FACTOR1 |
| JA | jasmonic acid |
| NF-YC10 | NUCLEAR FACTOR Y, SUBUNIT C10 |
| GAPC | glyceraldehyde-3-phosphate dehydrogenase |
| SPL | SQUAMOSA PROMOTER BINDING PROTEIN-LIKE |
| ROS | reactive oxygen species |
| Apx1 | ascorbate peroxidase 1 |
| FLM | FLOWERING LOCUS M |
| AS | alternative RNA splicing |
| PTC | premature stop codon |
| SR | serine/arginine-rich |
| NMD | nonsense-mediated mRNA decay |
References
- Prasad, P.; Bheemanahalli, R.; Jagadish, S.V.K. Field crops and the fear of heat stress—Opportunities, challenges and future directions. Field Crop. Res. 2017, 200, 114–121. [Google Scholar] [CrossRef]
- Hoegh-Guldberg, O.; Jacob, D.; Taylor, M.; Bindi, M.; Brown, S.; Camilloni, I.; Diedhiou, A.; Djalante, R.; Ebi, K.; Engelbrecht, F.; et al. Chapter 3: Impacts of 1.5 °C global warming on natural and human systems. In Global Warming of 1.5 °C. An IPCC Special Report on the Impacts of Global Warming of 1.5 °C above Preindustrial Levels and Related Global Greenhouse Gas Emission Pathways, in the Context of Strengthening the Global Response to the Threat of Climate Change; Specical Report; Intergovernmental Panel on Climate Change: Geneva, Switzerland, 2018; pp. 175–311. [Google Scholar]
- Frey, F.P.; Urbany, C.; Huettel, B.; Reinhardt, R.; Stich, B. Genome-wide expression profiling and phenotypic evaluation of European maize inbreds at seedling stage in response to heat stress. BMC Genomics 2015, 16, 123. [Google Scholar] [CrossRef] [PubMed]
- Shi, J.; Yan, B.; Lou, X.; Ma, H.; Ruan, S. Comparative transcriptome analysis reveals the transcriptional alterations in heat-resistant and heat-sensitive sweet maize (Zea mays L.) varieties under heat stress. BMC Plant Biol. 2017, 17, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Obata, T.; Witt, S.; Lisec, J.; Palacios-Rojas, N.; Florez-Sarasa, I.; Yousfi, S.; Araus, J.L.; Cairns, J.E.; Fernie, A.R. Metabolite profiles of maize leaves in drought, heat and combined stress field trials reveal the relationship between metabolism and grain yield. Plant Physiol. 2015, 169, 2665–2683. [Google Scholar] [CrossRef]
- Sun, C.X.; Li, M.Q.; Gao, X.X.; Liu, L.N.; Wu, X.F.; Zhou, J.H. Metabolic response of maize plants to multi-factorial abiotic stresses. Plant Biol. 2016, 18, 120–129. [Google Scholar] [CrossRef]
- Essemine, J.; Li, J.; Chen, G.; Qu, M. Analytical dataset of short-term heat stress induced reshuffling of metabolism and transcriptomes in maize grown under elevated CO2. Data Brief 2020, 28, 105004. [Google Scholar] [CrossRef]
- Tollenaar, M.; Lee, E. Yield potential, yield stability and stress tolerance in maize. Field Crop. Res. 2002, 75, 161–169. [Google Scholar] [CrossRef]
- Lobell, D.B.; Bänziger, M.; Magorokosho, C.; Vivek, B. Nonlinear heat effects on African maize as evidenced by historical yield trials. Nat. Clim. Chang. 2011, 1, 42–45. [Google Scholar] [CrossRef]
- Badu-Apraku, B.; Hunter, R.B.; Tollenaar, M. Effect of temperature during grain filling on whole plant and grain yield in maize (Zea mays L.). Can. J. Plant Sci. 1983, 63, 357–363. [Google Scholar] [CrossRef]
- Schoper, J.B.; Lambert, R.J.; Vasilas, B.L.; Westgate, M.E. Plant Factors Controlling Seed Set in Maize. Plant Physiol. 1987, 83, 121–125. [Google Scholar] [CrossRef]
- Lizaso, J.; Ruiz-Ramos, M.; Rodríguez, L.; Gabaldon-Leal, C.; Oliveira, J.; Lorite, I.; Sánchez, D.; García, E.; Rodríguez, A. Impact of high temperatures in maize: Phenology and yield components. Field Crop. Res. 2018, 216, 129–140. [Google Scholar] [CrossRef]
- Wilhelm, E.P.; Mullen, R.E.; Keeling, P.L.; Singletary, G.W. Heat Stress during Grain Filling in Maize: Effects on Kernel Growth and Metabolism. Crop Sci. 1999, 39, 1733–1741. [Google Scholar] [CrossRef]
- Li, Z.; Tang, J.; Srivastava, R.; Bassham, D.C.; Howell, S.H. The Transcription Factor bZIP60 Links the Unfolded Protein Response to the Heat Stress Response in Maize. Plant Cell 2020, 32, 3559–3575. [Google Scholar] [CrossRef] [PubMed]
- Siebers, M.H.; Slattery, R.A.; Yendrek, C.R.; Locke, A.M.; Drag, D.; Ainsworth, E.A.; Bernacchi, C.J.; Ort, D.R. Simulated heat waves during maize reproductive stages alter reproductive growth but have no lasting effect when applied during vegetative stages. Agric. Ecosyst. Environ. 2017, 240, 162–170. [Google Scholar] [CrossRef]
- Irwin, S.; Good, D. How Bad Was the 2012 Corn and Soybean Growing Season? Farmdoc Daily, 3 October 2012; 3–5. [Google Scholar]
- Poorter, H.; Fiorani, F.; Pieruschka, R.; Wojciechowski, T.; Van Der Putten, W.H.; Kleyer, M.; Schurr, U.; Postma, J. Pampered inside, pestered outside? Differences and similarities between plants growing in controlled conditions and in the field. New Phytol. 2016, 212, 838–855. [Google Scholar] [CrossRef]
- Wahid, A.; Gelani, S.; Ashraf, M.; Foolad, M. Heat tolerance in plants: An overview. Environ. Exp. Bot. 2007, 61, 199–223. [Google Scholar] [CrossRef]
- Akter, N.; Islam, M.R. Heat stress effects and management in wheat. A review. Agron. Sustain. Dev. 2017, 37, 37. [Google Scholar] [CrossRef]
- Wang, Y.; Wang, L.; Zhou, J.; Hu, S.; Chen, H.; Xiang, J.; Zhang, Y.; Zeng, Y.; Shi, Q.; Zhu, D.; et al. Research Progress on Heat Stress of Rice at Flowering Stage. Rice Sci. 2019, 26, 1–10. [Google Scholar] [CrossRef]
- Fragkostefanakis, S.; Röth, S.; Schleiff, E.; Scharf, K.-D. Prospects of engineering thermotolerance in crops through modulation of heat stress transcription factor and heat shock protein networks. Plant Cell Environ. 2015, 38, 1881–1895. [Google Scholar] [CrossRef]
- Guo, M.; Liu, J.-H.; Ma, X.; Luo, D.-X.; Gong, Z.-H.; Lu, M.-H. The Plant Heat Stress Transcription Factors (HSFs): Structure, Regulation, and Function in Response to Abiotic Stresses. Front. Plant Sci. 2016, 7, 114. [Google Scholar] [CrossRef]
- Shi, Y.; Mosser, D.D.; Morimoto, R.I. Molecularchaperones as HSF1-specific transcriptional repressors. Genes Dev. 1998, 12, 654–666. [Google Scholar] [CrossRef] [PubMed]
- Sistonen, L.; Sarge, K.D.; Morimoto, R.I. Human heat shock factors 1 and 2 are differentially activated and can synergistically induce hsp70 gene transcription. Mol. Cell. Biol. 1994, 14, 2087–2099. [Google Scholar] [CrossRef] [PubMed]
- Morimoto, R.I. Cells in stress: Transcriptional activation of heat shock genes. Science 1993, 259, 1409–1410. [Google Scholar] [CrossRef] [PubMed]
- Guo, L.; Chen, S.; Liu, K.; Liu, Y.; Ni, L.; Zhang, K.; Zhang, L. Isolation of Heat Shock Factor HsfA1a-Binding Sites in vivo Revealed Variations of Heat Shock Elements in Arabidopsis thaliana. Plant Cell Physiol. 2008, 49, 1306–1315. [Google Scholar] [CrossRef]
- Nover, L.; Bharti, K.; Döring, P.; Mishra, S.K.; Ganguli, A.; Scharf, K.-D. Arabidopsis and the heat stress transcription factor world: How many heat stress transcription factors do we need? Cell Stress Chaperones 2001, 6, 177–189. [Google Scholar] [CrossRef]
- Chauhan, H.; Khurana, N.; Agarwal, P.; Khurana, P. Heat shock factors in rice (Oryza sativa L.): Genome-wide expression analysis during reproductive development and abiotic stress. Mol. Genet. Genom. 2011, 286, 171–187. [Google Scholar] [CrossRef]
- Lin, Y.-X.; Jiang, H.; Chu, Z.-X.; Tang, X.-L.; Zhu, S.; Cheng, B. Genome-wide identification, classification and analysis of heat shock transcription factor family in maize. BMC Genom. 2011, 12, 76. [Google Scholar] [CrossRef]
- Zhang, H.; Li, G.; Fu, C.; Duan, S.; Hu, D.; Guo, X. Genome-wide identification, transcriptome analysis and alternative splicing events of Hsf family genes in maize. Sci. Rep. 2020, 10, 8073. [Google Scholar] [CrossRef]
- Harrison, C.J.; Bohm, A.A.; Nelson, H.C. Crystal structure of the DNA binding domain of the heat shock transcription factor. Science 1994, 263, 224–227. [Google Scholar] [CrossRef]
- Scharf, K.-D.; Berberich, T.; Ebersberger, I.; Nover, L. The plant heat stress transcription factor (Hsf) family: Structure, function and evolution. Biochim. Biophys. Acta Regul. Mech. 2012, 1819, 104–119. [Google Scholar] [CrossRef]
- Kotak, S.; Port, M.; Ganguli, A.; Bicker, F.; Von Koskull-Döring, P. Characterization of C-terminal domains of Arabidopsis heat stress transcription factors (Hsfs) and identification of a new signature combination of plant class A Hsfs with AHA and NES motifs essential for activator function and intracellular localization. Plant J. 2004, 39, 98–112. [Google Scholar] [CrossRef] [PubMed]
- Nishizawa-Yokoi, A.; Nosaka, R.; Hayashi, H.; Tainaka, H.; Maruta, T.; Tamoi, M.; Ikeda, M.; Ohme-Takagi, M.; Yoshimura, K.; Yabuta, Y.; et al. HsfA1d and HsfA1e Involved in the Transcriptional Regulation of HsfA2 Function as Key Regulators for the Hsf Signaling Network in Response to Environmental Stress. Plant Cell Physiol. 2011, 52, 933–945. [Google Scholar] [CrossRef] [PubMed]
- Nishizawa, A.; Yabuta, Y.; Yoshida, E.; Maruta, T.; Yoshimura, K.; Shigeoka, S. Arabidopsis heat shock transcription factor A2 as a key regulator in response to several types of environmental stress. Plant J. 2006, 48, 535–547. [Google Scholar] [CrossRef] [PubMed]
- Charng, Y.-Y.; Liu, H.-C.; Liu, N.-Y.; Chi, W.-T.; Wang, C.-N.; Chang, S.-H.; Wang, T.-T. A Heat-Inducible Transcription Factor, HsfA2, Is Required for Extension of Acquired Thermotolerance in Arabidopsis. Plant Physiol. 2007, 143, 251–262. [Google Scholar] [CrossRef] [PubMed]
- Ogawa, D.; Yamaguchi, K.; Nishiuchi, T. High-level overexpression of the Arabidopsis HsfA2 gene confers not only increased themotolerance but also salt/osmotic stress tolerance and enhanced callus growth. J. Exp. Bot. 2007, 58, 3373–3383. [Google Scholar] [CrossRef] [PubMed]
- Schramm, F.; Ganguli, A.; Kiehlmann, E.; Englich, G.; Walch, D.; Von Koskull-Döring, P. The heat stress transcription factor HsfA2 serves as a regulatory amplifier of a subset of genes in the heat stress response in Arabidopsis. Plant Mol. Biol. 2006, 60, 759–772. [Google Scholar] [CrossRef] [PubMed]
- Döring, P.; Treuter, E.; Kistner, C.; Lyck, R.; Chen, A.; Nover, L. The Role of AHA Motifs in the Activator Function of Tomato Heat Stress Transcription Factors HsfA1 and HsfA2. Plant Cell 2000, 12, 265–278. [Google Scholar] [CrossRef]
- Huang, Y.-C.; Niu, C.-Y.; Yang, C.-R.; Jinn, T.-L. The heat-stress factor HSFA6b connects ABA signaling and ABA-mediated heat responses. Plant Physiol. 2016, 172, 1182–1199. [Google Scholar] [CrossRef]
- Czarnecka-Verner, E.; Pan, S.; Salem, T.; Gurley, W.B. Plant class B HSFs inhibit transcription and exhibit affinity for TFIIB and TBP. Plant Mol. Biol. 2004, 56, 57–75. [Google Scholar] [CrossRef]
- Ikeda, M.; Mitsuda, N.; Ohme-Takagi, M. Arabidopsis HsfB1 and HsfB2b Act as Repressors of the Expression of Heat-Inducible Hsfs But Positively Regulate the Acquired Thermotolerance. Plant Physiol. 2011, 157, 1243–1254. [Google Scholar] [CrossRef]
- Qian, Y.; Ren, Q.; Zhang, J.; Chen, L. Transcriptomic analysis of the maize (Zea mays L.) inbred line B73 response to heat stress at the seedling stage. Gene 2019, 692, 68–78. [Google Scholar] [CrossRef] [PubMed]
- Lund, A.A.; Blum, P.H.; Bhattramakki, D.; Elthon, T.E. Heat-Stress Response of Maize Mitochondria. Plant Physiol. 1998, 116, 1097–1110. [Google Scholar] [CrossRef] [PubMed]
- Cooper, P.; Ho, T.-H.D. Heat Shock Proteins in Maize. Plant Physiol. 1983, 71, 215–222. [Google Scholar] [CrossRef]
- Cooper, P.; Ho, T.-H.D.; Hauptmann, R.M. Tissue Specificity of the Heat-Shock Response in Maize. Plant Physiol. 1984, 75, 431–441. [Google Scholar] [CrossRef] [PubMed]
- Abou-Deif, M.H.; Rashed, M.A.-S.; Khalil, K.M.; Mahmoud, F.E.-S. Proteomic analysis of heat shock proteins in maize (Zea mays L.). Bull. Natl. Res. Cent. 2019, 43. [Google Scholar] [CrossRef]
- Pegoraro, C.; Mertz, L.M.; Da Maia, L.C.; Rombaldi, C.V.; De Oliveira, A.C. Importance of heat shock proteins in maize. J. Crop Sci. Biotechnol. 2011, 14, 85–95. [Google Scholar] [CrossRef]
- Gao, H.; Brandizzi, F.; Benning, C.; Larkin, R.M. A membrane-tethered transcription factor defines a branch of the heat stress response in Arabidopsis thaliana. Proc. Natl. Acad. Sci. USA 2008, 105, 16398–16403. [Google Scholar] [CrossRef]
- Zhang, S.-S.; Yang, H.; Ding, L.; Song, Z.-T.; Ma, H.; Chang, F.; Liu, J.-X. Tissue-Specific Transcriptomics Reveals an Important Role of the Unfolded Protein Response in Maintaining Fertility upon Heat Stress in Arabidopsis. Plant Cell 2017, 29, 1007–1023. [Google Scholar] [CrossRef]
- Deng, Y.; Humbert, S.; Liu, J.-X.; Srivastava, R.; Rothstein, S.J.; Howell, S.H. Heat induces the splicing by IRE1 of a mRNA encoding a transcription factor involved in the unfolded protein response in Arabidopsis. Proc. Natl. Acad. Sci. USA 2011, 108, 7247–7252. [Google Scholar] [CrossRef]
- Deng, Y.; Srivastava, R.; Howell, S.H. Protein kinase and ribonuclease domains of IRE1 confer stress tolerance, vegetative growth, and reproductive development in Arabidopsis. Proc. Natl. Acad. Sci. USA 2013, 110, 19633–19638. [Google Scholar] [CrossRef]
- Li, Z.; Srivastava, R.; Tang, J.; Zheng, Z.; Howell, S.H. Cis-Effects Condition the Induction of a Major Unfolded Protein Response Factor, ZmbZIP60, in Response to Heat Stress in Maize. Front. Plant Sci. 2018, 9, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Larkindale, J.; Knight, M.R. Protection against Heat Stress-Induced Oxidative Damage in Arabidopsis Involves Calcium, Abscisic Acid, Ethylene, and Salicylic Acid. Plant Physiol. 2002, 128, 682–695. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.; Kang, J.-Y.; Cho, D.-I.; Park, J.H.; Kim, S.Y. ABF2, an ABRE-binding bZIP factor, is an essential component of glucose signaling and its overexpression affects multiple stress tolerance. Plant J. 2004, 40, 75–87. [Google Scholar] [CrossRef] [PubMed]
- Suzuki, N.; Bassil, E.; Nakano, R.; Rivero, R.M.; Verbeck, G.F.; Azad, R.K.; Blumwald, E.; Mittler, R.; Hamilton, J.S.; Inupakutika, M.A.; et al. ABA Is Required for Plant Acclimation to a Combination of Salt and Heat Stress. PLoS ONE 2016, 11, e0147625. [Google Scholar] [CrossRef]
- Gong, M.; Li, Y.-J.; Chen, S.-Z. Abscisic acid-induced thermotolerance in maize seedlings is mediated by calcium and associated with antioxidant systems. J. Plant Physiol. 1998, 153, 488–496. [Google Scholar] [CrossRef]
- Wang, X.; Zhuang, L.; Shi, Y.; Huang, B. Up-Regulation of HSFA2c and HSPs by ABA Contributing to Improved Heat Tolerance in Tall Fescue and Arabidopsis. Int. J. Mol. Sci. 2017, 18, 1981. [Google Scholar] [CrossRef]
- Cheng, M.-C.; Liao, P.-M.; Kuo, W.-W.; Lin, T. The Arabidopsis ETHYLENE RESPONSE FACTOR1 Regulates Abiotic Stress-Responsive Gene Expression by Binding to Different cis-Acting Elements in Response to Different Stress Signals. Plant Physiol. 2013, 162, 1566–1582. [Google Scholar] [CrossRef]
- Balfagón, D.; Sengupta, S.; Gómez-Cadenas, A.; Fritschi, F.B.; Azad, R.K.; Mittler, R.; Zandalinas, S.I. Jasmonic Acid Is Required for Plant Acclimation to a Combination of High Light and Heat Stress. Plant Physiol. 2019, 181, 1668–1682. [Google Scholar] [CrossRef]
- Zandalinas, S.I.; Fichman, Y.; Devireddy, A.R.; Sengupta, S.; Azad, R.K.; Mittler, R. Systemic signaling during abiotic stress combination in plants. Proc. Natl. Acad. Sci. USA 2020, 117, 13810–13820. [Google Scholar] [CrossRef]
- Sakuma, Y.; Maruyama, K.; Qin, F.; Osakabe, Y.; Shinozaki, K.; Yamaguchi-Shinozaki, K. Dual function of an Arabidopsis transcription factor DREB2A in water-stress-responsive and heat-stress-responsive gene expression. Proc. Natl. Acad. Sci. USA 2006, 103, 18822–18827. [Google Scholar] [CrossRef]
- Sato, H.; Mizoi, J.; Shinozaki, K.; Yamaguchi-Shinozaki, K.; Tanaka, H.; Maruyama, K.; Qin, F.; Osakabe, Y.; Morimoto, K.; Ohori, T.; et al. Arabidopsis DPB3-1, a DREB2A Interactor, Specifically Enhances Heat Stress-Induced Gene Expression by Forming a Heat Stress-Specific Transcriptional Complex with NF-Y Subunits. Plant Cell 2014, 26, 4954–4973. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.-C.; Guo, L.; Wang, X. Nuclear moonlighting of cytosolic glyceraldehyde-3-phosphate dehydrogenase regulates Arabidopsis response to heat stress. Nat. Commun. 2020, 11, 1–15. [Google Scholar] [CrossRef] [PubMed]
- Chao, L.-M.; Liu, Y.-Q.; Chen, D.-Y.; Xue, X.-Y.; Mao, Y.-B.; Chen, X.-Y. Arabidopsis Transcription Factors SPL1 and SPL12 Confer Plant Thermotolerance at Reproductive Stage. Mol. Plant 2017, 10, 735–748. [Google Scholar] [CrossRef] [PubMed]
- Song, Z.; Zhang, L.; Han, J.; Zhou, M.; Liu, J. Histone H3K4 methyltransferases SDG25 and ATX1 maintain heat-stress gene expression during recovery in Arabidopsis. Plant J. 2021. [Google Scholar] [CrossRef] [PubMed]
- Davletova, S.; Rizhsky, L.; Liang, H.; Shengqiang, Z.; Oliver, D.J.; Coutu, J.; Shulaev, V.; Schlauch, K.; Mittler, R. Cytosolic Ascorbate Peroxidase 1 Is a Central Component of the Reactive Oxygen Gene Network of Arabidopsis. Plant Cell 2005, 17, 268–281. [Google Scholar] [CrossRef]
- Volkov, R.A.; Panchuk, I.I.; Mullineaux, P.M.; Schöffl, F. Heat stress-induced H2O2 is required for effective expression of heat shock genes in Arabidopsis. Plant Mol. Biol. 2006, 61, 733–746. [Google Scholar] [CrossRef] [PubMed]
- Pnueli, L.; Liang, H.; Rozenberg, M.; Mittler, R. Growth suppression, altered stomatal responses, and augmented induction of heat shock proteins in cytosolic ascorbate peroxidase (Apx1)-deficient Arabidopsis plants. Plant J. 2003, 34, 187–203. [Google Scholar] [CrossRef] [PubMed]
- Li, Z.; Tang, J.; Bassham, D.C.; Howell, S.H. Differential expression of SR45a isoforms in maize in response to elevated daily temperatures alters their splicing activity toward RNA substrates. (In review).
- Sugio, A.; Dreos, R.; Aparicio, F.; Maule, A.J. The Cytosolic Protein Response as a Subcomponent of the Wider Heat Shock Response in Arabidopsis. Plant Cell 2009, 21, 642–654. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Sun, N.; Liu, M.; Liu, J.; Du, B.; Wang, X.; Qi, X. An Autoregulatory Loop Controlling Arabidopsis HsfA2 Expression: Role of Heat Shock-Induced Alternative Splicing. Plant Physiol. 2013, 162, 512–521. [Google Scholar] [CrossRef] [PubMed]
- Palusa, S.G.; Ali, G.S.; Reddy, A.S. Alternative splicing of pre-mRNAs of Arabidopsis serine/arginine-rich proteins: Regulation by hormones and stresses. Plant J. 2007, 49, 1091–1107. [Google Scholar] [CrossRef] [PubMed]
- Capovilla, G.; Symeonidi, E.; Wu, R.; Schmid, M. Contribution of major FLM isoforms to temperature-dependent flowering in Arabidopsis thaliana. J. Exp. Bot. 2017, 68, 5117–5127. [Google Scholar] [CrossRef] [PubMed]
- Jiao, Y.; Meyerowitz, E.M. Cell-type specific analysis of translating RNAs in developing flowers reveals new levels of control. Mol. Syst. Biol. 2010, 6, 419. [Google Scholar] [CrossRef] [PubMed]
- Balasubramanian, S.; Weigel, D. Temperature Induced Flowering in Arabidopsis thaliana. Plant Signal. Behav. 2006, 1, 227–228. [Google Scholar] [CrossRef] [PubMed]
- Balasubramanian, S.; Sureshkumar, S.; Lempe, J.; Weigel, D. Potent Induction of Arabidopsis thaliana Flowering by Elevated Growth Temperature. PLoS Genet. 2006, 2, e106. [Google Scholar] [CrossRef]
- Sureshkumar, S.; Dent, C.; Seleznev, A.; Tasset, C.; Balasubramanian, S. Nonsense-mediated mRNA decay modulates FLM-dependent thermosensory flowering response in Arabidopsis. Nat. Plants 2016, 2, 16055. [Google Scholar] [CrossRef]
- Stief, A.; Altmann, S.; Hoffmann, K.; Pant, B.D.; Scheible, W.-R.; Bäurle, I. Arabidopsis miR156 Regulates Tolerance to Recurring Environmental Stress through SPL Transcription Factors. Plant Cell 2014, 26, 1792–1807. [Google Scholar] [CrossRef]
- Pegler, J.L.; Oultram, J.M.; Grof, C.P.; Eamens, A.L. Profiling the Abiotic Stress Responsive microRNA Landscape of Arabidopsis thaliana. Plants 2019, 8, 58. [Google Scholar] [CrossRef]
- Zhang, M.; An, P.; Li, H.; Wang, X.; Zhou, J.; Dong, P.; Zhao, Y.; Wang, Q.; Li, C. The miRNA-Mediated Post-Transcriptional Regulation of Maize in Response to High Temperature. Int. J. Mol. Sci. 2019, 20, 1754. [Google Scholar] [CrossRef]
- Zhang, H.; Li, G.; Hu, D.; Zhang, Y.; Zhang, Y.; Shao, H.; Zhao, L.; Yang, R.; Guo, X. Functional characterization of maize heat shock transcription factor gene ZmHsf01 in thermotolerance. PeerJ 2020, 8, e8926. [Google Scholar] [CrossRef]
- Lindquist, S.; Biology, C.; Craig, E.A. The heat-shock proteins. Annu. Rev. Genet. 1988, 22, 631–677. [Google Scholar] [CrossRef] [PubMed]
- Swindell, W.R.; Huebner, M.; Weber, A.P. Transcriptional profiling of Arabidopsis heat shock proteins and transcription factors reveals extensive overlap between heat and non-heat stress response pathways. BMC Genom. 2007, 8, 125. [Google Scholar] [CrossRef] [PubMed]
- Gupta, S.C.; Sharma, A.; Mishra, M.; Mishra, R.K.; Chowdhuri, D.K. Heat shock proteins in toxicology: How close and how far? Life Sci. 2010, 86, 377–384. [Google Scholar] [CrossRef] [PubMed]
- Kotak, S.; Larkindale, J.; Lee, U.; Von Koskull-Döring, P.; Vierling, E.; Scharf, K.-D. Complexity of the heat stress response in plants. Curr. Opin. Plant Biol. 2007, 10, 310–316. [Google Scholar] [CrossRef] [PubMed]
- Schöffl, F.; Prändl, R.; Reindl, A. Regulation of the Heat-Shock Response. Plant Physiol. 1998, 117, 1135–1141. [Google Scholar] [CrossRef] [PubMed]
- Schlesinger, M.J. Heat shock proteins. J. Biol. Chem. 1990, 265, 12111–12114. [Google Scholar] [CrossRef]
- Vierling, E. The roles of heat shock proteins in tumor immunity. Annu. Rev. Plant Physiol. Plant Mol. Biol. 1991, 42, 579–620. [Google Scholar] [CrossRef]
- Hilton, G.R.; Lioe, H.; Stengel, F.; Baldwin, A.J.; Benesch, J.L.P. Small Heat-Shock Proteins: Paramedics of the Cell. In Molecular Chaperones; Springer: Berlin, Heidelberg, 2012; Volume 328. [Google Scholar] [CrossRef]
- Poór, P.; Czékus, Z.; Tari, I.; Ördög, A. The Multifaceted Roles of Plant Hormone Salicylic Acid in Endoplasmic Reticulum Stress and Unfolded Protein Response. Int. J. Mol. Sci. 2019, 20, 5842. [Google Scholar] [CrossRef]
- Yang, Y.-G.; Lv, W.-T.; Li, M.-J.; Wang, B.; Sun, D.-M.; Deng, X. Maize Membrane-Bound Transcription Factor Zmbzip17 is a Key Regulator in the Cross-Talk of ER Quality Control and ABA Signaling. Plant Cell Physiol. 2013, 54, 2020–2033. [Google Scholar] [CrossRef]
- Howell, S.H. Endoplasmic Reticulum Stress Responses in Plants. Annu. Rev. Plant Biol. 2013, 64, 477–499. [Google Scholar] [CrossRef]
- Larkindale, J.; Hall, J.D.; Knight, M.R.; Vierling, E. Heat Stress Phenotypes of Arabidopsis Mutants Implicate Multiple Signaling Pathways in the Acquisition of Thermotolerance. Plant Physiol. 2005, 138, 882–897. [Google Scholar] [CrossRef] [PubMed]
- Casaretto, J.A.; El-Kereamy, A.; Zeng, B.; Stiegelmeyer, S.M.; Chen, X.; Bi, Y.-M.; Rothstein, S.J. Expression of OsMYB55 in maize activates stress-responsive genes and enhances heat and drought tolerance. BMC Genom. 2016, 17, 312. [Google Scholar] [CrossRef] [PubMed]
- Hasanuzzaman, M.; Hossain, M.A.; da Silva, J.A.T.; Fujita, M. Plant Responses and Tolerance to Abiotic Oxidative Stress: Antioxidant Defenses is a Key Factor. In Crop Stress and Its Management: Perspectives and Strategies; Bandi, V., Shanker, A.K., Shanker, C., Mandapaka, M., Eds.; Springer: Berlin, Germany, 2012; pp. 261–316. [Google Scholar]
- Hasanuzzaman, M.; Nahar, K.; Fujita, M. Extreme Temperatures, Oxidative Stress and Antioxidant Defense in Plants. In Abiotic Stress—Plant Responses and Applications in Agriculture; Vahdati, K., Ed.; InTech: Rijeka, Croatia, 2013; pp. 169–205. [Google Scholar]
- Hasanuzzaman, M.; Nahar, K.; Alam, M.; Roychowdhury, R.; Fujita, M. Physiological, Biochemical, and Molecular Mechanisms of Heat Stress Tolerance in Plants. Int. J. Mol. Sci. 2013, 14, 9643–9684. [Google Scholar] [CrossRef] [PubMed]
- Suzuki, N.; Katano, K. Coordination Between ROS Regulatory Systems and Other Pathways Under Heat Stress and Pathogen Attack. Front. Plant Sci. 2018, 9, 490. [Google Scholar] [CrossRef]
- Suzuki, N.; Mittler, R. Reactive oxygen species and temperature stresses: A delicate balance between signaling and destruction. Physiol. Plant. 2006, 126, 45–51. [Google Scholar] [CrossRef]
- Hajiboland, R. Chapter 1—Reactive Oxygen Species and Photosynthesis. In Oxidative Damage to Plants; Parvaiz, A., Ed.; Elsevier: Waltham, MA, USA, 2014; pp. 1–63. ISBN 978-0-12-799963-0. [Google Scholar]
- Singh, R.; Singh, S.; Parihar, P.; Mishra, R.K.; Tripathi, D.K.; Singh, V.P.; Chauhan, D.K.; Prasad, S.M. Reactive Oxygen Species (ROS): Beneficial Companions of Plants’ Developmental Processes. Front. Plant Sci. 2016, 7, 1299. [Google Scholar] [CrossRef]
- Apel, K.; Hirt, H. Reactive oxygen species: Metabolism, oxidative stress, and signal transduction. Annu. Rev. Plant Biol. 2004, 55, 373–399. [Google Scholar] [CrossRef]
- Sharma, P.; Jha, A.B.; Dubey, R.S.; Pessarakli, M. Reactive Oxygen Species, Oxidative Damage, and Antioxidative Defense Mechanism in Plants under Stressful Conditions. J. Bot. 2012, 2012, 1–26. [Google Scholar] [CrossRef]
- Zhong, M.; Orosz, A.; Wu, C. Direct Sensing of Heat and Oxidation by Drosophila Heat Shock Transcription Factor. Mol. Cell 1998, 2, 101–108. [Google Scholar] [CrossRef]
- Ahn, S.-G.; Thiele, D.J. Redox regulation of mammalian heat shock factor 1 is essential for Hsp gene activation and protection from stress. Genes Dev. 2003, 17, 516–528. [Google Scholar] [CrossRef]
- Laloum, T.; Martín, G.; Duque, P. Alternative Splicing Control of Abiotic Stress Responses. Trends Plant Sci. 2018, 23, 140–150. [Google Scholar] [CrossRef] [PubMed]
- Punzo, P.; Grillo, S.; Batelli, G. Alternative splicing in plant abiotic stress responses. Biochem. Soc. Trans. 2020, 48, 2117–2126. [Google Scholar] [CrossRef] [PubMed]
- Chaudhary, S.; Khokhar, W.; Jabre, I.; Reddy, A.S.N.; Byrne, L.J.; Wilson, C.M.; Syed, N.H. Alternative Splicing and Protein Diversity: Plants Versus Animals. Front. Plant Sci. 2019, 10, 708. [Google Scholar] [CrossRef] [PubMed]
- Ishka, M.R.; Brown, E.; Weigand, C.; Tillet, R.L.; Schlauch, K.; Miller, G.; Harper, J.F. A comparison of heat-stress transcriptome changes between wild-type Arabidopsis pollen and a heat-sensitive mutant harboring a knockout of cyclic nucleotide-gated cation channel 16 (cngc16). BMC Genom. 2018, 19, 549. [Google Scholar] [CrossRef]
- Ding, Y.; Huang, L.; Jiang, Q.; Zhu, C. MicroRNAs as Important Regulators of Heat Stress Responses in Plants. J. Agric. Food Chem. 2020, 68, 11320–11326. [Google Scholar] [CrossRef] [PubMed]
- He, J.; Jiang, Z.; Gao, L.; You, C.; Ma, X.; Wang, X.; Xu, X.; Mo, B.; Chen, X.; Liu, L. Genome-Wide Transcript and Small RNA Profiling Reveals Transcriptomic Responses to Heat Stress. Plant Physiol. 2019, 181, 609–629. [Google Scholar] [CrossRef]
- Chen, J.; Xu, W.; Burke, J.J.; Xin, Z. Role of Phosphatidic Acid in High Temperature Tolerance in Maize. Crop Sci. 2010, 50, 2506–2515. [Google Scholar] [CrossRef]
- Chen, J.; Xu, W.; Velten, J.; Xin, Z.; Stout, J.E. Characterization of maize inbred lines for drought and heat tolerance. J. Soil Water Conserv. 2012, 67, 354–364. [Google Scholar] [CrossRef]
- Tesfaye, K.; Kruseman, G.; Cairns, J.E.; Zaman-Allah, M.; Wegary, D.; Zaidi, P.; Boote, K.J.; Rahut, D.; Erenstein, O. Potential benefits of drought and heat tolerance for adapting maize to climate change in tropical environments. Clim. Risk Manag. 2018, 19, 106–119. [Google Scholar] [CrossRef]
- Nelimor, C.; Badu-Apraku, B.; Tetteh, A.; N’Guetta, A.S.-P. Assessment of Genetic Diversity for Drought, Heat and Combined Drought and Heat Stress Tolerance in Early Maturing Maize Landraces. Plants 2019, 8, 518. [Google Scholar] [CrossRef]
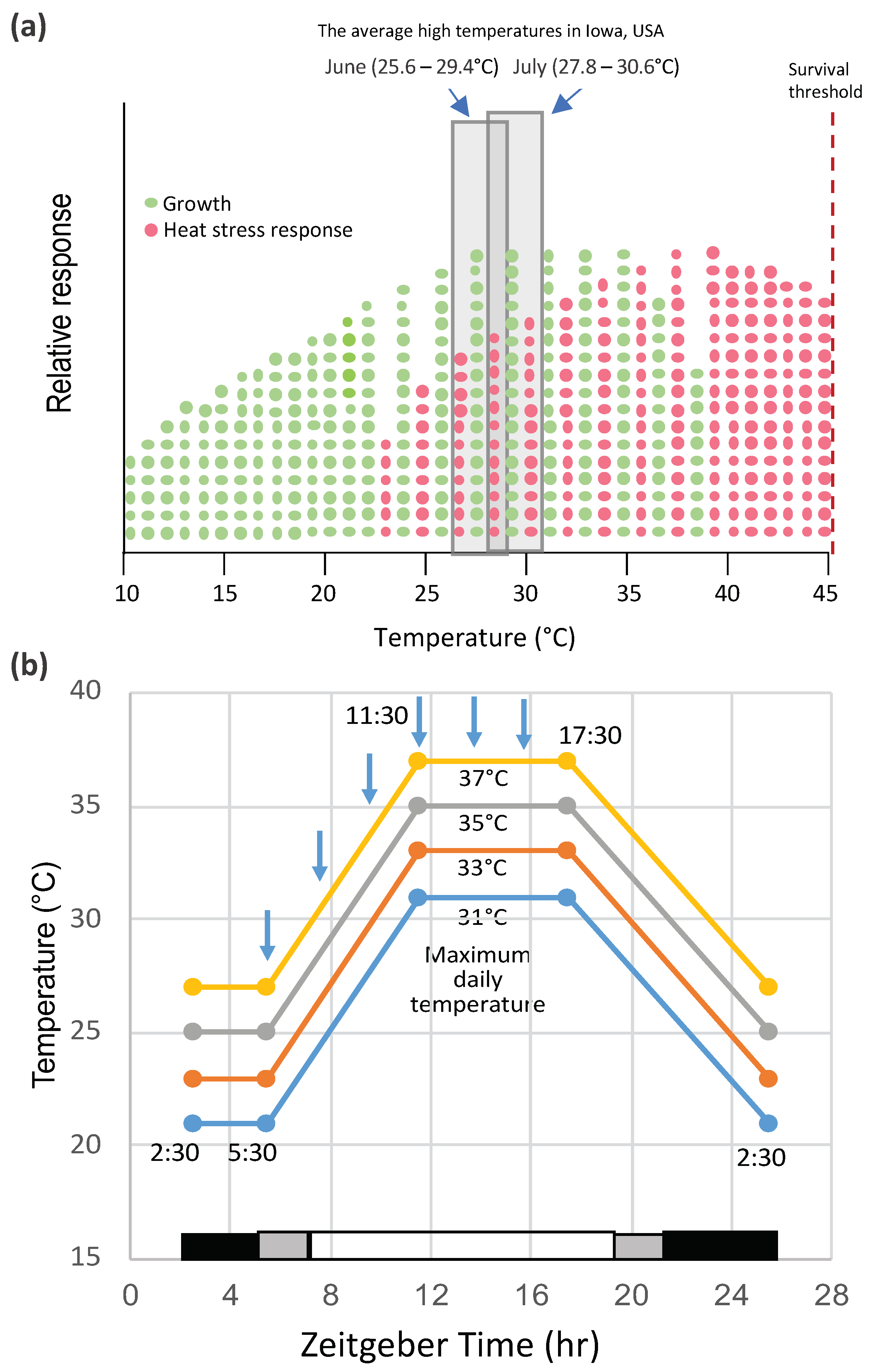

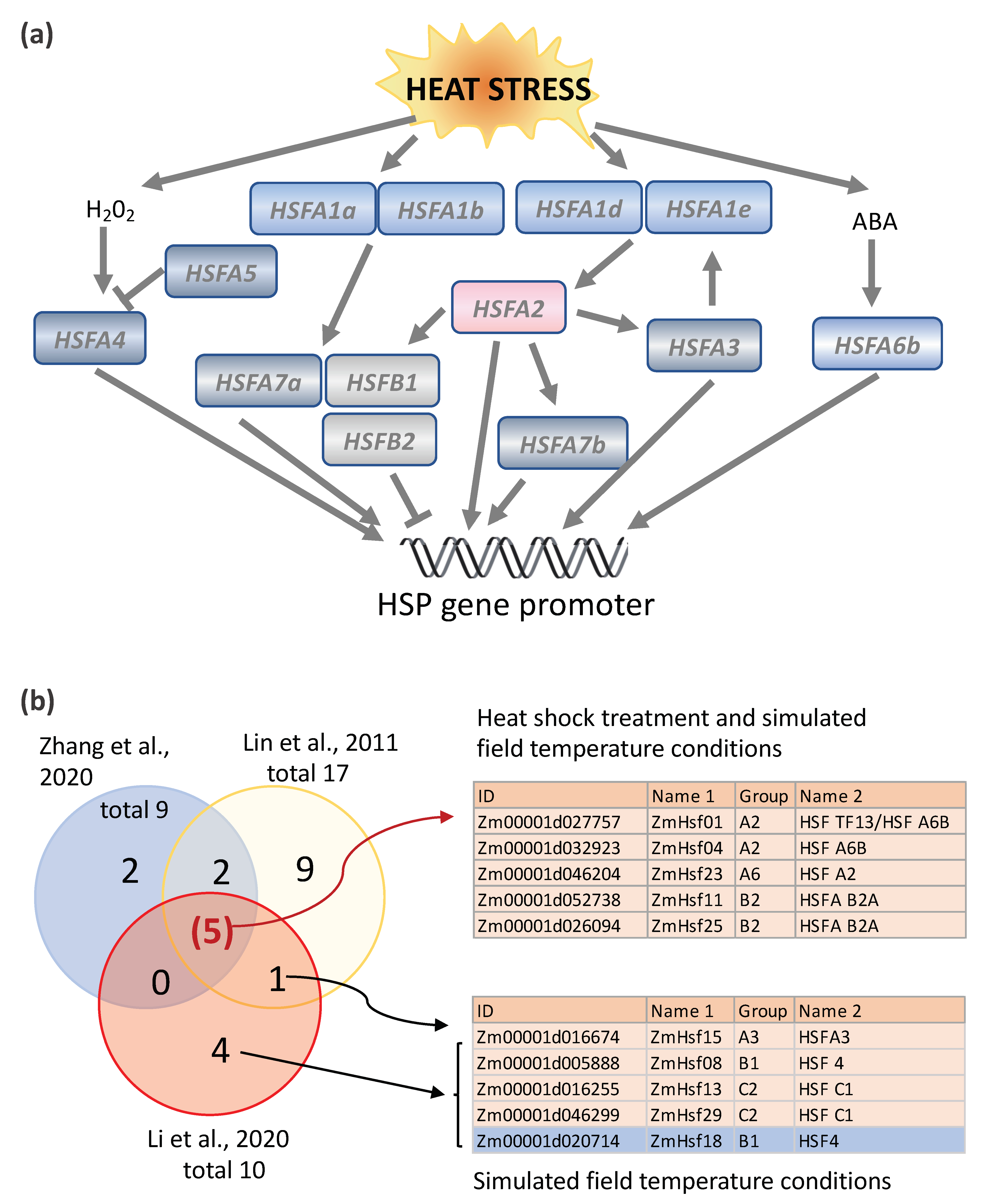
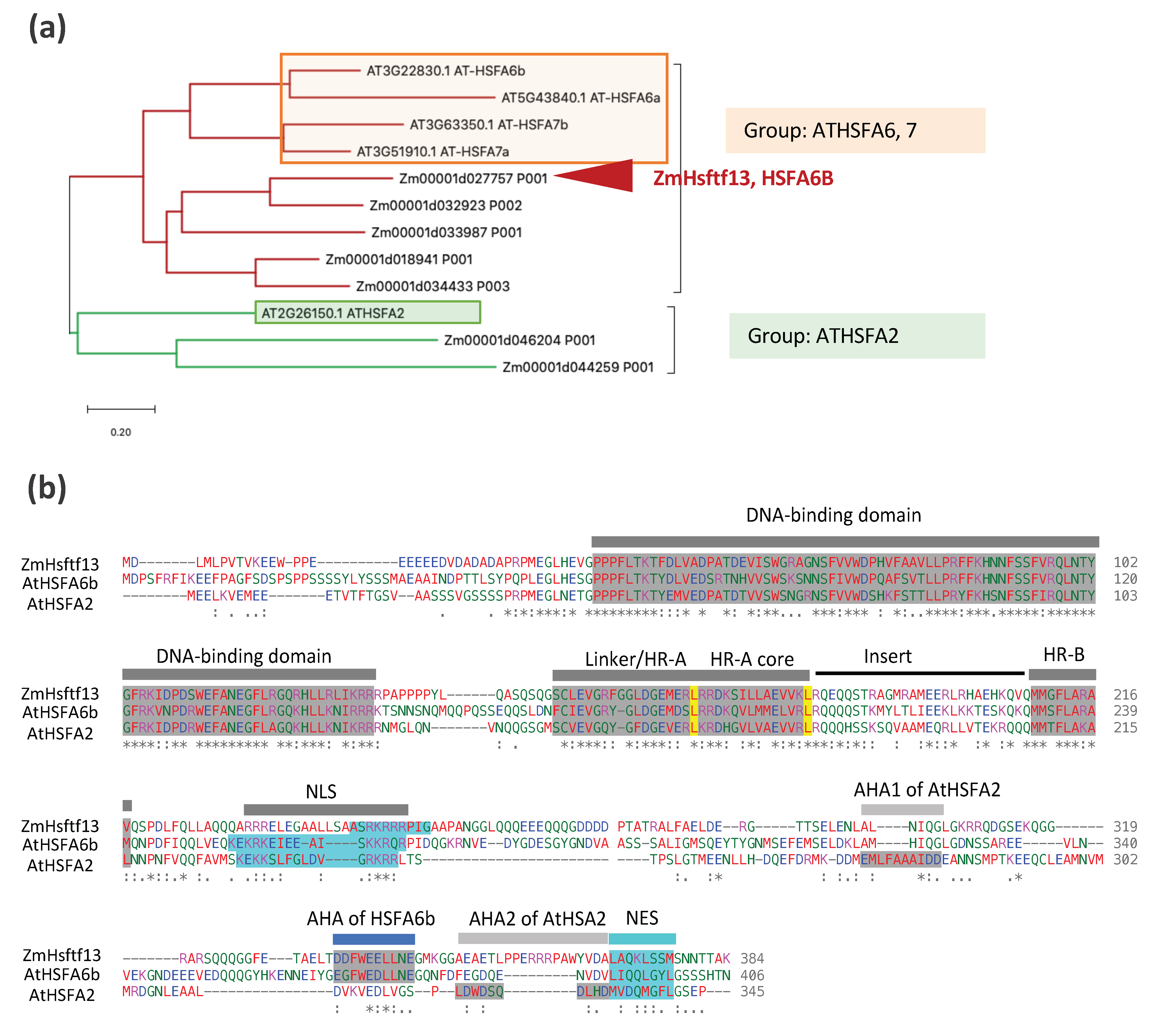
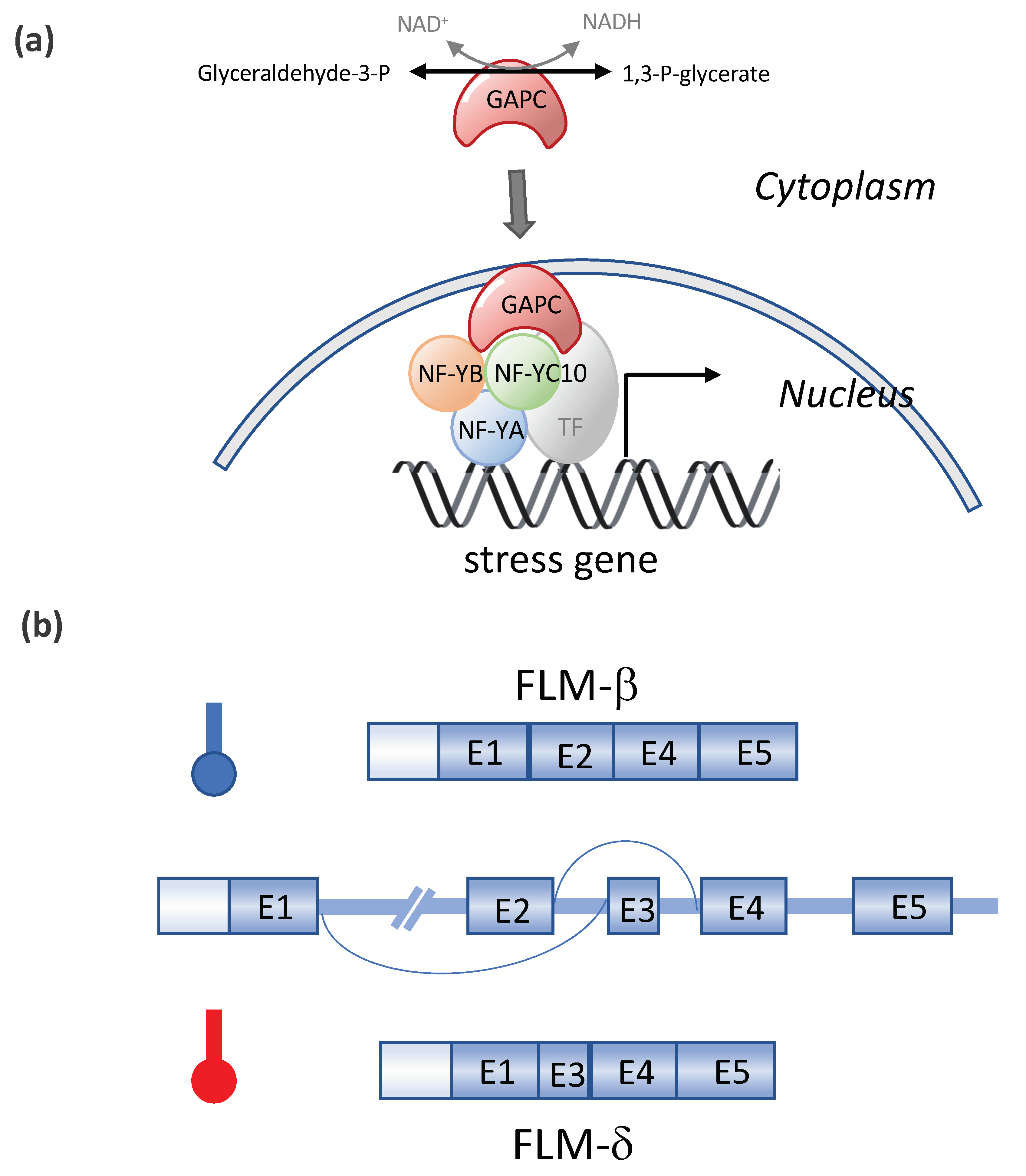
| Gene | Species | Main Results | References |
|---|---|---|---|
| HSR (heat stress response) -mediated transcriptional processes | |||
| HSF1 | Human | HSP (heat shock protein) 70 and the cochaperone Hdj1 interact directly with the transactivation domain of HSF (heat shock transcription factor) 1 and repress heat shock gene transcription | [23] |
| HSF1, HSF2 | Human | Activation of HSF1 and HSF2 including the formation of trimers concomitant with the acquisition of DNA-binding activity and nuclear localization | [24] |
| HSF family | Multiple | Survey of Arabidopsis, rice and maize HSFs, sequence, structure and domain analysis | [27,28,29,30,31,32,33] |
| HSFA1s, HSFA2 | Arabidopsis | HSFA1d and HSFA1e involved in the transcriptional regulation of HSFA2 function as key regulators for the HSF signaling | [34] |
| HSFA2 | Arabidopsis | HSFA2 function as key regulators of HSF signaling, HSFA2 expression levels, heat stress responses and thermotolerance | [34,35,36,37,38,39] |
| HSFA6b | Arabidopsis | HSFA6b connects ABA (abscisic acid) signaling and ABA-mediated heat responses, contributes to thermotolerance, drought and salt tolerance | [40] |
| HSFBs | Arabidopsis | Heat-inducible expression of HSFB1, HSFB2A, and HSFB2B and the repressor activity of HSFBs | [41,42] |
| HSPs | Maize | HSPs response to heat stress in maize | [43,44,45,46,47,48] |
| UPR-mediated transcriptional processes | |||
| bZIP28 | Arabidopsis | bZIP (basic leucine zipper) 28 contributes to the upregulation of heat-responsive genes and heat tolerance | [49] |
| bZIP28, bZIP60 | Arabidopsis | bzip28 bzip60 double-mutant plants are sensitive to heat stress; bZIP28 binds directly to the promoters of heat-responsive genes | [50] |
| IRE1 | Arabidopsis | Heat induces the splicing of bZIP60 by IRE1 (inositol-requiring enzyme 1); IRE1 confers heat stress tolerance, and this is attributable to its RNase domain | [51,52] |
| bZIP60, HSFTF13, HSPs | Maize | Maize heat stress response; bZIP60 links the UPR (unfolded protein response) to the HSR in maize by direct binding bZIP60 to the promoter of Hsftf13 to active its expression | [14] |
| bZIP60 | Maize | bZIP60 expression levels in tropical and temperate maize lines with different heat tolerances | [53] |
| Others transcriptional processes | |||
| ABA/ calcium/ SA/ethylene, etc. | Arabidopsis, maize | The involvement of calcium, ABA, ethylene, and SA (salicylic acid) in protecting against heat-induced oxidative damage in Arabidopsis or maize. | [54,55,56,57] |
| HSFA2c, HSPs, ABA | Tall Fescue, Arabidopsis | FaDREB (dehydration-responsive element-binding protein) 2, FaAREB3 directly bind to DRE/ABRE cis-elements in the FaHSF2c promoter to drive expression and improve heat tolerance in Tall Fescue and Arabidopsis | [58] |
| ERF1/JA/ ethylene | Arabidopsis | ERF (ETHYLENE RESPONSE FACTOR) 1 activates target genes by binding to GCC boxes or DRE/CRT elements in HSFA3 and HSP70, and enhances tolerance to heat and other abiotic stresses. | [59] |
| JA/AOS/ ROS | Arabidopsis | JA (jasmonic acid) and ROS (reactive oxygen species) are required for plant acclimation to a combination of high light and heat stress, at both local and systemic level | [60,61] |
| DREB2A | Arabidopsis | Thermotolerance was significantly increased in plants overexpressing DREB2A CA and decreased in DREB2A knockout plants | [62] |
| DREB2A/ NF-YC10 | Arabidopsis | As a DREB2A interactor, NF-YC10 (NUCLEAR FACTOR Y, SUBUNIT C 10) formed a transcriptional complex with NF-YA and NF-YB subunits, and the trimer enhanced heat stress-inducible gene expression in conjunction with DREB2A | [63] |
| NF-YC10/ GAPC | Arabidopsis | GAPC (glyceraldehyde-3-phosphate dehydrogenase) enters the nucleus in response to heat where it binds to NF- YC10 to increase the expression of heat-inducible genes, rendering Arabidopsis tolerant to heat stress. | [64] |
| SPL1, SPL12 | Arabidopsis | SPL (SQUAMOSA PROMOTER BINDING PROTEIN-LIKE) 1 and SPL12 act redundantly to enhance thermotolerance through PYL-mediated ABA signaling in Arabidopsis | [65] |
| SDG25, ATX1 | Arabidopsis | Histone H3K4 methyltransferases SDG25 and ATX1 regulate histone H3K4me3 level and prevent DNA methylation at loci associated with heat stress gene expression during stress recovery | [66] |
| APX1, 2 | Arabidopsis | HSF21 is key in the early sensing of H2O2 stress in KO-Apx1 (ascorbate peroxidase 1) plants. Increased H2O2 production during the early phase of heat stress is necessary for the HSFs-mediated induction of HSP17.6 and HSP18.2 | [67,68,69] |
| Post-transcriptional processes | |||
| AS, SR45a | Maize | Elevated maximum daily temperature induces alternative splicing and the roles of SR (serine/arginine-rich) 45a | [70] |
| HSF2A, HSFs | Arabidopsis, maize | HSF2A alternative splicing in response to heat stress | [30,71,72] |
| SR genes | Arabidopsis | Alternative splicing of SR genes in response to abiotic stress, including heat stress | [73] |
| FLM | Arabidopsis | Heat-induced alternative splicing of FLM (FLOWERING LOCUS M) isoforms and temperature dependent flowering | [74,75,76,77,78] |
| miR156-SPL | Arabidopsis | The miR156-SPL module mediates the response to recurring heat stress in Arabidopsis | [79] |
| miRNAs | Arabidopsis, maize | Identification of the miRNA in Arabidopsis and maize involved in post-transcriptional regulation of the response to heat stress | [80,81] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Li, Z.; Howell, S.H. Heat Stress Responses and Thermotolerance in Maize. Int. J. Mol. Sci. 2021, 22, 948. https://doi.org/10.3390/ijms22020948
Li Z, Howell SH. Heat Stress Responses and Thermotolerance in Maize. International Journal of Molecular Sciences. 2021; 22(2):948. https://doi.org/10.3390/ijms22020948
Chicago/Turabian StyleLi, Zhaoxia, and Stephen H. Howell. 2021. "Heat Stress Responses and Thermotolerance in Maize" International Journal of Molecular Sciences 22, no. 2: 948. https://doi.org/10.3390/ijms22020948
APA StyleLi, Z., & Howell, S. H. (2021). Heat Stress Responses and Thermotolerance in Maize. International Journal of Molecular Sciences, 22(2), 948. https://doi.org/10.3390/ijms22020948





