PlantForm-Grown Shoots of Rhaponticum carthamoides (Willd.) Iljin as a Source of Caffeoylquinic Acid Derivatives and Antioxidant Potential of Shoot Extract
Abstract
1. Introduction
2. Results
2.1. Caffeoylquinic Acid Derivative and Flavonoid Production
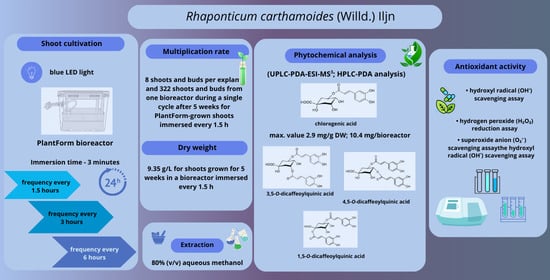
2.2. Shoot Micropropagation
2.3. Biomass Enhancement
2.4. Productivity of Caffeoylquinic Acid Derivatives
2.5. Antioxidant Activity
3. Discussion
4. Materials and Methods
4.1. Plant Materials
4.2. R. carthamoides Shoot Cultivation
4.3. Phytochemical Analysis
4.4. Productivity of CQAs and Flavonoids
4.5. Antioxidant Activity
4.6. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| TIS | Temporary immersion system |
| CQA | Caffeoylquinic acid derivatives |
References
- Kokoska, L.; Janovska, D. Chemistry and pharmacology of Rhaponticum carthamoides: A review. Phytochemistry 2009, 70, 842–855. [Google Scholar] [CrossRef]
- The World Flora Online Database. Available online: https://www.worldfloraonline.org/taxon/wfo-0000049011 (accessed on 19 October 2025).
- Todorova, V.; Ivanov, K.; Ivanova, S. Comparison between the biological active compounds in plants with adaptogenic properties (Rhaponticum carthamoides, Lepidium meyenii, Eleutherococcus senticosus and Panax ginseng). Plants 2022, 11, 64. [Google Scholar] [CrossRef]
- Shikov, A.N.; Narkevich, I.A.; Flisyuk, E.V.; Luzhanin, V.G.; Pozharitskaya, O.N. Medicinal plants from the 14th edition of the Russian Pharmacopoeia, recent updates. J. Ethnopharmacol. 2021, 268, 113685. [Google Scholar] [CrossRef]
- Todorova, V.; Ivanova, S.; Georgieva, Y.; Nalbantova, V.; Karcheva-Bahchevanska, D.; Benbassat, N.; Savova, M.S.; Georgiev, M.I.; Ivanov, K. Chemical composition and histochemical localization of essential oil from wild and cultivated Rhaponticum carthamoides roots and rhizomes. Plants 2022, 11, 2061. [Google Scholar] [CrossRef]
- Knížková, K.; Cihlářová, P.; Kuchař, M. Determination of selected biologically active substances in dietary supplements and the adaptogenic herb Rhaponticum carthamoides using HPLC–MS/MS. Food Anal. Methods 2025, 18, 1514–1531. [Google Scholar] [CrossRef]
- Skała, E.; Makowczyńska, J.; Wieczfinska, J.; Kowalczyk, T.; Sitarek, P. Caffeoylquinic acids with potential biological activity from plant in vitro cultures as alternative sources of valuable natural products. Curr. Pharm. Des. 2020, 26, 2817–2842. [Google Scholar] [CrossRef]
- Alcázar Magaña, A.; Kamimura, N.; Soumyanath, A.; Stevens, J.F.; Maier, C.S. Caffeoylquinic acids: Chemistry, biosynthesis, occurrence, analytical challenges, and bioactivity. Plant J. 2021, 107, 1299–1319. [Google Scholar] [CrossRef]
- Nekratova, N.A.; Kurovskiy, A.V.; Shurupova, M.N. Impact of elevation and slope exposure on abundance of rare medicinal plant Rhaponticum carthamoides (Maral root). Ukr. J. Ecol. 2020, 10, 210–217. [Google Scholar] [CrossRef] [PubMed]
- Raiser, O.; Tagimanova, D.; Turzhanova, A.; Magzumova, S.; Nagmetova, G.; Akhmetkarimova, Z.; Premina, N.; Filippova, N.; Khapilina, O. Propagation and long-term storage of Rhaponticum carthamoides under in vitro conditions. Horticulturae 2025, 11, 952. [Google Scholar] [CrossRef]
- Maysak, G.P.; Matolinets, D.A. Seed productivity of Maral root in the Perm Territory. Kormoproizvodstvo 2021, 2, 32–35. [Google Scholar]
- Timofeev, N.P. Experience of kni (Willd.) Iliin cultivation as a natural source of ecdysterone under the conditions of the Arkhangelsk region. Agric. Biol. 2023, 58, 114–141. [Google Scholar]
- Aka Kaçar, Y.; Biçen, B.; Şimşek, Ö.; Dönmez, D.; Erol, M.H. Evaluation and comparison of a new type of temporary immersion system (TIS) bioreactors for myrtle (Myrtus communis L.). Appl. Ecol. Environ. Res. 2020, 18, 1611–1620. [Google Scholar] [CrossRef]
- Benelli, C.; Faraloni, C.; İzgu, T.; Şimşek, Ö.; Tarraf, W. Optimizing micropropagation of Tanacetum balsamita L.: A machine learning approach to compare semisolid media and temporary immersion system. Horticulturae 2025, 11, 1173. [Google Scholar] [CrossRef]
- Bello-Bello, J.J.; Mancilla-Álvarez, E.; Spinoso-Castillo, J.L. Scaling-up procedures and factors for mass micropropagation using temporary immersion systems. Vitr. Cell. Dev.Biol.-Plant 2025, 61, 321–332. [Google Scholar] [CrossRef]
- Méndez-Hernández, H.A.; Loyola-Vargas, V.M. Plant micropropagation and temporary immersion systems. Methods Mol. Biol. 2024, 2827, 35–50. [Google Scholar] [CrossRef] [PubMed]
- Ramírez-Mosqueda, M.A.; Cruz-Cruz, C.A. Micropropagation Methods in Temporary Immersion Systems; Methods in Molecular Biology; Humana: New York, NY, USA, 2024; Volume 2759. [Google Scholar] [CrossRef]
- Gianguzzi, V.; Leto, L.; Agosti, A.; Di Fazio, A.; Marra, F.P.; Cirlini, M.; Chiancone, B. Influence of sucrose and immersion time on Humulus lupulus L., cv Columbus, plantlet in vitro proliferation and potentially bioactive compound content. Plants 2025, 14, 537. [Google Scholar] [CrossRef]
- Skała, E.; Grąbkowska, R.; Sitarek, P.; Kuźma, Ł.; Błauż, A.; Wysokińska, H. Rhaponticum carthamoides regeneration through direct and indirect organogenesis, molecular profiles and secondary metabolite production. Plant Cell Tissue Organ Cult. 2015, 123, 83–98. [Google Scholar] [CrossRef]
- Zand, A.; Babaei, A.; Omidbaigi, R.; Daneshfar, E. Study on callus induction and plant regeneration of Leuzea carthamoides via tissue culture system. J. Med. Plants Res. 2014, 8, 260–268. [Google Scholar] [CrossRef][Green Version]
- Duškova, J.; Dušek, J. Leuzea carthamoides DC in vitro. Herba Pol. 1995, 41, 165–169. [Google Scholar]
- Skała, E.; Olszewska, M.A.; Tabaka, P.; Kicel, A. Light-emitting diodes and liquid system affect the caffeoylquinic acid derivative and flavonoid production and shoot growth of Rhaponticum carthamoides (Willd.) Iljin. Molecules 2024, 29, 2145. [Google Scholar] [CrossRef]
- Skała, E.; Kicel, A.; Olszewska, M.A.; Kiss, A.K.; Wysokińska, H. Establishment of hairy root cultures of Rhaponticum carthamoides (Willd.) Iljin for the production of biomass and caffeic acid derivatives. BioMed Res. Int. 2015, 2015, 181098. [Google Scholar] [CrossRef]
- Chandimali, N.; Bak, S.G.; Park, E.H.; Lim, H.-J.; Won, Y.-S.; Kim, E.-K.; Park, S.-I.; Lee, S.J. Free radicals and their impact on health and antioxidant defenses: A review. Cell Death Discov. 2025, 11, 19. [Google Scholar] [CrossRef] [PubMed]
- De Carlo, A.; Tarraf, W.; Lambardi, M.; Benelli, C. Temporary immersion system for production of biomass and bioactive compounds from medicinal plants. Agronomy 2021, 11, 2414. [Google Scholar] [CrossRef]
- Hwang, H.-D.; Kwon, S.-H.; Murthy, H.N.; Yun, S.-W.; Pyo, S.-S.; Park, S.-Y. Temporary immersion bioreactor system as an efficient method for mass production of in vitro plants in horticulture and medicinal plants. Agronomy 2022, 12, 346. [Google Scholar] [CrossRef]
- Krasteva, G. Cultivation of Gardenia jasminoides shoots in temporary immersion systems and optimization of the immersion periods. BIO Web Conf. 2023, 58, 02008. [Google Scholar] [CrossRef]
- Available online: https://www.plantform.se (accessed on 20 October 2025).
- Kokotkiewicz, A.; Godlewska, S.; Sparzak-Stefanowska, B.; Panow, O.; Krol, A.; Szopa, A.; Krauze-Baranowska, M.; Łuczkiewicz, M. Accumulation of phenolic compounds in microshoot cultures of Rhododendron tomentosum Harmaja (Ledum palustre L.). Int. J. Mol. Sci. 2025, 26, 7999. [Google Scholar] [CrossRef]
- Klimek-Szczykutowicz, M.; Malinowska, M.A.; Gałka, A.; Blažević, I.; Đulović, A.; Paprocka, P.; Wrzosek, M.; Szopa, A. Nasturtium officinale microshoot culture multiplied in PlantForm bioreactor-phytochemical profiling and biological activity. Molecules 2025, 30, 936. [Google Scholar] [CrossRef]
- Szewczyk, A.; Trepa, M.; Zych, D. Optimization of the production of secondary metabolites from furanocoumarin and furoquinoline alkaloid groups in in vitro Ruta corsica cultures grown in temporary immersion bioreactors. Molecules 2024, 29, 5261. [Google Scholar] [CrossRef]
- Szewczyk, A.; Marino, A.; Taviano, M.F.; Cambria, L.; Davì, F.; Trepa, M.; Grabowski, M.; Miceli, N. Studies on the accumulation of secondary metabolites and evaluation of biological activity of in vitro cultures of Ruta montana L. in temporary immersion bioreactors. Int. J. Mol. Sci. 2023, 24, 7045. [Google Scholar] [CrossRef]
- Mosqueda Frómeta, O.; Escalona Morgado, M.M.; Teixeira da Silva, J.A.; Pina Morgado, D.T.; Daquinta Gradaille, M.A. In vitro propagation of Gerbera jamesonii Bolus ex Hooker f. in a temporary immersion bioreactor. Plant Cell Tissue Organ Cult. 2017, 129, 543–551. [Google Scholar] [CrossRef]
- Rico, S.; Garrido, J.; Sánchez, C.; Ferreiro-Vera, C.; Codesido, V.; Vidal, N. A temporary immersion system to improve Cannabis sativa micropropagation. Front. Plant Sci. 2022, 13, 895971. [Google Scholar] [CrossRef] [PubMed]
- Martínez-Estrada, E.; Islas-Luna, B.; Pérez-Sato, J.A.; Bello-Bello, J.J. Temporary immersion improves in vitro multiplication and acclimatization of Anthurium andreanum Lind. Sci. Hortic. 2019, 249, 185–191. [Google Scholar] [CrossRef]
- Liu, Z.C.; Murch, J.S.; El-Demerdash, M.; Saxena, K.P. Artemisia judaica L.: Micropropagation and antioxidant activity. J. Biotechnol. 2004, 110, 63–71. [Google Scholar] [CrossRef]
- Bayraktar, M. Micropropagation of Stevia rebaudiana Bertoni using RITA® bioreactor. HortScience 2019, 54, 725–731. [Google Scholar] [CrossRef]
- Ramirez-Mosqueda, M.A.; Iglesias-Andreu, L.G.; Ramirez-Madero, G.; Hernandez-Rincon, E.U. Micropropagation of Stevia rebaudiana Bert. in temporary immersion systems and evaluation of genetic fidelity. S. Afr. J. Bot. 2016, 106, 238–243. [Google Scholar] [CrossRef]
- Bello-Bello, J.J.; Schettino-Salomón, S.; Ortega-Espinoza, J.; Spinoso-Castillo, J.L. A temporary immersion system for mass micropropagation of pitahaya (Hylocereus undatus). 3 Biotech 2021, 11, 437. [Google Scholar] [CrossRef]
- Reyes-Beristain, B.; Mancilla-Álvarez, E.; López-Buenfil, J.A.; Bello-Bello, J.J. Temporary immersion bioreactor for in vitro multiplication of raspberry (Rubus idaeus L.). Horticulturae 2025, 11, 842. [Google Scholar] [CrossRef]
- Skała, E.; Olszewska, M.A.; Makowczyńska, J.; Kicel, A. Effect of sucrose concentration on Rhaponticum carthamoides (Willd.) Iljin transformed root biomass, caffeoylquinic acid derivative, and flavonoid production. Int. J. Mol. Sci. 2022, 23, 13848. [Google Scholar] [CrossRef]
- Makowski, W.; Królicka, A.; Tokarz, B.; Szopa, A.; Ekiert, H.; Tokarz, K. Temporary immersion bioreactors as a useful tool for obtaining high productivity of phenolic compounds with strong antioxidant properties from Pontechium maculatum. Plant Cell Tissue Organ Cult. 2023, 153, 525–537. [Google Scholar] [CrossRef]
- Skała, E.; Picot, L.; Bijak, M.; Saluk-Bijak, J.; Szemraj, J.; Kicel, A.; Olszewska, M.A.; Sitarek, P. An efficient plant regeneration from Rhaponticum carthamoides transformed roots, enhanced caffeoylquinic acid derivatives production in pRi-transformed plants and their biological activity. Ind. Crops Prod. 2019, 129, 327–338. [Google Scholar] [CrossRef]
- Grzegorczyk-Karolak, I.; Staniewska, P.; Lebelt, L.; Piotrowska, D.G. Optimization of cultivation conditions of Salvia viridis L. shoots in the Plantform bioreactor to increase polyphenol production. Plant Cell Tissue Organ Cult. 2022, 149, 269–280. [Google Scholar] [CrossRef]
- Makowski, W.; Mrzygłód, K.; Szopa, A.; Kubica, P.; Krychowiak-Maśnicka, M.; Tokarz, K.M.; Tokarz, B.; Ryngwelska, I.; Paluszkiewicz, E.; Królicka, A. Effect of agitation and temporary immersion on growth and synthesis of antibacterial phenolic compounds in genus Drosera. Biomolecules 2024, 14, 1132. [Google Scholar] [CrossRef]
- Biskup, E.; Lojkowska, E. Evaluation of biological activities of Rhaponticum carthamoides extracts. J. Med. Plants Res. 2009, 3, 1092–1098. [Google Scholar]
- Biskup, E.; Szynklarz, B.; Golebiowski, M.; Borsuk, K.; Stepnowski, P.; Lojkowska, E. Composition and biological activity of Rhaponticum carthamoides extracts obtained from plants collected in Poland and Russia. J. Med. Plants Res. 2013, 7, 687–695. [Google Scholar]
- Liu, W.; Li, J.; Zhang, X.; Zu, Y.; Yang, Y.; Liu, W.; Xu, Z.; Gao, H.; Sun, X.; Jiang, X.; et al. Current advances in naturally occurring caffeoylquinic acids: Structure, bioactivity, and synthesis. J. Agric. Food Chem. 2020, 68, 10489–10516. [Google Scholar] [CrossRef]
- Vo, Q.V.; Thuy Hoa, D.T.; Hoa, N.T.; Tran, M.D.; Mechler, A. The radical scavenging activity of monocaffeoylquinic acids: The role of neighboring hydroxyl groups and pH levels. RSC Adv. 2024, 30, 4179–4187. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Li, K.; Xie, H.; Xie, Y.; Li, Y.; Zhao, X.; Jiang, X.; Chen, D. Antioxidant and cytoprotective effects of the di-O-caffeoylquinic acid family: The mechanism, structure–activity relationship, and conformational effect. Molecules 2018, 23, 222. [Google Scholar] [CrossRef]
- Boulebd, H.; Carmena-Bargueño, M.; Pérez-Sánchez, H. Exploring the antioxidant properties of caffeoylquinic and feruloylquinic acids: A computational study on hydroperoxyl radical scavenging and xanthine oxidase inhibition. Antioxidants 2023, 12, 1669. [Google Scholar] [CrossRef]
- Murashige, T.; Skoog, F. A revised medium for rapid growth and bioassays with tobacco tissue cultures. Physiol. Plant. 1962, 15, 473–497. [Google Scholar] [CrossRef]
- Marchelak, A.; Owczarek, A.; Rutkowska, M.; Michel, P.; Kolodziejczyk-Czepas, J.; Nowak, P.; Olszewska, M.A. New insights into antioxidant activity of Prunus spinosa flowers: Extracts, model polyphenols and their phenolic metabolites in plasma towards multiple in vivo-relevant oxidants. Phytochem. Lett. 2019, 30, 288–295. [Google Scholar] [CrossRef]
- Granica, S.; Czerwińska, M.E.; Piwowarski, J.P.; Ziaja, M.; Kiss, A.K. Chemical composition, antioxidative and anti-inflammatory activity of extracts prepared from aerial parts of Oenothera biennis L. and Oenothera paradoxa Hudziok obtained after seeds cultivation. J. Agric. Food Chem. 2013, 61, 801–810. [Google Scholar] [CrossRef] [PubMed]









| Culture Condition | Content | Sum of CQAs | mono-CQAs | di-CQAs | tri-CQAs | Flavonoids |
|---|---|---|---|---|---|---|
| Flask | ||||||
| 3T | mg/g DW | 2.78 ± 0.15 d | 1.75 ± 0.10 c | 0.80 ± 0.03 b | 0.23 ± 0.02 cd | 0.44 ± 0.02 a |
| mg/flask 1 | 0.72 ± 0.04 E | 0.46 ± 0.02 F | 0.21 ± 0.008 E | 0.06 ± 0.006 C | 0.11 ± 0.004 F | |
| 5T | mg/g DW | 2.89 ± 0.14 d | 2.18 ± 0.13 b | 0.53 ± 0.02 c | 0.18 ± 0.003 e | 0.30 ± 0.01 b |
| mg/flask | 1.07 ± 0.05 F | 0.81 ± 0.05 E | 0.20 ± 0.01 E | 0.07 ± 0.001 C | 0.80 ± 0.03 D | |
| PlantForm 1.5 h | ||||||
| 3T | mg/g DW | 2.36 ± 0.06 e | 1.34 ± 0.03 e | 0.76 ± 0.01 b | 0.25 ± 0.02 b c | 0.13 ± 0.005 c |
| mg/bioreactor 2 | 6.02 ± 0.15 D | 3.43 ± 0.08 D | 1.95 ± 0.04 C | 0.65 ± 0.04 B | 0.34 ± 0.01 E | |
| 5T | mg/g DW | 2.17 ± 0.04 e | 1.55 ± 0.03 d | 0.48 ± 0.02 c | 0.14 ± 0.006 f | 0.32 ± 0.007 b |
| mg/bioreactor | 10.13 ± 0.19 C | 7.23 ± 0.14 B | 2.25 ± 0.07 B | 0.65 ± 0.03 B | 1.52 ± 0.03 B | |
| PlantForm 3 h | ||||||
| 3T | mg/g DW | 3.29 ± 0.10 c | 2.15 ± 0.04 b | 0.81 ± 0.03 b | 0.33 ± 0.03 a | 0.34 ± 0.01 b |
| mg/bioreactor | 6.70 ± 0.20 D | 4.37 ± 0.09 C | 1.65 ± 0.07 D | 0.67 ± 0.06 B | 0.69 ± 0.02 D | |
| 5T | mg/g DW | 4.46 ± 0.24 a | 3.27 ± 0.14 a | 1.00 ± 0.07 a | 0.18 ± 0.03 def | 0.47 ± 0.03 a |
| mg/bioreactor | 16.13 ± 0.85 A | 11.85 ± 0.49 A | 3.63 ± 0.27 A | 0.65 ± 0.09 B | 1.70 ± 0.12 A | |
| PlantForm 6 h | ||||||
| 3T | mg/g DW | 2.27 ± 0.08 e | 1.27 ± 0.03 e | 0.71 ± 0.03 b | 0.28 ± 0.02 ab | 0.43 ± 0.02 a |
| mg/bioreactor | 5.97 ± 0.20 D | 3.35 ± 0.09 D | 1.88 ± 0.07 CD | 0.75 ± 0.04 B | 1.14 ± 0.05 C | |
| 5T | mg/g DW | 3.75 ± 0.10 b | 2.39 ± 0.05 b | 1.04 ± 0.03 a | 0.31 ± 0.02 ab | 0.42 ± 0.01 a |
| mg/bioreactor | 12.56 ± 0.33 B | 8.01 ± 0.17 B | 3.49 ± 0.11 A | 1.05 ± 0.08 A | 1.40 ± 0.04 B |
| Culture Condition | Shoot Length | Ratio of Shoots to Buds (%) | Hyperhydricity Structures (%) |
|---|---|---|---|
| Flask | |||
| 3T | 2.46 ± 0.09 b | 71.9:28.1 | 6.1 |
| 5T | 3.28 ± 0.07 a | 99.6:0.4 | 3.8 |
| PlantForm 1.5 h | |||
| 3T | 1.27 ± 0.02 e | 75.7:24.3 | 13.0 |
| 5T | 2.37 ± 0.07 b | 90.2:9.8 | 4.4 |
| PlantForm 3 h | |||
| 3T | 1.49 ± 0.03 d | 74.5:25.5 | 6.3 |
| 5T | 2.46 ± 0.04 b | 97.7:2.3 | 4.1 |
| PlantForm 6 h | |||
| 3T | 1.96 ± 0.04 c | 94.7:5.3 | 4.9 |
| 5T | 2.06 ± 0.03 c | 99.8:0.2 | 9.6 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Skała, E.; Kicel, A. PlantForm-Grown Shoots of Rhaponticum carthamoides (Willd.) Iljin as a Source of Caffeoylquinic Acid Derivatives and Antioxidant Potential of Shoot Extract. Molecules 2025, 30, 4724. https://doi.org/10.3390/molecules30244724
Skała E, Kicel A. PlantForm-Grown Shoots of Rhaponticum carthamoides (Willd.) Iljin as a Source of Caffeoylquinic Acid Derivatives and Antioxidant Potential of Shoot Extract. Molecules. 2025; 30(24):4724. https://doi.org/10.3390/molecules30244724
Chicago/Turabian StyleSkała, Ewa, and Agnieszka Kicel. 2025. "PlantForm-Grown Shoots of Rhaponticum carthamoides (Willd.) Iljin as a Source of Caffeoylquinic Acid Derivatives and Antioxidant Potential of Shoot Extract" Molecules 30, no. 24: 4724. https://doi.org/10.3390/molecules30244724
APA StyleSkała, E., & Kicel, A. (2025). PlantForm-Grown Shoots of Rhaponticum carthamoides (Willd.) Iljin as a Source of Caffeoylquinic Acid Derivatives and Antioxidant Potential of Shoot Extract. Molecules, 30(24), 4724. https://doi.org/10.3390/molecules30244724