Abstract
Pulse beetle is the most harmful pest attacking stored grains and affecting quality and marketability. Continuous use of chemical-based pesticides against pulse beetle led to the development of insecticidal resistance; essential oils (EOs) can be an effective natural alternative against this pest. The main objective was to study the chemical composition of seven EOs viz., Acorus calamus, Hedychium spicatum, Lavandula angustifolia, Juniperus recurva, Juniperus communis, Cedrus deodara and Pinus wallichiana, their insecticidal and enzyme inhibition activities against pulse beetle. The primary compounds present in these EOs were cis-asarone, 1,8-cineole, linalyl isobutyrate, 2-β-pinene, camphene, α-dehydro-ar-himachalene and camphene. A. calamus oil showed promising fumigant toxicity to Callosobruchus maculatus and C. chinensis (LC50 = 1357.86 and 1379.54 µL/L, respectively). A combination of A. calamus + L. angustifolia was effective against C. maculatus and C. chinensis (LC50 = 108.58 and 92.18 µL/L, respectively). All the combinations of EOs showed synergistic activity. In the repellency study, A. calamus showed more repellence to C. maculatus and C. chinensis (RC50 = 53.98 and 118.91 µL/L, respectively). A. calamus and L. angustifolia oil at 2500, 5000 and 10,000 µL/L significantly inhibited the AChE and GST enzymes in C. maculatus and C. chinensis after 24 and 48 h.
1. Introduction
The global population is continuously rising which needs a huge amount of food to feed people, so the production of food grains creates a critical source of food for growing population. Among these, pulse crops are sustainable and high-quality protein sources [1]. However, increasing pest infestation causes stored grain loss, affecting food security worldwide [2]. During storage, pulse beetle can cause a 5–10% loss in temperate and a 20–30% loss in tropical regions [3]. If no control measures are employed, there will be a 50% loss of grains in store within 3-4 months [4]. At the global level, the pulses (green/black gram) are considered as major host of pulse beetle [5] and these pulses are rich source of proteins, carbohydrates, vitamins etc. [6]. Callosobruchus maculatus and Callosobruchus chinensis are the major pest of cowpea and French beans in Africa and Asia which contains rich source of proteins (20-25%) and carbohydrates (50-60%) [7,8]. The grubs of the C. maculatus and C. chinensis feed on grains affecting quantitative and qualitative damage to the stored grains [9]. Usually, fumigation with phosphine/aluminium phosphide and chemical pesticides are more effective for controlling pulse beetle where the grains are stored in small bins and big go downs. However, fumigation causes some minor side effects on consumer’s health [10]. Also, the indiscriminate use of synthetic insecticides for the control of stored grain pests has led to insect resistance, posed harm to non-target organisms and atmosphere [11].
Several studies reported that compounds from plants substitute for synthetic insecticides and have observed that essential oils (EOs) are a better replacement for controlling stored grain pests [12,13]. Contrary to synthetic insecticides, EOs reported higher toxicity/efficacy, safer use, and more biodegradability. The higher diversity of the composition of EOs reduces the resistance development in stored pests [14,15]. Several studies have reported the insecticidal activities of botanical products of many plant species to stored grain pests [16] and these plants are predominantly used as crude/solvent extracts, powders, slurries, and volatile/crude oils [17,18]. Plants comprise of many bioactive compounds such as phenolics, flavonoids, alkaloids, tannins, sterols and EOs [19]. Plant-derived botanicals such as extracts/fractions and EOs from non-host plants with toxicant, ovipositional and repellent properties may be a better alternative for chemical pesticides to control pests [20].
The monoterpenoids and sesquiterpenoids in the EOs possess insecticidal activities against stored grain pests [21]. In earlier studies there are few reports on the insecticidal activities of Acorus calamus but not much literature is available for the other targeted EOs. Similarly, the mechanism of action of EOs through enzyme (Glutathione-S-Transferase and Acetylcholine esterase) inhibition activities in pulse beetle has not been reported earlier. Based on this background, the EOs of A. calamus, Hedychium spicatum, Lavandula angustifolia, Juniperus recurva, Juniperus communis, Pinus wallichiana and Cedrus deodara were studied for their chemical composition, fumigant, synergistic, repellent, ovipositional, and enzyme inhibition activities against pulse beetle.
2. Results
2.1. Chemical Composition of Essential Oils
The chemical composition of EOs was analyzed via GC-MS, and the chemical composition of A. calamus was represented in Table 1. Further, the chemical composition of remaining EOs viz H. spicatum, J. recurva, J. communis, L. angustifolia, P. wallichiana, and C. deodara were presented in Tables S1–S6. Among different EOs, monoterpene hydrocarbons were found highest in J. communis (93.75%), followed by P. wallichiana (88.34%) and J. recurva (54.63%) as compared to others. In contrast, oxygenated monoterpene was found to be highest in L. angustifolia (75.36%), followed by P. wallichiana (1.99%) and H. spicatum (0.50%). Sesquiterpene hydrocarbon was found to be highest in C. deodara (76.55%), followed by H. spicatum (24.75%) and J. recurva (21.67%) as compared to others. However, oxygenated sesquiterpene was found to be highest in H. spicatum (3.72%), followed by C. deodara (12.9%) and A. calamus (3.93%) as compared to others. Different constituents were identified in different EOs. Among them, the major components present in the EOs of A. calamus, H. spicatum, J. recurva, J. communis, L. angustifolia, P. wallichiana, and C. deodara are cis-asarone (85.37%), 1,8-cineole (28.31%), 2-β-pinene (39.18%), camphene (61.03%), linalyl isobutyrate (40.16%), camphene (46.21%) and α-dehydro-ar-himachalene (44.81%), respectively as compared to other constituents.

Table 1.
Chemical composition of essential oil of A. calamus.
2.2. Fumigant Toxicity of Essential Oils against C. maculatus and C. chinensis
Fumigant toxicity of EOs viz., A. calamus, H. spicatum, J. recurva, J. communis, L. angustifolia, P. wallichiana, and C. deodara were screened against adults of pulse beetle.
2.2.1. C. maculatus
Toxicity of Different Essential Oils against C. maculatus
The fumigant toxicity of different individual essential oils (EOs) and its combinations against C. maculatus was presented in Table 2. Among EOs, C. deodara showed promising toxicity against C. maculatus (LC50 = 4116.25 µL/L) after 72 h, followed by A. calamus and L. angustifolia (LC50 = 4128.22 and 5204.72 µL/L, respectively) as compared to other EOs. Similarly, A. calamus was more effective (LC50 = 1357.86 µL/L) after 96 h of treatment, followed by L. angustifolia and H. spicatum (LC50 = 1876.15 and 2177.08 µL/L, respectively) as compared to other EOs (LC50 = 2818.88–6684.97 µL/L). C. maculatus was most susceptible to A. calamus after 120 h of treatment (LC50 = 701.48 µL/L) and was followed by H. spicatum and L. angustifolia (LC50 = 806.92 and 1220.93 µL/L, respectively). However, J. recurva was the least effective after 96 and 120 h of treatment (LC50 = 6684.97 and 2369.76 µL/L, respectively). All the tested EOs were superior to the positive control, i.e., aluminium phosphide (LC50 = 0.06 µg/mL) after 72 h.

Table 2.
Toxicity of different essential oils and their combinations against C. maculatus.
The experimental results on a combination of different EOs viz., A. calamus + L. angustifolia, A. calamus + H. spicatum, A. calamus + C. deodara, A. calamus + P. wallichiana, A. calamus + J. communis, L. angustifolia + H. spicatum, L. angustifolia + C. deodara, L. angustifolia + P. wallichiana and L. angustifolia + J. communis at a 1:1 ratio against C. maculatus for their toxicity (LC50 values) and synergistic activity after 24, 48, 72 and 96 h of treatment was also presented. The EOs combinations showed toxicity against C. maculatus after 24 h and continued up to 96 h after treatment. Among them, A. calamus + L. angustifolia showed promising toxicity (LC50 = 1148.59, 533.72, and 204.01 µL/L, respectively) against C. maculatus after 24, 48 and 72 h, followed by L. angustifolia + P. wallichiana (LC50 = 1322.93, 623.70 and 312.23 µL/L) and L. angustifolia + J. communis (LC50 = 1376.68, 664.26 and 372.88 µL/L, respectively). Further, A. calamus + L. angustifolia showed more toxicity (LC50 = 108.58 µL/L) after 96 h, followed by A. calamus + H. spicatum and A. calamus + P. wallichiana (LC50 = 164.31 and 169.89 µL/L, respectively) as compared to other combinations. All the combinations of EOs showed synergistic activity against C. maculatus.
Percent Mortality of Essential Oils against C. maculatus
The fumigant toxicity of EOs, concentrations, and interaction effect (oils × concentrations) against C. maculatus after 72, 96 and 120 h were presented in Figure 1. The pooled mean percent mortality was found to be significantly different across the oils (F6,174 = 32.71, 46.13 and 32.90; p < 0.0001), concentrations (F4,174 = 181.12, 218.23 and 302.09; p < 0.0001) and interaction (oils × concentrations) (F24,174 = 2.76, 2.19 and 3.59; p < 0.0001) after 72, 96 and 120 h of treatment. Among oils, pooled mean percent mortality was superior in A. calamus (41.60 ± 19.93) after 72 h and was at par with C. deodara (38.80 ± 24.89) and L. angustifolia (34.80 ± 24.17), followed by J. communis (25.60 ± 14.46) as compared to other EOs. Similarly, after 96 h, pooled mean mortality was maximum in A. calamus (61.20 ± 21.08), followed by L. angustifolia (56.00 ± 24.49) compared to others. Further, after 120 h, pooled mean mortality was higher in A. calamus (75.20 ± 19.39) and was at par with H. spicatum (72.40 ± 20.67), followed by L. angustifolia (68.00 ± 27.23) as compared to other EOs. Among concentrations, pooled mean mortality was more at 10,000 µL/L (56.28 ± 17.16, 76.86 ± 15.,49and 94.28 ± 8.84, respectively) after 72, 96 and 120 h, as compared to other concentrations.
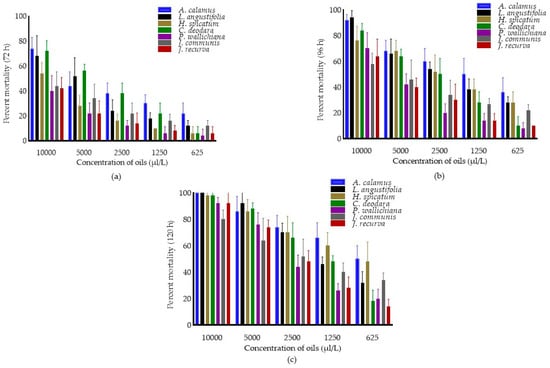
Figure 1.
Percent mortality of essential oils in C. maculatus after 72 h (a) 96 h (b) and 120 h (c). Bars represent the standard deviation (±SD) of five replications. Means ± SD in the error bars differs significantly by Tukey’s HSD (p < 0.0001).
2.2.2. C. chinensis
Toxicity of Different Essential Oils against C. chinensis
The fumigant toxicity of different EOs and their combinations against C. chinensis was presented in Table 3. Among EOs, L. angustifolia showed more toxicity against C. chinensis (LC50 = 4316.34 µL/L) after 72 h of treatment, followed by C. deodara and J. communis (LC50 = 4797.04 and 9895.41 µL/L, respectively) as compared to other oils (LC50 = 10,448.32–10,975.11 µL/L). Similarly, A. calamus (LC50 = 1379.54 µL/L) was more effective after 96 h and was followed by L. angustifolia and H. spicatum (LC50 = 1715.57 and 2598.47 µL/L, respectively) as compared to other oils (LC50 = 4875.82–7918.09 µL/L). However, after 120 h, C. chinensis was more susceptible to L. angustifolia (LC50 = 779.59 µL/L), followed by A. calamus and H. spicatum (LC50 = 1158.42 and 1586.15 µL/L, respectively) as compared to other oils (LC50 = 1694.87–2312.36 µL/L). P. wallichiana and J. communis were the least effective after 96 and 120 h (LC50 = 6684.97 and 2312.36 µL/L, respectively).

Table 3.
Toxicity of different essential oils and their combinations against C. chinensis.
The experimental results on a combination of different EOs viz., A. calamus + L. angustifolia, A. calamus + C. deodara, A. calamus + H. spicatum, A. calamus + J. communis, A. calamus + J. recurva, L. angustifolia + C. deodara, L. angustifolia + H. spicatum, L. angustifolia + J. communis and L. angustifolia + J. recurva at a 1:1 ratio against C. chinensis for their toxicity (LC50 values) and synergistic activity after 24, 48, 72 and 96 h of treatment was also presented. Among combinations, A. calamus + L. angustifolia showed more toxicity after 24, 48, 72 and 96 h after treatment (LC50 = 396.54, 201.22, 141.89 and 92.18 µL/L, respectively) and was followed by A. calamus + C. deodara (LC50 = 509.92, 258.76, 182.46 and 118.54 µL/L, respectively) and L. angustifolia + C. deodara (LC50 = 740.11, 432.83, 279.06 and 182.66 µL/L, respectively) as compared to other combinations. All the combinations of EOs showed synergistic activity against C. chinensis.
Percent Mortality of Different EOs against C. chinensis
The fumigant toxicity of EOs, concentrations, and interaction effect (oils × concentrations) against C. chinensis after 72, 96 and 120 h were presented in Figure 2. The pooled mean percent mortality was found to be significantly different across the oils (F6,174 = 34.31, 51.43 and 23.72; p < 0.0001), concentrations (F4,174 = 215.79, 222.99 and 258.57; p < 0.0001) after 72, 96 and 120 h of treatment.
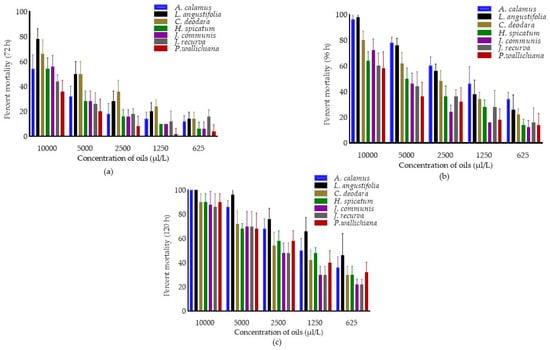
Figure 2.
Percent mortality in C. chinensis after 72 h (a) 96 h (b) and 120 h (c). Bars represent the standard deviation (±SD) of five replications. Means ± SD in the error bars differs significantly by Tukey’s HSD (p < 0.0001).
The interaction effect (oils × concentrations) showed significantly different against C. chinensis after 72 (F24,174 = 1.96; p < 0.0001) and 96 h (F24,174 = 1.89; p < 0.05). Results showed that among oils, pooled mean mortality was higher in L. angustifolia (38.00 ± 25.00) after 72 h of treatment and was at par with C. deodara as compared to other oils. Similarly, pooled mean mortality was maximum in A. calamus (62.80 ± 23.72) after 96 h and was at par with L. angustifolia (58.80 ± 27.43) followed by C. deodara (49.20 ± 21.78) as compared to EOs. After 120 h, mortality was higher in L. angustifolia (76.80 ± 22.49), followed by A. calamus (68.00 ± 24.66) compared to the remaining oils. Among concentrations, pooled mean mortality was higher in 10,000 µL/L (55.43 ± 15.40, 75.43 ± 17.71 and 92.00 ± 8.33, respectively) after 72, 96 and 120 h as compared to other concentrations.
2.3. Repellent Activity of Essential Oils against Pulse Beetle
The repellent activity of A. calamus, H. spicatum, J. recurva, J. communis, L. angustifolia, P. wallichiana and C. deodara against two species of pulse beetle after 1, 2, 3, 4, 5 and 24 h was presented in Table 4 and Table 5. The repellent activity of EOs against pulse beetle decreased over time. Results showed that A. calamus showed more repellence against C. maculatus and C. chinensis (RC50 = 53.98 and 118.91 µL/L, respectively) after 24 h and was followed by H. spicatum (RC50 = 293.77 and 226.85 µL/L, respectively) and J. recurva (RC50 = 309.75 and 383.51 µL/L, respectively) as compared to other EOs in C. maculatus (RC50 = 955.15–736.82 µL/L) and C. chinensis (617.11–947.27 µL/L).

Table 4.
Repellent activity of different essential oils against C. maculatus.

Table 5.
Repellent activity of different essential oils against C. chinensis.
2.4. Ovipositional Inhibition (OI) Effect of Essential Oils against Pulse Beetle
The ovipositional inhibition (OI) of different EOs and concentrations against pulse beetle reflected in the percent OI after 24, 48 and 72 h were summarized from Tables S7–S12.
2.4.1. C. maculatus
The percent OI of different EOs and concentrations against C. maculatus and its interaction effect (oils × concentrations) after 24, 48 and 72 h of treatment were presented in Tables S7–S9. The pooled mean percent OI was significantly different across the oils (F6,174 = 9.38, 3.77 and 8.35; p < 0.0001), concentrations (F4,174 = 27.30, 17.78 and 30.58; p < 0.0001) after 24, 48 and 72 h of treatment and whereas for interaction (oils × concentrations) (F24,174 = 2.54 and 2.20; p < 0.0001) after 24 and 72 h of treatment was found significantly different, but after 48 h, the interaction was not differed significantly (p > 0.05). Among the EOs, the pooled mean percent OI was more in J. communis (33.60 ± 13.98) after 24 h, followed by H. spicatum and J. recurva (29.72 ± 11.73 and 26.28 ± 13.40, respectively) as compared to other EOs. After 48 h, pooled mean OI was maximum in J. communis (32.88 ± 17.71) and was at par with C. deodara (32.36 ± 6.80), followed by H. spicatum (29.40 ± 8.59) as compared to others. However, after 72 h, pooled mean percent OI was higher in C. deodara (36.08 ± 5.58), followed by J. recurva and H. spicatum (28.76 ± 10.56 and 28.32 ± 8.09, respectively) as compared to other remaining oils. Among concentrations, pooled mean percent OI was higher in 10,000 µL/L (36.83 ± 12.69, 37.11 ± 7.91 and 36.57 ± 7.58, respectively) after 24, 48 and 72 h as compared to other concentrations.
2.4.2. C. chinensis
The percent OI of different EOs and concentrations against C. chinensis and its interaction effect (oils × concentrations) after 24, 48 and 72 h were presented in Tables S10–S12. The pooled mean percent OI was found to be significantly different across the oils (F6,174 = 7.45 and 3.77; p < 0.0001) after 24 and 72 h, concentrations (F4,174 = 25.18, 20.16 and 36.46; p < 0.0001) after 24, 48 and 72 h of treatment. However, the mean percent OI was not differed significantly (p > 0.05) in the interaction (oils × concentrations). Among oils, the pooled mean OI was highest in P. wallichiana (34.84 ± 7.28) after 24 h, followed by J. communis and C. deodara (28.72 ± 12.05 and 27.96 ± 10.31%, respectively) as compared to other EOs. Similarly, after 48 h, pooled mean percent OI was maximum in P. wallichiana (32.04 ± 9.52) and was followed by A. calamus (27.56 ± 8.98) as compared to other EOs. In the case of 72 h, pooled mean percent OI was higher in P. wallichiana (32.64 ± 7.58) and was at par with H. spicatum (31.04 ± 7.44) followed by A. calamus (29.20 ± 10.61) as compared to remaining oils. Among concentrations, pooled mean percent OI was higher in 10,000 µL/L (36.03 ± 11.03, 34.05 ± 9.02 and 37.28 ± 6.45%, respectively) after 24, 48 and 72 h as compared to other concentrations.
2.5. Detoxification Enzyme Activities of A. Calamus and L. Angustifolia against Pulse Beetle
Detoxification enzyme inhibition activities of A. calamus and L. angustifolia against C. maculatus and C. chinensis after 24 and 48 h of treatment were presented in Figure 3a–d and Figure 4a–d. Data showed that all the concentrations of A. calamus inhibited the AChE activity in C. chinensis after 24 and 48 h (F4,14 = 52.42 and 50.13; p < 0.0001, respectively) and C. maculatus (F4,14 = 142.06 and 116.74; p < 0.0001, respectively), as compared to the control.
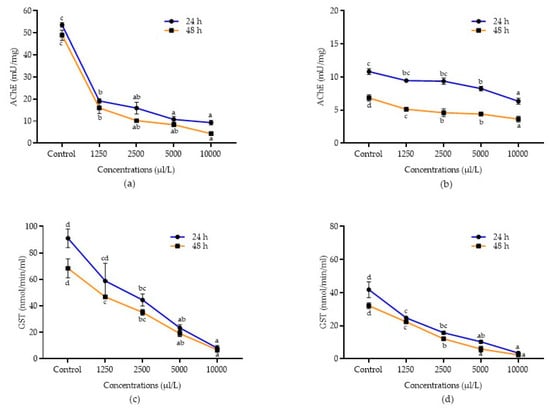
Figure 3.
Detoxification enzyme inhibition activities of essential oils. AChE inhibition in C. maculatus treated with A. calamus (a) and L. angustifolia (b). GST inhibition in C. maculatus treated with A. calamus (c) and L. angustifolia (d). Mean (±SE) of three replications. Figures in the same letters do not differ significantly by Tukey’s HSD (p ≥ 0.05).
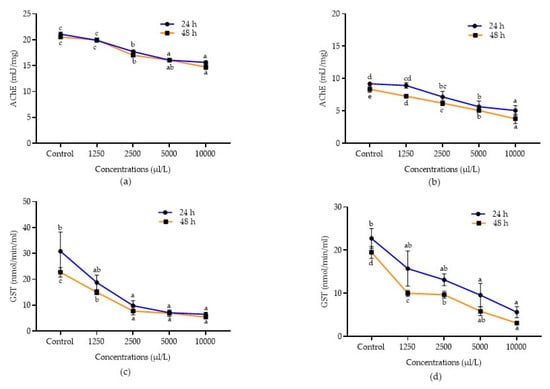
Figure 4.
Detoxification enzyme inhibition activities of essential oils. AChE inhibition in C. chinensis treated with A. calamus (a) and L. angustifolia (b). GST inhibition in C. chinensis treated with A. calamus (c) and L. angustifolia (d). Mean (±SE) of three replications. Figures in the same letters do not differ significantly by Tukey’s HSD (p ≥ 0.05).
Similarly, all the concentrations of L. angustifolia also inhibited the AChE activity after 24 and 48 h in both C. chinensis (F4,14 = 34.15 and 67.65; p < 0.0001, respectively) and C. maculatus (F4,14 = 25.88 and 160.50; p < 0.0001, respectively), as compared to the control. Among the different concentrations evaluated, A. calamus at 10,000 µL/L reported higher inhibition of AChE in both C. chinensis and C. maculatus (15.64 ± 0.28 and 9.31 ± 0.99 mU/mg, respectively) and was at par with 5000 µL/L (16.07 ± 0.29 and 10.77 ± 1.30 mU/mg, respectively) after 24 h followed by other lower concentrations (1250 – 2500 µL/L). Similarly, after 48 h, A. calamus at 10,000 µL/L reported higher inhibition of AChE in both C. chinensis and C. maculatus (14.73 ± 0.48 and 4.31 ± 0.86 mU/mg, respectively), followed by 5000 µL/L (16.07 ± 0.29 and 8.38 ± 0.79 mU/mg, respectively) as compared to other lower concentrations (1250–2500 µL/L). L. angustifolia at 10,000 µL/L also showed higher inhibition of AChE after 24 and 48 h in C. chinensis (5.07 ± 0.76 and 3.79 ± 0.73 mU/mg, respectively) and C. maculatus (6.34 ± 0.13 and 3.64 ± 0.43 mU/mg, respectively), followed by 5000 µL/L in C. chinensis (5.65 ± 0.88 and 5.08 ± 0.10 mU/mg, respectively) and C. maculatus (8.25 ± 0.34 and 4.40 ± 0.15 mU/mg, respectively) as compared to other lower concentrations.
Similarly, in C. maculatus, all the concentrations of A. calamus (F4,14 = 20.22; p < 0.0001 and F4,14 = 42.35; p < 0.0001) and L. angustifolia (F4,14 = 42.09; p < 0.0001 and F4,14 = 114.40; p < 0.0001) significantly inhibited the GST activity after 24 and 48 h, respectively. Among concentrations, A. calamus at 10,000 µL/L exhibited higher inhibition (8.10 ± 2.04 and 6.84 ± 1.90 nmol/min/mL, respectively) after 24 and 48 h, followed by 5000 µL/L (23.40 ± 2.36 and 19.08 ± 2.34 nmol/min/mL, respectively), as compared to the remaining concentrations (1250-2500 µL/L). However, L. angustifolia at 10,000 µL/L showed more inhibition (3.52 ± 0.78 nmol/min/mL) after 24 h and was followed by 5000 µL/L (10.27 ± 0.98 nmol/min/mL), as compared to lower concentrations (1250–2500 µL/L). Likewise, after 48 h, L. angustifolia at 10,000 µL/L showed higher inhibition (2.39 ± 0.70 nmol/min/mL) and was at par with 5000 µL/L (5.91 ± 1.71 nmol/min/mL), as compared to other concentrations.
3. Discussion
The chemical composition of selected EOs, insecticidal activities (fumigant toxicity, synergistic, repellence, ovipositional activities) and their effect on detoxification enzyme inhibition in C. chinensis and C. maculatus are discussed. EOs are volatile essences and aetheroleum found in aromatic plants as a mixture of compounds produced by secondary metabolites [23]. The EOs extracted by steam/water distillation which has a lower density than water [24,25]. The composition of EO is very complex, and the individual components have valuable applications in agriculture, cosmetics, human health, and the environment. EO has been discovered to be an effective complement to synthetic compounds used in the chemical industry [26]. EOs containing monoterpenes, sesquiterpenes, and terpenoids [27] play a significant role in pesticide activities. In the present study, the chemical composition of seven EOs viz., A. calamus, H. spicatum, J. recurva, J. communis, L. angustifolia, P. wallichiana, and C. deodara were studied and presented. In the present study, the major constituents are cis-asarone in the EO of A. calamus; 1,8-cineole in H. spicatum; α-dehydro-ar-himachalene and α-himachalene in C. deodara; camphene in J. communis and P. wallichiana; 2-β-pinene in J. recurva; linalyl isobutyrate and linalool in L. angustifolia. The constituents of EOs play a significant role in insecticidal activities against pests. In this study, the percent composition of 1,8-cineole is higher in H. spicatum than in EO of Artemisia maritima and M. piperita [9,21]. Similarly, the composition of 1,8-cineole is lesser (12.2–23.15%) in H. spicatum oil [28,29,30] than in the current results. The composition of asarone in the present study is higher than in earlier reports, which observed comparatively less (9.5 – 50.09%) [31,32]. Earlier studies showed that the composition of linalool in L. angustifolia in previous studies was also lesser (19.71, 28.06, and 31.17%) [33,34,35] than present study (33.30%). In the recent study, the composition of camphene was higher (46.21%) in P. wallichiana as compared to the previous (0.9 – 1%) studies [36,37]. The composition of α-himachalene (18.11%) in C. deodara was higher in the present study than in previous studies [38]. The variation in the chemical components of EOs may be due to geographical, seasonal, and climatic conditions, genetic/hereditary, chemotype, and nutrition of the plants [39].
Insecticidal activities of the EO depend upon the presence of significant constituents, mode of application, concentration, stage, and type of insect [40,41]. In the present study, A. calamus showed the highest fumigant toxicity against C. maculatus after 96 h, compared to the previous study (LC50 = 3043.94 µL/L) [42]. In a similar study, A. calamus showed fumigant toxicity against C. maculatus [43] and C. chinensis [44,45]. The EO of A. calamus was less effective against Trogaderma granarium [46] than in present studies. H. spicatum is the second-best oil that showed fumigant toxicity against C. maculatus (LC50 = 2177.08 and 806.92 µL/L) and C. chinensis (LC50 = 4875.82 and 1585.15 µL/L) after 96, and 120 h of treatment and these results are comparable with that of Plutella xylostella [47]. Similarly, the EOs of other species viz., H. forrestii, H. elatum, H. bousigonianum, H. flavum, H. thysiforme showed promising mortality against Stephanitis pyrioides [48] and H. gardnerianum against Artemia salina [49]. In the present findings, the EO of L. angustifolia was also found effective against C. chinensis and C. maculatus (LC50 = 1715.57–1876.15 µL/L) after 96 h. These results agree with previous studies of L. angustifolia against Ryzopertha dominica, Tribolium castaneum and L. dentata against C. maculatus [50,51,52].
The binary combinations of different EOs showed promising toxicity and synergistic activity against C. maculatus and C. chinensis. Due to the non-availability of reports against pulse beetle, other EOs were compared and discussed. In the present study, among combinations viz., A. calamus + L. angustifolia, A. calamus + H. spicatum, and A. calamus + P. wallichiana (LC50 = 108.58 to 169.89 µL/L) showed more promising toxicity against C. maculatus. Whereas A. calamus + L. angustifolia, A. calamus + C. deodara and L. angustifolia + C. deodara (LC50 = 92.18 to 182.66 µL/L) also showed toxicity against C. chinensis. Present results are confirmed with previous studies in which the EOs of Tagetes minuta + Mentha piperita and T. minuta + M. spicata oils also showed fumigant toxicity (0.87 to 2.40 µL/mL) against C. chinensis and C. maculatus after 48 h [9]. Other researchers also reported similar findings: Hymenocardia acida + Lippia adoensis and Lophira lanceolata + L. adoensis oils showed toxicity (LC50 = 2.4–1.61 g kg−1) against C. maculatus [53].
All the tested EOs in the present study showed more repellence against the pulse beetle. Among them, A. calamus showed higher repellence against C. maculatus and C. chinensis (RC50 = 53.98–118.91 µL/L) and was followed by H. spicatum (RC50 = 226.85–293.77 µL/L). The present results confirmed with EOs of eucalyptus and peppermint (RC50 = 29.5–57.1 µL/L) against Sitophilus oryzae [54] and Citrus sinensis, Rosmarinus officinalis and Pimenta racemosa against S. zeamais [55].
In the current study, EOs of C. deodara, J. recurva and A. calamus at higher concentrations showed significant ovipositional inhibition (37.8–42.6%) against both species of pulse beetle, and these findings agree with the earlier reports in which the EOs of C. tangerina, C. limonium, C. paradisi, C. aurantifolia and C. sinensis [56] and Eugenia caryophyllus and Illicium verum [57] showed promising ovipositional inhibition against C. maculatus as compared to the present results.
AChE is categorised as the enzymes that catalyse the hydrolysis of acetylcholine (ACh), a neurotransmitter converted into acetic acid and choline [58]. GST enzyme detoxifies various insecticides, including organochlorines, pyrethroids, organophosphates, and carbamates [59]. GST activity is mainly inhibited by the chemical compounds present in the EOs [60,61]. In the present study, EOs of A. calamus and L. angustifolia were significantly inhibiting the AChE and GST activity against C. maculatus and C. chinensis. The earlier reports confirmed the current results, which reported that the EO of A. maritima inhibited the GST activity in C. maculatus [21]. Similarly, EOs of A. monosperma, A. judaica, C. aurantifolia, C. viminals, C. lemon and Origanum vulgare also inhibited the AChE and adenosine triphosphatases (ATPases) activity against S. oryzae [62].
4. Materials and Methods
4.1. Essential Oils (EOs)
EOs of sweet flag (A. calamus), spiked ginger lily (H. spicatum), juniper leaf (J. recurva), juniper berry (J. communis), lavender (L. angustifolia), pine needle (P. wallichiana), and cedar wood (C. deodara) procured from M/S Natural Biotech Products, Baggi, Mandi, Himachal Pradesh, India and extracted through steam/hydro distillation.
4.2. Test Insect
C. maculatus and C. chinensis obtained from Council of Scientific and Industrial Research–Central Food Technological Research Institute (CSIR–CFTRI), Mysore, Karnataka, India, for further rearing in the Entomology laboratory, CSIR-Institute of Himalayan Bioresource Technology (IHBT), Palampur, H.P, India under controlled conditions at 25 ± 2 °C temperature, 60 ± 5% relative humidity. The adults were fed on the uninfected dried green gram (Vigna radiata (L.)) seeds in plastic jars and covered with a black muslin cloth. The adults were inspected for growth regularly (20–30 days intervals). The newly emerged adults were then transferred to 1 L plastic jars containing uninfested seeds for mating and egg-laying to ensure sufficient adults. The adults (1–4 days old) were used for bioassay and other experiments. The dead adults removed after their adult period competed, either by sieving the grains or handpicking, depending on the number of adults.
4.3. Gas Chromatography (GC) Analysis
The composition of EOs determined by gas chromatography (GC) on a Shimadzu GC 2010 equipped with DB–5 (J &W Scientific, Folsom, CA, USA) fused silica capillary column (30 m × 0.25 mm, i.e., 0.25 µm film thickness) with a flame ionisation detector (FID) [21,47]. The GC oven temperature was programmed at 70 °C (initial temperature), held for 4 min, then increased at a rate of 4 °C/min to 220 °C and held for 5 min. The injector temperature was 240 °C, the detector temperature was 260 °C, and the samples were injected in split mode. The carrier gas was nitrogen at a column flow rate of 1.05 mL/min (100 kPa). The sample’s retention indices (RI) were determined based on homologous n-alkane hydrocarbons under the same conditions.
4.4. GC-MS Analysis and Identification
The gas chromatography-mass spectrometry (GC-MS) analysis of EOs carried out using a Shimadzu QP 2010 using a DB–5 (J&W Scientific, Folsom, CA, USA) capillary column (30 m × 0.25 mm i.d., 0.25 µm film thickness) [9]. The GC oven temperature was 70 °C for 4 min and then increased to 220 °C at 4 °C /min and held for 5 min. The injector temperature was 240 °C, the interface temperature was 250 °C, the mass acquisition range was 800–50 amu, and the ionisation energy was 70 eV. The carrier gas used was Helium. Compounds were identified using a library search of the National Institute of Standards and Technology (NIST) database [63], as well as by comparing their RI and mass spectral frame pattern with those reported in the literature [22].
4.5. Fumigant Toxicity of Essential Oils and Their Combinations against the Pulse Beetle
Five different test concentrations (625 to 10,000 µL/L) of EOs viz., A. calamus, H. spicatum, J. recurva, J. communis, L. angustifolia, P. wallichiana, and C. deodara and their combinations (1:1 ratio) were taken for bioassay against the adults of pulse beetle for the synergistic activity. The experiments were carried out in 15 mL glass vials. Whatman No. 9 filter paper was inserted in the inner portion of the vial cap, and EO was released into the filter papers. The vials were kept in controlled laboratory conditions to record the adult mortality at different intervals. There were five treatments per EO, and each was replicated five times. Aluminium phosphide (0.5–0.9 mg/100 g grain) was also tested as a positive control against adults of C. maculatus. Observations on mortality were recorded from 24 to 120 h after treatment for EOs and their combinations/binary mixtures to calculate LC50 values and the co-toxicity coefficient for binary mixtures.
The co-toxicity coefficient (CTC) was calculated using the formula:
CTC = [LC50 of A/LC50 of A (in a mixture)] × 100
If the mixture gives a CTC > 100 (synergistic action), CTC < 100 (independent action), and CTC = 100 (similar action).
4.6. Repellent Activity of Essential Oils against the Pulse Beetle
The repellency of EOs against pulse beetle was studied as per the suggested methodology [21,64]. Five concentrations (62.5 to 1000 µL/L) were prepared, and each concentration/treatment was replicated five times. Whatman No. 9 filter paper (diameter 9 cm) was cut and marked with a pencil into two halves, each labelled as untreated (UT) and treated (T). Filter papers were transferred to Petri plates (diameter 9 cm), treated with required concentrations of EOs, and then allowed to air dry for 5 min. Ten adults (2–3 days old) were released in the centre of the Petri plate, and the Petri plates were sealed with parafilm to prevent the escape of adults. Observations on repellency were recorded after 1, 2, 3, 4, 5, and 24 h of treatment to calculate RC50 values.
4.7. Ovipositional Deterrent Activity of Essential Oils against the Pulse Beetle
Ovipositional deterrence of EOs against pulse beetle was studied as per the suggested methodology [64]. There were five concentrations (625 to 10,000 µL/L). Five concentrations were made by mixing EOs in Tween 80. Seeds (20 no./plate) were dipped in different concentrations for 10 s, then removed and placed on filter paper to air dry for 10-15 min. Treated seeds were placed in a Petri plate (diameter 9 cm), and then ten adults of one-day old (5 male and 5 female) were released. Petri plates were sealed with parafilm to prevent the escape of the adults. For the control, seeds were treated with Tween 80 only. Each treatment was replicated five times. The number of eggs laid on seeds of green gram was observed from 24 to 72 h.
Percent oviposition inhibition was calculated by using the formula [65].
where NT = No. of eggs in untreated and NT = No. of eggs laid in treated.
OI = [(NC − NT)/NC] ×100
4.8. Detoxification Enzyme Inhibition of A. Calamus and L. Angustifolia EO against the Pulse Beetle
4.8.1. Sample Preparation
Detoxification enzymes, i.e., Acetylcholinesterase (AChE) and Glutathione-S-Transferase (GST) inhibition activities, were completed as per the standard methods [21]. Four different concentrations of A. calamus and L. angustifolia EO (10,000, 5000, 2500, and 1250 µL/L) were taken for both C. maculatus and C. chinensis for detoxification enzyme inhibition activity. The adults alive after 24 and 48 h (5–8 adults weighing 20 mg/concentration) were collected for enzyme assay. The adults in each test concentration were transferred to a centrifuge tube and homogenized phosphate buffer (pH 7.4) in a ratio of 1:9. The weight of an adult (mg): the volume of buffer (mL) was kept in a ratio of 1:9. The adults were then homogenized with a homogenizer in a micro pestle mortar. The homogenate was transferred immediately under ice bath conditions and then centrifuged at 4 °C and 12,000 rpm for 30 min. The supernatant was taken into a new centrifuge tube for protein estimation by Bradford assay [66] for all the concentrations before proceeding with enzyme assays. The same assay was repeated thrice for separate homogenates, and then average values were taken for protein estimation.
4.8.2. Protein Estimation
Protein estimation was done using the Bradford method [66] by adding 2 µL of homogenate, 38 µL of MilliQ to 160 µL of Bradford reagent in triplicates. After incubation of the mixture for 15 min at room temperature, the absorbance was measured at 595 nm. Absorbance was converted into protein concentrations, and dilutions were made concerning lower concentrations for the AChE assay.
4.8.3. Acetylcholinesterase Assay
The diluted 25 µL homogenates in triplicates were incubated for 30 min at room temperature with 25 µL of the reaction mixture (50 µL of DTNB, 50 µL of Acetothiocholine, and 900 µL of assay buffer). The AChE activity was spectrophotometrically measured at 410 nm in a microplate reader (Biotek, Synergy H1 Hybrid Multi-Mode reader) and represented as milliunits per milligram of protein (mU/mg). For the determination of AChE, the Acetylcholinesterase Assay Kit was procured from Abcam, UK.
4.8.4. Glutathione-S-Transferase Assay
The reaction contains 100 µL of the solution, in which 75 µL of assay buffer, 10 µL of the homogenised sample, and 10 µL of glutathione were added. To start the reactions, 5 µL CDNB was added to each well in triplicates to the microplate at room temperature. These reaction mixtures were incubated at RT in a 96-well microplate. The enzyme kinetics were measured at the absorbance of 340 nm at 37 °C for 20 min in a microplate reader with continuous mixing for 10 s after 60 s of lag time. The extinction coefficient of 0.0096 μM−1 for CDNB was used to calculate the Glutathione-S-transferase activity and represented as nanomolar per minute per millilitre of a sample (nmol/min/mL). To determine the GST enzyme, the Glutathione-S-transferase Assay Kit was procured from Cayman Chemical, 1180 E, Ellsworth Road, Ann Arbor, MI, USA.
4.9. Statistical Analysis
The data on fumigant toxicity, synergistic activities, ovipositional inhibition, and repellence of different EOs was compiled. Lethal concentration (LC50) and repellent concentration (RC50) values were calculated by Probit analysis [67] using SPSS software v.16.0. The data on fumigant toxicity and ovipositional inhibition were subjected to multivariate analysis, and enzyme inhibition was subjected to one-way ANOVA by SPSS software. Means were compared by Tukey’s post hoc test to know the significant differences between treatments.
5. Conclusions
A. calamus, L. angustifolia and H. spicatum oil alone showed promising fumigant toxicity against both C. maculatus and C. chinensis. Among combinations, A. calamus oil with L. angustifolia and C. deodara against C. chinensis (LC50 = 92.18-118.54 µL/L) and A. calamus with L. angustifolia and P. wallichiana against C. maculatus (LC50 = 204.01-312.23 µL/L) showed promising toxicity/synergistic. A. calamus and H. spicatum also showed promising repellence to both species of pulse beetle. The insecticidal activities of promising EOs may be due to the presence of cis-asarone (85.37%), 1,8-cineole (28.31%) and 2-β-pinene (39.18%) are the major constituents. All the concentrations of A. calamus and L. angustifolia significantly inhibited the GST and AChE in both species of pulse beetle, so these enzymes may be the target site of action for tested oils in pulse beetle. Therefore, these promising EOs may be recommended to control pulse beetle where the grains are stored in bins and big godowns (staked grains) subject to large-scale trials.
Supplementary Materials
The following supporting information can be downloaded at: https://www.mdpi.com/article/10.3390/molecules28020492/s1, The chemical composition and ovipositional deterrent activities of seven EOs are represented in the supplementary tables: Table S1, Chemical composition of essential oil of H. spicatum. Table S2, Chemical composition of essential oil of C. deodara. Table S3, Chemical composition of essential oil of J. communis. Table S4, Chemical composition of essential oil of J. recurva. Table S5, Chemical composition of essential oil of L. angustifolia. Table S6, Chemical composition of essential oil of P. wallichiana. Table S7, Ovipositional inhibition effect of different oils against adults of C. maculatus. Table S8, Ovipositional inhibition effect of different oils against adults of C. maculatus. Table S9, Ovipositional inhibition effect of different oils against adults of C. maculatus. Table S10, Ovipositional inhibition effect of different oils against adults of C. chinensis. Table S11, Ovipositional inhibition effect of different oils against adults of C. chinensis. Table S12, Ovipositional inhibition effect of different oils against adults of C. chinensis.
Author Contributions
Conceptualization, S.G.E.R.; methodology, S.G.E.R., H.G., D. and U.; validation, S.G.E.R. and H.G.; formal analysis, S.G.E.R. and H.G.; investigation, S.G.E.R. and H.G.; data curation, H.G., D. and U.; writing original draft preparation, H.G.; writing review and editing, S.G.E.R. All authors have read and agreed to the published version of the manuscript.
Funding
This work was supported by the Council of Scientific and Industrial Research (CSIR), New Delhi, India, for funding the project entitled “CSIR-Aroma Mission Phase-II” (HCP-0007) and “CSIR-Floriculture Mission” (HCP-0037).
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
The original contribution presented in the study is included in the article. Further inquiries can be directed to the corresponding author.
Acknowledgments
The authors are thankful to the Council of Scientific and Industrial Research (CSIR), Government of India, for providing financial support under the project “CSIR-Aroma Mission Phase-II” (HCP-0007) and “CSIR-Floriculture Mission” (HCP-0037). The authors are thankful to the Director, CSIR-Institute of Himalayan Bioresource Technology (IHBT), Palampur, India for providing the necessary facilities. CSIR-IHBT publication no. for this manuscript is 5166.
Conflicts of Interest
The authors declare that they have no known competing financial interest or personal relationships that could have appeared to influence the work reported in this paper.
References
- Bessada, S.M.; Barreira, J.C.; Oliveira, M.B.P. Pulses and food security: Dietary protein, digestibility, bioactive and functional properties. Trends Food Sci. Technol. 2019, 93, 53–68. [Google Scholar] [CrossRef]
- Murugesan, R.; Vasuki, K.; Kaleeswaran, B.; Santhanam, P.; Ravikumar, S.; Alwahibi, M.S.; Soliman, D.A.; Mohsen Ahmed Almunqedhi, B.; Alkahtani, J. Insecticidal and repellent activities of Solanum torvum (Sw.) leaf extract against stored grain Pest, Callosobruchus maculatus (F.) (Coleoptera: Bruchidae). J. King Saud Univ.-Sci. 2021, 33, 101390. [Google Scholar] [CrossRef]
- Seni, A.; Mishra, K.M. Pulse beetle, Callosobruchus spp. (Coleoptera: Chrysomelidae); a major threat in legume grain storage and their management. Acta Phytopathol. Entomol. Hung. 2022, 57, 49–65. [Google Scholar] [CrossRef]
- Mogbo, T.C.; Okeke, T.E.; Akunne, C.E. Studies on the resistance of cowpea seeds (Vigna unguiculata) to weevil (Callosobruchus maculatus) infestations. Am. J. Zool. Res. 2014, 2, 37–40. [Google Scholar] [CrossRef]
- Tuda, M.; Chou, L.Y.; Niyomdham, C.; Buranapanichpan, S.; Tateishi, Y. Ecological factors associated with pest status in Callosobruchus (Coleoptera: Bruchidae): High host specificity of non-pests to Cajaninae (Fabaceae). J. Stored Prod. Res. 2005, 41, 31–45. [Google Scholar] [CrossRef]
- Ajayi, O. Bioactivity of the leaf extracts of Morinda lucida (Benth.) against cowpea Bruchid, Callosobruchus maculatus (F.) (Coleoptera: Chrysomelidae). Exp. Agric. Hortic. 2012, 1, 1–7. [Google Scholar]
- Beck, C.W.; Blumer, L.S. A Handbook on Bean Beetles, Callosobruchus maculatus; National Science Foundation: Alexandria, VA, USA, 2014; Available online: http://www.beanbeetles.org/handbook.pdf (accessed on 3 October 2022).
- França, A.F.J.; Araújo, J.N.; Santos, Y.Q.; Carelli, G.S.C.; Silva, D.A.; Amorim, T.M.L.; Migliolo, L.; Santos, E.A.; Oliveira, A.S.; Uchoa, A.F. Vicilin from Anadenanthera colubrina Seeds: An alternative tool to combat Callosobruchus maculatus. Saudi J. Biol. Sci. 2021, 28, 5229–5237. [Google Scholar] [CrossRef]
- Jayaram, C.S.; Chauhan, N.; Dolma, S.K.; Reddy, S.G.E. Chemical composition, and insecticidal activities of essential oils against the pulse beetle. Molecules 2022, 27, 568. [Google Scholar] [CrossRef] [PubMed]
- Garry, V.F.; Griffith, J.; Danzl, T.J.; Nelson, R.L.; Whorton, E.B.; Krueger, L.A.; Cervenka, J. Human genotoxicity: Pesticide applicators and phosphine. Science 1989, 246, 251–255. [Google Scholar] [CrossRef]
- Okonkwo, E.U.; Okoye, W.I. The efficacy of four seed powders and the essential oils as protectants of cowpea and maize grains against infestation by Callosobruchus maculatus (Fabricius) (Coleoptera: Bruchidae) and Sitophilus zeamais (Motschulsky) (Coleoptera: Curculionidae) in Nigeria. Int. J. Pest Manag. 1996, 42, 143–146. [Google Scholar] [CrossRef]
- Nerio, L.S.; Olivero-Verbel, J.; Stashenko, E.E. Repellent activity of essential oils from seven aromatic plants grown in Colombia against Sitophilus zeamais Motschulsky (Coleoptera). J. Stored Prod. Res. 2009, 45, 212–214. [Google Scholar] [CrossRef]
- Vendan, S.E.; Manivannan, S.; Sunny, A.M.; Murugesan, R. Phytochemical residue profiles in rice grains fumigated with essential oils for the control of rice weevil. PLoS ONE 2017, 12, e0186020. [Google Scholar] [CrossRef] [PubMed]
- Bakkali, F.; Averbeck, S.; Averbeck, D.; Idaomar, M. Biological effects of essential oils—A review. Food Chem. Toxicol. 2008, 46, 446–475. [Google Scholar] [CrossRef] [PubMed]
- Marques, D.M.; de Freitas Rocha, J.; de Almeida, T.S.; Mota, E.F. Essential oils of caatinga plants with deletary action for Aedes aegypti: A Review. S. Afr. J. Bot. 2021, 143, 69–78. [Google Scholar] [CrossRef]
- Rajendran, S.; Sriranjini, V. Plant products as fumigants for stored-product insect control. J. Stored Prod. Res. 2008, 44, 126–135. [Google Scholar] [CrossRef]
- Rajashekar, Y.; Bakthavatsalam, N.; Shivanandappa, T. Botanicals as Grain Protectants. Psyche 2012, e646740. [Google Scholar] [CrossRef]
- Singh, K.D.; Mobolade, A.J.; Bharali, R.; Sahoo, D.; Rajashekar, Y. Main plant volatiles as stored grain pest management approach: A review. J. Agric. Food Res. 2021, 4, 100127. [Google Scholar] [CrossRef]
- Dubey, V.S.; Bhalla, R.; Luthra, R. An overview of the non-mevalonate pathway for terpenoid biosynthesis in plants. J. Biosci. 2003, 28, 637–646. [Google Scholar] [CrossRef]
- Regnault-Roger, C.; Vincent, C.; Arnason, J.T. Essential oils in insect control: Low-risk products in a high-stakes world. Ann. Rev. Entomol. 2012, 57, 405–424. [Google Scholar] [CrossRef]
- Chauhan, N.; Kashyap, U.; Dolma, S.K.; Reddy, S.G.E. Chemical composition, insecticidal, persistence and detoxification enzyme inhibition activities of essential oil of Artemisia maritima against the pulse beetle. Molecules 2022, 27, 1547. [Google Scholar] [CrossRef]
- Adams, R.P. Identification of Essential Oil Components by Gas Chromatography/Quadrupole Mass Spectroscopy, 4th ed.; Allured Publication: Carol Stream, IL, USA, 2007. [Google Scholar]
- Bassolé, I.H.N.; Lamien-Meda, A.; Bayala, B.O.L.C.; Obame, L.C.; Ilboudo, A.J.; Franz, C.; Novak, J.; Nebié, R.C.; Dicko, M.H. Chemical composition and antimicrobial activity of Cymbopogon citratus and Cymbopogon giganteus essential oils alone and in combination. Phytomedicine 2011, 18, 1070–1074. [Google Scholar] [CrossRef] [PubMed]
- Bilia, A.R.; Guccione, C.; Isacchi, B.; Righeschi, C.; Firenzuoli, F.; Bergonzi, M.C. Essential oils loaded in nanosystems: A developing strategy for a successful therapeutic approach. Evid.-Based Complement. Altern. Med. 2014, 2014, 651593. [Google Scholar] [CrossRef] [PubMed]
- Perricone, M.; Arace, E.; Corbo, M.R.; Sinigaglia, M.; Bevilacqua, A. Bioactivity of essential oils: A review on their interaction with food components. Front. Microbiol. 2015, 6, 76. [Google Scholar] [CrossRef]
- Hanif, M.A.; Nisar, S.; Khan, G.S.; Mushtaq, Z.; Zubair, M. Essential oils. In Essential Oil Research; Malik, S., Ed.; Springer: Cham, Switzerland, 2019; pp. 3–17. [Google Scholar] [CrossRef]
- Kim, S.I.; Roh, J.Y.; Kim, D.H.; Lee, H.S.; Ahn, Y.J. Insecticidal activities of aromatic plant extracts and essential oils against Sitophilus oryzae and Callosobruchus chinensis. J. Stored Prod. Res. 2003, 39, 293–303. [Google Scholar] [CrossRef]
- Kalagatur, N.K.; Kamasani, J.R.; Siddaiah, C.; Gupta, V.K.; Krishna, K.; Mudili, V. Combinational inhibitory action of Hedychium spicatum L. essential oil and γ-radiation on growth rate and mycotoxins content of Fusarium graminearum in maize: Response surface methodology. Front. Microbiol. 2018, 9, 1511. [Google Scholar] [CrossRef] [PubMed]
- Rawat, A.; Thapa, P.; Prakash, O.; Kumar, R.; Pant, A.K.; Srivastava, R.M.; Rawat, D.S. Chemical composition, herbicidal, antifeedant and cytotoxic activity of Hedychium spicatum Sm.: A Zingiberaceae herb. Trends Phytochem. Res. 2019, 3, 123–136. [Google Scholar]
- Arya, S.; Kumar, R.; Prakash, O.; Kumar, S.; Mahawer, S.K.; Chamoli, S.; de Oliveira, M.S. Chemical composition and biological activities of Hedychium coccineum Buch.-Ham. ex Sm. essential oils from Kumaun hills of Uttarakhand. Molecules 2022, 27, 4833. [Google Scholar] [CrossRef]
- Miao, J.K.; Shi, R.H.; Li, C.; Li, X.W.; Chen, Q.X. Sweet Flag (Acorus calamus) Oils. In Essential Oils in Food Preservation, Flavor and Safety; Academic Press: Cambridge, MA, USA, 2016; pp. 775–782. [Google Scholar] [CrossRef]
- Brito, V.D.; Achimón, F.; Pizzolitto, R.P.; Ramírez Sánchez, A.; Gómez Torres, E.A.; Zygadlo, J.A.; Zunino, M.P. An alternative to reduce the use of the synthetic insecticide against the maize weevil Sitophilus zeamais through the synergistic action of Pimenta racemosa and Citrus sinensis essential oils with chlorpyrifos. J. Pest Sci. 2021, 94, 409–421. [Google Scholar] [CrossRef]
- Singh, P.; Andola, H.; Rawat, M.S.M.; Jangwan, J.S. GC-MS analysis of essential oil from Lavandula angustifolia cultivated in Garhwal Himalaya. J. Nat. Prod. 2015, 5, 268–272. [Google Scholar] [CrossRef]
- Beale, D.J.; Morrison, P.D.; Karpe, A.V.; Dunn, M.S. Chemometric analysis of lavender essential oils using targeted and untargeted GC-MS acquired data for the rapid identification and characterization of oil quality. Molecules 2017, 22, 1339. [Google Scholar] [CrossRef]
- Dong, G.; Bai, X.; Aimila, A.; Aisa, H.A.; Maiwulanjiang, M. Study on lavender essential oil chemical compositions by GC-MS and improved pGC. Molecules 2020, 25, 3166. [Google Scholar] [CrossRef] [PubMed]
- Dambolena, J.S.; Gallucci, M.N.; Luna, A.; Gonzalez, S.B.; Guerra, P.E.; Zunino, M.P. Composition, antifungal and antifumonisin activity of Pinus wallichiana, Pinus monticola and Pinus strobus essential oils from Patagonia Argentina. J. Essent. Oil-Bear. Plants 2016, 19, 1769–1775. [Google Scholar] [CrossRef]
- Rashed, K. Phytochemical and biological activities of Pinus wallichiana: A short review. Int. J. Pharm. Sci. Drug Anal. 2022, 2, 38–40. [Google Scholar]
- Kumar, S.; Mitra, B.; Kashyap, S.; Kumar, S. Physicochemical properties, GC-MS analysis and impact of different material size on yield of Himalayan C. deodara essential oil. Pharmacogn. Res. 2022, 14, 181–187. [Google Scholar] [CrossRef]
- Perry, N.B.; Anderson, R.E.; Brennan, N.J.; Douglas, M.H.; Heaney, A.J.; McGimpsey, J.A.; Smallfield, B.M. Essential oils from dalmatian sage (Salvia officinalis L.): Variations among individuals, plant parts, seasons, and sites. J. Agric. Food Chem. 1999, 47, 2048–2054. [Google Scholar] [CrossRef]
- Lee, S.E.; Lee, B.H.; Choi, W.S.; Park, B.S.; Kim, J.G.; Campbell, B.C. Fumigant toxicity of volatile natural products from Korean spices and medicinal plants towards the rice weevil, Sitophilus oryzae (L). Pest Manag. Sci. 2001, 57, 548–553. [Google Scholar] [CrossRef]
- Tripathi, A.K.; Prajapati, V.; Aggarwal, K.K.; Kumar, S. Toxicity, feeding deterrence, and effect of the activity of 1, 8-cineole from Artemisia annua on progeny production of Tribolium castanaeum (Coleoptera: Tenebrionidae). J. Econ. Entomol. 2001, 94, 979–983. [Google Scholar] [CrossRef]
- Hafez, M.; Dimetry, N.Z.; Abbass, M.H. Insecticidal and biological efficacy of two vegetable oils against Callosobruchus maculatus (Fabricius) (Coleoptera: Bruchidae) under laboratory conditions. Arch. Phytopathol. Pflanzenschutz. 2014, 47, 1942–1955. [Google Scholar] [CrossRef]
- Baskaran, J.; Dhanasekaran, S.; Krishnappa, K.; Elumalai, K. Toxicity of essential oils used for the protection of stored cowpea against Callosobruchus maculatus F. (Leoptera: Bruchidae). Int. J. Curr. Res. 2010, 9, 039–045. [Google Scholar]
- Su, H.C. Laboratory evaluation of toxicity of calamus oil against four species of stored-product insects. J. Entomol. Sci. 1991, 26, 76–80. [Google Scholar] [CrossRef]
- Shukla, R.; Singh, P.; Prakash, B.; Dubey, N.K. Assessment of essential oil of Acorus calamus L. and its major constituent β-Asarone in post-harvest management of Callosobruchus chinensis L. Acta Phytopathol. Entomol. Hung. 2016, 19, 542–552. [Google Scholar] [CrossRef]
- Hasan, M.S.; Ullah, E.; Ahmad, F.; Wakil, W. Insecticidal activity of different doses of Acorus calamus oil against Trogoderma granarium (Everts). Pak. J. Agric. Sci. 2006, 43, 55–58. [Google Scholar]
- Koundal, R.; Dolma, S.K.; Chand, G.; Agnihotri, V.K.; Reddy, S.G.E. Chemical composition, and insecticidal properties of essential oils against diamondback moth (Plutella xylostella L.). Toxin Rev. 2020, 39, 371–381. [Google Scholar] [CrossRef]
- Sakhanokho, H.F.; Sampson, B.J.; Tabanca, N.; Wedge, D.E.; Demirci, B.; Baser, K.H.C.; Spiers, J.M. Chemical composition, antifungal and insecticidal activities of Hedychium essential oils. Molecules 2013, 18, 4308–4327. [Google Scholar] [CrossRef] [PubMed]
- Arruda, M.; Viana, H.; Rainha, N.; Neng, N.R.; Rosa, J.S.; Nogueira, J.M.; Barreto, M.D.C. Anti-acetylcholinesterase and antioxidant activity of essential oils from Hedychium gardnerianum Sheppard ex Ker-Gawl. Molecules 2012, 17, 3082–3092. [Google Scholar] [CrossRef]
- Ncibi, S.; Barbouche, N.; Haouel-hamdi, S.; Ammar, M. Insecticidal activity of several Tunisian essential oils against two major pests of stored grain Rhyzopertha dominica (Fabricius, 1792) and Tribolium castaneum (Herbest 1797). J. New Sci. Agric. Biotech. 2019, 66, 4182–4194. [Google Scholar]
- Sayada, N.; Tine, S.; Soltani, N. Evaluation of a botanical insecticide, lavender (Lavandula angustifolia (M.)) essential oil as toxicant, repellent and antifeedant against lesser grain borer (Rhyzopertha dominica (F.)). Appl. Ecol. Environ. Res. 2008, 20, 1301–1324. [Google Scholar] [CrossRef]
- Abdali, E.Y.; Agour, A.; Allali, A.; Bourhia, M.; Moussaoui, E.A.; Eloutassi, N.; Bouia, A. Lavandula dentata L.: Phytochemical analysis, antioxidant, antifungal and insecticidal activities of its essential oil. Plants 2022, 11, 311. [Google Scholar] [CrossRef]
- Mazarin, A.; Nukenine, E.N.; Niu, C.; Vincent, F.V. Synergistic effects of wood ash and essential oil on fecundity, pupal eclosion and adult mortality of Callosobruchus maculatus (Coleoptera: Bruchidae) cowpea seed weevil. Am. J. Exp. Agric. 2016, 11, 1–12. [Google Scholar] [CrossRef]
- Hategekimana, A.; Erler, F. Comparative repellent activity of single, binary, and ternary combinations of plant essential oils and their major components against Sitophilus oryzae L. (Coleoptera: Curculionidae). J. Plant Prot. Res. 2020, 127, 873–881. [Google Scholar] [CrossRef]
- Brito, R.D.C.; Fontes, L.D.S.; da Silva, P.H.S.; Santana, C.D.S. Essential oils from Betula lenta, Cinnamomum cassia, Citrus aurantium var. Amara and Acorus calamus as biopesticides against cowpea weevil. Int. J. Trop. Insect Sci. 2022, 42, 261–268. [Google Scholar] [CrossRef]
- Rotimi, J.; Evbuomwan, C.O. Deterrent effects of citrus peel oils on oviposition and adult emergence of the cowpea weevil, Callosobruchus maculatus (F.) (Coleoptera: Bruchidae). Adv. Appl. Sci. Res. 2012, 3, 3545–3550. [Google Scholar]
- Matos, L.F.; da Cruz Lima, E.; de Andrade Dutra, K.; Navarro, D.M.D.A.F.; Alves, J.L.R.; Silva, G.N. Chemical composition, and insecticidal effect of essential oils from Illicium verum and Eugenia caryophyllus on Callosobruchus maculatus in cowpea. Ind. Crops Prod. 2020, 145, 112088. [Google Scholar] [CrossRef]
- Upadhyay, N.; Dwivedy, A.K.; Kumar, M.; Prakash, B.; Dubey, N.K. Essential oils as eco-friendly alternatives to synthetic pesticides for the control of Tribolium castaneum (Herbst) (Coleoptera: Tenebrionidae). J. Essent. Oil-Bear. Plants 2018, 21, 282–297. [Google Scholar] [CrossRef]
- Hu, Z.D.; Xia, F.E.N.G.; Lin, Q.S.; Chen, H.Y.; Li, Z.Y.; Fei, Y.I.N.; Gao, X.W. Biochemical mechanism of chlorantraniliprole resistance in the diamondback moth, Plutella xylostella Linnaeus. J. Integr. Agric. 2014, 13, 2452–2459. [Google Scholar] [CrossRef]
- Tak, J.H.; Isman, M.B. Metabolism of citral, the major constituent of lemongrass oil, in the cabbage looper, Trichoplusia ni, and effects of enzyme inhibitors on toxicity and metabolism. Pestic. Biochem. Physiol. 2016, 133, 20–25. [Google Scholar] [CrossRef] [PubMed]
- Yang, H.; Piao, X.; Zhang, L.; Song, S.; Xu, Y. Ginsenosides from the stems and leaves of Panax ginseng show antifeedant activity against Plutella xylostella. Ind. Crops Prod. 2018, 124, 412–417. [Google Scholar] [CrossRef]
- Abdelgaleil, S.A.; Mohamed, M.I.; Shawir, M.S.; Abou-Taleb, H.K. Chemical composition, insecticidal and biochemical effects of essential oils of different plant species from Northern Egypt on the rice weevil, Sitophilus oryzae L. J. Pest Sci. 2016, 89, 219–229. [Google Scholar] [CrossRef]
- Stein, S.E. Mass Spectral Database and Software, version 3.02; US Department of Commerce, National Institute of Standards and Technology: Gaithersburg, MD, USA, 2005.
- Eccles, K.; Powder-George, Y.L.; Mohammed, F.K.; Khan, A. Efficacy of Artocarpus altilis (Parkinson) Fosberg extracts on contact mortality, repellency, oviposition deterrency and fumigant toxicity of Callosobruchus maculatus (F.) (Coleoptera: Bruchidae). Int. J. Pest Manag. 2019, 65, 72–78. [Google Scholar] [CrossRef]
- Isman, M.B. Plant essential oils for pest and disease management. Crop Prot. 2000, 19, 603–608. [Google Scholar] [CrossRef]
- Bradford, M.M. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal. Biochem. 1976, 72, 248–254. [Google Scholar] [CrossRef] [PubMed]
- Finney, D.J. Probit Analysis, 3rd ed.; Cambridge University Press: Cambridge, UK, 1971. [Google Scholar]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).