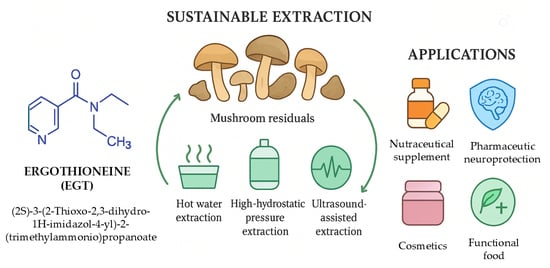
Ergothioneine: An Antioxidative, Neuroprotective and Anti-Inflammatory Compound from Mushroom Residuals
Abstract
1. Introduction
2. Ergothioneine Bioactivity
Antioxidant Mechanism of Ergothioneine
3. Mushroom Processing Residuals as an EGT Source
3.1. The Types of Mushroom Processing Residuals
3.2. Ergothioneine Content in Mushroom Species and Residuals
3.3. Factors Influencing EGT Content in Mushrooms and Residuals
3.4. Variability of Ergothioneine Content: Effects of Drying and Species Differences
3.5. Enhancing Ergothioneine Biosynthesis
4. Ergothioneine Extraction from Residuals and Purification
4.1. Extraction Principles
4.2. Extraction Methods
4.3. Purification Techniques
4.4. Optimisation of Extraction Conditions and Industrial Feasibility
4.5. Integration of Nano- and Molecular Encapsulation Technologies
4.6. Challenges Related to Substrate Complexity
5. Applications of Ergothioneine from Mushroom Residuals
5.1. Nutraceuticals and Functional Foods
5.2. Cosmetics
5.3. Pharmaceutical and Therapeutic Uses
5.4. Regulatory Status and Market Perspective
6. Economic Feasibility and Environmental Sustainability
6.1. Valorization of Waste and Circular Economy Principles
6.2. Cost-Effectiveness of EGT Recovery from Residuals
7. Future Perspectives and Research Gaps
8. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Paul, B.D. Ergothioneine: A stress vitamin with antiaging, vascular, and neuroprotective roles? Antioxid. Redox Signal. 2022, 36, 1306–1317. [Google Scholar] [CrossRef]
- Cheah, I.K.; Halliwell, B. Ergothioneine, recent developments. Redox Biol. 2021, 42, 101868. [Google Scholar] [CrossRef]
- Cheah, I.K.; Halliwell, B. Ergothioneine; antioxidant potential, physiological function and role in disease. Biochim. Biophys. Acta (BBA)—Mol. Basis Dis. 2012, 1822, 784–793. [Google Scholar] [CrossRef]
- Beelman, R.B.; Kalaras, M.D.; Phillips, A.T.; Richie, J.P., Jr. Is ergothioneine a ‘longevity vitamin’ limited in the American diet? J. Nutr. Sci. 2020, 9, e52. [Google Scholar] [CrossRef] [PubMed]
- Wijesekara, T.; Xu, B. Occurrence, dietary sources, quantification and bioactivities of natural antioxidant ergothioneine—A longavity vitamin? Int. J. Food Sci. Technol. 2024, 59, 5951–5963. [Google Scholar] [CrossRef]
- Sato, S.; Saika, A.; Koshiyama, T.; Higashiyama, Y.; Fukuoka, T.; Morita, T. Biosynthesis of ergothioneine: Current state, achievements, and perspectives. Appl. Microbiol. Biotechnol. 2025, 109, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Beelman, R.B.; Phillips, A.T.; Richie, J.P.; Ba, D.M.; Duiker, S.W.; Kalaras, M.D. Health consequences of improving the content of ergothioneine in the food supply. FEBS Lett. 2022, 596, 1231–1240. [Google Scholar] [CrossRef]
- Kalaras, M.D.; Richie, J.P.; Calcagnotto, A.; Beelman, R.B. Mushrooms: A rich source of the antioxidants ergothioneine and glutathione. Food Chem. 2017, 233, 429–433. [Google Scholar] [CrossRef]
- Lam-Sidun, D.; Peters, K.M.; Borradaile, N.M. Mushroom-Derived Medicine? Preclinical Studies Suggest Potential Benefits of Ergothioneine for Cardiometabolic Health. Int. J. Mol. Sci. 2021, 22, 3246. [Google Scholar] [CrossRef]
- Lee, K.; Lee, H.; Choi, Y.; Kim, Y.; Jeong, H.S.; Lee, J.S. Effect of different cooking methods on the true retention of vitamins, minerals, and bioactive compounds in shiitake mushrooms (Lentinula endodes). Food Sci. Technol. Res. 2019, 25, 115–122. [Google Scholar] [CrossRef]
- Zhang, L.; Cheah, I.K.; Zajac, I.; Brindal, E.; Kakoschke, N. Ergothioneine for cognitive health, longevity and healthy ageing: Where are we now? Proc. Nutr. Soc. 2025, 1–15. [Google Scholar] [CrossRef]
- Ames, B.N. Prolonging healthy aging: Longevity vitamins and proteins. Proc. Natl. Acad. Sci. USA 2018, 115, 10836–10844. [Google Scholar] [CrossRef]
- Bazela, K.; Solyga-Zurek, A.; Debowska, R.; Rogiewicz, K.; Bartnik, E.; Eris, I. l-Ergothioneine Protects Skin Cells against UV-Induced Damage—A Preliminary Study. Cosmetics 2014, 1, 51–60. [Google Scholar] [CrossRef]
- Zhao, A.; Berglund, L.; Völtz, L.R.; Swamy, R.; Antonopoulou, I.; Xiong, S.; Mouzon, J.; Bismarck, A.; Oksman, K. Fungal innovation: Harnessing mushrooms for production of sustainable functional materials. Adv. Funct. Mater. 2024, 35, 2412753. [Google Scholar] [CrossRef]
- Stavropoulou, N.A.; Lazou, A.E.; Giannakourou, M.C. Sustainable Utilization of Mushroom By-Products Processed with a Combined Osmotic Dehydration Pretreatment and a Hot-Air-Drying Step. Foods 2024, 13, 1339. [Google Scholar] [CrossRef]
- Navarro-Simarro, P.; Giménez-Bastida, J.A.; Vázquez-Carrillo, M.A.; Ortiz, C.; Pérez-Sanz, F. Valorization of mushroom by-products for sustainability: Exploring antioxidant and prebiotic properties. bioRxiv 2025. [Google Scholar] [CrossRef]
- Valenzuela-Cobos, J.D.; Guevara-Viejó, F.; Grijalva-Endara, A.; Vicente-Galindo, P.; Galindo-Villardón, P. Production and Evaluation of Pleurotus spp. Hybrids Cultivated on Ecuadorian Agro-Industrial Wastes: Using Multivariate Statistical Methods. Sustainability 2023, 15, 15546. [Google Scholar] [CrossRef]
- Zhu, M.; Han, Y.; Hu, X.; Gong, C.; Ren, L. Ergothioneine production by submerged fermentation of a medicinal mushroom Panus conchatus. Fermentation 2022, 8, 431. [Google Scholar] [CrossRef]
- Halliwell, B.; Tang, R.M.; Cheah, I.K. Diet-derived antioxidants: The special case of ergothioneine. Annu. Rev. Food Sci. Technol. 2023, 14, 323–345. [Google Scholar] [CrossRef]
- Takhor, N.H.; Phan, C.W. The role of Ergothioneine in cognition and age-related neurodegenerative disease: A systematic review. Inflammopharmacology 2025, 33, 2351–2375. [Google Scholar] [CrossRef]
- Kataoka, R.; Nigaki, A.; Barua, B.S.; Yamashita, K. Ergothioneine circulation in mushroom cultivation using food waste recycling. Recycling 2025, 10, 91. [Google Scholar] [CrossRef]
- Fu, Y.; Guan, H.; Yu, M.; Jiang, P.; He, X.; Bao, J.; Zhang, B.; Li, Y. The Current Situation and Future Trends of Ergothioneine in Biology and Medical Research: A Bibliometric Analysis. J. Multidiscip. Health 2025, 18, 6127–6149. [Google Scholar] [CrossRef]
- Gowda, G.A.N.; Zhu, W.; Pascua, V.; McMillen, T.; Tian, R.; Raftery, D. Identification and distribution of the dietary antioxidant ergothioneine in humans and animal models combining NMR, RANSY, and MS methods. Anal. Chem. 2025, 97, 19313–19320. [Google Scholar] [CrossRef]
- Tian, X.; Thorne, J.L.; Moore, J.B. Ergothioneine: An underrecognised dietary micronutrient required for healthy ageing? Br. J. Nutr. 2023, 129, 104–114. [Google Scholar] [CrossRef] [PubMed]
- Li, H.; Wang, X.; Li, R.; Chen, L.; Cheng, G.; Xiong, J. Synergistic neuroprotective effects of ergothioneine and lactoferrin in app/ps1 transgenic mice and mouse n2a cells. J. Agric. Food Chem. 2025, 73, 10186–10200. [Google Scholar] [CrossRef] [PubMed]
- Fu, T.-T.; Shen, L. Ergothioneine as a Natural Antioxidant Against Oxidative Stress-Related Diseases. Front. Pharmacol. 2022, 13, 850813. [Google Scholar] [CrossRef] [PubMed]
- Hartman, P.E. Ergothioneine as Antioxidant. Methods Enzymol. 1990, 186, 310–318. [Google Scholar] [CrossRef]
- Pang, L.; Wang, T.; Liao, Q.; Cheng, Y.; Wang, D.; Li, J.; Fu, C.; Zhang, C.; Zhang, J. Protective Role of Ergothioneine Isolated from Pleurotus ostreatus against Dextran Sulfate Sodium-Induced Ulcerative Colitis in a Rat Model. J. Food Sci. 2022, 87, 415–426. [Google Scholar] [CrossRef]
- Lei, Z.; Wang, Z.; Zhang, H.; Jia, Y.; Zhang, D.; Shi, R.; Wang, C.; Chen, W. Ergothioneine as a promising natural antioxidant: Bioactivities, therapeutic potential, and industrial applications. Food Funct. 2025, 16, 7473–7490. [Google Scholar] [CrossRef]
- Zhang, H.; Liu, Z.; Wang, Z.; Lei, Z.; Jia, Y.; Chen, W.; Shi, R.; Wang, C. A Review of Novel Antioxidant Ergothioneine: Biosynthesis Pathways, Production, Function and Food Applications. Foods 2025, 14, 1588. [Google Scholar] [CrossRef]
- Zhang, X.; Ma, H.; Guo, X.; Weng, Y.; Hu, X.; Ren, L. Cultivation and utilization of edible mushrooms: From extraction of active components to effective substrate utilization. J. Food Compos. Anal. 2025, 140, 107224. [Google Scholar] [CrossRef]
- Guo, J.; Zhang, M.; Fang, Z. Valorization of mushroom by-products: A review. J. Sci. Food Agric. 2022, 102, 5593–5605. [Google Scholar] [CrossRef] [PubMed]
- Zhang, C.; Xie, Y.; Liu, D.; Liu, R.; Han, J. Effects of drying process and high hydrostatic pressure on extraction of antioxidant ergothioneine from Pleurotus citrinopileatus singer. Foods 2024, 13, 878. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.-K. Natural products in cosmetics. Nat. Prod. Bioprospecting 2022, 12, 40. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, T.H.; Nagasaka, R.; Ohshima, T. Effects of extraction solvents, cooking procedures and storage conditions on the contents of ergothioneine and phenolic compounds and antioxidative capacity of the cultivated mushroom Flammulina velutipes. Int. J. Food Sci. Technol. 2012, 47, 1193–1205. [Google Scholar] [CrossRef]
- Ghahremani-Majd, H.; Mumivand, H.; Khanizadeh, P.; Bakhshipoor, F.; Argento, S. Optimizing Ergothioneine Biosynthesis and Antioxidant Activity in Agaricus spp. Through Amino Acid Supplementation and Yeast–Peptone Mixtures. Horticulturae 2025, 11, 348. [Google Scholar] [CrossRef]
- Chen, S.-Y.; Ho, K.-J.; Hsieh, Y.-J.; Wang, L.-T.; Mau, J.-L. Contents of lovastatin, γ-aminobutyric acid and ergothioneine in mushroom fruiting bodies and mycelia. LWT 2012, 47, 274–278. [Google Scholar] [CrossRef]
- Amaranthus, M. A Mushroom-Derived Compound That Could Change Your Life: Ergothioneine. Food Nutr. J. 2023, 8, 282. [Google Scholar] [CrossRef]
- Siti-Nuramira, J.; Farhana, R.; Nabil, S.; Jafari, S.M.; Raseetha, S. Impact of drying methods on the quality of grey (Pleurotus sajor caju) and pink (Pleurotus djamor) oyster mushrooms. J. Food Meas. Charact. 2022, 16, 3331–3343. [Google Scholar] [CrossRef]
- Villalobos-Pezos, M.; Muñoz-Fariña, O.; Ah-Hen, K.S.; Garrido-Figueroa, M.F.; García-Figueroa, O.; González-Esparza, A.; de Medina, L.G.-P.; Bastías-Montes, J.M. Optimization of Phenolic Content Extraction and Effects of Drying Treatments on Physicochemical Characteristics and Anti-oxidant Properties of Edible Mushroom Pleurotus ostreatus (Jacq.) P. Kumm (Oyster Mushroom). Antioxidants 2024, 13, 1581. [Google Scholar] [CrossRef]
- Pratti, P.G.; Junior, J.D.S.P.; de Lima Petito, N.; da Silva, B.D.; Conte-Junior, C.A.; Branco, V.N.C.; de Lima Araújo, K.G.; Domingues, J.R. Effect of Hot Air-Drying and Pasteurization on Ergothioneine Content in Edible Mushrooms. J. Food Compos. Anal. 2024, 126, 105865. [Google Scholar] [CrossRef]
- Podkowa, A.; Kryczyk-Poprawa, A.; Opoka, W.; Muszyńska, B. Culinary–medicinal mushrooms: A review of organic compounds and bioelements with antioxidant activity. Eur. Food Res. Technol. 2020, 247, 513–533. [Google Scholar] [CrossRef]
- Suzuki, K.; Kaneda, Y.; Izumo, T.; Nakao, Y.; Iidaka, T.; Horii, C.; Muraki, S.; Oka, H.; Kawaguchi, H.; Akune, T.; et al. The Association Between Serum Ergothioneine Concentration and Japanese Dietary Habits: The Third Survey of the ROAD Study. Nutrients 2025, 17, 517. [Google Scholar] [CrossRef] [PubMed]
- Barua, B.S.; Nigaki, A.; Kataoka, R. A New Recycling Method through Mushroom Cultivation Using Food Waste: Optimization of Mushroom Bed Medium Using Food Waste and Agricultural Use of Spent Mushroom Substrates. Recycling 2024, 9, 58. [Google Scholar] [CrossRef]
- Tsiantas, K.; Tsiaka, T.; Koutrotsios, G.; Siapi, E.; Zervakis, G.I.; Kalogeropoulos, N.; Zoumpoulakis, P. On the Identification and Quantification of Ergothioneine and Lovastatin in Various Mushroom Species: Assets and Challenges of Different Analytical Approaches. Molecules 2021, 26, 1832. [Google Scholar] [CrossRef]
- Lee, W.Y.; Park, E.-J.; Ahn, J.K.; Ka, K.-H. Ergothioneine contents in fruiting bodies and their enhancement in mycelial cultures by the addition of methionine. Mycobiology 2009, 37, 43–47. [Google Scholar] [CrossRef]
- Kim, M.-J.; Jeong, Y.; Park, M.-J.; Jang, Y.; Ka, K.-H. Effects of Blue Light on Fruiting Body Production and Ergothioneine Content during Sawdust Cultivation of Shiitake (Lentinula edodes). Mycobiology 2024, 52, 439–445. [Google Scholar] [CrossRef]
- Weng, Y.; Zhu, M.; Ma, H.; Hu, X.; Ren, L. Recent Progress in Mushroom-Derived Ergothioneine: Techniques and Applications. Food Biosci. 2024, 62, 105533. [Google Scholar] [CrossRef]
- Hu, D.-B.; Xue, R.; Zhuang, X.-C.; Zhang, X.-S.; Shi, S.-L. Ultrasound-Assisted Extraction Optimization of Polyphenols from Boletus bicolor and Evaluation of Its Antioxidant Activity. Front. Nutr. 2023, 10, 1135712. [Google Scholar] [CrossRef]
- Sirohi, R.; Negi, T.; Rawat, N.; Sagar, N.A.; Sindhu, R.; Tarafdar, A. Emerging Technologies for the Extraction of Bioactives from Mushroom Waste. J. Food Sci. Technol. 2024, 61, 1069–1082. [Google Scholar] [CrossRef]
- Parí, S.M.; Saldaña, E.; Rios-Mera, J.D.; Angulo, M.F.Q.; Huaman-Castilla, N.L. Emerging Technologies for Extracting Antioxidant Compounds from Edible and Medicinal Mushrooms: An Efficient and Sustainable Approach. Compounds 2025, 5, 29. [Google Scholar] [CrossRef]
- Yen, M.-T.; Chang, Y.-H.; Huang, S.-J.; Cheng, M.-C.; Mau, J.-L. Extraction of Ergothioneine from Pleurotus eryngii and P. citrinopileatus (Agaricomycetes) and Preparation of Its Product. Int. J. Med. Mushrooms 2018, 20, 381–392. [Google Scholar] [CrossRef] [PubMed]
- Wen, C.; Tang, J.; Cao, L.; Fan, M.; Lin, X.; Liu, G.; Liang, L.; Liu, X.; Zhang, J.; Li, Y.; et al. Strategic Approaches for Co-Encapsulation of Bioactive Compounds: Technological Advances and Mechanistic Insight. Foods 2025, 14, 2024. [Google Scholar] [CrossRef] [PubMed]
- Prandi, B.; Cigognini, I.M.; Faccini, A.; Zurlini, C.; Rodríguez, Ó.; Tedeschi, T. Comparative Study of Different Protein Extraction Technologies Applied on Mushroom By-Products. Food Bioprocess Technol. 2023, 16, 1570–1581. [Google Scholar] [CrossRef]
- Luo, J.; Chen, L. Status and development of spent mushroom substrate recycling: A review. J. Air Waste Manag. Assoc. 2024, 74, 843–860. [Google Scholar] [CrossRef]
- Sivakumar, D.; Bozzo, G. Mushroom Biomass Waste Is a Source of the Antioxidants Ergothioneine and Glutathione. Sustainability 2023, 15, 11961. [Google Scholar] [CrossRef]
- Muñoz-Tebar, N.; Pérez-Álvarez, J.A.; Fernández-López, J.; Viuda-Martos, M. Chitosan Edible Films and Coatings with Added Bioactive Compounds: Antibacterial and Antioxidant Properties and Their Application to Food Products: A Review. Polymers 2023, 15, 396. [Google Scholar] [CrossRef]
- Martín, C.; Zervakis, G.I.; Xiong, S.; Koutrotsios, G.; Strætkvern, K.O. Spent substrate from mushroom cultivation: Exploitation potential toward various applications and value-added products. Bioengineered 2023, 14, 2252138. [Google Scholar] [CrossRef]
- Leong, Y.K.; Ma, T.-W.; Chang, J.-S.; Yang, F.-C. Recent Advances and Future Directions on the Valorization of Spent Mushroom Substrate (SMS): A Review. Bioresour. Technol. 2022, 344, 126157. [Google Scholar] [CrossRef]
- Ravlikovsky, A.; Pinheiro, M.N.C.; Dinca, L.; Crisan, V.; Symochko, L. Valorization of Spent Mushroom Substrate: Establishing the Foundation for Waste-Free Production. Recycling 2024, 9, 44. [Google Scholar] [CrossRef]
- Tang, R.M.Y.; Cheah, I.K.-M.; Yew, T.S.K.; Halliwell, B. Distribution and accumulation of dietary ergothioneine and its metabolites in mouse tissues. Sci. Rep. 2018, 8, 1601. [Google Scholar] [CrossRef]
- Leow, D.M.-K.; Cheah, I.K.-M.; Chen, L.; Ng, Y.-K.; Yeo, C.J.-J.; Halliwell, B.; Ong, W.-Y. Ergothioneine-Mediated Neuroprotection of Human iPSC-Derived Dopaminergic Neurons. Antioxidants 2024, 13, 693. [Google Scholar] [CrossRef]
- Iglesias, H.; Ortiz, A.P.; Disla, J.M.S.; Lara-Guillén, A.J. Environmental and Economic Life Cycle Impacts of Using Spent Mushroom Substrate as a Soil Improver. Environments 2025, 12, 31. [Google Scholar] [CrossRef]




| Mushroom Species | Mushroom Part/Type | Ergothioneine Content (mg/kg) | Source |
|---|---|---|---|
| Pleurotus ostreatus (Oyster) | fruiting body | 11,800.00 | [25] |
| Pleurotus ostreatus (Oyster) | fruiting body | 118.91 | [8,21] |
| Pleurotus ostreatus (Oyster) | fruiting body | 2590.00 | [33] |
| Boletus edulis (King Bolete) | fruiting body | 528.14 | [8,21] |
| Boletus edulis (Porcini) | fruiting body | 7270.00 | [18,34] |
| Lentinula edodes (Shiitake) | fruiting body | 284.00 | [8] |
| Lentinula edodes (Shiitake) | fruiting body | 123 | [25] |
| Lentinula edodes (Shiitake) | fermented product | 1278.00–1775.00 | [25] |
| Flammulina velutipes (Enokitake) | fruiting body | 298.00 | [8] |
| Flammulina velutipes (Enokitake) | fruiting body | 151.00 | [25] |
| Cordyceps militaris | fruiting body | 382.00–799.00 | [25] |
| Cordyceps militaris | mycelium | 140.00 | [25] |
| Hericium erinaceus (Lion’s Mane) | mycelium | 376.20 | [8] |
| Agaricus bisporus (White Button) | fruiting body | 0.46 | [8] |
| Agaricus bisporus (White Button) | fruiting body | 210.00 | [35] |
| Agaricus bisporus (Brown) | fruiting body | 24.17 | [13,35] |
| Panus conchatus | submerged fermentation broth (crude extract) | 148.79 mg/L | [18] |
| Pleurotus pulmonarius | fruiting body (grown on sms) | 2170.00 | [18] |
| Agaricus bitorquis | mycelium (optimized conditions) | High | [36] |
| Armillaria mellea (Honey Mushroom) | mycelium | 219.60 | [37] |
| Coprinus comatus (Shaggy Mane) | mycelium | 399.00 | [38] |
| Coriolus versicolor (Turkey Tail) | mycelium | 13.00 | [38] |
| Ganoderma lucidum (Reishi) | mycelium | 16.50 | [38] |
| Pleurotus eryngii (King Oyster) | fruiting body | Fruiting Body | [25] |
| Pleurotus eryngii (King Oyster) | mycelium (regular) | Mycelium (regular) | [25] |
| Pleurotus eryngii (King Oyster) | mycelium (hi-ergo) | Mycelium (Hi-Ergo) | [25] |
| Pleurotus ostreatus (Oyster) | culls | Culls | [21,33] |
| Lentinula edodes (Shiitake) | culls | Culls | [32,33] |
| Agaricus bisporus (White Button) | stem waste | Stem Waste | [33] |
| Mushroom Species | Drying Method | Result on EGT | Source |
|---|---|---|---|
| Pleurotus ostreatus (oyster mushroom) | HAD (40 °C) vs. FD | Highest EGT retention among tested methods. | [40] |
| Pleurotus citrinopileatus (golden oyster mushroom) | ND (natural ventilation), FD, HD | EGT increased to 4.03 ± 0.01 mg/g d.w., higher than FD and HD. | [33] |
| Cyttaria espinosae (wild “digüeñe” mushroom) | FD, HAD | EGT not detected; negligible retention under HAD. | [40] |
| Various species (Auricularia spp., Pleurotus spp.) | Review of HAD, solar and microwave drying | EGT content is largely unchanged under moderate HAD. | [39] |
| Lentinula edodes (shiitake) | HAD + pasteurization | EGT decreased from 1.02 ± 0.07 to 0.58 ± 0.04 mg/g d.w., indicating heat sensitivity. | [41] |
| Pleurotus ostreatus (oyster mushroom) | HAD (40 °C) vs. FD | Highest EGT retention among tested methods. | [40] |
| Factor | Experimental Description/Observation | Effect on EGT Content | Representative Species/Model | Source |
|---|---|---|---|---|
| Species and strain | Comparative analysis of multiple mushroom species under identical growth conditions. | Significant interspecific variation in EGT concentration, reflecting species-specific biosynthetic capacity. | Agaricus bisporus, Pleurotus ostreatus, Lentinula edodes | [43] |
| Substrate composition | Use of food-waste–based substrate versus conventional sawdust medium. | EGT content nearly doubled in mushrooms cultivated on food-waste substrate. | Pleurotus ostreatus | [44] |
| Amino acid supplementation | Addition of histidine, methionine, cysteine, yeast extract, and peptone to growth media. | Marked enhancement of EGT biosynthesis; excessive (>0.5%) precursor concentration reduced growth rate. | Agaricus spp. | [47] |
| Light quality | Exposure to blue LED light (~450 nm) during fruiting body formation. | Increased EGT from 1.2 mg/g to ~2.8 mg/g dry weight; elevated antioxidant activity. | Lentinula edodes, Pleurotus ostreatus | [44,47] |
| Oxidative stress induction | Cultivation under elevated oxygen or late fruiting flush (3rd harvest). | Elevated EGT levels associated with oxidative stress response. | Agaricus bisporus, Panus conchatus | [18] |
| Drying and processing conditions | Comparison of HAD, FD, ND, and pasteurization treatments. | EGT retention pattern: FD ≥ Fresh ≥ HAD; HAD + pasteurization caused significant losses. | Pleurotus spp., Lentinula edodes | [39,41] |
| Method Type | Principle | Mushroom Source | Reported Yield/Purity | Advantages/Disadvantages | Source |
|---|---|---|---|---|---|
| Hot Water Extraction | Mycelial suspension in 1:40 water, 100 °C for 5 min (no stirring). | Pleurotus ostreatus mycelium | 1700 mL EGT aqueous solution (from 50 g mycelium) | Simple, low-cost, exploits thermal stability of EGT; limited selectivity. | [48,50] |
| Hot Water Extraction | Cells resuspended in water, 95 °C for 1 h. | Panus conchatus fermentation broth | Crude extract obtained | Efficient for crude extracts; maintains antioxidant integrity; may co-extract impurities. | [48,50] |
| Solvent Extraction (Ethanol) | Pulverized mushroom sonicated 30 min in ethanol and filtered. | Agaricus bisporus, Lentinula edodes, Pleurotus ostreatus | Better analytical signal than water extraction. | Effective for EGT + phenolics; easy lab-scale recovery; solvent removal required. | [49,50] |
| Solvent Extraction (70% Ethanol) | Extraction using 70% ethanol (v/v). | Flammulina velutipes | 2.05 ± 0.18 mg g−1 DW (EGT). | Maximizes recovery of polar + semi-polar metabolites; green-solvent alternative. | [48,50] |
| High-Hydrostatic-Pressure Extraction (HHPE) | 250 MPa, 52 min, distilled water (1:10 liquid–solid ratio). | Pleurotus citrinopileatus | 4.03 ± 0.01 mg g−1 DW (highest EGT content). | Disrupts tissues effectively, enhances mass transfer; high energy demand. | [48,50] |
| Ultrasound-Assisted Extraction (UAE) | Sonication of powdered biomass (20–40 kHz) to enhance solvent penetration. | Boletus bicolor (model species) | Polyphenols ↑; EGT retention confirmed. | Accelerated diffusion, mild temperature, scalable green method. | [49,50] |
| Submerged Fermentation | Liquid culture with molasses, soy peptone, and amino-acid precursors (His, Met, Cys). | Panus conchatus | 148.79 mg L−1 (highest EGT concentration). | High yield, low cost, easily scalable; downstream purification required. | [49,50] |
| Ultrafiltration (UF) | Hollow-fiber membrane (4 kDa or 6 kDa cut-off) for crude filtrate fractionation. | Mycelial fermentation liquid | 31.6% purity (4 kDa membrane). | Simple, low-energy separation; limited resolution for small molecules. | [48] |
| Ion-Exchange Chromatography (Specialized Resin) | LX-1880 uniform-particle resin under SSMB system. | EGT-rich fermentation liquids | >95% desalination, >98% EGT recovery. | High selectivity for charged species; industrial scalability; resin cost. | [48] |
| Traditional Chromatography | Column chromatography, TLC, ion-exchange, and paper electrophoresis. | Colibus genus, Agrocybe aegerita (tea tree mushroom) | High purity fractions obtained. | Robust, well-established; labor-intensive, multi-step. | [48] |
| HPLC (ANP Mode) | Cogent Diamond Hydride column, DI water/acetonitrile + 0.1% formic acid; UV 254 nm. | A. bisporus, L. edodes, P. ostreatus | 96.5–100.3% recovery (R2 = 0.99999). | Rapid, reproducible quantification of EGT in complex matrices. | [48] |
| UPLC–MS/MS (HILIC) | Methanolic extract analysed by HILIC–triple quadrupole MS. | Cosmetics samples (applicable to mushroom extracts) | LOD 25–50 μg kg−1; R2 > 0.999. | High sensitivity and specificity for EGT; cost-intensive instrumentation. | [48] |
| Region | Regulatory Category | Status for EGT | Key Document |
|---|---|---|---|
| European Union | Novel Food ingredient | Synthetic L-ergothioneine is authorised as a Novel Food ingredient under Regulation (EC) No 258/97 and later under Regulation (EU) 2015/2283. | Commission Implementing Decision (EU) 2017/1281—Authorising placing on the market of synthetic L-ergothioneine as a Novel Food ingredient. EUR-Lex |
| United States | GRAS (Generally Recognised As Safe) | EGT notified to the U.S. FDA as GRAS for specified food uses. | FDA GRAS Notice Response Letter (GRN 1191) for ergothioneine. U.S. Food and Drug Administration |
| Asia (examples) | Novel Food/Import Ingredient Control | In Asian markets (e.g., China), EGT is regulated as an import-controlled functional ingredient; specific national standards vary. | “Ergothioneine’s Regulatory Status in China.” Zmuni Industry News |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Harasym, J.; Tiupova, A.; Pejcz, E. Ergothioneine: An Antioxidative, Neuroprotective and Anti-Inflammatory Compound from Mushroom Residuals. Molecules 2025, 30, 4621. https://doi.org/10.3390/molecules30234621
Harasym J, Tiupova A, Pejcz E. Ergothioneine: An Antioxidative, Neuroprotective and Anti-Inflammatory Compound from Mushroom Residuals. Molecules. 2025; 30(23):4621. https://doi.org/10.3390/molecules30234621
Chicago/Turabian StyleHarasym, Joanna, Alona Tiupova, and Ewa Pejcz. 2025. "Ergothioneine: An Antioxidative, Neuroprotective and Anti-Inflammatory Compound from Mushroom Residuals" Molecules 30, no. 23: 4621. https://doi.org/10.3390/molecules30234621
APA StyleHarasym, J., Tiupova, A., & Pejcz, E. (2025). Ergothioneine: An Antioxidative, Neuroprotective and Anti-Inflammatory Compound from Mushroom Residuals. Molecules, 30(23), 4621. https://doi.org/10.3390/molecules30234621