Abstract
Dietary lipids not only enhance the flavor and nutritional value of food, but more importantly, they offer essential fatty acids and energy for metabolism. The importance of lipid unsaturation has gained increasing attention; however, the impact of the alky chain length on biofunction of dietary lipids remains unclear. This article discusses the effects of the alkyl chain length on the biological function of lipids, focusing on physical and chemical properties, digestion and absorption, and nutritional functions. Firstly, with the increase in the chain length, the melting point of the crystal increases, the symmetry increases, and the hypersensitivity induction decreases. Secondly, the alkyl chain length affects the contact between lipid droplets and lipase, as well as the fatty acids release rate. Finally, medium-chain and short-chain lipids can partially reverse the effect of long-chain lipids. Understanding the effect of the alkyl chain length on the biofunction of dietary lipids can provide valuable insights for designing nutritious diet.
1. Introduction
Dietary lipids confer specific functional, nutritional, and sensory properties to food. They supply energy for metabolism, participate in cell membrane composition, control intercellular and intracellular communication, and regulate gene expression that can trigger complex cascade reactions, thereby maintaining body homeostasis [1,2,3]. Dietary lipids primarily consist of triglycerides and lipoids (phospholipids, sterols). Triglycerides, which are the main components of dietary lipid, exist in various forms, such as free (oil), droplet (emulsion), or crystallization network (butter). Upon ingestion, triglycerides are first degraded into diglycerides with the assistance of gastric lipases. Subsequently, they are further digested into monoglycerides and fatty acids by various lipase [4]. These digestive products are absorbed and metabolized to provide energy. Beyond providing energy, triglycerides can also serve as bioactive substances or auxiliaries that help ameliorate metabolic syndrome, such as corpulency and insulin resistance [5,6]. Most nutritional molecules typically exert their effects by acting on membrane proteins or intracellular targets [7], necessitating passage through the cell membrane. Modification of triglyceride can facilitate better interactions between these biomolecules and cell membranes.
In the field of dietary triglycerides nutrition, the importance of lipid unsaturation has increasingly garnered attention. Several excellent reviews on lipid unsaturation have been published in recent years [8,9,10,11]. Saturated triglycerides (SATs) are less prone to oxidation; however, they cause a greater increase in insulin resistance, endotoxemia and multiple plasma ceramides compared to unsaturated triglycerides (UNSATs) [12]. Saturated triglyceride is more likely to have a negative impact on human liver metabolism than UNSAT. Additionally, the polyunsaturated structure of PUFAs complicates the spatial conformation of their glycerides, making them resistant to hydrolysis by lipases. Next, the digestive environment accelerates the oxidation of PUFAs, leading to a further reduction in their bioavailability [13].
Indeed, numerous studies have reported the chain length of the alkyl group is another crucial factor affecting the biofunction of dietary triglycerides. Triglycerides are composed of glycerol and fatty acids, and the category of fatty acids determines the category of triglycerides. Johannes et al. [14] coupled small molecules with triglycerides to explore the biological activity and cell permeability. The results indicated that compared to long-chain triglyceride (LCT, carbon number ≥ 14) conjugates, medium-chain triglyceride (MCT, carbon number 6–12) conjugates showed better uptake and biological activity, with effects comparable to those of short-chain triglyceride (SCT, carbon number < 6) conjugates. Zhang et al. found that under the intervention of dietary lipid for atherosclerosis, the aortic sinus plaque and the entire aorta acreage were remarkably smaller in the octanoic acid group compared to the decanoic acid and stearic acid groups [15]. Furthermore, another study revealed that the effect of palmitoleic acid was stronger than that of oleic acid in improving human endothelial cell inflammation [16]. These results appear to be mediated, at least in part, by inhibiting NF-κB gene expression and up-regulating PPAR-α.
However, no comprehensive in-depth reviews on the impact of the alkyl chain length have been published. The article aims to review the functions of the carbon chain length on the biofunction of dietary triglyceride. The roles of the chain length on physical and chemical properties, digestion, and absorption are also discussed. This review serves as a reference for developing a reasonable dietary ratio to prevent and treat intestinal and metabolic diseases.
2. The Role of Alkyl Length in Characteristics of Triglycerides
Triglycerides exhibit diverse structures and abundant contents in nature. Additionally, they have multiple metabolic pathways influenced by the alkyl length. Therefore, understanding the digestion, absorption, and metabolism of different triglycerides can help us better grasp the impact of alkyl chain length on the biological function of triglycerides and fatty acids (Figure 1).
2.1. Origin
Triglycerides are naturally present in multiple foods, e.g., meat, plants, nuts, and milk [17]. The primary plant sources of SCTs and MCTs are coconut oil and palm kernel oil [18,19]. Meanwhile, they are widely found in milk powder, especially in premature infant formula, where the content of medium-chain triglyceride can reach up to 50% of fat content [20,21]. Medium- and long-chain triacylglycerol (MLCT) are primarily found in mammalian milk fat, such as breast milk, goat milk, and related products, and have high nutritional value [22,23]. Ruminant milk fat contains short-chain fatty acids (SCFAs, carbon number < 6), including butyric acid (10 mol%), caproic acid (5 mol%), and acetic acid [24]. Short-chain fatty acids can also be generated by the fermentation of dietary fiber by intestinal anaerobic microorganisms [25]. For instance, Prevotella is a strong producer of SCFAs, primarily generating acetic acid [26]. Butyric acid is produced by the fermentation of Fecal bacilli [27]. Additionally, valeric acid is mainly produced by Megasphaera massiliensis [28]. Natural sources of medium-chain fatty acids (MCFAs, carbon number 6–12) include breast milk, where the uptake is about 15% of total fatty acids [29]; the content in mature milk was the highest, being about 42.18% higher than that in transition milk [30]. The quantity of MCFA in term milk was significantly higher than that in preterm milk [30]. Moreover, octanoic acid and lauric acid can also be derived from goat milk [31,32]. Long-chain fatty acids (LCFAs, carbon number ≥ 14), including long-chain saturated fatty acids (LCSFAs) and long-chain unsaturated fatty acids (LCUFAs), are widely found in various fats, in particular, vegetable oil, soybean oil, peanut oil, rapeseed oil, and animal lard and butter.
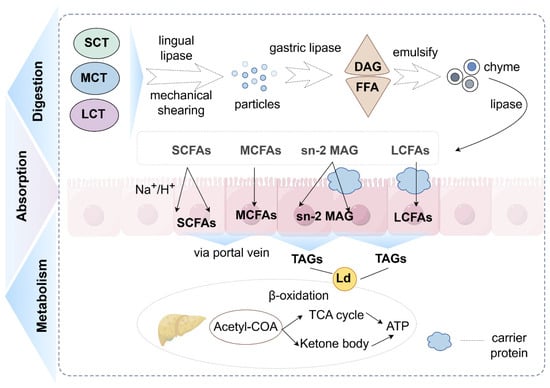
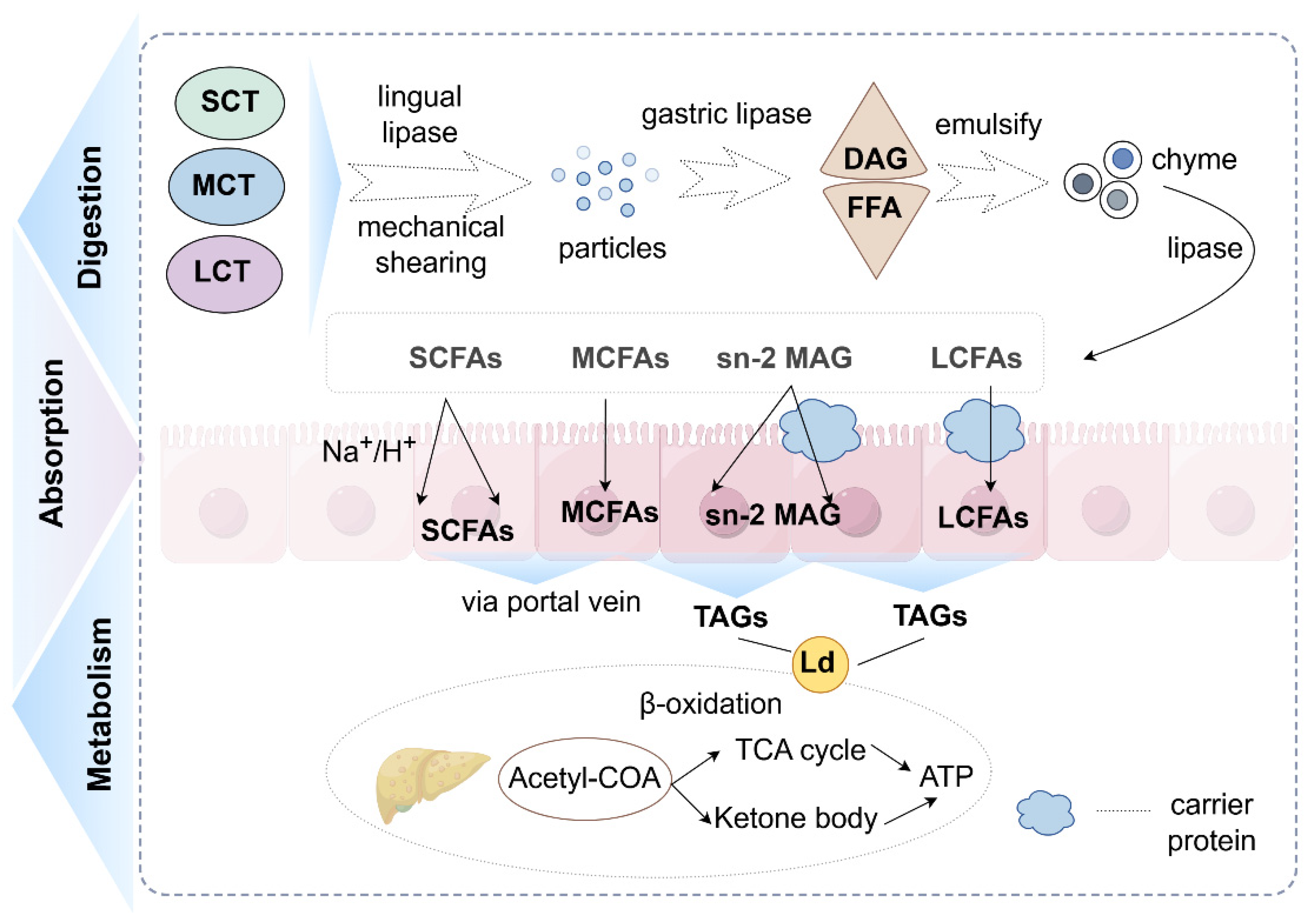
Figure 1.
Digestion, absorption and metabolism of triglycerides (created with Figdraw). (1) Triglycerides are degraded into monoglycerides and fatty acids by a series of enzymes. (2) Fatty acids enter intestinal epithelial cells in a variety of ways (SCFAs: depend on ion carriers and diffusion; MCFAs: diffusion effect; LCFAs: protein assistance). (3) They are metabolized in the liver (SCFAs and MCFAs: direct oxidation and providing a small amount of energy; LCFAs: oxidation after re-esterification and offering most of the energy required by the body). Abbreviations: SCT, short-chain triglycerides; MCT, medium-chain triglycerides; LCT, long-chain triglycerides; DAG, diacylglycerol; FFA, free fatty acids; Ld, lipid droplet; SCFAs, short-chain fatty acids; MCFAs, medium-chain fatty acids; LCFAs, long-chain fatty acids; sn-2 MAG, sn-2 mono-acylglycerol; TAGs, triacylglycerols; TCA cycle, tricarboxylic acid cycle; Acetyl-CoA, acetyl coen-zyme A.
Figure 1.
Digestion, absorption and metabolism of triglycerides (created with Figdraw). (1) Triglycerides are degraded into monoglycerides and fatty acids by a series of enzymes. (2) Fatty acids enter intestinal epithelial cells in a variety of ways (SCFAs: depend on ion carriers and diffusion; MCFAs: diffusion effect; LCFAs: protein assistance). (3) They are metabolized in the liver (SCFAs and MCFAs: direct oxidation and providing a small amount of energy; LCFAs: oxidation after re-esterification and offering most of the energy required by the body). Abbreviations: SCT, short-chain triglycerides; MCT, medium-chain triglycerides; LCT, long-chain triglycerides; DAG, diacylglycerol; FFA, free fatty acids; Ld, lipid droplet; SCFAs, short-chain fatty acids; MCFAs, medium-chain fatty acids; LCFAs, long-chain fatty acids; sn-2 MAG, sn-2 mono-acylglycerol; TAGs, triacylglycerols; TCA cycle, tricarboxylic acid cycle; Acetyl-CoA, acetyl coen-zyme A.
2.2. The Impacts on Physicochemical Properties
The alkyl chain length affects the thermal properties and polymorphism of aliphatic acids and esters. For saturated straight-chain aliphatic acids and esters, the melting point and heat capacity increase with the even number of the alkyl chain length, but the difference in the upward trend between the two is not significant. Moreover, the melting point of whole triglycerides is higher than that of free fatty acids [33,34]. However, the trend is not evident for unsaturated aliphatic acids, which are mainly influenced by the configuration and position of double bonds. Meanwhile, thermal conductivity and chemical structure do not change significantly with the chain length. Hagemann et al. revealed the stability of the β-form increased as the acyl chains extended from 16 to 22 carbons. In a recent study on the functions of triacylglycerol components in the crystallization behavior of milk fat [35], Yoga et al. confirmed that fully saturated LCTs were beneficial to the production of 2L crystals and triglycerides containing SCFAs, which conduce to the formation of 3L crystals. Moreover, triglycerides containing SCFAs restrained the crystallization process of milk fat to a certain extent. Furthermore, fatty acids can also combine with carbohydrates, such as starch, to form nanocomposite materials. As the chain length increased from C12 to C22, the encapsulation efficiency of fatty acids improved, and the compound’s emulsification enhanced [36].
The chain length of fatty acids affects the function of biological membranes. Long-chain fatty acids (e.g., C18–C28) enhance intermolecular van der Waals forces between lipid hydrocarbon tails, leading to a higher phase transition temperature (Tm) of the lipid bilayer [37]. This stronger intermolecular interaction reduces the lateral diffusion capacity of lipid molecules and membrane proteins, thereby decreasing membrane fluidity and rendering the membrane at a more rigid gel state. In contrast, short-chain fatty acids (e.g., C12–C16) have weaker intermolecular hydrophobic interactions due to their shorter hydrocarbon tails, resulting in a lower Tm and looser lipid packing; this loose arrangement increases the mobility of lipid molecules, improves membrane fluidity, and maintains the membrane in a dynamic liquid–crystalline state that facilitates material transport and signal transduction. Lipid packing density in the biological membrane is also closely associated with the fatty acid chain length. Long-chain fatty acids can extend deeper into the hydrophobic core of the lipid bilayer, reducing the gaps between adjacent lipid molecules and forming a more ordered and denser packing structure; this dense packing not only enhances the mechanical stability of the membrane but also limits the penetration of small molecules and ions across the membrane [38]. On the contrary, short-chain fatty acids have limited extension in the hydrophobic core, resulting in larger gaps between lipid molecules and relatively loose packing; such loose packing may increase the permeability of the membrane to small hydrophobic molecules while reducing the membrane’s resistance to external mechanical stress. Notably, the effect of the fatty acid chain length on lipid packing can further regulate the formation of lipid domains (e.g., raft domains) in the membrane, which are crucial for the localization and function of membrane proteins [39]. Collectively, the chain length of fatty acids incorporated into biological membranes serves as a key structural factor that modulates membrane biophysical properties and further mediates membrane functionality, laying a foundation for understanding the structure–bioactivity relationship.
In summary, the chain length is a crucial factor in the physicochemical properties of fatty acids, triglycerides, and other related compounds.
2.3. The Impacts on Digestion and Absorption Behaviors
Triglycerides are hydrophobic molecules, and their apolarity prevents them from being transported directly from the lumen of gut to the intestinal epithelium. Triglycerides are initially digested by the mouth, stomach, and intestine, and eventually form monoglycerides and free fatty acids. Triglyceride digestion typically involves three steps: first, triglycerides are broken down into smaller particles through mechanical shearing, such as chewing, swallowing, and hydrolysis of lingual lipase in the mouth; second, diacylglycerol and remaining triglycerides are further emulsified; and finally, various lipase act to hydrolyze fats into monoacylglycerol and free fatty acids. Various enzymes are involved in the digestion of dietary triglycerides within the body. Fatty acids are initially released by hydrolysis from Sn-1 and Sn-3 positions of triglycerides, while Sn-2 monoglyceride is retained to fulfill its critical nutritional and metabolic roles [40].
The chain length of alkyl is closely related to triacylglycerol digestion. As chain length increases, the degradation rate of triacylglycerol decreases. Previous research has indicated that short-chain and medium-chain triacylglycerol have higher rates of hydrolysis and metabolism when compared with long-chain triacylglycerol [41]. These results may be closely associated with the structure of triglyceride and its various digestive enzymes.
Short-chain triglycerides (SCTs) and medium-chain triglycerides (MCTs) They are more easily digested, which may be ascribed to the hydrolyzation characteristics of lipase. Compared to LCFAs, gastric lipase prefers to hydrolyze SCFAs and MCFAs at the Sn-3 position, while pancreatic lipase is more efficient in hydrolysis of Sn-1,3 MCFAs [17]. Moreover, short-chain triglycerides (SCTs) form smaller droplets when emulsified in the stomach, thereby increasing the surface area bound to lipase. Early in life, the content of pancreatic lipase and bile salts is limited in the body. Consequently, tongue and stomach lipases exist a necessary role in the decomposition of triglycerides. Lingual lipase has a higher affinity to MCTs than for LCTs [3]. Tongue lipase, secreted by the tongue gland along with the food from the mouth, hydrolyzes Sn-3 fatty acids in the stomach [42]. An in vitro study demonstrated that the activity of lingual lipase’s activity towards tricaprylate is 10 times greater than towards triolein. An in vivo study showed the digestion rate of LCT is lower than that of MCT in preterm infants, primarily due to the utilization of alternative digestion pathways involving in lingual and gastric lipase [43]. During digestion, the released LCFAs tend to accumulate at the oil–water boundary, significantly reducing the rivalrous absorption of bile salts, and limiting pancreatic lipase’s contact with fat droplets. Short-chain fatty acids and medium-chain fatty acids exhibit higher affinity for water, enabling them to rapidly enter the surrounding aqueous phase. This provides additional absorption sites at the oil–water interface of lipid droplets, facilitating easier access to the lipid surface for pancreatic lipases [44].
Triglycerides are primarily absorbed by the small bowel and oxidized in the liver to provide energy [45]. The length of the alkyl chain affects the absorption and metabolism of fatty acids and monoglycerides. It influences the absorption of substances through three mechanisms: intestinal cell absorption, intracellular processing, and mesenteric lymph reception [46]. McKimmie et al. found that the FFA absorption efficacy rate decreased with the increase in chain length, from 0.953 ± 0.017 for tetradecanoic acid to 0.798 ± 0.032 for octadecanoic acid, and extremely dropped to 0.264 ± 0.02 for eicosanoic acid [47]. This result can, at least in part, be attributed to the differing absorption pathways of multiple fatty acids and monoglycerides. Short-chain fatty acids enter enterocyte through four mechanisms [48]: a nonionized proton dispersion form, when converted to SCFA-2HCO3−, a hydrogen-coupled monocarboxylate transporter, and as a sodium-coupled monocarboxylate transporter. Medium-chain fatty acids are absorbed through passive diffusion due to their small molecular weight and strong water solubility [49]. Long-chain fatty acids are transported into colonic epithelial cells by binding with carrier proteins, such as fatty acid binding proteins (FABP) and fatty acid transport proteins (FATP) [50,51,52]. Additionally, long-chain fatty acids obtained through digestion may precipitate into fatty acid calcium soaps and form crystals at body temperature, thereby affecting the absorption of fatty acids. Monoglycerides and fatty acids are transported to the endoplasmic reticulum (ER) and synthesized into triglycerides again. They then form lipid droplets for energy storage in cells. However, medium-chain fatty acids exhibit low affinity with lipid anabolic enzymes (e.g., diglyceride acyltransferase) [18], which facilitate rapid oxidation. Diglyceride acyltransferase mainly exists in the endoplasmic reticulum (ER) and lipid droplets. It attaches to the superficies of lipid droplets through the C-terminal amphiphilic α-helix, thereby regulating fat absorption, triacylglycerol resynthesis and cholesterol metabolism [53]. Following intracellular processing, substances enter the lamina propria via exocytosis and ultimately reach the mesenteric lymph node.
Digested fatty acids are transported in various ways in the body. Short-chain fatty acids that enter into the systemic circulation by means of the vena portae are then delivered to the peripheral tissues for oxidation and energy supplying [54]. Medium-chain fatty acids, the main ketogenic fatty acids, are directly circulated through the portal vein and rapidly metabolized in the body [55]. Additionally, they can be transported as free fatty acids or connected to plasma albumin [56]. Long-chain fatty acids must be re-esterified in the small bowel to shape chylomicrons, which are then transported via lymph vessels and blood vessels [57]. The conjugation of aliphatic acid to serum albumin influences their transportation, thereby affecting the body’s uptake. Kragh-Hansen et al. [58] proved that the primary association constants of aliphatic acid with serum albumin increase with the chain length, but not linearly. The ratio between K1 for decanoic acid/caprylic acid is 2.6 and the K1 of laurate is 6.7 times that of decanoate. Besides that, the quantity and location of high-intimacy binding sites in the albumin molecular are likely determined by the chain length [59,60]. In addition, studies indicated that the macromolecule crowding affects the combination of aliphatic acid to serum albumin [56]; in a crowded nutrient environment, the configuration of BSA is loosened, promoting the binding of MCFA–BSA.
Regarding oxidative metabolism, butyrate is superior to ethane acid, propionic acid, and even glutamine, gluconate, and acetone bodies [61]. It serves as a crucial respiratory fuel for the body. Simultaneously, medium-chain fatty acids are preferentially metabolized to produce energy compared to LCFAs in the liver [62]. Long-chain fatty acids exhibit relatively slow metabolic rates and store a certain amount in tissues, and their oxidation is partially dependent on carnitine [63,64].
From the above, the alkyl chain length of triglycerides affects their digestion, absorption transport and metabolism. The given metabolic behavior of triglyceride is well linked to their profitable physiological function in vivo. Therefore, the alkyl chain length can be used as an important factor to optimize the proportion of fatty acids and enhance dietary nutrition.
3. The Role on Nutritional Properties
To date, numerous studies have been carried out on the intestinal health changes induced by dietary triglycerides and fatty acids (Table 1). Moreover, they can influence physiological state in vivo by acting on certain molecules or specific proteins (Figure 2). In series of studies, we observed that the alkyl chain can affect the nutritive peculiarities of fatty acids and triglycerides. These circumstances will be addressed below.

Table 1.
Effects of fatty acids and triglycerides on intestinal microenvironment.
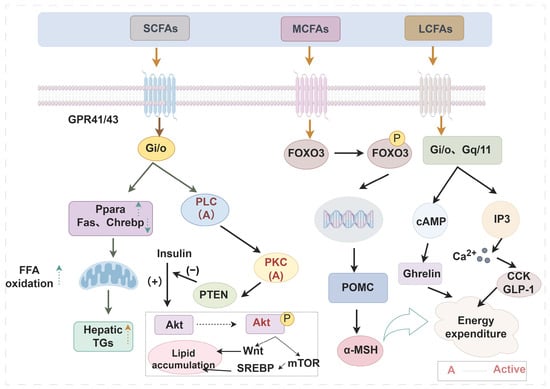
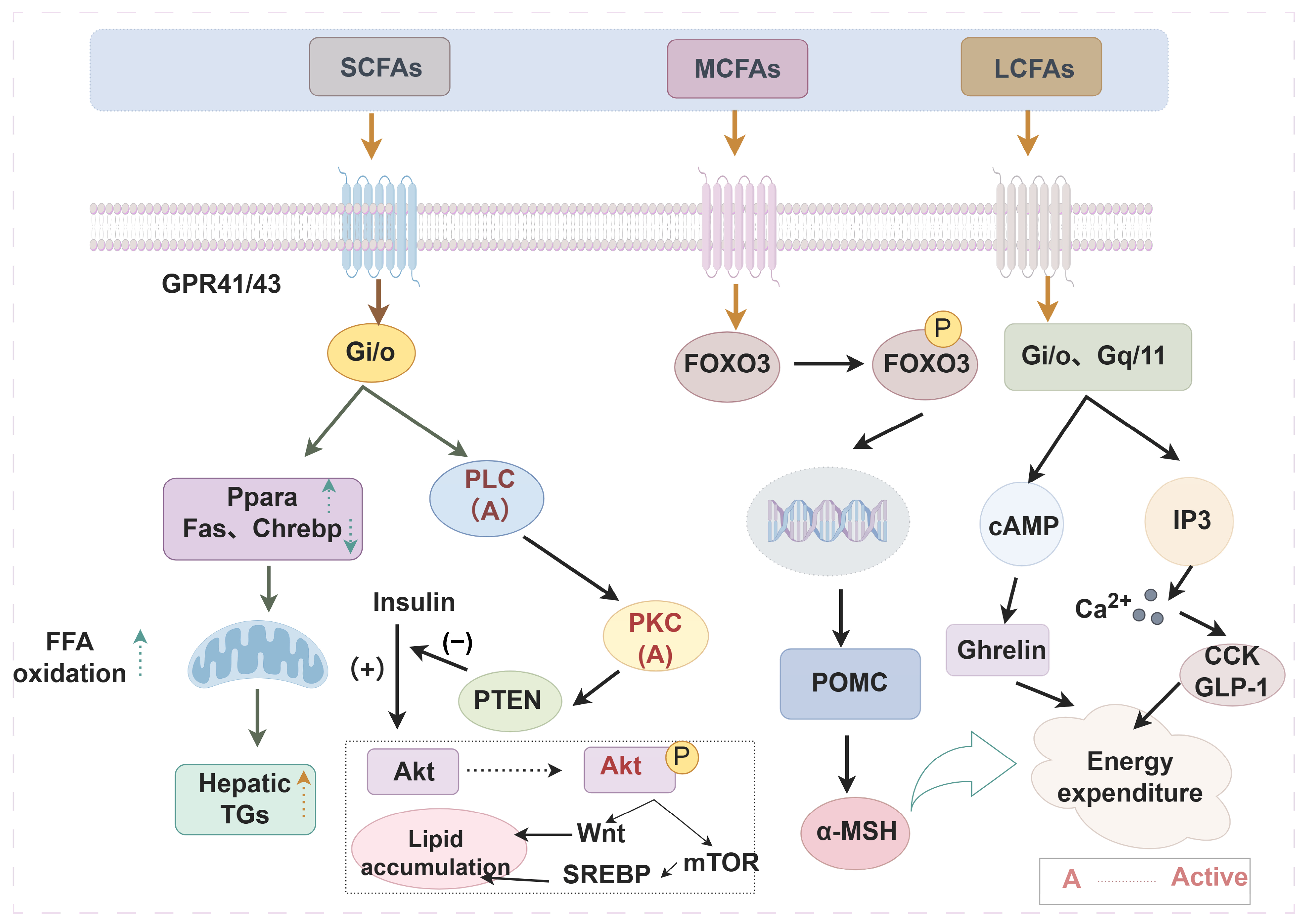
Figure 2.
Effects of different fatty acids on peripheral tissue metabolism or function (By Figdraw) The alkyl chain length affects the biological function of fatty acids through multiple mechanisms. Take regulating obesity as an example, SCFAs act on GPR41 and GPR43 to interfere with downstream gene expression, thereby promoting lipid metabolism. MCFAs and LCFAs apply to GPR40 and GPR120, respectively, regulate transcription factor expression and hormone secretion, and control energy metabolism, thus enhancing satiety. Arrows outside the text boxes indicate the subsequent induced reaction, while arrows inside the text boxes represent a trend of change. ↑—up-regulation; ↓—down-regulation. Abbreviations: SCFAs, short-chain fatty acids; MCFAs, medium-chain fatty acids; LCFAs, long-chain fatty acids; Gi/o, guanine nucleotide-binding protein, inhibitory α subunit; PLC, phospholipase C; PKC, protein kinase C, PTEN, phosphatase-tensin gene; SREBP, sterol-regulatory element binding protein; POMC, pro-opiomelanocortin; CCK, cholecystokinin; GLP-1, glucagon-like peptide-1; Ppara, peroxisome proliferator-activated receptor alpha; FFA, free fatty acids; TGs, triglycerides; Akt, ak strain transforming; Fas, fatty acid synthase; Chrebp, carbohydrate response element binding protein; mTOR, mammalian target of rapamycin; FOXO3, forkhead box protein O3; Gq/11, guanine nucleotide-binding protein, q/11 polypeptide; α-MSH, α-melanocyte-stimulating hormone; Ghrelin, growth hormone secretagogue.
3.1. Effects on Intestinal Microenvironment
Different types of triglycerides regulate the intestinal microenvironment in varied ways. The study by Kripke et al. [74] investigated the impacts of dietary intervention on gut health in short bowel syndrome (SBS). It was found that compared to the supplemental chemical definition (CD) group and the MCT diet group, the diet supplemented with SCT lead to a significant increase in mucosal weight, intestinal segment weight, mucosal protein and DNA expression level of jejunum and colon. Moreover, short-chain triglycerides promoted the adaptive growth in the jejunum and colon in SBS and maintained a comparable nutritional status. They ameliorated metabolic syndrome and maintained the intestinal microenvironment by multiple mechanisms. Studies have shown that butyrate glycerides not only abated the immune-inflammatory response by restraining the NF-κB/MAPK pathways but modulated the intestinal bacteria to improve intestinal health [72]. Meanwhile, valeric acid glycerides (GVA) obviously raised the proportion of the villus height to crypt depth in the jejunum and the density of enteroendocrine cells producing glucagon-like peptide-2 in order to diminish the occurrence rate of necrotic enteritis [69]. Tricaprylin mainly maintained intestinal morphology by improving digestive enzyme activity, the concentration of SCFAs, and the secretion of protein related to intestinal permeability, while reducing the inflammatory cytokine levels and the abundance of hazardous intestinal microbiota [73].
Fatty acids can influence the intestinal microenvironment. Infusing LCFAs into the ileum of humans and rats slowed small bowel transportation time. In contrast, short-chain fatty acids accelerated transport via a topical enteric reflex. Ethane acid (20 mM, 50 mM, 100 mM), butanoic acid (100 mM), and octoic acid (100 mM) significantly accelerated migration. The transportation velocity was inversely proportional to SCFA carbon chain length. Moreover, the colon can easily absorb these SCFAs, recovering some of the lost energy [75]. Short-chain fatty acids can promote intestinal impermeability and improve lipidic metabolism, lessening the occurrence of metabolic syndromes such as obesity [66,67]. They can also adjust the secretion of antifungal peptides from the intestinal epithelial cells. They can stimulate RegIIIγand defensins generation by reactivating mTOR and STAT3, thus strengthening the defense capability of intestinal epithelial barrier [68]. It was found that adding acetate to the growth medium could boost the level of histone acetylation in Salmonella enterica and Bacteria coli [37,76]. The acetylation of PhoP protein cut down the binding capacity of PhoP protein to DNA, thus weakening the toxicity of Salmonella. Butyrate inhibited the accumulation of enteric pernicious bacteria while accelerated the colonization of conducive bacteria, such as Actinobacteria, Bifidobacterium bifidum, and Bacteroidetes [77]. Butyrate also coordinated the gut barrier protection by improving epithelial O2 consumption which is conducive to the stability of transcription factor, such as hypoxia inducible factor (HIF) [66]. Medium-chain fatty acids have gained increasing attention owing to their possibly favorable antimicrobial effect. There were studies that proved MCFs were involved with a wide range of intestinal microflorae. They exerted protective effects by decreasing part of the colonization of the gut microbiome, including Campylobacter, Clostridium, Salmonella, and Escherichia coli, with an average of 44%, 78%, 79%, and 66%, respectively, thus promoting the nutritional and health status of poultry and decreasing the risk of increased antibiotic resistance in animals [70]. Lauric acid can be a naturally useful substance in restraining Clostridioides difficile growth. The generation of reactive oxygen species (ROS) and cell membrane lesions are part of the mechanism by which lauric acid can play a role [78]. Gastrointestinal function is closely related to the enteric nervous system, and the intestinal microenvironment can be maintained through the brain–gut axis. Palmitic acid (PA) caused an evident absence of myenteric neurons in both the ileum and colon by mediating the ADP-sensitive P2y13 receptor, thereby impairing intestinal health [65]. However, palmitoleic acid also increased the abundance of Akkermansia muciniphila, changed the content of SCFAs in the intestine, and ameliorated intestinal inflammation [79]. The increase in the carbon chain length of fatty acids follows the fundamental rules of their biosynthesis. In the classical de novo synthesis pathway catalyzed by fatty acid synthase, each elongation cycle uses malonyl-CoA as the donor of the two-carbon unit, resulting in the vast majority of natural fatty acids having an even number of carbon atoms. Despite exceptions, odd-chain fatty acids (e.g., pentadecanoic acid, heptadecanoic acid) synthesized using propionyl-CoA as the initiator can be of low abundance in nature, primarily derived from branched-chain amino acid metabolism, intestinal flora fermentation, and ruminant fat. Therefore, this study focused primarily on the properties of even-chain fatty acids.
3.2. Effects on Peripheral Tissue Metabolism or Function
The alkyl chain length changes the nutritional efficacy of triglycerides. In a double-blind randomized crossover research, sufferers with Crohn’s disease or bowel bypass syndrome were treated with triacylglycerol emulsion for four weeks. The results manifested when two of those treated with LCT emulsions exhibited aberrant liver function, which was then consequently reversed when switching to a structured triglyceride therapy. Hence, structured triglycerides including medium- and long-chain fatty acids may be relevant with a possible reduction in liver dysfunction [80]. In addition, supplementing MCTs in dietary LCTs can amplify postprandial caloric production. Research has indicated that MCTs can exert a key role in obesity management by increasing energy consumption and satiety [81,82]. This effect of MCTs may be related to brown fat (BAT) activation in animals [5]. When the presentation of β3_adrenergic receptor (β3_AR) protein and uncoupling protein 1 (UCP1) up-regulated in BAT and the noradrenaline (NE) pathway was activated, this can trigger many biochemical lipolysis reactions [83]. Furthermore, when there exists a lot of glycogen in vivo, MCTs can also undergo lipid metabolism to generate ketone B-hydroxy butyric acid (BHBA). B-hydroxy butyric acid can affect brain activity through the blood–brain barrier, regulat nerve center signals related to food intake, reduce appetite, and even produce anorexia [84]. Long-term consumption of LCTs may pose health risks, but moderate intake of LCTs is essential in daily life. This is because LCTs serve as the primary dietary source of essential fatty acids (linoleic acid and alpha-linolenic acid). Furthermore, LCTs act as vital carriers for the absorption and transport of fat-soluble vitamins (A, D, E, K).
The effects of dietary fatty acids on metabolism or on the function of peripheral tissues are different. Exploring the effects of SCFAs on adipose degeneration and inflammation in metabolic dysfunction-associated steatohepatitis (MASH) mice, three main SCFAs (acetate, propionate, and butyrate) were selected for intervention. The results showed that SCFAs could alleviate MASH, among which sodium acetate had the best effect [85]. The mechanism of action was to affect AMPK activation and inhibit macrophage pro-inflammatory differentiation. In human endothelial cells, studies have displayed that palmitoleic acid has better anti-inflammatory effects than oleic acid and palmitic acid, mainly by reducing the expression of NF-κ B and the release of inflammatory factors, up-regulating the presentation of the PPAR-α gene, and preventing the occurrence of endothelial dysfunction [16].
Diverse fatty acids can act on different fatty acid receptors, carry out signal transduction through various mechanisms, and regulate body metabolism. The fatty acid receptor FFAR2/3 can exhibit a common motif that SCFA can recognize. There is a PAM binding pocket in FFAR3. At the same time, AR420626 (selective agonist of FFAR3/GPR41) can interact with Gαi to provide a targeted receptor–transduction interface for the recognition of fatty acids with different carbon chain lengths [86]. Short-chain fatty acids could mediate the free fatty acid receptor FFAR43 and regulate the transcript of lipid metabolism genes (Fas, Ppara, Chrebp, etc.) in the liver, playing an anti-obesity role [87]. Acetate-mediated GPR43 activation can inhibit insulin-induced Akt phosphorylation through the G (i/o) βγ-PLC (phospholipase C)-PKC (protein kinase C)-PTEN (phosphatase-tensin gene) signaling pathway [88]. Inhibiting insulin biological activity can promote energy consumption, accelerate lipid burning in adipose tissue, and avoid lipid accumulation. Acetate could also regulate instantaneous contraction and control blood pressure through direct target receptor GPR43 [89]. Propionate can reduce blood pressure by mediating the activation of olfactory receptor 78 (olfr78) and G protein-coupled receptor 41 (GPR41) [45]. Propionate can also alleviate myocardial I/R injury aggravated by Angiotensin II dependent on caveolin-1/ACE2 (angiotensin-converting enzyme 2) axis through GPR41 [90]. Octanoic acid can control food intake and prevent various complications caused by obesity [91]. The mechanism mainly for this was that it selectively activated the excitability of pro-opiomelanocortin (POMC) neurons through GPR40 and stimulated changes in energy status via indirect non-synaptic, purines, and adenosine receptor-dependent mechanisms. Such changes were not observed in the oleic acid treatment group. Octanoic acid was also associated with the activation of GPR84, affecting downstream signal transduction and preventing disease [92]. G protein-coupled receptor 120 (GPR120, FFAR4) is a LCFA sensor, mainly long-chain unsaturated fatty acids (LCUFAs) [93]. LCUFAs lured GLP-1 secretion from STC-1 endocrine c’s of gut cells to control appetite and enhance sensitivity in a FFA4-dependent manner [94]. In addition, GPR120 mRNA was enriched in mice and human microglia; acting on this target contributed to neuroimmunomodulation and behavior improvement [95].
4. Conclusions and Prospects
Short- and medium-chain fatty acids (SCFAs/MCFAs) exhibit distinct functional advantages. Their shorter chains facilitate higher membrane fluidity and more efficient interaction with metabolic enzymes (e.g., lipases), leading to rapid absorption and oxidation. These properties underpin their therapeutic and nutraceutical potential in mitigating diet-induced obesity and metabolic disorders. In contrast, many LCFAs, such as palmitic acid, are often associated with pro-inflammatory responses and adverse metabolic effects, highlighting the critical importance of the chain length in functional outcomes.
However, the biological activity of lipids does not increase linearly with the chain length; it often diminishes beyond an optimal point, a phenomenon requiring further mechanistic exploration. Future research should move beyond one-dimensional analyses of the chain length. We propose establishing a multi-dimensional evaluation system that integrates the chain length, degree of unsaturation, and double-bond position to comprehensively predict lipid functionality. Additionally, investigating the synergistic interactions between designed lipids and other dietary components (proteins, carbohydrates) will be crucial for developing effective nutritional strategies to combat metabolic diseases.
Author Contributions
W.-H.S.: Writing—original draft, Conceptualization, Data curation, Visualization. S.L.: Data curation, Visualization. W.D.: Writing—original draft, Data curation. C.-P.T.: Writing—review and editing. Writing—review and editing. Y.-J.X.: Supervision, Writing—original draft, Writing—review and editing, Project administration. All authors have read and agreed to the published version of the manuscript.
Funding
This work was supported by the Pilot Research Program of WIIRI (XD24016), Science and Technology Program of the Nei Monggol (2025YFDZ0048) and 2024 Wuxi Municipal Health Commission Science Popularization Work Creation Project (P202405).
Data Availability Statement
No new data were created or analyzed in this study.
Conflicts of Interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
References
- Berland, C.; Small, D.M.; Luquet, S.; Gangarossa, G. Dietary lipids as regulators of reward processes: Multimodal integration matters. Trends Endocrin. Met. 2021, 32, 693–705. [Google Scholar]
- Yuan, Y.; Li, P.; Li, J.; Zhao, Q.; Chang, Y.; He, X. Protein lipidation in health and disease: Molecular basis, physiological function and pathological implication. Signal Transduct. Target Ther. 2024, 9, 60. [Google Scholar] [PubMed]
- Staggers, J.E.; Fernando-Warnakulasuriya, G.J.; Wells, M.A. Studies on fat digestion, absorption, and transport in the suckling rat. II. Triacylglycerols: Molecular species, stereospecific analysis, and specificity of hydrolysis by lingual lipase. J. Lipid Res. 1981, 22, 675–679. [Google Scholar] [CrossRef]
- Omer, E.; Chiodi, C. Fat digestion and absorption: Normal physiology and pathophysiology of malabsorption, including diagnostic testing. Nutr. Clin. Pract. 2024, 39, S6–S16. [Google Scholar] [CrossRef]
- Zhang, Y.; Qing, X.; Liu, Y.H.; Zhang, X.S.; Jin, W.; Yu, X.M.; Zhang, R.X.; Chao, X.; Yang, X.Y.; Xue, C.Y. Medium-chain triglyceride activated brown adipose tissue and induced reduction of fat mass in C57BL/6J mice fed high-fat diet. Biomed. Environ. Sci. 2015, 28, 97–104. [Google Scholar]
- Sung, M.-H.; Liao, F.-H.; Chien, Y.-W. Medium-chain triglycerides lower blood lipids and body weight in streptozotocin-induced type 2 diabetes rats. Nutrients 2018, 10, 963. [Google Scholar] [CrossRef]
- Jackson, I.J.; Patton, E.E. Red alert about lipid’s role in skin cancer. Nature 2017, 549, 337–339. [Google Scholar]
- Yan, X.; Huang, J.; Huang, L.; Luo, C.; Li, Z.; Xu, P.; Tan, K.; Cheong, K.-L.; Tan, K. Effects of dietary lipids on bioaccessibility and bioavailability of natural carotenoids. Food Sci. Technol. 2024, 200, 116171. [Google Scholar] [CrossRef]
- Baccouch, R.; Shi, Y.; Vernay, E.; Mathelié-Guinlet, M.; Taib-Maamar, N.; Villette, S.; Feuillie, C.; Rascol, E.; Nuss, P.; Lecomte, S.; et al. The impact of lipid polyunsaturation on the physical and mechanical properties of lipid membranes. Biochim. Biophys. Acta Biomembr. 2023, 1865, 184084. [Google Scholar] [CrossRef] [PubMed]
- Moreno, J.J.; Teresa Mitjavila, M. The degree of unsaturation of dietary fatty acids and the development of atherosclerosis (review). J. Nutr. Biochem. 2003, 14, 182–195. [Google Scholar] [CrossRef]
- Cao, M.; Yang, F.; McClements, D.J.; Guo, Y.; Liu, R.; Chang, M.; Wei, W.; Jin, J.; Wang, X. Impact of dietary n-6/n-3 fatty acid ratio of atherosclerosis risk: A review. Prog. Lipid Res. 2024, 95, 101289. [Google Scholar] [CrossRef]
- Luukkonen, P.K.; Sädevirta, S.; Zhou, Y.; Kayser, B.; Ali, A.; Ahonen, L.; Lallukka, S.; Pelloux, V.; Gaggini, M.; Jian, C.; et al. Saturated Fat Is More Metabolically Harmful for the Human Liver Than Unsaturated Fat or Simple Sugars. Diabetes Care 2018, 41, 1732–1739. [Google Scholar] [CrossRef]
- Auñon-Lopez, A.; Alberdi-Cedeño, J.; Martinez-Yusta, A.; Aichner, M.; Suwattanasophon, C.; Pignitter, M. Understanding the bioaccessibility of lipidic compounds and the formation of free oxylipins of encapsulated omega-3-rich marine oils during in vitro digestion. LWT 2025, 234, 118574. [Google Scholar] [CrossRef]
- Morstein, J.; Capecchi, A.; Hinnah, K.; Park, B.; Petit-Jacques, J.; Van Lehn, R.C.; Reymond, J.-L.; Trauner, D. Medium-chain lipid conjugation facilitates cell-permeability and bioactivity. J. Am. Chem. Soc. 2022, 144, 18532–18544. [Google Scholar]
- Zhang, X.; Xue, C.; Xu, Q.; Zhang, Y.; Li, H.; Li, F.; Liu, Y.; Guo, C. Caprylic acid suppresses inflammation via TLR4/NF-κB signaling and improves atherosclerosis in ApoE-deficient mice. Nutr. Metab. 2019, 16, 40. [Google Scholar]
- de Souza, C.O.; Valenzuela, C.A.; Baker, E.J.; Miles, E.A.; Neto, J.C.R.; Calder, P.C. Palmitoleic Acid has Stronger Anti-Inflammatory Potential in Human Endothelial Cells Compared to Oleic and Palmitic Acids. Mol. Nutr. Food Res. 2018, 62, 1800322. [Google Scholar]
- Gallier, S.; Singh, H. The physical and chemical structure of lipids in relation to digestion and absorption. Lipid Technol. 2012, 24, 271–273. [Google Scholar] [CrossRef]
- Ibrahim, K.S.; El-Sayed, E.M. Dietary conjugated linoleic acid and medium-chain triglycerides for obesity management. J. Biosci. 2021, 46, 12. [Google Scholar] [CrossRef] [PubMed]
- Nitbani, F.O.; Jumina; Siswanta, D.; Solikhah, E.N. Isolation and Antibacterial Activity Test of Lauric Acid from Crude Coconut Oil (Cocos nucifera L.). Procedia Chem. 2016, 18, 132–140. [Google Scholar] [CrossRef]
- Yuan, T.; Geng, Z.; Dai, X.; Zhang, X.; Wei, W.; Wang, X.; Jin, Q. Triacylglycerol Containing Medium-Chain Fatty Acids: Comparison of Human Milk and Infant Formulas on Lipolysis during In Vitro Digestion. J. Agric. Food Chem. 2020, 68, 4187–4195. [Google Scholar] [CrossRef]
- Jacobi, S.K.; Odle, J. Nutritional factors influencing intestinal health of the neonate. Adv. Nutr. 2012, 3, 687. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; Qi, C.; Zhang, Y.; Wei, W.; Jin, Q.; Xu, Z.; Tao, G.; Wang, X. Identification and quantification of triacylglycerols in human milk fat using ultra-performance convergence chromatography and quadrupole time-of-flight mass spectrometery with supercritical carbon dioxide as a mobile phase. Food Chem. 2019, 275, 712–720. [Google Scholar]
- Cheng, X.; Jiang, C.; Jin, J.; Jin, Q.; Akoh, C.C.; Wei, W.; Wang, X. Medium-and Long-Chain Triacylglycerol: Preparation, Health Benefits, and Food Utilization. Annu. Rev. Food Sci. Technol. 2024, 15, 381–408. [Google Scholar] [CrossRef] [PubMed]
- Bugaut, M.; Bentéjac, M. Biological effects of short-chain fatty acids in nonruminant mammals. Annu. Rev. Nutr. 1993, 13, 217–241. [Google Scholar] [CrossRef]
- Cong, J.; Zhou, P.; Zhang, R.Y. Intestinal Microbiota-Derived Short Chain Fatty Acids in Host Health and Disease. Nutrients 2022, 14, 1977. [Google Scholar] [CrossRef]
- Chen, T.T.; Long, W.M.; Zhang, C.H.; Liu, S.; Zhao, L.P.; Hamaker, B.R. Fiber-utilizing capacity varies in Prevotella- versus Bacteroides-dominated gut microbiota. Sci. Rep. 2017, 7, 2594. [Google Scholar] [CrossRef]
- Luo, Y.H.; Peng, H.W.; Wright, A.D.; Bai, S.P.; Ding, X.M.; Zeng, Q.F.; Li, H.; Zheng, P.; Su, Z.W.; Cui, R.Y.; et al. Broilers fed dietary vitamins harbor higher diversity of cecal bacteria and higher ratio of Clostridium, Faecalibacterium, and Lactobacillus than broilers with no dietary vitamins revealed by 16S rRNA gene clone libraries. Poul. Sci. 2013, 92, 2358–2366. [Google Scholar] [CrossRef] [PubMed]
- Yuille, S.; Reichardt, N.; Panda, S.; Dunbar, H.; Mulder, I.E. Human gut bacteria as potent class I histone deacetylase inhibitors in vitro through production of butyric acid and valeric acid. PLoS ONE 2018, 13, e0201073. [Google Scholar]
- Yuan, T.L.; Wang, L.; Jin, J.; Mi, L.J.; Pang, J.Z.; Liu, Z.D.; Gong, J.Y.; Sun, C.; Li, J.F.; Wei, W.; et al. Role Medium-Chain Fatty Acids in the Lipid Metabolism of Infants. Front. Nutr. 2022, 9, 804880. [Google Scholar] [CrossRef]
- Dai, X.; Yuan, T.; Zhang, X.; Zhou, Q.; Bi, H.; Yu, R.; Wei, W.; Wang, X. Short-chain fatty acid (SCFA) and medium-chain fatty acid (MCFA) concentrations in human milk consumed by infants born at different gestational ages and the variations in concentration during lactation stages. Food Funct. 2020, 11, 1869–1880. [Google Scholar]
- Altinoz, M.A.; Ozpinar, A.; Seyfried, T.N. Caprylic (Octanoic) Acid as a Potential Fatty Acid Chemotherapeutic for Glioblastoma. Prostaglandins Leukot. Essent. Fat. Acids 2020, 159, 102142. [Google Scholar]
- Lieberman, S.; Enig, M.G.; Preuss, H.G. A Review of Monolaurin and Lauric Acid: Natural Virucidal and Bactericidal Agents. Altern. Complement. Ther. 2006, 12, 310–314. [Google Scholar] [CrossRef]
- Knothe, G.; Dunn, R. A Comprehensive Evaluation of the Melting Points of Fatty Acids and Esters Determined by Differential Scanning Calorimetry. J. Am. Oil Chem. Soc. 2009, 86, 843–856. [Google Scholar] [CrossRef]
- Ravotti, R.; Worlitschek, J.; Pulham, C.R.; Stamatiou, A. Triglycerides as Novel Phase-Change Materials: A Review and Assessment of Their Thermal Properties. Molecules 2020, 25, 5572. [Google Scholar] [CrossRef]
- Pratama, Y.; Seilert, J.; Sadeghpour, A.; Simone, E.; Rappolt, M. Decoding the role of triacylglycerol composition in the milk fat crystallisation behaviour: A study using buffalo milk fat fractions. LWT Food Sci. Technol. 2023, 186, 115274. [Google Scholar] [CrossRef]
- Shin, H.Y.; Kim, J.H.; Kim, J.Y. Effect of fatty acid chain length on physicochemical properties of starch nanocomposites obtained via nanoprecipitation. Int. J. Biol. Macromol. 2023, 230, 123415. [Google Scholar] [CrossRef]
- Ríos-Covián, D.; Ruas-Madiedo, P.; Margolles, A.; Gueimonde, M.; de los Reyes-Gavilán, C.G.; Salazar, N. Intestinal Short Chain Fatty Acids and their Link with Diet and Human Health. Front. Microbiol. 2016, 7, 185. [Google Scholar] [CrossRef]
- Pinto, S.N.; Silva, L.C.; Futerman, A.H.; Prieto, M. Effect of ceramide structure on membrane biophysical properties: The role of acyl chain length and unsaturation. Biochim. Biophys. Acta Biomembr. 2011, 1808, 2753–2760. [Google Scholar] [CrossRef]
- Bianchetti, G.; Azoulay-Ginsburg, S.; Keshet-Levy, N.Y.; Malka, A.; Zilber, S.; Korshin, E.E.; Sasson, S.; De Spirito, M.; Gruzman, A.; Maulucci, G. Investigation of the Membrane Fluidity Regulation of Fatty Acid Intracellular Distribution by Fluorescence Lifetime Imaging of Novel Polarity Sensitive Fluorescent Derivatives. Int. J. Mol. Sci. 2021, 22, 3106. [Google Scholar] [CrossRef]
- Lísa, M.; Velínská, H.; Holčapek, M. Regioisomeric Characterization of Triacylglycerols Using Silver-Ion HPLC/MS and Randomization Synthesis of Standards. Anal. Chem. 2009, 81, 3903–3910. [Google Scholar] [CrossRef]
- Liang, R.; Jiang, Y.; Yokoyama, W.; Yang, C.; Cao, G.; Zhong, F. Preparation of Pickering emulsions with short, medium and long chain triacylglycerols stabilized by starch nanocrystals and their in vitro digestion properties. RSC Adv. 2016, 6, 99496–99508. [Google Scholar] [CrossRef]
- Mu, H.; Høy, C.-E. The digestion of dietary triacylglycerols. Prog. Lipid. Res. 2004, 43, 105–133. [Google Scholar] [CrossRef]
- Perretta, L.; Ouldibbat, L.; Hagadorn, J.I.; Brumberg, H.L. High versus low medium chain triglyceride content of formula for promoting short-term growth of preterm infants. Cochrane Database Syst. Rev. 2021, 2, CD002777. [Google Scholar]
- Wang, Y.; Zhang, T.; Liu, R.; Chang, M.; Wei, W.; Jin, Q.; Wang, X. Reviews of medium-and long-chain triglyceride with respect to nutritional benefits and digestion and absorption behavior. Food Res. Int. 2022, 155, 111058. [Google Scholar] [CrossRef] [PubMed]
- Kiela, P.R.; Ghishan, F.K. Physiology of intestinal absorption and secretion. Best Pract. Res. Clin. Gastroenterol. 2016, 30, 145–159. [Google Scholar] [CrossRef]
- Mansbach, C.M., 2nd; Gorelick, F. Development and physiological regulation of intestinal lipid absorption. II. Dietary lipid absorption, complex lipid synthesis, and the intracellular packaging and secretion of chylomicrons. J. Appl. Physiol. Gastrointest. Liver Physiol. 2007, 293, G645–G650. [Google Scholar] [CrossRef]
- McKimmie, R.L.; Easter, L.; Weinberg, R.B. Acyl chain length, saturation, and hydrophobicity modulate the efficiency of dietary fatty acid absorption in adult humans. J. Appl. Physiol. Gastrointest. Liver Physiol. 2013, 305, G620–G627. [Google Scholar] [CrossRef]
- Yang, F.; Chen, H.; Gao, Y.; An, N.; Li, X.; Pan, X.; Yang, X.; Tian, L.; Sun, J.; Xiong, X.; et al. Gut microbiota-derived short-chain fatty acids and hypertension: Mechanism and treatment. Biomed. Pharmacother. 2020, 130, 110503. [Google Scholar] [CrossRef]
- Schönfeld, P.; Wojtczak, L. Short-and medium-chain fatty acids in energy metabolism: The cellular perspective. J. Lipid Res. 2016, 57, 943–954. [Google Scholar] [CrossRef]
- Huang, X.; Zhou, Y.; Sun, Y.; Wang, Q. Intestinal fatty acid binding protein: A rising therapeutic target in lipid metabolism. Prog. Lipid Res. 2022, 87, 101178. [Google Scholar] [CrossRef] [PubMed]
- Stahl, A.; Hirsch, D.J.; Gimeno, R.E.; Punreddy, S.; Ge, P.; Watson, N.; Patel, S.; Kotler, M.; Raimondi, A.; Tartaglia, L.A.; et al. Identification of the Major Intestinal Fatty Acid Transport Protein. Mol. Cell 1999, 4, 299–308. [Google Scholar] [CrossRef]
- Stahl, A.; Gimeno, R.E.; Tartaglia, L.A.; Lodish, H.F. Fatty acid transport proteins: A current view of a growing family. Trends Endocrin. Met. 2001, 12, 266–273. [Google Scholar] [CrossRef] [PubMed]
- Stone, S.J. Mechanisms of intestinal triacylglycerol synthesis. Biochim. Biophys. Acta Mol. Cell Biol. Lipids 2022, 1867, 159151. [Google Scholar] [CrossRef] [PubMed]
- Canfora, E.E.; Jocken, J.W.; Blaak, E.E. Short-chain fatty acids in control of body weight and insulin sensitivity. Nat. Rev. Endocrinol. 2015, 11, 577–591. [Google Scholar] [CrossRef]
- Lee, Y.Y.; Tang, T.K.; Chan, E.S.; Phuah, E.T.; Lai, O.M.; Tan, C.P.; Wang, Y.; Ab Karim, N.A.; Dian, N.H.M.; Tan, J.S. Medium chain triglyceride and medium-and long chain triglyceride: Metabolism, production, health impacts and its applications—A review. Crit. Rev. Food Sci. Nutr. 2022, 62, 4169–4185. [Google Scholar] [CrossRef]
- Zhu, T.T.; Zhang, Y.; Luo, X.A.; Wang, S.Z.; Jia, M.Q.; Chen, Z.X. Difference in Binding of Long- and Medium-Chain Fatty Acids with Serum Albumin: The Role of Macromolecular Crowding Effect. J. Agric. Food Chem. 2018, 66, 1242–1250. [Google Scholar] [CrossRef]
- Zhou, S.; Wang, Y.; Jacoby, J.J.; Jiang, Y.; Zhang, Y.; Yu, L.L. Effects of Medium- and Long-Chain Triacylglycerols on Lipid Metabolism and Gut Microbiota Composition in C57BL/6J Mice. J. Agric. Food Chem. 2017, 65, 6599–6607. [Google Scholar] [CrossRef]
- Kragh-Hansen, U.; Watanabe, H.; Nakajou, K.; Iwao, Y.; Otagiri, M. Chain length-dependent binding of fatty acid anions to human serum albumin studied by site-directed mutagenesis. J. Mol. Biol. 2006, 363, 702–712. [Google Scholar] [CrossRef] [PubMed]
- Simard, J.R.; Zunszain, P.A.; Ha, C.-E.; Yang, J.S.; Bhagavan, N.V.; Petitpas, I.; Curry, S.; Hamilton, J.A. Locating high-affinity fatty acid-binding sites on albumin by x-ray crystallography and NMR spectroscopy. Proc. Natl. Acad. Sci. USA 2005, 102, 17958–17963. [Google Scholar] [CrossRef]
- Curry, S.; Brick, P.; Franks, N.P. Fatty acid binding to human serum albumin: New insights from crystallographic studies. BBA Mol. Cell Biol. Lipids 1999, 1441, 131–140. [Google Scholar] [CrossRef]
- Ameen, A.O.; Nielsen, S.W.; Kjær, M.W.; Andersen, J.V.; Westi, E.W.; Freude, K.K.; Aldana, B.I. Metabolic preferences of astrocytes: Functional metabolic mapping reveals butyrate outcompetes acetate. J. Cereb. Blood Flow Metab. 2025, 45, 528–541. [Google Scholar] [CrossRef]
- Noguchi, O.; Takeuchi, H.; Kubota, F.; Tsuji, H.; Aoyama, T. Larger diet-induced thermogenesis and less body fat accumulation in rats fed medium-chain triacylglycerols than in those fed long-chain triacylglycerols. J. Nutr. Sci. Vitaminol. 2002, 48, 524–529. [Google Scholar] [CrossRef]
- Lai, H.-S.; Chen, W.-J. Effects of medium-chain and long-chain triacylglycerols in pediatric surgical patients. Nutrition 2000, 16, 401–406. [Google Scholar] [CrossRef] [PubMed]
- Qu, Q.; Zeng, F.; Liu, X.; Wang, Q.J.; Deng, F. Fatty acid oxidation and carnitine palmitoyltransferase I: Emerging therapeutic targets in cancer. Cell Death Dis. 2016, 7, e2226. [Google Scholar] [CrossRef]
- Voss, U.; Turesson, M.F.; Robaye, B.; Boeynaems, J.-M.; Olde, B.; Erlinge, D.; Ekblad, E. The enteric nervous system of P2Y13 receptor null mice is resistant against high-fat-diet- and palmitic-acid-induced neuronal loss. Purinergic Signal. 2014, 10, 455–464. [Google Scholar] [CrossRef] [PubMed]
- Kelly, C.J.; Zheng, L.; Campbell, E.L.; Saeedi, B.; Scholz, C.C.; Bayless, A.J.; Wilson, K.E.; Glover, L.E.; Kominsky, D.J.; Magnuson, A.; et al. Crosstalk between Microbiota-Derived Short-Chain Fatty Acids and Intestinal Epithelial HIF Augments Tissue Barrier Function. Cell Host Microbe 2015, 17, 662–671. [Google Scholar] [CrossRef]
- Rial, S.A.; Karelis, A.D.; Bergeron, K.-F.; Mounier, C. Gut Microbiota and Metabolic Health: The Potential Beneficial Effects of a Medium Chain Triglyceride Diet in Obese Individuals. Nutrients 2016, 8, 281. [Google Scholar] [CrossRef]
- Zhao, Y.; Chen, F.D.; Wu, W.; Sun, M.M.; Bilotta, A.J.; Yao, S.X.; Xiao, Y.; Huang, X.S.; Eaves-Pyles, T.D.; Golovko, G.; et al. GPR43 mediates microbiota metabolite SCFA regulation of antimicrobial peptide expression in intestinal epithelial cells via activation of mTOR and STAT3. Mucosal Immunol. 2018, 11, 752–762. [Google Scholar] [CrossRef]
- Onrust, L.; Van Driessche, K.; Ducatelle, R.; Schwarzer, K.; Haesebrouck, F.; Van Immerseel, F. Valeric acid glyceride esters in feed promote broiler performance and reduce the incidence of necrotic enteritis. Poul. Sci. 2018, 97, 2303–2311. [Google Scholar] [CrossRef]
- Çenesiz, A.A.; Çiftci, İ. Modulatory effects of medium chain fatty acids in poultry nutrition and health. World’s Poult. Sci. J. 2020, 76, 234–248. [Google Scholar] [CrossRef]
- Mosińska, P.; Szczepaniak, A.; Wojciechowicz, T.; Skrzypski, M.; Nowak, K.; Fichna, J. Chain length of dietary fatty acids determines gastrointestinal motility and visceromotor function in mice in a fatty acid binding protein 4-dependent manner. Eur. J. Nutr. 2020, 59, 2481–2496. [Google Scholar] [CrossRef]
- Tian, M.; Li, L.; Tian, Z.; Zhao, H.; Chen, F.; Guan, W.; Zhang, S. Glyceryl butyrate attenuates enterotoxigenic Escherichia coli-induced intestinal inflammation in piglets by inhibiting the NF-κB/MAPK pathways and modulating the gut microbiota. Food Funct. 2022, 13, 6282–6292. [Google Scholar] [PubMed]
- Li, R.; He, Z.; Yan, W.; Yu, H.; Yi, X.; Sha, Y.; Zhang, Q.; Cai, R.; Pang, W. Tricaprylin, a medium-chain triglyceride, aggravates high-fat diet-induced fat deposition but improves intestinal health. Food Funct. 2023, 14, 8797–8813. [Google Scholar] [PubMed]
- Kripke, S.A.; De Paula, J.A.; Berman, J.M.; Fox, A.D.; Rombeau, J.L.; Settle, R.G. Experimental short-bowel syndrome: Effect of an elemental diet supplemented with short-chain triglycerides. Am. J. Clin. Nutr. 1991, 53, 954–962. [Google Scholar] [CrossRef]
- Jeppesen, P.B.; Mortensen, P.B. Colonic digestion and absorption of energy from carbohydrates and medium-chain fat in small bowel failure. J. Parenter. Enteral. Nutr. 1999, 23, S101–S105. [Google Scholar]
- Ren, J.; Sang, Y.; Qin, R.; Su, Y.; Cui, Z.L.; Mang, Z.G.; Li, H.; Lu, S.Y.; Zhang, J.; Cheng, S.; et al. Metabolic intermediate acetyl phosphate modulates bacterial virulence acetylation. Emerg. Microbes Infect. 2019, 8, 55–69. [Google Scholar] [CrossRef]
- Kang, J.J.; Sun, M.Z.; Chang, Y.; Chen, H.M.; Zhang, J.; Liang, X.D.; Xiao, T.F. Butyrate ameliorates colorectal cancer through regulating intestinal microecological disorders. Anti-Cancer Drugs 2023, 34, 227–237. [Google Scholar]
- Yang, H.-T.; Chen, J.-W.; Rathod, J.; Jiang, Y.-Z.; Tsai, P.-J.; Hung, Y.-P.; Ko, W.-C.; Paredes-Sabja, D.; Huang, I.-H. Lauric acid is an inhibitor of Clostridium difficile growth in vitro and reduces inflammation in a mouse infection model. Front. Microbiol. 2018, 8, 2635. [Google Scholar] [CrossRef]
- Chen, Y.; Mai, Q.; Chen, Z.; Lin, T.; Cai, Y.; Han, J.; Wang, Y.; Zhang, M.; Tan, S.; Wu, Z.; et al. Dietary palmitoleic acid reprograms gut microbiota and improves biological therapy against colitis. Gut Microbes 2023, 15, 2211501. [Google Scholar] [CrossRef]
- Rubin, M.; Moser, A.; Vaserberg, N.; Greig, F.; Levy, Y.; Spivak, H.; Ziv, Y.; Lelcuk, S. Structured triacylglycerol emulsion, containing both medium-and long-chain fatty acids, in long-term home parenteral nutrition: A double-blind randomized cross-over study. Nutrition 2000, 16, 95–100. [Google Scholar] [CrossRef]
- St-Onge, M.P.; Ross, R.; Parsons, W.D.; Jones, P.J. Medium-chain triglycerides increase energy expenditure and decrease adiposity in overweight men. Obes. Res. 2003, 11, 395–402. [Google Scholar] [CrossRef] [PubMed]
- Coleman, H.; Quinn, P.; Clegg, M.E. Medium-chain triglycerides and conjugated linoleic acids in beverage form increase satiety and reduce food intake in humans. Nutr. Res. 2016, 36, 526–533. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.-h.; Zhang, Y.; Xu, Q.; Yu, X.-m.; Zhang, X.-s.; Wang, J.; Xue, C.; Yang, X.-y.; Zhang, R.-x.; Xue, C.-y. Increased Norepinephrine by Medium-Chain Triglyceride Attributable to Lipolysis in White and Brown Adipose Tissue of C57BL/6J Mice. Biosci. Biotechol. Biochem. 2012, 76, 1213–1218. [Google Scholar] [CrossRef][Green Version]
- Laeger, T.; Metges, C.C.; Kuhla, B. Role of β-hydroxybutyric acid in the central regulation of energy balance. Appetite 2010, 54, 450–455. [Google Scholar] [CrossRef]
- Deng, M.; Qu, F.; Chen, L.; Liu, C.; Zhang, M.; Ren, F.; Guo, H.; Zhang, H.; Ge, S.; Wu, C.; et al. SCFAs alleviated steatosis and inflammation in mice with NASH induced by MCD. J. Endocrinol. 2020, 245, 425–437. [Google Scholar] [CrossRef]
- Li, F.; Tai, L.; Sun, X.; Lv, Z.; Tang, W.; Wang, T.; Zhao, Z.; Gong, D.; Ma, S.; Tang, S.; et al. Molecular recognition and activation mechanism of short-chain fatty acid receptors FFAR2/3. Cell Res. 2024, 34, 323–326. [Google Scholar] [CrossRef] [PubMed]
- Shimizu, H.; Masujima, Y.; Ushiroda, C.; Mizushima, R.; Taira, S.; Ohue-Kitano, R.; Kimura, I. Dietary short-chain fatty acid intake improves the hepatic metabolic condition via FFAR3. Sci. Rep. 2019, 9, 16574. [Google Scholar] [CrossRef]
- Kimura, I.; Ozawa, K.; Inoue, D.; Imamura, T.; Kimura, K.; Maeda, T.; Terasawa, K.; Kashihara, D.; Hirano, K.; Tani, T.; et al. The gut microbiota suppresses insulin-mediated fat accumulation via the short-chain fatty acid receptor GPR43. Nat. Commun. 2013, 4, 1829. [Google Scholar]
- Jiang, X.; Zhang, Y.; Zhang, H.; Zhang, X.; Yin, X.; Yuan, F.; Wang, S.; Tian, Y. Acetate suppresses myocardial contraction via the short-chain fatty acid receptor GPR43. Front. Physiol. 2022, 13, 1111156. [Google Scholar] [CrossRef] [PubMed]
- Deng, F.; Zhang, L.Q.; Wu, H.; Chen, Y.; Yu, W.Q.; Han, R.H.; Han, Y.; Zhang, X.Q.; Sun, Q.S.; Lin, Z.B.; et al. Propionate alleviates myocardial ischemia-reperfusion injury aggravated by Angiotensin II dependent on caveolin-1/ACE2 axis through GPR41. Int. J. Biol. Sci. 2022, 18, 858–872. [Google Scholar] [CrossRef] [PubMed]
- Haynes, V.R.; Michael, N.J.; van den Top, M.; Zhao, F.-Y.; Brown, R.D.; De Souza, D.; Dodd, G.T.; Spanswick, D.; Watt, M.J. A Neural basis for Octanoic acid regulation of energy balance. Mol. Metab. 2020, 34, 54–71. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Yuan, C.; Yang, B.; Pang, H.; Li, W.; Li, M.; Tang, Y.; Ma, D.; Xie, J.; Wang, J.; et al. Caprylic Acid (FFA C8:0) promotes the progression of prostate cancer by up-regulating G protein-coupled receptor 84/Krüppel-like factor 7. BMC Cancer 2023, 23, 426. [Google Scholar] [CrossRef]
- Oh, D.Y.; Talukdar, S.; Bae, E.J.; Imamura, T.; Morinaga, H.; Fan, W.; Li, P.; Lu, W.J.; Watkins, S.M.; Olefsky, J.M. GPR120 is an omega-3 fatty acid receptor mediating potent anti-inflammatory and insulin-sensitizing effects. Cell 2010, 142, 687–698. [Google Scholar] [CrossRef]
- Im, D.S. FFA4 (GPR120) as a fatty acid sensor involved in appetite control, insulin sensitivity and inflammation regulation. Mol. Aspects Med. 2018, 64, 92–108. [Google Scholar] [CrossRef]
- Nakajima, S.; Demers, G.; Machuca-Parra, A.I.; Pour, Z.D.; Bairamian, D.; Bouyakdan, K.; Fisette, A.; Kabahizi, A.; Robb, J.; Rodaros, D.; et al. Central activation of the fatty acid sensor GPR120 suppresses microglia reactivity and alleviates sickness- and anxiety-like behaviors. J. Neuroinflamm. 2023, 20, 302. [Google Scholar] [CrossRef] [PubMed]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.