Mitochondrial-Targeted Triphenylphosphonium-Conjugated Ionophores with Enhanced Cytotoxicity in Cancer Cells
Abstract
1. Introduction
2. Results and Discussion
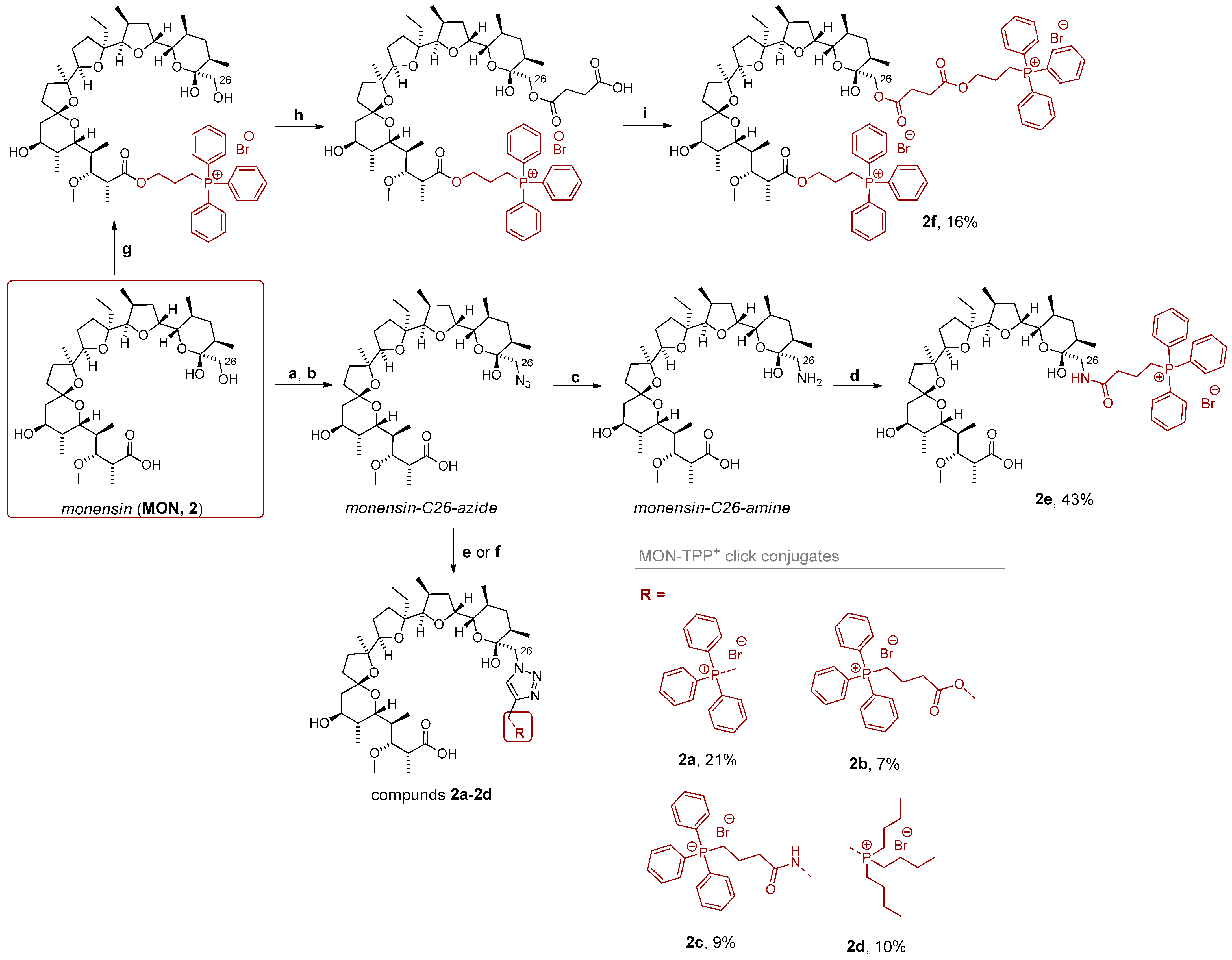
2.1. Analogs Design and Synthesis
2.2. Biological Activity
2.2.1. Cytotoxic Activity Studies
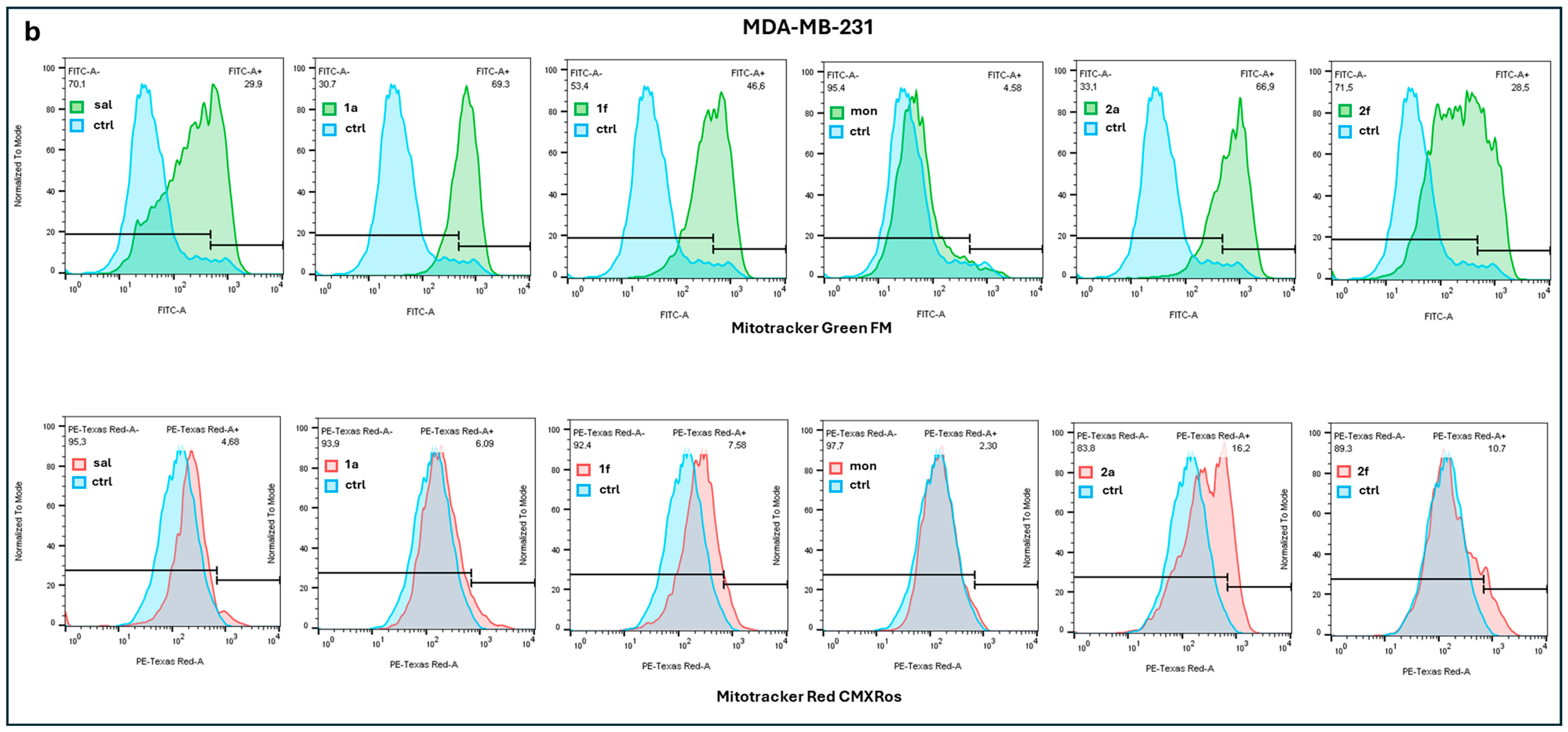
2.2.2. Mitochondria Activity
2.2.3. Mitochondrial ROS Production
2.3. Biophysical Studies
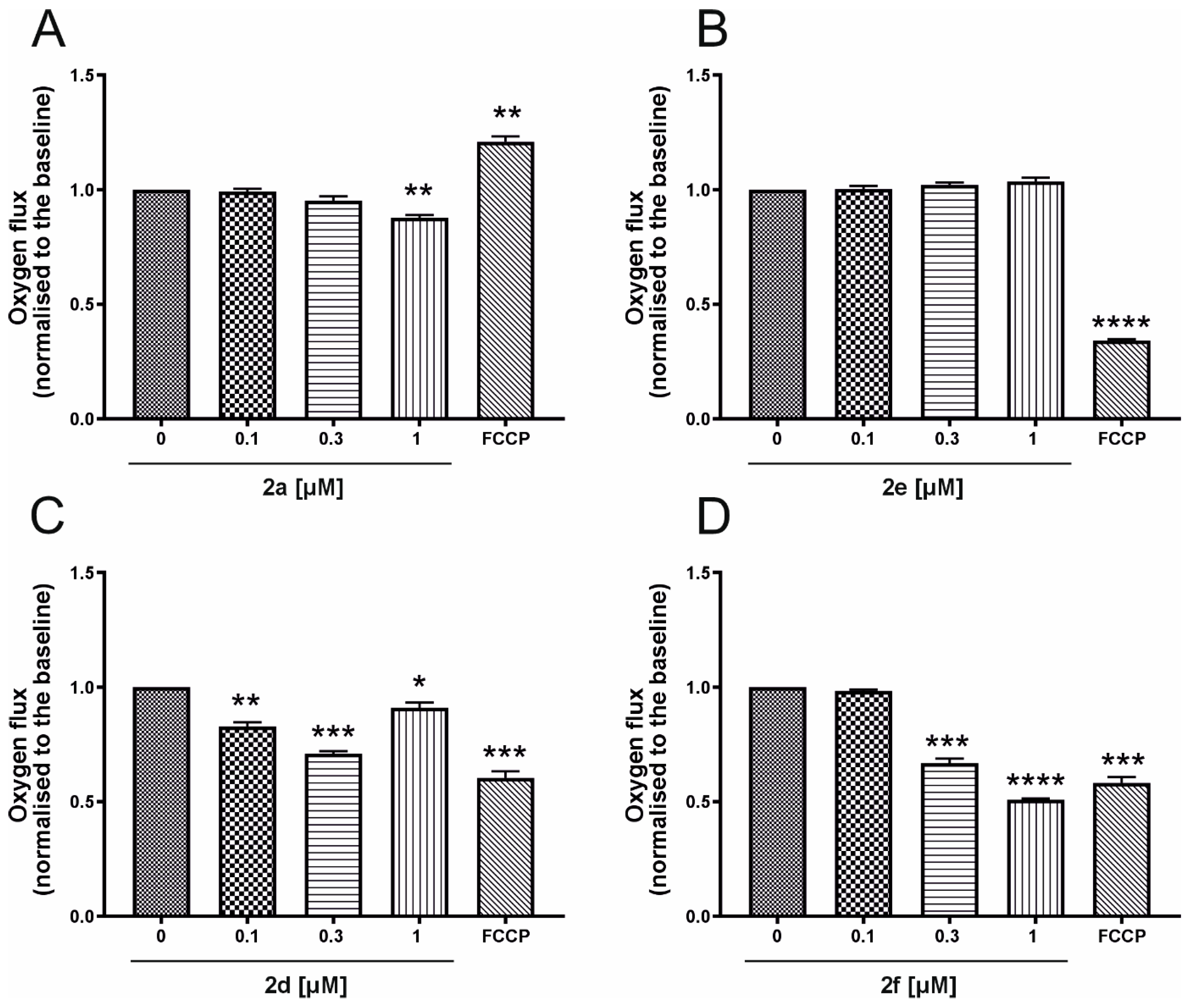
2.3.1. Mitochondrial Respiration Rate
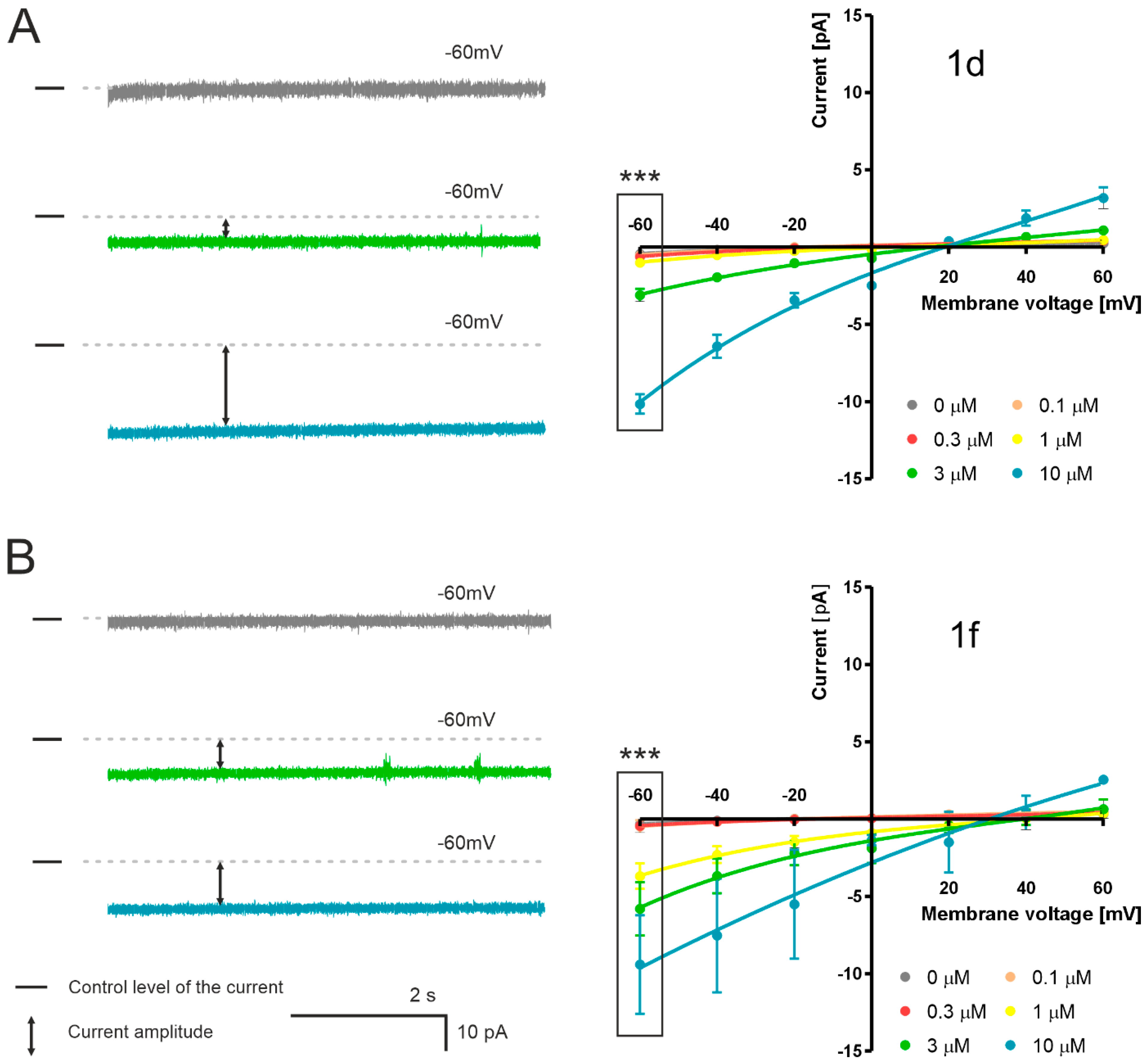
2.3.2. Interaction of Salinomycin Derivatives with Lipid Membranes
3. Materials and Methods
3.1. General Procedures
3.2. Isolation of Salinomycin and Monensin
3.3. Synthesis of (3-Carboxypropyl)triphenylphosphonium Bromide Propargyl Ester (Compound 3)
3.4. Synthesis of (3-Carboxypropyl)triphenylphosphonium Bromide Propargyl Amide (Compound 4)
3.5. Synthesis of Triphenylpropargylphosphonium Bromide (Compound 5)
3.6. Synthesis of C20-Azide of Salinomycin
3.7. General Procedure for Preparation of “Click”-Type Phosphonium Salts of Salinomycin (Analogs 1a–1d)
3.8. Synthesis of C20-Amine of Salinomycin
3.9. Synthesis of Amide-Type Phosphonium Salt of Salinomycin (Compound 1e)
3.10. Synthesis of Double Phosphonium Salt of Salinomycin (Compound 1f)
3.11. Synthesis of C26-Azide of Monensin
3.12. General Procedure for Preparation of “Click”-Type Phosphonium Salts of Monensin (Analogs 2a and 2c)
3.13. General Procedure for Preparation of “Click”-Type Phosphonium Salts of Monensin (Analogs 2b and 2d)
3.14. Synthesis of C26-Amine of Monensin
3.15. Synthesis of Amide-Type Phosphonium Salt of Momensin (Compound 2e)
3.16. Synthesis of Double Phosphonium Salt of Monensin (Compound 2f)
3.17. In Vitro Biological Studies
3.17.1. Culture Cell Lines
3.17.2. MTT Assay
3.17.3. Mitochondrial Activity
3.17.4. Mitochondrial ROS Generation
3.18. Biophysical Studies
3.18.1. Cell Culture
3.18.2. High-Resolution Respirometry
3.18.3. Black Lipid Membrane Technique (BLM)
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Liu, Y.; Sun, Y.; Guo, Y.; Shi, X.; Chen, X.; Feng, W.; Wu, L.-L.; Zhang, J.; Yu, S.; Wang, Y.; et al. An Overview: The Diversified Role of Mitochondria in Cancer Metabolism. Int. J. Biol. Sci. 2023, 19, 897–915. [Google Scholar] [CrossRef] [PubMed]
- Wen, S.; Zhu, D.; Huang, P. Targeting Cancer Cell Mitochondria as a Therapeutic Approach. Future Med. Chem. 2013, 5, 53–67. [Google Scholar] [CrossRef]
- Battogtokh, G.; Choi, Y.S.; Kang, D.S.; Park, S.J.; Shim, M.S.; Huh, K.M.; Cho, Y.-Y.; Lee, J.Y.; Lee, H.S.; Kang, H.C. Mitochondria-Targeting Drug Conjugates for Cytotoxic, Anti-Oxidizing and Sensing Purposes: Current Strategies and Future Perspectives. Acta Pharm. Sin. B 2018, 8, 862–880. [Google Scholar] [CrossRef]
- Pourahmad, J.; Salimi, A.; Seydi, E. Mitochondrial Targeting for Drug Development. In Toxicology Studies—Cells, Drugs and Environment; InTech: London, UK, 2015. [Google Scholar]
- Global Cancer Observatory. Available online: https://gco.iarc.fr/ (accessed on 14 October 2025).
- Jeena, M.T.; Kim, S.; Jin, S.; Ryu, J.-H. Recent Progress in Mitochondria-Targeted Drug and Drug-Free Agents for Cancer Therapy. Cancers 2019, 12, 4. [Google Scholar] [CrossRef]
- Bielski, E.R.; Zhong, Q.; Brown, M.; da Rocha, S.R.P. Effect of the Conjugation Density of Triphenylphosphonium Cation on the Mitochondrial Targeting of Poly(Amidoamine) Dendrimers. Mol. Pharm. 2015, 12, 3043–3053. [Google Scholar] [CrossRef]
- Murphy, M.P. Targeting Lipophilic Cations to Mitochondria. Biochim. Biophys. Acta (BBA)—Bioenerg. 2008, 1777, 1028–1031. [Google Scholar] [CrossRef]
- Zielonka, J.; Joseph, J.; Sikora, A.; Hardy, M.; Ouari, O.; Vasquez-Vivar, J.; Cheng, G.; Lopez, M.; Kalyanaraman, B. Mitochondria-Targeted Triphenylphosphonium-Based Compounds: Syntheses, Mechanisms of Action, and Therapeutic and Diagnostic Applications. Chem. Rev. 2017, 117, 10043–10120. [Google Scholar] [CrossRef]
- Huczyński, A. Polyether Ionophores—Promising Bioactive Molecules for Cancer Therapy. Bioorg. Med. Chem. Lett. 2012, 22, 7002–7010. [Google Scholar] [CrossRef]
- Antoszczak, M.; Rutkowski, J.; Huczyński, A. Structure and Biological Activity of Polyether Ionophores and Their Semisynthetic Derivatives. In Bioactive Natural Products; Wiley: Hoboken, NJ, USA, 2014; pp. 107–170. [Google Scholar]
- Gupta, P.B.; Onder, T.T.; Jiang, G.; Tao, K.; Kuperwasser, C.; Weinberg, R.A.; Lander, E.S. Identification of Selective Inhibitors of Cancer Stem Cells by High-Throughput Screening. Cell 2009, 138, 645–659. [Google Scholar] [CrossRef] [PubMed]
- Kopp, F.; Hermawan, A.; Oak, P.S.; Herrmann, A.; Wagner, E.; Roidl, A. Salinomycin Treatment Reduces Metastatic Tumor Burden by Hampering Cancer Cell Migration. Mol. Cancer 2014, 13, 16. [Google Scholar] [CrossRef] [PubMed]
- Schenk, M.; Aykut, B.; Teske, C.; Giese, N.A.; Weitz, J.; Welsch, T. Salinomycin Inhibits Growth of Pancreatic Cancer and Cancer Cell Migration by Disruption of Actin Stress Fiber Integrity. Cancer Lett. 2015, 358, 161–169. [Google Scholar] [CrossRef]
- Jangamreddy, J.R.; Ghavami, S.; Grabarek, J.; Kratz, G.; Wiechec, E.; Fredriksson, B.-A.; Rao Pariti, R.K.; Cieślar-Pobuda, A.; Panigrahi, S.; Łos, M.J. Salinomycin Induces Activation of Autophagy, Mitophagy and Affects Mitochondrial Polarity: Differences between Primary and Cancer Cells. Biochim. Biophys. Acta (BBA)—Mol. Cell Res. 2013, 1833, 2057–2069. [Google Scholar] [CrossRef]
- Wang, F.; He, L.; Dai, W.-Q.; Xu, Y.-P.; Wu, D.; Lin, C.-L.; Wu, S.-M.; Cheng, P.; Zhang, Y.; Shen, M.; et al. Salinomycin Inhibits Proliferation and Induces Apoptosis of Human Hepatocellular Carcinoma Cells In Vitro and In Vivo. PLoS ONE 2012, 7, e50638. [Google Scholar] [CrossRef]
- Lieke, T.; Ramackers, W.; Bergmann, S.; Klempnauer, J.; Winkler, M.; Klose, J. Impact of Salinomycin on Human Cholangiocarcinoma: Induction of Apoptosis and Impairment of Tumor Cell Proliferation in Vitro. BMC Cancer 2012, 12, 466. [Google Scholar] [CrossRef] [PubMed]
- Mao, J.; Fan, S.; Ma, W.; Fan, P.; Wang, B.; Zhang, J.; Wang, H.; Tang, B.; Zhang, Q.; Yu, X.; et al. Roles of Wnt/β-Catenin Signaling in the Gastric Cancer Stem Cells Proliferation and Salinomycin Treatment. Cell Death Dis. 2014, 5, e1039. [Google Scholar] [CrossRef]
- Lu, W.; Li, Y. Salinomycin Suppresses LRP6 Expression and Inhibits Both Wnt/Β-Catenin and MTORC1 Signaling in Breast and Prostate Cancer Cells. J. Cell Biochem. 2014, 115, 1799–1807. [Google Scholar] [CrossRef]
- Lu, Y.; Ma, W.; Mao, J.; Yu, X.; Hou, Z.; Fan, S.; Song, B.; Wang, H.; Li, J.; Kang, L.; et al. Salinomycin Exerts Anticancer Effects on Human Breast Carcinoma MCF-7 Cancer Stem Cells via Modulation of Hedgehog Signaling. Chem. Biol. Interact. 2015, 228, 100–107. [Google Scholar] [CrossRef]
- Riccioni, R.; Dupuis, M.L.; Bernabei, M.; Petrucci, E.; Pasquini, L.; Mariani, G.; Cianfriglia, M.; Testa, U. The Cancer Stem Cell Selective Inhibitor Salinomycin Is a P-Glycoprotein Inhibitor. Blood Cells Mol. Dis. 2010, 45, 86–92. [Google Scholar] [CrossRef]
- Fuchs, D.; Daniel, V.; Sadeghi, M.; Opelz, G.; Naujokat, C. Salinomycin Overcomes ABC Transporter-Mediated Multidrug and Apoptosis Resistance in Human Leukemia Stem Cell-Like KG-1a Cells. Biochem. Biophys. Res. Commun. 2010, 394, 1098–1104. [Google Scholar] [CrossRef] [PubMed]
- Verdoodt, B.; Vogt, M.; Schmitz, I.; Liffers, S.-T.; Tannapfel, A.; Mirmohammadsadegh, A. Salinomycin Induces Autophagy in Colon and Breast Cancer Cells with Concomitant Generation of Reactive Oxygen Species. PLoS ONE 2012, 7, e44132. [Google Scholar] [CrossRef] [PubMed]
- Ketola, K.; Hilvo, M.; Hyötyläinen, T.; Vuoristo, A.; Ruskeepää, A.-L.; Orešič, M.; Kallioniemi, O.; Iljin, K. Salinomycin Inhibits Prostate Cancer Growth and Migration via Induction of Oxidative Stress. Br. J. Cancer 2012, 106, 99–106. [Google Scholar] [CrossRef]
- Kim, J.; Chae, M.; Kim, W.K.; Kim, Y.; Kang, H.S.; Kim, H.S.; Yoon, S. Salinomycin Sensitizes Cancer Cells to the Effects of Doxorubicin and Etoposide Treatment by Increasing DNA Damage and Reducing P21 Protein. Br. J. Pharmacol. 2011, 162, 773–784. [Google Scholar] [CrossRef]
- Ketola, K.; Vainio, P.; Fey, V.; Kallioniemi, O.; Iljin, K. Monensin Is a Potent Inducer of Oxidative Stress and Inhibitor of Androgen Signaling Leading to Apoptosis in Prostate Cancer Cells. Mol. Cancer Ther. 2010, 9, 3175–3185. [Google Scholar] [CrossRef] [PubMed]
- Park, W.H.; Kim, E.S.; Kim, B.K.; Lee, Y.Y. Monensin-Mediated Growth Inhibition in NCI-H929 Myeloma Cells via Cell Cycle Arrest and Apoptosis. Int. J. Oncol. 2003, 23, 197–204. [Google Scholar] [CrossRef]
- Park, W.H.; Kim, E.S.; Jung, C.W.; Kim, B.K.; Lee, Y.Y. Monensin-Mediated Growth Inhibition of SNU-C1 Colon Cancer Cells via Cell Cycle Arrest and Apoptosis. Int. J. Oncol. 2003, 22, 377–382. [Google Scholar] [CrossRef] [PubMed]
- Park, W.H.; Seol, J.G.; Kim, E.S.; Kang, W.K.; Im, Y.H.; Jung, C.W.; Kim, B.K.; Lee, Y.Y. Monensin-Mediated Growth Inhibition in Human Lymphoma Cells Through Cell Cycle Arrest and Apoptosis. Br. J. Haematol. 2002, 119, 400–407. [Google Scholar] [CrossRef] [PubMed]
- Sulik, M.; Jędrzejczyk, M.; Mielczarek-Puta, M.; Lim, G.Y.; Podsiad, M.; Hoser, J.; Bednarczyk, P.; Struga, M.; Huczyński, A. Mitochondria-Targeting Phosphonium Salts of Monensin A as the Source of Novel Antiproliferative Agents. Eur. J. Med. Chem. Rep. 2025, 14, 100275. [Google Scholar] [CrossRef]
- Jędrzejczyk, M.; Sulik, M.; Mielczarek-Puta, M.; Lim, G.Y.; Podsiad, M.; Hoser, J.; Bednarczyk, P.; Struga, M.; Huczyński, A. Anticancer Activity of Salinomycin Quaternary Phosphonium Salts. Eur. J. Med. Chem. 2025, 282, 117055. [Google Scholar] [CrossRef]
- Antonenko, Y.N.; Jędrzejczyk, M.; Rokitskaya, T.I.; Khailova, L.S.; Kotova, E.A.; Huczyński, A. Rate of Translocation Across Lipid Bilayer of Triphenylphosphonium-Linked Salinomycin Derivatives Contributes Significantly to Their K+/H+ Exchange Activity on Membranes. Bioelectrochemistry 2022, 145, 108089. [Google Scholar] [CrossRef]
- Urbaniak, A.; Reed, M.R.; Heflin, B.; Gaydos, J.; Piña-Oviedo, S.; Jędrzejczyk, M.; Klejborowska, G.; Stępczyńska, N.; Chambers, T.C.; Tackett, A.J.; et al. Anti-Glioblastoma Activity of Monensin and Its Analogs in an Organoid Model of Cancer. Biomed. Pharmacother. 2022, 153, 113440. [Google Scholar] [CrossRef]
- Jędrzejczyk, M.; Stępczyńska, N.; Klejborowska, G.; Podsiad, M.; Stefańska, J.; Steverding, D.; Huczyński, A. Synthesis and Evaluation of Antibacterial and Trypanocidal Activity of Derivatives of Monensin A. Bioorg. Med. Chem. Lett. 2022, 58, 128521. [Google Scholar] [CrossRef] [PubMed]
- Czerwonka, D.; Mielczarek-Puta, M.; Antoszczak, M.; Cioch, A.; Struga, M.; Huczyński, A. Evaluation of the Anticancer Activity of Singly and Doubly Modified Analogues of C20-Epi-Salinomycin. Eur. J. Pharmacol. 2021, 908, 174347. [Google Scholar] [CrossRef]
- Czerwonka, D.; Barcelos, Y.; Steverding, D.; Cioch, A.; Huczyński, A.; Antoszczak, M. Singly and Doubly Modified Analogues of C20-Epi-Salinomycin: A New Group of Antiparasitic Agents against Trypanosoma Brucei. Eur. J. Med. Chem. 2021, 209, 112900. [Google Scholar] [CrossRef]
- Sulik, M.; Graniczny, R.; Janczak, J.; Kłopotowska, D.; Wietrzyk, J.; Huczyński, A. From Pseudocyclic to Macrocyclic Ionophores: Strategies Toward the Synthesis of Cyclic Monensin Derivatives. J. Org. Chem. 2025, 90, 1344–1353. [Google Scholar] [CrossRef] [PubMed]
- Antoszczak, M.; Gadsby-Davis, K.; Steverding, D.; Huczyński, A. Synthesis of Urea and Thiourea Derivatives of C20-Epi-Aminosalinomycin and Their Activity against Trypanosoma Brucei. Eur. J. Med. Chem. 2023, 250, 115241. [Google Scholar] [CrossRef]
- Sulik, M.; Workneh, E.A.; Santana, S.; Teixeira, B.; Prudêncio, M.; Janczak, J.; Huczyński, A. Chemical Modification of Monensin as a Source of Potent Antiplasmodial Agents. Bioorg. Med. Chem. 2025, 123, 118177. [Google Scholar] [CrossRef]
- Antoszczak, M.; Mielczarek-Puta, M.; Struga, M.; Huczynski, A. Urea and Thiourea Derivatives of Salinomycin as Agents Targeting Malignant Colon Cancer Cells. Anticancer Agents Med. Chem. 2025, 25, 330–338. [Google Scholar] [CrossRef] [PubMed]
- Sulik, M.; Maj, E.; Wietrzyk, J.; Huczyński, A.; Antoszczak, M. Synthesis and Anticancer Activity of Dimeric Polyether Ionophores. Biomolecules 2020, 10, 1039. [Google Scholar] [CrossRef]
- Czerwonka, D.; Müller, S.; Cañeque, T.; Colombeau, L.; Huczyński, A.; Antoszczak, M.; Rodriguez, R. Expeditive Synthesis of Potent C20-Epi-Amino Derivatives of Salinomycin against Cancer Stem-Like Cells. ACS Org. Inorg. Au 2022, 2, 214–221. [Google Scholar] [CrossRef]
- Chan, D.C. Mitochondrial Dynamics and Its Involvement in Disease. Annu. Rev. Pathol. Mech. Dis. 2020, 15, 235–259. [Google Scholar] [CrossRef]
- Friedman, J.R.; Nunnari, J. Mitochondrial Form and Function. Nature 2014, 505, 335–343. [Google Scholar] [CrossRef] [PubMed]
- Youle, R.J.; van der Bliek, A.M. Mitochondrial Fission, Fusion, and Stress. Science 2012, 337, 1062–1065. [Google Scholar] [CrossRef] [PubMed]
- Tilokani, L.; Nagashima, S.; Paupe, V.; Prudent, J. Mitochondrial Dynamics: Overview of Molecular Mechanisms. Essays Biochem. 2018, 62, 341–360. [Google Scholar] [CrossRef]
- Chen, S.; Li, Q.; Shi, H.; Li, F.; Duan, Y.; Guo, Q. New Insights into the Role of Mitochondrial Dynamics in Oxidative Stress-Induced Diseases. Biomed. Pharmacother. 2024, 178, 117084. [Google Scholar] [CrossRef] [PubMed]
- Shadel, G.S.; Horvath, T.L. Mitochondrial ROS Signaling in Organismal Homeostasis. Cell 2015, 163, 560–569. [Google Scholar] [CrossRef]
- Suski, J.M.; Lebiedzinska, M.; Bonora, M.; Pinton, P.; Duszynski, J.; Wieckowski, M.R. Relation Between Mitochondrial Membrane Potential and ROS Formation. Methods. Mol. Biol. 2012, 810, 183–205. [Google Scholar] [CrossRef]
- Mitani, M.; Yamanishi, T.; Miyazaki, Y.; Ōtake, N. Salinomycin Effects on Mitochondrial Ion Translocation and Respiration. Antimicrob. Agents Chemother. 1976, 9, 655–660. [Google Scholar] [CrossRef]
- Riddell, F.G.; Hayer, M.K. The Monensin-Mediated Transport of Sodium Ions Through Phospholipid Bilayers Studied by 23Na-NMR Spectroscopy. Biochim. Biophys. Acta (BBA)—Biomembr. 1985, 817, 313–317. [Google Scholar] [CrossRef]
- Antonenko, Y.N.; Rokitskaya, T.I.; Huczyński, A. Electrogenic and Nonelectrogenic Ion Fluxes Across Lipid and Mitochondrial Membranes Mediated by Monensin and Monensin Ethyl Ester. Biochim. Biophys. Acta (BBA)—Biomembr. 2015, 1848, 995–1004. [Google Scholar] [CrossRef]
- Itoh, Y.; Law, M.J.; Sokoloff, L. Effects of the Na+/H+ Exchanger Monensin on Intracellular PH in Astroglia. Brain Res. 2000, 882, 226–229. [Google Scholar] [CrossRef]
- Huczyński, A.; Janczak, J.; Stefańska, J.; Antoszczak, M.; Brzezinski, B. Synthesis and Antimicrobial Activity of Amide Derivatives of Polyether Antibiotic—Salinomycin. Bioorg. Med. Chem. Lett. 2012, 22, 4697–4702. [Google Scholar] [CrossRef] [PubMed]
- Klejborowska, G.; Jędrzejczyk, M.; Stępczyńska, N.; Maj, E.; Wietrzyk, J.; Huczyński, A. Antiproliferative Activity of Ester Derivatives of Monensin A at the C-1 and C-26 Positions. Chem. Biol. Drug. Des. 2019, 94, 1859–1864. [Google Scholar] [CrossRef] [PubMed]
- Hoser, J.; Dabrowska, A.; Zajac, M.; Bednarczyk, P. Changes in Ion Transport Across Biological Membranes Exposed to Particulate Matter. Membranes 2023, 13, 763. [Google Scholar] [CrossRef] [PubMed]







| Compound | Cancer Cells | Normal Cells | ||||||
|---|---|---|---|---|---|---|---|---|
| SW480 | SW620 | PC3 | MDA-MB-231 | A549 | MiaPaCa | HaCaT | V79 | |
| IC50 | IC50 | IC50 | IC50 | IC50 | IC50 | IC50 | IC50 | |
| SAL, 1 | 1.1 ± 0.04 | 1.5 ± 0.06 | 2.7 ± 0.1 | 1.0 ± 0.1 | 1.6 ± 0.1 | 2.6 ± 0.1 | 0.6 ± 0.1 | 1.2 ± 0.1 |
| 1a | 9.0 ± 3.6 | 16.4 ± 2.4 | 1.2 ± 0.2 | 3.4 ± 0.5 | 2.9 ± 0.5 | 7.6 ± 1.3 | 3.8 ± 0.3 | 4.8 ± 0.6 |
| 1b | 8.8 ± 2.4 | 16.8 ± 4.4 | 5.5 ± 1.4 | 5.2 ± 0.2 | 3.9 ± 0.8 | 8.5 ± 1.5 | 2.8 ± 0.2 | 4.5 ± 0.4 |
| 1c | 9.8 ± 2.0 | 14.0 ± 4.1 | 12.9 ± 1.3 | 11.4 ± 1.3 | 15.6 ± 9.1 | 14.1 ± 1.5 | 7.1 ± 1.2 | 17.0 ± 2.8 |
| 1d | 2.5 ± 0.6 | 1.8 ± 0.2 | 3.6 ± 1.6 | 5.0 ± 0.3 | 3.6 ± 0.5 | 2.9 ± 0.6 | 2.4 ± 0.1 | 4.8 ± 0.2 |
| 1e | 10.4 ± 2.3 | 10.9 ± 2.6 | 4.0 ± 0.7 | 6.7 ± 1.2 | 5.1 ± 0.8 | 6.4 ± 0.2 | 8.3 ± 0.7 | 8.8 ± 0.4 |
| 1f | 0.7 ± 0.2 | 0.6 ± 0.05 | 0.3 ± 0.05 | 1.7 ± 0.002 | 1.7 ± 0.01 | 0.4 ± 0.06 | 1.7 ± 0.002 | 1.7 ± 0.02 |
| MON, 2 | 2.5 ± 0.2 | 2.7 ± 0.1 | 3.4 ± 1.0 | 4.9 ± 0.3 | 2.6 ± 1.0 | 1.1 ± 0.2 | 3.5 ± 1.4 | 3.6 ± 0.5 |
| 2a | 0.5 ± 0.1 | 0.7 ± 0.2 | 0.4 ± 0.1 | 0.7 ± 0.2 | 0.3 ± 0.02 | 0.3 ± 0.1 | 0.6 ± 0.2 | 5.0 ± 0.04 |
| 2b | 0.8 ± 0.04 | 1.5 ± 0.2 | 0.7 ± 0.3 | 1.7 ± 0.1 | 3.4 ± 1.1 | 1.1 ± 0.3 | 1.1 ± 0.5 | 6.9 ± 1.9 |
| 2c | 18.3 ± 0.4 | 10.6 ± 0.4 | 14.5 ± 2.1 | 13.9 ± 2.6 | 14.9 ± 3.4 | 13.9 ± 0.7 | 14.5 ± 3.3 | 32.2 ± 1.0 |
| 2d | 3.5 ± 0.1 | 4.3 ± 0.01 | 3.1 ± 0.6 | 1.4 ± 0.2 | 1.4 ± 0.4 | 1.6 ± 0.3 | 1.6 ± 0.2 | 4.2 ± 1.4 |
| 2e | 46.7 ± 3.1 | 28.1 ± 2.1 | >100 | >100 | >100 | >100 | 39.2 ± 5.6 | >100 |
| 2f | 1.7 ± 0.3 | 2.3 ± 0.4 | 1.4 ± 0.1 | 1.4 ± 0.2 | 1.3 ± 0.2 | 0.6 ± 0.2 | 0.3 ± 0.1 | 3.2 ± 1.1 |
| DOX | 0.3 ± 0.08 | 0.3 ± 0.08 | 0.6 ± 0.02 | 0.8 ± 0.03 | 0.6 ± 0.2 | 1.1 ± 0.1 | 0.3 ± 0.01 | 2.0 ± 0.03 |
| Compound | HaCaT | V79 | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| SW480 | SW620 | PC3 | MDA-MB-231 | A549 | MiaPaCa | SW480 | SW620 | PC3 | MDA-MB-231 | A549 | MiaPaCa | |
| SAL, 1 | 0.5 | 0.4 | 0.2 | 0.6 | 0.4 | 0.2 | 1.1 | 0.8 | 0.4 | 1.2 | 0.8 | 0.5 |
| 1a | 0.4 | 0.2 | 3.2 | 1.1 | 1.3 | 0.5 | 0.5 | 0.3 | 4 | 1.4 | 1.7 | 0.6 |
| 1b | 0.3 | 0.2 | 0.5 | 0.5 | 0.7 | 0.3 | 0.5 | 0.3 | 0.8 | 0.9 | 1.2 | 0.5 |
| 1c | 0.7 | 0.5 | 0.6 | 0.6 | 0.5 | 0.5 | 1.7 | 1.2 | 1.3 | 1.5 | 1.1 | 1.2 |
| 1d | 1.0 | 1.3 | 0.7 | 0.5 | 0.7 | 0.8 | 1.9 | 2.7 | 1.3 | 1.0 | 1.3 | 1.7 |
| 1e | 0.8 | 0.8 | 2.1 | 1.2 | 1.6 | 1.3 | 0.8 | 0.8 | 2.2 | 1.3 | 1.7 | 1.4 |
| 1f | 2.4 | 2.8 | 5.7 | 1.0 | 1.0 | 4.3 | 2.4 | 2.8 | 5.7 | 1.0 | 1.0 | 4.3 |
| MON, 2 | 1.4 | 1.3 | 1.0 | 0.7 | 1.3 | 3.2 | 1.4 | 1.3 | 1.1 | 0.7 | 1.4 | 3.3 |
| 2a | 1.2 | 0.9 | 1.5 | 0.9 | 2.0 | 2.0 | 10.0 | 7.1 | 12.5 | 7.1 | 16.7 | 16.7 |
| 2b | 1.4 | 0.7 | 1.6 | 0.6 | 0.3 | 1.0 | 8.6 | 4.6 | 9.9 | 4.1 | 2.0 | 6.3 |
| 2c | 0.8 | 1.4 | 1.0 | 1.0 | 1.0 | 1.0 | 1.8 | 3.0 | 2.2 | 2.3 | 2.2 | 2.3 |
| 2d | 0.5 | 0.4 | 0.5 | 1.1 | 1.1 | 1.0 | 1.2 | 1.0 | 1.4 | 3.0 | 3.0 | 2.6 |
| 2e | 0.8 | 1.4 | - | - | - | - | - | - | - | - | - | - |
| 2f | 0.2 | 0.1 | 0.2 | 0.2 | 0.2 | 0.5 | 1.9 | 1.4 | 2.3 | 2.3 | 2.5 | 5.3 |
| DOX | 1.0 | 1.0 | 0.5 | 0.4 | 0.5 | 0.3 | 6.7 | 6.7 | 3.3 | 2.5 | 3.3 | 1.8 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sulik, M.; Jędrzejczyk, M.; Mielczarek-Puta, M.; Hoser, J.; Bednarczyk, P.; Struga, M.; Huczyński, A. Mitochondrial-Targeted Triphenylphosphonium-Conjugated Ionophores with Enhanced Cytotoxicity in Cancer Cells. Molecules 2025, 30, 4413. https://doi.org/10.3390/molecules30224413
Sulik M, Jędrzejczyk M, Mielczarek-Puta M, Hoser J, Bednarczyk P, Struga M, Huczyński A. Mitochondrial-Targeted Triphenylphosphonium-Conjugated Ionophores with Enhanced Cytotoxicity in Cancer Cells. Molecules. 2025; 30(22):4413. https://doi.org/10.3390/molecules30224413
Chicago/Turabian StyleSulik, Michał, Marta Jędrzejczyk, Magdalena Mielczarek-Puta, Jakub Hoser, Piotr Bednarczyk, Marta Struga, and Adam Huczyński. 2025. "Mitochondrial-Targeted Triphenylphosphonium-Conjugated Ionophores with Enhanced Cytotoxicity in Cancer Cells" Molecules 30, no. 22: 4413. https://doi.org/10.3390/molecules30224413
APA StyleSulik, M., Jędrzejczyk, M., Mielczarek-Puta, M., Hoser, J., Bednarczyk, P., Struga, M., & Huczyński, A. (2025). Mitochondrial-Targeted Triphenylphosphonium-Conjugated Ionophores with Enhanced Cytotoxicity in Cancer Cells. Molecules, 30(22), 4413. https://doi.org/10.3390/molecules30224413







