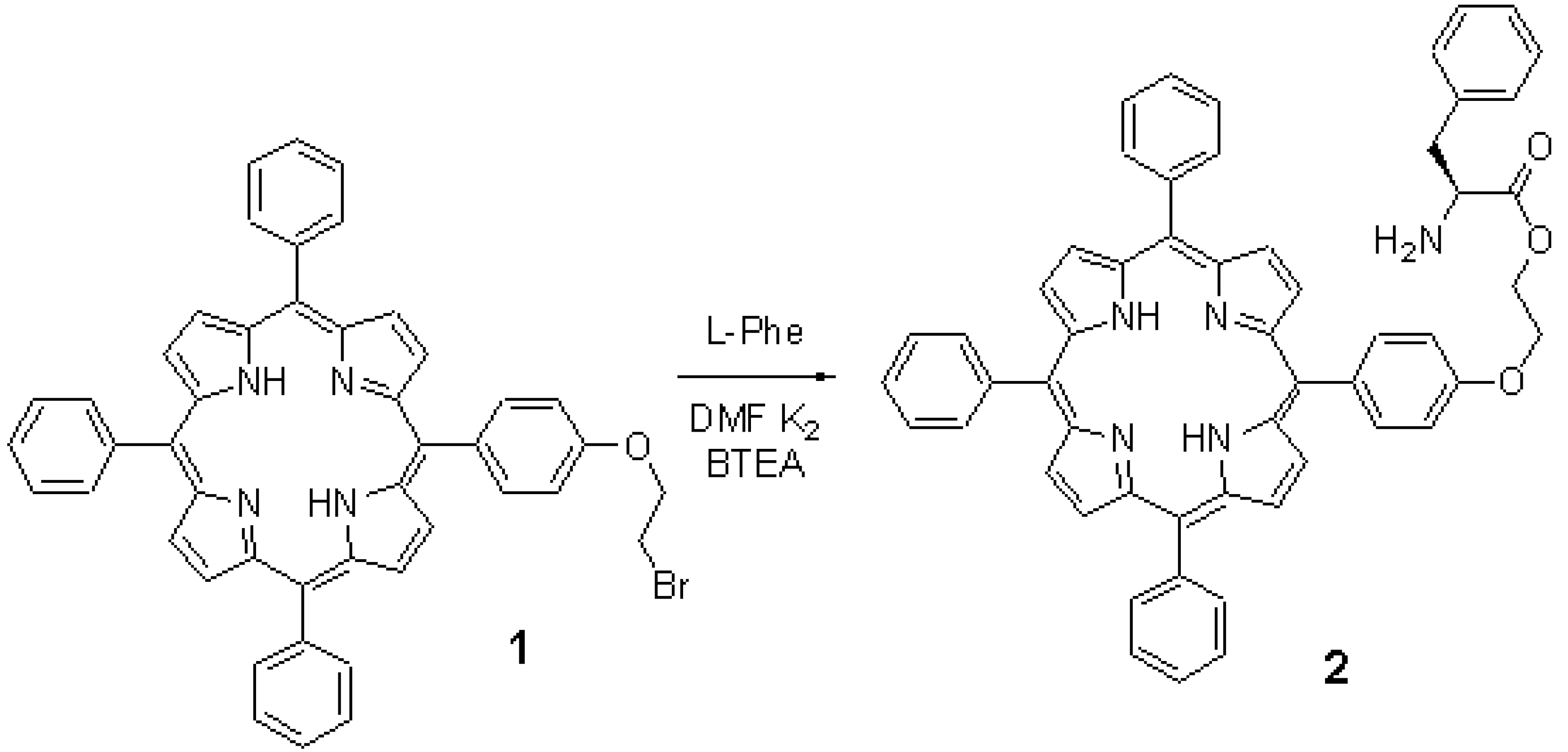
5-[p-(2-Bromo-1-ethoxy)]phenyl-10,15,20-triphenylporphyrin (1) was prepared by previously published method [1]. L-phenylalanine was chrom. pure. Chloroform (A.R. grade) was dried with P2O5 and distilled before use. Silica gel was C.P. grade (10-40 m). Other reagents were A.R. grade chemicals as supplied. The mixture of 1 (0.27mmol), L-phenylalanine (2.42mmol), BTEAC (25mg) and anhydrous potassium carbonate (4g) was stirred in DMF (50ml) at room temperature for 72 hours. Reaction mixture was then poured into saturated NaCl solution (150ml). The precipitate was filtered and washed several times with water. Product was then purified by means of chromatography on silica gel with chloroform as an eluent. Purple solid 5-[p-(1-L-phenylalanyloxy)ethoxyl]phenyl-10,15,20-triphenylporphyrin (2) was obtained, 0.122g. Yield: 55%.
IR (KBr): 3322.8, 3056.7, 3027.7, 2923.6, 2854.2, 2705.7, 2605.4, 2534.0, 1814.7, 1737.6 (C=O), 1631.5, 1602.6, 1562.1, 1506.2, 1471.4, 1442.5, 1402.0, 1349.9, 1284.4, 1243.9, 1222.7, 1178.3, 1112.7, 1074.2, 966.2, 929.5, 846.6, 800.3, 734.8, 702.0, 648.0, 555.4, 528.4.
UV-Vis l max (nm): 419.1, 516.1, 551.3, 591.5, 648.5.
1H-NMR (CDCl3): -2.77 (s, 2H, pyrrole NH), 1.75 (br. s, 2H, -NH2), 3.06 (m, 1H, CH2), 3.21 (m, 1H, CH2), 3.91 (t, 1H, C*H), 4.40 (m, 2H, -CH2-), 4.65 (m, 2H, -CH2-), 7.24-8.22 (several m, 24H, Ph-H), 8.85 (m, 8H, pyrrole b-H ).
FAB-MS ([M+1]+): 822.
Anal. calc. for C55H43N5O3 (821): C 80.39, H 5.24, N 8.53; found: C 80.21, H 5.13, N 8.46.
Supplementary materials
Supplementary File 1Supplementary File 2Acknowledgment
This research was supported by the Natural Science Foundation of Guangdong Province.
Reference
- Ji, L.-N.; Qin, X.; Huang, J.-W. Acta Scientiarum Natruralium Universitatis Sunyatseni, 1993; 32, 2, 1.
- Sample Availability: Available from the authors and from MDPI. MDPI 17056.
© 1999 MDPI. All rights reserved. Molecules website www.mdpi.org/molecules/
