Abstract
Essential oils (EOs) from combined plant materials offer a promising alternative to conventional extraction by enhancing chemical diversity and bioactivity. This study evaluated the chemical composition and insecticidal properties of individual and combined plant EOs from Cymbopogon citratus, Eucalyptus camaldulensis, Eucalyptus lehmannii, Salvia rosmarinus and Thymus vulgaris were evaluated against aphids. Binary and ternary combinations were prepared in equal proportions prior to hydrodistillation. GC-MS analysis revealed significant compositional shifts in EOs from combined plant materials. Major compounds in individual oils included citral (53.11%) and neral (29.14%) in C. citratus, thymol (70.84%) in T. vulgaris, and eucalyptol as the predominant compound in E. camaldulensis (66.51%), E. lehmannii (56.99%) and S. rosmarinus (46.56%), respectively. In the combined oils, the relative abundance of these constituents was altered, and in some cases new constituents were introduced. Principal Component Analysis (PCA) and Hierarchical Cluster Analysis (HCA) revealed that combined plant EOs clustered near their parental oils, indicating compositional inheritance. Contact toxicity assay against Aphis fabae demonstrated enhanced efficacy of the combined oils, with reduced LC50 values (1.39 µL mL−1 for E. camaldulensis + T. vulgaris) and synergistic interactions, indicated by a co-toxicity coefficient (CTC) of 221.58 and elevated synergistic factors. Pearson correlation analysis and Partial Least Squares (PLS) regression jointly identified Acorenone B and thymol as negatively, and caryophyllene as positively correlated compounds, all with relatively high contribution to insecticidal activity, ranking highest with a Variable Importance in Projection (VIP) scores > 1.0. While PLS model had modest predictive power, the integration of these statistical approaches supports the insecticidal potential of combined plant-derived EOS in laboratory bioassays and indicates their relevance to sustainable crop protection.
1. Introduction
The combination of essential oils (EOs) from different plant species, either through conventional blending of extracted oils, or through the distillation of combined plant materials, introduces a complex chemical matrix that can profoundly influence their biological activities [1,2]. These combinations can lead to specific interactions among the constituent compounds [3]. Additive effects occur when the combined bioactivity matches the cumulative activity of the individual oils [4]. In contrast, antagonistic interactions happen when one or more components reduce the mixture’s efficacy [5]. Synergistic effects are particularly interesting because they produce a combined activity that surpasses the predicted sum of the individual parts [6]. The nature of these interactions depends on several factors, including chemical composition, concentration ratios, and the method used to combine the oils [7]. The distillation of combined plant materials, entailing the simultaneous extraction of multiple botanical sources, can uniquely promote interactions among volatile compounds during the extraction process [8]. This approach has the potential to yield distinct chemical profiles with enhanced antimicrobial activity [9]. Despite its promise, the mechanisms underlying such interactions remain complex and insufficiently elucidated, thereby highlighting the need for further investigation [10].
Interest in EOs as sustainable alternatives to synthetic pesticides has grown due to their broad-spectrum antimicrobial activity and diverse biological targets, which help reduce resistance development in pests and pathogens [11,12,13]. While extensive research has focused on individual EOs and their mixtures, there is comparatively less information on EOs obtained by distilling combined plant materials (CPM-EOs). Unlike post-distillation blends, combining plant materials prior to distillation may alter chemical composition and produce unique synergistic biological effects, highlighting the novelty and necessity of the present study [14,15].
Aromatic and medicinal plants such as C. citratus, S. rosmarinus, T. vulgaris, E. camaldulensis and E. lehmannii are globally distributed and have been traditionally used for their medicinal and pesticidal properties [16,17]. These species have demonstrated significant antimicrobial and insecticidal potential individually [18]. However, the properties of EOs obtained from their combined plant materials remain underexplored [19]. Leveraging the synergistic potential of these plants species presents promising opportunities for the development of versatile and sustainable biopesticides, tailored to contemporary agricultural systems [20]. Agricultural production across many regions faces serious threats from insect pests including Aphis fabae (A. fabae), a major pest of legumes that imposes additional pressures on crop health and productivity [21]. A. fabae is also an important vector of viruses on potato, pepper and faba bean in Tunisia [22]. Effective biocontrol agents targeting such pests are crucial components of integrated pest management strategies that could include the use of EOS, representing a more sustainable approach to pest control with minimal environmental impact [23,24].
This study investigates EOs extracted from five aromatic species, C. citratus, E. camaldulensis, E. lehmannii, S. rosmarinus and T. vulgaris. The research focuses on both individual oils and selected binary and ternary combinations, aiming to evaluate how distillation of combined plant materials influences their chemical profiles and biological activities. Chemical composition was analyzed using gas chromatography–mass spectrometry (GC–MS). Insecticidal efficacy was evaluated against A. fabae through contact toxicity testing. Principal Component Analysis (PCA) and Hierarchical Cluster Analysis (HCA) were performed on the chemical composition data to evaluate the compositional patterns. In addition, Pearson correlation and Partial Least Squares (PLS) regression were performed to identify the main constituents associated with insecticidal activity based on LC50 values. These results contribute to a better understanding of the chemical basis of the bioactivity of combined EOs and support their potential role in sustainable crop protection.
2. Results
2.1. Chemical Composition of Individual and Combined Plant Material Essential Oils
The chemical analysis of individual and CPM-EOs from C. citratus, E. camaldulensis, E. lehmannii, S. rosmarinus, and T. vulgaris, as well as their binary and ternary mixtures, led to the identification of 35 compounds, representing 93.66% to 99.99% of the total oil composition. The identified constituents were grouped into four major chemical classes: monoterpene hydrocarbons, oxygenated monoterpenes, sesquiterpene hydrocarbons, and oxygenated sesquiterpenes. All EOs, whether individual or combined, were dominated by oxygenated monoterpenes, ranging from 62.99% to 90.39%, followed by monoterpene hydrocarbons (9.59% to 33.6%) (Table 1).

Table 1.
Chemical composition (%) and yield (% w/w, dry basis) of individual and CPM-EOs of C. citratus (CC), E. camaldulensis (EC), E. lehmannii (EL), S. rosmarinus (SR), and T. vulgaris (TV) identified by GC-MS analysis.
In C. citratus EO, five components accounted for 99.98% of the total oil, primarily oxygenated monoterpenes (90.39%) and monoterpene hydrocarbons (9.59%), with geranial (53.11%), neral (29.14%) and β-myrcene (9.59%) as the most abundant compounds. E. camaldulensis EO contained seven constituents (99.92%), dominated by oxygenated monoterpenes (70.79%) and monoterpene hydrocarbons (26.04%), particularly eucalyptol (66.51%) and α-pinene (24.38%). Similarly, E. lehmannii EO was rich in oxygenated monoterpenes (74.45%) and monoterpene hydrocarbons (24.78%), with six major chemicals representing 99.6% of the total composition, including eucalyptol (56.99%) and α-pinene (24.18%). S. rosmarinus EO comprised ten chemical constituents (99.98%), with eucalyptol (46.56%), borneol (11.35%), and α-pinene (9.06%) as predominant. Finally, T. vulgaris EO contained nine components (99.99%), rich in oxygenated monoterpenes (76.56%) and monoterpene hydrocarbons (22.07%), with thymol (70.84%), p-cymene (11.01%), and γ-terpinene (7.73%) as the major compounds.
Binary combinations involving C. citratus resulted in notable shifts in chemical profiles, reflecting both chemical reorganization and selective transformations depending on the companion species. In the C. citratus + E. camaldulensis oil, geranial (33.2%) and neral (17.23%) were the main constituents, indicating partial retention of the citrus profile, whereas eucalyptol, characteristic of E. camaldulensis, was completely absent. Remarkably, p-cymene (19.14%) appeared as a newly formed compound, indicating possible chemical transformation during the distillation process. In the blend with E. lehmannii, eucalyptol became the dominant compound (73.62%), a level exceeding that of E. lehmannii alone (56.99%) suggesting potential chemical interactions between the matrices during distillation, while geranial and neral decreased sharply to 5.11% and 2.69%, respectively. Additionally, α-pinene declined from 24.18% to 16.9%, reflecting rebalancing of the volatile fraction. The C. citratus + S. rosmarinus mixture showed a moderate retention of geranial (21.45%) and neral (9.8%), while camphor increased from 7.44% to 9.39%. In contrast, other major S. rosmarinus volatiles such as eucalyptol, α-pinene, camphene, and β-pinene were reduced to 26.13%, 3.5%, 2.28%, and 2.9%, respectively. In the C. citratus + T. vulgaris blend, thymol became the dominant constituent (45.78%), underscoring the strong phenolic imprint of T. vulgaris, while geranial (24.12%) and neral (12.34%) were well retained. The appearance of isoneral (2.45%) alongside reduced levels of p-cymene (2.92%) and γ-terpinene (3.57%), further highlights the emergence of new volatiles and reorganization of the monoterpene fraction during distillation.
Binary blends containing E. camaldulensis exhibited distinct compositional behaviors, ranging from preservation to transformation of key constituents depending on the combined species. Thus, in the E. camaldulensis + E. lehmannii oil, the major components, eucalyptol (71.27%) and α-pinene (23.39%) were largely retained, indicating compositional stability and chemical compatibility between the two species. Conversely, combination with S. rosmarinus led to a profile still dominated by eucalyptol (64.35%) and α-pinene (9.0%), alongside camphor (4.04%), while camphene (2.95%) and β-pinene (2.78%) were preserved at low levels, reflecting moderate rebalancing without major loss of original markers. In contrast, the blend with T. vulgaris revealed marked compositional shifts: thymol emerged as the dominant compound (55.69%), slightly reduced from its original concentration in T. vulgaris (70.84%). Notably, eucalyptol, originally the primary component of E. camaldulensis (66.51%) was no longer detectable, indicating possible chemical transformation. Monoterpene hydrocarbons, including α-pinene and β-pinene were also drastically reduced from 24.38% and 1.21% to 1.88% and undetectable levels, respectively. Meanwhile, p-cymene and γ-terpinene present in T. vulgaris at 11.01% and 7.73% were enriched to 20.52% and 10.11% respectively. These compositional modifications reflect a clear shift toward the phenolic and aromatic profile of T. vulgaris accompanied by a complete attenuation of the eucalyptol-rich signature characteristic of E. camaldulensis.
Combinations with E. lehmannii resulted in compositional changes influenced by the accompanying species. In the E. lehmannii + S. rosmarinus blend, eucalyptol remained the predominant compound at 49.62%, reflecting substantial retention from both original oils (56.99% and 46.56% respectively). Camphor, a key constituent of S. rosmarinus (7.44%), was slightly reduced to 6.28%, while α-pinene initially abundant in both oils (24.18% and 9.06% respectively) appeared at an intermediate level of 13.77%, indicating a compositional balance between the two sources. In contrast, the combination of E. lehmannii with T. vulgaris yielded an oil dominated by thymol (35.77%) and eucalyptol (35.09%), both substantially reduced from their original concentrations. p-cymene was completely eliminated, α-pinene declined to 11.97%, while γ-terpinene increased slightly from 7.73 to 7.96%, demonstrating a partial merging of the two oils.
Similar compositional patterns were observed in the combination of S. rosmarinus with T. vulgaris, where thymol (33.56%) and eucalyptol (29.02%) emerged as the predominant compounds shaping the blend’s chemical profile. p-cymene was completely eliminated, and α-pinene decreased to 5.09%, reflecting selective changes that enhance the blend’s chemical diversity.
Regarding ternary CPM-EOs, eucalyptol was detected exclusively in C. citratus + T. vulgaris + S. rosmarinus, where it accounted for 10.67%. Thymol, the principal phenolic compound of T. vulgaris, consistently dominated all ternary blends, with concentrations varying from 37.64% to 50.46%. p-cymene, another major compound from T. vulgaris, was present at 16.81% and 3.68% in blends containing E. camaldulensis and E. lehmannii, respectively, but was entirely absent in S. rosmarinus blend. Geranial (15.95–20.9%) and neral (6.43–10.7%) characteristic of C. citratus were well preserved across all blends, contributing to their distinctive citrus aroma. Camphor, a marker compound of S. rosmarinus, was detected only in its corresponding ternary blend at 3.99%. These compositional variations illustrate the complex interactions occurring in ternary CPM-EOs, where the phenolic dominance of thymol is balanced by the citrus freshness of geranial and neral, while other compounds such as eucalyptol, p-cymene, and camphor modulate the aroma profile and potentially the biological activity of the oils.
2.2. Multivariate Analysis of Essential Oil Compositions
2.2.1. Principal Component Analysis (PCA)
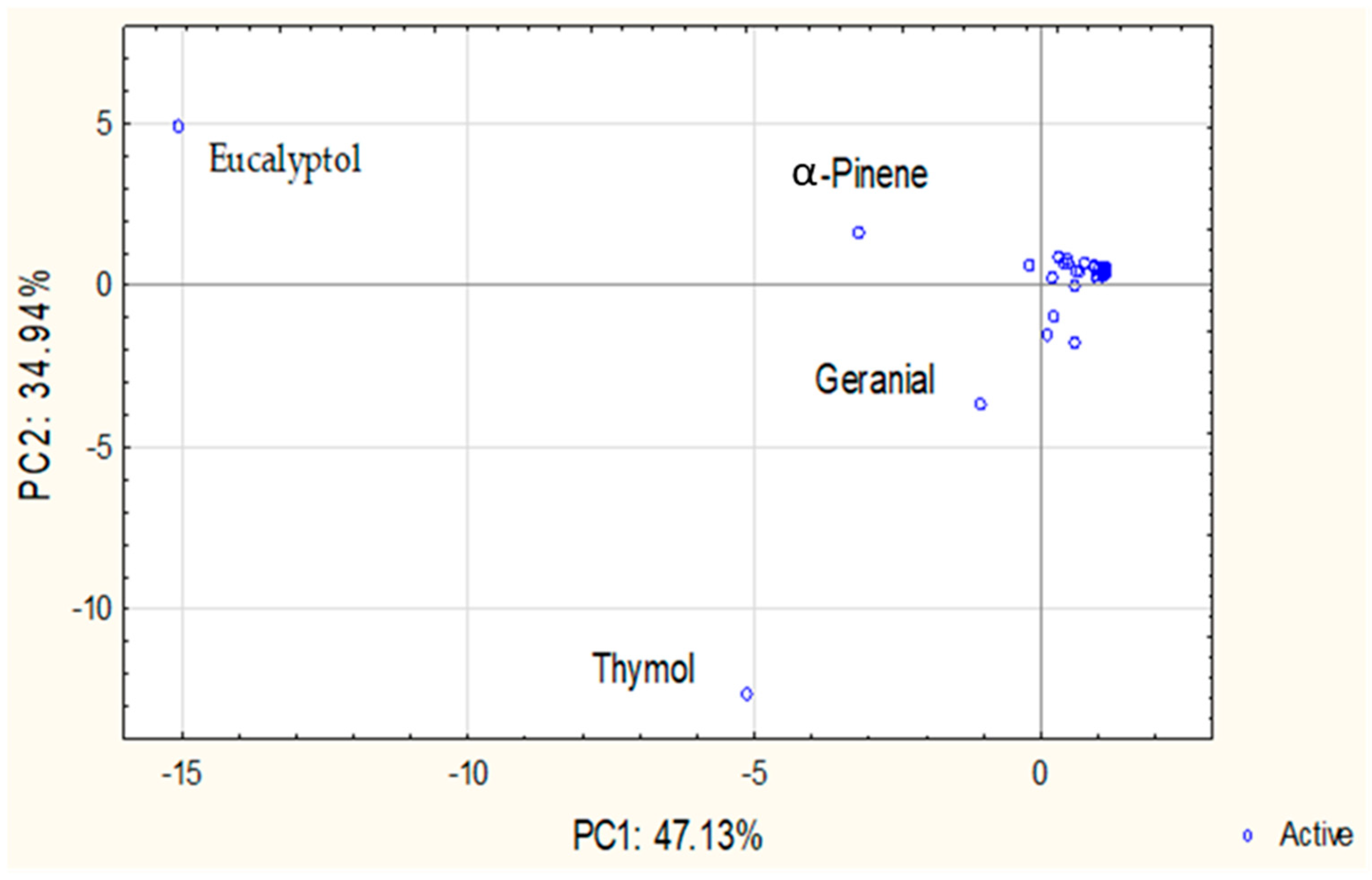
PCA was performed to explore patterns in the chemical composition of individual and CPM-EOs. The scree plot indicated that the first three principal components account for approximately 95.83% of the total variance, with PC1 explaining 47.13%, PC2 explaining 34.94%, and PC3 contributing an additional 13.76%. Given the sharp decline in eigenvalues after the third component, only PC1 and PC2, together explaining 82.07% of the variance, were considered for graphical representation.
The PCA score plot (Figure 1) revealed distinct groupings driven by the distribution of key chemical constituents in individual and CPM-EOs. Detailed PCA loadings (PC1, PC2 and PC3) for all individual, binary, and ternary CPM-EOs are provided in Supplementary Table S1. E. camaldulensis and E. lehmannii exhibited strong negative loadings on PC1 (−0.91 and −0.89, respectively), primarily influenced by eucalyptol, a dominant compound strongly correlated with this axis. Similarly, S. rosmarinus also loads strongly negatively on PC1 (−0.87) but displays a moderate positive loading on PC2 (0.40), suggesting some chemical similarity with the Eucalyptus species while being differentiated by compounds such as camphor, which contributes to its unique profile. T. vulgaris showed a strong negative loading on PC2 (−0.88), primarily associated with thymol, underscoring its distinct chemical signature. In contrast, C. citratus exhibited moderate loadings near the origin for both PC1 (−0.06) and PC2 (−0.28), indicating a balanced distribution across these components. However, it was primarily characterized by its high loading on PC3 (0.94), driven by its high citral content, which strongly influences this component and distinguishes this species’ chemical profile, reflecting variation that is not fully represented in the PC1/PC2 plot.
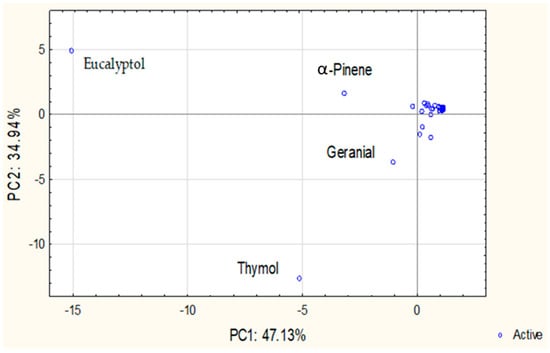
Figure 1.
Principal component analysis (PCA) of EOs composition: Projection on PC1 and PC2.
The binary and ternary CPM-EOs exhibited clear spatial distributions in the PCA space, shaped by the dominant constituents of their parent oils. Binary combinations involving Eucalyptus species, including pairings within the genus and with other plants such as C. citratus + E. lehmannii and E. camaldulensis + S. rosmarinus, showed strong negative loadings on PC1, reflecting high levels of eucalyptol and α-pinene. Mixtures involving T. vulgaris, such as C. citratus + T. vulgaris and E. camaldulensis + T. vulgaris were positioned along PC2 due to thymol content. Combinations with C. citratus, especially C. citratus + E. camaldulensis and C. citratus + S. rosmarinus, exhibited strong positive loadings on PC3, indicating a distinct geranial-driven profile. Ternary mixtures, such as C. citratus + T. vulgaris + E. camaldulensis and C. citratus + T. vulgaris + E. lehmannii, loaded strongly on PC2 (thymol-driven), with moderate PC3 contributions reflecting geranial influence. Similarly, C. citratus + T. vulgaris + S. rosmarinus showed strong negative loadings on both PC1 and PC2, indicating a combined influence of eucalyptol, α-pinene, and thymol, with minor geranial input. These spatial patterns illustrate the compositional interplay and synergistic effects emerging from plant material combinations.
2.2.2. Hierarchical Cluster Analysis (HCA)
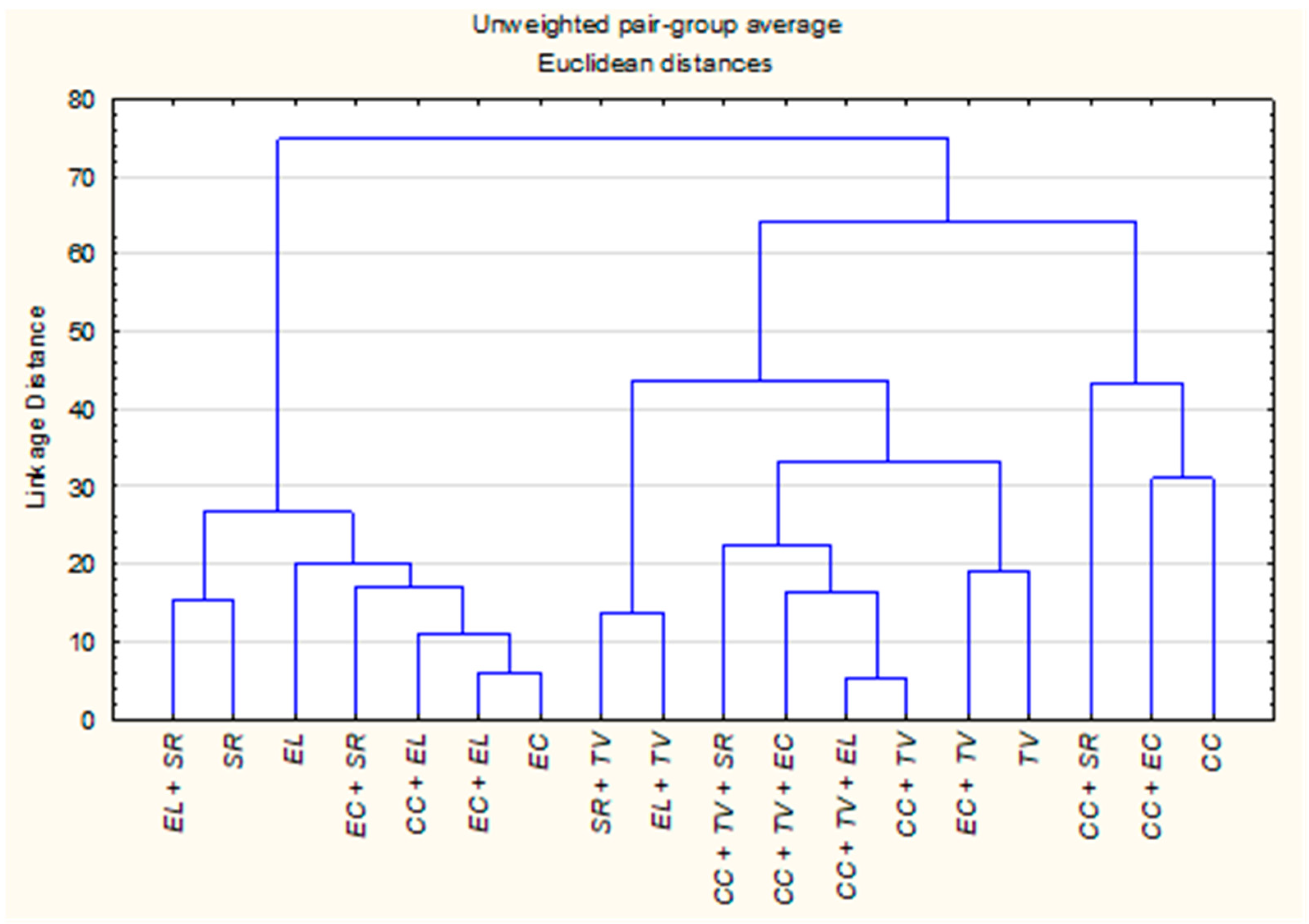
The HCA dendrogram, constructed using Euclidean distances and the UPGMA method, revealed clear groupings among the EOs and their combinations, in agreement with the patterns observed in the PCA. Three primary clusters emerged based on compositional similarity (Figure 2).
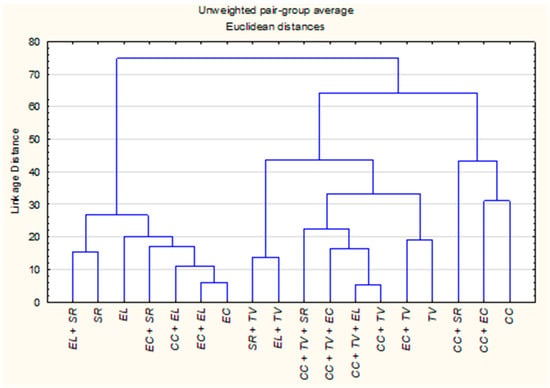
Figure 2.
Hierarchical clustering dendrogram of individual and CPM-EOs of C. citratus (CC), E. camaldulensis (EC), E. lehmannii (EL), S. rosmarinus (SR), and T. vulgaris (TV) based on their chemical composition. Clustering was performed using Euclidean distances and the UPGMA method.
Cluster A consisted of Eucalyptus species, S. rosmarinus and their mixtures. The close proximity between E. camaldulensis and E. lehmannii (distance = 16) reflects a high degree of shared constituents, further emphasized by the even closer distance between E. camaldulensis and its blend with E. lehmannii (distance = 6). Additionally, E. camaldulensis + S. rosmarinus (distance = 18 from E. camaldulensis and 22.3 from E. lehmannii) supports a trend of compositional alignment within this cluster, likely driven by common oxygenated monoterpenes such as eucalyptol and α-pinene. Cluster B was dominated by T. vulgaris and its mixtures, characterized by high phenolic content. While T. vulgaris alone was chemically distinct from E. camaldulensis (distance = 101), their combination showed a greatly reduced distance of 19, indicating a closer chemical relationship. Mixtures involving C. citratus and T. vulgaris also exhibited strong chemical affinity. For instance, C. citratus + T. vulgaris and C. citratus + T. vulgaris + E. lehmannii displayed a very low inter-sample distance of 5.4, while C. citratus + T. vulgaris + E. camaldulensis showed a slightly higher but still close distance of 17.9. These shifts suggest enhanced chemical integration, likely driven by synergistic interactions between thymol and geranial components. The mixture E. lehmannii + T. vulgaris (distance = 46) also supports the potential for moderate compositional blending between phenolic-rich and monoterpene-based oils. Cluster C encompassed C. citratus and its combined derivatives. Although C. citratus alone was chemically distant from most other oils (distance > 90 from E. camaldulensis and T. vulgaris), its mixtures consistently reduced inter-sample distances and frequently occupied intermediate positions between Clusters A and B. Notable examples include C. citratus + E. camaldulensis (distance = 31 from C. citratus), C. citratus + S. rosmarinus (48.1) all suggesting partial compositional blending. These results indicate that combining plant material modifies the volatile profile in a manner that reduces chemical distances between initially dissimilar oils.
2.3. Insecticidal Activity
2.3.1. Probit Analysis of Individual and Combined Plant Material Essential Oils
Probit regression analysis revealed notable variation in the insecticidal efficacy of both individual and CPM-EOs against A. fabae after 24 h of exposure. Among the individual oils tested, E. camaldulensis demonstrated the highest insecticidal potency, with the lowest LC50 value of 2.45 µL mL−1, indicating its efficiency was obtained with the smallest concentration to kill 50% of the aphid population. This was followed by E. lehmannii with LC50 of 2.90 µL mL−1, C. citratus at 3.24 µL mL−1, and T. vulgaris at 3.71 µL mL−1. The least effective individual oil was S. rosmarinus, which exhibited the highest LC50 value of 4.41 µL mL−1, reflecting lower toxicity (Table 2).

Table 2.
Probit regression results of individual and CPM-EOs against A. fabae (24 h post-treatment).
When evaluating CPM-EOs, binary mixtures generally lead to enhanced insecticidal activity compared to individual ones. The binary combination of E. camaldulensis + T. vulgaris was the most potent, achieving an LC50 of 1.39 µL mL−1, indicating good synergy between components inducing a strong increase in toxicity. Other binary mixtures involving C. citratus also showed substantial improvements, with LC50 values ranging from 1.75 to 2.38 µL mL−1, demonstrating a relative synergistic or additive effect. Ternary combinations resulted in complete mortality (100%) at the lowest tested concentration of 2 µL mL−1. While this precluded a fitting dose–response curve and calculating LC50 values, these results indicate potential synergistic effects among the three oils. However, further testing at lower concentrations would be required to accurately quantify the degree of synergy.
2.3.2. Assessment of Synergistic Interactions Using Co-Toxicity Coefficient and Synergistic Factors
The evaluation of CPM-EOs against A. fabae revealed a spectrum of interaction types, ranging from strong synergism to additive or slight antagonism. C. citratus stood out as a key synergistic component. Its binary combinations with E. camaldulensis, E. lehmannii, S. rosmarinus, and T. vulgaris consistently exhibited strong synergistic effects, with co-toxicity coefficients (CTC) ranging from 130.64 to 198.57 and synergistic factors (SF) exceeding 1.2 in all cases (Table 3).

Table 3.
Lethal concentration value (LC50), co-toxicity coefficient (CTC) and synergistic factors (SF) of CPM-EOs against A. fabae (24 h post-treatment).
T. vulgaris also contributed significantly to enhanced efficacy when paired with other species. Combinations with E. camaldulensis, E. lehmannii, and S. rosmarinus demonstrated particularly strong synergy, reflected by CTC values greater than 218 and SF values above 2, indicating substantial improvement in insecticidal activity. S. rosmarinus, despite being the least toxic as a single oil, displayed notable synergistic effects in combinations. When paired with C. citratus or T. vulgaris, the mixtures achieved high CTC values (up to 249.08) and strong SF values (>2.2), underscoring its supportive role in enhancing toxicity. However, in combination with E. camaldulensis, the interaction was weaker, classified as additive to moderate synergism (CTC = 119.52), and showed uneven SF values (0.85 and 1.54), suggesting variable contribution depending on the pairing. In contrast, the mixture of E. camaldulensis and E. lehmannii showed the weakest interaction, classified as additive to slight antagonism. This combination yielded a CTC of 81.58 and SF values of 0.75 and 0.88, indicating little to no enhancement and potential interference in insecticidal action.
Ternary mixtures comprising C. citratus, T. vulgaris, and either S. rosmarinus or, an Eucalyptus species caused complete mortality at the lowest tested dose (2 µL mL−1). This prevented LC50 estimation but clearly indicated highly effective interactions. In summary, C. citratus and T. vulgaris were the primary drivers of synergy across combinations, while S. rosmarinus enhanced their effects significantly. Conversely, pairings involving only Eucalyptus species provided limited or even slightly antagonistic results, emphasizing the importance of strategic plant material selection for optimal insecticidal efficacy.
2.3.3. Correlation and Regression Analysis Based on LC50
To identify the chemical constituents most strongly associated with insecticidal activity, correlation and regression analyses were performed using LC50 values as indicators of toxicity. Pearson correlation analysis provided insight into the strength and direction of linear relationships between key constituents and bioactivity. To further explore these associations in a multivariate context and account for potential interactions between compounds, PLS regression was applied. This approach allowed the development of a predictive model and highlighted the key constituents contributing to the overall insecticidal effect of EOs.
Pearson Correlation Between Major Constituents and LC50 Values
Among the 35 EOs constituents analyzed, three exhibited statistically significant correlations with LC50 values. Thymol (r = −0.5018, p = 0.034) and Acorenone B (r = −0.5119, p = 0.030) were negatively correlated, indicating that higher concentrations of these compounds corresponded to lower LC50 values and therefore greater insecticidal activity. Caryophyllene was positively correlated with LC50 (r = 0.5267, p = 0.025), suggesting that higher concentrations of this compound were associated with higher LC50 values and, therefore, reduced insecticidal potency. No other constituent of the EOs showed a statistically significant correlation with LC50 values. The complete list of correlation coefficients and p values for all analyzed compounds is provided in Table 4.

Table 4.
Pearson correlation coefficients (r) and p-values between EOs compounds and LC50 values.
Partial Least Square Regression Modeling
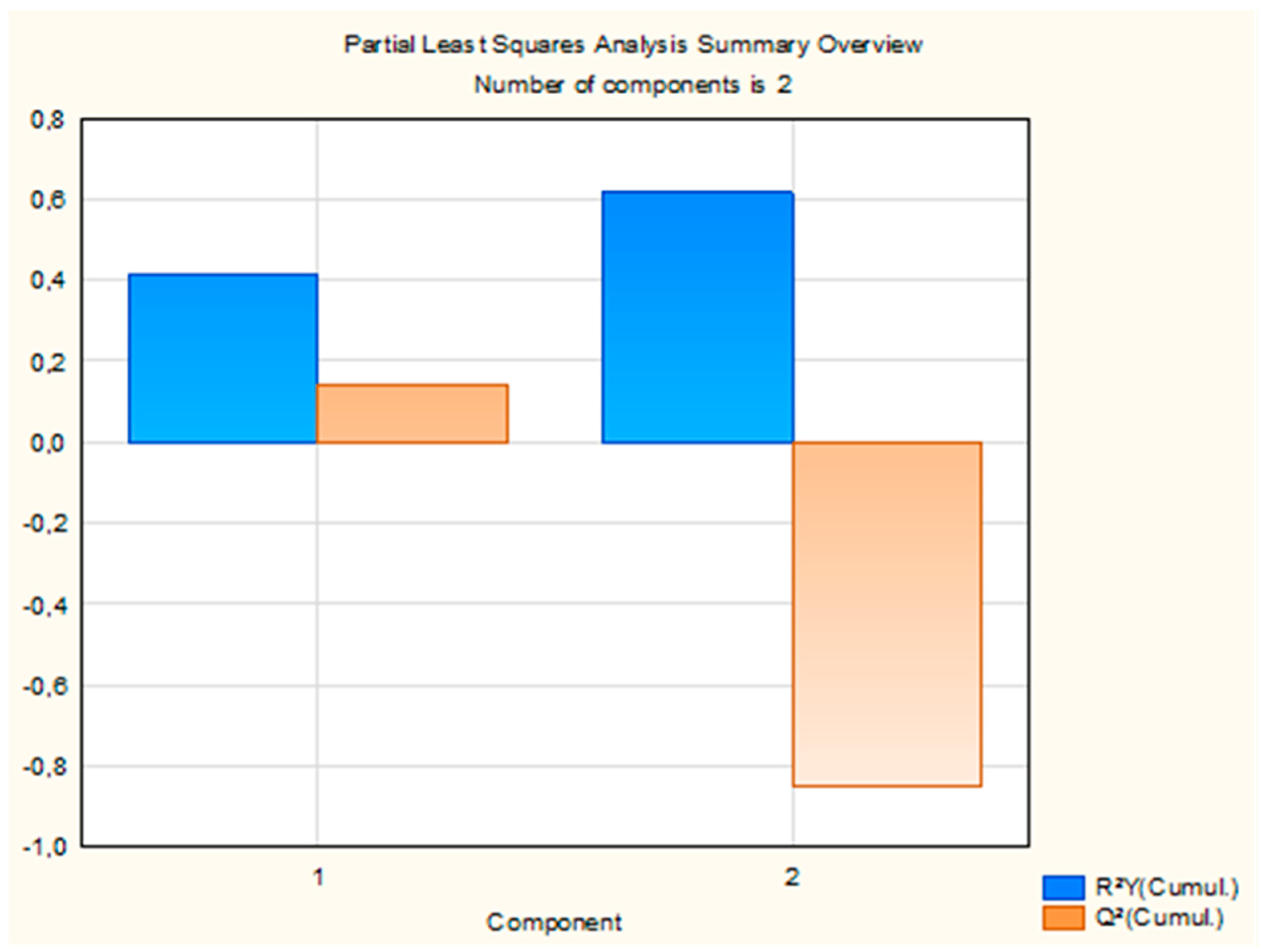
The PLS regression model extracted two components, which together explained 61.57% of the variance in insecticidal activity (LC50). Detailed statistics for each component, including R2X, R2Y, eigenvalues, Q2, and the number of iterations, are presented in Supplementary Table S2. The first component explained 21.86% of the variance in the predictor variables (R2X) and 41.62% of the variance in the response variable (R2Y), with an eigenvalue of 6.73. The associated Q2 value was 0.14, indicating moderate predictive accuracy. This component was statistically significant. The second component contributed 10.61% to R2X, bringing the cumulative R2X to 32.47% and R2Y to 61.57%. Its eigenvalue was 1.91, but the Q2 value was negative (−1.15) and the component was not statistically significant. Although the cumulative R2Y increased with the addition of the second component, the corresponding negative Q2 value indicates limited predictive contribution beyond the first component, which therefore dominates the predictive performance of the model. The overall performance of the model is summarized in Figure 3.
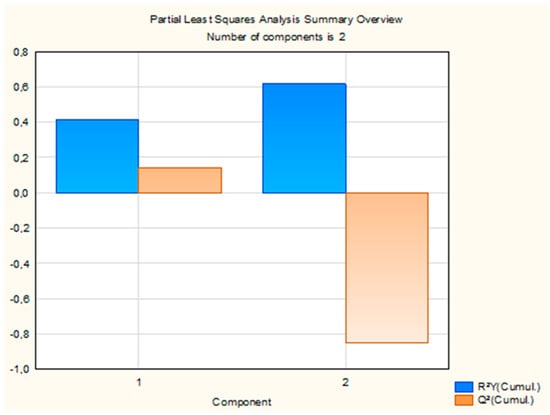
Figure 3.
Cumulative R2Y and Q2 statistics of the two PLS components explaining variance in LC50.
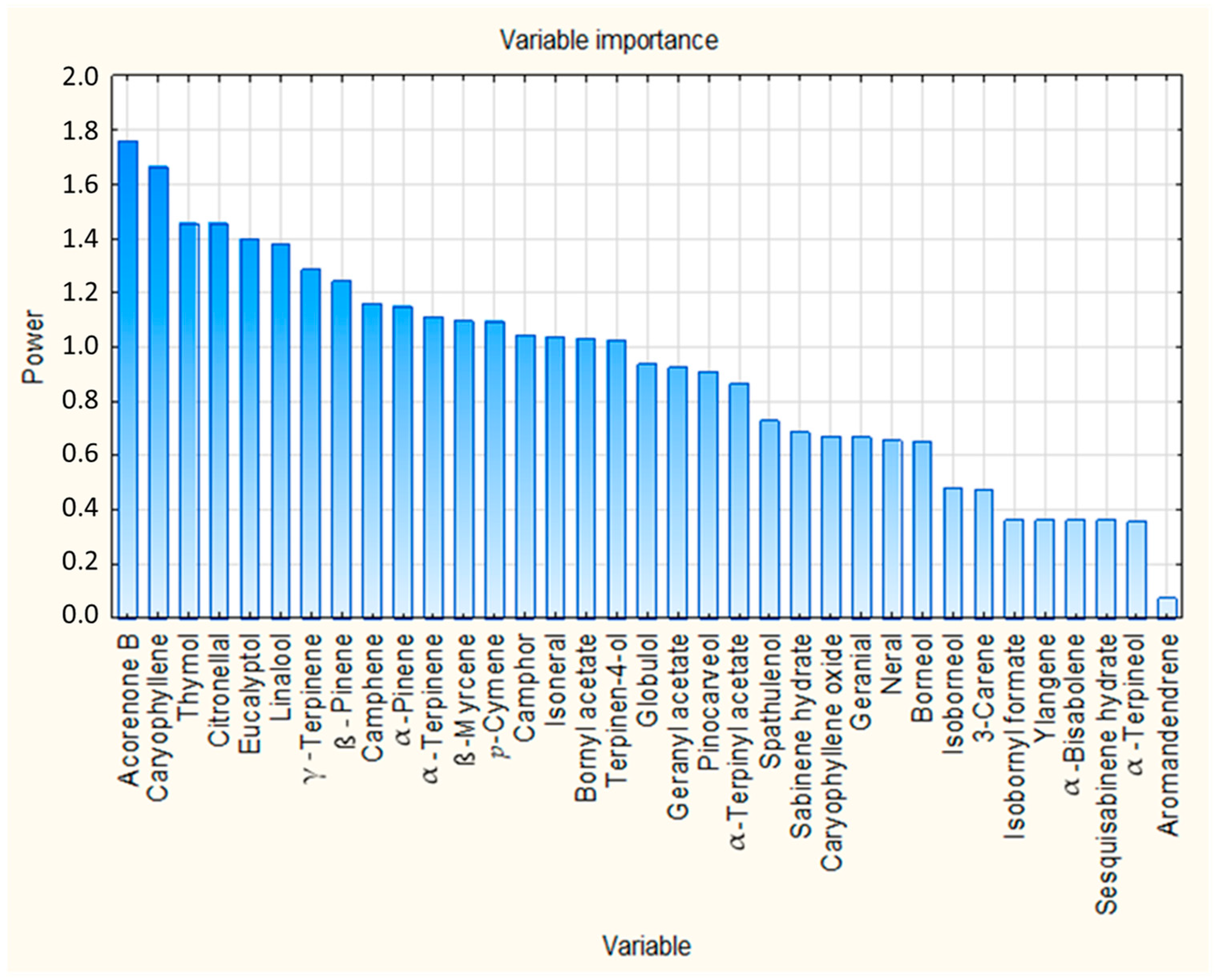
The VIP scores from the PLS regression model were used to assess the relative importance of the 35 identified compounds in predicting insecticidal activity. VIP scores quantify the contribution of each chemical constituent used as a predictor variable to explain variation in the response variable, and compounds with VIP scores greater than 1 are considered contributors to the model. As shown in Figure 4, 17 compounds exceeded the commonly used threshold of VIP > 1, indicating relatively high contribution to the model. These 17 compounds are summarized in Table 5, ranked according to their VIP scores. The complete list of compounds and their associated VIP scores are presented in Supplementary Table S3.
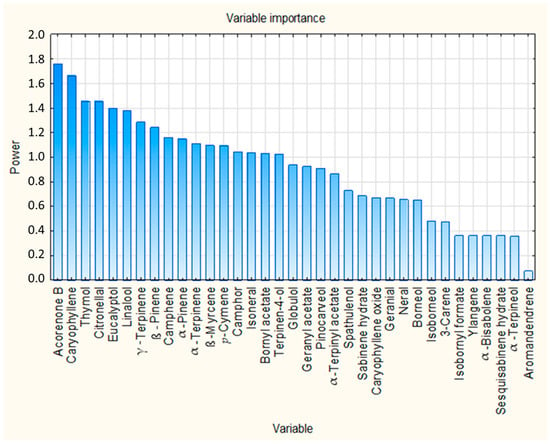
Figure 4.
VIP scores of all identified chemical compounds from PLS regression analysis.

Table 5.
Compounds with VIP scores greater than 1 in the PLS regression analysis.
Acorenone B (VIP = 1.762), caryophyllene (VIP = 1.663) and thymol (VIP = 1.457) were identified as the compounds with the highest VIP scores, indicating their relatively high influence on the PLS regression model. It is noteworthy that these same compounds also showed statistically significant correlations with insecticidal activity in the Pearson correlation analysis presented previously. The convergence of the results of these two independent statistical approaches reinforces their biological relevance and underlines their role as contributors to the observed insecticidal effect. Besides these compounds, several other compounds with VIP greater than 1, such as citronellal, eucalyptol, linalool, and γ-terpinene, also showed notable importance in the model and could contribute through additive or synergistic interactions. This observation supports the notion that insecticidal activity arises from the collective contribution of multiple constituents, rather than being solely driven by a single dominant compound.
3. Discussion
The chemical composition of EOs is a primary determinant of their biological activity, and in this study, the profiles of the tested EOs largely conformed to established chemotypes. Citral is the primary component of EOs derived from Cymbopogon species, with the present study showing high levels of geranial (53.11%) and neral (29.14%), closely aligning with Brasilian (geranial 55.48%, neral 35.40%) and Egyptian (geranial 31.57%, neral 13.42%) chemotypes [25,26]. Similar citral-dominant profiles have also been reported in Indonesian [27], Indian [28], and Vietnamese [29] C. citratus oils, confirming the consistency of this chemotype across regions. E. camaldulensis and E. lehmannii were both dominated by eucalyptol, consistent with previous findings, where eucalyptol typically ranges from 20 to 40% [30,31]. However, eucalyptol was more abundant in our samples (50–70%). Variability in chemical composition of Eucalpytus EOs can arise from geographic origin, climate, soil conditions, harvesting time, and extraction method [32,33]. S. rosmarinus EO was mainly composed of eucalyptol (46.56%) indicating a predominance of eucalyptol chemotype [34,35,36], though other chemotypes such as camphor [37], verbenone [38], α-pinene [39] and myrcene [40] have been described. T. vulgaris EO was characterized by a predominance of thymol, consistent with the literature [41,42], but with a higher proportion of thymol (reaching 70%) than previously reported (close to 40%) [43,44,45]. Additional chemotypes for T. vulgaris EO were reported including carvacrol [46], linalool [47] and α-terpineol [48].
Upon combining and distilling plant materials, the resulting EOs showed strong compositional inheritance from their respective parent profiles, as indicated by the clustering of major components. However, notable differences were observed, including suppression of certain constituents, emergence of new compounds, and significant shifts in relative abundance. In particular, combinations involving C. citratus with Eucalyptus species showed distinct profiles, likely due to the interaction of geranial and eucalyptol, whereas blends with T. vulgaris were dominated by thymol, reflecting its strong phenolic character. Chemically similar oils, such as E. camaldulensis, E. lehmannii and S. rosmarinus, showed limited differentiation, suggesting lower synergistic effects. These observations highlight the importance of strategic EO selection in combined distillation to achieve both compositional and functional diversity. Similar transformations were previously observed during the combined plant material distillation of C. citratus and Hyptis suaveolens, where new major constituents (piperitone (40.8%) and p-menth-4(8)-ene (13.2%)) appeared, while β-pinene, sabinene, neral and geranial disappeared, and limonene and α-pinene increased. These mechanisms are inferred from analytical evidence and literature precedent and were attributed to thermal and acid-catalyzed reactions, including cyclization, isomerization, and dehydration, leading to the conversion of citral isomers (neral and geranial) to piperitone, rearrangement of sabinene to p-menth-4(8)-ene and the transformation of myrcene and β-pinene into limonene [2]. Additionally, Sánchez-Velandia et al. [49] reported that in situ chemical reactions can occur during distillation, for example, α-pinene undergoes acid-catalyzed rearrangement to camphene, which is further oxidized to camphor, while myrcene, formed via β-pinene pyrolysis, can be converted to d-citronellal and ultimately cyclized to produce l-menthol. Consistent with these observations, in the present study, these compounds were not detected in the individual plant distillates processed under the same conditions, suggesting that contamination is unlikely. These transformations demonstrate how naturally occurring compounds can undergo significant structural changes during extraction and processing [50]. These reactions can generate new compounds not originally present in the biomass, influencing both chemical complexity and biological activity, with implications for therapeutic, antimicrobial, or preservative effects [51,52]. While these findings strongly suggest these reaction pathways, targeted experimental studies would be required to definitively confirm them.
These compositional changes arise from multiple chemical and physical factors inherent to the distillation process [53,54]. Hydrodistillation exposes EO constituents to elevated temperatures, prolonged contact with water, and oxygen, promoting chemical transformations [55,56]. Thermal degradation particularly affects heat-sensitive compounds like monoterpenes and sesquiterpenes, which can volatilize, decompose, or rearrange [57,58]. Hydrolysis can cleave esters and glycosides, generating alcohols and acids, with different volatilities and polarities, affecting extraction efficiency and oil composition [59,60]. Oxidative reactions, driven by dissolved oxygen or trace metals, can transform reactive aldehydes, phenolics, or unsaturated compounds into oxidized derivatives, that are not present in the raw plant material [61,62,63]. Physical factors such as differences in vapor pressure cause more volatile compounds to distill preferentially, potentially suppressing less volatile components or causing uneven release [64]. Moreover, the extraction procedure can influence physical properties such as viscosity, coloring, and odor, which may affect EOs quality [65]. When different plant matrices are combined, their anatomical and microstructural differences, such as trichome density, oil gland structure, and moisture content, can influence the release kinetics of EO constituents, leading to suppression or enhancement of certain compounds [66].
The insecticidal assay against A. fabae revealed variable toxicity levels among the tested oils. Among the individual ones, E. camaldulensis exhibited the highest toxicity, followed by E. lehmannii consistent with literature attributing strong insecticidal activity to eucalyptol-rich oils [67]. For instance, Khedhri et al. [68] demonstrated potent toxicity of four Eucalyptus species against A. fabae with LC50 ranging from 0.264 to 0.39 mg mL−1. Differences in LC50 units (mg mL−1 in Khedhri et al. vs. µL mL−1 in the present study) and variations in EO density should be taken into account when interpreting this comparison. C. citratus EO also showed considerable activity, likely due to its high content in geranial and neral, which have been reported to be strong insecticidal and repellent compounds against A. fabae [69]. Comparable efficacy was observed against related species including A. gossypii, A. citricola and M. persicae, further supporting the broad-spectrum activity of these oxygenated monoterpenes against the Aphididae family [69,70,71,72]. Similarly, T. vulgaris EO exhibited notable insecticidal activity which is attributed to its high thymol content, a monoterpene phenol with well-documented neurotoxic effects on insects [73]. In aphid species, several mechanisms have been described for thymol, including modulation of GABA-gated chloride channels and interference with the tyramine–octopamine signaling pathway, both of which are crucial for neural transmission and behavioral regulation in insects [74]. In the present study, S. rosmarinus EO exhibited the lowest activity, with LC50 value of 4.41 µL mL−1. These results are consistent with those obtained by Casas et al. [75] who reported a similar LC50 value (4.61 µL mL−1) against Myzus persicae.
When EOs were combined, they generally exhibited enhanced toxicity when compared to individual oils. For instance, E. camaldulensis + T. vulgaris mixture demonstrated the lowest LC50 value of 1.39 µL mL−1. The CTC and SF analyses confirmed strong synergy in combinations involving C. citratus, T. vulgaris, and S. rosmarinus. Statistical analyses identified acorenone B, thymol and caryophyllene as compounds with relatively high contributions to the insecticidal activity. In agreement, acorenone B has been reported to inhibit both acetylcholinesterase and butyrylcholinesterase, with IC50 values of 40.8 µg mL−1 and 10.9 µg mL−1 respectively, indicating a neuroinhibitory mode of action that disrupts cholinergic neurotransmission and leads to paralysis and death in insects. Consistently, acorenone-rich EOs exhibited high ecotoxicity, supporting the contribution of acorenone-type sesquiterpenes to EO-mediated toxicity [76]. These results are in line with the findings of Bora et al. [77], who reported that multi-component EO blends with different mechanism of action enhance pest control efficiency and delay resistance development. In line with these observations, a study on Sitophilus zeamais demonstrated that optimized mixtures of plant volatile compounds, including thymol, carvacrol, phellandrene, γ-terpinene, pulegone, and δ-3-carene, exhibit synergistic insecticidal activity through multitarget effects, impacting neurotransmission and reducing detoxification enzyme activities [78]. Similar synergism among the major constituents (Eucalyptol, carvacrol, pulegone, and eugenol) of Rosmarinus officinalis, Origanum compactum, Mentha pulegium, Thymus satureioides, Myrtus communis and Eugenia aromatica against Callosobruchus maculatus, has been shown to improve stability, absorption, cuticular penetration and multi-target neurophysiological effects [79]. Binary mixtures of thymol and eucalyptol demonstrated synergistic toxicity against Helicoverpa armigera, inhibiting detoxification and neurophysiological enzymes more effectively than single compounds [80]. Modeling-based research on Musca domestica demonstrated that the insecticidal activity of thyme EOs is primarily attributed to thymol, p-cymene and γ-terpinene, identified as the main active constituents through component effect analysis. Mixture modeling confirmed synergistic interactions among all binary combinations of these compounds, with the optimal ternary ratio (p-cymene: γ-terpinene: thymol = 32:23:45) achieving the highest mortality rate of 87.5%. This optimized blend significantly outperformed individual components, underscoring the crucial role of compositional optimization and synergistic interactions among terpenoid constituents in enhancing the efficacy and consistency of EO-based insecticides [81]. Several reports have highlighted the insecticidal potential of caryophyllene [82,83,84,85]. For example, Sun et al. [86] demonstrated that caryophyllene, α-pinene and β-myrcene act as major bioactive components responsible for the insecticidal and repellent activity of Peucedanum terebinthinaceum EO. Additionally, Chohan et al. [87] reported that β-caryophyllene and α-pinene exhibit strong fumigant toxicity against Myzus persicae, significantly affecting genes responsible for reproduction, dispersion, and insect growth. Moreover, β-caryophyllene-rich leaf EO of Psidium guajava showed strong contact toxicity against Sitophilus zeamais, highlighting the eco-friendly potential of this compound as natural insecticide and control agent [88].
4. Materials and Methods
4.1. Plant Material
During the spring of 2025 (March–April), five plant species (C. citratus, E. camaldulensis, E. lehmannii, S. rosmarinus and T. vulgaris) were collected from various regions across Tunisia. Leaves were obtained from C. citratus, E. camaldulensis and E. lehmannii, while the aerial parts were gathered for S. rosmarinus and T. vulgaris. C. citratus was cultivated, and the other species were collected from the wild. To ensure representative sampling, plant materials were randomly harvested from several individual plants or trees within each species. The samples were then combined to form homogenized samples, as summarized in Table 6. After collection, the plant materials were placed in a glass greenhouse and air-dried under ambient conditions for five days. Once dried, they were stored in paper bags at room temperature until further analysis. All plant species were taxonomically identified by Professor Lamia Hamrouni following standard procedures. Corresponding voucher specimens have been deposited in the herbarium division of the National Institute of Research on Rural Engineering, Water and Forests (INRGREF).

Table 6.
Plant species, used part, period and harvesting sites.
4.2. Essential Oil Extraction and Combinations
EOs were extracted by hydro-distillation of 50 g of dried plant material using a clevenger-type apparatus (SAF Wärmetechnik LabHEAT® KM-ME, 1000 mL, SAF GmbH, Hamm, Germany) with 500 mL distilled water at a plant material-to-water ratio of 10:1 (v/w) for 3 h [89]. Extractions were performed on individual species (C. citratus, E. camaldulensis, E. lehmannii, S. rosmarinus, and T. vulgaris) as well as on binary (1:1) and ternary (1:1:1) mixtures, prepared by mixing the dried materials in equal weight ratios prior to distillation [9]. Extractions were performed in three replicates. The extracted oils were dried over anhydrous sodium sulfate and stored at 4 °C in amber glass vials until further use. The EO yield was calculated as % (w/w) relative to the dry weight of plant material and is reported in Table 1. The overall experimental design is shown in Supplementary Figure S1.
4.3. Gas Chromatography Mass Spectrometric (GC-MS) Analysis
The GC-MS analysis was conducted using an HP 8890 gas chromatograph coupled to an HP 5977B mass spectrometer equipped with an HP-5MS UI column (30 m × 0.25 mm; 0.25 µm) (Agilent Technologies, Santa Clara, CA, USA). The injector and the detector temperatures were set at 250 °C and 280 °C, respectively. The oven temperature was programmed from 45 °C (held 1 min) to 250 °C (held 6 min) at a rate of 5 °C/min, for a total runtime of 48 min. Helium was used as the carrier gas at a constant flow rate of 1 mL min−1. Samples (1 µL) were injected in split mode with a 1:100 ratio. Mass spectrometry data were acquired in scan mode over a mass range of m/z 30 to 600. Components were identified by comparing their calculated retention indices (RI calc.), determined relative to a homologous n-alkane series, with literature retention indices (RI lit.), and by comparison of their mass spectra with those present in commercial libraries (Wiley 7, NIST 05) and/or reported in the literature [51].
4.4. Evaluation of Insecticidal Activity
4.4.1. Aphid Sampling and Rearing
Pathogen-free apterous parthenogenetic populations of A. fabae were collected from faba bean (Vicia faba) crops in the Cap Bon region (36.69° N, 10.49° E). Colonies were maintained on potted faba bean plants in a controlled-environment growth chamber at 23 °C ± 1, a relative humidity 60 ± 5%, and a 16:8 h light:dark photoperiod [67]. Aphids and host plants were kept in insect-proof cages equipped with fine mesh vents to ensure proper ventilation and prevent contamination.
4.4.2. Contact Toxicity Bioassay
The insecticidal activity of individual and CPM-EOs was evaluated against A. fabae using a direct contact toxicity test. The assays were conducted using concentrations of 2, 4, 6, 8, 10, and 12 μL mL−1 [90,91]. For each treatment, an emulsion was prepared by mixing the appropriate volume of EO with 1% (v/v) Tween 20 solution (Sigma-Aldrich, St. Louis, USA, P1379). Ten wingless aphids of similar size and developmental stage were gently transferred onto a fresh faba bean leaf placed on Whatman filter paper disks inside a sterile 90 mm Petri dish. All individuals were adult females, consistent with the parthenogenetic biology of A. fabae. Each group was then sprayed with 1 mL of the prepared EO emulsion, ensuring even coverage of both aphids and the leaf surface. Control groups were treated with 1 mL of 1% Tween 20 solution without EOs. No mortality was observed in the control, so Abbott’s correction was not required before estimating LC50 values. All treatments, including the control, were replicated five times. Mortality was assessed 24 h post-application. Aphids were considered dead if they showed no movement of legs or antennae upon gentle probing with a fine brush. Lethal concentration value (LC50) with 95% confidence interval was estimated using PROBIT analysis.
4.4.3. Interaction Assessment
The co-toxicity coefficient (CTC) was calculated to evaluate the interaction effects of CPM-EOs on A. fabae toxicity. For each mixture, the expected LC50 was calculated as the arithmetic mean of the LC50 values of the individual EOs. The CTC was then determined by dividing the expected LC50 by the observed LC50 of the mixture and multiplying by 100. A CTC value greater than 120 was considered indicative of strong synergism, values between 80 and 120 indicated additive (cumulative) effects, and values less than 80 suggested antagonism [92]. Additionally, the synergistic factor (SF) was calculated to assess further the interaction between EOs mixtures [93]. The SF was determined separately for each component of the mixture by dividing the LC50 of the individual EO by the LC50 of the corresponding mixture. Specifically, SF vs. A = LC50 of EO A/LC50 of the mixture, and SF vs. B = LC50 of EO B/LC50 of the mixture. An SF value greater than 1 indicated synergism, a value equal to 1 indicated an additive effect, and a value less than 1 suggested antagonism.
For interaction classification, CTC was used as the primary indicator of overall interaction, while SF values were used to refine the interpretation. When SF values were mixed (one SF > 1 and one < 1) and the CTC fell within the additive range (80–120), interactions were classified based on the position of the CTC within this range: values closer to 120, together with at least one SF > 1, were interpreted as additive to moderate synergism, whereas values closer to 80, together with SF values < 1, were interpreted as additive/slight antagonism. Accordingly, CTC and SF were interpreted jointly to ensure a consistent and transparent classification of interaction effects, particularly in borderline cases.
4.5. Statistical and Chemometric Analysis
All analyses were performed using STATISTICA software version 10 (StatSoft, Tulsa, OK, USA). EO yields were determined in three replicates and expressed as mean ± standard deviation. One-way ANOVA followed by Tukey’s HSD test (p < 0.05) was used to compare yields. Insecticidal bioassays were conducted in five replicates, and Probit analysis was performed to estimate lethal concentration (LC50) with 95% confidence interval. Principal Component Analysis (PCA) and Hierarchical Cluster Analysis (HCA) were conducted to examine patterns in the chemical composition of individual and CPM-EOs. HCA was based on Euclidean distance and clustering was performed using the Unweighted Pair Group Method with Arithmetic Mean (UPGMA). To study the relationship between EO composition and insecticidal activity, chemometric approaches were used. Pearson correlation analysis identified significant linear associations between the abundance of individual compounds and their insecticidal effects based on LC50 values. In addition, PLS regression was used to model the relationship between the chemical profiles (X variables) and insecticidal activity (Y variable). Prior to analysis, X variables were scaled to unit standard deviation, and the optimal number of components was determined by cross-validation to minimize prediction error and avoid overfitting. The VIP (Variable Importance in Projection) scores of the PLS model highlighted compounds with relatively high contributions to the activity, acknowledging that the model’s predictive power was modest. These multivariate chemometric analyses provided a better understanding of the associations between the chemical profiles of EOs and their insecticidal efficacy.
5. Conclusions
The combination of plant material facilitates effective compositional blending, as evidenced by multivariate analyses that revealed distinct chemical profiles predominantly influenced by key constituents such as eucalyptol, thymol, and geranial. These compositional alterations were observed in the insecticidal bioassay against A. fabae. Among the individual EOs tested, E. camaldulensis exhibited the highest toxicity. Notably, combinations involving C. citratus, Eucalyptus species and T. vulgaris demonstrated pronounced synergistic effects, significantly increasing toxicity and reducing LC50 values, with the E. camaldulensis + T. vulgaris mixture being the most effective. Althought, S. rosmarinus displayed the lowest individual toxicity, its inclusion in blends contributed positively to overall insecticidal efficacy, whereas, combinations composed solely of Eucalyptus species exhibited limited or mildly antagonistic interactions. Multivariate analysis further identified acorenone B, thymol and caryophyllene as compounds with relatively high contributions to the insecticidal activity. The observed activity may involve multiple mechanisms, including neurophysiological disruption and interference with reproductive, developmental, and behavioral processes, potentially enhanced by synergistic interactions. Although based on laboratory assays with a single pest species and no assessment of non-target organisms, these results support the potential of these bioactive-enriched EOs as environmentally sustainable alternatives to conventional synthetic insecticides. Further research is warranted to optimize formulation strategies, evaluate field-level applications, investigate the molecular mechanisms that may underlie their bioactivity, and test lower concentrations to accurately quantify their toxicity and potential insecticidal effects.
Supplementary Materials
The following supporting information can be downloaded at: https://www.mdpi.com/article/10.3390/molecules31060945/s1, Table S1: Factor loadings (PC1, PC2 and PC3) for individual and CPM-EOs based on PCA; Table S2: Summary of PLS regression relating chemical composition to LC50; Table S3: VIP scores of all identified chemical compounds from PLS regression analysis; Figure S1: Schematic diagram of the experimental design. Essential oils were extracted from individual plant species and from binary (1:1) and ternary (1:1:1) mixtures of plant material prior to hydro-distillation. Extracted oils were analyzed chemically and tested for insecticidal activity, followed by statistical and synergy analyses. Species included C. citratus, E. camaldulensis, E. lehmannii, S. rosmarinus, and T. vulgaris. EO = essential oil; GC-MS = gas chromatography–mass spectrometry; HCA = hierarchical cluster analysis; PCA = principal component analysis; PLS = partial least squares.
Author Contributions
Conceptualization, I.H., S.B.-B., I.A., M.L., A.B., J.J.R.C. and L.H.; writing—original draft preparation, I.H.; writing—review and editing, I.H., S.B.-B., M.L., A.B., J.J.R.C. and L.H.; funding acquisition, J.J.R.C. and L.H. All authors have read and agreed to the published version of the manuscript.
Funding
This research was funded by a PRIMA grant (Section 2-2021). J.J.R.C. was supported by the Ministry of Science and Innovation (MCIN), the State Investigation Agency (AEI) (https://doi.org/10.13039/501100011033, accessed on 17 November 2025) and by the European Union “NextGenerationEU”/Recovery Plant, Transformation and Resilience (PRTR) (Project PCI2022-132966). L.H was supported by PRIMA grant (Section 2-2021) and was funded by Ministry of Higher Education and Scientific Research (MHESR) Tunisia and the APC was funded by PRIMA grant (Section 2-2021), National Institute of Research on Rural Engineering, Water and Forests (INRGREF) and Ministry of Higher Education and Scientific Research (MHESR) Tunisia.
Data Availability Statement
The original contributions presented in this study are included in the article/Supplementary Materials. Further inquiries can be directed to the corresponding author.
Conflicts of Interest
The authors declare no conflicts of interest.
References
- Loukili, E.H.; Fadil, M.; Elrherabi, A.; Er-Rajy, M.; Taibi, M.; Azzaoui, K.; Salghi, R.; Sabbahi, R.; Alanazi, M.M.; Rhazi, L.; et al. Inhibition of carbohydrate digestive enzymes by a complementary essential oil blend: In silico and mixture design approaches. Front. Pharmacol. 2025, 16, 1522124. [Google Scholar] [CrossRef] [PubMed]
- Nebié, B.; Dabiré, C.M.; Bationo, R.K.; Soma, D.D.; Namountougou, M.; Sosso, S.; Nebié, R.C.H.; Dabiré, R.K.; Palé, E.; Duez, P. Investigation on chemical composition and insecticidal activity against Anopheles gambiae of essential oil obtained by co-distillation of Cymbopogon citratus and Hyptis suaveolens from western Burkina Faso. Malar. J. 2024, 23, 339. [Google Scholar] [CrossRef] [PubMed]
- Soulaimani, B. Comprehensive review of the combined antimicrobial activity of essential oil mixtures and synergism with conventional antimicrobials. Nat. Prod. Commun. 2025, 20, 1934578X251328241. [Google Scholar] [CrossRef]
- Chaachouay, N. Synergy, additive effects, and antagonism of drugs with plant bioactive compounds. Drugs Drug Candidates 2025, 4, 4. [Google Scholar] [CrossRef]
- Seukep, A.J.; Ojong, O.C.G.; Mbuntcha, H.G.; Matieta, V.Y.; Zeuko’o, E.M.; Kouam, A.F.; Kuete, V.; Ndip, L.A. In vitro antibacterial potential of herbal beverage extracts from cinnamon, clove, and thyme and their interactive antimicrobial profile with selected antibiotics against drug-resistant clinical pathogens. J. Trop. Med. 2025, 2025, 9916282. [Google Scholar] [CrossRef]
- Shahin, H.H.; Baroudi, M.; Dabboussi, F.; Ismail, B.; Salma, R.; Osman, M.; El Omari, K. Synergistic Antibacterial Effects of Plant Extracts and Essential Oils Against Drug-Resistant Bacteria of Clinical Interest. Pathogens 2025, 14, 348. [Google Scholar] [CrossRef]
- Elaissi, A.; Moumni, S.; Derbali, Y.; Khouja, M.; Abid, N.; Jlasssi, I.; Khaloud, M.A.; Frederic, L.; Khouja, M.L. Chemical compositions of Eucalyptus sp. essential oils and the evaluation of their combinations as a promising treatment against ear bacterial infections. BMC Complement. Med. Ther. 2024, 24, 220. [Google Scholar] [CrossRef]
- Nebié, B.; Dabire, C.M.; Bonzi, S.; Soma, D.D.; Bationo, R.K.; Sosso, S.; Namountougou, M.; Nebie, R.C.H.; Somda, I.; Pale, E.; et al. Chemical constituents and pesticide efficacy of two essential oil combinations of Cymbopogon citratus (DC.) Stapf and Mentha piperita L. from western Burkina Faso. Mediterr. J. Chem. 2024, 14, 79–87. [Google Scholar] [CrossRef]
- Nebié, B.; Dabiré, C.M.; Bonzi, S.; Bationo, R.K.; Sosso, S.; Nebi, R.C. Chemical composition and antifungal activity of the essential oil obtained by co-distillation of Cymbopogon citratus and Eucalyptus camaldulensis from Burkina Faso. J. Pharmacogn. Phytochem. 2023, 12, 43–48. [Google Scholar]
- Danzi, D.; Thomas, M.; Cremonesi, S.; Sadeghian, F.; Staniscia, G.; Andreolli, M.; Bovi, M.; Polverari, A.; Tosi, L.; Bonaconsa, M.; et al. Essential oil-based emulsions reduce bacterial canker on kiwifruit plants acting as antimicrobial and antivirulence agents against Pseudomonas syringae pv. actinidiae. Chem. Biol. Technol. Agric. 2025, 12, 23. [Google Scholar] [CrossRef]
- Nafis, A.; Kasrati, A.; Jamali, C.A.; Custódio, L.; Vitalini, S.; Iriti, M.; Hassani, L. A comparative study of the in vitro antimicrobial and synergistic effect of essential oils from Laurus nobilis L. and Prunus armeniaca L. from Morocco with antimicrobial drugs: New approach for health promoting products. Antibiotics 2020, 9, 140. [Google Scholar] [CrossRef] [PubMed]
- Kachkoul, R.; Benjelloun Touimi, G.; Bennani, B.; El Habbani, R.; El Mouhri, G.; Mohim, M.; Houssaini, T.S.; Chebaibi, M.; Koulou, A.; Lahrichi, A. The synergistic effect of three essential oils against bacteria responsible for the development of Lithiasis infection: An optimization by the mixture design. Evid. Based Complement. Altern. Med. 2021, 2021, 1305264. [Google Scholar] [CrossRef] [PubMed]
- Mihaylova, S.; Tsvetkova, A.; Stamova, S.; Ermenlieva, N.; Tsankova, G.; Georgieva, E.; Peycheva, K.; Panayotova, V.; Voynikov, Y. Antibacterial effects of Bulgarian oregano and thyme essential oils alone and in combination with antibiotics against Klebsiella pneumoniae and Pseudomonas aeruginosa. Microorganisms 2025, 13, 843. [Google Scholar] [CrossRef] [PubMed]
- Khan, N.; Fatima, S.; Sadiq, M.B. Innovative antifungal and food preservation potential of Eucalyptus citriodora essential oil in combination with modified potato Peel starch. Foods 2025, 14, 602. [Google Scholar] [CrossRef]
- Fratini, F.; Pecorini, C.; Resci, I.; Copelotti, E.; Nocera, F.P.; Najar, B.; Mancini, S. Evaluation of the synergistic antimicrobial activity of essential oils and cecropin a natural peptide on gram-negative bacteria. Animals 2025, 15, 282. [Google Scholar] [CrossRef]
- Aungtikun, J.; Soonwera, M.; Sittichok, S. Insecticidal synergy of essential oils from Cymbopogon citratus (Stapf.), Myristica fragrans (Houtt.), and Illicium verum Hook. f. and their major active constituents. Ind. Crop. Prod. 2021, 164, 113386. [Google Scholar] [CrossRef]
- Bejenaru, L.E.; Segneanu, A.E.; Bejenaru, C.; Biţă, A.; Tuţulescu, F.; Radu, A.; Ciocîlteu, M.V.; Mogosanu, G.D. Seasonal variations in chemical composition and antibacterial and antioxidant activities of Rosmarinus officinalis L. essential oil from southwestern Romania. Appl. Sci. 2025, 15, 681. [Google Scholar] [CrossRef]
- Xia, N.; Wang, J.; Jia, Y.; Duan, J.; Wang, X.; Li, J.; Zhou, P.; Xie, Y.; Shi, H.; Zhao, C.; et al. Optimization of the process of extracting essential oil of rosemary by hydro distillation with different auxiliary methods. LWT 2025, 215, 117266. [Google Scholar] [CrossRef]
- Khodaei, N.; Houde, M.; Bayen, S.; Karboune, S. Exploring the synergistic effects of essential oil and plant extract combinations to extend the shelf life and the sensory acceptance of meat products: Multi-antioxidant systems. J. Food Sci. Technol. 2023, 60, 679–691. [Google Scholar] [CrossRef]
- Alami, A.; Ez zoubi, Y.; Fadil, M.; Annemer, S.; Bassouya, M.; Moustaid, W.; Farah, A. Exploring ternary essential oil mixtures of Moroccan Artemisia species for larvicidal effectiveness against Culex pipiens mosquitoes: A mixture design approach. J. Parasitol. Res. 2025, 2025, 2379638. [Google Scholar] [CrossRef]
- Meradsi, F.; Lekbir, A.; Bensaci, O.A.; Tifferent, A.; Abbasi, A.; Djemoui, A.; Rebouh, N.Y.; Hashem, A.; Avila-Quezada, G.D.; Almutairi, K.F.; et al. Detection of the early sensitive stage and natural resistance of broad bean (Vicia faba L.) against black bean and cowpea aphids. Insects 2025, 16, 817. [Google Scholar] [CrossRef]
- Boukhris-Bouhachem, S.; Souissi, R.; Turpeau, E.; Rouzé-Jouan, J.; Fahem, M.; Brahim, N.B.; Hulle, M. Aphid (Hemiptera: Aphidoidea) diversity in Tunisia in relation to seed potato production. Ann. Société Entomol. Fr. 2007, 43, 311–318. [Google Scholar] [CrossRef]
- Baş, F.H.; Özgökçe, M.S. Life tables and predation rates of Hippodamia variegata (Goeze) and Adalia fasciatopunctata revelieri (Mulstant) (Coleoptera: Coccinellidae) on Aphis fabae (Scopoli) (Hemiptera: Aphididae). Phytoparasitica 2025, 53, 6. [Google Scholar] [CrossRef]
- Shaili, S.J.; Kabiraj, U.K.; Mahedi, M. Fungal biocontrol in agriculture: A sustainable alternative to chemical pesticides–A comprehensive review. World J. Adv. Res. Rev. 2025, 26, 2305–2316. [Google Scholar] [CrossRef]
- Mohamed, N.A.; Mohamed, A.M.; Sharaky, M.; Diab, Y.M.; Abdelbaky, A.S. Evaluation of antioxidant, antimicrobial, and cytotoxic effects of Cymbopogon citratus essential oil. Fayoum J. Agric. Res. Dev. 2025, 39, 372–382. [Google Scholar] [CrossRef]
- Sousa, D.L.D.; Bandeira, M.A.M.; Soares, I.L.; Duque, B.R.; Costa, M.D.R.D.; Viana, G.D.A.; Magalhães, E.P.; de Menezes, R.R.P.P.B.; Sampaio, T.L.; Marinho, M.M.; et al. Cymbopogon citratus essential oil protects tubular renal cells against Ischemia/Reoxygenation injury-involvement Nrf2/Keap1 pathway. Braz. Arch. Biol. Technol. 2025, 68, e25240758. [Google Scholar] [CrossRef]
- Khasanah, L.U.; Ariviani, S.; Purwanto, E.; Praseptiangga, D. Chemical composition and citral content of essential oil of lemongrass (Cymbopogon citratus (DC.) Stapf) leaf waste prepared with various production methods. J. Agric. Food Res. 2025, 19, 101570. [Google Scholar] [CrossRef]
- Schneider, G.; Schweitzer, B.; Steinbach, A.S.; Hodován, Á.S.; Horváth, M.; Bakó, E.; Mayer, A.; Pál, S. The therapeutic potential of west indian Lemongrass (Cymbopogon citratus) essential oil-based ointment in the treatment of pitted Keratolysis. Antibiotics 2025, 14, 241. [Google Scholar] [CrossRef] [PubMed]
- Margarita, V.; Nguyen, T.H.T.; Petretto, G.L.; Congiargiu, A.; Ligas, A.; Diaz, N.; Ton Nu, P.A.; Pintore, G.; Rappelli, P. Effect of essential oils from Cymbopogon citratus, Citrus grandis, and Mentha arvensis on Trichomonas vaginalis and role of its symbionts Mycoplasma hominis and Ca. Mycoplasma girerdii. Front. Parasitol. 2025, 4, 1610965. [Google Scholar] [CrossRef]
- Ammar, H.; M’Rabet, Y.; Hassan, S.; Chahine, M.; de Haro-Marti, M.; Soufan, W.; Andres, S.; López, S.; Hosni, K. Chemodiversity and antimicrobial activities of Eucalyptus spp. essential oils. Cogent Food Agric. 2024, 10, 2383318. [Google Scholar] [CrossRef]
- Rocha-Pimienta, J.; Espino, J.; Martillanes, S.; Delgado-Adámez, J. Essential oils as nature’s dual powerhouses for agroindustry and medicine: Volatile composition and bioactivities—Antioxidant, antimicrobial, and cytotoxic. Separations 2025, 12, 145. [Google Scholar] [CrossRef]
- Khedhri, S.; Polito, F.; Caputo, L.; Khammassi, M.; Dhaouadi, F.; Amri, I.; Hamrouni, L.; Mabrouk, Y.; Fratianni, F.; Nazzaro, F.; et al. Antimicrobial, herbicidal and pesticidal potential of Tunisian Eucalyptus species: Chemoprofiling and biological evaluation. Heliyon 2024, 10, e29905. [Google Scholar] [CrossRef]
- Rodrigues, M.; Mazzafera, P. Essential oils from Eucalyptus species: A review of their activities, applications, and the Brazilian market. Acta Bot. Bras. 2025, 39, e20240111. [Google Scholar] [CrossRef]
- Ivanova, S.; Nalbantova, V.; Benbassat, N.; Dzhoglova, V.; Dzhakova, Z.; Koleva, N.; Vasilev, V.; Grekova-Kafalova, D.; Ivanov, K. Comparison between the chemical composition of essential oils isolated from biocultivated Salvia rosmarinus Spenn. (Rosmarinus officinalis L.) and some commercial products. Pharmacia 2025, 72, e140155. [Google Scholar] [CrossRef]
- Jaïdi, A.; Boukhris, M.; Aydın, B.; Aytar, E.C.; Ayouni, W.; Salem, R.B.; Rigane, G. Chemical Composition, antioxidant activities, and molecular docking analysis of essential oils and hydrolates from two varieties of Salvia rosmarinus (laxiflorus and troglodytorum) growing in southern Tunisia. Chem. Biodivers. 2025, 22, e01266. [Google Scholar] [CrossRef] [PubMed]
- Nasraoui, S.; Mechergui, K.; Chargui, A.; Kammoun, M.; Ameur, M.; Melki, M.; Fauconnier, M.L.; Ammari, Y. Comparative analysis of essential oils, phenolic compounds, and bioactivity in wild and cultivated Salvia rosmarinus, Thymbra capitata, and Artemisia herba-alba under semi-arid Tunisian conditions. Chemoecology 2025, 35, 167–181. [Google Scholar] [CrossRef]
- Khan, N.F.; Sofi, A.B.; Ganaie, K.A.; Shah, S.Q.; Nazir, R.; Shakir, P.S.; Qazi, P.H. Chemical composition and antimicrobial potential of essential oils from morphologically distinct Salvia rosmarinus (Spenn.) cultivars from Kashmir, India. Front. Microbiol. 2025, 16, 1579383. [Google Scholar] [CrossRef] [PubMed]
- Liu, H.; Wang, Y.M.; Yu, G.F.; Chen, Z.H.; Ma, M.M.; Zhang, K.Y.; Zhang, Y.F.; Che, Z.P.; Hu, Z.J.; Chen, G.Q.; et al. Exploitation of Rosmarinus officinalis ct. verbenone essential oil as potential and eco-friendly attractant for Bactrocera dorsalis (Hendel). Ecotoxicol. Environ. Saf. 2025, 300, 118452. [Google Scholar] [CrossRef] [PubMed]
- Giordano, T.; Cerasa, G.; Marotta, I.; Conte, M.; Orlando, S.; Salamone, A.; Mammano, M.M.; Greco, C.; Tsolakis, H. Toxicity of essential oils of Origanum vulgare, Salvia rosmarinus, and Salvia officinalis Against Aculops lycopersici. Plants 2025, 14, 1462. [Google Scholar] [CrossRef]
- Toso, F.; Buldain, D.; Retta, D.; Di Leo Lira, P.; Marchetti, M.L.; Mestorino, N. Antimicrobial activity of Origanum vulgare L. And Salvia rosmarinus Spenn (syn Rosmarinus officinalis L.) essential oil combinations against Escherichia coli and Salmonella typhimurium isolated from poultry. Processes 2025, 13, 2856. [Google Scholar] [CrossRef]
- Ait Melloul, A.; Abbad, I.; Barakate, M. Efficacy of Thymus vulgaris, Syzygium aromaticum, and Marrubium vulgare essential oils against multi-drug resistant uropathogenic bacteria. In Proceedings of the E3S Web of Conferences, Oujda, Morocco, 21–23 February 2025; Volume 632, p. 01001. [Google Scholar]
- Alibeigi, Z.; Rakhshandehroo, E.; Saharkhiz, M.J.; Alavi, A.M. The acaricidal and repellent activity of the essential and nano essential oil of Thymus vulgaris against the larval and engorged adult stages of the brown dog tick, Rhipicephalus sanguineus (Acari: Ixodidae). BMC Vet. Res. 2025, 21, 135. [Google Scholar] [CrossRef] [PubMed]
- Veshareh, A.A.; Mohammadi, P.; Ahmadi, S. Hydrogels containing Thymus vulgaris essential oil as a novel approach for cleaning monument glazed tiles. Herit. Sci. 2025, 13, 130. [Google Scholar] [CrossRef]
- Galgano, M.; Pellegrini, F.; Mrenoshki, D.; Addante, L.; Sposato, A.; Del Sambro, L.; Capozzi, L.; Catalano, E.; Solito, M.; D’Amico, F.; et al. Inhibition of biofilm production and determination of in vitro time-kill Thymus vulgaris L. essential oil (TEO) for the control of Mastitis in small ruminants. Pathogens 2025, 14, 412. [Google Scholar] [CrossRef] [PubMed]
- Yaovi, A.B.; Das, A.; Behera, R.N.; Azokpota, P.; Farougou, S.; Baba-Moussa, L.; Michels, F.; Fauconnier, M.L.; Ambatipudi, K.; Sessou, P. Antibacterial activity of Cinnamomum verum and Thymus vulgaris essential oils on multidrug-resistant zoonotic bacteria isolated from dogs in southern Benin. Access Microbiol. 2025, 7, 000975-v3. [Google Scholar] [CrossRef]
- Jilani, S.; Ferjeni, M.; Al-Shammery, K.; AlTamimi, H.R.M.; Besbes, M.; Besbes, M.; Ahmed Lotfi, S.; Farouk, A.; Ben Selma, W. The synergistic effect of Thymus vulgaris essential oil and carvacrol with imipenem against carbapenem-resistant Acinetobacter baumannii: In vitro, molecular docking, and molecular dynamics studies. Front. Pharmacol. 2025, 16, 1582102. [Google Scholar] [CrossRef]
- Capdevila, S.; Grau, D.; Cristóbal, R.; Moré, E.; De las Heras, X. Chemical composition of wild populations of Thymus vulgaris and Satureja montana in central Catalonia, Spain. JSFA Rep. 2025, 5, 234–246. [Google Scholar] [CrossRef]
- Taibi, M.; Elbouzidi, A.; Bentouhami, N.E.; Haddou, M.; Baraich, A.; Hammouti, Y.; Belbachir, Y.; Bellaouchi, R.; Mothana, R.A.; Hawwal, M.F.; et al. Evaluation of the dermatoprotective properties of Clinopodium nepeta and Thymus vulgaris Essential Oils: Phytochemical analysis, anti-elastase, anti-tyrosinase, photoprotective activities, and antimicrobial potential against dermatopathogenic strains. Ski. Res. Technol. 2025, 31, e70191. [Google Scholar]
- Sánchez-Velandia, J.E.; Gallego-Villada, L.A.; Mäki-Arvela, P.; Sidorenko, A.; Murzin, D.Y. Upgrading biomass to high-added value chemicals: Synthesis of monoterpenes-based compounds using catalytic green chemical pathways. Catal. Rev. 2025, 67, 371–496. [Google Scholar] [CrossRef]
- Sell, C. Chemistry of essential oils. In Handbook of Essential Oils; CRC Press: Boca Raton, FL, USA, 2020; pp. 161–189. [Google Scholar]
- Sadgrove, N.J.; Padilla-González, G.F.; Phumthum, M. Fundamental chemistry of essential oils and volatile organic compounds, methods of analysis and authentication. Plants 2022, 11, 789. [Google Scholar] [CrossRef]
- da Cruz, E.P.; Souza, E.J.D.; Pail, G.L.; Siebeneichler, T.J.; Fonseca, L.M.; Rombaldi, C.V.; Zavareze, E.R.; Dias, A.R.G. Sweet orange and sour orange essential oils: A review of extraction methods, chemical composition, antioxidant and antimicrobial activities, and applications in innovative food technologies. Food Biophys. 2025, 20, 101. [Google Scholar] [CrossRef]
- Mugao, L. Factors influencing yield, chemical composition and efficacy of essential oils. Int. J. Multidiscip. Res. Growth Eval. 2024, 5, 169–178. [Google Scholar]
- Hedayati, S.; Tarahi, M.; Madani, A.; Mazloomi, S.M.; Hashempur, M.H. Towards a greener future: Sustainable innovations in the extraction of lavender (Lavandula spp.) essential oil. Foods 2025, 14, 100. [Google Scholar] [CrossRef] [PubMed]
- Abdelmohsen, U.R.; Elmaidomy, A.H. Exploring the therapeutic potential of essential oils: A review of composition and influencing factors. Front. Nat. Prod. 2025, 4, 1490511. [Google Scholar] [CrossRef]
- Sah, H.; Joshi, P. Conventional techniques for extracting essential Oil: Steam distillation and hydrodistillation. In Essential Oil Extraction from Food by-Products; Humana: New York, NY, USA, 2025; pp. 21–41. [Google Scholar]
- Akdağ, A.; Öztürk, E. Distillation methods of essential oils. Selçuk Üniversitesi Fen Fakültesi Fen Derg. 2019, 45, 22–31. [Google Scholar]
- Rani, N.; Kumar, V.; Chauhan, A. Exploring essential oils: Extraction, biological roles, and food applications. J. Food Qual. 2025, 2025, 9985753. [Google Scholar] [CrossRef]
- Souiy, Z. Essential oil extraction process. In Essential Oils-Recent Advances, New Perspectives and Applications; IntechOpen: London, UK, 2023. [Google Scholar]
- Fatima, A.; Ayub, M.A.; Choobkar, N.; Zubair, M.; Thomspon, K.D.; Hussain, A. The effect of different extraction techniques on the bioactive characteristics of dill (Anethum graveolens) essential oil. Food Sci. Nutr. 2025, 13, e70089. [Google Scholar] [CrossRef]
- Jakab, E.; Blazsó, M.; Barta-Rajnai, E.; Babinszki, B.; Sebestyén, Z.; Czégény, Z.; Nicol, J.; Clayton, P.; McAdam, K.; Liu, C. Thermo-oxidative decomposition of lime, bergamot and cardamom essential oils. J. Anal. Appl. Pyrolysis 2018, 134, 552–561. [Google Scholar] [CrossRef]
- Soltanbeigi, E.; Maral, H. Volatile oil content and composition in fresh and dried Lavandula species: The impact of distillation time. Biochem. Syst. Ecol. 2025, 123, 105066. [Google Scholar] [CrossRef]
- Xie, Y.; Zhang, L.; Wu, W.; Xie, J.; Gao, B.; Xiao, Y.; Zhu, D. Sustainable and green extraction of citrus peel essential oil using intermittent solvent-free microwave technology. Bioresour. Bioprocess. 2025, 12, 48. [Google Scholar] [CrossRef]
- Almeida, R.N.; Soares, R.D.P.; Cassel, E. Fractionation process of essential oils by batch distillation. Braz. J. Chem. Eng. 2018, 35, 1129–1140. [Google Scholar] [CrossRef]
- Nabi, M.H.B.; Ahmed, M.M.; Mia, M.S.; Islam, S.; Zzaman, W. Essential oils: Advances in extraction techniques, chemical composition, bioactivities, and emerging applications. Food Chem. Adv. 2025, 8, 101048. [Google Scholar] [CrossRef]
- Lainez-Cerón, E.; Jiménez-Munguía, M.T.; López-Malo, A.; Ramírez-Corona, N. Effect of process variables on heating profiles and extraction mechanisms during hydrodistillation of Eucalyptus essential oil. Heliyon 2021, 7, e08234. [Google Scholar] [CrossRef] [PubMed]
- Khedhri, S.; Khammassi, M.; Boukhris, B.S.; Pieracci, Y.; Flamini, G.; Gargouri, S.; Amri, I.; Hamrouni, L. Tunisian Eucalyptus essential oils: Exploring their potential for biological applications. Plant Biosyst.-Int. J. Deal. All Asp. Plant Biol. 2024, 158, 40–50. [Google Scholar] [CrossRef]
- Khedhri, S.; Khammassi, M.; Boukhris, B.S.; Pieracci, Y.; Mabrouk, Y.; Seçer, E.; Amri, I.; Flamini, G.; Hamrouni, L. Metabolite profiling of four Tunisian Eucalyptus essential oils and assessment of their insecticidal and antifungal activities. Heliyon 2023, 9, e22713. [Google Scholar] [CrossRef] [PubMed]
- Khaled-Gasmi, W.; Hamouda, A.B.; Chaieb, I.; Souissi, R.; Ascrizzi, R.; Flamini, G.; Boukhris, B.S. Natural repellents based on three botanical species essential oils as an eco-friendly approach against aphids. S. Afr. J. Bot. 2021, 141, 133–141. [Google Scholar] [CrossRef]
- Costa, A.V.; Pinheiro, P.F.; Rondelli, V.M.; de QUEIROZ, V.T.; Tuler, A.C.; Brito, K.B.; Stinguel, P.; Pratissoli, D. Cymbopogon citratus (poaceae) essential oil on frankliniella schultzei (Thysanoptera: Thripidae) and Myzus persicae (Hemiptera: Aphididae). Biosci. J. 2013, 29, 1840–1847. [Google Scholar]
- Zhang, C.; Liu, R.; He, J.; Ma, Z.; Zhang, X. Chemical compositions of Ligusticum chuanxiong oil and lemongrass oil and their joint action against Aphis citricola Van Der Goot (Hemiptera: Aphididae). Molecules 2016, 21, 1359. [Google Scholar] [CrossRef]
- Sjam, S.; Dewi, V.S.; Pujiati, W. Mortality and repellency of aphids (Aphis gossypii G) to orange peel extract (Citrus sinensis L.) and lemongrass emulsion oil (Cymbopogon citratus). In IOP Conference Series: Earth and Environmental Science; IOP Publishing: Philadelphia, PA, USA, 2025; p. 012029. [Google Scholar]
- Jan, A.; Ali, T.; Chirag, S.; Ahmed, S.; Ali, M.; Wali, S.; Basit, A.; Ibrahim, M.; Ullah, K. Eco-friendly management of insect pests and plant diseases using botanical extracts. Glob. Res. J. Nat. Sci. Technol. 2025, 3, 379–408. [Google Scholar]
- Ouknin, M.; Alahyane, H.; Ait Aabd, N.; Elgadi, S.; Lghazi, Y.; Majidi, L. Comparative analysis of five Moroccan thyme species: Insights into chemical composition, antioxidant potential, anti-enzymatic properties, and insecticidal effects. Plants 2025, 14, 116. [Google Scholar] [CrossRef]
- Casas, J.L.; López Santos-Olmo, M.; Sagarduy-Cabrera, A.; Marcos-García, M.Á. Evaluation of selected plant essential oils for aphid pest control in integrated pest management. Insects 2025, 16, 353. [Google Scholar] [CrossRef]
- Celaya, L.S.; Le Vraux, M.A.; Heit, C.I.; Viturro, C.I.; Martina, P.F. Phytochemical and biological profile of essential oils of Elionurus muticus (Spreng.) growing in Northeastern Argentina. Chem. Biodivers. 2023, 20, e202201105. [Google Scholar] [CrossRef] [PubMed]
- Bora, D.; Agrahari, J.; Phukan, A.; Kakoti, B.; Chhetry, S.; Puzari, K.; Saikia, I.; Bardhan, S.; Borah, H. Synergistic action of essential oil of Ageratum conyzoides, Cymbopogon citratus, Eucalyptus globulus, and synthetic insecticides against the mosquito vector, Aedes albopictus Skuse (Diptera: Culicidae). J. Basic Appl. Zool. 2025, 86, 24. [Google Scholar] [CrossRef]
- Galeano, L.J.N.; Prieto-Rodríguez, J.A.; Patiño-Ladino, O.J. Synergistic insecticidal activity of plant volatile compounds: Impact on neurotransmission and detoxification enzymes in Sitophilus zeamais. Insects 2025, 16, 609. [Google Scholar] [CrossRef] [PubMed]
- Et-Tazy, L.; Lamiri, A.; Krimi Bencheqroun, S.; Errati, H.; Hashem, A.; Avila-Quezada, G.D.; Abd-Allah, E.F.; Satrani, B.; Essahli, M.; Satia, L. Exploring synergistic insecticidal effects of binary mixtures of major compounds from six essential oils against Callosobruchus maculatus. Sci. Rep. 2025, 15, 15180. [Google Scholar] [CrossRef]
- Afrazeh, Z.; Sendi, J.J. Eco-friendly control of Helicoverpa armigera using synergistic mixtures of thymol and eucalyptol. Sci. Rep. 2025, 15, 26974. [Google Scholar] [CrossRef]
- Yoon, J.; Tak, J.H. Utilization and validation of the polynomial models to predict insecticidal synergy in the essential oils of Thymus vulgaris L. and Thymus zygis L. against Musca domestica L. Ind. Crop. Prod. 2025, 233, 121405. [Google Scholar] [CrossRef]
- Guarino, S.; Abbate, L.; Arif, M.A.; Peri, E. Insecticidal activity of single essential oil constituents against two stored-products insect pests. Int. J. Trop. Insect Sci. 2025, 45, 185–191. [Google Scholar] [CrossRef]
- Jamunkar, R.; Sinha, D.; Shrivas, K.; Patle, T.K.; Kumar, A.; Tandey, K.; Singh, T. Ultrasound-assisted extraction and RP-HPLC quantification of β-caryophyllene in plant essential oils: Separation efficiency and insecticidal activity. J. Mol. Struct. 2025, 1335, 141882. [Google Scholar] [CrossRef]
- Lhaosudto, S.; Sathantriphop, S.; Kongmee, M.; Karpkird, T.; Leepasert, T.; Ngoen-Klan, R.; Chareonviriyaphap, T. Comparative behavioral responses of β-caryophyllene against Anopheles mosquito species, potential vectors of malaria in Thailand. J. Am. Mosq. Control. Assoc. 2025, 41, 156–159. [Google Scholar] [CrossRef]
- Li, Y.-J.; Liu, T.-A.; Zhao, H.; Han, Y.; Lou, B.-H.; Lei, C.-Y.; Song, Y.-Q.; Jiang, H.-B. Repellency, toxicity, and chemical composition of plant essential oils from Myrtaceae against Asian citrus psyllid, Diaphorina citri Kuwayama (Hemiptera Liviidae). Molecules 2024, 29, 3390. [Google Scholar] [CrossRef]
- Sun, J.S.; Feng, Y.; Wang, Y.; Li, J.; Zou, K.; Liu, H.; Hu, Y.; Xue, Y.; Yang, L.; Du, S.; et al. α-Pinene, caryophyllene and β-myrcene from Peucedanum terebinthaceum essential oil: Insecticidal and repellent effects on three stored-product insects. Rec. Nat. Prod. 2020, 14, 189. [Google Scholar] [CrossRef]
- Chohan, T.A.; Chohan, T.A.; Mumtaz, M.Z.; Alam, M.W.; Naseer, I.; Riaz, A.; Tayyeba, N.; Areeba, I.; Ali, N.; Dur, E.; et al. Insecticidal Potential of α-Pinene and β-caryophyllene against Myzus persicae and their impacts on gene expression. Phyton 2023, 92, 1973. [Google Scholar]
- Usman, L.A.; Akpan, E.D.; Ojumoola, O.A.; Ismaeel, R.O.; Simbiat, A.B. Phytochemical profile and insecticidal potential of leaf essential oil of Psidium guajava growing in north central Nigeria. J. Mex. Chem. Soc. 2025, 69, 480–488. [Google Scholar] [CrossRef]
- Aabouch, F.; Annemer, S.; Satrani, B.; Ettaleb, I.; Kara, M.; Ghanmi, M.; Shahat, A.A.; Choudhary, R.; Farah, A.; Ouajdi, M.; et al. Assessing the optimal antibacterial action of Lavandula stoechas L., Thymus zygis L., and Eucalyptus camaldulensis Dehnh essential oils. Life 2024, 14, 1424. [Google Scholar] [CrossRef]
- Albouchi, F.; Ghazouani, N.; Souissi, R.; Abderrabba, M.; Boukhris-Bouhachem, S. Aphidicidal activities of Melaleuca styphelioides Sm. essential oils on three citrus aphids: Aphis gossypii Glover; Aphis spiraecola Patch and Myzus persicae (Sulzer). S. Afr. J. Bot. 2018, 117, 149–154. [Google Scholar] [CrossRef]
- Abbaci, H.; Nabti, E.-H.; Al-Bekairi, A.M.; Hagras, S.A.A.; Salem-Bekhit, M.M.; Adjaoud, A.; Alzahrani, H.A.; Bensidhoum, L.; Alenazy, R.; Piras, A.; et al. Comparative bioactivity evaluation of chemically characterized essential oils obtained from different aerial parts of Eucalyptus gunnii Hook. f. (Myrtaceae). Molecules 2023, 28, 2638. [Google Scholar] [CrossRef]
- Wei, Q.Y. A method for evaluating the toxicity interaction of binary mixtures. MethodsX 2020, 7, 101029. [Google Scholar] [CrossRef]
- Kalyanasundaram, M.; Das, P.K. Larvicidal and synergistic activity of plant extracts for mosquito control. Indian J. Med. Res. 1985, 82, 19–23. [Google Scholar]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.