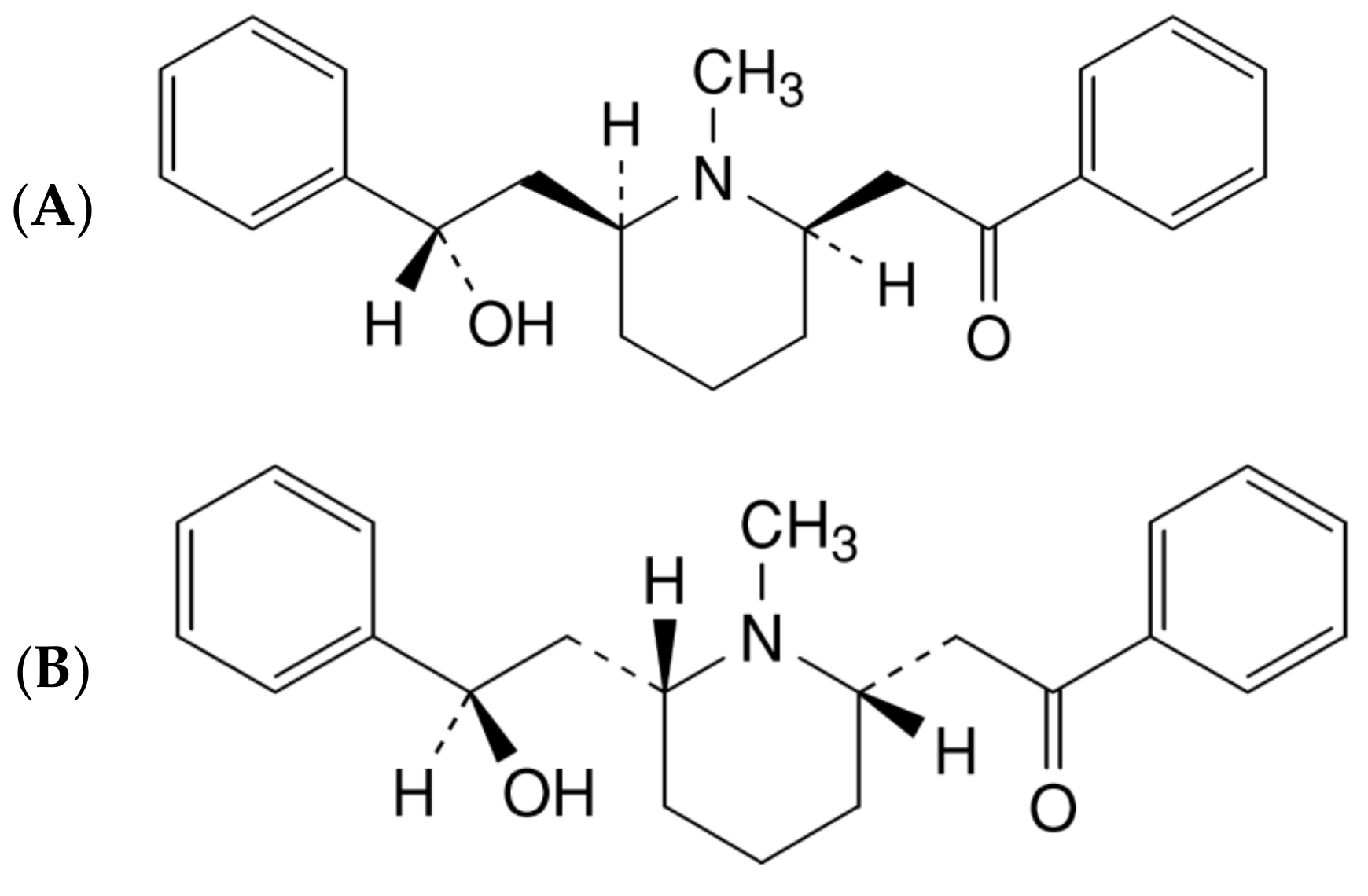
From cis-Lobeline to trans-Lobeline: Study on the Pharmacodynamics and Isomerization Factors
Abstract
1. Introduction
2. Results
2.1. Determination of trans-Lobeline Content
2.1.1. Optimized HPLC Conditions Were Feasible
2.1.2. Determination of trans-Lobeline Percentage Content
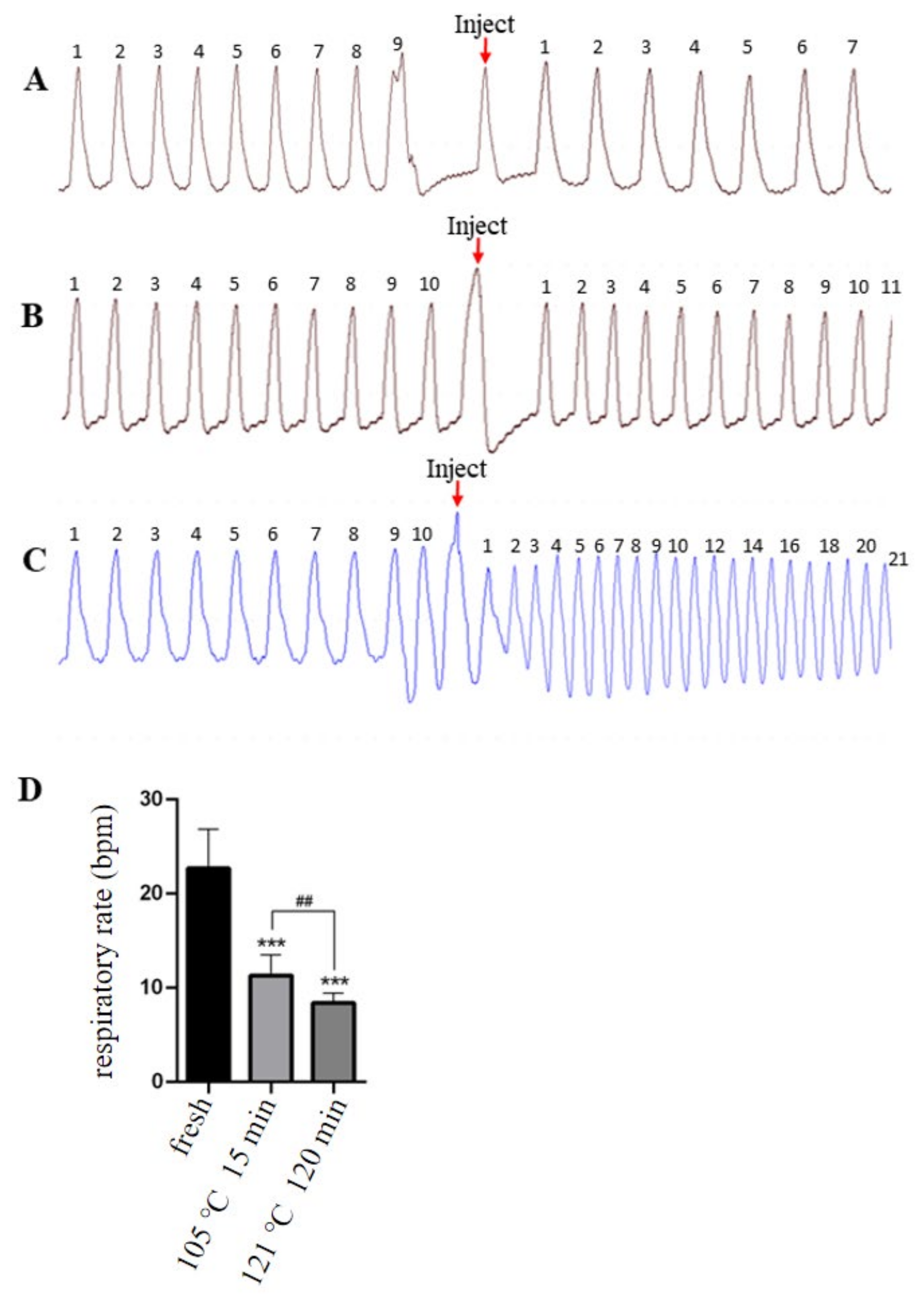
2.2. Effects of Two Isomers on Stimulation of Respiratory Are Significantly Different
2.3. Method Validation in the Experiment on Influencing Factors of Isomerization
2.3.1. Selectivity and Specificity
2.3.2. Linearity and LLOQ
2.3.3. Precision and Accuracy
2.3.4. Extraction Recovery
2.3.5. Stability
2.4. Influencing Factors of Isomerization
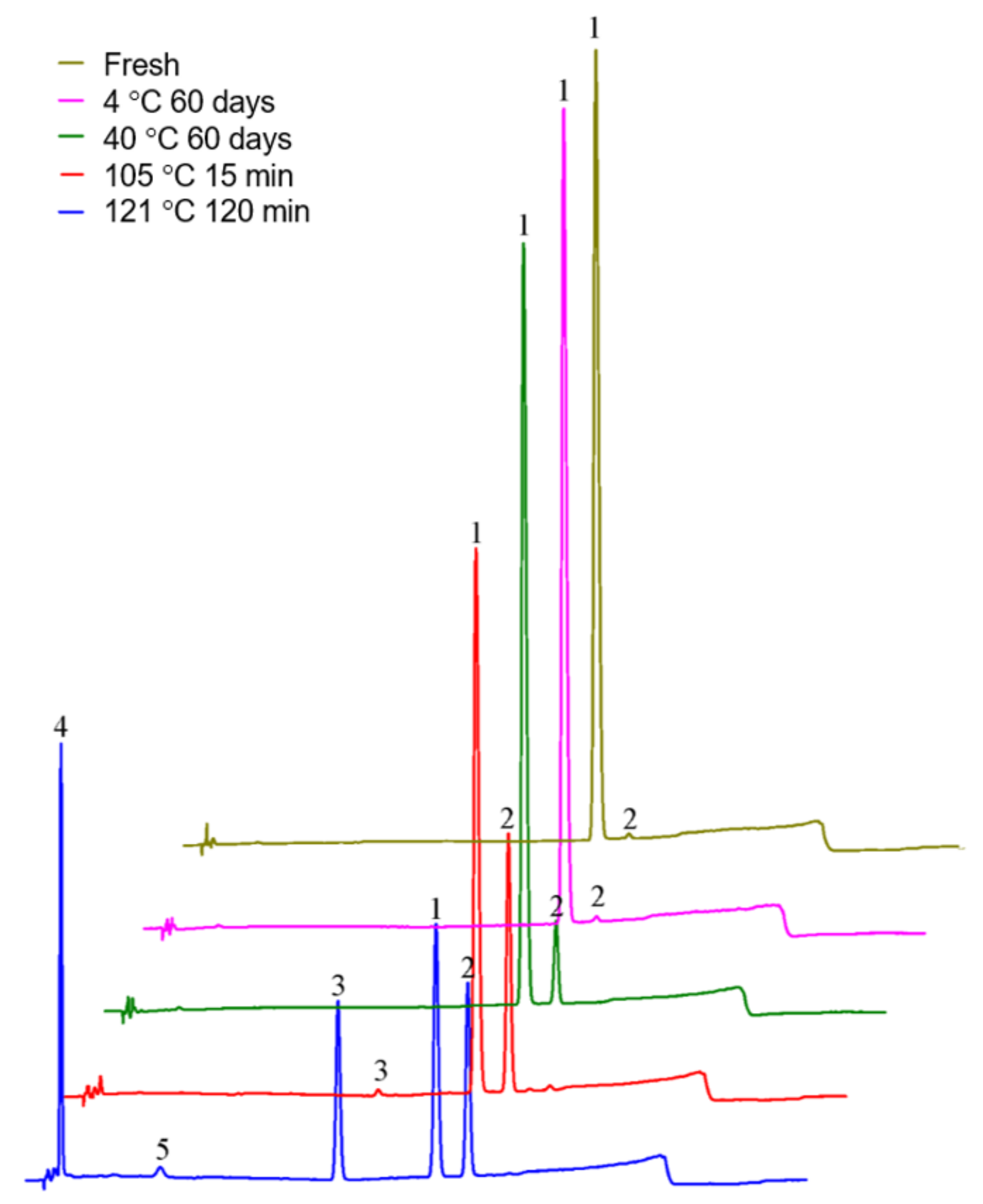
2.4.1. Temperature Is the Catalytic Factor of Isomerization
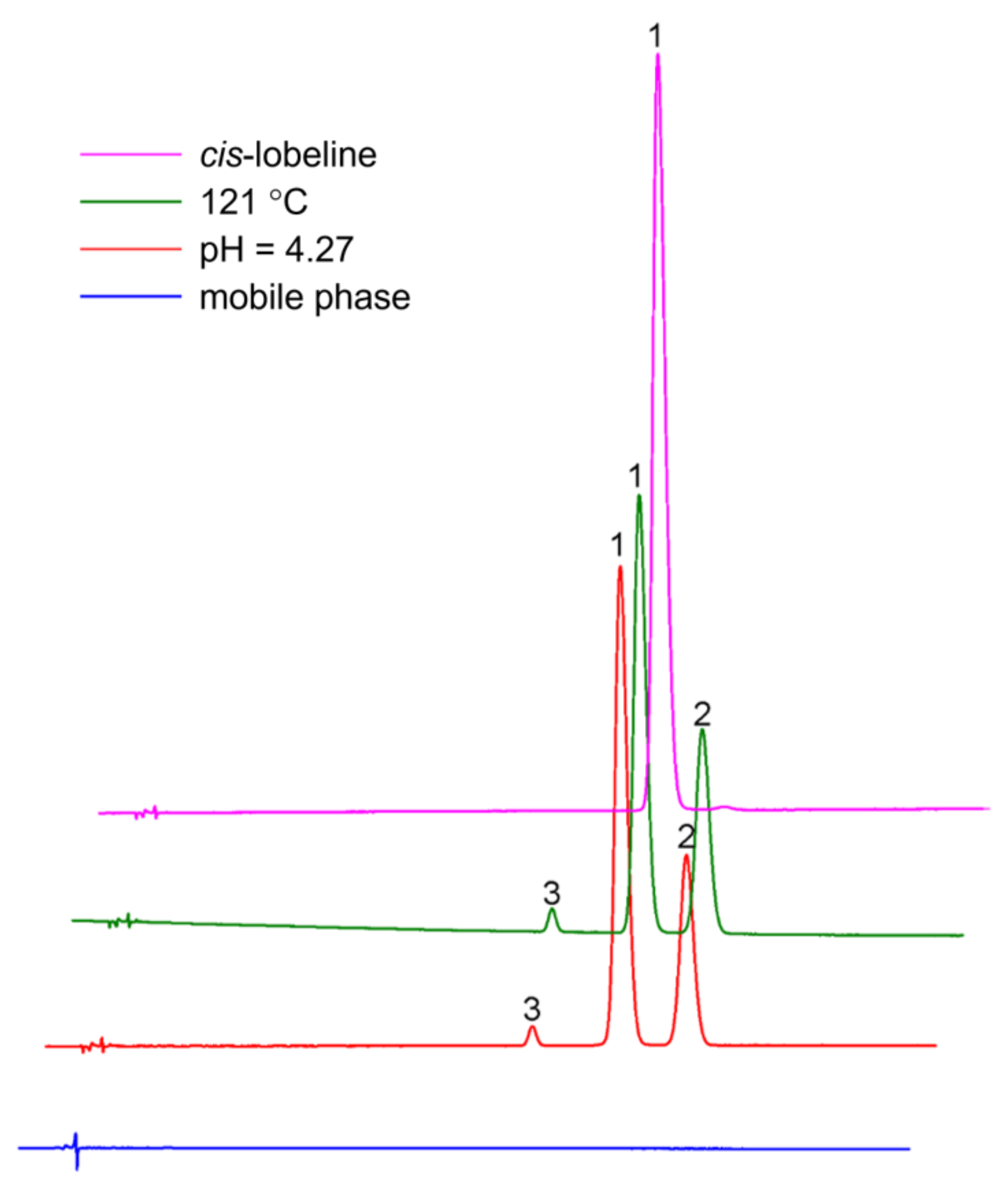
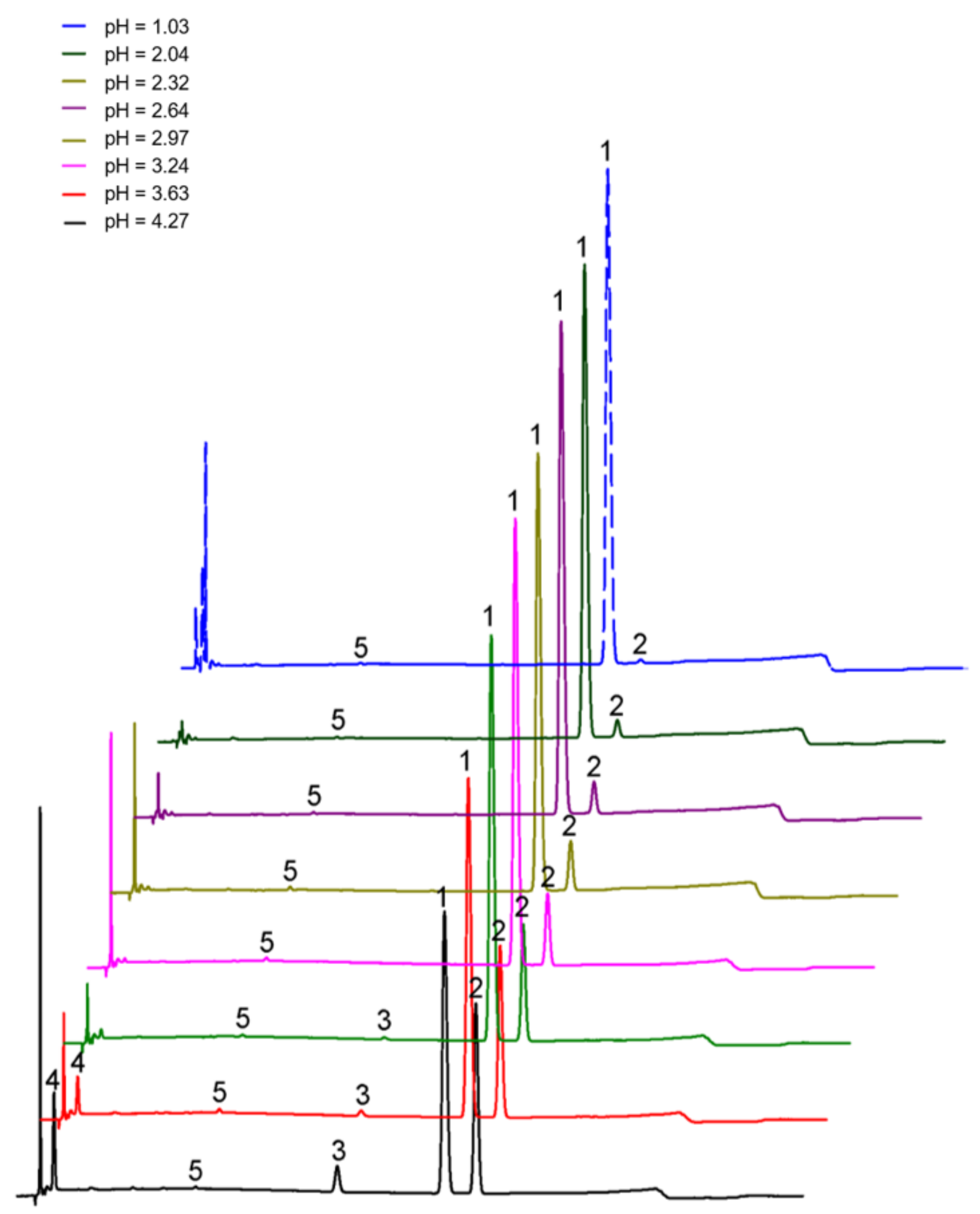
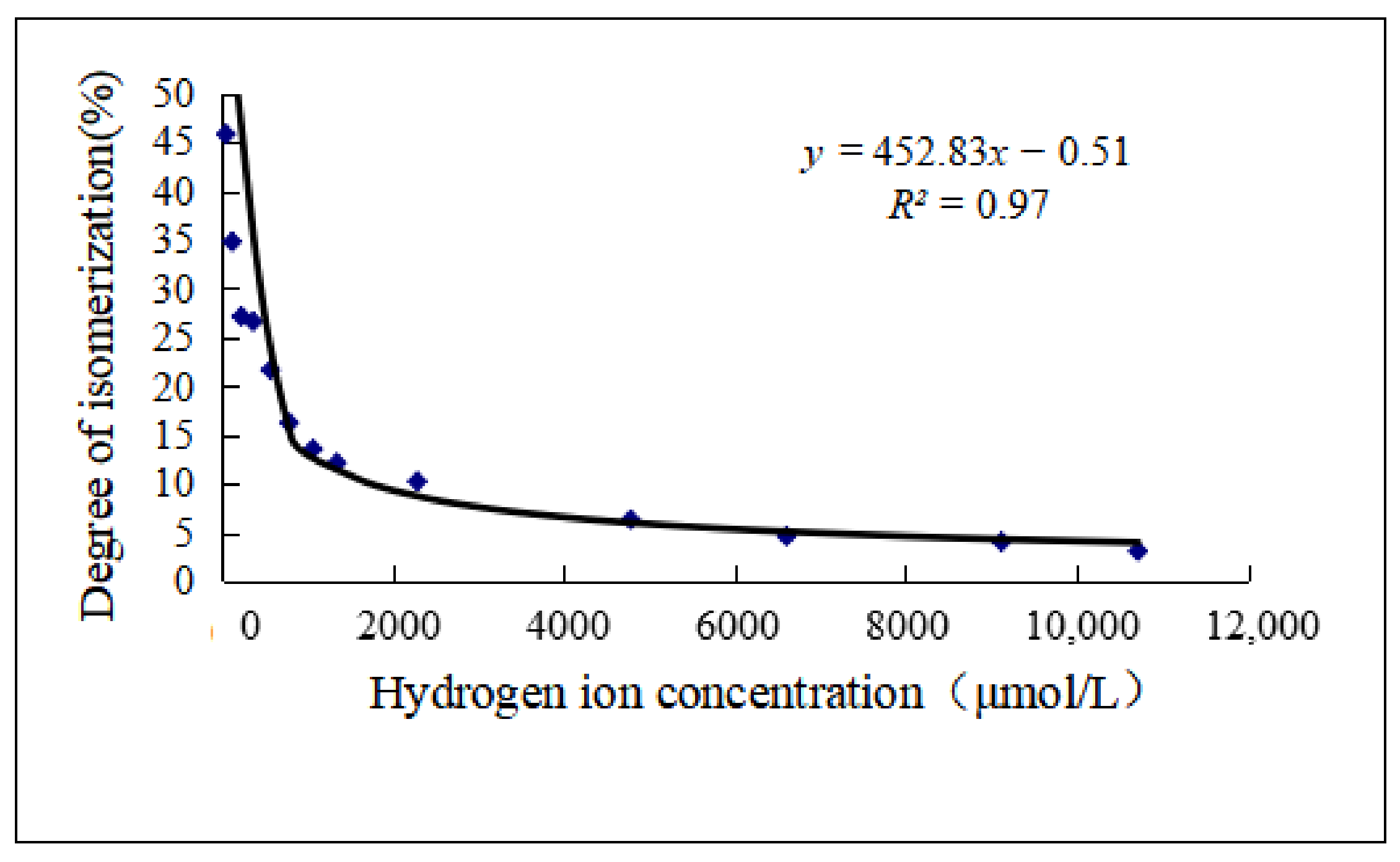
2.4.2. pH Is the Key Factor of Isomerization
3. Materials and Methods
3.1. Drugs and Reagents
3.2. Animals, Animal Management, and Experimental Design
3.3. Determination of cis-Lobeline and trans-Lobeline Content
3.4. Method Validation
3.5. Pharmacodynamic Study of trans-Lobeline Isomer
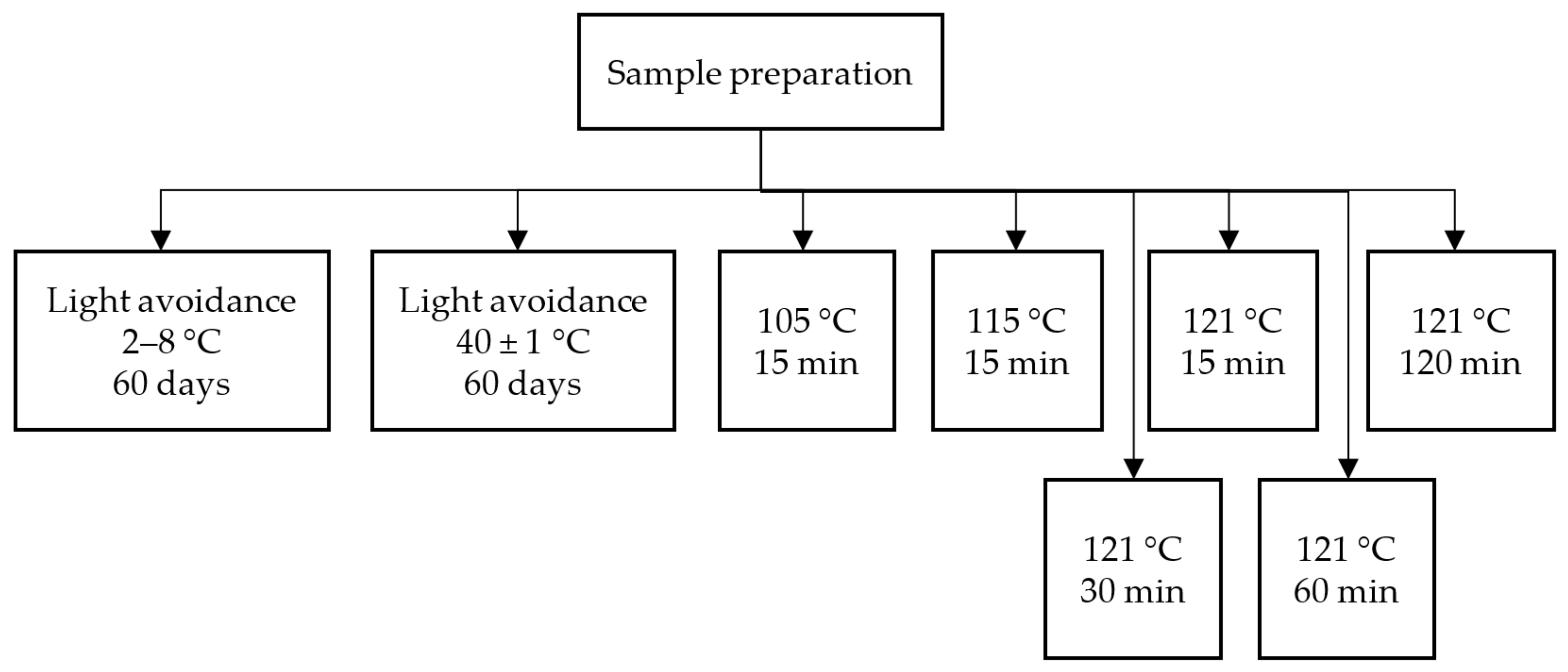
3.6. Study on the Influencing Factors of Isomerization
3.6.1. Investigation of the Relationship between Temperature and Isomerization
3.6.2. Investigation of the Relationship between pH and Isomerization
3.7. Statistical Analysis
4. Discussion
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Sample Availability
References
- Malcolmson, S.J. Racing against unwanted isomerization. Science 2021, 371, 345–346. [Google Scholar] [CrossRef] [PubMed]
- Ringle, M.L.; Loomba, R.; Dykes, J.C.; Khan, D.; Schidlow, D.; Wernovsky, G. The multisystem nature of isomerism: Left isomerism complicated by Abernethy malformation and portopulmonary hypertension. Cardiol. Young 2021, 31, 532–540. [Google Scholar] [CrossRef] [PubMed]
- Sengupta, A.; Rognoni, L.E.; Merkel, U.; Žoldák, G.; Rief, M. SlyD Accelerates trans-to-cis Prolyl Isomerization in a Mechanosignaling Protein under Load. J. Phys. Chem. B 2021, 125, 8712–8721. [Google Scholar] [CrossRef] [PubMed]
- Ganguly, S.; Murugan, N.A.; Ghosh, D.; Narayanaswamy, N.; Govindaraju, T.; Basu, G. DNA Minor Groove-Induced cis-trans Isomerization of a Near-Infrared Fluorescent Probe. Biochemistry 2021, 60, 2084–2097. [Google Scholar] [CrossRef]
- Gai, F.; Hasson, K.C.; McDonald, J.C.; Anfinrud, P.A. Chemical dynamics in proteins: The photoisomerization of retinal in bacteriorhodopsin. Science 1998, 279, 1886–1891. [Google Scholar] [CrossRef]
- Tian, X.Q.; Chen, T.; Matsuoka, L.; Wortsman, J.; Holick, M. Kinetic and thermodynamic studies of the conversion of previtamin D3 to vitamin D3 in human skin. J. Biol. Chem. 1993, 268, 14888–14892. [Google Scholar] [CrossRef]
- Delidovich, I.; Palkovits, R. Catalytic Isomerization of Biomass-Derived Aldoses: A Review. ChemSusChem 2016, 9, 547–561. [Google Scholar] [CrossRef]
- Meléndez, E.N.; Carreras, L.; Gijón, J.R. New alkaloid from Lobelia portoricensis Urban. J. Pharm. Sci. 1967, 56, 1677–1680. [Google Scholar] [CrossRef]
- Chenevert, R.; Morin, P. Synthesis of (−)-lobeline via enzymatic desymmetrization of lobelanidine. Bioorg. Med. Chem. 2009, 17, 1837–1839. [Google Scholar] [CrossRef]
- National Center for Biotechnology Information. PubChem Compound Summary for CID 101616, Lobeline. Available online: https://pubchem.ncbi.nlm.nih.gov/compound/Lobeline (accessed on 12 September 2022).
- Miller, D.K.; Crooks, P.A.; Zheng, G.; Grinevich, V.P.; Norrholm, S.D.; Dwoskin, L.P.; Rinaldi-Carmona, M.; Barth, F.; Congy, C.; Martinez, S.; et al. Lobeline analogs with enhanced affinity and selectivity for plasmalemma and vesicular monoamine transporters. J. Pharmacol. Exp. Ther. 2004, 310, 1035–1045. [Google Scholar] [CrossRef]
- Kim, H.L. Effects of SKF 525-A, phenobarbital and 3-methylcholanthrene on the toxicity of lobeline sulfate. Vet. Hum. Toxicol. 1985, 27, 1–2. [Google Scholar]
- Zernov, N.G. Effect of lobeline and cytiton in pneumonia in infants. Vopr. Pediatrii 1953, 21, 32–38. [Google Scholar]
- Martin, C.A.; Nuzzo, P.A.; Ranseen, J.D.; Kleven, M.S.; Guenthner, G.; Williams, Y.; Walsh, S.L.; Dwoskin, L.P. Lobeline Effects on Cognitive Performance in Adult ADHD. J. Atten Disord. 2018, 22, 1361–1366. [Google Scholar] [CrossRef]
- Roni, M.A.; Rahman, S. Lobeline attenuates ethanol abstinence-induced depression-like behavior in mice. Alcohol 2017, 61, 63–70. [Google Scholar] [CrossRef]
- Kulkarni, A.M.; Rampogu, S.; Lee, K.W. Computer-Aided Drug Discovery Identifies Alkaloid Inhibitors of Parkinson’s Disease Associated Protein, Prolyl Oligopeptidase. Evid.-Based Complement. Altern. Med. 2021, 2021, 6687572. [Google Scholar] [CrossRef]
- Stead, L.F.; Hughes, J.R. Lobeline for smoking cessation. Cochrane Database Syst. Rev. 2012, 2012, Cd000124. [Google Scholar] [CrossRef]
- Zheng, G.; Dwoskin, L.P.; Deaciuc, A.G.; Norrholm, A.S.D.; Crooks, P.A. Defunctionalized lobeline analogues: Structure-activity of novel ligands for the vesicular monoamine transporter. J. Med. Chem. 2005, 48, 5551–5560. [Google Scholar] [CrossRef][Green Version]
- Rosecrans, J.A.; Young, R. Discriminative Stimulus Properties of S(−)-Nicotine: “A Drug for All Seasons”. Curr. Top. Behav. Neurosci. 2018, 39, 51–94. [Google Scholar]
- Hojahmat, M.; Horton, D.B.; Norrholm, S.D.; Miller, D.K.; Grinevich, V.P.; Deaciuc, A.G.; Dwoskin, L.P.; Crooks, P.A. Lobeline esters as novel ligands for neuronal nicotinic acetylcholine receptors and neurotransmitter transporters. Bioorg. Med. Chem. 2010, 18, 640–649. [Google Scholar] [CrossRef]
- Xia, R.; Dai, H.; Che, B.; Zhang, J.; Guo, H. Determiniation of Related substances in Lobeline Hydrochloride Injection by RP-HPLC. Drug Stand. China 2009, 10, 46–50. [Google Scholar]
- Seniutinas, G.; Balčytis, A.; Tomašiūnas, R.; Petruškevičius, R.; Getautis, V. Freezing out all-optical poling dynamics of azophenylcarbazole molecules in polycarbonate. Phys. Chem. Chem. Phys. 2013, 15, 14219–14228. [Google Scholar] [CrossRef]
- Kanda, J.; Egami, N.; Sasamori, T.; Imayoshi, A.; Hosoya, T.; Tsubaki, K. Synthesis of Bridged Indigos and Their Thermoisomerization and Photoisomerization Behaviors. J. Org. Chem. 2021, 86, 17620–17628. [Google Scholar] [CrossRef]
- Mahatthananchai, J.; Dumas, A.M.; Bode, J.W. Catalytic selective synthesis. Angew. Chem. Int. Ed. Engl. 2012, 51, 10954–10990. [Google Scholar] [CrossRef]
- Crooks, P.A.; Zheng, G.; Vartak, A.P.; Culver, J.P.; Zheng, F.; Horton, B.D.; Dwoskin, P.L. Design, synthesis and interaction at the vesicular monoamine transporter-2 of lobeline analogs: Potential pharmacotherapies for the treatment of psychostimulant abuse. Curr. Top. Med. Chem. 2011, 11, 1103–1127. [Google Scholar] [CrossRef]
- Zheng, Q.; Wang, Y.; Zhang, S. Beyond Alkaloids: Novel Bioactive Natural Products From Lobelia Species. Front. Pharmacol. 2021, 12, 638210. [Google Scholar] [CrossRef]

| Sample Name | cis-Lobeline (%) | trans-Lobeline (%) | Degree of Isomerization (%) |
|---|---|---|---|
| Fresh | 100% | 0 | 0 |
| 105 °C 15 min | 61.3 ± 0.45% | 36.9 ± 0.68% | 38.7% |
| 121 °C 120 min | 32.3 ± 0.47% | 22.5 ± 1.05% | 67.7% |
| Sample Name | Degree of Isomerization (%) | Increased Respiratory Rate (bpm) |
|---|---|---|
| Fresh | 0 | 22.7 ± 4.14 |
| 105 °C 15 min | 38.7% | 11.3 ± 2.21 *** |
| 121 °C 120 min | 67.7% | 8.4 ± 1.07 ***, ## |
| Name | Standard Curves | R2 | Linear Range (μg/mL) | LLOQ (μg/mL) |
|---|---|---|---|---|
| cis-lobeline | y = 21.426x − 8.0696 | 0.9998 | 30–240 | 0.36 |
| Name | QC Conc. (μg/mL) | Intraday | Interday | ||||
|---|---|---|---|---|---|---|---|
| Calc Conc. (μg/mL) | RSD (%) | Accuracy (%) | Calc Conc. (μg/mL) | RSD (%) | Accuracy (%) | ||
| cis-lobeline | 60.52 | 60.08 ± 2.13 | 0.27 | 99.67 ± 0.28 | 61.03 ± 1.96 | 0.38 | 99.15 ± 1.06 |
| 121.48 | 120.61 ± 3.27 | 0.50 | 98.79 ± 1.02 | 120.57 ± 2.69 | 0.50 | 98.76 ± 0.94 | |
| 242.55 | 245.37 ± 4.95 | 0.69 | 97.34 ± 2.06 | 240 ± 3.54 | 0.63 | 96.25 ± 3.21 | |
| Group | Sample Intake (mg) | Reference Material Added (mg) | Content Determination (mg) | Recovery (%) | Average (%) | RSD (%) |
|---|---|---|---|---|---|---|
| 80% | 2.122 | 1.960 | 4.016 | 98.37 | 98.38 | 0.51 |
| 2.122 | 1.960 | 3.997 | 97.89 | |||
| 2.122 | 1.960 | 4.037 | 98.89 | |||
| 100% | 2.653 | 2.450 | 5.119 | 100.31 | 100.51 | 0.61 |
| 2.653 | 2.450 | 5.165 | 101.20 | |||
| 2.653 | 2.450 | 5.105 | 100.03 | |||
| 120% | 3.184 | 2.941 | 6.138 | 100.23 | 100.27 | 0.42 |
| 3.184 | 2.941 | 6.117 | 99.88 | |||
| 3.184 | 2.941 | 6.168 | 100.71 |
| Sample Name | cis-Lobeline (%) | trans-Lobeline (%) | Degree of Isomerization (%) |
|---|---|---|---|
| Fresh | 100 | 0 | 0 |
| 4 °C 60 days | 99.2 ± 0.39 | 0.6 ± 0.08 | 0.8 |
| 40 °C 60 days | 93.2 ± 0.63 | 6.7 ± 0.49 | 6.8 |
| 105 °C 15 min | 61.3 ± 0.45 | 36.9 ± 0.68 | 38.7 |
| 121 °C 120 min | 32.3 ± 0.47 | 22.5 ± 1.05 | 67.7 |
| pH | Hydrogen Ion Concentration (μmol/L) | Degree of Isomerization (%) |
|---|---|---|
| 1.97 | 10,715.2 | 3.084 |
| 2.04 | 9120.1 | 4.04 |
| 2.18 | 6606.9 | 4.629 |
| 2.32 | 4786.3 | 6.316 |
| 2.64 | 2290.9 | 10.237 |
| 2.87 | 1349.0 | 12.132 |
| 2.97 | 1071.5 | 13.537 |
| 3.1 | 794.3 | 16.236 |
| 3.24 | 575.4 | 21.66 |
| 3.43 | 371.5 | 26.61 |
| 3.63 | 234.4 | 27.134 |
| 3.9 | 125.9 | 34.796 |
| 4.27 | 53.7 | 45.824 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Xu, H.-H.; Yang, L.; Tang, M.-X.; Ye, A.-P.; Tu, B.-D.; Jiang, Z.-H.; Yi, J.-F. From cis-Lobeline to trans-Lobeline: Study on the Pharmacodynamics and Isomerization Factors. Molecules 2022, 27, 6253. https://doi.org/10.3390/molecules27196253
Xu H-H, Yang L, Tang M-X, Ye A-P, Tu B-D, Jiang Z-H, Yi J-F. From cis-Lobeline to trans-Lobeline: Study on the Pharmacodynamics and Isomerization Factors. Molecules. 2022; 27(19):6253. https://doi.org/10.3390/molecules27196253
Chicago/Turabian StyleXu, Huan-Hua, Liang Yang, Ming-Xia Tang, An-Ping Ye, Bo-Dan Tu, Zhen-Hong Jiang, and Jian-Feng Yi. 2022. "From cis-Lobeline to trans-Lobeline: Study on the Pharmacodynamics and Isomerization Factors" Molecules 27, no. 19: 6253. https://doi.org/10.3390/molecules27196253
APA StyleXu, H.-H., Yang, L., Tang, M.-X., Ye, A.-P., Tu, B.-D., Jiang, Z.-H., & Yi, J.-F. (2022). From cis-Lobeline to trans-Lobeline: Study on the Pharmacodynamics and Isomerization Factors. Molecules, 27(19), 6253. https://doi.org/10.3390/molecules27196253






