Polyphenols as Potential β-Lactamase Inhibitors: An Integrated Computational and Experimental Study
Abstract
1. Introduction
2. Results
2.1. Computational Study
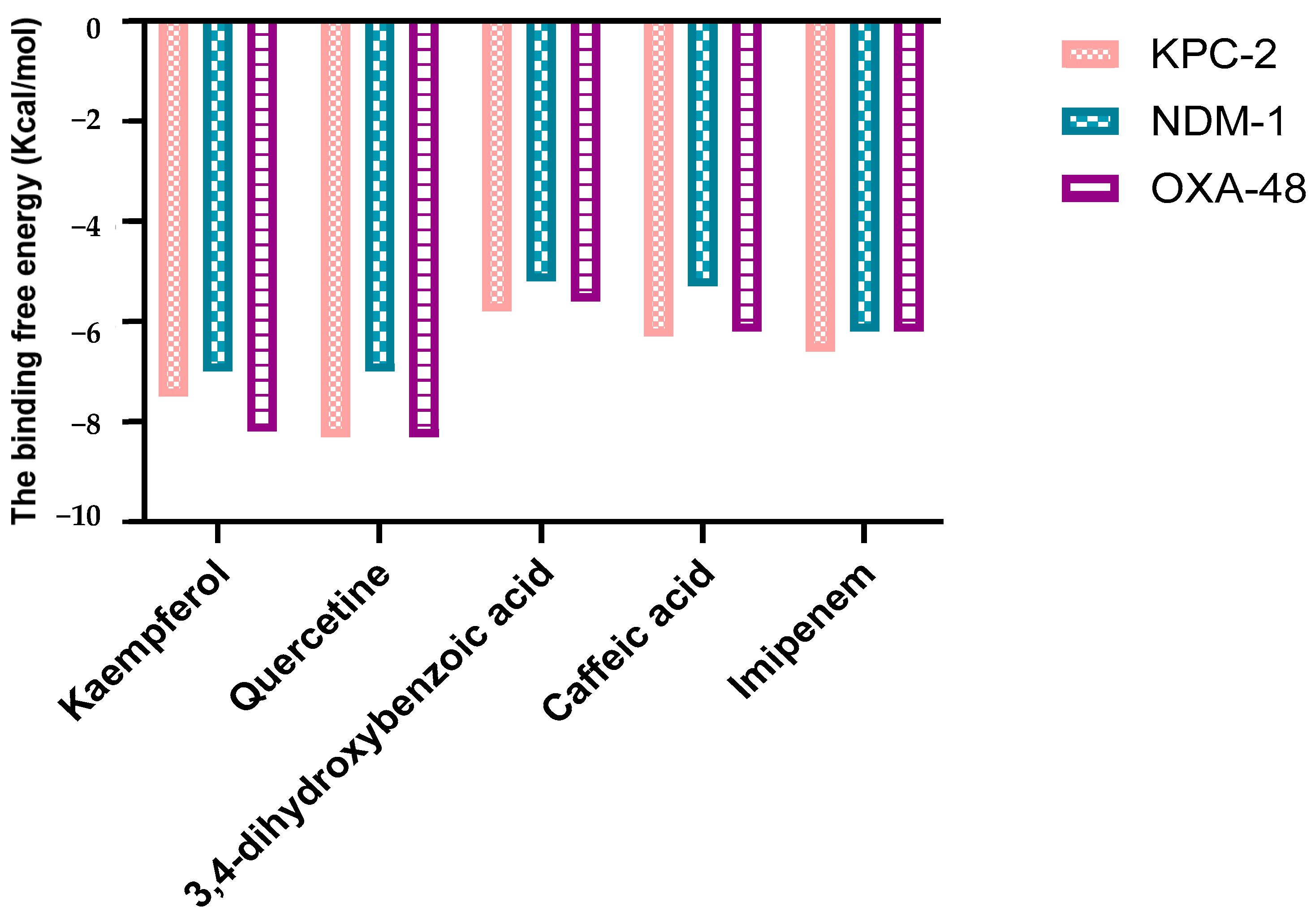
2.1.1. Molecular Docking Analysis
2.1.2. Molecular Dynamics Simulations
The Radius of Gyration Trajectories
Hydrogen Bond Number Analysis
RMSD
RMSF Profiles of KPC-2 and NDM-1, OXA-48-like Proteins for Each Polyphenol
2.1.3. Drug Likeness, ADMET Screening, and Toxicity Prediction
2.2. In Vitro Validation
2.2.1. Effects of the Four Polyphenols on the Bacterial Cell Viability with AlamarBlue Assay
Minimum Bactericidal Concentration (MBC) Determination
Checkerboard Assay
Polyphenols Inhibited β-Lactamase Activity
3. Discussion
4. Materials and Methods
4.1. Exploring Computational Methods and Predictions
4.1.1. Molecular Docking of Carbapenemase Inhibitors
Receptor Preparation, Active Site Prediction, and Grid Generation
Compound Preparation
Molecular Docking Simulations
4.1.2. Molecular Dynamics Simulations Using Gromacs
4.1.3. Drug Likeness, ADMET, and Toxicity Prediction
4.2. Experimental Confirmation and Biological Evaluation
4.2.1. Polyphenols and Antibiotic
4.2.2. Bacterial Strains
4.2.3. Assessment of the Antimicrobial Activity Using the AlamarBlue Assay
4.2.4. Minimum Bactericidal Concentration (MBC Determination)
4.2.5. Checkerboard Assay
4.2.6. Determination of % of β-Lactamase Inhibition
4.2.7. Statistical Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Bibi, Z.; Asghar, I.; Ashraf, N.M.; Zeb, I.; Rashid, U.; Hamid, A.; Ali, M.K.; Hatamleh, A.A.; Al-Dosary, M.A.; Ahmad, R.; et al. Prediction of Phytochemicals for Their Potential to Inhibit New Delhi Metallo β-Lactamase (NDM-1). Pharmaceuticals 2023, 16, 1404. [Google Scholar] [CrossRef]
- Abdel-Halim, M.S.; Askoura, M.; Mansour, B.; Yahya, G.; El-Ganiny, A.M. In Vitro Activity of Celastrol in Combination with Thymol against Carbapenem-Resistant Klebsiella pneumoniae Isolates. J. Antibiot. 2022, 75, 679–690. [Google Scholar] [CrossRef]
- Mourabiti, F.; Jouga, F.; Sakoui, S.; El Hosayny, O.; Zouheir, Y.; Soukri, A.; El Khalfi, B. Mechanisms, Therapeutic Strategies, and Emerging Therapeutic Alternatives for Carbapenem Resistance in Gram-Negative Bacteria. Arch. Microbiol. 2025, 207, 58. [Google Scholar] [CrossRef] [PubMed]
- Guo, Y.; Liu, H.; Yang, M.; Ding, R.; Gao, Y.; Niu, X.; Deng, X.; Wang, J.; Feng, H.; Qiu, J. Novel Metallo-β-Lactamases Inhibitors Restore the Susceptibility of Carbapenems to New Delhi Metallo-Lactamase-1 (NDM-1)-Harbouring Bacteria. Br. J. Pharmacol. 2024, 181, 54–69. [Google Scholar] [CrossRef] [PubMed]
- Terbtothakun, P.; Nwabor, O.F.; Siriyong, T.; Voravuthikunchai, S.P.; Chusri, S. Synergistic Antibacterial Effects of Meropenem in Combination with Aminoglycosides against Carbapenem-Resistant Escherichia coli Harboring BlaNDM-1 and BlaNDM-5. Antibiotics 2021, 10, 1023. [Google Scholar] [CrossRef]
- Aydemir, Ö.; Ormanoğlu, G.; Ayhancı, T.; Zengin, M.; Köroğlu, M. Investigation of in Vitro Efficacy of Quercetin-Meropenem Combination in Carbapenemase-Producing Klebsiella pneumoniae Isolates. J. Infect. Dev. Ctries. 2023, 17, 1325–1329. [Google Scholar] [CrossRef]
- Kuloglu, T.O.; Unuvar, G.K.; Cevahir, F.; Kilic, A.U.; Alp, E. Risk Factors and Mortality Rates of Carbapenem-Resistant Gram-Negative Bacterial Infections in Intensive Care Units. J. Intensive Med. 2024, 4, 347–354. [Google Scholar] [CrossRef]
- Ma, J.; Song, X.; Li, M.; Yu, Z.; Cheng, W.; Yu, Z.; Zhang, W.; Zhang, Y.; Shen, A.; Sun, H.; et al. Global Spread of Carbapenem-Resistant Enterobacteriaceae: Epidemiological Features, Resistance Mechanisms, Detection and Therapy. Microbiol. Res. 2023, 266, 127249. [Google Scholar] [CrossRef] [PubMed]
- Etminani, F.; Etminani, A.; Hasson, S.O.; Judi, H.K.; Akter, S.; Saki, M. In Silico Study of Inhibition Effects of Phytocompounds from Four Medicinal Plants against the Staphylococcus Aureus β-Lactamase. Inform. Med. Unlocked 2023, 37, 101186. [Google Scholar] [CrossRef]
- Tabti, K.; Abdessadak, O.; Sbai, A.; Maghat, H.; Bouachrine, M.; Lakhlifi, T. Design and Development of Novel Spiro-Oxindoles as Potent Antiproliferative Agents Using Quantitative Structure Activity Based Monte Carlo Method, Docking Molecular, Molecular Dynamics, Free Energy Calculations, and Pharmacokinetics /Toxicity Studies. J. Mol. Struct. 2023, 1284, 135404. [Google Scholar] [CrossRef]
- Daina, A.; Michielin, O.; Zoete, V. SwissADME: A Free Web Tool to Evaluate Pharmacokinetics, Drug-Likeness and Medicinal Chemistry Friendliness of Small Molecules. Sci. Rep. 2017, 7, 42717. [Google Scholar] [CrossRef]
- Mohammed, F.S.; Sevindik, M.; Uysal, İ.; Sabik, A.E. Quercetin: Derivatives, Biosynthesis, Biological Activity, Pharmacological and Therapeutic Effects. Prospect. Pharm. Sci. 2023, 21, 49–56. [Google Scholar] [CrossRef]
- Zhang, Y.; Chen, C.; Cheng, B.; Gao, L.; Qin, C.; Zhang, L.; Zhang, X.; Wang, J.; Wan, Y. Discovery of Quercetin and Its Analogs as Potent OXA-48 Beta-Lactamase Inhibitors. Front. Pharmacol. 2022, 13, 926104. [Google Scholar] [CrossRef]
- Pal, A.; Tripathi, A. Quercetin Potentiates Meropenem Activity among Pathogenic Carbapenem-Resistant Pseudomonas aeruginosa and Acinetobacter baumannii. J. Appl. Microbiol. 2019, 127, 1038–1047. [Google Scholar] [CrossRef] [PubMed]
- Vipin, C.; Saptami, K.; Fida, F.; Mujeeburahiman, M.; Rao, S.S.; Athmika; Arun, A.B.; Rekha, P.D. Potential Synergistic Activity of Quercetin with Antibiotics against Multidrug-Resistant Clinical Strains of Pseudomonas aeruginosa. PLoS ONE 2020, 15, e0241304. [Google Scholar] [CrossRef] [PubMed]
- Yang, Y.; Song, J.; Gao, Y.; Li, D.; Li, X.; Liu, Y. The Inhibition Mechanism of Quercetin Targeting β-Lactamases OXA-98 Based on Molecular Dynamics Simulation. Chem. Phys. Lett. 2023, 825, 140596. [Google Scholar] [CrossRef]
- Periferakis, A.; Periferakis, K.; Badarau, I.A.; Petran, E.M.; Popa, D.C.; Caruntu, A.; Costache, R.S.; Scheau, C.; Caruntu, C.; Costache, D.O. Kaempferol: Antimicrobial Properties, Sources, Clinical, and Traditional Applications. Int. J. Mol. Sci. 2022, 23, 15054. [Google Scholar] [CrossRef]
- Zhou, H.; Xu, M.; Guo, W.; Yao, Z.; Du, X.; Chen, L.; Sun, Y.; Shi, S.; Cao, J.; Zhou, T. The Antibacterial Activity of Kaempferol Combined with Colistin against Colistin-Resistant Gram-Negative Bacteria. Microbiol. Spectr. 2022, 10, e0226522. [Google Scholar] [CrossRef] [PubMed]
- Araújo, M.O.; Freire Pessoa, H.L.; Lira, A.B.; Castillo, Y.P.; De Sousa, D.P. Synthesis, Antibacterial Evaluation, and QSAR of Caffeic Acid Derivatives. J. Chem. 2019, 2019, 3408315. [Google Scholar] [CrossRef]
- Kauffmann, A.C.; Castro, V.S. Phenolic Compounds in Bacterial Inactivation: A Perspective from Brazil. Antibiotics 2023, 12, 645. [Google Scholar] [CrossRef]
- Xu, T.; Zhu, H.; Liu, R.; Wu, X.; Chang, G.; Yang, Y.; Yang, Z. The Protective Role of Caffeic Acid on Bovine Mammary Epithelial Cells and the Inhibition of Growth and Biofilm Formation of Gram-Negative Bacteria Isolated from Clinical Mastitis Milk. Front. Immunol. 2022, 13, 1005430. [Google Scholar] [CrossRef]
- Rohatgi, A.; Gupta, P. Benzoic Acid Derivatives as Potent Antibiofilm Agents against Klebsiella pneumoniae Biofilm. J. Biosci. Bioeng. 2023, 136, 190–197. [Google Scholar] [CrossRef] [PubMed]
- Yuan, J.J.; Yan, H.J.; He, J.; Liu, Y.Y. Antibacterial Activities of Polyphenols from Olive Leaves against Klebsiella pneumoniae. IOP Conf. Ser. Earth Environ. Sci. 2021, 680, 012060. [Google Scholar] [CrossRef]
- Farooq, A.; Drotleff, B.; Kroemer, N.; Han, M.L.; Li, J.; Decousser, J.W.; Schrey, D.; Buyck, J.; Grégoire, N.; Nordmann, P.; et al. Evaluation of in Vitro Pharmacodynamic Drug Interactions of Ceftazidime-Avibactam with Tigecycline in ESBL- and Carbapenemase Producing Escherichia coli and Klebsiella pneumoniae. Int. J. Antimicrob. Agents 2025, 65, 107457. [Google Scholar] [CrossRef]
- Forero-Doria, O.; Parra-Cid, C.; Venturini, W.; Espinoza, C.; Araya-Maturana, R.; Valenzuela-Riffo, F.; Saldias, C.; Leiva, A.; Duarte, Y.; Echeverría, J.; et al. Novel N-Benzoylimidazolium Ionic Liquids Derived from Benzoic and Hydroxybenzoic Acids as Therapeutic Alternative against Biofilm-Forming Bacteria in Skin and Soft-Tissue Infections. Bioorg. Chem. 2022, 126, 105914. [Google Scholar] [CrossRef] [PubMed]
- Pal, A.; Tripathi, A. Demonstration of Bactericidal and Synergistic Activity of Quercetin with Meropenem among Pathogenic Carbapenem Resistant Escherichia coli and Klebsiella pneumoniae. Microb. Pathog. 2020, 143, 104120. [Google Scholar] [CrossRef]
- Li, P.C.; Tong, Y.C.; Xiao, X.L.; Fan, Y.P.; Ma, W.R.; Liu, Y.Q.; Zhuang, S.; Qing, S.Z.; Zhang, W.M. Kaempferol Restores the Susceptibility of ESBLs Escherichia coli to Ceftiofur. Front. Microbiol. 2024, 15, 1474919. [Google Scholar] [CrossRef]
- Berendsen, H.J.C.; Van Der Spoel, D.; Van Drunen, R. GROMACS: A Message-Passing Parallel Molecular Dynamics Implementation. Comput. Phys. Commun. 1995, 91, 43–56. [Google Scholar] [CrossRef]
- Van Der Spoel, D.; Lindahl, E.; Hess, B.; Groenhof, G.; Mark, A.E.; Berendsen, H.J.C. GROMACS: Fast, Flexible, and Free. J. Comput. Chem. 2005, 26, 1701–1718. [Google Scholar] [CrossRef]
- Lipinski, C.A.; Dominy, B.W.; Feeney, P.J. Drug Delivery Reviews Experimental and Computational Approaches to Estimate Solubility and Permeability in Drug Discovery and Development Settings. Adv. Drug Deliv. Rev. 1997, 23, 3–25. [Google Scholar] [CrossRef]
- Mourabiti, F.; Jouga, F.; Mouzoun, Y.; Arslan, S.; Schena, R.; Zouheir, Y.; Soukri, A.; De Martino, L.; Nocera, F.P.; El Khalfi, B. Phenotypic and genotypic characterization of carbapenem encoding genes among carbapenem-resistant Gram-negative bacteria isolated from North Casablanca, Morocco. Comp. Immunol. Microbiol. Infect. Dis. 2025, 123, 102399. [Google Scholar]
- Lewis, J.S., II; Amy Mathers, F.J.; April Bobenchik, D.M.; Alexandra Lynn Bryson, D.; Shelley Campeau, D.; Sharon Cullen, D.K.; Tanis Dingle, R.; German Esparza, F.; Humphries, R.M.; Thomas Kirn, F.J.; et al. CLSI M100-Ed35 January 2025 Replaces CLSI M100-Ed34 Performance Standards for Antimicrobial Susceptibility Testing; CLSI: Wayne, PA, USA, 2021; ISBN 9781684402625. [Google Scholar]
- Calvo, L.G.; Villarino, R.A.; Rama, J.L.R.; Abril, A.G.; de Miguel, T. A Modification of the Resazurin Cell Viability Assay, Suitable for the Quantification of Lactic Acid Producing Bacteria. LWT 2025, 215, 117259. [Google Scholar] [CrossRef]
- Mourabiti, F.; Derdak, R.; El Amrani, A.; Momen, G.; Timinouni, M.; Soukri, A.; El Khalfi, B.; Zouheir, Y. The Antimicrobial Effectiveness of Rosmarinus officinalis, Lavandula angustifolia, and Salvia officinalis Essential Oils against Klebsiella pneumoniae and Pseudomonas aeruginosa in Vitro and in Silico. S. Afr. J. Bot. 2024, 168, 112–123. [Google Scholar] [CrossRef]
- Güran, M.; Şanlıtürk, G.; Kerküklü, N.R.; Altundağ, E.M.; Süha Yalçın, A. Combined Effects of Quercetin and Curcumin on Anti-Inflammatory and Antimicrobial Parameters in Vitro. Eur. J. Pharmacol. 2019, 859, 172486. [Google Scholar] [CrossRef] [PubMed]













| Polyphenols | MW (g/mol) | LogP | H-Bond Acceptors | H-Bond Donors | Lipinski Rule | TPSA (Å2) | Solubility | Bioavailability Score |
|---|---|---|---|---|---|---|---|---|
| Kaempferol | 286.24 | 2.28 | 6 | 4 | Yes | 111.13 | Pass | 0.55 |
| Quercetin | 302.24 | 1.99 | 7 | 5 | Yes | 131.36 | Pass | 0.55 |
| Caffeic acid | 180.16 | 1.09 | 4 | 3 | Yes | 77.76 | Pass | 0.56 |
| 3,4-Dihydroxybenzoic acid | 154.12 | 0.80 | 4 | 3 | Yes | 77.76 | Pass | 0.56 |
| Polyphenols | LD50 mg/kg | Toxicity Class |
|---|---|---|
| Kaempferol | 3919 | 5 |
| Quercetin | 159 | 3 |
| Caffeic acid | 2980 | 5 |
| 3,4-Dihydroxybenzoic acid | 2000 | 4 |
| Caffeic Acid | Quercetin | Kaempferol | 3,4-Dihydroxybenzoic Acid | |
|---|---|---|---|---|
| E. coli ATCC25922 | 12.5 | 12.5 | 25 | 25 |
| E. coli R | 25 | 25 | 25 | 25 |
| K. pneumoniae ATCC13883 | 25 | 25 | 25 | 25 |
| K. pneumoniae R | 25 | 25 | 25 | 25 |
| P. aeruginosa ATCC27853 | 12.5 | 25 | 25 | 12.5 |
| P. aeruginosa R | 12.5 | 25 | 25 | 25 |
| Ctx/Q | Ctx/CA | Ctx/K | Ctx/BA | |||||
|---|---|---|---|---|---|---|---|---|
| FICI | Effect | FICI | Effect | FICI | Effect | FICI | Effect | |
| KPR | 0.5283 | additive | 0.7267 | additive | 1.0667 | additive | 0.7078 | additive |
| PAR | 0.2784 | synergistic | 0.8667 | additive | 0.2584 | synergistic | 0.78 | additive |
| ECR | 0.2351 | synergistic | 0.52 | additive | 0.52 | additive | 0.8578 | additive |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mourabiti, F.; Jouga, F.; Calvo, L.G.; Villarino, R.-A.; Zouheir, Y.; Soukri, A.; de Miguel, T.; El Khalfi, B. Polyphenols as Potential β-Lactamase Inhibitors: An Integrated Computational and Experimental Study. Molecules 2025, 30, 4416. https://doi.org/10.3390/molecules30224416
Mourabiti F, Jouga F, Calvo LG, Villarino R-A, Zouheir Y, Soukri A, de Miguel T, El Khalfi B. Polyphenols as Potential β-Lactamase Inhibitors: An Integrated Computational and Experimental Study. Molecules. 2025; 30(22):4416. https://doi.org/10.3390/molecules30224416
Chicago/Turabian StyleMourabiti, Fatima, Fatimazahra Jouga, Lorena G. Calvo, Rosa-Antía Villarino, Yassine Zouheir, Abdelaziz Soukri, Trinidad de Miguel, and Bouchra El Khalfi. 2025. "Polyphenols as Potential β-Lactamase Inhibitors: An Integrated Computational and Experimental Study" Molecules 30, no. 22: 4416. https://doi.org/10.3390/molecules30224416
APA StyleMourabiti, F., Jouga, F., Calvo, L. G., Villarino, R.-A., Zouheir, Y., Soukri, A., de Miguel, T., & El Khalfi, B. (2025). Polyphenols as Potential β-Lactamase Inhibitors: An Integrated Computational and Experimental Study. Molecules, 30(22), 4416. https://doi.org/10.3390/molecules30224416







