Aptamer-Based Biosensors for Rapid Detection and Early Warning of Food Contaminants: From Selection to Field Applications
Abstract
1. Introduction
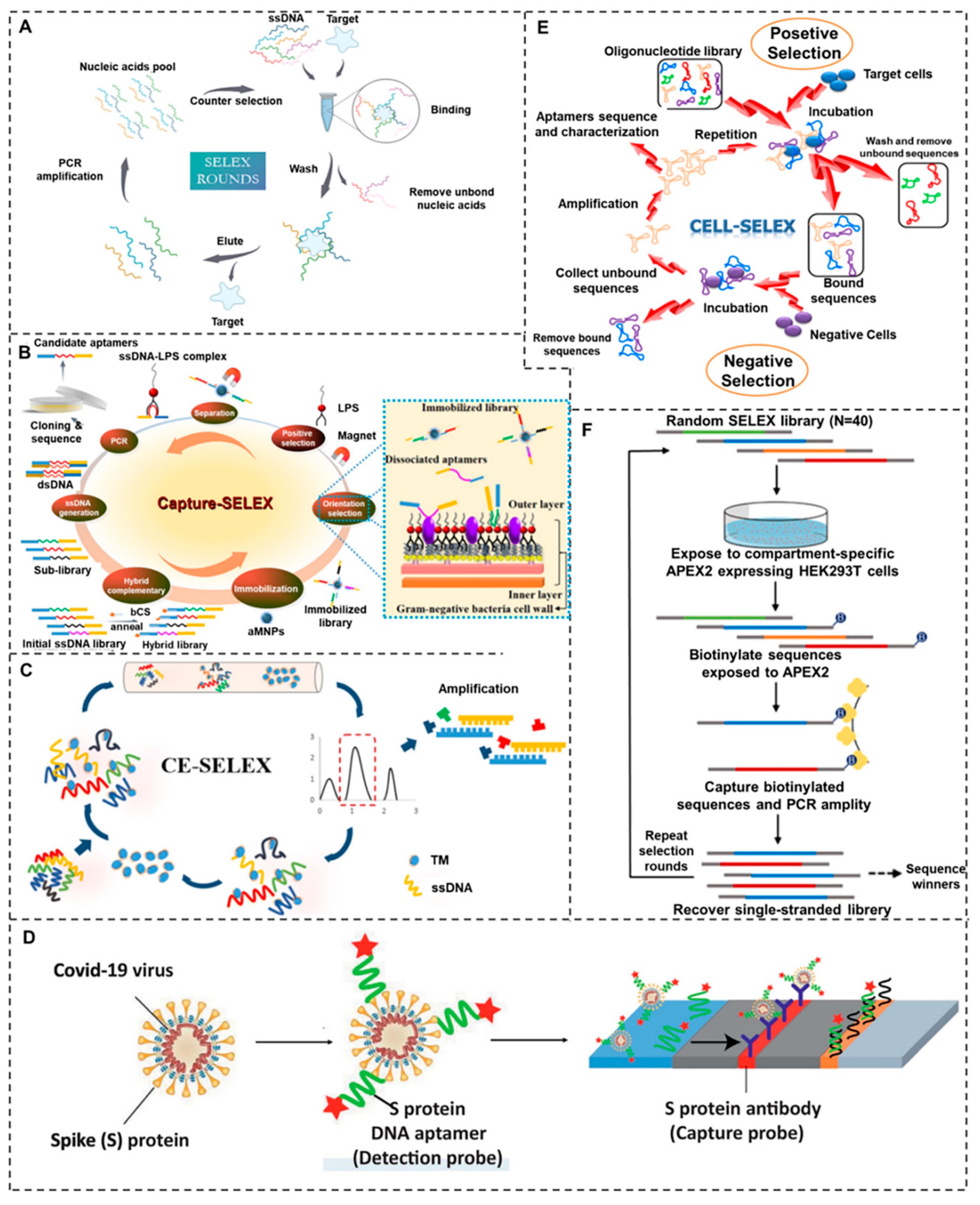
2. Aptamer Screening Methods
2.1. Capture-SELEX
2.2. CE-SELEX
2.3. Nitrocellulose SELEX
2.4. Cell-SELEX
2.5. Subcellular-SELEX
| Type of SELEX | Advantages | Limitations | Ref. |
|---|---|---|---|
| Capture-SELEX | Native Conformation, Label-Free, High-Throughput and Rapid, Small-Molecule Compatible | Low release efficiency, Non-specific binding, High cost | [33,34,35] |
| CE-SELEX | Large-Biomolecule Capable, High Precision, Complex Target Suitability, High Resolution, Iterative Enrichment | Limited target scope, Technical complexity | [36] |
| Nitrocellulose SELEX | Immobilization-Friendly, Broad-Spectrum, and High-Affinity | High non-specific binding, Low-throughput, and Labor-intensive | [38] |
| Cell-SELEX | In Vitro/In Vivo Compatible, High Sensitivity, Personalized Therapy Potential | Unidentified target, Complex process | [40] |
| Subcellular-SELEX | Native Conformation, Native PTMs | Target ambiguity, Technical complexity | [43] |
3. Detection of Food Contaminants
3.1. Heavy Metals

3.2. Antibiotics
3.2.1. Aminoglycoside Antibiotics
3.2.2. Tetracyclines
3.2.3. β-Lactam Antibiotic

3.2.4. Other Antibiotics
3.3. Hormonal Molecules
3.3.1. Steroid Hormones
3.3.2. Non-Steroidal Hormones
3.3.3. Harmful Substances with Hormone-like Activity

3.4. Bacteria and Viruses
3.4.1. Bacteria
3.4.2. Virus
3.5. Mycotoxins
3.5.1. Aflatoxin B1
3.5.2. Zearalenone
3.5.3. T-2 Toxin
3.5.4. Fumonisins
3.5.5. Ochratoxin A

3.6. Algal Toxins
3.6.1. Hepatotoxins
3.6.2. Neurotoxins
3.7. Pesticide Residues
3.7.1. Organophosphorus Pesticides
3.7.2. Neonicotinoids
3.7.3. Carbamates
3.7.4. Others

3.8. Preservatives
4. Challenges and Perspectives
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| AFB1 | Aflatoxin B1 |
| AgNCs | Aptamer-templated silver nanocluster |
| Apt | Aptamers |
| Apt-GFET | Aptamer-functionalized graphene field-effect transistor |
| AMP | mpicillin |
| BPA | Bisphenol A |
| BPB | Bisphenol B |
| BPE | Bisphenol E |
| BPF | Bisphenol F |
| CAP | Chloramphenicol |
| CE-SELEX | Capillary electrophoresis-SELEX |
| CE-MoS2 | Chemically exfoliated MoS2 |
| CHA | Catalytic hairpin assembly |
| CIP | Ciprofloxacin |
| CLCsolids | Cholesteric liquid crystals solid |
| CSCs | Cancer stem cells |
| CS | Chromatographic strip |
| DEHP | Di-2-ethylhexyl phthalate |
| DES | Diethylstilbestrol |
| DON | Deoxynivalenol |
| DOX | Doxycycline |
| EBFC | Enzyme biofuel cell |
| EC | Electrochemical |
| ECL | Electrochemiluminescence |
| ECL-RET | Electrochemiluminescence resonance energy transfer |
| ELC | Ehemiluminescence |
| ENR | Enrofloxacin |
| E2 | 17β-estradiol |
| FB1 | Fumonisin B1 |
| FL | Fluorescence |
| FUM | Fumonisins |
| FRET | Fluorescence resonance energy transfer |
| GEN | Gentamicin |
| GDY | Graphdiyne |
| GR | Graphene |
| KANA | Kanamycin |
| HES | Hexestrol |
| H-Gr | Hemoglobin–graphene |
| IARC | International Agency for Research on Cancer |
| LOD | Limit of detection |
| LPS | Lipopolysaccharides |
| LRET | Luminescence resonance energy transfer |
| MC-LR | Microcystin-LR |
| MIP | Molecularly imprinted membrane |
| MIPs | Molecularly imprinted polymers |
| MNPs | Magnetic nanoparticles |
| nAChRs | Nicotinic acetylcholine receptor |
| NOR | Norfloxacin |
| NoV | Norovirus |
| OTA | Ochratoxin A |
| OTC | Oxytetracycline |
| o-PD | O-phenylenediamine |
| PAE | Phthalate acid ester |
| PEC | Photoelectrochemical |
| PEN | Penicillin |
| PSP | Paralytic shellfish poisoning |
| pPtNP | Porous platinum nanoparticles |
| PPS | Peroxidase proximity selection |
| PT | Photothermal |
| rGO-ZnO-AuNPs | Reduced graphene oxide-zinc oxide-gold nanocomposites |
| SDZ | Sulfadiazine |
| SDM | Sulfamethoxypyridazine |
| SELEX | Systematic evolution of ligands by exponential enrichment |
| SERS | Surface-enhanced Raman scattering |
| SG | SYBR Green I |
| SMZ | Sulfamethoxazole |
| SPCEs | Screen-printed carbon electrodes |
| SQX | Sulfaquinoxaline |
| STR | Streptomycin |
| ssDNA | Single-stranded DNA |
| TCs | Tetracycline antibiotics |
| TET | Tetracycline |
| TISD | Target-induced strand displacement |
| TOB | Tobramycin |
| TST | Testosterone |
| TTX | Tetrodotoxin |
| ZEN | Zearalenone |
| Zn-PMOFs | Zinc porphyrin-based metal–organic frameworks |
References
- Gizaw, Z. Public health risks related to food safety issues in the food market: A systematic literature review. Environ. Health Prev. Med. 2019, 24, 68. [Google Scholar] [CrossRef]
- Azimi, A.; Azari, A.; Rezakazemi, M.; Ansarpour, M. Removal of Heavy Metals from Industrial Wastewaters: A Review. ChemBioEng Rev. 2017, 4, 37–59. [Google Scholar] [CrossRef]
- Li, B.; Xia, M.; Zorec, R.; Parpura, V.; Verkhratsky, A. Astrocytes in heavy metal neurotoxicity and neurodegeneration. Brain Res. 2021, 1752, 147234. [Google Scholar] [CrossRef]
- Zhang, P.; Yang, M.; Lan, J.; Huang, Y.; Zhang, J.; Huang, S.; Yang, Y.; Ru, J. Water Quality Degradation Due to Heavy Metal Contamination: Health Impacts and Eco-Friendly Approaches for Heavy Metal Remediation. Toxics 2023, 11, 828. [Google Scholar] [CrossRef] [PubMed]
- Nicolella, H.D.; de Assis, S. Epigenetic Inheritance: Intergenerational Effects of Pesticides and Other Endocrine Disruptors on Cancer Development. Int. J. Mol. Sci. 2022, 23, 4671. [Google Scholar] [CrossRef] [PubMed]
- Pfaffl, M.W.; Riedmaier-Sprenzel, I. New surveillance concepts in food safety in meat producing animals: The advantage of high throughput ‘omics’ technologies—A review. Asian-Australas. J. Anim. Sci. 2018, 31, 1062–1071. [Google Scholar] [CrossRef] [PubMed]
- Valsamakis, G.; Arapaki, A.; Balafoutas, D.; Charmandari, E.; Vlahos, N.F. Diet-Induced Hypothalamic Inflammation, Phoenixin, and Subsequent Precocious Puberty. Nutrients 2021, 13, 3460. [Google Scholar] [CrossRef]
- Saleh, N.A.; Ayoub, M.A.; Nossair, M.A.; Alqhtani, A.H.; Swelum, A.A.; Khojah, H.; Gamal, M.; Imam, M.S.; Khafaga, A.F.; Arif, M.; et al. Influence of Water Quality and Pollution on Broiler’s Performance, Vaccine and Antibiotic Efficiencies—A Review. Ann. Anim. Sci. 2023, 23, 1021–1036. [Google Scholar] [CrossRef]
- Nazhand, A.; Durazzo, A.; Lucarini, M.; Souto, E.B.; Santini, A. Characteristics, Occurrence, Detection and Detoxification of Aflatoxins in Foods and Feeds. Foods 2020, 9, 644. [Google Scholar] [CrossRef]
- Kim, S.-A.; Lee, Y.-M.; Choi, J.-Y.; Jacobs, D.R.; Lee, D.-H. Evolutionarily adapted hormesis-inducing stressors can be a practical solution to mitigate harmful effects of chronic exposure to low dose chemical mixtures. Environ. Pollut. 2018, 233, 725–734. [Google Scholar] [CrossRef]
- Focker, M.; van der Fels-Klerx, H.J. Economics applied to food safety. Curr. Opin. Food Sci. 2020, 36, 18–23, ISSN 2214-7993. [Google Scholar] [CrossRef]
- Ziani, I.; Bouakline, H.; El Guerraf, A.; El Bachiri, A.; Marie-Laure, F.; Sher, F. Integrating AI and advanced spectroscopic techniques for precision food safety and quality control. Trends Food Sci. Technol. 2025, 156, 104850. [Google Scholar] [CrossRef]
- Radomirović, M.; Gligorijević, N.; Rajković, A. Immuno-PCR in the Analysis of Food Contaminants. Int. J. Mol. Sci. 2025, 26, 3091. [Google Scholar] [CrossRef] [PubMed]
- Zhang, S.J.; Wu, C.; Walt, D.R. A Multiplexed Digital Platform Enables Detection of Attomolar Protein Levels with Minimal Cross-Reactivity. ACS Nano 2024, 18, 29891–29901. [Google Scholar] [CrossRef] [PubMed]
- Nieddu, M.; Baralla, E.; Pasciu, V.; Rimoli, M.G.; Boatto, G. Cross-reactivity of commercial immunoassays for screening of new amphetamine designer drugs. A review. J. Pharm. Biomed. Anal. 2022, 218, 114868. [Google Scholar] [CrossRef] [PubMed]
- He, Y.; Yuan, J.; Khan, I.M.; Zhang, L.; Ma, P.; Wang, Z. Research progress of aptasensor technology in the detection of foodborne pathogens. Food Control 2023, 153, 109891. [Google Scholar] [CrossRef]
- Tang, Y.; Yuan, J.; Zhang, Y.; Khan, I.M.; Ma, P.; Wang, Z. Lateral flow assays based on aptamers for food safety applications. Food Control 2024, 155, 110051. [Google Scholar] [CrossRef]
- Derkus, B. Applying the miniaturization technologies for biosensor design. Biosens. Bioelectron. 2016, 79, 901–913. [Google Scholar] [CrossRef]
- Chen, M.T.; Qileng, A.; Liang, H.Z.; Lei, H.T.; Liu, W.P.; Liu, Y.J. Advances in immunoassay-based strategies for mycotoxin detection in food: From single-mode immunosensors to dual-mode immunosensors. Compr. Rev. Food Sci. Food Saf. 2023, 22, 1285–1311. [Google Scholar] [CrossRef]
- Zhang, Z.; Lou, Y.; Guo, C.; Jia, Q.; Song, Y.; Tian, J.-Y.; Zhang, S.; Wang, M.; He, L.; Du, M. Metal–organic frameworks (MOFs) based chemosensors/biosensors for analysis of food contaminants. Trends Food Sci. Technol. 2021, 118, 569–588. [Google Scholar] [CrossRef]
- Boucheta, H.; Zouaoui, E.; Ferkous, H.; Madaci, A.; Yadav, K.K.; Benguerba, Y. Advancing Diabetes Management: The Future of Enzyme-Less Nanoparticle-Based Glucose Sensors-A Review. J. Diabetes Sci. Technol. 2024, 19, 1097–1108. [Google Scholar] [CrossRef]
- Soy, S.; Sharma, S.R.; Nigam, V.K. Bio-fabrication of thermozyme-based nano-biosensors: Their components and present scenario. J. Mater. Sci. Mater. Electron. 2022, 33, 5523–5533. [Google Scholar] [CrossRef]
- Li, X.; Kuchinski, L.M.; Park, A.; Murphy, G.S.; Soto, K.C.; Schuster, B.S. Enzyme purification and sustained enzyme activity for pharmaceutical biocatalysis by fusion with phase-separating intrinsically disordered protein. Biotechnol. Bioeng. 2024, 121, 3155–3168. [Google Scholar] [CrossRef] [PubMed]
- Jin, X.; Chen, J.; Zeng, X.; Xu, L.; Wu, Y.; Fu, F. A signal-on magnetic electrochemical immunosensor for ultra-sensitive detection of saxitoxin using palladium-doped graphitic carbon nitride-based non-competitive strategy. Biosens. Bioelectron. 2019, 128, 45–51. [Google Scholar] [CrossRef] [PubMed]
- Dosadina, E.E.; Savelyeva, E.E.; Belov, A.A. The effect of immobilization, drying and storage on the activity of proteinases immobilized on modified cellulose and chitosan. Process Biochem. 2018, 64, 213–220. [Google Scholar] [CrossRef]
- Alkhamis, O.; Byrd, C.; Canoura, J.; Bacon, A.; Hill, R.; Xiao, Y. Exploring the relationship between aptamer binding thermodynamics, affinity, and specificity. Nucleic Acids Res. 2025, 53, gkaf219. [Google Scholar] [CrossRef]
- Li, H.; Yao, S.; Wang, C.; Bai, C.; Zhou, P. Diverse applications and development of aptamer detection technology. Anal. Sci. 2023, 39, 1627–1641. [Google Scholar] [CrossRef]
- Wan, Q.; Liu, X.; Zu, Y. Oligonucleotide aptamers for pathogen detection and infectious disease control. Theranostics 2021, 11, 9133–9161. [Google Scholar] [CrossRef]
- Guo, W.; Zhang, C.; Ma, T.; Liu, X.; Chen, Z.; Li, S.; Deng, Y. Advances in aptamer screening and aptasensors’ detection of heavy metal ions. J. Nanobiotechnol. 2021, 19, 166. [Google Scholar] [CrossRef]
- Kang, M.; Zhao, Q. Recent advances of aptamer based sensors for aflatoxins, TrAC. Trends Anal. Chem. 2025, 189, 118250. [Google Scholar] [CrossRef]
- Azzouz, A.; Hejji, L.; Kumar, V.; Kim, K.H. Nanomaterials-based aptasensors: An efficient detection tool for heavy-metal and metalloid ions in environmental and biological samples. Environ. Res. 2023, 238, 117170. [Google Scholar] [CrossRef] [PubMed]
- Chen, Z.; Xie, M.; Zhao, F.; Han, S. Application of Nanomaterial Modified Aptamer-Based Electrochemical Sensor in Detection of Heavy Metal Ions. Foods 2022, 11, 1404. [Google Scholar] [CrossRef] [PubMed]
- Lyu, C.; Khan, I.M.; Wang, Z. Capture-SELEX for aptamer selection: A short review. Talanta 2021, 229, 122274. [Google Scholar] [CrossRef] [PubMed]
- Boussebayle, A.; Torka, D.; Ollivaud, S.; Braun, J.; Bofill-Bosch, C.; Dombrowski, M.; Groher, F.; Hamacher, K.; Suess, B. Next-level riboswitch development—Implementation of Capture-SELEX facilitates identification of a new synthetic riboswitch. Nucleic Acids Res. 2019, 47, 4883–4895. [Google Scholar] [CrossRef]
- Ye, H.; Duan, N.; Wu, S.J.; Tan, G.L.; Gu, H.J.; Li, J.H.; Wang, H.T.; Wang, Z.P. Orientation selection of broad-spectrum aptamers against lipopolysaccharides based on capture-SELEX by using magnetic nanoparticles. Microchim. Acta 2017, 184, 4235–4242. [Google Scholar] [CrossRef]
- Li, L.F.; Zhou, J.R.; Wang, K.W.; Chen, X.J.; Fu, L.L.; Wang, Y.B. Screening and Identification of Specific Aptamers for Shellfish Allergen Tropomyosin with Capillary Electrophoresis-SELEX. Food Anal. Methods 2022, 15, 1535–1544. [Google Scholar] [CrossRef]
- Zhao, L.P.; Yang, G.; Zhu, C.; Li, L.S.; Zhao, Y.; Luan, Y.X.; Qu, F. Three-step evolutionary enhanced capillary electrophoresis-SELEX for aptamer selection of exosome vesicles. Talanta 2024, 267, 125203. [Google Scholar] [CrossRef]
- Ellipilli, S.; Wang, H.Z.; Lee, W.J.; Shu, D.; Guo, P.X. Proof-of-concept for speedy development of rapid and simple at-home method for potential diagnosis of early COVID-19 mutant infections using nanogold and aptamer. Nanomed. Nanotechnol. Biol. Med. 2022, 45, 102590. [Google Scholar] [CrossRef]
- Kamarudin, N.; Sat, J.N.A.; Zaidi, N.F.M.; Mustaffa, K.M.F. Evolution of specific RNA aptamers via SELEX targeting recombinant human CD36 protein: A candidate therapeutic target in severe malaria. Asian Pac. J. Trop. Biomed. 2020, 10, 23–32. [Google Scholar] [CrossRef]
- Wu, Y.Y.; Hsieh, I.S.; Tung, C.H.; Weng, C.H.; Wu, J.E.; Yu, J.S.; Hong, T.M.; Chen, Y.L. A novel DNA aptamer targeting lung cancer stem cells exerts a therapeutic effect by binding and neutralizing Annexin A2. Mol. Ther. Nucleic Acids 2022, 27, 956–968. [Google Scholar] [CrossRef]
- Liu, Y.; Wang, Z.M.; Zhuo, Y.T.; Wu, H.; Peng, Y.; Wang, T.; Peng, T.H.; Qiu, L.P.; Tan, W.H. Aptamer-Based Multiparameter Analysis for Molecular Profiling of Hematological Malignancies. Anal. Chem. 2024, 96, 3429–3435. [Google Scholar] [CrossRef] [PubMed]
- Wilbanks, B.; Beimers, W.; Dugan, M.; Weiskittel, T.; Maher, L.J., III. Peroxidase proximity selection to identify aptamers targeting a subcellular location. PNAS Nexus 2023, 2, pgad151. [Google Scholar] [CrossRef] [PubMed]
- Liu, H.Q.; Zhou, Y.J.; Xu, Q.F.; Wong, S.M. Selection of DNA Aptamers for Subcellular Localization of RBSDV P10 Protein in the Midgut of Small Brown Planthoppers by Emulsion PCR-Based SELEX. Viruses 2020, 12, 1239. [Google Scholar] [CrossRef] [PubMed]
- Vignesh, A.; Amal, T.C.; Vasanth, K. Food contaminants: Impact of food processing, challenges and mitigation strategies for food security. Food Res. Int. 2024, 191, 114739. [Google Scholar] [CrossRef]
- Li, Y.; Gao, X.; Fang, Y.; Cui, B.; Shen, Y. Nanomaterials-driven innovative electrochemiluminescence aptasensors in reporting food pollutants, Coord. Chem. Rev. 2023, 485, 215136. [Google Scholar] [CrossRef]
- Liu, Y.; Cai, S.; He, X.; He, X.; Yue, T. Construction of a Food Safety Evaluation System Based on the Factor Analysis of Mixed Data Method. Foods 2024, 13, 2680. [Google Scholar] [CrossRef]
- Guo, Z.; Jayan, H. Fast Nondestructive Detection Technology and Equipment for Food Quality and Safety. Foods 2023, 12, 3744. [Google Scholar] [CrossRef]
- Xu, J.; Liu, Y.B.; Luo, X.Q.; Li, Y.J.; Xing, Y.Q.; Huang, K.J. Visual self-powered platform for ultrasensitive heavy metal detection designed on graphdiyne/graphene heterojunction and DNAzyme-triggered DNA circuit strategy. Chem. Eng. J. 2024, 485, 150151. [Google Scholar] [CrossRef]
- Li, X.C.; Liu, X.Y. A Sensitive Probe of Meso-Cyanophenyl Substituted BODIPY Derivative as Fluorescent Chemosensor for the Detection of Multiple Heavy Metal Ions. J. Fluoresc. 2024, 35, 1089–1098. [Google Scholar] [CrossRef]
- Li, H.H.; Haruna, S.A.; Sheng, W.; Bei, Q.Y.; Ahmad, W.; Zareef, M.; Chen, Q.S.; Ding, Z. SERS-activated platforms for chemical contaminants in food: Probes, encoding methods, and detection. TrAC Trends Anal. Chem. 2023, 169, 117365. [Google Scholar] [CrossRef]
- Hua, Z.; Yu, T.; Liu, D.H.; Xianyu, Y.L. Recent advances in gold nanoparticles-based biosensors for food safety detection. Biosens. Bioelectron. 2021, 179, 113076. [Google Scholar] [CrossRef]
- Yu, H.; Zhao, Q. Competitive fluorescence assay for Cd2+based on aptamer structure-switching. Microchem. J. 2023, 194, 109348. [Google Scholar] [CrossRef]
- Wang, Y.; Zhai, H.G.; Guo, Q.; Zhang, Y.H.; Sun, X.; Guo, Y.M.; Yang, Q.Q.; Zhang, Y.Y. Shared hairpin structure electrochemical aptasensor based on ZrO2@Ni/Co-MOFs@AuNPs for dual-target detection of Cd2+and S. aureus. Sens. Actuators B Chem. 2023, 396, 134648. [Google Scholar] [CrossRef]
- Feng, S.B.; Yang, L.J.; Yan, L.L.; Zhu, M.; Xu, H.L.; Li, G.F. Rapid detection of heavy metal ions based on a novel BMIMPF6-MWCNTs/GCE sensor and square wave voltammetry in actual water samples. J. Appl. Electrochem. 2024, 10, 1797–1806. [Google Scholar] [CrossRef]
- Duan, S.; Wu, X.Y.; Gong, Z.Y.; Wang, J.H.; Liu, X.D.; Wang, Q.; Wang, Y.L.; Dai, H. Curcumin-based ratiometric electrochemical sensing interface for the detection of Cd2+ and Pb2+ in grain products. Colloids Surf. A Physicochem. Eng. Asp. 2024, 684, 133125. [Google Scholar] [CrossRef]
- Yan, J.; Liu, R.; Shi, J.; Yang, W.; Jiang, S.; Zhang, Z.; Jiang, M. Ultra-sensitive, multi-component, and real-time detection of heavy metal ions via AgNPs@Cu-TCPP(Pt) sensitized strong coupling plasmonic sensor. Chem. Eng. J. 2025, 514, 162772. [Google Scholar] [CrossRef]
- Wang, R.; Cao, Y.; Qu, H.; Wang, Y.; Zheng, L. Label-free detection of Cu(II) in fish using a graphene field-effect transistor gated by structure-switching aptamer probes. Talanta 2022, 237, 122965. [Google Scholar] [CrossRef]
- Dong, Y.; Lee, A.; Ban, D.K.; Wang, K.; Bandaru, P. Femtomolar Level-Specific Detection of Lead Ions in Aqueous Environments, Using Aptamer-Derivatized Graphene Field-Effect Transistors. ACS Appl. Nano Mater. 2023, 6, 2228–2235. [Google Scholar] [CrossRef]
- Ye, H.; Yang, Z.X.; Khan, I.M.; Niazi, S.; Guo, Y.X.; Wang, Z.P.; Yang, H.S. Split aptamer acquisition mechanisms and current application in antibiotics detection: A short review. Crit. Rev. Food Sci. Nutr. 2023, 63, 9098–9110. [Google Scholar] [CrossRef]
- Wei, L.K.; Zhu, D.Z.; Cheng, Q.Y.; Gao, Z.H.; Wang, H.L.; Qiu, J.Q. Aptamer-Based fluorescent DNA biosensor in antibiotics detection. Food Res. Int. 2024, 179, 114005. [Google Scholar] [CrossRef]
- Mehlhorn, A.; Rahimi, P.; Joseph, Y. Aptamer-Based Biosensors for Antibiotic Detection: A Review. Biosensors 2018, 8, 54. [Google Scholar] [CrossRef] [PubMed]
- Zhanel, G.G.; Esquivel, J.; Zelenitsky, S.; Lawrence, C.K.; Adam, H.J.; Golden, A.; Hink, R.; Berry, L.; Schweizer, F.; Zhanel, M.A.; et al. Omadacycline: A Novel Oral and Intravenous Aminomethylcycline Antibiotic Agent. Drugs 2020, 80, 285–313. [Google Scholar] [CrossRef] [PubMed]
- Li, X.X.; Jia, M.D.; Yu, L.C.; Li, Y.J.; He, X.W.; Chen, L.X.; Zhang, Y.K. An ultrasensitive label-free biosensor based on aptamer functionalized two-dimensional photonic crystal for kanamycin detection in milk. Food Chem. 2023, 402, 134239. [Google Scholar] [CrossRef] [PubMed]
- Zhang, W.Q.; Sun, Z.C.; Tian, Y.H.; Mou, Y.T.; Guo, Y.M.; Sun, X.; Li, F.L. Ratiometric fluorescent sensor based on a truncated specific aptamer by MGO-SELEX screening for streptomycin detection. Sens. Actuators B Chem. 2024, 406, 135427. [Google Scholar] [CrossRef]
- Zhong, J.P.; Zhang, X.H.; Xu, J.; Zhou, L.; Zhou, Q.; Zhai, H.Y. A highly sensitive luminescent aptasensor utilizing MOF-74-co encapsulation of luminol in a “turn-on” mode for streptomycin detection. Food Chem. 2024, 458, 140306. [Google Scholar] [CrossRef]
- Guo, Q.; Li, S.Q.; Wu, C.H.; Deng, X.Y.; Zhang, B.J.; Xu, Y.; Luo, H.; Yu, H.M.; Huang, K. A dual-modal photothermal and smartphone colorimetric aptamer sensing platform for tobramycin assay. Microchem. J. 2024, 197, 109848. [Google Scholar] [CrossRef]
- Hu, R.; Xu, B.F.; Xue, Y.D.; Xu, Z.Z.; Wang, A.J.; Mei, L.P.; Song, P.; Feng, J.J. Tailoring enzymatic loading capacity on CdS nanorods@ZnIn2S4 nanosheets 1D/2D heterojunctions: Toward ultrasensitive photoelectrochemical bioassay of tobramycin. Chemosphere 2023, 316, 137808. [Google Scholar] [CrossRef]
- de-los-Santos-Alvarez, N.; Lobo-Castañón, M.J.; Miranda-Ordieres, A.J.; Tuñón-Blanco, P. Modified-RNA aptamer-based sensor for competitive impedimetric assay of neomycin B. J. Am. Chem. Soc. 2007, 129, 3808–3809. [Google Scholar] [CrossRef]
- Khavani, M.; Izadyar, M.; Housaindokht, M.R. Theoretical design and experimental study on the gold nanoparticles based colorimetric aptasensors for detection of neomycin B. Sens. Actuators B Chem. 2019, 300, 126947. [Google Scholar] [CrossRef]
- Ramalingam, S.; Collier, C.M.; Singh, A. A Paper-Based Colorimetric Aptasensor for the Detection of Gentamicin. Biosensors 2021, 11, 29. [Google Scholar] [CrossRef]
- Wang, S.; Sun, M.; Zhang, Y.; Ji, H.; Gao, J.; Song, S.; Sun, J.; Liu, H.; Zhang, Y.; Han, L. Ultrasensitive Antibiotic Perceiving Based on Aptamer-Functionalized Ultraclean Graphene Field-Effect Transistor Biosensor. Anal. Chem. 2022, 94, 14785–14793. [Google Scholar] [CrossRef]
- Mukherjee, P.; Sen, S.; Das, A.; Kundu, S.; RoyChaudhuri, C. Graphene FET biochip on PCB reinforced by machine learning for ultrasensitive parallel detection of multiple antibiotics in water. Biosens. Bioelectron. 2025, 271, 117023, ISSN 0956-5663. [Google Scholar] [CrossRef] [PubMed]
- Ahmed, S.R.; Sherazee, M.; Das, P.; Shalauddin, M.; Akhter, S.; Basirun, W.J.; Srinivasan, S.; Rajabzadeh, A.R. Electrochemical assisted enhanced nanozymatic activity of functionalized borophene for H2O2 and tetracycline detection. Biosens. Bioelectron. 2024, 246, 115857. [Google Scholar] [CrossRef] [PubMed]
- Kim, C.H.; Lee, L.P.; Min, J.R.; Lim, M.W.; Jeong, S.H. An indirect competitive assay-based aptasensor for detection of oxytetracycline in milk. Biosens. Bioelectron. 2014, 51, 426–430. [Google Scholar] [CrossRef] [PubMed]
- Guan, F.K.; Dong, H.W.; Xiang, Y.D.; Zhang, M.; Huang, J.C.; Wang, G.J.; Shen, Z.; Xu, D.Y.; Sun, X.; Guo, Y.M.; et al. Novel Electrochemical Aptasensor Based on Floral Zinc Oxide/Reduced Graphene Oxide/Colloidal Gold Composite for the Detection of Tetracycline, Doxycycline and Oxytetracycline in Milk. J. Electrochem. Soc. 2022, 169, 127501. [Google Scholar] [CrossRef]
- Yang, S.; Li, C.X.; Zhan, H.Y.; Liu, R.; Chen, W.L.; Wang, X.L.; Xu, K.X. A label-free fluorescent biosensor based on specific aptamer-templated silver nanoclusters for the detection of tetracycline. J. Nanobiotechnol. 2023, 21, 22. [Google Scholar] [CrossRef]
- Dou, X.Z.; Wu, Q.L.; Luo, S.J.; Yang, J.; Dong, B.L.; Wang, L.; Qu, H.; Zheng, L. A miniaturized biosensor for rapid detection of tetracycline based on a graphene field-effect transistor with an aptamer modified gate. Talanta 2024, 271, 125702. [Google Scholar] [CrossRef]
- Dou, L.P.; Lu, D.Q.; Guo, H.Y.; Liu, Z.; Sun, J.L.; Meng, L.X.; Xiao, K. Novel self-powered anti-interference photoelectrochemical sensor via zirconium porphyrin-based metal-organic framework as multifunctional signal label for oxytetracycline detection in food and environment. Chem. Eng. J. 2024, 496, 153979. [Google Scholar] [CrossRef]
- Flauzino, J.M.R.; Nalepa, M.A.; Chronopoulos, D.D.; Sedajová, V.; Panácek, D.; Jakubec, P.; Kührová, P.; Pykal, M.; Banás, P.; Panácek, A.; et al. Click and Detect: Versatile Ampicillin Aptasensor Enabled by Click Chemistry on a Graphene-Alkyne Derivative. Small 2023, 19, 2207216. [Google Scholar] [CrossRef]
- Ye, T.; Xu, Y.M.; Chen, H.H.; Yuan, M.; Cao, H.; Hao, L.L.; Wu, X.X.; Yin, F.Q.; Xu, F. A trivalent aptasensor by using DNA tetrahedron as scaffold for label-free determination of antibiotics. Biosens. Bioelectron. 2024, 251, 116127. [Google Scholar] [CrossRef]
- Climent, E.; Rurack, K. Combining Electrochemiluminescence Detection with Aptamer-Gated Indicator Releasing Mesoporous Nanoparticles Enables ppt Sensitivity for Strip-Based Rapid Tests. Angew. Chem. Int. Ed. 2021, 60, 26287–26297. [Google Scholar] [CrossRef] [PubMed]
- Hu, M.J.; Dong, J.W.; Wang, H.F.; Huang, J.C.; Geng, L.J.; Liu, M.Y.; Tao, C.; Liu, J.J.; Chen, X.F.; Ahmed, M.B.M.; et al. Novel ratiometric electrochemical aptasensor based on broad-spectrum aptamer recognition for simultaneous detection of penicillin antibiotics in milk. Food Chem. 2024, 456, 139946. [Google Scholar] [CrossRef] [PubMed]
- Li, Z.K.; Wang, B.; Dong, Y.X.; Jie, G.F. A multi-modal biosensing platform based on Ag-ZnIn2S4@Ag-Pt nanosignal probe-sensitized UiO-66 for ultra-sensitive detection of penicillin. Food Chem. 2024, 444, 138665. [Google Scholar] [CrossRef] [PubMed]
- Bao, X.L.; Wang, S.Y.; Hao, Q.F.; Bai, Y.; Li, S.Y.; Zhang, S.; Zhang, L.; Kang, X.X.; Lyu, M.; Wang, S.J. Enrofloxacin Rapid Detection in Aquatic Foods: Based on DNA Aptamer Sensor. Foods 2024, 13, 941. [Google Scholar] [CrossRef]
- Das, S.; Sil, S.; Pal, S.K.; Kula, P.; Roy, S.S. Label-free liquid crystal-based optical detection of norfloxacin using an aptamer recognition probe in soil and lake water. Analyst 2024, 149, 3828–3838. [Google Scholar] [CrossRef]
- Wu, W.W.; Hassan, M.M.; Ding, X.D.; Wu, J.Z.; Ouyang, Q.; Chen, Q.S. A novel acetylcholinesterase regulated upconversion-FITC fluorescence aptasensor for supersensitive detection of sulfadiazine in aquatic products. Microchem. J. 2023, 191, 108765. [Google Scholar] [CrossRef]
- Yadav, S.K.; Agrawal, B.; Chandra, P.; Goyal, R.N. In vitro chloramphenicol detection in a Haemophilus influenza model using an aptamer-polymer based electrochemical biosensor. Biosens. Bioelectron. 2014, 55, 337–342. [Google Scholar] [CrossRef]
- Jiang, J.H.; Wu, T.; Wei, M.; Jiang, D.; Jiang, L.; Shan, X.L.; Wang, W.C.; Shiigi, H.; Chen, Z.D. Self-powered photoelectrochemical sensing for sensitive detection of chloramphenicol based on sulfur-vacancy engineered MoS2 nanoribbons/ plasmonic Ti3C2 MXene with continual injection of photoinduced electrons. J. Environ. Chem. Eng. 2024, 12, 112067. [Google Scholar] [CrossRef]
- Chen, J.A.; Jin, Y.T.; Ren, T.T.; Wang, S.; Wang, X.H.; Zhang, F.Y.; Tang, Y.W. A novel terbium (III) and aptamer-based probe for label-free detection of three fluoroquinolones in honey and water samples. Food Chem. 2022, 386, 132751. [Google Scholar] [CrossRef]
- Li, X.M.; Yang, Z.H.; Waniss, M.; Liu, X.H.; Wang, X.Q.; Xu, Z.L.; Lei, H.T.; Liu, J.W. Multiplexed SELEX for Sulfonamide Antibiotics Yielding a Group-Specific DNA Aptamer for Biosensors. Anal. Chem. 2023, 95, 16366–16373. [Google Scholar] [CrossRef]
- Feng, H.; Luo, M.; Zhu, G.N.; Mokeira, K.D.; Yang, Y.X.; Lv, Y.X.; Tan, Q.; Lei, X.W.; Zeng, H.; Cheng, H.F.; et al. A facile electrochemical aptasensor for chloramphenicol detection based on synergistically photosensitization enhanced by SYBR Green I and MoS2. J. Colloid Interface Sci. 2024, 672, 236–243. [Google Scholar] [CrossRef]
- Tauchen, J.; Jurášek, M.; Huml, L.; Rimpelová, S. Medicinal Use of Testosterone and Related Steroids Revisited. Molecules 2021, 26, 1032. [Google Scholar] [CrossRef] [PubMed]
- Bereshchenko, O.; Bruscoli, S.; Riccardi, C. Glucocorticoids, Sex Hormones, and immunity. Front. Immunol. 2018, 9, 1332. [Google Scholar] [CrossRef] [PubMed]
- Rubin, E.J.; Baden, L.R.; Morrissey, S. Audio Interview: Dexamethasone and COVID-19. N. Engl. J. Med. 2020, 383, e52. [Google Scholar] [CrossRef] [PubMed]
- Qin, Y.D.; Qin, Y.A.; Bubiajiaer, H.; Chen, F.X.; Yao, J.; Zhang, M.W. Engineering constructed of high selectivity dexamethasone aptamer based on truncation and mutation technology. Front. Bioeng. Biotechnol. 2022, 10, 994711. [Google Scholar] [CrossRef]
- Sun, X.; Liu, M.; Liu, H.; Li, L.; Ding, Y. A molecularly imprinted electrochemical aptasensor-based dual recognition elements for selective detection of dexamethasone. Talanta 2024, 277, 126404. [Google Scholar] [CrossRef]
- Li, Y.; Liu, L.; Feng, J.; Ren, X.; Zhang, Y.; Yan, T.; Liu, X.; Wei, Q. A self-powered photoelectrochemical cathodic aptasensor for the detection of 17β-estradiol based on FeOOH/In2S3 photoanode. Biosens. Bioelectron. 2020, 154, 112089. [Google Scholar] [CrossRef]
- Nekooei, S.; Ghaedi, M.; Baezzat, M.R.; Tashkhourian, J.; Sillanpää, M. Electrochemical aptasensor for determination of testosterone using an aptamer-nanogold-metal-organic framework-ionic liquid modified carbon paste electrode. Anal. Biochem. 2025, 702, 115827. [Google Scholar] [CrossRef]
- Babić Leko, M.; Gunjača, I.; Pleić, N.; Zemunik, T. Environmental Factors Affecting Thyroid-Stimulating Hormone and Thyroid Hormone Levels. Int. J. Mol. Sci. 2021, 22, 6521. [Google Scholar] [CrossRef]
- Zou, L.; Liu, Y.; Liu, J.W. Capture-SELEX of DNA aptamers for label-free detection of epinephrine and norepinephrine in urine. Biosens. Bioelectron. 2025, 279, 117392. [Google Scholar] [CrossRef]
- Fredj, Z.; Marvi, F.; Ullah, F.; Sawan, M. A wearable electrochemical aptasensor based MOF on MOF heterostructure for multi-neurotransmitters monitoring. Microchim. Acta 2025, 192, 384. [Google Scholar] [CrossRef] [PubMed]
- Lu, L.X.; Shao, X.Y.; Gao, J.W.; Song, B.C.; Ding, L.H.; Zhou, J.H. Surface plasmon field enhanced upconversion luminescence for the screening and detection of phenolic environmental estrogens. Food Chem. 2023, 413, 135606. [Google Scholar] [CrossRef] [PubMed]
- Yao, L.; Zhi, J.; Wang, W.; Li, Q.; Jiang, D.; Chen, X.; Chen, Z. An efficient ECL aptasensor based on resonance energy transfer between NCDs@Ag3PO4 and Cu-doped Cu: Eu MOF for the detection of diethylstilbestrol. Bioelectrochemistry 2025, 165, 108987. [Google Scholar] [CrossRef] [PubMed]
- Ma, Y.; Liu, H.; Wu, J.; Yuan, L.; Wang, Y.; Du, X.; Wang, R.; Marwa, P.W.; Petlulu, P.; Chen, X.; et al. The adverse health effects of bisphenol A and related toxicity mechanisms. Environ. Res. 2019, 176, 108575. [Google Scholar] [CrossRef]
- Wu, S.; Liu, S.; Wang, Z.; Chen, Y.; Zhao, G. Comprehensive analysis of bisphenol analogues in complex water using a group-targeting aptamer engineered by base mutation. J. Hazard. Mater. 2023, 460, 132427. [Google Scholar] [CrossRef]
- Li, N.; Ren, C.; Hu, Q.; Wang, B.; Yang, Z.; Xiao, L.; Guan, T. Multiplex aptamer cluster detection platform and systems toxicology study for 17β-estradiol, bisphenol A, and diethylstilbestrol. Food Chem. 2025, 463, 141395. [Google Scholar] [CrossRef]
- Zhang, Y.; Han, Y.; Liu, Z.; Fan, L.; Guo, Y. Colorimetric and electrochemical assay for dual-mode detection of di-2-ethylhexyl phthalate based on hemin-graphene nanocomposites. Microchem. J. 2023, 191, 108788. [Google Scholar] [CrossRef]
- Zhang, L.; Xu, X.; Cao, L.; Zhu, Z.; Ding, Y.; Jiang, H.; Li, B.; Liu, J. Multi-aptamer-mediated hairpin allosteric and aptamer-assisted CRISPR system for detection of S. pneumoniae and S. aureus. Microchim. Acta 2023, 191, 29. [Google Scholar] [CrossRef]
- Shi, X.N.; Zhang, J.; Ding, Y.K.; Li, H.; Yao, S.; Hu, T.T.; Zhao, C.; Wang, J. Ultrasensitive detection platform for Staphylococcus aureus based on DNAzyme tandem blocking CRISPR/Cas12a system. Biosens. Bioelectron. 2024, 264, 116671. [Google Scholar] [CrossRef]
- Tao, Y.; Liu, Q.; Cheng, N.T. Sea hedgehog-inspired surface-enhanced Raman scattering biosensor probe for ultrasensitive determination of Staphylococcus aureus in food supplements. Biosens. Bioelectron. 2024, 252, 116146. [Google Scholar] [CrossRef]
- Zhao, W.; Yang, S.; Zhang, D.; Zhou, T.; Huang, J.; Gao, M.; Jiang, Y.; Liu, Y.; Yang, J. Ultrasensitive dual-enhanced sandwich strategy for simultaneous detection of Escherichia coli and Staphylococcus aureus based on optimized aptamers-functionalized magnetic capture probes and graphene oxide-Au nanostars SERS tags. J. Colloid Interface Sci. 2023, 634, 651–663. [Google Scholar] [CrossRef]
- Xie, J.L.; Jiang, H.; Zhao, Y.Q.; Zhong, M.; Jin, X.R.; Zhu, Z.X.; Li, B.L.; Guo, J.L.; Zhang, L.M.; Liu, J.B. Aptamer-based DNA-catalyzed amplification strategy for sensitive fluorescence resonance energy transfer detection of Acinetobacter baumannii. Talanta 2023, 255, 124212. [Google Scholar] [CrossRef]
- Zhang, Y.; Xing, H.; Li, R.; Andersson, J.; Bozdogan, A.; Strassl, R.; Draphoen, B.; Lindén, M.; Henkel, M.; Knippschild, U.; et al. Specific gFET-Based Aptasensors for Monitoring of Microbiome Quality: Quantification of the Enteric Health-Relevant Bacterium Roseburia Intestinalis. Adv. Healthc. Mater. 2025, 14, e2403827. [Google Scholar] [CrossRef]
- Yu, M.D.; Zhang, X.H.; Zhang, X.; Zahra, Q.U.; Huang, Z.H.; Chen, Y.; Song, C.X.; Song, M.; Jiang, H.J.; Luo, Z.F.; et al. An electrochemical aptasensor with N protein binding aptamer-complementary oligonucleotide as probe for ultra-sensitive detection of COVID-19. Biosens. Bioelectron. 2022, 213, 114436. [Google Scholar] [CrossRef]
- Shrikrishna, N.S.; Halder, S.; Kesarwani, V.; Nagamani, K.; Gandhi, S. Unveiling the Potential: High-Affinity aptamers for point of care detection of SARS-CoV-2 RBD protein and it’s validation in clinical samples. Chem. Eng. J. 2024, 493, 152841. [Google Scholar] [CrossRef]
- Song, T.; Sun, M.; Zhang, J.L.; Lu, Y.Z.; Wan, S.; Yang, C.Y.; Song, Y.L. Molecular Crowding Modulates SARS-CoV-2 Aptamer Affinity. Small Struct. 2023, 6, 2300089. [Google Scholar] [CrossRef]
- Wu, Q.; Luo, S.; Wang, L.; Dong, B.; Qu, H.; Zheng, L. Detection of SARS-CoV-2 and noroviruses in cold-chain food samples using aptamer-functionalized graphene field-effect transistors. Sens. Diagn. 2024, 3, 1947–1956. [Google Scholar] [CrossRef]
- Tseng, Y.T.; Wang, C.H.; Chang, C.P.; Lee, G.B. Integrated microfluidic system for rapid detection of influenza H1N1 virus using a sandwich-based aptamer assay. Biosens. Bioelectron. 2016, 82, 105–111. [Google Scholar] [CrossRef] [PubMed]
- Hushegyi, A.; Pihíková, D.; Bertok, T.; Adam, V.; Kizek, R.; Tkac, J. Ultrasensitive detection of influenza viruses with a glycan-based impedimetric biosensor. Biosens. Bioelectron. 2016, 79, 644–649. [Google Scholar] [CrossRef]
- Wang, M.; Chen, J.J.; Zhang, Z.L. Highly-Efficient Selection of Aptamers for Quantitative Fluorescence Detecting Multiple IAV Subtypes. Anal. Chem. 2024, 96, 15347–15354. [Google Scholar] [CrossRef]
- Ruslinda, A.R.; Tanabe, K.; Ibori, S.; Wang, X.F.; Kawarada, H. Effects of diamond-FET-based RNA aptamer sensing for detection of real sample of HIV-1 Tat protein. Biosens. Bioelectron. 2013, 40, 277–282. [Google Scholar] [CrossRef] [PubMed]
- Chekin, F.; Bagga, K.; Subramanian, P.; Jijie, R.; Singh, S.K.; Kurungot, S.; Boukherroub, R.; Szunerits, S. Nucleic aptamer modified porous reduced graphene oxide/MoS2 based electrodes for viral detection: Application to human papillomavirus (HPV). Sens. Actuators B Chem. 2018, 262, 991–1000. [Google Scholar] [CrossRef]
- Aspermair, P.; Mishyn, V.; Bintinger, J.; Happy, H.; Bagga, K.; Subramanian, P.; Knoll, W.; Boukherroub, R.; Szunerits, S. Reduced graphene oxide-based field effect transistors for the detection of E7 protein of human papillomavirus in saliva. Anal. Bioanal. Chem. 2021, 413, 779–787. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.Y.; Yang, J.Y.; Tang, L.; Luo, L.H.; Chen, C.Y.; Gong, H.; Cai, C.Q. Specific determination of HBV using a viral aptamer molecular imprinting polymer sensor based on ratiometric metal organic framework. Microchim. Acta 2021, 188, 221. [Google Scholar] [CrossRef]
- Kim, D.T.H.; Bao, D.T.; Park, H.; Ngoc, N.M.; Yeo, S.J. Development of a novel peptide aptamer-based immunoassay to detect Zika virus in serum and urine. Theranostics 2018, 8, 3629–3642. [Google Scholar] [CrossRef]
- Khoris, I.M.; Nasrin, F.; Chowdhury, A.D.; Park, E.Y. Advancement of dengue virus NS1 protein detection by 3D-nanoassembly complex gold nanoparticles utilizing competitive sandwich aptamer on disposable electrode. Anal. Chim. Acta 2022, 1207, 339817. [Google Scholar] [CrossRef]
- Basso, C.R.; Crulhas, B.P.; Magro, M.; Vianello, F.; Pedrosa, V.A. A new immunoassay of hybrid nanomater conjugated to aptamers for the detection of dengue virus. Talanta 2019, 197, 482–490. [Google Scholar] [CrossRef]
- Wu, Q.; Jezkova, A.; Yuan, Z.; Pavlikova, L.; Dohnal, V.; Kuca, K. Biological degradation of aflatoxins. Drug Metab. Rev. 2009, 41, 1–7. [Google Scholar] [CrossRef]
- Zhu, X.; Tang, J.; Ouyang, X.L.; Liao, Y.B.; Feng, H.P.; Yu, J.F.; Chen, L.; Lu, Y.T.; Yi, Y.Y.; Tang, L. A versatile CuCo@PDA nanozyme-based aptamer-mediated lateral flow assay for highly sensitive, on-site and dual-readout detection of Aflatoxin B1. J. Hazard. Mater. 2024, 465, 133178. [Google Scholar] [CrossRef]
- Liu, M.L.; Li, X.H.; Zhou, S.Y.; Men, D.; Duan, Y.; Liu, H.; Zhao, B.; Huo, D.Q.; Hou, C.J. Ultrasensitive detection of mycotoxins using a novel single-Atom, CRISPR/ Cas12a-Based nanozymatic colorimetric biosensor. Chem. Eng. J. 2024, 497, 154418. [Google Scholar] [CrossRef]
- Ma, L.L.; Jiang, Y.P.; Lu, F.G.; Wang, S.J.; Liu, M.; Liu, F.X.; Huang, L.B.; Li, Y.; Jiao, N.; Jiang, S.Z.; et al. Quantitative Proteomic Analysis of Zearalenone-Induced Intestinal Damage in Weaned Piglets. Toxins 2022, 14, 702. [Google Scholar] [CrossRef]
- Lai, H.H.; Ming, P.T.; Wu, M.Q.; Wang, S.M.; Sun, D.P.; Zhai, H.Y. An electrochemical aptasensor based on P-Ce-MOF@MWCNTs as signal amplification strategy for highly sensitive detection of zearalenone. Food Chem. 2023, 423, 136331. [Google Scholar] [CrossRef]
- Liu, M.; Zhang, J.; Liu, S.; Li, B. A label-free visual aptasensor for zearalenone detection based on target-responsive aptamer-cross-linked hydrogel and color change of gold nanoparticles. Food Chem. 2022, 389, 133078. [Google Scholar] [CrossRef] [PubMed]
- Yan, H.; He, B.; Zhao, R.; Ren, W.; Suo, Z.; Xu, Y.; Xie, D.; Zhao, W.; Wei, M.; Jin, H. Electrochemical aptasensor based on CRISPR/Cas12a-mediated and DNAzyme-assisted cascade dual-enzyme transformation strategy for zearalenone detection. Chem. Eng. J. 2024, 493, 152431. [Google Scholar] [CrossRef]
- Wang, Y.S.; Wang, B.; Wang, P.L.; Hua, Z.; Zhang, S.S.; Wang, X.B.; Yang, X.; Zhang, C. Review of neurotoxicity of T-2 toxin. Mycotoxin Res. 2024, 40, 85–95. [Google Scholar] [CrossRef] [PubMed]
- Ming, P.; Lai, H.; Liu, Y.; Wang, J.; You, F.; Sun, D.; Zhai, H. Aptasensor development for T-2 toxin detection utilizing a dual signal amplification strategy: Synergistic effects of bimetallic oxide (Ce-In)Ox and COFTAPB-DMTP. Sens. Actuator B-Chem. 2023, 396, 134602. [Google Scholar] [CrossRef]
- Zhang, Y.; He, B.; Zhao, R.; Bai, C.; Zhang, Y.; Jin, H.; Wei, M.; Ren, W.; Suo, Z.; Xu, Y. Electrochemical aptasensor based on the target-induced strand displacement strategy-driven for T-2 toxin detection. Sci. Total Environ. 2022, 849, 157769. [Google Scholar] [CrossRef]
- Ren, X.; Chen, J.G.; Gan, X.C.; Song, N.; Yang, X.R.; Zhao, J.X.; Ma, H.M.; Ju, H.X.; Wei, Q. Self-powered photoelectrochemical aptasensor for fumonisin B1 detection based on a Z-scheme ZnIn2S4WO3 photoanode. Biosens. Bioelectron. 2024, 259, 116387. [Google Scholar] [CrossRef]
- Song, N.; Chen, J.; Yang, X.; Ren, X.; Feng, J.; Wang, H.; Ma, H.; Wei, Q. Self-powered photoelectrochemical aptasensor based on in situ topological conversion of Bi2O2S/Bi2S3 for sensitive detection of fumonisin B1. Sens. Actuators B Chem. 2025, 431, 137395. [Google Scholar] [CrossRef]
- Geng, L.J.; Liu, J.J.; Zhang, W.B.; Wang, H.F.; Huang, J.C.; Wang, G.X.; Hu, M.J.; Dong, H.W.; Sun, J.S.; Fang, M.X.; et al. Preparation of dual recognition adsorbents based on molecularly imprinted polymers and aptamer for highly sensitive recognition and enrichment of ochratoxin A. J. Hazard. Mater. 2024, 476, 135112. [Google Scholar] [CrossRef]
- Lv, X.Q.; Foda, M.F.; He, J.L.; Zhou, J.J.; Cai, J. Robust and facile label-free colorimetric aptasensor for ochratoxin A detection using aptamer-enhanced oxidase-like activity of MnO2 nanoflowers. Food Chem. 2023, 401, 134144. [Google Scholar] [CrossRef] [PubMed]
- Tang, J.; Li, Y.; Yao, Y.; Hu, R. Colorimetric/electrochemical dual-mode aptasensor with phosphatase-like ceria nanozyme for fast and ultrasensitive detection of Ochratoxin A. Sens. Actuators B Chem. 2025, 432, 137482. [Google Scholar] [CrossRef]
- Nekrasov, N.; Jaric, S.; Kireev, D.; Emelianov, A.V.; Orlov, A.V.; Gadjanski, I.; Nikitin, P.I.; Akinwande, D.; Bobrinetskiy, I. Real-time detection of ochratoxin A in wine through insight of aptamer conformation in conjunction with graphene field-effect transistor. Biosens. Bioelectron. 2022, 200, 113890. [Google Scholar] [CrossRef] [PubMed]
- Falfushynska, H.; Kasianchuk, N.; Siemens, E.; Henao, E.; Rzymski, P. A Review of Common Cyanotoxins and Their Effects on Fish. Toxics 2023, 11, 118. [Google Scholar] [CrossRef]
- Labine, M.; Gong, Y.; Minuk, G.Y. Long-Term, Low-Dose Exposure to Microcystin-LR Does not Cause or Increase the Severity of Liver Disease in Rodents. Ann. Hepatol. 2017, 16, 959–965. [Google Scholar] [CrossRef]
- Hussain, S.; Al-Tabban, A.; Zourob, M. Aptameric photonic structure-based optical biosensor for the detection of microcystin. Biosens. Bioelectron. 2024, 260, 116413. [Google Scholar] [CrossRef]
- Zhang, J.; Yang, Z.; Chai, B.; Gao, L.; Zhao, J.; Xu, X. An ultrasensitive electrochemical aptasensor for microcystin-LR detection using core-satellite gold nanoparticle/silver nanocluster nanoassemblies as labels for signal amplification. Sens. Actuators B Chem. 2022, 371, 132586. [Google Scholar] [CrossRef]
- Patel, R.N.; Clare, R.H.; Ledsgaard, L.; Nys, M.; Kool, J.; Laustsen, A.H.; Ulens, C.; Casewell, N.R. An in vitro assay to investigate venom neurotoxin activity on muscle-type nicotinic acetylcholine receptor activation and for the discovery of toxin-inhibitory molecules. Biochem. Pharmacol. 2023, 216, 115758. [Google Scholar] [CrossRef]
- Liu, S.; Huo, Y.; Li, G.; Huang, L.; Wang, T.; Gao, Z. Aptamer-controlled reversible colorimetric assay: High-activity bimetallic organic frameworks for the efficient sensing of marine biotoxins. Chem. Eng. J. 2023, 469, 144027. [Google Scholar] [CrossRef]
- Park, J.A.; Kwon, N.; Park, E.; Kim, Y.; Jang, H.; Min, J.; Lee, T. Electrochemical biosensor with aptamer/porous platinum nanoparticle on round-type micro-gap electrode for saxitoxin detection in fresh water. Biosens. Bioelectron. 2022, 210, 114300. [Google Scholar] [CrossRef]
- Chen, Y.X.; Yang, Z.; Nian, B.; Yu, C.L.; Maimaiti, D.; Chai, M.; Yang, X.R.; Zang, X.X.; Xu, D.H. Mechanisms of Neurotoxicity of Organophosphate Pesticides and Their Relation to Neurological Disorders. Neuropsychiatr. Dis. Treat. 2024, 20, 2237–2254. [Google Scholar] [CrossRef]
- Wang, X.; Yang, Y.; Yin, Y.A.; Zeng, N.; Dong, Y.Y.; Liu, J.H.; Wang, L.; Yang, Z.; Yang, C.S. High-Throughput Aptamer Microarrays for Fluorescent Detection of Multiple Organophosphorus Pesticides in Food. Anal. Chem. 2022, 94, 3173–3179. [Google Scholar] [CrossRef] [PubMed]
- Li, J.S.; Yang, F.Z.; Chen, X.F.; Fang, H.G.; Zha, C.Y.; Huang, J.C.; Sun, X.; Ahmed, M.B.M.; Guo, Y.M.; Liu, Y. Dual-ratiometric aptasensor for simultaneous detection of malathion and profenofos based on hairpin tetrahedral DNA nanostructures. Biosens. Bioelectron. 2023, 227, 114853. [Google Scholar] [CrossRef] [PubMed]
- Guo, W.F.; Hu, C.C.; Li, S.; Wei, D.; Zhou, J.; Liu, X.Y.; Chen, H.; Li, S.; Deng, Y. Selection and electrochemical-sensor application of an DNA-aptamer for methyl parathion detection. Anal. Chim. Acta 2023, 1241, 340780. [Google Scholar] [CrossRef] [PubMed]
- Ruankham, W.; Tantimongcolwat, T.; Phopin, K.; Bausells, J.; Hangouët, M.; Martin, M.; Zine, N.; Errachid, A. Split aptamers immobilized array microelectrodes for detection of chlorpyrifos pesticide using electrochemical impedance spectroscopy. Sens. Actuator B-Chem. 2022, 372, 132614. [Google Scholar] [CrossRef]
- Li, P.S.; Sun, J.S.; Wang, H.F.; Huang, J.C.; Geng, L.J.; Dong, H.W.; Li, D.H.; Li, C.Q.; Fang, M.X.; Zhang, X.; et al. Novel electrochemiluminescence sensing platform for ultrasensitive detection of malathion residue in tea based on SiO2NSs doped Luminol/AgNPs as a signal amplification strategy. J. Hazard. Mater. 2024, 477, 135358. [Google Scholar] [CrossRef]
- Li, Y.B.; Li, T.T.; Chang, J.J.; Liu, Y.X.; Chen, J.; Nie, Z.Y.; Xie, J.W. Dual dye-labeled aptamers for detection of dichlorvos using ratiometric fluorescence resonance energy transfer. Anal. Chim. Acta 2024, 1329, 343158. [Google Scholar] [CrossRef]
- Costas-Ferreira, C.; Faro, L.R.F. Neurotoxic Effects of Neonicotinoids on Mammals: What Is There beyond the Activation of Nicotinic Acetylcholine Receptors?—A Systematic Review. Int. J. Mol. Sci. 2021, 22, 8413. [Google Scholar] [CrossRef]
- Fan, L.F.; Zhao, G.H.; Shi, H.J.; Liu, M.C.; Li, Z.X. A highly selective electrochemical impedance spectroscopy-based aptasensor for sensitive detection of acetamiprid. Biosens. Bioelectron. 2013, 43, 12–18. [Google Scholar] [CrossRef]
- Bor, G.; Man, E.; Ugurlu, O.; Ceylan, A.E.; Balaban, S.; Durmus, C.; Gumus, Z.P.; Evran, S. Timur, in vitro Selection of Aptamer for Imidacloprid Recognition as Model Analyte and Construction of a Water Analysis Platform. Electroanalysis 2020, 32, 1922–1929. [Google Scholar] [CrossRef]
- Li, S.H.; Deng, Y.; Pang, C.H.; Ma, X.H.; Wu, Y.W.; Wang, M.Y.; Li, J.P.; Xu, Z.; Zhang, L.M. An electrochemiluminescence sensor based on Fe/Zn-BTC@C-dots sensitisation for thiamethoxam detection. Sens. Actuator B-Chem. 2023, 394, 134415. [Google Scholar] [CrossRef]
- Yan, X.; Kong, D.S.; Jin, R.; Zhao, X.; Li, H.X.; Liu, F.M.; Lin, Y.H.; Lu, G.Y. Fluorometric and colorimetric analysis of carbamate pesticide via enzyme-triggered decomposition of Gold nanoclusters-anchored MnO2 nanocomposite. Sens. Actuator B-Chem. 2019, 290, 640–647. [Google Scholar] [CrossRef]
- Liu, Y.; Yang, G.J.; Li, T.T.; Deng, Y.; Chen, Z.; He, N.Y. Selection of a DNA aptamer for the development of fluorescent aptasensor for carbaryl detection. Chin. Chem. Lett. 2021, 32, 1957–1962. [Google Scholar] [CrossRef]
- Li, S.H.; Liu, C.H.; Han, B.J.; Luo, J.H.; Yin, G.H. An electrochemiluminescence aptasensor switch for aldicarb recognition via ruthenium complex-modified dendrimers on multiwalled carbon nanotubes. Microchim. Acta 2017, 184, 1669–1675. [Google Scholar] [CrossRef]
- Li, S.H.; Li, J.P.; Luo, J.H.; Xu, Z.; Ma, X.H. A microfluidic chip containing a molecularly imprinted polymer and a DNA aptamer for voltammetric determination of carbofuran. Microchim. Acta 2018, 185, 295. [Google Scholar] [CrossRef] [PubMed]
- Soderlund, D.M.; Bloomquist, J.R. Neurotoxic actions of pyrethroid insecticides. Annu. Rev. Entomol. 1989, 34, 77–96. [Google Scholar] [CrossRef]
- Yang, Y.X.; Tang, Y.; Wang, C.X.; Liu, B.Y.; Wu, Y.G. Selection and identification of a DNA aptamer for ultrasensitive and selective detection of λ-cyhalothrin residue in food. Anal. Chim. Acta 2021, 1179, 338837. [Google Scholar] [CrossRef]
- Wang, L.; Haruna, S.A.; Ahmad, W.; Wu, J.Z.; Chen, Q.S.; Ouyang, Q. Tunable multiplexed fluorescence biosensing platform for simultaneous and selective detection of paraquat and carbendazim pesticides. Food Chem. 2022, 388, 132950. [Google Scholar] [CrossRef]
- Liu, Y.Y.; Liu, B.Y.; Xia, L.; Yu, H.Y.; Wang, Q.D.; Wu, Y.E. Cationic polyelectrolyte as powerful capture molecule in aptamer-based chromatographic strip for rapid visual detection of paraquat residue in agricultural products. Sens. Actuator B-Chem. 2022, 368, 132237. [Google Scholar] [CrossRef]
- Park, S.J.; Seo, S.E.; Kim, K.H.; Lee, S.H.; Kim, J.; Ha, S.; Song, H.S.; Lee, S.H.; Kwon, O.S. Real-time monitoring of geosmin based on an aptamer-conjugated graphene field-effect transistor. Biosens. Bioelectron. 2021, 174, 112804, ISSN 0956-5663. [Google Scholar] [CrossRef]
- Aziz, M.; Karboune, S. Natural antimicrobial/antioxidant agents in meat and poultry products as well as fruits and vegetables: A review. Crit. Rev. Food Sci. Nutr. 2018, 58, 486–511. [Google Scholar] [CrossRef]
- Piper, J.D.; Piper, P.W. Benzoate and Sorbate Salts: A Systematic Review of the Potential Hazards of These Invaluable Preservatives and the Expanding Spectrum of Clinical Uses for Sodium Benzoate. Compr. Rev. Food Sci. Food Saf. 2017, 16, 868–880. [Google Scholar] [CrossRef] [PubMed]
- Noori, R.; Farahani, F.; Jun, C.; Aradpour, S.; Bateni, S.M.; Ghazban, F.; Hosseinzadeh, M.; Maghrebi, M.; Naseh, M.R.V.; Abolfathi, S. A non-threshold model to estimate carcinogenic risk of nitrate-nitrite in drinking water. J. Clean. Prod. 2022, 363, 132432. [Google Scholar] [CrossRef]
- Kathwate, L.H.; Chandak, V.S.; Mane, Y.S.; Lokhande, S.D.; Awale, M.B.; Kanwate, A.D. High-performance formaldehyde sensor based on Al-doped ZnO nanorods prepared by chemical bath deposition. Ceram. Int. 2025, 51, 0272–8842. [Google Scholar] [CrossRef]
- Hazman, Ö.; Bozkurt, M.F.; Fidan, A.F.; Uysal, F.E.; Çelik, S. The Effect of Boric Acid and Borax on Oxidative Stress, Inflammation, ER Stress and Apoptosis in Cisplatin Toxication and Nephrotoxicity Developing as a Result of Toxication. Inflammation 2018, 41, 1032–1048. [Google Scholar] [CrossRef]
- Zuo, W.C.; Wu, P.C.; He, W.; Xiao, Q.Q.; Yang, J.; Liu, X.M.; Jiang, H.; Dai, J.J.; Ju, Y.M. A fluorescent and ratiometric colorimetric biosensor for detection of different hazard contaminants in dairy products. Sens. Actuator B-Chem. 2023, 374, 132816. [Google Scholar] [CrossRef]
- Jing, L.; Qin, M.W.; Zhang, X.M.; Song, Y.Z.; Zhang, J.Y.; Xia, X.S.; Gao, K.; Han, Q.Q. A novel borax-specific ssDNA aptamer screened by high-throughput SELEX and its colorimetric assay with aggregation of AuNPs. J. Food Compos. Anal. 2021, 101, 103947. [Google Scholar] [CrossRef]
- Qin, Y.; Shen, J.; Qin, Y.; Hayilati, B.; Yao, J.; Zhang, M. Research progress on the application of aptamer optimization technology and portable sensing devices in food safety detection. Crit. Rev. Food Sci. Nutr. 2024, 3, 6176–6208. [Google Scholar] [CrossRef]
- McKeague, M.; Derosa, M.C. Challenges and opportunities for small molecule aptamer development. J. Nucleic Acids 2012, 2012, 748913. [Google Scholar] [CrossRef]
- Fu, L.; Qian, Y.; Zhou, J.; Zheng, L.; Wang, Y. Fluorescence-based quantitative platform for ultrasensitive food allergen detection: From immunoassays to DNA sensors. Compr. Rev. Food Sci. Food Saf. 2020, 19, 3343–3364. [Google Scholar] [CrossRef]
- Hu, Z.; Wang, Q.; Yang, Z.; Chen, M.; Wu, M.; Liang, H.; Pan, J.; Dong, J.; Zhang, L.; Sun, D. Switch-Type Electrochemiluminescence Aptasensor for AFB1 Detection Based on CoS Quantum Dots Encapsulated in Co-LDH and a Ferrocene Quencher. Anal. Chem. 2025, 97, 1366–1373. [Google Scholar] [CrossRef]
- Lichtenberg, J.Y.; Ling, Y.; Kim, S. Non-Specific Adsorption Reduction Methods in Biosensing. Sensors 2019, 19, 2488. [Google Scholar] [CrossRef]
- Choi, S.; Chae, J. Methods of reducing non-specific adsorption in microfluidic biosensors. J. Micromech. Microeng. 2010, 20, 075015. [Google Scholar] [CrossRef]
- Allemailem, K.S.; Almatroudi, A.; Alsahli, M.A.; Basfar, G.T.; Alrumaihi, F.; Rahmani, A.H.; Khan, A.A. Recent advances in understanding oligonucleotide aptamers and their applications as therapeutic agents. 3 Biotech 2020, 10, 551. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Li, Y.; Wang, Y.; Liang, P.; Lai, G. Distance-Regulated Photoelectrochemical Sensor “Signal-On” and “Signal-Off” Transitions for the Multiplexed Detection of Viruses Exposed in the Aquatic Environment. Anal. Chem. 2023, 95, 13922–13931. [Google Scholar] [CrossRef] [PubMed]
- Dias Neves, M.A.; Mendes Pinto, I. AptaShield: A Universal Signal-Transduction System for Fast and High-Throughput Optical Molecular Biosensing. ACS Sens. 2024, 9, 1756–1762. [Google Scholar] [CrossRef]
- Zhang, S.; Zhang, Y.; Ning, Z.; Duan, M.; Lin, X.; Duan, N.; Wang, Z.; Wu, S. Design and application of microfluidics in aptamer SELEX and Aptasensors. Biotechnol. Adv. 2024, 77, 108461. [Google Scholar] [CrossRef]
- Fu, C.; He, Z.; Liu, T.; Luo, Y.; Yi, S.; Chen, X.; Duan, X.; He, J.; Chen, H.; Jiang, M.; et al. Multi-group structure analysis and molecular docking of aptamers and small molecules: A case study of chloramphenicol. Biochem. Biophys. Res. Commun. 2025, 750, 151387. [Google Scholar] [CrossRef]
- Liu, Y.; Yang, J.; Chen, M.; Lu, X.; Wei, Z.; Tang, C.; Yu, P. Recent Advances in Computer-aided Virtual Screening and Docking Optimization for Aptamer. Curr. Top. Med. Chem. 2023, 23, 1985–2000. [Google Scholar] [CrossRef]
- Lee, E.; Kim, E.; Park, T.; Bae, D.-W.; Cha, S.; Kim, T.; Kim, Y. Gold nanoparticle-assisted SELEX as a visual monitoring platform for the development of small molecule-binding DNA aptasensors. Biosens. Bioelectron. 2021, 191, 113468. [Google Scholar] [CrossRef]
- Lee, J.; Park, B.; Ghang, H.; Song, H.; Yang, S.Y. Nano-Protrusive Gold Nanoparticle-Hybridized Polymer Thin Film as a Sensitive, Multipatternable, and Antifouling Biosensor Platform. ACS Appl. Mater. Interfaces 2018, 10, 13397–13405. [Google Scholar] [CrossRef]
- Su, L.; Wang, G.; Zhao, L.; Deng, Y.; Guo, Y.; Xiao, Y.; Wang, H.; Dong, C.; Fan, L. Ultrasensitive monitoring of PCB77 in environmental samples using a visible-driven photoelectrochemical sensing platform coupling with exonuclease I assisted in target recycling. Sci. Total Environ. 2024, 946, 173982. [Google Scholar] [CrossRef]
- Li, L.; Li, J.; Wang, S.; Dong, Y. Dual-mode CRISPR/Cas12a-assisted fluorescent and lateral flow aptasensor based on a newly truncated aptamer for Fumonisin B1 detection. Int. J. Biol. Macromol. 2025, 298, 139950. [Google Scholar] [CrossRef]
- Wang, L.; Zheng, Q.; Zhang, Q.A.; Xu, H.; Tong, J.; Zhu, C.; Wan, Y. Detection of single tumor cell resistance with aptamer biochip. Oncol. Lett. 2012, 4, 935–940. [Google Scholar] [CrossRef]


| Type | Structural Design of Sensor–Bioreceptor Complexes | Target | LOD | Dynamic Range | Time | Ref. |
|---|---|---|---|---|---|---|
| Electrochemical biosensors | Graphene/graphdiyne (GR/GDY) heterojunction-based sensing platform | Heavy metals | 0.005 nM | 0.003–5000 nM | - | [48] |
| FIS-based biosensor incorporating fully 2′-O-methylated RNA aptamers | Antibiotics | - | 0.75–500 µM | 5 min | [68] | |
| Molecularly imprinted electrochemical aptasensor based on dual-recognition elements | Hormonal molecules | 17.9 fM | 10−13–10−5 M | 30 min | [96] | |
| Optical Biosensor | Label-free photonic crystal aptasensor employing a SiO2-Au-ssDNA 2D photonic crystal architecture (2D PC) | Antibiotics | 1.10 pg/mL | 5 pg/mL–5 μg/mL | 45 min | [63] |
| Aptameric photonic structure-based optical biosensor | Algal toxins | 0.88 nM | 3.8 nM–150 nM | 110 min | [147] | |
| Fluorescent sensor | Ratiometric fluorescent aptasensor utilizing AgNCs-SMP@ZIF-8 as the responsive signal and aptamer-functionalized CQDs as the reference | Antibiotics | - | 0.98 nM | 90 s | [64] |
| Novel fluorescent probe leveraging Tb3+-enrofloxacin aptamer coordination | Antibiotics | - | 0.020–0.061 ng/mL | 30 min | [89] | |
| Photoelectrochemical sensors | Novel self-powered anti-interference photoelectrochemical sensor via zirconium porphyrin-based metal–organic (ZPM) framework as multifunctional signal label | Antibiotics | 0.03 pM | 0.1 pM–100 nM | - | [78] |
| Colorimetric sensor | Label-free biosensor constructed using DEX-specific aptamers and gold nanoparticle-modified graphene oxide | Hormonal molecules | - | 20–100 nmol/ml | 1 h | [95] |
| Colorimetric sensor constructed by combining Bor-A01, a high-affinity borax-specific aptamer obtained by SELEX technology screening, and gold nanoparticles (AuNPs) | Preservatives | - | 0.30–0.50 μg/mL | 60 min | [177] | |
| Surface-enhanced Raman scattering biosensor | Surface-enhanced Raman scattering (SERS) biosensor based on gold nanostars (AuNSs) | Bacteria | 1.0 CFU/mL | - | 30 min | [110] |
| Novel surface-enhanced Raman scattering (SERS) sandwich strategy biosensing platform | Bacteria | 10 CFU/mL | - | 55 min | [111] | |
| Electrochemiluminescence aptamer sensor | Electrochemiluminescence aptasensor via ruthenium complex-modified dendrimers on multiwalled carbon nanotubes | Pesticide residues | 9.6 pM | 40 pM–4 nM | 30 min | [164] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wang, C.; Ye, M.; Zhang, X.; Chai, X.; Yu, H.; Liu, B.; Zhang, C.; Wang, Y. Aptamer-Based Biosensors for Rapid Detection and Early Warning of Food Contaminants: From Selection to Field Applications. Molecules 2025, 30, 4332. https://doi.org/10.3390/molecules30224332
Wang C, Ye M, Zhang X, Chai X, Yu H, Liu B, Zhang C, Wang Y. Aptamer-Based Biosensors for Rapid Detection and Early Warning of Food Contaminants: From Selection to Field Applications. Molecules. 2025; 30(22):4332. https://doi.org/10.3390/molecules30224332
Chicago/Turabian StyleWang, Cong, Mengyu Ye, Ximeng Zhang, Xin Chai, Huijuan Yu, Boshi Liu, Chengyu Zhang, and Yuefei Wang. 2025. "Aptamer-Based Biosensors for Rapid Detection and Early Warning of Food Contaminants: From Selection to Field Applications" Molecules 30, no. 22: 4332. https://doi.org/10.3390/molecules30224332
APA StyleWang, C., Ye, M., Zhang, X., Chai, X., Yu, H., Liu, B., Zhang, C., & Wang, Y. (2025). Aptamer-Based Biosensors for Rapid Detection and Early Warning of Food Contaminants: From Selection to Field Applications. Molecules, 30(22), 4332. https://doi.org/10.3390/molecules30224332






