Abstract
Cancer is known for its cellular changes contributing to tumour growth and cell proliferation. As part of these changes, metabolic rearrangements are identified in several cancers, including multiple myeloma (MM), which is a condition whereby malignant plasma cells accumulate in the bone marrow (BM). These metabolic changes consist of generation, inhibition and accumulation of metabolites and metabolic shifts in MM cells. Changes in the BM micro-environment could be the reason for such adjustments. Enhancement of glycolysis and glutaminolysis is found in MM cells compared to healthy cells. Metabolites and enzymes can be upregulated or downregulated and play a crucial role in drug resistance. Therefore, this review will focus on changes in glucose and glutamine metabolism linked with the emergence of drug resistance. Moreover, metabolites do not only affect other metabolic components to benefit cancer development; they also interfere with transcription factors involved in proliferation and apoptotic regulation.
1. Introduction
In this review, we focus on the mechanisms of cancer metabolism in multiple myeloma (MM), which is a hematologic malignancy described as an expansion of dysfunctional plasma cells in the bone marrow (BM). This expansion and accumulation of malignant plasma cells is linked with high levels of monoclonal proteins (M-spike) in serum and urine [1]. The disease is usually preceded by a premalignant condition known as monoclonal gammopathy of undetermined significance (MGUS), whence MM arises [1,2]. The expansion of malignant plasma cells and the production of M-protein in excess causes the following frequently observed symptoms in MM: hypercalcemia, renal failure, anaemia and bone lesions (CRAB features) [3,4]. Reports estimate yearly 41,719 new cases in Europe and 20,462 deaths. Worldwide, MM affects 1–5 per 100,000 individuals every year. Despite the progress in treatment, MM remains an incurable disease and the prevalence continues to increase due to the ageing population, with a median age of 73–75 at diagnosis [5,6,7]. Early therapies were the combination of melphalan with prednisone followed by melphalan and autologous hematopoietic stem cell transplantation. Over the last decade, novel therapies improved survival in MM patients, such as the proteasome inhibitor bortezomib and the immunomodulatory drugs (IMiDs) thalidomide, lenalidomide and pomalidomide. Also, promising preliminary results with the monoclonal antibody daratumumab have been shown. These drugs are also combined with dexamethasone, an immunosuppressive drug [8,9]. The rate of complete response and the overall survival have improved with these drugs [9]. Despite these improvements in overall survival, MM patients eventually relapse. The main cause of this relapse is the development of drug resistance. This can be either intrinsically by mutational changes in genes or ribosomal proteins (monoallelic loss of 60S ribosomal proteins) where decreased ribosomal proteins are correlated with tumour progression and drug resistance; or extrinsically by the BM micro-environment [10,11].
It is well known that tumour growth and metastasis are increased by intense and complex neovascularisation in solid tumours [12,13]. Similarly, despite MM being a non-solid tumour situated in the BM, blood vessel development is increased through secretion of cytokines such as vascular endothelial growth factor (VEGF) and fibroblast growth factor-2 (FGF-2). Also, interleukin-6 (IL-6) is secreted by BM endothelial cells, which promote cell growth in MM cells. This causes an increase in vascularisation, which promotes MM progression and is correlated with poor survival. While anti-angiogenic therapy has been investigated as anti-cancer agent in MM, this could have some side effects. Angiogenesis leads to higher oxygen access, resulting in a less hypoxic environment. The use of anti-angiogenic therapy would therefore result in a more hypoxic environment, together with higher glycolysis rates of the tumour cells [1,14,15,16].
Besides the effect of IL-6, there is a synergistic activity of IL-6 and IL-3, leading to plasma cell differentiation from peripheral blood mononuclear cells [17].
The BM micro-environment consists of two different compartments: a cellular and a non-cellular compartment. The former consists of hematopoietic cells important for the immune system on the one hand and non-hematopoietic cells such as bone marrow stromal cells (BMSC), fibroblasts, osteoblasts and blood vessels on the other hand [1,18]. The non-cellular compartment consists of fibronectin, laminin and collagen [19]. Malignant cells have an impact on the functionality of the BM micro-environment that favours their survival and growth. Indeed, MM cells interact with BMSC by adherence, resulting in the activation of cell cycle pathways and anti-apoptotic pathways such as Janus kinase (JAK)/signal transducer and activator of transcription 3 (STAT3), which leads to upregulation of the anti-apoptotic proteins BcL-xL and Mcl-1. NF-κB signaling is also activated with inhibitors of apoptosis proteins (IAP) as an anti-apoptotic protein [1,20]. These effects of MM cells on the BM can explain the presence of anaemia in MM patients since the infiltrating MM cells can be disadvantageous to marrow erythropoietic niches, which could lead to erythroid cell apoptosis due to MM cells producing specific cytokines, such as Fas ligand (FL), tumour necrosis factor (TNF) and TNF-related apoptosis-inducing ligand (TRAIL) [21].
MM is further characterised by osteolysis, whereby malignant plasma cells activate osteoclast (OC) progenitors and initiate osteoclastic bone resorption. Osteoclast formation is stimulated by the adherence of MM cells in the BM via vascular cell adhesion molecule 1 (VCAM-1) and α4β1 integrin. Together with the receptor activator of nuclear factor κB ligand (RANKL), they belong to the osteoclastogenic factors inducing osteolysis [22,23]. Furthermore, interleukin-1β (IL-1β) and TNF-β, secreted by the malignant plasma cells, are defined as osteoclast-activating factors (OAF) due to their osteoclast activation and bone reabsorption function [24].
Besides the changes induced by MM cells in the BM micro-environment, these malignant plasma cells experience metabolic changes themselves compared to healthy plasma cells. Known metabolic rearrangements in MM cells are adjustments in the glucose pathway, glutamine pathway, serine metabolism, pentose phosphate pathway (PPP) and folate pathway. Furthermore, the overall changes in cell metabolism and the BM environment induce drug resistance in MM.
2. General Cancer Metabolism
Every healthy cell is obliged to import nutrients from the environment to fulfil biosynthetic demands. This allows proliferation, differentiation and migration. Glucose and glutamine are indispensable for survival of mammalian cells [25]. Indeed, glucose can enter the cell through different glucose transporters: GLUT1 (erythrocytes, vascular endothelium), GLUT2 (hepatocytes, pancreatic β-cells, intestinal mucosa and renal cells), GLUT3 (neurons) and GLUT4 (skeletal and cardiac muscle), wherein each membrane protein is expressed depending on the cell type [26]. The process of glycolysis starts once glucose enters the cell; it goes through several conversions in the cytosol, ending in the generation of 2 moles pyruvate and 2 moles adenosine triphosphate (ATP). At first glucose is converted into glucose-6-phosphate after addition of a phosphate group, which prevents the efflux of glucose. Fructose-6-phosphate is formed and followed by fructose-2,6-biphosphate, d-glyceraldehyde 3-phosphate, 1,3-biphosphoglycerate, 3-phosphoglycerate, 2-phosphoglycerate and phosphoenolpyruvate (PEP). In a final step, pyruvate is formed after the conversion of PEP, which is catalysed by pyruvate kinase (PK) [27].
Next, pyruvate comes into the matrix of the mitochondria and is oxidised to acetyl-CoA, by the enzymatic activity of pyruvate dehydrogenase (PDH), which is the beginning of the tricarboxylic acid cycle (TCA), also called the Krebs cycle, under aerobic conditions. The fusion of acetyl-CoA with oxaloacetate forms citrate as the next metabolite in the TCA cycle [28]. Next, isocitrate is generated, followed by α-ketoglutarate (αKG), succinate, fumarate, malate and oxaloacetate (Figure 1). The cycle is completed by arrival at oxaloacetate and then starts over again [29]. Each rearrangement step releases energy, in the form of electrons, which are accepted by the so-called electron shuttles such as nicotinamide adenine dinucleotide (NAD+) and flavin adenine dinucleotide (FAD). These molecules are responsible for the transportation of the high-energetic electrons. When energy is released, the electron shuttles capture the energy and are reduced to nicotinamide adenine dinucleotide hydrogen (NADH) and FADH2. At the end, the electron shuttles are transported to the electron transport chain, located in the inner membrane of the mitochondria, to generate ATP [30]. The oxidative phosphorylation (OXPHOS) is a very efficient mechanism that generates 36 moles ATP out of 1 mole glucose in aerobic conditions. However, under anaerobic conditions, lactate is generated as a result of the reduction of pyruvate in the cytosol and then excreted out of the cell through monocarboxylate transporters (MCTs) [28]. The reduction of pyruvate into lactate is realised through lactate dehydrogenase (LDH).
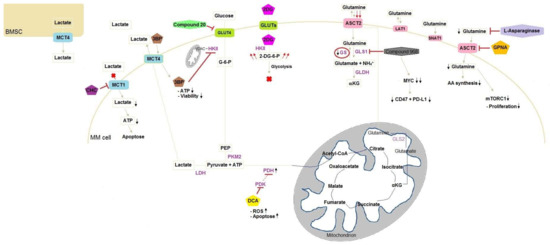
Figure 1.
Schematic presentation of targets in glucose and glutamine metabolism in MM cells. Metabolic rearrangements in MM cells after administering compounds inducing stimulation or inhibition. 3BP enters the cell through MCT4 and inhibits HKII connected to the outer mitochondrial membrane via VDAC leading to ATP reduction and the loss of viable cells. 2DG enters the cell through GLUT and is phosphorylated by HKII. The phosphorylated form cannot be metabolised and accumulates by consequence leading to glycolysis blockage. Compound 20 inhibits GLUT4 resulting in chemosensitising. The inhibition of PDK on PDH is suppressed after addition of DCA to MM cells, which inhibits PDK. As a result, PDH activity increases and lactate production decreases, whereby TCA activity is increased. CHC is a MCT1 competitive inhibitor and blocks the entrance of lactate into the cell. This lack of fuel results in apoptosis due to ATP reduction. However, BMSC supply MM cells with lactate. Glutamine enters the cell through three transporters: ASCT2, LAT1 and SNAT1, with ASCT2 being the major glutamine transporter. Compound 968 inhibits GLS inducing MYC degradation and decrease of CD47 and PD-L1. MM cells lack GS, resulting in low intracellular glutamine concentrations and leading to higher glutamine influx. GPNA is a glutamine transporter inhibitor and induces a lack of glutamine intracellularly, which disadvantages AA synthesis and reduces mTORC1 activity, resulting in less cell proliferation. Instead of inhibiting glutamine transporters, l-asparaginase hydrolyses glutamine. AA, amino acid; BMSC, bone marrow stromal cells; 3BP, 3-bromopyruvate; CHC, α-cyano-4-hydroxycinnamic acid; DCA, dichloroacetate; 2DG, 2-deoxyglucose; 2-DG-6-P, 2-deoxyglucose-6-phosphate; G-6-P, glucose-6-phosphate; GLDH, glutamate dehydrogenase; GLS1, glutaminase 1; GLS2, glutaminase 2; GPNA, l-γ-glutamyl-p-nitroanilide; GS, glutamine synthetase; HKII, hexokinase II; LDH, lactate dehydrogenase; MM, multiple myeloma; MYC, myelocytomatosis oncogene cellular homolog; PDH, pyruvate dehydrogenase; PDK, pyruvate dehydrogenase kinase; PEP, phosphoenolpyruvate; PKM2, pyruvate kinase M2; ROS, reactive oxygen species; VDAC, voltage-dependent anion channel;  , reduction;
, reduction;  , strong reduction;
, strong reduction;  , increase;
, increase;  , strong increase.
, strong increase.
 , reduction;
, reduction;  , strong reduction;
, strong reduction;  , increase;
, increase;  , strong increase.
, strong increase.
Besides glucose, glutamine is by far one of the most abundant amino acids and crucial for the maintenance and promotion of normal cell function. Glutamine enhances proliferation, differentiation, cytokine production and apoptosis. Also, it is the precursor for nucleotide and nucleic acid synthesis [31]. First, glutamine can be transported into the cell via several transporters such as the neutral amino acid transporter (ASCT2) (Figure 1). After the passage into the cell, glutamine can then be used for biosynthesis [32,33]. The process of glutaminolysis takes place in two different parts of the cell similar to glucose metabolism: the cytosol and mitochondria. First, glutamine can be converted to glutamate by glutaminase 1 (GLS1) in the cytosol. In the mitochondria it is converted by glutaminase 2 (GLS2), where it can further be oxidised to αKG by glutamate dehydrogenase (GLDH) or aminotransferases and then participate in the TCA cycle as explained above by generating mitochondrial NADH, NADPH and ammonia [29,34,35,36]. Besides the participation in the TCA cycle, glutamine is also used in biosynthesis of nucleic acids [36]. Formation of purine nucleotides start with the conversion of 5-phospho-α-ribosyl-1-pyrophosphate (PRPP) by glutamine PRPP amidotransferase where glutamine donates an amino group to PRPP and attributes further a nitrogen and ends in inosine monophosphate, which is a fully formed purine nucleotide, after a series of reactions. In this nucleic acid synthesis, glycine, aspartate, ATP and tetrahydrofolate are also used [37,38].
The entrance of glucose, glutamine and various other nutrients into the cell is the very start of this complex metabolic machinery, crucial for the cellular respiration that sparks the conversion of these nutrients into biochemical energy. When comparing glycolysis with glutaminolysis, both processes result in energy production and nucleotide synthesis. The difference is that glycolysis provides more lactate and reduces mitochondrial atrophy. Glutaminolysis results in amino acid synthesis and fatty acid synthesis. These mechanisms together contribute to the growth and survival of the cancer cell [39]. However, cancer cells are characterised by an altered metabolism and this varies from one cancer type to another. Due to the high heterogeneity of the disease, the metabolic features are very diverse depending on the cancer type [28,40]. Nevertheless, most malignant cells have an enhanced aerobic glycolysis, also known as the ‘Warburg effect’, in common. This effect is described as the conversion of glucose to lactate in the presence of oxygen [41]. As tumour cells often reside in a hypoxic environment, the constitutive and high glycolytic flux is probably an adaptation of the malignant cells to the environmental stress [30]. The mitochondrial OXPHOS was considered as weakened or impaired by Warburg for the explanation of the high glycolytic flux of cancer cells. However, recent investigations have found that the mitochondrial OXPHOS mechanism remains unharmed and that OXPHOS is suppressed due to the enhanced glycolysis rather than a defect in the mechanism [28,42]. This effect is reversible: when glycolysis is inhibited, mitochondrial OXPHOS can regain its function, according to observations made by Fantin et al. [28,43]. Further, the increased glucose consumption can be used as a successful diagnostic tool via the imaging technique positron emission tomography (PET) where a glucose analogue, radioactively fluorine labelled (18F-fluorodeoxyglucose (18F-FDG)), is used as a tracer [25,41].
After glucose, glutamine is most often used for energy in cancer cells. One of the processes that provide anaplerotic flux is glutaminolysis, through the generation of αKG [29]. The amino acid is responsible for various important formations of components that are used for cell proliferation [44]. As described earlier in this review, glutamine enters the cell through many different transporters, and can directly be useful as such by its amido nitrogen present in the molecular structure of this amino acid for hexosamine and nucleotide synthesis in cancer cells [44,45]. In a further step, glutamine can be converted to glutamate and then to αKG that enters the TCA cycle for energy production. Furthermore, de novo synthesis of glutamine has been shown by He et al. in C6 glioma cells in which glutamine synthetase (GS), located in the cytoplasm, catalyses glutamine synthesis through ammonia and glutamate [44,46]. He et al., also reported that when C6 cells were deprived of glutamine, GS expression was upregulated and caused de novo glutamine synthesis [46]. Moreover, this observation can be useful in PET tracer studies: 13N-ammonia is taken up by tumour cells executing de novo glutamine synthesis and this gives information about the glutaminolysis rate in tumour cells [46]. Another hallmark is the capacity of glutamine to import essential amino acids. Indeed, Nicklin et al. showed that the entry of l-leucine (an essential amino acid) via the human l-type amino acid transporter 1 (LAT1) causes efflux of glutamine at the same time [47]. Glutamine can import other essential amino acids through that same mechanism [28].
Additionally, it is important to mention that the extensive glucose and glutamine uptake in cancer cells is a result of extracellular stimuli such as growth factor signaling [48]. A cell deprived of growth factors is shown to be negatively affected in terms of cell size and ATP generation despite the presence of glucose in medium. The cell is unable to maintain normal cellular bioenergetics, which can lead to activation of programmed cell death [49]. Overall, it is evident that environmental factors alter tumour metabolism in several cancers.
Besides the importance of glucose and glutamine metabolism in cancer, the folate metabolism is also linked with cancer. It is reported that low folate levels promote carcinogenesis and are associated with cytogenetic abnormalities. Moreover, low folate levels are shown to play a role in the neoplastic process [50]. Next to folate, proline also has importance in cancer. Proline is an amino acid with high abundance in the micro-environment. Proline dehydrogenase/oxidase (PRODH/POX) catalyses the conversion of proline into pyrroline-5-carboxylate (P5C). During this conversion, PRODH/POX donates an electron to the electron transport chain and results in the generation of reactive oxygen species (ROS). This initiates apoptosis and inhibition of tumour growth and cell proliferation, which can be useful as a target in cancers. However, proline biosynthesis, enhanced by myelocytomatosis oncogene cellular homolog (MYC) through glutamine, contributes to tumorigenesis. Indeed, MYC stimulates glutaminolysis through miR-23a/b, which is connected with proline synthesis [51].
3. Glucose Metabolism in multiple myeloma (MM)
In cancer research, glucose metabolism is the most studied branch in cancer metabolism. However, glycolysis has not yet been fully elucidated in MM. A first interesting enzyme in the glycolysis pathway is hexokinase II (HKII), which is part of four HKs isoforms. It is a widely overexpressed enzyme in several cancers including MM [52]. The hexokinase family irreversibly catalyses the first step of glycolysis, in which glucose is converted into glucose-6-phosphate after entering the cell through glucose transporters [36]. Investigations showed that HKII binds to the voltage-dependent anion channel (VDAC) present on the outer membrane of mitochondria [36,52] (Figure 1). This interaction is promoted by phosphoinositide-3 kinase (PI3K)/Akt signalling, resulting in stabilised high HKII levels leading to the continuous proliferation of malignant cells [52,53,54]. The constitutive overexpressed HKII can be inhibited by the small molecule 3-bromopyruvate (3BP) with alkylating properties. This compound was first identified as an inhibitor of glycolysis and oxidative phosphorylation [36,55] (Figure 1). The highly reactive molecule, which is a structural analogue of pyruvic acid, enters the cell through MCTs and releases a bromide radical after alkylation of the targeted protein [55]. As a result, lactate is not the only component that passes through MCTs. Reasons for the entry of 3BP are: (1) the abundant expression of MCTs; (2) possibly the similar molecular structure of 3BP and lactate; (3) high lactate efflux generating an acidic extracellular milieu, which benefits 3BP uptake in malignant cells [55,56].
Niedźwiecka et al. demonstrated morphological changes induced by the presence of 3BP in MM cells and flow cytometric analysis showed an increase in apoptotic MM cells after 2 and 4 h, both in a dose-dependent manner [57]. Also, ATP production and viability are reduced in MM cells after the addition of 3BP. Additionally, MM cell lines appear to be more susceptible to 3BP than leukaemic cell lines [42].
Interestingly, 2-deoxyglucose (2DG) displayed similar effects to 3BP on ATP production and cell survival in MM cells. This second anti-cancer agent is a glucose analogue and is phosphorylated by HKII into 2-DG-6-phosphate after entering the cell. The phosphorylated form cannot be metabolised and subsequently accumulates in the cell and interferes with the glycolytic pathway [58,59]. However, 2DG needs to be combined with other therapeutics due to its limited therapeutic effects as single agent [59]. Thereby, 3BP is shown to be a more convincing anti-cancer agent in terms of cell death and ATP depletion in MM cells than 2DG [52] (Figure 1).
After several enzymes and conversions, the final step of glycolysis consists of the conversion of phosphoenolpyruvate (PEP) into pyruvate and ATP, which is catalysed by PK in the cytosol of the cell. Similar to HKII, PK exists in four isoforms, where PKM2 seems to be upregulated in such a way that it becomes an abundant isoform in cancer cells [36,60]. PKM2 plays a supportive role in tumour progression and suppresses apoptosis [61]. Also, the c-MYC oncogene induces high PKM2 expression through never in mitosis (NIMA)-related kinase 2 (NEK2), which is a kinase that regulates chromosome segregation in the G2/M phase of the cell cycle [60]. Furthermore, the high enzymatic activity of PKM2 is associated with an increase of acetyl-CoA, whereas the opposite generates more lactate, leading to the Warburg effect [62]. It has recently been revealed that PKM2 expression is increased in MM cells. Moreover, silencing PKM2 leads to a decrease of MM cell growth and a cell cycle arrest at the G1/S transition [61].
Besides the formation of pyruvate, lactate is highly generated in cancer cells and transported out of the cell through MCTs. However, several papers have indicated that lactate can be incorporated into the cell and used as fuel for oxidative phosphorylation [63,64,65]. Influx and efflux depend on the concentration of lactate intra- and extracellularly and the presence of other substrates that bind to MCTs as well [65]. Myeloma cells express MCT1 to incorporate lactate in cytoplasm and generate ATP. Indeed, knockdown of the transporter leads to a decrease in lactate influx and lactate-derived ATP production, which induces apoptosis [66]. Besides knocking down MCT1, a competitive inhibitor (α-cyano-4-hydroxycinnamic acid (CHC)) of MCT1 is also capable of reducing the ability of MM cells to incorporate lactate into the cell in a dose-dependent manner [63]. This phenomenon can be accelerated through the addition of a pyruvate dehydrogenase kinase inhibitor (dichloroacetate (DCA)), which reaches its goal by shifting the glucose metabolism from pyruvate to acetyl-CoA instead of pyruvate to lactate [63,67,68,69,70] (Figure 1). Knowing that lactate is incorporated into MM cells suggests the presence of the latter in the micro-environment. Indeed, myeloma cells are supplied with lactate originating from the surrounding environment, which is described as the ‘reverse Warburg effect’ [63,66]. More precisely, BM-derived stromal cells secrete lactate through MCT4 in contrast to MCT1 [66,71,72]. While MCT1 inhibition leads to apoptosis due to a lack of fuel, MCT4 inhibition results in lactate accumulation, terminating in acidosis [73].
4. Glutamine Metabolism in MM
Glutamine is widely known as a non-essential amino acid playing a crucial role in different mechanisms in the human organism. As previously explained, glutamine enters the cell and is metabolised, resulting in different outcomes. It is converted into glutamate and ammonia (NH4+) through the activity of GLS1 and GLS2. In vitro, human myeloma cell lines (HMCLs) show an excess of NH4+ produced from glutamine, which leads to the assumption that MM cells are glutamine addicted [74]. Indeed, BM aspirates of MM patients have been checked for NH4+ levels in purified CD138+ cells. As predicted, there were significantly higher NH4+ levels in the presence of glutamine in CD138+ cells than in the CD138− fraction [75]. It has also been shown that these malignant plasma cells lack GS and consequently rely on extracellular glutamine uptake, resulting in cytotoxic effects when glutamine is depleted. This lack of GS is further shown by the addition of methionine sulfoximine (MSO), a GS inhibitor, as the cytotoxic effects remained after glutamine depletion [74]. MM cells being highly dependent on glutamine could result in interesting therapeutic targets.
Next, l-asparaginase, a molecule used in the treatment of acute lymphoblastic leukaemia (ALL), has the capacity to hydrolyse glutamine, besides the degradation of asparagine, and leads to an intracellular depletion of amino acids and inhibition of mTOR activity [74,75,76]. Moreover, synergistic effects occur when l-asparaginase is combined with the proteasome inhibitor bortezomib, leading to increased cytotoxic effects in MM cells [74,77,78]. A second proteasome inhibitor, carfilzomib, also showed synergism with l-asparaginase, resulting in intensified anti-MM activity. IL-6 and insulin-like growth factor-1 (IGF-1), in combination with l-asparaginase and carfilzomib, did not reduce the anti-MM activity [76]. It is expected that the hypoxic environment of the BM, where the malignant plasma cells reside, should intensify the dependency on glutamine [77,78]. Affecting the glutamine transporter ASCT2 by inhibition with l-γ-glutamyl-p-nitroanilide (GPNA) and benzylserine decreases glutamine influx and results in lower proliferation rates [79] (Figure 1). Besides ASCT2, LAT1 and sodium-coupled neutral amino acid transporter 1 (SNAT1) are also major glutamine transporters expressed in MM cells. However, SNAT1 and LAT1 seem to contribute in a minor way to glutamine uptake [78]. Although optimal ASCT2 inhibitors are currently lacking, this target remains interesting due to the downstream effects, including the suppression of mTORC1 kinase activity, changes in cell proliferation, autophagy and protein synthesis [77,80].
The MYC oncogene is found in many human cancers and contributes to tumour growth, proliferation, DNA replication, transcription, protein biosynthesis and altered metabolism by escaping anti-tumoral mechanisms such as apoptosis, proliferative arrest and cellular senescence [81]. The transcriptional activity of MYC protein is upregulated in MM, more precisely during late stages of MM progression, and is correlated with poor survival [82]. Furthermore, MYC is involved in glutaminolysis, enhancing the expression of glutamine transporters and repressing inhibitors of glutaminolysis [82,83]. Also, glutamine consumption leads to accumulation of the oncometabolite 2-hydroxyglutarate via c-MYC [84]. It has been demonstrated that glutaminolysis inhibition results in apoptosis in HMCLs as well as the degradation of MYC. Indeed, inhibition of glutaminolysis with compound 968, which inhibits GLS, leads to MYC degradation. Also, glutamine removal from the media engenders degradation of MYC protein and apoptosis in HMCLs [82]. An interesting potential theory emerging from these findings is the possible susceptibility of MM cells to the immune response regulated by MYC. Degradation of MYC protein would enhance the antitumor immune response through reducing CD47 and PD-L1 [82,85].
Recent evidence has been shown that glutamine can influence proliferation, independent of glutamine metabolism. Depriving cancer cells from glutamine showed anti-proliferative effects without any rescue through addition of intermediate substrates generated in glutamine metabolism such as glutamate, 2-oxoglutarate and glutathione [33,86]. Moreover, Cacace et al., demonstrated that, while glutamine deprivation led to a reduction in glycolysis through downregulation of HIF-1α, activation of HIF-1α by dimethyl-2-oxoglutarate (DM-2-oxoglutarate) did not restore cell proliferation in cancer cells deprived of glutamine, indicating that the anti-proliferative effect occurs as a reaction to the absence of extracellular glutamine [86]. This leads to the conclusion that glutamine-induced cancer cell proliferation is not solely dependent on glutamine metabolism but also activation of other signalling pathways [86].
In this regard, Cacace et al., reported that glutamine activates (phosphorylates) the transcription factor STAT3 and controls hereby cell proliferation [86]. It seems that extracellular glutamine could activate certain cell surface receptors that can regulate STAT3. As a candidate, the authors suggest a receptor similar to Grp, a glutamine receptor identified in bacteria. This hypothesis is supported by the fact that other metabolites can also act on membrane receptors [86,87]. For example, lactate can activate GPR81, which is a Gi-coupled receptor mainly expressed in adipocytes and has been shown to be highly expressed in different cancers. GPR81 silencing results in reduced tumour growth, decreased cell proliferation and lower mitochondrial activity when lactate is the only energy source [87]. This confirms the hypothesis on the actions of metabolites on membrane receptors. These findings were shown in breast cancer and cervical cancer; however, these investigations need to be refined and performed in MM [86,88].
5. Drug Resistance in MM
5.1. Standard-of-Care Drugs
In MM, altered tumour cell metabolism reduces the therapeutic effects of standard of care drugs such as bortezomib and melphalan due to drug resistance. The major reason for altered metabolism is the hypoxic tumour environment [89]. Indeed, HIF-1 is activated in a hypoxic environment and shifts glucose metabolism by intensifying the conversion of pyruvate into lactate instead of oxidation of pyruvate in the mitochondria. Mitochondrial energy production decreases as a result of HIF-1 activation [90]. It has been suggested that residual cells are resistant to treatment due to hypoxia in the BM, which leads to relapse. When comparing the level of HIF-1α and HIF-2α pathways by analysing gene-expression datasets between primary MM patients and healthy donors, a clear enrichment of HIF-1α and HIF-2α was observed in newly diagnosed MM patients [90,91,92]. Enrichment of these pathways was also observed in relapsed MM patients and bortezomib-refractory myeloma patients and was more abundant compared to bortezomib responding patients [90]. Additionally, HKII and lactate dehydrogenase A (LDHA) are found to be highly upregulated in relapsed MM patients compared to newly diagnosed myeloma patients, indicating increased glucose metabolism [90,93]. HMCLs exposed to hypoxic conditions and treatment with bortezomib, dexamethasone and melphalan showed an elevated glucose metabolism activity with overexpressed HIF-1α and LDHA after treatment [90]. The knowledge that enhanced glucose metabolism is linked with drug resistance, through HIF, generates interesting opportunities to inhibit glucose uptake in MM cells. Indeed, the combination of phloretin (GLUT1 inhibitor) and daunorubicin, a chemotherapeutic, enhanced the effect of the latter in hypoxia [90,94,95]. Wei et al. showed that compound 20 (GLUT4 inhibitor) results in chemosensitising to dexamethasone and melphalan of MM cell lines and patient material [96]. Besides GLUT1 as a target, targeting MM cells with ritonavir (GLUT4 inhibitor) increases the cytotoxic sensitivity and, together with the BH3 mimetic venetoclax, synergistic effects occur [97,98].
Next to targeting glucose uptake, HK also seems to be an interesting target. HK inhibitors such as 3BP, 2DG and lonidamine (LND) enhance drug response in normoxia in vitro; however, no response was observed in vivo [90,99,100]. Under hypoxic conditions, bortezomib decreased the activity of HKII whereas the activity of LDHA did not decrease, indicating a role for LDHA in bortezomib resistance. Moreover, bortezomib-resistant cells lose their resistance after LDHA knockdown leading to a reduction of lactate formation and should by consequence increase mitochondrial activity, reduce proliferation under hypoxic condition and thereby decrease tumorigenicity [43,90,101].
Bortezomib resistance does not only occur through LDHA and HIF. Actually, serine metabolism has recently been shown to have its own role in bortezomib resistance in MM. Serine biosynthesis starts either by extracellular import or via intracellular synthesis from glucose. The latter is the most used biosynthetic route to serine in many cancers. First, glucose enters the cell as previously explained and is metabolised via glycolysis. After several conversions, serine synthesis initiates from 3-phosphoglycerate (3-PG), which is converted into 3-phosphohydroxypyruvate (PHP) due to the enzymatic activity of phosphoglycerate dehydrogenase (PHGDH), the rate-limiting step in the serine synthesis pathway (SSP). Phosphoserine aminotransferase (PSAT) converts PHP into 3-phosphoserine (P-Ser) and is finally transformed into serine, catalysed by phosphoserine phosphatase (PSPH) [102]. SSP is shown to be beneficial to cancer cells due to its involvement in growth and proliferation [36,103]. PHGDH is upregulated in bortezomib-resistant HMCLs (shown in RPMI-8226) as well as PSAT and PSPH in different HMCLs. Similar to HIF, an overexpression of PHGDH and PSPH was observed in CD138+ cells aspirated from bortezomib-refractory myeloma patients compared to drug responsive MM patients. Depriving cells of serine was reported to be beneficial to bortezomib activity in RPMI-8226 cells [104]. Indeed, the absence of serine in the diet caused a reduction of tumour growth in mice [104,105]. These findings could result in an attractive way of tackling MM disease and can be used as a diagnostic tool to link overexpressed PHGDH with tumorigenesis.
Next to SSP, the PPP has been actively investigated in cancer. PPP consists of two phases: the oxidative and non-oxidative phase. The first phase starts from glucose-6-phosphate present in glycolysis metabolism and generates ribulose-5-phoshate after several conversions. Once ribulose-5-phoshate is formed, nucleotides are generated and pyrimidine and purine synthesis starts. The non-oxidative phase can be redirected into glycolysis metabolism by converting ribulose-5-phoshate into xylulose-5-phosphate and then into fructose-6-phosphate, which can further generate ATP through glycolysis [106,107]. Investigations demonstrated an upregulation of PPP in MM cells and together with the overexpression of SSP led to a higher antioxidant activity of bortezomib-resistant MM cells [104]. Chen et al. showed that PPP is upregulated when epidermal growth factor receptor (EGFR) inhibitors, such as gefitinib and afatinib, are administered leading to a limited response in MM cells [108]. As EGFR is found to be increased in some cancer types, targeting the latter can be a new attractive therapy when added to standard of care drugs [109]. However, the upregulation of PPP can be explained by the fact that EGFR inhibition triggers metabolic rearrangements as a compensatory mechanism to adapt and survive the loss of EGFR signalling. This resistance can be diminished through the addition of 6-aminonicotinamide (6-AN), an antimetabolite that inhibits NADPH supply in PPP combined with gefitinib [108,110].
Reasons for drug resistance can also be found in glutamine metabolism. As mentioned earlier, expression of GS is lacking in MM cells, whereas GLS expression is increased. Targeting GLS with the selective inhibitor CB-839 and combined with proteasome inhibitors (bortezomib, carfilzomib, ixazomib and oprozomib) enhanced the cytotoxic effects of proteasome inhibitors in vitro and in vivo, with carfilzomib showing the strongest synergy [111] (Table 1).

Table 1.
Targets to decrease drug resistance in MM cells. 6-AN, 6-Aminonicotinamide; EGFR, epidermal growth factor; GLS, glutaminase; HKII; hexokinase II; LDHA, lactate dehydrogenase A; PPP, pentose phosphate pathway.
As mentioned before, the BM micro-environment and MM cells interact with one another and this interaction is known to be fundamental to the appearance of drug resistance in MM. Indeed, cell adhesion-mediated drug resistance (CAM-DR), a term introduced by Damiano and Dalton et al., is the manifestation of drug resistance after the adhesion to the extracellular matrix (ECM) [112,113,114]. This mechanism is diminished when PKM2 expression is promoted in MM cell lines and on the other hand it is supported after PKM2 knockdown [61]. PKM2 influences CAM-DR through the regulation of PI3/Akt and mitogen-activated protein kinase/extracellular signal-regulated kinase (MAPK/ERK1/2) signalling pathways, both known for their involvement in tumour progression [115,116,117]. Interestingly, the extracellular matrix glycoprotein Reelin leads to drug resistance by enhancing glycolysis and through HIF-1α [118].
5.2. Immunotherapy
Another form of therapy that is currently applied in MM is immune therapy. This consists of the use of the IMiDs lenalidomide and pomalidomide, immune checkpoint inhibitors, dendritic cells (DC)-based vaccines and allogeneic transplantation in MM patients [119]. The IMiDs can enhance the proliferation and function of NK and (NK) T cells. As mentioned earlier, the monoclonal antibody daratumumab also intensifies T cell immunity against myeloma [120]. Immune checkpoint inhibitors such as nivolumab, an anti-PD-1 antibody that targets the PD-1-PD-L1 pathway, enhance the antitumor T cell response. Furthermore, DC vaccines composed of DC fusion with tumour antigens are a potential mechanism to strengthen immunotherapy in MM [119]. Chimeric antigen receptor (CAR) T cell therapy is based on genetically modified autologous T cells, which express CAR and target specifically tumour antigens. Targets for CAR T cell therapy in MM have been developed and could be interesting for refractory MM patients [119,121].
Metabolic changes in the tumour micro-environment (TME) can also decrease the beneficial effects of immunotherapy [122]. The high rate of glucose and glutamine uptake, extensive lactate production and secretion shifts the TME into an advantageous place for tumour cells. However, this shaped TME is hostile to T cells due to nutrient deprivation, acidosis, accumulation of waste products and the hypoxic environment [122,123]. Acidification of the TME impairs T cell proliferation and decreases the NK cells’ functions. Buffering the pH improves immunotherapy outcomes with bicarbonate and can be useful in MM. Furthermore, anti-cancer agents such as 2DG are used in MM and shut glycolysis down; however, 2DG cannot be combined with immunotherapeutics due to the impairment of T cell metabolism and leads to decreased T cell antitumor effect. Similarly, DCA decreases the amount of lactate in the TME in MM, which could be a solution to the acidification issue. Unfortunately, DCA impairs the function of T cells [122]. Comparable to MM cells, immune cells utilise amino acids to be functional, such as l-arginine, which is a non-essential amino acid present in macrophages and DCs. When tumour cells secrete metabolites such as lactate, arginase is overexpressed and l-arginine is converted into urea and ornithine, leading to T cell dysfunction by interfering in the cell cycle progression. Since MM cells are known to secrete lactate, it is possible that MM cells could impair T cell function through this mechanism [124].
6. Conclusions and Future Perspectives
Metabolic changes are a general hallmark for most cancers. Investigating this field is a very complex task due to the large number of factors that have to be taken into account. However, an increasing number of papers on cancer metabolism have been published in the last decade. Cancer metabolism in MM is gradually being elucidated and new treatments have arisen from this process. The two most studied components in myeloma cells are glucose and glutamine, as in many other cancers. There are various targets present in glucose metabolism, as explained in this review. HKII is the first enzyme to catalyse the beginning of glucose metabolism. Together with other metabolites from glycolysis, these can be targeted with promising results. Similarly, metabolites and enzymes involved in glutamine metabolism can be targeted in myeloma cells. Despite preclinical evidence, no clinical trials with metabolism altering agents such as 3BP, DCA, GLUT4 inhibitor (compound 20) and GLS1 inhibitor (compound 968) have been performed as yet with MM patients. However, clinical trials on l-asparaginase combined with Doxil® (PEG-liposomal doxorubicin) and dexamethasone are in phase II trials. Also, clinical trials on 2DG have been completed and it has been shown to have a safe toxicity profile; however, it is not very potent as a single agent, so combination therapy with the proteasome inhibitors could be considered (according to ClinicalTrials.gov).
Promising data have been published involving drug resistance in MM. Resistance to the proteasome inhibitor, bortezomib, has been linked to the upregulation of different metabolic pathways in myeloma cells that can be successfully targeted. Moreover, resistance to the emerging immunotherapeutics has been linked to metabolism. However, resolving this resistance remains an issue since drugs such as 2DG and DCA also have immunosuppressive effects. More investigations on potential other combinations of metabolic drugs and immunotherapeutics would be interesting because of the high specificity and efficacy. Furthermore, immunotherapy can be adapted to each patient allowing for personalized treatment, which would further improve survival. Ideally, agents targeting MM metabolism without affecting the surrounded immune cells, combined with immunotherapy reacting on specific antigens present on MM cells, would be a huge improvement in myeloma treatment. Despite the current developments, MM patients still progress to a certain stage of treatment resistance. Drug resistance can be caused by drug efflux, apoptosis inhibition, drug inactivation, repair of DNA damage due to the drug and epigenetic effects [125]. Another issue is the presence of cancer progenitor cells. These cells are usually drug-resistant and remain present in patients after treatment. Unfortunately, cancer progenitor cells do not only seem to be responsible for cancer relapse; they can also migrate and cause metastasis [125,126,127]. Therefore, the need to investigate drug resistance remains a primary need in MM and other cancers.
In addition, there are still more metabolic pathways that require investigation in MM, such as the folate pathway and proline metabolism, which shows promising and very interesting targets in its biosynthesis and catabolism pathways. Also, hypoxia related with glucose and glutamine metabolism is an interesting path to explore, together with proline metabolism in MM, to reduce drug resistance and improve survival. An overall understanding of MM cancer metabolism is still needed to improve existing targets and drugs.
Acknowledgments
This work was supported by the International Myeloma Foundation. Chaima El Arfani is a PhD student supported by fonds voor wetenschappelijk onderzoek (FWO). Kim De Veirman and Ken Maes are postdoctoral fellows of FWO.
Author Contributions
Chaima El Arfani and Eline Menu conceived this review. Chaima El Arfani wrote the manuscript. Chaima El Arfani drew the figures. Eline Menu, Kim De Veirman, Ken Maes and Elke De Bruyne provided suggestions and revised the manuscript.
Conflicts of Interest
The authors declare no conflict of interest.
Abbreviations
| αKG | Alpha Ketoglutarate |
| ALL | Acute Lymphoblastic Leukaemia |
| 6-AN | 6-Aminonicotinamide |
| ASCT2 | Neutral Amino Acid Transporter 2 |
| ATP | Adenosine Triphosphate |
| BM | Bone Marrow |
| BMSC | Bone Marrow Stromal Cells |
| 3BP | 3-Bromopyruvate |
| CAM-DR | Cell Adhesion-Mediated Drug Resistance |
| CAR | Chimeric Antigen Receptor |
| CHC | α-Cyano-4-Hydroxycinnamic |
| DC | Dendritic Cell |
| DCA | Dichloroacetate |
| 2DG | 2-Deoxyglucose |
| DM | Dimethyl |
| ECM | Extracellular Matrix |
| EGFR | Epidermal Growth Factor |
| ERK1/2 | Extracellular signal-regulated Kinase |
| FAD | Flavin Adenine Dinucleotide |
| 18F-FDG | Fluorine-labelled Fluorodeoxyglucose |
| FGF-2 | Fibroblast Growth Factor-2 |
| FL | Fas Ligand |
| GLDH | Glutamate Dehydrogenase |
| GLS1 | Glutaminase 1 |
| GLS2 | Glutaminase 2 |
| GPNA | l-γ-Glutamyl-p-Nitroanilide |
| GS | Glutamine Synthetase |
| HKII | Hexokinase II |
| HMCL | Human Myeloma Cell Line |
| IAP | Inhibitors of Apoptosis Proteins |
| IGF-1 | Insulin-like Growth Factor-1 |
| IL-1β | Interleukin-1β |
| IL-6 | Interleukin-6 |
| IMiDs | Immunomodulatory |
| JAK | Janus Kinase |
| LAT1 | human L-type Amino acid Transporter 1 |
| LDH | Lactate Dehydrogenase |
| LDHA | Lactate Dehydrogenase A |
| MAPK | Mitogen-Activated Protein Kinase |
| MCT | Monocarboxylate Transporter |
| MGUS | Monoclonal Gammopathy of Undetermined Significance |
| MM | Multiple Myeloma |
| MSO | Methionine Sulfoximine |
| MYC | Myelocytomatosis oncogene cellular homolog |
| NAD | Nicotinamide Adenine Dinucleotide |
| NADH | Nicotinamide Adenine Dinucleotide Hydrogen |
| NH4+ | Ammonia |
| NK | Natural Killer |
| OAF | Osteoclast Activating Factors |
| OC | Osteoclast |
| OXPHOS | Oxidative Phosphorylation |
| PDH | Pyruvate Dehydrogenase |
| PEP | Phosphoenolpyruvate |
| PET | Positron Emission Tomography |
| PI3K | Phosphoinositide-3 Kinase |
| PK | Pyruvate Kinase |
| PPP | Pentose Phosphate Pathway |
| PRPP | 5-Phospho-α-Ribosyl-1-Pyrophosphate |
| RANKL | Receptor Activator of Nuclear factor κB Ligand |
| SNAT1 | Sodium-coupled Neutral Amino acid Transporter 1 |
| STAT3 | Signal Transducer and Activator of Transcription 3 |
| TCA | Tricarboxylic Acid cycle |
| TME | Tumour Micro-Environment |
| TNF | Tumour Necrosis Factor |
| TNF-β | Tumour Necrosis Factor-β |
| TRAIL | TNF-Related Apoptosis-Inducing Ligand |
| VCAM-1 | Vascular Cell Adhesion Molecule 1 |
| VDAC | Voltage-Dependent Anion Channel |
| VEGF | Vascular Endothelial Growth Factor |
References
- Kristinsson, S.Y.; Björkholm, M.; Goldin, L.R.; Blimark, C.; Mellqvist, U.H.; Wahlin, A.; Turesson, I.; Landgren, O. Patterns of hematologic malignancies and solid tumors among 37,838 first-degree relatives of 13,896 multiple myeloma patients in Sweden. Int. J. Cancer 2009, 125, 2147–2150. [Google Scholar] [CrossRef] [PubMed]
- Kawano, Y.; Moschetta, M.; Manier, S.; Glavey, S.; Görgün, GT.; Roccaro, A.M.; Anderson, K.C.; Ghobrial, I.M. Targeting the bone marrow microenvironment in multiple myeloma. Immunol. Rev. 2015, 263, 160–172. [Google Scholar] [CrossRef] [PubMed]
- Kühnel, A.; Blau, O.; Nogai, K.A.; Blau, I.W. The Warburg effect in Multiple Myeloma and its microenvironment. Med. Res. Arch. 2017, 5, 1–16. [Google Scholar]
- Rajkumar, S.V. Evolving diagnostic criteria for multiple myeloma. Hematol. Am. Soc. Hematol. Educ. Program 2015, 2015, 272–278. [Google Scholar] [CrossRef] [PubMed]
- Ramsenthaler, C.; Kane, P.; Gao, W.; Siegert, R.J.; Edmonds, P.M.; Schey, S.A.; Higginson, I.J. Prevalence of symptoms in patients with multiple myeloma: A systematic review and meta-analysis. Eur. J. Haematol. 2016, 97, 416–429. [Google Scholar] [CrossRef] [PubMed]
- Ferlay, J.; Soerjomataram, I.; Dikshit, R.; Eser, S.; Mathers, C.; Rebelo, M.; Parkin, D.M.; Forman, D.; Bray, F. Cancer incidence and mortality worldwide: Sources, methods and major patterns in GLOBOCAN 2012. Int. J. Cancer 2015, 136, E359–E386. [Google Scholar] [CrossRef] [PubMed]
- Kumar, S.K.; Rajkumar, S.V.; Dispenzieri, A.; Lacy, M.Q.; Hayman, S.R.; Buadi, F.K.; Zeldenrust, S.R.; Dingli, D.; Russell, S.J.; Lust, J.A.; et al. Improved survival in multiple myeloma and the impact of novel therapies. Blood 2008, 111, 2516–2520. [Google Scholar] [CrossRef] [PubMed]
- Bianchi, G.; Richardson, P.G.; Anderson, K.C. Promising therapies in multiple myeloma. Blood 2015, 126, 300–310. [Google Scholar] [CrossRef] [PubMed]
- Moreau, P.; Attal, M.; Facon, T. Frontline therapy of multiple myeloma. Blood 2015, 125, 3076–3084. [Google Scholar] [CrossRef] [PubMed]
- Russo, A.; Saide, A.; Smaldone, S.; Faraonio, R.; Russo, G. Role of uL3 in Multidrug Resistance in p53-Mutated Lung Cancer Cells. Int. J. Mol. Sci. 2017, 18, 547. [Google Scholar] [CrossRef] [PubMed]
- Russo, A.; Russo, G. Ribosomal Proteins Control or Bypass p53 during Nucleolar Stress. Int. J. Mol. Sci. 2017, 18, 140. [Google Scholar] [CrossRef] [PubMed]
- Folkman, J.; Watson, K.; Ingber, D.; Hanahan, D. Induction of angiogenesis during the transition from hyperplasia to neoplasia. Nature 1989, 339, 58–61. [Google Scholar] [CrossRef] [PubMed]
- Carmeliet, P. Angiogenesis in life, disease and medicine. Nature 2005, 438, 932–936. [Google Scholar] [CrossRef] [PubMed]
- Vacca, A.; Ribatti, D. Angiogenesis and vasculogenesis in multiple myeloma: Role of inflammatory cells. Recent Results Cancer Res. 2011, 183, 87–95. [Google Scholar] [CrossRef] [PubMed]
- Hose, D.; Moreaux, J.; Meissner, T.; Seckinger, A.; Goldschmidt, H.; Benner, A.; Mahtouk, K.; Hillengass, J.; Rème, T.; De Vos, J.; et al. Induction of angiogenesis by normal and malignant plasma cells. Blood 2009, 114, 128–143. [Google Scholar] [CrossRef] [PubMed]
- Vacca, A.; Ria, R.; Semeraro, F.; Merchionne, F.; Coluccia, M.; Boccarelli, A.; Scavelli, C.; Nico, B.; Gernone, A.; Battelli, F.; et al. Endothelial cells in the bone marrow of patients with multiple myeloma. Blood 2003, 102, 3340–3348. [Google Scholar] [CrossRef] [PubMed]
- Caligaris-Cappio, F.; Bergui, L.; Gregoretti, M.G.; Gaidano, G.; Gaboli, M.; Schena, M.; Zallone, A.Z.; Marchisio, P.C. Role of bone marrow stromal cells in the growth of human multiple myeloma. Blood 1991, 77, 2688–2693. [Google Scholar] [PubMed]
- Shafat, M.S.; Gnaneswaran, B.; Bowles, K.M.; Rushworth, S.A. The bone marrow microenvironment—Home of the leukemic blasts. Blood Rev. 2017, 31, 277–286. [Google Scholar] [CrossRef] [PubMed]
- Lemaire, M.; Deleu, S.; De Bruyne, E.; Van Valckenborgh, E.; Menu, E.; Vanderkerken, K. The microenvironment and molecular biology of the multiple myeloma tumor. Adv. Cancer Res. 2011, 110, 19–42. [Google Scholar] [CrossRef] [PubMed]
- Hideshima, T.; Bergsagel, P.L.; Kuehl, W.M.; Anderson, K.C. Advances in biology of multiple myeloma: Clinical applications. Blood 2004, 104, 607–618. [Google Scholar] [CrossRef] [PubMed]
- Moyo, T.K.; Bouchnita, A.; Eymard, N.; Volpert, V.; Koury, M.J. Effects of bone marrow infiltration by multiple myeloma on erythropoiesis. Blood 2015, 126, 2143. [Google Scholar]
- Christoulas, D.; Terpos, E.; Dimopoulos, M.A. Pathogenesis and Management of Myeloma Bone Disease. Expert Rev. Hematol. 2009, 2, 385–398. [Google Scholar] [CrossRef] [PubMed]
- Silvestris, F.; Ciavarella, S.; De Matteo, M.; Tucci, M.; Dammacco, F. Bone-Resorbing Cells in Multiple Myeloma: Osteoclasts, Myeloma Cell Polykaryons, or Both? Oncologist 2009, 14, 264–275. [Google Scholar] [CrossRef] [PubMed]
- Merico, F.; Bergui, L.; Gregoretti, M.G.; Ghia, P.; Aimo, G.; Lindley, I.J.; Caligaris-Cappio, F. Cytokines involved in the progression of multiple myeloma. Clin. Exp. Immunol. 1993, 92, 27–31. [Google Scholar] [CrossRef] [PubMed]
- Pavlova, N.N.; Thompson, C.B. The emerging hallmarks of cancer metabolism. Cell Metab. 2016, 23, 27–47. [Google Scholar] [CrossRef] [PubMed]
- Brown, G.K. Glucose transporters: Structure, function and consequences of deficiency. J. Inherit. Metab. Dis. 2000, 23, 237–246. [Google Scholar] [CrossRef] [PubMed]
- Alfarouk, K.O.; Verduzco, D.; Rauch, C.; Muddathir, A.K.; Bashir, H.H.; Elhassan, G.O.; Ibrahim, M.E.; Orozco, J.D.P.; Cardone, R.A.; Reshkin, S.J. Glycolysis, tumor metabolism, cancer growth and dissemination. A new pH-based etiopathogenic perspective and therapeutic approach to an old cancer question. Oncoscience 2014, 1, 777–802. [Google Scholar] [CrossRef] [PubMed]
- Zheng, J. Energy metabolism of cancer: Glycolysis versus oxidative phosphorylation. Oncol. Lett. 2012, 4, 1151–1157. [Google Scholar] [CrossRef] [PubMed]
- DeBerardinis, R.J.; Chandel, N.S. Fundamentals of cancer metabolism. Sci. Adv. 2016, 2, 2:1–2:18. [Google Scholar] [CrossRef] [PubMed]
- Pfeiffer, T.; Schuster, S.; Bonhoeffer, S. Cooperation and competition in the evolution of ATP-producing pathways. Science 2001, 292, 504–507. [Google Scholar] [CrossRef] [PubMed]
- Newsholme, P.; Procopio, J.; Lima, M.M.R.; Pithon-Curi, T.C.; Curi, R. Glutamine and glutamate—Their central role in cell metabolism and function. Cell Biochem. Funct. 2003, 21, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Pochini, L.; Scalise, M.; Galluccio, M.; Indiveri, C. Membrane transporters for the special amino acid glutamine: Structure/function relationships and relevance to human health. Front. Chem. 2014, 2, 61:1–61:23. [Google Scholar] [CrossRef] [PubMed]
- Altman, B.J.; Stine, Z.E.; Dang, C.V. From Krebs to clinic: Glutamine metabolism to cancer therapy. Nat. Rev. Cancer 2016, 16, 619–634. [Google Scholar] [CrossRef] [PubMed]
- Curthoys, N.P.; Watford, M. Regulation of glutaminase activity and glutamine metabolism. Annu. Rev. Nutr. 1995, 15, 133–159. [Google Scholar] [CrossRef] [PubMed]
- Moreadith, R.W.; Lehninger, A.L. The pathways of glutamate and glutamine oxidation by tumor cell mitochondria. Role of mitochondrial NAD(P)+-dependent malic enzyme. J. Biol. Chem. 1984, 259, 6215–6221. [Google Scholar] [PubMed]
- Hirschey, M.D.; DeBerardinis, R.J.; Diehl, A.M.; Drew, J.E.; Frezza, C.; Green, M.F.; Jones, L.W.; Ko, Y.H.; Le, A.; Lea, M.A.; et al. Dysregulated metabolism contributes to oncogenesis. Semin. Cancer Biol. 2015, 35, 129–150. [Google Scholar] [CrossRef] [PubMed]
- Lane, A.N.; Fan, T.W.M. Regulation of mammalian nucleotide metabolism and biosynthesis. Nucleic Acids Res. 2015, 43, 2466–2485. [Google Scholar] [CrossRef] [PubMed]
- Cory, J.G.; Cory, A.H. Critical roles of glutamine as nitrogen donors in purine and pyrimidine nucleotide synthesis: Asparaginase treatment in childhood acute lymphoblastic leukemia. In Vivo 2006, 20, 587–589. [Google Scholar] [PubMed]
- Alfarouk, K.O.; Shayoub, M.E.A.; Muddathir, A.K.; Elhassan, G.O.; Bashir, A.H.H. Evolution of Tumor Metabolism might Reflect Carcinogenesis as a Reverse Evolution process (Dismantling of Multicellularity). Cancers 2011, 3, 3002–3017. [Google Scholar] [CrossRef] [PubMed]
- Zu, X.L.; Guppy, M. Cancer metabolism: Facts, fantasy, and fiction. Biochem. Biophys. Res. Commun. 2004, 313, 459–465. [Google Scholar] [CrossRef] [PubMed]
- Gatenby, R.A.; Gillies, R.J. Why do cancers have high aerobic glycolysis? Nat. Rev. Cancer 2004, 4, 891–899. [Google Scholar] [CrossRef] [PubMed]
- Koppenol, W.H.; Bounds, P.L.; Dang, C.V. Otto Warburg’s contributions to current concepts of cancer metabolism. Nat. Rev. Cancer 2011, 11, 325–337. [Google Scholar] [CrossRef] [PubMed]
- Fantin, V.R.; St-Pierre, J.; Leder, P. Attenuation of LDH-A expression uncovers a link between glycolysis, mitochondrial physiology, and tumor maintenance. Cancer Cell 2006, 9, 425–434. [Google Scholar] [CrossRef] [PubMed]
- Martinez-Outschoorn, U.E.; Peiris-Pageès, M.; Pestell, R.G.; Sotgia, F.; Lisanti, M.P. Cancer metabolism: A therapeutic perspective. Nat. Rev. Clin. Oncol. 2017, 14, 11–31. [Google Scholar] [CrossRef] [PubMed]
- DeBerardinis, R.J.; Cheng, T. Q’s next: The diverse functions of glutamine in metabolism, cell biology and cancer. Oncogene 2010, 29, 313–324. [Google Scholar] [CrossRef] [PubMed]
- He, Q.; Shi, X.; Zhang, L.; Yi, C.; Zhang, X.; Zhang, X. De novo glutamine synthesis: Importance for the proliferation of glioma cells and potentials for its detection with 13N-ammonia. Mol. Imaging 2016, 15, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Nicklin, P.; Bergman, P.; Zhang, B.; Triantafellow, E.; Wang, H.; Nyfeler, B.; Yang, H.; Hild, M.; Kung, C.; Wilson, C.; et al. Bidirectional transport of amino acids regulates mTOR and autophagy. Cell 2009, 136, 521–534. [Google Scholar] [CrossRef] [PubMed]
- Thompson, C.B. Rethinking the regulation of cellular metabolism. Cold Spring Harb. Symp. Quant. Biol. 2011, 76, 23–29. [Google Scholar] [CrossRef] [PubMed]
- Rathmell, J.C.; vander Heiden, M.G.; Harris, M.H.; Frauwirth, K.A.; Thompson, C.B. In the absence of extrinsic signals, nutrient utilization by lymphocytes is insufficient to maintain either cell size or viability. Mol. Cell 2000, 6, 683–692. [Google Scholar] [CrossRef]
- Glynn, S.A.; Albanes, D. Folate and cancer: A review of the literature. Nutr. Cancer 1994, 22, 101–119. [Google Scholar] [CrossRef] [PubMed]
- Liu, W.; Phang, J.M. Oncogene and Cancer—From Bench to Clinic; InTech: London, UK, 2013; Volume 15, pp. 359–390. ISBN 978-953-51-0858-0. [Google Scholar]
- Nakano, A.; Miki, H.; Nakamura, S.; Harada, T.; Oda, A.; Amou, H.; Fujii, S.; Kagawa, K.; Takeuchi, K.; Ozaki, S.; et al. Up-regulation of hexokinaseII in myeloma cells: Targeting myeloma cells with 3-bromopyruvate. J. Bioenerg. Biomembr. 2012, 44, 31–38. [Google Scholar] [CrossRef] [PubMed]
- Mathupala, S.P.; Ko, Y.H.; Pedersen, P.L. Hexokinase II: Cancer’s double-edged sword acting as both facilitator and gatekeeper of malignancy when bound to mitochondria. Oncogene 2006, 25, 4777–4786. [Google Scholar] [CrossRef] [PubMed]
- Robey, R.B.; Hay, N. Mitochondrial hexokinases, novel mediators of the antiapoptotic effects of growth factors and Akt. Oncogene 2006, 25, 4683–4696. [Google Scholar] [CrossRef] [PubMed]
- Lis, P.; Dyląg, M.; Niedźwiecka, K.; Ko, Y.H.; Pedersen, P.L.; Goffeau, A.; Ułaszewski, S. The HK2 dependent “Warburg Effect” and mitochondrial oxidative phosphorylation in cancer: Targets for effective therapy with 3-bromopyruvate. Molecules 2016, 21, 1730. [Google Scholar] [CrossRef] [PubMed]
- Azevedo-Silva, J.; Queirós, O.; Ribeiro, A.; Baltazar, F.; Young, K.H.; Pedersen, P.L.; Preto, A.; Casal, M. The cytotoxicity of 3-bromopyruvate in breast cancer cells depends on extracellular pH. Biochem. J. 2015, 467, 247–258. [Google Scholar] [CrossRef] [PubMed]
- Niedźwiecka, K.; Dyląg, M.; Augustyniak, D.; Majkowska-Skrobek, G.; Cal-Bąkowska, M.; Ko, Y.H.; Pedersen, P.L.; Goffeau, A.; Ułaszewski, S. Glutathione may have implications in the design of 3-bromopyruvate treatment protocols for both fungal and algal infections as well as multiple myeloma. Oncotarget 2016, 7, 65614–65626. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.D.; Deslandes, E.; Villedieu, M.; Poulain, L.; Duval, M.; Gauduchon, P.; Schwartz, L.; Icard, P. Effect of 2-deoxy-d-glucose on various malignant cell lines in vitro. Anticancer Res. 2006, 26, 3561–3566. [Google Scholar] [PubMed]
- Zhang, D.; Li, J.; Wang, F.; Hu, J.; Wang, S.; Sun, Y. 2-Deoxy-d-glucose targeting of glucose metabolism in cancer cells as a potential therapy. Cancer Lett. 2014, 355, 176–183. [Google Scholar] [CrossRef] [PubMed]
- Gu, Z.; Xia, J.; Xu, H.; Frech, I.; Tricot, G.; Zhan, F. NEK2 Promotes Aerobic Glycolysis in Multiple Myeloma Through Regulating Splicing of Pyruvate Kinase. J. Hematol. Oncol. 2017, 10, 17:1–17:11. [Google Scholar] [CrossRef] [PubMed]
- He, Y.; Wang, Y.; Liu, H.; Xu, X.; He, S.; Tang, J.; Huang, Y.; Miao, X.; Wu, Y.; Wang, Q. Pyruvate kinase isoform M2 (PKM2) participates in multiple myeloma cell proliferation, adhesion and chemoresistance. Leuk. Res. 2015, 39, 1428–1436. [Google Scholar] [CrossRef] [PubMed]
- Tamada, M.; Suematsu, M.; Saya, H. Pyruvate Kinase M2: Multiple faces for conferring benefits on cancer cells. Clin. Cancer Res. 2012, 18, 5554–5561. [Google Scholar] [CrossRef] [PubMed]
- Fujiwara, S.; Wada, N.; Kawano, Y.; Okuno, Y.; Kikukawa, Y.; Endo, S.; Nishimura, N.; Ueno, N.; Mitsuya, H.; Hata, H. Lactate, a putative survival factor for myeloma cells, is incorporated by myeloma cells through monocarboxylate transporters 1. Exp. Hematol. Oncol. 2015, 4, 12:1–12:8. [Google Scholar] [CrossRef] [PubMed]
- Sonveaux, P.; Végran, F.; Schroeder, T.; Wergin, M.C.; Verrax, J.; Rabbani, Z.N.; de Saedeleer, C.J.; Kennedy, K.M.; Diepart, C.; Jordan, B.F. Targeting lactate-fueled respiration selectively kills hypoxic tumor cells in mice. J. Clin. Investig. 2008, 118, 3930–3942. [Google Scholar] [CrossRef] [PubMed]
- Doherty, J.R.; Cleveland, J.L. Targeting lactate metabolism for cancer therapeutics. J. Clin. Investig. 2013, 123, 3685–3692. [Google Scholar] [CrossRef] [PubMed]
- Fujiwara, S.; Wada, N.; Kawano, Y.; Kikukawa, Y.; Mitsuya, H.; Hata, H. Lactate is a crucial energy source for multiple myeloma (MM) cells in bone marrow microenvironment. Blood 2013, 122, 3109. [Google Scholar]
- Zhang, S.; Hulver, M.W.; McMillan, R.P.; Cline, M.A.; Gilbert, E.R. The pivotal role of pyruvate dehydrogenase kinases in metabolic flexibility. Nutr. Metab. 2014, 11, 10:1–10:9. [Google Scholar] [CrossRef] [PubMed]
- Niewisch, M.R.; Kuçi, Z.; Wolburg, H.; Sautter, M.; Krampen, L.; Deubzer, B.; Handgretinger, R.; Bruchelt, G. Influence of dichloroacetate (DCA) on lactate production and oxygen consumption in neuroblastoma cells: Is DCA a suitable drug for neuroblastoma therapy? Cell. Physiol. Biochem. 2012, 29, 373–380. [Google Scholar] [CrossRef] [PubMed]
- Sanchez, W.Y.; McGee, S.L.; Connor, T.; Mottram, B.; Wilkinson, A.; Whitehead, J.P.; Vuckovic, S.; Catley, L. Dichloroacetate inhibits aerobic glycolysis in multiple myeloma cells and increases sensitivity to bortezomib. Br. J. Cancer 2013, 108, 1624–1633. [Google Scholar] [CrossRef] [PubMed]
- Fujiwara, S.; Kawano, Y.; Yuki, H.; Okuno, Y.; Nosaka, K.; Mitsuya, H.; Hata, H. PDK1 inhibition is a novel therapeutic target in multiple myeloma. Br. J. Cancer 2013, 108, 170–178. [Google Scholar] [CrossRef] [PubMed]
- Romero-Garcia, S.; Moreno-Altamiranon, M.M.B.; Prado-Garcia, H.; Sánchez-García, F.J. Lactate contribution to the tumor microenvironment: Mechanisms, effects on immune cells and therapeutic relevance. Front. Immunol. 2016, 7, 52:1–52:11. [Google Scholar] [CrossRef] [PubMed]
- Rattigan, Y.I.; Patel, B.B.; Ackerstaff, E.; Sukenick, G.; Koutcher, J.A.; Glod, J.W.; Banerjee, D. Lactate is a mediator of metabolic cooperation between stromal carcinoma associated fibroblasts and glycolytic tumor cells in the tumor microenvironment. Exp. Cell Res. 2012, 318, 326–335. [Google Scholar] [CrossRef] [PubMed]
- Matsumoto, T.; Jimi, S.; Migita, K.; Takamatsu, Y.; Hara, S. Inhibition of glucose transporter 1 induces apoptosis and sensitizes multiple myeloma cells to conventional chemotherapeutic agents. Leuk. Res. 2016, 41, 103–110. [Google Scholar] [CrossRef] [PubMed]
- Bolzoni, M.; Chiu, M.; Accardi, F.; Vescovini, R.; Airoldi, I.; Storti, P.; Todoerti, K.; Agnelli, L.; Missale, G.; Andreoli, R.; et al. Dependence on glutamine uptake and glutamine addiction characterize myeloma cells: A new attractive target. Blood 2016, 128, 667–679. [Google Scholar] [CrossRef] [PubMed]
- Accardi, F.; Chiu, M.; Bolzoni, M.; Storti, P.; Todoerti, K.; Agnelli, L.; Ferrari, M.; Missale, G.; Aversa, F.; Bussolati, O.; et al. Ammonium Production and Glutamine-Addiction of Myeloma Cells: New Attractive Targets in Multiple Myeloma. Blood 2014, 124, 2067. [Google Scholar]
- Minetto, P.; Soncini, D.; Cagnetta, A.; Guolo, F.; Retali, V.; Rivoli, G.; Bisso, N.; Di Felice, N.; Miglino, M.; Canepa, L.; et al. Glutamine-Dependence Targeting By Asparaginase Significantly Increases Anti-Myeloma Activity of Proteasome Inhibitors. Blood 2017, 130, 1796. [Google Scholar]
- Giuliani, N.; Chiu, M.; Bolzoni, M.; Accardi, F.; Bianchi, M.G.; Toscani, D.; Aversa, F.; Bussolati, O. The potential of inhibiting glutamine uptake as a therapeutic target for multiple myeloma. Expert Opin. Ther. Targets 2017, 21, 231–234. [Google Scholar] [CrossRef] [PubMed]
- Corbet, C.; Feron, O. Metabolic and mind shifts: From glucose to glutamine and acetate addictions in cancer. Curr. Opin. Clin. Nutr. Metab. Care 2015, 18, 346–353. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Chen, L.; Cui, H. Targeting Glutamine Induces Apoptosis: A Cancer Therapy Approach. Int. J. Mol. Sci. 2015, 16, 22830–22855. [Google Scholar] [CrossRef] [PubMed]
- Jeon, Y.J.; Khelifa, S.; Ratnikov, B.; Scott, D.A.; Feng, Y.; Parisi, F.; Ruller, C.; Lau, E.; Kim, H.; Brill, L.M.; et al. Regulation of glutamine carrier proteins by RNF5 determines breast cancer response to ER stressinducing chemotherapies. Cancer Cell 2015, 27, 354–369. [Google Scholar] [CrossRef] [PubMed]
- Gabay, M.; Li, Y.; Felsher, D.W. MYC activation is a hallmark of cancer initiation and maintenance. Cold Spring Harb. Perspect. Med. 2014, 4, 6:1–6:13. [Google Scholar] [CrossRef] [PubMed]
- Effenberger, M.; Bommert, K.S.; Kunz, V.; Kruk, J.; Leich, E.; Rudelius, M.; Bargou, R.; Bommert, K. Glutaminase inhibition in multiple myeloma induces apoptosis via MYC degradation. Oncotarget 2017, 8, 85858–85867. [Google Scholar] [CrossRef] [PubMed]
- Wise, D.R.; DeBerardinis, R.J.; Mancuso, A.; Sayed, N.; Zhang, X.Y.; Pfeiffer, H.K.; Nissim, I.; Daikhin, E.; Yudkoff, M.; McMahon, S.B.; et al. Myc regulates a transcriptional program that stimulates mitochondrial glutaminolysis and leads to glutamine addiction. Proc. Natl. Acad. Sci. USA 2008, 105, 18782–18787. [Google Scholar] [CrossRef] [PubMed]
- Gonsalves, W.I.; Ramakrishnan, V.; Hitosugi, T.; Ghosh, T.; Jevremovic, D.; Dutta, T.; Sakrikar, D.; Petterson, X.M.; Wellik, L.; Kumar, S.K.; et al. Glutamine-derived 2-hydroxyglutarate is associated with disease progression in plasma cell malignancies. JCI Insight 2018, 3, e94543. [Google Scholar] [CrossRef] [PubMed]
- Casey, S.C.; Tong, L.; Li, Y.; Do, R.; Walz, S.; Fitzgerald, K.N.; Gouw, A.M.; Baylot, V.; Gütgemann, I.; Eilers, M.; et al. MYC regulates the antitumor immune response through CD47 and PD-L1. Science 2016, 352, 227–231. [Google Scholar] [CrossRef] [PubMed]
- Cacace, A.; Sboarina, M.; Vazeille, T.; Sonveaux, P. Glutamine activates STAT3 to control cancer cell proliferation independently of glutamine metabolism. Oncogene 2017, 36, 2074–2084. [Google Scholar] [CrossRef] [PubMed]
- Roland, C.L.; Arumugam, T.; Deng, D.; Liu, S.H.; Philip, B.; Gomez, S.; Burns, W.R.; Ramachandran, V.; Wang, H.; Cruz-Monserrate, Z.; et al. Cell surface lactate receptor GPR81 is crucial for cancer cell survival. Cancer Res. 2014, 74, 5301–5310. [Google Scholar] [CrossRef] [PubMed]
- Yao, C.; Li, Y.Y.; Li, J.; Zhang, H.Y.; Wang, F.; Bai, X.; Li, S.S. STAT3 regulates hypoxia-induced epithelial mesenchymal transition in oesophageal squamous cell cancer. Oncol. Rep. 2016, 36, 108–116. [Google Scholar] [CrossRef]
- Denko, N.C. Hypoxia, HIF1 and glucose metabolism in the solid tumour. Nat. Rev. Cancer 2008, 8, 705–713. [Google Scholar] [CrossRef] [PubMed]
- Maiso, P.; Huynh, D.; Moschetta, M.; Sacco, A.; Aljawai, Y.; Mishima, Y.; Asara, J.M.; Roccaro, A.M.; Kimmelman, A.C.; Ghobrial, I.M. Metabolic signature identifies novel targets for drug resistance in Multiple Myeloma. Cancer Res. 2015, 75, 2071–2082. [Google Scholar] [CrossRef] [PubMed]
- Schaefer, C.F.; Anthony, K.; Krupa, S.; Buchoff, J.; Day, M.; Hannay, T.; Buetow, K.H. PID: The Pathway Interaction Database. Nucleic Acids Res. 2009, 37, D674–679. [Google Scholar] [CrossRef] [PubMed]
- Mulligan, G.; Mitsiades, C.; Bryant, B.; Zhan, F.; Chng, W.J.; Roels, S.; Koenig, E.; Fergus, A.; Huang, Y.; Richardson, P.; et al. Gene expression profiling and correlation with outcome in clinical trials of the proteasome inhibitor bortezomib. Blood 2007, 109, 3177–3188. [Google Scholar] [CrossRef] [PubMed]
- Liu, Z.; Jia, X.; Duan, Y.; Xiao, H.; Sundqvist, K.G.; Permert, J.; Wang, F. Excess glucose induces hypoxia-inducible factor-1α in pancreatic cancer cells and stimulates glucose metabolism and cell migration. Cancer Biol. Ther. 2013, 14, 428–435. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Zub, K.A.; de Sousa, M.M.L.; Sarno, A.; Sharma, A.; Demirovic, A.; Rao, S.; Young, C.; Aas, P.A.; Ericsson, I.; Sundan, A.; et al. Modulation of Cell Metabolic Pathways and Oxidative Stress Signaling Contribute to Acquired Melphalan Resistance in Multiple Myeloma Cells. PLoS ONE 2015, 10, 3:1–3:20. [Google Scholar] [CrossRef] [PubMed]
- Cao, X.; Fang, L.; Gibbs, S.; Huang, Y.; Dai, Z.; Wen, P.; Zheng, X.; Sadee, W.; Sun, D. Glucose uptake inhibitor sensitizes cancer cells to daunorubicin and overcomes drug resistance in hypoxia. Cancer Chemother. Pharmacol. 2007, 59, 495–505. [Google Scholar] [CrossRef] [PubMed]
- Wei, C.; Bajpai, R.; Sharma, H.; Heitmeier, M.; Jain, A.D.; Matulis, S.M.; Nooka, A.K.; Mishra, R.K.; Hruz, P.W.; Schiltz, G.E.; et al. Development of GLUT4-selective antagonists for multiple myeloma therapy. Eur. J. Med. Chem. 2017, 139, 573–586. [Google Scholar] [CrossRef] [PubMed]
- Bajpai, R.; Matulis, S.M.; Wei, C.; Nooka, A.K.; Von Hollen, H.E.; Lonial, S.; Boise, L.H.; Shanmugam, M. Targeting glutamine metabolism in multiple myeloma enhances BIM binding to BCL-2 eliciting synthetic lethality to venetoclax. Oncogene 2016, 35, 3955–3964. [Google Scholar] [CrossRef] [PubMed]
- Dalva-Aydemir, S.; Bajpai, R.; Martinez, M.; Adekola, K.U.; Kandela, I.; Wei, C.; Singhal, S.; Koblinski, J.E.; Raje, N.S.; Rosen, S.T.; et al. Targeting the Metabolic Plasticity of Multiple Myeloma with FDA-Approved Ritonavir and Metformin. Clin. Cancer Res. 2015, 21, 1161–1171. [Google Scholar] [CrossRef] [PubMed]
- Kurtoglu, M.; Gao, N.; Shang, J.; Maher, J.C.; Lehrman, M.A.; Wangpaichitr, M.; Savaraj, N.; Lane, A.N.; Lampidis, T.J. Under normoxia, 2-deoxy-D-glucose elicits cell death in select tumor types not by inhibition of glycolysis but by interfering with N-linked glycosylation. Mol. Cancer Ther. 2007, 6, 3049–3058. [Google Scholar] [CrossRef] [PubMed]
- Maher, J.C.; Krishan, A.; Lampidis, T.J. Greater cell cycle inhibition and cytotoxicity induced by 2-deoxy-D-glucose in tumor cells treated under hypoxic vs aerobic conditions. Cancer Chemother. Pharmacol. 2004, 53, 116–122. [Google Scholar] [CrossRef] [PubMed]
- Miao, P.; Sun, X.; Liu, J.; Huang, G. Lactate dehydrogenase a in cancer: A promising target for diagnosis and therapy. IUBMB Life 2013, 65, 904–910. [Google Scholar] [CrossRef] [PubMed]
- Mattaini, K.R.; Sullivan, M.R.; Vander Heiden, M.G. The importance of serine metabolism in cancer. J. Cell Biol. 2016, 214, 249–257. [Google Scholar] [CrossRef] [PubMed]
- Locasale, J.W. Serine, glycine and one-carbon units: Cancer metabolism in full circle. Nat. Rev. Cancer 2013, 13, 572–583. [Google Scholar] [CrossRef] [PubMed]
- Zaal, E.A.; Wu, W.; Jansen, G.; Zweegman, S.; Cloos, J.; Berkers, C.R. Bortezomib resistance in multiple myeloma is associated with increased serine synthesis. Cancer Metab. 2017, 5, 7:1–7:12. [Google Scholar] [CrossRef] [PubMed]
- Maddocks, O.D.K.; Athineos, D.; Cheung, E.C.; Lee, P.; Zhang, T.; van den Broek, N.J.F.; Mackay, G.M.; Labuschagne, C.F.; Gay, D.; Kruiswijk, F.; et al. Modulating the therapeutic response of tumours to dietary serine and glycine starvation. Nature 2017, 544, 372–376. [Google Scholar] [CrossRef] [PubMed]
- Patra, K.C.; Hay, N. The pentose phosphate pathway and cancer. Trends Biochem. Sci. 2014, 39, 347–354. [Google Scholar] [CrossRef]
- Horecker, B.L. The pentose phosphate pathway. J. Biol. Chem. 2002, 277, 47965–47971. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Huang, R.; Ding, J.; Ji, D.; Song, B.; Yuan, L.; Chang, H.; Chen, G. Multiple myeloma acquires resistance to EGFR inhibitor via induction of pentose phosphate pathway. Sci. Rep. 2015, 5, 9925:1–9925:8. [Google Scholar] [CrossRef] [PubMed]
- Normanno, N.; De Luca, A.; Bianco, C.; Strizzi, L.; Mancino, M.; Maiello, M.R.; Carotenuto, A.; de Feo, G.; Caponigro, F.; Salomon, D.S. Epidermal growth factor receptor (EGFR) signaling in cancer. Gene 2006, 366, 2–16. [Google Scholar] [CrossRef] [PubMed]
- Hothersall, J.S.; Gordge, M.; Noronha-Dutra, A.A. Inhibition of NADPH supply by 6-aminonicotinamide: Effect on glutathione, nitric oxide and superoxide in J774 cells. FEBS Lett. 1998, 434, 97–100. [Google Scholar] [CrossRef]
- Thompson, R.M.; Dytfeld, D.; Reyes, L.; Robinson, R.M.; Smith, B.; Manevich, Y.; Jakubowiak, A.; Komarnicki, M.; Przybylowicz-Chalecka, A.; Szczepaniak, T.; et al. Glutaminase inhibitor CB-839 synergizes with carfilzomib in resistant multiple myeloma cells. Oncotarget 2017, 8, 35863–35876. [Google Scholar] [CrossRef] [PubMed]
- Katz, B.Z. Adhesion molecules—The lifelines of multiple myeloma cells. Semin. Cancer Biol. 2010, 20, 186–195. [Google Scholar] [CrossRef] [PubMed]
- Burger, J.A.; Ghia, P.; Rosenwald, A.; Caligaris-Cappio, F. The microenvironment in mature B-cell malignancies: A target for new treatment strategies. Blood 2009, 114, 3367–3375. [Google Scholar] [CrossRef] [PubMed]
- Damiano, J.S.; Cress, A.E.; Hazlehurst, L.A.; Shtil, A.A.; Dalton, W.S. Cell adhesion mediated drug resistance (CAM-DR): Role of integrins and resistance to apoptosis in human myeloma cell lines. Blood 1999, 93, 1658–1667. [Google Scholar] [PubMed]
- Zhu, J.; Wang, M.; Cao, B.; Hou, T.; Mao, X. Targeting the phosphatidylinositol 3-kinase/AKT pathway for the treatment of multiple myeloma. Curr. Med. Chem. 2014, 21, 3173–3187. [Google Scholar] [CrossRef] [PubMed]
- Steinbrunn, T.; Stühmer, T.; Sayehli, C.; Chatterjee, M.; Einsele, H.; Bargou, R.C. Combined targeting of MEK/MAPK and PI3K/Akt signalling in multiple myeloma. Br. J. Haematol. 2012, 159, 430–440. [Google Scholar] [CrossRef] [PubMed]
- Medina, E.A.; Oberheu, K.; Polusani, S.R.; Ortega, V.; Velagaleti, G.V.; Oyajobi, B.O. PKA/AMPK signaling in relation to adiponectin’s antiproliferative effect on multiple myeloma cells. Leukemia 2014, 28, 2080–2089. [Google Scholar] [CrossRef] [PubMed]
- Qin, X.; Lin, L.; Cao, L.; Zhang, X.; Song, X.; Hao, J.; Zhang, Y.; Wei, R.; Huang, X.; Lu, J.; et al. Extracellular matrix protein Reelin promotes myeloma progression by facilitating tumor cell proliferation and glycolysis. Sci. Rep. 2017, 7, 45305:1–45305:12. [Google Scholar] [CrossRef] [PubMed]
- Tamura, H. Immunopathogenesis and immunotherapy of multiple myeloma. Int. J. Hematol. 2018, 107, 278–285. [Google Scholar] [CrossRef] [PubMed]
- Krejcik, J.; Frerichs, K.A.; Nijhof, I.S.; van Kessel, B.; van Velzen, J.F.; Bloem, A.C.; Broekmans, M.E.C.; Zweegman, S.; van Meerloo, J.; Musters, R.J.P. Monocytes and Granulocytes Reduce CD38 Expression Levels on Myeloma Cells in Patients Treated with Daratumumab. Clin. Cancer Res. 2017, 23, 7498–7511. [Google Scholar] [CrossRef] [PubMed]
- Hosen, N.; Matsunaga, Y.; Hasegawa, K.; Matsuno, H.; Nakamura, Y.; Makita, M.; Watanabe, K.; Yoshida, M.; Satoh, K.; Morimoto, S. The activated conformation of integrin β7 is a novel multiple myeloma-specific target for CAR T cell therapy. Nat. Med. 2017, 23, 1436–1443. [Google Scholar] [CrossRef] [PubMed]
- Kouidhi, S.; Ben Ayed, F.; Benammar Elgaaied, A. Targeting Tumor Metabolism: A New Challenge to Improve Immunotherapy. Front. Immunol. 2018, 9, 353. [Google Scholar] [CrossRef] [PubMed]
- Beckermann, K.E.; Dudzinski, S.O.; Rathmell, J.C. Dysfunctional T cell metabolism in the tumor microenvironment. Cytokine Growth Factor Rev. 2017, 35, 7–14. [Google Scholar] [CrossRef] [PubMed]
- Noël, G.; Fontsa, M.L.; Willard-Gallo, K. The impact of tumor cell metabolism on T cell-mediated immune responses and immuno-metabolic biomarkers in cancer. Semin. Cancer Biol. 2018, 1–28. [Google Scholar] [CrossRef] [PubMed]
- Housman, G.; Byler, S.; Heerboth, S.; Lapinska, K.; Longacre, M.; Snyder, N.; Sarkar, S. Drug resistance in cancer: An overview. Cancers 2014, 6, 1769–1792. [Google Scholar] [CrossRef] [PubMed]
- Parkin, B.; Ouillette, P.; Li, Y.; Keller, J.; Lam, C.; Roulston, D.; Li, C.; Shedden, K.; Malek, S.N. Clonal evolution and devolution after chemotherapy in adult acute myelogenous leukemia. Blood 2013, 121, 369–377. [Google Scholar] [CrossRef] [PubMed]
- Navin, N.; Krasnitz, A.; Rodgers, L.; Cook, K.; Meth, J.; Kendall, J.; Riggs, M.; Eberling, Y.; Troge, J.; Grubor, V.; et al. Inferring tumor progression from genomic heterogeneity. Genome Res. 2010, 20, 68–80. [Google Scholar] [CrossRef] [PubMed]
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).