Plasma Dimethylarginine Levels and Carotid Intima–Media Thickness are related to Atrial Fibrillation in Patients with Embolic Stroke
Abstract
1. Introduction
2. Results
2.1. Epidemiological Data
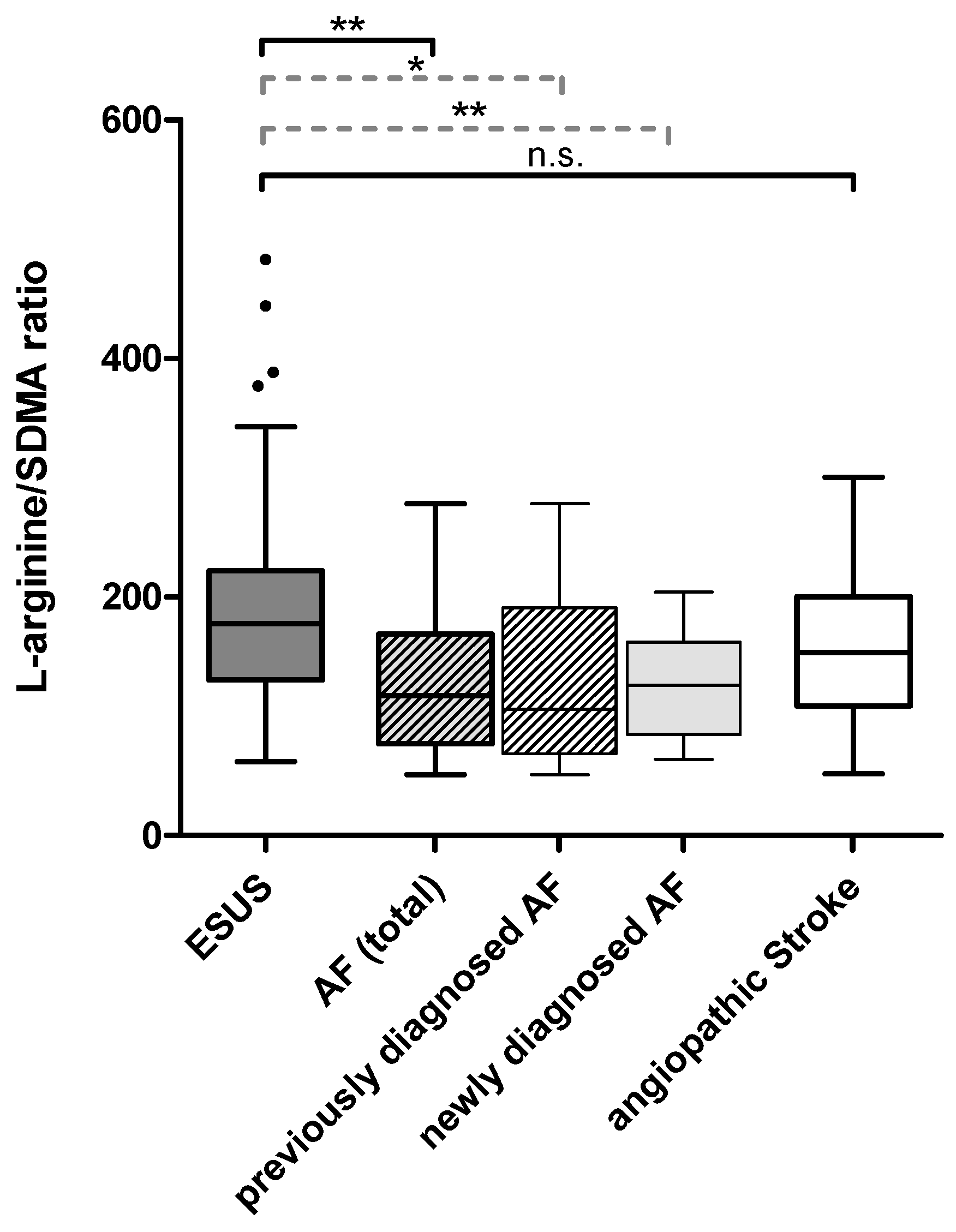
2.2. Plasma Dimethylarginine Levels in Ischemic Stroke of Different Causes
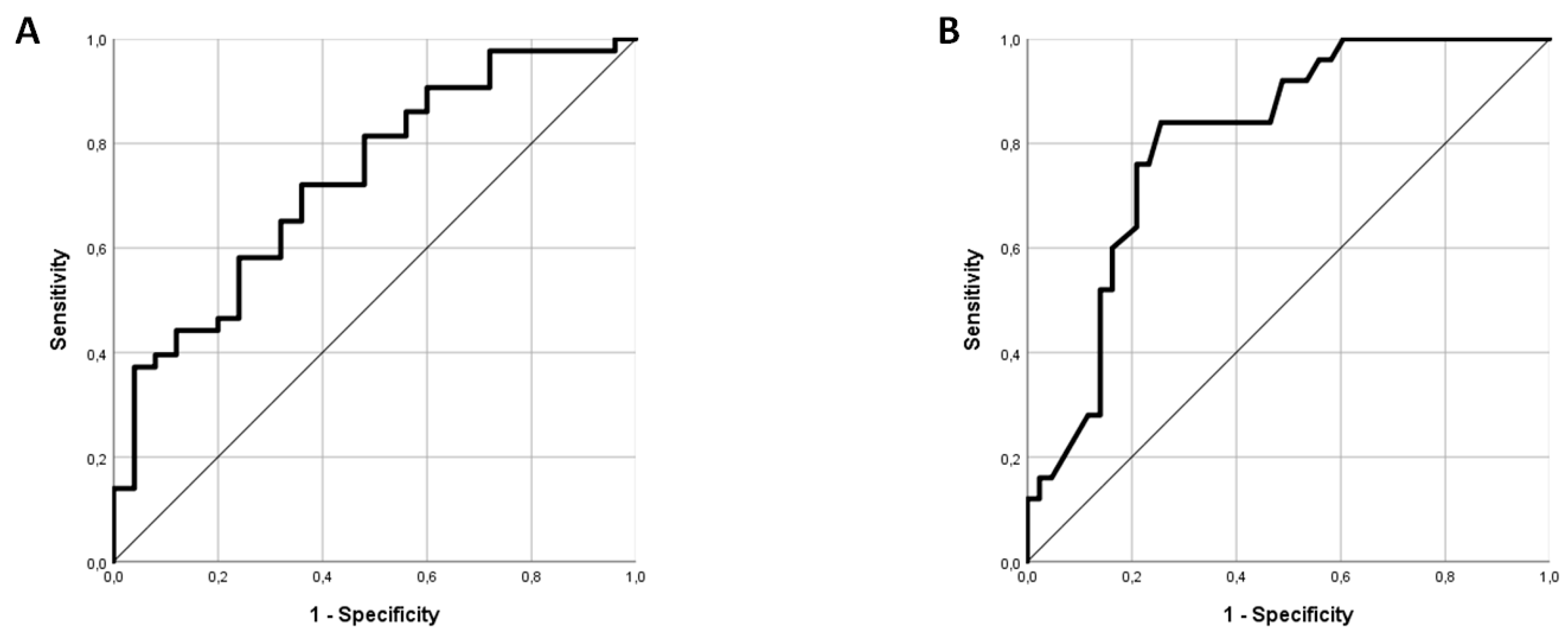
2.3. L-arginine/SDMA Ratio as A Potential Marker for Identifying AF in Patients Admitted with ESUS
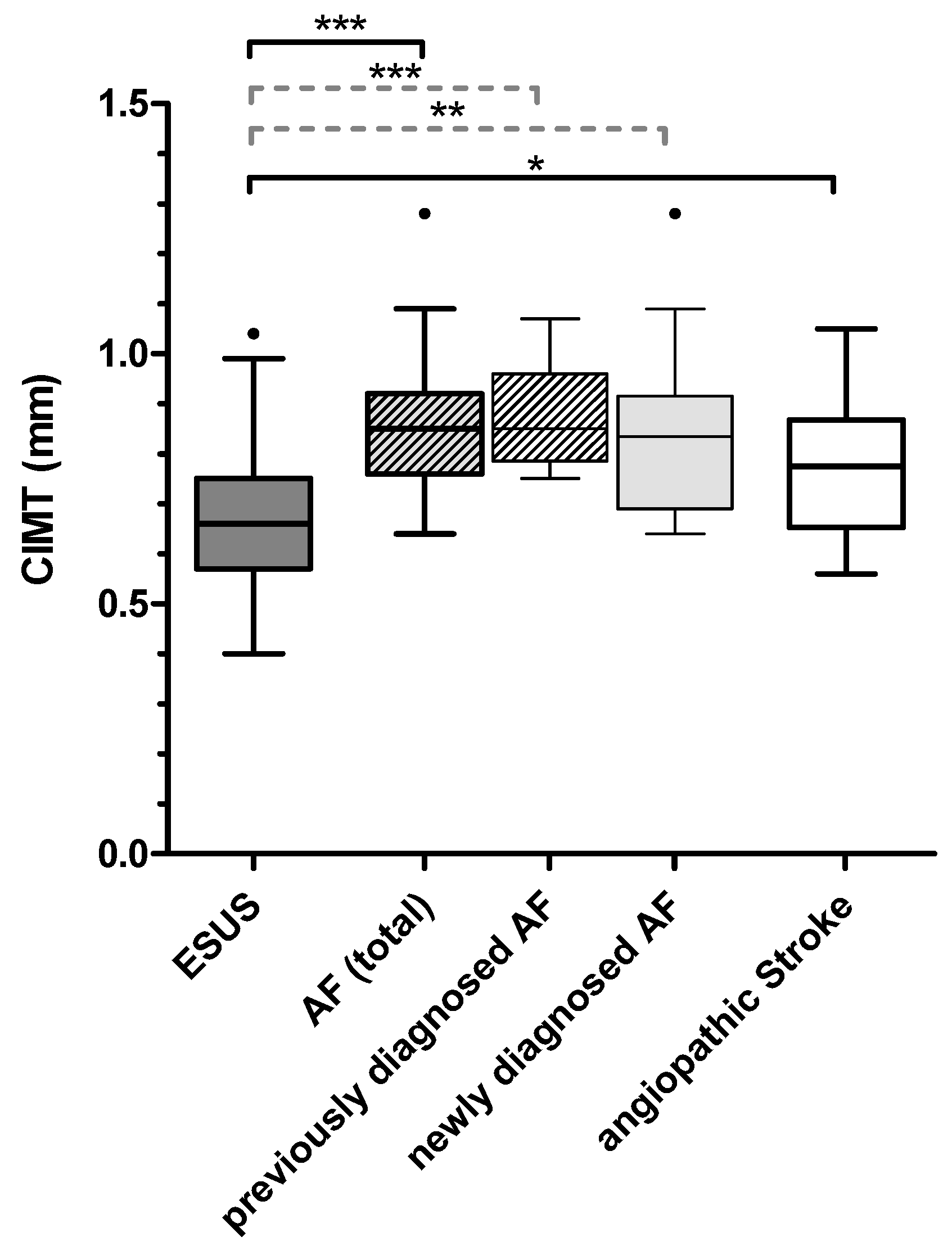
2.4. Carotid Intima–Media Thickness Differs between Patients with ESUS and Other Etiologies
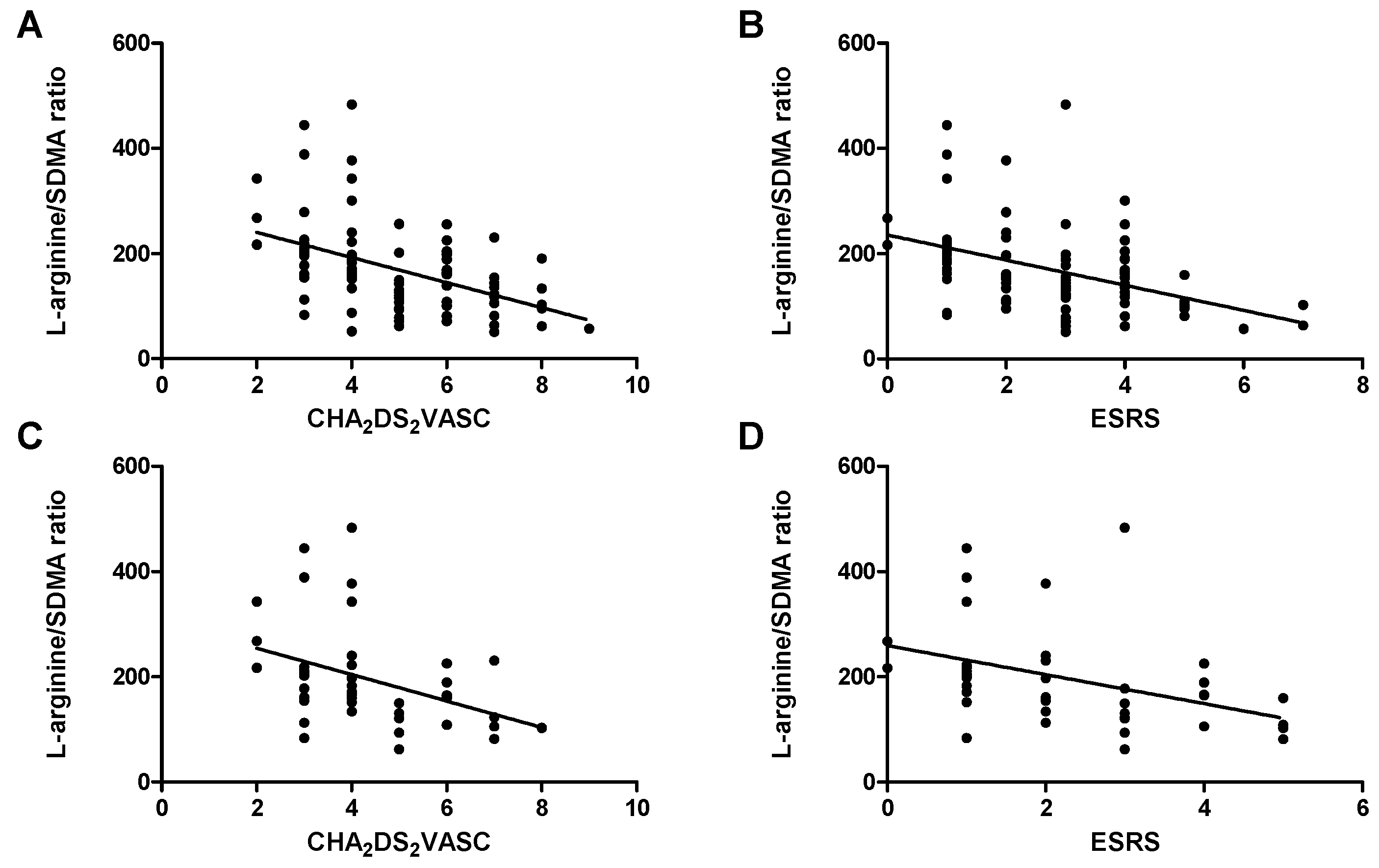
2.5. Markers of Endothelial Dysfunction and Thrombembolic Risk
2.6. Markers of Endothelial Dysfunction and Renal Function
3. Discussion
4. Materials and Methods
4.1. Study Population
4.2. Clinical Evaluation
4.3. Biomarker Analysis
4.4. Statistical Analysis
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
Abbreviations
| ADMA | asymmetric dimethylarginine |
| AF | atrial fibrillation |
| ASA | acetylsalicylic acid |
| AUC | area under the curve |
| BMI | body mass index |
| CI | confidence interval |
| CIMT | carotid intima–media thickness |
| DWI | diffusion-weighted imaging |
| ECG | electrocardiography |
| ESRS | Essen Stroke Risk Score |
| ESUS | embolic stroke of undetermined source |
| MRI | magnetic resonance imaging |
| NIHSS | National Institutes of Health Stroke Scale |
| NO | nitric oxide |
| NOAC | non-vitamin K oral anticoagulant |
| PFO | patent foramen ovale |
| ROC | receiver operating characteristic |
| SDMA | symmetric dimethylarginine |
| TEE | transesophageal echocardiography |
References
- Hart, R.G.; Diener, H.C.; Coutts, S.B.; Easton, J.D.; Granger, C.B.; O’Donnell, M.J.; Sacco, R.L.; Connolly, S.J. Cryptogenic Stroke/ESUS International Working Group. Embolic Strokes of Undetermined Source: The Case for a New Clinical Construct. Lancet Neurol. 2014, 13, 429–438. [Google Scholar] [CrossRef]
- Hart, R.G.; Sharma, M.; Mundl, H.; Kasner, S.E.; Bangdiwala, S.I.; Berkowitz, S.D.; Swaminathan, B.; Lavados, P.; Wang, Y.; Wang, Y.; et al. Rivaroxaban for Stroke Prevention After Embolic Stroke of Undetermined Source. N. Engl. J. Med. 2018, 378, 2191–2201. [Google Scholar] [CrossRef] [PubMed]
- Wachter, R.; Groschel, K.; Gelbrich, G.; Hamann, G.F.; Kermer, P.; Liman, J.; Seegers, J.; Wasser, K.; Schulte, A.; Jurries, F.; et al. Holter-Electrocardiogram-Monitoring in Patients with Acute Ischaemic Stroke (Find-AFRANDOMISED): An Open-Label Randomised Controlled Trial. Lancet Neurol. 2017, 16, 282–290. [Google Scholar] [CrossRef]
- Guo, Y.; Lip, G.Y.; Apostolakis, S. Inflammation in Atrial Fibrillation. J. Am. Coll. Cardiol. 2012, 60, 2263–2270. [Google Scholar] [CrossRef] [PubMed]
- Wijesurendra, R.S.; Casadei, B. Atrial Fibrillation: Effects Beyond the Atrium? Cardiovasc. Res. 2015, 105, 238–247. [Google Scholar] [CrossRef] [PubMed]
- Goette, A.; Kalman, J.M.; Aguinaga, L.; Akar, J.; Cabrera, J.A.; Chen, S.A.; Chugh, S.S.; Corradi, D.; D’Avila, A.; Dobrev, D.; et al. EHRA/HRS/APHRS/SOLAECE Expert Consensus on Atrial Cardiomyopathies: Definition, Characterization, and Clinical Implication. Europace 2016, 18, 1455–1490. [Google Scholar] [CrossRef]
- Willeit, K.; Kiechl, S. Atherosclerosis and Atrial Fibrillation--Two Closely Intertwined Diseases. Atherosclerosis 2014, 233, 679–681. [Google Scholar] [CrossRef]
- Kim, S.J.; Choisy, S.C.; Barman, P.; Zhang, H.; Hancox, J.C.; Jones, S.A.; James, A.F. Atrial Remodeling and the Substrate for Atrial Fibrillation in Rat Hearts with Elevated Afterload. Circ. Arrhythm Electrophysiol. 2011, 4, 761–769. [Google Scholar] [CrossRef]
- Skalidis, E.I.; Zacharis, E.A.; Tsetis, D.K.; Pagonidis, K.; Chlouverakis, G.; Yarmenitis, S.; Hamilos, M.; Manios, E.G.; Vardas, P.E. Endothelial Cell Function during Atrial Fibrillation and After Restoration of Sinus Rhythm. Am. J. Cardiol. 2007, 99, 1258–1262. [Google Scholar] [CrossRef]
- Guazzi, M.; Belletti, S.; Lenatti, L.; Bianco, E.; Guazzi, M.D. Effects of Cardioversion of Atrial Fibrillation on Endothelial Function in Hypertension Or Diabetes. Eur. J. Clin. Investig. 2007, 37, 26–34. [Google Scholar] [CrossRef]
- Tveit, A.; Arnesen, H.; Smith, P.; Bratseth, V.; Seljeflot, I. L-Arginine, Asymmetric Dimethylarginine and Rhythm Outcome After Electrical Cardioversion for Atrial Fibrillation. Cardiology 2010, 117, 176–180. [Google Scholar] [CrossRef] [PubMed]
- Willeit, P.; Freitag, D.F.; Laukkanen, J.A.; Chowdhury, S.; Gobin, R.; Mayr, M.; Di Angelantonio, E.; Chowdhury, R. Asymmetric Dimethylarginine and Cardiovascular Risk: Systematic Review and Meta-Analysis of 22 Prospective Studies. J. Am. Heart Assoc. 2015, 4, e001833. [Google Scholar] [CrossRef] [PubMed]
- Bai, Y.; Sun, L.; Du, L.; Zhang, T.; Xin, W.; Lan, X.; Du, G. Association of Circulating Levels of Asymmetric Dimethylarginine (ADMA) with Carotid Intima-Media Thickness: Evidence from 6168 Participants. Ageing Res. Rev. 2013, 12, 699–707. [Google Scholar] [CrossRef] [PubMed]
- Chen, S.; Li, N.; Deb-Chatterji, M.; Dong, Q.; Kielstein, J.T.; Weissenborn, K.; Worthmann, H. Asymmetric Dimethyarginine as Marker and Mediator in Ischemic Stroke. Int. J. Mol. Sci. 2012, 13, 15983–16004. [Google Scholar] [CrossRef]
- Chen, S.; Martens-Lobenhoffer, J.; Weissenborn, K.; Kielstein, J.; Lichtinghagen, R.; Deb, M.; Li, N.; Tryc, A.; Goldbecker, A.; Dong, Q.; et al. Association of Dimethylarginines and Mediators of Inflammation After Acute Ischemic Stroke. J. Neuroinflamm. 2012, 9, 251. [Google Scholar] [CrossRef]
- Worthmann, H.; Li, N.; Martens-Lobenhoffer, J.; Dirks, M.; Schuppner, R.; Lichtinghagen, R.; Kielstein, J.T.; Raab, P.; Lanfermann, H.; Bode-Boger, S.M.; et al. Dimethylarginines in Patients with Intracerebral Hemorrhage: Association with Outcome, Hematoma Enlargement, and Edema. J. Neuroinflamm. 2017, 14, 247. [Google Scholar] [CrossRef] [PubMed]
- Bode-Boger, S.M.; Scalera, F.; Kielstein, J.T.; Martens-Lobenhoffer, J.; Breithardt, G.; Fobker, M.; Reinecke, H. Symmetrical Dimethylarginine: A New Combined Parameter for Renal Function and Extent of Coronary Artery Disease. J. Am. Soc. Nephrol. 2006, 17, 1128–1134. [Google Scholar] [CrossRef] [PubMed]
- Kielstein, J.T.; Salpeter, S.R.; Bode-Boeger, S.; Cooke, J.P.; Fliser, D. Symmetric Dimethylarginine (SDMA) as Endogenous Marker of Renal Function-a Meta-Analysis. Nephrol. Dial. Transplant. 2006, 21, 2446–2451. [Google Scholar] [CrossRef] [PubMed]
- Schlesinger, S.; Sonntag, S.R.; Lieb, W.; Maas, R. Asymmetric and Symmetric Dimethylarginine as Risk Markers for Total Mortality and Cardiovascular Outcomes: A Systematic Review and Meta-Analysis of Prospective Studies. PLoS ONE 2016, 11, e0165811. [Google Scholar] [CrossRef] [PubMed]
- Schulze, F.; Carter, A.M.; Schwedhelm, E.; Ajjan, R.; Maas, R.; von Holten, R.; Atzler, D.; Grant, P.J.; Boger, R.H. Symmetric Dimethylarginine Predicts all-Cause Mortality Following Ischemic Stroke. Atherosclerosis 2010, 208, 518–523. [Google Scholar] [CrossRef]
- Israel, C.; Kitsiou, A.; Kalyani, M.; Deelawar, S.; Ejangue, L.E.; Rogalewski, A.; Hagemeister, C.; Minnerup, J.; Schabitz, W.R. Detection of Atrial Fibrillation in Patients with Embolic Stroke of Undetermined Source by Prolonged Monitoring with Implantable Loop Recorders. Thromb. Haemost. 2017, 117, 1962–1969. [Google Scholar] [CrossRef] [PubMed]
- Martinez-Majander, N.; Aarnio, K.; Pirinen, J.; Lumikari, T.; Nieminen, T.; Lehto, M.; Sinisalo, J.; Kaste, M.; Tatlisumak, T.; Putaala, J. Embolic Strokes of Undetermined Source in Young Adults: Baseline Characteristics and Long-Term Outcome. Eur. J. Neurol. 2017, 25, 535–541. [Google Scholar] [CrossRef] [PubMed]
- Kasner, S.E.; Lavados, P.; Sharma, M.; Wang, Y.; Wang, Y.; Davalos, A.; Shamalov, N.; Cunha, L.; Lindgren, A.; Mikulik, R.; et al. Characterization of Patients with Embolic Strokes of Undetermined Source in the NAVIGATE ESUS Randomized Trial. J. Stroke Cerebrovasc. Dis. 2018, 27, 1673–1682. [Google Scholar] [CrossRef]
- Arauz, A.; Morelos, E.; Colin, J.; Roldan, J.; Barboza, M.A. Comparison of Functional Outcome and Stroke Recurrence in Patients with Embolic Stroke of Undetermined Source (ESUS) Vs. Cardioembolic Stroke Patients. PLoS ONE 2016, 11, e0166091. [Google Scholar] [CrossRef] [PubMed]
- Bisson, A.; Clementy, N.; Bodin, A.; Angoulvant, D.; Babuty, D.; Lip, G.Y.H.; Fauchier, L. Relationship of Preexisting Cardiovascular Comorbidities to Newly Diagnosed Atrial Fibrillation After Ischemic Stroke. Stroke 2017, 48, 2878–2880. [Google Scholar] [CrossRef] [PubMed]
- Bisson, A.; Bodin, A.; Clementy, N.; Babuty, D.; Lip, G.Y.H.; Fauchier, L. Prediction of Incident Atrial Fibrillation According to Gender in Patients with Ischemic Stroke from a Nationwide Cohort. Am. J. Cardiol. 2017, 121, 437–444. [Google Scholar] [CrossRef] [PubMed]
- Hov, G.G.; Sagen, E.; Bigonah, A.; Asberg, A. Health-Associated Reference Values for Arginine, Asymmetric Dimethylarginine (ADMA) and Symmetric Dimethylarginine (SDMA) Measured with High-Performance Liquid Chromatography. Scand. J. Clin. Lab. Investig. 2007, 67, 868–876. [Google Scholar] [CrossRef]
- Schnabel, R.B.; Maas, R.; Wang, N.; Yin, X.; Larson, M.G.; Levy, D.; Ellinor, P.T.; Lubitz, S.A.; McManus, D.D.; Magnani, J.W.; et al. Asymmetric Dimethylarginine, Related Arginine Derivatives, and Incident Atrial Fibrillation. Am. Heart J. 2016, 176, 100–106. [Google Scholar] [CrossRef]
- Ramuschkat, M.; Appelbaum, S.; Atzler, D.; Zeller, T.; Bauer, C.; Ojeda, F.M.; Sinning, C.R.; Hoffmann, B.; Lackner, K.J.; Boger, R.H.; et al. ADMA, Subclinical Changes and Atrial Fibrillation in the General Population. Int. J. Cardiol. 2016, 203, 640–646. [Google Scholar] [CrossRef]
- Sattler, K.; Behnes, M.; Barth, C.; Wenke, A.; Sartorius, B.; El-Battrawy, I.; Mashayekhi, K.; Kuschyk, J.; Hoffmann, U.; Papavasiliu, T.; et al. Occlusion of Left Atrial Appendage Affects Metabolomic Profile: Focus on Glycolysis, Tricarboxylic Acid and Urea Metabolism. Metabolomics 2017, 13, 127. [Google Scholar] [CrossRef]
- Seppala, I.; Kleber, M.E.; Bevan, S.; Lyytikainen, L.P.; Oksala, N.; Hernesniemi, J.A.; Makela, K.M.; Rothwell, P.M.; Sudlow, C.; Dichgans, M.; et al. Associations of Functional Alanine-Glyoxylate Aminotransferase 2 Gene Variants with Atrial Fibrillation and Ischemic Stroke. Sci. Rep. 2016, 6, 23207. [Google Scholar] [CrossRef] [PubMed]
- Bansal, N.; Zelnick, L.R.; Alonso, A.; Benjamin, E.J.; de Boer, I.H.; Deo, R.; Katz, R.; Kestenbaum, B.; Mathew, J.; Robinson-Cohen, C.; et al. eGFR and Albuminuria in Relation to Risk of Incident Atrial Fibrillation: A Meta-Analysis of the Jackson Heart Study, the Multi-Ethnic Study of Atherosclerosis, and the Cardiovascular Health Study. Clin. J. Am. Soc. Nephrol. 2017, 12, 1386–1398. [Google Scholar] [CrossRef] [PubMed]
- MacAllister, R.J.; Rambausek, M.H.; Vallance, P.; Williams, D.; Hoffmann, K.H.; Ritz, E. Concentration of Dimethyl-L-Arginine in the Plasma of Patients with End-Stage Renal Failure. Nephrol. Dial. Transplant. 1996, 11, 2449–2452. [Google Scholar] [CrossRef] [PubMed]
- Chen, L.Y.; Leening, M.J.; Norby, F.L.; Roetker, N.S.; Hofman, A.; Franco, O.H.; Pan, W.; Polak, J.F.; Witteman, J.C.; Kronmal, R.A.; et al. Carotid Intima-Media Thickness and Arterial Stiffness and the Risk of Atrial Fibrillation: The Atherosclerosis Risk in Communities (ARIC) Study, Multi-Ethnic Study of Atherosclerosis (MESA), and the Rotterdam Study. J. Am. Heart Assoc. 2016, 5. [Google Scholar] [CrossRef] [PubMed]
- Proietti, M.; Calvieri, C.; Malatino, L.; Signorelli, S.; Corazza, G.R.; Perticone, F.; Vestri, A.R.; Loffredo, L.; Davi, G.; Violi, F.; et al. Relationship between Carotid Intima-Media Thickness and Non Valvular Atrial Fibrillation Type. Atherosclerosis 2015, 238, 350–355. [Google Scholar] [CrossRef] [PubMed]
- Adamsson Eryd, S.; Ostling, G.; Rosvall, M.; Persson, M.; Smith, J.G.; Melander, O.; Hedblad, B.; Engstrom, G. Carotid Intima-Media Thickness is Associated with Incidence of Hospitalized Atrial Fibrillation. Atherosclerosis 2014, 233, 673–678. [Google Scholar] [CrossRef] [PubMed]
- Cui, R.; Yamagishi, K.; Muraki, I.; Hayama-Terada, M.; Umesawa, M.; Imano, H.; Li, Y.; Eshak, E.S.; Ohira, T.; Kiyama, M.; et al. Association between Markers of Arterial Stiffness and Atrial Fibrillation in the Circulatory Risk in Communities Study (CIRCS). Atherosclerosis 2017, 263, 244–248. [Google Scholar] [CrossRef]
- Mitchell, G.F.; Vasan, R.S.; Keyes, M.J.; Parise, H.; Wang, T.J.; Larson, M.G.; D’Agostino RB, S.; Kannel, W.B.; Levy, D.; Benjamin, E.J. Pulse Pressure and Risk of New-Onset Atrial Fibrillation. JAMA 2007, 297, 709–715. [Google Scholar] [CrossRef]
- Lau, D.H.; Middeldorp, M.E.; Brooks, A.G.; Ganesan, A.N.; Roberts-Thomson, K.C.; Stiles, M.K.; Leong, D.P.; Abed, H.S.; Lim, H.S.; Wong, C.X.; et al. Aortic Stiffness in Lone Atrial Fibrillation: A Novel Risk Factor for Arrhythmia Recurrence. PLoS ONE 2013, 8, e76776. [Google Scholar] [CrossRef]
- Sporns, P.B.; Hanning, U.; Schwindt, W.; Velasco, A.; Minnerup, J.; Zoubi, T.; Heindel, W.; Jeibmann, A.; Niederstadt, T.U. Ischemic Stroke: What does the Histological Composition Tell Us about the Origin of the Thrombus? Stroke 2017, 48, 2206–2210. [Google Scholar] [CrossRef]
- Boeckh-Behrens, T.; Kleine, J.F.; Zimmer, C.; Neff, F.; Scheipl, F.; Pelisek, J.; Schirmer, L.; Nguyen, K.; Karatas, D.; Poppert, H. Thrombus Histology Suggests Cardioembolic Cause in Cryptogenic Stroke. Stroke 2016, 47, 1864–1871. [Google Scholar] [CrossRef] [PubMed]
- Elkind, M.S.V. Atrial Cardiopathy and Stroke Prevention. Curr. Cardiol. Rep. 2018, 20, 103. [Google Scholar] [CrossRef] [PubMed]
- Jalini, S.; Rajalingam, R.; Nisenbaum, R.; Javier, A.D.; Woo, A.; Pikula, A. Atrial Cardiopathy in Patients with Embolic Strokes of Unknown Source and Other Stroke Etiologies. Neurology 2018. [Google Scholar] [CrossRef] [PubMed]
- Worthmann, H.; Chen, S.; Martens-Lobenhoffer, J.; Li, N.; Deb, M.; Tryc, A.B.; Goldbecker, A.; Dong, Q.; Kielstein, J.T.; Bode-Boger, S.M.; et al. High Plasma Dimethylarginine Levels are Associated with Adverse Clinical Outcome After Stroke. J. Atheroscler. Thromb. 2011, 18, 753–761. [Google Scholar] [CrossRef] [PubMed]
- Martens-Lobenhoffer, J.; Bode-Boger, S.M. Quantification of L-Arginine, Asymmetric Dimethylarginine and Symmetric Dimethylarginine in Human Plasma: A Step Improvement in Precision by Stable Isotope Dilution Mass Spectrometry. J. Chromatogr. B. Analyt Technol. Biomed. Life. Sci. 2012, 904, 140–143. [Google Scholar] [CrossRef] [PubMed]
| ESUS | AF (N = 25) | Angiopathic Stroke | p-Value | ||
|---|---|---|---|---|---|
| Previously Diagnosed | Newly Diagnosed | ||||
| N | 43 | 9 | 16 | 20 | |
| Age (a) (IQR) | 65.00 $$ (52–75) | 83.00 **ßß (74–91) | 77.50 (70–82.25) | 68.50 §§ (59.50–78.00) | 0.001 |
| Male | 28 $ (65%) | 3 $ (33%) | 15 *§ (94%) | 13 (65%) | 0.019 |
| Female | 15 $ (35%) | 6 $ (66%) | 1 *§ (6%) | 7 (35%) | |
| ESRS (IQR) | 2.00 (1–3) | 3.00 (2.5–3.5) | 3.50 (2.25–4) | 3.00 (2–4) | 0.016 |
| CHA2DS2 VASC (IQR) | 4.00 $ (3–5) | 6.00 * (5–7) | 6.00 (5–7) | 5.00 (4–6.75) | 0.001 |
| Previous ischemic stroke | 8 (19%) | 1 (11%) | 3 (19%) | 8 (40%) | 0.199 |
| BMI (kg/m2) (IQR) | 26.06 (23.15–28.76) | 24.45 $ß (23.42–25.77) | 26.23 § (24.69–29.91) | 27.49 § (24.93–31.38) | 0.108 |
| NIHSS on admission (IQR) | 2 (1–5) | 4.00 (2–15.5) | 4.00 (1.25–6.75) | 3.00 (2–5) | 0.045 |
| Symptom to venous puncture time (days) (IQR) | 7.00 (6–7) | 7.00 (7–7) | 7.00 (6–7) | 7.00 (7–8) | 0.127 |
| Serum creatinine (µmol/L) (IQR) | 78.00 (65–93) | 75.00 (61–108) | 89 (74–112) | 81.50 (71.50–93) | 0.215 |
| ESUS | Newly Diagnosed AF | AF (Total) | |||
|---|---|---|---|---|---|
| L-arginine (µmol/L) (median (IQR)) | 85.80 (69.70–106.10) | 73.25 (58.40–88.80) | p = 0.03 | 74.40 (57.24–93.52) | p = 0.031 |
| ADMA (µmol/L) (median (IQR)) | 0.48 (0.41–0.55) | 0.51 (0.46–0.56) | p = 0.223 | 0.50 (0.46–0.56) | p = 0.26 |
| SDMA (µmol/L) (median (IQR)) | 0.49 (0.41–0.61) | 0.56 (0.52–0.77) | p = 0.026 | 0.58 (0.52–0.80) | p = 0.004 |
| L-arginine/ADMA ratio (median (IQR)) | 176.51 (153.39–236.50) | 148.75 (120.83–171.65) | p = 0.009 | 148.64 (121.68–181.60) | p = 0.006 |
| L-arginine/SDMA ratio (median (IQR)) | 177.84 (130.85–222.12) | 126.17 (84.99–162.44) | p = 0.004 | 118.03 (80.16–175.79) | p = 0.002 |
| ADMA/SDMA ratio (median (IQR)) | 0.96 (0.77–1.17) | 0.85 (0.68–0.95) | p = 0.046 | 0.83 (0.67–0.95) | p = 0.013 |
| CIMT (mm) (median (IQR)) | 0.66 (0.57–0.75) | 0.83 (0.69–0.91) | p = 0.001 | 0.85 (0.76–0.92) | p < 0.001 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Grosse, G.M.; Biber, S.; Sieweke, J.-T.; Martens-Lobenhoffer, J.; Gabriel, M.M.; Putzer, A.-S.; Hasse, I.; van Gemmeren, T.; Schuppner, R.; Worthmann, H.; et al. Plasma Dimethylarginine Levels and Carotid Intima–Media Thickness are related to Atrial Fibrillation in Patients with Embolic Stroke. Int. J. Mol. Sci. 2019, 20, 730. https://doi.org/10.3390/ijms20030730
Grosse GM, Biber S, Sieweke J-T, Martens-Lobenhoffer J, Gabriel MM, Putzer A-S, Hasse I, van Gemmeren T, Schuppner R, Worthmann H, et al. Plasma Dimethylarginine Levels and Carotid Intima–Media Thickness are related to Atrial Fibrillation in Patients with Embolic Stroke. International Journal of Molecular Sciences. 2019; 20(3):730. https://doi.org/10.3390/ijms20030730
Chicago/Turabian StyleGrosse, Gerrit M., Saskia Biber, Jan-Thorben Sieweke, Jens Martens-Lobenhoffer, Maria M. Gabriel, Anne-Sophie Putzer, Isabel Hasse, Till van Gemmeren, Ramona Schuppner, Hans Worthmann, and et al. 2019. "Plasma Dimethylarginine Levels and Carotid Intima–Media Thickness are related to Atrial Fibrillation in Patients with Embolic Stroke" International Journal of Molecular Sciences 20, no. 3: 730. https://doi.org/10.3390/ijms20030730
APA StyleGrosse, G. M., Biber, S., Sieweke, J.-T., Martens-Lobenhoffer, J., Gabriel, M. M., Putzer, A.-S., Hasse, I., van Gemmeren, T., Schuppner, R., Worthmann, H., Lichtinghagen, R., Bode-Böger, S. M., Bavendiek, U., & Weissenborn, K. (2019). Plasma Dimethylarginine Levels and Carotid Intima–Media Thickness are related to Atrial Fibrillation in Patients with Embolic Stroke. International Journal of Molecular Sciences, 20(3), 730. https://doi.org/10.3390/ijms20030730





