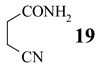Abstract
The NaY zeolite catalysed hydrolysis of nitriles to primary amides is reported. It is found that aryl nitriles with strong electron-withdrawing substituents and cyanopyridines are readily hydrolysed in the water suspension, while aliphatic nitriles do not react.
Introduction
Zeolites are effective catalysts in organic chemistry and their specificity in gas phase transforma- tions is greatly utilised in industry [1]. Alkylation reaction, polymerisation, cyclization [2], pho- toreduction [3], or preparation of nitroalkenes [4], occur in gas phase or with reactants sorbed within zeolite in inert solvent. Recently, several reports on the use of acidic zeolites (HY) in macrolactoniza- tion [5], acetalization [6], acetylation [7] and gem-diacetalization [8], as well as the synthesis and ap- plication of the first organic-functionalized zeolite-beta [9], prompted us to investigate the new cata- lytic possibilities of NaY zeolite.
Here we wish to report the application of NaY zeolite as reusable catalyst in the hydrolysis of nitriles to primary amides.
Results and Discussion
In our experiments the suspension of NaY zeolite and a nitrile in water (or methanol) was heated to reflux for a given period (Table 1), zeolite was filtered off, and products were separated (or directly crystallised). Most important observation is that nitriles are hydrolysed only to the amide stage. Cya- nopyridines and benzonitriles with electron-withdrawing substituents are readily hydrolysed in good yield (Table 1, entries 1-3, 7).

Table 1.
Hydrolysis of nitriles into amidesa. 
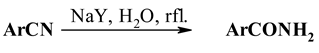
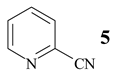
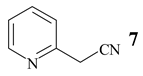
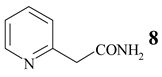
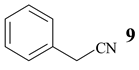
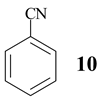
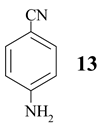
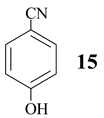
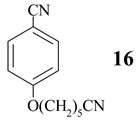
Benzonitrile (10, entry 6) was totally resistant to hydrolysis, while benzonitriles substituted with week inductive electron-withdrawing groups (with strong +R, 4-aminobenzonitrile (13) and 4-(5- cyanopentoxy)benzonitrile (16) [10], Table 1, entries 8 and 10) were hydrolysed, although in low yield (35% and 21%, respectively). Hydroxy substituent completely prevented the hydrolysis (15, Table 1, entry 9) probably as a consequence of phenoxy ion formation. The case of di-nitrile 16 is very interesting: it shows that hydrolysis proceeds by blocking the formation of phenoxy ion, while at the same time pointing to the resistance of aliphatic nitriles to hydrolysis. The resistance of aliphatic nitriles was confirmed by attempted hydrolysis of CH3CN (not shown) and by hydrolysis of ethyl 3- cyanopropanoate 20 only to cyanoacid 21 (Table 1, entry 12). Benzylic cyano group was also found to be resistant to applied conditions (9, Table 1, entry 5), or was very slightly hydrolysed when CH2CN was attached to the electron-withdrawing pyridine ring (7 →8, 16%, Table 1, entry 4).
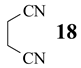
Hydrolysis of succinodinitrile (18, Table 1, entry 11) afforded only 35% of 2-cyanopropanamide (19) and 65% of educt. It is interesting to note that much greater site-differentiation was achieved in enzymatic hydrolysis of α,ω-dinitriles into corresponding ω-cyanoacids [11].
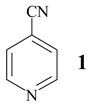
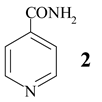
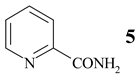
In some cases, prolonged reaction time resulted in higher yields, as is given for 4-cyanopyridine (1) (Table 1, entry 1, 6 h (67%) → 24 h (87%)). The reusability of NaY catalyst was tested using 3- cyanopyridine (3, Table 1, entry 2). Four runs were performed with the same batch of the catalyst without significant loss of its activity.
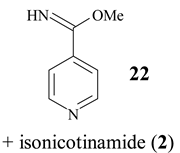
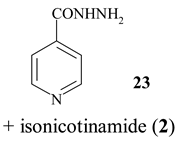
In Table 2 the influence of reactant (solvent) is shown. Using methanol instead of water the imino ester 22 was obtained in good yield (67%; entry 2), while ethanol and higher homologues (propanol and 1-butanol) were ineffective. However, hydrazine hydrate afforded isoniazide (23, 67%) along with 18% of isonicotinamide (2).

Table 2.
Solvolysis of 4-cyanopyridine (1).
The influence of the amount of catalyst on product distribution is exemplified with methanolysis of 4-cyanopyridine (1) (Table 3). Product formation started with 6% (w/w) of catalyst, and the increase of zeolite part did not significantly affect the product distribution, what, beside its already shown reus- ability, confirms the true catalytic nature of NaY zeolite.

Table 3.
Methanolysis of 4-cyanopyridine (1): dependence on catalyst-to-substrate ratio (reflux 15 h).
Conclusion
We have shown that NaY zeolite can be used in the simple procedure as a reusable catalyst for hy- drolysis of aromatic nitriles, primarily of cyanopyridines and benzonitriles possessing electron- withdrawing groups. Contrary to hydrolysis under acidic conditions, benzyl- and alkanenitriles are sta- ble under conditions applied, so enabling their further selective transformations. In addition, NaY zeo- lite can also be used for imino ester preparation as an alternative to Pinner synthesis.
Experimental
General
Melting points were determined on a Mikro-Heiztisch Boetius PHMK apparatus and were not corrected. IR spectra were recorded on Perkin-Elmer spectrophotometer FT-IR 1725X. 1H and 13C NMR spectra were recorded on a Varian Gemini-200 or Bruker AM-250 spectrometers. Chemical shifts were expressed as ppm (δ) values and coupling constants (J) in Hz. Mass spectra were taken on a Finnigan- MAT 8230 spectrometer, as indicated below.
In our experiments, NaY zeolite with following characteristics was used [12]: crystallinity 100%; SiO2 [%] 63.80; Al2O3 [%] 22.90; NaO [%] 13.30; molar ratio SiO2 / Al2O3: 4.73; specific area (B.E.T) 850 m2 / g; pore volume 0.32 cm3 / g; diameter of crystallite 3.5 μm; diameter of granulate 150 μm; pH of water suspension 10.05. Zeolite was pre-dried only for methanolysis reaction, in order to suppress the formation of an amide, and for acetylation reactions.
Hydrolysis of Nitriles - General
The suspension of a nitrile (200 mg) and zeolite (800 mg) in water (5 ml) was heated to reflux (for details see Table 1). The hot reaction mixture was filtered and zeolite was washed with water (and/or methanol). When catalyst was reused, it was dried on air overnight. Pure amides were crystallised di- rectly from the crude product mixture or were purified by column chromatography (SiO2 or RP-18). All isolated compounds were fully characterised by spectroscopic and analytical methods.
The data of known compounds were compared with literature data given in [13], and that refers to:
- 2: mp 152-154°C, [14] mp 155-157°C
- 4: mp 128-129°C, [13, p. C-474] mp. 129-131°C
- 6: mp 106-108°C, [13, p. C-474] mp 107-108°C
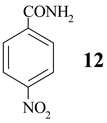
- 12: mp 201-202.5°C, [13, p. C-197] mp 201.4°C
- 14: mp 184-186°C, [13, p. C-183] mp 183°C
- 21: mp 48-50°C, [11] mp 49.5-51°C.
2-Pyridylacetamide (8)
Mp 120-121°C. (colourless needles, water). IR (KBr) cm−1: 3377, 3188, 3112, 3017, 1678, 1646, 1597, 1570, 1439, 1402. 1H NMR (CD3OD, 200 MHz): 8.51-8.43 (m, 1H, H-C(6')), 7.78 (td, J = 7.8, 1.8, H-C(4')), 7.40 (d, J = 8.0, H-C(3')), 7.30 (ddd, J = 8, 5, 1, H-C(5')), 3.75 (s, 2H-C(2)). 13C NMR (CD3OD, 50 MHz): 174.97 (C1), 156.82 (C2'), 149.86 (C6'), 138.76 (C4'), 125.69 (C3'), 123.59 (C5'), 45.12 (C2). Anal. calc. for C7H8N2O (136.06): C 61.75, H 5.92, N 20.57, found: C 62.08, H 5.57, N 20.83.
3-Cyanopropanamide (19)
Mp 86-88°C (colourless amorphous solid). IR (KBr) cm−1: 3414, 3225, 2293, 2248, 1681, 1619, 1421. 1H NMR (D2O, 200 MHz) δ: 2.80-2.65 (m, 4H, 2H-C(2), 2H-C(3)). 13C NMR (D2O, 50 MHz) δ: 177.90 (C1), 123.04 (C4), 32.38 (C2), 15.12 (C3). MS (CI, isobutane): 197 (2 M++1). Anal. calc. for C4H6N2O (98.11): C 48.97, H 6.16, found: C 49.34, H 6.08.
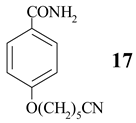
4-(5-Cyanopentoxy)benzamide (17)
Colourless solid mp 97-101°C. IR (KBr) cm−1: 3466, 2140, 1614, 1566, 1401. 1H NMR (CD3OD, 200 MHz): 7.84 (AA'BB', J = 6.8, 2, H-C(2), H-C(6)), 6.98 (AA'BB', J = 6.8, 2, H-C(3), H-C(5)), 4.06 (t, J = 6.2, 2H-C(1')), 2.49 (t, J = 6.8, 2H-C(5')), 1.90-1.57 (m, 6H). MS (EI, 70 eV): 232 (M+, 25), 137 (33), 121 (100), 96 (14), 55 (8), 41(5). Anal. calc. for C13H16N2O2 (232.12): C 67.22, H 6.94, N 12.06, found: C 67.54, H 6.90, N 12.54.
Iminoester 22
The suspension of 4-cyanopyridine (1, 200 mg) and zeolite (800 mg) in methanol (5 ml) was heated to reflux for 14 h. Hot reaction mixture was filtered and zeolite was washed with methanol. Crude product was chromatographed on Lobar RP-18 column (eluent: CH3OH / H2O = 40 : 60) affording the analytical sample of iminoester 22 (175 mg, 67%), isonicotinamide (2, 28 mg, 12%) and educt 1 (36 mg, 18%). 22: mp = 45°C (colourless needles, diisopropyl ether). IR (KBr) cm−1: 3290, 3220, 3034, 1651, 1603, 1556, 1445, 1348, 1313, 1108, 1087. 1H NMR (200 MHz, DMSO-d6): δ 9.50 (bs, HN=C), 8.76 (AB, J = 4, H-C(2), H-C(6)), 7.80 (AB, J = 4, H-C(3), H-C(5)), 3.82 (s, CH3O-). 13C NMR (50 MHz, DMSO-d6, DEPT): δ 164.14 (C=N), 150.60 (C2 and C6), 138.82 (C4) 121.08 (C3 and C5), 53.46 CH3. MS CI (isobutane): 137 (MH+). Anal. calc. for C7H8N2O (136.15): C 61.75, H 5.92, N 20.57, found: C 62.04, H 5.89, N 19.96.
Isoniazid (23)
The suspension of 4-cyanopyridine (1, 1.00 g) and zeolite (1.00 g) in hydrazine hydrate (1.00 ml) and water (8 ml) was heated at 90°C for 2 h. Hot reaction mixture was filtered and zeolite was washed with hot ethanol. Crude product crystallised affording 478 mg of 23, and the rest was chromatographed on SiO2 column (eluent: methanol / EtOAc (1:9)). Isoniazid (23) was obtained in combined yield of 67% (883 mg), together with 211 mg (18%) of isonicotinamide (2). 23: mp 169°C (colourless needles, ethanol). [13, p. C-475] mp 171-173°C. IR (KBr) cm −1: 3111, 3050, 1667, 1635, 1557. 1H NMR (DMSO-d6, 250 MHz): 10.13 (s, 1H), 8.76-8.67 (m, 2H, H-C(2), H-C(6)), 7.80-7.68 (m, 2H, H-C(3), H-C(5)), 4.77-4.60 (m, 2H). 13C NMR (DMSO-d6, 62.5 MHz): 164.02 (C=O), 150.26 (C2 and C6), 140.30 (C4), 121.06 (C3 and C5). Anal. calc. for C6H7N3O (137.17): C 52.54, H 5.15, N 30.64; found: C 52.31, H 5.27, N 30.83.
Acknowledgements:
This work was supported in part by Epsilon Research Ltd., High Wycombe, Bucks, UK, and by Serbian Academy of Sciences and Arts.
References and Notes
- Hölderich, W.; Hesse, M.; Naumann, F. Zeolites: catalysts for organic synthesis. Angew. Chem. Int. Ed. Engl. 1988, 27, 226–246. [Google Scholar] [CrossRef]
- Sen, S. E.; Zhang, Y. Z.; Roach, S. L. Zeolite-mediated cyclization of an epoxide-containing polyene. J. Org. Chem. 1996, 61, 9534–9537. [Google Scholar] [CrossRef]
- Rao, J. V.; Uppili, S. R.; Corbin, D. R.; Schwarz, S.; Lustig, S. R.; Ramamurthy, V. Facial selec- tive photoreduction of steroids: role of zeolites. J. Am. Chem. Soc. 1998, 120, 2480–2481. [Google Scholar] [CrossRef]
- Sreekumar, R.; Padmakumar, S. R.; Rugmini, P. Regioselective reduction of epoxides and conju- gated carbonyl compounds using zeolite supported zinc borohydride. Tetrahedron Lett. 1998, 39, 5151–5154. [Google Scholar] [CrossRef]
- a) Ookishi, T.; Onaka, M. Zeolite-catalyzed macrolactonization of ω-hydroxyalkanoic acids in a highly concentrated solution. Tetrahedron Lett. 1998, 39, 293–296. [Google Scholar] ; b) Tatsumi, T.; Sakashita, H.; Asanao, K. Selective macrolactonization using zeolite molecular sieves. J. Chem. Soc., Chem. Commun. 1993, 1264–1265. [Google Scholar]
- a) Ballini, R.; Bosica, G.; Frullanti, B.; Maggi, R.; Sartori, G.; Schroer, F. 1,3-Dioxolanes from carbonyl compounds over zeolite HSZ-360 as a reusable, heterogenous catalyst. Tetrahedron Lett. 1998, 39, 1615–1618. [Google Scholar] ; b) Corma, A.; Climent, M. J.; Carcia, H.; Primo, J. Applied Catalysis 1990, 59, 333–340.
- Ballini, R.; Bosica, G.; Carloni, S.; Ciaralli, L.; Maggi, R.; Sartori, G. Zeolite HSZ-360 as a new reusable catalyst for the direct acetylation of alcohols and phenols under solventless conditions. Tetrahedron Lett. 1998, 39, 6049–6052. [Google Scholar] [CrossRef]
- Ballini, R.; Bordoni, M.; Bosica, G.; Maggi, R.; Sartori, G. Solvent free synthesis and deprotec- tion of 1,1-diacetates over a commercially available zeolite Y as a reusable catalyst. Tetrahedron Lett. 1998, 39, 7587–7590. [Google Scholar] [CrossRef]
- a) Jones, C. W.; Tsuji, K.; Davis, M. E. Organic-functionalized molecular sieves as shape- selective catalysts. Nature 1998, 393, 52–54. [Google Scholar] ; b) Creyghton, E. J. Organic groups cling to the pores. Nature 1998, 393, 21–22. [Google Scholar]
- The structure of 4-(5-cyanopentoxy)benzamide (17) was deduced (beside IR and microanalytical data) by comparison of its spectral data with that of 16 (vide infra). 1H NMR: replacing of CN group with CONH2 on benzene ring leads to downfield shift of ortho-protons (0.25 ppm) and small upfield shift of meta-protons (-0.08 ppm), what is in good agreement with observed values (0.25 and -0.04 ppm, respectively). Source: Pretsch, E.; Clerc, T.; Seibl, J.; Simon, W. Tabellen zur Strukturaufklä rung organischer Verbindungen, 3rd Ed. ed; Springer-Verlag: Berlin - Heidelberg - New York - Tokyo, 1986; p. H260. [Google Scholar] Also, in mass spectra of both compounds cyanoalkyl chain and McLafferty rearrangement could be observed (m/e 96 and 41, respectively). Data for 4-(5- cyanopentoxy)benzonitrile (16): colourless oil. 1H NMR (CDCl3, 200 MHz): 7.59 (AA'BB', J = 6.8, 2, H-C(2), H-C(6)), 6.94 (AA'BB', J = 6.8, 2, H-C(3), H-C(5)), 4.03 (t, J = 6.2, 2H-C(1')), 2.41 (t, J = 6.8, 2H-C(5')), 1.93-1.56 (m, 6H). 13C NMR (CDCl3, 50 MHz): 162.13 (C4), 133.99 (C2 and C6), 119.97 (CN), 119.20 (CN), 115.09 (C3 and C5), 103.89 (C1), 67.66 (C1'), 28.19, 25.20, 25.04, 17.08 (C5'). MS (EI, 70 eV): 215 (44), 214 (M+, 95), 119 (100), 96 (35), 55 (22), 41 (39). Anal. calc. for C13H14N2O (214.11): C 72.87, H 6.59, found: C 72.96, H 6.87.
- Gavagan, J. E.; Fager, S. K.; Fallon, R. D.; Folsom, P. W.; Herkes, F. E.; Eisenberg, A.; Hann, E. C.; DiCosimo, R. Chemoenzymatic production of lactams from aliphatic α,ω-dinitriles. J. Org. Chem. 1998, 63, 4792–4801. [Google Scholar] [CrossRef]
- Stojković, S.; Gajinov, S.; Adnadević, B. Correlation between a degrees of crystallinity of acid treated NaA and NaY zeolites as determined by infrared and X-ray diffraction. Spectroscopy Lett. 1991, 24, 801–815. [Google Scholar] [CrossRef]
- CRC Handbook of Chemistry and Physics, 57th Ed. ed; CRC Press Inc.: Cleveland, Ohio; pp. 1976–1977.
- Beilstein Handbuch Der Organischen Chemie; Springer-Verlag: Berlin - Göttingen - Heidelberg, 1953; E-II; Volume 22, p. 37.
- Samples Availability: Available from the authors.
© 2000 by MDPI (http://www.mdpi.org). Reproduction is permitted for noncommercial purposes.