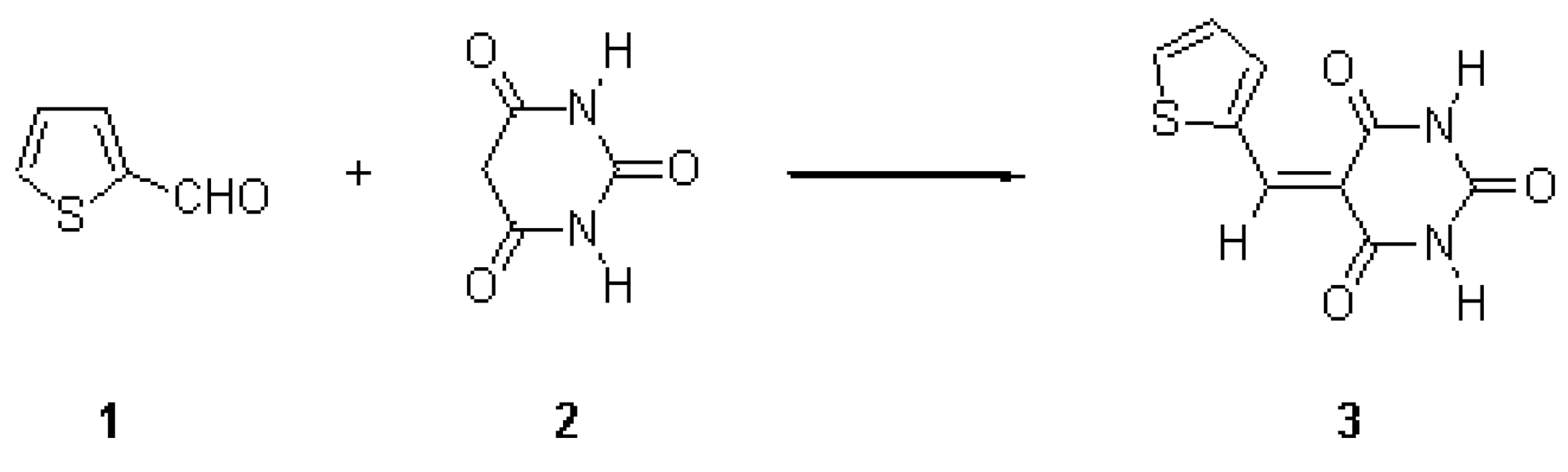
5-(2-Thienylidene)barbituric acid 3 was prepared by Knoevenagel condensation of thiophene- 2-carboxaldehyde 1 and barbituric acid 2 in ethanol using piperidine as a base [1,2]. Barbituric acid 2 ( 2.82 g, 0.02 mol) and thiophene-2-carboxaldehyde 1 (2.50 g, 0.02 mol ) in ethanol ( 50 mL) was heated under reflux for three minutes. Piperidine (0.5 mL) was added in one portion and the reflux was continued for additional two hours. The reaction mixture was cooled to room temperature and the solid formed was filtered, washed with cooled ethanol (2x20 mL) and dried. 5-(2-Thienylidene)barbituric acid 3 was recrystallized from ethanol as yellow powder (4.65g, 95%).
M.p. 220-222 (EtOH, uncorrected).
UV (EtOH) (e dm3.mol-1.cm-1): 260 (2009), 370 (2875), 220 (2475).
IR (KBr): 3204 (N-H), 1694 (C=O), 1670 (N-CO-N), 1632 (C=C).
1H-NMR (400 MHz, CDCl3): 11.3 (1H, s, NH), 8.55 (1H, s, HC=), 8.27 (1H, d, H-5, J54= 5.0 Hz ), 8.17 (1H, d, H-3, J34=3.9 Hz ), 7.34 (1H, dd, H-4, J43= 3.9Hz , J45= 5.0 Hz ).
13C-NMR (100 MHz, CDCl3): 111.6 (CH=C), 128.4 (CH=C), 136.3 (C-2 thiophene), 142.2, 145.7, 145.8 (C-5 thiophene), 150.3 (N-CON), 163.0 (C=O ), 163.6 (C=O).
Anal.Calc. for C9H6N2O3S (222.262): C 48.64, H 2.72 , N 12.60; found : C 48.31, H 2.86, N 12.47.
References
- Jones, G. Org. React. 1967, 15, 204.
- Tanaka, K.; Chen, X.; Yoneda, F. Tetrahedron 1988, 44, 3241. [CrossRef]
Sample Availability: Available from the authors and from MDPI |
© 2000 MDPI. All rights reserved. Molecules website www.mdpi.org/molecules/.
