Abstract
During the last years, many bisbibenzylic macrocyclic ethers were isolated and identified in Hepaticae. One of them is MARCHANTINQUINONE, a quinonic macrocycle with interesting biological activity. In the following report, we present the last steps of the total synthesis.
Introduction
Bisbibenzylic systems such as Marchantins, Perrottetins, Riccardins are found only in Hepatica and have been shown to display a wide range of biological activities [1,2]. Marchantinquinone (1), from extracts of Reboulia hemisphaerica, formerly described as Mannia subpilosa, [3,4] was the first bisbibenzylic diether possessing a quinone structure isolated from Bryophytes. Herein its first synthesis is reported.
Experimental
Relevant steps of the synthesis are shown in the following retrosynthetic scheme:
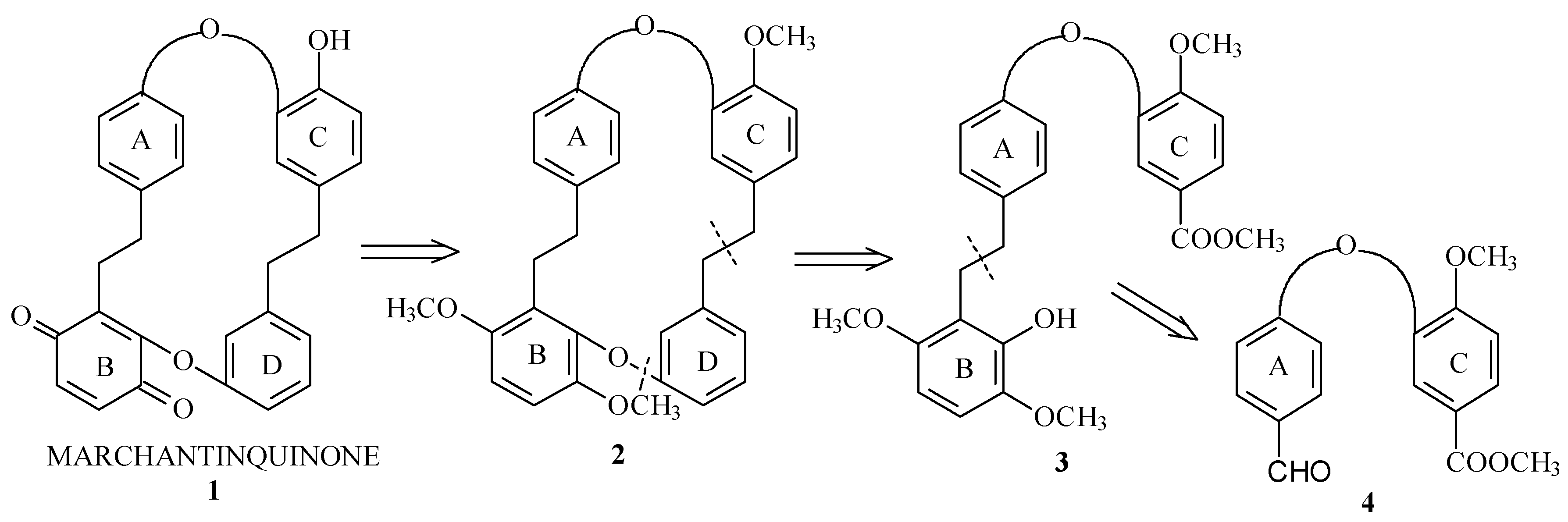
The global strategy of this synthesis is based on standard organic synthesis reactions: nucleophilic aromatic substitution, Wittig reaction, catalytic hydrogenation. It also includes redox reactions and macrocyclization using Ni° complex.
Results and Discussion
Previously, we described the synthesis of macrocycle (2) [5] an advance precursor of Marchantinquinone (1). Different conditions of macrocyclization, deprotection and oxidation to obtain the quinonic structure will be disclosed.
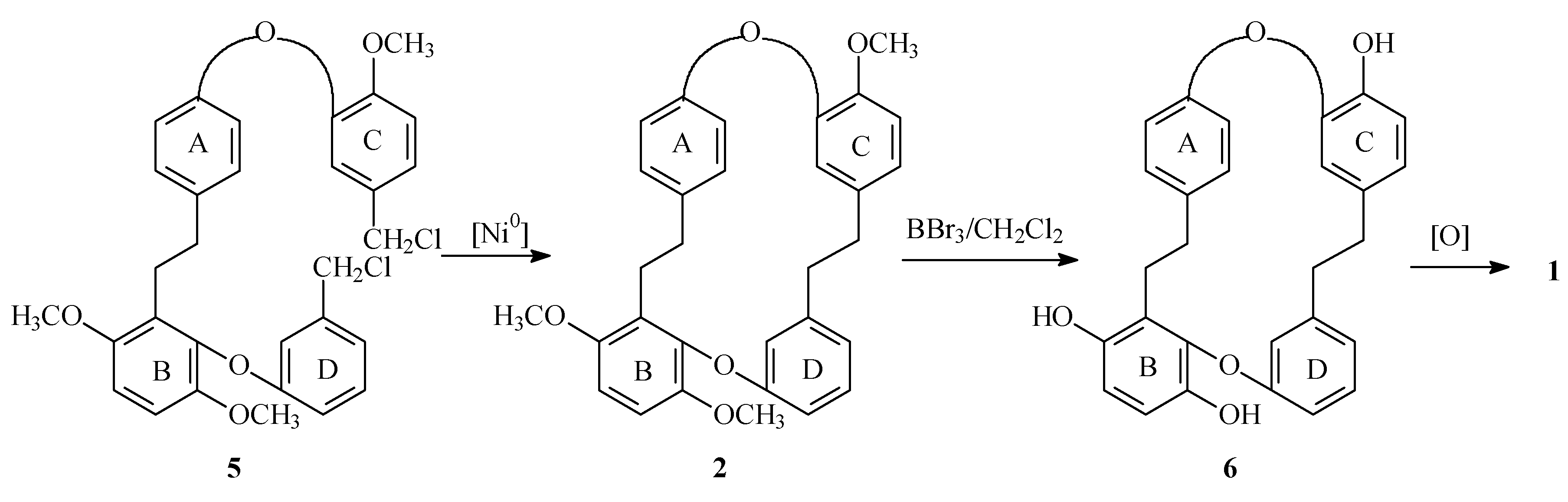
References and Notes
- Zinsmeister, H.D.; Becker, H.; Eicher, Th. Angew. Chem. 1991, 103, 134.
- Asakawa, Y. Progress in the Chemistry of Organic Natural Products; Herz, E., Kirby, G.W., Moore, R.E., Steglick, W. C., Tamm., Eds.; Springer: Wien, New York, 1995; p. 5. [Google Scholar]
- Wei, H.-C.; Wu, C.-L. J. Chem. Research (S) 1991, 230.
- Wei, H.-C.; Ma, S.-J.; Wu, C.-L. Phytochemistry 1995, 39(1), 91.
- López, V.; Pandolfi, E.; Seoane, G. VII Jornadas de Jovens Pesquisadores do Grupo Montevideo; Curitiba, Brasil, 9-11 de setiembre de 1999. [Google Scholar]
