Prenylated Flavonoids with Potential Antimicrobial Activity: Synthesis, Biological Activity, and In Silico Study
Abstract
1. Introduction
2. Results and Discussion
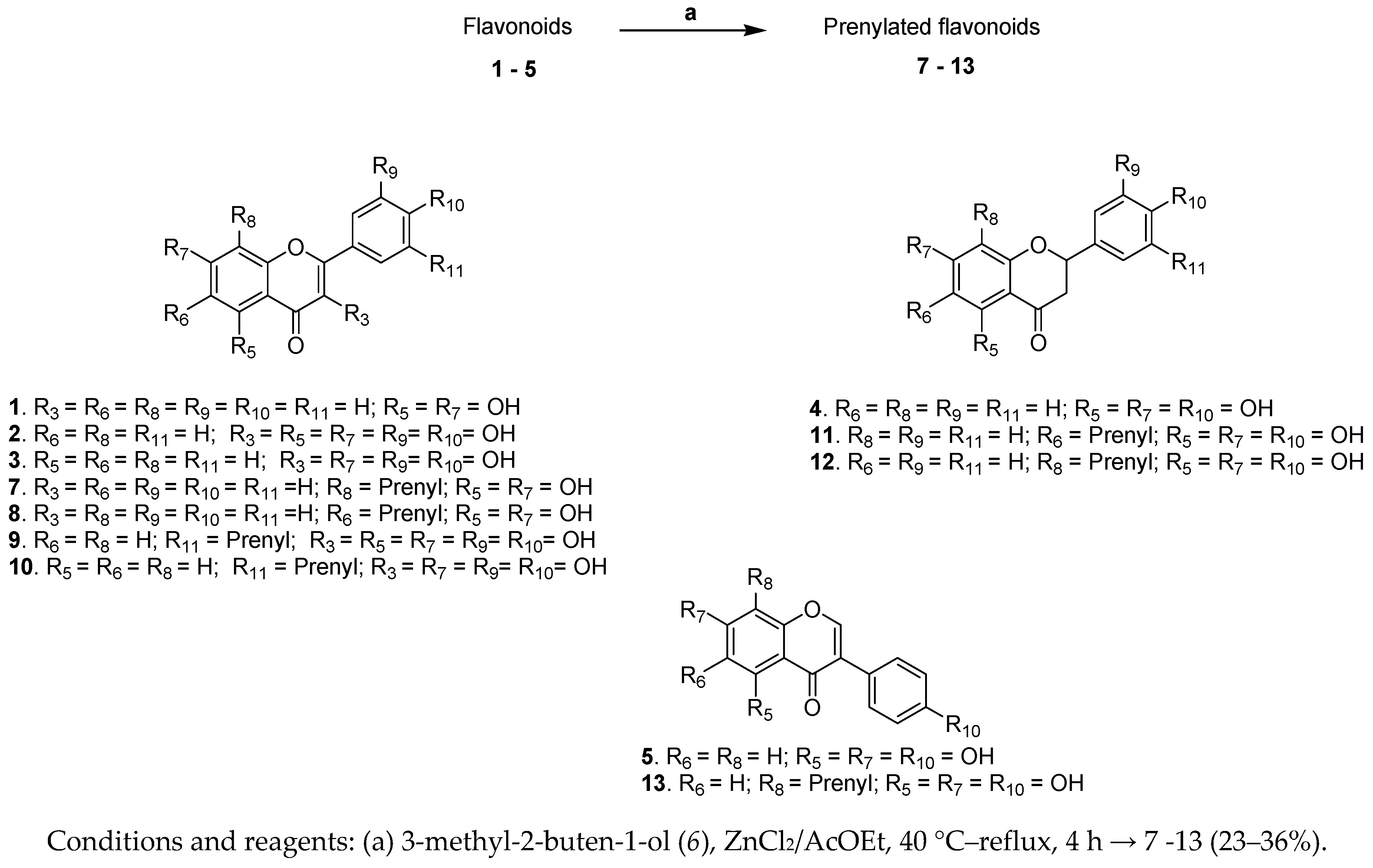
2.1. Synthesis
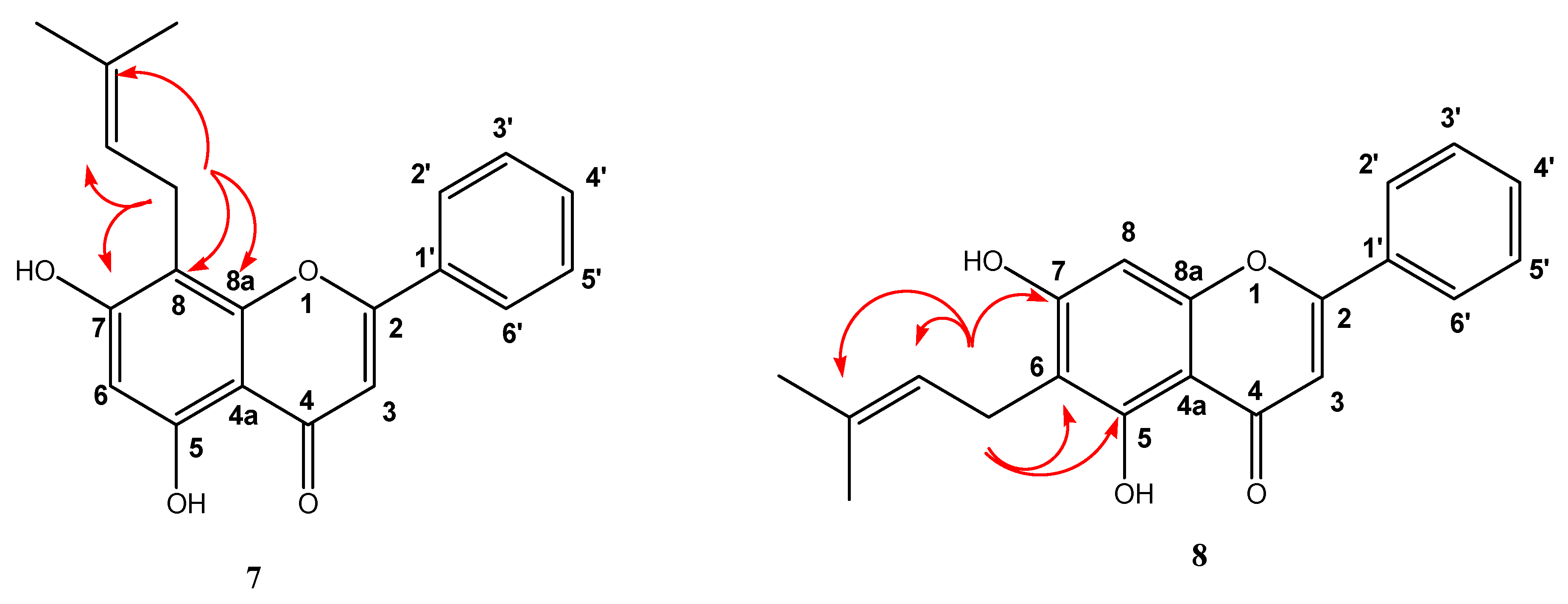
Structure Determination
2.2. In Vitro Antibacterial Effects on Human Pathogen Bacteria
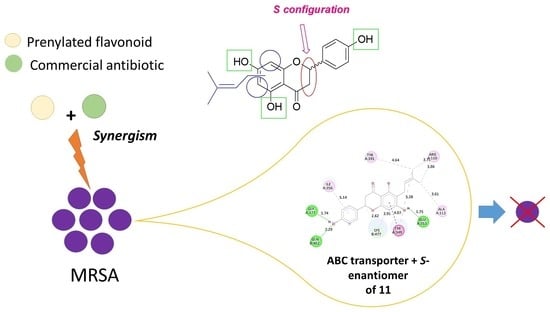
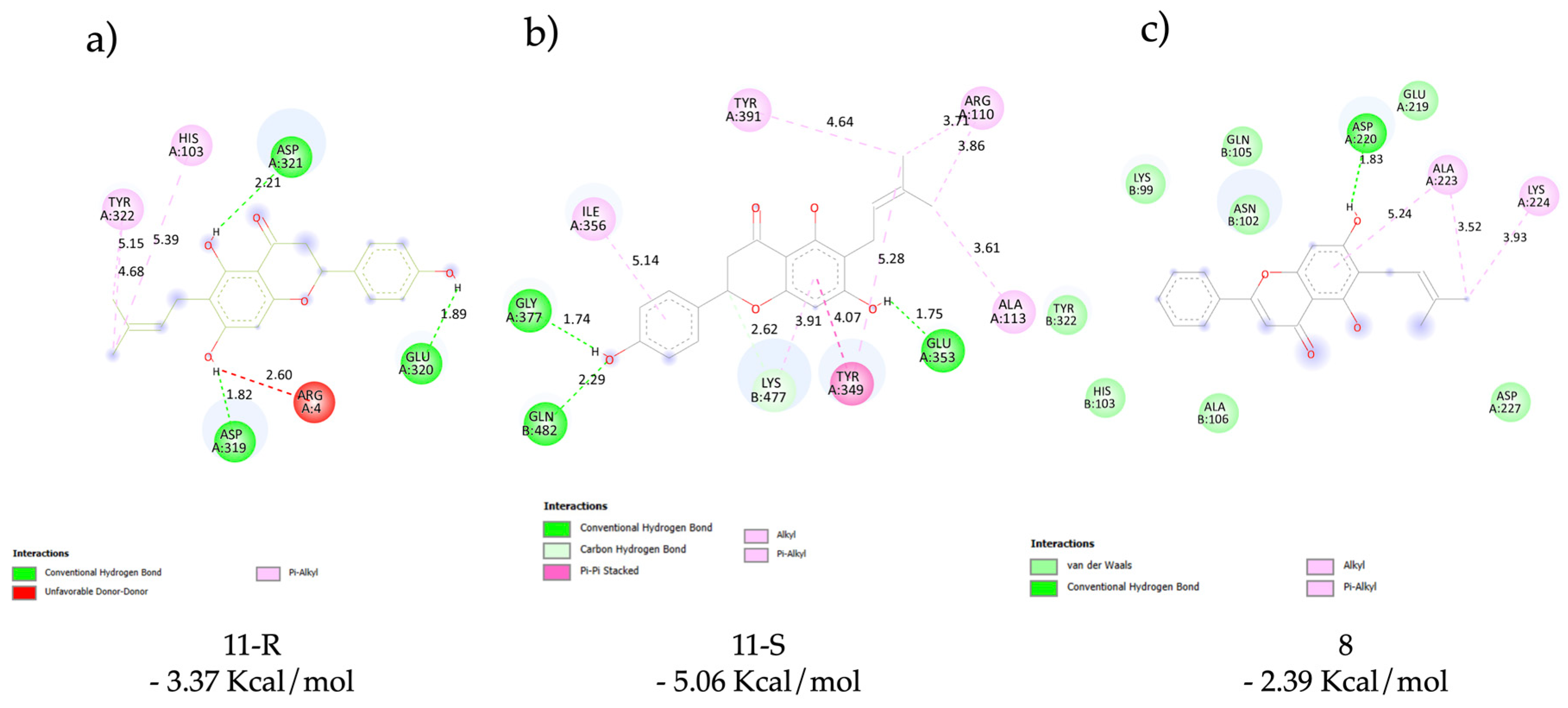
Docking of 11 (S and R Enantiomers) and 8 with ATP-Binding Cassette (Multi-Drug ABC Transporter Sav1866 from S. aureus in Complex with AMP-PNP)
2.3. In Vitro Antifungal Effects on Human Pathogenic fungi
2.4. In Vitro Antibacterial Effects on Plant Pathogen bacteria
2.5. In Vitro 5-hLOX Enzyme Inhibition Assay
2.6. DPPH Radical Scavenging Activity
2.7. Cytotoxic Activity of 11 and 12
3. Materials and Methods
3.1. Chemistry
3.1.1. General Data
3.1.2. General Experimental Procedure for the Prenylation of Flavonoids
3.1.3. Physical Data of Prenylated Flavonoids
3.2. Biological Assays
3.2.1. In Vitro Antibacterial Activity Assays: Human Pathogens
Minimum Inhibitory Concentration Assay
Checkerboard Dilution Test
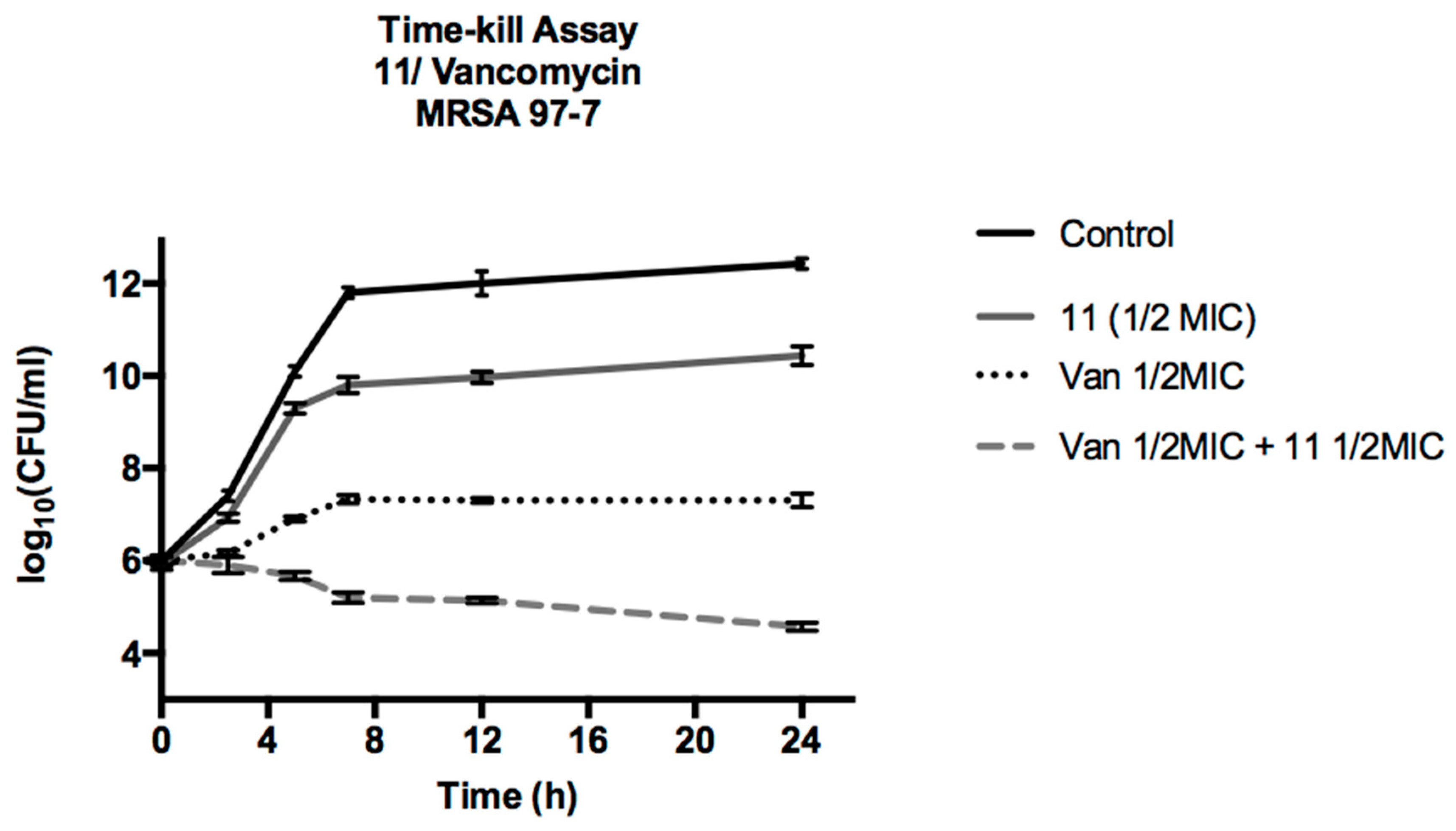
Time Kill Assay
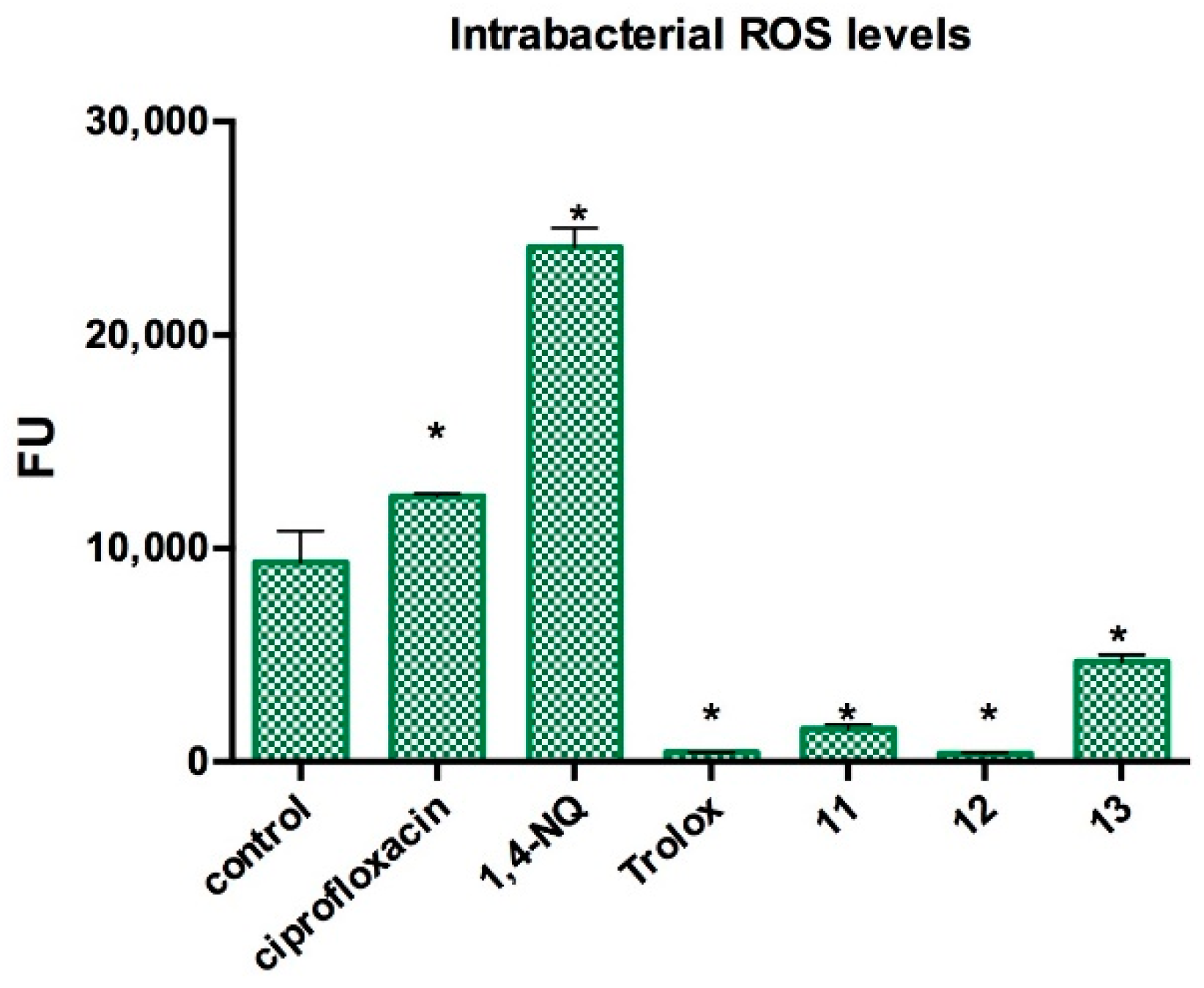
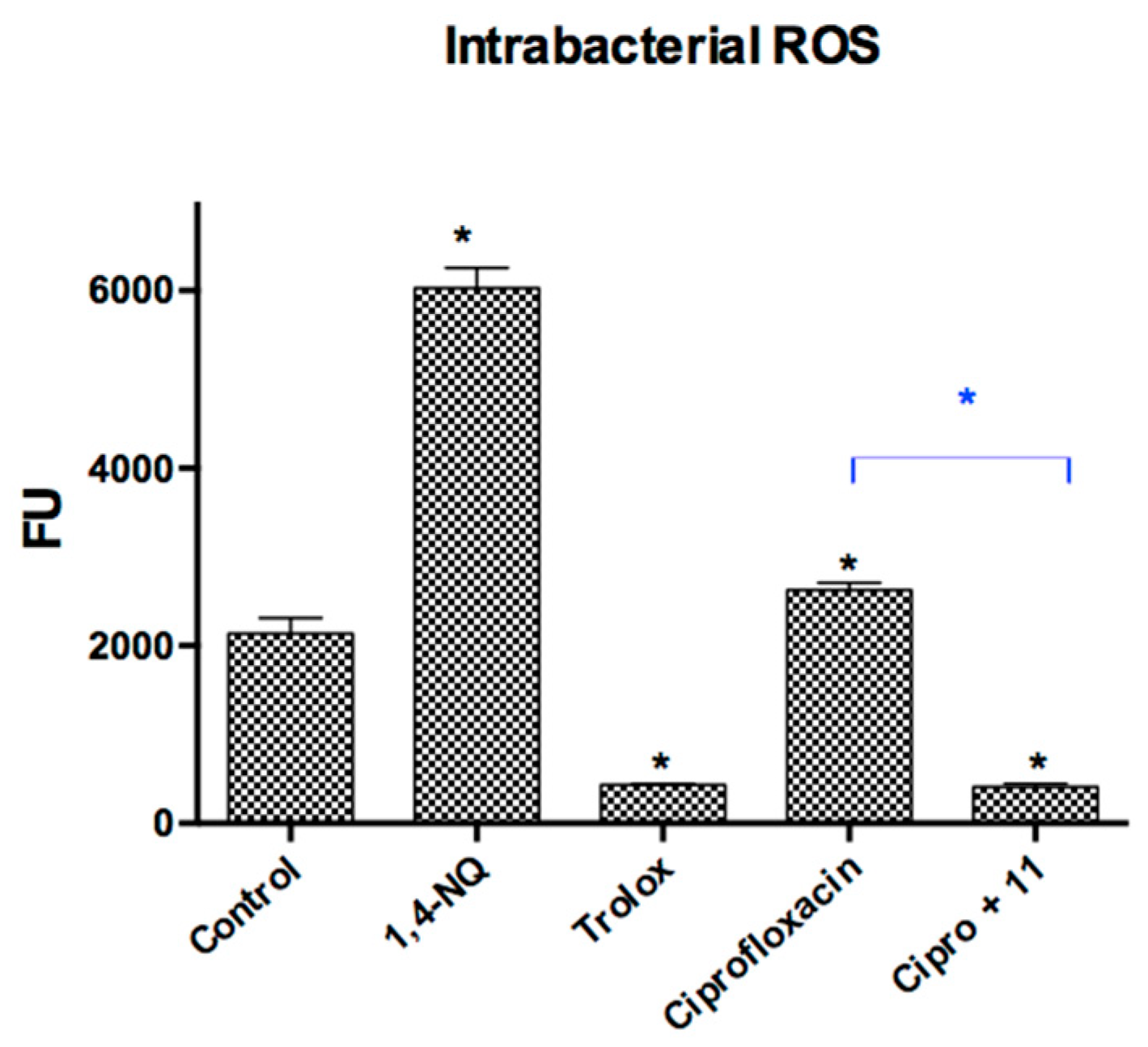
Determination of Reactive Oxygen Species (ROS) Intrabacterial Levels
3.2.2. In Vitro Antifungal Activity Assays against Human Pathogens
Microorganisms and Media
Antifungal Susceptibility Testing
3.2.3. In Vitro Antibacterial Activity against Plant Pathogens
3.2.4. Statistics
3.3. In Vitro 5-LOX Enzyme Inhibition Assay
3.4. Docking of 11 (S and R Enantiomers) and with ABC Transporter
3.5. General Procedure to Determine the DPPH Radical Scavenging Activity
3.6. In Vitro Anticancer Activity of 11 and 12
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Acknowledgments
Conflicts of Interest
References
- Oh, I.; Yang, W.-Y.; Chung, S.-C.; Kim, T.-Y.; Oh, K.-B.; Shin, J. In vitro sortase A inhibitory and antimicrobial activity of flavonoids isolated from the roots of Sophora flavescens. Arch. Pharm. Res. 2011, 34, 217–222. [Google Scholar] [CrossRef] [PubMed]
- Yazaki, K.; Sasaki, K.; Tsurumaru, Y. Prenylation of aromatic compounds, a key diversification of plant secondary metabolites. Phytochemistry 2009, 70, 1739–1745. [Google Scholar] [CrossRef]
- Kushwaha, P.P.; Prajapati, S.K.; Pothabathula, S.V.; Singh, A.K.; Shuaib, M.; Joshi, K.; Kumar, S. Prenylated flavonoids as a promising drug discovery candidate. In Phytochemicals as Lead Compounds for New Drug Discovery; Elsevier: Amsterdam, The Netherlands, 2020; pp. 347–355. ISBN 9780128178911. [Google Scholar]
- Kumano, T.; Richard, S.B.; Noel, J.P.; Nishiyama, M.; Kuzuyama, T. Chemoenzymatic syntheses of prenylated aromatic small molecules using Streptomyces prenyltransferases with relaxed substrate specificities. Bioorg. Med. Chem. 2008, 16, 8117–8126. [Google Scholar] [CrossRef] [PubMed]
- Simmonds, M.S.J. Importance of flavonoids in insect–plant interactions: Feeding and oviposition. Phytochemistry 2001, 56, 245–252. [Google Scholar] [CrossRef]
- Quiroga, E.N.; Sampietro, D.A.; Sgariglia, M.A.; Soberón, J.R.; Vattuone, M.A. Antimycotic activity of 5′-prenylisoflavanones of the plant Geoffroea decorticans, against Aspergillus species. Int. J. Food Microbiol. 2009, 132, 42–46. [Google Scholar] [CrossRef]
- Edziri, H.; Mastouri, M.; Mahjoub, M.A.; Mighri, Z.; Mahjoub, A.; Verschaeve, L. Antibacterial, Antifungal and Cytotoxic Activities of Two Flavonoids from Retama raetam Flowers. Molecules 2012, 17, 7284–7293. [Google Scholar] [CrossRef]
- Du, J.; He, Z.-D.; Jiang, R.-W.; Ye, W.-C.; Xu, H.-X.; But, P.P.-H. Antiviral flavonoids from the root bark of Morus alba L. Phytochemistry 2003, 62, 1235–1238. [Google Scholar] [CrossRef]
- Liu, M.; Hansen, P.; Wang, G.; Qiu, L.; Dong, J.; Yin, H.; Qian, Z.; Yang, M.; Miao, J. Pharmacological Profile of Xanthohumol, a Prenylated Flavonoid from Hops (Humulus lupulus). Molecules 2015, 20, 754–779. [Google Scholar] [CrossRef]
- Jin, J.H.; Kim, J.S.; Kang, S.S.; Son, K.H.; Chang, H.W.; Kim, H.P. Anti-inflammatory and anti-arthritic activity of total flavonoids of the roots of Sophora flavescens. J. Ethnopharmacol. 2010, 127, 589–595. [Google Scholar] [CrossRef]
- Molčanová, L.; Janošíková, D.; Dall’Acqua, S.; Šmejkal, K. C-prenylated flavonoids with potential cytotoxic activity against solid tumor cell lines. Phytochem. Rev. 2019, 18, 1051–1100. [Google Scholar] [CrossRef]
- Yang, Z.-G.; Matsuzaki, K.; Takamatsu, S.; Kitanaka, S. Inhibitory Effects of Constituents from Morus alba var. multicaulis on Differentiation of 3T3-L1 Cells and Nitric Oxide Production in RAW264.7 Cells. Molecules 2011, 16, 6010–6022. [Google Scholar] [CrossRef]
- Venturelli, S.; Burkard, M.; Biendl, M.; Lauer, U.M.; Frank, J.; Busch, C. Prenylated chalcones and flavonoids for the prevention and treatment of cancer. Nutrition 2016, 32, 1171–1178. [Google Scholar] [CrossRef] [PubMed]
- Yang, X.; Yang, J.; Jiang, Y.; Yang, H.; Yun, Z.; Rong, W.; Yang, B. Regiospecific synthesis of prenylated flavonoids by a prenyltransferase cloned from Fusarium oxysporum. Sci. Rep. 2016, 6, 24819. [Google Scholar] [CrossRef] [PubMed]
- Levisson, M.; Araya-Cloutier, C.; de Bruijn, W.J.C.; van der Heide, M.; Salvador López, J.M.; Daran, J.-M.; Vincken, J.-P.; Beekwilder, J. Toward Developing a Yeast Cell Factory for the Production of Prenylated Flavonoids. J. Agric. Food Chem. 2019, 67, 13478–13486. [Google Scholar] [CrossRef] [PubMed]
- Sasaki, K.; Tsurumaru, Y.; Yamamoto, H.; Yazaki, K. Molecular Characterization of a Membrane-bound Prenyltransferase Specific for Isoflavone from Sophora flavescens. J. Biol. Chem. 2011, 286, 24125–24134. [Google Scholar] [CrossRef]
- Yang, X.; Jiang, Y.; Yang, J.; He, J.; Sun, J.; Chen, F.; Zhang, M.; Yang, B. Prenylated flavonoids, promising nutraceuticals with impressive biological activities. Trends Food Sci. Technol. 2015, 44, 93–104. [Google Scholar] [CrossRef]
- Osorio, M.E.; Qiuroz, K.A.; Carvajal, M.A.; Vergara, A.P.; Sánchez, E.Y.; González, C.E.; Catalán, K.S. Synthesis, anti-phytopathogenic and DPPH radical Scavenging activities of C-prenylated ace tophenones and benzaldehydes. J. Chil. Chem. Soc. 2016, 61, 3095–3101. [Google Scholar] [CrossRef]
- Global Antimicrobial Resistance Surveillance System (GLASS); Report; WHO: Geneva, Switzerland, 2017; ISBN 9789241513449.
- Vandeputte, P.; Ferrari, S.; Coste, A.T. Antifungal Resistance and New Strategies to Control Fungal Infections. Int. J. Microbiol. 2012, 2012, 1–26. [Google Scholar] [CrossRef]
- Araya-Cloutier, C.; den Besten, H.M.W.; Aisyah, S.; Gruppen, H.; Vincken, J.-P. The position of prenylation of isoflavonoids and stilbenoids from legumes (Fabaceae) modulates the antimicrobial activity against Gram positive pathogens. Food Chem. 2017, 226, 193–201. [Google Scholar] [CrossRef] [PubMed]
- State of the World’s Antibiotics, 2015; Center for Disease Dynamics, Economics & Policy: Washington, DC, USA, 2015.
- Cheesman, M.; Ilanko, A.; Blonk, B.; Cock, I. Developing new antimicrobial therapies: Are synergistic combinations of plant extracts/compounds with conventional antibiotics the solution? Pharm. Rev. 2017, 11, 57. [Google Scholar] [CrossRef]
- Sousa Carvalho, G.F.; Marques, L.K.; Sousa, H.G.; Silva, L.R.; Leão Ferreira, D.C.; Pires de Moura do Amaral, F.; Martins Maia Filho, A.L.; Figueredo-Silva, J.; Alves, W.d.S.; Oliveira, M.d.D.A.d.; et al. Phytochemical study, molecular docking, genotoxicity and therapeutic efficacy of the aqueous extract of the stem bark of Ximenia americana L. in the treatment of experimental COPD in rats. J. Ethnopharmacol. 2020, 247, 112259. [Google Scholar] [CrossRef] [PubMed]
- Zhao, X.; Chen, R.; Shi, Y.; Zhang, X.; Tian, C.; Xia, D. Antioxidant and Anti-Inflammatory Activities of Six Flavonoids from Smilax glabra Roxb. Molecules 2020, 25, 5295. [Google Scholar] [CrossRef] [PubMed]
- Tonk, R.K.; Bawa, S.; Chawla, G.; Deora, G.S.; Kumar, S.; Rathore, V.; Mulakayala, N.; Rajaram, A.; Kalle, A.M.; Afzal, O. Synthesis and pharmacological evaluation of pyrazolo [4, 3-c] cinnoline derivatives as potential anti-inflammatory and antibacterial agents. Eur. J. Med. Chem. 2012, 57, 176–184. [Google Scholar] [CrossRef]
- Atala, E.; Fuentes, J.; Wehrhahn, M.J.; Speisky, H. Quercetin and related flavonoids conserve their antioxidant properties despite undergoing chemical or enzymatic oxidation. Food Chem. 2017, 234, 479–485. [Google Scholar] [CrossRef]
- Zuo, G.-Y.; Yang, C.-X.; Han, J.; Li, Y.-Q.; Wang, G.-C. Synergism of prenylflavonoids from Morus alba root bark against clinical MRSA isolates. Phytomedicine 2018, 39, 93–99. [Google Scholar] [CrossRef] [PubMed]
- Sohn, H.-Y.; Son, K.H.; Kwon, C.-S.; Kwon, G.-S.; Kang, S.S. Antimicrobial and cytotoxic activity of 18 prenylated flavonoids isolated from medicinal plants: Morus alba L., Morus mongolica Schneider, Broussnetia papyrifera (L.) Vent, Sophora flavescens Ait and Echinosophora koreensis Nakai. Phytomedicine 2004, 11, 666–672. [Google Scholar] [CrossRef] [PubMed]
- Tsuchiya, H.; Sato, M.; Miyazaki, T.; Fujiwara, S.; Tanigaki, S.; Ohyama, M.; Tanaka, T.; Iinuma, M. Comparative study on the antibacterial activity of phytochemical flavanones against methicillin-resistant Staphylococcus aureus. J. Ethnopharmacol. 1996, 50, 27–34. [Google Scholar] [CrossRef]
- Sato, M.; Tanaka, H.; Tani, N.; Nagayama, M.; Yamaguchi, R. Different antibacterial actions of isoflavones isolated from Erythrina poeppigiana against methicillin-resistant Staphylococcus aureus. Lett. Appl. Microbiol. 2006, 43, 243–248. [Google Scholar] [CrossRef]
- das Chagas Almeida, A.; Azevedo Rodrigues, L.; dos Santos Paulino, G.; Pereira Aguilar, A.; Andrade Almeida, A.; Olavo Ferreira, S.; Brandão, G.C.; Viana Leite, J.P.; de Oliveira Barros Ribon, A. Prenylated flavonoid-enriched fraction from Maclura tinctoria shows biological activity against Staphylococcus aureus and protects Galleria mellonella larvae from bacterial infection. BMC Complement. Altern. Med. 2019, 19, 189. [Google Scholar] [CrossRef]
- Araya-Cloutier, C.; Vincken, J.-P.; van de Schans, M.G.M.; Hageman, J.; Schaftenaar, G.; den Besten, H.M.W.; Gruppen, H. QSAR-based molecular signatures of prenylated (iso)flavonoids underlying antimicrobial potency against and membrane-disruption in Gram positive and Gram negative bacteria. Sci. Rep. 2018, 8, 1–14. [Google Scholar] [CrossRef]
- Mun, S.-H.; Joung, D.-K.; Kim, S.-B.; Park, S.-J.; Seo, Y.-S.; Gong, R.; Choi, J.-G.; Shin, D.-W.; Rho, J.-R.; Kang, O.-H.; et al. The Mechanism of Antimicrobial Activity of Sophoraflavanone B Against Methicillin-Resistant Staphylococcus aureus. Foodborne Pathog. Dis. 2014, 11, 234–239. [Google Scholar] [CrossRef] [PubMed]
- Wagner, H.; Ulrich-Merzenich, G. Synergy research: Approaching a new generation of phytopharmaceuticals. Phytomedicine 2009, 16, 97–110. [Google Scholar] [CrossRef]
- Jung, H.A.; Jung, M.J.; Kim, J.Y.; Chung, H.Y.; Choi, J.S. Inhibitory activity of flavonoids from Prunus davidiana and other flavonoids on total ROS and hydroxyl radical generation. Arch. Pharm. Res. 2003, 26, 809–815. [Google Scholar] [CrossRef] [PubMed]
- Becerra, M.; Albesa, I. Oxidative stress induced by ciprofloxacin in Staphylococcus aureus. Biochem. Biophys. Res. Commun. 2002, 297, 1003–1007. [Google Scholar] [CrossRef]
- Lage, H. ABC-transporters: Implications on drug resistance from microorganisms to human cancers. Int. J. Antimicrob. Agents 2003, 22, 188–199. [Google Scholar] [CrossRef]
- CLSI. Reference Method for Broth Dilution Antifungal Susceptibility Testing of Yeast, Approved Standard-Third Edition; CLSI document M27-A3; Clinical and Laboratory Standards Institute: Wayne, PA, USA, 2008. [Google Scholar]
- CLSI. Reference Method for Broth Dilution Antifungal Susceptibility Testing of Filamentous Fungi, Approved Standard-Second Edition; CLSI document M38-A2; Clinical and Laboratory Standards Institute: Wayne, PA, USA, 2008. [Google Scholar]
- Pfaller, M.A.; Messer, S.A.; Boyken, L.; Rice, C.; Tendolkar, S.; Hollis, R.J.; Doern, G.V.; Diekema, D.J. Global Trends in the Antifungal Susceptibility of Cryptococcus neoformans (1990 to 2004). J. Clin. Microbiol. 2005, 43, 2163–2167. [Google Scholar] [CrossRef] [PubMed]
- Pfaller, M.A.; Diekema, D.J. Epidemiology of Invasive Candidiasis: A Persistent Public Health Problem. Clin. Microbiol. Rev. 2007, 20, 133–163. [Google Scholar] [CrossRef] [PubMed]
- Pankey, G.A.; Sabath, L.D. Clinical Relevance of Bacteriostatic versus Bactericidal Mechanisms of Action in the Treatment of Gram-Positive Bacterial Infections. Clin. Infect. Dis. 2004, 38, 864–870. [Google Scholar] [CrossRef]
- Valdés, E.; González, C.; Díaz, K.; Vásquez-Martínez, Y.; Mascayano, C.; Torrent, C.; Cabezas, F.; Mejias, S.; Montoya, M.; Cortez-San Martín, M.; et al. Biological Properties and Absolute Configuration of Flavanones from Calceolariathyrsiflora Graham. Front. Pharm. 2020, 11, 1125. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Q.; Yang, W.; Liu, J.; Liu, H.; Lv, Z.; Zhang, C.; Chen, D.; Jiao, Z. Identification of Six Flavonoids as Novel Cellular Antioxidants and Their Structure-Activity Relationship. Oxid. Med. Cell. Longev. 2020, 2020, 1–12. [Google Scholar] [CrossRef]
- Choi, J.; Lee, D.-H.; Jang, H.; Park, S.-Y.; Seol, J.-W. Naringenin exerts anticancer effects by inducing tumor cell death and inhibiting angiogenesis in malignant melanoma. Int. J. Med. Sci. 2020, 17, 3049–3057. [Google Scholar] [CrossRef] [PubMed]
- Stompor, M.; Uram, Ł.; Podgórski, R. In Vitro Effect of 8-Prenylnaringenin and Naringenin on Fibroblasts and Glioblastoma Cells-Cellular Accumulation and Cytotoxicity. Molecules 2017, 22, 1092. [Google Scholar] [CrossRef] [PubMed]
- Wang, R.; Wang, J.; Dong, T.; Shen, J.; Gao, X.; Zhou, J. Naringenin has a chemoprotective effect in MDA-MB-231 breast cancer cells via inhibition of caspase-3 and -9 activities. Oncol. Lett. 2018, 17, 1217–1222. [Google Scholar] [CrossRef] [PubMed]
- Li, Q.; Wang, Y.; Zhang, L.; Chen, L.; Du, Y.; Ye, T.; Shi, X. Naringenin exerts anti-angiogenic effects in human endothelial cells: Involvement of ERRα/VEGF/KDR signaling pathway. Fitoterapia 2016, 111, 78–86. [Google Scholar] [CrossRef]
- Maatouk, M.; Elgueder, D.; Mustapha, N.; Chaaban, H.; Bzéouich, I.M.; Loannou, I.; Kilani, S.; Ghoul, M.; Ghedira, K.; Chekir-Ghedira, L. Effect of heated naringenin on immunomodulatory properties and cellular antioxidant activity. Cell Stress Chaperones 2016, 21, 1101–1109. [Google Scholar] [CrossRef]
- Beirne, J.J.; Carroll, N.M.; O’Sullivan, W.I.; Woods, J. Alkylation of the chrysin dianion. Tetrahedron 1975, 31, 265–267. [Google Scholar] [CrossRef]
- Hänsel, R.; Schulz, J. Desmethylxanthohumol: Isolierung aus Hopfen und Cyclisierung zu Flavanonen. Arch. Pharm. 1988, 321, 37–40. [Google Scholar] [CrossRef]
- Dong, X.; Fan, Y.; Yu, L.; Hu, Y. Synthesis of Four Natural Prenylflavonoids and Their Estrogen-like Activities. Arch. Pharm. 2007, 340, 372–376. [Google Scholar] [CrossRef]
- Kawamura, T.; Hayashi, M.; Mukai, R.; Terao, J.; Nemoto, H. The First Synthesis of Uralenol, 5′-Prenylated Quercetin, via Palladium-Catalyzed O-Dimethylallylation Reaction with Concurrent Acetyl Migration. Synthesis 2013, 46, 170–174. [Google Scholar] [CrossRef]
- Al-Maharik, N.; Botting, N.P. Synthesis of lupiwighteone via a para-Claisen–Cope rearrangement. Tetrahedron 2003, 59, 4177–4181. [Google Scholar] [CrossRef]
- CLSI. Reference Method for Determining Bactericidal Activity of Antibacterial Agents; Approved Guideline. CLSI document M26-A; Clinical and Laboratory Standards Institute: Wayne, PA, USA, 1999. [Google Scholar]
- Iten, F.; Saller, R.; Abel, G.; Reichling, J. Additive Antmicrobial Effects of the Active Components of the Essential Oil of Thymus vulgaris—Chemotype Carvacrol. Planta Med. 2009, 75, 1231–1236. [Google Scholar] [CrossRef]
- Doern, C.D. When Does 2 Plus 2 Equal 5? A Review of Antimicrobial Synergy Testing. J. Clin. Microbiol. 2014, 52, 4124–4128. [Google Scholar] [CrossRef] [PubMed]
- Petersen, P.J.; Jones, C.H.; Bradford, P.A. In vitro antibacterial activities of tigecycline and comparative agents by time-kill kinetic studies in fresh Mueller-Hinton broth. Diagn. Microbiol. Infect. Dis. 2007, 59, 347–349. [Google Scholar] [CrossRef]
- Ong, K.S.; Cheow, Y.L.; Lee, S.M. The role of reactive oxygen species in the antimicrobial activity of pyochelin. J. Adv. Res. 2017, 8, 393–398. [Google Scholar] [CrossRef]
- Cole, M.D. Key antifungal, antibacterial and anti-insect assays-a critical review. Biochem. Syst. Ecol. 1994, 22, 837–856. [Google Scholar] [CrossRef]
- Zampini, I.C.; Vattuone, M.A.; Isla, M.I. Antibacterial activity of Zuccagnia punctata Cav. ethanolic extracts. J. Ethnopharmacol. 2005, 102, 450–456. [Google Scholar] [CrossRef]
- Cos, P.; Vlietinck, A.J.; Berghe, D.V.; Maes, L. Anti-infective potential of natural products: How to develop a stronger in vitro ‘proof-of-concept’. J. Ethnopharmacol. 2006, 106, 290–302. [Google Scholar] [CrossRef]
- McManus, P.S.; Stockwell, V.O.; Sundin, G.W.; Jones, A.L. Antibiotic use in plant agriculture. Annu. Rev. Phytopathol. 2002, 40, 443–465. [Google Scholar] [CrossRef] [PubMed]
- Carvajal, M.A.; Vergara, A.P.; Santander, R.; Osorio, M.E. Chemical Composition and Anti-phytopathogenic Activity of the Essential Oil of Beilschmiedia miersii. Nat. Prod. Commun. 2016, 11, 1367–1372. [Google Scholar] [CrossRef] [PubMed]
- Molecular Operating Environment (MOE), 2013.08; Chemical Computing Group ULC: Montreal, QC, Canada, 2018.
- Frisch, M.J.; Trucks, G.W.; Schlegel, H.B.; Scuseria, G.E.; Robb, M.A.; Cheeseman, J.R.; Scalmani, G.; Barone, V.; Petersson, G.A.; Nakatsuji, H.; et al. Gaussian 09. Revision A.02; Gaussian Inc.: Wallingford, CT, USA, 2016. [Google Scholar]
- Morris, G.M.; Huey, R.; Lindstrom, W.; Sanner, M.F.; Belew, R.K.; Goodsell, D.S.; Olson, A.J. AutoDock4 and AutoDockTools4: Automated docking with selective receptor flexibility. J. Comput. Chem. 2009, 30, 2785–2791. [Google Scholar] [CrossRef] [PubMed]
- GraphPad Prism, Version 6.00 for Mac OS X; GraphPad Software: La Jolla, CA, USA, 2015.
| Compound | MRSA 97-7 | MRSA 622-4 | ATCC 6538 |
|---|---|---|---|
| 7 | 21% | 0% | 6% |
| 8 | 5% | 0% | 26% |
| 9 | 61% | 3% | 52% |
| 10 | 13% | 18% | 17% |
| 11 | 95% | 93% | 100% |
| 12 | 98% | 98% | 99% |
| 13 | 91% | 96% | 99% |
| Compound | MRSA 97-7 | MRSA 622-4 | ATCC6538 |
|---|---|---|---|
| 4 | >50 | >50 | >50 |
| 11 | 5 | 25 | 10 |
| 12 | 15 | 25 | 15 |
| 13 | 10 | 50 | 10 |
| Vancomycin | 25 | 5 | 3.5 |
| Ciprofloxacin | 100 | 10 | 2.5 |
| Methicillin | 50 | 50 | 10 |
| Agents | MIC Alone (µg/mL) | MIC Combined (µg/mL) | FICI | Outcome | Fold Reduction in Commercial Antibiotic MIC |
|---|---|---|---|---|---|
| 11 Vancomycin | 5 25 | 0.5 2.5 | 0.2 | Synergy | 10 |
| 12 Vancomycin | 15 25 | 5 2.5 | 0.43 | Synergy | 10 |
| 13 Vancomycin | 10 25 | 0.5 2.5 | 0.15 | Synergy | 10 |
| 11 Ciprofloxacin | 5 100 | 2.5 10 | 0.6 | Partial Synergy | 10 |
| 12 Ciprofloxacin | 15 100 | 1 10 | 0.16 | Synergy | 10 |
| 11 Methicillin | 5 50 | 0.02 0.5 | 0.01 | Synergy | 100 |
| Agents | MIC Alone (µg/mL) | MIC Combined (µg/mL) | FICI | Outcome | Fold Reduction in Commercial Antibiotic MIC |
|---|---|---|---|---|---|
| 11 Ciprofloxacin | 25 10 | 2.5 0.1 | 0.1 | Synergy | 100 |
| 12 Ciprofloxacin | 25 10 | 5 0.1 | 0.2 | Synergy | 100 |
| 13 Ciprofloxacin | 50 10 | 5 0.1 | 0.1 | Synergy | 100 |
| Strain | Agents | MIC Alone | MIC Combined | FICI | Outcome |
|---|---|---|---|---|---|
| MRSA 97-7 | 11 12 | 5 15 | 5 15 | 2 | Indifferent |
| MRSA 622-4 | 11 12 | 25 25 | 2.5 25 | 1.1 | Indifferent |
| Comp. | Candida albicans ATCC10231 | Cryptococcus neoformans ATCC32264 | Aspergillus flavus ATCC 9170 | Aspergillus fumigatus ATCC26934 | Aspergillus niger ATCC9029 | Microsporum gypseum CCC115, | Trichophyton rubrum CCC113 | Trichophyton mentagrophytes ATCC9972 |
|---|---|---|---|---|---|---|---|---|
| 1 | >250 | >250 | >250 | >250 | >250 | >250 | >250 | >250 |
| 2 | >250 | >250 | >250 | >250 | >250 | >250 | >250 | >250 |
| 3 | >250 | >250 | >250 | >250 | >250 | >250 | >250 | >250 |
| 4 | >250 | >250 | >250 | >250 | >250 | >250 | >250 | >250 |
| 5 | >250 | >250 | >250 | >250 | >250 | >250 | >250 | >250 |
| 7 | >250 | >250 | >250 | >250 | >250 | >250 | >250 | >250 |
| 8 | >250 | >250 | >250 | >250 | >250 | >250 | >250 | >250 |
| 9 | 62.5/62.5 | 62.5/62.5 | >250 | >250 | >250 | 62.5/62.5 | 62.5/62.5 | 62.5/62.5 |
| 11 | >250 | 125/125 | >250 | >250 | >250 | 62.5/62.5 | 62.5/62.5 | 62.5/62.5 |
| 12 | 125/250 | 62.5/125 | >250 | >250 | >250 | 31.25/31.25 | 15.6/31.25 | 15.6/31.25 |
| 10 | >250 | 250/>250 | >250 | >250 | >250 | 250/250 | 250/250 | 250/250 |
| 13 | >250 | >250 | >250 | >250 | >250 | >250 | >250 | >250 |
| Amph | 1.0/1.0 | 1.0/2.0 | 2.0/2.0 | 2.0/2.0 | 2.0/2.0 | - | - | - |
| Terb | - | - | - | - | - | 0.008/0.015 | 0.004/0.008 | 0.004/0.015 |
| Compound | C. neoformans Clinical Isolates | ||||
|---|---|---|---|---|---|
| IM983040 | IM972724 | IM042074 | IM983036 | IM00319 | |
| 9 | 125/250 | 125/250 | 125/125 | 125/125 | 125/125 |
| 11 | 250/>250 | 250/>250 | 250/>250 | 250/>250 | 250/>250 |
| 12 | 125/125 | 125/125 | 62.5/62.5 | 62.5/125 | 125/125 |
| Amph | 0.25 | 0.25 | 0.12 | 0.25 | 0.5 |
| Compound | C. albicans Clinical Strains | ||||
|---|---|---|---|---|---|
| C. albicans CCC 126 | C. albicans CCC 127 | C. albicans CCC 128 | C. albicans CCC 129 | C. albicans CCC 130 | |
| 9 | 62.5/62.5 | 62.5/62.5 | 62.5/62.5 | 62.5/62.5 | 62.5/62.5 |
| 12 | 125/125 | 125/125 | 125/125 | 125/125 | 125/125 |
| Amph | 1.56 | 0.78 | 1.56 | 0.78 | 0.50 |
| Compound | IC50/MIC (µM) | ||
|---|---|---|---|
| P. syringae | A. tumefaciens | P. carotovorum | |
| 1 | >31.3/>250 | >250/>250 | >250/>250 |
| 2 | >62.5/>250 | >250/>250 | >250/>250 |
| 3 | >250/>250 | >250/>250 | >250/>250 |
| 4 | >250/>250 | >250/>250 | >250/>250 |
| 5 | >250/>250 | >250/>250 | >250/>250 |
| 7 | <3.9/>250 | <3.9/>250 | <3.9/>250 |
| 8 | <3.9/>250 | <3.9/>250 | <3.9/>250 |
| 9 | <3.9/>250 | <3.9/>250 | <3.9/>250 |
| 11 | <3.9/>250 | <3.9/>250 | <3.9/>250 |
| 12 | <3.9/>250 | <3.9/>250 | <7.8/>250 |
| 10 | <15.6/>250 | <3.9/>250 | <3.9/>250 |
| 13 | <3.9/125 | <3.9/>250 | <3.9/125 |
| Compound | % Inhibition |
|---|---|
| 7 | 4 |
| 8 | 1 |
| 9 | 51 |
| 10 | 38 |
| 11 | 20 |
| 12 | 12 |
| 13 | 17 |
| Compound | IC50 µM ± SD | Compound | IC50 µM ± SD |
|---|---|---|---|
| 1 | NA | 7 | NA |
| 2 | 13.22 ± 0.49 | 8 | NA |
| 3 | 12.82 ± 0.48 | 9 | 9.49 ± 0.36 |
| 4 | NA | 10 | 10.32 ± 0.03 |
| 5 | NA | 11 | NA |
| 12 | NA | ||
| Trolox | 22.54 ± 0.61 | 13 | NA |
| Compound | IC50 (µM) ± SD | ||
|---|---|---|---|
| MDA-MB-231 | B16-F10 | MEF | |
| 11 | 53.94 ± 9.66 | 49.14 ± 3.38 | 48.45 ± 3.44 |
| 12 | 59.02 ± 3.25 | 45.53 ± 3.82 | 54.66 ± 3.35 |
| Taxol | 0.21 ± 0.05 | 0.48 ± 0.08 | 0.40 ± 0.07 |
| Etoposide | 1.33 ± 0.28 | 1.74 ± 0.32 | 1.03 ± 0.17 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Osorio, M.; Carvajal, M.; Vergara, A.; Butassi, E.; Zacchino, S.; Mascayano, C.; Montoya, M.; Mejías, S.; Martín, M.C.-S.; Vásquez-Martínez, Y. Prenylated Flavonoids with Potential Antimicrobial Activity: Synthesis, Biological Activity, and In Silico Study. Int. J. Mol. Sci. 2021, 22, 5472. https://doi.org/10.3390/ijms22115472
Osorio M, Carvajal M, Vergara A, Butassi E, Zacchino S, Mascayano C, Montoya M, Mejías S, Martín MC-S, Vásquez-Martínez Y. Prenylated Flavonoids with Potential Antimicrobial Activity: Synthesis, Biological Activity, and In Silico Study. International Journal of Molecular Sciences. 2021; 22(11):5472. https://doi.org/10.3390/ijms22115472
Chicago/Turabian StyleOsorio, Mauricio, Marcela Carvajal, Alejandra Vergara, Estefania Butassi, Susana Zacchino, Carolina Mascayano, Margarita Montoya, Sophia Mejías, Marcelo Cortez-San Martín, and Yesseny Vásquez-Martínez. 2021. "Prenylated Flavonoids with Potential Antimicrobial Activity: Synthesis, Biological Activity, and In Silico Study" International Journal of Molecular Sciences 22, no. 11: 5472. https://doi.org/10.3390/ijms22115472
APA StyleOsorio, M., Carvajal, M., Vergara, A., Butassi, E., Zacchino, S., Mascayano, C., Montoya, M., Mejías, S., Martín, M. C.-S., & Vásquez-Martínez, Y. (2021). Prenylated Flavonoids with Potential Antimicrobial Activity: Synthesis, Biological Activity, and In Silico Study. International Journal of Molecular Sciences, 22(11), 5472. https://doi.org/10.3390/ijms22115472