Abstract
Easy and effective preparations of the nearly pure [bis(trifluoroacetoxy)-iodo]arenes, ArI(OCOCF)3, from some iodoarenes, ArI, are reported, using an anhydrous sodium percarbonate/(CF3CO)2O/CH2Cl2 system. The colorless, freshly prepared ArI(OCOCF3)2 thus obtained were 98-99% pure (by iodometry).
Introduction
[Bis(trifluoroacetoxy)iodo]arenes, ArI(OCOCF3)2, are potent and selective oxidants, widely used in modern organic synthesis. So far, they have been synthesized from either ArI, or ArIO, or ArICl2, or directly from ArH, but the most common method depends on their preparation (in moderate yields) from the corresponding (diacetoxyiodo)arenes, ArI(OAc)2, by their recrystallization from CF3COOH [1,2].
Results and Discussion
Recently, in our laboratory we have devised a simple, easy, and efficient method for the direct preparation of ArI(OCOCF3)2 from the respective iodoarenes, ArI, in a binary anhydrous solvent system, (CF3CO)2O/CH2Cl2, using commercially available sodium percarbonate, 2Na2CO3. 3H2O2, as the oxidant (Scheme 1 and Table 1):
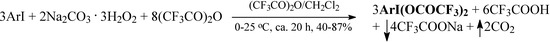
Scheme 1.
Scheme 1.
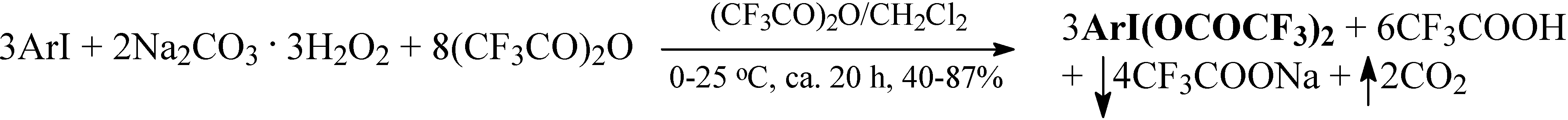
In practice, only the fairly stable parent compound, PhI(OCOCF3)2 has found widespread use in organic synthesis [1,2,3], although it should be noted that p-Cl- and p-F-C6H4I(OCOCF3)2 are even more stable. A limited number of other ring-substituted ArI(OCOCF3)2 have been reported in the literature [4,5,6]. We have unexpectedly found that several other ArI(OCOCF3)2 compounds are less stable, particularly p-MeC6H4I(OCOCF3)2, and they usually deteriorate within several days to finally form oily residues with a strong acidic odor – even when stored in a dark cooler. As far as limitations of the reported method are concerned, we have found that it is not suitable for the preparation of the three O2N-C6H4I(OCOCF3)2 isomers, as well as p-MeOC6H4I(OCOCF3)2. It is also seen from the data in Table 1 that 3-F3CC6H4I(OCOCF3)2 was obtained in only 40% yield by our new method, cf. [6].
Experimental
General
Chemical structures of the compounds in Table 1 were confirmed by satisfactory microanalyses of the crude, freshly prepared products (C ± 0.2, H ± 0.1, I ± 0.2%) obtained at the Institute of Organic Chemistry, the Polish Academy of Sciences, Warsaw.
Optimized Procedure for Preparing [Bis(trifluoroacetoxy)iodo]arenes from Iodoarenes:
A solution of an appropriate iodoarene (5 mmol) in a mixture of (CF3CO)2O (12 mL) and CH2Cl2 (40 mL) was cooled with stirring to 0-2 oC. Next, sodium percarbonate (2.1 g; 13.4 mmol; 300% excess) was added portionwise, and the stirring was continued for 2 h at 0-2 oC, and further at room temperature for ca. 18 h. The precipitated CF3COONa was collected by filtration under reduced pressure, washed with CH2Cl2 (2 x 20 mL), and discarded. The filtrates were evaporated under vacuum. The solid residues were triturated with hexane (10 mL), collected by filtration, washed with hexane on the filter, and quickly air-dried by the suction, leaving the colourless title products, ArI(OCOCF3)2 (40-87% yields). Their yields and melting points are given in Table 1 below. In practice these products need not to be recrystallized. They should be stored in the dark, preferably in a cooler, avoiding any moisture [3]. The freshly prepared products were 98-99% pure (by iodometry [7]).

Table 1.
Yields and melting points (uncorrected) for the prepared [bis(trifluoroacetoxy)iodo]arenes, ArI(OCOCF3)2
| Ar | Yield [%] | Mp [oC] | Lit. Mp [oC] |
|---|---|---|---|
| phenyl | 87 | 122-124 | 120-122 [4] |
| 2-methylphenyl | 81 | 89-92 | 88-90 [4] |
| 3-methylphenyl | 86 | 93-96 | 87-90 [5] |
| 4-methylphenyl | 76 | 112-115 | 114-116 [4] |
| 4-fluorophenyl | 77 | 103-105 | 88-90 [4] |
| 4-chlorophenyl | 74 | 129-132 | 131-133 [5] |
| 3-(trifluoromethyl)phenyl | 40 | 97-100 | 99-100 [6] |
References
- Varvoglis, A. The Organic Chemistry of Polycoordinated Iodine; VCH: Weinheim, 1992. [Google Scholar]
- Varvoglis, A. Hypervalent Iodine in Organic Synthesis; Academic Press: San Diego, 1997. [Google Scholar]
- Aldrich Handbook of Fine Chemicals and Laboratory Equipment 2003-2004; p. 241. a m.p. of 121-125 oC is reported for 97% PhI(OCOCF3)2, with a note: MOISTURE-SENSITIVE.
- Kasumov, T.M.; Brel, V.K.; Grishin, Y.K.; Zefirov, N.S.; Stang, P.J. Oxidative Properties of Xenon(II) Compounds. A New, Convenient Synthesis of [Bis(trifluoroacetoxy)iodo]arenes, [Bis(trifluoroacetoxy)iodo]perfluoroalkanes and μ-Oxo-bridged Aryliodoso-Derivatives. Tetrahedron 1997, 53, 1145–1150. [Google Scholar]
- White, J.D.; Caravatti, G.; Kline, T.B.; Edstrom, E.; Rice, K.C.; Brossi, A. Biomimetic Total Synthesis of (-)-Codeine. Tetrahedron 1983, 39, 2393–2397, (p. 2396). [Google Scholar] [CrossRef]
- Yagupolskii, L.M.; Maletina, I.I.; Kondratenko, N.V.; Orda, V.V. Tetrakis[perfluoroacyloxy]iodoarenes and Bis[acetoxy]iodylarenes; New Pentavalent Iodine Derivatives. Synthesis 1977, 574–575. [Google Scholar]
- Lucas, H.J.; Kennedy, E.R.; Formo, M.W. Org. Synth. 1942, 22, 70.
- Sample Availability: Available from the authors.
© 2002 by MDPI (http://www.mdpi.org). Reproduction is permitted for noncommercial purposes.
