CD39 Regulation and Functions in T Cells
Abstract
:1. Adenosine Pathway and CD39/CD73 Expression in the Tumor Microenvironment
2. CD39 Expression and Functions in Conventional CD4+ T Cells and Tregs
3. CD39 Expression and Functions in CD8+ T Cells
4. CD39 Regulation by Cytokines
5. CD39 Regulation by Genetic Factors
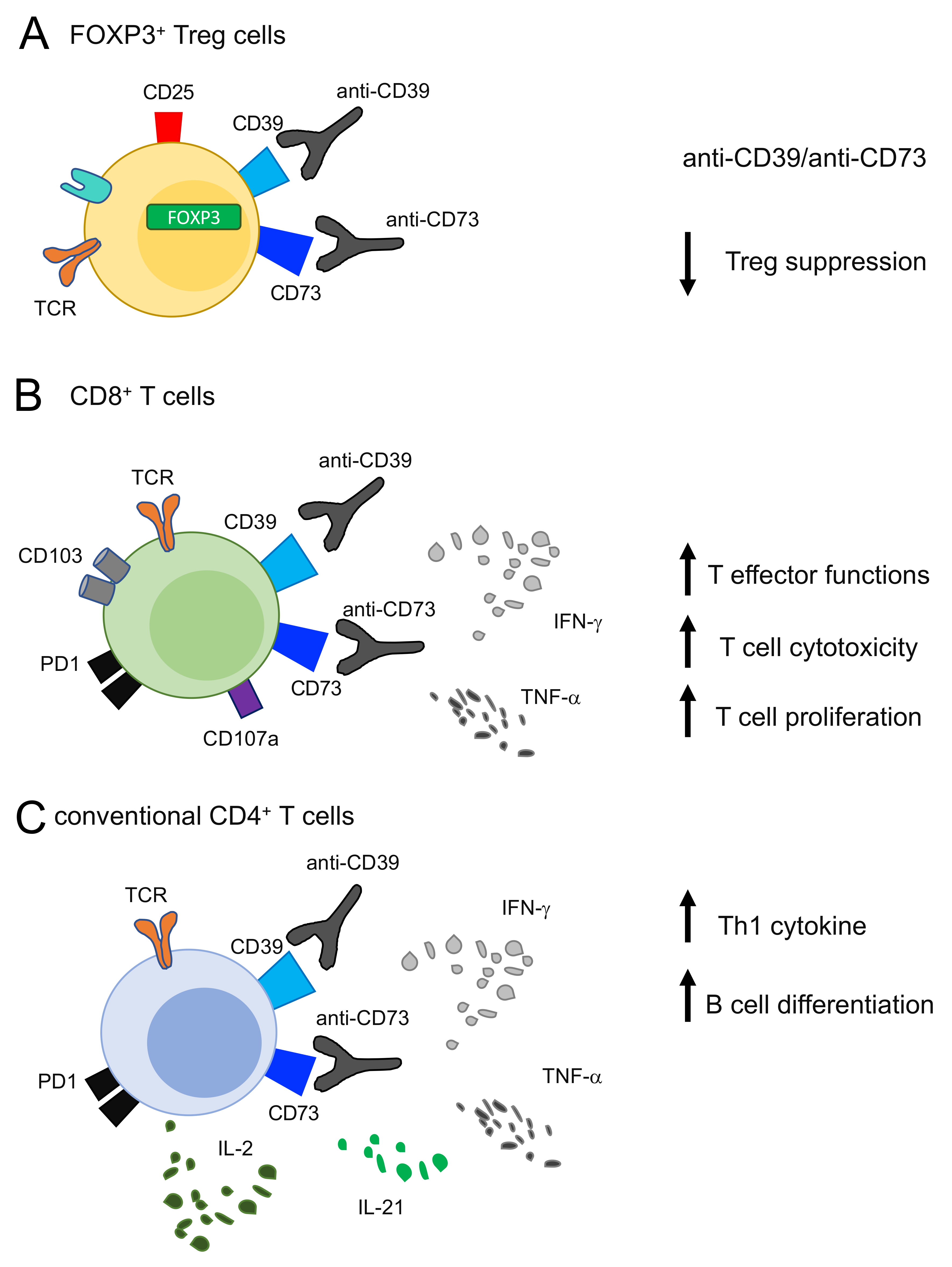
6. Targeting Adenosine Pathway, CD73 and CD39 as Anti-Tumor Strategy
7. CD39 Targeting for Immunotherapy Strategies
8. Future Perspectives
Funding
Conflicts of Interest
Abbreviations
| regulatory T cells (Tregs) |
| conventional CD4 T cells (cCD4+ T cells) |
| cancer-associated fibroblasts (CAF) |
| multiple sclerosis (MS) |
| tumor-infiltrating (TI) |
| tumor-infiltrating lymphocytes (TILs) |
| natural killer (NK) |
| tumor-associated-macrophages (TAM) |
| tumor-microenvironment (TME) |
| adenosine triphosphate (ATP) |
| adenosine diphosphate (ADP) |
| cyclic adenosine monophosphate (cAMP) |
| extracellular ATP (eATP) |
| immune check point blocked (ICB) |
| single nucleotide polymorphism (SNP) |
References
- Zimmermann, H. 5′-Nucleotidase: Molecular Structure and Functional Aspects. Biochem. J. 1992, 285, 345–365. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Graeff, R.M.; Walseth, T.F.; Fryxell, K.; Branton, W.D.; Lee, H.C. Enzymatic Synthesis and Characterizations of Cyclic GDP-Ribose. A Procedure for Distinguishing Enzymes with ADP-Ribosyl Cyclase Activity. J. Biol. Chem. 1994, 269, 30260–30267. [Google Scholar] [CrossRef]
- Robson, S.C.; Sévigny, J.; Zimmermann, H. The E-NTPDase Family of Ectonucleotidases: Structure Function Relationships and Pathophysiological Significance. Purinergic Signal. 2006, 2, 409–430. [Google Scholar] [CrossRef] [Green Version]
- Kaczmarek, E.; Koziak, K.; Sévigny, J.; Siegel, J.B.; Anrather, J.; Beaudoin, A.R.; Bach, F.H.; Robson, S.C. Identification and Characterization of CD39/Vascular ATP Diphosphohydrolase. J. Biol. Chem. 1996, 271, 33116–33122. [Google Scholar] [CrossRef] [Green Version]
- Enjyoji, K.; Sévigny, J.; Lin, Y.; Frenette, P.S.; Christie, P.D.; Esch, J.S.; Imai, M.; Edelberg, J.M.; Rayburn, H.; Lech, M.; et al. Targeted Disruption of Cd39/ATP Diphosphohydrolase Results in Disordered Hemostasis and Thromboregulation. Nat. Med. 1999, 5, 1010–1017. [Google Scholar] [CrossRef] [PubMed]
- Ohta, A.; Sitkovsky, M. Role of G-Protein-Coupled Adenosine Receptors in Downregulation of Inflammation and Protection from Tissue Damage. Nature 2001, 414, 916–920. [Google Scholar] [CrossRef] [Green Version]
- Huang, S.; Apasov, S.; Koshiba, M.; Sitkovsky, M. Role of A2a Extracellular Adenosine Receptor-Mediated Signaling in Adenosine-Mediated Inhibition of T-Cell Activation and Expansion. Blood 1997, 90, 1600–1610. [Google Scholar] [CrossRef] [PubMed]
- Wolberg, G.; Zimmerman, T.P.; Hiemstra, K.; Winston, M.; Chu, L.C. Adenosine Inhibition of Lymphocyte-Mediated Cytolysis: Possible Role of Cyclic Adenosine Monophosphate. Science 1975, 187, 957–959. [Google Scholar] [CrossRef] [PubMed]
- Moesta, A.K.; Li, X.-Y.; Smyth, M.J. Targeting CD39 in Cancer. Nat. Rev. Immunol. 2020, 20, 739–755. [Google Scholar] [CrossRef] [PubMed]
- Ohta, A.; Gorelik, E.; Prasad, S.J.; Ronchese, F.; Lukashev, D.; Wong, M.K.K.; Huang, X.; Caldwell, S.; Liu, K.; Smith, P.; et al. A2A Adenosine Receptor Protects Tumors from Antitumor T Cells. Proc. Natl. Acad. Sci. USA 2006, 103, 13132–13137. [Google Scholar] [CrossRef] [Green Version]
- Mittal, D.; Sinha, D.; Barkauskas, D.; Young, A.; Kalimutho, M.; Stannard, K.; Caramia, F.; Haibe-Kains, B.; Stagg, J.; Khanna, K.K.; et al. Adenosine 2B Receptor Expression on Cancer Cells Promotes Metastasis. Cancer Res. 2016, 76, 4372–4382. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chen, S.; Akdemir, I.; Fan, J.; Linden, J.; Zhang, B.; Cekic, C. The Expression of Adenosine A2B Receptor on Antigen-Presenting Cells Suppresses CD8+ T-Cell Responses and Promotes Tumor Growth. Cancer Immunol. Res. 2020, 8, 1064–1074. [Google Scholar] [CrossRef]
- Sciaraffia, E.; Riccomi, A.; Lindstedt, R.; Gesa, V.; Cirelli, E.; Patrizio, M.; De Magistris, M.T.; Vendetti, S. Human Monocytes Respond to Extracellular CAMP through A2A and A2B Adenosine Receptors. J. Leukoc. Biol. 2014, 96, 113–122. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mirabet, M.; Herrera, C.; Cordero, O.J.; Mallol, J.; Lluis, C.; Franco, R. Expression of A2B Adenosine Receptors in Human Lymphocytes: Their Role in T Cell Activation. J. Cell Sci. 1999, 112, 491–502. [Google Scholar] [CrossRef] [PubMed]
- Di Virgilio, F.; Adinolfi, E. Extracellular Purines, Purinergic Receptors and Tumor Growth. Oncogene 2017, 36, 293–303. [Google Scholar] [CrossRef]
- Di Virgilio, F.; Dal Ben, D.; Sarti, A.C.; Giuliani, A.L.; Falzoni, S. The P2 × 7 Receptor in Infection and Inflammation. Immunity 2017, 47, 15–31. [Google Scholar] [CrossRef] [Green Version]
- Di Virgilio, F.; Vuerich, M. Purinergic Signaling in the Immune System. Auton. Neurosci. 2015, 191, 117–123. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ferrari, D.; Chiozzi, P.; Falzoni, S.; Dal Susino, M.; Melchiorri, L.; Baricordi, O.R.; Di Virgilio, F. Extracellular ATP Triggers IL-1 Beta Release by Activating the Purinergic P2Z Receptor of Human Macrophages. J. Immunol. 1997, 159, 1451–1458. [Google Scholar]
- Wu, J.; Wang, Y.-C.; Xu, W.-H.; Luo, W.-J.; Wan, F.-N.; Zhang, H.-L.; Ye, D.-W.; Qu, Y.-Y.; Zhu, Y.-P. High Expression of CD39 Is Associated with Poor Prognosis and Immune Infiltrates in Clear Cell Renal Cell Carcinoma. Onco Targets Ther. 2020, 13, 10453–10464. [Google Scholar] [CrossRef]
- Hayes, G.M.; Cairns, B.; Levashova, Z.; Chinn, L.; Perez, M.; Theunissen, J.-W.; Liao-Chan, S.; Bermudez, A.; Flory, M.R.; Schweighofer, K.J.; et al. CD39 Is a Promising Therapeutic Antibody Target for the Treatment of Soft Tissue Sarcoma. Am. J. Transl. Res. 2015, 7, 1181–1188. [Google Scholar]
- Bastid, J.; Regairaz, A.; Bonnefoy, N.; Déjou, C.; Giustiniani, J.; Laheurte, C.; Cochaud, S.; Laprevotte, E.; Funck-Brentano, E.; Hemon, P.; et al. Inhibition of CD39 Enzymatic Function at the Surface of Tumor Cells Alleviates Their Immunosuppressive Activity. Cancer Immunol. Res. 2015, 3, 254–265. [Google Scholar] [CrossRef] [Green Version]
- Häusler, S.F.M.; Montalbán del Barrio, I.; Strohschein, J.; Chandran, P.A.; Engel, J.B.; Hönig, A.; Ossadnik, M.; Horn, E.; Fischer, B.; Krockenberger, M.; et al. Ectonucleotidases CD39 and CD73 on OvCA Cells Are Potent Adenosine-Generating Enzymes Responsible for Adenosine Receptor 2A-Dependent Suppression of T Cell Function and NK Cell Cytotoxicity. Cancer Immunol. Immunother. 2011, 60, 1405–1418. [Google Scholar] [CrossRef]
- Simoni, Y.; Becht, E.; Fehlings, M.; Loh, C.Y.; Koo, S.-L.; Teng, K.W.W.; Yeong, J.P.S.; Nahar, R.; Zhang, T.; Kared, H.; et al. Bystander CD8+ T Cells Are Abundant and Phenotypically Distinct in Human Tumour Infiltrates. Nature 2018, 557, 575–579. [Google Scholar] [CrossRef]
- Borsellino, G.; Kleinewietfeld, M.; Di Mitri, D.; Sternjak, A.; Diamantini, A.; Giometto, R.; Höpner, S.; Centonze, D.; Bernardi, G.; Dell’Acqua, M.L.; et al. Expression of Ectonucleotidase CD39 by Foxp3+ Treg Cells: Hydrolysis of Extracellular ATP and Immune Suppression. Blood 2007, 110, 1225–1232. [Google Scholar] [CrossRef] [Green Version]
- Li, X.-Y.; Moesta, A.K.; Xiao, C.; Nakamura, K.; Casey, M.; Zhang, H.; Madore, J.; Lepletier, A.; Aguilera, A.R.; Sundarrajan, A.; et al. Targeting CD39 in Cancer Reveals an Extracellular ATP- and Inflammasome-Driven Tumor Immunity. Cancer Discov. 2019, 9, 1754–1773. [Google Scholar] [CrossRef] [Green Version]
- Yan, J.; Li, X.-Y.; Roman Aguilera, A.; Xiao, C.; Jacoberger-Foissac, C.; Nowlan, B.; Robson, S.C.; Beers, C.; Moesta, A.K.; Geetha, N.; et al. Control of Metastases via Myeloid CD39 and NK Cell Effector Function. Cancer Immunol. Res. 2020, 8, 356–367. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhang, H.; Vijayan, D.; Li, X.-Y.; Robson, S.C.; Geetha, N.; Teng, M.W.L.; Smyth, M.J. The Role of NK Cells and CD39 in the Immunological Control of Tumor Metastases. OncoImmunology 2019, 8, e1593809. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Canale, F.P.; Ramello, M.C.; Núñez, N.; Araujo Furlan, C.L.; Bossio, S.N.; Gorosito Serrán, M.; Tosello Boari, J.; Del Castillo, A.; Ledesma, M.; Sedlik, C.; et al. CD39 Expression Defines Cell Exhaustion in Tumor-Infiltrating CD8+ T Cells. Cancer Res. 2018, 78, 115–128. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cai, X.-Y.; Wang, X.-F.; Li, J.; Dong, J.-N.; Liu, J.-Q.; Li, N.-P.; Yun, B.; Xia, R.-L.; Qin, J.; Sun, Y.-H. High Expression of CD39 in Gastric Cancer Reduces Patient Outcome Following Radical Resection. Oncol. Lett. 2016, 12, 4080–4086. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Muñóz-Godínez, R.; de Lourdes Mora-García, M.; Weiss-Steider, B.; Montesinos-Montesinos, J.J.; Del Carmen Aguilar-Lemarroy, A.; García-Rocha, R.; Hernández-Montes, J.; Azucena Don-López, C.; Ávila-Ibarra, L.R.; Torres-Pineda, D.B.; et al. Detection of CD39 and a Highly Glycosylated Isoform of Soluble CD73 in the Plasma of Patients with Cervical Cancer: Correlation with Disease Progression. Mediat. Inflamm. 2020, 2020, 1678780. [Google Scholar] [CrossRef]
- Cai, X.-Y.; Ni, X.-C.; Yi, Y.; He, H.-W.; Wang, J.-X.; Fu, Y.-P.; Sun, J.; Zhou, J.; Cheng, Y.-F.; Jin, J.-J.; et al. Overexpression of CD39 in Hepatocellular Carcinoma Is an Independent Indicator of Poor Outcome after Radical Resection. Medicine 2016, 95, e4989. [Google Scholar] [CrossRef] [PubMed]
- Ma, X.-L.; Hu, B.; Tang, W.-G.; Xie, S.-H.; Ren, N.; Guo, L.; Lu, R.-Q. CD73 Sustained Cancer-Stem-Cell Traits by Promoting SOX9 Expression and Stability in Hepatocellular Carcinoma. J. Hematol. Oncol. 2020, 13, 11. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Young, A.; Ngiow, S.F.; Barkauskas, D.S.; Sult, E.; Hay, C.; Blake, S.J.; Huang, Q.; Liu, J.; Takeda, K.; Teng, M.W.L.; et al. Co-Inhibition of CD73 and A2AR Adenosine Signaling Improves Anti-Tumor Immune Responses. Cancer Cell 2016, 30, 391–403. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chen, S.; Fan, J.; Zhang, M.; Qin, L.; Dominguez, D.; Long, A.; Wang, G.; Ma, R.; Li, H.; Zhang, Y.; et al. CD73 Expression on Effector T Cells Sustained by TGF-β Facilitates Tumor Resistance to Anti-4-1BB/CD137 Therapy. Nat. Commun. 2019, 10, 150. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Neo, S.Y.; Yang, Y.; Record, J.; Ma, R.; Chen, X.; Chen, Z.; Tobin, N.P.; Blake, E.; Seitz, C.; Thomas, R.; et al. CD73 Immune Checkpoint Defines Regulatory NK Cells within the Tumor Microenvironment. J. Clin. Investig. 2020, 130, 1185–1198. [Google Scholar] [CrossRef] [Green Version]
- Wettstein, M.S.; Buser, L.; Hermanns, T.; Roudnicky, F.; Eberli, D.; Baumeister, P.; Sulser, T.; Wild, P.; Poyet, C. CD73 Predicts Favorable Prognosis in Patients with Nonmuscle-Invasive Urothelial Bladder Cancer. Dis. Markers 2015, 2015, 785461. [Google Scholar] [CrossRef]
- Chen, Q.; Pu, N.; Yin, H.; Zhang, J.; Zhao, G.; Lou, W.; Wu, W. CD73 Acts as a Prognostic Biomarker and Promotes Progression and Immune Escape in Pancreatic Cancer. J. Cell Mol. Med. 2020, 24, 8674–8686. [Google Scholar] [CrossRef]
- Ren, Z.-H.; Lin, C.-Z.; Cao, W.; Yang, R.; Lu, W.; Liu, Z.-Q.; Chen, Y.-M.; Yang, X.; Tian, Z.; Wang, L.-Z.; et al. CD73 Is Associated with Poor Prognosis in HNSCC. Oncotarget 2016, 7, 61690–61702. [Google Scholar] [CrossRef] [Green Version]
- Monteiro, I.; Vigano, S.; Faouzi, M.; Treilleux, I.; Michielin, O.; Ménétrier-Caux, C.; Caux, C.; Romero, P.; de Leval, L. CD73 Expression and Clinical Significance in Human Metastatic Melanoma. Oncotarget 2018, 9, 26659–26669. [Google Scholar] [CrossRef] [Green Version]
- Kashyap, A.S.; Thelemann, T.; Klar, R.; Kallert, S.M.; Festag, J.; Buchi, M.; Hinterwimmer, L.; Schell, M.; Michel, S.; Jaschinski, F.; et al. Antisense Oligonucleotide Targeting CD39 Improves Anti-Tumor T Cell Immunity. J. Immunother. Cancer 2019, 7, 67. [Google Scholar] [CrossRef] [Green Version]
- Allard, B.; Longhi, M.S.; Robson, S.C.; Stagg, J. The Ectonucleotidases CD39 and CD73: Novel Checkpoint Inhibitor Targets. Immunol. Rev. 2017, 276, 121–144. [Google Scholar] [CrossRef] [Green Version]
- Stagg, J.; Divisekera, U.; McLaughlin, N.; Sharkey, J.; Pommey, S.; Denoyer, D.; Dwyer, K.M.; Smyth, M.J. Anti-CD73 Antibody Therapy Inhibits Breast Tumor Growth and Metastasis. Proc. Natl. Acad. Sci. USA 2010, 107, 1547–1552. [Google Scholar] [CrossRef] [Green Version]
- Stagg, J.; Divisekera, U.; Duret, H.; Sparwasser, T.; Teng, M.W.L.; Darcy, P.K.; Smyth, M.J. CD73-Deficient Mice Have Increased Antitumor Immunity and Are Resistant to Experimental Metastasis. Cancer Res. 2011, 71, 2892–2900. [Google Scholar] [CrossRef] [Green Version]
- Gallerano, D.; Ciminati, S.; Grimaldi, A.; Piconese, S.; Cammarata, I.; Focaccetti, C.; Pacella, I.; Accapezzato, D.; Lancellotti, F.; Sacco, L.; et al. Genetically Driven CD39 Expression Shapes Human Tumor-Infiltrating CD8+ T-Cell Functions. Int. J. Cancer 2020, 147, 2597–2610. [Google Scholar] [CrossRef]
- Dang, E.V.; Barbi, J.; Yang, H.-Y.; Jinasena, D.; Yu, H.; Zheng, Y.; Bordman, Z.; Fu, J.; Kim, Y.; Yen, H.-R.; et al. Control of T(H)17/T(Reg) Balance by Hypoxia-Inducible Factor 1. Cell 2011, 146, 772–784. [Google Scholar] [CrossRef] [Green Version]
- Liao, H.; Hyman, M.C.; Baek, A.E.; Fukase, K.; Pinsky, D.J. CAMP/CREB-Mediated Transcriptional Regulation of Ectonucleoside Triphosphate Diphosphohydrolase 1 (CD39) Expression. J. Biol. Chem. 2010, 285, 14791–14805. [Google Scholar] [CrossRef] [Green Version]
- Ostapchuk, Y.O.; Perfilyeva, Y.V.; Kustova, E.A.; Urazalieva, N.T.; Omarbaeva, N.A.; Talaeva, S.G.; Belyaev, N.N. Functional Heterogeneity of Circulating T Regulatory Cell Subsets in Breast Cancer Patients. Breast Cancer 2018, 25, 687–697. [Google Scholar] [CrossRef] [PubMed]
- Głowala-Kosińska, M.; Chwieduk, A.; Nieckula, J.; Saduś-Wojciechowska, M.; Grosicki, S.; Rusin, A.; Nowara, E.; Giebel, S. Association of Circulating Regulatory T Cell Number with the Incidence and Prognosis of Diffuse Large B-Cell Lymphoma. Eur. J. Haematol. 2013, 91, 122–128. [Google Scholar] [CrossRef] [PubMed]
- Retseck, J.; Nasr, A.; Lin, Y.; Lin, H.; Mendiratta, P.; Butterfield, L.H.; Tarhini, A.A. Long Term Impact of CTLA4 Blockade Immunotherapy on Regulatory and Effector Immune Responses in Patients with Melanoma. J. Transl. Med. 2018, 16, 184. [Google Scholar] [CrossRef]
- Mandapathil, M.; Szczepanski, M.J.; Szajnik, M.; Ren, J.; Lenzner, D.E.; Jackson, E.K.; Gorelik, E.; Lang, S.; Johnson, J.T.; Whiteside, T.L. Increased Ectonucleotidase Expression and Activity in Regulatory T Cells of Patients with Head and Neck Cancer. Clin. Cancer Res. 2009, 15, 6348–6357. [Google Scholar] [CrossRef] [Green Version]
- Timperi, E.; Pacella, I.; Schinzari, V.; Focaccetti, C.; Sacco, L.; Farelli, F.; Caronna, R.; Del Bene, G.; Longo, F.; Ciardi, A.; et al. Regulatory T Cells with Multiple Suppressive and Potentially Pro-Tumor Activities Accumulate in Human Colorectal Cancer. Oncoimmunology 2016, 5, e1175800. [Google Scholar] [CrossRef] [Green Version]
- Ahlmanner, F.; Sundström, P.; Akeus, P.; Eklöf, J.; Börjesson, L.; Gustavsson, B.; Lindskog, E.B.; Raghavan, S.; Quiding-Järbrink, M. CD39+ Regulatory T Cells Accumulate in Colon Adenocarcinomas and Display Markers of Increased Suppressive Function. Oncotarget 2018, 9, 36993–37007. [Google Scholar] [CrossRef] [Green Version]
- Piconese, S.; Timperi, E.; Pacella, I.; Schinzari, V.; Tripodo, C.; Rossi, M.; Guglielmo, N.; Mennini, G.; Grazi, G.L.; Di Filippo, S.; et al. Human OX40 Tunes the Function of Regulatory T Cells in Tumor and Nontumor Areas of Hepatitis C Virus-Infected Liver Tissue. Hepatology 2014, 60, 1494–1507. [Google Scholar] [CrossRef] [Green Version]
- Kortekaas, K.E.; Santegoets, S.J.; Sturm, G.; Ehsan, I.; van Egmond, S.L.; Finotello, F.; Trajanoski, Z.; Welters, M.J.P.; van Poelgeest, M.I.E.; van der Burg, S.H. CD39 Identifies the CD4+ Tumor-Specific T-Cell Population in Human Cancer. Cancer Immunol. Res. 2020, 8, 1311–1321. [Google Scholar] [CrossRef]
- Schenk, U.; Frascoli, M.; Proietti, M.; Geffers, R.; Traggiai, E.; Buer, J.; Ricordi, C.; Westendorf, A.M.; Grassi, F. ATP Inhibits the Generation and Function of Regulatory T Cells through the Activation of Purinergic P2X Receptors. Sci. Signal. 2011, 4, ra12. [Google Scholar] [CrossRef]
- Chalmin, F.; Mignot, G.; Bruchard, M.; Chevriaux, A.; Végran, F.; Hichami, A.; Ladoire, S.; Derangère, V.; Vincent, J.; Masson, D.; et al. Stat3 and Gfi-1 Transcription Factors Control Th17 Cell Immunosuppressive Activity via the Regulation of Ectonucleotidase Expression. Immunity 2012, 36, 362–373. [Google Scholar] [CrossRef] [Green Version]
- Gupta, P.K.; Godec, J.; Wolski, D.; Adland, E.; Yates, K.; Pauken, K.E.; Cosgrove, C.; Ledderose, C.; Junger, W.G.; Robson, S.C.; et al. CD39 Expression Identifies Terminally Exhausted CD8+ T Cells. PLoS Pathog. 2015, 11, e1005177. [Google Scholar] [CrossRef] [Green Version]
- Qi, Y.; Xia, Y.; Lin, Z.; Qu, Y.; Qi, Y.; Chen, Y.; Zhou, Q.; Zeng, H.; Wang, J.; Chang, Y.; et al. Tumor-Infiltrating CD39+CD8+ T Cells Determine Poor Prognosis and Immune Evasion in Clear Cell Renal Cell Carcinoma Patients. Cancer Immunol. Immunother. 2020, 69, 1565–1576. [Google Scholar] [CrossRef]
- Yost, K.E.; Satpathy, A.T.; Wells, D.K.; Qi, Y.; Wang, C.; Kageyama, R.; McNamara, K.L.; Granja, J.M.; Sarin, K.Y.; Brown, R.A.; et al. Clonal Replacement of Tumor-Specific T Cells Following PD-1 Blockade. Nat. Med. 2019, 25, 1251–1259. [Google Scholar] [CrossRef] [PubMed]
- Duhen, T.; Duhen, R.; Montler, R.; Moses, J.; Moudgil, T.; de Miranda, N.F.; Goodall, C.P.; Blair, T.C.; Fox, B.A.; McDermott, J.E.; et al. Co-Expression of CD39 and CD103 Identifies Tumor-Reactive CD8 T Cells in Human Solid Tumors. Nat. Commun. 2018, 9, 2724. [Google Scholar] [CrossRef] [Green Version]
- Workel, H.H.; van Rooij, N.; Plat, A.; Spierings, D.C.J.; Fehrmann, R.S.N.; Nijman, H.W.; de Bruyn, M. Transcriptional Activity and Stability of CD39+CD103+CD8+ T Cells in Human High-Grade Endometrial Cancer. Int. J. Mol. Sci. 2020, 21, 3770. [Google Scholar] [CrossRef]
- Webb, J.R.; Milne, K.; Watson, P.; Deleeuw, R.J.; Nelson, B.H. Tumor-Infiltrating Lymphocytes Expressing the Tissue Resident Memory Marker CD103 Are Associated with Increased Survival in High-Grade Serous Ovarian Cancer. Clin. Cancer Res. 2014, 20, 434–444. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kim, H.-D.; Jeong, S.; Park, S.; Lee, Y.J.; Ju, Y.S.; Kim, D.; Song, G.-W.; Lee, J.H.; Kim, S.-Y.; Shin, J.; et al. Implication of CD69+ CD103+ Tissue-Resident-like CD8+ T Cells as a Potential Immunotherapeutic Target for Cholangiocarcinoma. Liver Int. 2021, 41, 764–776. [Google Scholar] [CrossRef] [PubMed]
- Vieyra-Lobato, M.R.; Vela-Ojeda, J.; Montiel-Cervantes, L.; López-Santiago, R.; Moreno-Lafont, M.C. Description of CD8+ Regulatory T Lymphocytes and Their Specific Intervention in Graft-versus-Host and Infectious Diseases, Autoimmunity, and Cancer. J. Immunol. Res. 2018, 2018, 3758713. [Google Scholar] [CrossRef] [Green Version]
- Noble, A.; Mehta, H.; Lovell, A.; Papaioannou, E.; Fairbanks, L. IL-12 and IL-4 Activate a CD39-Dependent Intrinsic Peripheral Tolerance Mechanism in CD8(+) T Cells. Eur. J. Immunol. 2016, 46, 1438–1448. [Google Scholar] [CrossRef] [Green Version]
- Parodi, A.; Battaglia, F.; Kalli, F.; Ferrera, F.; Conteduca, G.; Tardito, S.; Stringara, S.; Ivaldi, F.; Negrini, S.; Borgonovo, G.; et al. CD39 Is Highly Involved in Mediating the Suppression Activity of Tumor-Infiltrating CD8+ T Regulatory Lymphocytes. Cancer Immunol. Immunother. 2013, 62, 851–862. [Google Scholar] [CrossRef]
- Schuler, P.J.; Schilling, B.; Harasymczuk, M.; Hoffmann, T.K.; Johnson, J.; Lang, S.; Whiteside, T.L. Phenotypic and Functional Characteristics of CD4+ CD39+ FOXP3+ and CD4+ CD39+ FOXP3neg T-Cell Subsets in Cancer Patients. Eur. J. Immunol. 2012, 42, 1876–1885. [Google Scholar] [CrossRef] [Green Version]
- Gerner, M.C.; Ziegler, L.S.; Schmidt, R.L.J.; Krenn, M.; Zimprich, F.; Uyanik-Ünal, K.; Konstantopoulou, V.; Derdak, S.; Del Favero, G.; Schwarzinger, I.; et al. The TGF-b/SOX4 Axis and ROS-Driven Autophagy Co-Mediate CD39 Expression in Regulatory T-Cells. FASEB J. 2020, 34, 8367–8384. [Google Scholar] [CrossRef] [Green Version]
- Park, Y.-J.; Ryu, H.; Choi, G.; Kim, B.-S.; Hwang, E.S.; Kim, H.S.; Chung, Y. IL-27 Confers a Protumorigenic Activity of Regulatory T Cells via CD39. Proc. Natl. Acad. Sci. USA 2019, 116, 3106–3111. [Google Scholar] [CrossRef] [Green Version]
- Kochetkova, I.; Golden, S.; Holderness, K.; Callis, G.; Pascual, D.W. IL-35 Stimulation of CD39+ Regulatory T Cells Confers Protection against Collagen II-Induced Arthritis via the Production of IL-10. J. Immunol. 2010, 184, 7144–7153. [Google Scholar] [CrossRef] [Green Version]
- Raczkowski, F.; Rissiek, A.; Ricklefs, I.; Heiss, K.; Schumacher, V.; Wundenberg, K.; Haag, F.; Koch-Nolte, F.; Tolosa, E.; Mittrücker, H.-W. CD39 Is Upregulated during Activation of Mouse and Human T Cells and Attenuates the Immune Response to Listeria Monocytogenes. PLoS ONE 2018, 13, e0197151. [Google Scholar] [CrossRef]
- Shevchenko, I.; Mathes, A.; Groth, C.; Karakhanova, S.; Müller, V.; Utikal, J.; Werner, J.; Bazhin, A.V.; Umansky, V. Enhanced Expression of CD39 and CD73 on T Cells in the Regulation of Anti-Tumor Immune Responses. Oncoimmunology 2020, 9, 1744946. [Google Scholar] [CrossRef] [Green Version]
- Zheng, Y.; Li, Y.; Tang, B.; Zhao, Q.; Wang, D.; Liu, Y.; Guo, M.; Zhao, S.; Qi, Y.; Zhang, Y.; et al. IL-6-Induced CD39 Expression on Tumor-Infiltrating NK Cells Predicts Poor Prognosis in Esophageal Squamous Cell Carcinoma. Cancer Immunol. Immunother. 2020, 69, 2371–2380. [Google Scholar] [CrossRef]
- Amadori, A.; Zamarchi, R.; De Silvestro, G.; Forza, G.; Cavatton, G.; Danieli, G.A.; Clementi, M.; Chieco-Bianchi, L. Genetic Control of the CD4/CD8 T-Cell Ratio in Humans. Nat. Med. 1995, 1, 1279–1283. [Google Scholar] [CrossRef]
- Orrù, V.; Steri, M.; Sole, G.; Sidore, C.; Virdis, F.; Dei, M.; Lai, S.; Zoledziewska, M.; Busonero, F.; Mulas, A.; et al. Genetic Variants Regulating Immune Cell Levels in Health and Disease. Cell 2013, 155, 242–256. [Google Scholar] [CrossRef] [Green Version]
- Roederer, M.; Quaye, L.; Mangino, M.; Beddall, M.H.; Mahnke, Y.; Chattopadhyay, P.; Tosi, I.; Napolitano, L.; Terranova Barberio, M.; Menni, C.; et al. The Genetic Architecture of the Human Immune System: A Bioresource for Autoimmunity and Disease Pathogenesis. Cell 2015, 161, 387–403. [Google Scholar] [CrossRef] [Green Version]
- Rissiek, A.; Baumann, I.; Cuapio, A.; Mautner, A.; Kolster, M.; Arck, P.C.; Dodge-Khatami, A.; Mittrücker, H.-W.; Koch-Nolte, F.; Haag, F.; et al. The Expression of CD39 on Regulatory T Cells Is Genetically Driven and Further Upregulated at Sites of Inflammation. J. Autoimmun. 2015, 58, 12–20. [Google Scholar] [CrossRef]
- Friedman, D.J.; Künzli, B.M.; A-Rahim, Y.I.; Sevigny, J.; Berberat, P.O.; Enjyoji, K.; Csizmadia, E.; Friess, H.; Robson, S.C. From the Cover: CD39 Deletion Exacerbates Experimental Murine Colitis and Human Polymorphisms Increase Susceptibility to Inflammatory Bowel Disease. Proc. Natl. Acad. Sci. USA 2009, 106, 16788–16793. [Google Scholar] [CrossRef] [Green Version]
- Melchiotti, R.; Puan, K.J.; Andiappan, A.K.; Poh, T.Y.; Starke, M.; Zhuang, L.; Petsch, K.; Lai, T.S.; Chew, F.T.; Larbi, A.; et al. Genetic Analysis of an Allergic Rhinitis Cohort Reveals an Intercellular Epistasis between FAM134B and CD39. BMC Med. Genet. 2014, 15, 73. [Google Scholar] [CrossRef] [Green Version]
- Timperi, E.; Folgori, L.; Amodio, D.; De Luca, M.; Chiurchiù, S.; Piconese, S.; Di Cesare, S.; Pacella, I.; Martire, C.; Bonatti, G.; et al. Expansion of Activated Regulatory T Cells Inversely Correlates with Clinical Severity in Septic Neonates. J. Allergy Clin. Immunol. 2016, 137, 1617–1620.e6. [Google Scholar] [CrossRef] [Green Version]
- Adhikary, S.R.; Cuthbertson, P.; Turner, R.J.; Sluyter, R.; Watson, D. A Single-Nucleotide Polymorphism in the Human ENTPD1 Gene Encoding CD39 Is Associated with Worsened Graft-versus-Host Disease in a Humanized Mouse Model. Immunol. Cell Biol. 2020, 98, 397–410. [Google Scholar] [CrossRef]
- Nikolova, M.; Carriere, M.; Jenabian, M.-A.; Limou, S.; Younas, M.; Kök, A.; Huë, S.; Seddiki, N.; Hulin, A.; Delaneau, O.; et al. CD39/Adenosine Pathway Is Involved in AIDS Progression. PLoS Pathog. 2011, 7, e1002110. [Google Scholar] [CrossRef] [Green Version]
- Fang, F.; Yu, M.; Cavanagh, M.M.; Hutter Saunders, J.; Qi, Q.; Ye, Z.; Le Saux, S.; Sultan, W.; Turgano, E.; Dekker, C.L.; et al. Expression of CD39 on Activated T Cells Impairs Their Survival in Older Individuals. Cell Rep. 2016, 14, 1218–1231. [Google Scholar] [CrossRef] [Green Version]
- Cao, W.; Fang, F.; Gould, T.; Li, X.; Kim, C.; Gustafson, C.; Lambert, S.; Weyand, C.M.; Goronzy, J.J. Ecto-NTPDase CD39 Is a Negative Checkpoint That Inhibits Follicular Helper Cell Generation. J. Clin. Investig. 2020, 130, 3422–3436. [Google Scholar] [CrossRef]
- Perrot, I.; Michaud, H.-A.; Giraudon-Paoli, M.; Augier, S.; Docquier, A.; Gros, L.; Courtois, R.; Déjou, C.; Jecko, D.; Becquart, O.; et al. Blocking Antibodies Targeting the CD39/CD73 Immunosuppressive Pathway Unleash Immune Responses in Combination Cancer Therapies. Cell Rep. 2019, 27, 2411–2425.e9. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jin, D.; Fan, J.; Wang, L.; Thompson, L.F.; Liu, A.; Daniel, B.J.; Shin, T.; Curiel, T.J.; Zhang, B. CD73 on Tumor Cells Impairs Antitumor T-Cell Responses: A Novel Mechanism of Tumor-Induced Immune Suppression. Cancer Res. 2010, 70, 2245–2255. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Stagg, J.; Beavis, P.A.; Divisekera, U.; Liu, M.C.P.; Möller, A.; Darcy, P.K.; Smyth, M.J. CD73-Deficient Mice Are Resistant to Carcinogenesis. Cancer Res. 2012, 72, 2190–2196. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yegutkin, G.G.; Marttila-Ichihara, F.; Karikoski, M.; Niemelä, J.; Laurila, J.P.; Elima, K.; Jalkanen, S.; Salmi, M. Altered Purinergic Signaling in CD73-Deficient Mice Inhibits Tumor Progression. Eur. J. Immunol. 2011, 41, 1231–1241. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Fan, J.; Thompson, L.F.; Zhang, Y.; Shin, T.; Curiel, T.J.; Zhang, B. CD73 Has Distinct Roles in Nonhematopoietic and Hematopoietic Cells to Promote Tumor Growth in Mice. J. Clin. Investig. 2011, 121, 2371–2382. [Google Scholar] [CrossRef] [PubMed]
- Sun, X.; Han, L.; Seth, P.; Bian, S.; Li, L.; Csizmadia, E.; Junger, W.G.; Schmelzle, M.; Usheva, A.; Tapper, E.B.; et al. Disordered Purinergic Signaling and Abnormal Cellular Metabolism Are Associated with Development of Liver Cancer in Cd39/ENTPD1 Null Mice. Hepatology 2013, 57, 205–216. [Google Scholar] [CrossRef] [Green Version]
- Sattler, C.; Steinsdoerfer, M.; Offers, M.; Fischer, E.; Schierl, R.; Heseler, K.; Däubener, W.; Seissler, J. Inhibition of T-Cell Proliferation by Murine Multipotent Mesenchymal Stromal Cells Is Mediated by CD39 Expression and Adenosine Generation. Cell Transplant. 2011, 20, 1221–1230. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yang, R.; Elsaadi, S.; Misund, K.; Abdollahi, P.; Vandsemb, E.N.; Moen, S.H.; Kusnierczyk, A.; Slupphaug, G.; Standal, T.; Waage, A.; et al. Conversion of ATP to Adenosine by CD39 and CD73 in Multiple Myeloma Can Be Successfully Targeted Together with Adenosine Receptor A2A Blockade. J. Immunother. Cancer 2020, 8, e000610. [Google Scholar] [CrossRef]
- Deaglio, S.; Dwyer, K.M.; Gao, W.; Friedman, D.; Usheva, A.; Erat, A.; Chen, J.-F.; Enjyoji, K.; Linden, J.; Oukka, M.; et al. Adenosine Generation Catalyzed by CD39 and CD73 Expressed on Regulatory T Cells Mediates Immune Suppression. J. Exp. Med. 2007, 204, 1257–1265. [Google Scholar] [CrossRef] [Green Version]
- Sun, X.; Wu, Y.; Gao, W.; Enjyoji, K.; Csizmadia, E.; Müller, C.E.; Murakami, T.; Robson, S.C. CD39/ENTPD1 Expression by CD4+Foxp3+ Regulatory T Cells Promotes Hepatic Metastatic Tumor Growth in Mice. Gastroenterology 2010, 139, 1030–1040. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Enjyoji, K.; Kotani, K.; Thukral, C.; Blumel, B.; Sun, X.; Wu, Y.; Imai, M.; Friedman, D.; Csizmadia, E.; Bleibel, W.; et al. Deletion of Cd39/Entpd1 Results in Hepatic Insulin Resistance. Diabetes 2008, 57, 2311–2320. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lanser, A.J.; Rezende, R.M.; Rubino, S.; Lorello, P.J.; Donnelly, D.J.; Xu, H.; Lau, L.A.; Dulla, C.G.; Caldarone, B.J.; Robson, S.C.; et al. Disruption of the ATP/Adenosine Balance in CD39-/- Mice Is Associated with Handling-Induced Seizures. Immunology 2017, 152, 589–601. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Nardi-Schreiber, A.; Sapir, G.; Gamliel, A.; Kakhlon, O.; Sosna, J.; Gomori, J.M.; Meiner, V.; Lossos, A.; Katz-Brull, R. Defective ATP Breakdown Activity Related to an ENTPD1 Gene Mutation Demonstrated Using 31P NMR Spectroscopy. Chem. Commun. 2017, 53, 9121–9124. [Google Scholar] [CrossRef] [PubMed]
- Thompson, E.A.; Powell, J.D. Inhibition of the Adenosine Pathway to Potentiate Cancer Immunotherapy: Potential for Combinatorial Approaches. Annu. Rev. Med. 2021, 72, 331–348. [Google Scholar] [CrossRef]
- Borghaei, H.; Paz-Ares, L.; Horn, L.; Spigel, D.R.; Steins, M.; Ready, N.E.; Chow, L.Q.; Vokes, E.E.; Felip, E.; Holgado, E.; et al. Nivolumab versus Docetaxel in Advanced Nonsquamous Non-Small-Cell Lung Cancer. N. Engl. J. Med. 2015, 373, 1627–1639. [Google Scholar] [CrossRef]
- Sharma, P.; Allison, J.P. The Future of Immune Checkpoint Therapy. Science 2015, 348, 56–61. [Google Scholar] [CrossRef]
- Topalian, S.L.; Hodi, F.S.; Brahmer, J.R.; Gettinger, S.N.; Smith, D.C.; McDermott, D.F.; Powderly, J.D.; Carvajal, R.D.; Sosman, J.A.; Atkins, M.B.; et al. Safety, Activity, and Immune Correlates of Anti-PD-1 Antibody in Cancer. N. Engl. J. Med. 2012, 366, 2443–2454. [Google Scholar] [CrossRef] [PubMed]
- Topalian, S.L.; Drake, C.G.; Pardoll, D.M. Immune Checkpoint Blockade: A Common Denominator Approach to Cancer Therapy. Cancer Cell 2015, 27, 450–461. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hay, C.M.; Sult, E.; Huang, Q.; Mulgrew, K.; Fuhrmann, S.R.; McGlinchey, K.A.; Hammond, S.A.; Rothstein, R.; Rios-Doria, J.; Poon, E.; et al. Targeting CD73 in the Tumor Microenvironment with MEDI9447. Oncoimmunology 2016, 5, e1208875. [Google Scholar] [CrossRef]
- Allard, B.; Pommey, S.; Smyth, M.J.; Stagg, J. Targeting CD73 Enhances the Antitumor Activity of Anti-PD-1 and Anti-CTLA-4 MAbs. Clin. Cancer Res. 2013, 19, 5626–5635. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cekic, C.; Linden, J. Adenosine A2A Receptors Intrinsically Regulate CD8+ T Cells in the Tumor Microenvironment. Cancer Res. 2014, 74, 7239–7249. [Google Scholar] [CrossRef] [Green Version]
- Kjaergaard, J.; Hatfield, S.; Jones, G.; Ohta, A.; Sitkovsky, M. A2A Adenosine Receptor Gene Deletion or Synthetic A2A Antagonist Liberate Tumor-Reactive CD8+ T Cells from Tumor-Induced Immunosuppression. J. Immunol. 2018, 201, 782–791. [Google Scholar] [CrossRef] [PubMed] [Green Version]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Timperi, E.; Barnaba, V. CD39 Regulation and Functions in T Cells. Int. J. Mol. Sci. 2021, 22, 8068. https://doi.org/10.3390/ijms22158068
Timperi E, Barnaba V. CD39 Regulation and Functions in T Cells. International Journal of Molecular Sciences. 2021; 22(15):8068. https://doi.org/10.3390/ijms22158068
Chicago/Turabian StyleTimperi, Eleonora, and Vincenzo Barnaba. 2021. "CD39 Regulation and Functions in T Cells" International Journal of Molecular Sciences 22, no. 15: 8068. https://doi.org/10.3390/ijms22158068
APA StyleTimperi, E., & Barnaba, V. (2021). CD39 Regulation and Functions in T Cells. International Journal of Molecular Sciences, 22(15), 8068. https://doi.org/10.3390/ijms22158068






