Abstract
The elimination of nitrogen and sulfur compounds from liquid fuel is a critical aspect of reducing environmental pollution. However, the widely utilized hydrodesulfurization and hydrodenitrogenation technologies require harsh operating conditions. Moreover, when operated simultaneously, these processes induce mutual competition and inhibition between the two reactions, thereby limiting the actual removal efficiency. Conversely, non-hydrogenation technologies offer substantial advantages in terms of operating conditions and provide high levels of desulfurization and denitrogenation. Nevertheless, the presence of nitrogen-containing compounds has also been demonstrated to engender competition and inhibition. It is imperative to develop environmentally friendly technologies that can simultaneously desulfurize and denitrogenate. This paper reviews research progress in this field over the past decade, providing a detailed assessment and comparison of hydrogenation and non-hydrogenation technologies, including adsorption, extraction, oxidation and biological methods. Furthermore, it considers future research directions. The article’s aim is to furnish a novel perspective on the development of clean fuel sources and to investigate more economical, sustainable, and commercially viable desulfurization and denitrogenation methods.
1. Introduction
Energy and environmental issues have long been recognized as a major stumbling block to social development [1]. The continuous increase in energy consumption has led to the depletion of low-sulfur and low-nitrogen crude oil, resulting in increasingly serious air pollution. Concurrently, unconventional fuel resources such as oil shale, oil sands and coal have been exploited to obtain high-sulfur crude oil [2]. The sulfur and nitrogen-containing compounds (SCCs and NCCs) found in liquid fuels are the primary cause of air pollution and extreme weather conditions, including haze and acid rain [3]. This phenomenon gives rise to a series of issues, including, but not limited to, soil acidification and building corrosion. The presence of SOx in the atmosphere has been demonstrated to combine with other pollutants to form carcinogens such as PM2.5, which have been shown to cause severe damage to the human respiratory system [4,5]. Concurrently, NOx undergoes a chemical reaction with VOCs under ultraviolet radiation to generate photochemical smog, which has been demonstrated to irritate the skin and respiratory system. The organic nitrogen present in fuel displays strong oxidizing activity. While in storage, it is susceptible to a reaction with oxygen, resulting in the formation of gum and sediment. In fuel vehicle systems, the resulting sulfur oxides and nitrogen oxides are absorbed by the engine oil, accelerating its ageing and causing corrosion of the engine and exhaust system [6]. In addition, diesel engines produce more nitrogen oxides [7]. The six pollutants announced by the EPA include particulate matter, ozone, carbon monoxide, sulfur oxides, nitrogen oxides and lead. In the Tier 3 standards implemented by EPA, the NOx limit is further reduced to 0.01 g/mile, and it is expected that in the new regulations in 2027, the NOx limit will be further reduced to 0.02 g/bhp hr. The minimum requirement for SO2 emissions from factories is an emission limit of 100 mg/m3. In 2021, the US Environmental Protection Agency also recognized the close connection between sulfur and nitrogen compounds in air pollution and atmospheric chemistry, combining the study of the two substances.
Fuel is inextricably linked to daily life and industrial development. With clean energy and renewable resources impacting the traditional fuel sector and increasingly strict standards being set for the sulfur and nitrogen content of fuel, in order to reduce extreme weather conditions, it is essential to utilize advanced scientific and technological methodologies in order to control air pollution at its source, such as photochemical pollution and acid rain [8,9]. Currently, most literature covers studies on fuel desulfurization [10,11,12,13,14] and denitrogenation [15,16], but reports on simultaneous desulfurization and denitrogenation are much rarer [17]. Although several review articles have examined various aspects of the simultaneous desulfurization and denitrogenation of fuels, most have focused on specific fields or technologies. There has been no systematic and comprehensive work to integrate the reaction mechanisms, catalytic materials and key influencing parameters of all major methods into a unified framework, with a lack of systematic summaries and comprehensive comparisons. In consideration of this restriction, this review comprehensively discusses the hydroprocessing processes and underlying mechanisms of major nitrogenous and sulfur-containing compounds in liquid fuels, while presenting an in-depth analysis of simultaneous hydrodesulfurization and hydrodenitrogenation (HDS/HDN) technologies. Subsequently, it focuses on the fundamental principles and technical details (including reaction mechanisms) of four emerging non-hydrogenation approaches—simultaneous adsorptive desulfurization and denitrogenation (ADS/ADN), simultaneous extractive desulfurization and denitrogenation (EDS/EDN), simultaneous oxidative desulfurization and denitrogenation (ODS/ODN), simultaneous biological desulfurization and denitrogenation (BDS/BDN)—and systematically evaluates the advantages and limitations of each technology for concurrent fuel desulfurization and denitrogenation. Overall, this review aims to address the existing fundamental gap by providing a comprehensive. The aim of this review is to address the existing gap in the field by offering a comprehensive comparison and specific analysis of traditional hydrodesulfurization and denitrogenation and emerging non-hydrotreatment technologies. It also provides valuable discussions and promotes the development of clean fuel.
2. Typical Sulfur and Nitrogen Compounds in Fuel Oil
There is significant competition and inhibition between traditional hydrodesulfurization and hydrodenitrogenation. The specific mechanisms will be explained in detail in the following sections. Therefore, a full understanding of the different types of sulfur- and nitrogen-containing compounds in fuel is necessary.
2.1. Types of Sulfur Compounds
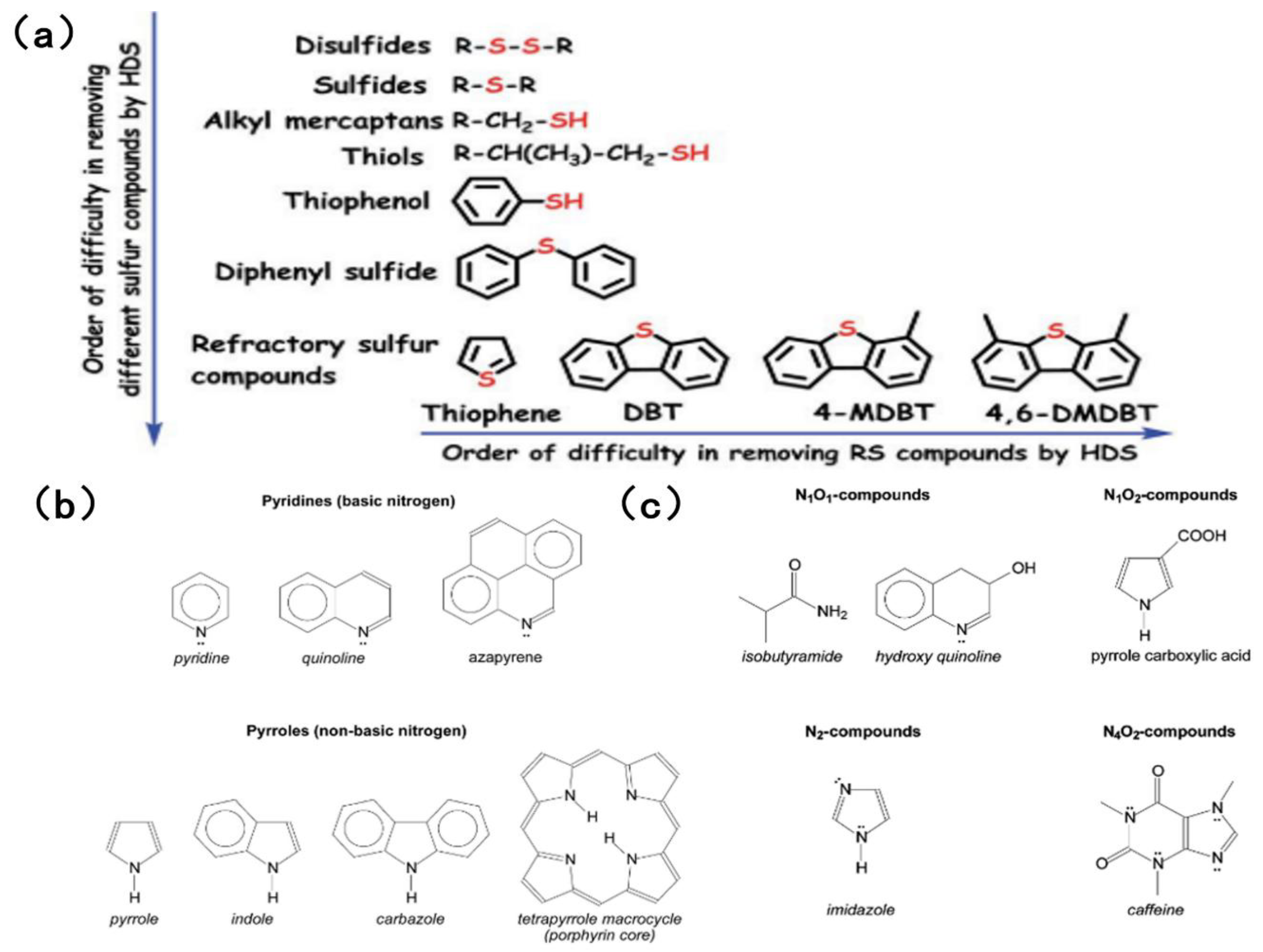
The sulfur content of fuel oil varies depending on the storage layer and type of petroleum, ranging from 0.05% to 14% [18,19]. Sulfur compounds can be classified as acidic or non-acidic based on their acidity or alkalinity. Simple sulfides, such as disulfide and hydrogen sulfide, can be easily removed by hydrogenation. However, aromatic sulfides with complex structures, such as thiophenes, benzothiophenes and dibenzothiophenes, pose a greater challenge in terms of their elimination, owing to resonance stability and spatial site resistance effects. Figure 1a shows common sulfides in HDS and their order of difficulty in removal [20]. When simulating sulfur compounds in fuel, BT, DBT and 4,6-DMDBT are most commonly chosen as the simulated sulfur compounds. This is because traditional HDS is relatively inefficient at removing aromatic sulfides.
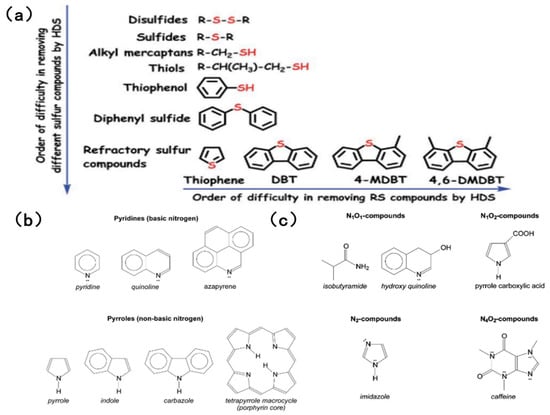
Figure 1.
(a) Order of organic sulfur compounds in the HDS process based on removal difficulty in fuel feedstock. Reproduced with permission from Ref. [20]. Copyright 2020 RSC. (b) Examples of pyridinic and pyrrolic compounds. Reproduced with permission from Ref. [6]. Copyright 2017 ACS. (c) Examples of nitrogen-containing compounds with more than one heteroatom that were identified in crude oil samples. Reproduced with permission from Ref. [6]. Copyright 2017 ACS.
2.2. Types of Nitrogen Compounds
Compared with the sulfur content, the nitrogen content of fuel is generally lower [21]. The nitrogen oxides in fuel are typically organic nitrogen, accounting for over 95% of the total, with only trace amounts of inorganic nitrogen present. Fuel is categorized as high-nitrogen (≥0.25 wt%) or low-nitrogen (<0.25 wt%) according to its nitrogen content. The fuel’s boiling point also varies with the concentration of nitrogen-containing compounds [6]. Organic nitrogen can be further categorized as either acyclic or aromatic compounds. The former are mainly organic amines, which have a low content and are easy to remove. The latter can be further classified as either alkaline (pyridine derivatives) or non-alkaline (pyrrole derivatives). An excessively high content of aromatic hydrocarbons reduces the quality of the fuel [22]. In most fuels, the ratio of alkaline to non-alkaline nitrogen fluctuates within the range 0.25–0.35 [23,24]. It is evident that basic nitrogen, a six-ring pyridine compound, which includes pyridine and quinoline, primarily exists in light fuel oil. These compounds are highly alkaline and interact strongly with acidic sites on the catalyst surface. Non-basic nitrogen, namely pentacyclic pyrrole compounds, mainly exist in heavy fuel and mainly include pyrrole, indole and carbazole, which makes the deep desulfurization of fuel difficult [25].
Although there are many types of nitrogen-containing substances, references [21,25] generally agree that the most abundant in crude oil are heterocyclic compounds containing a single nitrogen atom, such as pyridine, quinoline and carbazole. Despite the fact that the content of nitrogen compounds is much lower than that of sulfur compounds, the toxicity of nitrogen compounds to the catalyst is significant in the traditional hydrodesulfurization and hydrodenitrogenation process, forcing the catalyst to lose its desulfurization activity [21]. Figure 1b [6] shows common nitrogen-containing compounds in fuel.
3. Hydrodesulfurization and Hydrodenitrogenation
The HDS and HDN methodologies have been extensively applied in actual industrial production contexts. In the presence of elevated temperatures and pressures, in a hydrogen atmosphere, and with the aid of catalysts, the C-S and C-N bonds in sulfur and nitrogen compounds, respectively, are broken and subsequently converted into hydrogen sulfide (H2S) and ammonia (NH3) for removal.
3.1. Hydrodesulfurization (HDS)
Hydrodesulfurization is typically performed under harsh conditions (a temperature of 300–400 °C and a pressure of 3–10 MPa) using hydrogenation to break the C–S bond and release H2S and sulfur-free hydrocarbons [26]. This process occurs through two main reaction mechanisms: In the direct mechanism, the C-S bond is broken and replaced with hydrogen through hydrogenolysis or direct desulfurization (DDS) [27]. Aromatic sulfur compounds with linear structures, such as sulfides and mercaptans, are prone to reacting and can be removed by DDS. The other mechanism is indirect, also known as the hydrogenation pathway (HYD). This involves hydrogenating the sulfur compounds to generate corresponding saturated intermediate products, thereby achieving cleavage of the C-S bonds and desulfurization.
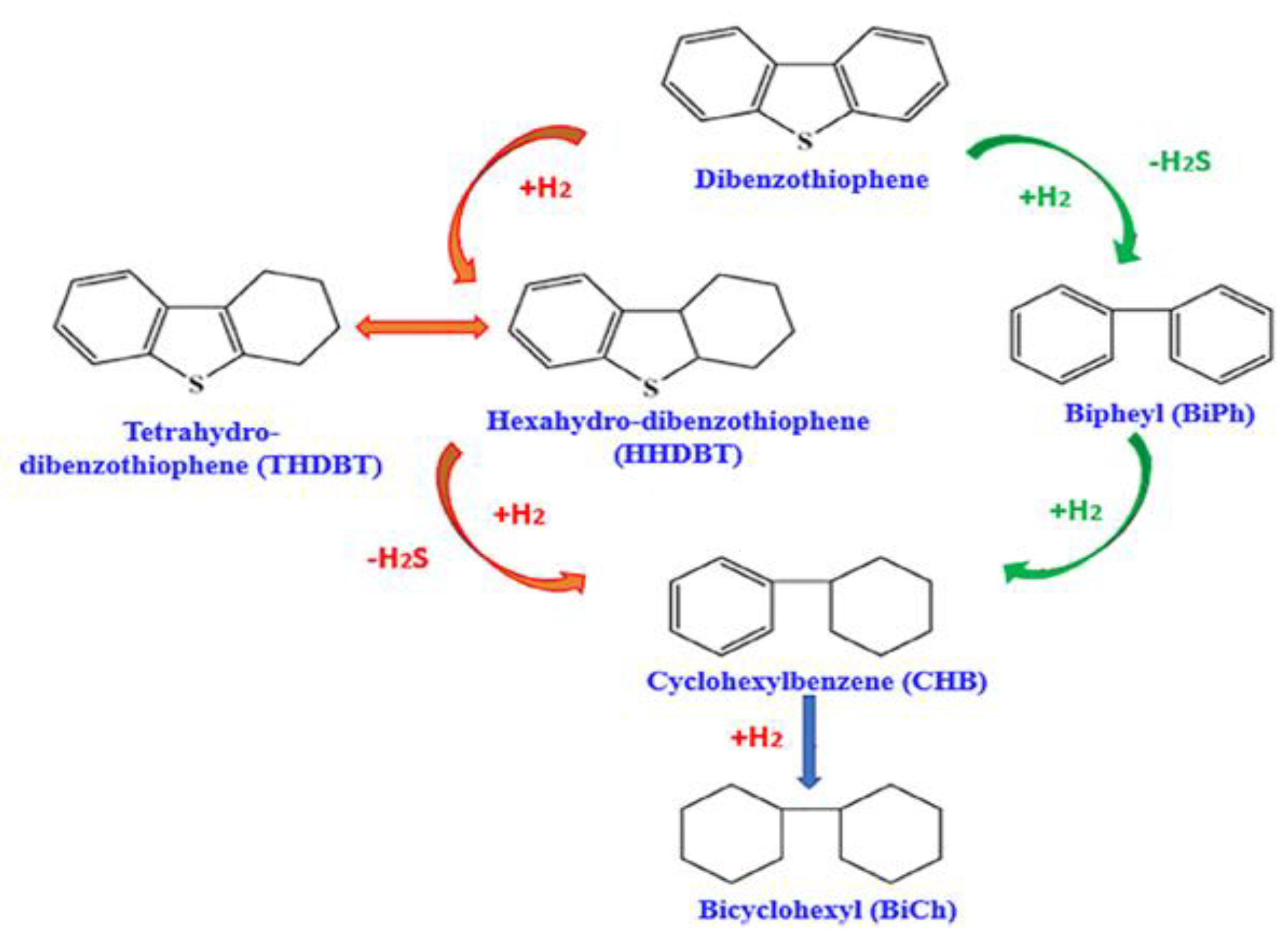
Dibenzothiophene (DBT) and its derivatives are the most difficult sulfur compounds to remove. They are mainly produced through the HYD method, which involves hydrogenation to form saturated aromatic rings, followed by C-S bond cleavage [28]. The specific reaction is shown in Figure 2 [29]. During the actual desulfurization reaction process, many sulfides react through different pathways during hydrodesulfurization (HDS). The research results of Sharifvaghefi et al. [30] indicate this. First, DBT undergoes hydrogenolysis, which directly breaks the C-S bond to release H2S; the final product is biphenyl. Secondly, via the hydrogenation pathway, DBT is first converted to tetrahydrodibenzothiophene (THDBT). The two pathways depend on the type of catalytic material, promoter, and support material used. Zheng et al. [31] reported that, in the thiophene HDS process, the final products of the DDS and HYD routes were all four compounds.

Figure 2.
Possible pathways of HDS of DBT compound, reproduced with permission from Ref. [29]. Copyright 2018 Elsevier.
3.2. Hydrodenitrogenation (HDN)
The process of hydrodenitrogenation has been demonstrated to achieve ring-opening denitrogenation through the hydrogenation of aromatic rings. Subsequent to the cleavage of the C-N bond, alkenes, ammonia and various nitrogen compounds are released. The various reaction mechanisms in hydrodenitrogenation (HDN) are primarily contingent on the molecular structure, basicity, and the interaction mode with the active site of the catalyst. Prior to the cleavage of the C-N bond, there are substantial disparities in the structural characteristics of the five-membered N-ring and six-membered N-ring amine intermediates formed during the reaction [32]. In the context of polycyclic nitrogen compounds, two predominant HDN mechanisms have been identified: aniline-like and pyridine-like. The aniline-like mechanism focuses on the hydrogenation of the benzene ring and the cleavage of the C-N bond [33], while the pyridine-like mechanism involves the hydrogenation saturation of the nitrogen heterocyclic ring and the cleavage of the C-N bond [34]. It has been established that the remaining nitrogen compounds possess a single reaction mechanism. In comparison with non-alkaline compounds, nitrogen compounds with strong alkalinity exhibit enhanced adsorption properties on the active sites of the catalyst. This phenomenon has been observed to induce catalyst poisoning and result in competition with sulfur compounds for the active sites. It has a significant impact on the reaction process of HDN and HDS [35,36]. Furthermore, the low resolution of nitrogen compounds at the active sites results in irreversible poisoning [37].
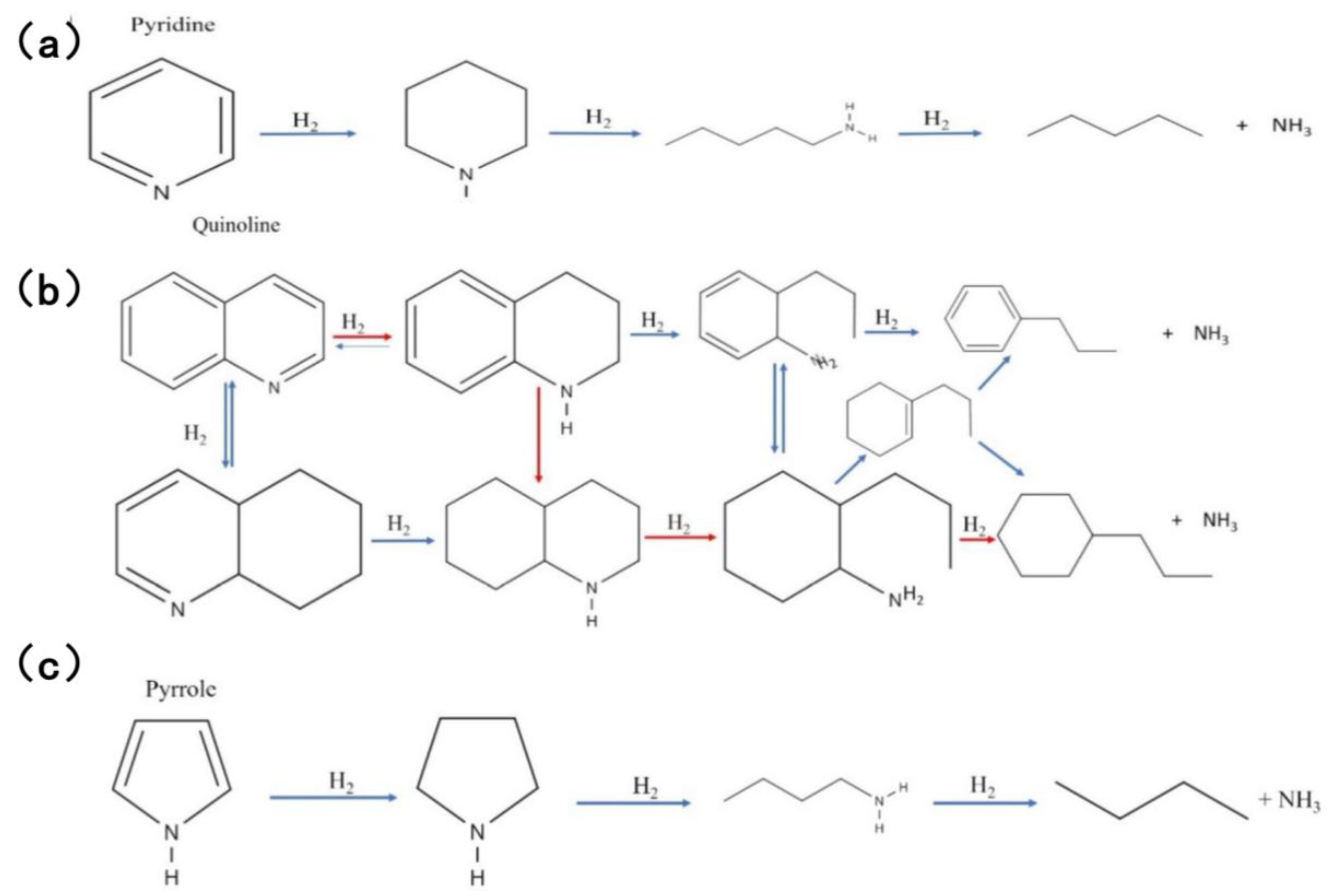
The reaction process of pyridine HDN, the simplest aromatic compound in fuel oil, is shown in Figure 3a. The process of hydrogenation of pyridine is gradual, resulting in the formation of piperidine and Amylamine. Subsequent removal of NH3 leads to the final formation of a C5 hydrocarbon [38,39,40]. Quinoline, a nitrogen compound with a high molecular weight and a boiling point above 340 °C, has been found to possess two HDN pathways [39,41,42]. The initial pathway entails hydrogenation, the process of saturating the ring, with the objective of synthesizing 1,2,3,4-tetrahydroquinoline (THQ). Subsequent to this, ring cleavage is initiated, resulting in the formation of orthopropylaniline (OPA). The final step in this process involves denitrogenation. The second pathway involves the saturation of THQ to decahydroquinoline (DHQ), followed by the opening of the ring to release hydrocarbons through PCHA. The specific reaction process is illustrated in Figure 3b [26]. In contrast, the reaction mechanism of pyrrole is relatively straightforward. Moravek et al. proposed a reaction process that is analogous to that depicted in Figure 3c.
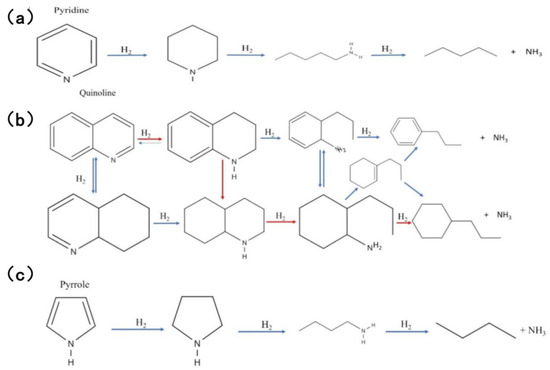
Figure 3.
Reaction HDN network for basic N-compounds: (a) pyridine, (b) quinoline, (c) pyrrole. Reproduced with permission from Ref. [26]. Copyright 2021 ACS.
3.3. Simultaneous Hydrodesulfurization and Hydrodenitrogenation
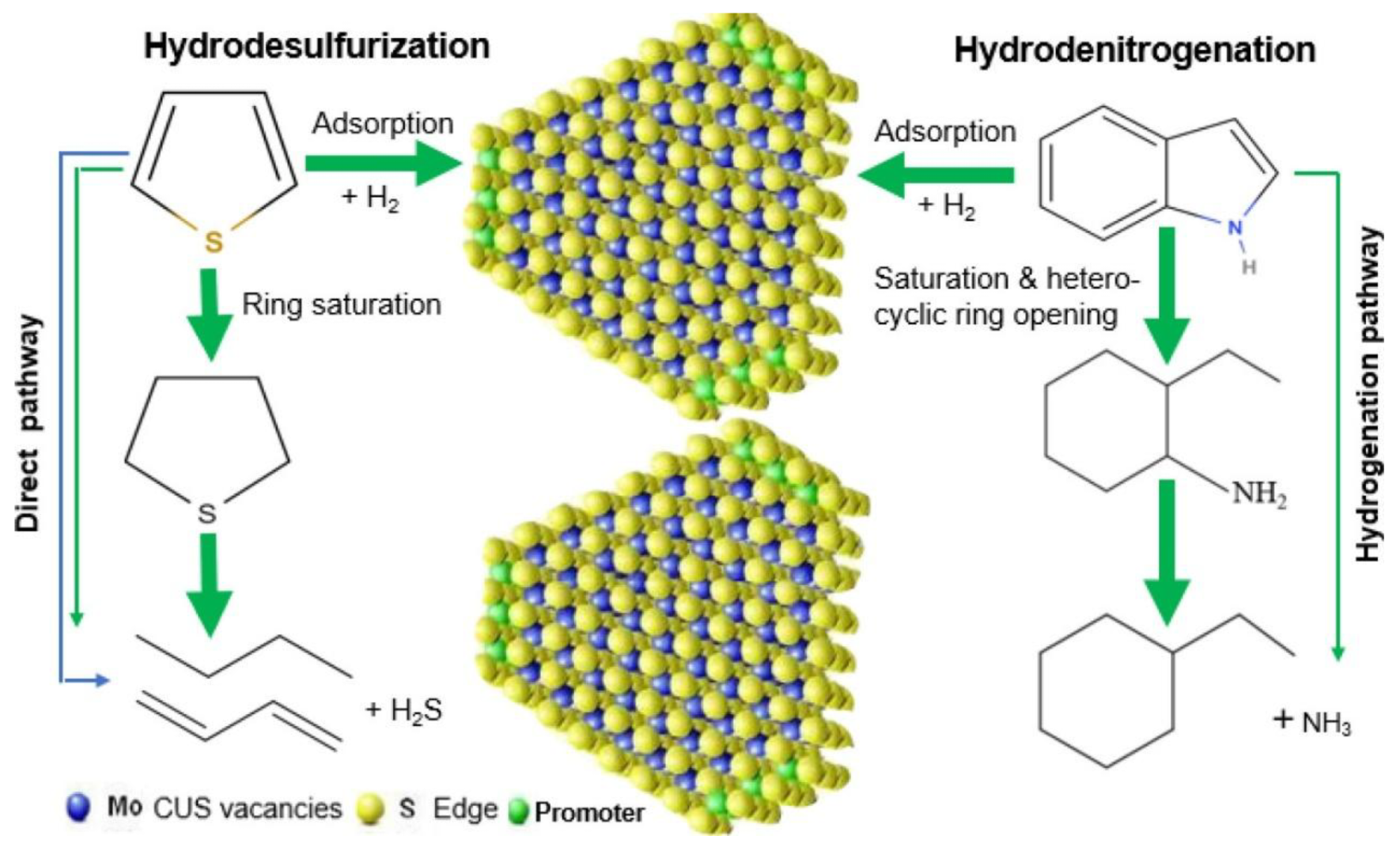
In the process of actual fuel purification, hydrodesulfurization (HDS) and hydrodenitrogenation (HDN) occur in a concurrent manner. The most prevalent catalyst systems for this purpose involve loading Al2O3 with sulfide cobalt–molybdenum (Co-Mo) or nickel–molybdenum (Ni-Mo), with the coordination unsaturated sites (CUS) constituting the fundamental element of the catalyst activity for HDS and HDN. The formation of this compound is primarily attributable to the exposure of coordinated unsaturated molybdenum (or tungsten) atoms at the edges of MoS2 or WS2 crystals [43]. These sites possess Lewis acidity and have been demonstrated to adsorb and activate sulfur and nitrogen compounds, thereby promoting the cleavage of C-S and C-N bonds [44]. The specific catalytic pathways are illustrated in Figure 4.
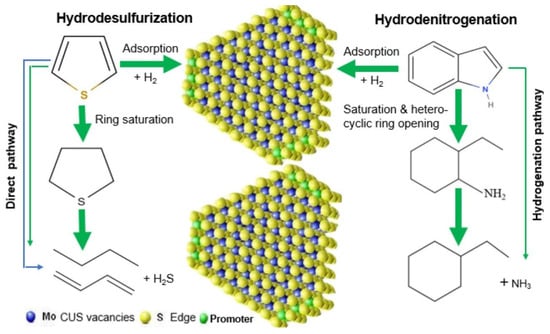
Figure 4.
Catalysis of HDS and HDN for sulfur and nitrogen removal. Reproduced with permission from Ref. [26]. Copyright 2021 ACS.
As previously stated, the competition of nitrogen compounds for the active site, in conjunction with the irreversible poisoning of the catalyst, significantly impedes the HDS reaction. Therefore, it is essential to comprehend the interplay between these two reactions and the selection of the active site of the catalyst. When De Mello et al. [45] simultaneously carried out HDS and HDN using the NiMoP/Al2O3 catalyst, a similar result was discovered: the competition of active sites led to a decrease in HDS efficiency. Specifically, the conversion rate of 4-6-DMDBT decreased from 85% for individual conversion to 65%, and the conversion rate of Q decreased from 80% to 75%. In subsequent studies, Albersberger et al. [46] discovered that when DBT and o-propyl aniline were HDS and HDN simultaneously, they would share the same active site. Due to the preferential adsorption of o-propyl aniline by the active site, the HDS reaction process was greatly inhibited. The conversion rate of DBT decreased by 31% compared to individual conversion, while the conversion rate of Q or OPA only decreased by about 6% during competition. The research of Farag et al. [47] indicates that the presence of quinoline in HDS and HDN without MoS2 has an adverse effect on the HDS of DBT, but the presence of DBT promotes the HDN process of quinoline by increasing the acidity of potential active sites. Similarly, under competitive reactions, the conversion rate of DBT decreased by 31%, while the conversion rate of Q or OPA only decreased by 4% during competition.
In order to surmount the competitive and inhibitory relationship between HDS and HDN, the design of hydrogenation treatment catalysts with higher activity than traditional catalysts has always been a significant issue. Oliver et al. [48] sought to explore different catalyst carriers of (nickel) molybdenum sulfide catalysts with a view to improving catalytic performance. The research findings indicate that the characteristics of the carrier have a significant impact on the dispersion of the unpromoted catalyst and the degree of modification of nickel at the periphery of the Ni-Mo-S phase. In addition, the acidity of the carrier has been observed to influence the acidity of the loaded sulfide phase, a factor that may play a crucial role in HDN. The catalyst designed in this experiment, in the system with the best reaction efficiency, showed a 17% decrease in the conversion rate of sulfides and a 3% decrease in the conversion rate of nitrides even under competitive conditions, which is much higher than the previous research results. In their study, Idia et al. [49] investigated the kinetic modelling of the hydrodesulfurization (HDS) reactions of 4,6-dimethyldibenzothiophene (4,6-DMDBT) and the hydrodenitrogenation (HDN) reactions of quinoline with CoMoP and NiMoP catalysts in a stacked bed configuration. The research results show that a higher reaction temperature can not only increase the total conversion rate of 4,6-DMDBT and quinoline, but also enhance the hydrogenation selectivity. Even during competitive conversion, the CoMoP and NiMoP stacking configuration at 340 °C can achieve a conversion rate of 85% for 4,6-DMDBT and 95% for Q. Matheus et al. [45] conducted a study on the competitive effect of phosphorus content on hydrodesulfurization (HDS) of 4,6-dimethyldibenzothiophene (4,6-DMDBT) and hydrodenitrogenation (HDN) of quinoline. It has been demonstrated that when the content of quinoline is relatively high, the HDS activity of all catalysts is inhibited. Among these catalysts, the inhibition of the hydrogenation (HYD) pathway is more obvious. In instances where the concentration of quinoline is minimal, the addition of phosphorus has been observed to augment the activity of HDS. For specific quinoline concentrations and temperatures, the catalyst containing 1 wt% phosphorus is more active for HDS, while the catalyst containing 2 wt% phosphorus has a stronger inhibitory effect on HDS but is active for HDN.
In conclusion, although hydrogenation is widely used in actual production, to deal with fuel with high sulfur and nitrogen content, not only is an additional 50% or more of hydrogen required, but also several times the amount of catalyst is needed. Concurrently, the HDN and HDS reactions characteristically exhibit a robust interaction in hydrogenation treatment. By summarizing the literature, it is found that in hydrodesulfurization and hydrodenitrogenation, the inhibition effect of nitrides on sulfides is much higher than that of sulfides on nitrides. The core reason is that the adsorption affinity and competitive priority of nitrides on catalytically active sites are significantly higher than those of sulfides. Specifically, nitrogen atoms containing strong alkaline lone pair electrons in nitrides can form a strong σ-bond adsorption with Lewis acid sites on the catalyst surface. Sulfides are adsorbed by sulfur atoms or π electrons in aromatic rings, and the adsorption bond energy is only 1/3–1/2 of that of nitrides. The removal of sulfide is more dependent on the HYD path, while nitrogen compounds just strongly inhibit the HYD path, forcing sulfide to fail to complete aromatic hydrogenation, resulting in a sudden decline in desulfurization efficiency. A comprehensive understanding of the mechanisms of active site competition, reaction pathway differences, and product inhibition, in conjunction with the meticulous design of catalysts, has enabled the development of novel catalysts that exhibit high activity and selectivity. These catalysts are well-suited to meet the progressively stringent requirements for fuel desulfurization and denitrogenation.
4. Non-Hydrogenation Process
The conventional hydrodesulfurization and hydrodenitrogenation (HDS/HDN) processes necessitate the use of hydrogen, a costly reactant, as a fundamental component of the reaction. These processes are characterized by their relatively low overall efficiency in the production of ultra-clean fuel and their inability to adequately remove refractory aromatic compounds. Consequently, researchers have developed a range of non-hydrogenation technologies, including oxidative denitrogenation (ODN)/oxidative desulfurization (ODS), adsorption denitrogenation (ADN)/adsorption desulfurization (ADS), extractive denitrogenation (EDN)/extractive desulfurization (EDS), and biological denitrogenation (BDN)/biological desulfurization (BDS). These have been complemented by photocatalytic and ultrasonic-assisted oxidative desulfurization and denitrogenation technologies.
4.1. Simultaneous Extraction Desulfurization and Denitrogenation
Extraction is a process that involves the removal of sulfur and nitrogen compounds from fuel under mild conditions. It achieves this by selectively dissolving these heteroatom compounds, thereby separating them from the fuel components. The core of this methodology is the selection of an efficient and highly selective extractant. In the present study, ionic liquids (ILs) and deep eutectic solvents (DESs) are regarded as highly promising extractants due to their unique properties.
4.1.1. Ionic Liquids (ILs)
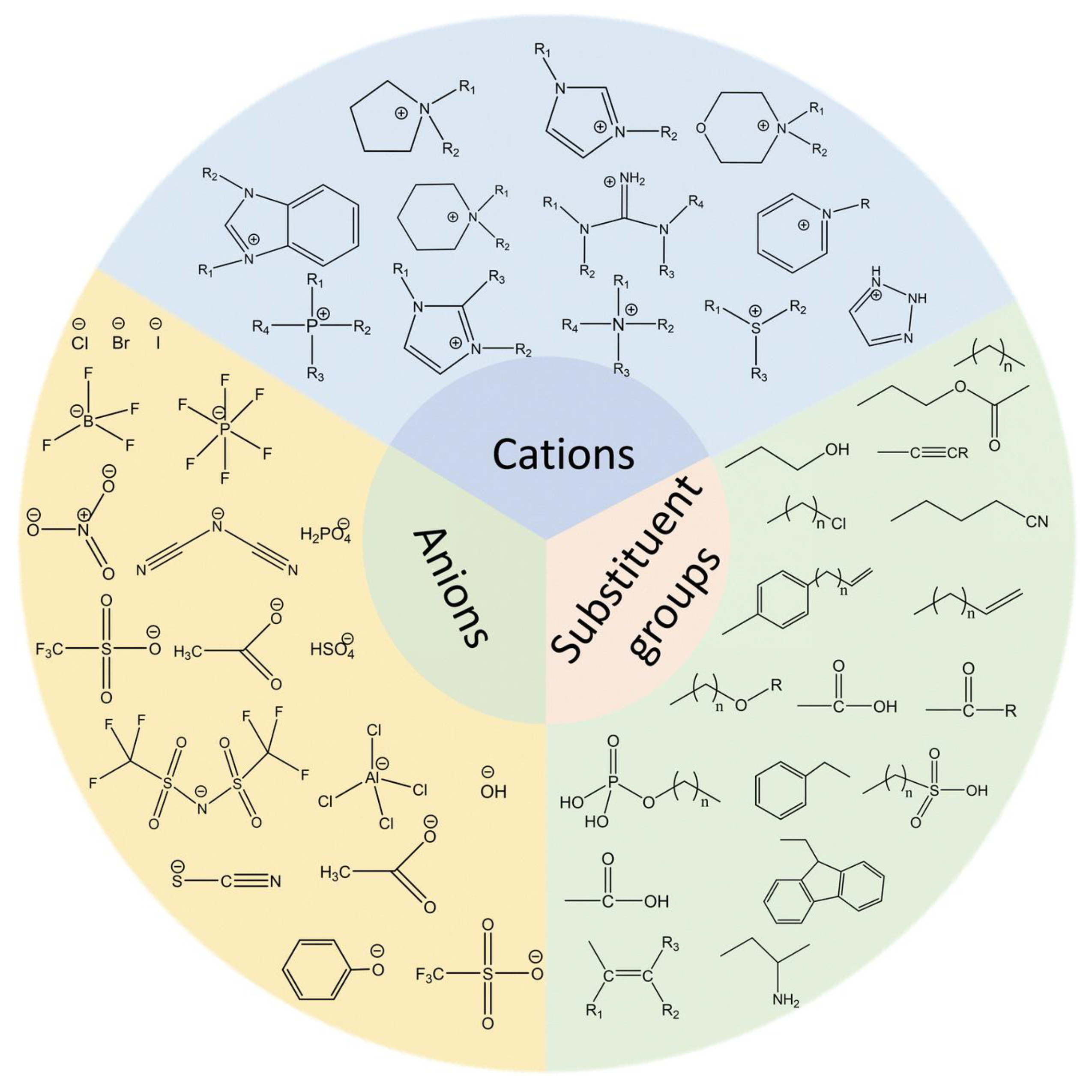
Ionic liquids have been shown to exhibit a series of advantageous physical and chemical properties when compared with traditional aprotic solvents. This renders them an optimal choice for a range of applications, including extraction, desulfurization and denitrogenation [35,50,51]. The extraction and separation of aromatic compounds from any fuel source is possible, thereby ensuring higher extraction yields. Furthermore, these substances exhibit minimal vapor pressure and remarkably low evaporation during the extraction process, thus classifying them as a novel type of green solvent. The substance under scrutiny is chiefly constituted of molten salts of organic cations with deficient coordination and inorganic or organic anions [52,53]. It is noteworthy that even the most minimal alteration in its structural configuration has the capacity to induce significant alterations in its physical and chemical properties [54]. In practical applications, the selectivity, extraction capacity, surface activity and other properties of ionic liquids as extractants must be considered [55]. The properties of these liquids are determined by the structure of the ionic liquids themselves, as well as the different anions and cations that are present. Common anions, cations and substituents are illustrated in Figure 5 [56].

Figure 5.
Structures of the typical IL cations, anions, and substituent groups. Reproduced with permission from Ref. [56]. Copyright 2023 RSC.
The mechanism of simultaneous desulfurization and denitrogenation by ionic liquids is intricate and typically involves multiple synergistic effects. The primary mechanisms encompass acid–base interactions, hydrogen bond interactions, π–π interactions, Lewis acid–base interactions, and others. Yi Nie et al. [57] were responsible for the design and synthesis of three types of bifunctional ionic liquids for the purpose of simultaneous desulfurization and denitrogenation. The findings indicate that the three nitrogen-containing compounds (pyridine, pyrrole, and quinoline) can be effectively removed, with a removal rate approaching 100%. It has been demonstrated that quinoline exerts no influence on the ultimate DBT removal; its effect is merely negligible in the preliminary phase of the reaction. It has been demonstrated that both pyridine and pyrrole have a deleterious effect on desulfurization. Of these, pyrrole has been shown to be the most significant inhibitor. This phenomenon is primarily attributable to the oxidative competition between nitrogen-containing compounds and sulfides, as well as the interference with the extraction capacity of ionic liquids. Compared with the traditional Sulfolane and conventional imidazole ionic liquids, the extraction effect of bifunctional ionic liquids is remarkable. After 4 times of reuse, the DBT removal rate was still 91.2%, the rate constant decreased from 0.9323 min−1 to 0.9141 min−1, and the activity decreased by only 2%. In their seminal work, Hansmeier et al. [58] developed an ionic liquid with a specific structure (pyridinium group containing anions) as an efficient extractant for fuel desulfurization and denitrogenation. The performance of the developed ionic liquid was found to be significantly superior to that of the industrial standard sulfoxide. The extraction selectivity of ionic liquids for nitrogen compounds is significantly higher than that for sulfur compounds. Compared with the reference solution, the extraction efficiency of the four designed extractants for nitrides and sulfides was increased by more than 10%, the extraction efficiency of nitrides was up to 99%, and the extraction efficiency of sulfides was higher than 84%. After repeated use for 5–8 times, the single extraction rate of sulfide remained above 75%, while the extraction rate of nitride remained above 95%. Chen et al. [59] selected four acidic ionic liquids (two Lewis acidic and two Brown acidic) for simultaneous desulfurization and denitrogenation. The findings demonstrate that the Lewis acid type exhibits superior performance. The extraction efficiency of this acidic ionic liquid is much higher than that of the traditional solvent. The desulfurization rate is up to 95.9%, and the denitrogenation rate is nearly 100%. It can be reused five times and still maintain stable activity and the regeneration method is simple. These are primarily attributable to the coordination of cations with sulfur and nitrogen atoms, and the synergistic acidic effect of anions. Furthermore, the study revealed that acidic ionic liquids exhibited remarkably high selectivity for the removal of basic nitrogen compounds (e.g., pyridine), thus providing a viable solution to the poisoning problem of HDS catalysts.
4.1.2. Deep Eutectic Solvents (DESs)
Deep eutectic solvents are typically constituted of two components: hydrogen bond acceptors and hydrogen bond donors. These are low-eutectic mixtures formed under the interaction of hydrogen bonds [60,61]. DES shares numerous similarities with ILs, including low vapor pressure, wide liquid range, high thermal stability, low flammability and high solubility [62]. However, DESs have a low preparation cost, a simple preparation process, better economic appeal, and are biodegradable with low toxicity. It is also a green solvent. As demonstrated in earlier research, the extractant in question has been shown to have the capacity to remove sulfur oxides (EDS) [63,64] and nitrogen oxides (EDN) [65] in a simultaneous manner [66]. Its remarkable performance in simultaneous desulfurization and denitrogenation has led to a surge in research interest in DESs.
Lima et al. [67] made a significant contribution to the field by achieving research progress in the simultaneous EDS and EDN. The efficacy of four types of DES was investigated through the preparation of these substances and the subsequent simulation of the fuel oil and gasoline systems. The findings of this investigation revealed that the four types of DES exhibited an efficiency range of 65% to 99% in their capacity to target nitrogen and sulfur compounds. Sulfur compounds were the most challenging to eliminate, with Th exhibiting the most significant difficulty and EE demonstrating the least. The removal efficiency of nitrogen compounds is higher (Py can reach over 99%). The findings demonstrate that DES exhibits notable stability in the removal of nitrogen compounds, unaffected by the complexity of the fuel matrix. The deep removal of sulfur compounds can be achieved through multi-stage extraction. The extractant can be completely recovered by precipitation with water, vacuum drying and regeneration, and can be recycled for more than five times. This conclusion was corroborated by Rogosic et al. [68]. In the course of the experiment, ChCl was utilized as the hydrogen bond acceptor, while PG was employed as the hydrogen bond donor. This approach enabled the preparation of DES, which exhibited a molar ratio of 1:3 for desulfurization and denitrogenation. Compared with sulfolane, the selectivity of Des for nitrides was significantly improved, but the selectivity for sulfides was only 30–40%. MK et al. [69] designed three DES with imidazole as the hydrogen bond acceptor and natural compounds as the hydrogen bond donor for the purpose of removing sulfur and nitrogen from the fuel. The high polarity of DES is attributable to the strong hydrogen bond formed between the N atom of imidazole and pyridine, and the π–π interaction between the aromatic rings of imidazole and HBD and thiophene (Figure 6). The enhanced selectivity for polar impurities (pyridine, thiophene) is a consequence of these interactions. The synergistic effect of multiple mechanisms enables all three DES to efficiently and concurrently desulfurize and denitrify, although the denitrogenation effect is significantly superior to that of desulfurization. In the study by Zarin et al. [70], the authors prepared acidic DES utilizing triphenylphosphine bromide as the hydrogen bond acceptor and p-aminobenzoic acid as the hydrogen bond donor, with a molar ratio of 2:3. The removal performance of thiophene and pyridine in synthetic gasoline was systematically evaluated. The experimental results show that the removal efficiency of pyridine is 50% higher than that of thiophene, and the RMSD of extractant is less than 0.005, which is higher than that of conventional solvents. Furthermore, the efficacy of the device in removing impurities from real gasoline is significantly superior to that of analogous products. It is widely acknowledged that the efficacy of DES as a denitrifying agent is contingent upon its capacity to selectively extract nitrogen and sulfur compounds. This property is a consequence of the agent’s unique extraction mechanisms, which are distinct for both nitrogen and sulfur compounds.
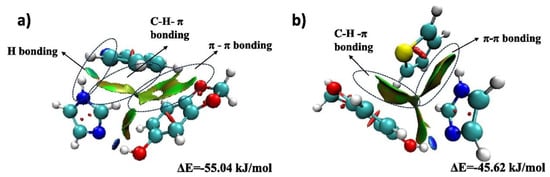
Figure 6.
Optimized conformations for SES: IMI-1 with (a) pyridine (b) thiophene. Reproduced with permission from Ref. [69]. Copyright 2025 Elsevier.
4.1.3. Other New Types of Extractants
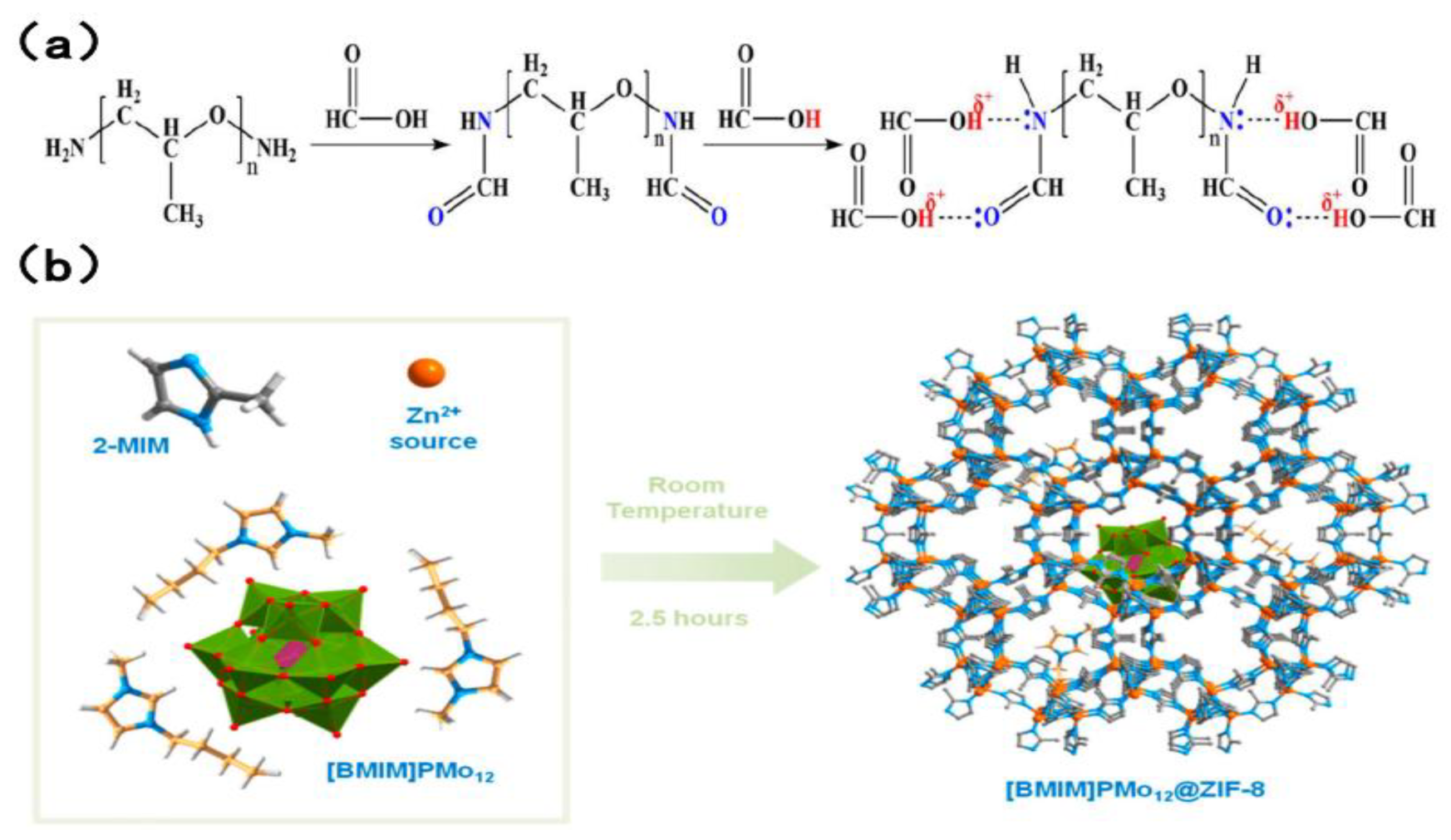
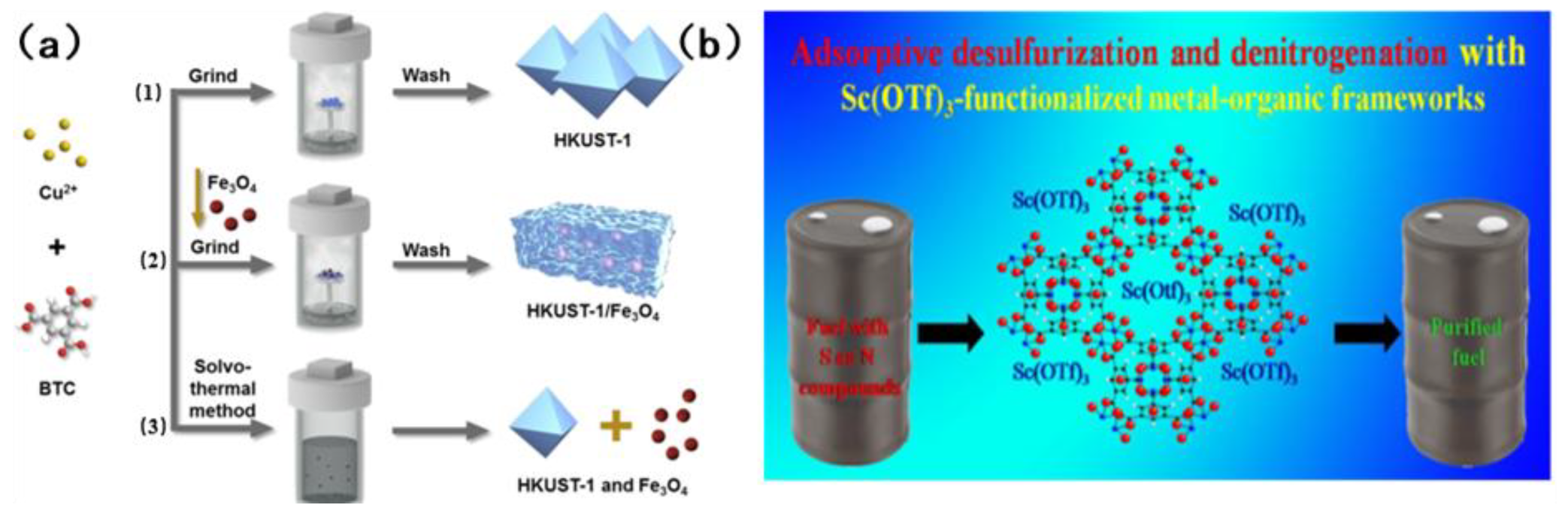
In the context of industrial extraction, desulfurization, and denitrogenation, both ILS and DESs have been shown to exhibit certain undesirable characteristics. The use of EDS extractant, for instance, has been associated with significant environmental damage, cumbersome recovery processes, and considerable expense. Conversely, EDN necessitates the simultaneous removal of both alkaline and non-alkaline nitrogen, a task that is rarely accomplished by available extractants. Consequently, there is a pressing need to develop an extractant that is both cost-effective and highly efficient, with the capacity to facilitate desulfurization and denitrogenation processes. Zhu et al. [66] made a significant contribution to this aspect of research. A novel composite solvent was developed, comprising a new type of polyetheramine (PEA/formic acid FA complex). It has been established that PEA reacts with FA to form amide groups. Concurrently, a complexation effect is observed between excess FA and amide groups. The complex is characterized by the presence of both sufficient Lewis basicity and mild Brownst acidity. The interaction between sulfur and nitrogen compounds is primarily characterized by weak hydrogen bonds and mild acid–base interactions. The extractant, with its dual Lewis alkalinity and mild Brunst acidity, enables efficient removal of sulfur and nitrogen compounds from fuel. Silva et al. [71] encapsulated imidazole-based polyoxometriate ([BMIM]3PMo12) into the ZIF-8 molecular sieve framework to prepare the [BMIM]PMo12@ZIF-8 heterogeneous catalyst, whose structure is shown in Figure 7. The catalyst can be repeatedly recycled up to ten times. The desulfurization efficiency has been found to be consistently above 98.5%, while the denitrogenation efficiency has been found to be stable at approximately 82%. The apparatus is capable of achieving efficient simultaneous desulfurization and denitrogenation, and complete removal of the target impurities in the fuel within a time frame of one hour.
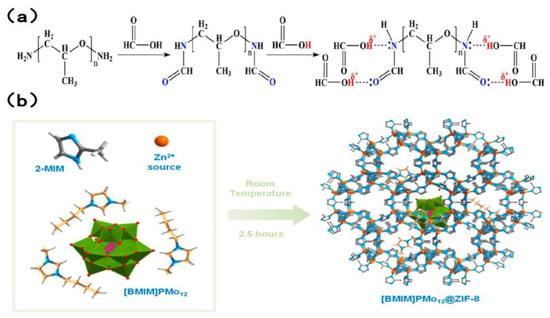
Figure 7.
(a) Formation of PEA/FA. Reproduced with permission from Ref. [66]. Copyright 2020 ACS. (b) Representation of the preparation of the [BMIM]PMo12@ZIF-8 compound by a “bottlearound-the ship” approach and using a sustainable room temperature synthetic strategy (one-pot and in situ). Reproduced with permission from Ref. [71]. Copyright 2020 MDPI.
In conclusion, the extraction and removal of sulfur and nitrogen compounds is a very promising method that can efficiently remove most of the pollutants and impurities in a short period of time. In comparison with aprotic solvents, both ILS and DESs have been demonstrated to exhibit significant superiority, thus providing valuable references for future research and application [72]. However, the nitrogen removal efficiency of these two extractants is significantly higher than that of desulfurization. This significant disadvantage makes the extraction method need to be used many times in desulfurization, which undoubtedly increases the cost.
4.2. Simultaneous Adsorptive Desulfurization-Denitrogenation
Adsorption desulfurization and adsorption denitrogenation (ADS/ADN) are comparable to extraction processes, and the selection of an appropriate adsorbent is pivotal to achieving optimal results. The principal requirements for adsorbents encompass operational conditions, methodologies for preparation, elevated levels of porosity, and environmental sustainability. The most commonly used adsorbents include activated carbon (ACs), mesoporous silica, metal oxides (Al2O3, TiO2, ZnO), metal–organic frameworks (MOFs), and zeolites [73]. The subsequent section will provide a comprehensive discussion of several significant adsorbents, a comparative analysis of the distinctions between various systems, and an in-depth examination of the subject.
4.2.1. Metal-Supported Catalyst
Common metal-supported catalysts comprise metals with denitrogenation and desulfurization activities (e.g., Pt, Cu) that are loaded onto high specific surface area carriers (e.g., activated carbon, zeolite, alumina). It is evident that there is a high degree of similarity between the adsorption design concepts and mechanisms of the two systems. To illustrate this point, consider activated carbon (AC). It is a highly mature adsorbent in practical application, featuring a high specific surface area and different functional groups attached to its surface [74]. Typically, it is loaded with metal ions, such as Cu+ and Ni2+, or modified with acids and bases to enhance its adsorption capacity for sulfur/nitrogen compounds. The adsorption mechanism principally incorporates a series of intermolecular interactions, including π–π interaction, acid–base interaction, hydrogen bonding, and π complexation bonding. It is noteworthy that these mechanisms bear a striking resemblance to those involved in extraction.
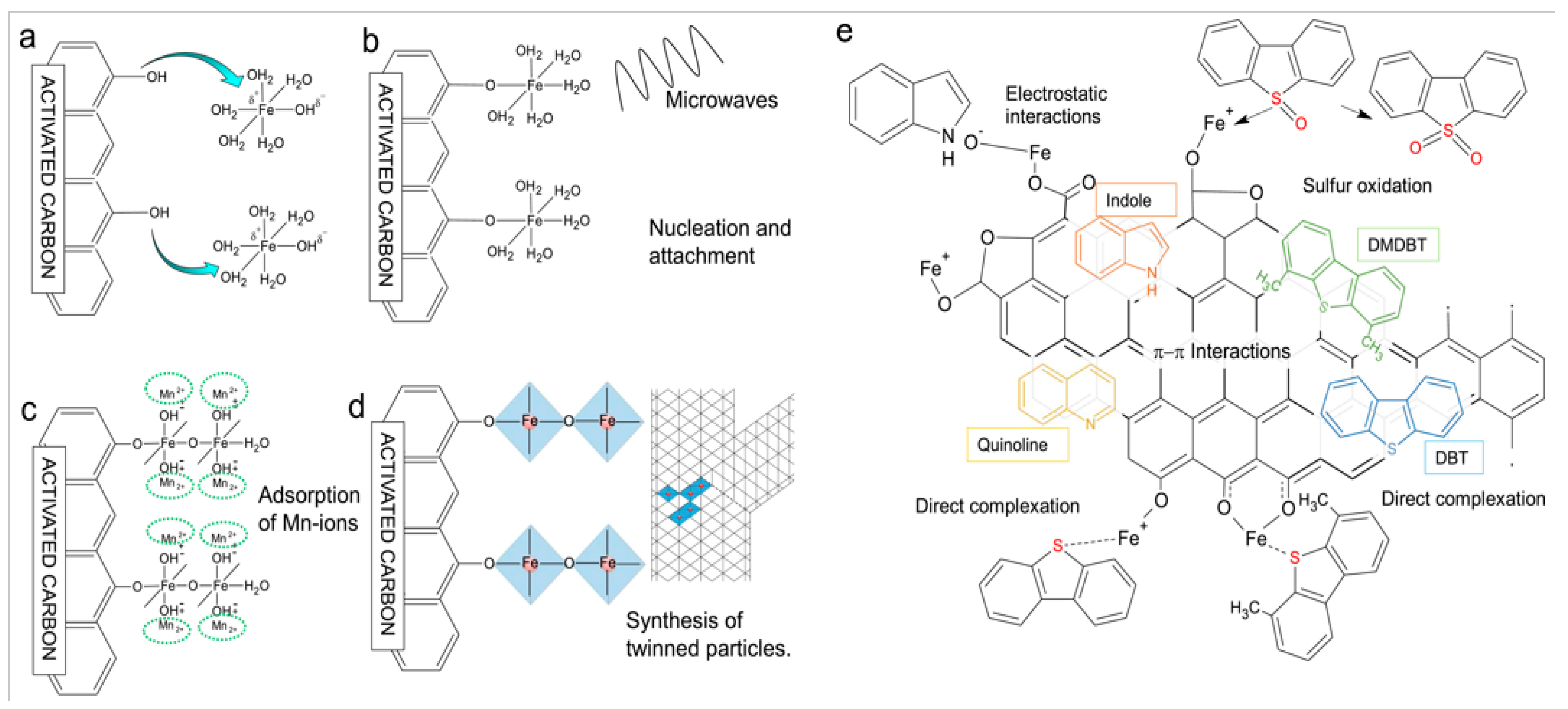
As demonstrated in Figure 8a–d, Arcibar et al. [75] concentrated on the development of iron-manganese microwave-modified activated carbon adsorbents for the purpose of achieving synchronous desulfurization and denitrogenation of fuel oil. It has been demonstrated that the synergistic mechanism of inducing the formation of twinked needle-like structures of iron oxides, the π–π interaction between the activated carbon matrix and aromatic impurities, the acid–base interaction between the acidic sites of iron oxides and nitrogen compounds, and the surface complexation between iron ions and sulfur atoms (Figure 8b), it was found that the comprehensive adsorption performance of F4-FeMn was the best. The maximum adsorption capacity for DMDBT is 0.391 mmol/g, and the adsorption capacities for indole and quinoline are 1.322 mmol/g and 0.756 mmol/L, respectively. Furthermore, it demonstrates a high degree of selectivity for the recalcitrant DMDBT. Arcibar et al. [76] modified activated carbon (AC-I) with iron nanoparticles with a view to enhancing the adsorption capacity of traditional nitrogen compounds. Concurrently, they investigated the influence of sulfur compounds on denitrogenation performance. The results demonstrate that the specific surface area of the modified material (AC-I) undergoes a slight increase, and the surface charge undergoes a transition from alkaline to weakly acidic. In the presence of both indole and quinoline, AC-I exhibited an indole adsorption capacity that was 32% higher than that of AC, while its quinoline adsorption capacity increased by 9%, indicative of its high selectivity for indole. In the presence of 50 ppm sulfur, the adsorption capacity of AC-I for indole and quinoline is still 20% and 8% higher than that of AC, respectively. However, the desulfurization capacity of AC-I is lower than that of AC. This indicates that iron modification enhances the selectivity of the material for nitrogen compounds, and the iron-nitrogen interaction is stronger than the iron-sulfur interaction. Thaligari et al. [77] developed a nickel-modified granular activated carbon (Ni-GAC) catalyst for the simultaneous desulfurization and denitrogenation of fuels. The successful loading of nickel results in the enhancement of the GAC surface uniformity, with the presence of nickel oxides. It is evident that the synchronous adsorption capacity of activated carbon for DBT and QN in liquid fuels was significantly enhanced by nickel modification. This phenomenon can be attributed to the acid–base interaction between Ni2+ and pollutants, as well as the hydrogen bond interaction of surface functional groups. Concurrently, experimental findings indicated that the adsorption capacity of QN with elevated alkalinity was superior to that of DBT, exhibiting a competitive dynamic between the two.
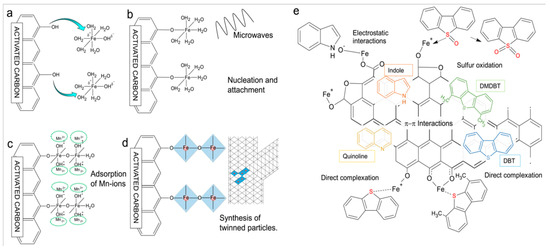
Figure 8.
(a–d) Proposed steps for the formation of twinned particles in material F4-FeMn. Reproduced with permission from Ref. [75]. Copyright 2019 Elsevier. (e) Proposed adsorption mechanism of several molecules studied on the surface of the iron-modified activated carbon. DMDBT stands for 4,6-dimethyldibenzotiophene and DBT for dibenzothiophene. Reproduced with permission from Ref. [75]. Copyright 2019 Elsevier.
Numerous studies have modified carriers to enhance adsorption performance; however, in actual fuel, resins, aromatics, etc. compete with sulfur and nitrogen compounds for adsorption sites, resulting in limited adsorption capacity. The majority of modified activated carbons demonstrate high selectivity for nitrogen compounds; however, they frequently exhibit inadequate removal capacity for challenging pollutants such as 4,6-dimethyldibenzothiophene. In the context of industrial production, although the cycle times and specific thermal degradation points of adsorbents are obviously mentioned in the above three modified activated carbon experiments, according to the commonness of these adsorbents, the oxidation of activated carbon matrix will occur above 600 °C, and the regeneration method is single, which mainly depends on thermal regeneration, with high energy consumption and high cost, preventing its large-scale application.
4.2.2. Metal–Organic Frameworks
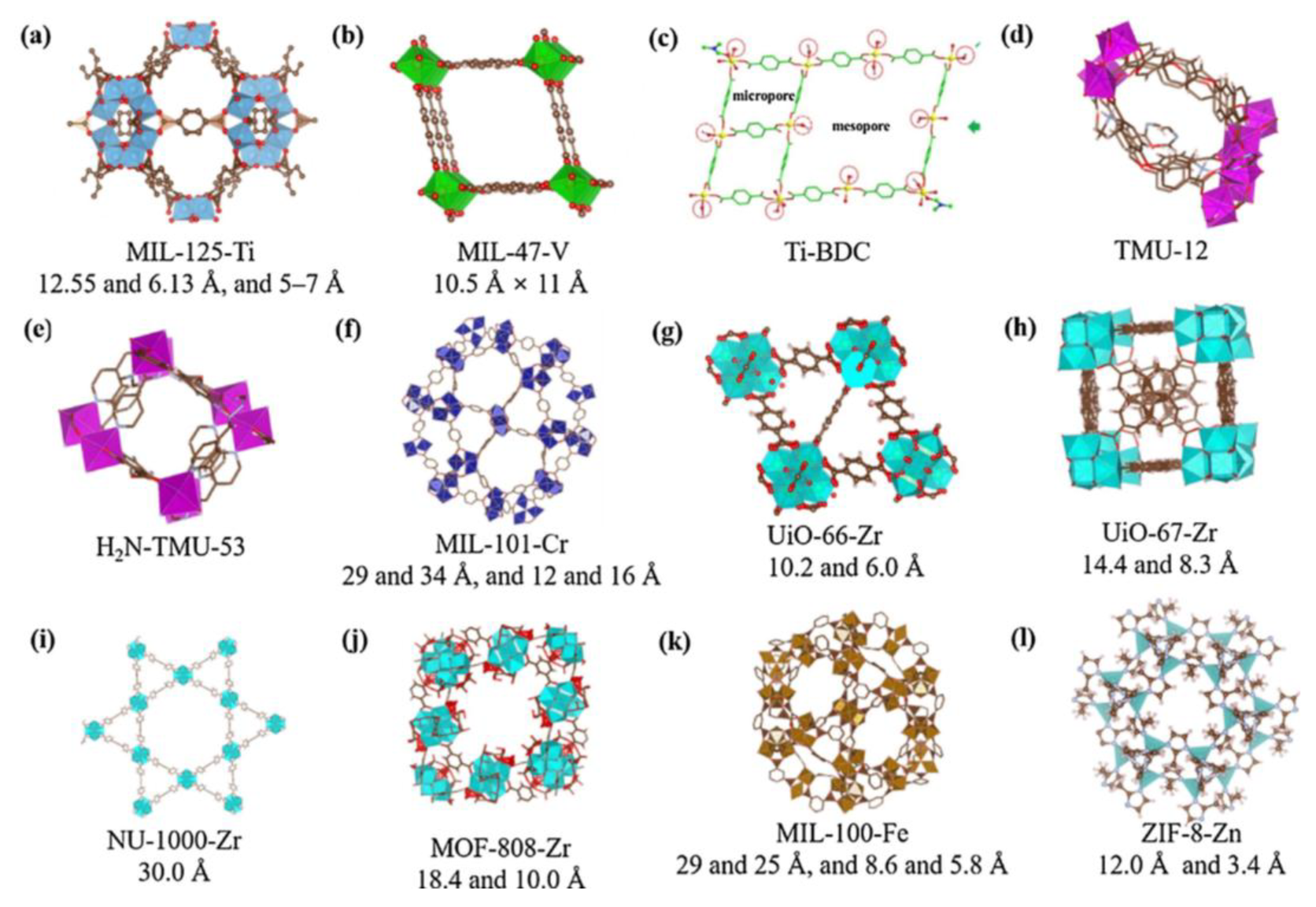
MOFs are defined as porous materials formed by the connection of inorganic metal ions or clusters with organic ligands through coordination bonds. The structural organization of these materials is such that they self-assemble into a three-dimensional network framework through coordination bonds between inorganic metal nodes (including single metal ions and multi-nuclear metal clusters) and organic ligands. This results in high porosity, high specific surface area and structural tunability [78,79]. The synthesis of diverse materials can be achieved through the alteration of metal ions or ligands, with these materials finding wide application in fields such as adsorption and degradation. As illustrated in Figure 9, a range of MOF structures is presented [80]. In comparison with inorganic zeolites, aluminium phosphate, and analogous structures, MOFs can be formed by different metals and a variety of organic ligands. These materials typically exhibit a highly porous structure, which is essential for their functionality in adsorption processes [81,82].
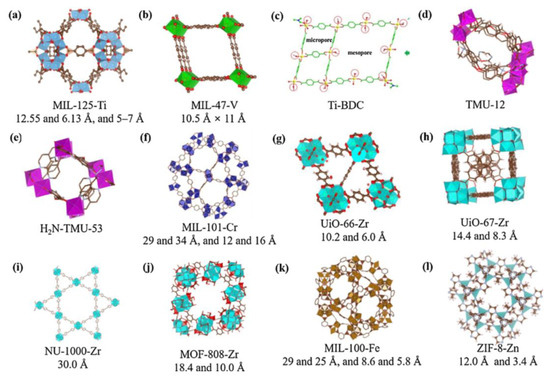
Figure 9.
Structures of various MOFs. Reproduced with permission from Ref. [80]. Copyright 2019 Elsevier.
Tan et al. [83] prepared HKUST-1/Fe3O4 composites using a novel dry gel transformation method (see Figure 10a). The composite in question is a magnetic MOF, which is characterized by the presence of both micropores and mesoporous pores. The open metal sites of quinoline and HKUST-1 are adsorbed through acid–base interaction. Indole, due to the presence of N-H bonds, has the capacity to form hydrogen bonds with the framework and thus ignores the effect of steric hindrance. This results in an adsorption capacity that is much higher than that of quinoline. However, the adsorption of sulfur compounds is contingent upon acid–base interaction and π complexation. The present study investigates the efficacy of an adsorption system, subject to an externally applied magnetic field, in the removal of aromatic sulfur and nitrogen compounds in fuel. The adsorption capacity of this adsorbent is superior to that of traditional adsorbents such as activated carbon and graphite. The adsorbent was separated by a magnetic field and regenerated. The recovery rate of the adsorbent was more than 99% each time. After six consecutive cycles, the cycle stability was outstanding. In the seminal study, Khan et al. [84] pioneered the loading of Sc(OTF)3 onto MOFs, marking a pivotal advancement in the field. Utilizing the inherent acidity of Sc(OTF)3, they demonstrated a remarkable enhancement in the adsorption selectivity for sulfur and nitrogen compounds through the judicious exploitation of acid–base interactions. Figure 10b illustrates the structure of the absorbent that undergoes modifications upon loading. The study found that the adsorption effects on benzothiophene, dibenzothiophene and quinoline were significantly increased by 64.7%, 20.7% and 34.2%, respectively. However, the absorption of indole did not show a significant increase. The underlying reason for this phenomenon is that it is incapable of forming an acid–base interaction with acidic Sc(OTF)3. Furthermore, following the loading process, the porosity of MOFs is reduced, thereby hindering the process of adsorption. The adsorbent can be stably recycled for 4 times, and the regeneration process is mild.
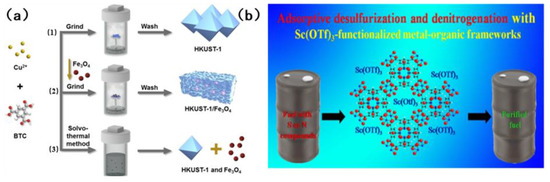
Figure 10.
(a) Preparation method of HKUST-1/Fe3O4 composite material. Reproduced with permission from Ref. [83]. Copyright 2016 Elsevier. (b) The structure of Sc (OTF)3 adsorbent. Reproduced with permission from Ref. [84]. Copyright 2015 ACS.
Metal–organic frameworks (MOFs) have demonstrated considerable potential in the domains of fuel desulfurization and denitrogenation, attributable to their elevated specific surface area and porous structural characteristics. In comparison with materials such as activated carbon and zeolite, the diversity of its preparation types and the variability of its structure greatly enhance the adsorption performance. However, when dealing with large-volume sulfur or nitrogen compounds that are difficult to remove, the clogging of pores leads to slow adsorption kinetics. Concurrently, the majority of MOF materials are confronted with defects such as competition for adsorption sites, elevated preparation costs, constrained thermal stability, and degradation of regeneration performance. These practical application problems bear a strong resemblance to those encountered in the field of activated carbon.
4.3. Simultaneous Oxidative Desulfurization-Denitrogenation
Oxidation desulfurization (ODS) and oxidation denitrogenation (ODN) in fuel are usually carried out in a system of oxidant and catalyst at normal temperature and pressure. Therefore, the selection of the catalyst and oxidant is a key link in the oxidation system. Sulfur oxides will be oxidized to the corresponding sulfoxides or sulfones [2], while the oxidation products of nitrogen oxides are more complex. Aniline is oxidized to azoxylbenzene, carbazole is oxidized to carbazole 1,4-dione, and the oxidation of indole produces polymer materials. In ODS/ODN, ozone and hydrogen peroxide are used as oxidants to destroy the C-S bond of organic sulfur compounds and functional groups of nitrogen-containing compounds in fuel oil through selective oxidation, and the low valent sulfur and nitrogen are oxidized into sulfur oxides such as sulfones and sulfonic acids and nitrogen oxides, carboxylic acids and other nitrogen-containing oxidation products, respectively. This oxidation method fundamentally changes the problem of the weak polarity of original sulfur compounds and nitrogen compounds and the small solubility difference with alkanes, and the generated oxides have stronger polarity and higher electronegativity, providing good conditions for subsequent separation. At the same time, the adsorbent/extractant used in the actual production can not only remove the nitrogen base, heavy metal ions and other components that may poison the oxidation catalyst in the real fuel, but also remove the oxidation products in time to reduce the probability of blocking the catalyst channel. Therefore, the efficient separation of oxidation products and fuel oil is often achieved by means of extraction, centrifugation and other separation methods, and ultimately, the purpose of deep purification of fuel oil is achieved. The oxidation method is always used in combination with extraction or adsorption [2], and can also be used in conjunction with auxiliary technologies such as visible or ultraviolet light irradiation and ultrasound [26] to enhance the removal rate. High selectivity catalysts are more convenient to control and improve removal efficiency (Table 1).
4.3.1. Choice of Oxidant
In a reaction system where oxidation, desulfurization and denitrogenation are carried out simultaneously, the selection of the oxidant is of vital importance, which is usually dependent on the hydrophobicity of the catalyst. Oxidants, during the reaction process, release peroxide free radicals, provide reactive oxygen species to break the C-S and C-N bond of sulfur nitrogen compounds and oxidize them into polar and easily separable products. Consequently, the reaction rate of the oxidation process can be affected by the presence of inappropriate oxidants. As demonstrated in the extant research, the most prevalent oxidants encompass TBHP, hydrogen peroxide, molecular oxygen, O3, air, amongst others. O3 [85] has become an alternative to traditional catalysts due to its high selectivity, strong oxidizing properties, and environmental friendliness. It can oxidize pollutants in a short period of time by breaking the O-O bond. H2O2 is a widely used and environmentally friendly oxidant, with only H2O, but its biodegradability requires excessive addition to ensure sufficient active oxides. H2O2, as a byproduct, can be used as a co oxidant for heterogeneous thermal catalysts and also as the main oxidant in catalysis. TBHP, as an oxidant and heterogeneous catalyst in oxidation reactions, often leads to the formation of complexes and metal peroxide intermediates through nucleophilic attacks. The use of TBHP or H2O2 as an oxidant is usually in the form of an aqueous solution. Two phases are introduced into the reaction system to inhibit mass transfer and affect the reaction rate. Although molecular oxygen or air can overcome this obstacle when used as an oxidant, it requires high pressure and high purification, which greatly increases the cost [86]. It is evident that H2O2 demonstrates elevated reaction rates and selectivity under mild conditions, thereby exhibiting superior performance in comparison to other oxidants. It has been demonstrated in previous studies that, in the event of H2O2 being utilized as an oxidant, the presence of a hydrophobic catalyst is an essential requisite [87]. The efficacy of the process is optimized by the utilization of silicate catalysts, while metal oxide catalysts belonging to the IV-VI group demonstrate compatibility with tert-butyl hydrogen peroxide. Furthermore, as the temperature rises, H2O2 decomposes into O2 and H2O, thereby inhibiting the reaction. Excessive H2O2 has been shown to eliminate hydroxyl radicals with oxidizing effects, thereby seriously affecting the efficiency of desulfurization and denitrogenation [88]. Furthermore, the mechanism of oxidation reactions can also be subject to alteration depending on the different oxidants. When molecular oxygen is utilized as the oxidant, the predominant reaction mechanism is that of free radicals [89]. When H2O2 is utilized as the oxidant, the oxidation reaction progresses through the generation of metal peroxide complexes of the catalyst.

Table 1.
Various Important Parameters of Catalysts in ODS.
Table 1.
Various Important Parameters of Catalysts in ODS.
| Catalyst | n (Catalyst) | Oxidant | Reactant | O/S | Temperature (°C) | Time (min) | Conversion Rate | Ref. |
|---|---|---|---|---|---|---|---|---|
| [cetrimonium]11P2W13V5O62 | 7.5 g/L | H2O2 | DBT; Quinoline | 8 | 70 | 45 | DBT:94% Q:100% | [90] |
| Tris-LDH-LaW1 | N(DBT)/n(catalyst) = 20 | H2O2 | Sulfides: DBT, BT, 4,6-DMDBT; Nitrides: Quinoline, Pyridine, Indole | 6 | 65 | 60 | S:99%: N:99% | [91] |
| HPA-5 | 0.5 mmol | O2 | Sulfides: (Benzothiophene, DBT 4,6-DMDBT); Nitrides (indole, 1-methylindole, 2-methylindole, 3-methylindole, quinoline, etc.); Quinalidine | — | 120 | — | N:100% (5 min); S:88% (24 h) | [92] |
| TA-700; AM-700 | 200 mg | H2O2 | Sulfides (DBT, 4-methylDBT, 4,6-dimethylDBT, benzothiophene, thiophene), nitrides; SRGO; diesel | 20 | 60 | 5 | DBT:100% Diesel oil:S:97%;N:96%; SRGO:S:70%; N:89% | [93] |
| MoO3/Al2O3 | 5 wt% | CHP | Sulfides (4-methylDBT, 4,6-dimethylDBT) Nitrides, polycyclic aromatic hydrocarbons (PAHs); HGO1, HGO2 | 20 | 75 | — | HGO1:S:83%; HGO2:S:39% | [94] |
| Mn−Co−Mo/Al2O3 | 0.1 g | H2O2 NaClO | Sulfides (DBT, BT, 4,6-DMDBT) Nitrides (pyridine, indole, carbazole) | 4 O/N = 8 | 25 | 15 | DBT:100% PY:100% | [95] |
| MAX-phase@rGO@PW11Zn | 0.01 g | H2O2 | Sulfides (thiophene, TH, thiols) Nitrides (pyridine, PY); gasoline | — | 35 | 60 | TH:97.72% PY:98.94% | [96] |
The ‘—’ represents a value where the parameter does not appear.
As shown in Table 2, a detailed comparison of various commonly used oxidants was made from different perspectives. Combined with the research reports mentioned in the following section, in most cases, H2O2 and O2 are the better choices.

Table 2.
Multidimensional comparison of different oxidants.
4.3.2. Choice of Catalyst
Catalysts are the most crucial reactants in the oxidation system. In contrast to the complexity and inconvenience of the homogeneous catalytic system, the heterogeneous catalytic system is more favored. It has advantages such as easier separation, regeneration, and reusability of the catalyst. Nevertheless, regardless of the reaction system type, the time consumed for the reaction is extremely short.
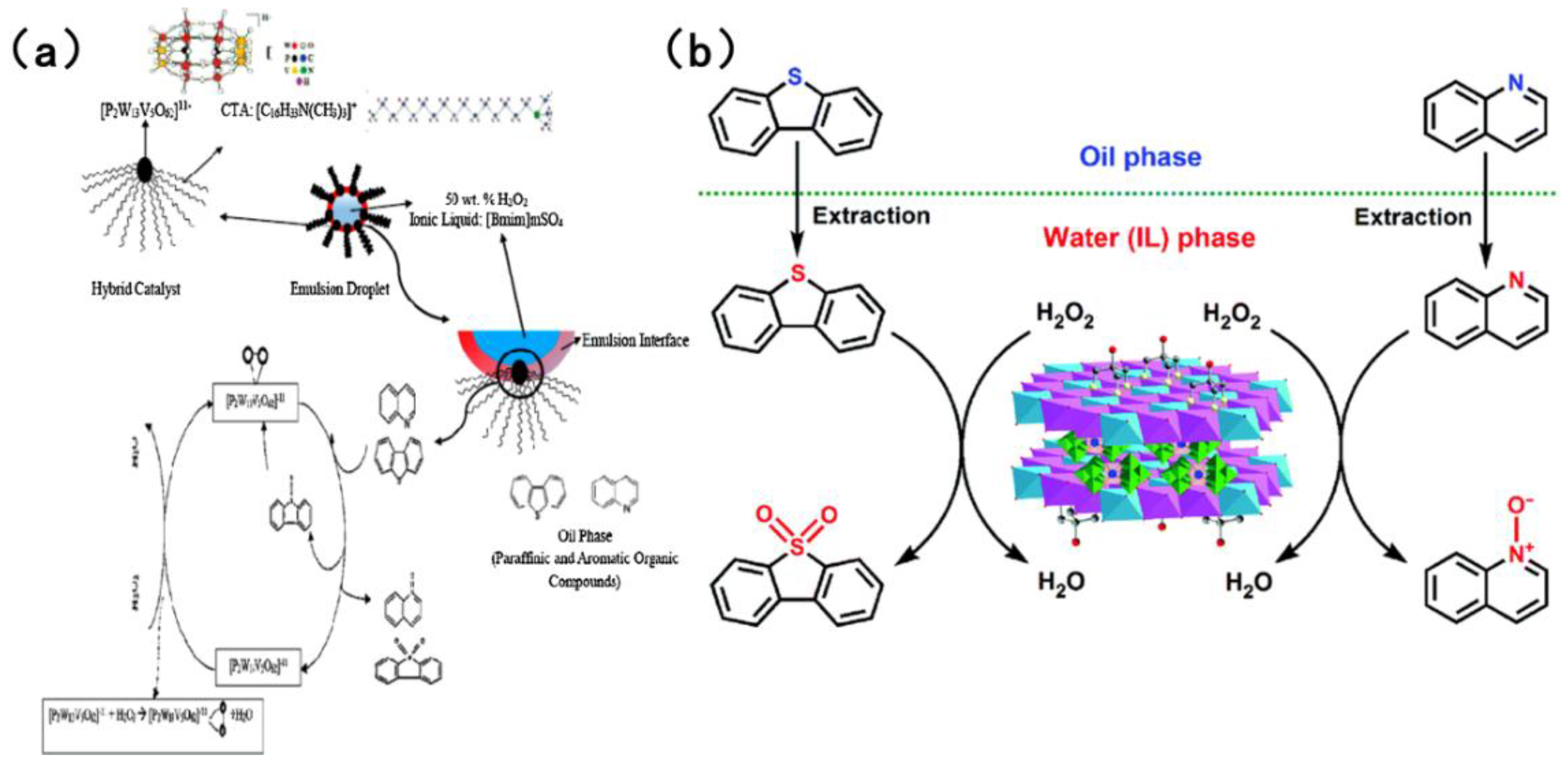
In the field of catalysts for the simultaneous oxidation-based denitrogenation and desulfurization of organic compounds, polyoxometalate and heteropolyacid catalysts have attracted considerable interest [92]. The most prevalent of these are Keggin-type and Dawson-type polyoxometalate catalysts. These catalysts can be modified by incorporating transition metals such as vanadium to enhance their catalytic efficacy. As shown in Figure 11a, Banisharif et al. [90] developed an ECODS system that uses a vanadium-substituted Dawson-type emulsion catalyst alongside an ionic liquid (IL). Using H2O2 as the oxidant, this system efficiently removed dibenzothiophene (DBT) and quinoline from model fuels simultaneously. The long alkyl chains of cetyltrimethylammonium bromide (CTAB) in the emulsion catalyst were found to act as phase-transfer agents, facilitating the diffusion of DBT and quinoline from the non-polar oil phase into the polar solvent phase. Furthermore, the vanadium-substituted Dawson-type catalyst provided potent oxidation sites, activating H2O2 to generate reactive oxygen species. When in contact with -O-O- species on the catalyst surface, DBT is oxidized to DBTO2 and quinoline is oxidized to quinoline-N-oxide. Yao et al. [91] fabricated a Tris-modified layered double hydroxide (LDH) that was utilized as a support for the synthesis of a LaW10-type polyoxometalate (Tris-LDH-LaW10). Through electrostatic interactions and hydrogen bonding, the [LaW10O36]9− active sites were immobilized within the LDH interlayers. Utilizing 30 wt% H2O2 as the oxidant, as demonstrated in Figure 11b, at 65 °C and atmospheric pressure, the ionic liquid facilitated the extraction of sulfur- and nitrogen-containing compounds from the oil phase to the aqueous phase. The [LaW10O36]9− in the catalyst reacted with H2O2 to generate stable W-peroxide active species, oxidizing DBT to dibenzothiophene sulfones and quinoline to quinoline nitrogen oxides. Subsequently, the oxidation products were efficiently separated by the ionic liquid (via extraction), thus achieving simultaneous removal. Quinoline and DBT are both adsorbed on the active sites of the catalyst through π–π stacking and acid–base interactions. Quinoline has a slightly higher adsorption affinity than DBT, but due to sufficient active sites on the catalyst, the oxidation rate of DBT only decreases by less than 10%. Bertleff et al. [92] synthesized vanadium-substituted heteropolyacids with a Keggin structure. In high-temperature and high-pressure systems, molecular oxygen was selected as the oxidant. Conversely, in other systems, air was utilized as the oxidant. V5+ and Mo6+ in HPA-5 have multi electron redox ability. O2 reacts with the catalyst to generate reactive oxygen species (such as •OH). The introduction of vanadium enhances the oxidation selectivity of the catalyst for nitrogen compounds. Nitrogen compounds such as indole and quinoline first adsorb at the active site of the catalyst and are oxidized by the reactive oxygen species. Indole compounds generate carboxylic acids and CO2, while quinoline compounds generate quinoline N-oxides, which are further degraded into small molecule acids. The gaseous product is mainly N2. In both reaction systems, nitrogen compounds, excluding 1-methylindole, could be removed within an extremely short period. Furthermore, it was established that 1-methylindole, functioning as a reducing agent, effectively activated the catalyst, reduce part of V5+ in the catalyst to V4+ to form V4+/V5+ redox cycle and significantly enhancing the desulfurization efficiency across a range of simulated fuels.
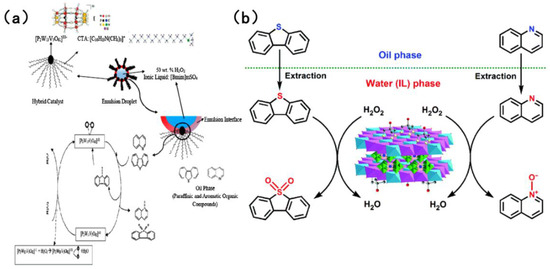
Figure 11.
(a) Proposed mechanism of extractive-catalytic oxidative desulfurization and denitrogenation using ionic liquid. Reproduced with permission from Ref. [90]. Copyright 2017 Elsevier. (b) Proposed extraction and catalytic oxidative desulfurization and denitrogenation process by the catalytic system of H2O2/[bmim]BF4/TrisLDH-LaW10. Reproduced with permission from Ref. [91]. Copyright 2017 RSC.
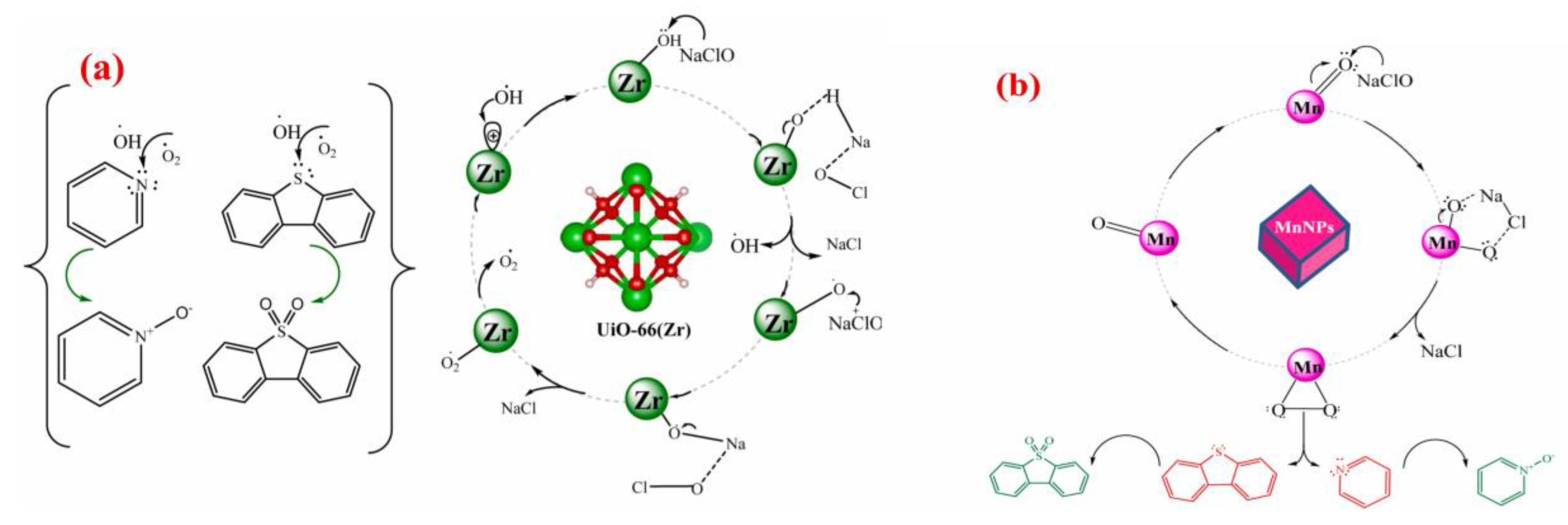
Metal–organic frameworks (MOFs) are porous materials combining organic and inorganic characteristics [10]. They not only exhibit remarkable performance in adsorption but also demonstrate excellent capabilities in catalytic oxidation. MOFs provide adsorption sites for adsorption processes and mainly offer active sites in oxidation reactions, relying on active centers modified by metal ions or ligands [97,98] to activate oxidants and generate free radicals or reactive oxygen species. The previous section has reviewed various common MOF structures, and most of these structures can form derivatives via methods such as oxidation, pyrolysis, and coordination exchange [99], retaining some MOF characteristics or generating new structures. Palomeque et al. [93] selected ZrO2 (a UiO-66 derivative) as the support and tungstic acid (H2WO4) as the tungsten precursor, fabricating WOx/ZrO2 catalysts through two distinct methods: anion exchange (TA) and impregnation (AM), targeting the synergistic oxidative abatement of sulfur and nitrogen compounds in fuel oil. H2O2 forms the intermediate of hydroperoxytungstate, releases reactive oxygen species through peroxide bond breaking, and reacts with tetrahedral WO42− to generate W-O-O-H. Tungsten peroxide species attack the sulfur atom of sulfur compounds and the nitrogen atom of nitrogen compounds, and are oxidized to N-oxide sulfone and carboxylic acid products through nucleophilic attack. Due to the formation of hydroperoxytungstate intermediates, there is a problem of activity allocation caused by non-selective competition. Detailed characterizations revealed that the TA catalyst was dominated by tetrahedral tungsten species, which contributed to the formation of stronger Brønsted and Lewis acid sites—key active centers for ODS/ODN reactions. Consequently, it achieved a maximum desulfurization efficiency of 97% and denitrogenation efficiency of 95%, with superior reactivity toward refractory S/N compounds (e.g., sterically hindered thiophenes and pyridines). In contrast, the AM catalyst exhibited octahedral tungsten configurations, accompanied by reduced acid site density and the formation of crystalline WO3 phases. These structural features induced the agglomeration of tungsten species, weakened the metal-support interaction, and ultimately compromised catalytic performance compared to the TA catalyst. Density functional theory (DFT) calculations further elucidated the intrinsic activity mechanism of tetrahedral tungsten, verifying its lower reaction energy barrier for oxidant activation and pollutant cleavage. Subhan et al. [98] constructed a MnO2/UiO-66 composite catalyst by anchoring MnO2 nanoparticles onto UiO-66, employing NaClO as the oxidant for the simultaneous oxidative removal of DBT, indole, and other typical S/N contaminants. Experimental results demonstrated that the introduction of MnO2 nanoparticles significantly increased the specific surface area of UiO-66 by 88%, which facilitated the exposure of active sites (Mn4+ and Zr4+) and enhanced mass transfer during reactions. As depicted in Figure 12, the dissociation of NaClO produces ClO−, which interacts specifically with Mn4+ and Zr4+ active sites on the composite catalyst. This interaction triggers the heterolytic cleavage of ClO−, generating two highly reactive oxygen species with strong oxidizing capacity. These radicals selectively attack the sulfur atom in DBT and the nitrogen atom in pyridine, converting them into dibenzothiophene sulfone and pyridine N-oxide, respectively—both of which are easily separable from fuel oil. Notably, this catalytic system achieved complete (100%) removal of DBT and pyridine under ambient temperature and pressure, realizing rapid desulfurization and denitrogenation. Moreover, the by-products (NaCl and H2O) are inert and non-toxic, avoiding the introduction of impurities that could degrade fuel quality. Regarding the potential competitive relationship between sulfide and nitride removal, previous studies [100] have shown that nitrides such as pyridine and pyrrole have smaller sizes and adsorb into the micropores of the catalyst, preventing effective adsorption of thiophene and other compounds on the catalyst. By excessive use of oxidants, it had been demonstrated that sulfide removal is only affected by the competitive adsorption between the two, and the possibility of significant competitive oxidation has been ruled out. Therefore, the oxidation efficiency is highly dependent on the catalyst porosity.
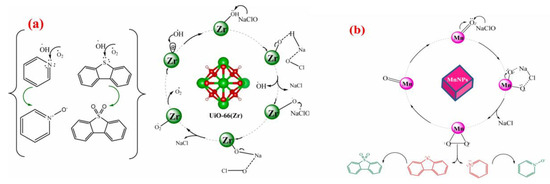
Figure 12.
Proposed catalytic oxidation mechanism of Zr (a), and Mn (b) active species in MnO2/UiO-66 using NaClO as oxidant. Reproduced with permission from Ref. [98]. Copyright 2021 Elsevier.
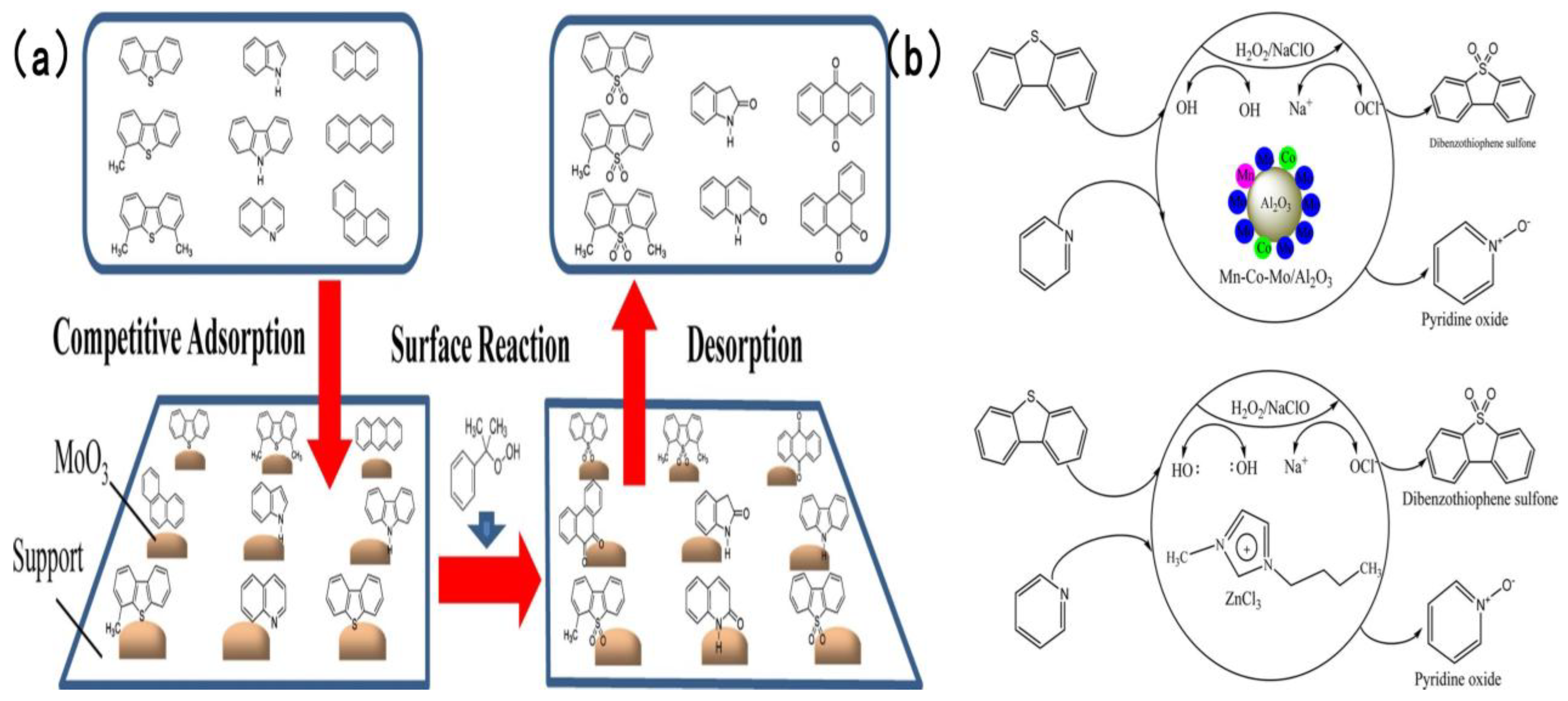
In addition to the two types of catalysts discussed earlier, numerous studies have adopted supports including Al2O3 to fabricate highly efficient catalysts for simultaneous oxidative desulfurization and denitrogenation (ODS/ODN). Safa et al. [94] prepared a 12 wt% MoO3/γ-Al2O3 catalyst, using 80% cumene hydroperoxide as the oil-soluble oxidant, the active site of the catalyst adsorbs CHP and decomposed it into two strong oxidation species, t-Buo• and Mo6+-O-O-t-Bu and carried out an in-depth investigation into the mechanism underlying the influence of nitrogen compounds and polycyclic aromatic hydrocarbons (PAHs) on the oxidative removal of sulfur compounds. Experimental findings revealed that as the nitrogen concentration increased from 0 to 237 ppmw, the oxidation rates of 4,6-dimethyldibenzothiophene (4,6-DMDBT) and 4-methyldibenzothiophene (4-MDBT) declined remarkably as the content of impurities increased. Notably, 4-MDBT exhibited higher sensitivity toward impurities than 4,6-DMDBT. As illustrated in Figure 13a [94], gas chromatography-nitrogen chemiluminescence detection (GC-NCD) analysis validated that nitrogen compounds were partially oxidized during ODS. This suggests that nitrogen compounds compete against sulfur compounds for catalyst active sites and can be simultaneously removed. Subban et al. [95] developed a series of Al2O3-supported catalysts, encompassing Co-Mo/Al2O3, Ni-Mo/Al2O3, Mn-Co-Mo/Al2O3 and Mn-Ni-Mo/Al2O3. They evaluated and compared the oxidative removal efficiency of two oxidant systems: hydrogen peroxide (H2O2) and sodium hypochlorite (NaClO). Experimental results indicated that in the NaClO system at 25 °C, complete removal of dibenzothiophene (DBT) and pyridine was attained within 15 and 20 min, respectively—markedly superior to that in the H2O2 system. Figure 13b [95] depicts the detailed reaction mechanism: hypochlorite ions (ClO−) produced via NaClO dissociation possess stronger oxidizing power, integrated to the Mn active site and released after cleavage •OCl and enhanced coordination with Mn active sites than the peroxy species generated by H2O2, thus minimizing the unproductive consumption of the oxidant.
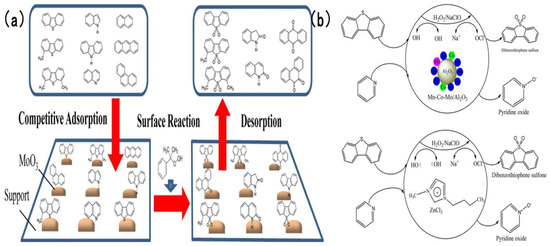
Figure 13.
(a) Proposed ODS mechanism of dibenzothiophenes in the presence of nitrogen compounds and PAH. Reproduced with permission from Ref. [94]. Copyright 2020 Elsevier. (b) Proposed reaction mechanism for the CODS and CODN of DBT and pyridine in the presence of H2O2 and NaClO as oxidants over Mn–Co–Mo/Al2O3 and [Bmim]Cl/ZnCl2 as catalysts. Reproduced with permission from Ref. [95]. Copyright 2019 ACS.
Most of the oxidants studied are mainly H2O2 or O2, while ozone, as a strong oxidant, has a very similar oxidation principle to other oxidants, which oxidizes organic sulfides to corresponding sulfones or sulfoxides. These oxidation products have enhanced polarity and are easily separated from the fuel by extraction, adsorption, or precipitation [101]. The difference is that O3 is oxidized without a catalyst. Ma et al. [101] used ozone generated by dielectric barrier discharge plasma combined with ionic liquid extraction to conduct deep oxidative desulfurization of simulated fuel. The order of oxidation activity of the four sulfides was TS > BT >> DBT > 4,6-dmdbt, which was different from that of the traditional hydrogen peroxide oxidation system. The [bmim] AC used has a stronger extraction ability for oxidation products, and the desulfurization rate of BT can reach 99.9%. Adhami et al. [102] used ozone microbubbles for oxidation to improve the contact efficiency between ozone and fuel and accelerate the oxidation reaction. The reaction is carried out through two paths: direct oxidation and free radical chain reaction. In the direct oxidation path, ozone molecule directly reacts with sulfide, breaks the C-S bond and gradually oxidizes the sulfur atom to sulfoxide (S=O, +4) and sulfones (O=S=O, +6). In the free radical chain reaction path, ozone first splits into •Roh, which grabs the hydrogen atom of sulfide to form R-S•, then the sulfur center radical reacts with ozone to form R-SO•, and finally the sulfoxide radical reacts with ozone or hydroxyl radical to form sulfone products. At present, there are few research papers on ozone denitrogenation or simultaneous denitrogenation and desulfurization, which is a challenging but promising field.
4.3.3. Auxiliary Technology
To further enhance the performance of oxidative desulfurization and denitrogenation (ODS/ODN), integrating auxiliary approaches such as light irradiation or ultrasound into catalytic oxidation systems has notably boosted oxidation kinetics and process efficiency [96,103]. Photocatalysis operates by light excitation, prompting valence band electrons to undergo transition, leaving behind holes with strong oxidizing capacity; these can synergize with active sites generated by transition metals contained in the catalyst. Ultrasound-assisted technology employs ultrasound waves to generate physical effects that disrupt the aggregated state of sulfur and nitrogen compounds, alongside chemical effects that induce the cleavage of water molecules or oxygen to produce free radicals.
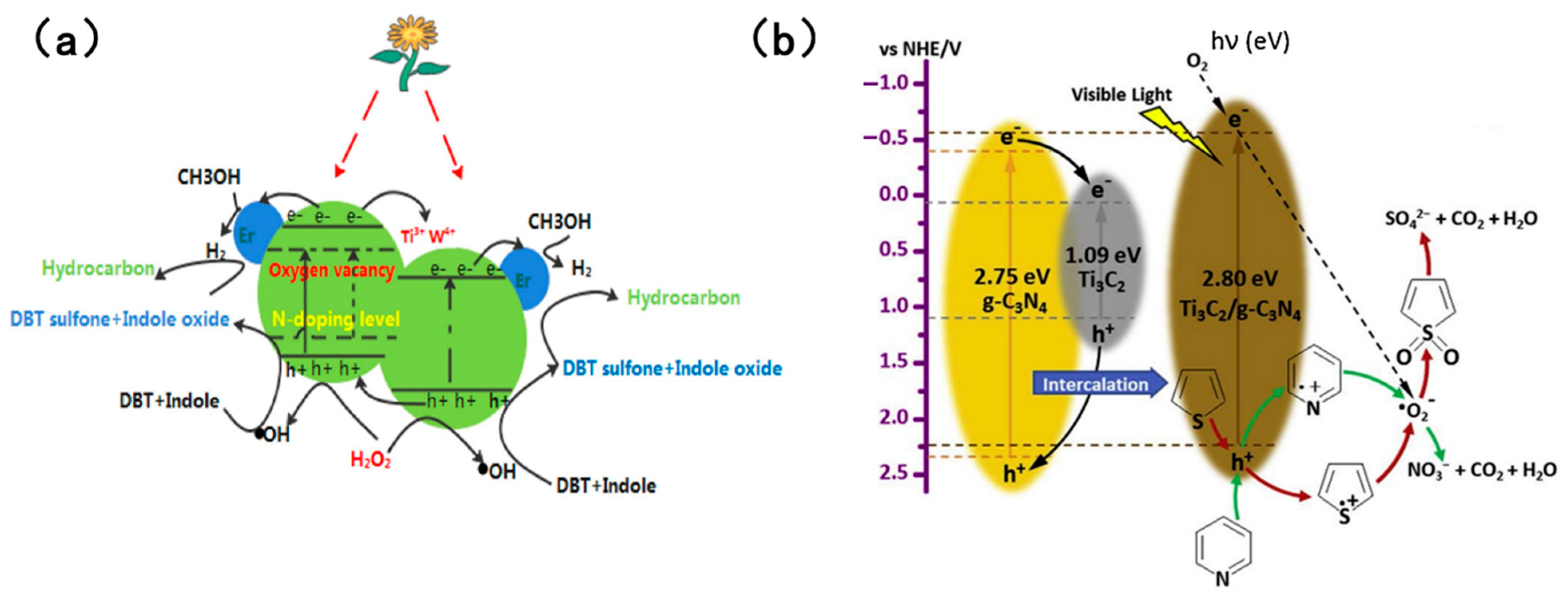
A noteworthy study was reported by Aghbolagh et al. [96], who innovatively constructed a MAX-phase@rGO@PW11Zn composite photocatalyst to address the limitations of single-component catalysts, achieving efficient photocatalytic removal of sulfur and nitrogen compounds in fuel oil. When the catalyst dosage was 0.01 g and the reaction time was 60 min, the removal efficiencies of thiophene (TH) and pyridine (PY) reached 97.72% and 98.84%, respectively—markedly outperforming those of single-component catalysts. Wang et al. [104] fabricated an Er, W, N co-doped photocatalyst (Er/W-N-TiO2) and employed H2O2 as the oxidant for simultaneous denitrogenation and desulfurization. Figure 14a [104] illustrates the specific reaction mechanism: under visible light irradiation, valence band electrons of TiO2 are excited to the conduction band, generating photogenerated electrons and holes. Meanwhile, W4+/W6+ redox pairs, Er3+ ions, and oxygen vacancies in the catalyst effectively trap electrons, significantly suppressing electron–hole recombination. The holes oxidize H2O2 to produce hydroxyl radicals (•OH), which serve as the core active species for oxidizing sulfur and nitrogen compounds, while electrons are captured by methanol to generate hydrogen radicals (•H). Ultimately, sulfur compounds are oxidized by •OH into more polar sulfones, and nitrogen compounds are converted into nitrogen oxides, realizing deep purification of fuel oil. However, alkaline nitrogen oxides compete fiercely with sulfides for the active sites of the catalyst. Li et al. [79] developed a photocatalytic reaction system using Ti3C2/g-C3N4 as the catalyst and air as the oxygen source. Figure 14b [79] depicts the core mechanism: holes act as the primary active species driving desulfurization and denitrogenation reactions, preferentially undergoing electrophilic oxidation with pyridine and thiophene to form corresponding cationic intermediates (C5H5N+ and C4H4S+). In contrast, superoxide radicals () generated by the combination of conduction band electrons with dissolved oxygen in air function as auxiliary active species, promoting further deep oxidation of the intermediates.

Figure 14.
(a) Schematic presentation of charge carrier separation in the desulfurization and denitrogenation over Er/W-N-TiO2 aided by visible light. Reproduced with permission from Ref. [104]. Copyright 2021 Elsevier. (b) Proposed photocatalytic mechanism of PODN and PODS. Reproduced with permission from Ref. [86]. Copyright 2020 Elsevier.
Eventually, pyridine is mineralized into NO3−, CO2, and H2O, while thiophene is converted into SO42− via sulfone intermediates.
Ultrasound assists oxidative desulfurization and denitrogenation (ODS/ODN) through its induced physical and chemical effects, featuring affordable economic cost and high removal efficiency for aromatic compounds. Notably, a review has elaborated on the key parameters in the auxiliary oxidation process, including temperature, ultrasonic power and frequency, pH value, initial concentrations of sulfur and nitrogen compounds, the role of phase transfer agents, extraction solvents, reaction kinetics, and fuel properties. Despite being a promising auxiliary technology, few studies have focused on simultaneous desulfurization and denitrogenation in recent years. Consequently, this technology remains largely at the theoretical stage, lacking sufficient in-depth experiments to be convincing.
In actual industrial production, sulfur oxides and nitrogen oxides are mostly removed by extraction [105]. Using the polarity difference between oxidation products and unreacted hydrocarbon fuels, sulfur oxides are extracted from non-polar fuels by selecting appropriate polar solvents [106]. Common traditional polar solvents include acetonitrile and methanol. Many studies [106] have shown that the solubility of oxidized organic sulfur compounds in acetonitrile is significantly increased, which can effectively separate sulfone products. In addition, ionic liquids have attracted extensive attention due to their designability, low vapor pressure and good extraction selectivity for sulfur/nitrogen compounds [54,107]. Ionic liquids can simultaneously or selectively extract sulfur and nitrogen compounds. Benzyl ionic liquids such as and have been proven to have high extraction desulfurization capacity for BT and DBT. Therefore, the combination of oxidation and extraction methods can achieve the goal of efficient removal of nitrides and sulfides, while avoiding secondary pollution. In addition, activated carbon, alumina, molecular sieve, MOF and other adsorption materials have been used for the removal of sulfur oxides [108] when the concentration of sulfur oxides is high or the selected oxidation system leads to a significant decrease in the solubility of the product. Oxidation products may form precipitates, which can be separated by physical methods such as filtration or centrifugation.
Simultaneous oxidative desulfurization and denitrogenation offers mild reaction conditions and high selectivity toward refractory sulfur and nitrogen compounds. It can also incorporate external auxiliary technologies like photocatalysis or ultrasound, enabling deep pollutant removal. However, challenges persist in practical applications: the commonly used H2O2 oxidant in reaction systems exhibits poor stability, catalyst costs are relatively high, and pollutants compete for catalytically active sites. Moreover, catalyst regeneration involves irreversible deactivation of active sites, making it difficult to fully restore performance, significant differences in regeneration mechanisms, and a lack of practical solutions. These issues result in a mismatch between oxidation methods and complex industrial systems, often remaining in the experimental stage.
4.4. Simultaneous Biodesulfurization-Biodenitrogenation
Biodesulfurization (BDS) and biodenitrogenation (BDN) utilize enzymes secreted by microorganisms to decompose sulfur compounds and nitrogen compounds in fuel oil. This technology boasts merits including low cost, minimal environmental pollution, and no adverse by-products. However, the slow growth rate of microorganisms and sluggish reaction kinetics necessitate prolonged degradation periods. In biocatalytic systems, attention must be paid to microbial selection, medium composition, reaction conditions, and the contact efficiency between microorganisms and substrates. Among the latest research advances, biofilms have been identified as a bridge facilitating metabolic and physiological interactions among microorganisms, and BDS-BDN systems based on biofilms are believed to surpass the limitations associated with suspended-cell BDS-BDN systems [2]. Currently, most studies have focused solely on either desulfurization or denitrogenation, with few reports documenting simultaneous biological desulfurization and denitrogenation. The most extensively studied pathway for BDS is the 4S pathway, which involves sulfoxidation, sulfonation, C-S bond cleavage to form sulfonates, and final oxidation to sulfates or sulfites. Beyond this, microorganisms can also achieve degradation via C-C bond cleavage: sulfur-containing compounds first undergo preliminary oxidation to weaken the stability of C-C bonds, followed by C-C bond cleavage catalyzed by specific enzymes. The fragmented carbon skeletons then enter the tricarboxylic acid (TCA) cycle of microorganisms and are ultimately mineralized into CO2 and H2O. The degradation mechanism of nitrogen compounds is strikingly similar to that of sulfur compounds; the key difference lies in the cleavage of C-N bonds instead of C-S bonds for nitrogen compound removal.
Dinamarca et al. [109] employed two bacterial strains, Rhodococcus and Cobetia sp. strain MM1IDA2H-1, to investigate the removal of dibenzothiophene (DBT) and quinoline (QN). In the mixed biofilm system, the shared carrier surface between the two bacterial strains results in reduced metabolic activity per unit area. Meanwhile, 2-hydroxybiphenyl—an intermediate product generated during DBT desulfurization by Rhodococcus—exerts a slight inhibitory effect on quinoline degradation. Consequently, the removal efficiencies of both DBT and QN by the mixed biofilm failed to exceed 80%. Similarly, Maass et al. [110] also utilized Rhodococcus for simultaneous denitrogenation and desulfurization of heavy gas oil (HGO). Notably, HGO concentration not only did not inhibit the growth of Rhodococcus but also significantly enhanced its growth rate at concentrations ranging from 10% to 50%. The overall trends of desulfurization and denitrogenation were consistent: the maximum removal efficiency was achieved at 40% HGO concentration, with desulfurization and denitrogenation efficiencies reaching 42.7% and 43.2%, respectively. Under the extreme condition of 100% HGO, severe cell aggregation occurred, accompanied by no obvious metabolic activity, and desulfurization/denitrogenation completely ceased. This verified that the deterioration of cell physiological status is the direct cause of the loss of catalytic activity. Subsequently, Maass et al. [111] conducted further investigations and identified the optimal culture conditions for Rhodococcus, achieving a 240% increase in biomass yield. However, experimental results showed that 10% HGO was the optimal concentration, which improved the denitrogenation efficiency by 8.3%. In contrast, the sulfur removal capacity was only 250 mg, significantly lower than the denitrogenation performance.
Simultaneous biological denitrogenation and desulfurization boasts core advantages such as mild reaction conditions and environmental benignity, making it well-suited for low-energy-consumption removal of sulfur and nitrogen compounds. Nevertheless, it suffers from limitations, including sluggish reaction rates and poor strain stability, leading to lower removal efficiency compared to technologies like hydrotreatment or oxidation. Despite these drawbacks, its unparalleled environmental friendliness endows biological methods with tremendous potential for future applications. With the development of various fields in biology, although various technologies have not been fully adapted to fuel desulfurization and denitrogenation, these other fields have demonstrated strong operability. For example, biofilm reactors [112,113] provide stable growth environments and efficient reaction interfaces for microorganisms, which can efficiently enrich microorganisms, enhance substrate mass transfer, and improve reaction stability. Metabolic engineering [114] aims to modify cellular metabolism and improve the degradation rate of sulfur and nitrogen compounds by optimizing microbial metabolic pathways. Synthetic biology [115] enables precise genetic modification of microorganisms to become efficient cells specifically designed for the removal of sulfides and nitrates.
5. Conclusions and Future Perspectives
Desulfurization and denitrogenation of fuel oil have long been a major challenge in industrial development. Over decades of exploration, most studies have centered on desulfurization, with its theoretical research relatively well-established. In contrast, research focusing on simultaneous desulfurization and denitrogenation remains extremely limited, and the mechanistic investigations into the co-removal of different pollutants are still incomplete. This review presents a comprehensive review and overview of hydroprocessing technologies and non-hydroprocessing technologies (adsorption, extraction, oxidation, and biological methods), highlighting the types of materials employed in each technology. It concludes each section with a summary and outlines the current challenges faced by each method or material, primarily including competition for adsorption sites or active sites between sulfur and nitrogen compounds. Furthermore, issues such as the high preparation cost of catalysts and their reusability have seriously hindered the industrialization process of various technologies. In addition, most of the research in the paper is on simulated fuel, which has certain differences in composition from real fuel. The aromatic hydrocarbons present in real fuels not only occupy the adsorption active sites, but also, due to their dense structure, easily form an adsorption layer on the catalyst surface, hindering the mass transfer of nitride and sulfide reactions. Meanwhile, the interaction between aromatic hydrocarbons and oxidation products such as sulfones delays product desorption, leading to blockage of catalyst pores. The most fatal aspect of simultaneous desulfurization and denitrogenation is the formation of stable coordination bonds between the nitrogen base and the active site, resulting in permanent deactivation of the active site.
Therefore, in the next 5 to 10 years, the industrialization process of non-hydrogen desulfurization and denitrogenation technologies for fuel will accelerate under the dual drive of the global ultra-low fuel standard “dual carbon” policy. The core path revolves around technological iteration, industrial adaptation, and ecological construction. Mainstream technologies represented by ODS/ODN, BDS/BDN, and ADS/ADN will break through the current bottlenecks of catalyst deactivation, high extractant cost, and slow degradation rate through the research and development of high-performance catalysts or extractants, and the optimization of reaction separation integrated processes. Among them, ODS and ODN need to focus on improving the balance between catalytic efficiency and oil yield, BDS/BDN relies on new reactors such as biological drip filtration towers to achieve continuous and automated operation, while ADS/ADN needs to enhance the cycling stability and large-scale preparation capability of MOF-based adsorbents. The following future research directions have been specifically proposed:
- (1)
- Accelerate the transition of non-hydroprocessing technologies (e.g., adsorption, extraction, oxidation) from theoretical and experimental research to large-scale practical industrial applications. Reduce the fabrication costs of catalysts, extractants, and related materials, while developing catalysts/extractants with high activity, stability, and reusability.
- (2)
- By making certain improvements, biotechnology that has been successfully applied in other fields can be adapted to the field of fuel desulfurization and denitrogenation. It is possible to cultivate highly efficient organisms in the fields of metabolic engineering and biosynthesis of solution fuels through techniques such as genetic modification.
- (3)
- Integrate multiple technologies—for instance, coupling ultrasound-assisted oxidation with biological methods—to lower energy consumption while improving pollutant removal rates.
- (4)
- Employ advanced in situ characterization techniques (e.g., STEM, FT-IR) to further unravel the specific processes and mechanisms underlying the competition for active sites and adsorption sites between sulfur and nitrogen compounds, laying a theoretical foundation for the design of high-performance catalysts and adsorbents.
- (5)
- Most existing studies focus on simulated fuels, whose composition and component concentrations differ from those of real industrial fuels. Thus, the application of actual industrial fuels in experimental research is strongly advocated. Gradient processing technology is required for the treatment of oil products with high nitrogen and sulfur content, which involves first removing most of the target species through extraction and other techniques, and then using oxidation desulfurization and other methods to meet the standards.
- (6)
- By adopting defect engineering and acid treatment or calcination strategies, controllable defects are constructed in catalytic materials such as MOFs and metal oxides, significantly improving catalytic activity and stability. Organic–inorganic hybrid phase transfer catalysts or molybdenum based composite materials should be designed to achieve synergistic oxidation conversion of sulfides and nitrides and deepen the reaction mechanism through multi metal active site synergistic design to solve the problem of decreased catalytic efficiency caused by competitive adsorption of sulfur and nitrogen compounds.
Author Contributions
Writing—original draft preparation, J.W. and R.W.; writing—review and editing, R.W.; supervision, R.W. All authors have read and agreed to the published version of the manuscript.
Funding
This research was funded by the Natural Science Foundation of Shandong Province, China (ZR2025MS970).
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
Not applicable.
Conflicts of Interest
The authors declare no conflicts of interest.
References
- Haghighi, M.; Gooneh-Farahani, S. Insights to the Oxidative Desulfurization Process of Fossil Fuels over Organic and Inorganic Heterogeneous Catalysts: Advantages and Issues. Environ. Sci. Pollut. Res. 2020, 27, 39923–39945. [Google Scholar] [CrossRef] [PubMed]
- Kumari, S.; Sengupta, S. Non-Hydrogen Processes for Simultaneous Desulfurization and Denitrogenation of Light Petroleum Fuels—An Elaborative Review. Environ. Sci. Pollut. Res. 2021, 28, 61873–61907. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Cheng, M.; Gao, J.; Li, J. Review of the Influencing Factors of Secondary Organic Aerosol Formation and Aging Mechanism Based on Photochemical Smog Chamber Simulation Methods. J. Environ. Sci. 2023, 123, 545–559. [Google Scholar] [CrossRef] [PubMed]
- Ren, X.; Liu, Z.; Dong, L.; Miao, G.; Liao, N.; Li, Z.; Xiao, J. Dynamic Catalytic Adsorptive Desulfurization of Real Diesel over Ultra-stable and Low-cost Silica Gel-supported TiO2. AIChE J. 2018, 64, 2146–2159. [Google Scholar] [CrossRef]
- Qiu, X.; Wang, B.; Wang, R.; Kozhevnikov, I.V. New Adsorption Materials for Deep Desulfurization of Fuel Oil. Materials 2024, 17, 1803. [Google Scholar] [CrossRef]
- Prado, G.H.C.; Rao, Y.; Klerk, A. Nitrogen Removal from Oil: A Review. Energy Fuels 2017, 31, 14–36. [Google Scholar] [CrossRef]
- Zolotareva, D.; Zazybin, A.; Rafikova, K.; Dembitsky, V.M.; Dauletbakov, A.; Yu, V. Ionic Liquids Assisted Desulfurization and Denitrogenation of Fuels. Vietnam J. Chem. 2019, 57, 133–163. [Google Scholar] [CrossRef]
- Zhang, K.; Hu, J.; Gao, S.; Liu, Y.; Huang, X.; Bao, X. Sulfur Content of Gasoline and Diesel Fuels in Northern China. Energy Policy 2010, 38, 2934–2940. [Google Scholar] [CrossRef]
- Liu, Z.; An, K.; Cao, Y. Undefined-Oriented Programming: Detecting and Chaining Prototype Pollution Gadgets in Node.Js Template Engines for Malicious Consequences. In Proceedings of the 2024 IEEE Symposium on Security and Privacy (SP), San Francisco, CA, USA, 19–23 May 2024; pp. 4015–4033. [Google Scholar]
- Sun, Z.; Wang, R. A Critical Review of Catalytic Oxidative Desulfurization (COD) in Petroleum-Based Liquid Fuels. Fuel 2025, 402, 136031. [Google Scholar] [CrossRef]
- Saha, B.; Vedachalam, S.; Dalai, A.K. Review on Recent Advances in Adsorptive Desulfurization. Fuel Process. Technol. 2021, 214, 106685. [Google Scholar] [CrossRef]
- Majid, M.F.; Mohd Zaid, H.F.; Kait, C.F.; Jumbri, K.; Yuan, L.C.; Rajasuriyan, S. Futuristic Advance and Perspective of Deep Eutectic Solvent for Extractive Desulfurization of Fuel Oil: A Review. J. Mol. Liq. 2020, 306, 112870. [Google Scholar] [CrossRef]
- Yuan, B.; Li, X.; Sun, Y. A Short Review of Aerobic Oxidative Desulfurization of Liquid Fuels over Porous Materials. Catalysts 2022, 12, 129. [Google Scholar] [CrossRef]
- Zhou, X.; Wang, T.; Liu, H.; Gao, X.; Wang, C.; Wang, G. Desulfurization through Photocatalytic Oxidation: A Critical Review. ChemSusChem 2021, 14, 492–511. [Google Scholar] [CrossRef] [PubMed]
- Leng, L.; Yang, L.; Chen, J.; Leng, S.; Li, H.; Li, H.; Yuan, X.; Zhou, W.; Huang, H. A Review on Pyrolysis of Protein-Rich Biomass: Nitrogen Transformation. Bioresour. Technol. 2020, 315, 123801. [Google Scholar] [CrossRef]
- Li, F.; Srivatsa, S.C.; Bhattacharya, S. A Review on Catalytic Pyrolysis of Microalgae to High-Quality Bio-Oil with Low Oxygeneous and Nitrogenous Compounds. Renew. Sustain. Energy Rev. 2019, 108, 481–497. [Google Scholar] [CrossRef]
- Roman, F.F.; Tuesta, J.L.D.D.; Silva, A.M.T.; Faria, J.L.; Gomes, H.T. Carbon-Based Materials for Oxidative Desulfurization and Denitrogenation of Fuels: A Review. Catalysts 2021, 11, 1239. [Google Scholar] [CrossRef]
- Tissot, B.P.; Welte, D.H. Geochemical Fossils and Their Significance in Petroleum Formation. In Petroleum Formation and Occurrence; Tissot, B.P., Welte, D.H., Eds.; Springer: Berlin/Heidelberg, Germany, 1984; pp. 93–130. ISBN 978-3-642-87813-8. [Google Scholar]
- Shi, Q.; Wu, J. Review on Sulfur Compounds in Petroleum and Its Products: State-of-the-Art and Perspectives. Energy Fuels 2021, 35, 14445–14461. [Google Scholar] [CrossRef]
- Rajendran, A.; Cui, T.; Fan, H.; Yang, Z.; Feng, J.; Li, W. A Comprehensive Review on Oxidative Desulfurization Catalysts Targeting Clean Energy and Environment. J. Mater. Chem. A 2020, 8, 2246–2285. [Google Scholar] [CrossRef]
- Silva, S.R.C.; Moncioso, N.A.P.; Sad, C.M.S.; Tosta, C.L.; Souza, L.M.; Cipriano, D.F.; Romão, W.; Freitas, J.C.C.; Kuster, R.M.; Castro, E.V.R.; et al. Preparation of a Nitrogen Oil Compound Fraction by Modified Gel Silica Column Chromatography. Energy Fuels 2020, 34, 5652–5664. [Google Scholar] [CrossRef]
- Alli, R.D.; Kroon, M.C. Extraction of Benzothiazole and Thiophene from Their Mixtures with N-Heptane Using Tetrahexylammonium Bromide-Based Deep Eutectic Solvents as Extractive Denitrogenation and Desulfurization Agents. Fluid Phase Equilib. 2018, 477, 1–11. [Google Scholar] [CrossRef]
- Akhtar, M.S.; Ali, S.; Zaman, W. Recent Advancements in Catalysts for Petroleum Refining. Catalysts 2024, 14, 841. [Google Scholar] [CrossRef]
- Fathy, D.; El-Balkiemy, A.F.; Makled, W.A.; Hosny, A.M. Organic Geochemical Signals of Paleozoic Rocks in the Southern Tethys, Siwa Basin, Egypt: Implications for Source Rock Characterization and Petroleum System. Phys. Chem. Earth Parts A/B/C 2023, 130, 103393. [Google Scholar] [CrossRef]
- Li, N.; Ma, X.; Zha, Q.; Song, C. Analysis and Comparison of Nitrogen Compounds in Different Liquid Hydrocarbon Streams Derived from Petroleum and Coal. Energy Fuels 2010, 24, 5539–5547. [Google Scholar] [CrossRef]
- Bello, S.S.; Wang, C.; Zhang, M.; Gao, H.; Han, Z.; Shi, L.; Su, F.; Xu, G. A Review on the Reaction Mechanism of Hydrodesulfurization and Hydrodenitrogenation in Heavy Oil Upgrading. Energy Fuels 2021, 35, 10998–11016. [Google Scholar] [CrossRef]
- Liu, K.; Ng, F.T.T. Effect of the Nitrogen Heterocyclic Compounds on Hydrodesulfurization Using in Situ Hydrogen and a Dispersed Mo Catalyst. Catal. Today 2010, 149, 28–34. [Google Scholar] [CrossRef]
- Ja’fari, M.; Ebrahimi, S.L.; Khosravi-Nikou, M.R. Ultrasound-Assisted Oxidative Desulfurization and Denitrogenation of Liquid Hydrocarbon Fuels: A Critical Review. Ultrason. Sonochem. 2018, 40, 955–968. [Google Scholar] [CrossRef]
- Páez, D.E.; Andriollo, A.; Morfes, G. Aqueous-Phase Catalytic Hydrogenation and Hydrogenolysis Reactions of Sulfur- and Nitrogen-Containing Compounds in Oil Fractions. Catal. Today 2015, 247, 139–146. [Google Scholar] [CrossRef]
- Sharifvaghefi, S.; Zheng, Y. Development of a Magnetically Recyclable Molybdenum Disulfide Catalyst for Direct Hydrodesulfurization. ChemCatChem 2015, 7, 3397–3403. [Google Scholar] [CrossRef]
- Zheng, M.; Zhao, L.; Cao, L.; Zhang, C.; Gao, J.; Xu, C. Catalysis Performance of Nonpromoted and Co-Promoted MoS2 Catalysts on a Hydrodesulfurization Reaction: A DFT Study. Mol. Catal. 2019, 467, 38–51. [Google Scholar] [CrossRef]
- Jian, M.; Prins, R. Kinetics of the Hydrodenitrogenation of Decahydroquinoline over NiMo(P)/Al2O3 Catalysts. Ind. Eng. Chem. Res. 1998, 37, 834–840. [Google Scholar] [CrossRef]
- Li, Y.; Guo, W.; Zhu, H.; Zhao, L.; Li, M.; Li, S.; Fu, D.; Lu, X.; Shan, H. Initial Hydrogenations of Pyridine on MoP(001): A Density Functional Study. Langmuir 2012, 28, 3129–3137. [Google Scholar] [CrossRef]
- Qian, E.W.; Abe, S.; Kagawa, Y.; Ikeda, H. Hydrodenitrogenation of Porphyrin on Ni-Mo Based Catalysts. Chin. J. Catal. 2013, 34, 152–158. [Google Scholar] [CrossRef]
- Ding, C.; Sun, Z.; Zhang, L. Efficient Removal of Basic Heterocyclic Nitrogen Compounds from Fuel Oils by Functionalized Acidic Ionic Liquids: Extraction Experiment and Interaction Exploration. J. Mol. Liq. 2023, 386, 122455. [Google Scholar] [CrossRef]
- Liu, J.; Li, W.-Y.; Feng, J.; Gao, X. Molecular Insights into the Hydrodenitrogenation Mechanism of Pyridine over Pt/γ-Al2O3 Catalysts. Mol. Catal. 2020, 495, 111148. [Google Scholar] [CrossRef]
- Laredo, G.C.; Vega-Merino, P.M.; Trejo-Zárraga, F.; Castillo, J. Denitrogenation of Middle Distillates Using Adsorbent Materials towards ULSD Production: A Review. Fuel Process. Technol. 2013, 106, 21–32. [Google Scholar] [CrossRef]
- Hu, S.; Luo, G.; Shima, T.; Luo, Y.; Hou, Z. Hydrodenitrogenation of Pyridines and Quinolines at a Multinuclear Titanium Hydride Framework. Nat. Commun. 2017, 8, 1866. [Google Scholar] [CrossRef]
- Sheng, Q.; Wang, G.; Liu, Y.; Husein, M.M.; Gao, C.; Gao, J. Pilot-Scale Evaluation of Hydrotreating Inferior Coker Gas Oil Prior to Its Fluid Catalytic Cracking. Fuel 2018, 226, 27–34. [Google Scholar] [CrossRef]
- Raghuveer, C.S.; Thybaut, J.W.; Bruycker, R.; Metaxas, K.; Bera, T.; Marin, G.B. Pyridine Hydrodenitrogenation over Industrial NiMo/γ-Al2O3 Catalyst: Application of Gas Phase Kinetic Models to Liquid Phase Reactions. Fuel 2014, 125, 206–218. [Google Scholar] [CrossRef]
- Tian, S.; Li, X.; Wang, A.; Chen, Y.; Li, H.; Hu, Y. Hydrodenitrogenation of Quinoline and Decahydroquinoline over a Surface Nickel Phosphosulfide Phase. Catal. Lett. 2018, 148, 1579–1588. [Google Scholar] [CrossRef]
- Bassm, R.; Villarroel, M.; Gil-Llambias, F.J.; Baeza, P.; García-Fierro, J.L.; Martínez, N.; Olivera, P.; Leiva, K.; Escalona, N. Support effect on conversion of quinoline over ReS2 catalyst. J. Chil. Chem. Soc. 2016, 61, 3170–3176. [Google Scholar] [CrossRef]
- Guo, C.; Zhang, T.; Niu, M.; Cao, S.; Wei, S.; Wang, Z.; Guo, W.; Lu, X.; Wu, C.-M.L. Impact of Diverse Active Sites on MoS2 Catalyst: Competition on Active Site Formation and Selectivity of Thiophene Hydrodesulfurization Reaction. Mol. Catal. 2019, 463, 67–76. [Google Scholar] [CrossRef]
- He, S.-S.; Huang, T.-T.; Chen, C.; Fan, Y. Tuning Active Sites in MoS2-Based Catalysts via H2O2 Etching to Enhance Hydrodesulfurization Performance. Pet. Sci. 2023, 20, 3875–3886. [Google Scholar] [CrossRef]
- Mello, M.D.; Braggio, F.A.; Magalhães, B.C.; Zotin, J.L.; Silva, M.A.P. Effects of Phosphorus Content on Simultaneous Ultradeep HDS and HDN Reactions over NiMoP/Alumina Catalysts. Ind. Eng. Chem. Res. 2017, 56, 10287–10299. [Google Scholar] [CrossRef]
- Albersberger, S.; Hein, J.; Schreiber, M.W.; Guerra, S.; Han, J.; Gutiérrez, O.Y.; Lercher, J.A. Simultaneous Hydrodenitrogenation and Hydrodesulfurization on Unsupported Ni-Mo-W Sulfides. Catal. Today 2017, 297, 344–355. [Google Scholar] [CrossRef]
- Farag, H.; Kishida, M.; Al-Megren, H. Competitive Hydrodesulfurization of Dibenzothiophene and Hydrodenitrogenation of Quinoline over Unsupported MoS2 Catalyst. Appl. Catal. A 2014, 469, 173–182. [Google Scholar] [CrossRef]
- Gutiérrez, O.Y.; Singh, S.; Schachtl, E.; Kim, J.; Kondratieva, E.; Hein, J.; Lercher, J.A. Effects of the Support on the Performance and Promotion of (Ni)MoS2 Catalysts for Simultaneous Hydrodenitrogenation and Hydrodesulfurization. ACS Catal. 2014, 4, 1487–1499. [Google Scholar] [CrossRef]
- Nascimento, I.G.; Machado, M.D.S.C.; Mello, M.D.; Segtovich, I.S.V.; Zotin, J.L.; Silva, M.A.P. Enhancing Simultaneous Hydrodesulfurization and Hydrodenitrogenation Reactions: Kinetic Modeling of Stacked NiMoP and CoMoP Catalysts Beds. Catal. Today 2025, 443, 114954. [Google Scholar] [CrossRef]
- Abro, R.; Abro, M.; Gao, S.; Bhutto, A.W.; Ali, Z.M.; Shah, A.; Chen, X.; Yu, G. Extractive Denitrogenation of Fuel Oils Using Ionic Liquids: A Review. RSC Adv. 2016, 6, 93932–93946. [Google Scholar] [CrossRef]
- Salah, H.B.; Nancarrow, P.; Al-Othman, A. Ionic Liquid-Assisted Refinery Processes—A Review and Industrial Perspective. Fuel 2021, 302, 121195. [Google Scholar] [CrossRef]
- Welton, T. Ionic Liquids: A Brief History. Biophys. Rev. 2018, 10, 691–706. [Google Scholar] [CrossRef]
- Nusaibah Masri, A.; Mutalib Mi, A.; Leveque, J.M. A Review on Dicationic Ionic Liquids: Classification and Application. Ind. Eng. Manag. 2016, 5, 197. [Google Scholar] [CrossRef]
- Paucar, N.E.; Kiggins, P.; Blad, B.; Jesus, K.; Afrin, F.; Pashikanti, S.; Sharma, K. Ionic Liquids for the Removal of Sulfur and Nitrogen Compounds in Fuels: A Review. Environ. Chem. Lett. 2021, 19, 1205–1228. [Google Scholar] [CrossRef]
- Hanamertani, A.S.; Pilus, R.M.; Irawan, S. A Review on the Application of Ionic Liquids for Enhanced Oil Recovery. In Icipeg 2016; Awang, M., Negash, B.M., Akhir, N.A., Lubis, L.A., Rafek, A.G., Eds.; Springer: Singapore, 2017; pp. 133–147. ISBN 978-981-10-3649-1. [Google Scholar]
- Hu, Y.; Xing, Y.; Yue, H.; Chen, T.; Diao, Y.; Wei, W.; Zhang, S. Ionic Liquids Revolutionizing Biomedicine: Recent Advances and Emerging Opportunities. Chem. Soc. Rev. 2023, 52, 7262–7293. [Google Scholar] [CrossRef] [PubMed]
- Nie, Y.; Gong, X.; Gao, H.; Zhang, X.; Zhang, S. Simultaneous Desulfurization and Denitrogen of Liquid Fuels Using Two Functionalized Group Ionic Liquids. Sci. China Chem. 2014, 57, 1766–1773. [Google Scholar] [CrossRef]
- Hansmeier, A.R.; Meindersma, G.W.; Haan, A.B. Desulfurization and Denitrogenation of Gasoline and Diesel Fuels by Means of Ionic Liquids. Green Chem. 2011, 13, 1907. [Google Scholar] [CrossRef]
- Chen, X.; Yuan, S.; Abdeltawab, A.A.; Al-Deyab, S.S.; Zhang, J.; Yu, L.; Yu, G. Extractive Desulfurization and Denitrogenation of Fuels Using Functional Acidic Ionic Liquids. Sep. Purif. Technol. 2014, 133, 187–193. [Google Scholar] [CrossRef]
- Veríssimo, N.V.P.; Mussagy, C.U.; Bento, H.B.S.; Pereira, J.F.B.; Santos-Ebinuma, V.D.C. Ionic Liquids and Deep Eutectic Solvents for the Stabilization of Biopharmaceuticals: A Review. Biotechnol. Adv. 2024, 71, 108316. [Google Scholar] [CrossRef]
- Zhang, Q.; Vigier, K.O.; Royer, S.; Jérôme, F. Deep Eutectic Solvents: Syntheses, Properties and Applications. Chem. Soc. Rev. 2012, 41, 7108. [Google Scholar] [CrossRef]
- Warrag, S.E.E.; Peters, C.J.; Kroon, M.C. Deep Eutectic Solvents for Highly Efficient Separations in Oil and Gas Industries. Curr. Opin. Green Sustain. Chem. 2017, 5, 55–60. [Google Scholar] [CrossRef]
- Li, Z.; Cui, Y.; Li, C.; Shen, Y. Deep Desulfurization of Fuels Based on Deep Eutectic Theory. Sep. Purif. Technol. 2019, 219, 9–15. [Google Scholar] [CrossRef]
- Wu, X.; Ning, Q.; Tu, Y.; Du, C.; Ren, Z. Extractive Desulfurization of Fuel Oil with Dual Imidazolium-Based Deep Eutectic Solvents. Chem. Eng. Sci. 2025, 317, 122092. [Google Scholar] [CrossRef]
- Khan, R.; Xu, D.; Sadrmousavi-Dizaj, A.; Gao, J.; Sun, L.; Wang, Y. Rational Design and Extractive Performance of Novel Ternary Deep Eutectic Solvents for Oil Denitrogenation. Chem. Eng. Sci. 2026, 321, 122864. [Google Scholar] [CrossRef]
- Zhu, S.; Cheng, H.; Dai, Y.; Gao, J.; Jiang, X. Extractive Desulfurization and Denitrogenation from Fuel Oil by a Polyether-Amine-Based Solvent. Energy Fuels 2020, 34, 8186–8194. [Google Scholar] [CrossRef]
- Lima, F.; Dave, M.; Silvestre, A.J.D.; Branco, L.C.; Marrucho, I.M. Concurrent Desulfurization and Denitrogenation of Fuels Using Deep Eutectic Solvents. ACS Sustain. Chem. Eng. 2019, 7, 11341–11349. [Google Scholar] [CrossRef]
- Rogošić, M.; Sander, A.; Pantaler, M. Application of 1-Pentyl-3-Methylimidazolium Bis(Trifluoromethylsulfonyl) Imide for Desulfurization, Denitrification and Dearomatization of FCC Gasoline. J. Chem. Thermodyn. 2014, 76, 1–15. [Google Scholar] [CrossRef]
- Mk, S.B.; Mehra, S.; Kumar, A.; Kancharla, S. Diesel Purification through Imidazole-Based Deep Eutectic Solvents: Desulfurization, Dearomatization, and Denitrogenation. Fuel 2025, 387, 134317. [Google Scholar] [CrossRef]
- Zarin, L.; Saien, J.; Dastan, D.; Jafari, F. Desulfurization and Denitrogenation of a Synthesized Gasoline Utilizing an Acidic Deep Eutectic Solvent: Experimental and Thermodynamic Studies. J. Mol. Liq. 2025, 425, 127197. [Google Scholar] [CrossRef]
- Silva, D.F.; Viana, A.M.; Santos-Vieira, I.; Balula, S.S.; Cunha-Silva, L. Ionic Liquid-Based Polyoxometalate Incorporated at ZIF-8: A Sustainable Catalyst to Combine Desulfurization and Denitrogenation Processes. Molecules 2022, 27, 1711. [Google Scholar] [CrossRef]
- Qiu, X.; Ren, R.; Wang, B.; Wang, R.; Kozhevnikov, I. Advancements and Insights into Single-Atom Catalysts for Environmental and Energy Applications. Renew. Sustain. Energy Rev. 2025, 218, 115842. [Google Scholar] [CrossRef]
- Kwon, J.-M.; Moon, J.-H.; Bae, Y.-S.; Lee, D.-G.; Sohn, H.-C.; Lee, C.-H. Adsorptive Desulfurization and Denitrogenation of Refinery Fuels Using Mesoporous Silica Adsorbents. ChemSusChem 2008, 1, 307–309. [Google Scholar] [CrossRef]
- Li, N.; Almarri, M.; Ma, X.; Zha, Q. The Role of Surface Oxygen-Containing Functional Groups in Liquid-Phase Adsorptive Denitrogenation by Activated Carbon. New Carbon Mater. 2011, 26, 470–478. [Google Scholar] [CrossRef]
- Arcibar-Orozco, J.A.; Acosta-Herrera, A.A.; Rangel-Mendez, J.R. Simultaneous Desulfuration and Denitrogenation of Model Diesel Fuel by Fe-Mn Microwave Modified Activated Carbon: Iron Crystalline Habit Influence on Adsorption Capacity. J. Clean. Prod. 2019, 218, 69–82. [Google Scholar] [CrossRef]
- Arcibar-Orozco, J.A.; Rangel-Mendez, J.R. Model Diesel Denitrogenation by Modified Activated Carbon with Iron Nanoparticles: Sulfur Compounds Effect. Chem. Eng. J. 2013, 230, 439–446. [Google Scholar] [CrossRef]
- Thaligari, S.K.; Gupta, S.; Srivastava, V.C.; Prasad, B. Simultaneous Desulfurization and Denitrogenation of Liquid Fuel by Nickel-Modified Granular Activated Carbon. Energy Fuels 2016, 30, 6161–6168. [Google Scholar] [CrossRef]
- Liu, J.; Chen, L.; Cui, H.; Zhang, J.; Zhang, L.; Su, C.-Y. Applications of Metal–Organic Frameworks in Heterogeneous Supramolecular Catalysis. Chem. Soc. Rev. 2014, 43, 6011–6061. [Google Scholar] [CrossRef]
- Qiu, S.; Xue, M.; Zhu, G. Metal–Organic Framework Membranes: From Synthesis to Separation Application. Chem. Soc. Rev. 2014, 43, 6116–6140. [Google Scholar] [CrossRef]
- Bhadra, B.N.; Jhung, S.H. Oxidative Desulfurization and Denitrogenation of Fuels Using Metal-Organic Framework-Based/-Derived Catalysts. Appl. Catal. B 2019, 259, 118021. [Google Scholar] [CrossRef]
- Ahmed, I.; Jhung, S.H. Adsorptive Desulfurization and Denitrogenation Using Metal-Organic Frameworks. J. Hazard. Mater. 2016, 301, 259–276. [Google Scholar] [CrossRef]
- Lee, G.; Jhung, S.H. Effective and Selective Removal of Carbazole from Model Bitumen-Derived Fuel via Adsorption Using Amine-Grafted Metal–Organic Frameworks. J. Mol. Liq. 2023, 390, 123169. [Google Scholar] [CrossRef]
- Tan, P.; Xie, X.-Y.; Liu, X.-Q.; Pan, T.; Gu, C.; Chen, P.-F.; Zhou, J.-Y.; Pan, Y.; Sun, L.-B. Fabrication of magnetically responsive HKUST-1/Fe3O4 composites by dry gel conversion for deep desulfurization and denitrogenation. J. Hazard. Mater. 2017, 321, 344–352. [Google Scholar] [CrossRef]
- Khan, N.A.; Jhung, S.H. Scandium-Triflate/Metal–Organic Frameworks: Remarkable Adsorbents for Desulfurization and Denitrogenation. Inorg. Chem. 2015, 54, 11498–11504. [Google Scholar] [CrossRef] [PubMed]
- Akopyan, A.V.; Grigoriev, D.A.; Polikarpova, P.L.; Eseva, E.A.; Litvinova, V.V.; Anisimov, A.V. Ozone-Assisted Oxidative Desulfurization of Light Oil Fractions. Pet. Chem. 2017, 57, 904–907. [Google Scholar] [CrossRef]
- Banisharif, F.; Dehghani, M.R.; Capel-Sanchez, M.C.; Campos-Martin, J.M. Extractive-Oxidative Removals of Dibenzothiophene and Quinoline Using Vanadium Substituted Dawson Emulsion Catalyst and Ionic Liquid Based Solvents. J. Ind. Eng. Chem. 2017, 47, 348–359. [Google Scholar] [CrossRef]
- Yao, Z.; Miras, H.N.; Song, Y.-F. Efficient Concurrent Removal of Sulfur and Nitrogen Contents from Complex Oil Mixtures by Using Polyoxometalate-Based Composite Materials. Inorg. Chem. Front. 2016, 3, 1007–1013. [Google Scholar] [CrossRef]
- Bertleff, B.; Haider, M.S.; Claußnitzer, J.; Korth, W.; Wasserscheid, P.; Jess, A.; Albert, J. Extractive Catalytic Oxidative Denitrogenation of Fuels and Their Promoting Effect for Desulfurization Catalyzed by Vanadium Substituted Heteropolyacids and Molecular Oxygen. Energy Fuels 2020, 34, 8099–8109. [Google Scholar] [CrossRef]
- Palomeque-Santiago, J.F.; López-Medina, R.; Oviedo-Roa, R.; Navarrete-Bolaños, J.; Mora-Vallejo, R.; Fuente, J.A.M.; Martínez-Magadán, J.M. Deep Oxidative Desulfurization with Simultaneous Oxidative Denitrogenation of Diesel Fuel and Straight Run Gas Oil. Appl. Catal. B 2018, 236, 326–337. [Google Scholar] [CrossRef]
- Safa, M.A.; Ma, X.; Bouresli, R.; Albazzaz, H. Effects of Co-Existing Nitrogen Compounds and Polycyclic Aromatic Hydrocarbons on Catalytic Oxidative Desulfurization of Refractory Sulfur Compounds in Middle Distillates. Catal. Today 2021, 371, 258–264. [Google Scholar] [CrossRef]
- Subhan, S.; Muhammad, Y.; Sahibzada, M.; Subhan, F.; Tong, Z. Studies on the Selection of a Catalyst–Oxidant System for the Energy-Efficient Desulfurization and Denitrogenation of Fuel Oil at Mild Operating Conditions. Energy Fuels 2019, 33, 8423–8439. [Google Scholar] [CrossRef]
- Aghbolagh, Z.S.; Maleki, A. Photocatalytic Oxidative Demercaptanization/Denitrogenation of Gasoline Catalyzed by MAX-phase@rGO@PW11Zn as a High-Performance Nanocatalyst. Mater. Today Commun. 2023, 37, 107252. [Google Scholar] [CrossRef]
- Li, B.; Song, H.; Han, F.; Wei, L. Photocatalytic Oxidative Desulfurization and Denitrogenation for Fuels in Ambient Air over Ti3C2/g-C3N4 Composites under Visible Light Irradiation. Appl. Catal. B 2020, 269, 118845. [Google Scholar] [CrossRef]
- Caero, L.C.; Hernández, E.; Pedraza, F.; Murrieta, F. Oxidative Desulfurization of Synthetic Diesel Using Supported Catalysts. Catal. Today 2005, 107–108, 564–569. [Google Scholar] [CrossRef]
- Moradi, S.; Vossoughi, M.; Feilizadeh, M.; Zakeri, S.M.E.; Mohammadi, M.M.; Rashtchian, D.; Booshehri, A.Y. Photocatalytic Degradation of Dibenzothiophene Using La/PEG-Modified TiO2 under Visible Light Irradiation. Res. Chem. Intermed. 2015, 41, 4151–4167. [Google Scholar] [CrossRef]
- Sundararaman, R.; Ma, X.; Song, C. Oxidative Desulfurization of Jet and Diesel Fuels Using Hydroperoxide Generated in Situ by Catalytic Air Oxidation. Ind. Eng. Chem. Res. 2010, 49, 5561–5568. [Google Scholar] [CrossRef]
- Zhang, Y.; Wang, B.; Wang, R. Functionally Decorated Metal–Organic Frameworks in Environmental Remediation. Chem. Eng. J. 2023, 455, 140741. [Google Scholar] [CrossRef]
- Subhan, S.; Yaseen, M.; Ahmad, B.; Tong, Z.; Subhan, F.; Ahmad, W.; Sahibzada, M. Fabrication of MnO2 NPs Incorporated UiO-66 for the Green and Efficient Oxidative Desulfurization and Denitrogenation of Fuel Oils. J. Environ. Chem. Eng. 2021, 9, 105179. [Google Scholar] [CrossRef]
- Li, J.; Luo, H.; Li, B.; Ma, J.-G.; Cheng, P. Application of MOF-Derived Materials as Electrocatalysts for CO2 Conversion. Mater. Chem. Front. 2023, 7, 6107–6129. [Google Scholar] [CrossRef]
- Jia, Y.; Li, G.; Ning, G.; Jin, C. The Effect of N-Containing Compounds on Oxidative Desulphurization of Liquid Fuel. Catal. Today 2009, 140, 192–196. [Google Scholar] [CrossRef]
- Ma, C.; Dai, B.; Liu, P.; Zhou, N.; Shi, A.; Ban, L.; Chen, H. Deep Oxidative Desulfurization of Model Fuel Using Ozone Generated by Dielectric Barrier Discharge Plasma Combined with Ionic Liquid Extraction. J. Ind. Eng. Chem. 2014, 20, 2769–2774. [Google Scholar] [CrossRef]
- Adhami, M.; Movahedirad, S.; Sobati, M.A. Novel Method for Desulfurization of Mixed Fuel via Microbubble Oxidation Followed by Microtube Extraction. Energy Fuels 2024, 38, 2153–2166. [Google Scholar] [CrossRef]
- Kumari, S.; Sengupta, S. Multi-Objective Optimization of Simultaneous Oxidative Desulfurization and Denitrogenation of Model Fuel. J. Ind. Eng. Chem. 2024, 134, 457–473. [Google Scholar] [CrossRef]
- Wang, L.; Xie, D.; Ma, Y.; Sun, M.; Mominou, N.; Jiang, W.; Shufeng, C.; Jing, C. Simultaneous Desulfurization and Denitrogenation of Diesel over Er/W-N-TiO2 Photocatalyst. Fuel Process. Technol. 2021, 216, 106802. [Google Scholar] [CrossRef]
- Maslova, O.; Senko, O.; Stepanov, N.; Gladchenko, M.; Gaydamaka, S.; Akopyan, A.; Polikarpova, P.; Lysenko, S.; Anisimov, A.; Efremenko, E. Formation and Use of Anaerobic Consortia for the Biotransformation of Sulfur-Containing Extracts from Pre-Oxidized Crude Oil and Oil Fractions. Bioresour. Technol. 2021, 319, 124248. [Google Scholar] [CrossRef] [PubMed]
- Fan, J.; Khan, H.A.; Wang, T.; Ruiz-Martinez, J.; Saxena, S.; Emwas, A.-H.; Samaras, V.G.; Roberts, W.L. Oxidative Desulfurization of Fuel Oil and Molecular Characterization of the Sulfone Compound Distribution in the Different Extractants. Sep. Purif. Technol. 2023, 327, 124902. [Google Scholar] [CrossRef]
- Li, M.; Zhou, Z.; Zhang, F.; Chai, W.; Zhang, L.; Ren, Z. Deep Oxidative–Extractive Desulfurization of Fuels Using Benzyl-Based Ionic Liquid. AIChE J. 2016, 62, 4023–4034. [Google Scholar] [CrossRef]
- Pan, Y.; Huang, W.; Liu, Z.H.; Yang, Z.Z.; Tan, J.J.; Xu, B.M. Recent Progress of Metal-Organic Frameworks for Liquid Fuels Desulfurization. Sep. Purif. Rev. 2025, 54, 128–148. [Google Scholar] [CrossRef]
- Dinamarca, M.A.; Eyzaguirre, J.; Baeza, P.; Aballay, P.; Canales, C.; Ojeda, J. A New Functional Biofilm Biocatalyst for the Simultaneous Removal of Dibenzothiophene and Quinoline Using Rhodococcus rhodochrous and Curli Amyloid Overproducer Mutants Derived from Cobetia sp. Strain MM1IDA2H-1. Biotechnol. Rep. 2018, 20, e00286. [Google Scholar] [CrossRef]
- Maass, D.; Todescato, D.; Moritz, D.E.; Oliveira, J.V.; Oliveira, D.; Souza, A.A.U.D.; Souza, S.M.A.G. Desulfurization and Denitrogenation of Heavy Gas Oil by Rhodococcus erythropolis ATCC 4277. Bioprocess Biosyst. Eng. 2015, 38, 1447–1453. [Google Scholar] [CrossRef]
- Todescato, D.; Maass, D.; Mayer, D.A.; Oliveira, J.V.; Oliveira, D.; Souza, S.M.A.G.U.D.; Souza, A.A.U.D. Optimal Production of a Rhodococcus erythropolis ATCC 4277 Biocatalyst for Biodesulfurization and Biodenitrogenation Applications. Appl. Biochem. Biotechnol. 2017, 183, 1375–1389. [Google Scholar] [CrossRef]
- Xia, G.; Sun, Z.; Huang, J.; Qi, J.; Yao, J. Biodegradation of Carbon Disulfide and Hydrogen Sulfide Using a Moving Bed Biofilm Reactor Coupled with Sulfur Recycling: Performance, Mechanism, and Potential Application. J. Environ. Manag. 2024, 367, 121943. [Google Scholar] [CrossRef]
- Cui, Y.-X.; Biswal, B.K.; Loosdrecht, M.C.M.; Chen, G.-H.; Wu, D. Long Term Performance and Dynamics of Microbial Biofilm Communities Performing Sulfur-Oxidizing Autotrophic Denitrification in a Moving-Bed Biofilm Reactor. Water Res. 2019, 166, 115038. [Google Scholar] [CrossRef]
- Han, T.; Nazarbekov, A.; Zou, X.; Lee, S.Y. Recent Advances in Systems Metabolic Engineering. Curr. Opin. Biotechnol. 2023, 84, 103004. [Google Scholar] [CrossRef]
- Ma, R.; Zhao, S.; Jin, Y.; Li, Y.; Tang, H.; Hu, M.; Hu, X.; Xu, Y.; Ding, W. Advancing Biomaterial Innovation for Tissue Engineering through Microbial Synthetic Biology: A Review. Int. J. Biol. Macromol. 2025, 322, 146771. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.