Abstract
The alterations in serum trace element levels are common phenomena observed in patients with different psychiatric conditions such as schizophrenia, autism spectrum disorder, or major depressive disorder. The fluctuations in the trace element concentrations might act as potential diagnostic and prognostic biomarkers of many psychiatric and neurological disorders. This paper aimed to assess the alterations in serum trace element concentrations in patients with a diagnosed schizophrenia. The authors made a systematic review, extracting papers from the PubMed, Web of Science, and Scopus databases according to the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines. Among 5009 articles identified through database searching, 59 of them were assessed for eligibility. Ultimately, 33 articles were included in the qualitative synthesis. This review includes the analysis of serum levels of the following trace elements: iron, nickel, molybdenum, phosphorus, lead, chromium, antimony, uranium, magnesium, aluminum, zinc, copper, selenium, calcium, and manganese. Currently, there is no consistency regarding serum trace element levels in schizophrenic patients. Thus, it cannot be considered as a reliable prognostic or diagnostic marker of schizophrenia. However, it can be assumed that altered concentrations of those elements are crucial regarding the onset and exaggeration of either psychotic or negative symptoms or cognitive dysfunctions.
1. Introduction
Schizophrenia is a multifactorial psychiatric condition with a complex pathophysiological theory. It is described as a disruption of thought processes and an inconsistency between thoughts, emotions, and behavior. Schizophrenia prevalence is about 0.7%, with an onset in the early 20s among males and approximately 3 to 4 years later among females; its incidence is also 1.15-fold higher for men [1,2]. Patients diagnosed with schizophrenia suffer from psychotic symptoms: hallucinations, delusions, and negative symptoms, which include social withdrawal or flat affect, as well as cognitive impairments [3,4,5]. As a multifaceted condition, schizophrenia has a wide spectrum of hypotheses, among which alterations and abnormalities within neurotransmission (dopamine hypothesis) seem to be of primary relevance [6,7,8,9,10,11]. Other hypotheses refer to genetic predisposition, altered γ-aminobutyric acid (GABA) levels, nicotinic receptors, the endocannabinoid system, inflammation with oxidative stress, sex and developmental differences in brain anatomy, the protective role of estrogens, gut microbiota, or vitamin D deficiency [12,13,14,15,16,17,18,19,20,21,22,23].
The onset of psychiatric conditions or exaggerations of their symptoms might be induced by numerous factors including genetic predispositions, infections, inflammations, trace elements and vitamin levels, severe stress, postpartum complications, intellectual disability, neurodegenerative diseases, trauma, stimulants, and socio-economic and psychological factors [24,25,26,27,28,29,30,31]. The importance of various trace elements’ concentrations in human organisms cannot be underestimated since they are a crucial factor responsible for either induction or exaggeration of numerous disease symptoms (Table 1) [32,33,34,35,36,37,38].

Table 1.
Physiological ranges and biological functions of trace elements in the human organism.
Furthermore, trace elements’ imbalances might constitute indicators of disease remission or progression. It is still debatable whether altered trace elements’ concentrations could be implicated in the increased probability of schizophrenia onset. Numerous trace elements are crucial in the maintenance of proper functioning within the central nervous system (CNS) and these include physiological ranges of chromium (Cr), Cu, Fe, Mn, Se, or Zn. Therefore, pathological alterations in trace elements’ levels might contribute to the adverse impairments of the biological processes, inducing various diseases of the CNS or exacerbate already existing symptoms of autism spectrum disorder (ASD), neurodegenerative disorders (Alzheimer, Parkinson, Huntington diseases), dementia, major depressive disorder, or attention deficit hyperactivity disorder (ADHD) [144,145,146,147,148,149,150]. Several studies have shown altered levels of serum trace elements’ concentrations, as well as their impaired distribution within CNS in schizophrenic patients. Among numerous triggering factors, nutritional deprivation significantly affects the levels of certain trace elements, inducing oxidative stress, irrational behavior, and impaired cognition [151]. Besides, an association between metal exposure with subsequent concentration imbalances and psychotic symptoms’ severity was stated [152].
2. Aim of the Review and Search Strategy
The objective of this paper was to conduct a review of the available literature regarding serum trace elements’ concentrations in schizophrenic patients. A systematic literature review of PubMed, Web of Science, and Scopus databases was performed by two identifications in March 2020. The first identification included the search strategy as follows: (schizophrenia OR schizophrenic) AND (trace element). After reviewing 373 articles searching for serum trace elements’ concentrations that had already been investigated in sera of schizophrenic patients, we chose the following ones to include in this review: magnesium, aluminum, zinc, copper, selenium, rubidium, potassium, cadmium, calcium, lithium, molybdenum, phosphorus, antimony, uranium, and manganese. We continued as the second identification with the use of the following search string: (schizophrenia OR schizophrenic) AND (magnesium OR aluminum OR cobalt OR zinc OR copper OR selenium OR rubidium OR potassium OR cadmium OR calcium OR lithium OR molybdenum OR phosphorus OR antimony OR uranium OR manganese). Eventually, 33 articles were included in a qualitative synthesis. The literature search included both human and animal studies. There were no restrictions regarding the year of a publication. The authors chose only articles in English. In the final analysis, regarding serum trace element concentrations in schizophrenics, we included only human studies. Twenty-six articles were excluded for several reasons: concentrations were measured in blood plasma, within erythrocytes, or in hair samples only; the aim of the study was not specifically focused on serum level of particular trace element; there were no sufficient, relevant, or accurate data; or it was investigated in a study, but lack of specific significant results disabled it from further analysis and description (Figure 1).
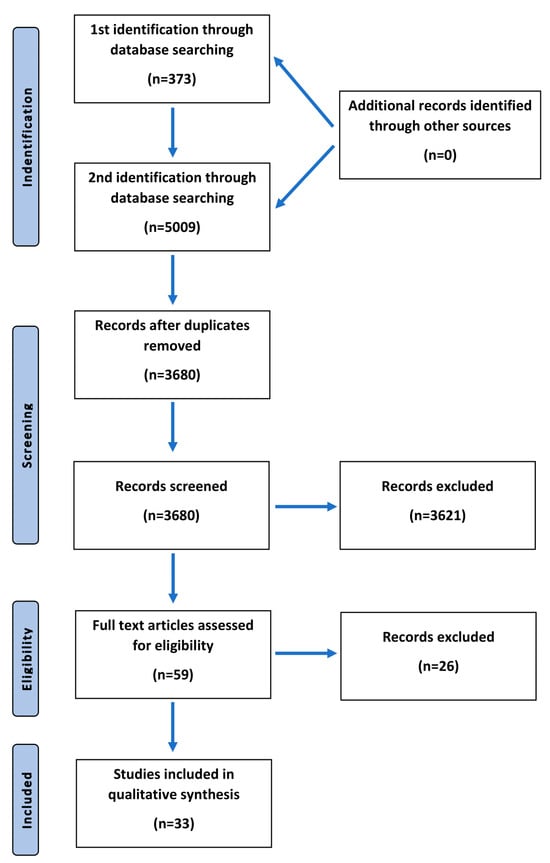
Figure 1.
Flowchart presenting the process of article selection, according to Preferred Reporting Items for Systematic Review and Meta-Analyses (PRISMA) guidelines.
3. Results
After two identifications and duplicates were removed, we assessed 59 articles for eligibility, among which 33 of them were chosen in a qualitative synthesis. The articles included concerned the studies performed on humans. The time range of the published articles was 1950–2020. Two major inclusion criteria were the diagnosis of schizophrenia among patients and the examined trace elements’ concentrations in patients’ sera. Trace elements that were taken into consideration in this analysis included iron, nickel, molybdenum, phosphorus, lead, chromium, antimony, uranium, magnesium, aluminum, zinc, copper, selenium, calcium, and manganese. The results of the systematic review are included in Table 2.

Table 2.
The studies about serum trace element concentrations in schizophrenic patients included in the systematic review.
4. Iron
Iron (Fe) metabolism is crucial regarding all neurobehavioral aspects since this element is involved in the metabolism of various neurotransmitters (serotonin, norepinephrine, dopamine), myelin synthesis, proper cellular functioning, and brain development [186,187,188,189]. The abovementioned neurotransmitters are synthesized by several Fe-dependent enzymes including phenylalanine hydroxylase, tryptophan hydroxylase, and tyrosine hydroxylase [190]. Therefore, Fe deficiency might decrease serotonin and dopamine levels, as well as increase urinary norepinephrine; this phenomenon is also associated with the direct involvement of Fe on hydroxyl radicals and reactive oxygen species’ formation [191]. Fe, being a required cofactor of cholesterol, is directly involved in the myelination processes [192]. Fe-mediated dopamine neurotoxicity is associated with quinones’ formation [193]. Animal studies of Fe concentrations showed an association between Fe deficiency, abnormal behavior, and alterations of dopamine metabolism [194,195]. Further, several human studies, as well as animal models of Fe depletion, presented significant behavioral and electrophysiological findings [196,197,198,199,200]. Fe deficiency is associated with developmental delays in children and cognitive impairments in adolescents; furthermore, it might affect motor development, recognition memory, socio-emotional behavior, and the overall processes of CNS maturation. Fe deficiency alters the density and activity of dopamine type 2 (D2) receptors by mimicking their blockade, which is implicated in the pathophysiology of schizophrenia [169]. Contrarily, increased Fe concentrations can lead to neuronal death primarily due to the enhanced oxidative stress, which might subsequently promote a progression of a wide range of neuropsychiatric disorders and neurodegenerative diseases [201,202,203,204,205]. Animal models have also shown that Fe overload might reduce dopamine levels in the striatum, induce neurodegeneration in the midbrain, and enhance the vulnerability to toxic injury [206]. Besides, increased Fe levels influence emotional behavior, which is determined by various biological and physiological indicators such as neurotransmitters’ concentration, density and affinity of neurotransmitter receptors, or Fe concentrations in specific brain regions [207]. Disturbed Fe metabolism impairs dopaminergic activity, resulting in a progression of negative symptoms of schizophrenia [169]. In the available literature, there is still no consensus on the altered levels of Fe in the serum of schizophrenic patients.
4.1. Serum Iron Levels
The results of the studies regarding serum Fe levels remain inconsistent. Generally, acutely schizophrenic patients present with decreased serum Fe concentrations [174]. Apart from lowered Fe levels, schizophrenic patients display decreased Mn and elevated Cu concentrations [208]. However, other researchers showed increased serum Fe and Ni levels among schizophrenic patients [156]. There is no correlation between Fe, Mn, Cu, Se, and Zn levels and duration of schizophrenia, doses of antipsychotic drugs, protein content, or smoking habits of patients. Despite altered serum Fe levels, there is no significant difference in ferroxidase II levels and Fe-binding capacity; besides schizophrenic patients present elevated levels of ceruloplasmin by approximately 20% [165]. Furthermore, schizophrenic patients exhibit a positive correlation between serum Fe concentrations and aspartate aminotransferase (AST) with alanine transaminase (ALT) levels [156]. It was suggested that altered serum Fe levels in schizophrenic patients might be due to the imbalanced diet, rather than being a separate characteristic of schizophrenia [181]. Moreover, increased Fe, Cu, Pb, Ca, Se, and Mn serum levels might be protective, whereas increased B, Cr, Mg, K, and As levels might constitute risk factors for schizophrenia [161]. Due to significant inconsistencies regarding serum Fe levels in schizophrenic patients, serum Fe levels can be considered neither as diagnostic nor as a prognostic factor of schizophrenia.
4.2. Iron Levels in the CNS
Casanova et al. observed a greater mean content of Fe per internal segment of globus pallidus in the post-mortem study of schizophrenic patients; however, these findings might be limited due to the small number of patients [209]. Even though several post-mortem studies showed the presence of basal ganglia mineralization (especially Fe deposition) in schizophrenic patients, the results remain inconsistent among researchers [210,211]. Microscopic examination of basal ganglia of schizophrenics revealed Fe deposition in the walls of small blood vessels [209]. Other studies showed no significant alterations in Fe, Cu, Zn, Mg, and Ca concentrations in the post-mortem brains of schizophrenic patients and control groups. However, both schizophrenic patients and control groups present with increased Fe and Cu concentrations in the caudate nucleus compared to the hippocampus and amygdala [212,213]. Demmel et al. presented a case of a schizophrenic patient whose element concentrations (Fe, Co, Rb, Se, and Zn) were at normal range in contrast to patients with alcohol abuse (decreased levels of Rb in cerebral nuclei and Co with Rb in cortical regions) or endogenous psychosis (increased levels of Fe, Co, Rb, Se, and Zn in the caudate nucleus) [214].
To the best of authors’ knowledge, there is no study that was investigating serum Fe and Fe levels in the brain at the same time. According to the reviewed literature, serum Fe levels tend to be decreased in schizophrenic patients. Contrarily, post-mortem studies focusing on Fe levels in the CNS showed that Fe might accumulate in several regions such as globus pallidus or basal ganglia.
4.3. Prenatal Iron Deficiency
Maternal Fe deficiency is an early environmental factor, which significantly enhances the probability of schizophrenia onset in offspring since Fe is involved in early behavioral and reflex neurodevelopment [169,186,215,216,217,218]. Moreover, low maternal hemoglobin concentrations might be associated with a nearly 4-fold elevated risk for schizophrenia in offspring [219]. The mechanism involves anemia induced by maternal Fe depletion and further reduction of oxygen delivery in the developing fetus [220]. Since proper Fe metabolism is a crucial component in myelination and dopaminergic transmission, any pathological alterations in its levels might disrupt fetal neurodevelopment. Apart from schizophrenia, fetal or postnatal Fe deficiency might influence the development of other psychiatric conditions including ASD, intellectual disability, hyperactivity disorder, anemia, anxiety, or depression in offspring [219,220,221].
4.4. Schizophrenic Patients with Akathisia
There is a significant inconsistency in the results of the studies regarding serum Fe levels in schizophrenic patients with coexisting symptoms such as akathisia. Barnes et al. showed that schizophrenic patients with chronic akathisia do not present lowered serum Fe concentrations; further, there is no relationship between serum Fe levels and the severity of akathisia [167]. Similar results were presented by Soni et al., where serum Fe, ferritin, and total iron-binding capacity (TIBC) parameters were not altered in schizophrenic patients with neuroleptic-induced akathisia [171]. Likewise, schizophrenic patients with acute dystonia are not characterized by low serum Fe, transferrin, or ferritin levels [172]. Wirshing et al. suggested that serum ferritin levels are associated with choreoathetoid movements especially among male schizophrenic patients treated permanently [173].
Contrarily, other studies showed that schizophrenic patients (either akathisic or non-akathisic) have significantly lowered serum Fe and ferritin levels compared to control groups [170]. The difference was more pronounced in schizophrenics with akathisia; those patients also presented elevated TIBC values. Furthermore, male schizophrenic patients present lowered ferritin but higher serum Fe levels comparing to females [168].
4.5. Serum Iron Levels during Treatment
The majority of studies have proven that antipsychotic treatment induces alterations in trace elements serum concentrations in schizophrenic patients, among which Na, Ca, K, Mg, Al, P, Zn, and Fe are mostly affected. However, several studies proved that Fe, ferritin, or transferrin levels may remain unchanged during treatment with neuroleptics [172]. Sussulini et al. (2017) observed that antipsychotic treatment (risperidone, olanzapine, quetiapine) induces the increase of Fe levels only in good responders, which provides the possibility to investigate Fe levels in monitoring responses to antipsychotic treatment among schizophrenic patients [222]. Contrarily, Chen et al. showed that antipsychotic drugs decrease Fe and increase P serum levels [157]. A long-term haloperidol treatment leads to Fe deficiency anemia, possibly due to haloperidol chelation abilities [166]. Furthermore, haloperidol-induced Fe deficiency alters the dopamine receptor D2 (DRD2) number. The reduction of serum Fe and ferritin levels with physiological TIBC status might be due to lurasidone treatment [223]. Besides, Li treatment significantly increases serum Fe concentrations in schizophrenics when compared to healthy controls [154].
5. Serum Nickel, Molybdenum, Phosphorus, Lead, Chromium, Antimony, and Uranium Levels
Increased Ni levels might induce alterations within the central as well as peripheral nervous systems, proving its cytotoxicity by various underlying mechanisms including mitochondrial dysfunctions [224]. Accumulation of Ni within the CNS disrupts neurotransmission and leads to the apoptosis of olfactory sensory and cerebral cortex neurons [191]. Further, some animal research showed that Ni exposure might increase the probability of aggressive behavior and affective disorders [225]. Ni affects the acetylcholine release, as well as decreases dopamine, norepinephrine, and serotonin levels within the CNS; it might also affect the gene expression of Glu receptors [226]. Nevertheless, literature specifically concerned with serum Ni levels in schizophrenic patients is scarce. Cao et al. (2019) showed that compared to healthy controls, patients with recurrent schizophrenic episodes presented elevated serum Ni and Fe concentrations [156]. Furthermore, an adjusted odds ratio (OD) revealed that serum Ni and Co levels are positively associated with schizophrenia.
Liu et al. did not point to any correlation between serum Mo or Zn levels and increased risk of schizophrenia [153]. However, patients with schizophrenia tend to present significantly lower serum concentrations of Mo [156].
Several studies suggest that altered P metabolism might reflect the degree of psychiatric symptoms and correlate with the presence of positive or negative symptoms [227,228]. Chen et al. observed that lower P levels are associated with increased schizophrenia risk and antipsychotic treatment might increase its serum levels, presenting its impact on the P metabolism [157]. These results were supported by Jamilian et al. who reported significantly lower levels of serum P in schizophrenics compared to healthy controls [181].
Ma et al. showed that increased serum antimony (Sb) and U levels might be associated with an elevated risk of schizophrenia [184]. Further studies showed that also increased serum concentrations of Pb and Cr constitute a risk factor for schizophrenia [185]. Even though, more research should be carried out in order to assess whether any of the above-described elements could act as a potential diagnostic and/or prognostic factor of schizophrenia.
6. Magnesium
Mg is the fourth most abundant element in human organism, involved in more than 300 enzymatic reactions including protein, DNA, and RNA synthesis [68]. Several human and animal molecular studies have proven its neuroprotective role. Its protective role in the nervous system includes the prevention of excessive excitation and potential excitotoxicity involved in the onset of numerous neurological disorders [71]. Therefore, any imbalances in Mg concentrations (primarily hypomagnesemia) might be associated with various disorders, including those with psychiatric/psychotic background such ADHD, depression, anxiety, Alzheimer and Parkinson diseases, migraine pain, epilepsy, or schizophrenia [229,230]. However, higher Mg and lower Zn levels are related to more severe psychopathology after metal exposure [152]. There is no consistency regarding serum Mg levels in schizophrenic patients [178,231,232]. However, it might be probably due to researchers’ tendency to determine only extracellular concentrations, while Mg constitutes an intracellular ion [233]. Furthermore, there are still no data regarding the concentration of ionized Mg levels in schizophrenic patients. Likewise, no altered levels in brain Mg concentrations in post-mortem studies have been reported [212]. Nevertheless, there is a significant number of studies regarding altered Mg levels after different types of medications used to treat schizophrenia.
Serum Magnesium Levels during Treatment of Schizophrenia
Pimozide and fluphenazine induced a decrease of both serum Mg and Ca levels in the study by Alexander et al. on schizophrenics treated with neuroleptics [175]. Further studies confirmed the fact that treatment with pimozide, fluphenazine, or loxapine might slightly alter both serum Mg and Ca levels, also inducing extrapyramidal symptoms; however, according to researchers, the slight increase of Mg levels is not statistically significant [177]. Schizophrenic patients who tolerate higher doses of neuroleptics present higher serum Mg and Ca levels. Besides, Mg and Ca concentrations together correlate with pimozide dosage at which extrapyramidal symptoms might develop; this might be a result of impaired dopaminergic and cholinergic activity affected by altered Mg and Ca levels [176]. Contrarily, patients treated with major tranquilizers did not show any differences in serum Mg levels [178]. Mg levels in schizophrenics might be higher compared to healthy control groups, however, antipsychotic treatment (risperidone, clozapine, olanzapine, aripiprazole, quetiapine, perphenazine, sulpiride, ziprasidone) leads to a decrease of serum Mg levels [157,161].
7. Aluminum
Al, as a toxic metal, can accumulate in every organ particularly within the CNS, which is implicated in the onset or progression of neurodegenerative disorders. Increased Al concentrations are associated with the induction of oxidative stress, modification of membrane biophysics, impaired cell signaling, and neurotransmission [234]. Al increases the permeability of the blood–brain barrier and accumulation of this metal might induce the impairments of higher cognitive functions [235,236]. Besides, chronic Al exposure is associated with neurochemical, behavioral, and neuropathological impairments.
Kaya et al. observed higher Al, Fe, Cu, and Mg levels and lower Zn and Mg levels in the sera of schizophrenic patients compared to healthy controls [237]. Nevertheless, a more recent study by Liu et al. (2015) showed that lower concentrations of Al, Fe, Cu, Se, and As along with higher concentrations of Mg and Cr are associated with the increased risk of schizophrenia onset; however, these results were not statistically significant [153]. This inconsistency should be further evaluated in order to provide statistically significant results.
8. Zinc
Zn constitutes an essential trace element across the human lifespan, being involved in the control of early neonatal brain development and proper functioning of the CNS. Zn-containing neurons are found in the highest amounts primarily in the forebrain, being responsible for the interconnections between the limbic system and the majority of cerebral cortices [82]. At the molecular level, Zn is responsible for neuronal metabolism and plasticity, as well as synaptic activity. Thus, imbalances in Zn levels (especially increased concentrations) and further Zn accumulation within the CNS leads to neuronal damage via excitotoxicity, oxidative stress, and impaired cellular energy generation [80,238,239]. Zn deficiency during pregnancy and its insufficient levels during child development are associated with a higher probability of mental retardation, apathy, lethargy, and impaired learning ability [240,241]. Imbalanced Zn homeostasis is associated with an induction of various neurological and psychiatric conditions such as Alzheimer and Parkinson diseases, depression, amyotrophic lateral sclerosis, epilepsy, or schizophrenia [242,243,244].
8.1. Serum Zinc Levels
There is an inconsistency in the current literature regarding serum Zn levels in schizophrenic patients; nonetheless, in a majority of cases, serum Zn levels are usually reported to be decreased [183,245]. Cruz et al. showed significantly lower serum Zn, Se, and Fe levels, as well as higher Cu/Zn ratio among patients with schizophrenia compared to healthy controls [154]. Furthermore, the authors have suggested that Cu/Zn ratio might constitute a useful parameter related to oxidative stress in schizophrenics. Other researchers have suggested that there is no association between serum Zn and Mo levels and the increased risk of schizophrenia [153]. These results were confirmed by Cao et al., who showed that serum Zn, Mn, and Mo levels are negatively correlated, whereas serum Ni and Co levels are positively correlated with schizophrenia [156]. Besides, Saghazadeh et al. showed that schizophrenic patients tend to present lower Zn, Fe, and Mn serum concentrations, along with excessive Cu amounts [246]. Based on the results of the evaluated studies it can be assumed that Zn levels are rather decreased in schizophrenic patients; however, it cannot yet be stated whether serum Zn concentrations can be treated as either a prognostic or diagnostic factor of schizophrenia.
8.2. Zinc Levels in the CNS
A post-mortem study, which included 10 brains obtained from the schizophrenic patients during the autopsy, revealed the ionic Zn staining especially within the hippocampal formation [247]. Terminal zones of the mossy fiber system within the dentate hilus and the CA3 region of the hippocampus presented the highest density of Zn staining. A lighter Zn staining was present within the outer and inner layers of the dentate gyrus molecular layer, as well as within the CA1 region of the hippocampus. Another study, which included 12 subjects, showed that Zn levels were highest in the hippocampus, with lower Zn concentrations within the amygdala and the caudate nucleus; however, there were no significant differences between Zn levels among schizophrenics and a control group [212]. The results of these studies are intriguing since serum Zn levels in schizophrenic patients tend to be rather decreased. However, there are no studies that were investigating serum Zn and Zn levels in the CNS of schizophrenic patients at the same time.
8.3. Serum Zinc Levels during Treatment
Antipsychotic treatment leads to a decrease of serum Zn levels regardless of the treatment duration. Chen et al. observed that serum of both Zn and Cu were lowered after the antipsychotic treatment (which included risperidone, clozapine, olanzapine, aripiprazole, quetiapine, perphenazine, sulpiride, and ziprasidone) immediately, as well as after three weeks of treatment duration [157]. The authors also showed a significant decrease in serum Zn levels after risperidone treatment, whereas there was no association between Zn levels and olanzapine treatment. Valproate treatment was also reported to stabilize decreased Zn and K levels [248].
9. Copper
Cu is an essential micronutrient required for numerous cellular functions such as cellular respiration, neurotransmitter synthesis, neuronal myelination, or proper maintenance of Fe metabolism [249]. Cu constitutes a crucial element during the synthesis of ceruloplasmin, zyklopen, hephaestin, and dopamine-β-monooxygenase, which is crucial in dopamine metabolism and dopaminergic activity. Since Cu participates in reactions that generate free radicals’ contribution to oxidative stress, excessively high amounts of this transition metal might be toxic. Cu distribution within the brain parenchyma is generally even. However, the subventricular zone of brain ventricles was observed with higher Cu levels compared to other regions in the brain [250]. Post-mortem studies of Cu concentration within the CNS showed increased Cu concentrations mainly in the caudate nucleus in schizophrenic patients [212]. Disturbed distribution and homeostasis of Cu within the CNS contributes to the promotion of many neurodegenerative diseases: Alzheimer and Parkinson diseases, Huntington disease, amyotrophic lateral sclerosis, as well as psychiatric conditions such as depressive disorder, ASD, epilepsy, or schizophrenia primarily due to the impaired neuronal myelination, catecholamine imbalances, and disturbed brain architecture [251,252,253,254,255]. Neurobehavioral abnormalities might be a consequence of maternal Cu deficiency as well [186]. Schoonover et al. showed impaired intracellular Cu binding and transport into the cell via high-affinity Cu uptake protein 1 (Ctr1) in schizophrenic patients [256].
9.1. Serum Copper Levels
Regarding serum Cu concentrations among patients diagnosed with schizophrenia, there is still no consensus, as in the case of serum Fe and Zn levels. Several studies did not show any significant difference in serum Cu or ceruloplasmin levels in schizophrenics [159,160,162,163]. Other results have shown increased serum Cu levels among schizophrenic males and females compared to the control group [164]. Besides, serum Cu levels were higher in females, however, without statistical significance. Further, Cruz et al. observed increased serum Cu levels among patients with schizophrenia or bipolar disorder compared to control groups; however, these results were not statistically significant [154]. Cao et al. did not observe any significant differences in serum Cu, Co, and Fe levels in sera of schizophrenics [156]. A study on 40 male schizophrenic patients without any treatment for four weeks before the study revealed higher Cu, ceruloplasmin, LDL-cholesterol, total cholesterol, and high-sensitivity C-reactive protein (hs-CRP) levels, as well as a significant decrease in HDL-level [158]. Further, there was a significant correlation between serum Cu and hs-CRP levels. Wolf et al. also proved significantly elevated serum Cu levels among schizophrenics [165]. Furthermore, as approximately 95% of blood Cu is bound to ceruloplasmin, the researchers confirmed that increased Cu levels correlate with increased ceruloplasmin levels. Therefore, it can be assumed that schizophrenic patients tend to present greater serum Cu concentrations compared to healthy controls.
9.2. Serum Copper Levels during Treatment
Herran et al. showed that schizophrenic patients treated with a depot neuroleptic tend to have higher serum Cu levels compared to patients treated with oral antipsychotics [155]. The researchers observed that either typical or atypical antipsychotic treatment or neuroleptic dosage does not affect serum Cu levels. However, Saghazadeh et al. showed that schizophrenic patients treated with antipsychotic drugs tend to present higher serum Cu levels compared to controls [246]. Clozapine and aripiprazole treatment significantly decrease serum Cu levels compared to levels before treatment [157]. Intake of Se supplements elevates serum Cu and Zn levels [161].
10. Selenium
Various antioxidant selenoproteins such as glutathione peroxidase (GPx), thioredoxin reductase (TrxR), or selenoproteins—P, S, or H ((SelP), (SelS), or (SelH)) require incorporation of Se. Selenoproteins are involved in the free radical defense system, due to their antioxidant activity. They are responsible for proper CNS functioning. Furthermore, Se provides the response to oxidative stress primarily via mitochondrial biogenesis and regulation of Ca2+ channels [257]. SelP, being found primarily in neurons, also exhibits a neuroprotective effect. A neuroprotective role of Se supplementation is achieved via several mechanisms including antioxidant activity and selenoprotein synthesis de novo. Deficient or excessive Se exposure might be associated with the onset and progression of various diseases; however, the knowledge concerning the amount of exposure, as well as Se ranges and specific health outcomes, is still scarce [258]. Animal studies proved that Se deficiency induces neurological dysfunctions, examples of which are seizures, or neurodegeneration especially within the brain areas related to auditory and motor functions [259,260]. Indeed, patients with deficient Se supplementation or mutations in the gene encoding selenocysteine synthase are more susceptible to neurological impairments [261]. Imbalances in Se levels, as well as their disturbed distribution within CNS, might be a trigger of such conditions as schizophrenia, depression, bipolar disorder, or Alzheimer disease [100,149,262,263]. Schizophrenia and bipolar disorder are characterized by an elevated level of selenium binding protein 1 (SELENBP1) gene expression; however, it is yet unclear whether enhanced expression of SELENBP1 correlates with a higher incidence of psychosis [264,265]. It was hypothesized that schizophrenia onset might be not due to the Se deficiency itself, but rather due to the impaired Se transport [266,267].
Serum Selenium Levels
Alertsen et al. did not show a statistically significant difference in serum Se levels between schizophrenics and the healthy controls [179]. A study on 114 schizophrenic patients revealed decreased levels of serum Se; the researchers also observed that lower serum Se, Fe, Cu, Al, and As might be associated with the increased risk of schizophrenia onset [153]. Schizophrenic patients usually present with reduced concentrations of serum Se [154,161,183]. Li et al. showed that Se supplementation by schizophrenics might increase appetite, as well as improve memory abilities—these results concern 60% and 18% of studied females and males, respectively [161]. Besides, the researchers observed that higher concentrations of Se, Mn, Ca, Pb, Fe, and Cu act protectively regarding schizophrenic patients. Thus, serum Se levels seem to be rather decreased in schizophrenics and the elevation of its concentrations might potentially improve the course of schizophrenia.
11. Calcium
Under physiological conditions, serum Ca levels and brain interstitial fluids maintain equilibrium; therefore, any changes in physiological Ca concentrations might affect neuronal and cognitive functions. Free intracellular Ca concentration in neurons is fairly low (a range of 10–100 nM), but some of the organelles such as mitochondria act as ‘Ca stores’ [268]. Numerous neuronal processes such as neurotransmitter release, intracellular signaling, proper homeostasis of glial cells, or N-methyl-D-aspartate (NMDA)-mediated neuroplasticity require the influx of extracellular Ca and its balanced homeostasis. Sharma et al. reported a relationship between higher Ca levels and poorer cognitive functioning in individuals aged 75 and above [269]. Recently, Ca2+/cAMP interaction, the so-called ‘calcium paradox’, was implemented to enhance neurotransmission and neuroprotection [270]. Further, voltage-gated Ca channels are implemented in the pathogenesis of schizophrenia. Other distortions associated with schizophrenia include altered C2+-dependent gene transcription, which affects signaling components. Schizophrenic patients present with increased Ca2+ intracellular levels, involving plasma membrane Ca2+ ATPase (PMCA) in the alterations of Ca levels [271]. The literature concerning serum Ca concentrations in schizophrenic patients is still scarce. Ruljancic et al. pointed to decreased serum Ca levels in suicidal schizophrenic patients compared to the nonsuicidal group of schizophrenics [180]. Besides, there is evidence that schizophrenic patients show an upregulation of Ca-binding proteins particularly within the cerebellum, which indicates impaired Ca signaling and disrupted cerebellar circuits [272]. Even though, more research should be conducted in order to assess whether any differences in serum Ca levels in schizophrenic patients are significant and whether they are truly associated with the course of this psychiatric condition.
12. Manganese
Studies suggest that some metals (Mn, Zn, Cd, Pb) are associated with schizophrenia or psychotic conditions [116,273]. These metals are involved in the induction of alterations in neurotransmission, excitotoxicity, and inflammation [112,274]. Excessive exposure to Mn might induce initial clinical psychotic manifestations, which include mood changes, emotional lability, hallucinations, and uncontrolled laughter, collectively referred to as ‘Mn psychosis’ [275]. Excessive Mn exposure is associated with behavioral and cognitive impairments, decreased IQ levels, reduced educational achievement, or abnormalities within the frontal lobe [276,277,278,279,280]. High levels of Mn were documented to cause Parkinson-like neuromuscular condition and neurocognitive dysfunctions that were primarily found in miners [274]. There is also evidence that large and long-lasting administration of manganese chloride (MnCl2) in rats, due to decreased activity of tyrosine hydroxylase, causes the development of schizophrenia-like behaviors [246]. It was also noted that higher concentrations of Mn decrease Gln uptake into astrocytes and reduce the activity of Gln synthetase in the neurons and astrocytes of the striatum and globus pallidus in the brain [281]. High level of free radicals due to the impaired activity of manganese superoxide dismutase (MnSOD) damage cell membranes causing malfunctioning of neurotransmission, abnormal neuronal apoptosis, and neurodegeneration, which is believed to induce the development of symptomatology in schizophrenia [282]. Researchers suggest that lower concentrations of Mn are associated with reduced MnSOD activity, which might be associated with schizophrenia [156]. So far, the majority of studies have shown that schizophrenic patients present with increased blood concentrations of Mn. However, Yanik et al. showed significantly lower plasma Mn concentrations in schizophrenics compared to control groups [208]. Besides, Cao et al. observed decreased serum Mn levels in cases of first-episode and recurrent schizophrenia, compared to healthy controls [156]. Contrarily, Saghazadeh et al. concluded that there are no significant changes in serum Mn concentrations in patients with schizophrenia; however, the researchers found lower levels of Mn in the subgroups of patients with previously diagnosed or chronic schizophrenia [246]. Plasma MnSOD activity is decreased in schizophrenic patients either with or without tardive dyskinesia [283]. Further, decreased MnSOD activity is associated with impaired cognitive functions in schizophrenics. Serum Mn levels might also be associated with an increased risk of schizophrenia [153]. Some studies suggest that Mn serum levels can be altered by psychoactive substances: They increase with the usage of tobacco or alcohol and decrease by antipsychotic medication [39,284,285]. Another evidence indicates that antipsychotic drugs do not alter the Mn concentrations but instead chelate Mn, making it less available as an enzyme activator [237]. Therefore, at the current state of knowledge, it is quite hard to assess what serum Mn levels might be associated with schizophrenia.
13. Conclusions
At the current state of knowledge, none of the investigated trace elements has presented its utility as a diagnostic or treatment tool of schizophrenia. The results of the analyzed studies were usually contradictory. Thus, the epidemiological evidence of altered serum trace element levels and the risk of schizophrenia onset is slightly controversial. The differences in the results of the abovementioned studies can be due to the heterogeneity of methodology, as well as different inclusion and exclusion criteria. Analytical methods regarding concentrations of trace elements are to some extent limited since, during gel electrophoresis or liquid chromatography, chelated metals can be easily released from proteins, preventing an accurate analysis [286]. Accuracy of the results might be to some extent limited particularly because the concentrations of trace elements are determined usually only in extracellular compartments, while some of the elements (e.g., Mg) are specifically intracellular ions; besides, for the majority of elements, there is a lack of strict concentrations’ ranges.
According to the results of the analyzed studies, maternal deficiency of essential trace elements with micro- and macronutrients along with subsequent dysregulation of early life trace elements’ concentrations might be associated with an increased risk of psychotic disorders including schizophrenia. Furthermore, the causation might be related to maternal stress, as it disrupts the placental transfer of nutrients. Prenatal deficiency of essential trace elements such as Zn induces epigenetic alterations, affecting the levels of other trace elements [152,287]. Alterations in serum trace elements’ levels in schizophrenic patients might be caused by numerous factors, among which current treatment (antipsychotics), exposure and inhalation of particular elements, or dietary intake are crucial.
Since the amount of literature is still scarce and the results are often contradictory, more research should be done in this field with the aim of investigating potential diagnostic and prognostic parameters of schizophrenia.
Funding
This research received no external funding.
Conflicts of Interest
The authors declare no conflict of interest.
Abbreviations
| AChE | acetylcholinesterase |
| ADHD | attention deficit hyperactivity disorder |
| Al | aluminum |
| ALT | alanine aminotransferase |
| ASD | autism spectrum disorder |
| AST | aspartate transaminase |
| Ca | calcium |
| Cd | cadmium |
| CNS | central nervous system |
| Cu | copper |
| Fe | iron |
| GABA | γ-aminobutyric acid |
| GABAA | γ-aminobutyric acid type A |
| Gln | glutamine |
| Glu | glutamate |
| GPx | glutathione peroxidase |
| HDL | high-density lipoprotein |
| hs-CRP | high-sensitivity C-reactive protein |
| IDD | iodothyronine deiodinases |
| K | potassium |
| LDL | low-density lipoprotein |
| Li | lithium |
| Mg | magnesium |
| Mn | manganese |
| MnSOD | manganese superoxide dismutase |
| Mo | molybdenum |
| Ni | nickel |
| NMDA | N-methyl-D-aspartate |
| P | phosphorus |
| PMCA | plasma membrane Ca2+-ATPase |
| Rb | rubidium |
| Sb | antimony |
| Se | selenium |
| SELENBP1 | selenium binding protein 1 gene |
| SelH | selenoprotein H |
| SelP | selenoprotein P |
| SelS | selenoprotein S |
| TIBC | total iron-binding capacity |
| TrxR | thioredoxin reductase |
| U | uranium |
| VLDL | very-low-density lipoprotein |
| Zn | zinc |
References
- Howes, O.; Mccutcheon, R.; Stone, J. Glutamate and dopamine in schizophrenia: An update for the 21st century. J. Psychopharmacol. 2015, 29, 97–115. [Google Scholar] [CrossRef] [PubMed]
- Werf, M.V.D.; Hanssen, M.; Köhler, S.; Verkaaik, M.; Verhey, F.R.; Winkel, R.V.; van Os, J.; Allardyce, J. Systematic review and collaborative recalculation of 133 693 incident cases of schizophrenia. Psychol. Med. 2012, 44, 9–16. [Google Scholar] [CrossRef] [PubMed]
- Picchioni, M.M.; Murray, R.M. Schizophrenia. BMJ 2007, 335, 91–95. [Google Scholar] [CrossRef] [PubMed]
- Tandon, R.; Gaebel, W.; Barch, D.M.; Bustillo, J.; Gur, R.E.; Heckers, S.; Malaspina, D.; Owen, M.J.; Schultz, S.; Tsuang, M.; et al. Definition and description of schizophrenia in the DSM-5. Schizophr. Res. 2013, 150, 3–10. [Google Scholar] [CrossRef]
- Valton, V.; Romaniuk, L.; Steele, J.D.; Lawrie, S.; Seriès, P. Comprehensive review: Computational modelling of schizophrenia. Neurosci. Biobehav. Rev. 2017, 83, 631–646. [Google Scholar] [CrossRef]
- Jauhar, S.; Nour, M.M.; Veronese, M.; Rogdaki, M.; Bonoldi, I.; Azis, M.; Turkheimer, F.; McGuire, P.; Young, A.H.; Howes, O.D. A Test of the Transdiagnostic Dopamine Hypothesis of Psychosis Using Positron Emission Tomographic Imaging in Bipolar Affective Disorder and Schizophrenia. JAMA Psychiatry 2017, 74, 1206. [Google Scholar] [CrossRef]
- Szczypiński, J.J.; Gola, M. Dopamine dysregulation hypothesis: The common basis for motivational anhedonia in major depressive disorder and schizophrenia? Rev. Neurosci. 2018, 29, 727–744. [Google Scholar] [CrossRef]
- Howes, O.D.; McCutcheon, R.; Owen, M.J.; Murray, R.M. The Role of Genes, Stress, and Dopamine in the Development of Schizophrenia. Biol. Psychiatry 2017, 81, 9–20. [Google Scholar] [CrossRef]
- Mccutcheon, R.A.; Abi-Dargham, A.; Howes, O.D. Schizophrenia, Dopamine and the Striatum: From Biology to Symptoms. Trends Neurosci. 2019, 42, 205–220. [Google Scholar] [CrossRef]
- Howes, O.D.; Kapur, S. The Dopamine Hypothesis of Schizophrenia: Version III--The Final Common Pathway. Schizophr. Bull. 2009, 35, 549–562. [Google Scholar] [CrossRef]
- Lau, C.-I.; Wang, H.-C.; Hsu, J.-L.; Liu, M.-E. Does the dopamine hypothesis explain schizophrenia? Rev. Neurosci. 2013, 24, 389–400. [Google Scholar] [CrossRef] [PubMed]
- Zhuo, C.; Hou, W.; Li, G.; Mao, F.; Li, S.; Lin, X.; Jiang, D.; Xu, Y.; Tian, H.; Wang, W.; et al. The genomics of schizophrenia: Shortcomings and solutions. Prog. Neuro Psychopharmacol. Biol. Psychiatry 2019, 93, 71–76. [Google Scholar] [CrossRef] [PubMed]
- Lichtenstein, P.; Yip, B.H.; Björk, C.; Pawitan, Y.; Cannon, T.D.; Sullivan, P.F.; Hultman, C.M. Common genetic determinants of schizophrenia and bipolar disorder in Swedish families: A population-based study. Lancet 2009, 373, 234–239. [Google Scholar] [CrossRef]
- Hoftman, G.D.; Dienel, S.J.; Bazmi, H.H.; Zhang, Y.; Chen, K.; Lewis, D.A. Altered Gradients of Glutamate and Gamma-Aminobutyric Acid Transcripts in the Cortical Visuospatial Working Memory Network in Schizophrenia. Biol. Psychiatry 2018, 83, 670–679. [Google Scholar] [CrossRef]
- Tregellas, J.R.; Wylie, K.P. Alpha7 Nicotinic Receptors as Therapeutic Targets in Schizophrenia. Nicotine Tob. Res. 2019, 21, 349–356. [Google Scholar] [CrossRef]
- Rodríguez-Muñoz, M.; Sánchez-Blázquez, P.; Callado, L.F.; Meana, J.J.; Garzón-Niño, J. Schizophrenia and depression, two poles of endocannabinoid system deregulation. Transl. Psychiatry 2017, 7, 1–12. [Google Scholar] [CrossRef]
- Sawa, A.; Sedlak, T.W. Oxidative stress and inflammation in schizophrenia. Schizophr. Res. 2016, 176, 1–2. [Google Scholar] [CrossRef]
- Mendrek, A.; Mancini-Marïe, A. Sex/gender differences in the brain and cognition in schizophrenia. Neurosci. Biobehav. Rev. 2016, 67, 57–78. [Google Scholar] [CrossRef]
- Gonçalves, V.F.; Cuperfain, A.B.; Kennedy, J.L. Sex differences in schizophrenia: Estrogen and mitochondria. Neuropsychopharmacology 2018, 44, 216–217. [Google Scholar] [CrossRef]
- Mcgregor, C.; Riordan, A.; Thornton, J. Estrogens and the cognitive symptoms of schizophrenia: Possible neuroprotective mechanisms. Front. Neuroendocrinol. 2017, 47, 19–33. [Google Scholar] [CrossRef]
- Rodrigues-Amorim, D.; Rivera-Baltanás, T.; Regueiro, B.; Spuch, C.; Heras, M.E.D.L.; Méndez, R.V.-N.; Nieto-Araujo, M.; Barreiro-Villar, C.; Olivares, J.M.; Agis-Balboa, R.C. The role of the gut microbiota in schizophrenia: Current and future perspectives. World J. Biol. Psychiatry 2018, 19, 571–585. [Google Scholar] [CrossRef] [PubMed]
- Kočovská, E.; Gaughran, F.; Krivoy, A.; Meier, U.-C. Vitamin-D Deficiency as a Potential Environmental Risk Factor in Multiple Sclerosis, Schizophrenia, and Autism. Front. Psychiatry 2017, 8, 47. [Google Scholar] [CrossRef] [PubMed]
- Mayne, P.E.; Burne, T.H. Vitamin D in Synaptic Plasticity, Cognitive Function, and Neuropsychiatric Illness. Trends Neurosci. 2019, 42, 293–306. [Google Scholar] [CrossRef] [PubMed]
- Cirulli, F.; Laviola, G.; Ricceri, L. Risk factors for mental health: Translational models from behavioural neuroscience. Neurosci. Biobehav. Rev. 2009, 33, 493–497. [Google Scholar] [CrossRef] [PubMed]
- Martin, J.; Taylor, M.J.; Lichtenstein, P. Assessing the evidence for shared genetic risks across psychiatric disorders and traits. Psychol. Med. 2017, 48, 1759–1774. [Google Scholar] [CrossRef] [PubMed]
- Baj, J.; Sitarz, E.; Forma, A.; Wróblewska, K.; Karakuła-Juchnowicz, H. Alterations in the Nervous System and Gut Microbiota after β-Hemolytic Streptococcus Group A Infection—Characteristics and Diagnostic Criteria of PANDAS Recognition. Int. J. Mol. Sci. 2020, 21, 1476. [Google Scholar] [CrossRef]
- Altmann, D.M. Neuroimmunology and neuroinflammation in autoimmune, neurodegenerative and psychiatric disease. Immunology 2018, 154, 167–168. [Google Scholar] [CrossRef]
- Owen, L.; Corfe, B. The role of diet and nutrition on mental health and wellbeing. Proc. Nutr. Soc. 2017, 76, 425–426. [Google Scholar] [CrossRef]
- Meltzer-Brody, S.; Howard, L.M.; Bergink, V.; Vigod, S.; Jones, I.; Munk-Olsen, T.; Honikman, S.; Milgrom, J. Postpartum psychiatric disorders. Nat. Rev. Dis. Primers 2018, 4, 1–18. [Google Scholar] [CrossRef]
- Pickard, H.; Fazel, S. Substance abuse as a risk factor for violence in mental illness. Curr. Opin. Psychiatry 2013, 26, 349–354. [Google Scholar] [CrossRef]
- Teicher, M.H.; Samson, J.A.; Anderson, C.M.; Ohashi, K. The effects of childhood maltreatment on brain structure, function and connectivity. Nat. Rev. Neurosci. 2016, 17, 652–666. [Google Scholar] [CrossRef] [PubMed]
- Wood, H. Brain iron correlates with cognitive change in Parkinson disease. Nat. Rev. Neurol. 2020, 16, 184. [Google Scholar] [CrossRef] [PubMed]
- Qian, R.; Ma, Y.; You, L.; Zhao, Y.; Li, S.; Shen, J.; Jiang, L.; Yang, C.; Jiang, P.; Yuan, Z.; et al. The Blood Levels of Trace Elements Are Lower in Children with Tic Disorder: Results from a Retrospective Study. Front. Neurol. 2019, 10, 1324. [Google Scholar] [CrossRef] [PubMed]
- Martínez-Hernanz, Á.; González-Estecha, M.; Blanco, M.; Fuentes, M.; Ordóñez-Iriarte, J.M.; Palazón-Bru, I.; Calvo-Manuel, E.; Bodas-Pindeo, A. Blood lead in children and associations with trace elements and sociodemographic factors. J. Trace Elem. Med. Biol. 2020, 58, 126424. [Google Scholar] [CrossRef] [PubMed]
- Grochowski, C.; Blicharska, E.; Baj, J.; Mierzwińska, A.; Brzozowska, K.; Forma, A.; Maciejewski, R. Serum iron, Magnesium, Copper, and Manganese Levels in Alcoholism: A Systematic Review. Molecules 2019, 24, 1361. [Google Scholar] [CrossRef]
- Grochowski, C.; Blicharska, E.; Bogucki, J.; Proch, J.; Mierzwińska, A.; Baj, J.; Litak, J.; Podkowiński, A.; Flieger, J.; Teresiński, G.; et al. Increased Aluminum Content in Certain Brain Structures is Correlated with Higher Silicon Concentration in Alcoholic Use Disorder. Molecules 2019, 24, 1721. [Google Scholar] [CrossRef]
- Grochowski, C.; Szukała, M.; Litak, J.; Budny, A.; Proch, J.; Majerek, D.; Blicharska, E.; Niedzielski, P. Correlations Between Trace Elements in Selected Locations of the Human Brain in Individuals with Alcohol Use Disorder. Molecules 2020, 25, 359. [Google Scholar] [CrossRef]
- Grochowski, C.; Blicharska, E.; Krukow, P.; Jonak, K.; Maciejewski, M.; Szczepanek, D.; Jonak, K.; Flieger, J.; Maiejewski, R. Analysis of Trace Elements in Human Brain: Its Aim, Methods, and Concentration Levels. Front. Chem. 2019, 7, 115. [Google Scholar] [CrossRef]
- Scassellati, C.; Bonvicini, C.; Benussi, L.; Ghidoni, R.; Squitti, R. Neurodevelopmental disorders: Metallomics studies for the identification of potential biomarkers associated to diagnosis and treatment. J. Trace Elem. Med. Biol. 2020, 60, 126499. [Google Scholar] [CrossRef]
- Abbaspour, N.; Hurrell, R.; Kelishadi, R. Review on iron and its importance for human health. J. Res. Med. Sci. 2014, 19, 164–174. [Google Scholar]
- Zhang, D.-L.; Ghosh, M.C.; Rouault, T.A. The physiological functions of iron regulatory proteins in iron homeostasis—An update. Front. Pharmacol. 2014, 5, 124. [Google Scholar] [CrossRef] [PubMed]
- Murray-Kolb, L.E. Iron and brain functions. Curr. Opin. Clin. Nutr. Metab.Care 2013, 16, 703–707. [Google Scholar] [CrossRef] [PubMed]
- Jáuregui-Lobera, I. Iron deficiency and cognitive functions. Neuropsychiatr. Dis. Treat. 2014, 10, 2087. [Google Scholar] [CrossRef] [PubMed]
- Muñoz, M.; Villar, I.; García-Erce, J.A. An update on iron physiology. World J. Gastroenterol. 2009, 15, 4617. [Google Scholar] [CrossRef] [PubMed]
- Dev, S.; Babitt, J.L. Overview of iron metabolism in health and disease. Hemodial. Int. 2017, 21, S6–S20. [Google Scholar] [CrossRef]
- Cairo, G.; Bernuzzi, F.; Recalcati, S. A precious metal: Iron, an essential nutrient for all cells. Genes Nutr. 2006, 1, 25–39. [Google Scholar] [CrossRef]
- Coughlan, M.P. The role of molybdenum in human biology. J. Inherit. Metab. Dis. 1983, 6, 70–77. [Google Scholar] [CrossRef]
- Mendel, R.R.; Kruse, T. Cell biology of molybdenum in plants and humans. Biochim. Biophys. Acta Mol. Cell Res. 2012, 1823, 1568–1579. [Google Scholar] [CrossRef]
- Schwarz, G. Molybdenum cofactor and human disease. Curr. Opin. Chem. Biol. 2016, 31, 179–187. [Google Scholar] [CrossRef]
- Novotny, J.A.; Peterson, C.A. Molybdenum. Adv Nutr. 2018, 9, 272–273. [Google Scholar] [CrossRef]
- Novotny, J.A. Molybdenum Nutriture in Humans. J. Evid. Based Complementary Altern. Med. 2011, 16, 164–168. [Google Scholar] [CrossRef]
- Bourke, C.A. Molybdenum Deficiency Produces Motor Nervous Effects That Are Consistent with Amyotrophic Lateral Sclerosis. Front. Neurol. 2016, 7, 28. [Google Scholar] [CrossRef] [PubMed]
- Calvo, M.S. Phosphorus. Adv Nutr. 2015, 6, 860–862. [Google Scholar] [CrossRef] [PubMed]
- Takeda, E.; Taketani, Y.; Sawada, N.; Sato, T.; Yamamoto, H. The regulation and function of phosphate in the human body. Biofactors 2004, 21, 345–355. [Google Scholar] [CrossRef]
- Penido, M.G.M.G.; Alon, U.S. Phosphate homeostasis and its role in bone health. Pediatric Nephrol. 2012, 27, 2039–2048. [Google Scholar] [CrossRef]
- Kendrick, J.; Chonchol, M. The Role of Phosphorus in the Development and Progression of Vascular Calcification. Am. J. Kidney Dis. 2011, 58, 826–834. [Google Scholar] [CrossRef]
- Moore, L.W.; Nolte, J.V.; Gaber, A.O.; Suki, W.N. Association of dietary phosphate and serum phosphorus concentration by levels of kidney function. Am. J. Clin Nutr. 2015, 102, 444–453. [Google Scholar] [CrossRef]
- Bacchetta, J.; Salusky, I.B. Evaluation of Hypophosphatemia: Lessons from Patients with Genetic Disorders. Am. J. Kidney Dis. 2012, 59, 152–159. [Google Scholar] [CrossRef]
- Shaman, A.M.; Kowalski, S.R. Hyperphosphatemia Management in Patients with Chronic Kidney Disease. Saudi Pharm. J. 2016, 24, 494–505. [Google Scholar] [CrossRef]
- Kumar, S.; Trivedi, A. A Review on Role of Nickel in the Biological System. Int. J. Curr. Microbiol. Appl. Sci. 2016, 5, 719–727. [Google Scholar] [CrossRef]
- Duda-Chodak, A.; Blaszczyk, U. The Impact of Nickel on human health. J. Elementol. 2008, 13, 685–696. [Google Scholar]
- Zambelli, B.; Ciurli, S. Nickel and human health. Met. Ions Life Sci. 2013, 13, 321–357. [Google Scholar] [PubMed]
- Das, K.K.; Reddy, R.C.; Bagoji, I.B.; Das, S.; Bagali, S.; Mullur, L.; Khodnapur, J.P.; Biradar, M.S. Primary concept of nickel toxicity—An overview. J. Basic Clin. Physiol. Pharmacol. 2018, 30, 141–152. [Google Scholar] [CrossRef] [PubMed]
- Genchi, G.; Carocci, A.; Lauria, G.; Sinicropi, M.S.; Catalano, A. Nickel: Human Health and Environmental Toxicology. Int. J. Environ. Res. Public Health 2020, 17, 679. [Google Scholar] [CrossRef]
- Al Alawi, A.M.; Majoni, S.W.; Falhammar, H. Magnesium and Human Health: Perspectives and Research Directions. Int. J. Endocrinol. 2018, 2018, 9041694. [Google Scholar] [CrossRef]
- Jahnen-Dechent, W.; Ketteler, M. Magnesium basics. Clin. Kideny J. 2012, 5 (Suppl. 1), i3–i14. [Google Scholar] [CrossRef]
- Schwalfenberg, G.K.; Genuis, S.J. The Importance of Magnesium in Clinical Healthcare. Scientifica 2017, 2017, 1–14. [Google Scholar] [CrossRef]
- Gröber, U.; Schmidt, J.; Kisters, K. Magnesium in Prevention and Therapy. Nutrients 2015, 7, 8199–8226. [Google Scholar] [CrossRef]
- Dinicolantonio, J.J.; O’Keefe, J.H.; Wilson, W. Subclinical magnesium deficiency: A principal driver of cardiovascular disease and a public health crisis. Open Heart 2018, 5, e000668. [Google Scholar] [CrossRef]
- Ahmed, F.; Mohammed, A. Magnesium: The Forgotten Electrolyte—A Review on Hypomagnesemia. Med. Sci. 2019, 7, 56. [Google Scholar] [CrossRef]
- Kirkland, A.; Sarlo, G.; Holton, K. The Role of Magnesium in Neurological Disorders. Nutrients 2018, 10, 730. [Google Scholar] [CrossRef] [PubMed]
- Inan-Eroglu, E.; Ayaz, A. Is aluminium exposure a risk factor for neurological disorders? J. Res. Med. Sci. 2018, 23, 51. [Google Scholar] [PubMed]
- Krewski, D.; Yokel, R.A.; Nieboer, E.; Borchelt, E.; Cohen, J.; Harry, J.; Kacew, S.; Lindsay, J.; Mahfouz, A.M.; Rondeau, V. Human health risk assessment for aluminium, aluminium oxide, and aluminium hydroxide. J. Toxicol. Environ. Health B Crit. Rev. 2007, 10 (Suppl. 1), 1–269. [Google Scholar] [CrossRef] [PubMed]
- Akinrinade, I.D.; Memudu, A.E.; Ogundele, O.M. Fluoride and aluminium disturb neuronal morphology, transport functions, cholinesterase, lysosomal and cell cycle activities. Pathophysiology 2015, 22, 105–115. [Google Scholar] [CrossRef] [PubMed]
- Sood, P.K.; Nahar, U.; Nehru, B. Stress Proteins and Glial Cell Functions during Chronic Aluminium Exposures: Protective Role of Curcumin. Neurochem. Res. 2011, 37, 639–646. [Google Scholar] [CrossRef] [PubMed]
- Ivanovski, I.; Ivanovski, A.; Nikolić, D.; Ivanovski, P. Aluminium in brain tissue in autism. J. Trace Elem. Med. Biol. 2019, 51, 138–140. [Google Scholar] [CrossRef]
- Exley, C. The Chemistry of Human Exposure to Aluminium. In Neurotoxicity of Aluminum; Advances in Experimental Medicine and Biology; Niu, Q., Ed.; Springer: Singapore, 2018; Volume 1091, pp. 33–37. [Google Scholar]
- Lukiw, W.J. Evidence supporting a biological role for aluminum in chromatin compaction and epigenetics. J. Inorg. Biochem. 2010, 104, 1010–1012. [Google Scholar] [CrossRef]
- Klotz, K.; Weistenhofer, W.; Neff, F.; Hartwig, A.; van Thriel, C.; Drexler, H. The Health Effects of Aluminium Exposure. Dtsch Arztebl Int. 2017, 114, 653–659. [Google Scholar]
- Gower-Winter, S.D.; Levenson, C.W. Zinc in the central nervous system: From molecules to behavior. BioFactors 2012, 38, 186–193. [Google Scholar] [CrossRef]
- Blakemore, L.J.; Trombley, P.Q. Zinc as a Neuromodulator in the Central Nervous System with a Focus on the Olfactory Bulb. Front. Cell. Neurosci. 2017, 11, 297. [Google Scholar] [CrossRef]
- Frederickson, C.J.; Suh, S.W.; Silva, D.; Frederickson, C.J.; Thompson, R.B. Importance of Zinc in the Central Nervous System: The Zinc-Containing Neuron. J. Nutr. 2000, 130, 1471S–1483S. [Google Scholar] [CrossRef] [PubMed]
- Gammoh, N.Z.; Rink, L. Zinc in Infection and Inflammation. Nutrients 2017, 9, 624. [Google Scholar] [CrossRef] [PubMed]
- Prasad, A.S. Zinc in Human Health: Effect of Zinc on Immune Cells. Mol. Med. 2008, 14, 353–357. [Google Scholar] [CrossRef]
- Collins, J.F. Copper. Adv. Nutr. 2011, 2, 520–522. [Google Scholar] [CrossRef] [PubMed]
- Tapiero, H.; Townsend, D.M.; Tew, K.D. Trace elements in human physiology and pathology. Copp. Biomed. Pharmacother. 2003, 57, 386–398. [Google Scholar] [CrossRef]
- Scheiber, I.F.; Mercer, J.F.; Dringen, R. Metabolism and functions of copper in brain. Prog. Neurobiol. 2014, 116, 33–57. [Google Scholar] [CrossRef] [PubMed]
- Gaetke, L.M.; Chow-Johnson, H.S.; Chow, C.K. Copper: Toxicological relevance and mechanisms. Arch. Toxicol. 2014, 88, 1929–1938. [Google Scholar] [CrossRef]
- Kardos, J.; Héja, L.; Simon, Á.; Jablonkai, I.; Kovács, R.; Jemnitz, K. Copper signalling: Causes and consequences. Cell Commun. Signal. 2018, 16, 1–22. [Google Scholar] [CrossRef]
- Gaier, E.; Eipper, B.; Mains, R. Copper signaling in the mammalian nervous system: Synaptic effects. J. Neurosci. Res. 2012, 91, 2–19. [Google Scholar] [CrossRef]
- Opazo, C.M.; Greenough, M.A.; Bush, A.I. Copper: From neurotransmission to neuroproteostasis. Front. Aging Neurosci. 2014, 6, 143. [Google Scholar] [CrossRef]
- Schlief, M.L.; Gitlin, J.D. Copper Homeostasis in the CNS: A Novel Link Between the NMDA Receptor and Copper Homeostasis in the Hippocampus. Mol. Neurobiol. 2006, 33, 81–90. [Google Scholar] [CrossRef]
- Desai, V.; Kaler, S.G. Role of copper in human neurological disorders. Am. J. Clin. Nutr. 2008, 88, 855S–858S. [Google Scholar] [CrossRef] [PubMed]
- Wazir, S.M.; Ghobrial, I. Copper deficiency, a new triad: Anemia, leucopenia, and myeloneuropathy. J. Community Hosp. Intern. Med. Perspect. 2017, 7, 265–268. [Google Scholar] [CrossRef] [PubMed]
- Tinggi, U. Selenium: Its role as antioxidant in human health. Environ. Health Prev. Med. 2008, 13, 102–108. [Google Scholar] [CrossRef]
- Mehdi, Y.; Hornick, J.-L.; Istasse, L.; Dufrasne, I. Selenium in the Environment, Metabolism and Involvement in Body Functions. Molecules 2013, 18, 3292–3311. [Google Scholar] [CrossRef]
- Kurokawa, S.; Berry, M.J. Selenium. Role of the Essential Metalloid in Health. In Metal Ions in Life Sciences Interrelations between Essential Metal Ions and Human Diseases; Springer: Dordrecht, The Netherlands, 2013; pp. 499–534. [Google Scholar]
- Rederstorff, M.; Krol, A.; Lescure, A. Understanding the importance of selenium and selenoproteins in muscle function. Cell. Mol. Life Sci. 2005, 63, 52–59. [Google Scholar] [CrossRef]
- Rayman, M.P. The importance of selenium to human health. Lancet 2000, 356, 233–241. [Google Scholar] [CrossRef]
- Solovyev, N.D. Importance of selenium and selenoprotein for brain function: From antioxidant protection to neuronal signalling. J. Inorg. Biochem. 2015, 153, 1–12. [Google Scholar] [CrossRef]
- Cormick, G.; Belizán, J.M. Calcium Intake and Health. Nutrients 2019, 11, 1606. [Google Scholar] [CrossRef]
- Vannucci, L.; Fossi, C.; Quattrini, S.; Guasti, L.; Pampaloni, B.; Gronchi, G.; Giusti, F.; Romagnoli, C.; Cianferotti, L.; Marcucci, G.; et al. Calcium Intake in Bone Health: A Focus on Calcium-Rich Mineral Waters. Nutrients 2018, 10, 1930. [Google Scholar] [CrossRef]
- Young, W. Role of calcium in central nervous system injuries. J. Neurotrauma 1992, 9 (Suppl. 1), S9–S25. [Google Scholar] [PubMed]
- Braet, K.; Cabooter, L.; Paemeleire, K.; Leybaert, L. Calcium signal communication in the central nervous system. Biol. Cell 2004, 96, 79–91. [Google Scholar] [CrossRef] [PubMed]
- Nedergaard, M.; Rodríguez, J.J.; Verkhratsky, A. Glial calcium and diseases of the nervous system. Cell Calcium 2010, 47, 140–149. [Google Scholar] [CrossRef] [PubMed]
- Mata, A.M.; Sepulveda, M.R. Calcium pumps in the central nervous system. Brain Res. Brain Res. Rev. 2005, 49, 398–405. [Google Scholar] [CrossRef]
- Nicholls, D.G. Mitochondrial calcium function and dysfunction in the central nervous system. Biochim. Biophys. Acta Bioenerg. 2009, 1787, 1416–1424. [Google Scholar] [CrossRef]
- Ruat, M.; Traiffort, E. Roles of the calcium sensing receptor in the central nervous system. Best Pract. Res. Clin. Endocrinol. Metab. 2013, 27, 429–442. [Google Scholar] [CrossRef]
- Andersen, T.L.; Sondergaard, T.E.; Skorzynska, K.E.; Dagnaes-Hansen, F.; Plesner, T.L.; Hauge, E.M.; Plesner, T.; Delaisse, J.-M. A Physical Mechanism for Coupling Bone Resorption and Formation in Adult Human Bone. Am. J. Pathol. 2009, 174, 239–247. [Google Scholar] [CrossRef]
- Aschner, J.L.; Aschner, M. Nutritional aspects of manganese homeostasis. Mol. Asp. Med. 2005, 26, 353–362. [Google Scholar] [CrossRef]
- Chen, P.; Bornhorst, J.; Aschner, M. Manganese metabolism in humans. Front. Biosci. 2018, 123, 1655–1679. [Google Scholar] [CrossRef]
- Horning, K.J.; Caito, S.W.; Tipps, K.G.; Bowman, A.B.; Aschner, M. Manganese Is Essential for Neuronal Health. Annu. Rev. Nutr. 2015, 35, 71–108. [Google Scholar] [CrossRef]
- Bowman, A.B.; Kwakye, G.F.; Hernandez, E.H.; Aschner, M. Role of manganese in neurodegenerative diseases. J. Trace Elem. Med. Biol. 2011, 25, 191–203. [Google Scholar] [CrossRef] [PubMed]
- Sidoryk-Wegrzynowicz, M.; Aschner, M. Manganese toxicity in the central nervous system: The glutamine/glutamate-γ-aminobutyric acid cycle. J. Intern. Med. 2013, 273, 466–477. [Google Scholar] [CrossRef] [PubMed]
- Rivera-Mancía, S.; Ríos, C.; Montes, S. Manganese accumulation in the CNS and associated pathologies. BioMetals 2011, 24, 811–825. [Google Scholar] [CrossRef] [PubMed]
- Chen, P.; Chakraborty, S.; Mukhopadhyay, S.; Lee, E.; Paoliello, M.M.B.; Bowman, A.B.; Aschner, M. Manganese Homeostasis in the Nervous System. J. Neurochem. 2015, 134, 601–610. [Google Scholar] [CrossRef] [PubMed]
- Won, E.; Kim, Y.K. An Oldie but Goodie: Lithium in the Treatment of Bipolar Disorder through Neuroprotective and Neurotrophic Mechanisms. Int. J. Mol. Sci. 2017, 18, 2679. [Google Scholar] [CrossRef]
- Demling, J.; Eglau, M.; Autenrieth, T. On the physiological function oflithium from a psychiatric view point. Med. Hypotheses 2001, 57, 506–509. [Google Scholar] [CrossRef]
- Malhi, G.S.; Outhred, T. Therapeutic Mechanisms of Lithium in Bipolar Disorder: Recent Advances and Current Understanding. CNS Drugs 2016, 30, 931–949. [Google Scholar] [CrossRef]
- Eker, C.; Simsek, F.; Yılmazer, E.E.; Kitis, O.; Cinar, C.; Eker, O.D.; Coburn, K.; Gonul, A.S. Brain regions associated with risk and resistance for bipolar I disorder: A voxel-based MRI study of patients with bipolar disorder and their healthy siblings. Bipolar Disord. 2014, 16, 249–261. [Google Scholar] [CrossRef]
- Wijeratne, C.; Sachdev, S.; Wen, W.; Piguet, O.; Lipnicki, D.M.; Malhi, G.S.; Mitchell, P.B.; Sachdev, P.S. Hippocampal and amygdala volumes in an older bipolar disorder sample. Int. Psychogeriatr. 2012, 25, 54–60. [Google Scholar] [CrossRef]
- Chen, X.; Wen, W.; Malhi, G.S.; Ivanovski, B.; Sachdev, P.S. Regional Gray Matter Changes in Bipolar Disorder: A Voxel-Based Morphometric Study. Aust. N. Z. J. Psychiatry 2007, 41, 327–336. [Google Scholar] [CrossRef]
- Sousa, R.T.D.; Streck, E.L.; Zanetti, M.V.; Ferreira, G.K.; Diniz, B.S.; Brunoni, A.R.; Busatto, G.F.; Gattaz, W.F.; Machado-Vieira, R. Lithium increases leukocyte mitochondrial complex I activity in bipolar disorder during depressive episodes. Psychopharmacology 2014, 232, 245–250. [Google Scholar] [CrossRef] [PubMed]
- Macêdo, D.S.; Lucena, D.F.D.; Queiroz, A.I.G.; Cordeiro, R.C.; Araújo, M.M.; Sousa, F.C.; Vasconcelos, S.M.; Hyphantis, T.N.; Quevedo, J.; McIntyre, R.S.; et al. Effects of lithium on oxidative stress and behavioral alterations induced by lisdexamfetamine dimesylate: Relevance as an animal model of mania. Prog. Neuro Psychopharmacol. Biol. Psychiatry 2013, 43, 230–237. [Google Scholar] [CrossRef] [PubMed]
- Nielsen, F.H. Other elements: Sb, Ba, B, Br, Cs, Ge, Rb, Ag, Sr, Sn, Ti, Zr, Be, Bi, Ga, Au, In, Nb, Sc, Te, Tl, W. In Trace Elements in Human and Animal Nutrition; Mertz, W., Ed.; Academic Press: San Diego, CA, USA, 2012; Volume 2, pp. 415–463. [Google Scholar]
- Paschalis, C.; Jenner, F.A.; Lee, C.R. Effects of rubidium chloride on the course of manic-depressive illness. J. R Soc. Med. 1978, 71, 343–352. [Google Scholar] [CrossRef] [PubMed]
- Meltzer, H.L. A pharmacokinetic analysis of long-term administration of rubidium chloride. J. Clin. Pharmacol. 1991, 31, 179–184. [Google Scholar] [CrossRef]
- Mehri, A. Trace Elements in Human Nutrition (II)—An Update. Int. J. Prev. Med. 2020, 11, 2. [Google Scholar]
- Sun, Y.; Byon, C.H.; Yang, Y.; Bradley, W.E.; Dell’Italia, L.J.; Sanders, P.W.; Agarwal, A.; Wu, H.; Chen, Y. Dietary potassium regulates vascular calcification and arterial stiffness. JCI Insight 2017, 2, e94920. [Google Scholar] [CrossRef]
- D’Elia, L.; Barba, G.; Cappuccio, F.; Strazzullo, P. Potassium Intake, Stroke, and Cardiovascular Disease: A Meta-Analysis of Prospective Studies. J. Am. Coll. Cardiol. 2011, 57, 1210–1219. [Google Scholar] [CrossRef]
- Lanham-New, S.A.; Lambert, H.; Frassetto, L. Potassium. Adv. Nutr. 2012, 3, 820–821. [Google Scholar] [CrossRef]
- Yue, Y.C.; Li, M.H.; Wang, H.B.; Zhang, B.L.; He, W. The toxicological mechanisms and detoxification of depleted uranium exposure. Environ. Health Prev. Med. 2018, 23, 18. [Google Scholar] [CrossRef]
- Hao, Y.; Li, R.; Leng, Y.; Ren, J.; Liu, J.; Ai, G.; Xu, H.; Su, Y.; Cheng, T. A study assessing the genotoxicity in rats after chronic oral exposure to a low dose of depleted uranium. J. Radiat. Res. 2009, 50, 521–528. [Google Scholar] [CrossRef]
- Carmona, A.; Malard, V.; Avazeri, E.; Roudeau, S.; Porcaro, F.; Paredes, E.; Vidaud, C.; Bresson, C.; Ortega, R. Uranium exposure of human dopaminergic cells results in low cytotoxicity, accumulation within sub-cytoplasmic regions, and down regulation of MAO-B. Neurotoxicology 2018, 68, 177–188. [Google Scholar] [CrossRef] [PubMed]
- Briner, W. The toxicity of depleted uranium. Int J. Environ. Res. Public Health 2010, 7, 303–313. [Google Scholar] [CrossRef] [PubMed]
- Dublineau, I.; Grandcolas, L.; Grison, S.; Baudelin, C.; Paquet, F.; Voisin, P.; Aigueperse, J.; Gourmelon, P. Modifications of inflammatory pathways in rat intestine following chronic ingestion of depleted uranium. Toxicol. Sci. 2007, 98, 458–468. [Google Scholar] [CrossRef] [PubMed]
- Bertin, G.; Averbeck, D. Cadmium: Cellular effects, modifications of biomolecules, modulation of DNA repair and genotoxic consequences (a review). Biochimie 2006, 88, 1549–1559. [Google Scholar] [CrossRef]
- Waalkes, M.P. Cadmium carcinogenesis. Mutat. Res. 2003, 533, 107–120. [Google Scholar] [CrossRef]
- Satarug, S.; Baker, J.R.; Reilly, P.E.; Moore, M.R.; Williams, D.J. Cadmium levels in the lung, liver, kidney cortex, and urine samples from Australians without occupational exposure to metals. Arch. Environ. Health 2002, 57, 69–77. [Google Scholar] [CrossRef]
- Uetani, M.; Kobayashi, E.; Suwazono, Y.; Honda, R.; Nishijo, M.; Nakagawa, H.; Kido, T.; Nogawa, K. Tissue cadmium (Cd) concentrations of people living in a Cd polluted area, Japan. Biometals 2006, 19, 521–525. [Google Scholar] [CrossRef]
- Wills, N.K.; Ramanujam, V.M.; Chang, J.; Kalariya, N.; Lewis, J.R.; Weng, T.X.; van Kuijk, F.J. Cadmium accumulation in the human retina: Effects of age, gender, and cellular toxicity. Exp. Eye Res. 2008, 86, 41–51. [Google Scholar] [CrossRef]
- Satarug, S.; Garrett, S.H.; Sens, M.A.; Sens, D.A. Cadmium, environmental exposure, and health outcomes. Environ. Health Perspect 2010, 118, 182–190. [Google Scholar] [CrossRef]
- Rodríguez-Barranco, M.; Lacasaña, M.; Gil, F.; Lorca, A.; Alguacil, J.; Rohlman, D.S.; González-Alzaga, B.; Molina-Villalba, I.; Mendoza, R.; Aguilar-Garduño, C. Cadmium exposure and neuropsychological development in school children in southwestern Spain. Environ. Res. 2014, 134, 66–73. [Google Scholar] [CrossRef]
- Saghazadeh, A.; Ahangari, N.; Hendi, K.; Saleh, F.; Rezaei, N. Status of essential elements in autism spectrum disorder: Systematic review and meta-analysis. Rev. Neurosci. 2017, 28, 783–809. [Google Scholar] [CrossRef] [PubMed]
- Ayton, S.; Lei, P.; Bush, A.I. Biometals and Their Therapeutic Implications in Alzheimer’s Disease. Neurotherapeutics 2014, 12, 109–120. [Google Scholar] [CrossRef] [PubMed]
- Kim, M.-J.; Oh, S.-B.; Kim, J.; Kim, K.; Ryu, H.-S.; Kim, M.S.; Ayton, S.; Bush, A.I.; Lee, J.-Y.; Chung, S.J. Association of metals with the risk and clinical characteristics of Parkinsons disease. Parkinsonism Relat. Disord. 2018, 55, 117–121. [Google Scholar] [CrossRef] [PubMed]
- Dominguez, J.F.D.; Ng, A.C.L.; Poudel, G.; Stout, J.C.; Churchyard, A.; Chua, P.; Egan, G.F.; Georgiou-Karistianis, N. Iron accumulation in the basal ganglia in Huntingtons disease: Cross-sectional data from the IMAGE-HD study. J. Neurol. Neurosurg. Psychiatry 2015, 87, 545–549. [Google Scholar] [CrossRef]
- Bartnicka, J.J.; Blower, P.J. Insights into Trace Metal Metabolism in Health and Disease From PET: “PET Metallomics”. J. Nucl. Med. 2018, 59, 1355–1359. [Google Scholar] [CrossRef]
- Wang, J.; Um, P.; Dickerman, B.A.; Liu, J. Zinc, Magnesium, Selenium and Depression: A Review of the Evidence, Potential Mechanisms and Implications. Nutrients 2018, 10, 584. [Google Scholar] [CrossRef]
- Tseng, P.-T.; Cheng, Y.-S.; Yen, C.-F.; Chen, Y.-W.; Stubbs, B.; Whiteley, P.; Carvalho, A.F.; Li, D.-J.; Chen, T.-Y.; Yang, W.-C.; et al. Peripheral Iron Levels in Children with Attention-Deficit Hyperactivity Disorder: A Systematic Review and Meta-Analysis. Sci. Rep. 2018, 8, 788. [Google Scholar] [CrossRef]
- Royal, B. Schizophrenia: Nutrition and Alternative Treatment Approaches. Schizophr. Bull. 2015, 42, 1083–1085. [Google Scholar] [CrossRef]
- Velthorst, E.; Smith, L.; Bello, G.; Austin, C.; Gennings, C.; Modabbernia, A.; Franke, N.; Frangou, S.; Wright, R.; de Haan, L.; et al. New Research Strategy for Measuring Pre- and Postnatal Metal Dysregulation in Psychotic Disorders. Schizophr. Bull. 2017, 43, 1153–1157. [Google Scholar] [CrossRef]
- Liu, T.; Lu, Q.-B.; Yan, L.; Guo, J.; Feng, F.; Qiu, J.; Wang, J. Comparative Study on Serum Levels of 10 Trace Elements in Schizophrenia. PLoS ONE 2015, 10, e0133622. [Google Scholar] [CrossRef]
- Cruz, E.C.S.; Madrid, K.C.; Arruda, M.A.; Sussulini, A. Association between trace elements in serum from bipolar disorder and schizophrenia patients considering treatment effects. J. Trace Elem. Med. Biol. 2020, 59, 126467. [Google Scholar] [CrossRef] [PubMed]
- Herrán, A.; García-Unzueta, M.T.; Fernández-González, M.D.; Vázquez-Barquero, J.L.; Alvarez, C.; Amado, J.A. Higher levels of serum copper in schizophrenic patients treated with depot neuroleptics. Psychiatry Res. 2000, 94, 51–58. [Google Scholar] [CrossRef]
- Cao, B.; Yan, L.; Ma, J.; Jin, M.; Park, C.; Nozari, Y.; Kazmierczak, O.P.; Zukerman, H.; Lee, Y.; Pan, Z.; et al. Comparison of serum essential trace metals between patients with schizophrenia and healthy controls. J. Trace Elem. Med. Biol. 2019, 51, 79–85. [Google Scholar] [CrossRef] [PubMed]
- Chen, X.; Li, Y.; Zhang, T.; Yao, Y.; Shen, C.; Xue, Y. Association of Serum Trace Elements with Schizophrenia and Effects of Antipsychotic Treatment. Biol. Trace Elem. Res. 2017, 181, 22–30. [Google Scholar] [CrossRef] [PubMed]
- Devanarayanan, S.; Nandeesha, H.; Kattimani, S.; Sarkar, S.; Jose, J. Elevated copper, hs C-reactive protein and dyslipidemia in drug free schizophrenia: Relation with psychopathology score. Asian J. Psychiatry 2016, 24, 99–102. [Google Scholar] [CrossRef]
- Edelstein, E.L.; Krassilowsky, D.; Gdalia, J. A Study on Serum Copper Level in 20 Acute Schizophrenics. J. Ment. Sci. 1959, 105, 511–513. [Google Scholar] [CrossRef]
- Frohman, C.E. Ceruloplasmin, Transferrin, and Tryptophan in Schizophrenia. Arch. Neurol. Psychiatry 1958, 79, 730. [Google Scholar] [CrossRef]
- Li, Z.; Liu, Y.; Li, X.; Ju, W.; Wu, G.; Yang, X.; Fu, X.; Gao, X. Association of Elements with Schizophrenia and Intervention of Selenium Supplements. Biol. Trace Elem. Res. 2017, 183, 16–21. [Google Scholar] [CrossRef]
- Maas, J.W.; Gleser, G.C.; Gottschalk, L.A. Schizophrenia, Anxiety, and Biochemical Factors: The Rate of Oxidation of N,N-Dimethyl-p-Phenylenediamine by Plasma and Levels of Serum Copper and Plasma Ascorbic Acid. Arch. Gen. Psychiatry 1961, 4, 109–118. [Google Scholar] [CrossRef]
- Munch-Petersen, S. On Serum Copper in Patients with Schizophrenia. Acta Psychiatr. Scand. 1950, 25, 423–427. [Google Scholar] [CrossRef]
- Olatunbosun, D.A.; Akindele, M.O.; Adadevoh, B.K.; Asuni, T. Serum Copper in Schizophrenia in Nigerians. Br. J. Psychiatry 1975, 127, 119–121. [Google Scholar] [CrossRef] [PubMed]
- Wolf, T.L.; Kotun, J.; Meador-Woodruff, J.H. Plasma copper, iron, ceruloplasmin and ferroxidase activity in schizophrenia. Schizophr. Res. 2006, 86, 167–171. [Google Scholar] [CrossRef] [PubMed]
- Wasti, A.Z.; Zahid, S.; Ahmed, N. Antipsychotic Drugs Induced Iron Deficiency Anemia in Schizophrenic Patients. Int. J. Adv. Res. 2013, 1, 111–118. [Google Scholar]
- Barnes, T.R.E.; Halstead, S.M.; Little, P.W.A. Relationship Between Iron Status and Chronic Akathisia in an In-patient Population with Chronic Schizophrenia. Br. J. Psychiatry 1992, 161, 791–796. [Google Scholar] [CrossRef]
- Hofmann, M.; Seifritz, E.; Botschev, C.; Kräuchi, K.; Müller-Spahn, F. Serum iron and ferritin in acute neuroleptic akathisia. Psychiatry Res. 2000, 93, 201–207. [Google Scholar] [CrossRef]
- Kim, S.-W.; Stewart, R.; Park, W.-Y.; Jhon, M.; Lee, J.-Y.; Kim, S.-Y.; Kim, J.-M.; Amminger, P.; Chung, Y.-C.; Yoon, J.-S. Latent Iron Deficiency as a Marker of Negative Symptoms in Patients with First-Episode Schizophrenia Spectrum Disorder. Nutrients 2018, 10, 1707. [Google Scholar] [CrossRef]
- Kuloglu, M.; Atmaca, M.; Üstündag, B.; Canatan, H.; Gecici, O.; Tezcan, E. Serum iron levels in schizophrenic patients with or without akathisia. Eur. Neuropsychopharmacol. 2003, 13, 67–71. [Google Scholar] [CrossRef]
- Soni, S.D.; Tench, D.; Routledge, R.C. Serum Iron Abnormalities in Neuroleptic-Induced Akathisia in Schizophrenic Patients. Br. J. Psychiatry 1993, 163, 669–672. [Google Scholar] [CrossRef]
- Spina, E.; Ancione, M.; Rosa, A.E.D.; Artemisia, A.; Natoli, C.; Caputi, A.P. Iron status in schizophrenic patients with acute neuroleptic-induced dystonic reactions. Prog. Neuro Psychopharmacol. Biol. Psychiatry 1994, 18, 891–898. [Google Scholar] [CrossRef]
- Wirshing, D.A.; Bartzokis, G.; Pierre, J.M.; Wirshing, W.C.; Sun, A.; Tishler, T.A.; Marder, S.R. Tardive dyskinesia and serum iron indices. Biol. Psychiatry 1998, 44, 493–498. [Google Scholar] [CrossRef]
- Wiser, M.; Levkowitch, Y.; Neuman, M.; Yehuda, S. Decrease of Serum Iron in Acutely Psychotic Schizophrenic Patients. Int. J. Neurosci. 1994, 78, 49–52. [Google Scholar] [CrossRef] [PubMed]
- Alexander, P.E.; Kammen, D.P.V.; Bunney, W.E. Serum Calcium and Magnesium in Schizophrenia: Relationship to Clinical Phenomena and Neuroleptic Treatment. Br. J. Psychiatry 1978, 133, 143–149. [Google Scholar] [CrossRef] [PubMed]
- Alexander, P.E. Serum Calcium and Magnesium Levels in Schizophrenia. Arch. Gen. Psychiatry 1979, 36, 1372. [Google Scholar] [CrossRef] [PubMed]
- Athanassenas, G.; Papadopolos, E.; Kourkoubas, A.; Tsitourides, S.; Gabriel, J.; Hoidas, S.; Frangos, E. Serum Calcium and Magnesium Levels in Chronic Schizophrenics. J. Clin. Psychopharmacol. 1983, 3, 212–216. [Google Scholar] [CrossRef] [PubMed]
- Levine, J.; Rapoport, A.; Mashiah, M.; Dolev, E. Serum and Cerebrospinal Levels of Calcium and Magnesium in Acute versus Remitted Schizophrenic Patients. Neuropsychobiology 1996, 33, 169–172. [Google Scholar] [CrossRef]
- Alertsen, A.R.; Aukrust, A.; Skaug, O.E. Selenium concentrations in blood and serum from patients with mental diseases. Acta Psychiatr. Scand. 1986, 74, 217–219. [Google Scholar] [CrossRef]
- Ruljancic, N.; Mihanovic, M.; Cepelak, I.; Bakliza, A. Platelet and serum calcium and magnesium concentration in suicidal and non-suicidal schizophrenic patients. Psychiatry Clin. Neurosci. 2013, 67, 154–159. [Google Scholar] [CrossRef]
- Jamilian, H.; Bagherzadeh, K.; Nazeri, Z.; Hassanijirdehi, M. Vitamin D, parathyroid hormone, serum calcium and phosphorus in patients with schizophrenia and major depression. Int. J. Psychiatry Clin. Pract. 2012, 17, 30–34. [Google Scholar] [CrossRef]
- Lin, T.; Liu, T.; Lin, Y.; Yan, L.; Chen, Z.; Wang, J. Comparative study on serum levels of macro and trace elements in schizophrenia based on supervides learning methods. J. Trace Elem. Med. Biol. 2017, 43, 202–208. [Google Scholar] [CrossRef]
- Cai, L.; Chen, T.; Yang, J.; Zhou, K.; Yan, X.; Chen, W.; Sun, L.; Li, L.; Qin, S.; Wang, P.; et al. Serum trace element differences between Schizophrenia patients and controls in the Han Chinese population. Sci. Rep. 2015, 5, 15013. [Google Scholar] [CrossRef]
- Ma, J.; Wang, B.; Gao, X.; Wu, H.; Wang, D.; Li, N.; Tan, J.; Wang, J.; Yan, L. A comparative study of the typical toxic metals in serum by patients of schizophrenia and healthy controls in China. Psychiatry Res. 2018, 269, 558–564. [Google Scholar] [CrossRef] [PubMed]
- Ma, J.; Yan, L.; Guo, T.; Yang, S.; Guo, C.; Liu, Y.; Xie, Q.; Wang, J. Association of Typical Toxic Heavy Metals with Schizophrenia. Int. J. Environ. Res. Public Health 2019, 16, 4200. [Google Scholar] [CrossRef] [PubMed]
- Gambling, L.; Kennedy, C.; Mcardle, H.J. Iron and copper in fetal development. Semin. Cell Dev. Biol. 2011, 22, 637–644. [Google Scholar] [CrossRef] [PubMed]
- Piñero, D.J.; Connor, J.R. Iron in the Brain: An Important Contributor in Normal and Diseased States. Neuroscientist 2000, 6, 435–453. [Google Scholar] [CrossRef]
- Moreno-Fernandez, J.; Ochoa, J.J.; Latunde-Dada, G.O.; Diaz-Castro, J. Iron Deficiency and Iron Homeostasis in Low Birth Weight Preterm Infants: A Systematic Review. Nutrients 2019, 11, 1090. [Google Scholar] [CrossRef]
- Wessling-Resnick, M. Excess iron: Considerations related to development and early growth. Am. J. Clin. Nutr. 2017, 10 (Suppl. 6), 1600S–1605S. [Google Scholar] [CrossRef]
- Hare, D.; Ayton, S.; Bush, A.; Lei, P. A Delicate Balance: Iron Metabolism and Diseases of the Brain. Front. Aging Neurosci. 2013, 5. [Google Scholar] [CrossRef]
- Lamtai, M.; Chaibat, J.; Ouakki, S.; Zghari, O.; Mesfioui, A.; Hessni, A.E.; Rifi, E.-H.; Marmouzi, I.; Essamri, A.; Ouichou, A. Effect of Chronic Administration of Nickel on Affective and Cognitive Behavior in Male and Female Rats: Possible Implication of Oxidative Stress Pathway. Brain Sci. 2018, 8, 141. [Google Scholar] [CrossRef]
- Todorich, B.; Pasquini, J.M.; Garcia, C.I.; Paez, P.M.; Connor, J.R. Oligodendrocytes and myelination: The role of iron. Glia 2009, 57, 467–478. [Google Scholar] [CrossRef]
- Hare, D.J.; Double, K.L. Iron and dopamine: A toxic couple. Brain 2016, 139, 1026–1035. [Google Scholar] [CrossRef]
- Felt, B.T.; Beard, J.L.; Schallert, T.; Shao, J.; Aldridge, J.W.; Connor, J.R.; Georgieff, M.K.; Lozoff, B. Persistent neurochemical and behavioral abnormalities in adulthood despite early iron supplementation for perinatal iron deficiency anemia in rats. Behav. Brain Res. 2006, 171, 261–270. [Google Scholar] [CrossRef] [PubMed]
- Beard, J.L.; Felt, B.; Schallert, T.; Burhans, M.; Connor, J.R.; Georgieff, M.K. Moderate iron deficiency in infancy: Biology and behavior in young rats. Behav. Brain Res. 2006, 170, 224–232. [Google Scholar] [CrossRef]
- Pisansky, M.T.; Wickham, R.J.; Su, J.; Fretham, S.; Yuan, L.-L.; Sun, M.; Gewirtz, J.C.; Georgieff, M.K. Iron deficiency with or without anemia impairs prepulse inhibition of the startle reflex. Hippocampus 2013, 23, 952–962. [Google Scholar] [CrossRef] [PubMed]
- Iglesias, L.; Canals, J.; Arija, V. Effects of prenatal iron status on child neurodevelopment and behavior: A systematic review. Crit. Rev. Food Sci. Nutr. 2017, 58, 1604–1614. [Google Scholar] [CrossRef] [PubMed]
- Lozoff, B. Early Iron Deficiency Has Brain and Behavior Effects Consistent with Dopaminergic Dysfunction. J. Nutr. 2011, 141, 740S–746S. [Google Scholar] [CrossRef] [PubMed]
- East, P.; Delker, E.; Lozoff, B.; Delva, J.; Castillo, M.; Gahagan, S. Associations among Infant Iron Deficiency, Childhood Emotion and Attention Regulation, and Adolescent Problem Behaviors. Child. Dev. 2017, 89, 593–608. [Google Scholar] [CrossRef]
- Wenger, M.J.; Dellavalle, D.M.; Murray-Kolb, L.E.; Haas, J.D. Effect of iron deficiency on simultaneous measures of behavior, brain activity, and energy expenditure in the performance of a cognitive task. Nutr. Neurosci. 2017, 22, 196–206. [Google Scholar] [CrossRef]
- Ferreira, A.C.; Sousa, N.; Bessa, J.M.; Sousa, J.C.; Marques, F. Metabolism and adult neurogenesis: Towards an understanding of the role of lipocalin-2 and iron-related oxidative stress. Neurosci. Biobehav. Rev. 2018, 95, 73–84. [Google Scholar] [CrossRef]
- Imam, M.; Zhang, S.; Ma, J.; Wang, H.; Wang, F. Antioxidants Mediate Both Iron Homeostasis and Oxidative Stress. Nutrients 2017, 9, 671. [Google Scholar] [CrossRef]
- Ng, F.; Berk, M.; Dean, O.; Bush, A.I. Oxidative stress in psychiatric disorders: Evidence base and therapeutic implications. Int. J. Neuropsychopharmacol. 2008, 11, 851–876. [Google Scholar] [CrossRef]
- Smythies, J. The neurotoxicity of glutamate, dopamine, iron and reactive oxygen species: Functional interrelationships in health and disease: A review—Discussion. Neurotox. Res. 1999, 1, 27–39. [Google Scholar] [CrossRef] [PubMed]
- Andersen, H.H.; Johnsen, K.B.; Moos, T. Iron deposits in the chronically inflamed central nervous system and contributes to neurodegeneration. Cell. Mol. Life Sci. 2013, 71, 1607–1622. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Wu, Y.; Li, T.; Wang, X.; Zhu, C. Iron Metabolism and Brain Development in Premature Infants. Front. Physiol. 2019, 10, 463. [Google Scholar] [CrossRef]
- Kim, J.; Wessling-Resnick, M. Iron and mechanisms of emotional behavior. J. Nutr. Biochem. 2014, 25, 1101–1107. [Google Scholar] [CrossRef] [PubMed]
- Yanik, M.; Kocyigit, A.; Tutkun, H.; Vural, H.; Herken, H. Plasma manganese, selenium, zinc, copper, and iron concentrations in patients with schizophrenia. Biol. Trace Elem. Res. 2004, 98, 109–117. [Google Scholar] [CrossRef]
- Casanova, M.F.; Waldman, I.N.; Kleinman, J.E. A postmortem quantitative study of iron in the globus pallidus of schizophrenic patients. Biol. Psychiatry 1990, 27, 143–149. [Google Scholar] [CrossRef]
- Casanova, M.F.; Prasad, C.M.; Waldman, I.; Illowsky Karp, B.; Stein, B.; Weinberger, D.R.; Kleinman, J.B. No difference in basal ganglia mineralization between schizophrenic and nonschizophrenic patients: A quantitative computerized tomographic study. Biol. Psychiatry 1990, 27, 138–142. [Google Scholar] [CrossRef]
- Sachdev, P. The neuropsychiatry of brain iron. J. Neuropsychiatry Clin. Neurosci. 1993, 5, 18–29. [Google Scholar]
- Kornhuber, J.; Lange, K.W.; Kruzik, P.; Rausch, W.-D.; Gabriel, E.; Jellinger, K.; Riederer, P. Iron, copper, zinc, magnesium, and calcium in postmortem brain tissue from schizophrenic patients. Biol. Psychiatry 1994, 36, 31–34. [Google Scholar] [CrossRef]
- Casanova, M.F.; Comparini, S.O.; Kim, R.W.; Kleinman, J.E. Staining intensity of brain iron in patients with schizophrenia: A postmortem study. J. Neuropsychiatry Clin. Neurosci. 1992, 4, 36–41. [Google Scholar]
- Demmel, U.; Höck, A.; Feinendegen, L.; Sebek, P. Trace elements in brains of patients with alcohol abuse, endogeneous psychosis and schizophrenia. Sci. Total Environ. 1984, 38, 69–77. [Google Scholar] [CrossRef]
- Insel, B.J.; Schaefer, C.A.; Mckeague, I.W.; Susser, E.S.; Brown, A.S. Maternal Iron Deficiency and the Risk of Schizophrenia in Offspring. Arch. Gen. Psychiatry 2008, 65, 1136. [Google Scholar] [CrossRef] [PubMed]
- Tran, P.V.; Kennedy, B.C.; Pisansky, M.T.; Won, K.-J.; Gewirtz, J.C.; Simmons, R.A.; Georgieff, M.K. Prenatal Choline Supplementation Diminishes Early-Life Iron Deficiency–Induced Reprogramming of Molecular Networks Associated with Behavioral Abnormalities in the Adult Rat Hippocampus. J. Nutr. 2016, 146, 484–493. [Google Scholar] [CrossRef] [PubMed]
- Mireku, M.O.; Davidson, L.L.; Boivin, M.J.; Zoumenou, R.; Massougbodji, A.; Cot, M.; Bodeau-Livinec, F. Prenatal Iron Deficiency, Neonatal Ferritin, and Infant Cognitive Function. Pediatrics 2016, 138, e20161319. [Google Scholar] [CrossRef] [PubMed]
- Lozoff, B.; Beard, J.; Connor, J.; Barbara, F.; Georgieff, M.; Schallert, T. Longlasting neural and behavioral effects of iron deficiency in infancy. Nutr. Rev. 2006, 64, S34–S43. [Google Scholar] [CrossRef] [PubMed]
- Sorensen, H.J.; Nielsen, P.R.; Pedersen, C.B.; Mortensen, P.B. Association Between Prepartum Maternal Iron Deficiency and Offspring Risk of Schizophrenia: Population-Based Cohort Study with Linkage of Danish National Registers. Schizophr. Bull. 2010, 37, 982–987. [Google Scholar] [CrossRef] [PubMed]
- Wiegersma, A.M.; Dalman, C.; Lee, B.K.; Karlsson, H.; Gardner, R.M. Association of Prenatal Maternal Anemia with Neurodevelopmental Disorders. JAMA Psychiatry 2019, 76, 1294. [Google Scholar] [CrossRef]
- Berglund, S.K.; Torres-Espínola, F.J.; García-Valdés, L.; Segura, M.T.; Martínez-Zaldívar, C.; Padilla, C.; Rueda, R.; Garcia, M.P.; McArdle, H.J.; Campoy, C. The impacts of maternal iron deficiency and being overweight during pregnancy on neurodevelopment of the offspring. Br. J. Nutr. 2017, 118, 533–540. [Google Scholar] [CrossRef]
- Sussulini, A.; Erbolato, H.M.; Pessôa, G.D.S.; Arruda, M.A.Z.; Steiner, J.; Martins-De-Souza, D. Elemental fingerprinting of schizophrenia patient blood plasma before and after treatment with antipsychotics. Eur. Arch. Psychiatry Clin. Neurosci. 2017, 268, 565–570. [Google Scholar] [CrossRef]
- Faye, A.; Kirpekar, V.; Bhave, S.; Tadke, R.; Gawande, S. Lurasidone-induced anemia: Is there a need for hematological monitoring? Indian J. Pharmacol. 2019, 51, 276. [Google Scholar] [CrossRef]
- Song, X.; Kenston, S.S.F.; Kong, L.; Zhao, J. Molecular mechanisms of nickel induced neurotoxicity and chemoprevention. Toxicology 2017, 392, 47–54. [Google Scholar] [CrossRef] [PubMed]
- He, M.D.; Xu, S.C.; Zhang, X.; Wang, Y.; Xiong, J.C.; Zhang, X.; Lu, Y.H.; Zhang, L.; Yu, Z.P.; Zhou, Z. Disturbance of aerobic metabolism accompanies neurobehavioral changes induced by nickel in mice. Neurotoxicology 2013, 38, 9–16. [Google Scholar] [CrossRef] [PubMed]
- Fatehyab, S.; Hasan, M.; Hasan, M.Z.; Anwar, J. Effect of Nickel on the Levels of Dopamine, Noradrenaline and Serotonin in Different Regions of the Rat Brain. Acta Pharmarol. Toxicol. 1980, 47, 318–320. [Google Scholar]
- Deicken, R.F.; Calabrese, G.; Merrin, E.L.; Meyerhoff, D.J.; Dillon, W.P.; Weiner, M.W.; Fein, G. 31phosphorus Magnetic Resonance Spectroscopy of the Frontal and Parietal Lobes in Chronic Schizophrenia. Biol. Psychiatry 1994, 36, 503–510. [Google Scholar] [CrossRef]
- Fukuzako, H.; Fukuzako, T.; Takeuchi, K.; Ohbo, Y.; Ueyama, K.; Takigawa, M.; Fujimoto, T. Phosphorus magnetic resonance spectroscopy in schizophrenia: Correlation between membrane phospholipid metabolism in the temporal lobe and positive symptoms. Prog. Neuro Psychopharmacol. Biol. Psychiatry 1996, 20, 629–640. [Google Scholar] [CrossRef]
- Yamanaka, R.; Shindo, Y.; Oka, K. Magnesium is a Key Player in Neuronal Maturation and Neuropathology. Int. J. Mol. Sci. 2019, 20, 3439. [Google Scholar] [CrossRef]
- Modabbernia, A.; Velthorst, E.; Gennings, C.; Haan, L.D.; Austin, C.; Sutterland, A.; Mollon, J.; Frangou, S.; Wright, R.; Arora, M.; et al. Early-life metal exposure and schizophrenia: A proof-of-concept study using novel tooth-matrix biomarkers. Eur. Psychiatry 2016, 36, 1–6. [Google Scholar] [CrossRef]
- Kirov, G.K.; Tsachev, K.N. Magnesium, Schizophrenia and Manic-Depressive Disease. Neuropsychobiology 1990, 23, 79–81. [Google Scholar] [CrossRef]
- Plisza, S.R.; Rogeness, G.A. Calcium and magnesium in children with schizophrenia and major depression. Biol. Psychiatry 1983, 19, 871–876. [Google Scholar]
- Ordak, M.; Matras, J.; Muszynska, E.; Nasierowski, T.; Bujalska-Zadrozny, M. Magnesium in schizophrenia. Pharmacol. Rep. 2017, 69, 929–934. [Google Scholar] [CrossRef]
- Verstraeten, S.V.; Aimo, L.; Oteiza, P.I. Aluminium and lead: Molecular mechanisms of brain toxicity. Arch. Toxicol. 2008, 82, 789–802. [Google Scholar] [CrossRef] [PubMed]
- Banks, W.; Kastin, A. Aluminium increases permeability of the blood-brain barrier to labelled dsip and β-endorphin: Possible implications for senile and dialysis dementia. Lancet 1983, 322, 1227–1229. [Google Scholar] [CrossRef]
- Meiri, H.; Banin, E.; Roll, M.; Rousseau, A. Toxic effects of aluminium on nerve cells and synaptic transmission. Prog. Neurobiol. 1993, 40, 89–121. [Google Scholar] [CrossRef]
- Kaya, B.; Akdaǧ, N.; Fadillioǧlu, E.; Taycan, S.E.; Emre, M.H.; Unal, S.; Sayal, A.; Erdogan, H. Elements levels and glucose-6-phosphate dehydrogenase activity in blood of patients with schizophrenia. Düşünen Adam J. Psychiatry Neurol. Sci. 2012, 25, 198–205. [Google Scholar]
- Marger, L.; Schubert, C.; Bertrand, D. Zinc: An underappreciated modulatory factor of brain function. Biochem. Pharmacol. 2014, 91, 426–435. [Google Scholar] [CrossRef]
- Zhifeng Qi, K.E.J.L. The Interaction of Zinc and the Blood-Brain Barrier Under Physiological and Ischemic Conditions. Toxicol. Appl. Pharmacol. 2019, 364, 114–119. [Google Scholar]
- Pfeiffer, C.C.; Braverman, E.R. Zinc, the Brain and Behavior. Biol. Psychiatry 1982, 17, 513–532. [Google Scholar]
- Vela, G.; Stark, P.; Socha, M.; Sauer, A.K.; Hagmeyer, S.; Grabrucker, A.M. Zinc in Gut-Brain Interaction in Autism and Neurological Disorders. Neural Plast. 2015, 2015, 1–15. [Google Scholar] [CrossRef]
- Portbury, S.; Adlard, P. Zinc Signal in Brain Diseases. Int. J. Mol. Sci. 2017, 18, 2506. [Google Scholar] [CrossRef]
- Pochwat, B.; Nowak, G.; Szewczyk, B. Relationship between Zinc (Zn2+) and Glutamate Receptors in the Processes Underlying Neurodegeneration. Neural Plast. 2015, 2015, 591563. [Google Scholar] [CrossRef]
- Doboszewska, U.; Młyniec, K.; Wlaź, A.; Poleszak, E.; Nowak, G.; Wlaź, P. Zinc signaling and epilepsy. Pharmacol. Ther. 2019, 193, 156–177. [Google Scholar] [CrossRef] [PubMed]
- Joe, P.; Petrilli, M.; Malaspina, D.; Weissman, J. Zinc in schizophrenia: A meta-analysis. Gen. Hosp. Psychiatry 2018, 53, 19–24. [Google Scholar] [CrossRef] [PubMed]
- Saghazadeh, A.; Mahmoudi, M.; Shahrokhi, S.; Mojarrad, M.; Dastmardi, M.; Mirbeyk, M.; Rezaei, N. Trace elements in schizophrenia: A systematic review and meta-analysis of 39 studies (N = 5151 participants). Nutr. Rev. 2019, 78, 278–303. [Google Scholar] [CrossRef] [PubMed]
- Adams, C.E.; Demasters, B.; Freedman, R. Regional zinc staining in postmortem hippocampus from schizophrenic patients. Schizophr. Res. 1995, 18, 71–77. [Google Scholar] [CrossRef]
- Paulsen, B.D.S.; Cardoso, S.C.; Stelling, M.P.; Cadilhe, D.V.; Rehen, S.K. Valproate reverts zinc and potassium imbalance in schizophrenia-derived reprogrammed cells. Schizophr. Res. 2014, 154, 30–35. [Google Scholar] [CrossRef]
- Dambrosi, N.; Rossi, L. Copper at synapse: Release, binding and modulation of neurotransmission. Neurochem. Int. 2015, 90, 36–45. [Google Scholar] [CrossRef]
- Lutsenko, S.; Washington-Hughes, C.; Ralle, M.; Schmidt, K. Copper and the brain noradrenergic system. JBIC J. Biol. Inorg. Chem. 2019, 24, 1179–1188. [Google Scholar] [CrossRef]
- Davies, K.M.; Hare, D.J.; Cottam, V.; Chen, N.; Hilgers, L.; Halliday, G.; Mercer, J.F.B.; Double, K.L. Localization of copper and copper transporters in the human brain. Metallomics 2013, 5, 43–51. [Google Scholar] [CrossRef]
- Mital, M.; Bal, W.; Frączyk, T.; Drew, S.C. Interplay between Copper, Neprilysin, and N-Truncation of β-Amyloid. Inorg. Chem. 2018, 57, 6193–6197. [Google Scholar] [CrossRef]
- Gokhale, A.; Vrailas-Mortimer, A.; Larimore, J.; Comstra, H.S.; Zlatic, S.A.; Werner, E.; Manvich, D.F.; Iuvone, P.M.; Weinshenker, D.; Faundez, V. Neuronal copper homeostasis susceptibility by genetic defects in dysbindin, a schizophrenia susceptibility factor. Hum. Mol. Genet. 2015, 24, 5512–5523. [Google Scholar] [CrossRef]
- Ni, M.; You, Y.; Chen, J.; Zhang, L. Copper in depressive disorder: A systematic review and meta-analysis of observational studies. Psychiatry Res. 2018, 267, 506–515. [Google Scholar] [CrossRef] [PubMed]
- Bowman, M.B.; Lewis, M.S. The copper hypothesis of schizophrenia: A review. Neurosci. Biobehav. Rev. 1982, 6, 321–328. [Google Scholar] [CrossRef]
- Schoonover, K.E.; Queern, S.L.; Lapi, S.E.; Roberts, R.C. Impaired copper transport in schizophrenia results in a copper-deficient brain state: A new side to the dysbindin story. World J. Biol. Psychiatry 2018, 21, 13–28. [Google Scholar] [CrossRef] [PubMed]
- Cardoso, B.R.; Roberts, B.R.; Bush, A.I.; Hare, D.J. Selenium, selenoproteins and neurodegenerative diseases. Metallomics 2015, 7, 1213–1228. [Google Scholar] [CrossRef]
- Vinceti, M.; Filippini, T.; Wise, L.A. Environmental Selenium and Human Health: An Update. Curr. Environ. Health Rep. 2018, 5, 464–485. [Google Scholar] [CrossRef]
- Valentine, W.M.; Abel, T.W.; Hill, K.E.; Austin, L.M.; Burk, R.F. Neurodegeneration in mice resulting from loss of functional selenoprotein P or its receptor apolipoprotein E receptor 2. J. Neuropathol. Exp. Neurol. 2008, 67, 68–77. [Google Scholar] [CrossRef]
- Pitts, M.W.; Kremer, P.M.; Hashimoto, A.C.; Torres, D.J.; Byrns, C.N.; Williams, C.S.; Berry, J.B. Competition between the Brain and Testes under Selenium-Compromised Conditions: Insight into Sex Differences in Selenium Metabolism and Risk of Neurodevelopmental Disease. J. Neurosci. 2015, 35, 15326–15338. [Google Scholar] [CrossRef]
- Steinbrenner, H.; Sies, H. Selenium homeostasis and antioxidant selenoproteins in brain: Implications for disorders in the central nervous system. Arch. Biochem. Biophys. 2013, 536, 152–157. [Google Scholar] [CrossRef]
- Reddy, V.S.; Bukke, S.; Dutt, N.; Rana, P.; Pandey, A.K. A systematic review and meta-analysis of the circulatory, erythrocellular and CSF selenium levels in Alzheimers disease: A metal meta-analysis (AMMA study-I). J. Trace Elem. Med. Biol. 2017, 42, 68–75. [Google Scholar] [CrossRef]
- Pillai, R.; Uyehara-Lock, J.H.; Bellinger, F.P. Selenium and selenoprotein function in brain disorders. Iubmb Life 2014, 66, 229–239. [Google Scholar] [CrossRef]
- Kanazawa, T.; Glatt, S.J.; Faraone, S.V.; Hwu, H.G.; Yoneda, H.; Tsuang, M.T. Family-based association study of SELENBP1 in schizophrenia. Schizophr. Res. 2009, 113, 268–272. [Google Scholar] [CrossRef] [PubMed]
- Kanazawa, T.; Chana, G.; Glatt, S.J.; Mizuno, H.; Masliah, E.; Yoneda, H.; Tsuang, M.T.; Everall, I.P. The utility of SELENBP1 gene expression as a biomarker for major psychotic disorders: Replication in schizophrenia and extension to bipolar disorder with psychosis. Am. J. Med. Genet. B Neuropsychiatr Genet. 2008, 147, 686–689. [Google Scholar] [CrossRef] [PubMed]
- Berry, T. A selenium transport protein model of a sub-type of schizophrenia. Med. Hypotheses 1994, 43, 409–414. [Google Scholar] [CrossRef]
- Benton, D. Selenium Intake, Mood and Other Aspects of Psychological Functioning. Nutr. Neurosci. 2002, 5, 363–374. [Google Scholar] [CrossRef]
- Segal, M.; Korkotian, E. Roles of Calcium Stores and Store-Operated Channels in Plasticity of Dendritic Spines. Neuroscientist 2016, 22, 477–485. [Google Scholar] [CrossRef]
- Sharma, A.; Schray, A.; Bartolovic, M.; Roesch-Ely, D.; Aschenbrenner, S.; Weisbrod, M. Relationship between serum calcium and neuropsychological performance might indicate etiological heterogeneity underlying cognitive deficits in schizophrenia and depression. Psychiatry Res. 2017, 252, 80–86. [Google Scholar] [CrossRef]
- Bergantin, L.B.; Caricati-Neto, A. Challenges for the pharmacological treatment of neurological and psychiatric disorders: Implications of the Ca 2 /cAMP intracellular signalling interaction. Eur. J. Pharmacol. 2016, 788, 255–260. [Google Scholar] [CrossRef]
- Lisek, M.; Boczek, T.; Zylinska, L. Calcium as a Trojan horse in mental diseases—The role of PMCA and PMCA-interacting proteins in bipolar disorder and schizophrenia. Neurosci. Lett. 2018, 663, 48–54. [Google Scholar] [CrossRef]
- Vidal-Domènech, F.; Riquelme, G.; Pinacho, R.; Rodriguez-Mias, R.; Vera, A.; Monje, A.; Ferrer, I.; Callado, L.F.; Meana, J.J.; Villen, J.; et al. Calcium-Binding Proteins are Altered in the Cerebellum in Schizophrenia. bioRxiv 2020. [Google Scholar] [CrossRef]
- Li, L.; Yang, X. The Essential Element Manganese, Oxidative Stress, and Metabolic Diseases: Links and Interactions. Oxid. Med. Cell. Longev. 2018, 2018, 7580707. [Google Scholar] [CrossRef]
- Finley, J.W. Does environmental exposure to manganese pose a health risk to healthy adults. Nutr. Rev. 2004, 62, 148–153. [Google Scholar] [CrossRef] [PubMed]
- Sidoryk-Wegrzynowicz, M.; Lee, E.; Albrecht, J.; Aschner, M. Manganese disrupts astrocyte glutamine transporter expression and function. J. Neurochem. 2009, 110, 822–830. [Google Scholar] [CrossRef] [PubMed]
- Dobson, A.; Erikson, K.; Aschner, M. Manganese Neurotoxicity. Ann. N. Y. Acad. Sci. 2004, 1012, 115–128. [Google Scholar] [CrossRef] [PubMed]
- Peres, T.V.; Schettinger, M.R.C.; Chen, P.; Carvalho, F.; Avila, D.S.; Bowman, A.B.; Aschner, M. Manganese-induced neurotoxicity: A review of its behavioral consequences and neuroprotective strategies. BMC Pharm. Toxicol. 2016, 17, 57. [Google Scholar] [CrossRef] [PubMed]
- Firth, J.; Carney, R.; Stubbs, B.; Teasdale, S.B.; Vancampfort, D.; Ward, P.B.; Berk, M.; Sarris, J. Nutritional Deficiencies and Clinical Correlates in First-Episode Psychosis: A Systematic Review and Meta-analysis. Schizophr. Bull. 2017, 44, 1275–1292. [Google Scholar] [CrossRef] [PubMed]
- Wu, J.; Kosten, T.; Zhang, X. Free radicals, antioxidant defense systems, and schizophrenia. Prog. Neuro Psychopharmacol. Biol. Psychiatry 2013, 46, 200–2006. [Google Scholar] [CrossRef]
- Wang, D.-F.; Cao, B.; Xu, M.-Y.; Liu, Y.-Q.; Yan, L.-L.; Liu, R.; Wang, J.-Y.; Lu, Q.-B. Meta-Analyses of Manganese Superoxide Dismutase Activity, Gene Ala-9Val Polymorphism, and the Risk of Schizophrenia. Medicine 2015, 94, e1507. [Google Scholar] [CrossRef]
- Morello, M.; Zatta, P.; Zambenedetti, P.; Martorana, A.; D’Angelo, V.; Melchiorri, G.; Bernardi, G.; Sancesario, G. Manganese intoxication decreases the expression of manganoproteins in the rat basal ganglia: An immunohistochemical study. Brain Res. Bull. 2007, 74, 406–415. [Google Scholar] [CrossRef]
- Flatow, J.; Buckley, P.; Miller, B. Meta-Analysis of Oxidative Stress in Schizophrenia. Biol. Psychiatry 2013, 74, 400–409. [Google Scholar] [CrossRef]
- Wu, J.Q.; Chen, D.C.; Tan, Y.L.; Tan, S.P.; Wang, Z.R.; Xiu, M.H.; Yang, F.D.; Zhang, X.Y. Cognition impairment in schizophrenia patients with tardive dyskinesia: Association with plasma superoxide dismutase activity. Schizophr. Res. 2014, 152, 210–216. [Google Scholar] [CrossRef]
- Arinola, O.; Idonije, O. Status of plasma nitric oxide and non-enzymatic antioxidants before and after antipsychotic treatment in Nigerian patients with schizophrenia. J. Res. Med. Sci. 2009, 14, 37–42. [Google Scholar] [PubMed]
- Rivera-Mancía, S.; Colin-Ramirez, E.; Montes, S.; Cartas, R.; Vargas-Barrón, J.; Vallejo, M. Dyslipidemia and tobacco smoking synergistically increase serum manganese. Investig. Clínica 2017, 58, 238–249. [Google Scholar]
- Bulska, E.; Ruszczyńska, A. Analytical Techniques for Trace Element Determination. Phys. Sci. Rev. 2017, 2, 20178002. [Google Scholar] [CrossRef]
- Kurita, H.; Ohsako, S.; Hashimoto, S.; Yoshinaga, J.; Tohyama, C. Prenatal zinc deficiency-dependent epigenetic alterations of mouse metallothionein-2 gene. J. Nutr. Biochem. 2013, 24, 256–266. [Google Scholar] [CrossRef] [PubMed]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).