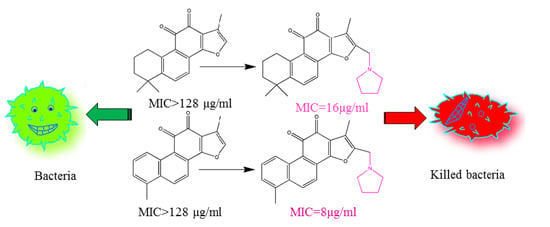
Unveiling the Mode of Action of Two Antibacterial Tanshinone Derivatives
Abstract
:1. Introduction
2. Results and Discussion
2.1. Preparation of Compound A and Compound B

2.2. Determination of Minimal Inhibitory Concentration (MIC) and Minimal Bactericidal Concentration (MBC)
| MIC/MBC (μg/mL) | |||||
|---|---|---|---|---|---|
| Gram Category | Microorganisms | Tan IIA | Compound A | Tan Ι | Compound B |
| G− | E. coli | >128/n d | >64/n d | >128/n d | >64/n d |
| P. aerurginosa | >128/n d | >64/n d | >128/n d | >64/n d | |
| S. typhimurium | >128/n d | >64/n d | >128/n d | >64/n d | |
| G+ | S. aureus | >128/n d | 16/64 | >128/n d | 8/16 |
| L. monocytogenes | >128/n d | 16/128 | >128/n d | 8/16 | |
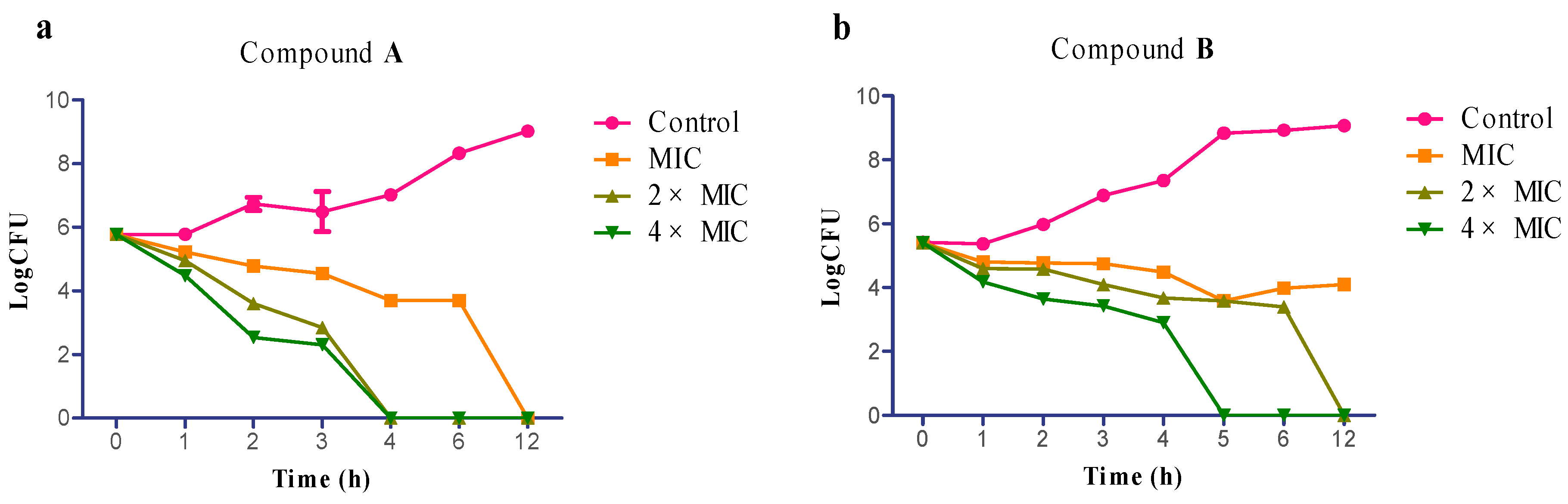
2.3. Time-Kill Assay
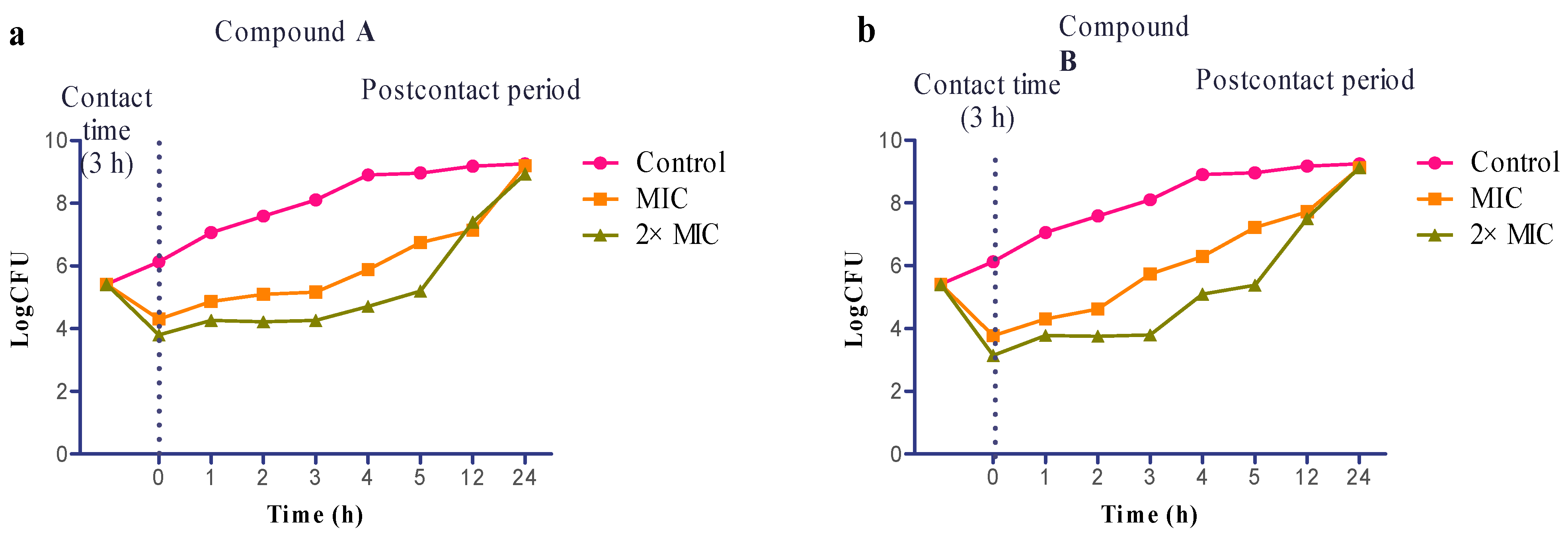
2.4. Evaluation of Postcontact Effect (PCE)
2.5. Involvement of Hydroxyl Radical in Staphylococcus aureus Inactivation
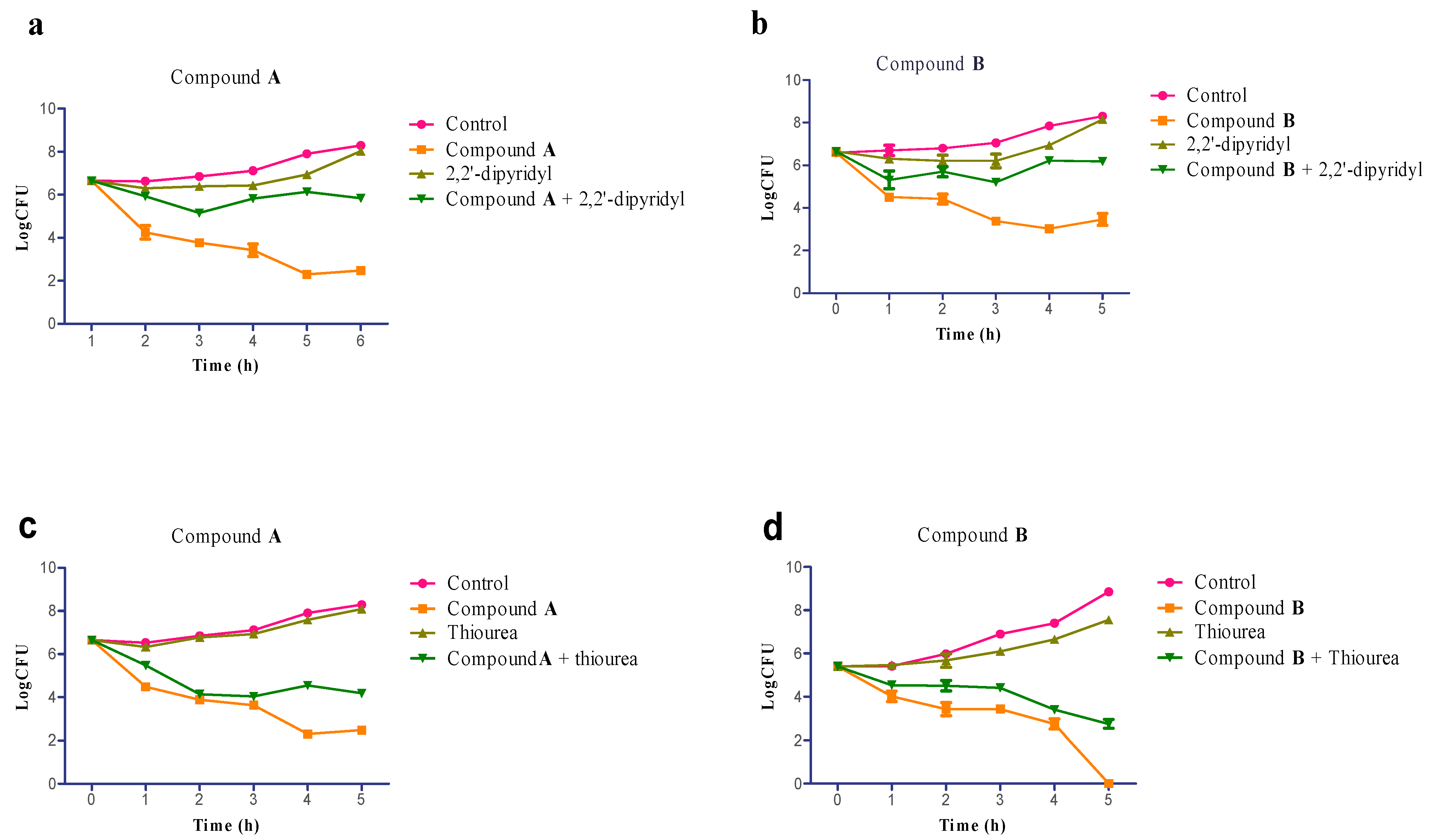
2.6. Scanning Electron Microscope (SEM)

2.7. Cell Constituents’ Release
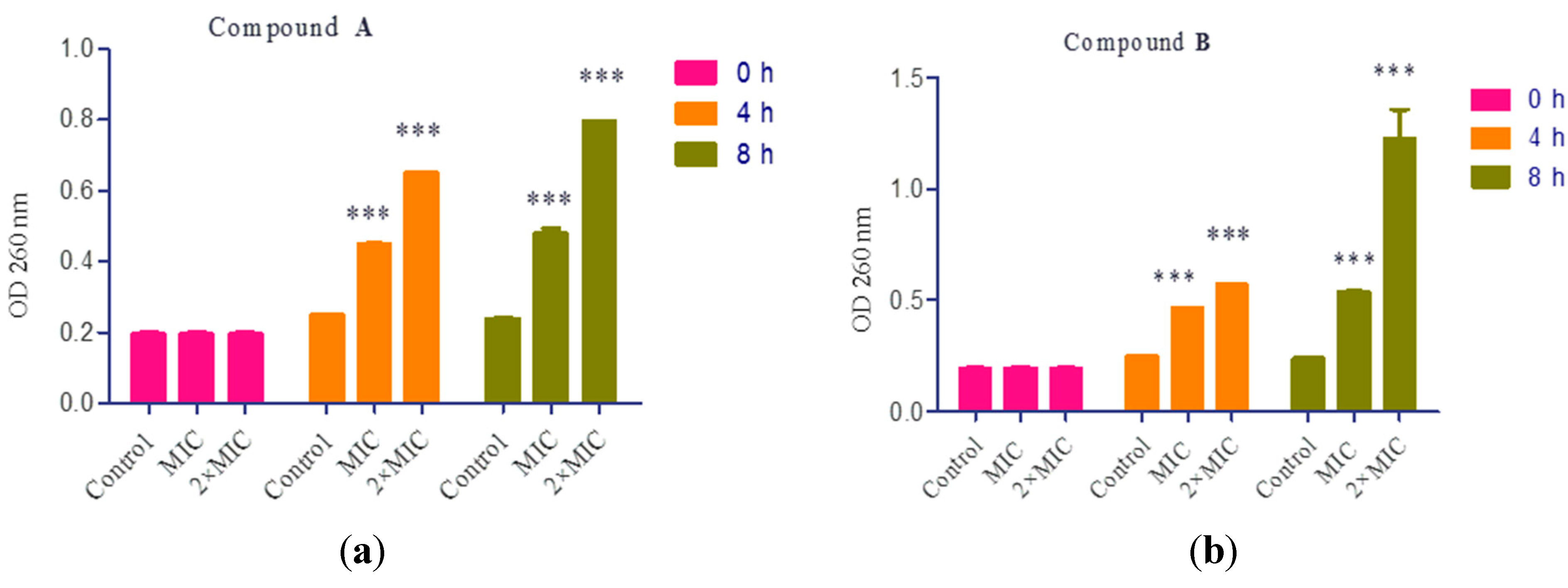
3. Materials and Methods
3.1. Chemistry
3.1.1. Preparation of Compound A
3.1.2. Preparation of Compound B
3.2. Biology
3.2.1. Bacterial Strains and Growth Conditions
3.2.2. Minimal Inhibitory Concentration (MIC) and Minimal Bactericidal Concentration (MBC)
3.2.3. Time-Kill Assay
3.2.4. Evaluation of Postcontact Effect (PCE)
3.2.5. Iron Chelator and Hydroxyl Radical Quenching Experiments
3.2.6. Scanning Electron Microscope (SEM)
3.2.7. Cell Constituents’ Release
4. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Butler, M.S.; Buss, A.D. Natural products—The future scaffolds for novel antibiotics? Biochem. Pharmacol. 2006, 71, 919–929. [Google Scholar] [CrossRef] [PubMed]
- Saleem, M.; Nazir, M.; Ali, M.S.; Hussain, H.; Lee, Y.S.; Riaz, N.; Jabbar, A. Antimicrobial natural products: An update on future antibiotic drug candidates. Nat. Prod. Rep. 2010, 27, 238–254. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Jiang, P.; Ye, M.; Kim, S.H.; Jiang, C.; Lu, J. Tanshinones: Sources, pharmacokinetics and anti-cancer activities. Int. J. Mol. Sci. 2012, 13, 13621–13666. [Google Scholar] [CrossRef] [PubMed]
- Lee, W.Y.; Chiu, L.C.; Yeung, J.H. Cytotoxicity of major tanshinones isolated from Danshen (Salvia miltiorrhiza) on HepG2 cells in relation to glutathione perturbation. Food Chem. Toxicol. 2008, 46, 328–338. [Google Scholar] [CrossRef] [PubMed]
- Nakao, M.; Fukushima, T. On the chemical composition of Salvia miltiorrhiza (Chinese drug Tan-shen). J. Pharm. Soc. Jpn. 1934, 54, 154–162. [Google Scholar]
- Han, J.Y.; Fan, J.Y.; Horie, Y.; Miura, S.; Cui, D.H.; Ishii, H.; Hibi, T.; Tsuneki, H.; Kimura, I. Ameliorating effects of compounds derived from Salvia miltiorrhiza root extract on microcirculatory disturbance and target organ injury by ischemia and reperfusion. Pharmacol. Ther. 2008, 117, 280–295. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Morris-Natschke, S.L.; Lee, K.H. New developments in the chemistry and biology of the bioactive constituents of Tanshen. Med. Res. Rev. 2007, 27, 133–148. [Google Scholar] [CrossRef] [PubMed]
- Zhao, J.; Lou, J.; Mou, Y.; Li, P.; Wu, J.; Zhou, L. Diterpenoid tanshinones and phenolic acids from cultured hairy roots of Salvia miltiorrhiza Bunge and their antimicrobial activities. Molecules 2011, 16, 2259–2267. [Google Scholar] [CrossRef] [PubMed]
- Tian, X.H.; Wu, J.H. Tanshinone derivatives: A patent review (January 2006–September 2012). Expert Opin. Ther. Pat. 2013, 23, 19–29. [Google Scholar] [CrossRef] [PubMed]
- Dong, Y.; Morris-Natschke, S.L.; Lee, K.H. Biosynthesis, total syntheses, and antitumor activity of tanshinones and their analogs as potential therapeutic agents. Nat. Prod. Rep. 2011, 28, 529–542. [Google Scholar] [CrossRef] [PubMed]
- Sun, C.J.; Bai, D.L. Synthesis of some compounds related to tanshinquinone. Yao Xue Xue Bao 1985, 20, 39–43. [Google Scholar] [PubMed]
- Bi, Y.F.; Wang, Z.J.; Guan, R.F.; Ye, Y.T.; Chen, Y.Y.; Zhang, Y.B.; Liu, H.M. Design, synthesis and vasodilative activity of tanshinone II A derivatives. Bioorg. Med. Chem. Lett. 2012, 22, 5141–5143. [Google Scholar] [CrossRef] [PubMed]
- Xu, S.; Liu, P. Tanshinone II-A: New perspectives for old remedies. Expert Opin. Ther. Pat. 2013, 23, 149–153. [Google Scholar] [CrossRef] [PubMed]
- Domagala, J.M. Structure-activity and structure-side-effect relationships for the quinolone antibacterials. J. Antimicrob. Chemother. 1994, 33, 685–706. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.L.; Fang, K.C.; Sheu, J.Y.; Hsu, S.L.; Tzeng, C.C. Synthesis and antibacterial evaluation of certain quinolone derivatives. J. Med. Chem. 2001, 44, 2374–2377. [Google Scholar] [CrossRef] [PubMed]
- Roman, G. Mannich bases in medicinal chemistry and drug design. Eur. J. Med. Chem. 2015, 89, 743–816. [Google Scholar] [CrossRef] [PubMed]
- Ait-Ouazzou, A.; Espina, L.; Gelaw, T.; Lamo-Castellví, S.; Pagán, R.; García-Gonzalo, D. New insights in mechanisms of bacterial inactivation by carvacrol. J. Appl. Microbiol. 2013, 114, 173–185. [Google Scholar] [CrossRef] [PubMed]
- Chueca, B.; Pagan, R.; Garcia-Gonzalo, D. Oxygenated monoterpenes citral and carvacrol cause oxidative damage in Escherichia coli without the involvement of tricarboxylic acid cycle and Fenton reaction. Int. J. Food Microbiol. 2014, 189, 126–131. [Google Scholar] [CrossRef] [PubMed]
- Carson, C.F.; Mee, B.J.; Riley, T.V. Mechanism of action of Melaleuca alternifolia (Tea Tree) oil on Staphylococcus aureus determined by time-kill, lysis, leakage, and salt tolerance assays and electron microscopy. Antimicrob. Agents Chemother. 2002, 46, 1914–1920. [Google Scholar] [CrossRef] [PubMed]
- Plech, T.; Wujec, M.; Siwek, A.; Kosikowska, U.; Malm, A. Synthesis and antimicrobial activity of thiosemicarbazides, s-triazoles and their Mannich bases bearing 3-chlorophenyl moiety. Eur. J. Med. Chem. 2011, 46, 241–248. [Google Scholar] [CrossRef] [PubMed]
- Wikler, M.A. Methods for Dilution Antimicrobial Susceptibility Tests for Bacteria that Grow Aerobically, Approved Standard—Eighth ed.; CLSI Document M07-A8: Wayne, PA, USA, 2003. [Google Scholar]
- Benincasa, M.; Zahariev, S.; Pelillo, C.; Milan, A.; Gennaro, R.; Scocchi, M. PEGylation of the peptide Bac7 (1–35) reduces renal clearance while retaining antibacterial activity and bacterial cell penetration capacity. Eur. J. Med. Chem. 2015, 95, 210–219. [Google Scholar] [CrossRef] [PubMed]
- Danac, R.; Mangalagiu, II. Antimycobacterial activity of nitrogen heterocycles derivatives: Bipyridine derivatives. Part III. Eur. J. Med. Chem. 2014, 74, 664–670. [Google Scholar] [CrossRef] [PubMed]
- Castro, M.A.; Gamito, A.M.; Tangarife-Castano, V.; Zapata, B.; Miguel del Corral, J.M.; Mesa-Arango, A.C.; Betancur-Galvis, L.; San Feliciano, A. Synthesis and antifungal activity of terpenyl-1,4-naphthoquinone and 1,4-anthracenedione derivatives. Eur. J. Med. Chem 2013, 67, 19–27. [Google Scholar] [CrossRef] [PubMed]
- Pournaras, S.; Vrioni, G.; Neou, E.; Dendrinos, J.; Dimitroulia, E.; Poulou, A.; Tsakris, A. Activity of tigecycline alone and in combination with colistin and meropenem against Klebsiella pneumoniae carbapenemase (KPC)-producing enterobacteriaceae strains by time-kill assay. Int. J. Antimicrob. Agents 2011, 37, 244–247. [Google Scholar] [CrossRef] [PubMed]
- Marceau, A.H.; Bernstein, D.A.; Walsh, B.W.; Shapiro, W.; Simmons, L.A.; Keck, J.L. Protein interactions in genome maintenance as novel antibacterial targets. PLoS ONE 2013, 8, e58765. [Google Scholar] [CrossRef] [PubMed]
- Daglia, M.; Papetti, A.; Gietroli, P.; Aceti, C.; Dacarro, C.; Gazzani, G. Antibacterial activity of red and white wine against oral streptococci. J. Agric. Food Chem. 2007, 55, 5038–5042. [Google Scholar] [CrossRef] [PubMed]
- Craig, W.A. The postantibiotic effect. Clin. Microbiol. Newsl. 1991, 13, 121–124. [Google Scholar] [CrossRef]
- Giguere, S.; Lee, E.A.; Guldbech, K.M.; Berghaus, L.J. In vitro synergy, pharmacodynamics, and postantibiotic effect of 11 antimicrobial agents against Rhodococcus equi. Vet. Microbiol. 2012, 160, 207–213. [Google Scholar] [CrossRef] [PubMed]
- Kohanski, M.A.; Dwyer, D.J.; Hayete, B.; Lawrence, C.A.; Collins, J.J. A common mechanism of cellular death induced by bactericidal antibiotics. Cell 2007, 130, 797–810. [Google Scholar] [CrossRef] [PubMed]
- Jensen, P.O.; Briales, A.; Brochmann, R.P.; Wang, H.; Kragh, K.N.; Kolpen, M.; Hempel, C.; Bjarnsholt, T.; Hoiby, N.; Ciofu, O. Formation of hydroxyl radicals contributes to the bactericidal activity of ciprofloxacin against Pseudomonas aeruginosa biofilms. Pathog. Dis. 2014, 70, 440–443. [Google Scholar] [CrossRef] [PubMed]
- Chueca, B.; Pagan, R.; Garcia-Gonzalo, D. Differential mechanism of Escherichia coli Inactivation by (+)-limonene as a function of cell physiological state and drug’s concentration. PLoS ONE 2014, 9, e94072. [Google Scholar] [CrossRef] [PubMed]
- Ghosh, S.; Indukuri, K.; Bondalapati, S.; Saikia, A.K.; Rangan, L. Unveiling the mode of action of antibacterial labdane diterpenes from Alpinia nigra (Gaertn.) B. L. Burtt seeds. Eur. J. Med. Chem. 2013, 66, 101–105. [Google Scholar] [CrossRef] [PubMed]
- Aidah, N.; Abdullah, N.; Oskoueian, E.; Sieo, C.C.; Saad, W.Z. Membrane-active antibacterial compounds in methanolic extracts of Jatropha curcas and their mode of action against Staphylococcus aureus S1434 and Escherichia coli E216. Int. J. Agric. Biol. 2014, 16, 723–730. [Google Scholar]
- Kaur, G.; Singh, R.P. Antibacterial and membrane damaging activity of Livistona chinensis fruit extract. Food Chem. Toxicol. 2008, 46, 2429–2934. [Google Scholar] [CrossRef] [PubMed]
- Sikkema, J.; de Bont, J.A.; Poolman, B. Mechanisms of membrane toxicity of hydrocarbons. Microbiol. Rev. 1995, 59, 201–222. [Google Scholar] [PubMed]
- Somolinos, M.; Garc, D.; Condon, S.; Mackey, B.; Pagan, R. Inactivation of Escherichia coli by citral. J. Appl. Microbiol. 2010, 108, 1928–1939. [Google Scholar] [PubMed]
- Trombetta, D.; Castelli, F.; Sarpietro, M.G.; Venuti, V.; Cristani, M.; Daniele, C.; Saija, A.; Mazzanti, G.; Bisignano, G. Mechanisms of antibacterial action of three monoterpenes. Antimicrob. Agents Chemother. 2005, 49, 2474–2478. [Google Scholar] [CrossRef] [PubMed]
- Janssen, A.M.; Scheffer, J.J.C.; Baerheim, S.A. Antimicrobial activity of Essential oils: A 1976–1986 literature review. Aspects of the test methods. Planta Med. 1987, 53, 395–398. [Google Scholar] [CrossRef] [PubMed]
- Bandyopadhyay, N.; Zhu, M.; Lu, L.; Mitra, D.; Das, M.; Das, P.; Samanta, A.; Naskar, J.P. Synthesis, structure, spectral characterization, electrochemistry and evaluation of antibacterial potentiality of a novel oxime-based palladium(II) compound. Eur. J. Med. Chem. 2015, 89, 59–66. [Google Scholar] [CrossRef] [PubMed]
- Sun, D.; Zhang, W.; Lv, M.; Yang, E.; Zhao, Q.; Wang, W. Antibacterial activity of ruthenium(II) polypyridyl complex manipulated by membrane permeability and cell morphology. Bioorg. Med. Chem. Lett. 2015, 25, 2068–2073. [Google Scholar] [CrossRef] [PubMed]
- Schallenberger, M.A.; Newhouse, T.; Baran, P.S.; Romesberg, F.E. The psychotrimine natural products have antibacterial activity against Gram-positive bacteria and act via membrane disruption. J. Antibiot. 2010, 63, 685–687. [Google Scholar] [CrossRef] [PubMed]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wang, D.; Zhang, W.; Wang, T.; Li, N.; Mu, H.; Zhang, J.; Duan, J. Unveiling the Mode of Action of Two Antibacterial Tanshinone Derivatives. Int. J. Mol. Sci. 2015, 16, 17668-17681. https://doi.org/10.3390/ijms160817668
Wang D, Zhang W, Wang T, Li N, Mu H, Zhang J, Duan J. Unveiling the Mode of Action of Two Antibacterial Tanshinone Derivatives. International Journal of Molecular Sciences. 2015; 16(8):17668-17681. https://doi.org/10.3390/ijms160817668
Chicago/Turabian StyleWang, Dongdong, Wuxia Zhang, Tingting Wang, Na Li, Haibo Mu, Jiwen Zhang, and Jinyou Duan. 2015. "Unveiling the Mode of Action of Two Antibacterial Tanshinone Derivatives" International Journal of Molecular Sciences 16, no. 8: 17668-17681. https://doi.org/10.3390/ijms160817668
APA StyleWang, D., Zhang, W., Wang, T., Li, N., Mu, H., Zhang, J., & Duan, J. (2015). Unveiling the Mode of Action of Two Antibacterial Tanshinone Derivatives. International Journal of Molecular Sciences, 16(8), 17668-17681. https://doi.org/10.3390/ijms160817668