Abstract
Cadmium carbonate was found to be a useful promoter in the Koenigs-Knorr synthesis of 2-(4-methoxybenzyl)cyclohexyl-β-d-glycopyranosides. Using this promoter model glucoside and galactoside derivatives of cyclic (i.e., secondary) alcohols were synthesized in 50–60 % overall yields. Diastereoisomeric mixtures of products were obtained in these syntheses, which started from racemic isomers of 2-(4-methoxy-benzyl)cyclohexanol. The prepared compounds have been purified and characterized by their 1H- and 13C-NMR spectra, as well as by their IR and MS spectra, in order to use them as reference compounds in planned subsequent research.
Introduction
The Koenigs-Knorr method for the synthesis of glycosides and alkyl glycosides probably represents the most widely used procedure in the chemistry of carbohydrate derivatives. Application of the Koenigs-Knorr method confers a strong glycosyl donor character to the activated species by exchange of the anomeric hydroxyl functionality by a bromine or chlorine atom in the activation step [1,2,3]. The α-halosubstituted carbohydrate generated in the activation step, can be readily further activated in the glycosylation step by halophilic promoters, i.e. heavy metal salts, resulting in an irreversible glycosyl transfer to the acceptor. This method is the basis of a valuable technique for the synthesis of complex alkyl glycosides, oligosaccharides or glycoconjugates. It has been continuously developed over the years and is widely employed [1,2,3,4,5], but in spite of the generality of the Koenigs-Knorr method, its requirement for up to 4 equivalents of heavy metal salts as promoter can be a limiting factor for large-scale preparations.
More than 20 years ago, we had used cadmium carbonate [6] for a successful synthesis of 2-[4-(3-ethoxy-3-methyl-1-butoxy)benzyl]cyclohexyl-β-d-glucopyranosides, derived from the separated racemic cis and trans isomers of 2-[4-(3-ethoxy-3-methyl-1-butoxy)benzyl]cyclohexanol, insect juvenile hormone bioanalogs [7]. At that time, only 100 MHz NMR spectra of 2-[4-(3-ethoxy-3-methyl-1-butoxy)benzyl]cyclohexyl-β-d-glucopyranoside products were measured, which ultimately were not published [6]. Recently, we have again focused our attention on alkyl glycosides. Although a remarkable variety of heavy metal salts have been reported to successfully promote the Koenigs-Knorr synthesis [2,3,4,5], we have applied our original method [6] again to thus obtain several reference compounds for a more comprehensive study of the glycosylation reaction. The currently used modification of the Koenigs-Knorr synthesis of alkyl glycosides has been proven to result in the formation of only the β-anomers of the alkyl glycoside products [2,3,4,5,6]. This selectivity is the key advantage of the method employed for the present study, undertaken with the purpose of obtaining detailed analytical data for the products. Using cadmium carbonate as the promoter of the Koenigs-Knorr synthesis this time we obtained 2-(4-methoxybenzyl)cyclohexyl-β-d-glucopyranosides and -β-d-galactopyranosides. Our synthesis started from the separated racemic isomers of 2-(4-methoxybenzyl)cyclohexanol, useful model compounds in the investigation of several reactions used for transformations of 2-substituted cycloalkanols into other desirable derivatives, and resulted in the obtention of diastereoisomeric mixtures of the respective products. From the 1H- and 13C-NMR spectra of these products, as well as their IR and MS (FAB) spectra, valuable analytical data have been obtained. This study was also performed in order to develop and to prove the usefulness of a convenient method for our ongoing preparations of a series of biologically active alkyl glycosides, using the racemic isomers of 2-(4-methoxybenzyl)cyclohexanol as a model secondary alcohol.
Results and Discussion
The modification of the Koenigs-Knorr synthesis of alkyl glycosides used in the first synthetic step of the experiments described started from the separated racemic cis and trans isomers of 2-(4-methoxybenzyl)cyclohexanol (1 and 2), and, therefore, the prepared products 3a/3b – 6a/6b, and subsequently also the products of the deprotection reaction (7a/7b – 10a/10b), are diastereoisomeric mixtures (Scheme 1). A 1:1 ratio of the respective diastereoisomers was found in each mixture, which indicates that there was no detectable preference for either enantiomer of the isomeric racemic alcohols 1 and 2 during the Koenigs-Knorr synthesis.
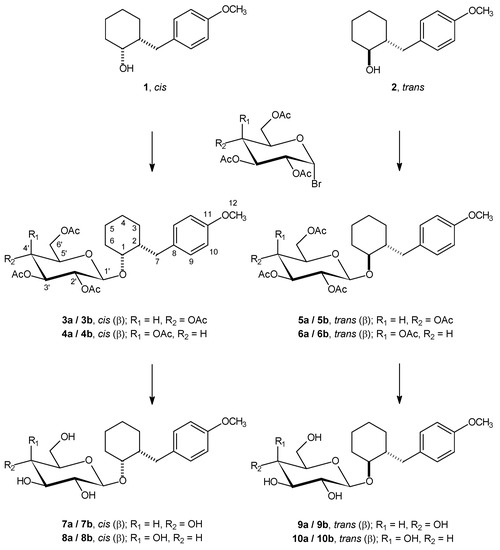
Scheme 1.
Scheme 1.
Our modification of the Koenigs-Knorr method consisted of maintaining rigorously anhydrous conditions for the whole duration of the reaction via continuous azeotropic removal of any humidity potentially present or entering the system, an approach that always resulted in the most advantageous reaction course for the Koenigs-Knorr synthesis of the alkyl glycoside derivatives 3a/3b – 6a/6b. The second synthetic step is a simple deprotection (removal of acetyl protecting groups from the carbohydrate moieties) of compounds 3a/3b – 6a/6b, using potassium carbonate in a 4:1 methanol / water medium [8] to give the final products 7a/7b – 10a/10b in good yields (see Experimental).
It was found that the Koenigs-Knorr synthesis resulted in higher yields of the products 5a/5b and 6a/6b derived from the trans-alcohol 2, a result which was expected due to higher steric hindrance during the substitution of the axial hydroxyl functionality in the cis-alcohol 1. In turn, deprotection of compounds 3a/3b – 6a/6b took place faster and was more successful in the case of compounds 3a/3b and 4a/4b, a finding that can be explained by the same steric hindrance, on account of the expected different molecular volumes of the respective glycosyloxy and 2’,3’,4’,6’-tetra-O-acetylglycosyloxy groups, which also display different spatial orientations: thus, the C(1)-O bond (Scheme 1) is axial in compounds 3a/3b, 4a/4b, 7a/7b and 8a/8b, and equatorial in compounds 5a/5b, 6a/6b, 9a/9b and 10a/10b [9].
The synthesized diastereoisomeric mixtures of products 3a/3b – 10a/10b were used as reference compounds for recording the desired model 1H- and 13C-NMR spectra, supported by standard recording of their MS (FAB) and IR spectra. It is a great advantage of the applied modification of the Koenigs-Knorr synthesis [2] that only β-anomers of the products occur in the reaction mixtures. The 1H-1H PFG COSY and the 1H-13C PFG HSQC experiments [10,11] were performed using standard pulse programs, as provided by the spectrometer manufacturer, to unambiguously assign the signals in the recorded spectra. Characteristic parameters of the experiments are given in the Experimental part. The obtained data are presented in the Experimental part (1H-NMR data) and in Table 1 and Table 2 (13C-NMR data).
Conclusions
The used modification of the Koenigs-Knorr synthesis [1,2,3] of alkyl glycosides has proven to be a valuable and convenient procedure for preparation of pure 2-(4-methoxybenzyl)cyclohexyl-β-d-glucopyranosides (3a/3b and 5a/5b) and -β-d-galactopyranosides (4a/4b and 6a/6b), i.e. glycosides derived from the secondary cyclic alcohols. It has also been proven by analysis of all minor compounds in the reaction mixtures resulting from the synthesis of the compounds 3a/3b – 6a/6b that no α-anomers of 3a/3b – 6a/6b were formed during the reaction. Finally, it has been proven that this synthetic approach is a convenient method to be applied in the ongoing synthesis of biologically active alkyl-β-d-glucopyranosides and alkyl-β-d-galactopyranosides.
Experimental
General
The 1H-NMR and the 13C-NMR spectra were recorded on a Bruker AVANCE 500 spectrometer (in FT mode) at 500.1 MHz and 125.8 MHz, respectively, in CDCl3 or in CD3OD, using either tetramethylsilane (δ 0.0 – CDCl3) or a solvent signal (CD3OD, δ 3.31 for 1H- and δ 49.50 for 13C-) as internal references. 2D-NMR experiments were measured using the following characteristic parameters: 1H-1H PFG COSY – spectral width 9 ppm in both f1, f2 dimensions, delay 1 s, data matrix for processing 2048 × 2048 data points; 1H-13C PFG HSQC – spectral width 9 ppm in f2 and 160 ppm in f1, delay 1 s, data matrix for processing 2048 × 2048 data points. IR spectra were recorded in solution (CCl4) or by the KBr technique on a Bruker IFS 88 instrument. MS (FAB) were recorded on a VG analytical 70 - 250 SE mass spectrometer, ZAB-EQ (BEQQ configuration) at 70 eV. Preparative column chromatography was performed on a silica gel type 60 (particle size 0.04 – 0.063 mm, Fluka, Switzerland). TLC was performed on aluminum sheets precoated with silica gel 60 (Merck, Germany). Analytical HPLC was carried out on a TSP (Thermoseparation Products, USA) instrument equipped with a ConstaMetric 4100 Bio pump and a SpectroMonitor 5000 UV DAD. Product analyses were performed on a Sepharon SGX C-18 reverse phase column (250 × 4 mm, particle size 7 mm, Watrex, Czech Republic) or on a chiral Nucleodex β-OH column (150 x 4 mm, Macherey-Nagel, Germany) using 4:1 (v/v) methanol-water as mobile phase at 0.5 mL min-1 or at 0.3 mL min-1. The eluate was monitored at 220, 254 and 275 nm and UV spectra were run from 200 to 300 nm.
2-(4-Methoxybenzyl)cyclohexyl-2’,3’,4’,6’-tetra-O-acetyl-β-d-glucopyranoside (3a/3b and 5a/5b) and 2-(4-methoxybenzyl)cyclohexyl-2’,3’,4’,6’-tetra-O-acetyl-β-d-galactopyranoside (4a/4b and 6a/6b).
The respective isomers of 2-(4-methoxybenzyl)cyclohexanol (0.272 g; 1.24 mmol) were dissolved in toluene (30 mL), and cadmium carbonate (0.656 g; 3.72 mmol) was added. A portion of the toluene (7 to 10 mL) was distilled off under azeotropic conditions to remove all traces of water from the system. A solution of 2,3,4,6-tetra-O-acetyl-α-d-glucopyranosyl bromide or 2,3,4,6-tetra-O-acetyl-α-d-galactopyranosyl bromide (1.528 g; 3.72 mmol) in toluene (2 mL) was then added over 5 minutes to the reaction mixture, which was further stirred and heated under azeotropic conditions for an additional 6 h. A mixture of inorganic salts was removed by filtration, and the solvent was evaporated from the filtrate, affording a dry residue. Silica gel column chromatography (eluent: 3:1 to 1:1 step-wise gradient of light petroleum – ether) yielded the pure products in yields of 61.6 % (3a/3b), 61.8 % (4a/4b), 83.6 % (5a/5b) and 73.7 % (6a/6b), respectively.
3a/3b: 1H-NMR (CDCl3): Diastereoisomer A: 1.17 – 1.43 (m, 6H, H-3a, 3b, 4a, 5a, 5b and 6a), 1.57 – 1.68 (m, 2H, H-2 and 4b), 1.84 (m, 1H, H-6b), 2.05 (s, 3H, OAc), 2.06 (s, 3H, OAc), 2.08 (s, 6H, 2 × OAc), 2.41 (dd, J = 6.4, 13.8Hz, 1H, H-7a), 2.72 (dd, J = 7.8, 13.8Hz, 1H, H-7b), 3.65 (ddd, J = 2.6, 4.7, 10.0Hz, 1H, H-5’), 3.80 (s, 3H, H-12), 3.81 (dt, J = 2.4, 2.4, 4.3Hz, 1H, H-1), 4.14 (dd, J = 2.6, 12.2Hz, 1H, H-6’a), 4.28 (dd, J = 4.7, 12.2Hz, 1H, H-6’b), 4.56 (d, J = 7.9Hz, 1H, H-1’), 5.11 (dd, J = 7.9, 9.9Hz, 1H, H-2’), 5.15 (t, J = 9.8Hz, 1H, H-4’), 5.22 (t, J = 9.8Hz, 1H, H-3’), 6.77 (m, 2H, H-9), 7.13 (m, 2H, H-8); Diastereoisomer B: 1.17 – 1.43 (m, 6H, H-3a, 3b, 4a, 5a, 5b and 6a), 1.57 – 1.68 (m, 2H, H-2 and 4b), 1.84 (m, 1H, H-6b), 2.02 (s, 6H, 2 × OAc), 2.03 (s, 6H, 2 × OAc), 2.39 (dd, J = 7.7, 13.6Hz, 1H, H-7a), 2.53 (dd, J = 7.0, 13.6Hz, 1H, H-7b), 3.64 (ddd, J = 2.6, 5.5, 10.1Hz, 1H, H-5’), 3.65 (dt, J = 2.4, 2.4, 4.5Hz, 1H, H-1), 3.77 (s, 3H, H-12), 4.11 (dd, J = 2.6, 12.2Hz, 1H, H-6’a), 4.21 (dd, J = 5.5, 12.2Hz, 1H, H-6’b), 4.55 (d, J = 7.9Hz, 1H, H-1’), 5.06 (dd, J = 9.3, 10.1Hz, 1H, H-4’), 5.09 (dd, J = 7.9, 9.8Hz, 1H, H-2’), 5.23 (t, J = 9.5Hz, 1H, H-3’), 6.84 (m, 2H, H-10), 7.06 (m, 2H, H-9). IR (cm-1, CCl4): 3027 (w), 2960 (w), 2936 (w), 2836 (w), 1756 (s), 1613 (w), 1513 (m), 1441 (w), 1388 (m), 1300 (w), 1245 (s), 1220 (s), 1042 (s), 895 (w), 843 (w). MS (FAB): [M]+ 550 (4), 331 (28), 289 (5), 229 (6), 203 (24), 169 (56), 121 (100), 109 (40), 77 (8). [α]D20 = -99 (c 0.22, CHCl3). For C28H38O11 (550.61): C 61.08, H 6.96; found: C 60.92, H 6.95.
4a/4b: 1H-NMR (CDCl3): Diastereoisomer A: 0.89 (m, 1H, H-3a), 1.18 (m, 1H, H-5a), 1.22 (m, 1H, H-4a), 1.36 (m, 1H, H-6), 1.57 m, 1H, H-5b), 1.62 (m, 1H, H-2), 1.63 (m, 1H, H-3b), 1.67 (m, 1H, H-4b), 1.84 (m, 1H, H-6b), 2.00 (s, 3H, OAc), 2.02 (s, 3H, OAc), 2.09 (s, 3H, OAc), 2.17 (s, 3H, OAc), 2.44 (dd, J = 7.0, 13.8Hz, 1H, H-7a), 2.74 (dd, J = 7.4, 13.8Hz, 1H, H-7b), 3.80 (s, 3H, H-12), 3.81 (dt, J = 2.3, 2.3, 4.5Hz, 1H, H-1), 3.87 (dt, J = 1.2, 6.7, 6.7Hz, 1H, H-5’), 4.16 (d, J = 6.7Hz, 2H, H-6a, H-6b), 4.51 (d, J = 7.9Hz, 1H, H-1’), 5.05 (dd, J = 3.5, 10.5Hz, 1H, H-3’), 5.30 (dd, J = 7.9, 10.5Hz, 1H, H-2’), 5.40 (dd, J = 1.2, 3.5Hz, 1H, H-4’), 6.80 (m, 2H, H-10), 7.13 (m, 2H, H-9); Diastereoisomer B: 0.89 (m, 1H, H-3a), 1.18 (m, 1H, H-5a), 1.22 (m, 1H, H-4a), 1.36 (m, 1H, H-6), 1.57 m, 1H, H-5b), 1.62 (m, 1H, H-2), 1.63 (m, 1H, H-3b), 1.67 (m, 1H, H-4b), 1.84 (m, 1H, H-6b), 2.00 (s, 3H, OAc), 2.02 (s, 3H, OAc), 2.06 (s, 3H, OAc), 2.09 (s, 3H, OAc), 2.41 (dd, J = 8.0, 13.6Hz, 1H, H-7a), 2.54 (dd, J = 6.8, 13.6Hz, 1H, H-7b), 3.66 (dt, J = 2.6, 2.6, 4.6Hz, 1H, H-1), 3.78 (s, 3H, H-12), 3.84 (dt, J = 1.2, 6.7, 6.7Hz, 1H, H-5’), 4.08 (dd, J = 6.8, 11.2Hz, 1H, H-6’a), 4.16 (dd, J = 6.6, 11.2Hz, 1H, H-6’b), 4.50 (d, J = 7.9, 1H, H-1’), 5.04 (dd, J = 3.5, 10.5Hz, 1H, H-3’), 5.26 (dd, J = 7.9, 10.5Hz, 1H, H-2’), 5.38 (dd, J = 1.2, 3.5Hz, 1H, H-4’), 6.84 (m, 2H, H-10), 7.02 (m, 2H, H-9). IR (cm-1, CCl4): 3063 (w), 3030 (w), 2936 (m), 2859 (w), 2836 (w), 1757 (s), 1613 (w), 1513 (s), 1441 (w), 1369 (s), 1300 (w), 1246 (s), 1221 (s), 1173 (s), 1056 (s), 1045 (s), 898 (w), 842 (w). MS (FAB): [M]+ 550 (1), 331 (10), 203 (12), 169 (24), 121 (100), 109 (28), 91 (10). [α]D20 = -70 (c 0.24, CHCl3). For C28H38O11 (550.61): C 61.08, H 6.96; found: C 61.00, H 6.92.
5a/5b: 1H-NMR (CDCl3): Diastereoisomer A: 0.88 (m, 1H, H-3a), 1.10 (m, 1H, H-5a), 1.22 (m, 1H, H-4a), 1.42 (m, 1H, H-6a), 1.56 (m, 1H, H-5b), 1.58 (m, 1H, H-2), 1.63 (m, 1H, H-3b), 1.72 (m, 1H, H-4b), 2.00 (s, 3H, OAc), 2.03 (s, 3H, OAc), 2.05 (s, 3H, OAc), 2.08 (s, 3H, OAc), 2.16 (m, 1H, H-6b), 2.19 (dd, J = 9.3, 13.6Hz, 1H, H-7a), 3.13 (dd, J = 3.6, 13.6Hz, 1H, H-7b), 3.32 (dt, J = 4.2, 9.5, 9.5Hz, 1H, H-1), 3.71 (ddd, J = 2.6, 5.4, 10.1Hz, 1H, H-5’), 3.78 (s, 3H, H-12), 4.17 (dd, J = 2.6, 12.1Hz, 1H, H-6’a), 4.27 (dd, J = 5.4, 12.1Hz, 1H, H-6’b), 4.63 (d, J = 8.0, 1H, H-1’), 5.08 (dd, J = 8.0, 9.6Hz, 1H, H-2’), 5.11 (t, J = 9.8Hz, 1H, H-4’), 5.22 (t, J = 9.6Hz, 1H, H-3’), 6.81 (m, 2H), 7.09 (m, 2H); Diastereoisomer B: 0.88 (m, 1H, H-3a), 1.10 (m, 1H, H-5a), 1.22 (m, 1H, H-4a), 1.42 (m, 1H, H-6a), 1.56 (m, 1H, H-5b), 1.58 (m, 1H, H-2), 1.63 (m, 1H, H-3b), 1.72 (m, 1H, H-4b), 2.00 (s, 3H, OAc), 2.03 (s, 3H, OAc), 2.05 (s, 3H, OAc), 2.08 (s, 3H, OAc), 2.09 (dd, J = 8.9, 13.2Hz, 1H, H-7a), 3.07 (dd, J = 3.3, 13.2Hz, 1H, H-7b), 3.21 (dt, J = 4.3, 9.7, 9.7Hz, 1H, H-1), 3.68 (ddd, J = 2.7, 4.7, 10.0Hz, 1H, H-5’), 3.78 (s, 3H, H-12), 4.13 (dd, J = 2.7, 12.1Hz, 1H, H-6’a), 4.25 (dd, J = 4.7, 12.1Hz, 1H, H-6’b), 4.62 (d, J = 8.0, 1H, H-1’), 5.01 (dd, J = 8.0, 9.8Hz, 1H, H-2’), 5.10 (t, J = 9.8Hz, 1H, H-4’), 5.22 (t, J = 9.6Hz, 1H, H-3’), 6.80 (m, 2H, H-10), 7.02 (m, 2H, H-9). IR (cm-1, CCl4): 3031 (w), 2935 (m), 2859 (w), 2853 (w), 1761 (s), 1753 (s), 1613 (w), 1513 (m), 1442 (w), 1300 (w), 1246 (s), 1225 (s), 1040 (s), 880 (w), 856 (w). MS (FAB): [M]+ 550 (2), 331 (25), 289 (10), 229 (8), 202 (14), 169 (62), 121 (100), 109 (46), 91 (16). [α]D20 = -103 (c 0.25, CHCl3). For C28H38O11 (550.61): C 61.08, H 6.96; found: C 61.05, H 6.98.
6a/6b: 1H-NMR (CDCl3): Diastereoisomer A: 0.88 (m, 1H, H-3a), 1.06 (m, 1H, H-5a), 1.19 (m, 1H, H-6a), 1.20 (m, 1H, H-4a), 1.52 (m, 1H, H-5b), 1.58 (m, 1H, H-2), 1.64 (m, 1H, H-3b), 1.71 (m, 1H, H-4b), 1.95 (m, 1H, H-6b), 2.09 (s, 3H), 2.09-2.14 (m, 4H, H-7a, OAc), 2.12 (s, 3H, OAc), 2.14 (s, 3H, OAc), 2.16 (s, 3H, OAc), 3.19 (dd, J = 3.8, 13.4Hz, 1H, H-7b), 3.32 (dt, J = 4.1, 9.5, 9.5Hz, 1H, H-1), 3.78 (s, 3H, H-12), 3.92 (dt, J = 1.3, 5.8, 6.8Hz, H-5’), 4.10 (dd, J = 5.8, 11.2Hz, 1H, H-6’a), 4.22 (dd, J = 6.8, 11.2Hz, 1H, H-6’b), 4.60 (d, J = 8.0Hz, 1H, H-1’), 5.04 (dd, J = 3.5, 10.5Hz, 1H, H-3’), 5.31 (dd, J = 8.0, 10.5Hz, 1H, H-2’), 5.39 (dd, J = 1.3, 3.5Hz, 1H, H-4’), 6.82 (m, 2H, H-10), 7.09 (m, 2H, H-9); Diastereoisomer B: 0.88 (m, 1H, H-3a), 1.06 (m, 1H, H-5a), 1.19 (m, 1H, H-6a), 1.20 (m, 1H, H-4a), 1.52 (m, 1H, H-5b), 1.58 (m, 1H, H-2), 1.64 (m, 1H, H-3b), 1.71 (m, 1H, H-4b), 1.95 (m, 1H, H-6b), 2.03 (s, 3H, OAc), 2.03-2.06 (m, 1H, H-7a), 2.04 (s, 3H, OAc), 2.05 (s, 3H, OAc), 2.06 (s, 3H, OAc), 3.08 (dd, J = 3.3, 13.2Hz, 1H, H-7b), 3.22 (dt, J = 4.3, 9.9, 9.9Hz, 1H, H-1), 3.78 (s, 3H, H-12), 3.89 (dt, J = 1.3, 6.3, 6.3Hz, 1H, H-5’), 4.11 (dd, J = 6.2, 11.1Hz, 1H, H-6’a), 4.19 (dd, J = 6.3, 11.1Hz, 1H, H-6’b), 4.56 (d, J = 7.9Hz, 1H, H-1’), 5.04 (dd, J = 3.5, 10.5Hz, 1H, H-3’), 5.20 (dd, J = 7.9, 10.5Hz, 1H, H-2’), 5.39 (dd, J = 1.3, 3.5Hz, 1H, H-4’), 6.80 (m, 2H, H-10), 7.03 (m, 2H, H-9). IR (cm-1, CCl4): 3063 (w), 3031 (w), 2935 (m), 2859 (m), 2836 (w), 1757 (s), 1613 (w), 1513 (s), 1441 (w), 1369 (s), 1300 (w), 1245 (s), 1221 (s), 1172 (s), 1044 (s), 880 (w), 856 (w). MS (FAB): [M]+ 550 (1), 331 (16), 202 (18), 169 (36), 121 (100), 109 (34), 81 (10). [α]D20 = -72 (c 0.21, CHCl3). For C28H38O11 (550.61): C 61.08, H 6.96; found: C 60.99, H 6.99.
2-(4-Methoxybenzyl)cyclohexyl-β-d-glucopyranoside (7a/7b and 9a/9b) and 2-(4-methoxybenzyl)-cyclohexyl-β-d-galactopyranoside (8a/8b and 10a/10b).
A solution of the respective compounds 3a/3b – 6a/6b (0.206 g; 0.374 mmol) in a mixture of methanol (20 mL) and water (4 mL) was heated to reflux in the presence of potassium carbonate (0.3 g) for 2 h. Methanol and water were removed under reduced pressure, and the residue was applied on the top of a column filled with silica gel and purified by elution with a 15:1 to 5:1 stepwise gradient of chloroform / methanol mixture, affording the pure products in yields of 92.4 % (7a/7b), 88.6 % (8a/8b), 53.8 % (9a/9b) and 66.4 % (10a/10b), respectively.
7a/7b: 1H-NMR (CD3OD): Diastereoisomer A: 1.24 (m, 1H, H-4a), 1.32 (m, 1H, H-3a), 1.41 (m, 2H, H-5a, H-6a), 1.52 (m, 1H, H-3b), 1.65 (m, 1H, H-4b), 1.72 (m, 1H, H-5b), 1.76 (m, 1H, H-2), 2.04 (m, 1H, H-6b), 2.52 (dd, J = 8.4, 13.7Hz, 1H, H-7a), 2.84 (dd, J = 6.6, 13.7Hz, 1H, H-7b), 3.25 (ddd, J = 2.5, 5.6, 9.7Hz, 1H, H-5’), 3.27 (dd, J = 7.7, 9.2Hz, 1H, H-2’), 3.32 (t, J = 9.5Hz, 1H, H-4’), 3.37 (t, J = 9.2Hz, 1H, H-3’), 3.72 (dd, J = 5.6, 11.7Hz, 1H, H-6’a), 3.75 (s, 3H, H-12), 3.89 (dd, J = 2.5, 11.7Hz, 1H, H-6’b), 3.92 (dt, J = 2.5, 2.5, 4.9Hz, 1H, H-1), 4.34 (d, J = 7.7Hz, 1H, H-1’), 6.79 (m, 2H, H-10), 7.15 (m, 2H, H-9); Diastereoisomer B: 1.24 (m, 1H, H-4a), 1.32 (m, 2H, H-3a, H-6a), 1.36 (m, 1H, H-5a), 1.52 (m, 1H, H-3b), 1.65 (m, 1H, H-4b), 1.70 (m, 1H, H-2), 1.77 (m, 1H, H-5b), 1.96 (m, 1H, H-6b), 2.42 (dd, J = 7.9, 13.6Hz, 1H, H-7a), 2.81 (dd, J = 6.5, 13.6Hz, 1H, H-7b), 3.20 (ddd, J = 2.4, 5.7, 9.6Hz, 1H, H-5’), 3.25 (dd, J = 7.8, 9.1Hz, 1H, H-2’), 3.27 (t, J = 9.7Hz, 1H, H-4’), 3.35 (dd, J = 9.2, 9.8Hz, 1H, H-3’), 3.65 (dd, J = 5.7, 11.8Hz, 1H, H-6’a), 3.73 (dt, J = 2.4, 2.4, 4.8Hz, 1H, H-1), 3.75 (s, 3H, H-12), 3.82 (dd, J = 2.4, 11.8Hz, 1H, H-6’b), 4.33 (d, J = 7.8Hz, 1H, H-1’), 6.80 (m, 2H, H-10), 7.15 (m, 2H, H-9). IR (cm-1, KBr): 3401 (s), 2994 (w), 2932 (s), 1612 (m), 1513 (s), 1177 (s), 1096 (s), 1074 (s), 1036 (s), 1018 (s), 895 (w), 842 (w). MS (FAB): [M+H]+ 383 (1), 325 (1), 273 (1), 261 (1), 241 (1), 203 (16), 121 (100), 91 (5), 79 (7). [α]D20 = -63 (c 0.19, CH3OH). For C20H30O7 (382.46): C 62.81, H 7.91; found: C 62.75, H 7.92.
8a/8b: 1H-NMR (CD3OD): Diastereoisomer A: 1.25 (m, 1H, H-4a), 1.33 (m, 1H, 3a), 1.37 (m, 1H, H-5a), 1.41 (m, 1H, H-6a), 1.52 (m, 1H, H-3b), 1.66 (m, 1H, H-4b), 1.72 (m, 1H, H-5b), 1.76 (m, 1H, H-2), 2.02 (m, 1H, H-6b), 2.52 (dd, J = 8.4, 13.6Hz, 1H, H-7a), 2.85 (dd, J = 6.6, 13.6Hz, 1H, H-7b), 3.48 (dd, J = 3.5, 9.7Hz, 1H, H-3’), 3.48 (dt, J = 1.2, 6.2, 6.2Hz, 1H, H-5’), 3.60 (dd, J = 7.7, 9.7Hz, 1H, H-2’), 3.75 (s, 3H, H-12), 3.76 (dd, J = 6.1, 11.0Hz, 1H, H-6’a), 3.80 (dd, J = 6.3, 11.0Hz, 1H, H-6’b), 3.88 (dt, J = 1.2, 3.5Hz, 1H, H-4’), 3.91 (dt, J = 2.4, 2.4, 4.9Hz, 1H, H-1), 4.30 (d, J = 7.7Hz, 1H, H-1’), 6.79 (m, 2H, H-10), 7.15 (m, 2H, H-9); Diastereoisomer B: 1.25 (m, 1H, H-4a), 1.31 (m, 1H, H-6a), 1.33 (m, 1H, H-3a), 1.35 (m, 1H, H-5a), 1.52 (m, 1H, H-3b), 1.66 (m, 1H, H-4b), 1.69 (m, 1H, H-2), 1.78 (m, 1H, H-5b), 1.96 (m, 1H, H-6b), 2.42 (dd, J = 7.8, 13.7Hz, 1H, H-7a), 2.81 (dd, J = 6.6, 13.7Hz, 1H, H-7b), 3.44 (dt, J = 1.2, 6.2, 6.2Hz, 1H, H-5’), 3.48 (dd, J = 3.4, 9.7Hz, 1H, H-3’), 3.58 (dd, J = 7.8, 9.7Hz, 1H, H-2’), 3.69 (dd, J = 6.4, 11.1Hz, 1H, H-6’a), 3.72 (dd, J = 6.1, 11.1Hz, 1H, H-6’b), 3.74 (dt, J = 2.4, 2.4, 4.8Hz, 1H, H-1), 3.75 (s, 3H, H-12), 3.84 (dd, J = 1.1, 3.4Hz, 1H, H-4’), 4.28 (d, J = 7.8Hz, 1H, H-1’), 6.80 (m, 2H, H-10), 7.14 (m, 2H, H-9). IR (cm-1, KBr): 3413 (s), 2923 (s), 2908 (m), 1611 (w), 1512 (s) 1105 (m), 1090 (s), 1082 (s), 1049 (s), 1038 (s), 848 (w). MS (FAB): [M+H]+ 383 (1), 231 (2), 203 (9), 121 (100), 91 (8), 79 (17). [α]D20 = -82 (c 0.20, CH3OH). For C20H30O7 (382.46): C 62.81, H 7.91; found: C 62.87, H 7.85.
9a/9b: 1H-NMR (CD3OD): Diastereoisomer A: 0.90 (m, 1H, H-3a), 1.08 (m, 1H, H-5a), 1.22 (m, 1H, H-6a), 1.24 (m, 1H, H-4a), 1.55 (m, 1H, H-5b), 1.58 (m, 1H, H-2), 1.60 (m, 1H, H-3b), 1.73 (m, 1H, H-4b), 2.13 (m, 1H, H-6b), 2.22 (dd, J = 9.4, 13.7Hz, 1H, H-7a), 3.23 – 3.41 (m, 3H, H-3’, 4’ and 5’), 3.24 (dd, J = 7.8, 9.2Hz, 1H, H-2’), 3.39 (dd, J = 2.9, 13.7Hz, 1H, H-7b), 3.49 (dt, J = 4.1, 9.6, 9.6Hz, 1H, H-1), 3.70 (dd, J = 5.6, 11.7Hz, 1H, H-6’a), 3.75 (s, 3H, H-12), 3.90 (dd, J = 2.3, 11.7Hz, 1H, H-6’b), 4.41 (d, J = 7.8Hz, 1H, H-1’), 6.80 (m, 2H, H-10), 7.09 (m, 2H, H-9); Diastereoisomer B: 0.90 (m, 1H, H-3a), 1.08 (m, 1H, H-5a), 1.22 (m, 1H, H-6a), 1.24 (m, 1H, H-4a), 1.55 (m, 1H, H-5b), 1.58 (m, 1H, H-2), 1.60 (m, 1H, H-3b), 1.73 (m, 1H, H-4b), 2.13 (m, 1H, H-6b), 2.15 (dd, J = 10.0, 13.3Hz, 1H, H-7a), 3.22 (dd, J = 7.8, 9.2, 1H, H-2’), 3.23 – 3.41 (m, 4H, H-7b, H-3’, H-4’ and H-5’), 3.68 (dd, J = 5.2, 11.6Hz, 1H, H-6’a), 3.75 (s, 3H, H-12), 3.85 (dd, J = 2.2, 11.7Hz, 1H, H-6’b), 4.40 (d, J = 7.8Hz, 1H, H-1’), 6.80 (m, 2H, H-10), 7.08 (m, 2H, H-9). IR (cm-1, KBr): 3429 (s), 2995 (w), 2927 (m), 1612 (m), 1513 (m), 1099 (m), 1074 (m), 1036 (s), 1024 (s), 879 (w). MS (FAB): [M+H]+ 383 (1), 261 (1), 241 (2), 203 (8), 121 (100), 91 (5), 79 (16). [α]D20 = -67 (c 0.26, CH3OH). For C20H30O7 (382.46): C 62.81, H 7.91; found: C 62.85, H 7.88.
10a/10b: 1H-NMR (CD3OD): Diastereoisomer A: 0.89 (m, 1H, H-3a), 1.08 (m, 1H, H-5a), 1.23 (m, 1H, H-4a), 1.37 (m, 1H, H-6a), 1.56 (m, 1H, 5b), 1.61 (m, 2H, H-2, H-3b), 1.72 (m, 1H, 4b), 2.14 (dd, J = 9.9, 13.4Hz, 1H, H-7a), 2.22 (m, 1H, H-6b), 3.40 (dd, J = 3.3, 13.4Hz, 1H), 3.48 (m, 1H), 3.48 (dd, J = 3.3, 9.7Hz, 1H), 3.50 (dt, J = 1.1, 6.2, 6.2Hz, 1H), 3.55 (dd, J = 7.5, 9.7Hz, 1H), 3.75 (s, 3H), 3.75 (dd, J = 6.2, 11.2Hz, 1H), 3.78 (dd, J = 6.3, 11.2Hz, 1H), 3.86 (dd, J = 1.1, 3.3Hz, 1H), 4.36 (d, J = 7.5Hz, 1H), 6.79 (m, 2H), 7.09 (m, 2H); Diastereoisomer B: 0.89 (m, 1H, H-3a), 1.08 (m, 1H, H-5a), 1.23 (m, 1H, H-4a), 1.28 (m, 1H, H-6a), 1.56 (m, 1H, 5b), 1.57 (m, 1H, H-2), 1.61 (m, 1H, H-3b), 1.72 (m, 1H, 4b), 2.14 (m, 1H, H-6b), 2.21 (dd, J = 9.5, 13.7Hz, 1H, H-7a), 3.24 (dd, J = 3.6, 13.7Hz, 1H, H-7b), 3.32 (m, 1H, H-1), 3.47 (dd, J = 3.4, 9.7Hz, 1H, H-3’), 3.50 (dt, J = 1.0, 6.2, 6.2Hz, 1H, H-5’), 3.57 (dd, J = 7.7, 9.7Hz, 1H, H-2’), 3.72 (dd, J = 6.1, 11.2Hz, 1H, H-6’a), 3.75 (s, 3H, H-12), 3.75 (dd, J = 6.3, 11.2Hz, 1H, H-6’b), 3.85 (dd, J = 1.0, 3.4Hz, 1H, H-4’), 4.36 (d, J = 7.7Hz, 1H, H-1’), 6.80 (m, 2H, H-10), 7.08 (m, 2H, H-9). IR (cm-1, KBr): 3520 (s), 3420 (s), 2990 (w), 1613 (m), 1514 (s) 1100 (m), 1084 (s), 1078 (m), 1052 (s), 1035 (s), 880 (w). MS (FAB): [M+H]+ 383 (1), 231 (1), 203 (10), 121 (100), 91 (8), 79 (11). [α]D20 = -86 (c 0.25, CH3OH). For C20H30O7 (382.46): C 62.81, H 7.91; found: C 62.78, H 7.87.

Table 1.
13C-NMR data of the protected alkyl glycosides 3a – 6b (measured in CDCl3)
| Carbon atom No. | Compound | |||||||
|---|---|---|---|---|---|---|---|---|
| 3a a | 3b a | 4a b | 4b b | 5a c | 5b c | 6a d | 6b d | |
| 1 | 74.65 (d) | 79.93 (d) | 75.18 (d) | 80.07 (d) | 80.94 (d) | 85.60 (d) | 81.25 (d) | 85.77 (d) |
| 2 | 43.89 (d) | 43.80 (d) | 43.81 (d) | 43.72 (d) | 45.14 (d) | 44.32 (d) | 45.12 (d) | 44.28 (d) |
| 3 | 26.82 (t) | 26.68 (t) | 26.73 (t) | 26.62 (t) | 29.76 (t) | 29.68 (t) | 29.76 (t) | 29.60 (t) |
| 4 | 25.37 (t) | 25.06 (t) | 25.30 (t) | 24.96 (t) | 24.76 (t) | 24.43 (t) | 24.76 (t) | 24.41 (t) |
| 5 | 20.76 (t) | 20.70 (t) | 20.98 (t) | 20.84 (t) | 24.91 (t) | 24.88 (t) | 24.93 (t) | 24.81 (t) |
| 6 | 31.93 (t) | 28.78 (t) | 31.93 (t) | 28.88 (t) | 33.72 (t) | 31.23 (t) | 33.79 (t) | 31.26 (t) |
| 7 | 37.23 (t) | 37.17 (t) | 37.22 (t) | 37.03 (t) | 37.30 (t) | 37.26 (t) | 37.34 (t) | 37.31 (t) |
| 8 | 133.43 (s) | 133.01 (s) | 133.51 (s) | 133.04 (s) | 132.91 (s) | 132.44 (s) | 132.99 (s) | 132.47 (s) |
| 9 | 130.30 (d) | 129.84 (d) | 130.27 (d) | 129.84 (d) | 130.40 (d) | 130.08 (d) | 130.35 (d) | 130.10 (d) |
| 10 | 113.78 (d) | 113.34 (d) | 113.77 (d) | 113.40 (d) | 113.64 (d) | 113.41 (d) | 113.64 (d) | 113.44 (d) |
| 11 | 157.83 (s) | 157.67 (s) | 157.83 (s) | 157.65 (s) | 157.81 (s) | 157.66 (s) | 157.80 (s) | 157.66 (s) |
| 12 | 55.24 (q) | 55.19 (q) | 55.24 (q) | 55.21 (q) | 55.21 (q) | 55.21 (q) | 55.21 (q) | 55.21 (q) |
| 1’ | 101.76 (d) | 98.13 (d) | 102.32 (d) | 98.85 (d) | 102.04 (d) | 98.49 (d) | 102.56 (d) | 99.24 (d) |
| 2’ | 71.76 (d) | 7159 (d) | 69.34 (d) | 69.19 (d) | 71.67 (d) | 7158 (d) | 69.32 (d) | 69.25 (d) |
| 3’ | 73.11 (d) | 73.08 (d) | 71.17 (d) | 71.16 (d) | 73.09 (d) | 73.05 (d) | 71.18 (d) | 71.18 (d) |
| 4’ | 68.94 (d) | 68.67 (d) | 67.21 (d) | 67.19 (d) | 68.77 (d) | 68.75 (d) | 67.13 (d) | 67.13 (d) |
| 5’ | 71.59 (d) | 71.52 (d) | 70.48 (d) | 70.45 (d) | 71.79 (d) | 71.60 (d) | 70.52 (d) | 70.46 (d) |
| 6’ | 62.28 (t) | 62.10 (t) | 61.37 (t) | 61.36 (t) | 62.27 (t) | 62.10 (t) | 61.36 (t) | 61.36 (t) |
Signals of the protecting acetate functionalities on the carbohydrate part of the molecules: a 20.60 (q), 20.60 (q), 20.64 (q), 20.64 (q), 20.70 (q), 20.70 (q), 20.76 (q), 20.84 (q), 169.20 (s), 169.23 (s), 169.43 (s), 169.45 (s), 170.37 (s), 170.40 (s), 170.59 (s), 170.76 (s); b 20.61 (q), 20.62 (q), 20.62 (q), 20.66 (q), 20.71 (q), 20.71 (q), 20.73 (q), 20.90 (q), 169.95 (s), 170.21 (s), 170.26 (s), 170.39 (s), 170.39 (s), 170.40 (s), 170.43 (s), 170.48 (s); c 20.60 (q), 20.60 (q), 20.60 (q), 20.63 (q), 20.69 (q), 20.69 (q), 20.69 (q), 20.71 (q), 169.27 (s), 169.32 (s), 169.41 (s), 169.42 (s), 170.35 (s), 170.37 (s), 170.62 (s), 170.68 (s); d 20.60 (q), 20.62 (q), 20.64 (q), 20.69 (q), 20.70 (q), 20.70 (q), 20.80 (q), 20.83 (q), 169.40 (s), 169.86 (s), 169.95 (s), 170.22 (s), 170.35 (s), 170.38 (s), 170.40 (s), 170.48 (s).

Table 2.
13C-NMR data of the alkyl glycosides 7a – 10b (measured in CD3OD)
| Carbon atom No. | Compound | |||||||
|---|---|---|---|---|---|---|---|---|
| 7a | 7b | 8a | 8b | 9a | 9b | 10a | 10b | |
| 1 | 76.58 (d) | 80.54 (d) | 76.48 (d) | 80.55 (d) | 81.22 (d) | 85.90 (d) | 81.26 (d) | 85.84 (d) |
| 2 | 45.61 (d) | 45.24 (d) | 45.71 (d) | 45.33 (d) | 47.44 (d) | 46.72 (d) | 47.46 (d) | 46.73 (d) |
| 3 | 28.30 (t) | 28.23 (t) | 28.30 (t) | 28.27 (t) | 31.57 (t) | 31.38 (t) | 31.58 (t) | 31.39 (t) |
| 4 | 26.59 (t) | 26.09 (t) | 26.55 (t) | 26.14 (t) | 26.43 (t) | 26.21 (t) | 26.43 (t) | 26.22 (t) |
| 5 | 23.30 (t) | 22.34 (t) | 23.28 (t) | 22.37 (t) | 26.85 (t) | 26.70 (t) | 26.86 (t) | 26.71 (t) |
| 6 | 33.25 (t) | 30.13 (t) | 33.29 (t) | 30.18 (t) | 35.71 (t) | 32.73 (t) | 35.75 (t) | 32.78 (t) |
| 7 | 38.36 (t) | 37.41 (t) | 38.25 (t) | 37.46 (t) | 39.03 (t) | 38.99 (t) | 39.01 (t) | 39.01 (t) |
| 8 | 135.61 (s) | 135.44 (s) | 135.65 (s) | 135.49 (s) | 135.14 (s) | 134.95 (s) | 135.13 (s) | 134.99 (s) |
| 9 | 131.79 (d) | 131.69 (d) | 131.81 (d) | 131.70 (d) | 131.84 (d) | 131.76 (d) | 131.86 (d) | 131.76 (d) |
| 10 | 115.04 (d) | 114.95 (d) | 115.03 (d) | 114.93 (d) | 114.99 (d) | 114.95 (d) | 114.98 (d) | 114.93 (d) |
| 11 | 159.70 (s) | 159.64 (s) | 159.68 (s) | 159.62 (s) | 159.70 (s) | 159.66 (s) | 159.69 (s) | 159.65 (s) |
| 12 | 56.12 (q) | 56.12 (q) | 56.12 (q) | 56.12 (q) | 56.12 (q) | 56.12 (q) | 56.12 (q) | 56.12 (q) |
| 1’ | 105.66 (d) | 102.23 (d) | 106.32 (d) | 102.98 (d) | 106.20 (d) | 101.81 (d) | 106.83 (d) | 102.54 (d) |
| 2’ | 76.08 (d) | 75.77 (d) | 73.54 (d) | 73.24 (d) | 76.13 (d) | 75.68 (d) | 73.57 (d) | 73.13 (d) |
| 3’ | 78.83 (d) | 78.83 (d) | 75.74 (d) | 75.70 (d) | 78.76 (d) | 78.74 (d) | 75.66 (d) | 75.64 (d) |
| 4’ | 72.39 (d) | 72.29 (d) | 70.75 (d) | 70.75 (d) | 72.42 (d) | 72.21 (d) | 70.81 (d) | 70.75 (d) |
| 5’ | 78.29 (d) | 78.22 (d) | 76.90 (d) | 76.86 (d) | 78.41 (d) | 78.28 (d) | 77.01 (d) | 76.92 (d) |
| 6’ | 63.47 (t) | 63.35 (t) | 62.83 (t) | 62.83 (t) | 63.52 (t) | 63.31 (t) | 62.92 (t) | 62.87 (t) |
Acknowledgments
Financial support through the grant 203/02/0166 (Grant Agency of the Czech Republic) is gratefully acknowledged. The authors thank Mrs. M. Wimmerová for her skillful technical assistance.
References
- Igarashi, K. Adv. Carbohydr. Chem. Biochem. 1977, 34, 243–283.
- Schmidt, R. R. Angew. Chem. Int. Ed. Engl. 1986, 25, 212–235.
- Schmidt, R. R.; Jung, K.-H. ‘Preparative Carbohydrate Chemistry’; Hanessian, S., Ed.; Dekker: New York, 1997; pp. 283–312. [Google Scholar]
- Nicolaou, K. C.; Ueno, H. ‘Preparative Carbohydrate Chemistry’; Hanessian, S., Ed.; Dekker: New York, 1997; pp. 313–338. [Google Scholar]
- Hanessian, S. ‘Preparative Carbohydrate Chemistry’; Hanessian, S., Ed.; Dekker: New York, 1997; pp. 381–388. [Google Scholar]
- Wimmer, Z.; Romaňuk, M. Coll. Czech. Chem. Commun. 1982, 47, 1878–1883.
- Wimmer, Z.; Rejzek, M.; Zarevúcka, M.; Kuldová, J.; Hrdý, I.; Němec, V.; Romaňuk, M. J. Chem. Ecol. 1997, 23, 605–628.
- Plattner, J. J.; Gless, R. D.; Rapoport, H. J. Am. Chem. Soc. 1972, 94, 8613–8615.
- Buděšínský, M.; Šaman, D.; Wimmer, Z. Coll. Czech. Chem. Commun. 1982, 47, 2763–2767.
- von Kienlin, M.; Moonen, C. T. W.; van der Toorn, A.; van Zijl, C. M. J. Magn. Reson. 1991, 93, 423–429.
- Willker, W.; Leibfritz, D.; Kerssebaum, R.; Bermel, W. Magn. Reson. Chem. 1993, 31, 287–292.
- Sample availability: Available from the corresponding author (ZW).
© 2004 by MDPI (http://www.mdpi.org). Reproduction is permitted for noncommercial purposes.
