Copper Binding Features of Tropomyosin-Receptor-Kinase-A Fragment: Clue for Neurotrophic Factors and Metals Link
Abstract
1. Introduction
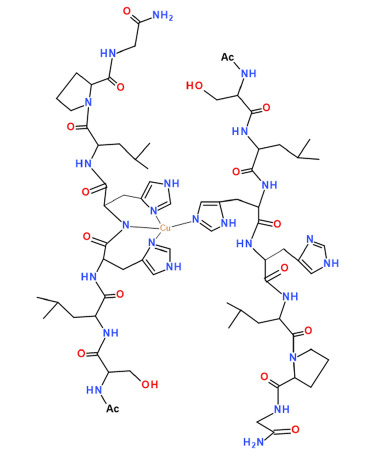
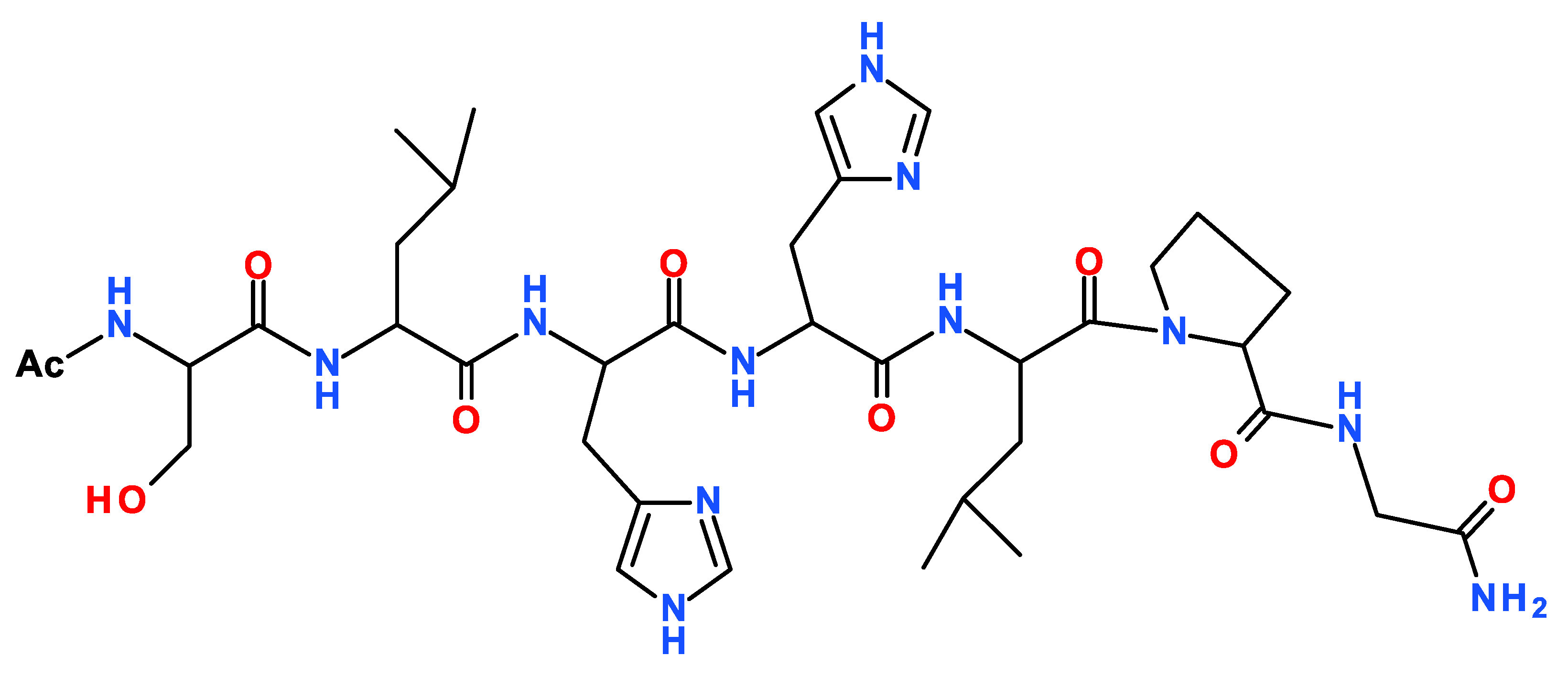
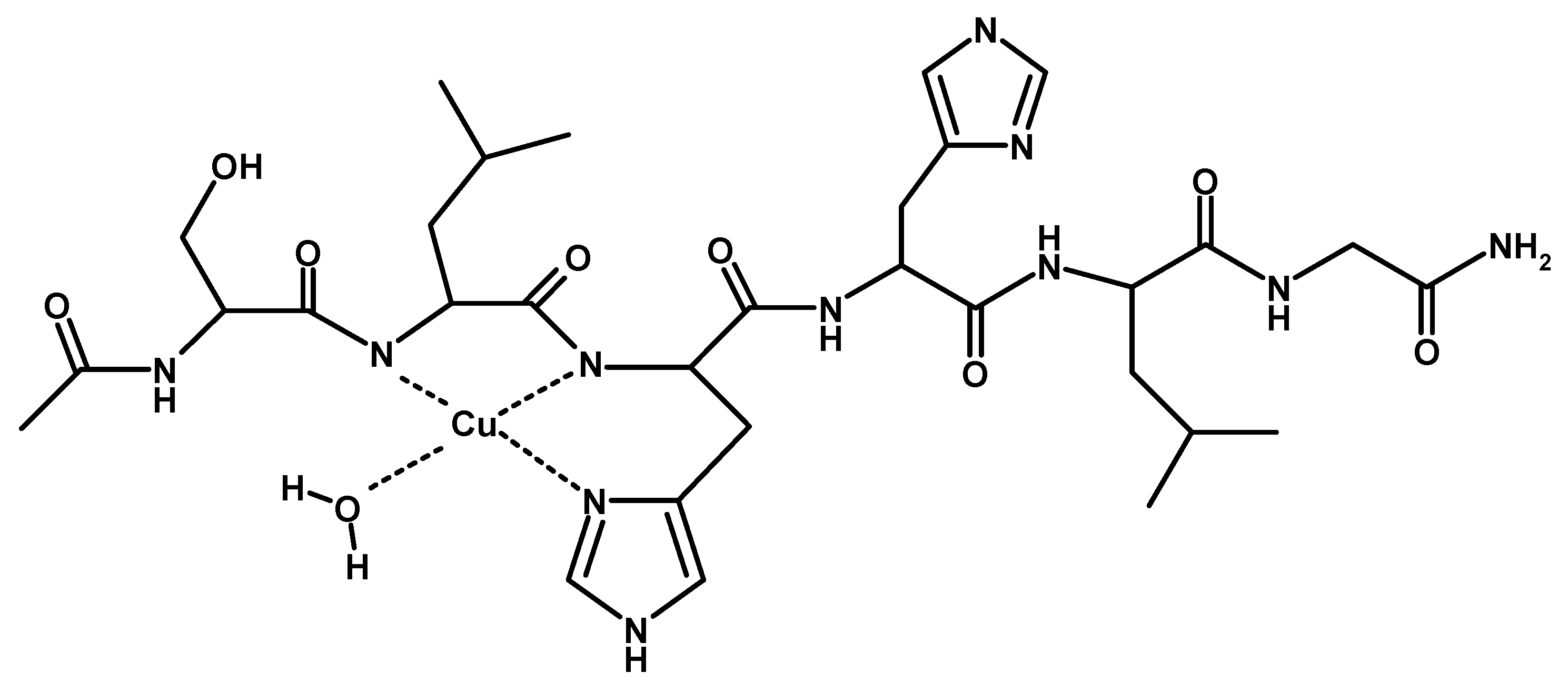
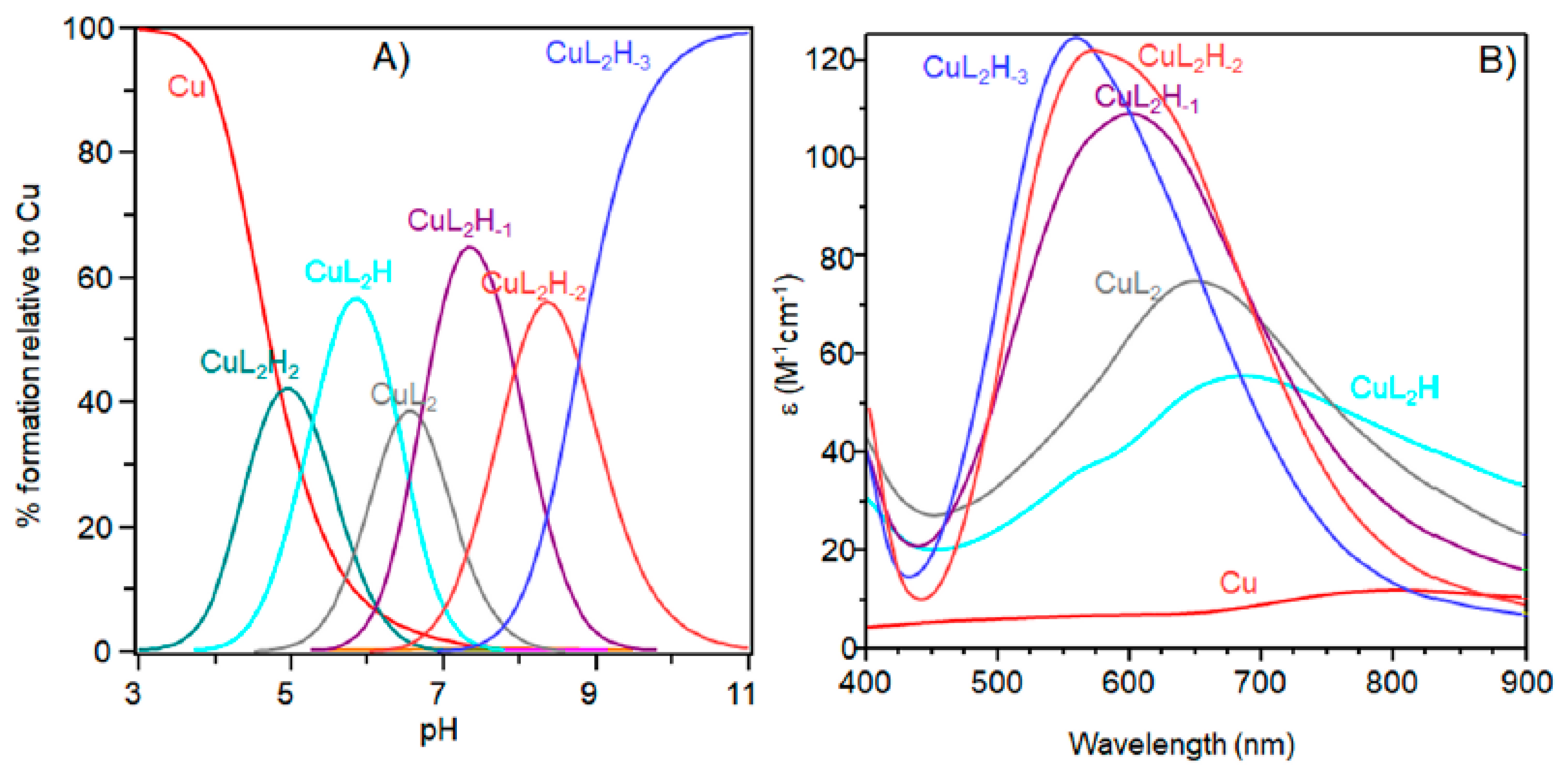
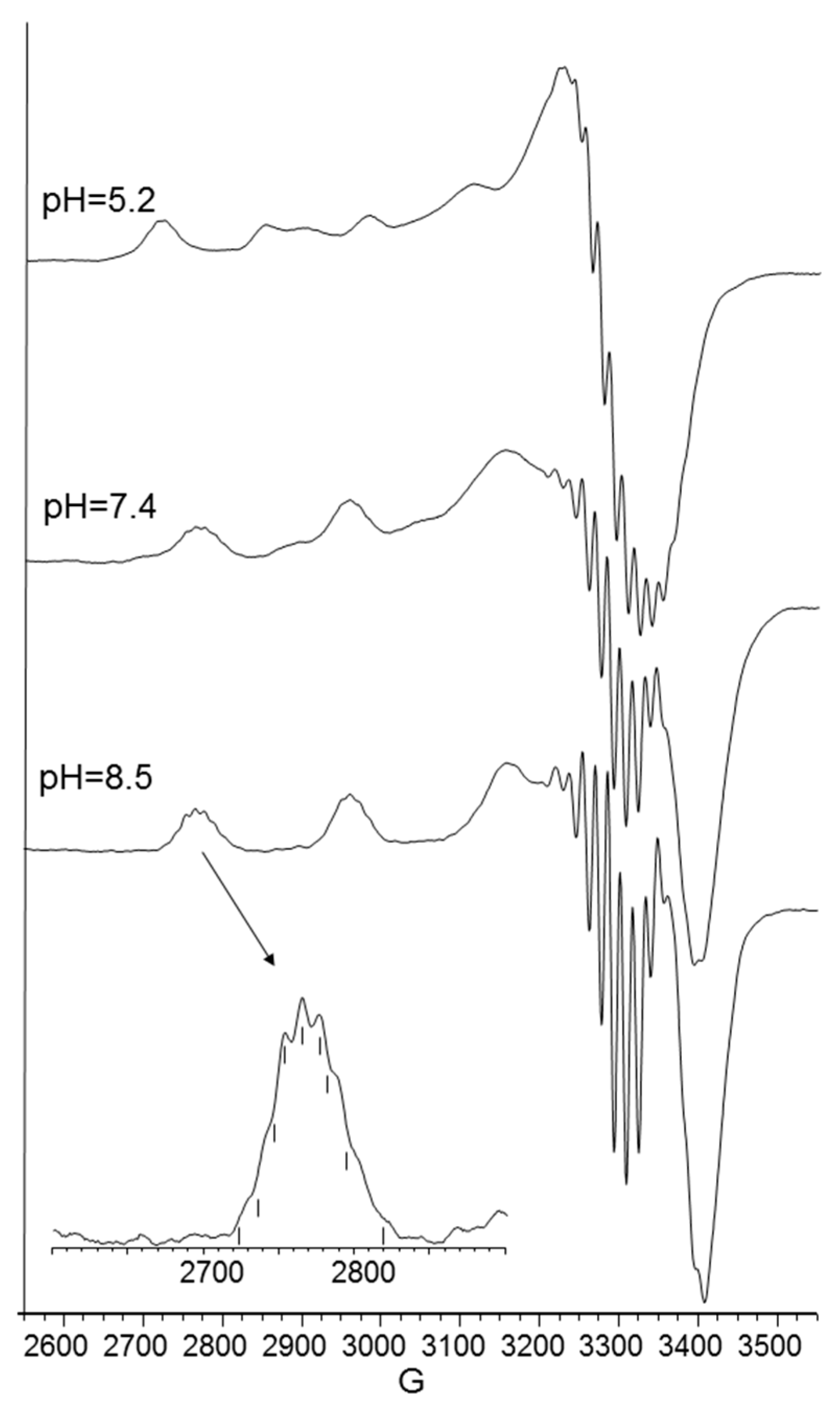
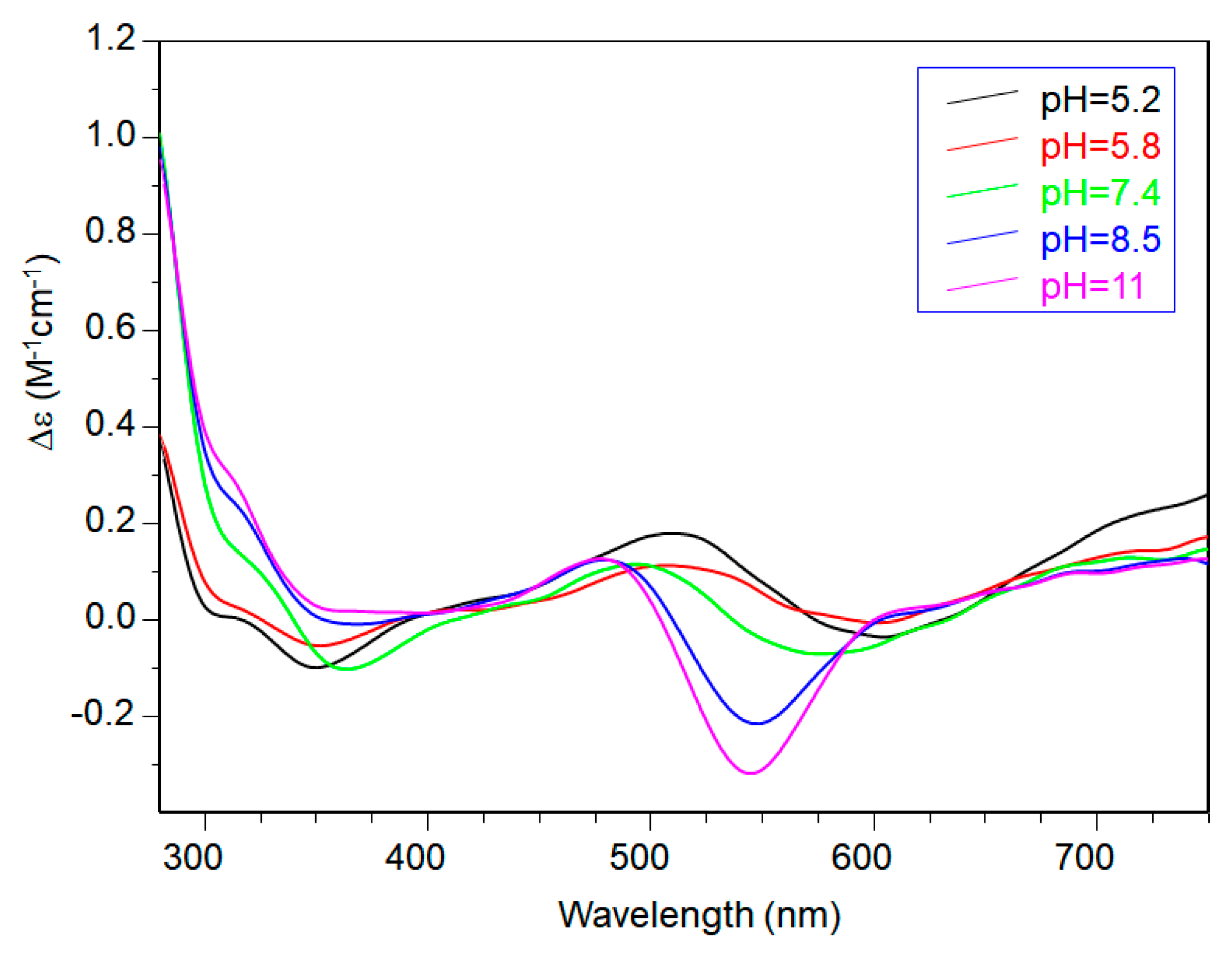
2. Results
3. Discussion
4. Materials and Methods
4.1. Materials
4.2. Peptide Synthesis and Purification
4.3. Potentiometric Titrations
4.4. Spectroscoscopic Studies
4.4.1. UV-Vis Measurements
4.4.2. CD Measurements
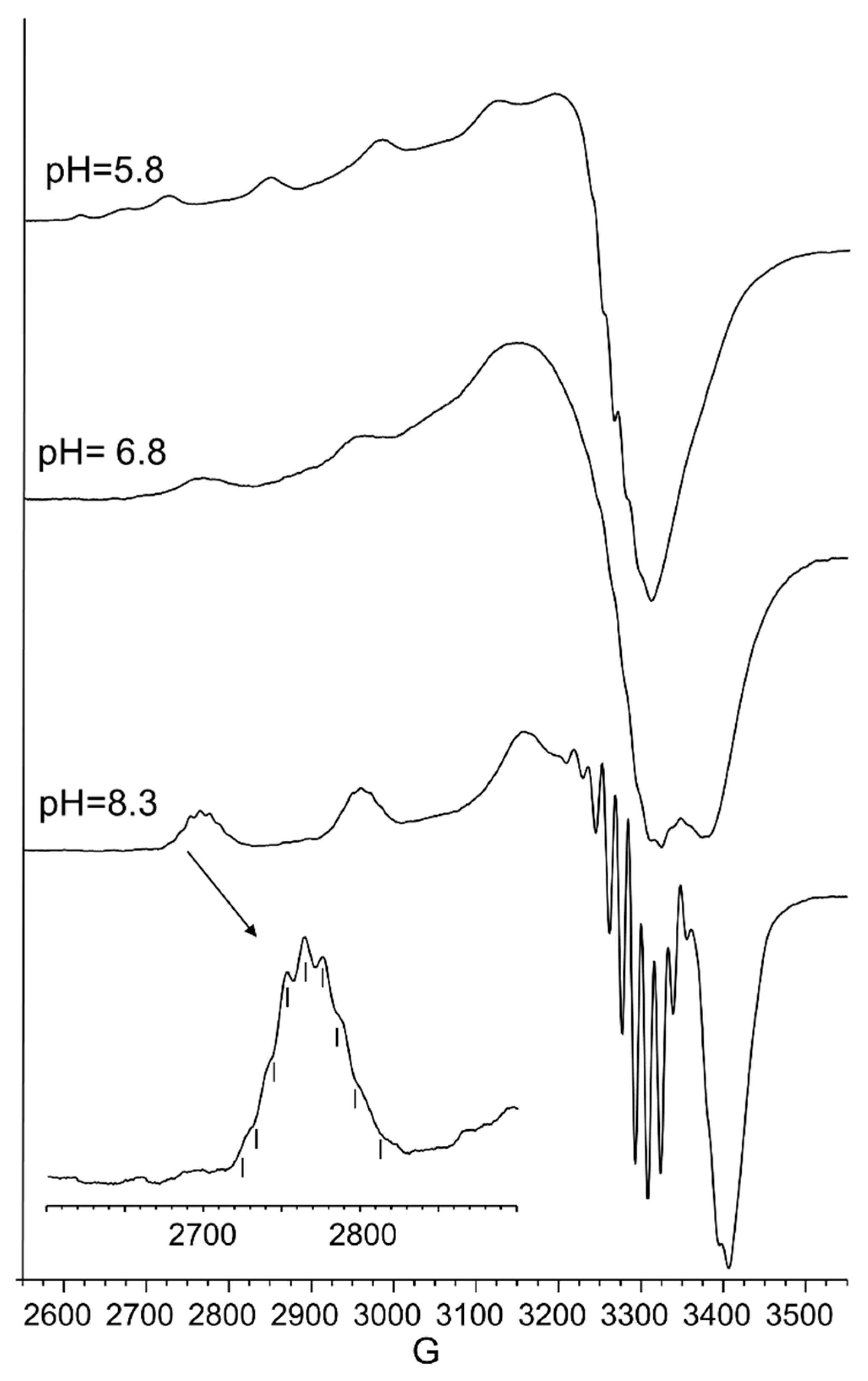
4.4.3. EPR Measurements
Author Contributions
Funding
Conflicts of Interest
References
- Huang, J.E.; Reichardt, L.F. Neurotrophins: Roles in neuronal development and function. Ann. Rev. Neurosci. 2001, 24, 677–736. [Google Scholar] [CrossRef] [PubMed]
- Twiss, J.L.; Chang, J.H.; Schanen, N.C. Pathophysiological Mechanisms for Actions of the Neurotrophins. Brain Pathol. 2006, 16, 320–332. [Google Scholar] [CrossRef] [PubMed]
- Skaper, S.D. Neurotrophic Factors: An Overview. Methods Mol. Biol. 2018, 1727, 1–17. [Google Scholar] [CrossRef] [PubMed]
- Bothwell, M. NGF, BDNF, NT3, and NT4. Handb. Exp. Pharmacol. 2014, 220, 3–15. [Google Scholar] [CrossRef] [PubMed]
- Chao, M.V. Neurotrophins and their receptors: A convergence point for many signalling pathways. Nat. Rev. Neurosci. 2003, 4, 299–309. [Google Scholar] [CrossRef] [PubMed]
- Haddad, Y.; Adam, V.; Heger, Z. Trk Receptors and Neurotrophin Cross-Interactions: New Perspectives Toward Manipulating Therapeutic Side-Effects. Front. Mol. Neurosci. 2017, 10, 130. [Google Scholar] [CrossRef] [PubMed]
- Levi-Montalcini, R. The nerve growth factor 35 years later. Science 1987, 237, 1154–1162. [Google Scholar] [CrossRef] [PubMed]
- Campenot, R.B. NGF uptake and retrograde signaling mechanisms in sympathetic neurons in compartmented cultures. Results Probl. Cell Differ. 2009, 48, 141–158. [Google Scholar] [CrossRef] [PubMed]
- Iulita, M.F.; Cuello, A.C. Nerve growth factor metabolic dysfunction in Alzheimer’s disease and down syndrome. Trends Pharmacol. Sci. 2014, 35, 338–348. [Google Scholar] [CrossRef] [PubMed]
- Triaca, V.; Calissano, P. Impairment of the nerve growth factor pathway driving amyloid accumulation in cholinergic neurons: the incipit of the Alzheimer’s disease story? Neural Regen Res. 2016, 11, 1553–1556. [Google Scholar] [CrossRef] [PubMed]
- Xu, C.J.; Wang, J.L.; Jin, W.L. The Emerging Therapeutic Role of NGF in Alzheimer’s Disease. Neurochem. Res. 2016, 41, 1211–1218. [Google Scholar] [CrossRef] [PubMed]
- Bush, A.I. Drug Development Based on the Metals Hypothesis of Alzheimer’s Disease. J. Alzheimers Dis. 2008, 15, 223–240. [Google Scholar] [CrossRef] [PubMed]
- Barnham, K.J.; Bush, A.I. Metals in Alzheimer’s and Parkinson’s diseases. Curr. Opin. Chem. Biol. 2008, 12, 222–228. [Google Scholar] [CrossRef] [PubMed]
- Mathys, Z.K.; White, A.R. Copper and Alzheimer’s Disease. Adv. Neurobiol. 2017, 18, 199–216. [Google Scholar] [CrossRef] [PubMed]
- Ross, G.M.; Shamovsky, I.L.; Woo, S.B.; Post, J.I.; Vrkljan, P.N.; Lawrence, G.; Solc, M.; Dostaler, S.M.; Neet, K.E.; Riopelle, R.J. The binding of zinc and copper ions to nerve growth factor is differentially affected by pH: implications for cerebral acidosis. J. Neurochem. 2001, 78, 515–523. [Google Scholar] [CrossRef] [PubMed]
- Travaglia, A.; Pietropaolo, A.; La Mendola, D.; Nicoletti, V.G.; Rizzarelli, E. The inorganic perspectives of neurotrophins and Alzheimer’s disease. J. Inorg. Biochem. 2012, 111, 130–137. [Google Scholar] [CrossRef] [PubMed]
- Birkaya, B.; Aletta, J.M. NGF promotes copper accumulation required for optimum neurite outgrowth and protein methylation. J. Neurobiol. 2005, 63, 49–61. [Google Scholar] [CrossRef] [PubMed]
- Ross, G.M.; Shamovsky, I.L.; Lawrance, G.; Solc, M.; Dostaler, S.M.; Jimmo, S.L.; Weaver, D.F.; Riopelle, R.J. Zinc alters conformation and inhibits biological activities of nerve growth factor and related neurotrophins. Nat. Med. 1997, 3, 872–878. [Google Scholar] [CrossRef] [PubMed]
- Wang, W.; Post, J.I.; Dow, K.E.; Shin, S.H.; Riopelle, R.J.; Ross, G.M. Zinc and copper inhibit nerve growth factor-mediated protection from oxidative stress-induced apoptosis. Neurosci. Lett. 1999, 259, 115–118. [Google Scholar] [CrossRef]
- Allington, C.; Shamovsky, I.L.; Ross, G.M.; Riopelle, R.J. Zinc inhibits p75NTR-mediated apoptosis in chick neural retina. Cell Death Differ. 2001, 8, 451–456. [Google Scholar] [CrossRef] [PubMed]
- Travaglia, A.; Arena, G.; Fattorusso, R.; Isernia, C.; La Mendola, D.; Malgeri, G.; Nicoletti, V.G.; Rizzarelli, E. The inorganic perspective of nerve growth factor: interactions of Cu2+ and Zn2+ with the N-terminus fragment of nerve growth factor encompassing the recognition domain of the TrkA receptor. Chem. Eur. J. 2011, 17, 3726–3738. [Google Scholar] [CrossRef] [PubMed]
- Pandini, G.; Satriano, C.; Pietropaolo, A.; Gianì, F.; Travaglia, A.; La Mendola, D.; Nicoletti, V.G.; Rizzarelli, E. The Inorganic Side of NGF: Copper(II) and Zinc(II) Affect the NGF Mimicking Signaling of the N-Terminus Peptides Encompassing the Recognition Domain of TrkA Receptor. Front. Neurosci. 2016, 10, 569. [Google Scholar] [CrossRef] [PubMed]
- Takeda, A.; Tamano, H. Significance of the degree of synaptic Zn²⁺ signaling in cognition. Biometals 2016, 29, 177–185. [Google Scholar] [CrossRef] [PubMed]
- Sindreu, C.; Storm, D.R. Modulation of neuronal signal transduction and memory formation by synaptic zinc. Front. Behav. Neurosci. 2011, 5, 68. [Google Scholar] [CrossRef] [PubMed]
- Grubman, A.; White, A.R. Copper as a key regulator of cell signalling pathways. Expert Rev. Mol. Med. 2014, 16, e11. [Google Scholar] [CrossRef] [PubMed]
- Hwang, J.J.; Park, M.H.; Choi, S.Y.; Koh, J.Y. Activation of the Trk signaling pathway by extracellular zinc. Role of metalloproteinases. J. Biol. Chem. 2005, 280, 11995–12001. [Google Scholar] [CrossRef] [PubMed]
- Pietropaolo, A.; Magrì, A.; Greco, V.; Losasso, V.; la Mendola, D.; Sciuto, S.; Carloni, P.; Rizzarelli, E. Binding of Zn(II) to Tropomyosin Receptor Kinase A in Complex with Its Cognate Nerve Growth Factor: Insights from Molecular Simulation and in Vitro Essays. ACS Chem. Neurosci. 2018, 9, 1095–1103. [Google Scholar] [CrossRef] [PubMed]
- Witkowska, D.; Politano, R.; Rowinska-Zyrek, M.; Guerrini, R.; Remelli, M.; Kozlowski, H. The Coordination of NiII and CuII Ions to the Polyhistidyl Motif of Hpn Protein: Is It as Strong as We Think? Chem. Eur. J. 2012, 18, 11088–11099. [Google Scholar] [CrossRef] [PubMed]
- Pontecchiani, F.; Simonovsky, E.; Wieczorek, R.; Barbosa, N.; Rowinska-Zyrek, M.; Potocki, S.; Remelli, M.; Miller, Y.; Kozlowski, H. The unusual binding mechanism of Cu(II) ions to the poly-histidyl domain of a peptide found in the venom of an African viper. Dalton Trans. 2014, 43, 16680–16689. [Google Scholar] [CrossRef] [PubMed]
- Hernández-Guzmán, J.; Sun, L.; Mehta, A.K.; Dong, J.; Lynn, D.G.; Warncke, K. Copper(II)-bis-Histidine Coordination Structure in a Fibrillar Amyloid β-Peptide Fragment and Model Complexes Revealed by Electron Spin Echo Envelope modulation Spectroscopy. Chembiochem 2013, 14, 1762–1771. [Google Scholar] [CrossRef] [PubMed]
- Mital, M.; Wezynfeld, N.E.; Frączyk, T.; Wiloch, M.Z.; Wawrzyniak, U.E.; Bonna, A.; Tumpach, C.; Barnham, K.J.; Haigh, C.L.; Bal, W.; et al. A Functional Role for Aβ in Metal Homeostasis? N-Truncation and High-Affinity Copper Binding. Angew. Chem. Int. Ed. Engl. 2015, 54, 10460–10464. [Google Scholar] [CrossRef] [PubMed]
- Magrì, A.; La Mendola, D.; Nicoletti, V.G.; Pappalardo, G.; Rizzarelli, E. New Insight in Copper-Ion Binding to Human Islet Amyloid: The Contribution of Metal-Complex Speciation to Reveal the Polypeptide Toxicity. Chem. Eur. J. 2016, 22, 13287–13300. [Google Scholar] [CrossRef]
- Sanna, D.; Micera, G.; Kallay, C.; Rigo, V.; Sovago, I. Copper(II) complexes of N-terminal protected tri- and tetra-peptides containing histidine residues. Dalton Trans. 2004, 2702–2707. [Google Scholar] [CrossRef] [PubMed]
- Jancsó, A.; Kolozsi, A.; Gyurcsik, B.; Nagy, N.V.; Gajda, T. Probing the Cu2+ and Zn2+ binding affinity of histidine-rich glycoprotein. J. Inorg. Biochem. 2009, 103, 1634–1643. [Google Scholar] [CrossRef] [PubMed]
- Matera, A.; Brasuń, J.; Cebrat, M.; Świątek-Kozłowska, J. The role of the histidine residue in the coordination abilities of peptides with a multi-histidine sequence towards copper(II) ions. Polyhedron 2008, 27, 1539–1555. [Google Scholar] [CrossRef]
- Timári, S.; Kállay, C.S.; Osz, K.; Sóvágó, I.; Várnagy, K. Transition metal complexes of short multihistidine peptides. Dalton Trans. 2009, 1962–1971. [Google Scholar] [CrossRef] [PubMed]
- Kulon, K.; Valensin, D.; Kamysz, W.; Valensin, G.; Nadolski, P.; Porciatti, E.; Gaggelli, E.; Kozlowski, H. The His-His sequence of the antimicrobial peptide demegen P-113 makes it very attractive ligand for Cu2+. J. Inorg. Biochem. 2008, 102, 960–972. [Google Scholar] [CrossRef] [PubMed]
- Myari, A.; Malandrinos, G.; Deligiannakis, Y.; Plakatouras, J.C.; Hadjiliadis, N.; Nagy, Z.; Sovago, I. Interaction of Cu2+ with His-Val-His and of Zn2+ with His-Val-Gly-Asp, two peptides surrounding metal ions in Cu,Zn-superoxide dismutase enzyme. J. Inorg. Biochem. 2001, 85, 253–261. [Google Scholar] [CrossRef]
- Valensin, D.; Luczkowski, M.; Mancini, F.M.; Legowska, A.; Gaggelli, E.; Valensin, G.; Rolka, K.; Kozlowski, H. The dimeric and tetrameric octarepeat fragments of prion protein behave differently to its monomeric unit. Dalton Trans. 2004, 1284–1293. [Google Scholar] [CrossRef] [PubMed]
- Sóvágó, I.; Osz, K. Metal ion selectivity of oligopeptides. Dalton Trans. 2006, 3841–3854. [Google Scholar] [CrossRef] [PubMed]
- Magrì, A.; Tabbì, G.; Breglia, R.; de Gioia, L.; Fantucci, P.; Bruschi, M.; Bonomo, R.P.; la Mendola, D. Copper ion interaction with the RNase catalytic site fragment of the angiogenin protein: An experimental and theoretical investigation. Dalton Trans. 2017, 46, 8524–8538. [Google Scholar] [CrossRef] [PubMed]
- La Mendola, D.; Magrì, A.; Santoro, A.M.; Nicoletti, V.G.; Rizzarelli, E. Copper(II) interaction with peptide fragments of histidine-proline-rich glycoprotein: Speciation, stability and binding details. J. Inorg. Biochem. 2012, 111, 59–69. [Google Scholar] [CrossRef] [PubMed]
- Hecel, A.; Wątły, J.; Rowińska-Zyrek, M.; Świątek-Kozłowska, J.; Kozłowski, H. Histidine tracts in human transcription factors: insight into metal ion coordination ability. J. Biol. Inorg. Chem. 2018, 23, 81–90. [Google Scholar] [CrossRef] [PubMed]
- Kowalik-Jankowska, T.; Ruta, M.; Wiśniewska, K.; Lankiewicz, L. Coordination abilities of the 1-16 and 1-28 fragments of beta-amyloid peptide towards copper(II) ions: A combined potentiometric and spectroscopic study. J. Inorg. Biochem. 2003, 95, 270–282. [Google Scholar] [CrossRef]
- Amorini, A.M.; Bellia, F.; di Pietro, V.; Giardina, B.; la Mendola, D.; Lazzarino, G.; Sortino, S.; Tavazzi, B.; Rizzarelli, E.; Vecchio, G. Synthesis and antioxidant activity of new homocarnosine beta-cyclodextrin conjugates. Eur. J. Med. Chem. 2007, 42, 910–920. [Google Scholar] [CrossRef] [PubMed]
- Rajković, S.; Kállay, C.; Serényi, R.; Malandrinos, G.; Hadjiliadis, N.; Sanna, D.; Sovago, I. Complex formation processes of terminally protected peptides containing two or three histidyl residues. Characterization of the mixed metal complexes of peptides. Dalton Trans. 2008, 5059–5071. [Google Scholar] [CrossRef] [PubMed]
- Potocki, S.; Valensin, D.; Kozlowski, H. The specificity of interaction of Zn2+, Ni2+ and Cu2+ ions with the histidine-rich domain of the TjZNT1 ZIP family transporter. Dalton Trans. 2014, 10215–10223. [Google Scholar] [CrossRef] [PubMed]
- La Mendola, D.; Bonomo, R.P.; Caminati, S.; Di Natale, G.; Emmi, S.S.; Hansson, Ö.; Maccarrone, G.; Pappalardo, G.; Pietropaolo, A.; Rizzarelli, E. Copper(II) complexes with an avian prion N-terminal region and their potential SOD-like activity. J. Inorg. Biochem. 2009, 103, 195–204. [Google Scholar] [CrossRef] [PubMed]
- Gans, P.; Sabatini, A.; Vacca, A. Investigation of equilibria in solution. Determination of equilibrium constants with the HYPERQUAD suite of programs. Talanta 1996, 43, 1739–1753. [Google Scholar] [CrossRef]
- Alderighi, L.; Gans, P.; Ienco, A.; Peters, D.; Sabatini, A.; Vacca, A. Hyperquad simulation and speciation (HySS): A utility program for the investigation of equilibria involving soluble and partially soluble species. Coord. Chem. Rev. 1999, 184, 311–318. [Google Scholar] [CrossRef]
- Remenyi, C.; Reviakine, R.; Kaupp, M. Density Functional Study of EPR Parameters and Spin-Density Distribution of Azurin and Other Blue Copper Proteins. J. Phys. Chem. B 2007, 111, 8290–8304. [Google Scholar] [CrossRef] [PubMed]


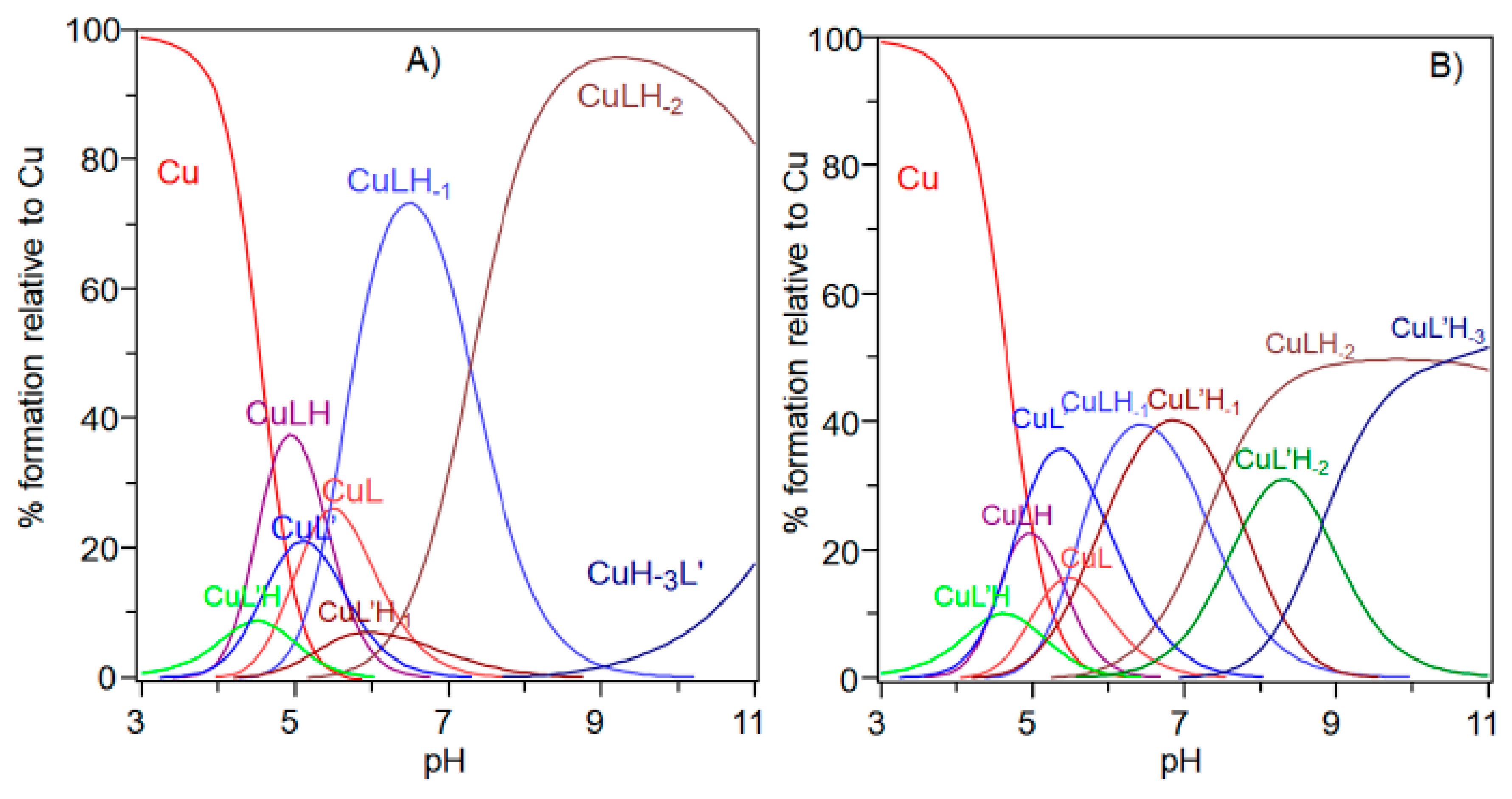
| Species (pqr) | logβ | pK (n/m) |
|---|---|---|
| CuLH | 10.61 (9) | - |
| CuL | 6.19 (3) | 4.42 |
| CuLH−1 | 0.23 (5) | 5.97 |
| CuLH−2 | −7.55 (4) | 7.78 |
| CuLH−3 | −16.36 (6) | 8.80 |
| pH | Species | UV-Vis | CD | EPR | ||
|---|---|---|---|---|---|---|
| λ (nm) (ε (M−1·cm−1)) | λ (nm) (Δε (M−1·cm−1)) | g║║ | A║║ (10−4 cm−1) | |||
| 5.8 | CuL | 650 | (50) | 280 (0.65), 350 (−0.30), 511 (0.30), 723 (0.25) | 2.315 (2) | 145 (3) |
| 6.8 | CuLH−1 | 595 | (95) | 280 (1.40), 362 (−0.22), 482 (0.17), 585 (−0.71), 723 (0.30) | 2.210 (3) | 194 (5) |
| 8.3 | CuLH−2 | 550 | (105) | 280 (1.52), 362 (−0.10), 474 (0.24), 549 (−0.80), 717 (0.19) | 2.206 (1) | 202 (2) |
| 9–11 | CuLH−3 | 550 | (95) | 280 (1.90), 477 (0.23), 547 (−0.78) | 2.206 (1) | 202 (2) |
| Species (pqr) | logβ | pK (n/m) |
|---|---|---|
| CuL2H2 | 22.12 (5) | - |
| CuL2H | 16.85 (3) | 5.27 |
| CuL2 | 10.46 (5) | 6.39 |
| CuL2H−1 | 3.77 (4) | 6.69 |
| CuL2H−2 | −4.17 (4) | 7.94 |
| pH | Species | UV-Vis | CD | EPR | ||
|---|---|---|---|---|---|---|
| λ (nm) (ε (M−1·cm−1)) | λ (nm) (Δε (M−1·cm−1)) | g║ | A║ (10−4 cm−1) | |||
| 5.2 | CuL2H2 | 675 | (55) | − | 2.310 (2) | 142 (3) |
| 5.8 | CuL2H | 650 | (75) | 280 (0.44), 349 (0.06), 516 (0.13), 600 (−0.03), 725 (0.13) | − | − |
| 7.4 | CuL2H−1 | 598 | (110) | 280 (1.16), 360 (−0.11), 491 (0.14), 568 (−0.09), 723 (0.12) | 2.249 (2) | 192 (3) |
| 8.5 | CuL2H−2 | 555 | (120) | 280 (1.20), 474 (0.15), 549 (−0.23), 723 (0.12) | 2.206 (1) | 202 (2) |
| 11 | CuL2H−3 | 550 | (125) | 280 (1.05), 481 (0.15), 547 (−0.35) | 2.206 (1) | 202 (2) |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Magrì, A.; La Mendola, D. Copper Binding Features of Tropomyosin-Receptor-Kinase-A Fragment: Clue for Neurotrophic Factors and Metals Link. Int. J. Mol. Sci. 2018, 19, 2374. https://doi.org/10.3390/ijms19082374
Magrì A, La Mendola D. Copper Binding Features of Tropomyosin-Receptor-Kinase-A Fragment: Clue for Neurotrophic Factors and Metals Link. International Journal of Molecular Sciences. 2018; 19(8):2374. https://doi.org/10.3390/ijms19082374
Chicago/Turabian StyleMagrì, Antonio, and Diego La Mendola. 2018. "Copper Binding Features of Tropomyosin-Receptor-Kinase-A Fragment: Clue for Neurotrophic Factors and Metals Link" International Journal of Molecular Sciences 19, no. 8: 2374. https://doi.org/10.3390/ijms19082374
APA StyleMagrì, A., & La Mendola, D. (2018). Copper Binding Features of Tropomyosin-Receptor-Kinase-A Fragment: Clue for Neurotrophic Factors and Metals Link. International Journal of Molecular Sciences, 19(8), 2374. https://doi.org/10.3390/ijms19082374