Odor Characteristics of Novel Non-Canonical Terpenes
Abstract
:1. Introduction
2. Results
2.1. Determination of Purities, Response Factors, and Mass Spectra of the Methylated Compounds
2.2. Odor Description of Methylated Hemi-, Mono-, and Sesquiterpenes
2.3. Odor Threshold of (2E)-Decenal in Water and Air
- OTair,IS (participant 1) = 8.0 ± 2.7 ng L−1;
- OTair,IS (participant 2) = 4.0 ± 1.3 ng L−1;
- OTair,IS (participant 3) = 16.0 ± 5.3 ng L−1.
2.4. Odor Thresholds in Air
3. Discussion
4. Materials and Methods
4.1. Chemicals
4.2. Sensory Analysis
4.3. Gas Chromatographic Analysis
4.4. Odor Thresholds of the Internal Standard (2E)-Decenal
4.5. Odor Thresholds in Air
5. Conclusions
6. Patents
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Sample Availability
Abbreviations
| DMAPP | dimethylallyl pyrophosphate |
| DT | detection threshold |
| GC-O | gas chromatography-olfactometry |
| ee | enantiomeric excess |
| FPP | farnesyl pyrophosphate |
| IPP | isopentenyl pyrophosphate |
| IS | internal standard |
| n.a. | not available |
| ODP | olfactory detection port |
| OT | odor threshold |
| OTW | odor threshold in water |
| OTair | odor threshold in air |
| n.d. | not determined |
| RT | recognition threshold |
| SAM | S-adenosyl methionine |
References
- Ružička, L. The isoprene rule and the biogenesis of terpenic compounds. Experientia 1953, 9, 357–367. [Google Scholar] [CrossRef] [PubMed]
- Breitmaier, E. Terpenes; Wiley: Hoboken, NJ, USA, 2006; ISBN 9783527317868. [Google Scholar]
- Shiota, H.; Young, H.; Paterson, V.J.; Irie, M. Volatile aroma constituents of pepino fruit. J. Sci. Food Agric. 1988, 43, 343–354. [Google Scholar] [CrossRef]
- Persson, P.-E. Sensory properties and analysis of two muddy odour compounds, geosmin and 2-methylisoborneol, in water and fish. Water Res. 1980, 14, 1113–1118. [Google Scholar] [CrossRef]
- Drummond, L.; von Wallbrunn, C.; Buchhaupt, M. Microbial Dehydration of 2-Methylisoborneol in Forest Soil. Chem. Biodivers. 2022, 19, e2021007. [Google Scholar] [CrossRef]
- Brock, N.L.; Ravella, S.R.; Schulz, S.; Dickschat, J.S. A detailed view of 2-methylisoborneol biosynthesis. Angew. Chem. Int. Ed. 2013, 52, 2100–2104. [Google Scholar] [CrossRef]
- Harms, V.; Schröder, B.; Oberhauser, C.; Tran, C.D.; Winkler, S.; Dräger, G.; Kirschning, A. Methyl-Shifted Farnesyldiphosphate Derivatives Are Substrates for Sesquiterpene Cyclases. Org. Lett. 2020, 22, 4360–4365. [Google Scholar] [CrossRef]
- Ignea, C.; Pontini, M.; Motawia, M.S.; Maffei, M.E.; Makris, A.M.; Kampranis, S.C. Synthesis of 11-carbon terpenoids in yeast using protein and metabolic engineering. Nat. Chem. Biol. 2018, 14, 1090–1098. [Google Scholar] [CrossRef]
- Kschowak, M.J.; Wortmann, H.; Dickschat, J.S.; Schrader, J.; Buchhaupt, M. Heterologous expression of 2-methylisoborneol / 2 methylenebornane biosynthesis genes in Escherichia coli yields novel C11-terpenes. PLoS ONE 2018, 13, e0196082. [Google Scholar] [CrossRef]
- Drummond, L.; Kschowak, M.J.; Breitenbach, J.; Wolff, H.; Shi, Y.-M.; Schrader, J.; Bode, H.B.; Sandmann, G.; Buchhaupt, M. Expanding the Isoprenoid Building Block Repertoire with an IPP Methyltransferase from Streptomyces monomycini. ACS Synth. Biol. 2019, 8, 1303–1313. [Google Scholar] [CrossRef]
- Dalal, T.; Gupta, N.; Haddad, R. Bilateral and unilateral odor processing and odor perception. Commun. Biol. 2020, 3, 150. [Google Scholar] [CrossRef] [Green Version]
- Genva, M.; Kenne Kemene, T.; Deleu, M.; Lins, L.; Fauconnier, M.-L. Is It Possible to Predict the Odor of a Molecule on the Basis of its Structure? Int. J. Mol. Sci. 2019, 20, 3018. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Haring, H.G.; Rijkens, F.; Boelens, H.; van der Gen, A. Olfactory studies on enantiomeric eremophilane sesquiterpenoids. J. Agric. Food Chem. 1972, 20, 1018–1021. [Google Scholar] [CrossRef]
- Leitereg, T.J.; Guadagni, D.G.; Harris, J.; Mon, T.R.; Teranishi, R. Evidence for the difference between the odours of the optical isomers (4)- and (-)-carvone. Nature 1971, 230, 455–456. [Google Scholar] [CrossRef] [PubMed]
- Egawa, T.; Kameyama, A.; Takeuchi, H. Structural determination of vanillin, isovanillin and ethylvanillin by means of gas electron diffraction and theoretical calculations. J. Mol. Struct. 2006, 794, 92–102. [Google Scholar] [CrossRef] [Green Version]
- The Good Scents Information System. 2-Decanone. Available online: www.thegoodscentscompany.com/data/rw1047071.html (accessed on 17 November 2021).
- The Good Scents Information System. 2-Nonanone. Available online: www.thegoodscentscompany.com/data/rw1008331.html (accessed on 17 November 2021).
- Czerny, M.; Christlbauer, M.; Christlbauer, M.; Fischer, A.; Granvogl, M.; Hammer, M.; Hartl, C.; Hernandez, N.M.; Schieberle, P. Re-investigation on odour thresholds of key food aroma compounds and development of an aroma language based on odour qualities of defined aqueous odorant solutions. Eur. Food Res. Technol. 2008, 228, 265–273. [Google Scholar] [CrossRef]
- Teranishi, R.; Buttery, R.G.; Guadagni, D.G. Odor quality and chemical structure in fruit and vegetable flavors. Ann. N. Y. Acad. Sci. 1974, 237, 209–216. [Google Scholar] [CrossRef]
- Ullrich, F.; Grosch, W. Identification of the most intense volatile flavour compounds formed during autoxidation of linoleic acid. Z. Lebensm. Unters. Forsch. 1987, 184, 277–282. [Google Scholar] [CrossRef]
- Hammer, A.K.; Emrich, N.O.; Ott, J.; Birk, F.; Fraatz, M.A.; Ley, J.P.; Geissler, T.; Bornscheuer, U.T.; Zorn, H. Biotechnological Production and Sensory Evaluation of ω1-Unsaturated Aldehydes. J. Agric. Food Chem. 2021, 69, 345–353. [Google Scholar] [CrossRef]
- Elsharif, S.A.; Banerjee, A.; Buettner, A. Structure-odor relationships of linalool, linalyl acetate and their corresponding oxygenated derivatives. Front. Chem. 2015, 3, 57. [Google Scholar] [CrossRef] [Green Version]
- Elsharif, S.A.; Buettner, A. Influence of the chemical structure on the odor characters of β-citronellol and its oxygenated derivatives. Food Chem. 2017, 232, 704–711. [Google Scholar] [CrossRef]
- Yang, D.S.; Shewfelt, R.L.; Lee, K.-S.; Kays, S.J. Comparison of odor-active compounds from six distinctly different rice flavor types. J. Agric. Food Chem. 2008, 56, 2780–2787. [Google Scholar] [CrossRef] [PubMed]
- Mori, K.; Manabe, H. Unique Characteristics of the Olfactory System. In The Olfactory System; Mori, K., Ed.; Springer: Tokyo, Japan, 2014; pp. 1–18. ISBN 978-4-431-54375-6. [Google Scholar]
- Rothe, M.; Specht, M. Bemerkungen zur Molmasse von Aromastoffen. Nahrung 1976, 20, 281–286. [Google Scholar] [CrossRef] [PubMed]
- Zarzo, M. The sense of smell: Molecular basis of odorant recognition. Biol. Rev. Camb. Philos. Soc. 2007, 82, 455–479. [Google Scholar] [CrossRef] [PubMed]
- Buck, L.; Axel, R. A novel multigene family may encode odorant receptors: A molecular basis for odor recognition. Cell 1991, 65, 175–187. [Google Scholar] [CrossRef]
- Kuhwald, C.; Kirschning, A. Matteson Reaction under Flow Conditions: Iterative Homologations of Terpenes. Org. Lett. 2021, 23, 4300–4304. [Google Scholar] [CrossRef]
- Stepanyuk, A.; Kirschning, A. Synthetic terpenoids in the world of fragrances: Iso E Super® is the showcase. Beilstein J. Org. Chem. 2019, 15, 2590–2602. [Google Scholar] [CrossRef] [Green Version]
- Chen, W.; Viljoen, A.M. Geraniol—A review of a commercially important fragrance material. S. Afr. J. Bot. 2010, 76, 643–651. [Google Scholar] [CrossRef] [Green Version]
- Letizia, C.; Cocchiara, J.; Lalko, J.; Api, A. Fragrance material review on linalool. Food Chem. Toxicol. 2003, 41, 943–964. [Google Scholar] [CrossRef]
- Elsharif, S.A.; Buettner, A. Structure-Odor Relationship Study on Geraniol, Nerol, and Their Synthesized Oxygenated Derivatives. J. Agric. Food Chem. 2018, 66, 2324–2333. [Google Scholar] [CrossRef]
- Schoenauer, S.; Schieberle, P. Structure-Odor Activity Studies on Monoterpenoid Mercaptans Synthesized by Changing the Structural Motifs of the Key Food Odorant 1-p-Menthene-8-thiol. J. Agric. Food Chem. 2016, 64, 3849–3861. [Google Scholar] [CrossRef]
- Rychlik, M.; Schieberle, P.; Grosch, W. Compilation of Odor Thresholds, Odor Qualities and Retention Indices of Key Food Odorants; Universität München: Garching, Germany, 1998; ISBN 3980342654. [Google Scholar]
- Trimmer, C.; Mainland, J.D. The Olfactory System. In Conn’s Translational Neuroscience; Elsevier: Amsterdam, The Netherlands, 2017; pp. 363–377. ISBN 9780128023815. [Google Scholar]
- van den Dool, H.; Kratz, P.D. A generalization of the retention index system including linear temperature programmed gas—liquid partition chromatography. J. Chromatogr. A 1963, 11, 463–471. [Google Scholar] [CrossRef]

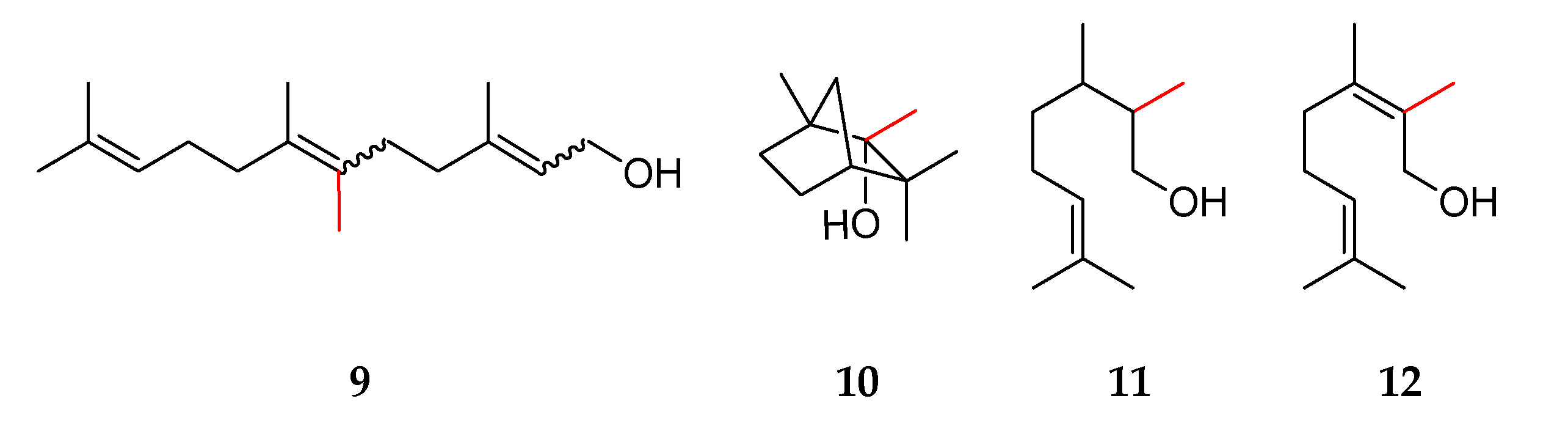
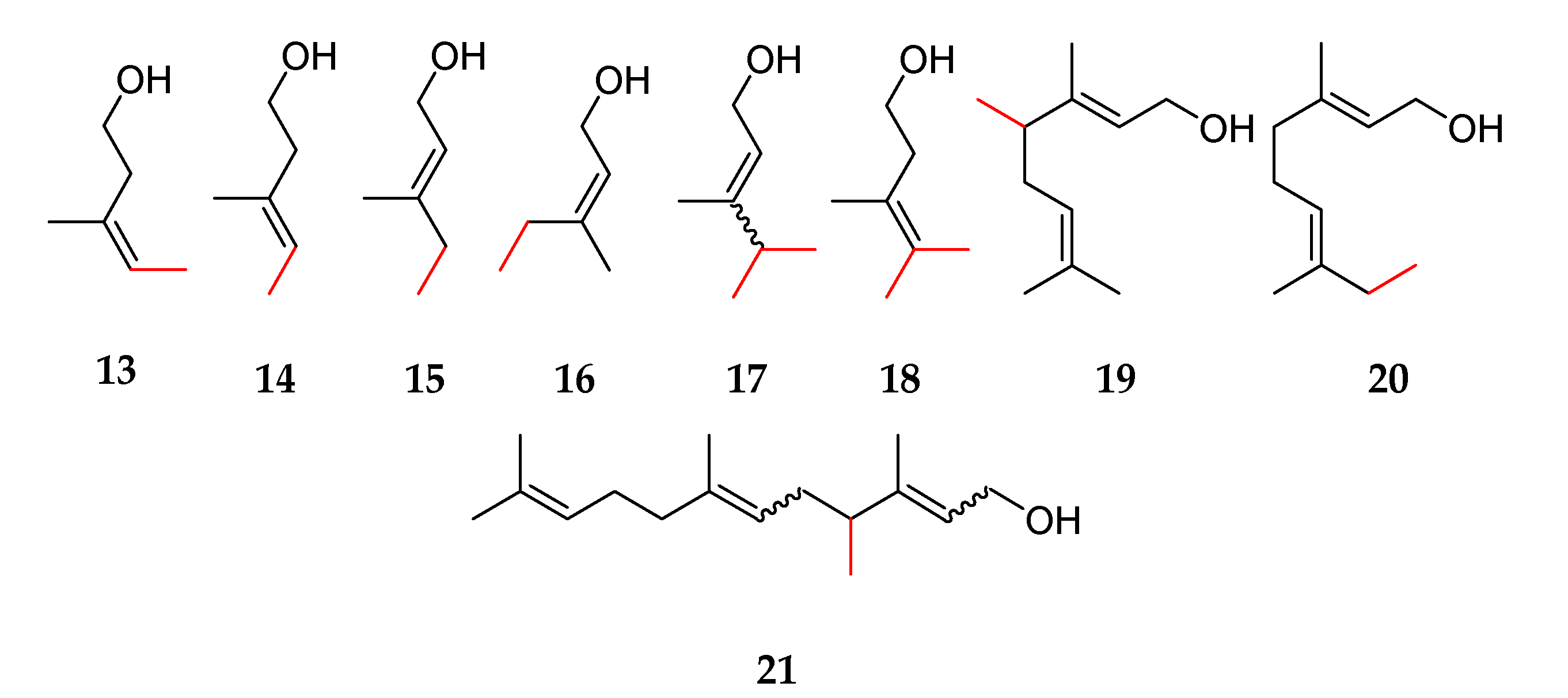

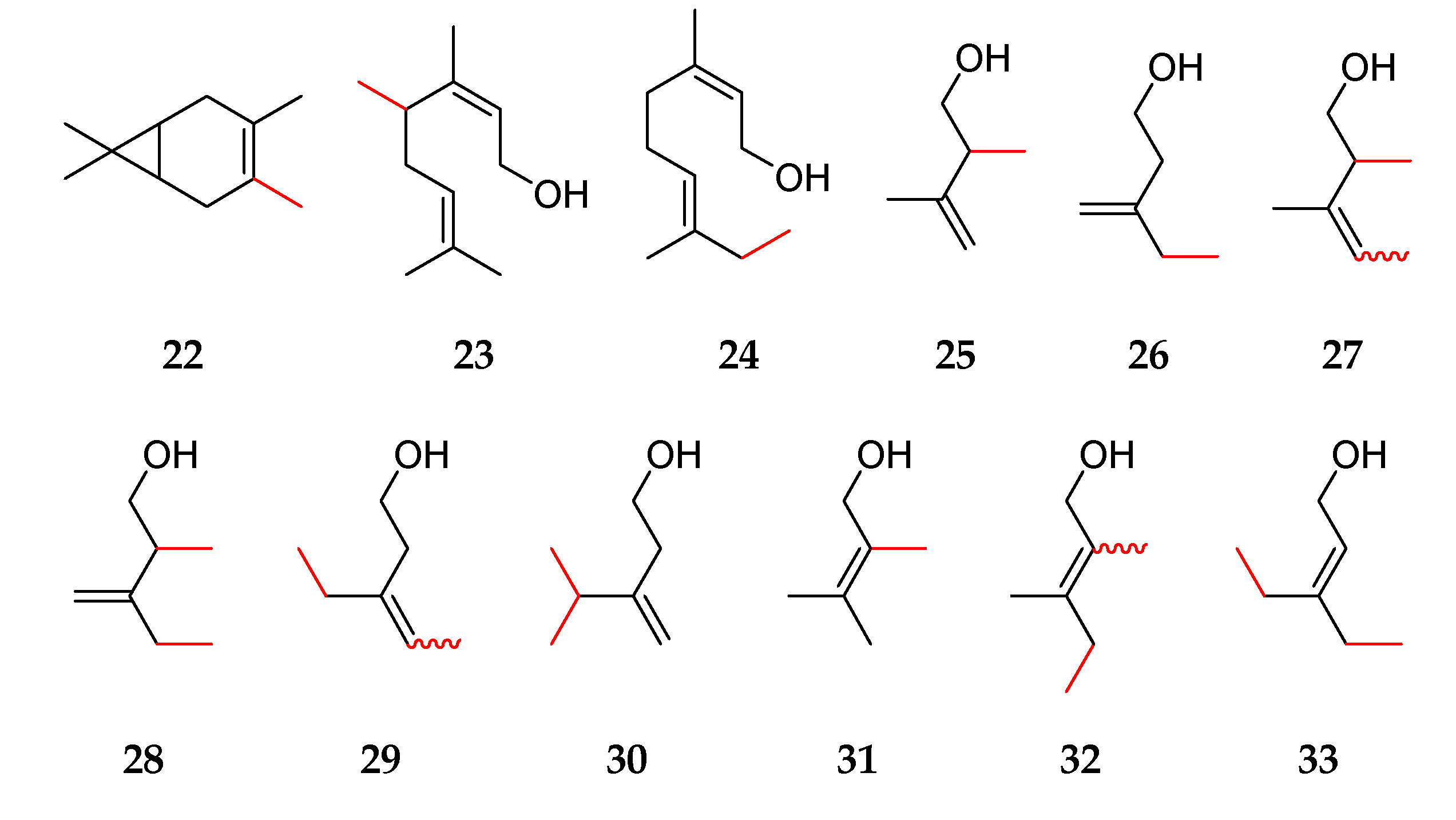
| (a) | ||||
| Compound | RI | Ratios/% | ee/% | |
| 1 | 2-methylenebornane 4 | VF-WAXms: 1120 | - | - |
| DB-5ms: 1017 | - | |||
| 2 | (S)-1-methylcamphene 3 | VF-WAXms: 1075 | - | 100% |
| DB-5ms: 985 | (R/S): 0/100 | |||
| 3 | 4-methyl-3-carene 22 | VF-WAXms: 1229 | - | - |
| DB-5ms: 1091 | - | |||
| 4 | 2-methylcitronellol 11 | VF-WAXms: 1824, 1834 | (E/Z): 64/36 # | 30% |
| DB-5ms: 1301, 1305 | (R/S): 65 +/35 +,# | |||
| 5 | 4-methylfarnesol 21 | VF-WAXms: 2348 | - | - |
| DB-5ms: 1749 | - | |||
| 6 | 6-methylfarnesol 9 | VF-WAXms: 2380, 2430 | - | - |
| DB-5ms: 1790 | - | |||
| 7 | (S)-2-methyl-α-fenchol 10 | VF-WAXms: 1606 | - | 100% |
| DB-5ms: 1199 | (R/S): 0/100 | |||
| 8 | 2-methylgeraniol 5/2-methylnerol 12 | VF-WAXms: 1843 Z,*, 1884 E | (E/Z): 50/50 | - |
| DB-5ms: 1299 Z,*, 1317 E | - | |||
| 9 | 4-methylgeraniol 19/4-methylnerol 23 | VF-WAXms: 1807 Z,*, 1857 E | (E/Z): 8/14 | 100% |
| DB-5ms: 1265 Z,*, 1293 E | (R/S): 100/0 # | |||
| 10 | 8-methylgeraniol 20/8-methylnerol 24 | VF-WAXms: 1919 Z,*, 1923 E | (E/Z): 75/25 | - |
| DB-5ms: 1334 Z,*, 1341 E | - | |||
| 11 | 2-methylisoprenol 25 | VF-WAXms: 1283 | - | - |
| DB-5ms: 812 | - | |||
| 12 | 5-methylisoprenol 26 | VF-WAXms: 1348 | - | - |
| DB-5ms: 842 | - | |||
| 13 | (E)-4-methylisoprenol 14 | VF-WAXms: 1363 | - | - |
| DB-5ms: 861 | - | |||
| 14 | (Z)-4-methylisoprenol 13 | VF-WAXms: 1374 | - | - |
| DB-5ms: 856 | - | |||
| (b) | ||||
| Compound | RI | Ratios/% | ee/% | |
| 15 | 2,4-dimethylisoprenol 27 | VF-WAXms: 1393, 1401 | (E/Z): 86/14 # | - |
| DB-5ms: 916, 924 | - | |||
| 16 | 2,5-dimethylisoprenol 28 | VF-WAXms: 1378 | - | - |
| DB-5ms: 904 | - | |||
| 17 | 4,4-dimethylisoprenol 18 | VF-WAXms: 1477 | - | - |
| DB-5ms: 958 | - | |||
| 18 | 4,5-dimethylisoprenol 29 | VF-WAXms: 1441, 1467 | (E/Z): 63/37 # | - |
| DB-5ms: 944, 949 | - | |||
| 19 | 5,5-dimethylisoprenol 30 | VF-WAXms: 1400 | - | - |
| DB-5ms: 906 | - | |||
| 20 | 2-methyllimonene 7 | VF-WAXms: 1299 | - | 0% |
| DB-5ms: 1122 | (R/S): 50/50 | |||
| 21 | 2-methyllinalool 6 | VF-WAXms: 1620 | - | 0% |
| DB-5ms: 1190 | (R/S): 50/50 | |||
| 22 | 2-methylprenol 31 | VF-WAXms: 1407 | - | - |
| DB-5ms: 877 | - | |||
| 23 | (Z)-4-methylprenol 16 | VF-WAXms: 1393 | - | - |
| DB-5ms: 866 | - | |||
| 24 | (E)-4-methylprenol 15 | VF-WAXms: 1416 | - | - |
| DB-5ms: 881 | - | |||
| 25 | 2,4-dimethylprenol 32 | VF-WAXms: 1467, 1478 | (E/Z): 50/50 | - |
| DB-5ms: 951, 956 | - | |||
| 26 | 4,4-dimethylprenol 17 | VF-WAXms: 1448, 1470 | (E/Z): 13/87 # | - |
| DB-5ms: 929, 944 | - | |||
| 27 | 4,5-dimethylprenol 33 | VF-WAXms: 1487 | - | - |
| DB-5ms: 959 | - | |||
| 28 | 2-methyl-α-terpineol 8 | VF-WAXms: 1785 | - | - |
| DB-5ms: 1286 | - | |||
| Substances | Odor Impression | Intensity | |
|---|---|---|---|
| 1 | 2-methylenebornane 4 | earthy (4), coniferous forest (3), resinous (3) | 0.9 ± 0.8 |
| 2 | (S)-1-methylcamphene 3 | resinous (10), coniferous forest (9), woody (3), fruity (3) | 3.3 ± 1.0 |
| 3 | 4-methyl-3-carene 22 | fruity (7), coniferous forest (7), resinous (6), sweetish (4), pepper (4), mint (3), citrus (3) | 3.5 ± 0.8 |
| 4 | 2-methylcitronellol 11 | flowery (8), citrus (6), rose (4), sweetish (3), ethereal (3), fruity (3) | 3.9 ± 0.8 |
| 5 | 4-methylfarnesol 21 | citrus (5), resinous (5), green (3) | 1.7 ± 1.0 |
| 6 | 6-methylfarnesol 9 | -# | 0.7 ± 0.8 |
| 7 | (S)-2-methyl-α-fenchol 10 | earthy (13), moldy (9), moss (3), beetroot (3) | 4.8 ± 0.4 |
| 8 | 2-methylgeraniol 5/2-methylnerol 12 * | flowery (8), citrus (5), resinous (4), rose (4), sweetish (3) | 2.9 ± 1.4 |
| 9 | 4-methylgeraniol 19/4-methylnerol 23 * | citrus (8), lemon (3), lemon peel (3) | 3.7 ± 0.8 |
| 10 | 8-methylgeraniol 20/8-methylnerol 24 * | flowery (8), resinous (6), sweetish (5), citrus (4), varnish (4) | 2.5 ± 0.8 |
| 11 | 2-methylisoprenol 25 | resinous (5), sweetish (3), coniferous forest (3), fruity (3) | 1.6 ± 1.2 |
| 12 | (E)-4-methylisoprenol 14 | green (8), grass (4), herbal (4), coniferous forest (3), apple (3) | 2.5 ± 0.8 |
| 13 | (Z)-4-methylisoprenol 13 | flowery (9), green (6), fruity (5), apple (4) | 3.1 ± 1.0 |
| 14 | 5-methylisoprenol 26 | pungent (6), solvent (6), glue (4), varnish (3) | 4.6 ± 0.8 |
| 15 | 2,4-dimethylisoprenol 27 | coniferous forest (5), green (4), resinous (4) | 2.5 ± 0.7 |
| 16 | 2,5-dimethylisoprenol 28 | resinous (9), coniferous forest (7), mint (3), green (3), varnish (3) | 3.2 ± 1.2 |
| 17 | 4,4-dimethylisoprenol 18 | green (6), citrus (4), flowery (3), soapy (3), grass (3) | 2.5 ± 1.1 |
| 18 | 4,5-dimethylisoprenol 29 | resinous (4), woody (3), coniferous forest (3) | 1.5 ± 1.1 |
| 19 | 5,5-dimethylisoprenol 30 | coniferous forest (10), resinous (8), woody (3) | 3.4 ± 1.1 |
| 20 | 2-methyllimonene 7 | resinous (6), terpene (4), mushroom (4) | 3.7 ± 0.8 |
| 21 | 2-methyllinalool 6 | flowery (11), citrus (9), sweetish (8), fruity (6), bergamot (5), blueberry (4), lavender (3) | 3.7 ± 0.5 |
| 22 | 2-methylprenol 31 | plastic (3), terpene-like (3) | 1.5 ± 0.9 |
| 23 | (Z)-4-methylprenol 16 | plastic (3), terpene-like (3), chemical (3) | 1.7 ± 0.8 |
| 24 | (E)-4-methylprenol 15 | sweetish (5), flowery (5), green (5), citrus (3), fresh (3), resinous (3) | 2.3 ± 1.4 |
| 25 | 2,4-dimethylprenol 32 | resinous (4), woody (3), coniferous forest (3), glue (3), sweetish (3) | 2.8 ± 1.3 |
| 26 | 4,4-dimethylprenol 17 | sweetish (6), fruity (3) | 2.1 ± 1.3 |
| 27 | 4,5-dimethylprenol 33 | woody (6), resinous (3), plastic (3) | 3.1 ± 1.1 |
| 28 | 2-methyl-α-terpineol 8 | sweetish (4), green (3) | 1.1 ± 1.1 |
| Compound | OT (Literature)/ng L−1 | OT (This Study)/ng L−1 | |
|---|---|---|---|
| 1 | (R/S)-citronellol | 11 [33] | n.d. |
| 2 | (R)-citronellol | 1.1 [34] | 24 ± 19 |
| 3 | (S)-citronellol | 0.57 [34] | 19 ± 23 |
| 4 | geraniol | 0.067 [34], 11.5 [33] | 5.7 ± 5.4 |
| 5 | nerol | 61 [34], 68 [35] | 61 ± 100 |
| 6 | (R)-limonene | 135 [35] | 100 ± 67 |
| 7 | (S)-limonene | 270 [35] | 81 ± 84 |
| 8 | (R/S)-linalool | 0.26 [34], 3.2 [33] | n.d. |
| 9 | (R)-linalool | 0.036 [35] | 0.098 ± 0.064 |
| Compounds | |
|---|---|
| Mixture 1 | (R)-camphene (578 mg L−1), (S)-limonene (310 mg L−1), 2-methyllimonene (304 mg L−1), (R)-linalool (38.0 mg L−1), 2-methylfenchol (38.0 mg L−1), (2E)-decenal (38.0 mg L−1), (R)-citronellol (77.4 mg L−1), geraniol (39.5 mg L−1), and 8-methylgeraniol ((E/Z)-mixture, 152 mg L−1) |
| Mixture 2 | 4-methyl-3-carene (405 mg L−1), (R)-α-fenchol (38.4 mg L−1), 2-methyllinalool (38.0 mg L−1), (2E)-decenal (38.0 mg L−1), (S)-citronellol (78.3 mg L−1), and 2-methylgeraniol ((E/Z)-mixture, 152 mg L−1) |
| Mixture 3 | (R/S)-camphene (500 mg L−1), (S)-3-carene (576 mg L−1), (R)-limonene (621 mg L−1), (2E)-decenal (38.0 mg L−1), nerol (78.4 mg L−1), and 4-methylgeraniol ((E/Z)-mixture, 152 mg L−1) |
| Mixture 4 | (S)-1-methylcamphene (456 mg L−1), (2E)-decenal (38.0 mg L−1), and 2-methylcitronellol ((E/Z)-mixture, 152 mg L−1) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sommer, S.; Lang, L.M.; Drummond, L.; Buchhaupt, M.; Fraatz, M.A.; Zorn, H. Odor Characteristics of Novel Non-Canonical Terpenes. Molecules 2022, 27, 3827. https://doi.org/10.3390/molecules27123827
Sommer S, Lang LM, Drummond L, Buchhaupt M, Fraatz MA, Zorn H. Odor Characteristics of Novel Non-Canonical Terpenes. Molecules. 2022; 27(12):3827. https://doi.org/10.3390/molecules27123827
Chicago/Turabian StyleSommer, Svenja, Leon M. Lang, Laura Drummond, Markus Buchhaupt, Marco A. Fraatz, and Holger Zorn. 2022. "Odor Characteristics of Novel Non-Canonical Terpenes" Molecules 27, no. 12: 3827. https://doi.org/10.3390/molecules27123827
APA StyleSommer, S., Lang, L. M., Drummond, L., Buchhaupt, M., Fraatz, M. A., & Zorn, H. (2022). Odor Characteristics of Novel Non-Canonical Terpenes. Molecules, 27(12), 3827. https://doi.org/10.3390/molecules27123827






