Abstract
α-Alkoxynitrile 1 in the presence of tris(methylthio)methyllithium 2 at –78°C gave the dimer 5 instead of the expected C1-elongated product 3. The formation of com-pound 5 is explained in terms of anion formation and self-condensation, a variant of the Thorpe reaction. Scrutinizing the 1H NMR spectra revealed that the enamine tautomer 5b is predominant over the imine 5a in the solvents investigated.
Introduction
Nitrile 1 (Scheme 1) was obtained en route to acyclic analogues of the C-nucleoside tiazofurin [1]. This compound was deemed to be a good starting material for the synthesis of α-keto ester 4 via in-termediate 3. This assumption was based on analogy with the Grignard reaction of nitriles giving rise to ketones [2] or the Blaise reaction (Reformatsky reaction of nitriles affording β-keto esters) [3].
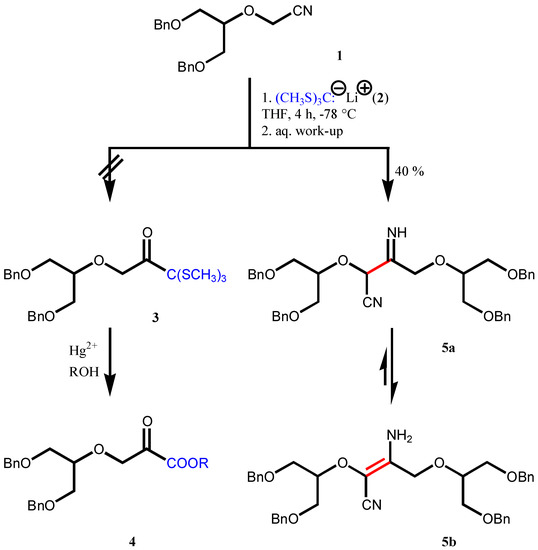
Scheme 1.
For the required carboxylate anion synthon different tris(alkylthio or arylthio)methyllithiums, de-rived from tris(alkyl or arylthio)methanes [4], were used successfully in the C1 elongation of ketones [5] and lactones [6]. Most often [tris(methylthio)methyl]lithium (2) was applied which is stable up to –40°C [7]. The obtained trithioorthoesters can be transformed into the corresponding esters using the common mercury(II) salt-assisted demercaptalisation procedure. In principle, alkyl chloroformates could also be employed as C1 synthons. However, in the single reported Reformatsky-type reaction of ethyl chloroformate with ethyl acetoacetate in the presence of a zinc-copper couple leading to diethyl 2-hydroxy-2-methylsuccinate, the yield was rather low (20%) [8]. In a recent application, ethyl chloro-formate was allowed to react with methyl phenylglyoxylate in a titanium(IV)-promoted reaction to af-ford ethyl methyl 2-hydroxy-2-phenylmalonate in 60% yield [9].
Results and Discussion
Nitrile 1 was allowed to react with compound 2 at –78°C for 4 h. Contrary to our expectations, compound 3 was not obtained. The isolated substance exhibited surprisingly complicated 1H NMR spectra in different solvents, according to the microanalysis it contained no sulfur and its elemental composition was identical with that of nitrile 1. Its low-voltage (15 eV) EI mass spectrum showed a molecular ion at m/z 622 (the molecular mass of the starting nitrile is 311) and a fragmentation pattern compatible only with the dimeric structures 5a and 5b. Scrutiny of the 1H NMR spectrum revealed that the substance exists as an enamine (5b) rather than an imine (5a). The IR spectrum also confirmed the presence of a nitrile group (νCN 2180 cm-1).
The literature search for precedents (Beilstein’s CrossFire 4.0, release BS9902PRPR using Beilstein Commander 4.0) showed that α-alkoxynitriles react normally with Grignard reagents [10] while orga-nolithium [11] and organosodium compounds [12,13] provoke self-condensation. This can be ex-plained by the strong basicity of organolithiums and the result is, just as in the above instance, the spe-cial case of the well-known Thorpe reaction [14] (Scheme 2).
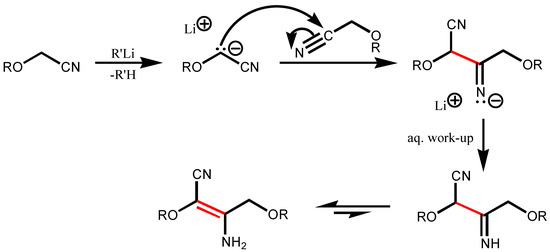
Scheme 2.
It is noteworthy that even acetonitrile tends to react abnormally with Grignard reagents, e.g. with phenylmagnesium bromide, only 42% of the expected acetophenone has been obtained along with 32% of benzene [15]. Nitriles lacking α-hydrogens react normally and afford ketones.
Experimental
General
To tris(methylthio)methane [4] (0.927 g, 6.0 mmol) dissolved in anhydrous THF (2.0 mL) was added butyllithium (1.26 M solution, 5.20 mL, 6.60 mL) at -78°C under nitrogen. Nitrile 1 [1] (1.40 g, 4.5 mmol) dissolved in anhydrous THF (10.0 mL) was added dropwise to this solution after 20 min and kept at –78°C for 4 h. The reaction mixture was quenched at –60°C with satd. NH4Cl solution. Af-ter allowing to warm up to room temperature, the mixture was diluted and extracted with chloroform (25 mL). The aqueous phase was extracted with chloroform (2 × 50 mL). The combined organic phases were dried (MgSO4) and evaporated in vacuo. Chromatographic purification using 20% (v/v) ethyl acetate in hexanes afforded 3-amino-2,4-bis{2-(benzyloxy)-1-[(benzyloxy)methyl]-ethoxy}-2-butene-nitrile (5, 0.56 g, 40%) as a yellow oil.
Spectral Data
IR (KBr, ν, cm-1): 3450m, 3330m, 3020m, 2910s, 2860s, 2180m, 1680m, 1493m, 1450m, 1090m.
1H NMR (CDCl3, 200 MHz, δ, ppm): 3.55 (d, J 6 Hz, 4 H, 2 x CH2); 3.65 (d, J 6 Hz, 4 H, 2 x CH2); 3.71 (m, 1 H, CH); 3.94 (m, 1 H, CH); 4.37 (s, 2 H, 4-CH2); 4.49 (s, 4 H, 2 x PhCH2); 4.52 (s, 4 H, 2 x PhCH2); 5.45 (br s, exchangeable with D2O, 2 H, NH2); 7.30 (m, 20 H, 4 x C6H5).
1H NMR (DMSO-d6, 200 MHz, δ, ppm): 3.52 (d, J 5 Hz, 4 H, 2 x CH2); 3.65 (d, J 5 Hz, 4 H, 2 x CH2); 3.72 (m, 1 H, CH); 3.88 (m, 1 H, H); 4.20 (s, 2 H, 4-CH2); 4.45 (s, 8 H, 4 x PhCH2); 6.00 (br s, exchangeable with D2O, 2 H, NH2); 7.30 (m, 20 H, 4 x C6H5).
EI-MS (15 eV, m/z, %): 622 (5, M+); 531 (1, M-91); 418 (4); 387 (4); 367 [4, M-CH(CH2OBn)2]; 331 (6); 292 (9); 278 (24); 220 (14); 181 (75); 102 (88); 91 (100, C7H7+).
Anal. calcd. for C38H42N2O6 (622.750): C, 73.29; H, 6.80; N, 4.50; found: C, 73.49; H, 6.71; N, 4.18.
References and Notes
- Kovács, L.; Herczegh, P.; Batta, G.; Farkas, I. Two acyclic analogues of 2-β-D-ribofuranosyl-thiazole-4-carboxamide (tiazofurin). Heterocycles 1987, 26, 947–960. [Google Scholar]
- March, J. Advanced Organic Chemistry, 4th ed.; John Wiley and Sons: New York, 1992; pp. 935–936. [Google Scholar]
- Gaudemar, M. The Reformatsky reaction in the course of the last thirty years. Organometal. Chem. Rev. A 1972, 8, 183–233. [Google Scholar]
- Holmberg, B. On the esters of orthotrithioformic acid. Ber. Dtsch. Chem. Ges. 1907, 40, 1740–1743. [Google Scholar] [CrossRef]
- Dailey, O. D.; Fuchs, P. L. Synthesis of a model for the BCE ring system of bruceantin. A caveat on the cyclohexene→trans diaxial diol conversion. J. Org. Chem. 1980, 45, 216–236. [Google Scholar] [CrossRef]
- Hengeveld, J. E.; Grief, V.; Tadanier, J.; Lee, C.-M.; Riley, D.; Lartey, P. A. A general synthesis of methyl aldulosonates using tris(methylthio)methyl lithium as the ester anion equivalent. Tetra-hedron Lett. 1984, 25, 4075–4078. [Google Scholar] [CrossRef]
- Damon, R. E.; Schlessinger, R. H. An efficient and stereospecific total synthesis of DL-protoli-chesterinic acid. Tetrahedron Lett. 1976, 1561–1564. [Google Scholar] [CrossRef]
- Antia, M. B.; Kaushal, R. Reformatsky condensation of ketonic esters with halo esters. Agra Univ. J. Res., Sci. 1954, 3, 197–201, (Chem. Abstr. 1955, 49, 6121h). [Google Scholar]
- Clerici, A.; Clerici, L.; Porta, O. Acylation of a transient Ti(IV)-enolate by acyl halides and anhy-drides. Facile synthesis of α-hydroxy-β-ketones. Tetrahedron 1996, 52, 11037–11044. [Google Scholar] [CrossRef]
- Lingo, S. P.; Henze, H. R. Substituted ethers derived from ethylene chlorohydrin. J. Am. Chem. Soc. 1939, 61, 1574–1576. [Google Scholar] [CrossRef]
- Gauthier, R.; Axiotis, G. P.; Chastrette, M. Double addition of organometallic compounds to β-oxygenated nitriles RCN. Preparation of primary amines of RCR1R2NH2 type. J. Organomet. Chem. 1977, 140, 245–255. [Google Scholar] [CrossRef]
- Allen, C. F. H.; van Allan, J. A. Some macrocyclic oxalactones and related substances. J. Org. Chem. 1949, 14, 754–760. [Google Scholar] [CrossRef]
- Guibert, S.; Cariou, M.; Simonet, J. Research on novel organic solvents with potential use in lith-ium batteries 2. The properties of certain aliphatic nitriles substituted by donor groups. Bull. Soc. Chim. Fr. 1988, 924–929. [Google Scholar]
- March, J. Advanced Organic Chemistry, 4th ed.; John Wiley and Sons: New York, 1992; pp. 963–964. [Google Scholar]
- Jones, R. L.; Pearson, D. E.; Gordon, M. Studies on the reaction of phenylmagnesium bromides with acetonitrile. J. Org. Chem. 1972, 37, 3369–3370. [Google Scholar] [CrossRef]
- Samples Availability: not available.
© 2000 by MDPI (http://www.mdpi.org). Reproduction is permitted for noncommercial purposes.