Host-Guest Complexation Studied by Fluorescence Correlation Spectroscopy: Adamantane–Cyclodextrin Inclusion
Abstract
:1. Introduction
2. Theory
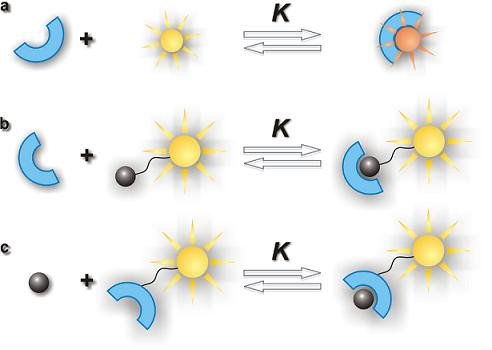
2.1. Mechanism
2.2. FCS
3. Results and Discussion
4. Experimental Section
4.1. Materials
4.2. Sample Preparation
4.3. FCS Measurements
4.4. Synthesis of the Ada-A488 Compound
Preparation of compound 1 (see Scheme 1)
Preparation of compound 2
Preparation of compound 4
Abbreviations Used
Suppliers
5. Conclusions
Acknowledgments
References and Notes
- Turro, NJ. Supramolecular structure and dynamics special feature: Molecular structure as a blueprint for supramolecular structure chemistry in confined spaces. Proc. Nat. Acad. Sci. USA 2005, 102, 10766–10770. [Google Scholar]
- Dodziuk, H. Introduction to Supramolecular Chemistry; Springer: Dordrecht, The Netherlands, 2002. [Google Scholar]
- Rigler, R; Elson, ES. Fluorescence Correlation Spectroscopy: Theory and Applications; Springer: Berlin, Germany, 2001. [Google Scholar]
- Al-Soufi, W; Reija, B; Novo, M; Felekyan, S; Kühnemuth, R; Seidel, CAM. Fluorescence correlation spectroscopy, a tool to investigate supramolecular dynamics: Inclusion complexes of pyronines with cyclodextrin. J. Am. Chem. Soc 2005, 127, 8775–8784. [Google Scholar]
- Al-Soufi, W; Reija, B; Felekyan, S; Seidel, CA; Novo, M. Dynamics of supramolecular association monitored by fluorescence correlation spectroscopy. Chemphyschem 2008, 9, 1819–1827. [Google Scholar]
- Cramer, F; Hettler, H. Inclusion compounds of cyclodextrins. Naturwissenschaften 1967, 54, 625–32. [Google Scholar]
- Szejtli, J. Introduction and general overview of cyclodextrin chemistry. Chem. Rev 1998, 98, 1743–1754. [Google Scholar]
- Dodziuk, H. Cyclodextrins and Their Complexes: Chemistry, Analytical Methods, Applications; Wiley: Weinheim, Germany, 2006. [Google Scholar]
- Carrazana, J; Reija, B; Ramos Cabrer, P; Al-Soufi, W; Novo, M; Vázquez Tato, J. Complexation of methyl orange with beta-cyclodextrin: Detailed analysis and application to quantification of polymer-bound cyclodextrin. Supramol. Chem 2004, 16, 549–559. [Google Scholar]
- Bordello, J; Reija, B; Al-Soufi, W; Novo, M. Host-assisted guest self-assembly: Enhancement of the dimerization of pyronines Y and B by gamma-cyclodextrin. Chemphyschem 2009, 10, 931–939. [Google Scholar]
- Reija, B; Al-Soufi, W; Novo, M; Vázquez Tato, J. Specific interactions in the inclusion complexes of Pyronines Y and B with beta-cyclodextrin. J. Phys. Chem. B 2005, 109, 1364–1370. [Google Scholar]
- Bohne, C. Supramolecular dynamics of guest complexation to cyclodextrins. Spectrum 2000, 13, 14–19. [Google Scholar]
- Harada, A; Li, J; Kamachi, M. Synthesis of a tubular polymer from threaded cyclodextrins. Nature 1993, 364, 516. [Google Scholar]
- Lehn, JM. Supramolecular Chemistry; VCH: Weinheim, Germany, 1995. [Google Scholar]
- Nepogodiev, SA; Stoddart, JF. Cyclodextrin-based catenanes and rotaxanes. Chem. Rev 1998, 98, 1959–1976. [Google Scholar]
- Alvarez Parrilla, E; Ramos Cabrer, P; Al-Soufi, W; Meijide del Río, F; Rodríguez Núñez, EA; Vázquez Tato, J. Dendritic growth of a supramolecular complex. Angew. Chem. Int. Ed. Engl 2000, 39, 2856–2858. [Google Scholar]
- Ogoshi, T; Harada, A. Chemical sensors based on cyclodextrin derivatives. Sensors 2008, 8, 4961–4982. [Google Scholar]
- Hennig, A; Bakirci, H; Nau, WM. Label-free continuous enzyme assays with macrocycle-fluorescent dye complexes. Nat. Methods 2007, 4, 629–632. [Google Scholar]
- Breslow, R; Belvedere, S; Gershell, L; Leung, D. The chelate effect in binding, catalysis, and chemotherapy. Pure Appl. Chem 2000, 72, 333–342. [Google Scholar]
- Loftsson, T; Brewster, ME. Pharmaceutical applications of cyclodextrins. 1. Drug solubilization and stabilization. J. Pharm. Sci 1996, 85, 1017–1025. [Google Scholar]
- Uekama, K; Hirayama, F; Irie, T. Cyclodextrin drug carrier systems. Chem. Rev 1998, 98, 2045–2076. [Google Scholar]
- Hirayama, F; Uekama, K. Cyclodextrin-based controlled drug release system. Adv. Drug Deliv. Rev 1999, 36, 125–141. [Google Scholar]
- Lezcano, M; Al-Soufi, W; Novo, M; Rodríguez-Núñez, E; Vázquez Tato, J. Complexation of several benzimidazole-type fungicides with alpha- and beta-cyclodextrins. J. Agric. Food Chem 2002, 50, 108–112. [Google Scholar]
- Ritter, H; Tabatabai, M. Cyclodextrin in polymer synthesis: A green way to polymers. Progr. Polym. Sci 2002, 27, 1713–1720. [Google Scholar]
- Davis, ME; Brewster, ME. Cyclodextrin-based pharmaceutics: Past, present and future. Nat. Rev. Drug Discov 2004, 3, 1023–1035. [Google Scholar]
- Eftink, MR; Andy, ML; Bystrom, K; Perlmutter, HD; Kristol, DSJ. Cyclodextrin inclusion complexes: Studies of the variation in the size of alicyclic guests. J. Am. Chem. Soc 1989, 111, 6765–6772. [Google Scholar]
- Cromwell, WC; Bystrom, K; Eftink, MR. Cyclodextrin-adamantanecarboxylate inclusion complexes: Studies of the variation in cavity size. J. Phys. Chem 1985, 89, 326–332. [Google Scholar]
- Gelb, RI; Schwartz, LM. Complexation of admantane-ammonium substrates by beta-cyclodextrin and its O-methylated derivatives. J. Incl. Phenom. Mol. Recogn. Chem 1989, 7, 537–543. [Google Scholar]
- Palepu, R; Reinsborough, VC. β-cyclodextrin inclusion of adamantane derivatives in solution. Aust. J. Chem 1990, 43, 2119–2123. [Google Scholar]
- Kwak, ES; Gomez, FA. Determination of the binding of β-cyclodextrin derivatives to adamantane carboxylic acids using capillary electrophoresis. Chromatographia 1996, 43, 659–662. [Google Scholar]
- Harries, D; Rau, DC; Parsegian, VA. Solutes probe hydration in specific association of cyclodextrin and adamantane. J. Am. Chem. Soc 2005, 127, 2184–2190. [Google Scholar]
- Koopmans, C; Ritter, H. Formation of physical hydrogels via host-guest interactions of b-cyclodextrin polymers and copolymers bearing adamantyl groups. Macromolecules 2008, 41, 7418–7422. [Google Scholar]
- Holzinger, M; Bouffier, L; Villalonga, R; Cosnier, S. Adamantane/β-cyclodextrin affinity biosensors based on single-walled carbon nanotubes. Biosens. Bioelectron 2009, 24, 1128–1134. [Google Scholar]
- Park, I; von Recum, HA; Jiang, S; Pun, SH. Spatially-controlled delivery of cyclodextrin-based polyplexes from solid surfaces. Mol. Ther 2006, 13, S67–S67. [Google Scholar]
- Bellocq, NC; Pun, SH; Jensen, GS; Davis, ME. Transferrin-containing, cyclodextrin polymer-based particles for tumor-targeted gene delivery. Bioconjug. Chem 2003, 14, 1122–1132. [Google Scholar]
- Munteanu, M; Choi, S; Ritter, H. Cyclodextrin-click-cucurbit[6]uril: Combi-receptor for supramolecular polymer systems in water. Macromolecules 2009, 42, 3887–3891. [Google Scholar]
- Soto Tellini, VH; Jover, A; Carrazana Garcia, J; Galantini, L; Meijide, F; Vázquez Tato, J. Thermodynamics of formation of host-guest supramolecular polymers. J. Am. Chem. Soc 2006, 128, 5728–5734. [Google Scholar]
- Widengren, J; Rigler, R. Fluorescence correlation spectroscopy as a tool to investigate chemical reactions in solutions and on cell surfaces. Cell Mol. Biol 1998, 44, 857–879. [Google Scholar]
- Widengren, J. Photophysical aspects of FCS Measurements; Rigler, R, Elson, ES, Eds.; Springer Verlag: Berlin, Germany, 2001; p. 276. [Google Scholar]
- Haustein, E; Schwille, P. Fluorescence Correlation Spectroscopy in Vitro and in Vivo; Selvin, PR, Ha, T, Eds.; Cold Spring Harbor Laboratory Press: New York, NY, USA, 2008; p. 259. [Google Scholar]
- Elson, EL; Magde, D. Fluorescence correlation spectroscopy. I. Conceptual basis and theory. Biopolymers 1974, 13, 1–27. [Google Scholar]
- Enderlein, J; Gregor, I; Patra, D; Dertinger, T; Kaupp, UB. Performance of fluorescence correlation spectroscopy for measuring diffusion and concentration. Chemphyschem 2005, 6, 2324–2336. [Google Scholar]
- Alcor, D; Allemand, JF; Cogne-Laage, E; Croquette, V; Ferrage, F; Jullien, L; Kononov, A; Lemarchand, A. Stochastic resonance to control diffusive motion in chemistry. J. Phys. Chem. B 2005, 109, 1318–1328. [Google Scholar]
- Carrazana, J; Jover, A; Meijide, F; Soto, VH; Vázquez Tato, J. Complexation of adamantyl compounds by beta-cyclodextrin and monoaminoderivatives. J. Phys. Chem. B 2005, 109, 9719–9726. [Google Scholar]
- Felekyan, S; Kuhnemuth, R; Kudryavtsev, V; Sandhagen, C; Becker, W; Seidel, CAM. Full correlation from picoseconds to seconds by time-resolved and time-correlated single photon detection. Rev Sci Instrum 2005, 76, 083104:1–083104:14. [Google Scholar]
- Eggeling, C; Widengren, J; Rigler, R; Seidel, CAM. Photobleaching of fluorescent dyes under conditions used for single-molecule detection: Evidence of two-step photolysis. Anal. Chem 1998, 70, 2651–2659. [Google Scholar]
- Gendron, PO; Avaltroni, F; Wilkinson, KJ. Diffusion coefficients of several rhodamine derivatives as determined by pulsed field gradient-nuclear magnetic resonance and fluorescence correlation spectroscopy. J. Fluoresc 2008, 18, 1093–1101. [Google Scholar]
- Muller, C; Loman, A; Pacheco, V; Koberling, F; Willbold, D; Richtering, W. Precise measurement of diffusion by multi-color dual-focus fluorescence correlation spectroscopy. Europhys Lett 2008, 83, 46001:1–46001:6. [Google Scholar]






| Ada-A488 + βCD | |
|---|---|
| K/103 M−1 | 52 ± 2 |
| τf/ms | 0.222 ± 0.002 |
| τb/ms | 0.300 ± 0.002 |
| AT | 0.20 |
| τT/μs | 4.8 |
| Df/10−10 m2s−1 | 3.15 ± 0.30 |
| Db/10−10 m2s−1 | 2.33 ± 0.20 |
| Rh,f/Å | 7.8 ± 0.7 |
| Rh,b/Å | 10.5 ± 0.9 |
© 2010 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Granadero, D.; Bordello, J.; Pérez-Alvite, M.J.; Novo, M.; Al-Soufi, W. Host-Guest Complexation Studied by Fluorescence Correlation Spectroscopy: Adamantane–Cyclodextrin Inclusion. Int. J. Mol. Sci. 2010, 11, 173-188. https://doi.org/10.3390/ijms11010173
Granadero D, Bordello J, Pérez-Alvite MJ, Novo M, Al-Soufi W. Host-Guest Complexation Studied by Fluorescence Correlation Spectroscopy: Adamantane–Cyclodextrin Inclusion. International Journal of Molecular Sciences. 2010; 11(1):173-188. https://doi.org/10.3390/ijms11010173
Chicago/Turabian StyleGranadero, Daniel, Jorge Bordello, Maria Jesus Pérez-Alvite, Mercedes Novo, and Wajih Al-Soufi. 2010. "Host-Guest Complexation Studied by Fluorescence Correlation Spectroscopy: Adamantane–Cyclodextrin Inclusion" International Journal of Molecular Sciences 11, no. 1: 173-188. https://doi.org/10.3390/ijms11010173
APA StyleGranadero, D., Bordello, J., Pérez-Alvite, M. J., Novo, M., & Al-Soufi, W. (2010). Host-Guest Complexation Studied by Fluorescence Correlation Spectroscopy: Adamantane–Cyclodextrin Inclusion. International Journal of Molecular Sciences, 11(1), 173-188. https://doi.org/10.3390/ijms11010173