Comparison of Antioxidant Capacity and Network Pharmacology of Phloretin and Phlorizin against Neuroinflammation in Traumatic Brain Injury
Abstract
1. Introduction
2. Results
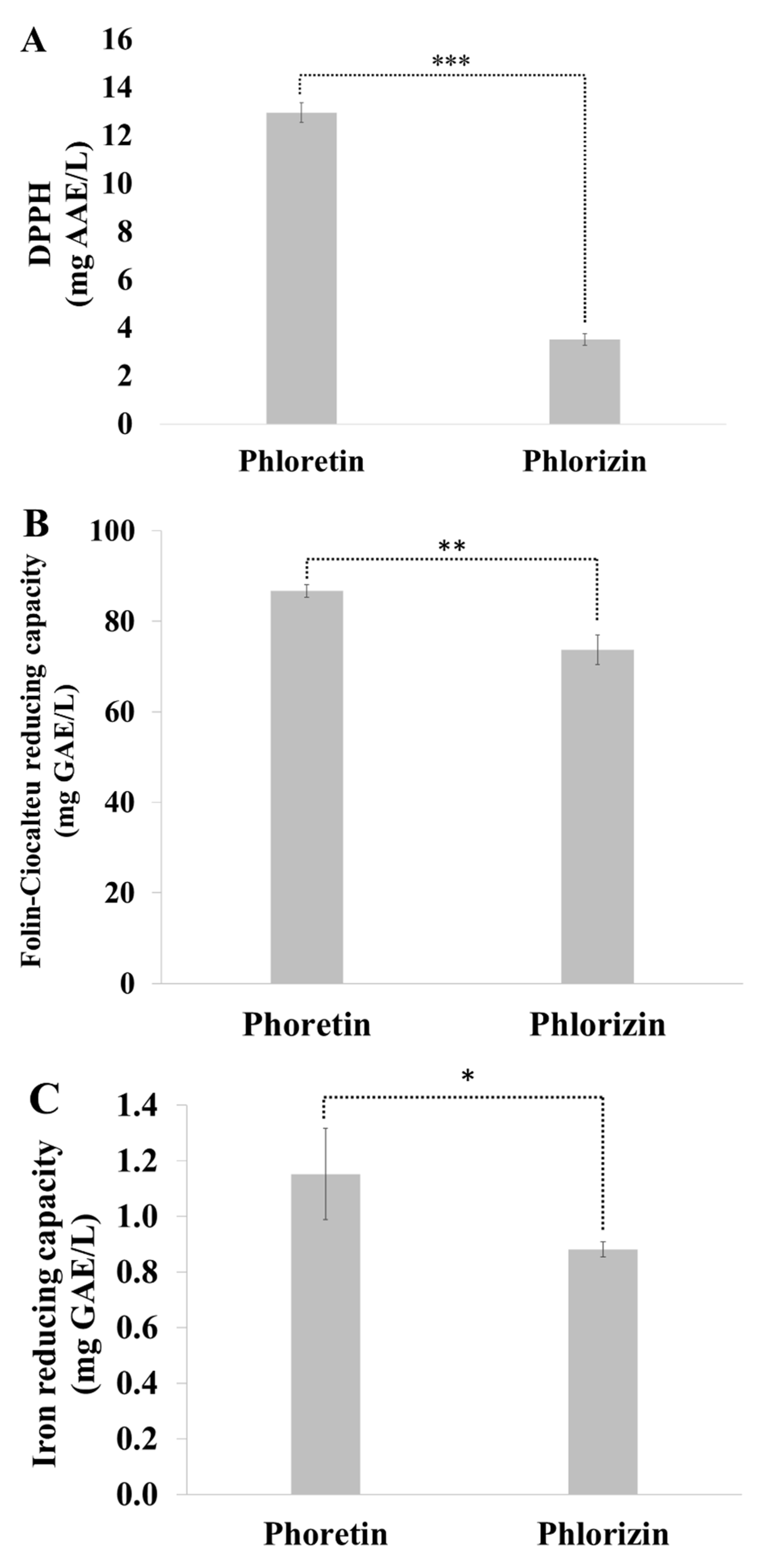
2.1. Antioxidant Activities of Phlorizin and Phloretin
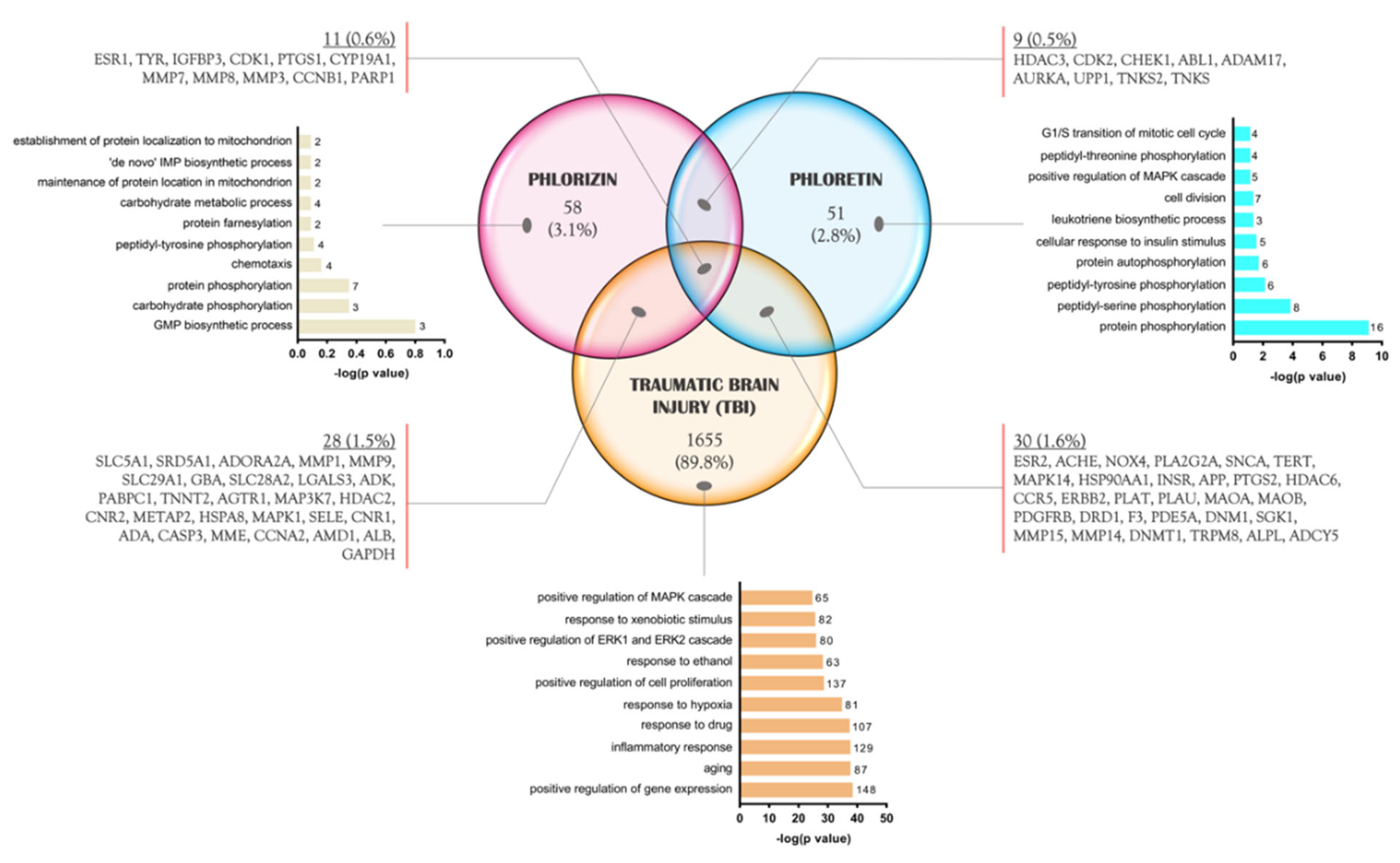
2.2. Potential Molecular Targets of Phlorizin and Phloretin in TBI Pathology
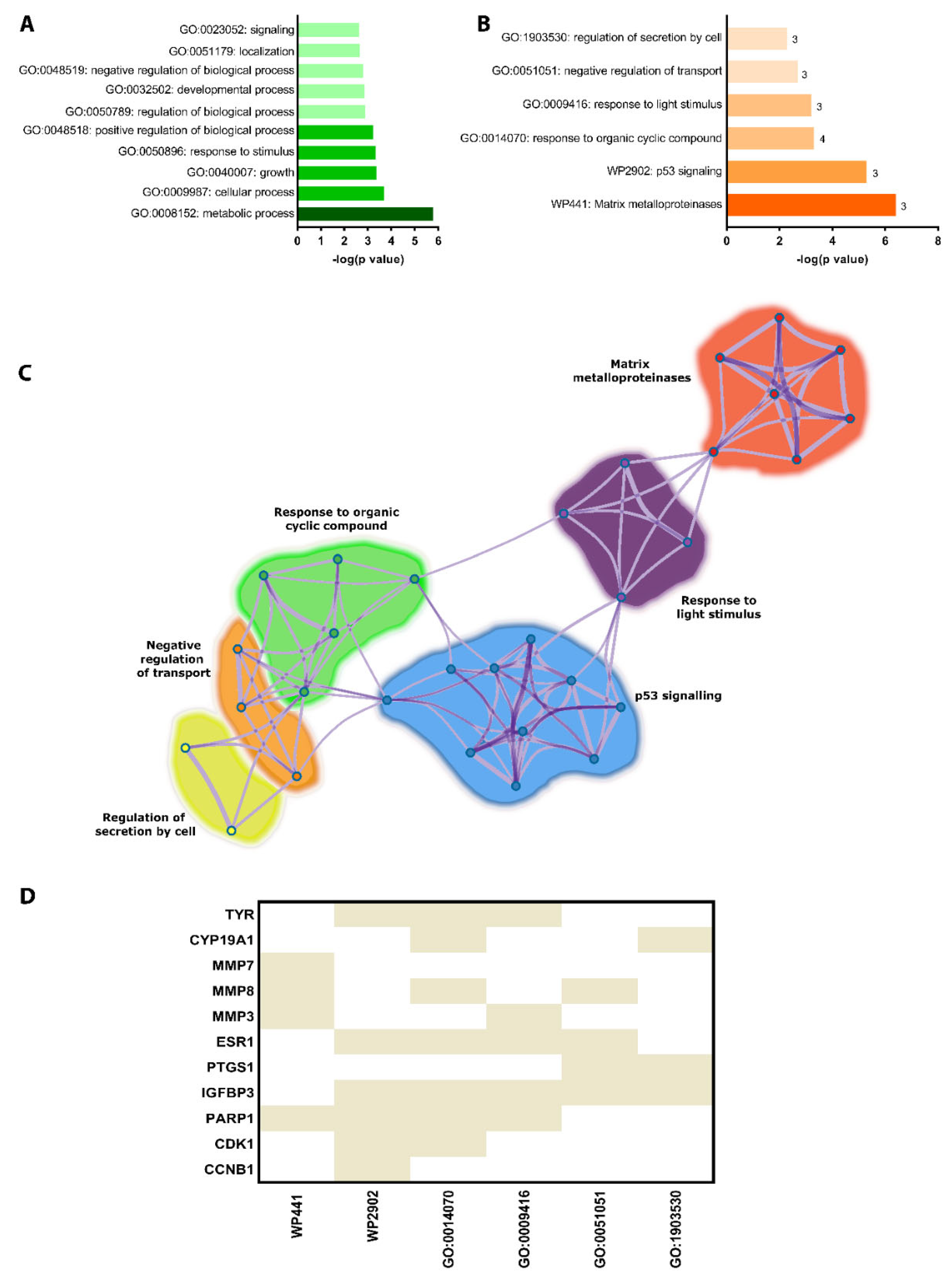
2.3. Enrichment Analysis and Meta-Analysis of Common Proteins
2.4. PPI Analysis of Target Proteins
3. Discussion
4. Materials and Methods
4.1. Identification of Proteins Regulated by Phlorizin and Phloretin in TBI Pathology
4.2. Protein–Protein Interaction (PPI) Network and Functional Enrichment Analysis
4.3. Meta-Analysis of Common Proteins among Phlorizin, Phloretin, and TBI
4.4. Antioxidant Capacity In Vitro
4.5. Statistical Analysis
5. Conclusions and Future Perspectives
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Sample Availability
References
- Li, G.; Liu, H.; He, Y.; Hu, Z.; Gu, Y.; Li, Y.; Ye, Y.; Hu, J. Neurological Symptoms and Their Associations with Inflammatory Biomarkers in the Chronic Phase Following Traumatic Brain Injuries. Front. Psychiatry 2022, 13, 895852. [Google Scholar] [CrossRef] [PubMed]
- Sabet, N.; Soltani, Z.; Khaksari, M. Multipotential and systemic effects of traumatic brain injury. J. Neuroimmunol. 2021, 357, 577619. [Google Scholar] [CrossRef] [PubMed]
- Brazinova, A.; Rehorcikova, V.; Taylor, M.; Buckova, V.; Majdan, M.; Psota, M.; Peeters, W.; Feigin, V.L.; Theadom, A.; Holkovic, L.; et al. Epidemiology of Traumatic Brain Injury in Europe: A Living Systematic Review. J. Neurotrauma 2021, 38, 1411–1440. [Google Scholar] [CrossRef]
- Haarbauer-Krupa, J.; Pugh, M.J.; Prager, E.M.; Harmon, N.; Wolfe, J.; Yaffe, K.C. Epidemiology of Chronic Effects of Traumatic Brain Injury. J. Neurotrauma 2021, 38, 3235–3247. [Google Scholar] [CrossRef]
- De Silva, M.J.; Roberts, I.; Perel, P.; Edwards, P.; Kenward, M.G.; Fernandes, J.; Shakur, H.; Patel, V.; CRASH Trial Collaborators. Patient outcome after traumatic brain injury in high-, middle- and low-income countries: Analysis of data on 8927 patients in 46 countries. Int. J. Epidemiol. 2009, 38, 452–458. [Google Scholar] [CrossRef] [PubMed]
- Galgano, M.; Toshkezi, G.; Qiu, X.; Russell, T.; Chin, L.; Zhao, L.-R. Traumatic Brain Injury: Current Treatment Strategies and Future Endeavors. Cell Transplant. 2017, 26, 1118–1130. [Google Scholar] [CrossRef]
- Capizzi, A.; Woo, J.; Verduzco-Gutierrez, M. Traumatic Brain Injury: An Overview of Epidemiology, Pathophysiology, and Medical Management. Med. Clin. N. Am. 2020, 104, 213–238. [Google Scholar] [CrossRef] [PubMed]
- van Erp, I.A.M.; Michailidou, I.; van Essen, T.A.; van der Jagt, M.; Moojen, W.; Peul, W.C.; Baas, F.; Fluiter, K. Tackling Neuroinflammation After Traumatic Brain Injury: Complement Inhibition as a Therapy for Secondary Injury. Neurotherapeutics 2022, 1–20. [Google Scholar] [CrossRef] [PubMed]
- Hui, Y.; Zhao, H.; Shi, L.; Zhang, H. Traumatic Brain Injury-Mediated Neuroinflammation and Neurological Deficits are Improved by 8-Methoxypsoralen Through Modulating PPARgamma/NF-kappaB Pathway. Neurochem. Res. 2022, 1–16. [Google Scholar] [CrossRef]
- Postolache, T.T.; Wadhawan, A.; Can, A.; Lowry, C.A.; Woodbury, M.; Makkar, H.; Hoisington, A.J.; Scott, A.J.; Potocki, E.; Benros, M.E.; et al. Inflammation in Traumatic Brain Injury. J. Alzheimer’s Dis. 2020, 74, 1–28. [Google Scholar] [CrossRef]
- Bergold, P.J. Treatment of traumatic brain injury with anti-inflammatory drugs. Exp. Neurol. 2016, 275, 367–380. [Google Scholar] [CrossRef]
- Chakrabarti, M.; Das, A.; Samantaray, S.; Smith, J.A.; Banik, N.L.; Haque, A.; Ray, S.K. Molecular mechanisms of estrogen for neuroprotection in spinal cord injury and traumatic brain injury. Rev. Neurosci. 2016, 27, 271–281. [Google Scholar] [CrossRef] [PubMed]
- Schneider, H.J.; Kreitschmann-Andermahr, I.; Ghigo, E.; Stalla, G.K.; Agha, A. Hypothalamopituitary dysfunction following traumatic brain injury and aneurysmal subarachnoid hemorrhage: A systematic review. JAMA 2007, 298, 1429–1438. [Google Scholar] [CrossRef] [PubMed]
- Emelifeonwu, J.A.; Flower, H.; Loan, J.J.; McGivern, K.; Andrews, P.J. Prevalence of Anterior Pituitary Dysfunction Twelve Months or More following Traumatic Brain Injury in Adults: A Systematic Review and Meta-Analysis. J. Neurotrauma 2020, 37, 217–226. [Google Scholar] [CrossRef] [PubMed]
- Brotfain, E.; Gruenbaum, S.E.; Boyko, M.; Kutz, R.; Zlotnik, A.; Klein, M. Neuroprotection by Estrogen and Progesterone in Traumatic Brain Injury and Spinal Cord Injury. Curr. Neuropharmacol. 2016, 14, 641–653. [Google Scholar] [CrossRef] [PubMed]
- Hohl, A.; Mazzuco, T.L.; Coral, M.H.C.; Schwarzbold, M.; Walz, R. Hypogonadism after traumatic brain injury. Arq. Bras. de Endocrinol. Metabol. 2009, 53, 908–914. [Google Scholar] [CrossRef] [PubMed]
- Fortress, A.M.; Avcu, P.; Wagner, A.K.; Dixon, C.E.; Pang, K.C. Experimental traumatic brain injury results in estrous cycle disruption, neurobehavioral deficits, and impaired GSK3beta/beta-catenin signaling in female rats. Exp. Neurol. 2019, 315, 42–51. [Google Scholar] [CrossRef]
- McGovern, A.J.; González, J.; Ramírez, D.; Barreto, G.E. Identification of HMGCR, PPGARG and prohibitin as potential druggable targets of dihydrotestosterone for treatment against traumatic brain injury using system pharmacology. Int. Immunopharmacol. 2022, 108, 108721. [Google Scholar] [CrossRef]
- Giatti, S.; Garcia-Segura, L.M.; Barreto, G.E.; Melcangi, R.C. Neuroactive steroids, neurosteroidogenesis and sex. Prog. Neurobiol. 2019, 176, 1–17. [Google Scholar] [CrossRef]
- Acaz-Fonseca, E.; Avila-Rodriguez, M.; Garcia-Segura, L.M.; Barreto, G.E. Regulation of astroglia by gonadal steroid hormones under physiological and pathological conditions. Prog. Neurobiol. 2016, 144, 5–26. [Google Scholar] [CrossRef]
- Barreto, G.; Veiga, S.; Azcoitia, I.; Garcia-Segura, L.M.; Garcia-Ovejero, D. Testosterone decreases reactive astroglia and reactive microglia after brain injury in male rats: Role of its metabolites, oestradiol and dihydrotestosterone. Eur. J. Neurosci. 2007, 25, 3039–3046. [Google Scholar] [CrossRef] [PubMed]
- Barreto, G.; McGovern, A.; Garcia-Segura, L. Role of Neuroglobin in the Neuroprotective Actions of Estradiol and Estrogenic Compounds. Cells 2021, 10, 1907. [Google Scholar] [CrossRef]
- Azcoitia, I.; Barreto, G.E.; Garcia-Segura, L.M. Molecular mechanisms and cellular events involved in the neuroprotective actions of estradiol. Analysis of sex differences. Front. Neuroendocr. 2019, 55, 100787. [Google Scholar] [CrossRef] [PubMed]
- Hidalgo-Lanussa, O.; Baez-Jurado, E.; Echeverria, V.; Ashraf, G.M.; Sahebkar, A.; Garcia-Segura, L.M.; Melcangi, R.C.; Barreto, G.E. Lipotoxicity, neuroinflammation, glial cells and oestrogenic compounds. J. Neuroendocr. 2020, 32, e12776. [Google Scholar] [CrossRef] [PubMed]
- Crespo-Castrillo, A.; Yanguas-Casás, N.; Arevalo, M.A.; Azcoitia, I.; Barreto, G.E.; Garcia-Segura, L.M. The Synthetic Steroid Tibolone Decreases Reactive Gliosis and Neuronal Death in the Cerebral Cortex of Female Mice After a Stab Wound Injury. Mol. Neurobiol. 2018, 55, 8651–8667. [Google Scholar] [CrossRef] [PubMed]
- Barreto, G.E.; Santos-Galindo, M.; Diz-Chaves, Y.; Pernía, O.; Carrero, P.; Azcoitia, I.; Garcia-Segura, L. Selective Estrogen Receptor Modulators Decrease Reactive Astrogliosis in the Injured Brain: Effects of Aging and Prolonged Depletion of Ovarian Hormones. Endocrinology 2009, 150, 5010–5015. [Google Scholar] [CrossRef]
- Khaksari, M.; Abbasloo, E.; Dehghan, F.; Soltani, Z.; Asadikaram, G. The brain cytokine levels are modulated by estrogen following traumatic brain injury: Which estrogen receptor serves as modulator? Int. Immunopharmacol. 2015, 28, 279–287. [Google Scholar] [CrossRef]
- Khaksari, M.; Soltani, Z.; Shahrokhi, N. Effects of Female Sex Steroids Administration on Pathophysiologic Mechanisms in Traumatic Brain Injury. Transl. Stroke Res. 2018, 9, 393–416. [Google Scholar] [CrossRef]
- Kövesdi, E.; Szabó-Meleg, E.; Abrahám, I.M. The Role of Estradiol in Traumatic Brain Injury: Mechanism and Treatment Potential. Int. J. Mol. Sci. 2020, 22, 11. [Google Scholar] [CrossRef]
- Sachdev, K.R.; Lynch, K.J.; Barreto, G.E. Exploration of novel ligands to target C-C Motif Chemokine Receptor 2 (CCR2) as a promising pharmacological treatment against traumatic brain injury. Biomed. Pharmacother. 2022, 151, 113155. [Google Scholar] [CrossRef]
- Mahmoudi, A.; Heydari, S.; Markina, Y.V.; Barreto, G.E.; Sahebkar, A. Role of statins in regulating molecular pathways following traumatic brain injury: A system pharmacology study. Biomed. Pharmacother. 2022, 153, 113304. [Google Scholar] [CrossRef] [PubMed]
- Areiza-Mazo, N.; Robles, J.; Zamudio-Rodriguez, J.A.; Giraldez, L.; Echeverria, V.; Barrera-Bailon, B.; Aliev, G.; Sahebkar, A.; Ashraf, G.M.; Barreto, G.E. Extracts of Physalis peruviana Protect Astrocytic Cells Under Oxidative Stress With Rotenone. Front. Chem. 2018, 6, 276. [Google Scholar] [CrossRef] [PubMed]
- Keshavarzi, Z.; Shakeri, F.; Barreto, G.E.; Bibak, B.; Sathyapalan, T.; Sahebkar, A. Medicinal plants in traumatic brain injury: Neuroprotective mechanisms revisited. Biofactors 2019, 45, 517–535. [Google Scholar] [CrossRef] [PubMed]
- Sahebkar, A.; Sathyapalan, T.; Guest, P.C.; Barreto, G.E. Identification of difluorinated curcumin molecular targets linked to traumatic brain injury pathophysiology. Biomed. Pharmacother. 2022, 148, 112770. [Google Scholar] [CrossRef] [PubMed]
- Lucke-Wold, B.P.; Logsdon, A.F.; Nguyen, L.; Eltanahay, A.; Turner, R.C.; Bonasso, P.; Knotts, C.; Moeck, A.; Maroon, J.C.; Bailes, J.E.; et al. Supplements, nutrition, and alternative therapies for the treatment of traumatic brain injury. Nutr. Neurosci. 2018, 21, 79–91. [Google Scholar] [CrossRef]
- Mazo, N.A.; Echeverria, V.; Cabezas, R.; Avila-Rodríguez, M.; Tarasov, V.V.; Yarla, N.S.; Aliev, G.; Barreto, G.E. Medicinal Plants as Protective Strategies Against Parkinson’s Disease. Curr. Pharm. Des. 2017, 23, 4180–4188. [Google Scholar] [CrossRef]
- Albarracin, S.L.; Stab, B.; Casas, Z.; Sutachan, J.-J.; Samudio, I.; Gonzalez, J.; Gonzalo, L.; Capani, F.; Morales, L.; Barreto, G.E. Effects of natural antioxidants in neurodegenerative disease. Nutr. Neurosci. 2012, 15, 1–9. [Google Scholar] [CrossRef]
- Battaglini, D.; Siwicka-Gieroba, D.; Rocco, P.R.; Cruz, F.F.; Silva, P.L.; Dabrowski, W.; Brunetti, I.; Patroniti, N.; Pelosi, P.; Robba, C. Novel Synthetic and Natural Therapies for Traumatic Brain Injury. Curr. Neuropharmacol. 2021, 19, 1661–1687. [Google Scholar] [CrossRef]
- Mendes, R.A.; e Silva, B.L.S.; Takeara, R.; Freitas, R.G.; Brown, A.; de Souza, G.L.C. Probing the antioxidant potential of phloretin and phlorizin through a computational investigation. J. Mol. Model. 2018, 24, 101. [Google Scholar] [CrossRef]
- Zielinska, D.; Laparra-Llopis, J.M.; Zielinski, H.; Szawara-Nowak, D.; Giménez-Bastida, J.A. Role of Apple Phytochemicals, Phloretin and Phloridzin, in Modulating Processes Related to Intestinal Inflammation. Nutrients 2019, 11, 1173. [Google Scholar] [CrossRef]
- Barreca, D.; Currò, M.; Bellocco, E.S.; Ficarra, S.; Laganà, G.; Tellone, E.; Giunta, M.L.; Visalli, G.; Caccamo, D.; Galtieri, A.; et al. Neuroprotective effects of phloretin and its glycosylated derivative on rotenone-induced toxicity in human SH-SY5Y neuronal-like cells. Biofactors 2017, 43, 549–557. [Google Scholar] [CrossRef] [PubMed]
- Park, S.; Park, S.-K. Anti-Oxidant and Anti-Aging Effects of Phlorizin Are Mediated by DAF-16-Induced Stress Response and Autophagy in Caenorhabditis elegans. Antioxidants 2022, 11, 1996. [Google Scholar] [CrossRef] [PubMed]
- Un, H.; Ugan, R.A.; Gurbuz, M.A.; Bayir, Y.; Kahramanlar, A.; Kaya, G.; Cadirci, E.; Halici, Z. Phloretin and phloridzin guard against cisplatin-induced nephrotoxicity in mice through inhibiting oxidative stress and inflammation. Life Sci. 2021, 266, 118869. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Y.; Liu, C.; Lai, X.; Hou, S.; Zeng, X.; Li, X. Immunomodulatory activities of phlorizin metabolites in lipopolysaccharide-stimulated RAW264.7 cells. Biomed. Pharmacother. 2017, 91, 49–53. [Google Scholar] [CrossRef] [PubMed]
- Xie, L.; Deng, Z.; Zhang, J.; Dong, H.; Wang, W.; Xing, B.; Liu, X. Comparison of Flavonoid O-Glycoside, C-Glycoside and Their Aglycones on Antioxidant Capacity and Metabolism during In Vitro Digestion and In Vivo. Foods 2022, 11, 882. [Google Scholar] [CrossRef]
- Xiao, Z.; He, L.; Hou, X.; Wei, J.; Ma, X.; Gao, Z.; Yuan, Y.; Xiao, J.; Li, P.; Yue, T. Relationships between Structure and Antioxidant Capacity and Activity of Glycosylated Flavonols. Foods 2021, 10, 849. [Google Scholar] [CrossRef]
- Crespy, V.; Aprikian, O.; Morand, C.; Besson, C.; Manach, C.; Demigné, C.; Rémésy, C. Bioavailability of Phloretin and Phloridzin in Rats. J. Nutr. 2001, 131, 3227–3230. [Google Scholar] [CrossRef]
- Costa, A.J.; Oliveira, R.B.; Wachilewski, P.; Nishino, M.S.; Bassani, T.B.; Stilhano, R.S.; Cerutti, J.M.; Nozima, B.; Porto, C.S.; da Silva Pereira, G.J.; et al. Membrane estrogen receptor ERalpha activation improves tau clearance via autophagy induction in a tauopathy cell model. Brain Res. 2022, 1795, 148079. [Google Scholar] [CrossRef]
- Qu, N.; Wang, X.-M.; Zhang, T.; Zhang, S.-F.; Li, Y.; Cao, F.-Y.; Wang, Q.; Ning, L.-N.; Tian, Q. Estrogen Receptor α Agonist is Beneficial for Young Female Rats Against Chronic Unpredicted Mild Stress-Induced Depressive Behavior and Cognitive Deficits. J. Alzheimer’s Dis. 2020, 77, 1077–1093. [Google Scholar] [CrossRef]
- Lai, Y.; Zhu, B.; Sun, F.; Luo, D.; Ma, Y.; Luo, B.; Tang, J.; Xiong, M.; Liu, L.; Long, Y.; et al. Estrogen receptor α promotes Cav1.2 ubiquitination and degradation in neuronal cells and in APP/PS1 mice. Aging Cell 2019, 18, e12961. [Google Scholar] [CrossRef]
- Stary, C.M.; Xu, L.; Li, L.; Sun, X.; Ouyang, Y.-B.; Xiong, X.; Zhao, J.; Giffard, R.G. Inhibition of miR-181a protects female mice from transient focal cerebral ischemia by targeting astrocyte estrogen receptor-α. Mol. Cell. Neurosci. 2017, 82, 118–125. [Google Scholar] [CrossRef] [PubMed]
- Cordeau, P.; Lalancette-Hébert, M.; Weng, Y.C.; Kriz, J. Estrogen receptors alpha mediates postischemic inflammation in chronically estrogen-deprived mice. Neurobiol. Aging 2016, 40, 50–60. [Google Scholar] [CrossRef] [PubMed]
- Chen, S.-H.; Chang, C.-Y.; Chang, H.-K.; Chen, W.-C.; Lin, M.-T.; Wang, J.-J.; Chen, J.C.-Y.; Chang, F.-M. Premarin stimulates estrogen receptor-α to protect against traumatic brain injury in male rats. Crit. Care Med. 2009, 37, 3097–3106. [Google Scholar] [CrossRef] [PubMed]
- Li, L.-Z.; Bao, Y.-J.; Zhao, M. 17Beta-estradiol attenuates programmed cell death in cortical pericontusional zone following traumatic brain injury via upregulation of ERalpha and inhibition of caspase-3 activation. Neurochem. Int. 2011, 58, 126–133. [Google Scholar] [CrossRef]
- Bao, Y.-J.; Li, L.-Z.; Li, X.-G.; Wang, Y.-J. 17Beta-estradiol differentially protects cortical pericontusional zone from programmed cell death after traumatic cerebral contusion at distinct stages via non-genomic and genomic pathways. Mol. Cell. Neurosci. 2011, 48, 185–194. [Google Scholar] [CrossRef]
- Asl, S.Z.; Khaksari, M.; Khachki, A.S.; Shahrokhi, N.; Nourizade, S. Contribution of estrogen receptors alpha and beta in the brain response to traumatic brain injury. J. Neurosurg. 2013, 119, 353–361. [Google Scholar] [CrossRef]
- Na, W.; Lee, J.Y.; Kim, W.-S.; Yune, T.Y.; Ju, B.-G. 17β-Estradiol Ameliorates Tight Junction Disruption via Repression of MMP Transcription. Mol. Endocrinol. 2015, 29, 1347–1361. [Google Scholar] [CrossRef]
- Napieralski, J.A.; Raghupathi, R.; McIntosh, T.K. The tumor-suppressor gene, p53, is induced in injured brain regions following experimental traumatic brain injury. Brain Res. Mol. Brain Res. 1999, 71, 78–86. [Google Scholar] [CrossRef]
- Raz, L.; Zhang, Q.-G.; Han, D.; Dong, Y.; De Sevilla, L.; Brann, D.W. Acetylation of the Pro-Apoptotic Factor, p53 in the Hippocampus following Cerebral Ischemia and Modulation by Estrogen. PLoS ONE 2011, 6, e27039. [Google Scholar] [CrossRef]
- Breinholt, V.; Larsen, J.C. Detection of Weak Estrogenic Flavonoids Using a Recombinant Yeast Strain and a Modified MCF7 Cell Proliferation Assay. Chem. Res. Toxicol. 1998, 11, 622–629. [Google Scholar] [CrossRef]
- Ye, H.; Shaw, I.C. Food flavonoid ligand structure/estrogen receptor-α affinity—Toxicity or food functionality? Food Chem. Toxicol. 2019, 129, 328–336. [Google Scholar] [CrossRef] [PubMed]
- Shannon, P.; Markiel, A.; Ozier, O.; Baliga, N.S.; Wang, J.T.; Ramage, D.; Amin, N.; Schwikowski, B.; Ideker, T. Cytoscape: A software environment for integrated models of Biomolecular Interaction Networks. Genome Res. 2003, 13, 2498–2504. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Y.; Zhou, B.; Pache, L.; Chang, M.; Khodabakhshi, A.H.; Tanaseichuk, O.; Benner, C.; Chanda, S.K. Metascape provides a biologist-oriented resource for the analysis of systems-level datasets. Nat. Commun. 2019, 10, 1523. [Google Scholar] [CrossRef] [PubMed]
- Santos, J.S.; Escher, G.B.; Carmo, M.V.D.; Azevedo, L.; Marques, M.B.; Daguer, H.; Molognoni, L.; Genovese, M.I.; Wen, M.; Zhang, L.; et al. A new analytical concept based on chemistry and toxicology for herbal extracts analysis: From phenolic composition to bioactivity. Food Res. Int. 2020, 132, 109090. [Google Scholar] [CrossRef]
- Granato, D.; Reshamwala, D.; Korpinen, R.; Azevedo, L.; do Carmo, M.A.V.; Cruz, T.M.; Marques, M.B.; Wen, M.; Zhang, L.; Marjomäki, V.; et al. From the forest to the plate—Hemicelluloses, galactoglucomannan, glucuronoxylan, and phenolic-rich extracts from unconventional sources as functional food ingredients. Food Chem. 2022, 381, 132284. [Google Scholar] [CrossRef]

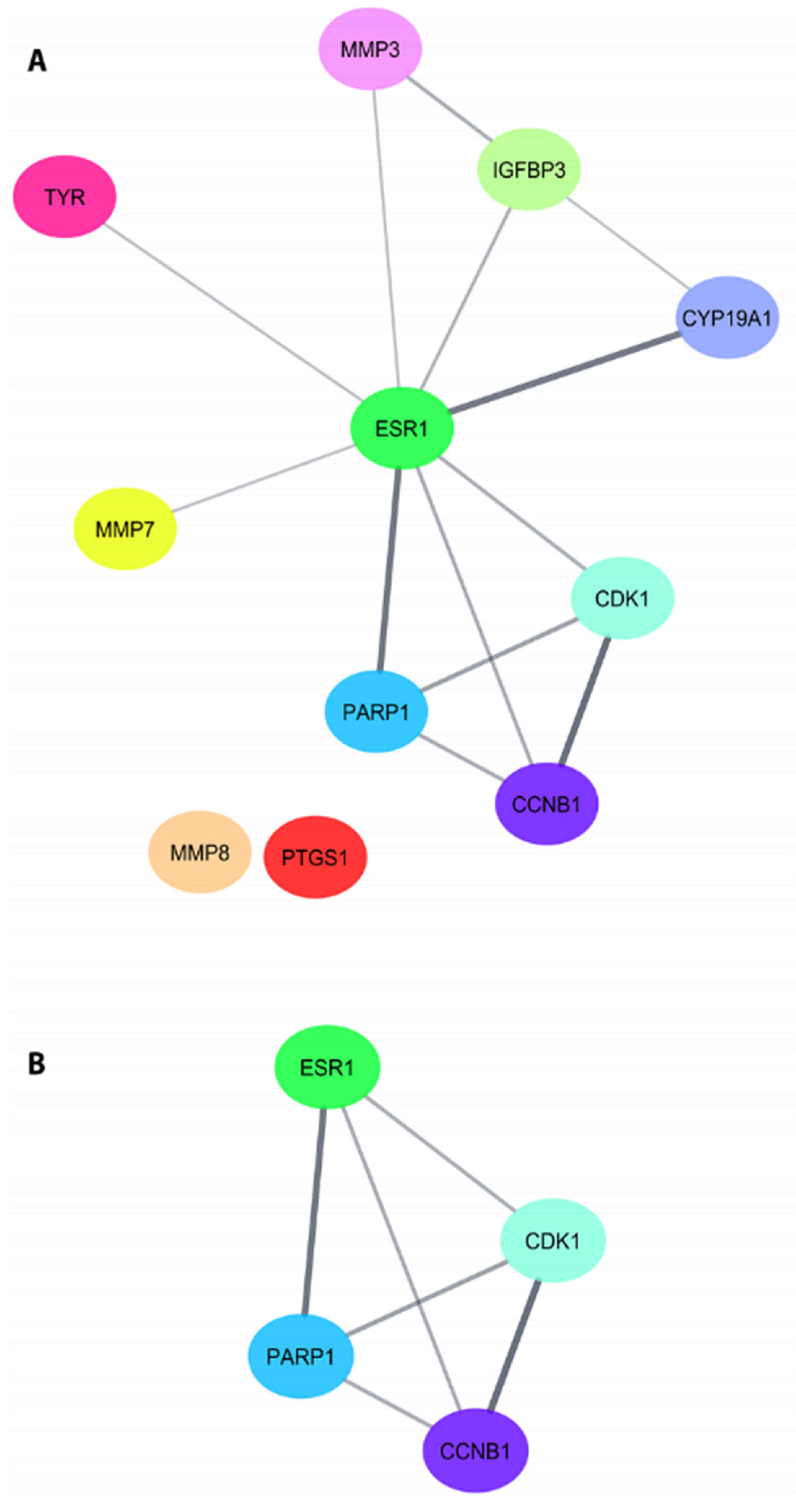
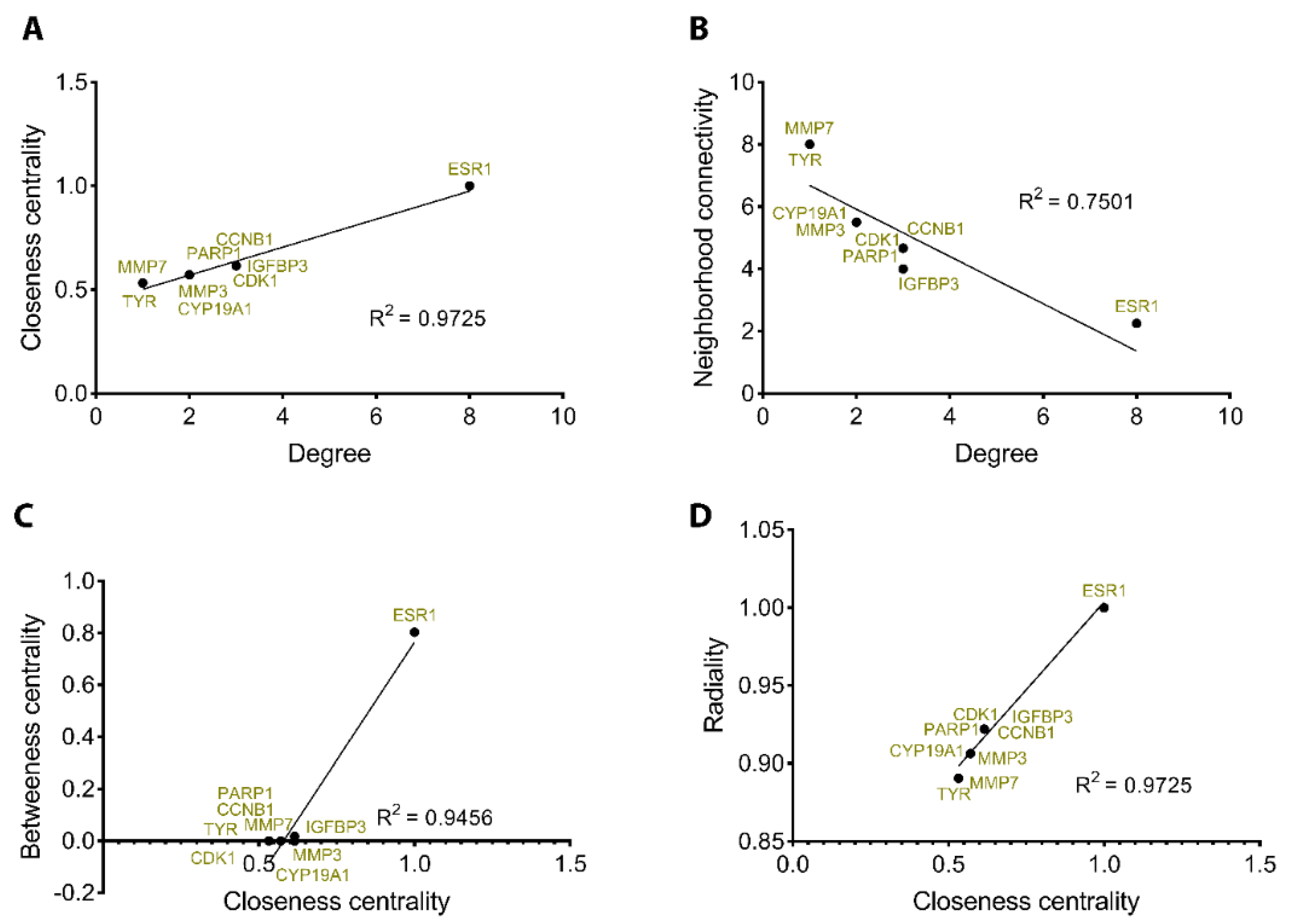
| Protein | Degree | Betweenness Centrality | Closeness Centrality | Neighborhood Connectivity | Radiality |
|---|---|---|---|---|---|
| ESR1 | 8 | 0.803571429 | 1 | 2.25 | 1 |
| IGFBP3 | 3 | 0.017857143 | 0.615384615 | 4 | 0.921875 |
| CCNB1 | 3 | 0 | 0.615384615 | 4.666666667 | 0.921875 |
| PARP1 | 3 | 0 | 0.615384615 | 4.666666667 | 0.921875 |
| CDK1 | 3 | 0 | 0.615384615 | 4.666666667 | 0.921875 |
| MMP3 | 2 | 0 | 0.571428571 | 5.5 | 0.90625 |
| CYP19A1 | 2 | 0 | 0.571428571 | 5.5 | 0.90625 |
| TYR | 1 | 0 | 0.533333333 | 8 | 0.890625 |
| MMP7 | 1 | 0 | 0.533333333 | 8 | 0.890625 |
| MMP8 | 0 | 0 | 0 | 0 | Infinity |
| PTGS1 | 0 | 0 | 0 | 0 | Infinity |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ongay, K.K.; Granato, D.; Barreto, G.E. Comparison of Antioxidant Capacity and Network Pharmacology of Phloretin and Phlorizin against Neuroinflammation in Traumatic Brain Injury. Molecules 2023, 28, 919. https://doi.org/10.3390/molecules28030919
Ongay KK, Granato D, Barreto GE. Comparison of Antioxidant Capacity and Network Pharmacology of Phloretin and Phlorizin against Neuroinflammation in Traumatic Brain Injury. Molecules. 2023; 28(3):919. https://doi.org/10.3390/molecules28030919
Chicago/Turabian StyleOngay, Kubra Kizil, Daniel Granato, and George E. Barreto. 2023. "Comparison of Antioxidant Capacity and Network Pharmacology of Phloretin and Phlorizin against Neuroinflammation in Traumatic Brain Injury" Molecules 28, no. 3: 919. https://doi.org/10.3390/molecules28030919
APA StyleOngay, K. K., Granato, D., & Barreto, G. E. (2023). Comparison of Antioxidant Capacity and Network Pharmacology of Phloretin and Phlorizin against Neuroinflammation in Traumatic Brain Injury. Molecules, 28(3), 919. https://doi.org/10.3390/molecules28030919







