Selective Deoxygenation of Biomass Polyols into Diols
Abstract
1. Introduction
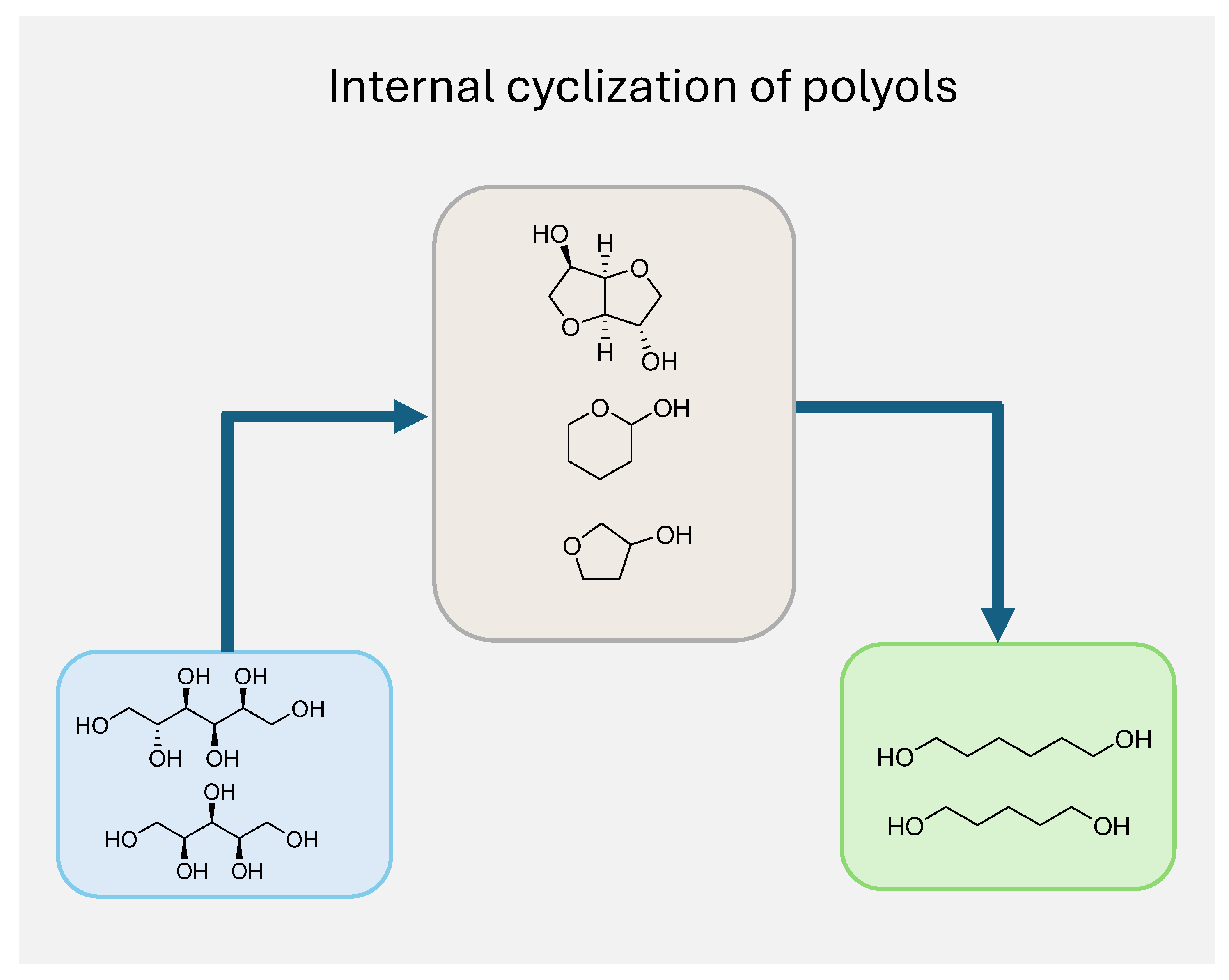
2. Biomass-Derived Polyols: Characteristics and Reactivity
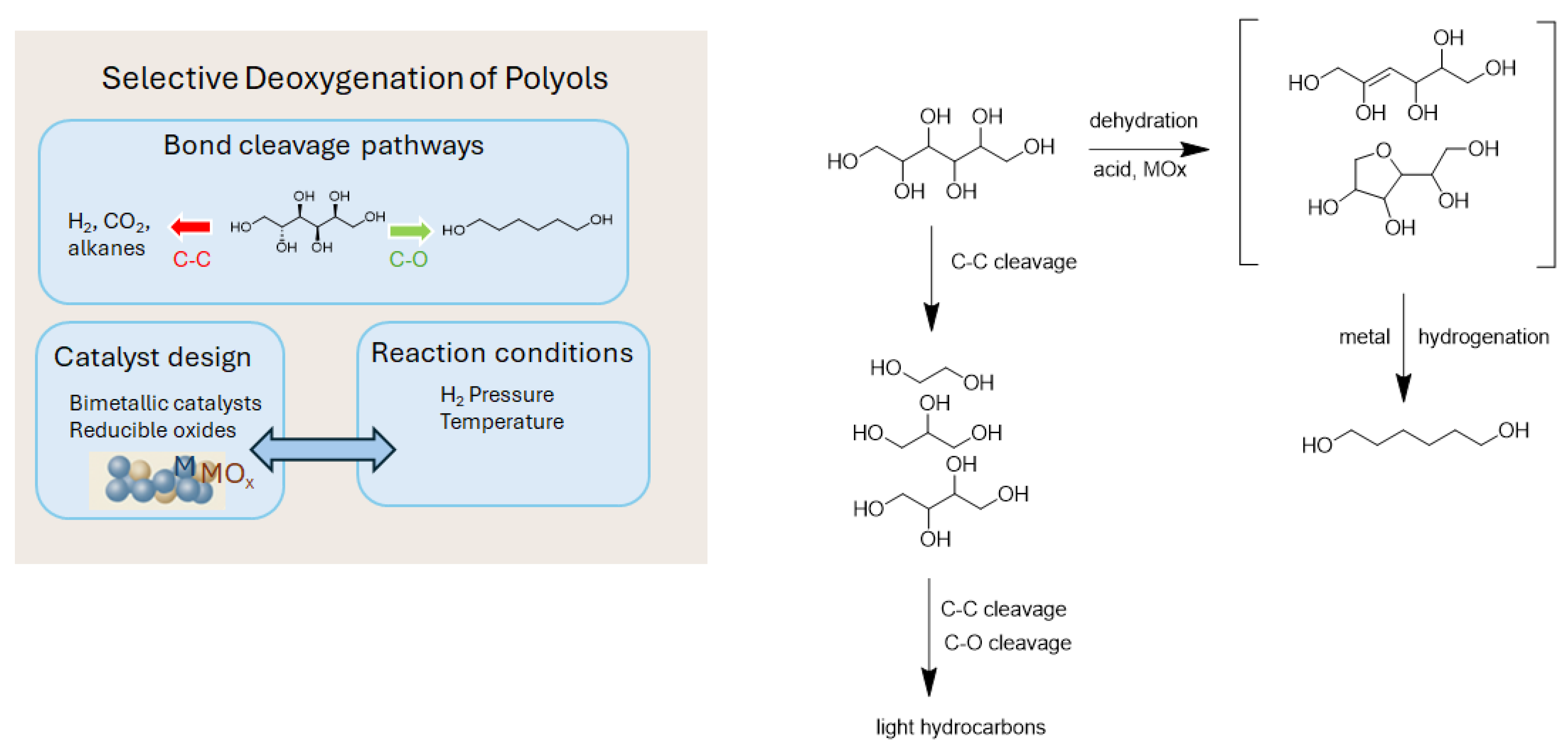
3. Catalytic Deoxygenation of Polyols to Diols
3.1. Glycerol
| Catalyst System | Reaction Conditions | Selectivity to PDs | Reference |
|---|---|---|---|
| Ir–ReOx/TiO2 | 160 °C, 3 MPa H2, aqueous, batch reactor, 4 g (43 mmol) of glycerol in 2 g of water, 150 mg of 4 wt% Ir Ir-ReOx/TiO2; 4 h reaction time; TOF 830 h−1 | up to 70% (1,3-PD) | [43] |
| Pt–WOx/SBA-15 | 210 °C, 0.1 MPa H2, aqueous, fixed bed reactor; 10wt% glycerol in water; 0.5 g of catalyst; WHSV: 1.02 h−1 | 45% (1,3-PD) | [44] |
| Ru NPs | 160 °C, 2 MPa H2, aqueous; batch reactor; 5 wt% of glycerol in water, metal-to-substrate molar ratio of 1:200; 16 h reaction time; | 15% (1,3-PD) | [45] |
| Ni–Mo2C/Al2O3 | 200 °C, 3 MPa H2, aqueous; batch reactor; 95 wt% of glycerol in water, 1.2 g of catalyst; 6 h reaction time; | 61% (1,2 PD) | [47] |
| Pt–WOx/Al2O3 | 240 °C, 5 MPa H2 aqueous; batch reactor; 5 wt% of glycerol in water, 0.3 g of catalyst/g glycerol; 4 h reaction time; | 40% (1,3-PD), | [53] |
| Pt/WOx | 140 °C, 1 MPa H2 aqueous; fixed bed reactor; 5 wt% of glycerol in water; 75 h reaction time; LHSV: 1 h−1 | 46% (1,3-PD), 7% (1,2-PD) | [54] |
| egg-shell Ir/ReOx | 130 °C, 8 MPa H2, aqueous; fixed bed reactor; 5 wt% of glycerol in water, 1.5 g of catalyst; 36 h reaction time | 31% (1,3-PD) | [55] |
| Pt–WOx/r–TiO2 | 150 °C; 4 MPa H2, batch reactor; 30 wt% of glycerol in water, 0.2 g of catalyst; 24 h reaction time | 51.2% (1,3-PD) | [60] |
| Pt/WOx | 160 °C, 5.0 MPa H2, batch reactor; 30 wt% aqueous glycerol; 0.2 g of catalyst; 24 h reaction time | 51% (1,3-PD) | [59] |
3.2. Xylitol
3.3. Sorbitol
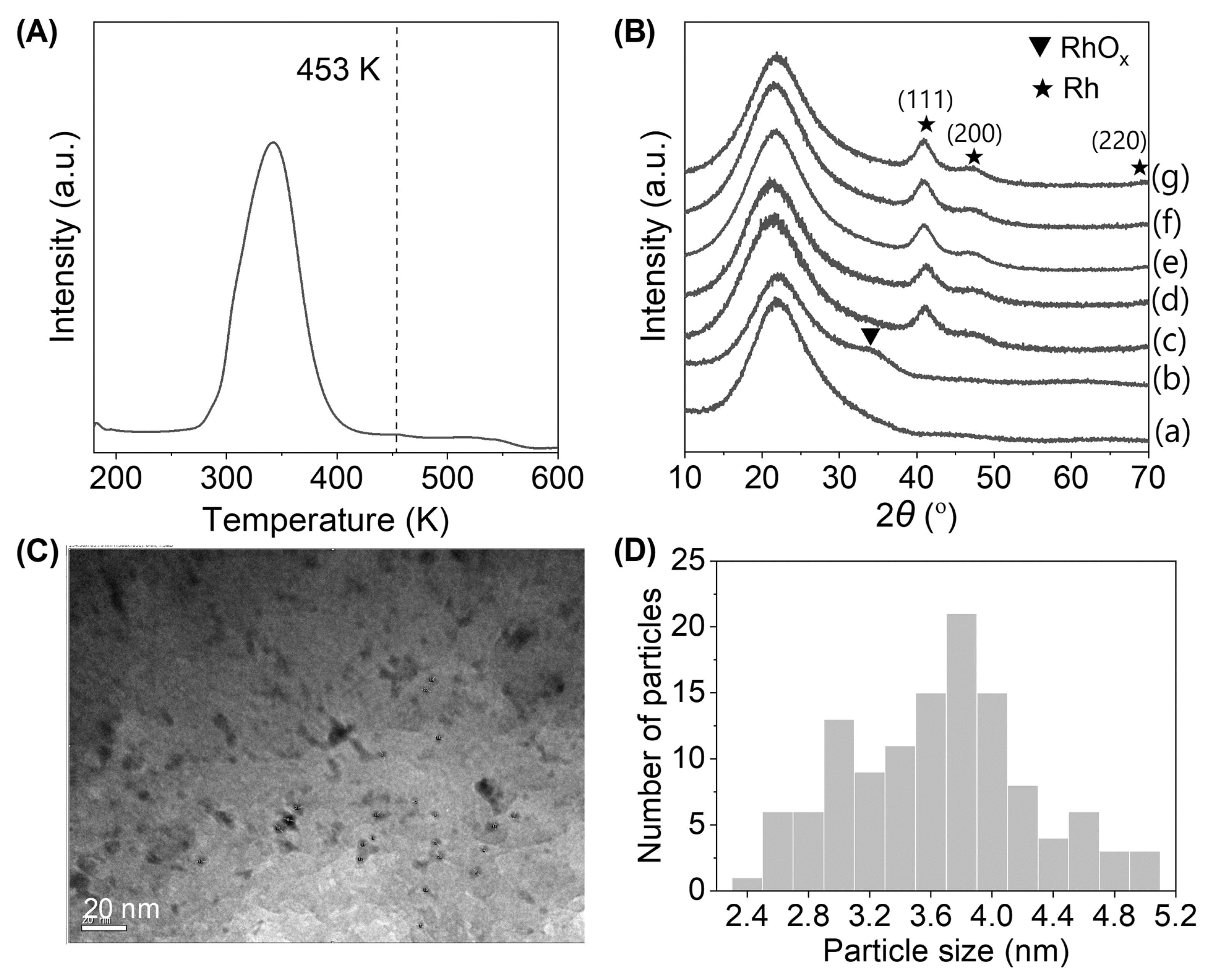
| Catalyst | Conditions | Main Product Selectivity (%) | Reference |
|---|---|---|---|
| Rh–ReOx/ZrO2 | 473 K, 8.0 MPa H2; batch reactor; 5 wt % of sorbitol in water; 0.5 g catalyst | 55 (1,4-sorbitan) | [79] |
| Pd/TiO2 +ZnO + Mg3AlOx | 493 K, 5.0 MPa H2; batch reactor; 10 wt % of sorbitol in water; 1 g catalyst; reaction time 4 h | 42 (1,2-PG) | [68] |
| Pt–Pd (lattice-strained core–shell) | 473 K, 1 MPa N2; batch reactor; 0.3 kmol/m3 of sorbitol in water; 0.1 g catalyst; reaction time 8.5 h | 43 (glycols) | [69] |
| Pt/WOx–SiO2 | 463 K, 8.0 MPa H2; batch reactor; W/Pt = 0.25, molar ratio | 65 (C6 triols and diols) | [70] |
| Rh/SiO2 | 453 K, 8.0 MPa H2, batch reactor; isosorbide: 4 g in 2 g H2O, 0.6 g catalyst, 4 h. | 58 (C5 and C6 diols and triols) | [78] |
| Ni/La2O3/ZrO2 | 493 K, 4.0 MPa H2; batch reactor; 10% sorbitol; catalyst amount = 1 g; 4 h. | 60 (EG + 1,2-PD) | [71] |
| Ni/Mg6Al4Ox | 523 K, 6.0 MPa H2; sorbitol 4 g in 40 mL of water; 0.2 g of catalyst; 6 h | 44 (EG + 1,2-PD) | [72] |
| Ru/WOx supported on N-doped carbon | 473 K, 4.0 MPa H2; batch reactor; 2 g sorbitol in 20 mL of water, 0.1 g catalyst, 0.2 g Ca(OH)2; 6 h | 85 (EG + 1,2-PD) | [73] |
| Ru/C in combination with mixed metal oxides of alkaline earth metals and Ti or Zr | 473 K, 8.0 MPa H2; batch reactor; 0.5 g sorbitol in 10 mL of water, 0.025 g of 5 %Ru/C and 0.225 g of mixed metal oxide; 4 h | 50 (1,2-PD) | [74] |
| Ru on N-doped Carbon | 473 K, 8.0 MPa H2; batch reactor; 0.2 g catalyst; 0.3 g Ca(OH)2, 20 mL of H2O; 4 h | 80 (EG + 1,2-PD) | [75] |
| Cu/AC | 513 K, 5.0 MPa H2; batch reactor; 0.18 M sorbitol, 0.08 g Cu, 0.2 g Ca(OH)2, 20 ml H2O; 25 h. | 84 (EG + 1,2-PD) | [76] |
| Cr2O3-SiO2 | 493 K, 3.0 MPa H2; batch reactor; 10 wt% of aqueous sorbitol solution; 0.4 g of catalyst; 2 h | 60 (EG + 1,2-PD) | [77] |
4. Scalability Challenges and Continuous-Flow Systems
5. Overcoming Barriers in Selective Hydrogenolysis of Polyols to Diols
6. Conclusions
Funding
Acknowledgments
Conflicts of Interest
References
- Wu, J.; Chen, T.; Ge, S.; Fan, W.; Wang, H.; Zhang, Z.; Huang, R. Synthesis and applications of carbon quantum dots derived from biomass waste: A review. Environ. Chem. Lett. 2023, 21, 3393–3424. [Google Scholar] [CrossRef]
- Yin, Y.; Suo, C.; Ma, C.; Liu, S. Recent progress towards fuels and value-added chemicals through lignin depolymerization. J. Environ. Chem. Eng. 2025, 13, 116321. [Google Scholar] [CrossRef]
- Sun, D.; Zhang, Y.; Zhou, Y.; Nie, Y.; Ban, L.; Wu, D.; Zhang, K. Photocatalytic and electrochemical synthesis of biofuel via efficient valorization of biomass. Adv. Energy Mater. 2025, 15, 2406098. [Google Scholar] [CrossRef]
- Mohanta, Y.K.; Mishra, A.K.; Lakshmayya, N.S.V.; Panda, J.; Thatoi, H.; Sarma, H.; Mishra, B. Agro-Waste-Derived Bioplastics: Sustainable Innovations for a Circular Economy. Waste Biomass Valorization 2025, 16, 3331–3355. [Google Scholar] [CrossRef]
- Rifna, E.J.; Dwivedi, M.; Seth, D.; Pradhan, R.C.; Sarangi, P.K.; Tiwari, B.K. Transforming the potential of renewable food waste biomass towards food security and supply sustainability. Sustain. Chem. Pharm. 2024, 38, 101515. [Google Scholar] [CrossRef]
- Hsiao, C.J.; Hu, J.L. Biomass and circular economy: Now and the future. Biomass 2024, 4, 720–739. [Google Scholar] [CrossRef]
- Khandaker, T.; Islam, T.; Nandi, A.; Anik, M.A.A.M.; Hossain, M.S.; Hasan, M.K.; Hossain, M.S. Biomass-derived carbon materials for sustainable energy applications: A comprehensive review. Sustain. Energy Fuels 2025, 9, 693–723. [Google Scholar] [CrossRef]
- Pandey, A.; Sharma, Y.C. Advancements in biomass valorization in integrated biorefinery systems. Biofuels Bioprod. Biorefin. 2024, 18, 2078–2090. [Google Scholar] [CrossRef]
- Long, B.; Zhang, F.; Dai, S.Y.; Foston, M.; Tang, Y.J.; Yuan, J.S. Engineering strategies to optimize lignocellulosic biorefineries. Nat. Rev. Bioeng. 2024, 3, 230–244. [Google Scholar] [CrossRef]
- Sessa, A.; Prete, P.; Cespi, D.; Scotti, N.; Tabanelli, T.; Antonetti, C.; Cucciniello, R. Levulinic acid biorefinery in a life cycle perspective. Curr. Opin. Green Sustain. Chem. 2024, 50, 100963. [Google Scholar] [CrossRef]
- Li, Z.; Waghmare, P.R.; Dijkhuizen, L.; Meng, X.; Liu, W. Research advances on the consolidated bioprocessing of lignocellulosic biomass. Eng. Microbiol. 2024, 4, 100139. [Google Scholar] [CrossRef]
- Ashokkumar, V.; Chandramughi, V.P.; Kumar, G.; Ngamcharussrivichai, C.; Piechota, G.; Igliński, B.; Chen, W.H. Advancements in lignocellulosic biomass: A critical appraisal of fourth-generation biofuels and value-added bioproduct. Fuel 2024, 365, 130751. [Google Scholar] [CrossRef]
- Li, W.; Xu, Y.; Wang, G.; Xu, T.; Wang, K.; Zhai, S.; Si, C. Sustainable Carbon-Based Catalyst Materials Derived From Lignocellulosic Biomass for Energy Storage and Conversion: Atomic Modulation and Properties Improvement. Carbon Energy 2025, 7, e708. [Google Scholar] [CrossRef]
- Zhou, Q.; Shen, Y.; Gu, X. Progress in torrefaction pretreatment for biomass gasification. Green Chem. 2024, 26, 9652–9670. [Google Scholar] [CrossRef]
- Song, L.; Yang, Y.; Lei, T.; Li, Y.; Shen, Y.; Wang, G.; Zheng, H. Regulation of Biomass Physicochemical Properties for Fuel Quality Improvement by Pretreatment Technology: A Review. Energy 2025, 360, 100015. [Google Scholar] [CrossRef]
- Mishra, R.K.; Kumar, D.J.P.; Sankannavar, R.; Binnal, P.; Mohanty, K. Hydro-deoxygenation of pyrolytic oil derived from pyrolysis of lignocellulosic biomass: A review. Fuel 2024, 360, 130473. [Google Scholar] [CrossRef]
- Webber, M.S.; Watson, J.; Zhu, J.; Jang, J.H.; Çağlayan, M.; Heyne, J.S.; Román-Leshkov, Y. Lignin deoxygenation for the production of sustainable aviation fuel blendstocks. Nat. Mater. 2024, 23, 1622–1638. [Google Scholar] [CrossRef] [PubMed]
- Carrasco Díaz, A.; Abdelouahed, L.; Brodu, N.; Montes-Jiménez, V.; Taouk, B. Upgrading of pyrolysis bio-oil by catalytic hydrodeoxygenation, a review focused on catalysts, model molecules, deactivation, and reaction routes. Molecules 2024, 29, 4325. [Google Scholar] [CrossRef]
- Aghamiri, A.R.; Lahijani, P. Catalytic conversion of biomass and plastic waste to alternative aviation fuels: A review. Biomass Bioenergy 2024, 183, 107120. [Google Scholar] [CrossRef]
- Jian, Y.; Meng, Y.; Li, H. Selectivity Control of C-O Bond Cleavage for Catalytic Biomass Valorization. Front. Energy Res. 2021, 9, 827680. [Google Scholar] [CrossRef]
- Feng, S.; Nguyen, P.T.; Ma, X.; Yan, N. Photorefinery of biomass and plastics to renewable chemicals using heterogeneous catalysts. Angew. Chem. Int. Ed. 2024, 63, e202408504. [Google Scholar] [CrossRef]
- Redina, E.; Tkachenko, O.; Salmi, T. Recent Advances in C5 and C6 Sugar Alcohol Synthesis by Hydrogenation of Monosaccharides and Cellulose Hydrolytic Hydrogenation over Non-Noble Metal Catalysts. Molecules 2022, 27, 1353. [Google Scholar] [CrossRef]
- Tran, M.H.; Lee, E.Y. Production of polyols and polyurethane from biomass: A review. Environ. Chem. Lett. 2023, 21, 2199–2223. [Google Scholar] [CrossRef]
- Diols. In Kirk-Othmer Encyclopedia of Chemical Technology; John Wiley & Sons, Inc.: Hoboken, NJ, USA, 2004.
- Główka, M.; Krawczyk, T. New Trends and Perspectives in Production of 1,2-Propanediol. ACS Sustain. Chem. Eng. 2023, 11, 7274–7287. [Google Scholar] [CrossRef]
- Wool, R.P.; Sun, X.S. Bio-Based Polymers and Composites; Academic Press: Cambridge, MA, USA, 2005. [Google Scholar]
- Cao, F.; Schwartz, T.J.; McClelland, D.J.; Krishna, S.H.; Dumesic, J.A.; Huber, G.W. Dehydration of cellulose to levoglucosenone using polar aprotic solvents. Green Chem. 2018, 20, 2954–2959. [Google Scholar] [CrossRef]
- Yang, L.; Su, J.; Yang, X.; Lin, H. Catalytic Oxidation Pathways for the Production of Carboxylic Acids from Biomass. In Green Chemistry and Sustainable Technology; Springer: Singapore, 2016; pp. 171–202. [Google Scholar]
- Rossi, A.; Pellegrini, G.C.S.C.; Pellegrini, F.C.S.C.; Caporali, S. A Short Overview on the Hydrogen Production Via Aqueous Phase Reforming (APR) of Cellulose, C6-C5 Sugars and Polyols. Catalysts 2019, 9, 917. [Google Scholar] [CrossRef]
- Yamada, T.; Kimura, A.; Takenouchi, S.; Sugahara, M.; Yoshioka, M.; Monta, Y.; Matsuda, M.; Hatano, M. Concise Synthesis of Polyacyloxy Cyclic Ethers Using the p-Toluenesulfonic Acid-Catalyzed Cascade Esterification/Cyclization of Polyols. ChemCatChem 2025, 17, e202401723. [Google Scholar] [CrossRef]
- Sadier, A.; Perret, N.; Da Silva Perez, D.; Besson, M.; Pinel, C. Effect of carbon chain length on catalytic CO bond cleavage of polyols over Rh-ReOx/ZrO2 in aqueous phase. Appl. Catal. A 2019, 586, 117213. [Google Scholar] [CrossRef]
- Sawadjoon, S.; Lundstedt, A.; Samec, J.S.M. Pd-Catalyzed Transfer Hydrogenolysis of Primary, Secondary, and Tertiary Benzylic Alcohols by Formic Acid: A Mechanistic Study. ACS Catal. 2013, 3, 635–642. [Google Scholar] [CrossRef]
- Tomishige, K.; Nakagawa, Y.; Tamura, M. Selective hydrogenolysis and hydrogenation using metal catalysts directly modified with metal oxide species. Green Chem. 2017, 19, 2876–2924. [Google Scholar] [CrossRef]
- Bhowmik, S.; Darbha, S. Advances in solid catalysts for selective hydrogenolysis of glycerol to 1, 3-propanediol. Catal. Rev. 2021, 63, 639–703. [Google Scholar] [CrossRef]
- Luo, W.; Cao, W.; Bruijnincx, P.C.A.; Lin, L.; Wang, A.; Zhang, T. Zeolite-supported metal catalysts for selective hydrodeoxygenation of biomass-derived platform molecules. Green Chem. 2019, 21, 3744–3768. [Google Scholar] [CrossRef]
- Xiao, Z.; Hu, Y.; Hu, J.; Wang, H.; Ji, J.; Huang, J.; Xing, C. Coordination environment tuning of Ni-Sn catalysts by glu-carbon to optimize cellulose hydrogenolysis to oxygenated chemicals. Fuel 2023, 350, 128833. [Google Scholar] [CrossRef]
- Yan, H.; Zhao, M.; Cao, Y.; Zhou, X.; Liu, Y.; Chen, X.; Zhou, H.; Yang, C. Crystal-facet-dependent, electron sink effect for the enhanced selective oxidation of polyols at the secondary hydroxyl position. J. Catal. 2024, 431, 115401. [Google Scholar] [CrossRef]
- Reinhold, J.S.; Pang, J.; Zhang, B.; Kühn, F.E.; Zhang, T. Rhenium-based catalysts for biomass conversion. Green Chem. 2024, 26, 312–353. [Google Scholar] [CrossRef]
- Mondelli, C.; Gözaydın, G.; Yan, N.; Pérez-Ramírez, J. Biomass valorisation over metal-based solid catalysts from nanoparticles to single atoms. Chem. Soc. Rev. 2020, 49, 3764–3782. [Google Scholar] [CrossRef]
- Jin, X.; Fang, T.; Wang, J.; Liu, M.; Pan, S.; Subramaniam, B.; Shen, J.; Yang, C.; Chaudhari, R.V. Nanostructured metal catalysts for selective hydrogenation and oxidation of cellulosic biomass to chemicals. Chem. Rec. 2019, 19, 1952–1994. [Google Scholar] [CrossRef]
- Emanuel, M.; Virgilio, C.L.; Padró, M.E. Sad. Ru/ReOx/TiO2 Selective and Reusable Catalyst for C-O Hydrogenolysis of C4 Polyols. ChemCatChem 2023, 15, e202201618. [Google Scholar]
- Ai, S.; Huang, Z.; Yu, W.; Huang, C. Separation of 1, 2-diols from ethylene glycol via biphasic-solvent extraction and thermodynamic insights. Chem. Eng. Res. Des. 2023, 200, 22–31. [Google Scholar] [CrossRef]
- Liu, L.; Asano, T.; Nakagawa, Y.; Tamura, M.; Okumura, K.; Tomishige, K. Selective Hydrogenolysis of Glycerol to 1,3-Propanediol over Rhenium-Oxide-Modified Iridium Nanoparticles Coating Rutile Titania Support. ACS Catal. 2019, 9, 10913–10930. [Google Scholar] [CrossRef]
- Priya, S.S.; Kumar, V.P.; Kantam, M.L.; Bhargava, S.K.; Srikanth, A.; Chary, K.V. High Efficiency Conversion of Glycerol to 1,3-Propanediol Using a Novel Platinum–Tungsten Catalyst Supported on SBA-15. Ind. Eng. Chem. Res. 2015, 54, 9104–9115. [Google Scholar] [CrossRef]
- Guadix-Montero, S.; Santos-Hernández, A.; Lei, N.; Morgan, D.J.; He, Q.; Zhang, T.; Roldán, A.; Sankar, M. Controlling the Selectivity of Supported Ru Nanoparticles During Glycerol Hydrogenolysis: C–O vs. C–C Cleavage. Catal. Sci. Technol. 2021, 11, 1595–1606. [Google Scholar] [CrossRef]
- Wang, C.; Jiang, H.; Chen, C.; Chen, R.; Xing, W. Solvent Effect on Hydrogenolysis of Glycerol to 1,2-Propanediol over Cu–ZnO Catalyst. Chem. Eng. J. 2015, 264, 344–350. [Google Scholar] [CrossRef]
- Duarte, R.B.; Corazza, M.L.; Pimenta, J.L.C.W.; de Matos Jorge, L.M. Kinetic Study of Glycerol Hydrodeoxygenation on Al2O3 and NiMo2C/Al2O3 Catalysts. Fuel 2023, 354, 129257. [Google Scholar] [CrossRef]
- García-Fernández, S.; Gandarias, I.; Requies, J.; Soulimani, F.; Arias, P.L.; Weckhuysen, B.M. The Role of Tungsten Oxide in the Selective Hydrogenolysis of Glycerol to 1,3-Propanediol over Pt/WOx/Al2O3. Appl. Catal. B Environ. 2017, 204, 260–272. [Google Scholar] [CrossRef]
- Liu, Y.; Rempel, G.L.; Ng, F.T.T. Kinetic Study of Pd-Promoting Effect on Cu/ZnO/Al2O3 Catalyst for Glycerol Hydrogenolysis to Produce 1,2-Propanediol at Low Hydrogen Pressure. Biomass 2022, 2, 27–45. [Google Scholar] [CrossRef]
- Nakagawa, Y.; Koso, S.; Tomishige, K. Catalytic materials for the hydrogenolysis of glycerol to 1,3-propanediol. J. Mater. Chem. A 2014, 2, 6688–6702. [Google Scholar] [CrossRef]
- da Silva Ruy, A.D.; de Brito Alves, R.M.; Hewer, T.L.R.; de Aguiar Pontes, D.; Teixeira, L.S.G.; Pontes, L.A.M. Catalysts for Glycerol Hydrogenolysis to 1,3-Propanediol: A Review of Chemical Routes and Market. Catal. Today 2021, 381, 243–253. [Google Scholar] [CrossRef]
- Chaminand, J.; Djakovitch, L.; Gallezot, P.; Marion, P.; Pinel, C.; Rosier, C. Glycerol Hydrogenolysis on Heterogeneous Catalysts. Green Chem. 2004, 6, 359–361. [Google Scholar] [CrossRef]
- Jarauta-Córdoba, C.; Oregui Bengoechea, M.; Agirrezabal-Telleria, I.; Arias, P.-L.; Gandarias, I. Insights into the Nature of the Active Sites of Pt–WOx/Al2O3 Catalysts for Glycerol Hydrogenolysis into 1,3-Propanediol. Catalysts 2021, 11, 1171. [Google Scholar] [CrossRef]
- Wang, J.; Zhao, X.; Lei, N.; Li, L.; Zhang, L.; Xu, S.; Zhang, T. Hydrogenolysis of Glycerol to 1,3-propanediol under Low Hydrogen Pressure over WOx-Supported Single/Pseudo-Single Atom Pt Catalyst. ChemSusChem 2016, 9, 784–790. [Google Scholar] [CrossRef]
- Luo, W.; Lyu, Y.; Gong, L.; Du, H.; Wang, T.; Ding, Y. Selective hydrogenolysis of glycerol to 1,3-propanediol over egg-shell type Ir–ReO x catalysts. RSC Adv. 2016, 6, 13600–13608. [Google Scholar] [CrossRef]
- Xi, Z.; Cui, G.; Wang, W.; Wu, T.; Zhu, K.; Wei, J.; Liu, Y.; Yang, M.; Zeng, J.; Wei, M. Platinum–copper single atom alloy catalysts with high performance towards glycerol hydrogenolysis. Nat. Commun. 2019, 10, 5812. [Google Scholar] [CrossRef]
- Chen, J.-Y.; Xiao, Y.; Guo, F.-S.; Li, K.-M.; Huang, Y.-B.; Lu, Q. Single-Atom Metal Catalysts for Catalytic Chemical Conversion of Biomass to Chemicals and Fuels. ACS Catal. 2024, 14, 5198–5226. [Google Scholar] [CrossRef]
- Wang, S.; Guo, X.; Zhou, Z.; Cheng, H.; Wang, L. Atomic-Precision Engineering of Single-Atom Alloy Materials for Green Catalysis and Energy Conversion. ACS Mater. Lett. 2025, 7, 1654–1697. [Google Scholar] [CrossRef]
- Zhao, B.; Zou, J.; Yao, B.; Wade, A.; Liu, L.; Dong, J.; He, Q. Discovering positively charged Pt for enhanced hydrogenolysis of glycerol to 1,3-propanediol. Green Chem. 2020, 22, 8254–8261. [Google Scholar] [CrossRef]
- Zeng, Y.; Jiang, L.; Zhang, X.; Li, H.; Liu, S.; Fu, Y. Effect of titania polymorphs on the structure and catalytic performance of the PtWOx/TiO2 catalyst in glycerol hydrogenolysis to 1,3-propanediol. ACS Sustain. Chem. Eng. 2022, 10, 9532–9545. [Google Scholar] [CrossRef]
- Heisig, C.; Turek, T.; Glotzbach, C.; Schirrmeister, S. Selective Hydrogenolysis of Biomass-Derived Xylitol to Glycols: Reaction Network and Kinetics. Chem. Eng. Technol. 2021, 44, 761–772. [Google Scholar] [CrossRef]
- Rivière, M.; Perret, N.; Pinel, C.; Cabiac, A.; Delcroix, D. Xylitol Hydrogenolysis over Ruthenium-Based Catalysts: Effect of Alkaline Promoters and Basic Oxide-Modified Catalysts. ChemCatChem 2017, 9, 2145–2159. [Google Scholar] [CrossRef]
- Zhou, Z.; Dai, S.; Yu, P.; Wu, W.; Qin, J. Preparation of nano-Ni/meso-Ce-TiO2 by one-step in a sol-gel system and its catalytic performance for hydrogenolysis of xylitol. RSC Adv. 2015, 5, 70410–70416. [Google Scholar] [CrossRef]
- Xia, Q.; Zhang, G.; Wang, J.; Jin, X.; Shen, J. Synergistic Bimetallic Pd-Pt/TiO2 Catalysts for Hydrogenolysis of Xylitol with in Situ-Formed H2. Ind. Eng. Chem. Res. 2020, 59, 13879–13891. [Google Scholar] [CrossRef]
- Li, S.; Zan, Y.; Sun, Y.; Kong, L.Z.; Sun, Y. Efficient one-pot hydrogenolysis of biomass-derived xylitol into ethylene glycol and 1,2-propylene glycol over Cu–Ni–ZrO2 catalyst without solid bases. J. Energy Chem. 2019, 28, 101–106. [Google Scholar] [CrossRef]
- Liu, H.; Huang, Z.; Kang, H.; Chen, J.; Liu, H. Efficient bimetallic NiCu-SiO2 catalysts for selective hydrogenolysis of xylitol to ethylene glycol and propylene glycol. Appl. Catal. B 2018, 220, 251–263. [Google Scholar] [CrossRef]
- Rivière, M.; Perret, N.; Pinel, C.; Delcroix, D.; Cabiac, A. Solvent Effect in Hydrogenolysis of Xylitol over Bifunctional Ru/MnO/C Catalysts under Alkaline-Free Conditions. ACS Sustain. Chem. Eng. 2018, 6, 4076–4085. [Google Scholar] [CrossRef]
- Jia, Y.; Sun, Q.; Liu, H. Selective hydrogenolysis of biomass-derived sorbitol to propylene glycol and ethylene glycol on in-situ formed PdZn alloy catalysts. Appl. Catal. A 2020, 603, 117770. [Google Scholar] [CrossRef]
- Jin, X.; Zhang, W.; Zhang, D.; Yin, B.; Liu, X.; Zhang, G.; Chaudhari, R.V. Lattice strained bimetallic PtPd nanocatalysts display multifunctional nature for transfer hydrogenolysis of sorbitol in base-free medium. Mater. Today Sustain. 2020, 10, 100047. [Google Scholar] [CrossRef]
- Liu, L.; Cao, J.; Nakagawa, Y.; Betchaku, M.; Tamura, M.; Yabushita, M.; Tomishige, K. Hydrodeoxygenation of C4–C6 sugar alcohols to diols or mono-alcohols with the retention of the carbon chain over a silica-supported tungsten oxide-modified platinum catalyst. Green Chem. 2021, 23, 5665–5679. [Google Scholar] [CrossRef]
- Cai, C.; Wang, H.; Xin, H.; Zhu, C.; Zhang, Q.; Zhang, X.; Ma, L. Hydrogenolysis of biomass-derived sorbitol over La-promoted Ni/ZrO2 catalysts. RSC Adv. 2020, 10, 3993–4001. [Google Scholar] [CrossRef] [PubMed]
- Zhu, X.; Feng, X.; Yao, C.; Sun, W.; Ma, J.; Zhong, F.; Zhang, J. Glycol Production from Sorbitol Hydrogenolysis over Hydrothermally Stable Ni Catalysts and the Mechanistic Study. Ind. Eng. Chem. Res. 2024, 63, 8175–8186. [Google Scholar] [CrossRef]
- Chen, X.; Yang, Y.; Xiao, Q.; Guo, X.; Chen, F.; Liu, X.; Lu, W. Controlling transformation of sorbitol into glycols over Ru-WOx modified biomass-derived N-doped carbon. J. Mater. Sci. 2024, 59, 8186–8204. [Google Scholar] [CrossRef]
- Beine, A.K.; Albano, Y.; Reichenberger, S.; Gupta, K.; Palkovits, R. Biobased Glycols–Towards the Use of Solid Bases in the Hydrogenolysis of Sorbitol. ChemistrySelect 2024, 9, e202302659. [Google Scholar] [CrossRef]
- Beine, A.K.; Ludovicy, J.; Chai, J.; Hofmann, J.P.; Glotzbach, C.; Hausoul, P.J.; Palkovits, R. Ru on N-doped carbon for the selective hydrogenolysis of sugars and sugar alcohols. ChemCatChem 2022, 14, e202101908. [Google Scholar] [CrossRef]
- Wang, X.; Beine, A.K.; Hausoul, P.J.; Palkovits, R. Cu/C-catalyzed hydrogenolysis of sorbitol to glycols–on the influence of particle size and base. ChemCatChem 2019, 11, 4123–4129. [Google Scholar] [CrossRef]
- Alexzman, Z.A.; Salamun, N.; Ibrahim, M.L.; Sidi, S.M.; Annuar, N.H.R. Catalytic hydrogenolysis of sorbitol to glycols over chromium oxide silica: Effect of chromium loading. J. Environ. Chem. Eng. 2024, 12, 112531. [Google Scholar] [CrossRef]
- Chen, P.; Onodera, W.; Akatsuka, M.; Kita, Y.; Tamura, M. Hydrogenolysis of isosorbide to diols and triols over a heterogeneous SiO2-supported Rh catalyst. Catal. Sci. Technol. 2024, 14, 3001–3006. [Google Scholar] [CrossRef]
- Sun, J.; Liu, H. Selective hydrogenolysis of biomass-derived xylitol to ethylene glycol and propylene glycol on Ni/C and basic oxide-promoted Ni/C catalysts. Catal. Today 2014, 234, 75–82. [Google Scholar] [CrossRef]
- Kang, P.L.; Shi, Y.F.; Shang, C.; Liu, Z.P. Artificial intelligence pathway search to resolve catalytic glycerol hydrogenolysis selectivity. Chem. Sci. 2022, 13, 8148–8160. [Google Scholar] [CrossRef]
- Nakagawa, Y.; Kasumi, T.; Ogihara, J.; Tamura, M.; Arai, T.; Tomishige, K. Erythritol: Another C4 platform chemical in biomass refinery. ACS Omega 2020, 5, 2520–2530. [Google Scholar] [CrossRef]
- Li, B.; Zhang, Y.; Chen, J.; Wang, H.; Liu, Z.; Sun, W. Progress in 1,3-Propanediol Biosynthesis: Metabolic Engineering of the Glycerol Reductive Pathway toward High Yield. Microb. Cell Fact. 2024, 23, 114. [Google Scholar]
- Kong, D.S.; Wei, Y.; Wang, S.; Zhang, L.; Feng, J.; Li, H. Bioconversion of Glycerol to 1,3-Propanediol Using Klebsiella pneumoniae L17 with Microbially Influenced Corrosion of Zero-Valent Iron. Fermentation 2023, 9, 233. [Google Scholar] [CrossRef]
- Frazão, C.J.R.; Silva, I.J.; Pereira, A.R.; Carvalho, A.L.; Santos, C.N.S.; Rodrigues, J.L.; Belo, I.; Ferreira, B.S. Construction of a Synthetic Metabolic Pathway for the Production of 1,3-Propanediol from Glucose. Sci. Rep. 2019, 9, 11838. [Google Scholar] [CrossRef] [PubMed]

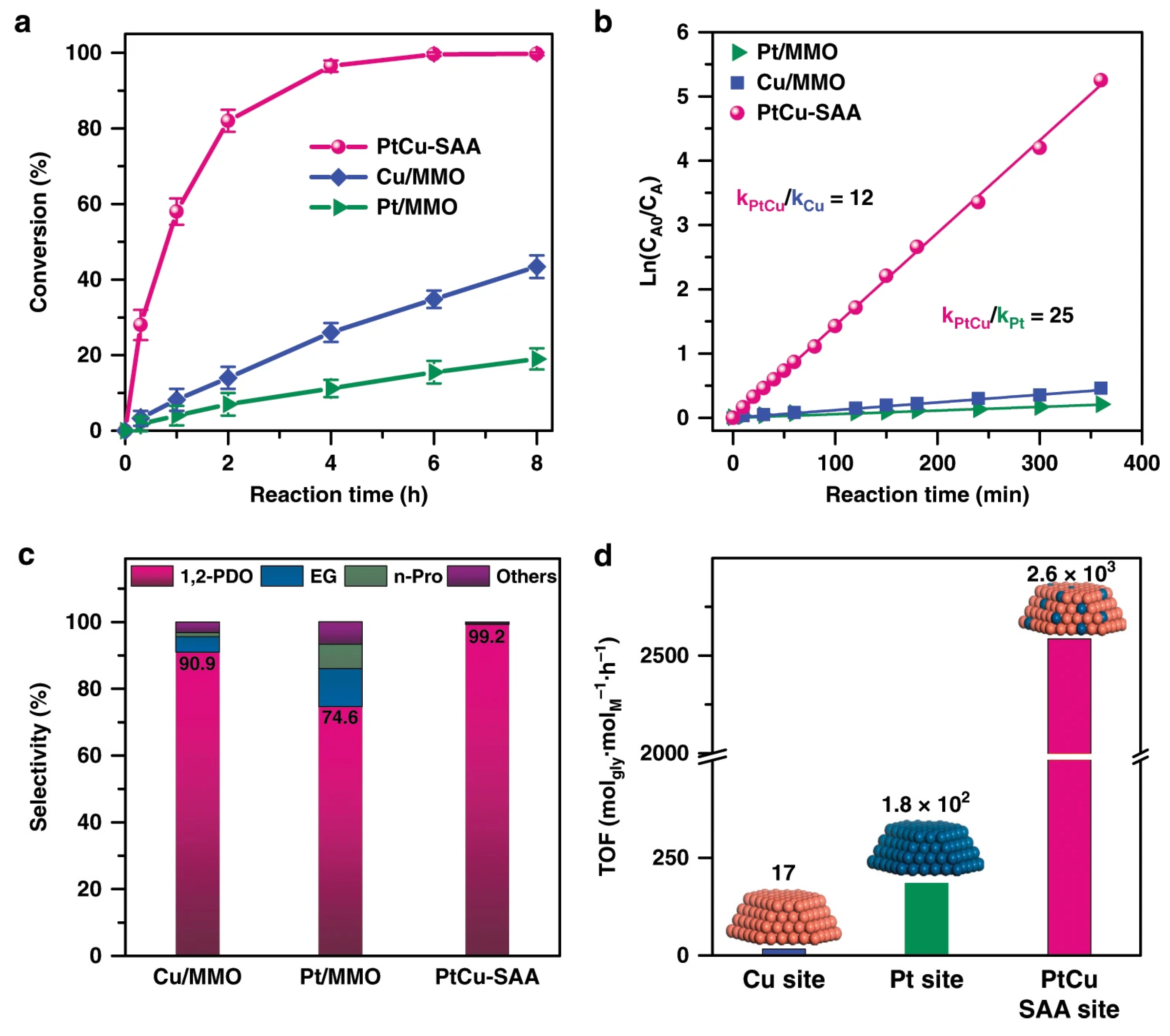
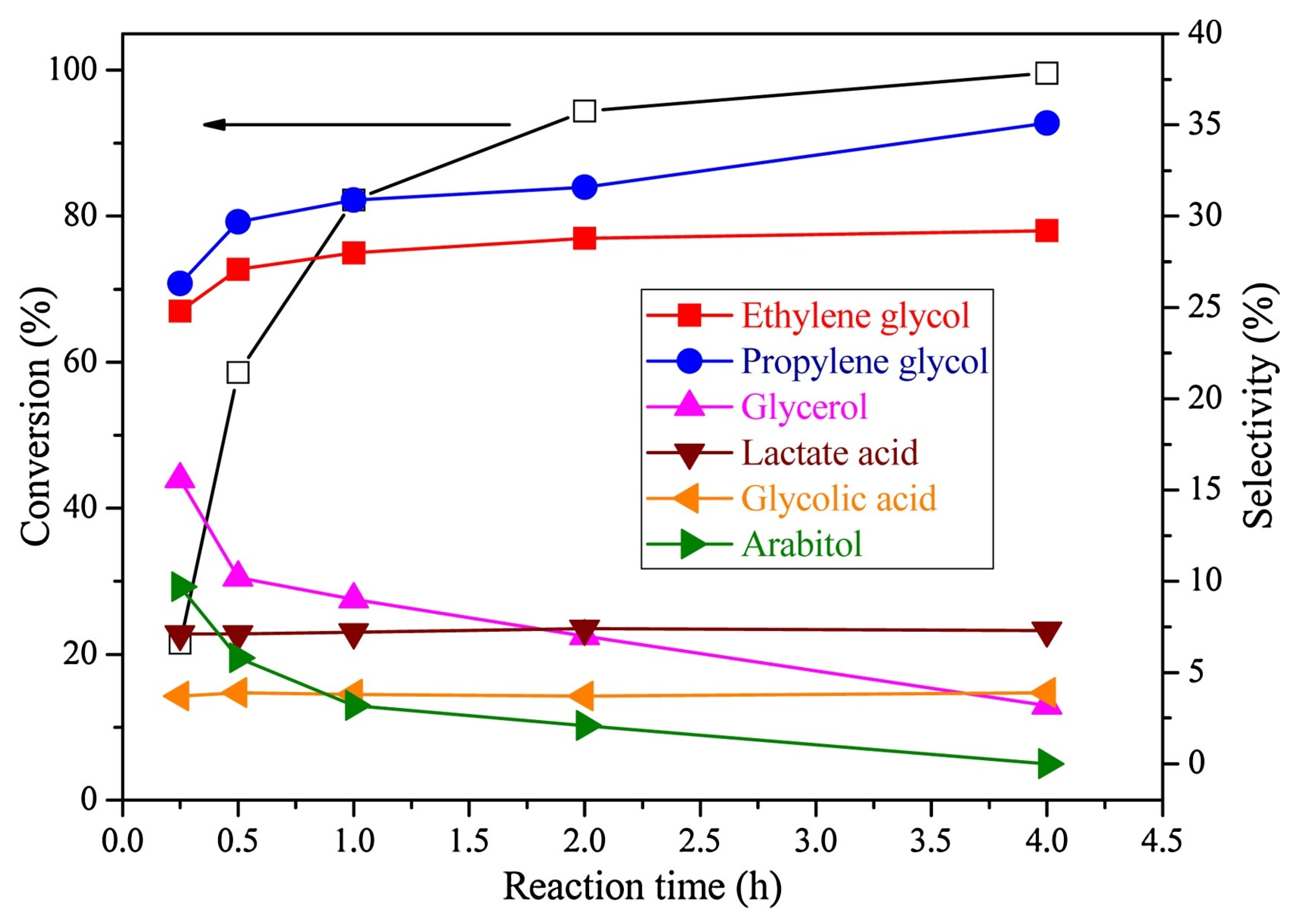
| Catalyst System | Reaction Conditions | Selectivity to PDs | Reference |
|---|---|---|---|
| Ru/C | 423–463 K, 3–5 MPa H2, H2O, alkaline promoters | EG (40–50%), 1,2-PD (30–40%) | [61] |
| Ru/C + KOH or MnO | 433–473 K, 4 MPa H2, aqueous phase | EG (35–45%), 1,2-PD (25–30%) | [62] |
| Ni/Ce–TiO2 (mesoporous) | 493 K, 3 MPa H2, H2O | EG (45–55%), 1,2-PD (25–35%) | [63] |
| Pd–Pt/TiO2 | 453 K, ~1 MPa H2 (in situ from formic acid), H2O | EG (30–35%), 1,2-PD (40–45%) | [64] |
| Cu–Ni–ZrO2 | 493 K, 5 MPa H2, H2O, base-free | EG (40–50%), 1,2-PD (30–35%) | [65] |
| Ni–Cu/SiO2 | 473–493 K, 4 MPa H2, H2O | EG (45–55%), 1,2-PD (25–35%) | [66] |
| Ru/MnO/C | 453–493 K, 4 MPa H2, H2O, no base | EG (50%), 1,2-PD (30%) | [67] |
| Ni/C, Ni/C + basic promoters | 473–493 K, 5 MPa H2, H2O | EG (40–45%), 1,2-PD (35–40%) | [31] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Serrano-Ruiz, J.C. Selective Deoxygenation of Biomass Polyols into Diols. Molecules 2025, 30, 3559. https://doi.org/10.3390/molecules30173559
Serrano-Ruiz JC. Selective Deoxygenation of Biomass Polyols into Diols. Molecules. 2025; 30(17):3559. https://doi.org/10.3390/molecules30173559
Chicago/Turabian StyleSerrano-Ruiz, Juan Carlos. 2025. "Selective Deoxygenation of Biomass Polyols into Diols" Molecules 30, no. 17: 3559. https://doi.org/10.3390/molecules30173559
APA StyleSerrano-Ruiz, J. C. (2025). Selective Deoxygenation of Biomass Polyols into Diols. Molecules, 30(17), 3559. https://doi.org/10.3390/molecules30173559






