N-Alkyl Derivatives of Deoxynojirimycin (DNJ) as Antiviral Agents: Overview and Update
Abstract
1. Introduction
2. A Brief History of the Discovery of the Antiviral Activity of N-Alkyl Analogs of DNJ
3. An Overview of N-Alkyl Derivatives of DNJ
3.1. Most Common N-Alkyl Derivatives of DNJ as Antivirals
3.1.1. N-Butyldeoxynojirimycin (N-Butyl-DNJ or NB-DNJ or UV-1 or AT2221, Miglustat)
3.1.2. N-Nonyl-DNJ (NN-DNJ or UV-2)
3.1.3. N-7-Oxadecyl-DNJ (UV-3) (SP116)
3.1.4. N-9′-Methoxynonyl-1-deoxynojirimycin (MON-DNJ or UV-4)
3.1.5. N-9′-Methoxynonyl-1-deoxynojirimycin Hydrochloride (UV-4B)
3.1.6. N-[N-(4-Azido-2-nitrophenyl)-6-aminohexyl]-1-deoxynojirimycin (NAP-DNJ or UV-5)
3.2. N-Alkyl Derivatives Synthesized in Recent Years
3.2.1. 5′-Tocopheroxypentyl-DNJ (ToP-DNJ, 1)
3.2.2. 2-(3,4-Dihydroxyphenyl)-7-hydroxy-5-((10-((2R,3R,4R,5S)-3,4,5-trihydroxy-2-(hydroxymethyl)piperidin-1-yl)decyl)oxy)-4H-chromen-4-one (DNJ-20, 2)
3.2.3. N-8′-(2′′-Tetrahydrofuranyl)octyldeoxynojirimycin (2THO-DNJ or UV-12, 3) and (N-(8′-Ethoxyoctyl)deoxynojirimycin) (EOO-DNJ, 4)
3.2.4. UV-5 like DNJ-Valiolamine Derivatives (EB-0128, 5; EB-0442, 6; EB-0450, 7; and EB-0686, 8)
3.2.5. (2R,3R,4R,5S)-1–(5-(Adamantan-1-ylmethoxy)pentyl)-2-(hydroxymethyl)piperidine-3,4,5-triol (9) and (2R,3R,4R,5S)-1–(5-(Bicyclo[1.1.1]pentan-1-yl)pentyl)-3,4,5-trihydroxy-2-(hydroxymethyl)piperidin-1-ium Chloride (10)
3.2.6. (3-(tert-Butyl)-1-cyclohexyl-1-(6-((2R,3R,4R,5S)-3,4,5-trihydroxy-2-(hydroxymethyl)piperidin-1-yl)hexyl)urea (IHVR-19029 or BSBI-1902, 11); (2R,3R,4R,5S)-1-(6-(2,5-Difluorophenoxy)hexyl)-2-(hydroxymethyl)piperidine-3,4,5-triol, phenylether DNJ (IHVR-11029, 12) and N-Cyclohexyl-N-(6-((2R,3R,4R,5S)-3,4,5-trihydroxy-2-(hydroxymethyl)piperidin-1-yl)hexyl)pivalamide, Pivalamide DNJ (IHVR-17028, 13)
4. Synthesis of Deoxynojirimycin and Its Derivatives
5. Mechanisms of Action Proposed for the GluII Inhibition of Iminosugars
6. Considerations on Toxicity
7. Conclusions
8. Future Perspectives
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| BVDV | Bovine viral diarrhea virus |
| CC50 | Half maximum cell cytotoxicity |
| CCHFV | Crimean–Congo hemorrhagic fever virus |
| CNX | Calnexin |
| COVID-19 | Coronavirus Disease 2019 |
| CRT | Calreticulin |
| DENV | Dengue virus |
| DENV-2 | Dengue virus serotype 2 |
| DNJ | Deoxynojirimycin |
| EC50 | Half maximal effective concentration |
| EBOV | Ebola virus |
| ER | Endoplasmic reticulum |
| ERQC | Endoplasmic reticulum quality control |
| GluI | α-Glucosidase I |
| GluII | α-Glucosidase II |
| HAZV | Hazara virus |
| HBV | Hepatitis B virus |
| HCV | Hepatitis C virus |
| HIV | Human immunodeficiency virus |
| HPV | Human papillomavirus |
| IC50 | Half maximal inhibitory concentration |
| MARV | Monocyte-derived macrophages |
| PsV | SARS-CoV-2 pseudovirus |
| SARS-CoV-1 | Severe acute respiratory syndrome coronavirus 1 |
| SARS-CoV-2 | Severe acute respiratory syndrome coronavirus 2 |
| TCRV | Tacaribe virus |
| VSV | Vesicular stomatitis virus |
| WNV | West Nile virus |
References
- Flanagan, J.J.; Rossi, B.; Tang, K.; Wu, X.; Mascioli, K.; Donaudy, F.; Tuzzi, M.R.; Fontana, F.; Cubellis, M.V.; Porto, C.; et al. The Pharmacological Chaperone 1-Deoxynojirimycin Increases the Activity and Lysosomal Trafficking of Multiple Mutant Forms of Acid Alpha-glucosidase. Hum. Mutat. 2009, 30, 1683–1692. [Google Scholar] [CrossRef]
- Tricase, A.F.; Cavalluzzi, M.M.; Catalano, A.; De Bellis, M.; De Palma, A.; Basile, G.; Sinicropi, M.S.S.; Lentini, G. Insights into the Activities and Usefulness of Deoxynojirimycin and Morus alba: A Comprehensive Review. Molecules 2025, 30, 3213. [Google Scholar] [CrossRef]
- Datema, R.; Romero, P.A.; Rott, R.; Schwarz, R.T. On the Role of Oligosaccharide Trimming in the Maturation of Sindbis and Influenza Virus. Arch. Virol. 1984, 81, 25–39. [Google Scholar] [CrossRef]
- Schlesinger, S.; Malfer, C.; Schlesinger, M.J. The Formation of Vesicular Stomatitis Virus (San Juan Strain) Becomes Temperature-Sensitive when Glucose Residues are Retained on the Oligosaccharides of the Glycoprotein. J. Biol. Chem. 1984, 259, 7597–7601. [Google Scholar] [CrossRef] [PubMed]
- McDowell, W.; Romero, P.A.; Datema, R.; Schwarz, R.T. Glucose Trimming and Mannose Trimming Affect Different Phases of the Maturation of Sindbis Virus in infected BHK Cells. Virology 1984, 161, 37–44. [Google Scholar] [CrossRef] [PubMed]
- Datema, R.; Olofsson, S.; Romero, P.A. Inhibitors of Protein Glycosylation and Glycoprotein Processing in Viral Systems. Pharmacol. Ther. 1987, 33, 221–286. [Google Scholar] [CrossRef]
- Oo, T.Z.M.; Wuttiin, Y.; Choocheep, K.; Kumsaiyai, W.; Bunpo, P.; Cressey, R. Exploring Small-Molecule Inhibitors of Glucosidase II: Advances, Challenges, and Therapeutic Potential in Cancer and Viral Infection. Int. J. Mol. Sci. 2025, 26, 11867. [Google Scholar] [CrossRef]
- Sunkara, P.S.; Bowlin, T.L.; Liu, P.S.; Sjoerdsma, A. Antiretroviral Activity of Castanospermine and Deoxynojirimycin, Specific Inhibitors of Glycoprotein Processing. Biochem. Biophys. Res. Commun. 1987, 148, 206–210. [Google Scholar] [CrossRef] [PubMed]
- Gruters, R.A.; Neefjes, J.J.; Tersmette, M.; de Goede, R.E.Y.; Tulp, A.; Huisman, H.G.; Miedema, F.; Ploegh, H.L. Interference with HIV-induced Syncytium Formation and Viral Infectivity by Inhibitors of Trimming Glucosidase. Nature 1987, 330, 74–77. [Google Scholar] [CrossRef]
- Papandréou, M.J.; Barbouche, R.; Guieu, R.; Kieny, M.P.; Fenouillet, E. The Alpha-glucosidase Inhibitor 1-Deoxynojirimycin blocks Human Immunodeficiency Virus Envelope Glycoprotein-Mediated Membrane Fusion at the CXCR4 Binding Step. Mol. Pharmacol. 2002, 61, 186–193. [Google Scholar] [CrossRef]
- Bhushan, G.; Lim, L.; Bird, I.; Chothe, S.K.; Nissly, R.H.; Kuchipudi, S.V. Iminosugars with Endoplasmic Reticulum α-Glucosidase Inhibitor Activity Inhibit ZIKV Replication and Reverse Cytopathogenicity in Vitro. Front. Microbiol. 2020, 11, 531. [Google Scholar] [CrossRef]
- Iftikhar, M.; Lu, Y.; Zhou, M. An Overview of Therapeutic Potential of N-alkylated 1-Deoxynojirimycin Congeners. Carbohydr. Res. 2021, 504, 108317. [Google Scholar] [CrossRef]
- Wang, H.; Shen, Y.; Zhao, L.; Ye, Y. 1-Deoxynojirimycin and Its Derivatives: A Mini Review of the Literature. Curr. Med. Chem. 2021, 28, 628–643. [Google Scholar] [CrossRef]
- Liu, Q.; Liu, Y.; Liu, T.; Fan, J.; Xia, Z.; Zhou, Y.; Deng, X. Expanding Horizons of Iminosugars as Broad-Spectrum Anti-Virals: Mechanism, Efficacy and Novel Developments. Nat. Prod. Bioprospect. 2024, 14, 55. [Google Scholar] [CrossRef]
- Pathak, S.R.; Senwar, K.R.; Sharma, K.N. Alpha-glucosidase in Diabetes Mellitus. In Diabetes Mellitus; Academic Press: Cambridge, MA, USA, 2025; Chapter 4; pp. 63–78. [Google Scholar] [CrossRef]
- Kim, H.M.; Hyun, C.G. Miglitol, an Oral Antidiabetic Drug, Downregulates Melanogenesis in B16F10 Melanoma Cells through the PKA, MAPK, and GSK3β/β-catenin Signaling Pathways. Molecules 2022, 28, 115. [Google Scholar] [CrossRef]
- Liu, J.; Li, W.; Jiao, R.; Liu, Z.; Zhang, T.; Chai, D.; Meng, L.; Yang, Z.; Liu, Y.; Wu, H.; et al. Miglustat Ameliorates Isoproterenol-Induced Cardiac Fibrosis via Targeting UGCG. Mol. Med. 2025, 31, 55. [Google Scholar] [CrossRef] [PubMed]
- Tang, L.; Xu, Y.; He, J.; Huang, G.; Jiang, X.; Li, Y.; Li, H.; Zhang, R.; Gui, Z. 1-Deoxynojirimycin Derivative Containing Tegafur Induced HCT-116 Cell Apoptosis through Mitochondrial Dysfunction and Oxidative Stress Pathway. ACS Med. Chem. Lett. 2024, 15, 1947–1952. [Google Scholar] [CrossRef] [PubMed]
- Esposito, A.; Rossi, A.; Stabile, M.; Pinto, G.; De Fino, I.; Melessike, M.; Tamanini, A.; Cabrini, G.; Lippi, G.; Aureli, M.; et al. Assessing the Potential of N-Butyl-l-deoxynojirimycin (L-NBDNJ) in Models of Cystic Fibrosis as a Promising Antibacterial Agent. ACS Pharmacol. Transl. Sci. 2024, 7, 1807–1822. [Google Scholar] [CrossRef]
- Tsuda, S.; Sato, T.; Kako, S.; Tabuchi, M.; Sugita, Y.; Maeda, H.; Hamamura, K.; Miyazawa, K. Miglustat Suppresses Alveolar Bone Resorption in Mouse Models of Periodontitis. J. Hard Tissue Biol. 2025, 34, 1–8. [Google Scholar] [CrossRef]
- Lee, H.; Kim, D.; Kim, B.; Joung, D.; Jeon, J.; Kim, T.O.; Youn, H.; Youn, B. UDP-Glucose Ceramide Glucosyltransferase Promotes Radioresistance via Membrane Reorganization to Maintain Redox Balance in Glioblastoma: Molecular Diagnostics. Br. J. Cancer 2025, 133, 1720–1732. [Google Scholar] [CrossRef] [PubMed]
- Kang, J.W.; Chan, K.W.K.; Vasudevan, S.G.; Low, J.G. α-Glucosidase Inhibitors as Broad-Spectrum Antivirals: Current Knowledge and Future Prospects. Antivir. Res. 2025, 238, 106147. [Google Scholar] [CrossRef] [PubMed]
- Alonzi, D.S.; Scott, K.A.; Dwek, R.A.; Zitzmann, N. Iminosugar Antivirals: The Therapeutic Sweet Spot. Biochem. Soc. Trans. 2017, 45, 571–582. [Google Scholar] [CrossRef] [PubMed]
- Chang, J.; Block, T.M.; Guo, J.T. Antiviral Therapies Targeting Host ER Alpha-Glucosidases: Current Status and Future Directions. Antivir. Res. 2013, 99, 251–260. [Google Scholar] [CrossRef]
- Ji, X.; Li, Z. Medicinal Chemistry Strategies toward Host Targeting Antiviral Agents. Med. Res. Rev. 2020, 40, 1519–1557. [Google Scholar] [CrossRef]
- Dahiya, J.; Kumar, G.; Narula, A.K. The Multifaceted Potential of Azasugars: Synthetic Approaches, Molecular Interactions and Therapeutic Usage. J. Chem. Lett. 2025, 6, 79–92. [Google Scholar] [CrossRef]
- Gu, B.; Mason, P.; Wang, L.; Norton, P.; Bourne, N.; Moriarty, R.; Mehta, A.; Despande, M.; Shah, R.; Block, T. Antiviral Profiles of Novel Iminocyclitol Compounds against Bovine Viral Diarrhea Virus, West Nile Virus, Dengue Virus and Hepatitis B Virus. Antiviral Chem. Chemother. 2007, 18, 49–59. [Google Scholar] [CrossRef]
- Timokhova, A.V.; Bakinovskii, L.V.; Zinin, A.I.; Popenko, V.I.; Ivanov, A.V.; Rubtsov, P.M.; Kochetkov, S.N.; Belzhelarskaya, S.N. Effect of Deoxynojirimycin Derivatives on Morphogenesis of Hepatitis C Virus. Mol. Biol. 2012, 46, 579–587. [Google Scholar] [CrossRef]
- Williams, S.J.; Goddard-Borger, E.D. α-Glucosidase Inhibitors as Host-Directed Antiviral Agents with Potential for the Treatment of COVID-19. Biochem. Soc. Trans. 2020, 48, 1287–1295. [Google Scholar] [CrossRef] [PubMed]
- Fukushi, M.; Yoshinaka, Y.; Matsuoka, Y.; Hatakeyama, S.; Ishizaka, Y.; Kirikae, T.; Sasazuki, T.; Miyoshi-Akiyama, T. Monitoring of S Protein Maturation in the Endoplasmic Reticulum by Calnexin is Important for the Infectivity of Severe Acute Respiratory Syndrome Coronavirus. J. Virol. 2012, 86, 11745–11753. [Google Scholar] [CrossRef]
- Brun, J.; Arman, B.Y.; Hill, M.L.; Kiappes, J.L.; Alonzi, D.S.; Makower, L.L.; Witt, K.D.; Gileadi, C.; Rangel, V.; Dwek, R.A.; et al. Assessment of Repurposed Compounds against Coronaviruses Highlights the Antiviral Broad-Spectrum Activity of Host-Targeting Iminosugars and Confirms the Activity of Potent Directly Acting Antivirals. Antivir. Res. 2025, 237, 106123. [Google Scholar] [CrossRef]
- Hubbard, S.C.; Robbins, P.W. Synthesis and Processing of Protein-Linked Oligosaccharides In Vivo. J. Biol. Chem. 1979, 254, 4568–4576. [Google Scholar] [CrossRef]
- Romero, P.A.; Datema, R.; Schwarz, R.T. N-Methyl-1-deoxynojirimycin, a Novel Inhibitor of Glycoprotein Processing, and Its Effect on Fowl Plague Virus Maturation. Virology 1983, 130, 238–242. [Google Scholar] [CrossRef]
- Hettkamp, H.; Legler, G.; Bause, E. Purification by Affinity Chromatography of Glucosidase I, an Endoplasmic Reticulum Hydrolase Involved in the Processing of Asparagine-linked Oligosaccharides. Eur. J. Biochem. 1984, 142, 85–90. [Google Scholar] [CrossRef]
- Tan, A.; Van den Broek, L.; Van Boeckel, S.; Ploegh, H.; Bolscher, J. Chemical Modification of the Glucosidase Inhibitor 1-Deoxynojirimycin. Structure-Activity Relationships. J. Biol. Chem. 1991, 266, 14504–14510. [Google Scholar] [CrossRef] [PubMed]
- Block, T.M.; Lu, X.; Platt, F.M.; Foster, G.R.; Gerlich, W.H.; Blumberg, B.S.; Dwek, R.A. Secretion of Human Hepatitis B Virus Is Inhibited by the Imino Sugar N-Butyldeoxynojirimycin. Proc. Natl. Acad. Sci. USA 1994, 91, 2235–2239. [Google Scholar] [CrossRef]
- Fischer, P.; Collin, M.; Karlsson, G.; James, W.; Butters, T.; Davis, S.; Gordon, S.; Dwek, R.; Platt, F. The Glucosidase Inhibitor N-Butyldeoxynojirimycin Inhibits Human Immunodeficiency Virus Entry at the Level of Post-CD4 Binding. J. Virol. 1995, 69, 5791–5797. [Google Scholar] [CrossRef]
- Block, T.M.; Lu, X.; Mehta, A.S.; Blumberg, B.S.; Tennant, B.; Ebling, M.; Korba, B.; Lansky, D.M.; Jacob, G.S.; Dwek, R.A. Treatment of Chronic Hepadnavirus Infection in a Woodchuck Animal Model with an Inhibitor of Protein Folding and Trafficking. Nat. Med. 1998, 4, 610–614. [Google Scholar] [CrossRef]
- Mehta, A.; Zitzmann, N.; Rudd, P.M.; Block, T.M.; Dwek, R.A. α-Glucosidase Inhibitors as Potential Broad Based Anti-Viral Agents. FEBS Lett. 1998, 430, 17–22. [Google Scholar] [CrossRef] [PubMed]
- Zitzmann, N.; Mehta, A.S.; Carrouée, S.; Butters, T.D.; Platt, F.M.; McCauley, J.; Blumberg, B.S.; Dwek, R.A.; Block, T.M. Imino Sugars Inhibit the Formation and Secretion of Bovine Viral Diarrhea Virus, a Pestivirus Model of Hepatitis C Virus: Implications for the Development of Broad Spectrum Anti-Hepatitis Virus Agents. Proc. Natl. Acad. Sci. USA 1999, 96, 11878–11882. [Google Scholar] [CrossRef] [PubMed]
- Mehta, A.; Ouzounov, S.; Jordan, R.; Simsek, E.; Lu, X.; Moriarty, R.M.; Jacob, G.; Dwek, R.A.; Block, T.M. Imino Sugars That Are Less Toxic But More Potent As Antivirals, In Vitro, Compared with N-n-nonyl DNJ. Antivir. Chem. Chemother. 2002, 13, 299–304. [Google Scholar] [CrossRef]
- Mehta, A.; Conyers, B.; Tyrrell, D.L.J.; Walters, K.-A.; Tipples, G.A.; Dwek, R.A.; Block, T.M. Structure-Activity Relationship of a New Class of Anti-Hepatitis B Virus Agents. Antimicrob. Agents Chemother. 2002, 46, 4004–4008. [Google Scholar] [CrossRef]
- Sayce, A.C.; Alonzi, D.S.; Killingbeck, S.S.; Tyrrell, B.E.; Hill, M.L.; Caputo, A.T.; Iwaki, R.; Kinami, K.; Ide, D.; Kiappes, J.L.; et al. Iminosugars Inhibit Dengue Virus Production via Inhibition of ER Alpha-Glucosidases—Not Glycolipid Processing Enzymes. PLoS Negl. Trop. Dis. 2016, 10, e0004524. [Google Scholar] [CrossRef]
- Warfield, K.L.; Warren, T.K.; Qiu, X.; Wells, J.; Mire, C.E.; Geisbert, J.B.; Stuthman, K.S.; Garza, N.L.; Van Tongeren, S.A.; Shurtleff, A.C.; et al. Assessment of the Potential for Host-Targeted Iminosugars UV-4 and UV-5 Activity against Filovirus Infections In Vitro and In Vivo. Antivir. Res. 2017, 138, 22–31. [Google Scholar] [CrossRef]
- Durantel, D.; Branza-Nichita, N.; Carrouee-Durantel, S.; Butters, T.D.; Dwek, R.A.; Zitzmann, N. Study of the Mechanism of Antiviral Action of Iminosugar Derivatives against Bovine Viral Diarrhea Virus. J. Virol. 2001, 75, 8987–8998. [Google Scholar] [CrossRef] [PubMed]
- Wu, S.F.; Lee, C.J.; Liao, C.L.; Dwek, R.A.; Zitzmann, N.; Lin, Y.L. Antiviral Effects of an Iminosugar Derivative on Flavivirus Infections. J. Virol. 2002, 76, 3596–3604. [Google Scholar] [CrossRef] [PubMed]
- Howe, J.D.; Smith, N.; Lee, M.J.R.; Ardes-Guisot, N.; Vauzeilles, B.; Désiré, J.; Baron, A.; Blériot, Y.; Sollogoub, M.; Alonzi, D.S.; et al. Novel Imino Sugar α-Glucosidase Inhibitors as Antiviral Compounds. Bioorg. Med. Chem. 2013, 21, 4831–4838. [Google Scholar] [CrossRef] [PubMed]
- Yu, W.; Gill, T.; Wang, L.; Du, Y.; Ye, H.; Qu, X.; Guo, J.T.; Cuconati, A.; Zhao, K.; Block, T.M.; et al. Design, Synthesis, and Biological Evaluation of N-alkylated Deoxynojirimycin (DNJ) Derivatives for the Treatment of Dengue Virus Infection. J. Med. Chem. 2012, 55, 6061–6075. [Google Scholar] [CrossRef][Green Version]
- Qu, X.; Pan, X.; Weidner, J.; Yu, W.; Alonzi, D.; Xu, X.; Butters, T.; Block, T.; Guo, J.T.; Chang, J. Inhibitors of Endoplasmic Reticulum Alpha-Glucosidases Potently Suppress Hepatitis C Virus Virion Assembly and Release. Antimicrob. Agents Chemother. 2011, 55, 1036–1044. [Google Scholar] [CrossRef]
- Evans DeWald, L.; Starr, C.; Butters, T.; Treston, A.; Warfield, K.L. Iminosugars: A Host-Targeted Approach to Combat Flaviviridae Infections. Antivir. Res. 2020, 184, 104881. [Google Scholar] [CrossRef]
- Miller, J.L.; Tyrrell, B.E.; Zitzmann, N. Mechanisms of Antiviral Activity of Iminosugars against Dengue Virus. Adv. Exp. Med. Biol. 2018, 1062, 277–301. [Google Scholar] [CrossRef]
- Kiappes, J.L.; Hill, M.L.; Alonzi, D.S.; Miller, J.L.; Iwaki, R.; Sayce, A.C.; Caputo, A.T.; Kato, A.; Zitzmann, N. ToP-DNJ, a Selective Inhibitor of Endoplasmic Reticulum α-Glucosidase II Exhibiting Antiflaviviral Activity. ACS Chem. Biol. 2018, 13, 60–65. [Google Scholar] [CrossRef]
- Catalano, A.; Iacopetta, D.; Ceramella, J.; Maio, A.C.; Basile, G.; Giuzio, F.; Bonomo, M.G.; Aquaro, S.; Walsh, T.J.; Sinicropi, M.S.; et al. Are Nutraceuticals Effective in COVID-19 and Post-COVID Prevention and Treatment? Foods 2022, 11, 2884. [Google Scholar] [CrossRef]
- Catalano, A. COVID-19: Could Irisin Become the Handyman Myokine of the 21st Century? Coronaviruses 2020, 1, 32–41. [Google Scholar] [CrossRef]
- Asano, N.; Kato, A.; Watson, A.A. Therapeutic Applications of Sugar-mimicking Glycosidase Inhibitors. Mini Rev. Med. Chem. 2001, 1, 145–154. [Google Scholar] [CrossRef]
- Miller, J.L.; Lachica, R.; Sayce, A.C.; Williams, J.P.; Bapat, M.; Dwek, R.; Beatty, P.R.; Harris, E.; Zitzmann, N. Liposome-mediated Delivery of Iminosugars Enhances Efficacy against Dengue Virus In Vivo. Antimicrob. Agents Chemother. 2012, 56, 6379–6386. [Google Scholar] [CrossRef] [PubMed]
- Perry, S.T.; Buck, M.D.; Plummer, E.M.; Penmasta, R.A.; Batra, H.; Stavale, E.J.; Warfield, K.L.; Dwek, R.A.; Butters, T.D.; Alonzi, D.S.; et al. An Iminosugar with Potent Inhibition of Dengue Virus Infection In Vivo. Antivir. Res. 2013, 98, 35–43. [Google Scholar] [CrossRef]
- Rajasekharan, S.; Bonotto, R.M.; Alves, L.N.; Kazungu, Y.; Poggianella, M.; Martinez-Orellana, P.; Skoko, N.; Polez, S.; Marcello, A. Inhibitors of Protein Glycosylation Are Active against the Coronavirus Severe Acute Respiratory Syndrome Coronavirus SARS-CoV-2. Viruses 2021, 13, 808. [Google Scholar] [CrossRef] [PubMed]
- Wetherill, L.F.; Wasson, C.W.; Swinscoe, G.; Kealy, D.; Foster, R.; Griffin, S.; Macdonald, A. Alkyl-imino Sugars Inhibit the Pro-Oncogenic Ion Channel Function of Human Papillomavirus (HPV) E5. Antivir. Res. 2018, 158, 113–121. [Google Scholar] [CrossRef] [PubMed]
- Perera, N.; Brun, J.; Alonzi, D.S.; Tyrrell, B.E.; Miller, J.L.; Zitzmann, N. Antiviral Effects of Deoxynojirimycin (DNJ)-Based Iminosugars in Dengue Virus-Infected Primary Dendritic Cells. Antivir. Res. 2022, 199, 105269. [Google Scholar] [CrossRef]
- Hussain, S.; Miller, J.L.; Harvey, D.J.; Gu, Y.; Rosenthal, P.B.; Zitzmann, N.; McCauley, J.W. Strain-specific Antiviral Activity of Iminosugars against Human Influenza A Viruses. J. Antimicrob. Chemother. 2015, 70, 136–152. [Google Scholar] [CrossRef]
- Warfield, K.L.; Plummer, E.M.; Sayce, A.C.; Alonzi, D.S.; Tang, W.; Tyrrell, B.E.; Hill, M.L.; Caputo, A.T.; Killingbeck, S.S.; Beatty, P.R.; et al. Inhibition of Endoplasmic Reticulum Glucosidases is Required for In Vitro and In Vivo Dengue Antiviral Activity by the Iminosugar UV-4. Antivir. Res. 2016, 129, 93–98. [Google Scholar] [CrossRef]
- Franco, E.J.; Warfield, K.L.; Brown, A.N. UV-4B Potently Inhibits Replication of Multiple SARS-CoV-2 Strains in Clinically Relevant Human Cell Lines. Front. Biosci.-Landmark Ed. 2022, 27, 3. [Google Scholar] [CrossRef]
- Karade, S.S.; Franco, E.J.; Rojas, A.C.; Hanrahan, K.C.; Kolesnikov, A.; Yu, W.; MacKerell, A.D., Jr.; Hill, D.C.; Weber, D.J.; Brown, A.N.; et al. Structure-Based Design of Potent Iminosugar Inhibitors of Endoplasmic Reticulum α-Glucosidase I with Anti-SARS-CoV-2 Activity. J. Med. Chem. 2023, 66, 2744–2760. [Google Scholar] [CrossRef]
- Rawlings, A.J.; Lomas, H.; Pilling, A.W.; Lee, M.J.-R.; Alonzi, D.S.; Rountree, J.S.S.; Jenkinson, S.F.; Fleet, G.W.J.; Dwek, R.A.; Jones, J.H.; et al. Synthesis and Biological Characterisation of Novel N-Alkyl-Deoxynojirimycin α-Glucosidase Inhibitors. ChemBioChem 2009, 10, 1101–1105. [Google Scholar] [CrossRef] [PubMed]
- Ficicioglu, C. Review of Miglustat for Clinical Management in Gaucher Disease Type 1. Ther. Clin. Risk Manag. 2008, 4, 425–431. [Google Scholar] [CrossRef] [PubMed]
- Lyseng-Williamson, K.A. Miglustat: A Review of its Use in Niemann-Pick Disease Type C. Drugs 2014, 74, 61–74. [Google Scholar] [CrossRef] [PubMed]
- Patterson, M.C.; Vecchio, D.; Prady, H.; Abel, L.; Wraith, J.E. Miglustat for Treatment of Niemann-Pick C Disease: A Randomised Controlled Study. Lancet Neurol. 2007, 6, 765–772. [Google Scholar] [CrossRef]
- Mishra, N.; Tiwari, V.K.; Schmidt, R.R. Recent Trends and Challenges on Carbohydrate-Based Molecular Scaffolding: General Consideration toward Impact of Carbohydrates in Drug Discovery and Development. In Carbohydrates in Drug Discovery and Development; Elsevier: Amsterdam, The Netherlands, 2020; Chapter 1; pp. 1–69. [Google Scholar] [CrossRef]
- Kishnani, P.; Schoser, B.; Bratkovic, D.; Byrned, B.J.; Clemense, P.R.; Goker-Alpanf, O.; Mingg, X.; Robertsh, M.; Schwenkreisi, P.; Sivakumarj, K.; et al. First-in-human Study of Advanced and Targeted Acid α-glucosidase (AT-GAA) (ATB200/AT2221) in Patients with Pompe Disease: Preliminary Functional Assessment Results from the ATB200-02 Trial. Mol. Genet. Metab. 2019, 126, S86. [Google Scholar] [CrossRef]
- Roberts, M.E.; Proskorovsky, I.; Guyot, P.; Shukla, P.; Thibault, N.; Hamed, A.; Pulikottil-Jacob, R.; O’Callaghan, L.; Pollissard, L. An Indirect Treatment Comparison of Avalglucosidase Alfa versus Cipaglucosidase Alfa Plus Miglustat in Patients with Late-Onset Pompe Disease. Adv. Ther. 2025, 42, 5578–5599. [Google Scholar] [CrossRef]
- Schoser, B.; Attarian, S.; Graham, R.; Holdbrook, F.; Goldman, M.; Díaz-Manera, J.; ATB200-03 study group. Challenges in Multinational Rare Disease Clinical Studies during COVID-19: Regulatory Assessment of Cipaglucosidase Alfa Plus Miglustat in Adults with Late-Onset Pompe Disease. J. Neurol. 2025, 272, 103. [Google Scholar] [CrossRef]
- Jacob, G.S.; Bryant, M.L. Iminosugar Glycosylation Inhibitors as Anti-HIV Agents. Perspect. Drug Discov. Design 1993, 1, 211–224. [Google Scholar] [CrossRef]
- Ratner, L.E.E.; Heyden, N.V. Mechanism of Action of N-Butyl Deoxynojirimycin in Inhibiting HIV-1 Infection and Activity in Combination with Nucleoside Analogs. AIDS Res. Hum. Retroviruses 1993, 9, 291–297. [Google Scholar] [CrossRef]
- Fischl, M.A.; Resnick, L.; Coombs, R.; Kremer, A.B.; Pottage, J.C., Jr.; Fass, R.J.; Fife, K.H.; Powderly, W.G.; Collier, A.C.; Aspinall, R.L.; et al. The Safety and Efficacy of Combination N-Butyl-deoxynojirimycin (SC-48334) and Zidovudine in Patients with HIV-1 Infection and 200–500 CD4 Cells/mm. J. Acquir. Immune Defic. Syndr. 1994, 7, 139–147. [Google Scholar]
- Clarke, E.C.; Nofchissey, R.A.; Ye, C.; Bradfute, S.B. The Iminosugars Celgosivir, Castanospermine and UV-4 Inhibit SARS-CoV-2 Replication. Glycobiology 2021, 31, 378–384. [Google Scholar] [CrossRef] [PubMed]
- El Khoury, M.; Wanes, D.; Lynch-Miller, M.; Hoter, A.; Naim, H.Y. Glycosylation Modulation Dictates Trafficking and Interaction of SARS-CoV-2 S1 Subunit and ACE2 in Intestinal Epithelial Caco-2 Cells. Biomolecules 2024, 14, 537. [Google Scholar] [CrossRef] [PubMed]
- Borie-Guichot, M.; Tran, M.L.; Garcia, V.; Oukhrib, A.; Rodriguez, F.; Turrin, C.O.; Levade, T.; Génisson, Y.; Bellereau, S.; Dehoux, C. Multivalent Pyrrolidines Acting as Pharmacological Chaperones Against Gaucher Disease. Bioorg. Chem. 2024, 146, 107295. [Google Scholar] [CrossRef]
- Montebugnoli, T.; Grootaert, C.; Bordoni, A.; Rajković, A.; Alderweireldt, E.; Rombaut, J.; De Maeseneire, S.L.; Van Camp, J.; De Mol, M.L. Food Iminosugars and Related Synthetic Derivatives Shift Energy Metabolism and Induce Structural Changes in Colon Cancer Cell Lines. Foods 2025, 14, 1713. [Google Scholar] [CrossRef]
- Stavale, E.J.; Vu, H.; Sampath, A.; Ramstedt, U.; Warfield, K.L. In Vivo Therapeutic Protection against Influenza A (H1N1) Oseltamivir-Sensitive and Resistant Viruses by the Iminosugar UV-4. PLoS ONE 2015, 10, e0121662. [Google Scholar] [CrossRef]
- Sayce, A.C.; Martinez, F.O.; Tyrrell, B.E.; Perera, N.; Hill, M.L.; Dwek, R.A.; Miller, J.L.; Zitzmann, N. Pathogen-induced Inflammation Is Attenuated by the Iminosugar MON-DNJ via Modulation of the Unfolded Protein Response. Immunology 2021, 164, 587–601. [Google Scholar] [CrossRef] [PubMed]
- Callahan, M.; Treston, A.M.; Lin, G.; Smith, M.; Kaufman, B.; Khaliq, M.; Evans DeWald, L.; Spurgers, K.; Warfield, K.L.; Lowe, P.; et al. Randomized Single Oral Dose Phase 1 Study of Safety, Tolerability, and Pharmacokinetics of Iminosugar UV-4 Hydrochloride (UV-4B) in Healthy Subjects. PLoS Negl. Trop. Dis. 2022, 16, e0010636. [Google Scholar] [CrossRef]
- Dwek, R.A.; Bell, J.I.; Feldmann, M.; Zitzmann, N. Host-Targeting Oral Antiviral Drugs to Prevent Pandemics. Lancet 2022, 399, 1381–1382. [Google Scholar] [CrossRef]
- Tian, Y.-S.; Zhou, Y.; Takagi, T.; Kameoka, M.; Kawashita, N. Dengue Virus and Its Inhibitors: A Brief Review. Chem. Pharm. Bull. 2018, 66, 191–206. [Google Scholar] [CrossRef] [PubMed]
- Warfield, K.L.; Alonzi, D.S.; Hill, J.C.; Caputo, A.T.; Roversi, P.; Kiappes, J.L.; Sheets, N.; Duchars, M.; Dwek, R.A.; Biggins, J.; et al. Targeting Endoplasmic Reticulum α-Glucosidase I with a Single-Dose Iminosugar Treatment Protects against Lethal Influenza and Dengue Virus Infections. J. Med. Chem. 2020, 63, 4205–4214. [Google Scholar] [CrossRef]
- Franco, E.; de Mello, C.P.; Brown, A. Antiviral Evaluation of UV-4B and Interferon-Alpha Combination Regimens against Dengue Virus. Viruses 2021, 13, 771. [Google Scholar] [CrossRef] [PubMed]
- Franco, E.J.; Drusano, G.L.; Hanrahan, K.C.; Warfield, K.L.; Brown, A.N. Combination Therapy with UV-4B and Molnupiravir Enhances SARS-CoV-2 Suppression. Viruses 2023, 15, 1175. [Google Scholar] [CrossRef] [PubMed]
- Caputo, A.T.; Alonzi, D.S.; Marti, L.; Reca, I.B.; Kiappes, J.L.; Struwe, W.B.; Cross, A.; Basu, S.; Lowe, E.D.; Darlot, B.; et al. Structures of Mammalian ER α-Glucosidase II Capture the Binding Modes of Broad-Spectrum Iminosugar Antivirals. Proc. Natl. Acad. Sci. USA 2016, 113, E4630–E4638. [Google Scholar] [CrossRef]
- Ferjancic, Z.; Bihelovic, F.; Vulovic, B.; Matovic, R.; Trmcic, M.; Jankovic, A.; Pavlovic, M.; Djurkovic, F.; Prodanovic, R.; Djelmas, A.D.; et al. Development of Iminosugar-based Glycosidase Inhibitors as Drug Candidates for SARS-CoV-2 Virus Via Molecular Modelling and In Vitro Studies. J. Enzym. Inhib. Med. Chem. 2024, 39, 2289007. [Google Scholar] [CrossRef]
- Wennekes, T.; Lang, B.; Leeman, M.; Van Der Marel, G.A.; Smits, E.; Weber, M.; Van Wiltenburg, J.; Wolberg, M.; Aerts, J.M.F.G.; Overkleeft, H.S. Large-scale Synthesis of the Glucosylceramide Synthase Inhibitor N-[5-(adamantan-1-yl-methoxy)-pentyl]-1-Deoxynojirimycin. Org. Process Res. Dev. 2008, 12, 414–423. [Google Scholar] [CrossRef]
- Chang, J.; Warren, T.K.; Zhao, X.; Gill, T.; Guo, F.; Wang, L.; Comunale, M.A.; Du, Y.; Alonzi, D.S.; Yu, W.; et al. Small Molecule Inhibitors of ER Alpha-Glucosidases Are Active against Multiple Hemorrhagic Fever Viruses. Antiviral. Res. 2013, 98, 432–440. [Google Scholar] [CrossRef]
- Ma, J.; Wu, S.; Zhang, X.; Guo, F.; Yang, K.; Guo, J.; Su, Q.; Lu, H.; Lam, P.; Li, P.; et al. Ester Prodrugs of IHVR-19029 with Enhanced Oral Exposure and Prevention of Gastrointestinal Glucosidase Interaction. ACS Med. Chem. Lett. 2017, 8, 157–162. [Google Scholar] [CrossRef]
- Reyes, H.; Du, Y.; Zhou, T.; Xie, X.; Shi, P.Y.; Weiss, S.; Block, T.M. Glucosidase Inhibitors Suppress SARS-CoV-2 in Tissue Culture and May Potentiate. bioRxiv 2021, in press. [Google Scholar] [CrossRef]
- Liu, Y.Y.; Li, Z.A.; Zhou, Y.Z.; Wang, S.L.; Chen, Z.P.; Liu, S.X.; Zhan, P.; Zhou, Y.J.; Xia, Z.X.; Deng, X. TCM Theory-Inspired Discovery of DNJ-Flavonoid Conjugates as Broad-Spectrum Anti-SARS-CoV-2 Agents by Primarily Targeting ER-Associated Glycoprotein Folding Process. Eur. J. Med. Chem. 2025, 290, 117582. [Google Scholar] [CrossRef]
- Tyrrell, B.E.; Kumar, A.; Gangadharan, B.; Alonzi, D.; Brun, J.; Hill, M.; Bharucha, T.; Bosworth, A.; Graham, V.; Dowall, S.; et al. Exploring the Potential of Iminosugars as Antivirals for Crimean-Congo Haemorrhagic Fever Virus, Using the Surrogate Hazara Virus: Liquid-Chromatography-Based Mapping of Viral N-Glycosylation and in Vitro Antiviral Assays. Pathogens 2023, 12, 399. [Google Scholar] [CrossRef]
- Overkleeft, H.S.; Renkema, G.H.; Neele, J.; Vianello, P.; Hung, I.O.; Strijland, A.; van der Burg, A.M.; Koomen, G.J.; Pandit, U.K.; Aerts, J.M. Generation of Specific Deoxynojirimycin-Type Inhibitors of the Non-Lysosomal Glucosylceramidase. J. Biol. Chem. 1998, 273, 26522–26527. [Google Scholar] [CrossRef]
- Ma, J.; Zhang, X.; Soloveva, V.; Warren, T.; Guo, F.; Wu, S.; Lu, H.; Guo, J.; Su, Q.; Shen, H.; et al. Enhancing the Antiviral Potency of ER α-Glucosidase Inhibitor IHVR-19029 against Hemorrhagic Fever Viruses In Vitro and In Vivo. Antivir. Res. 2018, 150, 112–122. [Google Scholar] [CrossRef] [PubMed]
- Yesudhas, D.; Srivastava, A.; Gromiha, M.M. COVID-19 Outbreak: History, Mechanism, Transmission, Structural Studies and Therapeutics. Infection 2021, 49, 199–213. [Google Scholar] [CrossRef]
- Nishimura, Y. The Synthesis and Biological Activity of Glycosidase Inhibitors. J. Synth. Org. Chem. 1991, 49, 846–857. [Google Scholar] [CrossRef]
- Dhara, D.; Dhara, A.; Bennett, J.; Murphy, P.V. Cyclisations and Strategies for Stereoselective Synthesis of Piperidine Iminosugars. Chem. Rec. 2021, 21, 2958–2979. [Google Scholar] [CrossRef]
- Kim, I.S.; Lee, H.Y.; Jung, Y.H. Stereoselective Synthesis of D-1-deoxynojirimycin and its Stereoisomers. Heterocycles 2007, 71, 1787–1800. [Google Scholar] [CrossRef]
- Ikota, N.; Hirano, J.I.; Gamage, R.; Nakagawa, H.; Hama-Inaba, H. Improved Synthesis of 1-Deoxynojirimycin and Facile Synthesis of its Stereoisomers from (S)-Pyroglutamic Acid Derivative. Heterocycles 1997, 46, 637. [Google Scholar] [CrossRef]
- Schaller, C.; Vogel, P.; Jäger, V. Total Syntheses of (+)-and (−)-1-Deoxynojirimycin (1, 5-dideoxy-1,5-imino D-and L-glucitol) and of (+)-and (−)-1-Deoxyidonojirimycin (1,5-dideoxy-1,5-imino D-and L-iditol) via furoisoxazoline-3-aldehydes. Carbohydr. Res. 1998, 314, 25–35. [Google Scholar] [CrossRef]
- Somfai, P.; Marchand, P.; Torsell, S.; Lindström, U.M. Asymmetric Synthesis of (+)-1-Deoxynojirimycin and (+)-Castanospermine. Tetrahedron 2003, 59, 1293–1299. [Google Scholar] [CrossRef]
- Best, D.; Wang, C.; Weymouth-Wilson, A.C.; Clarkson, R.A.; Wilson, F.X.; Nash, R.J.; Miyauchi, S.; Kato, A.; Fleet, G.W. Looking Glass Inhibitors: Scalable Syntheses of DNJ, DMDP, and (3R)-3-Hydroxy-L-Bulgecinine from D-Glucuronolactone and of L -DNJ, L-DMDP, and (3S)-3-Hydroxy-D-bulgecinine from L-Glucuronolactone. DMDP Inhibits β-Glucosidases and β-Galactosidases whereas L-DMDP is a Potent and Specific Inhibitor of α-Glucosidases. Tetrahedron Asymmetry 2010, 21, 311–319. [Google Scholar] [CrossRef]
- Bagal, S.K.; Davies, S.G.; Lee, J.A.; Roberts, P.M.; Scott, P.M.; Thomson, J.E. Syntheses of the Enantiomers of 1-Deoxynojirimycin and 1-Deoxyaltronojirimycin Via Chemo--and Diastereoselective Olefinic Oxidation of Unsaturated Amines. J. Org. Chem. 2010, 75, 8133–8146. [Google Scholar] [CrossRef]
- Iftikhar, M.; Wang, L.; Fang, Z. Synthesis of 1-Deoxynojirimycin: Exploration of Optimised Conditions for Reductive Amidation and Separation of Epimers. J. Chem. Res. 2017, 41, 460–464. [Google Scholar] [CrossRef]
- Iftikhar, M.; Zhou, M.; Lu, Y. An Overview of Debenzylation Methods to Obtain 1-Deoxynojirimycin (DNJ). Mini-Rev. Org. Chem. 2021, 18, 1037–1048. [Google Scholar] [CrossRef]
- Tyrrell, B.E.; Sayce, A.C.; Warfield, K.L.; Miller, J.L.; Zitzmann, N. Iminosugars: Promising Therapeutics for Influenza Infection. Crit. Rev. Microbiol. 2017, 43, 521–545. [Google Scholar] [CrossRef]
- Dwek, R.A.; Feldmann, M.; Fotinou, C.; Zitzmann, N. If Vaccines Falter, Broad-Spectrum Antivirals Provide a Global Shield. Lancet 2026, 407, 25–26. [Google Scholar] [CrossRef] [PubMed]
- Beran, R.K.; Vijjapurapu, A.; Nair, V.; Du Pont, V. Host-targeted Antivirals as Broad-spectrum Inhibitors of Respiratory Viruses. Curr. Opin. Virol. 2025, 73, 101492. [Google Scholar] [CrossRef]


| Compound | Name | Biological Activity | Ref. |
|---|---|---|---|
 | N-butyl DNJ (NB-DNJ or UV-1 or AT2221 or miglustat) | IC50 = 125–200 μM (BVDV) CC50 > 5000 μM (MDBK cells) IC50 = 100–500 μM (HBV) CC50 > 5000 μM (HepG2 2.2.15 cells) | [41] |
| IC50 = 6.00 ± 7.31 µM (DENV-2-infected MDMΦs) CC50 = 24.903 ± 10.506 µM (MDMΦs) | [56] | ||
| IC50 = 162 μM (DENV) CC50 > 500 μM (Vero cells) | [57] | ||
| EC50 = 10.6 μM (DENV-infected MDMΦs) | [43] | ||
| IC50 = 32.95 μM (EBOV) IC50 = 47.72 μM (MARV) CC50 > 500 μM (Vero cells) | [44] | ||
| EC50 = 41 ± 22 µM (Vero E6 infected with SARS-CoV-2) CC50 > 1000 µM (Vero cells) EC50 = 80.5 ± 23 µM (Calu-3 infected with SARS-CoV-2) CC50 > 1000 µM (Calu-3 cells) | [58] | ||
| IC50 = 170 µM (SARS-CoV-2) CC50 > 1000 µM (Calu-3 cells) | [31] | ||
 | N-nonyl-DNJ (NN-DNJ or UV-2) | IC50 = 6 μM (BVDV) CC50 = 175 μM (MDBK cells) IC50 = 1–10 μM (HBV) CC50 = 175 μM (HepG2 2.2.15 cells) | [41] |
| EC50 = 4.0 ± 0.5 μM (Huh7.5 infected with HCV) IC50 = 0.54 ± 0.08 (GluI) CC50 = 87 ± 8.7 μM (Huh7.5 cells) | [48] | ||
| IC50 = 0.91 ± 0.4 µM (DENV-2 infected MDMΦs) CC50 = 317 µM (MDMΦs) | [57] | ||
| IC50 = 9 μM (DENV) CC50 = 125 μM (Vero cells) | [56] | ||
| EC50 = 1.25 μM (DENV-infected MDMΦs) | [43] | ||
| IC50 = 15.22 μM (EBOV) IC50 = 28.66 μM (MARV) CC50 = 125 μM (Vero cells) | [44] | ||
| IC50 ~ 6.84 μM (HPV E5 viroporin) | [59] | ||
| IC50 = 3.3 ± 1.5 μM (DENV-2-infected imDCs) CC50 = 479 ± 211 µM (imDCs) | [60] | ||
| IC50 = 0.4 ± 0.2 µM (H3N2-infected MDCK) IC50 = 1.9 ± 0.8 µM (H1N1-infected MDCK) | [61] | ||
| IC50 = 4.63 µM (SARS-CoV-2 ENG2/20) CC50 > 500.0 μM (Calu-3 cells) | [31] | ||
 | N-7-oxadecyl-DNJ (UV-3) | IC50 = 30 μM (BVDV) CC50 > 2000 μM (MDBK cells) IC50 = 100 μM (HBV) CC50 > 2000 μM (HepG2 2.2.15 cells) | [41] |
| IC50 = 41 μM (DENV) CC50 > 500 μM (Vero cells) | [57] | ||
| IC50 = 34.98 μM (EBOV) IC50 = 47.72 μM (MARV) CC50 > 500 μM (Vero cells) | [44] | ||
 | N-9-methoxy-nonyl-DNJ (MON-DNJ or UV-4) | IC50 = 3.0 μM (BVDV) CC50 > 2000 μM (MDBK cells) IC50 = 3.0 μM (HBV) CC50 > 2000 μM (HepG2 2.2.15 cells) | [41] |
| IC50 = 3.09 ± 3.93 μM (DENV-2-infected MDMΦs) CC50 = 3.150 ± 1.211 μM (MDMΦs) | [56] | ||
| IC50 = 17 μM (DENV) CC50 > 500 μM (Vero cells) | [57] | ||
| IC50 = 29.97 μM (EBOV) IC50 = 47.72 μM (MARV) CC50 > 500 μM (Vero cells) | [44] | ||
| IC50 = 51.7 µM (SARS-CoV-2 ENG2/20) IC50 = 14.1 µM (SARS-CoV-2 Omicron B.1.1.529) IC50 = 21.3 µM (HCoV OC43) IC50 = 0.5371 µM (GluI) CC50 > 1000 μM (Calu-3 and HuH-7 cells) | [31] | ||
| UV-4 HCl | UV-4B | IC50 = 2.10 µM (DENV-1 SH29177) IC50 = 6.49 µM (DENV-2 NGC) IC50 = 3.64 µM (DENV-3 SL 5-29-04) IC50 = 2.78 µM (DENV-4 H241) CC50 > 1 mM (Vero cells) IC50 = 0.16 µM (Mouse ER GluI) | [62] |
| EC50 = 2.694 µM (SARS-CoV-2-infected ACE2-A549 cells) EC50 = 2.489 µM (SARS-CoV-2-infected Caco-2 cells) EC50 = 4.369 µM (SARS-CoV-2 beta variant-infected ACE2-A549 cells) EC50 = 6.816 µM (SARS-CoV-2 beta variant-infected Caco-2 cells) CC50 > 400 μM (ACE2-A549 and Caco-2 cells) | [63] | ||
| EC50 = 3.32 µM (SARS-CoV-2) CC50 > 100 μM (ACE2-A549 cells) | [64] | ||
 | N-[N-(4-azido-2-nitrophenyl)-6-aminohexyl]-DNJ (NAP-DNJ or UV-5) | IC50 = 2 μM (DENV) CC50 = 350 μM (Vero cells) | [57] |
| IC50 = 7.859 μM (EBOV) IC50 = 6.359 μM (MARV) CC50 = 350 μM (Vero cells) | [44] | ||
| IC50 = 0.017 ± 0.001 (purified rat liver GluI) | [65] | ||
| IC50 = 9.92 µM (SARS-CoV-2 ENG2/20) CC50 = 132 μM (Calu-3 cells) | [31] | ||
| EC50 = 0.58 µM (SARS-CoV-2) CC50 > 100 μM (ACE2-A549 cells) | [64] |
| Structure | Compound | Antiviral Activity | Ref. |
|---|---|---|---|
 | 1 ToP-DNJ | IC50 = 12.7 μM (DENV-infected MDMΦs) | [52] |
 | 2 DNJ-20 | EC50 = 5.13 µM (PsV entry) EC50 = 7.52 µM (PsV packaging) | [88] |
 | 3 2THO-DNJ or UV-12 | IC50 = 1.6 ± 0.8 μM (DENV-2 infected imDCs) CC50 = 443 µM (imDCs) | [60] |
 | 4 EOO-DNJ | IC50 = 3.1 ± 1.3 μM (DENV-2 infected imDCs) CC50 > 1000 µM (imDCs) | [60] |
 | 5 EB-0128 | EC50 = 0.42 µM (SARS-CoV-2) CC50 > 100 μM (ACE2-A549 cells) | [64] |
 | 6 EB-0442 | EC50 = 0.44 µM (SARS-CoV-2) CC50 > 100 μM (ACE2-A549 cells) | [64] |
 | 7 EB-0450 | EC50 = 0.72 µM (SARS-CoV-2) CC50 = 40.74 μM (ACE2-A549 cells) | [64] |
 | 8 EB-0686 | EC50 = 1.61 µM (SARS-CoV-2) CC50 > 100 μM (ACE2-A549 cells) | [64] |
 | 9 | EC90 = 1.94 µM (SARS-CoV-2 Omicron BA.1 strain-infected ACE2-A549 cells) | [89] |
 | 10 | EC90 = 3.83 µM (SARS-CoV-2 Omicron BA.1 strain-infected ACE2-A549 cells) | [90] |
 | 11 IHVR-19029 or BSBI-19029 (R = H) 11 Tetrabutyrate IHVR-19029 or BSBI-19029 tetrabutyrate (R = CH3CH2CH2CO) | EC50 = 0.25 ± 0.05 µM (BVDV) EC90 = 16.3 ± 7.8 µM (BVDV) CC50 > 500 µM (MDBK cells) EC50 = 0.74 ± 0.3 µM (TCRV) EC90 = 52.5 ± 38.9 µM (TCRV) CC50 > 500 µM (Huh7.5 cells) EC50 = 1.25 ± 1.1 µM (DENV) EC90 = 22.5 ± 10.6 µM (DENV) CC50 > 500 µM (BHK cells) EC90 = 4 µM (SARS-CoV-2 in A549-ACE2 cells) | [91,92,93] |
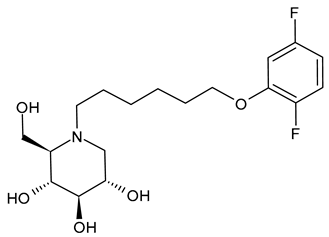 | 12 IHVR-11029 | EC50 = 1.3 ± 0.7 µM (BVDV) EC90 = 16 ± 7.9 µM (BVDV) CC50 > 500 µM (MDBK cells) EC50 = 3.3 ± 2.7 µM (TCRV) EC90 = 69 ± 37.7 µM (TCRV) CC50 > 500 µM (Huh7.5 cells) EC50 = 0.75 ± 0.06 µM (DENV) EC90 = 6.3 ± 3.5 µM (DENV) CC50 > 500 µM (BHK cells) | [7,91] |
 | 13 IHVR-17028 | EC50 = 0.4 ± 0.6 µM (BVDV) EC90 = 16 ± 12µM (BVDV) CC50 > 500 µM (MDBK cells) EC50 = 0.26 ± 0.08 µM (TCRV) EC90 = 26.7 ± 20.9 µM (TCRV) CC50 > 500 µM (Huh7.5 cells) EC50 = 0.3 ± 0.03 µM (DENV) EC90 = 1.7 ± 0.8 µM (DENV) CC50 > 500 µM (BHK cells) | [7,91] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Checconi, P.; Iacopetta, D.; Catalano, A.; Ceramella, J.; Cavalluzzi, M.M.; Mariconda, A.; Marsico, S.; Aquaro, S.; Longo, P.; Sinicropi, M.S.; et al. N-Alkyl Derivatives of Deoxynojirimycin (DNJ) as Antiviral Agents: Overview and Update. Molecules 2026, 31, 399. https://doi.org/10.3390/molecules31030399
Checconi P, Iacopetta D, Catalano A, Ceramella J, Cavalluzzi MM, Mariconda A, Marsico S, Aquaro S, Longo P, Sinicropi MS, et al. N-Alkyl Derivatives of Deoxynojirimycin (DNJ) as Antiviral Agents: Overview and Update. Molecules. 2026; 31(3):399. https://doi.org/10.3390/molecules31030399
Chicago/Turabian StyleChecconi, Paola, Domenico Iacopetta, Alessia Catalano, Jessica Ceramella, Maria Maddalena Cavalluzzi, Annaluisa Mariconda, Stefania Marsico, Stefano Aquaro, Pasquale Longo, Maria Stefania Sinicropi, and et al. 2026. "N-Alkyl Derivatives of Deoxynojirimycin (DNJ) as Antiviral Agents: Overview and Update" Molecules 31, no. 3: 399. https://doi.org/10.3390/molecules31030399
APA StyleChecconi, P., Iacopetta, D., Catalano, A., Ceramella, J., Cavalluzzi, M. M., Mariconda, A., Marsico, S., Aquaro, S., Longo, P., Sinicropi, M. S., & Lentini, G. (2026). N-Alkyl Derivatives of Deoxynojirimycin (DNJ) as Antiviral Agents: Overview and Update. Molecules, 31(3), 399. https://doi.org/10.3390/molecules31030399















