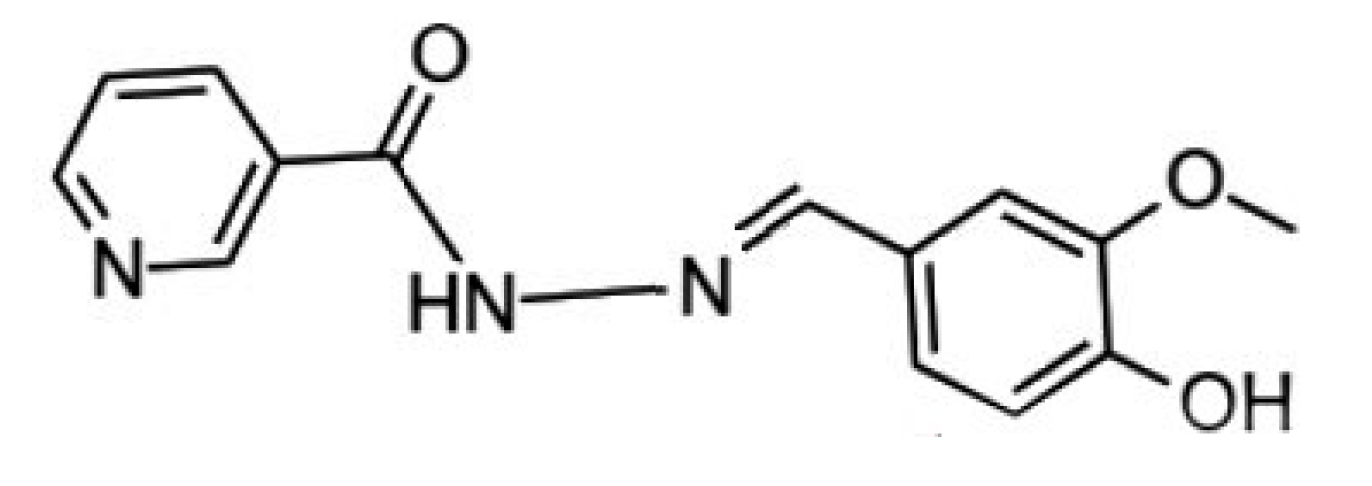
Experimental and Quantum Chemical Investigations on the Anticorrosion Efficiency of a Nicotinehydrazide Derivative for Mild Steel in HCl
Abstract
:1. Introduction
2. Results and Discussion
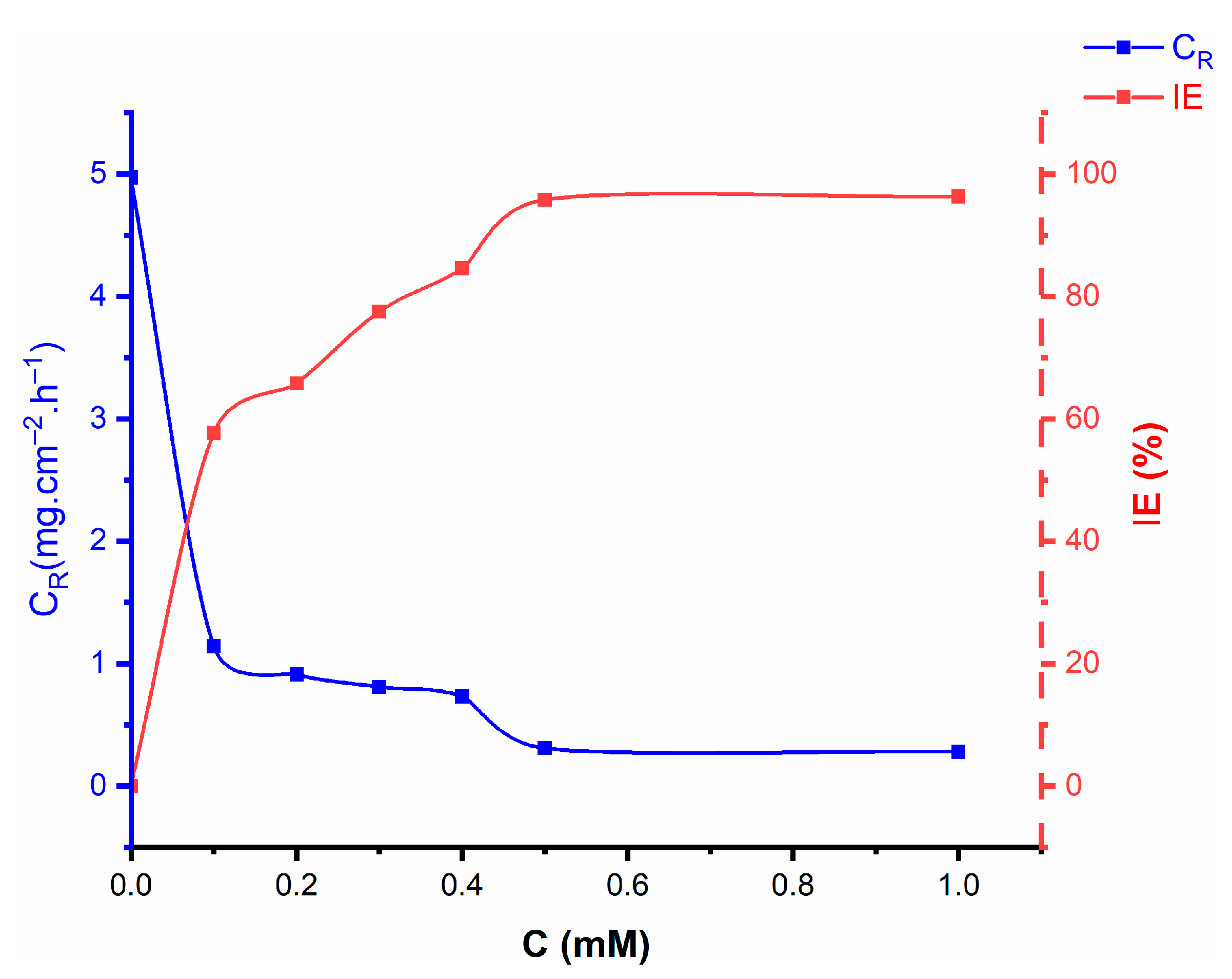
2.1. Weight Loss Investigations
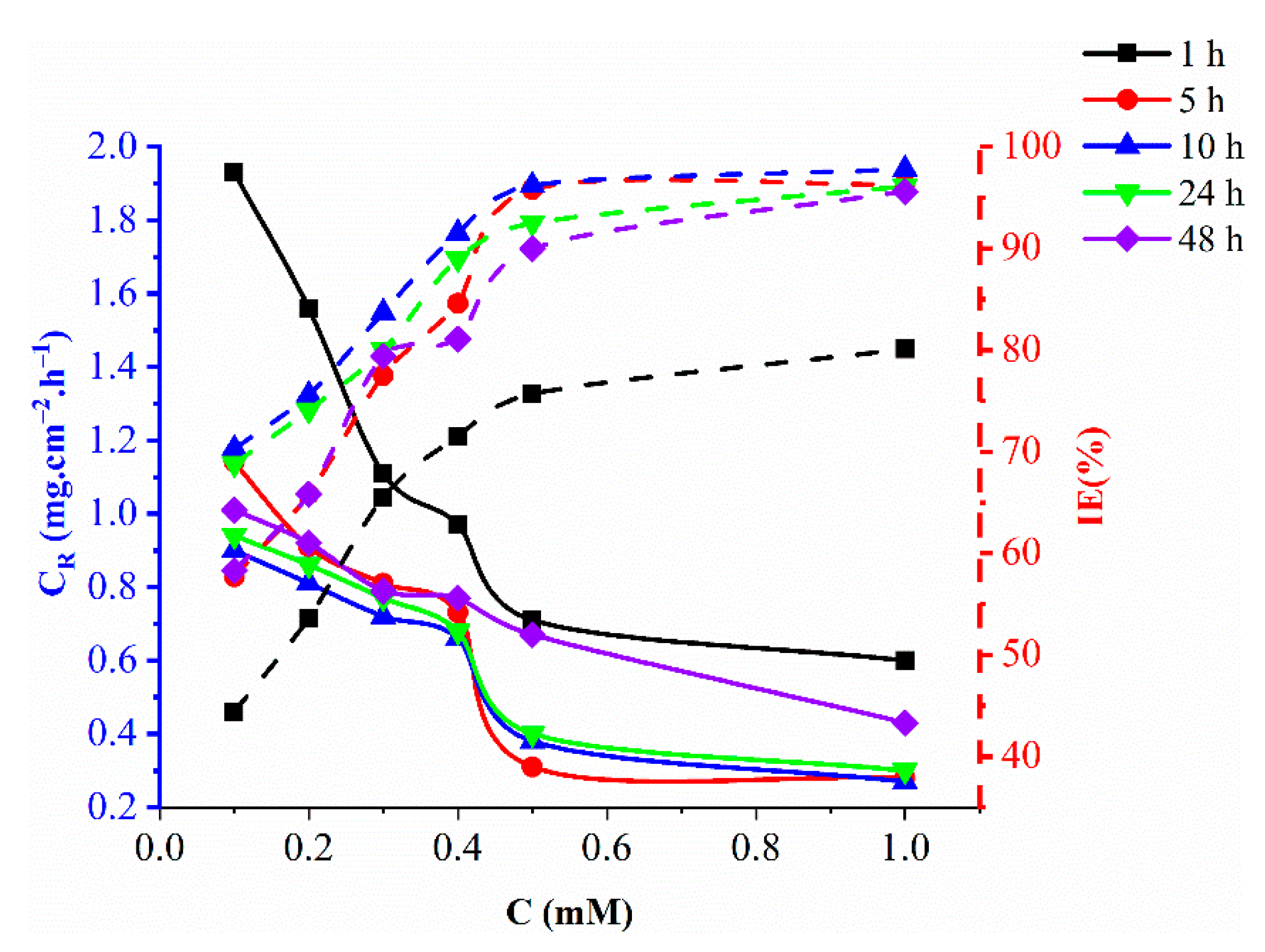
2.2. The Effect of Exposure Time
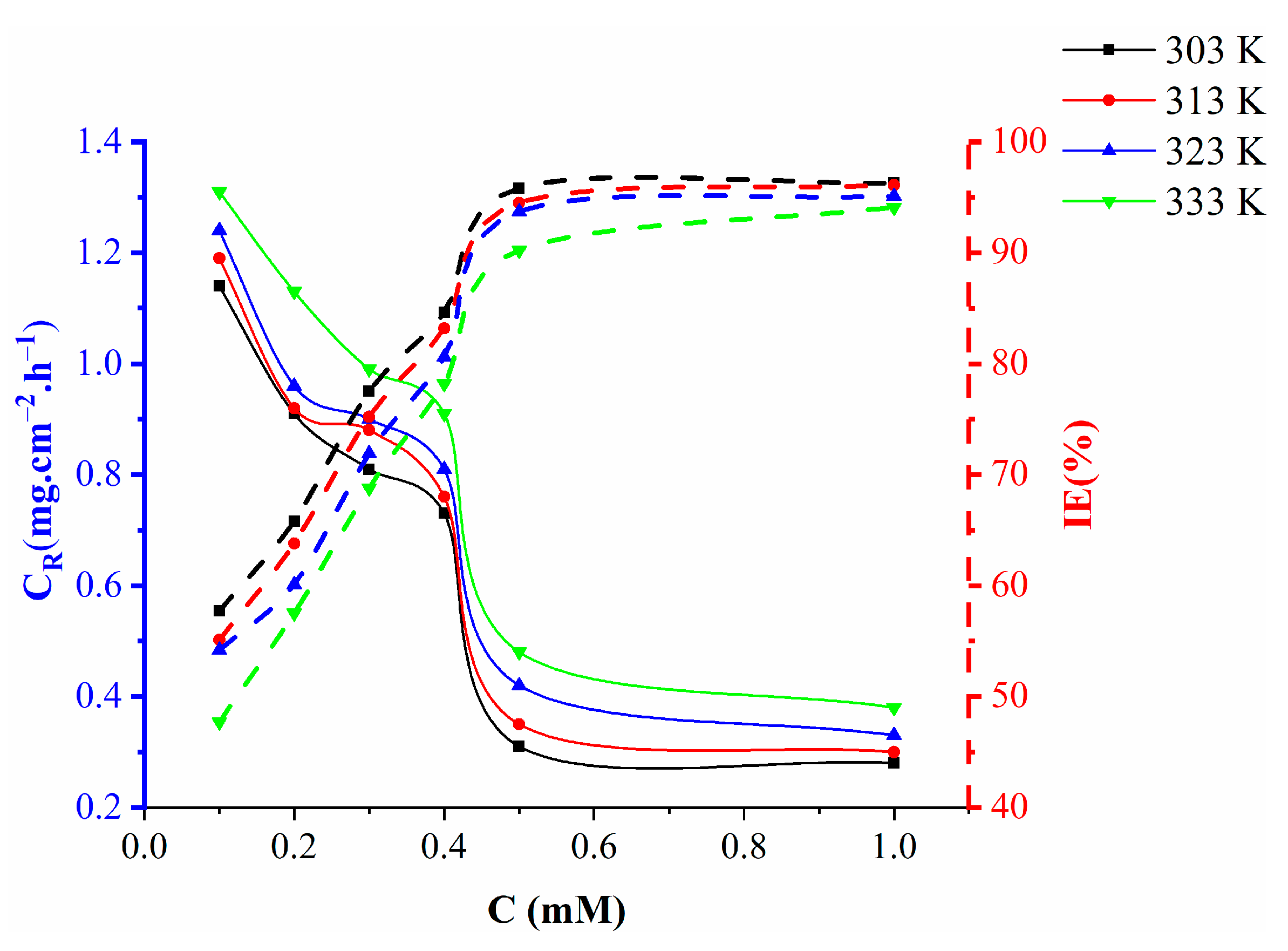
2.3. The Effect of Temperature
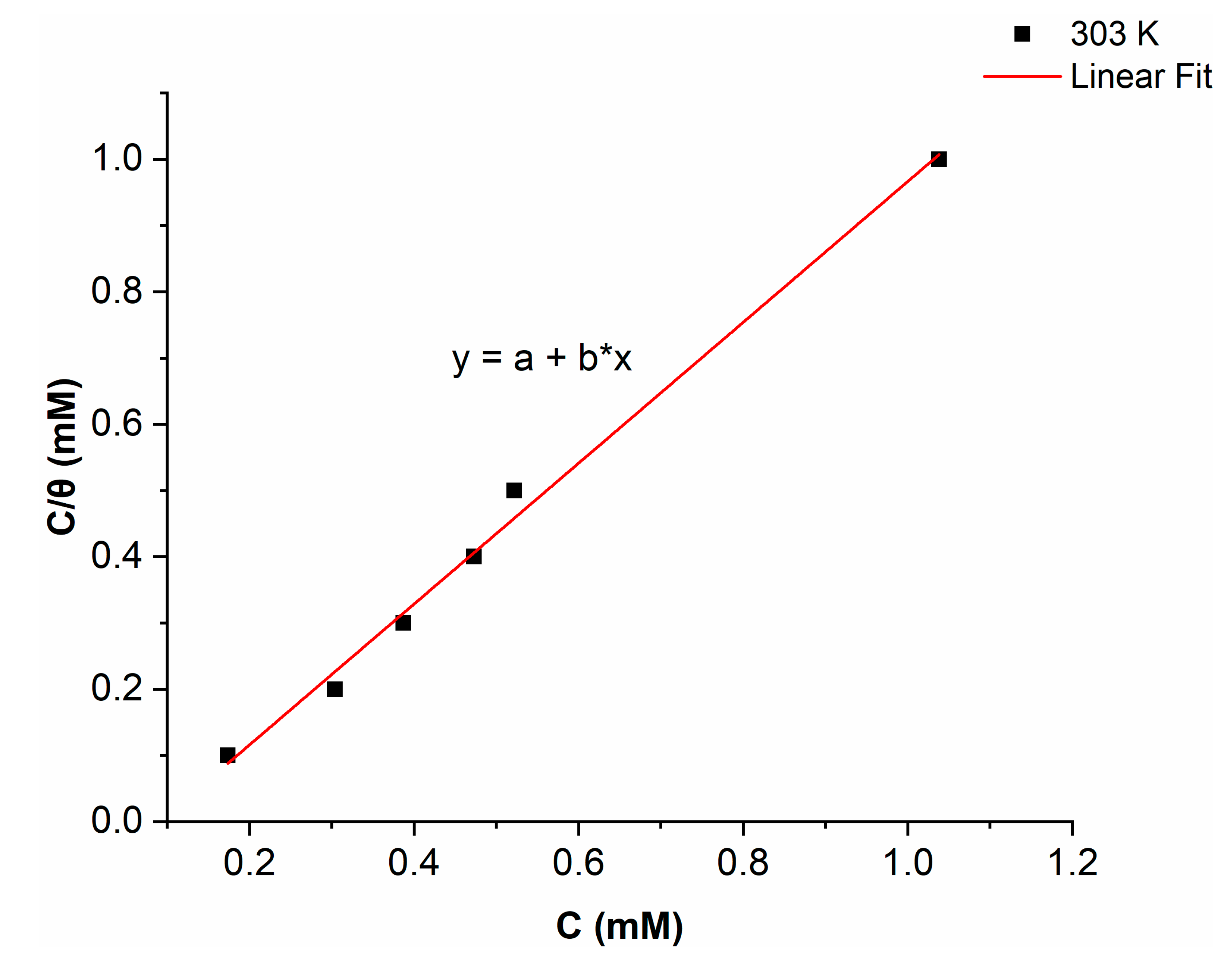
2.4. Adsorption Isotherm
2.5. Potentiodynamic Polarisation Measurements
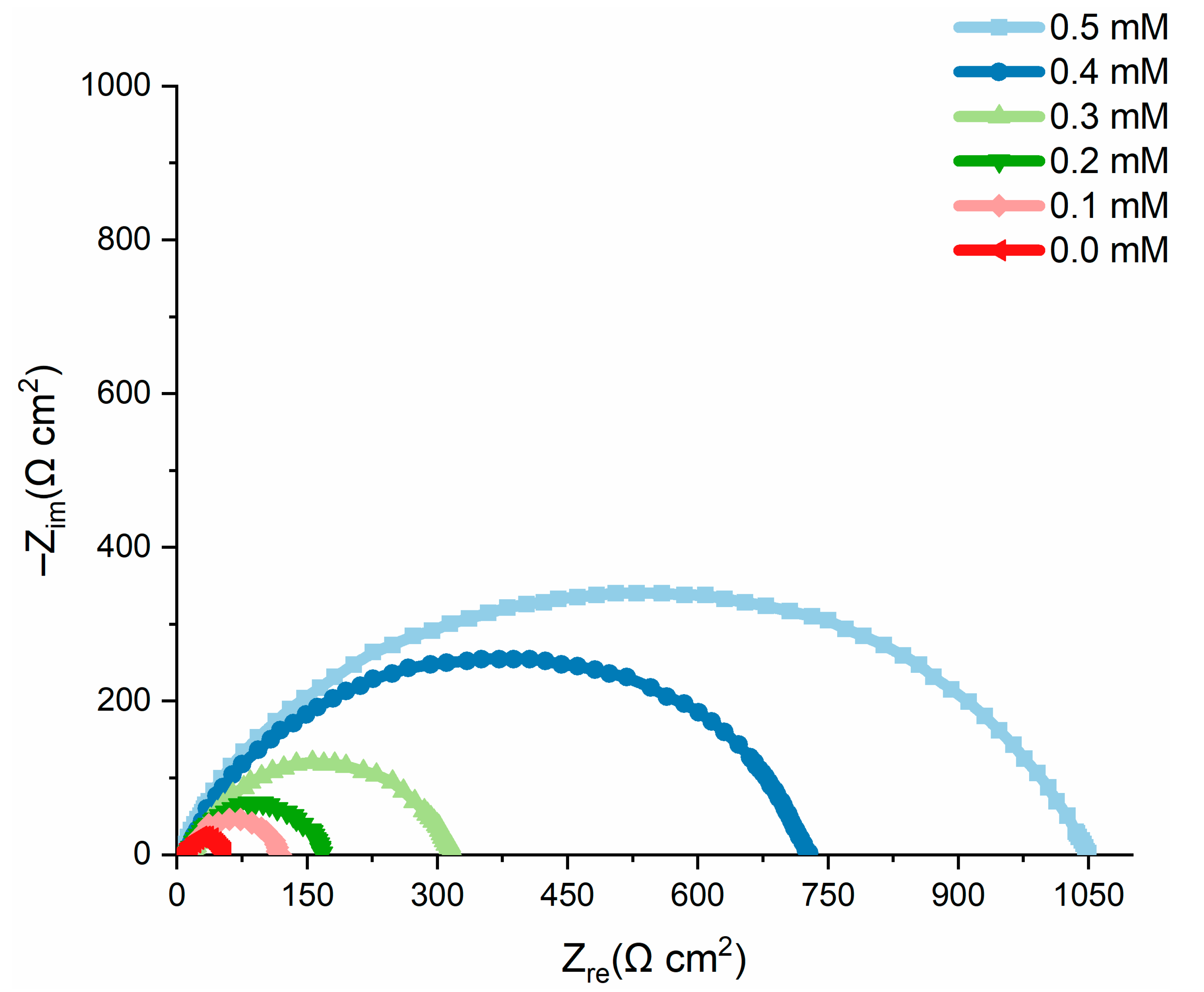
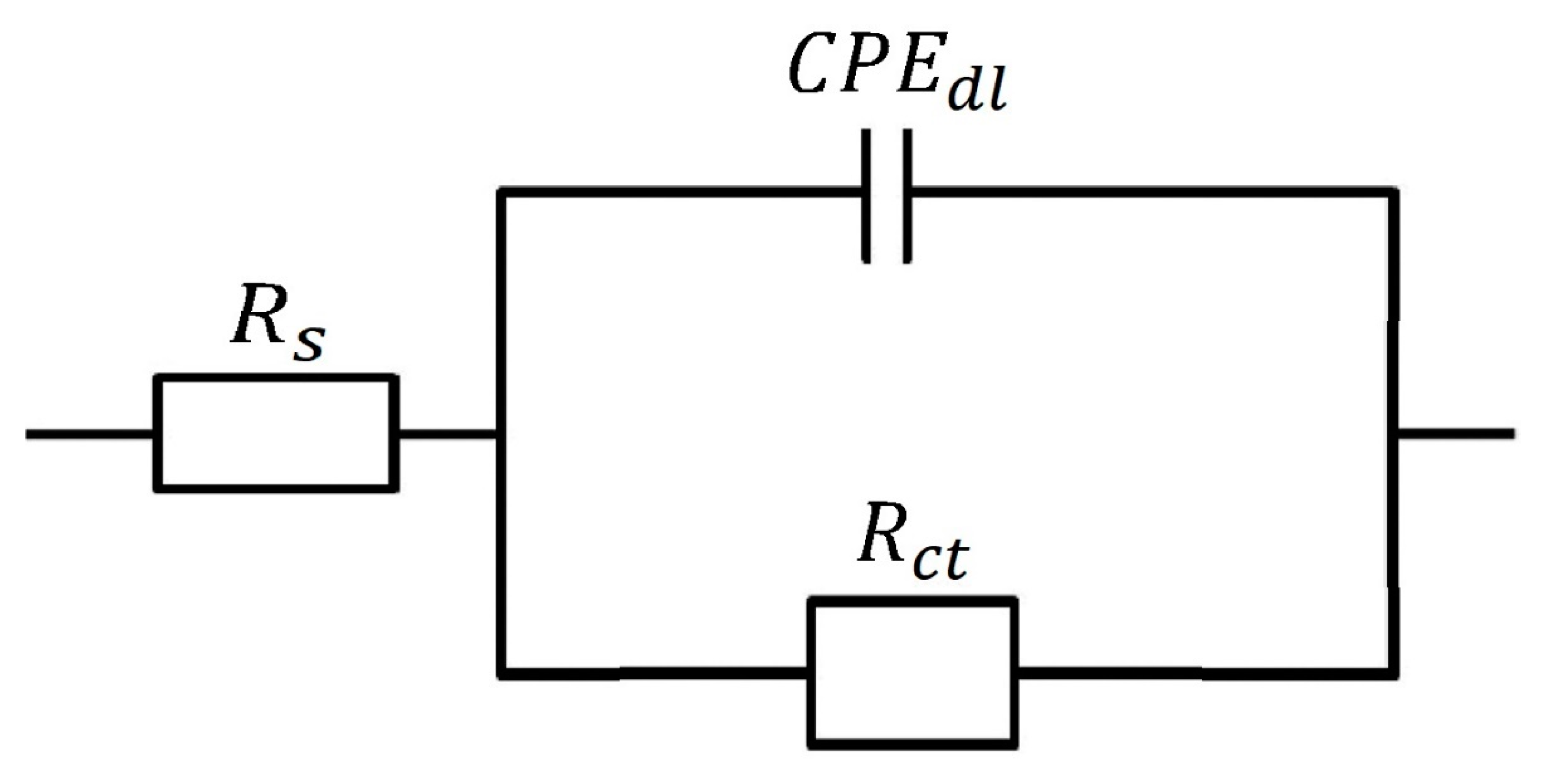
2.6. Electrochemical Measurements
2.7. Surface Morphology
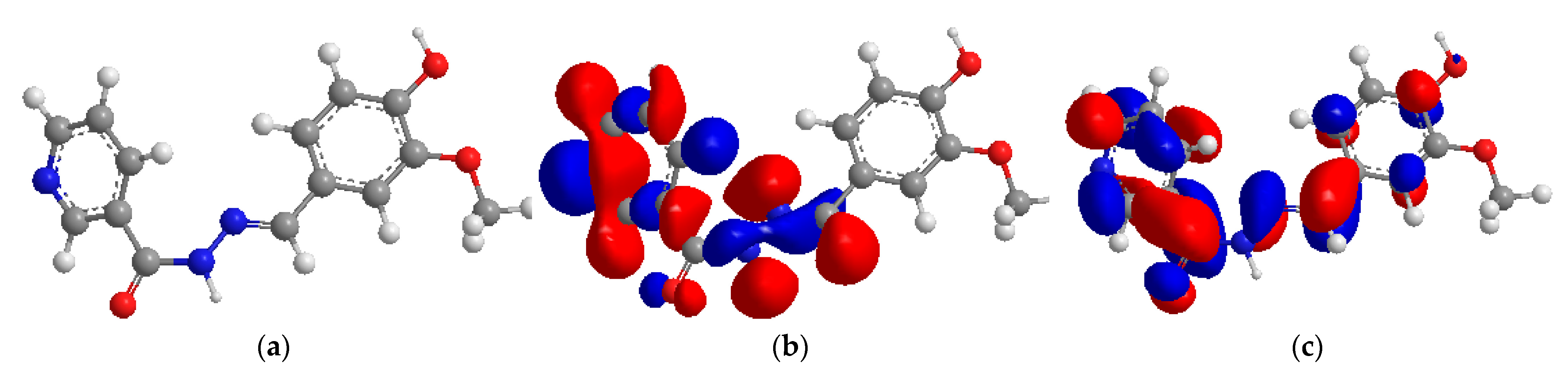
2.8. Theoretical Calculation
2.9. Mulliken Atomic Charges
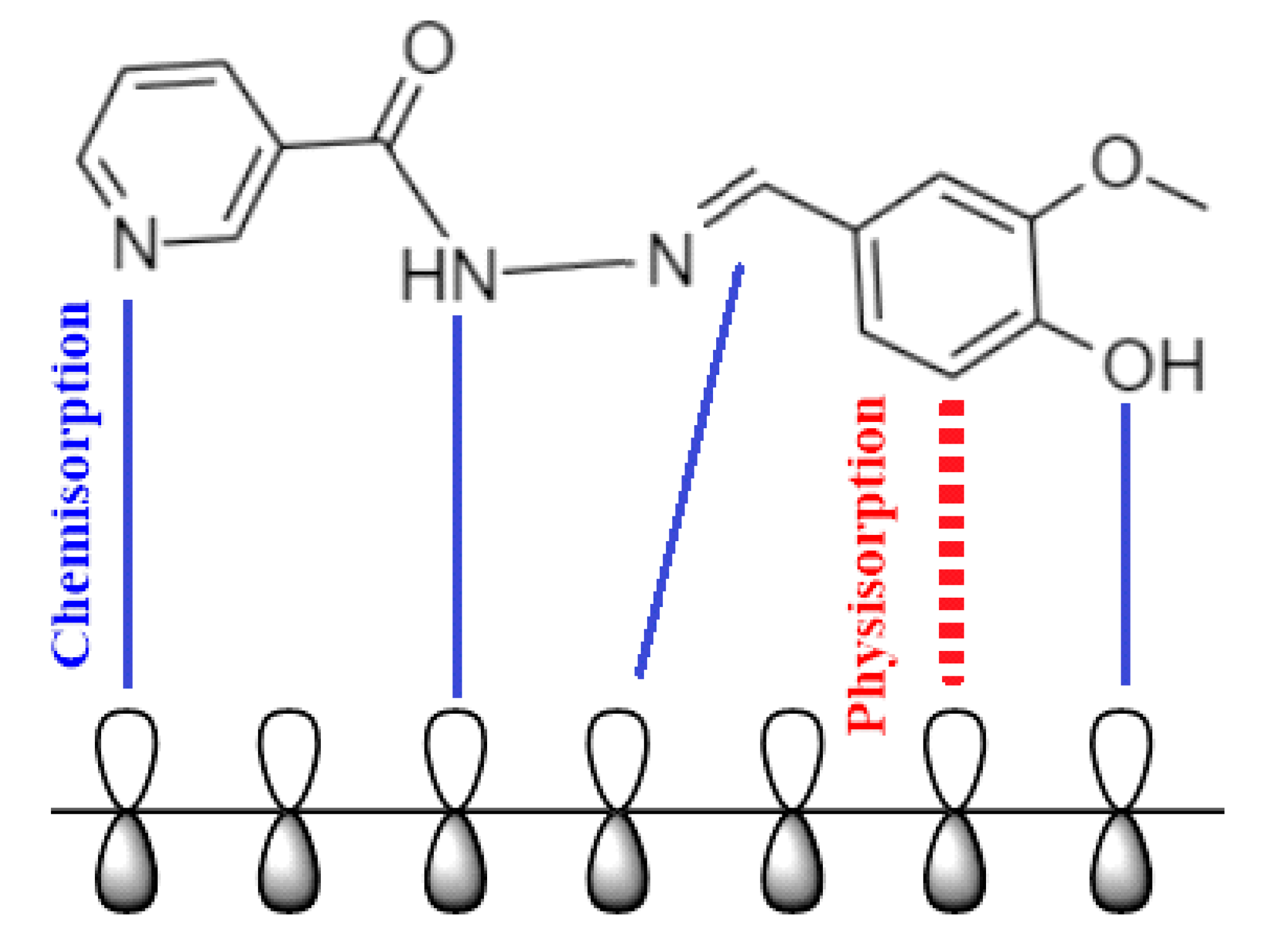
2.10. Mechanism of Inhibition
3. Materials and Methods
3.1. Materials
3.2. Weight Loss Techniques
3.3. Computations
3.4. Electrochemical Data
3.5. Surface Scanning Electron Microscope
4. Conclusions
- The synthesized nicotinehydrazide derivative shows good inhibition efficiency for the mild steel corrosion in 1 M HCl environment and the inhibition efficiency increases on increasing the concentration of nicotinehydrazide and decreases with the increase in temperature. The highest inhibition efficiency was 97% at 303 K in 1 M HCl solution.
- Nicotinehydrazide participates in chemical adsorption on metallic surfaces and weakly bonds to the metal surface with the inhibition efficacy decreasing as the temperature increases.
- Nicotinehydrazide prevents mild steel corrosion due to the creation of a protective layer of inhibitor molecules at the steel–electrolyte interface.
- The Gads suggests a chemisorption and physisorption phenomena, and the adsorption mechanism is spontaneous.
- The quantum chemical simulations indicate that nicotinehydrazide uses oxygen and nitrogen to adsorb onto a mild steel surface.
- There was good agreement between the experimental results and the theoretical analysis.
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Sample Availability
References
- Guo, L.; Kaya, S.; Obot, I.; Zheng, X.; Qiang, Y. Toward understanding the anticorrosive mechanism of some thiourea derivatives for carbon steel corrosion: A combined DFT and molecular dynamics investigation. J. Colloid Interface Sci. 2017, 506, 478–485. [Google Scholar] [CrossRef] [PubMed]
- Hanoon, M.M.; Resen, A.M.; Shaker, L.M.; Kadhum, A.A.H.; Al-Amiery, A.A. Corrosion investigation of mild steel in aqueous hydrochloric acid environment using n-(Naphthalen-1yl)-1-(4-pyridinyl)methanimine complemented with antibacterial studies. Biointerface Res. Appl. Chem. 2021, 11, 9735–9743. [Google Scholar]
- Al-Baghdadi, S.; Gaaz, T.S.; Al-Adili, A.; Al-Amiery, A.A.; Takriff, M.S. Experimental studies on corrosion inhibition performance of acetylthiophene thiosemicarbazone for mild steel in HCl complemented with DFT investigation. Int. J. Low-Carbon Technol. 2021, 16, 181–188. [Google Scholar] [CrossRef]
- Al-Amiery, A.A. Anti-corrosion performance of 2-isonicotinoyl-n-phenylhydrazinecarbothioamide for mild steel hydrochloric acid solution: Insights from experimental measurements and quantum chemical calculations. Surf. Rev. Lett. 2021, 28, 2050058. [Google Scholar] [CrossRef]
- Mahdi, B.S.; Abbass, M.K.; Mohsin, M.K.; Al-azzawi, W.K.; Hanoon, M.M.; Al-kaabi, M.H.H.; Shaker, L.M.; Al-amiery, A.A.; Isahak, W.N.R.W.; Kadhum, A.A.H.; et al. Corrosion Inhibition of Mild Steel in Hydrochloric Acid Environment Using Terephthaldehyde Based on Schiff Base: Gravimetric, Thermodynamic, and Computational Studies. Molecules 2022, 27, 4857. [Google Scholar] [CrossRef] [PubMed]
- Al-Baghdadi, S.B.; Al-Amiery, A.A.; Gaaz, T.S.; Kadhum, A.A.H. Terephthalohydrazide and isophthalo-hydrazide as new corrosion inhibitors for mild steel in hydrochloric acid: Experimental and theoretical approaches. Koroze Ochr. Mater. 2021, 65, 12–22. [Google Scholar] [CrossRef]
- Koch, G.H.; Brongers, M.P.; Thompson, N.G.; Virmani, Y.P.; Payer, J. Corrosion Cost and Preventive Strategies in the United States; Federal Highway Administration: Washington, DC, USA, 2002. [Google Scholar]
- De Damborenea, J.; Conde, A.; Arenas, M. Corrosion Inhibition with Rare Earth Metal Compounds in Aqueous Solutions, Rare Earth-Based Corrosion Inhibitors; Elsevier: Amsterdam, The Netherlands, 2014; pp. 84–116. [Google Scholar]
- Verma, C.; Obot, I.B.; Bahadur, I.; Sherif, E.-S.M.; Ebenso, E.E. Choline based ionic liquids as sustainable corrosion inhibitors on mild steel surface in acidic medium: Gravimetric, electrochemical, surface morphology, DFT and Monte Carlo simulation studies. Appl. Surf. Sci. 2018, 457, 134–149. [Google Scholar] [CrossRef]
- Obot, I.B.; Obi-Egbedi, N.O.; Eseola, A.O. Anticorrosion Potential of 2-Mesityl-1H-imidazo [4, 5-f] [1, 10] phenanthroline on Mild Steel in Sulfuric Acid Solution: Experimental and Theoretical Study. Ind. Eng. Chem. Res. 2011, 50, 2098–2110. [Google Scholar] [CrossRef]
- Aziz, I.A.A.; Abdulkareem, M.H.; Annon, I.A.; Hanoon, M.M.; Al-Kaabi, M.H.H.; Shaker, L.M.; Alamiery, A.A.; Isahak, W.N.R.W.; Takriff, M.S. Weight Loss, Thermodynamics, SEM, and Electrochemical Studies on N-2-Methylbenzylidene-4-antipyrineamine as an Inhibitor for Mild Steel Corrosion in Hydrochloric Acid. Lubricants 2022, 10, 23. [Google Scholar] [CrossRef]
- Antonijevic, M.M.; Petrovic, M.B. Copper corrosion inhibitors. A review. Int. J. Electrochem. Sci. 2008, 3, 1–28. [Google Scholar]
- Tan, T.; Lan, W.; Zhang, S.; Deng, H.; Qiang, Y.; Fu, A.; Ran, Y.; Xiong, J.; Marzouki, R.; Li, W. Passiflora edulia Sims leaves Extract as renewable and degradable inhibitor for copper in sulfuric acid solution. Colloids Surf. A Physicochem. Eng. Asp. 2022, 645, 128892. [Google Scholar] [CrossRef]
- Verma, C.; Lgaz, H.; Verma, D.; Ebenso, E.E.; Bahadur, I.; Quraishi, M. Molecular dynamics and Monte Carlo simulations as powerful tools for study of interfacial adsorption behavior of corrosion inhibitors in aqueous phase: A review. J. Mol. Liq. 2018, 260, 99–120. [Google Scholar] [CrossRef]
- Chile, N.E.; Haldhar, R.; Godffrey, U.K.; Chijioke, O.C.; Umezuruike, E.A.; Ifeoma, O.P.; Oke, M.O.; Ichou, H.; Arrousse, N.; Kim, S.-C.; et al. Theoretical Study and Adsorption Behavior of Urea on Mild Steel in Automotive Gas Oil (AGO) Medium. Lubricants 2022, 10, 157. [Google Scholar] [CrossRef]
- Mishra, A.; Verma, C.; Lgaz, H.; Srivastava, V.; Quraishi, M.A.; Ebenso, E.E. Synthesis, characterization and corrosion inhibition studies of N-phenyl-benzamides on the acidic corrosion of mild steel: Experimental and computational studies. J. Mol. Liq. 2018, 251, 317–332. [Google Scholar] [CrossRef]
- Fernine, Y.; Arrousse, N.; Haldhar, R.; Merzouki, O.; Dalbouha, S.; Mabrouk, E.H.; Hajjaji, F.E.L.; Touhami, M.E.; Kim, S.-C.; Taleb, M. Economical, efficient, and environmentally friendly synthesis strategy of O-Alkylation strategy based on phenolphthalein reactions with electrophiles: Characterization, DFT study, and molecular docking. J. Mol. Liq. 2022, 1265, 133424. [Google Scholar] [CrossRef]
- Mrani, S.A.; Arrousse, N.; Haldhar, R.; Lahcen, A.A.; Amine, A.; Saffaj, T.; Kim, S.-C.; Taleb, M. In Silico Approaches for Some Sulfa Drugs as Eco-Friendly Corrosion Inhibitors of Iron in Aqueous Medium. Lubricants 2022, 10, 43. [Google Scholar] [CrossRef]
- Ferreira, E.S.; Giacomelli, C.; Giacomelli, F.C.; Spinelli, A. Evaluation of the inhibitor effect of L-ascorbic acid on the corrosion of mild steel. Mater. Chem. Phys. 2004, 83, 129–134. [Google Scholar] [CrossRef]
- Saliyan, V.R.; Adhikari, A.V. Quinolin-5-ylmethylene-3-{[8-(trifluoromethyl)quinolin-4-yl]thio}propanohydrazide as an effective inhibitor of mild steel corrosion in HCl solution. Corros. Sci. 2008, 50, 55–61. [Google Scholar] [CrossRef]
- Pardo, A.; Merino, M.C.; Arrabal, R.; Feliu, S., Jr.; Viejo, F.; Carboneras, M. Enhanced corrosion resistance of A3xx.x/SiCp composites in chloride media by La surface treatments. Electrochim. Acta 2006, 51, 4367–4378. [Google Scholar] [CrossRef]
- Popova, A.; Christov, M. Evaluation of impedance measurements on mild steel corrosion in acidmedia in the presence of heterocyclic compounds. Corros. Sci. 2005, 48, 3208–3221. [Google Scholar] [CrossRef]
- Tan, B.; Zhang, S.; Cao, X.; Fuc, A.; Guo, L.; Marzouki, R.; Li, W. Insight into the anti-corrosion performance of two food flavors as eco-friendly and ultra-high performance inhibitors for copper in sulfuric acid medium. J. Colloid Interface Sci. 2022, 609, 838–851. [Google Scholar] [CrossRef] [PubMed]
- Khaled, K.F. The inhibition of benzimidazole derivatives on corrosion of iron in 1 M HClsolutions. Electrochim. Acta 2003, 48, 2493–2503. [Google Scholar] [CrossRef]
- Hleli, S.; Abdelghani, A.; Tlili, A. Impedance spectroscopy technique for DNA hybridization. Sensors 2003, 3, 472–479. [Google Scholar] [CrossRef]
- Alkadir Aziz, I.A.; Annon, I.A.; Abdulkareem, M.H.; Hanoon, M.M.; Alkaabi, M.H.; Shaker, L.M.; Alamiery, A.A.; Wan Isahak, W.N.R.; Takriff, M.S. Insights into Corrosion Inhibition Behavior of a 5-Mercapto-1, 2, 4-triazole Derivative for Mild Steel in Hydrochloric Acid Solution: Experimental and DFT Studies. Lubricants 2021, 9, 122. [Google Scholar] [CrossRef]
- Bonatti, R.S.; Meyer, Y.A.; Padilha, P.G.; Bortolozo, P.A.; Osório, W.R. Silicon Content Affecting Corrosion Behavior of Alp/Sip Composites in a Biodiesel Blend. Corrosion 2020, 76, 1109–1121. [Google Scholar] [CrossRef]
- Verma, C.; Olasunkanmi, L.O.; Ebenso, E.E.; Quraishi, M.A.; Obot, I.B. Adsorption Behavior of Glucosamine-Based, Pyrimidine-Fused Heterocycles as Green Corrosion Inhibitors for Mild Steel: Experimental and Theoretical Studies. J. Phys. Chem. C 2016, 120, 11598–11611. [Google Scholar] [CrossRef]
- Singh, P.; Quraishi, M.A. Corrosion inhibition of mild steel using Novel Bis Schiff’s Bases as corrosion inhibitors: Electrochemical and Surface measurement. Measurement 2016, 86, 114–124. [Google Scholar] [CrossRef]
- Alamiery, A.A.; Wan Isahak, W.N.R.; Takriff, M.S. Inhibition of Mild Steel Corrosion by 4-benzyl-1-(4-oxo-4-phenylbutanoyl)thiosemicarbazide: Gravimetrical, Adsorption and Theoretical Studies. Lubricants 2021, 9, 93. [Google Scholar] [CrossRef]
- Ozcan, M.; Dehri, I.; Erbil, M. Organic sulphur-containing compounds as corrosion inhibitors for mild steel in acidic media: Correlation between inhibition efficiency and chemical structure. Appl. Surf. Sci. 2004, 236, 155–164. [Google Scholar] [CrossRef]
- Ozcan, M.; Dehri, I. Electrochemical and quantum chemical studies of some sulphur-containing organic compounds as inhibitors for the acid corrosion of mild steel. Prog. Org. Coat. 2004, 51, 181–187. [Google Scholar] [CrossRef]
- Sastri, V.S.; Perumareddi, J.R. Molecular orbital theoretical studies of some organic corrosion inhibitors. Corrosion 1997, 53, 617–622. [Google Scholar] [CrossRef]
- Assad, N.; Abdul-Hameed, F.; Yousif, E.; Balakit, A. Synthesis of Thiophene Derivative and its Use as Zinc Metal Corrosion Inhibitor in Hydrochloric Acid Solution. ANJS 2018, 18, 69–78. [Google Scholar] [CrossRef]
- Standard Practice for Preparing, Cleaning, and Evaluating Corrosion Test; ASTM International: West Conshohocken, PA, USA, 2011; G1-03; pp. 1–9.
- Jawad, Q.; Zinad, D.; Dawood Salim, R.; Al-Amiery, A.; Sumer Gaaz, T.; Takriff, M.S.; Kadhum, A.A. Synthesis, Characterization, and Corrosion Inhibition Potential of Novel Thiosemicarbazone on Mild Steel in Sulfuric Acid Environment. Coatings 2019, 9, 729. [Google Scholar] [CrossRef]
- Junaedi, S.; Al-Amiery, A.A.; Kadihum, A.; Kadhum, A.A.H.; Mohamad, A.B. Inhibition Effects of a Synthesized Novel 4-Aminoantipyrine Derivative on the Corrosion of Mild Steel in Hydrochloric Acid Solution together with Quantum Chemical Studies. Int. J. Mol. Sci. 2013, 14, 11915–11928. [Google Scholar] [CrossRef]
- Koopmans, T. Ordering of wave functions and eigenenergy’s to the individual electrons of an atom. Physica 1933, 1, 104–113. [Google Scholar] [CrossRef]
- Meyer, Y.A.; Menezes, I.; Bonatti, R.S.; Bortolozo, A.D.; Osório, W.R. EIS Investigation of the Corrosion Behavior of Steel Bars Embedded into Modified Concretes with Eggshell Contents. Metals 2022, 12, 417. [Google Scholar] [CrossRef]
- Zhang, X.L.; Jiang, Z.H.; Yao, Z.P.; Song, Y.; Wu, Z.D. Effects of scan rate on the potentiodynamic polarization curve obtained to determine the Tafel slopes and corrosion current density. Corros. Sci. 2009, 51, 581–587. [Google Scholar] [CrossRef]
- McCafferty, E. Validation of corrosion rates measured by Tafel extrapolation method. Corros. Sci. 2005, 47, 3202–3215. [Google Scholar] [CrossRef]
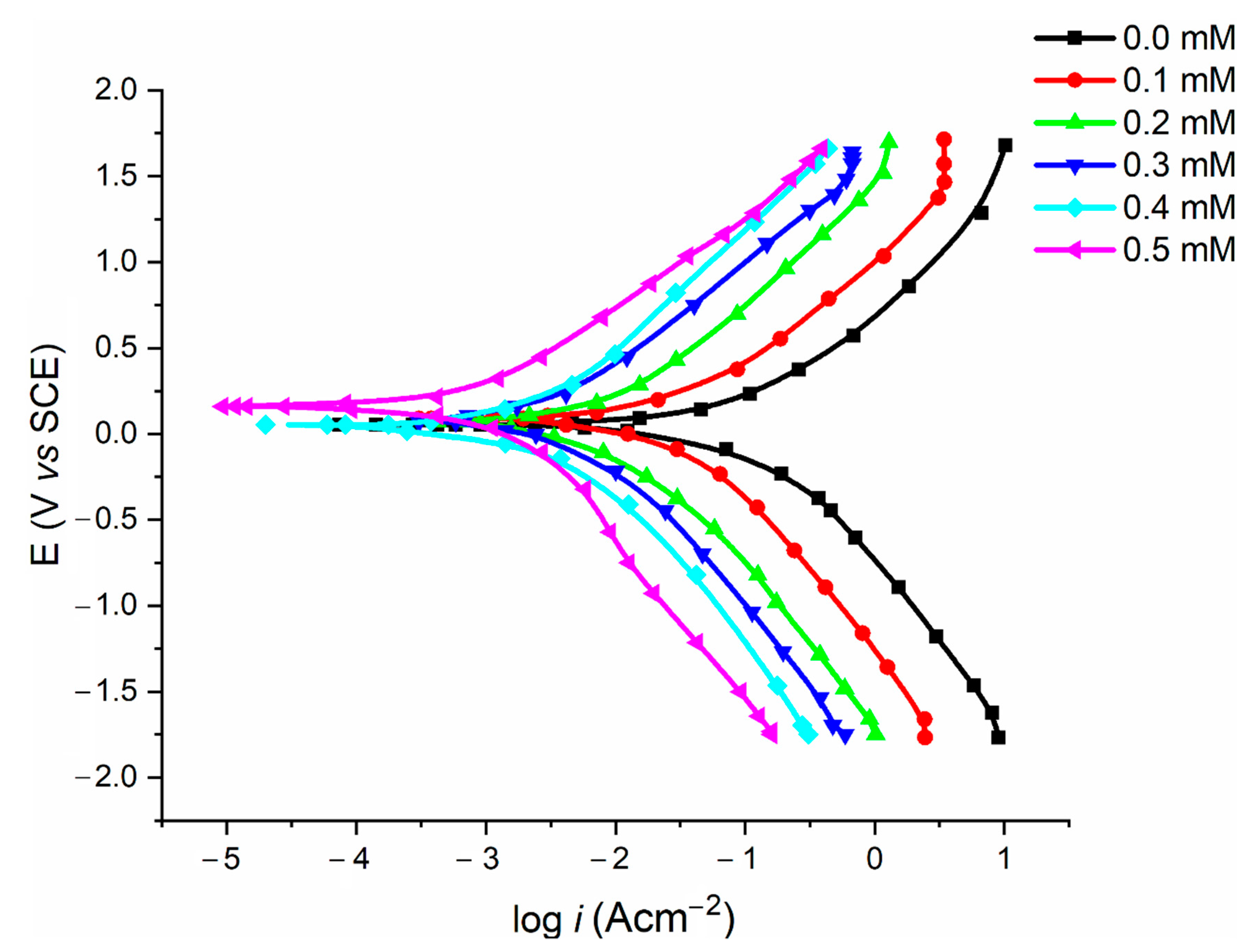

| Conc. mM | Ecorr (V) | βa (mV/dec) | βc (mV/dec) | icorr (μA·cm−2) | IE (%) |
|---|---|---|---|---|---|
| 0.0 | –0.47 | 240 | 220 | 515.3 ± 1.83 | 0 |
| 0.1 | –0.51 | 125.7 | 188.6 | 355.7 ± 5.03 | 73.6 |
| 0.2 | –0.49 | 91.8 | 151.8 | 199.3 ± 3.70 | 82.8 |
| 0.3 | –0.55 | 83.4 | 131.2 | 95.7 ± 2.93 | 86.4 |
| 0.4 | –0.53 | 56.7 | 126.7 | 84.8 ± 1.84 | 92.8 |
| 0.5 | –0.46 | 48.9 | 102.8 | 58.3 ± 4.77 | 97.1 |
| Conc. (mM) | Rs (Ω cm2) | Rct (Ω cm2) | Cdl (μF) | IE % |
|---|---|---|---|---|
| 0.0 | 2.4 | 50.64 | 530 | 0 |
| 0.1 | 2.1 | 78.65 | 310 | 55.2 |
| 0.2 | 2.3 | 172.37 | 311 | 72.8 |
| 0.3 | 2.2 | 280.76 | 244 | 84.8 |
| 0.4 | 2.4 | 327.33 | 180 | 92.5 |
| 0.5 | 2.3 | 466.13 | 130 | 96.8 |
| EHOMO (eV) | ELUMO (eV) | ΔE (eV) | I (eV) | A (eV) | χ (eV) | η (eV) | σ (eV−1) | µ | ΔN |
|---|---|---|---|---|---|---|---|---|---|
| −9.884 | −3.922 | −5.962 | 9.884 | 3.922 | 6.903 | 2.981 | 0.335 | −1.455 | 0.145 |
| Atom | Charge | Atom | Charge | Atom | Charge | 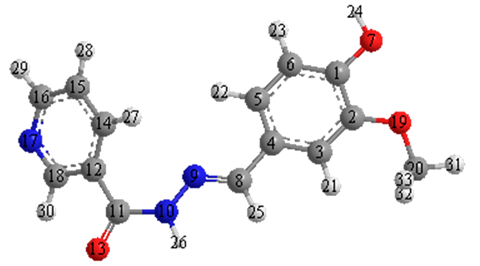 |
| C(1) | 0.0726 | C(8) | −0.1199 | C(15) | −0.1996 | |
| C(2) | 0.0539 | N(9) | −0.0346 | C(16) | −0.0467 | |
| C(3) | 0.1627 | N(10) | −0.3113 | N(17) | −0.1446 | |
| C(4) | −0.0450 | C(11) | 0.3789 | C(18) | −0.0192 | |
| C(5) | −0.0874 | C(12) | −0.1612 | O(19) | −0.1862 | |
| C(6) | −0.1985 | O(13) | −0.3420 | C(20) | −0.0797 | |
| O(7) | −0.2252 | C(14) | −0.0327 | H(21) | 0.1407 |
| C | Mn | Si | Al | S | P | Fe |
|---|---|---|---|---|---|---|
| 0.21% | 0.05% | 0.38% | 0.01% | 0.05% | 0.09% | balance |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Betti, N.; Al-Amiery, A.A.; Al-Azzawi, W.K. Experimental and Quantum Chemical Investigations on the Anticorrosion Efficiency of a Nicotinehydrazide Derivative for Mild Steel in HCl. Molecules 2022, 27, 6254. https://doi.org/10.3390/molecules27196254
Betti N, Al-Amiery AA, Al-Azzawi WK. Experimental and Quantum Chemical Investigations on the Anticorrosion Efficiency of a Nicotinehydrazide Derivative for Mild Steel in HCl. Molecules. 2022; 27(19):6254. https://doi.org/10.3390/molecules27196254
Chicago/Turabian StyleBetti, Nadia, Ahmed A. Al-Amiery, and Waleed Khalid Al-Azzawi. 2022. "Experimental and Quantum Chemical Investigations on the Anticorrosion Efficiency of a Nicotinehydrazide Derivative for Mild Steel in HCl" Molecules 27, no. 19: 6254. https://doi.org/10.3390/molecules27196254
APA StyleBetti, N., Al-Amiery, A. A., & Al-Azzawi, W. K. (2022). Experimental and Quantum Chemical Investigations on the Anticorrosion Efficiency of a Nicotinehydrazide Derivative for Mild Steel in HCl. Molecules, 27(19), 6254. https://doi.org/10.3390/molecules27196254








