Pharmacological Effects of Shikonin and Its Potential in Skin Repair: A Review
Abstract
1. Introduction
2. Biological Activity
2.1. Anti-Inflammatory
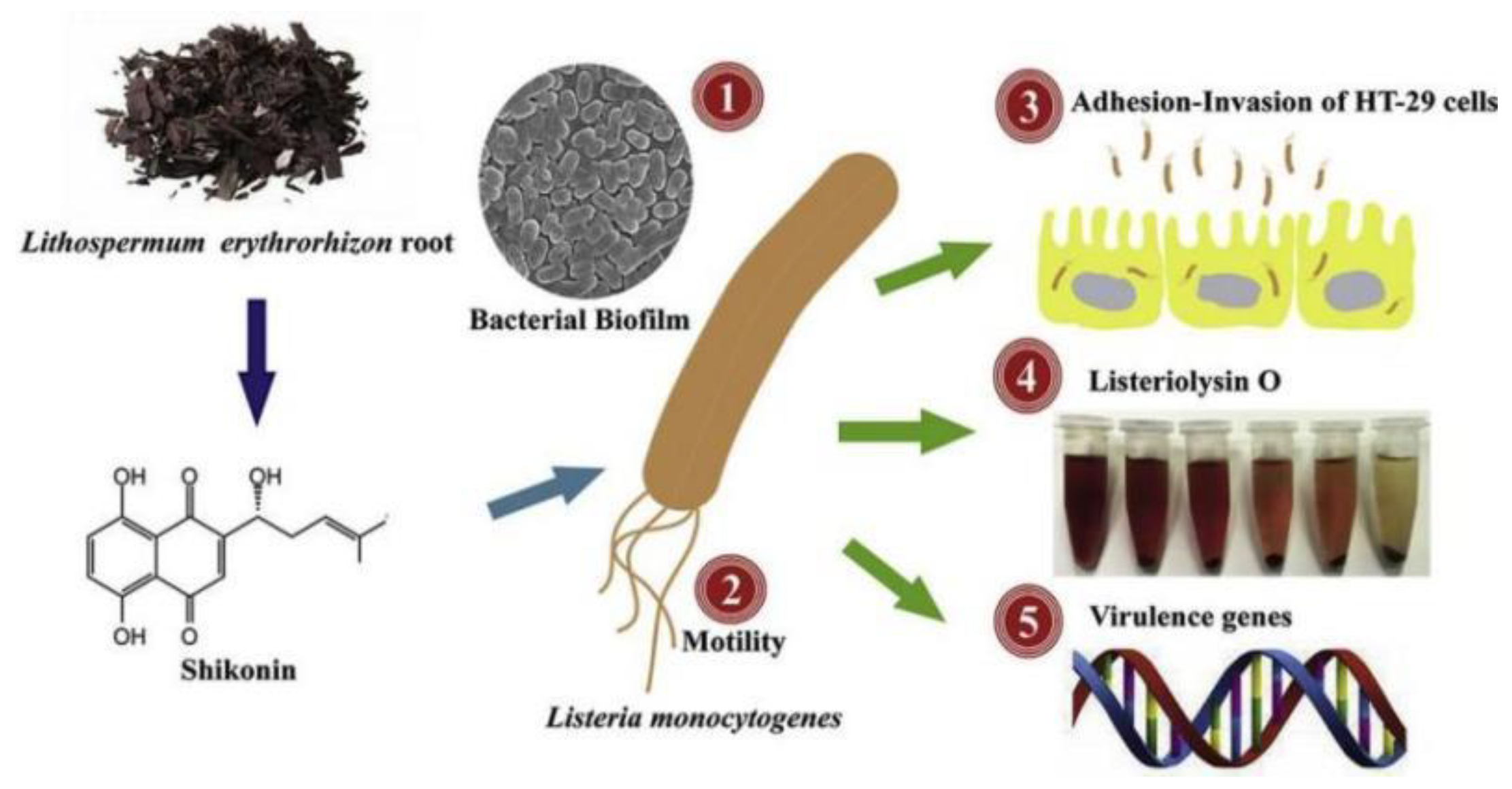
2.2. Antimicrobial
2.3. Antiviral
2.4. Anti-Tumor
2.5. Anti-Hepatic Injury
2.6. Promoting Wound Healing
2.7. Reduce Cognitive and Behavioral Disorders
3. Progress of Research Related to the Treatment of Skin Diseases Using Shikonin
3.1. Dermatitis
3.2. Psoriasis
3.3. Skin Cancer
3.4. Scarring
3.5. New Dosage Forms of Shikonin in Diseases
4. Conclusions and Outlook
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Sun, Q.; Gong, T.; Liu, M.; Ren, S.; Yang, H.; Zeng, S.; Zhao, H.; Chen, L.; Ming, T.; Meng, X.; et al. Shikonin, a naphthalene ingredient: Therapeutic actions, pharmacokinetics, toxicology, clinical trials and pharmaceutical researches. Phytomedicine 2022, 94, 153805. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Z.; Bai, J.; Zeng, Y.; Cai, M.; Yao, Y.; Wu, H.; You, L.; Dong, X.; Ni, J. Pharmacology, toxicity and pharmacokinetics of acetylshikonin: A review. Pharm. Biol. 2020, 58, 950–958. [Google Scholar] [CrossRef] [PubMed]
- Kaur, K.; Sharma, R.; Singh, A.; Attri, S.; Arora, S.; Kaur, S.; Bedi, N. Pharmacological and analytical aspects of alkannin/shikonin and their derivatives: An update from 2008 to 2022. Chin. Herb. Med. 2022, 14, 511–527. [Google Scholar] [CrossRef] [PubMed]
- Sevimli-Gur, C.; Akgun, I.H.; Deliloglu-Gurhan, I.; Korkmaz, K.S.; Bedir, E. Cytotoxic Naphthoquinones from Alkanna cappadocica (perpendicular). J. Nat. Prod. 2010, 73, 860–864. [Google Scholar] [CrossRef] [PubMed]
- Malik, S.; Brudzynska, P.; Khan, M.R.; Sytar, O.; Makhzoum, A.; Sionkowska, A. Natural Plant-Derived Compounds in Food and Cosmetics: A Paradigm of Shikonin and Its Derivatives. Materials 2023, 16, 4377. [Google Scholar] [CrossRef]
- Lu, L.; Qin, A.; Huang, H.; Zhou, P.; Zhang, C.; Liu, N.; Li, S.; Wen, G.; Zhang, C.; Dong, W.; et al. Shikonin extracted from medicinal Chinese herbs exerts anti-inflammatory effect via proteasome inhibition. Eur. J. Pharmacol. 2011, 658, 242–247. [Google Scholar] [CrossRef]
- Shen, C.C.; Syu, W.J.; Li, S.Y.; Lin, C.H.; Lee, G.H.; Sun, C.M. Antimicrobial activities of naphthazarins from Arnebia euchroma. J. Nat. Prod. 2002, 65, 1857–1862. [Google Scholar] [CrossRef] [PubMed]
- Hu, J.; Feng, K.; Cong, Y.; Li, X.; Jiang, Y.; Jiao, X.; Li, Y.; Zhang, Y.; Dong, X.; Lu, W.; et al. Nanosized Shikonin-Fe(III) Coordination Material for Synergistic Wound Treatment: An Initial Explorative Study. ACS Appl. Mater. Interfaces 2022, 14, 56510–56524. [Google Scholar] [CrossRef]
- Yadav, S.; Sharma, A.; Nayik, G.A.; Cooper, R.; Bhardwaj, G.; Sohal, H.S.; Mutreja, V.; Kaur, R.; Areche, F.O.; Aloudat, M.; et al. Review of Shikonin and Derivatives: Isolation, Chemistry, Biosynthesis, Pharmacology and Toxicology. Front. Pharmacol. 2022, 13, 905755. [Google Scholar] [CrossRef]
- Huang, X.Y.; Fu, H.L.; Tang, H.Q.; Yin, Z.Q.; Zhang, W.; Shu, G.; Yin, L.Z.; Zhao, L.; Yan, X.R.; Lin, J.C. Optimization Extraction of Shikonin Using Ultrasound-Assisted Response Surface Methodology and Antibacterial Studies. Evid. Based Complement Altern. Med. 2020, 2020, 1208617. [Google Scholar] [CrossRef]
- Liu, T.; Ma, C.; Yang, L.; Wang, W.; Sui, X.; Zhao, C.; Zu, Y. Optimization of shikonin homogenate extraction from Arnebia euchroma using response surface methodology. Molecules 2013, 18, 466–481. [Google Scholar] [CrossRef] [PubMed]
- Dabiri, M.; Salimi, S.; Ghassempour, A.; Rassouli, A.; Talebi, M. Optimization of microwave-assisted extraction for alizarin and purpurin in Rubiaceae plants and its comparison with conventional extraction methods. J. Sep. Sci. 2005, 28, 387–396. [Google Scholar] [CrossRef] [PubMed]
- Slominski, A.T.; Slominski, R.M.; Raman, C.; Chen, J.Y.; Athar, M.; Elmets, C. Neuroendocrine signaling in the skin with a special focus on the epidermal neuropeptides. Am. J. Physiol. Cell Physiol. 2022, 323, C1757–C1776. [Google Scholar] [CrossRef]
- Lianhua, H.E.; Huijie, L.; Qingxia, Q.; Juan, H.E.; Jian, C.; Yiping, H.U.; Yueming, C.; Desheng, S.; Yu, S.; Qingwen, W. Shikonin alleviates collagen-induced arthritis mice by inhibiting M1 macrophage polarization. J. Tradit. Chin. Med. 2022, 42, 932–939. [Google Scholar] [CrossRef] [PubMed]
- Wang, A.; Fang, S.; Zhong, L.; Lu, M.; Zhou, H.; Huang, W.; Li, L.; Gao, W.; Yin, Z. Shikonin, a promising therapeutic drug for osteoarthritis that acts via autophagy activation. Int. Immunopharmacol. 2022, 106, 108563. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.Y.; Liu, X.; Zhang, X.; Zhang, J. Shikonin improve sepsis-induced lung injury via regulation of miRNA-140-5p/TLR4-a vitro and vivo study. J. Cell. Biochem. 2020, 121, 2103–2117. [Google Scholar] [CrossRef] [PubMed]
- Biscaia, M.; Llorente, R.; Gomez, J.; Grassi, D.; Vega-Avelaira, D. Shikonin inhibits microglia activation and reduces CFA-induced mechanical hyperalgesia in an animal model of pain. Biomed. Pharmacother. 2022, 150, 112961. [Google Scholar] [CrossRef]
- Andujar, I.; Rios, J.L.; Giner, R.M.; Recio, M.C. Shikonin promotes intestinal wound healing in vitro via induction of TGF-beta release in IEC-18 cells. Eur. J. Pharm. Sci. 2013, 49, 637–641. [Google Scholar] [CrossRef] [PubMed]
- Cardoso, M.; Gaspar, V.M.; Ferreira, C.; Silvestre, R.; Duarte, I.F.; Mano, J.F. Macrophage-targeted shikonin-loaded nanogels for modulation of inflammasome activation. Nanomedicine 2022, 42, 102548. [Google Scholar] [CrossRef] [PubMed]
- Maliszewska, I.; Czapka, T. Electrospun polymer nanofibers with antimicrobial activity. Polymers 2022, 14, 1661. [Google Scholar] [CrossRef]
- Zou, Y.; Sun, Y.; Shi, W.; Wan, B.; Zhang, H. Dual-functional shikonin-loaded quaternized chitosan/polycaprolactone nanofibrous film with pH-sensing for active and intelligent food packaging. Food Chem. 2023, 399, 133962. [Google Scholar] [CrossRef] [PubMed]
- Arampatzis, A.S.; Kontogiannopoulos, K.N.; Theodoridis, K.; Aggelidou, E.; Rat, A.; Willems, A.; Tsivintzelis, I.; Papageorgiou, V.P.; Kritis, A.; Assimopoulou, A.N. Electrospun wound dressings containing bioactive natural products: Physico-chemical characterization and biological assessment. Biomater. Res. 2021, 25, 23. [Google Scholar] [CrossRef] [PubMed]
- Priyadarshi, R.; Rhim, J. Shikonin: Extraction, properties and applications in active and intelligent packaging. Packag. Technol. Sci. 2022, 35, 863–877. [Google Scholar] [CrossRef]
- Lee, Y.S.; Lee, D.Y.; Kim, Y.B.; Lee, S.W.; Cha, S.W.; Park, H.W.; Kim, G.S.; Kwon, D.Y.; Lee, M.H.; Han, S.H. The mechanism underlying the antibacterial activity of shikonin against methicillin-resistant staphylococcus aureus. Evid. Based Complement Altern. Med. 2015, 2015, 520578. [Google Scholar] [CrossRef] [PubMed]
- Roy, S.; Kim, H.; Rhim, J. Synthesis of carboxymethyl cellulose and agar-based multifunctional films reinforced with cellulose nanocrystals and shikonin. ACS Appl. Polym. Mater. 2021, 3, 1060–1069. [Google Scholar] [CrossRef]
- Roy, S.; Kim, H.J.; Rhim, J.W. Effect of blended colorants of anthocyanin and shikonin on carboxymethyl cellulose/agar-based smart packaging film. Int. J. Biol. Macromol. 2021, 183, 305–315. [Google Scholar] [CrossRef] [PubMed]
- Roy, S.; Rhim, J. Preparation of gelatin/carrageenan-based color-indicator film integrated with shikonin and propolis for smart food packaging applications. ACS Appl. Bio Mater. 2021, 4, 770–779. [Google Scholar] [CrossRef]
- Wan, Y.; Wang, X.; Zhang, P.; Zhang, M.; Kou, M.; Shi, C.; Peng, X.; Wang, X. Control of foodborne staphylococcus aureus by shikonin, a natural extract. Foods 2021, 10, 2954. [Google Scholar] [CrossRef]
- Pang, C.; Chen, J.; Liu, S.; Cao, Y.; Miao, H. In vitro antifungal activity of shikonin against candida albicans by inducing cellular apoptosis and necrosis. Mol. Biol. Rep. 2023, 50, 1079–1087. [Google Scholar] [CrossRef]
- Li, J.; Li, S.; Li, H.; Guo, X.; Du, G.; Yang, Y.; Wang, X.; Zhang, C.; Shan, Z.; Xia, X.; et al. Antibiofilm activity of shikonin against listeria monocytogenes and inhibition of key virulence factors. Food Control 2021, 120, 107558. [Google Scholar] [CrossRef]
- Balaha, M.F.; Alamer, A.A.; Eisa, A.A.; Aljohani, H.M. Shikonin Alleviates Gentamicin-Induced Renal Injury in Rats by Targeting Renal Endocytosis, SIRT1/Nrf2/HO-1, TLR-4/NF-κB/MAPK, and PI3K/Akt Cascades. Antibiotics 2023, 12, 826. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Han, H.; Qiu, H.; Lin, H.; Yu, L.; Zhu, W.; Qi, J.; Yang, R.; Pang, Y.; Wang, X.; et al. Antiviral activity of a synthesized shikonin ester against influenza A (H1N1) virus and insights into its mechanism. Biomed. Pharmacother. 2017, 93, 636–645. [Google Scholar] [CrossRef] [PubMed]
- Chen, X.; Yang, L.; Zhang, N.; Turpin, J.A.; Buckheit, R.W.; Osterling, C.; Oppenheim, J.J.; Howard, O.M. Shikonin, a component of chinese herbal medicine, inhibits chemokine receptor function and suppresses human immunodeficiency virus type 1. Antimicrob. Agents Chemother. 2003, 47, 2810–2816. [Google Scholar] [CrossRef] [PubMed]
- Gong, K.; Li, W. Shikonin, a Chinese plant-derived naphthoquinone, induces apoptosis in hepatocellular carcinoma cells through reactive oxygen species: A potential new treatment for hepatocellular carcinoma. Free Radic. Biol. Med. 2011, 51, 2259–2271. [Google Scholar] [CrossRef] [PubMed]
- Mao, X.; Yu, C.R.; Li, W.H.; Li, W.X. Induction of apoptosis by shikonin through a ROS/JNK-mediated process in Bcr/Abl-positive chronic myelogenous leukemia (CML) cells. Cell Res. 2008, 18, 879–888. [Google Scholar] [CrossRef] [PubMed]
- Moon, J.; Koh, S.S.; Malilas, W.; Cho, I.R.; Kaewpiboon, C.; Kaowinn, S.; Lee, K.; Jhun, B.H.; Choi, Y.W.; Chung, Y.H. Acetylshikonin induces apoptosis of hepatitis b virus x protein-expressing human hepatocellular carcinoma cells via endoplasmic reticulum stress. Eur. J. Pharmacol. 2014, 735, 132–140. [Google Scholar] [CrossRef]
- Guo, C.; He, J.; Song, X.; Tan, L.; Wang, M.; Jiang, P.; Li, Y.; Cao, Z.; Peng, C. Pharmacological properties and derivatives of shikonin-a review in recent years. Pharmacol. Res. 2019, 149, 104463. [Google Scholar] [CrossRef]
- Ji, W.; Sun, X.; Gao, Y.; Lu, M.; Zhu, L.; Wang, D.; Hu, C.; Chen, J.; Cao, P. Natural Compound Shikonin Is a Novel PAK1 Inhibitor and Enhances Efficacy of Chemotherapy against Pancreatic Cancer Cells. Molecules 2022, 27, 2747. [Google Scholar] [CrossRef]
- Lohberger, B.; Glanzer, D.; Kaltenegger, H.; Eck, N.; Leithner, A.; Bauer, R.; Kretschmer, N.; Steinecker-Frohnwieser, B. Shikonin derivatives cause apoptosis and cell cycle arrest in human chondrosarcoma cells via death receptors and MAPK regulation. BMC Cancer 2022, 22, 758. [Google Scholar] [CrossRef]
- Ma, X.; Yu, M.; Hao, C.; Yang, W. Shikonin induces tumor apoptosis in glioma cells via endoplasmic reticulum stress, and Bax/Bak mediated mitochondrial outer membrane permeability. J. Ethnopharmacol. 2020, 263, 113059. [Google Scholar] [CrossRef]
- Lee, J.H.; Han, S.H.; Kim, Y.M.; Kim, S.H.; Yoo, E.S.; Woo, J.S.; Jung, G.H.; Jung, S.H.; Kim, B.S.; Jung, J.Y. Shikonin inhibits proliferation of melanoma cells by MAPK pathway-mediated induction of apoptosis. Biosci. Rep. 2021, 41, BSR20203834. [Google Scholar] [CrossRef] [PubMed]
- Wang, W.; Wu, Y.; Chen, S.; Liu, X.; He, J.; Wang, S.; Lu, W.; Tang, Y.; Huang, J. Shikonin is a novel and selective IMPDH2 inhibitor that target triple-negative breast cancer. Phytother. Res. 2021, 35, 463–476. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Zeng, X. Shikonin suppresses progression and epithelial-mesenchymal transition in hepatocellular carcinoma (HCC) cells by modulating miR-106b/SMAD7/TGF-β signaling pathway. Cell Biol. Int. 2020, 44, 467–476. [Google Scholar] [CrossRef] [PubMed]
- Chang, Y.H.; Lin, Y.J.; Huang, C.Y.; Harnod, T.; Ding, D.C. Shikonin impedes type 2 ovarian cancer progression via FasL/caspase-8 and mir-874-3p/XIAP axis and prohibits the properties of stemness. Am. J. Cancer Res. 2022, 12, 4584–4601. [Google Scholar]
- Boulos, J.C.; Rahama, M.; Hegazy, M.F.; Efferth, T. Shikonin derivatives for cancer prevention and therapy. Cancer Lett. 2019, 459, 248–267. [Google Scholar] [CrossRef]
- Wang, Q.; Wang, J.; Wang, J.; Ju, X.; Zhang, H. Molecular mechanism of shikonin inhibiting tumor growth and potential application in cancer treatment. Toxicol. Res. 2021, 10, 1077–1084. [Google Scholar] [CrossRef]
- Liu, T.; Xia, Y.; Li, J.; Li, S.; Feng, J.; Wu, L.; Zhang, R.; Xu, S.; Cheng, K.; Zhou, Y.; et al. Shikonin attenuates concanavalin a-induced acute liver injury in mice via inhibition of the JNK pathway. Mediat. Inflamm. 2016, 2016, 2748367. [Google Scholar] [CrossRef]
- Guo, H.; Sun, J.; Li, D.; Hu, Y.; Yu, X.; Hua, H.; Jing, X.; Chen, F.; Jia, Z.; Xu, J. Shikonin attenuates acetaminophen-induced acute liver injury via inhibition of oxidative stress and inflammation. Biomed. Pharmacother. 2019, 112, 108704. [Google Scholar] [CrossRef]
- Tian, Y.; Li, Z.; Shen, B.; Wu, L.; Han, L.; Zhang, Q.; Feng, H. The protective effects of Shikonin on lipopolysaccharide/d-galactosamine-induced acute liver injury via inhibiting MAPK and NF-κB and activating Nrf2/HO-1 signaling pathways. RSC Adv. 2017, 7, 34846–34856. [Google Scholar] [CrossRef]
- Lin, M.X.; Yi, Y.X.; Fang, P.P.; Huang, S.S.; Pan, C.W.; Jin, L.X.; Zhang, T.; Zhou, G.Y. Shikonin protects against d-galactosamine and lipopolysaccharide-induced acute hepatic injury by inhibiting TLR4 signaling pathway. Oncotarget 2017, 8, 91542–91550. [Google Scholar] [CrossRef][Green Version]
- Li, H.; Chen, Y.; Zhang, J.; Chen, X.; Li, Z.; Liu, B.; Zhang, L. Shikonin Attenuates Acetaminophen-Induced Hepatotoxicity by Upregulation of Nrf2 through Akt/GSK3β Signaling. Molecules 2018, 24, 110. [Google Scholar] [CrossRef] [PubMed]
- Song, M.; Zhang, H.; Chen, Z.; Yang, J.; Li, J.; Shao, S.; Liu, J. Shikonin reduces hepatic fibrosis by inducing apoptosis and inhibiting autophagy via the platelet-activating factor-mitogen-activated protein kinase axis. Exp. Ther. Med. 2021, 21, 28. [Google Scholar] [CrossRef] [PubMed]
- Liu, T.; Xu, L.; Wang, C.; Chen, K.; Xia, Y.; Li, J.; Li, S.; Wu, L.; Feng, J.; Xu, S.; et al. Alleviation of hepatic fibrosis and autophagy via inhibition of transforming growth factor-β1/Smads pathway through shikonin. J. Gastroenterol. Hepatol. 2019, 34, 263–276. [Google Scholar] [CrossRef] [PubMed]
- Nikita, G.; Vivek, P.; Chhaya, G. Wound-healing activity of an oligomer of alkannin/shikonin, isolated from root bark of onosma echioides. Nat. Prod. Res. 2015, 29, 1584–1588. [Google Scholar] [CrossRef] [PubMed]
- Karayannopoulou, M.; Tsioli, V.; Loukopoulos, P.; Anagnostou, T.L.; Giannakas, N.; Savvas, I.; Papazoglou, L.G.; Kaldrymidou, E. Evaluation of the effectiveness of an ointment based on alkannins/shikonins on second intention wound healing in the dog. Can. J. Vet. Res. Rev. Can. Rech. Vet. 2011, 75, 42–48. [Google Scholar]
- Yang, X.; Fan, W.; Huang, R.; Liu, G. β-acetoxyisovaleryl alkannin (AAN-II) from Alkanna tinctoria promotes the healing of pressure-induced venous ulcers in a rabbit model through the activation of TGF-β/Smad3 signaling. Cell. Mol. Biol. Lett. 2021, 26, 35. [Google Scholar] [CrossRef]
- He, Y.; Luo, K.; Hu, X.; Liu, J.; Hao, M.; Li, Y.; Xia, X.; Lu, X.; Shi, C. Antibacterial mechanism of shikonin against vibrio vulnificus and its healing potential on infected mice with full-thickness excised skin. Foodborne Pathog. Dis. 2023, 20, 67–79. [Google Scholar] [CrossRef]
- Mani, H.; Sidhu, G.S.; Singh, A.K.; Gaddipati, J.; Banaudha, K.K.; Raj, K.; Maheshwari, R.K. Enhancement of wound healing by shikonin analogue 93/637 in normal and impaired healing. Skin Pharmacol. Physiol. 2004, 17, 49–56. [Google Scholar] [CrossRef]
- Imai, K.; Kato, H.; Taguchi, Y.; Umeda, M. Biological Effects of Shikonin in Human Gingival Fibroblasts via ERK 1/2 Signaling Pathway. Molecules 2019, 24, 3542. [Google Scholar] [CrossRef]
- Shu, G.; Xu, D.; Zhang, W.; Zhao, X.; Li, H.; Xu, F.; Yin, L.; Peng, X.; Fu, H.; Chang, L.J.; et al. Preparation of shikonin liposome and evaluation of its in vitro antibacterial and in vivo infected wound healing activity. Phytomedicine 2022, 99, 154035. [Google Scholar] [CrossRef]
- Guo, L.; Li, Y.; Li, W.; Qiu, J.; Du, J.; Wang, L.; Zhang, T. Shikonin ameliorates oxidative stress and neuroinflammation via the Akt/ERK/JNK/NF-κB signalling pathways in a model of Parkinson’s disease. Clin. Exp. Pharmacol. Physiol. 2022, 49, 1221–1231. [Google Scholar] [CrossRef] [PubMed]
- Oh, J.S.; Lee, S.J.; Choung, S.Y. Lithospermum erythrorhizon Alleviates Atopic Dermatitis-like Skin Lesions by Restoring Immune Balance and Skin Barrier Function in 2.4-Dintrochlorobenzene-Induced NC/Nga Mice. Nutrients 2021, 13, 3209. [Google Scholar] [CrossRef]
- Choi, J.H.; Song, Y.S.; Lee, H.J.; Kim, G.C.; Hong, J.W. The topical application of low-temperature argon plasma enhances the anti-inflammatory effect of Jaun-ointment on DNCB-induced NC/Nga mice. BMC Complement. Altern. Med. 2017, 17, 340. [Google Scholar] [CrossRef]
- Ku, J.M.; Hong, S.H.; Kim, S.R.; Choi, H.S.; Kim, H.I.; Kim, D.U.; Oh, S.M.; Seo, H.S.; Kim, T.Y.; Shin, Y.C.; et al. The prevention of 2,4-dinitrochlorobenzene-induced inflammation in atopic dermatitis-like skin lesions in BALB/c mice by Jawoongo. BMC Complement Altern. Med. 2018, 18, 215. [Google Scholar] [CrossRef] [PubMed]
- Yen, C.; Chiang, W.; Liu, S.; Yu, S.; Hsieh, C. Shikonin inhibits der p2-induced cytokine and chemokine expression in dendritic cells in patients with atopic dermatitis. Evid. Based Complement Altern. Med. 2020, 2020, 9506363. [Google Scholar] [CrossRef]
- Yan, Y.; Furumura, M.; Gouya, T.; Iwanaga, A.; Teye, K.; Numata, S.; Karashima, T.; Li, X.G.; Hashimoto, T. Shikonin promotes skin cell proliferation and inhibits nuclear factor-kappab translocation via proteasome inhibition in vitro. Chin. Med. J. 2015, 128, 2228–2233. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Hayashi, S.; Umezaki, M.; Yamamoto, T.; Kageyama-Yahara, N.; Kondo, T.; Kadowaki, M. Shikonin, a constituent of Lithospermum erythrorhizon exhibits anti-allergic effects by suppressing orphan nuclear receptor Nr4a family gene expression as a new prototype of calcineurin inhibitors in mast cells. Chem. Biol. Interact. 2014, 224, 117–127. [Google Scholar] [CrossRef]
- Deng, S.; May, B.H.; Zhang, A.L.; Lu, C.; Xue, C.C. Topical herbal formulae in the management of psoriasis: Systematic review with meta-analysis of clinical studies and investigation of the pharmacological actions of the main herbs. Phytother. Res. 2014, 28, 480–497. [Google Scholar] [CrossRef]
- Yu, Y.J.; Xu, Y.Y.; Lan, X.O.; Liu, X.Y.; Zhang, X.L.; Gao, X.H.; Geng, L. Shikonin induces apoptosis and suppresses growth in keratinocytes via CEBP-δ upregulation. Int. Immunopharmacol. 2019, 72, 511–521. [Google Scholar] [CrossRef]
- Zhang, X.; Li, J.; Yu, Y.; Lian, P.; Gao, X.; Xu, Y.; Geng, L. Shikonin Controls the Differentiation of CD4+CD25+ Regulatory T Cells by Inhibiting AKT/mTOR Pathway. Inflammation 2019, 42, 1215–1227. [Google Scholar] [CrossRef]
- Lan, X.; Wang, H.; Qi, R.; Xu, Y.; Yu, Y.; Yang, Y.; Guo, H.; Gao, X.; Geng, L. Shikonin inhibits CEBPD downregulation in IL-17-treated HaCaT cells and in an imiquimod-induced psoriasis model. Mol. Med. Rep. 2020, 22, 2263–2272. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Liu, L.; Sun, X.Y.; Zhang, S.; Zhou, Y.Q.; Ze, K.; Chen, S.T.; Lu, Y.; Cai, X.C.; Chen, J.L.; et al. Evidence and potential mechanism of action of lithospermum erythrorhizon and its active components for psoriasis. Front. Pharmacol. 2022, 13, 781850. [Google Scholar] [CrossRef] [PubMed]
- Tao, T.; Chen, Y.; Lai, B.; Wang, J.; Wang, W.; Xiao, W.; Cha, X. Shikonin combined with methotrexate regulate macrophage polarization to treat psoriasis. Bioengineered 2022, 13, 11146–11155. [Google Scholar] [CrossRef] [PubMed]
- Li, W.; Zhang, C.; Ren, A.; Li, T.; Jin, R.; Li, G.; Gu, X.; Shi, R.; Zhao, Y. Shikonin suppresses skin carcinogenesis via inhibiting cell proliferation. PLoS ONE 2015, 10, e126459. [Google Scholar] [CrossRef] [PubMed]
- Kretschmer, N.; Hufner, A.; Durchschein, C.; Popodi, K.; Rinner, B.; Lohberger, B.; Bauer, R. Synthesis and pharmacological in vitro investigations of novel shikonin derivatives with a special focus on cyclopropane bearing derivatives. Int. J. Mol. Sci. 2021, 22, 2774. [Google Scholar] [CrossRef]
- Cao, H.; Liu, D.; Lai, Y.; Chen, Y.; Yu, L.; Shao, M.; Liu, J. Inhibition of the STAT3 Signaling Pathway Contributes to the Anti-Melanoma Activities of Shikonin. Front. Pharmacol. 2020, 11, 748. [Google Scholar] [CrossRef]
- Cui, J.; Zhou, X.; Huang, J.; Cui, J.; Chen, J. Selective Antitumor Effect of Shikonin Derived DMAKO-20 on Melanoma through CYP1B1. Curr. Cancer Drug Targets 2020, 21, 223–231. [Google Scholar] [CrossRef]
- Fan, C.; Xie, Y.; Dong, Y.; Su, Y.; Upton, Z. Investigating the potential of shikonin as a novel hypertrophic scar treatment. J. Biomed. Sci. 2015, 22, 70. [Google Scholar] [CrossRef]
- Zhou, R.; Wang, C.; Lv, D.; Sun, Y.; Liang, Y. TNF-α inhibits fibrosis and migration of fibroblasts in hypertrophic scar by miR-141-3p. Acta Biochim. Biophys. Sin. 2021, 53, 1106–1108. [Google Scholar] [CrossRef]
- Fan, C.; Lim, L.; Loh, S.Q.; Ying, L.K.; Upton, Z.; Leavesley, D. Application of “macromolecular crowding” in vitro to investigate the naphthoquinones shikonin, naphthazarin and related analogues for the treatment of dermal scars. Chem.-Biol. Interact. 2019, 310, 108747. [Google Scholar] [CrossRef]
- Deng, X.; Chen, Q.; Qiang, L.; Chi, M.; Xie, N.; Wu, Y.; Yao, M.; Zhao, D.; Ma, J.; Zhang, N.; et al. Development of a porcine full-thickness burn hypertrophic scar model and investigation of the effects of shikonin on hypertrophic scar remediation. Front. Pharmacol. 2018, 9, 590. [Google Scholar] [CrossRef] [PubMed]
- Lu, Y.; Yue, Z.; Xie, J.; Wang, W.; Zhu, H.; Zhang, E.; Cao, Z. Micelles with ultralow critical micelle concentration as carriers for drug delivery. Nat. Biomed. Eng. 2018, 2, 318–325. [Google Scholar] [CrossRef] [PubMed]
- Kesharwani, S.S.; Jain, V.; Dey, S.; Sharma, S.; Mallya, P.; Kumar, V.A. An overview of advanced formulation and nanotechnology-based approaches for solubility and bioavailability enhancement of silymarin. J. Drug Deliv. Sci. Technol. 2020, 60, 102021. [Google Scholar] [CrossRef]
- Yan, C.; Li, Q.; Sun, Q.; Yang, L.; Liu, X.; Zhao, Y.; Shi, M.; Li, X.; Luo, K. Promising nanomedicines of shikonin for cancer therapy. Int. J. Nanomed. 2023, 18, 1195–1218. [Google Scholar] [CrossRef] [PubMed]
- Ding, Q.; Chen, K.; Liu, X.; Ding, C.; Zhao, Y.; Sun, S.; Zhang, Y.; Zhang, J.; Liu, S.; Liu, W. Modification of taxifolin particles with an enteric coating material promotes repair of acute liver injury in mice through modulation of inflammation and autophagy signaling pathway. Biomed. Pharmacother. 2022, 152, 113242. [Google Scholar] [CrossRef]
- Wen, X.; Li, J.; Cai, D.; Yue, L.; Wang, Q.; Zhou, L.; Fan, L.; Sun, J.; Wu, Y. Anticancer Efficacy of Targeted Shikonin Liposomes Modified with RGD in Breast Cancer Cells. Molecules 2018, 23, 268. [Google Scholar] [CrossRef] [PubMed]
- He, J.; Xie, Y.; Zhong, J.; Chen, W.; Fang, S.; Chen, X.; Peng, S.; Liu, W.; Liu, C. Improving shikonin solubility and stability by encapsulation in natural surfactant-coated shikonin nanoparticles. J. Food Sci. 2023, 88, 825–836. [Google Scholar] [CrossRef] [PubMed]
- Chen, T.; Tu, L.; Wang, G.; Qi, N.; Wu, W.; Zhang, W.; Feng, J. Multi-functional chitosan polymeric micelles as oral paclitaxel delivery systems for enhanced bioavailability and anti-tumor efficacy. Int. J. Pharm. 2020, 578, 119105. [Google Scholar] [CrossRef]
- Li, G.; Shang, C.; Li, Q.; Chen, L.; Yue, Z.; Ren, L.; Yang, J.; Zhang, J.; Wang, W. Combined Shikonin-Loaded MPEG-PCL Micelles Inhibits Effective Transition of Endothelial-to-Mesenchymal Cells. Int. J. Nanomed. 2022, 17, 4497–4508. [Google Scholar] [CrossRef]


| Key Numbers | Tumor Type | Mechanism of Action | References |
|---|---|---|---|
| 1 | Lung cancer | Inhibits the viability, proliferation, invasion, and migration of non-small cell lung cancer A549 and PC9 cells. | [37] |
| 2 | Pancreatic cancer | Shikonin inhibits the activation of PAK1 and its downstream signaling pathway proteins. | [38] |
| 3 | Chondrosarcoma | It can inhibit pSTAT3 and increase pAKT, MAPKs, pERK, pJNK, and p-p38 MAPK. | [39] |
| 4 | Glioma | Tumor apoptosis mediated by interference with endoplasmic reticulum (ER) stress; induction of mitochondrial outer membrane permeability (MOMP) triggers apoptosis of cancer cells. | [40] |
| 5 | Melanoma | Apoptosis was induced by the MAPK pathway. | [41] |
| 6 | Triple-negative breast cancer | The growth of human triple-negative breast cancer cell line MDA-MB-231 and mouse triple-negative breast cancer cell line 4T1 was inhibited. | [42] |
| 7 | Liver cancer | The miR-106b/SMAD7/TGF-β signaling pathway inhibits cell progression and EMT and accelerates cell death. | [43] |
| 8 | Ovarian cancer | Shikonin inhibited cell viability, migration, and invasion of type 2 ovarian cancer cells. The expression of CSC-related markers and the number of cocci colonies were reduced. The tumorigenicity of Kuramochi cells was also reduced. | [44] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Song, Y.; Ding, Q.; Hao, Y.; Cui, B.; Ding, C.; Gao, F. Pharmacological Effects of Shikonin and Its Potential in Skin Repair: A Review. Molecules 2023, 28, 7950. https://doi.org/10.3390/molecules28247950
Song Y, Ding Q, Hao Y, Cui B, Ding C, Gao F. Pharmacological Effects of Shikonin and Its Potential in Skin Repair: A Review. Molecules. 2023; 28(24):7950. https://doi.org/10.3390/molecules28247950
Chicago/Turabian StyleSong, Yanping, Qiteng Ding, Yuewen Hao, Bing Cui, Chuanbo Ding, and Feng Gao. 2023. "Pharmacological Effects of Shikonin and Its Potential in Skin Repair: A Review" Molecules 28, no. 24: 7950. https://doi.org/10.3390/molecules28247950
APA StyleSong, Y., Ding, Q., Hao, Y., Cui, B., Ding, C., & Gao, F. (2023). Pharmacological Effects of Shikonin and Its Potential in Skin Repair: A Review. Molecules, 28(24), 7950. https://doi.org/10.3390/molecules28247950








