Abstract
We present 1-pentafluorophenyl-1H-pyrrole and some of its electrophilic substitution reactions: formylation and acetylations. The 2-substituted products formed in these reactions are selectively and in high yield converted into 3-substituted products by treatment with trifluoromethanesulfonic acid. A general synthesis of 3-acylpyrroles was developed by the trifluoromethanesulfonic acid–mediated rearrangement of the corresponding 2-acylpyrroles.
Introduction
Pyrrole itself and substituted pyrroles are important starting materials for the synthesis of naturally-occuring porphynoid compounds and other porphyrins, potential chemotherapeutic agents and conducting polymers [1]. Polypyrrole films are currently being explored in aplications such as biosensors, adressable gene-ships and interactive conduits for neuronal tissue engineering [2]. Control of molecular architecture of electronic functionalities to the backbone or to the side chains of the conjugated polymers have got very various properties. The pentafluorophenyl group is known for face-to-face stacking interactions between phenyl and pentafluorophenyl group as common non-covalent interactions. With this supramolecular synthon it should be possible to design molecules that have high probability of packing in the desired crystalline arrangement [3].
Results and Discussion
In 1993 Chen and Li described pentafluorophenylation of pyrrole by a photo-induced electron-transfer cation-diradical coupling process. By this route they prepared 2- and 3- pentafluorophenyl substituted pyrroles [4]. Henrie II and Yeager [5] and Biemans et al. [6] were interested in the reaction of hexafluorobenzene with the sodium salt of pyrrole. They prepared several substituted products but failed to prepare the monosubstituted product 1-pentafluorophenyl-1H-pyrrole (1). The synthesis of this compound starts of pentafluoroaniline and provides compound 1 by cyclization with 2,5-dimethoxytetrahydrofuran by a slightly modified Clauson-Kass procedure [7a] (yield 78%) or by P2O5 initiated cyclization [7b] (yield 64%).
These reactions are suitable for a multi-gram preparation of 1-pentafluorophenyl-1H-pyrrole. The product is a stable colorless crystalline compound; it can easily be purified by steam distillation or by vacuum sublimation. Electrophilic substitution reactions of compound 1 such as Vilsmeyer-Haack formylation and acylations with aliphatic acid anhydrides lead in very good yields predominantly to 2-substituted products (2a-c) [8] with no more than 5% of the corresponding 3-substituted products (3a-c) (Scheme 1).
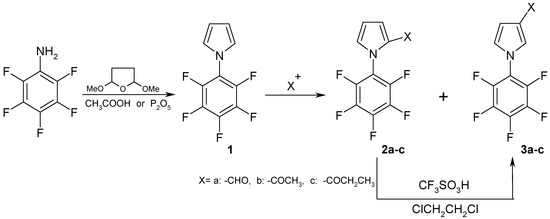
Scheme 1.
Preparation and some reactions of compound 1
Scheme 1.
Preparation and some reactions of compound 1
3-Substituted products were prepared in near quantitative yields by isomerisation of 2-substituted products using trifluoromethanesulfonic acid [9]. Only one example of isomerisation of carboxaldehyde group [9] has been reported in the literature. We used this method for isomerisation of a keto group in near quantitative yield. The acid-promoted conversion of 2-substituted into 3-substituted pyrroles has preparative significance. Such compounds, heretofore prepared by multi-step sequences can now often be obtained in one step from the readily available 2-substituted pyrroles. All 19F-NMR spectra displayed two triplets and one doublet in a 2:1:2 ratio which characterize the pentafluorophenyl group.
Conclusions
In this paper we have described a straightforward and efficient preparation of new 1-pentafluorophenyl-1H-pyrrole derivatives, all of which are important precursors for preparation of new oligo- and polypyrrole derivatives with optoelectronical properties. Our objective has been to produce well-defined polypyrroles containing electroactive functionalities, such as those giving rise to self-compensated materials and well-organized self-assembled structures.
Experimental
General
1H-, 13C- and 19F NMR spectra were obtained using a Varian VXR 300 NMR and were recorded at 300, 75.4 and 282.2 MHz respectively. Chemical shifts are reported in parts per million downfield using the CDCl3 peak (7.27 ppm, 1H; 77.4 ppm, 13C) or trifluoroacetic acid (-78.5 ppm, 19F) as internal references. Coupling constants are recorded in Hz. MS measurements were made with an MS 902-S mass spectrophotometer. Melting points were measured on a Kofler block and are uncorrected. Analytical TLC was performed on precoated 0.25 mm silica gel 60F-254 plates (E. Merck), developed with toluene and visualized with UV irradiation.
Synthesis of 1-Pentafluorophenyl-1H-Pyrrole (1).
A solution of the pentafluoroaniline (3.0 g, 16.4 mmol) and 2,5-dimethoxytetrahydrofuran (2.6 g, 19.7 mmol) in acetic acid (30 mL) is first refluxed for 3 hours, then the solvent is removed under reduced pressure, and the oily residue is taken up with diethyl ether. The organics were washed sequentially with water and saturated sodium bicarbonate solution. Purification by steam distillation or vacuum sublimation gives pure pyrrole 1 (2.98 g, 78% yield), m.p. 58-60oC; Rf 0.67; 1H-NMR δ: 6.78 (t, 2H, J=2.0), 6.41 (d, 2H, J=2.1); 13C-NMR δ: 110.9, 116.7, 122.4, 136.5, 139.9, 140.7; 19F-NMR δ: -145.3 (d, 2F, J=15.6), -152.7 (t, 1F, J=23.1), -157.5 (t, 2F, J=20.3); HRMS (CI) for C10H4F5N (MH+, 100): 233.
Synthesis of 1-Pentafluorophenyl-1H-Pyrrole-2-Carbaldehyde (2a).
Phosphorus oxychloride (0.35 g, 2.26 mmol) was added dropwise to cooled N,N-dimethyl-formamide (0.17 g, 2.26 mmol). The mixture was brought to room temperature and a solution of 1-pentafluorophenyl-1H-pyrrole (1) (0.44 g, 1.89 mmol) in 1,2-dichloroethane (10 mL) is added in small portions over 15 min. After 7 hours warming at 80oC the reaction mixture was cooled and poured into ice. The solution was neutralized with saturated aqueous sodium carbonate and extracted with diethyl ether (3x20 mL). The combined organic layers were dried over calcium chloride. Removal of the solvent in vacuo gave the crude product, which was then purified by chromatography on a silica gel column (eluent = toluene) to yield 2a as a colorless crystalline solid (0.38 g, 76% yield); m.p. 77-79oC; Rf 0.49; 1H-NMR δ: 9.59 (s, 1H), 7.19 (d, 1H, J=3.9), 6.99 (s, 1H), 6.55 (t, 1H, J= 3.5); 13C-NMR δ: 112.4, 125.0, 131.6, 136.0, 139.5, 141.9, 142.1, 145.4, 178.7; 19F-NMR δ: -143.44 (d, 2F, J=17.7), -149.82 (t, 1F, J=22.8), -158.22 (dd, 2F, J=17.7); HRMS (CI) for C11H4F5NO (MH+, 100): 261
Synthesis of Compounds 2b,c.
A mixture of 1-pentafluorophenyl-1H-pyrrole (1) (1.0 g, 4.29 mmol), Mg(ClO4)2·2H2O (0.1 g) and the appropriate anhydride (10 mL) was heated for 4 hours at 80oC. After cooling it was poured onto ice. The solution was neutralized with saturated aqueous sodium carbonate solution and extracted with diethyl ether (3x20 mL). The combined organic layers were dried over calcium chloride. Removal of the solvent in vacuo afforded the crude products, which were then purified by chromatography on a silica gel column (eluent = toluene) to yield 2b or 2c as colorless crystalline solids.
Analytical and Spectral Data: 1-(1-Pentafluorophenyl-1H-pyrrole-2-yl)-ethanone (2b): 0.83 g (70% yield); m.p: 45-48oC; Rf 0.45; 1H-NMR δ: 7.20 (d, 1H, J=2.3), 6.87 (s, 1H), 6.45 (t, 1H, J=3.5), 2.44 (s, 3H); 13C-NMR δ: 26.11, 111.3, 120.6, 130.6, 132.1, 135.8, 139.6, 142.2, 145.4, 187.7; 19F-NMR δ: -143.63 (d, 2F, J=17.4), -150.60 (t, 1F, J=22.8), -158.62 (dd, 2F, J=17.4); HRMS (CI) for C12H6F5NO (MH+, 100): 275.
1-(1-Pentafluorophenyl-1H-pyrrole-2-yl)-propan-1-one (2c): 0.89 g (72% yield); m.p. 75-77oC; Rf 0.55; 1H-NMR δ: 7.17 (d, 1H, J=2.4), 6.87 (s, 1H), 6.45 (t, 1H, J=3.5), 2.87 (q, 1H, J= 7.5), 1.13 (t, 3H, J=7.5); 13C-NMR δ: 8.3, 31.8, 111.2, 119.5, 130.3, 131.7, 135.9, 139.3, 142.1, 145.6, 191.2; 19F-NMR δ: -69.76 (d, 2F, J=17.4), -76.81 (t, 1F, J=22.8), -84.70 (dd, 2F, J=17.7); HRMS (CI) for C13H8F5NO (MH+, 100): 289.
Synthesis of Compounds 3a-c.
Triflic acid (1.8 ml, 20 mmol) was added to a solution of 2a-c (5 mmol) in dichloroethane (10 mL). The reaction mixture was refluxed for 3 hours and then evaporated to dryness under reduced pressure. The residue was dissolved in diethyl ether and the solution was washed twice with a saturated aqueous solution of sodium carbonate. The organic layer was separated, dried over magnesium sulphate and the solvent was removed under reduced pressure. Vacuum sublimation gives the compounds 3a, 3b or 3c, respectively, in nearly quantitative yields.
Analytical and Spectral Data: 1-Pentafluorophenyl-1H-pyrrole-3-carbaldehyde (3a): m.p. 53-56oC; Rf 0.38; 1H-NMR δ: 9.89 (s, 1H), 7.49 (t, 1H, J=1.8), 6.86-6.91 (m, 2H); 13C-NMR δ: 109.6, 125.0, 128.6, 130.3, 136.6, 139.7, 140.6, 142.6, 144.0, 185.2; 19F-NMR δ: -65.91 (d, 2F, J=18.0), -70.97 (t, 1F, J=23.1), -77.54 (t, 2F, J=18.0); HRMS (CI) for C11H4F5NO (MH+, 100): 261.
1-(1-Pentafluorophenyl-1H-pyrrole-3-yl)-ethanone (3b): m.p. 114-115oC; Rf 0.33; 1H-NMR δ: 7.45 (t, 1H, J=1.8), 6.82-6.85 (m, 2H), 2.46 (s, 3H); 13C-NMR δ: 27.4, 110.9, 124.1, 127.0, 128.2, 136.7, 139.7, 140.8, 142.1, 144.0, 193.0; 19F-NMR δ: -65.97 (d, 2F, J=18.0), -71.43 (t, 1F, J=23.1), -77.81 (t, 2F, J=18.0); HRMS (CI) for C12H6F5NO (MH+, 100): 275.
1-(1-Pentafluorophenyl-1H-pyrrole-3-yl)-propan-1-one (3c): m.p. 102-104oC; Rf 0.52; 1H-NMR δ: 7.46 (t, 1H, J=1.8), 6.83-6.85 (m, 2H), 2.82 (q, 2H, J=7.2), 1.21 (t, 3H, J= 7.5); 13C-NMR δ: 196.2, 144.0, 143.8, 142.4, 140.7, 127.6, 126.4, 123.9, 110.8, 32.9, 8.4; 19F-NMR δ: -65.99 (d, 2F, J=18.6), -71.58 (t, 1F, J=23.1), -77.85 (t, 2F, J=18.0); HRMS (CI) for C13H8F5NO (MH+, 100): 289.
References
- Müllen, K.; Wegner, G. Electronic Materials: The Oligomer Approach; Wiley-VCH: New York, 1998. [Google Scholar]
- Shastri, V. R.; Pishko, M. V. Electrical and Optical Polymer Systems; Wnek, G. E., Trantolo, D. J., Cooper, T. M., Gresser, J. D., Eds.; Marcel Dekker: New York, 1998; p. 1031. [Google Scholar] Buchanan, J. G.; Sable, H.Z. Selective Organic Transformations; Thyagarajan, B. S., Ed.; Wiley-Interscience: New York, 1972; Vol. 2, pp. 1–95. [Google Scholar]
- Coates, G. W.; Dunn, A. R.; Henling, I. M.; Ziller, J. W.; Lobkovsky, E. B.; Grubbs, R. H. Phenyl-Perfluorophenyl Stacking Intercations: Topochemical [2+2] Photodimerization and Photopolymerization of Olefinic Compounds. J. Am. Chem. Soc. 1998, 120, 3641–3649. [Google Scholar] [CrossRef]
- Chen, Q.-Y.; Li, Z.-T. Pentafluorophenylation of Aromatics with Pentafluorophenyl Perfluoro- and Polyfluoroalkanesulfonates. A Photoinduced Electron Transfer Cation Diradical Coupling Process. J. Org. Chem. 1993, 58, 2599–2604. [Google Scholar] [CrossRef]
- Henrie, R. N., II; Yeager, W. H. Reaction of Diazole Anions with Hexafluorobenzene: an Unexpectedly Facile Entry Into Hexa(Diazol-1-yl)-Benzenes. Heterocycles 1993, 35, 415–425. [Google Scholar]
- Biemans, H. A. M.; Ahang, C.; Smith, P.; Koojiman, H.; Smeets, W. J. J.; Spek, A. L.; Meijer, E. W. Hexapyrrolylbenzene and Octapyrrolylnaphtalene. J. Org. Chem. 1996, 61, 9012–9015. [Google Scholar] [CrossRef]
- Clauson-Kaas, N.; Tyle, Z. Preparation of Cis and Trans 2,5-Dimethoxy-2-(acetamidomethyl)-2,5-dihydrofuran, of Cis and Trans 2,5-Dimethoxy-2-(acetamidomethyl)-tetrahydrofuran and of 1-Phenyl-2-(acetamidomethyl)pyrrole. Acta Chem. Scand. 1952, 6, 667–670. [Google Scholar] Elming, N.; Clauson-Kaas, N. The preparation Pyrroles from Furans. Acta Chem. Scand. 1952, 6, 867–874. [Google Scholar]
- Fang, Y.; Leysen, D.; Ottenheijm, H. C. J. A Facile Synthesis of N-Substituted Pyrroles. Synth. Commun. 1995, 25, 1857–1861. [Google Scholar] [CrossRef]
- Dellemagne, P.; Rault, S.; Fabis, F.; Dumoulin, H.; Robba, M. A Convenient Rearrangement of 1-Phenylpyrrole-2-Carboxaldehydes into Their 3-Isomers Synth. Commun. 1994, 24, 1855–1857. [Google Scholar]
- Sample Availability: Available from the authors.
© 2003 by MDPI ( http://www.mdpi.org). Reproduction is permitted for noncommercial purposes.
