Production of Dichostereum sordulentum Laccase and Its Entrapment in Lignocellulosic Biopolymers for Estrogen Biodegradation
Abstract
1. Introduction
2. Results and Discussion
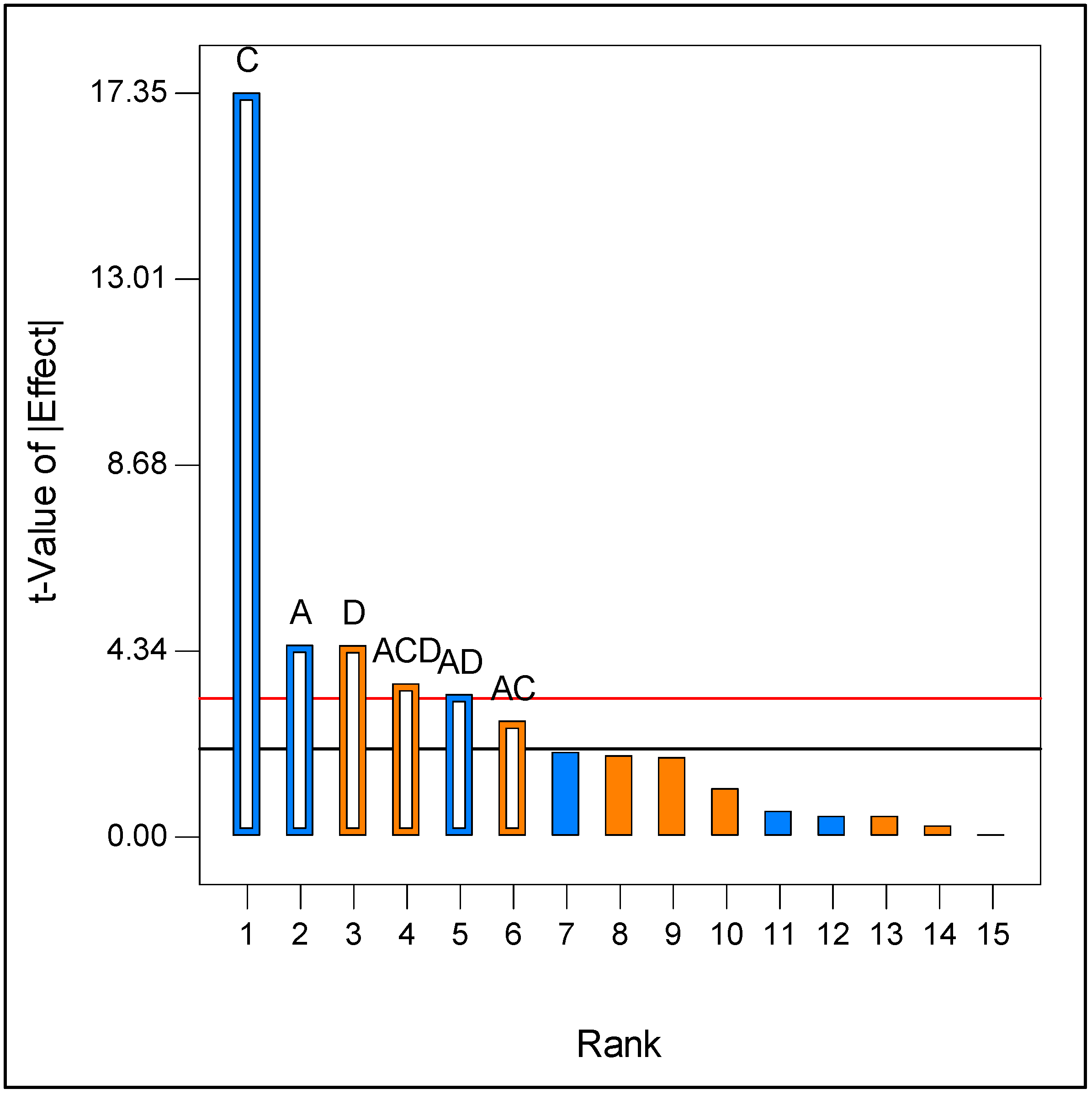
2.1. Optimization of Culture Medium for Laccase Production
− 12,850.66832 × Peptone − 9.42430 × Inoculum × Bark − 4.09822 × Inoculum
× Peptone − 1399.42989 × Bark × Peptone − 0.41247 × Inoculum2 − 1510.33822
× Bark2 + 830.79382 × Peptone2
2.2. Evaluation of EE2 Removal by Soluble and Immobilized Laccase
3. Materials and Methods
3.1. Chemicals
3.2. Laccase Activity Assay
3.3. Protein Determination
3.4. Laccase Production
3.5. Optimisation of Culture Medium for Laccase Production
3.5.1. Multilevel Categoric Model
3.5.2. Full 24 Factorial Design
3.5.3. Central Composite Design
3.6. Active Hydrogel Formation
3.7. Estrogen Biodegradation
3.8. Yeast Estrogen Screen (YES) Bioassay
4. Conclusions and Future Work
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Hejna, M.; Kapuścińska, D.; Aksmann, A. Pharmaceuticals in the Aquatic Environment: A Review on Eco-Toxicology and the Remediation Potential of Algae. Int. J. Environ. Res. Public Health 2022, 19, 7717. [Google Scholar] [CrossRef] [PubMed]
- Larsson, D.G.J. Pollution from Drug Manufacturing: Review and Perspectives. Philos. Trans. R. Soc. B Biol. Sci. 2014, 369, 20130571. [Google Scholar] [CrossRef]
- Yazdan, M.M.S.; Kumar, R.; Leung, S.W. The Environmental and Health Impacts of Steroids and Hormones in Wastewater Effluent, as Well as Existing Removal Technologies: A Review. Ecologies 2022, 3, 206–224. [Google Scholar] [CrossRef]
- Brian, J.V.; Harris, C.A.; Scholze, M.; Backhaus, T.; Booy, P.; Lamoree, M.; Pojana, G.; Jonkers, N.; Runnalls, T.; Bonfà, A.; et al. Accurate Prediction of the Response of Freshwater Fish to a Mixture of Estrogenic Chemicals. Environ. Health Perspect. 2005, 113, 721–728. [Google Scholar] [CrossRef]
- Beronius, A.; Rudén, C.; Hanberg, A.; Garai, J.; Håkansson, H. Assessing the Risks of Endocrine-Disrupting Chemicals. In Endocrine-Disrupting Chemicals in Food; Woodhead Publishing: Sawston, UK, 2009; pp. 356–380. ISBN 9781845692186. [Google Scholar]
- Zoeller, R.T. Environmental Chemicals Impacting the Thyroid: Targets and Consequences. Thyroid 2007, 17, 811–817. [Google Scholar] [CrossRef]
- Sanderson, J.T.; Letcher, R.J.; Heneweer, M.; Giesy, J.P.; Van Den Berg, M. Effects of Chloro-s-Triazine Herbicides and Metabolites on Aromatase Activity in Various Human Cell Lines and on Vitellogenin Production in Male Carp Hepatocytes. Environ. Health Perspect. 2001, 109, 1027–1031. [Google Scholar] [CrossRef]
- Moukhtari, F.E.; Martín-Pozo, L.; Zafra-Gómez, A. Strategies Based on the Use of Microorganisms for the Elimination of Pollutants with Endocrine-Disrupting Activity in the Environment. J. Environ. Chem. Eng. 2023, 11, 109268. [Google Scholar] [CrossRef]
- Johnson, A.C.; Dumont, E.; Williams, R.J.; Oldenkamp, R.; Cisowska, I.; Sumpter, J.P. Do Concentrations of Ethinylestradiol, Estradiol, and Diclofenac in European Rivers Exceed Proposed EU Environmental Quality Standards? Environ. Sci. Technol. 2013, 47, 12297–12304. [Google Scholar] [CrossRef]
- Du, B.; Fan, G.; Yu, W.; Yang, S.; Zhou, J.; Luo, J. Occurrence and Risk Assessment of Steroid Estrogens in Environmental Water Samples: A Five-Year Worldwide Perspective. Environ. Pollut. 2020, 267, 115405. [Google Scholar] [CrossRef] [PubMed]
- Griffero, L.; Alcántara-Durán, J.; Alonso, C.; Rodríguez-Gallego, L.; Moreno-González, D.; García-Reyes, J.F.; Molina-Díaz, A.; Pérez-Parada, A. Basin-Scale Monitoring and Risk Assessment of Emerging Contaminants in South American Atlantic Coastal Lagoons. Sci. Total Environ. 2019, 697, 134058. [Google Scholar] [CrossRef] [PubMed]
- Caldwell, D.J.; Mastrocco, F.; Anderson, P.D.; Länge, R.; Sumpter, J.P. Predicted-No-Effect Concentrations for the Steroid Estrogens Estrone, 17β-Estradiol, Estriol, and 17α-Ethinylestradiol. Environ. Toxicol. Chem. 2012, 31, 1396–1406. [Google Scholar] [CrossRef]
- European Commission. Amending Directive 2000/60/EC Establishing a Framework for Community Action in the Field of Water Policy, Directive 2006/118/EC on the Protection of Groundwater against Pollution and Deterioration and Directive 2008/105/EC on Environmental Quality Standards in the Field of Water Policy; European Commission: Bruselas, Belgium, 2022; Volume 0344.
- Klaic, M.; Jirsa, F. 17α-Ethinylestradiol (EE2): Concentrations in the Environment and Methods for Wastewater Treatment—An Update. RSC Adv. 2022, 12, 12794–12805. [Google Scholar] [CrossRef] [PubMed]
- Chafi, S.; Azzouz, A.; Ballesteros, E. Occurrence and Distribution of Endocrine Disrupting Chemicals and Pharmaceuticals in the River Bouregreg (Rabat, Morocco). Chemosphere 2022, 287, 132202. [Google Scholar] [CrossRef] [PubMed]
- Haakenstad, A.; Angelino, O.; Irvine, C.M.S.; Bhutta, Z.A.; Bienhoff, K.; Bintz, C.; Causey, K.; Ashworth Dirac, M.; Fullman, N.; Gakidou, E.; et al. Measuring Contraceptive Method Mix, Prevalence, and Demand Satisfied by Age and Marital Status in 204 Countries and Territories, 1970–2019: A Systematic Analysis for the Global Burden of Disease Study 2019. Lancet 2022, 400, 295–327. [Google Scholar] [CrossRef] [PubMed]
- Tang, Z.; Liu, Z.; Wang, H.; Dang, Z.; Liu, Y. Occurrence and Removal of 17α-Ethynylestradiol (EE2) in Municipal Wastewater Treatment Plants: Current Status and Challenges. Chemosphere 2021, 271, 129551. [Google Scholar] [CrossRef]
- Zhou, Y.; Zha, J.; Wang, Z. Occurrence and Fate of Steroid Estrogens in the Largest Wastewater Treatment Plant in Beijing, China. Environ. Monit. Assess. 2012, 184, 6799–6813. [Google Scholar] [CrossRef]
- Pessoa, G.P.; de Souza, N.C.; Vidal, C.B.; Alves, J.A.C.; Firmino, P.I.M.; Nascimento, R.F.; dos Santos, A.B. Occurrence and Removal of Estrogens in Brazilian Wastewater Treatment Plants. Sci. Total Environ. 2014, 490, 288–295. [Google Scholar] [CrossRef]
- López-Velázquez, K.; Guzmán-Mar, J.L.; Saldarriaga-Noreña, H.A.; Murillo-Tovar, M.A.; Villanueva-Rodríguez, M. Ecological Risk Assessment Associated with Five Endocrine-Disrupting Compounds in Wastewater Treatment Plants of Northeast Mexico. Environ. Sci. Pollut. Res. 2023, 30, 30714–30726. [Google Scholar] [CrossRef]
- Gomes, I.B.; Maillard, J.Y.; Simões, L.C.; Simões, M. Emerging Contaminants Affect the Microbiome of Water Systems—Strategies for Their Mitigation. npj Clean Water 2020, 3, 39. [Google Scholar] [CrossRef]
- Lloret, L.; Eibes, G.; Moreira, M.T.; Feijoo, G.; Lema, J.M. Removal of Estrogenic Compounds from Filtered Secondary Wastewater Effluent in a Continuous Enzymatic Membrane Reactor. Identification of Biotransformation Products. Environ. Sci. Technol. 2013, 47, 4536–4543. [Google Scholar] [CrossRef]
- Zdarta, J.; Jankowska, K.; Strybel, U.; Marczak, Ł.; Nguyen, L.N.; Oleskowicz-Popiel, P.; Jesionowski, T. Bioremoval of Estrogens by Laccase Immobilized onto Polyacrylonitrile/Polyethersulfone Material: Effect of Inhibitors and Mediators, Process Characterization and Catalytic Pathways Determination. J. Hazard. Mater. 2022, 432, 128688. [Google Scholar] [CrossRef]
- Dzionek, A.; Wojcieszyńska, D.; Guzik, U. Natural Carriers in Bioremediation: A Review. Electron. J. Biotechnol. 2016, 23, 28–36. [Google Scholar] [CrossRef]
- Siddiqui, N.M.; Dahiya, P. Enzyme-Based Biodegradation of Toxic Environmental Pollutants. In Development in Wastewater Treatment Research and Processes; Rodriguez-Couto, S., Shah, M.P., Eds.; Elsevier: Amsterdam, The Netherlands, 2022; pp. 311–333. ISBN 9780323858397. [Google Scholar]
- Zdarta, J.; Nguyen, L.N.; Jankowska, K.; Jesionowski, T.; Nghiem, L.D. A Contemporary Review of Enzymatic Applications in the Remediation of Emerging Estrogenic Compounds. Crit. Rev. Environ. Sci. Technol. 2021, 52, 2661–2690. [Google Scholar] [CrossRef]
- Macellaro, G.; Pezzella, C.; Cicatiello, P.; Sannia, G.; Piscitelli, A. Fungal Laccases Degradation of Endocrine Disrupting Compounds. BioMed Res. Int. 2014, 2014, 614038. [Google Scholar] [CrossRef] [PubMed]
- Bilal, M.; Iqbal, H.M.N. Persistence and Impact of Steroidal Estrogens on the Environment and Their Laccase-Assisted Removal. Sci. Total Environ. 2019, 690, 447–459. [Google Scholar] [CrossRef] [PubMed]
- Brugnari, T.; Braga, D.M.; dos Santos, C.S.A.; Torres, B.H.C.; Modkovski, T.A.; Haminiuk, C.W.I.; Maciel, G.M. Laccases as Green and Versatile Biocatalysts: From Lab to Enzyme Market—An Overview. Bioresour. Bioprocess. 2021, 8, 131. [Google Scholar] [CrossRef] [PubMed]
- Janusz, G.; Skwarek, E.; Pawlik, A. Potential of Laccase as a Tool for Biodegradation of Wastewater Micropollutants. Water 2023, 15, 3770. [Google Scholar] [CrossRef]
- Chaudhary, S.; Singh, A.; Varma, A.; Porwal, S. Recent Advancements in Biotechnological Applications of Laccase as a Multifunctional Enzyme. J. Pure Appl. Microbiol. 2022, 16, 1479–1491. [Google Scholar] [CrossRef]
- Zofair, S.F.F.; Ahmad, S.; Hashmi, M.A.; Khan, S.H.; Khan, M.A.; Younus, H. Catalytic Roles, Immobilization and Management of Recalcitrant Environmental Pollutants by Laccases: Significance in Sustainable Green Chemistry. J. Environ. Manag. 2022, 309, 114676. [Google Scholar] [CrossRef]
- Yu, S.; Cheng, J.; Li, K.; Zhang, S.; Dong, X.; Paizullakhanov, M.S.; Chen, D. Preparation and Application of Laccase-Immobilized Magnetic Biochar for Effective Degradation of Endocrine Disruptors: Efficiency and Mechanistic Analysis. Int. J. Biol. Macromol. 2025, 305, 141167. [Google Scholar] [CrossRef]
- Rybarczyk, A.; Smułek, W.; Ejsmont, A.; Goscianska, J.; Jesionowski, T.; Zdarta, J. The Role of Metal–Organic Framework (MOF) in Laccase Immobilization for Advanced Biocatalyst Formation for Use in Micropollutants Removal. Environ. Pollut. 2025, 371, 125954. [Google Scholar] [CrossRef]
- Pourkhanali, K.; Khayati, G.; Mizani, F.; Raouf, F. Isolation, Identification and Optimization of Enhanced Production of Laccase from Galactomyces geotrichum under Solid-State Fermentation. Prep. Biochem. Biotechnol. 2021, 51, 659–668. [Google Scholar] [CrossRef] [PubMed]
- He, X.J.; Jiao, J.; Gai, Q.Y.; Fu, J.X.; Fu, Y.J.; Zhang, Z.Y.; Gao, J. Bioprocessing of Pigeon Pea Roots by a Novel Endophytic Fungus Penicillium rubens for the Improvement of Genistein Yield Using Semi-Solid-State Fermentation with Water. Innov. Food Sci. Emerg. Technol. 2023, 90, 103519. [Google Scholar] [CrossRef]
- Sheldon, R.A.; Woodley, J.M. Role of Biocatalysis in Sustainable Chemistry. Chem. Rev. 2018, 118, 801–838. [Google Scholar] [CrossRef] [PubMed]
- Tikhonov, B.B.; Sulman, E.M.; Stadol’nikova, P.Y.; Sulman, A.M.; Golikova, E.P.; Sidorov, A.I.; Matveeva, V.G. Immobilized Enzymes from the Class of Oxidoreductases in Technological Processes: A Review. Catal. Ind. 2019, 11, 251–263. [Google Scholar] [CrossRef]
- Wang, H.; Tang, L.X.; Ye, Y.F.; Ma, J.X.; Li, X.; Si, J.; Cui, B.K. Laccase Immobilization and Its Degradation of Emerging Pollutants: A Comprehensive Review. J. Environ. Manag. 2024, 359, 120984. [Google Scholar] [CrossRef]
- Daronch, N.A.; Kelbert, M.; Pereira, C.S.; de Araújo, P.H.H.; de Oliveira, D. Elucidating the Choice for a Precise Matrix for Laccase Immobilization: A Review. Chem. Eng. J. 2020, 397, 125506. [Google Scholar] [CrossRef]
- Serbent, M.P.; Magario, I.; Saux, C. Immobilizing White-Rot Fungi Laccase: Toward Bio-Derived Supports as a Circular Economy Approach in Organochlorine Removal. Biotechnol. Bioeng. 2024, 121, 434–455. [Google Scholar] [CrossRef]
- Hasanov, I.; Raud, M.; Kikas, T. The Role of Ionic Liquids in the Lignin Separation from Lignocellulosic Biomass. Energies 2020, 13, 4864. [Google Scholar] [CrossRef]
- Meyer, J.; Meyer, L.E.; Kara, S. Enzyme Immobilization in Hydrogels: A Perfect Liaison for Efficient and Sustainable Biocatalysis. Eng. Life Sci. 2022, 22, 165–177. [Google Scholar] [CrossRef]
- Imam, H.T.; Marr, P.C.; Marr, A.C. Enzyme Entrapment, Biocatalyst Immobilization without Covalent Attachment. Green Chem. 2021, 23, 4980–5005. [Google Scholar] [CrossRef]
- Kim, M.H.; An, S.; Won, K.; Kim, H.J.; Lee, S.H. Entrapment of Enzymes into Cellulose-Biopolymer Composite Hydrogel Beads Using Biocompatible Ionic Liquid. J. Mol. Catal. B Enzym. 2012, 75, 68–72. [Google Scholar] [CrossRef]
- Vázquez, V.; Giorgi, V.; Bonfiglio, F.; Menéndez, P.; Gioia, L.; Ovsejevi, K. Lignocellulosic Residues from Bioethanol Production: A Novel Source of Biopolymers for Laccase Immobilization. RSC Adv. 2023, 13, 13463–13471. [Google Scholar] [CrossRef] [PubMed]
- Botto, E.; D’annibale, A.; Petruccioli, M.; Galetta, A.; Martínez, S.; Bettucci, L.; Menéndez, P. Ligninolytic Enzymes Production by Dichostereum sordulentum Cultures in the Presence of Eucalyptus Bark as a Natural Laccase Stimulator. J. Adv. Biotechnol. 2015, 5, 478–484. [Google Scholar] [CrossRef]
- Montgomery, D.C. Design and Analysis of Experiments, 8th ed.; John Wiley: New York, NY, USA, 2012. [Google Scholar]
- Hadiyat, M.A.; Sopha, B.M.; Wibowo, B.S. Response Surface Methodology Using Observational Data: A Systematic Literature Review. Appl. Sci. 2022, 12, 10663. [Google Scholar] [CrossRef]
- Fonseca, M.I.; Shimizu, E.; Zapata, P.D.; Villalba, L.L. Copper Inducing Effect on Laccase Production of White Rot Fungi Native from Misiones (Argentina). Enzym. Microb. Technol. 2010, 46, 534–539. [Google Scholar] [CrossRef]
- Dirección General Forestal MGAP. Superficie Forestal Del Uruguay 2022 (Bosques Plantados); Dirección General Forestal MGAP: Montevideo, Uruguay, 2022. [Google Scholar]
- Gioia, L.; Manta, C.; Ovsejevi, K.; Burgueño, J.; Menéndez, P.; Rodriguez-Couto, S. Enhancing Laccase Production by a Newly-Isolated Strain of Pycnoporus sanguineus with High Potential for Dye Decolouration. RSC Adv. 2014, 4, 34096–34103. [Google Scholar] [CrossRef]
- Gupta, A.; Jana, A.K. Production of Laccase by Repeated Batch Semi-Solid Fermentation Using Wheat Straw as Substrate and Support for Fungal Growth. Bioprocess Biosyst. Eng. 2019, 42, 499–512. [Google Scholar] [CrossRef]
- Ripollés, C.; Ibáñez, M.; Sancho, J.V.; López, F.J.; Hernández, F. Determination of 17β-Estradiol and 17α-Ethinylestradiol in Water at Sub-Ppt Levels by Liquid Chromatography Coupled to Tandem Mass Spectrometry. Anal. Methods 2014, 6, 5028–5037. [Google Scholar] [CrossRef]
- Turull, M.; Buttiglieri, G.; Vazquez, V.; Rodriguez-Mozaz, S.; Santos, L.H.M.L.M. Analytical Upgrade of a Methodology Based on UHPLC-MS/MS for the Analysis of Endocrine Disrupting Compounds in Greywater. Water Emerg. Contam. Nanoplast. 2023, 2, 17. [Google Scholar] [CrossRef]
- Nahar, L.; Onder, A.; Sarker, S.D. A Review on the Recent Advances in HPLC, UHPLC and UPLC Analyses of Naturally Occurring Cannabinoids (2010–2019). Phytochem. Anal. 2020, 31, 413–457. [Google Scholar] [CrossRef]
- Auriol, M.; Filali-Meknassi, Y.; Tyagi, R.D.; Adams, C.D. Laccase-Catalyzed Conversion of Natural and Synthetic Hormones from a Municipal Wastewater. Water Res. 2007, 41, 3281–3288. [Google Scholar] [CrossRef]
- Pamidipati, S.; Ahmed, A. A First Report on Competitive Inhibition of Laccase Enzyme by Lignin Degradation Intermediates. Folia Microbiol. 2020, 65, 431–437. [Google Scholar] [CrossRef] [PubMed]
- Chmelová, D.; Ondrejovič, M.; Miertuš, S. Laccases as Effective Tools in the Removal of Pharmaceutical Products from Aquatic Systems. Life 2024, 14, 230. [Google Scholar] [CrossRef] [PubMed]
- Bezerra, T.M.D.S.; Bassan, J.C.; Santos, V.T.D.O.; Ferraz, A.; Monti, R. Covalent Immobilization of Laccase in Green Coconut Fiber and Use in Clarification of Apple Juice. Process Biochem. 2015, 50, 417–423. [Google Scholar] [CrossRef]
- Posilipo, A.; Russo, M.E.; Marzocchella, A. Characterization of Laccase Adsorption on Lignocellulosic Biomasses. Chem. Eng. Trans. 2024, 110, 271–276. [Google Scholar] [CrossRef]
- Beck, I.C.; Bruhn, R.; Gandrass, J. Analysis of Estrogenic Activity in Coastal Surface Waters of the Baltic Sea Using the Yeast Estrogen Screen. Chemosphere 2006, 63, 1870–1878. [Google Scholar] [CrossRef]
- Gałązka, A.; Jankiewicz, U.; Szczepkowski, A. Biochemical Characteristics of Laccases and Their Practical Application in the Removal of Xenobiotics from Water. Appl. Sci. 2023, 13, 4394. [Google Scholar] [CrossRef]
- Baskar, A.V.; Bolan, N.; Hoang, S.A.; Sooriyakumar, P.; Kumar, M.; Singh, L.; Jasemizad, T.; Padhye, L.P.; Singh, G.; Vinu, A.; et al. Recovery, Regeneration and Sustainable Management of Spent Adsorbents from Wastewater Treatment Streams: A Review. Sci. Total Environ. 2022, 822, 153555. [Google Scholar] [CrossRef]
- Smith, P.K.; Krohn, R.I.; Hermanson, G.T.; Mallia, A.K.; Gartner, F.H.; Provenzano, M.D.; Fujimoto, E.K.; Goeke, N.M.; Olson, B.J.; Klenk, D.C. Measurement of Protein Using Bicinchoninic Acid. Anal. Biochem. 1985, 150, 76–85. [Google Scholar] [CrossRef]
- Martínez, S.; Nakasone, K.K. New Records of Interesting Corticioid Basidiomycota from Uruguay. Check List 2014, 10, 1237–1242. [Google Scholar] [CrossRef][Green Version]
- Viswanath, B.; Rajesh, B.; Janardhan, A.; Kumar, A.P.; Narasimha, G. Fungal Laccases and Their Applications in Bioremediation. Enzym. Res. 2014, 2014, 163242. [Google Scholar] [CrossRef] [PubMed]
- Routledge, E.J.; Sumpter, J.P. Estrogenic Activity of Surfactants and Some of Their Degradation Products Assessed Using a Recombinant Yeast Screen. Environ. Toxicol. Chem. 1996, 15, 241–248. [Google Scholar] [CrossRef]
- Bila, D.; Montalvão, A.F.; Azevedo, D.d.A.; Dezotti, M. Estrogenic Activity Removal of 17β-Estradiol by Ozonation and Identification of by-Products. Chemosphere 2007, 69, 736–746. [Google Scholar] [CrossRef] [PubMed]


| Source | Sum of Squares | df | Mean Square | F Value | p-Value Prob > F |
|---|---|---|---|---|---|
| Model | 3.339 × 108 | 6 | 5.566 × 107 | 9.17 | 0.0021 |
| A-Source of N | 2.433 × 108 | 1 | 2.433 × 108 | 40.07 | 0.0001 |
| B-Glucose | 2.085 × 107 | 1 | 2.085 × 107 | 3.43 | 0.0969 |
| C-Bark | 1.150 × 107 | 1 | 1.150 × 107 | 1.89 | 0.2020 |
| AB | 7.373 × 106 | 1 | 7.373 × 106 | 1.21 | 0.2991 |
| AC | 9.213 × 106 | 1 | 9.213 × 106 | 1.52 | 0.2492 |
| BC | 4.172 × 107 | 1 | 4.172 × 107 | 6.87 | 0.0277 |
| Residual | 5.464 × 107 | 9 | 6.071 × 106 | ||
| Lack of Fit | 1.141 × 106 | 1 | 1.141 × 106 | 0.17 | 0.6904 |
| Pure Error | 5.350 × 107 | 8 | 6.688 × 106 | ||
| Cor Total | 3.886 × 108 | 15 |
| Source | Sum of Squares | df | Mean Square | F Value | p-Value Prob > F |
|---|---|---|---|---|---|
| Model | 2.768 × 108 | 5 | 5.535 × 107 | 4.07 | 0.0283 |
| A-Source of N | 1.739 × 108 | 1 | 1.739 × 108 | 12.78 | 0.0051 |
| B-Glycerol | 6.395 × 106 | 1 | 6.395 × 106 | 0.47 | 0.5087 |
| C-Bark | 5.839 × 107 | 1 | 5.839 × 107 | 4.29 | 0.0652 |
| AB | 1.666 × 107 | 1 | 1.666 × 107 | 1.22 | 0.2945 |
| AC | 2.143 × 107 | 1 | 2.143 × 107 | 1.57 | 0.2382 |
| Residual | 1.361 × 108 | 10 | 1.361 × 107 | ||
| Lack of Fit | 1.066 × 107 | 2 | 5.328 × 106 | 0.34 | 0.7218 |
| Pure Error | 1.255 × 108 | 8 | 1.568 × 107 | ||
| Cor Total | 4.129 × 108 | 15 |
| Source | Sum of Squares | df | Mean Square | F Value | p-Value Prob > F |
|---|---|---|---|---|---|
| Model | 1.769 × 109 | 6 | 2.948 × 108 | 58.68 | <0.0001 |
| A-Inoculum | 1.004 × 108 | 1 | 1.004 × 108 | 19.97 | 0.0001 |
| C-Bark | 1.513 × 109 | 1 | 1.513 × 109 | 301.04 | <0.0001 |
| D-Peptone | 9.977 × 107 | 1 | 9.977 × 107 | 19.86 | 0.0002 |
| AC | 3.649 × 107 | 1 | 3.649 × 107 | 7.26 | 0.0124 |
| AD | 5.522 × 107 | 1 | 5.522 × 107 | 10.99 | 0.0028 |
| ACD | 6.394 × 107 | 1 | 6.394 × 107 | 12.73 | 0.0015 |
| Curvature | 1.057 × 109 | 1 | 1.057 × 109 | 210.28 | <0.0001 |
| Residual | 1.256 × 108 | 25 | 5.024 × 106 | ||
| Lack of Fit | 6.914 × 107 | 9 | 7.682 × 106 | 2.18 | 0.0837 |
| Pure Error | 5.647 × 107 | 16 | 3.529 × 106 | ||
| Cor Total | 2.951 × 109 | 32 |
| Source | Squares | df | Square | Value | Prob > F |
|---|---|---|---|---|---|
| Model | 8.302 × 108 | 9 | 9.224 × 107 | 49.49 | 0.0002 |
| A-Inoculum | 6.184 × 106 | 1 | 6.184 × 106 | 3.32 | 0.1282 |
| B-Bark | 8.426 × 107 | 1 | 8.426 × 107 | 45.20 | 0.0011 |
| C-Peptone | 1.538 × 107 | 1 | 1.538 × 107 | 8.25 | 0.0349 |
| AB | 3.291 × 107 | 1 | 3.291 × 107 | 17.66 | 0.0085 |
| AC | 1.107 × 107 | 1 | 1.107 × 107 | 5.94 | 0.0589 |
| BC | 7.258 × 107 | 1 | 7.258 × 107 | 38.94 | 0.0015 |
| A2 | 2.574 × 107 | 1 | 2.574 × 107 | 13.81 | 0.0138 |
| B2 | 1.661 × 106 | 1 | 1.661 × 106 | 0.89 | 0.3885 |
| C2 | 1.081 × 106 | 1 | 1.081 × 106 | 0.58 | 0.4806 |
| Residual | 9.320 × 106 | 5 | 1.864 × 106 | ||
| Lack of Fit | 7.317 × 106 | 2 | 3.659 × 106 | 5.48 | 0.0996 |
| Pure Error | 2.003 × 106 | 3 | 6.675 × 105 | ||
| Cor Total | 8.395 × 108 | 14 |
| Sample | EE2 Recovered (%) |
|---|---|
| EE2 | 102.6 ± 7.1 |
| R-SLac 0.1 | 27.2 ± 2.1 |
| R-SLac 0.3 | 9.6 ± 1.0 |
| R-SLac 0.5 | 7.1 ± 1.5 |
| R-ILac | 1.2 ± 0.5 |
| B-EE2 | 3.0 ± 0.2 |
| B-ILac | <LOD |
| SLac | <LOD |
| R-ILac W | 10.6 ± 5.7 |
| B-ILac W | <LOD |
| B-EE2 W | 20.2 * |
| Sample | EC50 (µg/L) | Relative Potency |
|---|---|---|
| E2 | 0.047 ± 0.02 | |
| EE2 | 0.028 ± 0.02 | 1.68 |
| EE2 + soluble laccase (R-SLac 0.5) | 0.391 ± 0.33 | 1.20 × 10−1 |
| EE2 + hydrogel beads (B-EE2) | 0.598 ± 0.13 | 7.90 × 10−2 |
| EE2 + active hydrogel beads (R-ILac) | 4.819 ± 0.22 | 9.75 × 10−3 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Vázquez, V.; Botto, E.; Bertone, A.; Turull, M.; Santos, L.H.M.L.M.; Giorgi, V.; Bonfiglio, F.; García-Alonso, J.; Menéndez, P.; Ovsejevi, K.; et al. Production of Dichostereum sordulentum Laccase and Its Entrapment in Lignocellulosic Biopolymers for Estrogen Biodegradation. Molecules 2025, 30, 4713. https://doi.org/10.3390/molecules30244713
Vázquez V, Botto E, Bertone A, Turull M, Santos LHMLM, Giorgi V, Bonfiglio F, García-Alonso J, Menéndez P, Ovsejevi K, et al. Production of Dichostereum sordulentum Laccase and Its Entrapment in Lignocellulosic Biopolymers for Estrogen Biodegradation. Molecules. 2025; 30(24):4713. https://doi.org/10.3390/molecules30244713
Chicago/Turabian StyleVázquez, Valeria, Emiliana Botto, Alejandra Bertone, Marta Turull, Lúcia H. M. L. M. Santos, Victoria Giorgi, Fernando Bonfiglio, Javier García-Alonso, Pilar Menéndez, Karen Ovsejevi, and et al. 2025. "Production of Dichostereum sordulentum Laccase and Its Entrapment in Lignocellulosic Biopolymers for Estrogen Biodegradation" Molecules 30, no. 24: 4713. https://doi.org/10.3390/molecules30244713
APA StyleVázquez, V., Botto, E., Bertone, A., Turull, M., Santos, L. H. M. L. M., Giorgi, V., Bonfiglio, F., García-Alonso, J., Menéndez, P., Ovsejevi, K., & Gioia, L. (2025). Production of Dichostereum sordulentum Laccase and Its Entrapment in Lignocellulosic Biopolymers for Estrogen Biodegradation. Molecules, 30(24), 4713. https://doi.org/10.3390/molecules30244713







