Abstract
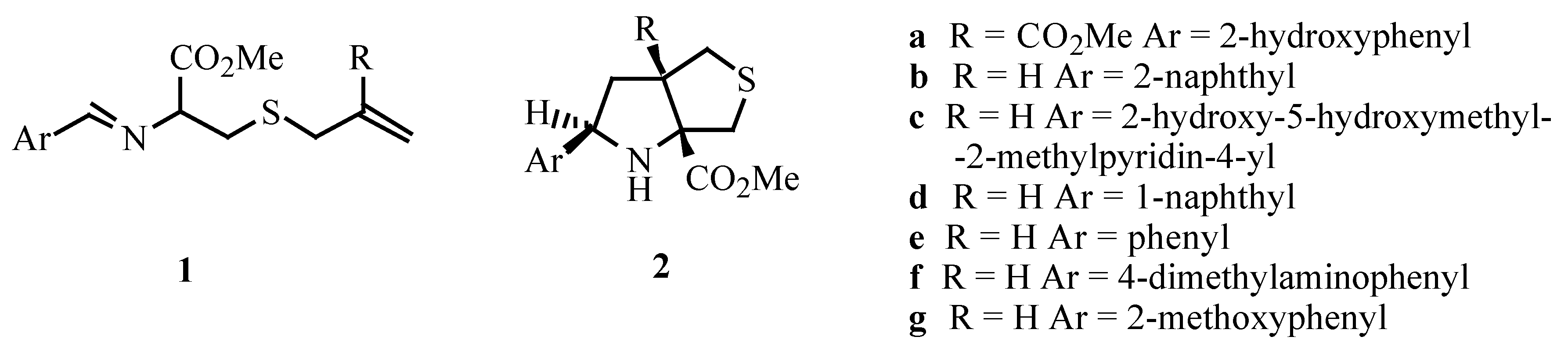
Azomethine ylides were generated from Schiffs bases of S-allylcysteine methyl ester and their intramolecular 1,3-dipolar cycloadditions were studied. These reactions led to the synthesis of thieno[3,4-b]pyrrole derivatives in good yield.
Introduction
Intramolecular 1,3-dipolar cycloaddition is an important method for the construction of ring fused heterocycles. The prototropic generation of azomethine ylides from imines of α-amino acid esters is an example of this type of process [1]. We have previously reported the synthesis of tetrahydro-1H-thieno[3,4-b]pyrrole 2a through an intramolecular 1,3-dipolar cycloaddition of the azomethine ylide which was generated from the Schiffs base 1a by proton transfer [2]. Grigg has also observed the dipolar cycloaddition of azomethine ylides derived from Schiffs bases of S-allylcysteine methyl ester (1b and 1c) [1,3]. We decided to investigate the possibility of making more extensive use of this type of reaction and to gather consistent structural information on the products. Thieno[3,4-b]pyrroles, a class of compounds isosteric with indoles, are of potential pharmacological importance [4].
Scheme 1.
Scheme 1.
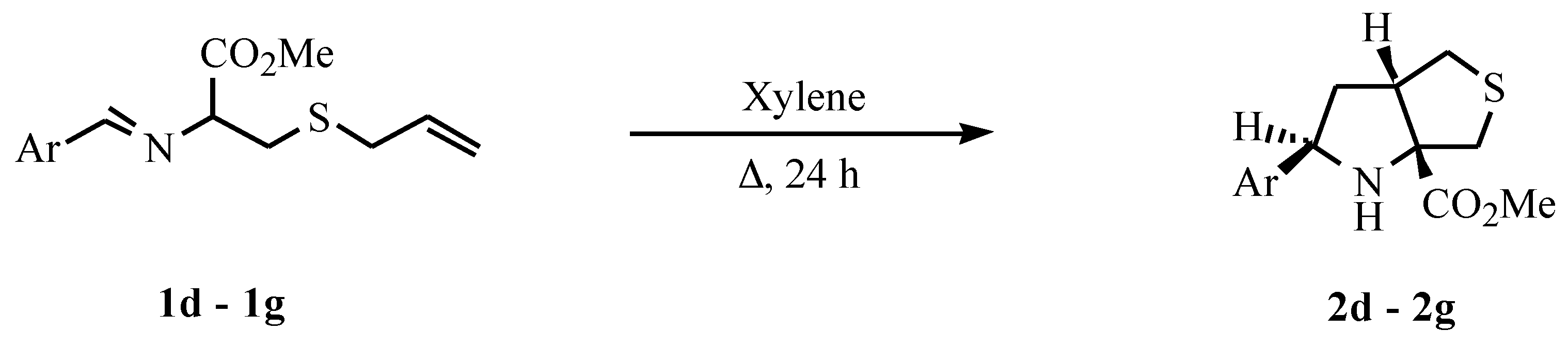
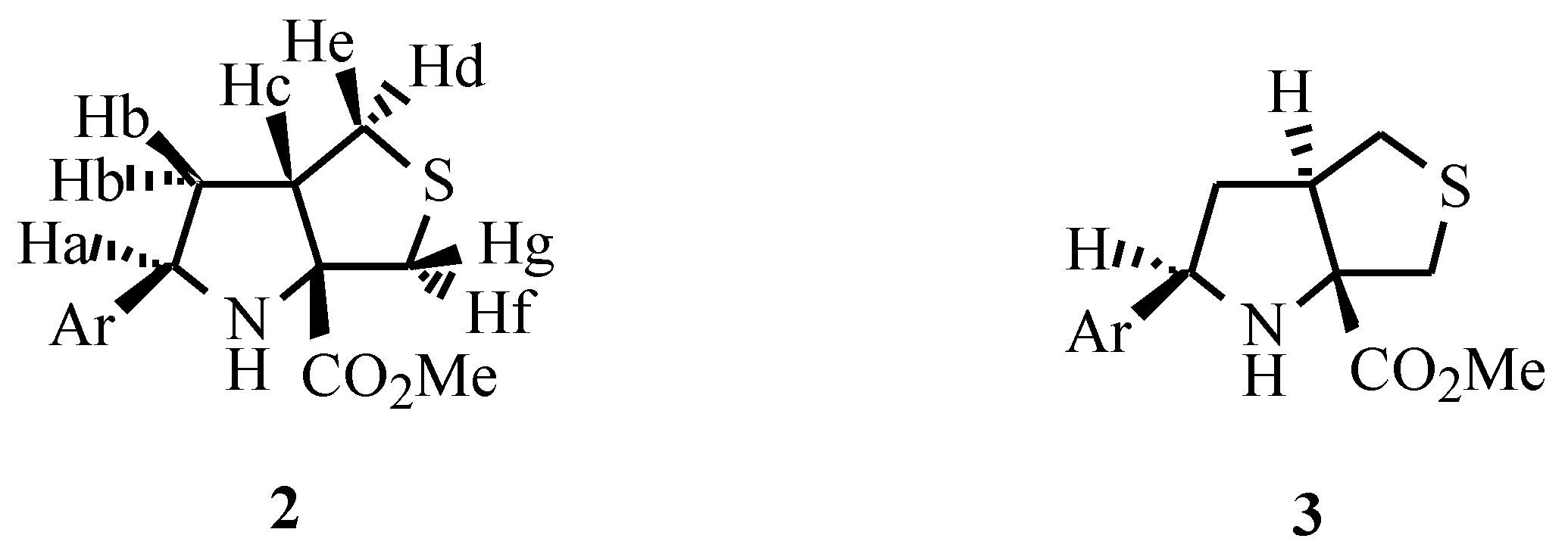
Results and Discussions
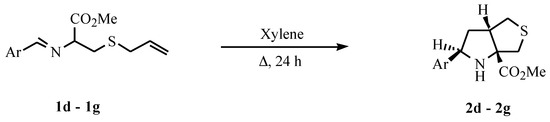
S-Allylcysteine methyl ester was converted into the corresponding imines (1d, 1e, 1f and 1g) in good yield by condensing with aromatic aldehydes in a solution of NaOMe/MeOH at room temperature. Each of the imines was dissolved in xylene and the solution was heated under reflux for 24 hours giving the cycloadducts 2d, 2e, 2f and 2g (Scheme 1). Compounds 2d, 2f and 2g were isolated in good yield after crystallization. Compound 2e was isolated as an oil in moderate yield.
Reactions at lower temperature were also attempted. At 85°C 2d was isolated in good yield after 24 hours, but 1e gave no evidence of reaction at this temperature over this time. Only at 100°C could a moderate yield of 2e be isolated. It was proved however that the cycloaddition of imine 1e can be performed and is an extremely clean reaction at room temperature although a very slow reaction. A solution of 1e in CDCl3 was left at room temperature and the reaction followed by 1H NMR. The formation of adduct 2e was monitored by integration of the triplet assigned to proton H-2 (δ = 4.53 ppm) and by following the disappearance of the singlet (δ = 8.31 ppm) assigned to the imine hydrogen of 1e: after 25 days the adduct was formed in 18% yield; after 76 days in 58% yield; after 130 days in 82% yield; and after 150 days in 87% yield. These results allow us to conclude that the intramolecular 1,3-dipolar cycloaddition occurs at room temperature but is a very slow process.
The reaction of the Schiffs base 1b was also performed leading to the formation of compound 2b as the major product together with the formation of a minor amount of isomer 3 (Ar = 2-naphthyl) as described in the literature [1,3]. In the cycloaddition reactions of imines 1d, 1e, 1f and 1g the isomers with the stereochemistry of 3 were detected in the 1H NMR of the crude products but were present in less than 5% yield. Only products 2d, 2e, 2f and 2g could be isolated.
The X-ray structure of 2g was obtained confirming the stereochemistry of the adducts: cis stereochemistry at the ring junction resulting from an exo intramolecular 1,3-dipolar cycloaddition [5]. The unit-cell is formed by two molecules which are an enantiomeric pair.
Conclusion
Azomethine ylides were generated from Schiffs bases of S-allylcysteine methyl ester and its intramolecular 1,3-dipolar cycloaddition was performed. The reported reaction involves a non-activated terminal alkene as the internal dipolarophile leading to a slower cycloaddition when compared with the formation of 2a. In this work it was proved that the 1,3-dipolar cycloaddition can be exploited with a broad range of structures to give tetrahydro-1H-thieno[3,4-b]pyrroles in good yield and well defined stereochemistry.
Experimental Section
General
1H NMR spectra were recorded on a Brucker AMX300 instrument operating at 300 MHz. 13C were recorded on a Brucker AMX300 instrument operating at 75.5 MHz. The solvent was deuteriochloroform. IR spectra were recorded on a Perkin Elmer 1720X FTIR spectrometer. Mass spectra were recorded under electron impact at 70 eV on a VG Micromass 7070E instrument. M.p.’s were recorded on a Reichert hot stage. Flash column chromatography was performed with Merck 9385 silica as the stationary phase.
General procedure for the preparation of imines
S-Allylcysteine methyl ester hydrochloride [1] (20 mmol) was treated with a solution of NaOMe (from 20 mmol of Na in 30 ml of dry MeOH). The aldehyde (20 mmol) was added and the resulting mixture was stirred at room temperature for 24 hours. The solvent was evaporated and the residue dissolved in CH2Cl2 and washed with water. The organic solvent was dried (MgSO4) and evaporated off. The crude imines were used directly for the cycloaddition (except for 1f) as distillation resulted in cyclization and/or in decomposition.
Methyl N-(1-naphthylidene)-S-allylcysteine (1d)
An oil. Yield 97%. 1H NMR δ 3.00 (1H, dd, J 8.2 and 13.7), 3.16 (2H, d, J 7.3), 3.20 (1H, dd, J 5.2 and 13.7), 3.78 (3H, s, CO2CH3), 4.21 (1H, dd, J 5.2 and 8.2, CHCO2CH3), 5.08-5.15 (2H, m, CH=CH2), 5.69-5.81 (1H, m, CH=CH2), 7.47-7.61 (3H, m, ArH), 7.85-7.96 (4H, m, ArH) and 8.95 (1H, s, CH=N). MS m/z 313 (M+, 5%), 254 (36), 186 (4), 127 (20) and 169 (100).
Methyl N-phenylidene-S-allylcysteine (1e)
An oil. Yield 75%. 1H NMR δ 2.92 (1H, dd, J 7.9 and 13.8), 3.08-3.15 (3H, m), 3.74 (3H, s, CO2CH3), 4.13 (1H, dd, J 5.46 and 7.95, CHCO2CH3), 5.07-5.13 (2H, m, CH=CH2), 5.68-5.82 (1H, m, CH=CH2), 7.37-7.44 (3H, m, ArH), 7.77-7.81 (2H, m, ArH) and 8.31 (1H, s, CH=N). MS m/z 278 (M+, 1%), 205 (2), 201 (3), 190 (100), 174 (11) and 142 (2).
Methyl N-(4-dimethylaminophenylidene)-S-allylcysteine (1f)
Yield 63%. M.p. 74-76°C (from ethyl ether-petroleum ether 40/60). Analysis: Found C, 63.11; H, 7.14; N, 9.08; C16H22N2O2S requires C, 62.72; H, 7.24; N, 9.14%. 1H NMR δ 2.88 (1H, dd, J 7.7 and 13.8), 3.01 (6H, s, NMe2), 3.06-3.16 (3H, m), 3.75 (3H, s, CO2CH3), 4.06 (1H, dd, J 5.8 and 7.7, CHCO2CH3), 5.07-5.14 (2H, m, CH=CH2), 5.69-5.83 (1H, m, CH=CH2), 6.68 (2H, d, J 8.9, ArH), 7.65 (2H, d, J 8.9, ArH) and 8.17 (1H, s, CH=N). MS m/z 306 (M+, 29%), 265 (15), 247 (13), 219 (100) and 83 (64).
Methyl N-(2-methoxyphenylidene)-S-allylcysteine (1g)
An oil. Yield 70%. 1H NMR δ 2.77 (1H, dd, J 7.7 and 13.8), 2.93-3.07 (3H, m), 3.48 (3H, s), 3.66 (3H, s), 4.03 (1H, dd, J 5.7 and 7.7, CHCO2CH3), 4.94-4.97 (2H, m, CH=CH2), 5.55-5.73 (1H, m, CH=CH2), 6.67-6.85 (2H, m, ArH), 7.18-7.27 (2H, m, ArH) and 8.63 (1H, s, CH=N). MS m/z 293 (M+, 20%), 278 (30), 262 (9), 186 (50) 127 (82) and 117 (100).
General procedure for the intramolecular cycloaddition
A solution of the imine 1 (2 mmol) in xylene (10 ml) was heated under reflux, under nitrogen, for 24 hours. The solvent was evaporated and the crude product was purified by crystallization (2d, 2f and 2g crystallization from ethyl ether-petroleum ether 40/60) or by flash chromatography (2e).
Methyl 2-(1-naphthyl)tetrahydro-1H-thieno[3,4-b]pyrrole-6a(6H)-carboxylate (2d)
Yield 51%. M.p. 122-123°C (from ethyl ether-petroleum ether 40/60). Analysis: Found C, 68.54; H, 6.04; N, 4.10; C18H19NO2S requires C, 68.98; H, 6.11; N, 4.47%. 1H NMR δ 2.16-2.34 (2H, m, Hb), 2.65 (1H, brs, NH), 2.80 (1H, dd, J 4.1 and 11.7, Hd), 2.95 (1H, d, J 12.2, Hf), 3.24 (1H, dd, J 7.4 and 11.7, He), 3.39 (1H, d, J 12.2, Hg), 3.34-3.41 (1H, m, Hc), 3.63 (3H, s, OCH3), 5.29 (1H, t, J 7.5, Ha), 7.38-7.54 (3H, m, ArH), 7.60 (1H, d, J 7.1, ArH), 7.72 (1H, d, J 8.2, ArH), 7.83 (1H, dd, J 5.7 and 1.5, ArH) and 8.16 (1H, d, J 8.2, ArH). 13C NMR δ 38.89, 39.76, 44.32, 52.16, 52.49, 58.32, 79.41, 122.02, 123.38, 125.26, 125.42, 125.93, 127.54, 128.65, 131.31, 133.67, 138.18 and 175.15. MS m/z 313 (M+, 33%), 254 (43), 206 (28), 169 (100) and 127 (23).
Methyl 2-phenyltetrahydro-1H-thieno[3,4-b]pyrrole-6a(6H)-carboxylate (2e)
An oil. Yield 37%. 1H NMR δ 2.03-2.10 (2H, m, Hb), 2.65 (1H, brs, NH), 2.71 (1H, dd, J 2.7 and 11.5, Hd), 2.90 (1H, d, J 12.2, Hf), 3.18 (1H, dd, J 9.7 and 11.5, He), 3.26-3.33 (1H, m, Hc), 3.40 (1H, d, J 12.2, Hg), 3.73 (3H, s, OCH3), 4.53 (1H, t, J 6.8, Ha) and 7.37-7.44 (5H, m, ArH); m/z 278 (M+, 1%), 263 (10), 231 (1), 201 (3), 190 (100), 130 (89) and 103 (74).
Methyl 2-(4-dimethylaminophenyl)tetrahydro-1H-thieno[3,4-b]pyrrole-6a(6H)-carboxylate (2f)
Yield 71%. M.p. 80-81°C (from ethyl ether-petroleum ether 40/60). Analysis: Found C, 62.64; H, 7.24; N, 8.80; S, 10.85. C16H22N2O2S requires C, 62.72; H, 7.24; N, 9.14; S, 10.46%). 1H NMR δ 2.00-2.04 (2H, m, Hb), 2.65 (1H, brs, NH), 2.72 (1H, dd, J 5.3 and 11.4, Hd), 2.88 (1H, d, J 12, Hf), 2.88 (6H, s, 2xCH3), 3.15 (1H, dd, J 7.8 and 11.4, He), 3.24-3.29 (1H, m, Hc), 3.39 (1H, d, J 12, Hg), 3.73 (3H, s, OCH3), 4.42 (1H, t, J 7.8, Ha), 6.65 (2H, d, J 8.6, ArH) and 7.21 (2H, d, J 8.6, ArH); m/z 306 (M+, 30%), 233 (100) and 162 (58).
Methyl 2-(2-methoxyphenyl)tetrahydro-1H-thieno[3,4-b]pyrrole-6a(6H)-carboxylate (2g)
Yield 67%. M.p. 114-117°C (from ethyl ether-petroleum ether 40/60). Analysis: Found C, 61.47; H, 6.43; N, 4.22; S, 10.40 C15H19NO3S requires C, 61.41; H, 6.33; N, 4.79; S, 10.93%. 1H NMR δ 2.07-2.21 (2H, m, Hb), 2.75 (1H, dd, J 4.4 and 11.7, Hd), 2.88 (1H, d, J 12.2, Hf), 2.96 (1H, brs, NH), 3.19 (1H, dd, J 7.5 and 11.7, He), 3.30 (1H, d, J 12.2, Hg), 3.33-3.39 (1H, m, Hc), 3.64 (3H, s, CO2CH3), 3.83 (3H, s), 4.76 (1H, t, J 6.8, Ha), 6.82-6.93 (2H, m, ArH) ,7.18-7.24 (1H, m, ArH) and 7.31 (1H, dd, J 1.4 and 7.5, ArH). MS m/z 293 (M+, 4%), 234 (98), 220 (60), 186 (37), 149 (100) and 134 (94).
Acknowledgements
We wish to thank Faculdade de Farmácia, Universidade de Coimbra for leave of absence of Ana M. T. D. P. V. Cabral. We also wish to thank Chymiotechnon for their financial support.
References
- Grigg, R. Chem. Soc. Rev. 1987, 16, 89–121.
- Barkley, J. V.; Gilchrist, T. L.; Gonsalves, A. M. D. R.; Pinho e Melo, T. M. V. D. Tetrahedron 1995, 51, 13455–13460.
- Grigg, R.; Armstrong, P. Tetrahedron 1989, 45, 7581–7585.
- Garcia, F.; Galvez, C. Synthesis 1985, 143–156.
- Beja, A. M.; Paixao, J. A.; Silva, M. R.; Alte da Veiga, L.; Gonsalves, A. M. D. R.; Pinho e Melo, T. M. V. D.; Cabral, A. M. T. D. P. V. Acta Crystt. 1997, in press.
- Sample Availability: Available from the authors.
© 1998 MDPI. All rights reserved. Molecules website http://www.mdpi.org/molecules/
