Phenethyl Ester of Gallic Acid Ameliorates Experimental Autoimmune Encephalomyelitis
Abstract
1. Introduction
2. Results
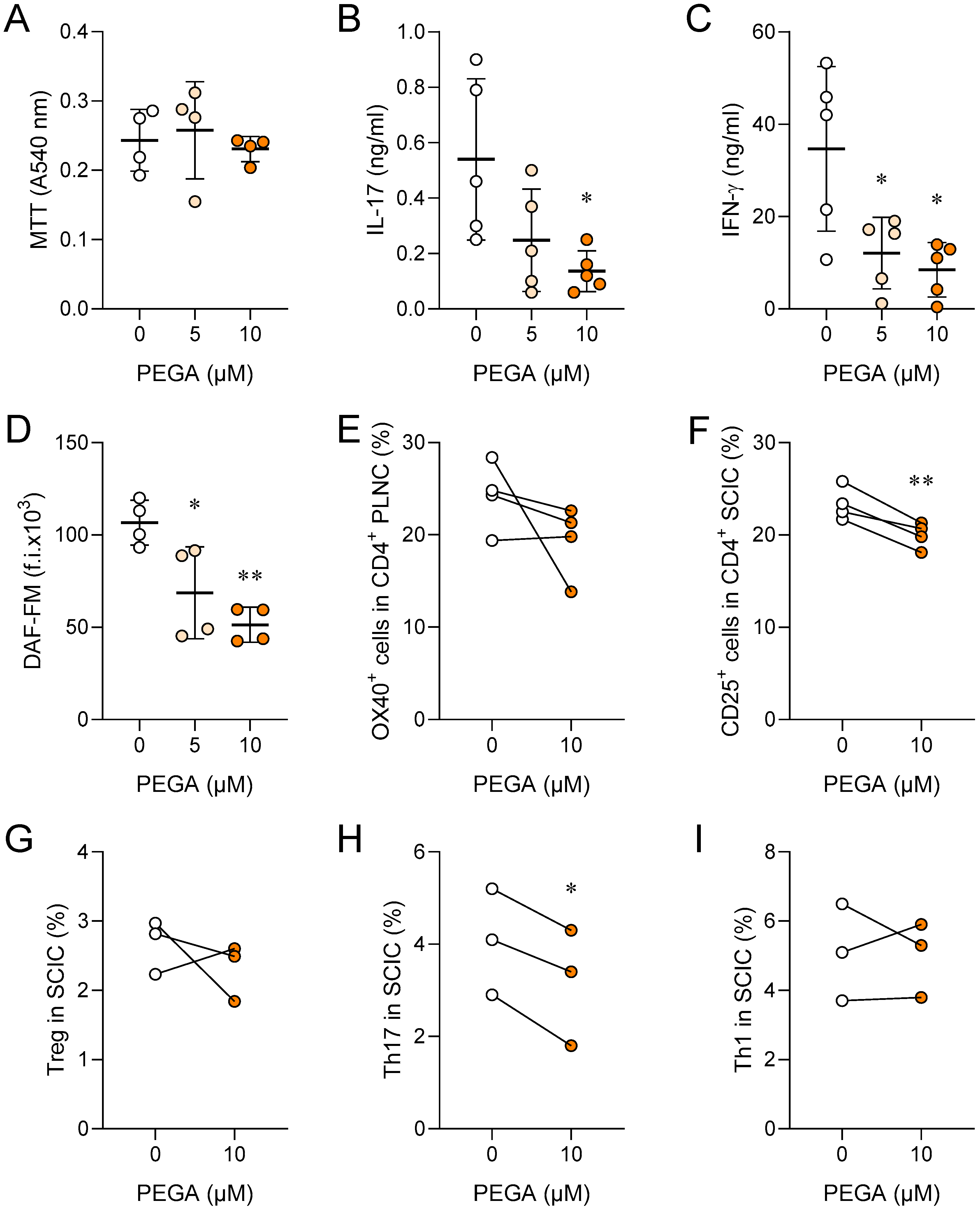
2.1. PEGA Affects Popliteal Lymph Node Cells (PLNC)
2.2. PEGA Affects Spinal Cord Immune Cells (SCIC)
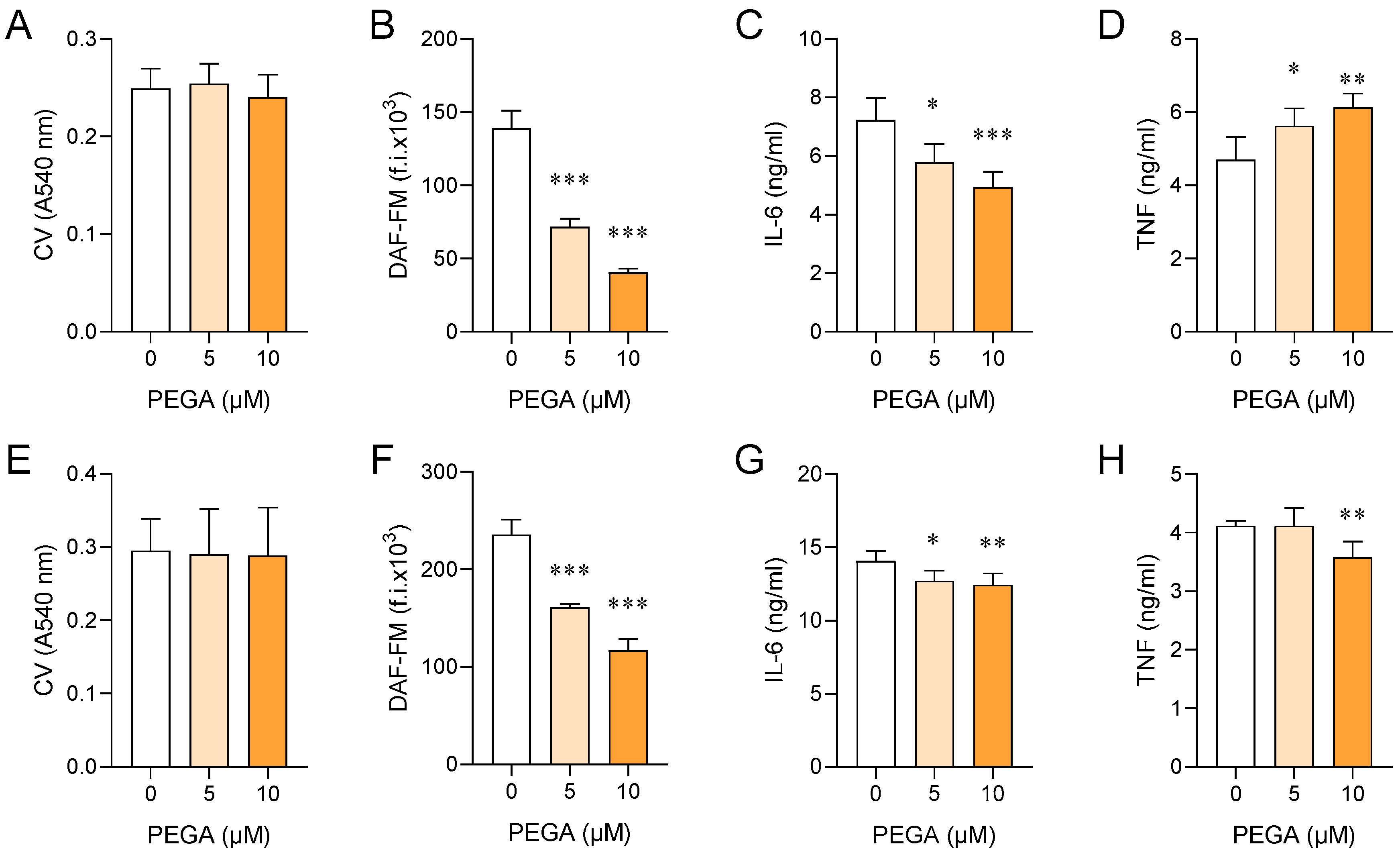
2.3. PEGA Affects Macrophages/Microglia
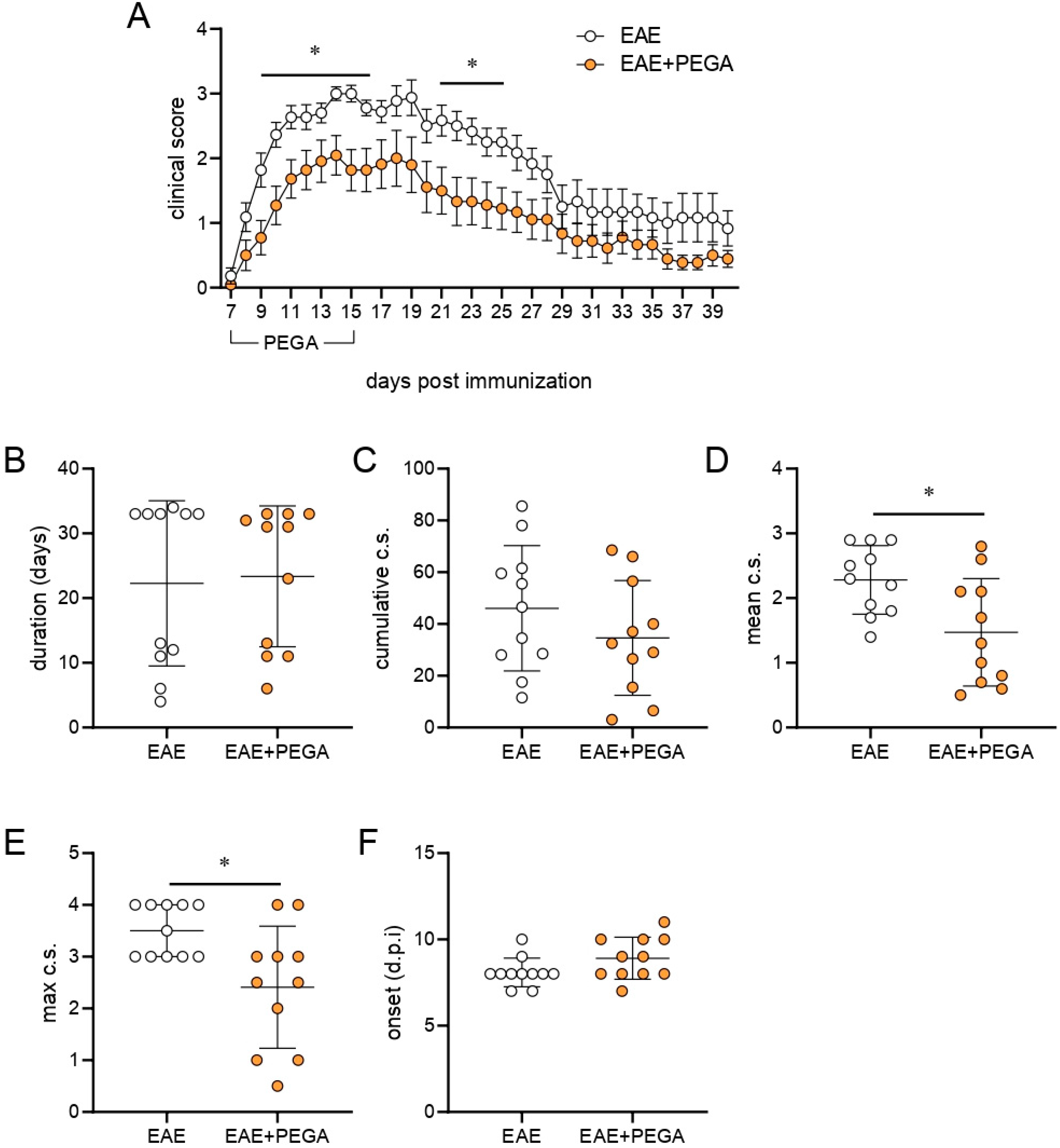
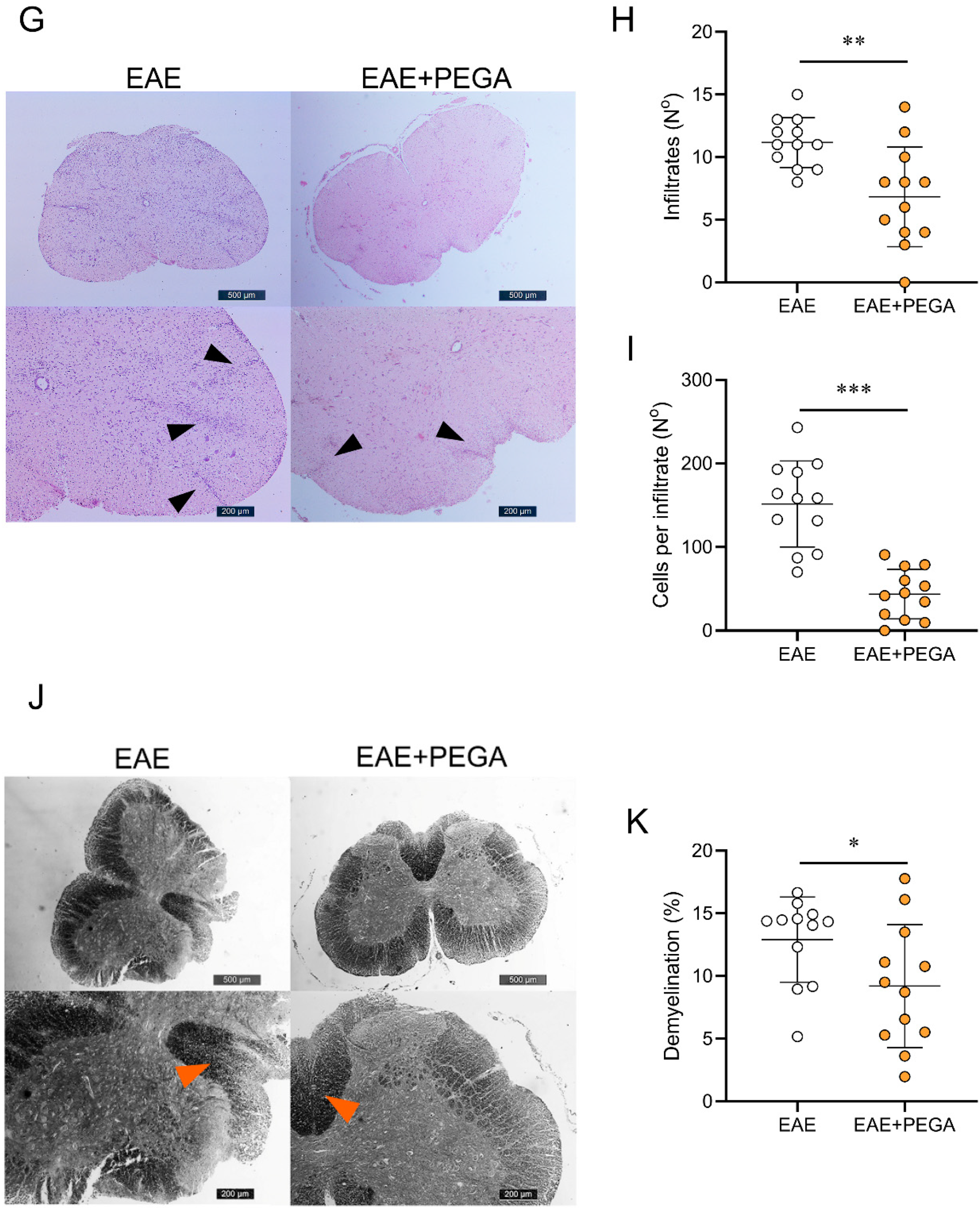
2.4. PEGA Ameliorates EAE
3. Discussion
4. Materials and Methods
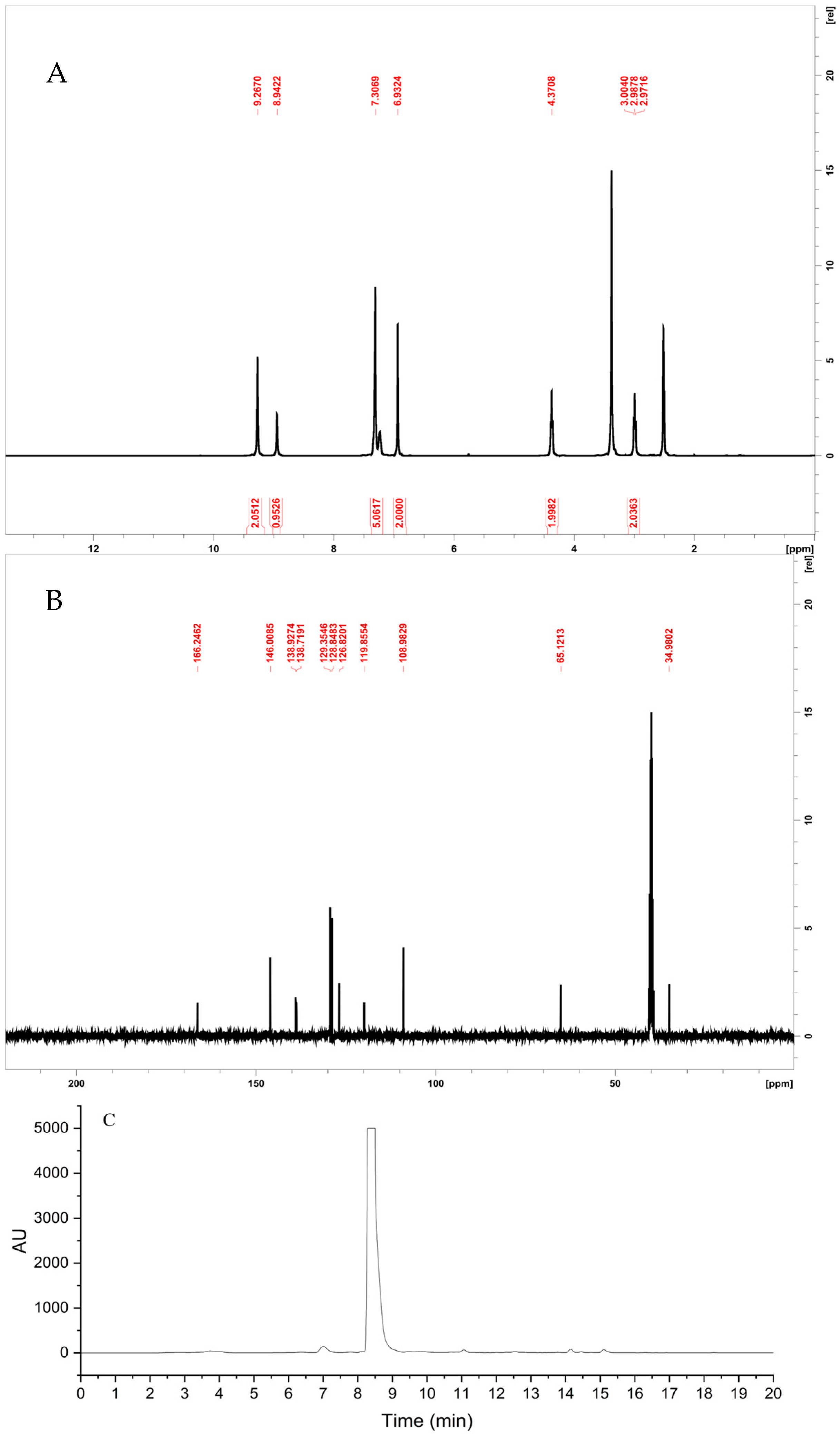
4.1. Synthesis and Application of PEGA
4.2. Experimental Animals, EAE, and PEGA Application
4.3. Histological Assessment of EAE
4.4. Isolation of Cells and Cell Cultures
4.5. Cell Viability Assays
4.6. ELISA
4.7. NO Detection
4.8. Cytofluorimetry
4.9. “Real-Time” RT-PCR
4.10. Statistical Analysis
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Rodríguez Murúa, S.; Farez, M.F.; Quintana, F.J. The Immune Response in Multiple Sclerosis. Annu. Rev. Pathol. 2022, 17, 121–139. [Google Scholar] [CrossRef] [PubMed]
- Saleem, S.; Anwar, A.; Fayyaz, M.; Anwer, F. An Overview of Therapeutic Options in Relapsing-remitting Multiple Sclerosis. Cureus 2019, 11, e5246. [Google Scholar] [CrossRef] [PubMed]
- Farzaei, M.H.; Shahpiri, Z.; Bahramsoltani, R.; Nia, M.M.; Najafi, F.; Rahimi, R. Efficacy and Tolerability of Phytomedicines in Multiple Sclerosis Patients: A Review. CNS Drugs 2017, 31, 867–889. [Google Scholar] [CrossRef] [PubMed]
- Mohtashami, L.; Shakeri, A.; Javadi, B. Neuroprotective natural products against experimental autoimmune encephalomyelitis: A review. Neurochem. Int. 2019, 129, 104516. [Google Scholar] [CrossRef] [PubMed]
- Bai, J.; Zhang, Y.; Tang, C.; Hou, Y.; Ai, X.; Chen, X.; Zhang, Y.; Wang, X.; Meng, X. Gallic acid: Pharmacological activities and molecular mechanisms involved in inflammation-related diseases. Biomed. Pharmacother. 2021, 133, 110985. [Google Scholar] [CrossRef] [PubMed]
- Ho, T.Y.; Lo, H.Y.; Liu, I.C.; Huang, S.L.; Hsiang, C.Y. Rosmarinic acid ameliorated psoriatic skin inflammation in mice through the novel inhibition of the interleukin-17A/interleukin-17A receptor interaction. Food Funct. 2022, 13, 6802–6812. [Google Scholar] [CrossRef]
- Zhu, L.; Gu, P.; Shen, H. Gallic acid improved inflammation via NF-κB pathway in TNBS-induced ulcerative colitis. Int. Immunopharmacol. 2019, 67, 129–137. [Google Scholar] [CrossRef]
- Fan, Y.; Piao, C.H.; Hyeon, E.; Jung, S.Y.; Eom, J.E.; Shin, H.S.; Song, C.H.; Chai, O.H. Gallic acid alleviates nasal inflammation via activation of Th1 and inhibition of Th2 and Th17 in a mouse model of allergic rhinitis. Int. Immunopharmacol. 2019, 70, 512–519. [Google Scholar] [CrossRef]
- Hu, G.; Zhou, X. Gallic Acid Ameliorates Atopic Dermatitis-like Skin Inflammation Through Immune Regulation in a Mouse Model. Clin. Cosmet. Investig. Dermatol. 2021, 14, 1675–1683. [Google Scholar] [CrossRef]
- Chan, B.C.; Li, L.F.; Hu, S.Q.; Wat, E.; Wong, E.C.; Zhang, V.X.; Lau, C.B.; Wong, C.K.; Hon, K.L.; Hui, P.C.; et al. Gallic Acid Is the Major Active Component of Cortex Moutan in Inhibiting Immune Maturation of Human Monocyte-Derived Dendritic Cells. Molecules 2015, 20, 16388–16403. [Google Scholar] [CrossRef]
- Abdullah, A.; Maged, M.; Hairul-Islam, M.I.; Osama, I.A.; Maha, H.; Manal, A.; Hamza, H. Activation of aryl hydrocarbon receptor signaling by a novel agonist ameliorates autoimmune encephalomyelitis. PLoS ONE 2019, 14, e0215981. [Google Scholar] [CrossRef] [PubMed]
- Diamantis, D.A.; Oblukova, M.; Chatziathanasiadou, M.; Gemenetzi, A.; Papaemmanouil, C.; Gerogianni, P.S.; Syed, N.; Crook, T.; Galaris, D.; Deligiannakis, Y.; et al. Bioinspired tailoring of fluorogenic thiol responsive antioxidant precursors to protect cells against H2O2-induced DNA damage. Free. Radic. Biol. Med. 2020, 160, 540–551. [Google Scholar] [CrossRef] [PubMed]
- Gerogianni, P.S.; Chatziathanasiadou, M.V.; Diamantis, D.A.; Tzakos, A.G.; Galaris, D. Lipophilic ester and amide derivatives of rosmarinic acid protect cells against H2O2-induced DNA damage and apoptosis: The potential role of intracellular accumulation and labile iron chelation. Redox Biol. 2018, 15, 548–556. [Google Scholar] [CrossRef] [PubMed]
- Karpus, W.J. Cytokines and Chemokines in the Pathogenesis of Experimental Autoimmune Encephalomyelitis. J. Immunol. 2020, 204, 316–326. [Google Scholar] [CrossRef] [PubMed]
- Hiltensperger, M.; Korn, T. The Interleukin (IL)-23/T helper (Th)17 Axis in Experimental Autoimmune Encephalomyelitis and Multiple Sclerosis. Cold Spring Harb. Perspect. Med. 2018, 8, a029637. [Google Scholar] [CrossRef]
- Waisman, A.; Johann, L. Antigen-presenting cell diversity for T cell reactivation in central nervous system autoimmunity. J. Mol. Med. 2018, 96, 1279–1292. [Google Scholar] [CrossRef]
- Kaskow, B.J.; Baecher-Allan, C. Effector T Cells in Multiple Sclerosis. Cold Spring Harb. Perspect. Med. 2018, 8, a029025. [Google Scholar] [CrossRef]
- Miljković, D.; Spasojević, I. Multiple sclerosis: Molecular mechanisms and therapeutic opportunities. Antioxid. Redox Signal. 2013, 19, 2286–2334. [Google Scholar] [CrossRef]
- Cross, A.H.; Manning, P.T.; Stern, M.K.; Misko, T.P. Evidence for the production of peroxynitrite in inflammatory CNS demyelination. J. Neuroimmunol. 1997, 80, 121–130. [Google Scholar] [CrossRef]
- Lan, M.; Tang, X.; Zhang, J.; Yao, Z. Insights in pathogenesis of multiple sclerosis: Nitric oxide may induce mitochondrial dysfunction of oligodendrocytes. Rev. Neurosci. 2018, 29, 39–53. [Google Scholar] [CrossRef]
- Miljković, D.; Momčilović, M.; Stanojević, Z.; Rašić, D.; Mostarica-Stojković, M. It is still not for the old iron: Adjuvant effects of carbonyl iron in experimental autoimmune encephalomyelitis induction. J. Neurochem. 2011, 118, 205–214. [Google Scholar] [CrossRef] [PubMed]
- Petković, F.; Blaževski, J.; Momčilović, M.; Mostarica Stojkovic, M.; Miljković, D. Nitric oxide inhibits CXCL12 expression in neuroinflammation. Immunol. Cell Biol. 2013, 91, 427–434. [Google Scholar] [CrossRef] [PubMed]
- Cowden, W.B.; Cullen, F.A.; Staykova, M.A.; Willenborg, D.O. Nitric oxide is a potential down-regulating molecule in autoimmune disease: Inhibition of nitric oxide production renders PVG rats highly susceptible to EAE. J. Neuroimmunol. 1998, 88, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Gold, D.P.; Schroder, K.; Powell, H.C.; Kelly, C.J. Nitric oxide and the immunomodulation of experimental allergic encephalomyelitis. Eur. J. Immunol. 1997, 27, 2863–2869. [Google Scholar] [CrossRef]
- O’Loughlin, E.; Madore, C.; Lassmann, H.; Butovsky, O. Microglial Phenotypes and Functions in Multiple Sclerosis. Cold Spring Harb. Perspect. Med. 2018, 8, a028993. [Google Scholar] [CrossRef]
- Shemer, A.; Jung, S. Differential roles of resident microglia and infiltrating monocytes in murine CNS autoimmunity. Semin. Immunopathol. 2015, 37, 613–623. [Google Scholar] [CrossRef]
- BenSaad, L.A.; Kim, K.H.; Quah, C.C.; Kim, W.R.; Shahimi, M. Anti-inflammatory potential of ellagic acid, gallic acid and punicalagin A&B isolated from Punica granatum. BMC Complement. Altern. Med. 2017, 17, 47. [Google Scholar]
- Jeon, W.; Park, S.J.; Kim, B.C. n-Propyl gallate suppresses lipopolysaccharide-induced inducible nitric oxide synthase activation through protein kinase Cδ-mediated up-regulation of heme oxygenase-1 in RAW264.7 macrophages. Eur. J. Pharmacol. 2017, 801, 86–94. [Google Scholar] [CrossRef]
- Correa, L.B.; Pádua, T.A.; Seito, L.N.; Costa, T.E.; Silva, M.A.; Candéa, A.L.; Rosas, E.C.; Henriques, M.G. Anti-inflammatory Effect of Methyl Gallate on Experimental Arthritis: Inhibition of Neutrophil Recruitment, Production of Inflammatory Mediators, and Activation of Macrophages. J. Nat. Prod. 2016, 79, 1554–1566. [Google Scholar] [CrossRef]
- Park, P.H.; Hur, J.; Kim, Y.C.; An, R.B.; Sohn, D.H. Involvement of heme oxygenase-1 induction in inhibitory effect of ethyl gallate isolated from Galla Rhois on nitric oxide production in RAW 264.7 macrophages. Arch. Pharm. Res. 2011, 34, 1545–1552. [Google Scholar] [CrossRef]
- Mehta, A.K.; Gracias, D.T.; Croft, M. TNF activity and T cells. Cytokine 2018, 101, 14–18. [Google Scholar] [CrossRef] [PubMed]
- Salomon, B.L.; Leclerc, M.; Tosello, J.; Ronin, E.; Piaggio, E.; Cohen, J.L. Tumor Necrosis Factor α and Regulatory T Cells in Oncoimmunology. Front. Immunol. 2018, 9, 444. [Google Scholar] [CrossRef] [PubMed]
- Agbanoma, G.; Li, C.; Ennis, D.; Palfreeman, A.C.; Williams, L.M.; Brennan, F.M. Production of TNF-α in macrophages activated by T cells, compared with lipopolysaccharide, uses distinct IL-10-dependent regulatory mechanism. J. Immunol. 2012, 188, 1307–1317. [Google Scholar] [CrossRef]
- Hubbard, J.S.; Chen, P.H.; Boyd, K.L. Effects of Repeated Intraperitoneal Injection of Pharmaceutical-grade and Nonpharmaceutical-grade Corn Oil in Female C57BL/6J Mice. J. Am. Assoc. Lab. Anim. Sci. 2017, 56, 779–785. [Google Scholar] [PubMed]
- Lazarević, M.; Djedovic, N.; Stanisavljević, S.; Dimitrijević, M.; Stegnjaić, G.; Krishnamoorthy, G.; Mostarica Stojković, M.; Miljković, Đ.; Jevtić, B. Complete Freund’s adjuvant-free experimental autoimmune encephalomyelitis in Dark Agouti rats is a valuable tool for multiple sclerosis studies. J. Neuroimmunol. 2021, 354, 577547. [Google Scholar] [CrossRef] [PubMed]
- Esser, C.; Rannug, A. The aryl hydrocarbon receptor in barrier organ physiology, immunology, and toxicology. Pharmacol. Rev. 2015, 67, 259–279. [Google Scholar] [CrossRef] [PubMed]
- Esplugues, E.; Huber, S.; Gagliani, N.; Hauser, A.E.; Town, T.; Wan, Y.Y.; O’Connor, W., Jr.; Rongvaux, A.; Van Rooijen, N.; Haberman, A.M.; et al. Control of TH17 cells occurs in the small intestine. Nature 2011, 475, 514–518. [Google Scholar] [CrossRef]
- Lee, Y.K.; Menezes, J.S.; Umesaki, Y.; Mazmanian, S.K. Proinflammatory T-cell responses to gut microbiota promote experimental autoimmune encephalomyelitis. Proc. Natl. Acad. Sci. USA 2011, 108 (Suppl. 1), 4615–4622. [Google Scholar] [CrossRef]
- Miljković, Đ.; Jevtić, B.; Stojanović, I.; Dimitrijević, M. ILC3, a Central Innate Immune Component of the Gut-Brain Axis in Multiple Sclerosis. Front. Immunol. 2021, 12, 657622. [Google Scholar] [CrossRef]
- Ben-Nun, A.; Kaushansky, N.; Kawakami, N.; Krishnamoorthy, G.; Berer, K.; Liblau, R.; Hohlfeld, R.; Wekerle, H. From classic to spontaneous and humanized models of multiple sclerosis: Impact on understanding pathogenesis and drug development. J. Autoimmun. 2014, 54, 33–50. [Google Scholar] [CrossRef]
- Lavrnja, I.; Smiljanic, K.; Savic, D.; Mladenovic-Djordjevic, A.; Tesovic, K.; Kanazir, S.; Pekovic, S. Expression profiles of cholesterol metabolism-related genes are altered during development of experimental autoimmune encephalomyelitis in the rat spinal cord. Sci. Rep. 2017, 7, 2702. [Google Scholar] [CrossRef] [PubMed]
- Miljković, D.; Blaževski, J.; Petković, F.; Djedović, N.; Momčilović, M.; Stanisavljević, S.; Jevtić, B.; Mostarica Stojković, M.; Spasojević, I. A comparative analysis of multiple sclerosis-relevant anti-inflammatory properties of ethyl pyruvate and dimethyl fumarate. J. Immunol. 2015, 194, 2493–2503. [Google Scholar] [CrossRef] [PubMed]


Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Stegnjaić, G.; Tsiailanis, A.D.; Lazarević, M.; Gkalpinos, V.K.; Djedovic, N.; Antoniou, T.; Stanisavljević, S.; Dimitrijević, M.; Momčilović, M.; Miljković, Đ.; et al. Phenethyl Ester of Gallic Acid Ameliorates Experimental Autoimmune Encephalomyelitis. Molecules 2022, 27, 8770. https://doi.org/10.3390/molecules27248770
Stegnjaić G, Tsiailanis AD, Lazarević M, Gkalpinos VK, Djedovic N, Antoniou T, Stanisavljević S, Dimitrijević M, Momčilović M, Miljković Đ, et al. Phenethyl Ester of Gallic Acid Ameliorates Experimental Autoimmune Encephalomyelitis. Molecules. 2022; 27(24):8770. https://doi.org/10.3390/molecules27248770
Chicago/Turabian StyleStegnjaić, Goran, Antonios D. Tsiailanis, Milica Lazarević, Vasileios K. Gkalpinos, Neda Djedovic, Thomas Antoniou, Suzana Stanisavljević, Mirjana Dimitrijević, Miljana Momčilović, Đorđe Miljković, and et al. 2022. "Phenethyl Ester of Gallic Acid Ameliorates Experimental Autoimmune Encephalomyelitis" Molecules 27, no. 24: 8770. https://doi.org/10.3390/molecules27248770
APA StyleStegnjaić, G., Tsiailanis, A. D., Lazarević, M., Gkalpinos, V. K., Djedovic, N., Antoniou, T., Stanisavljević, S., Dimitrijević, M., Momčilović, M., Miljković, Đ., Tzakos, A. G., & Jevtić, B. (2022). Phenethyl Ester of Gallic Acid Ameliorates Experimental Autoimmune Encephalomyelitis. Molecules, 27(24), 8770. https://doi.org/10.3390/molecules27248770







