The Impact of Composites with Silicate-Based Glasses and Gold Nanoparticles on Skin Wound Regeneration
Abstract
1. Introduction
2. Results and Discussions
2.1. Glass Composite Ointments Characterization
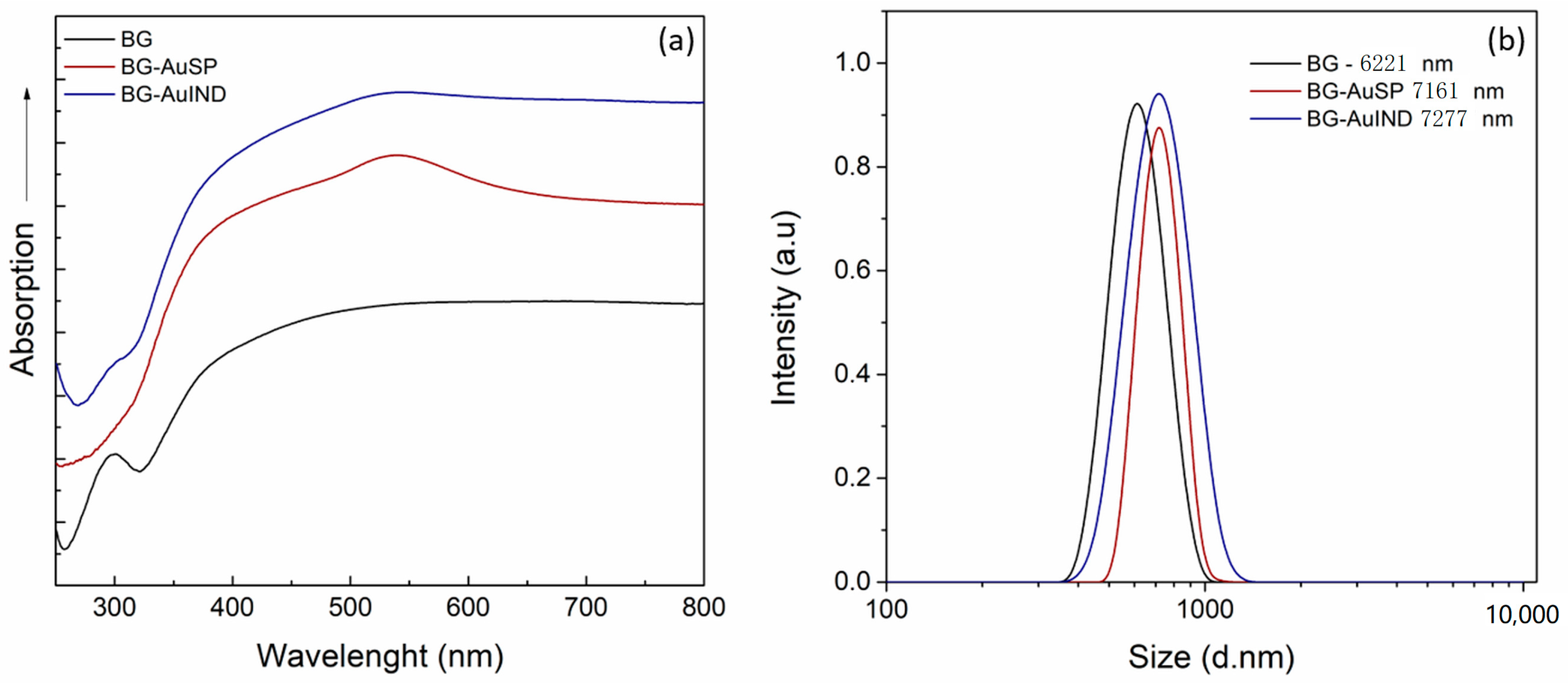
2.1.1. Glasses Characterization
2.1.2. Ointments Characterization
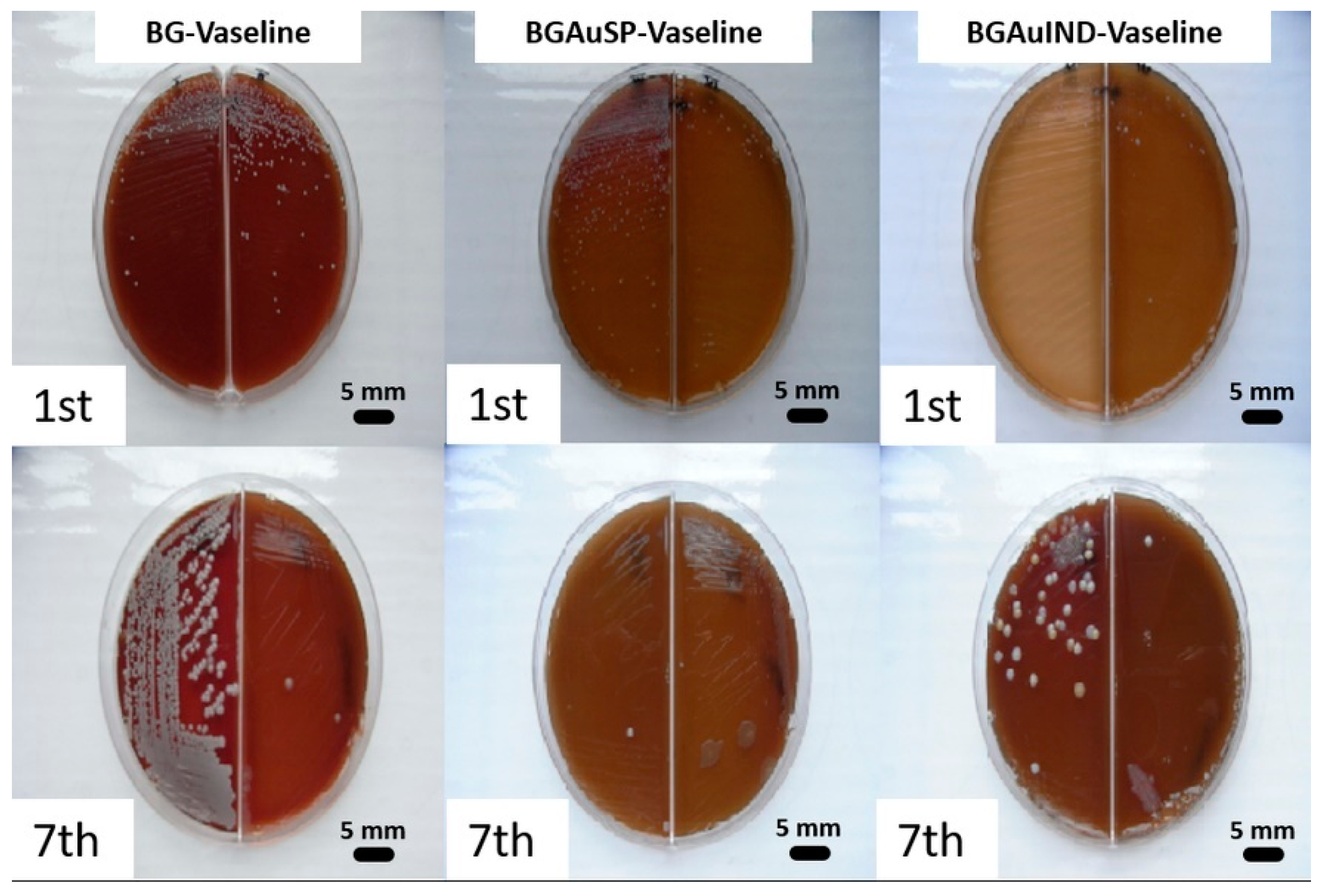
2.2. Immunological and Bacteriological Assays
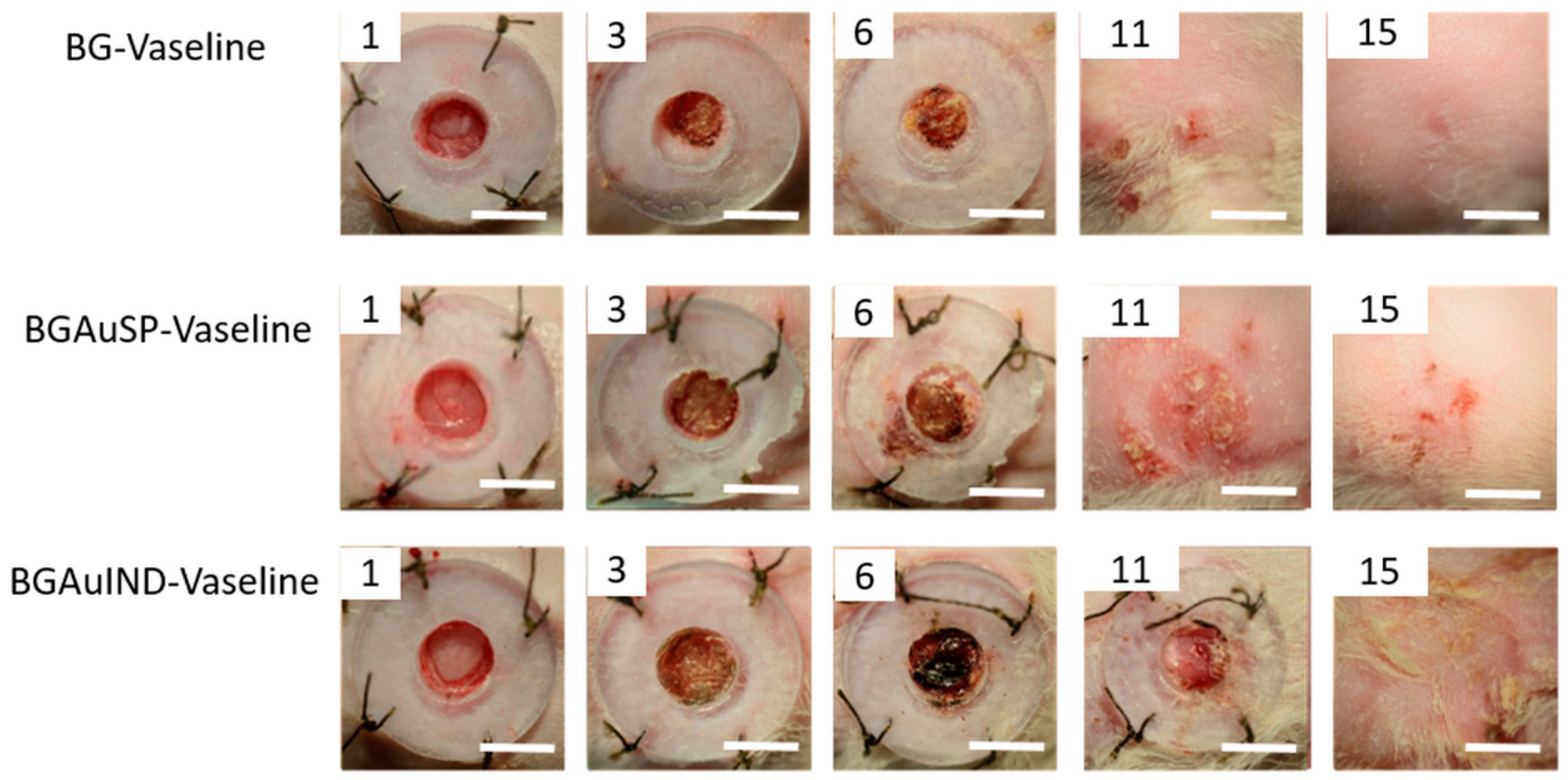
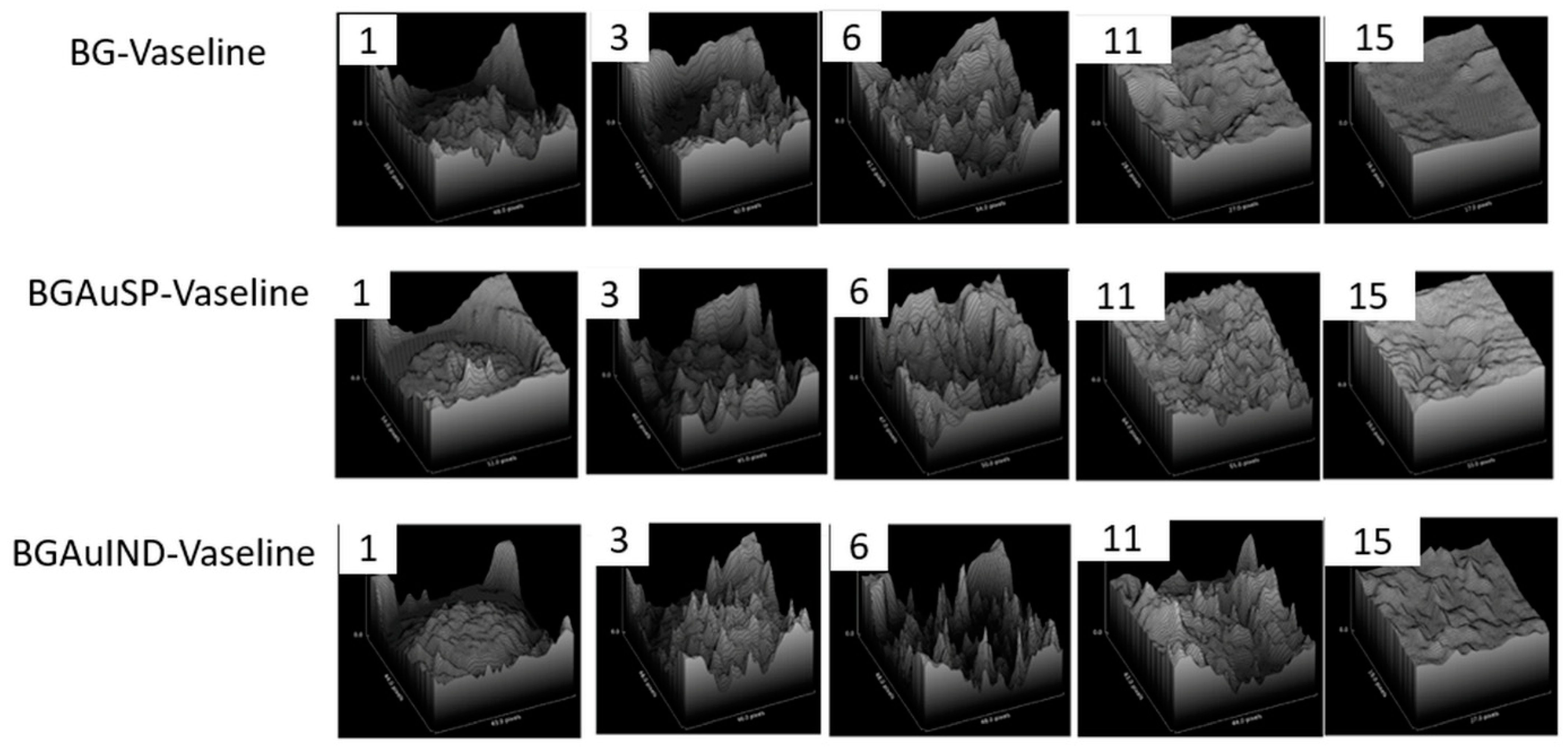
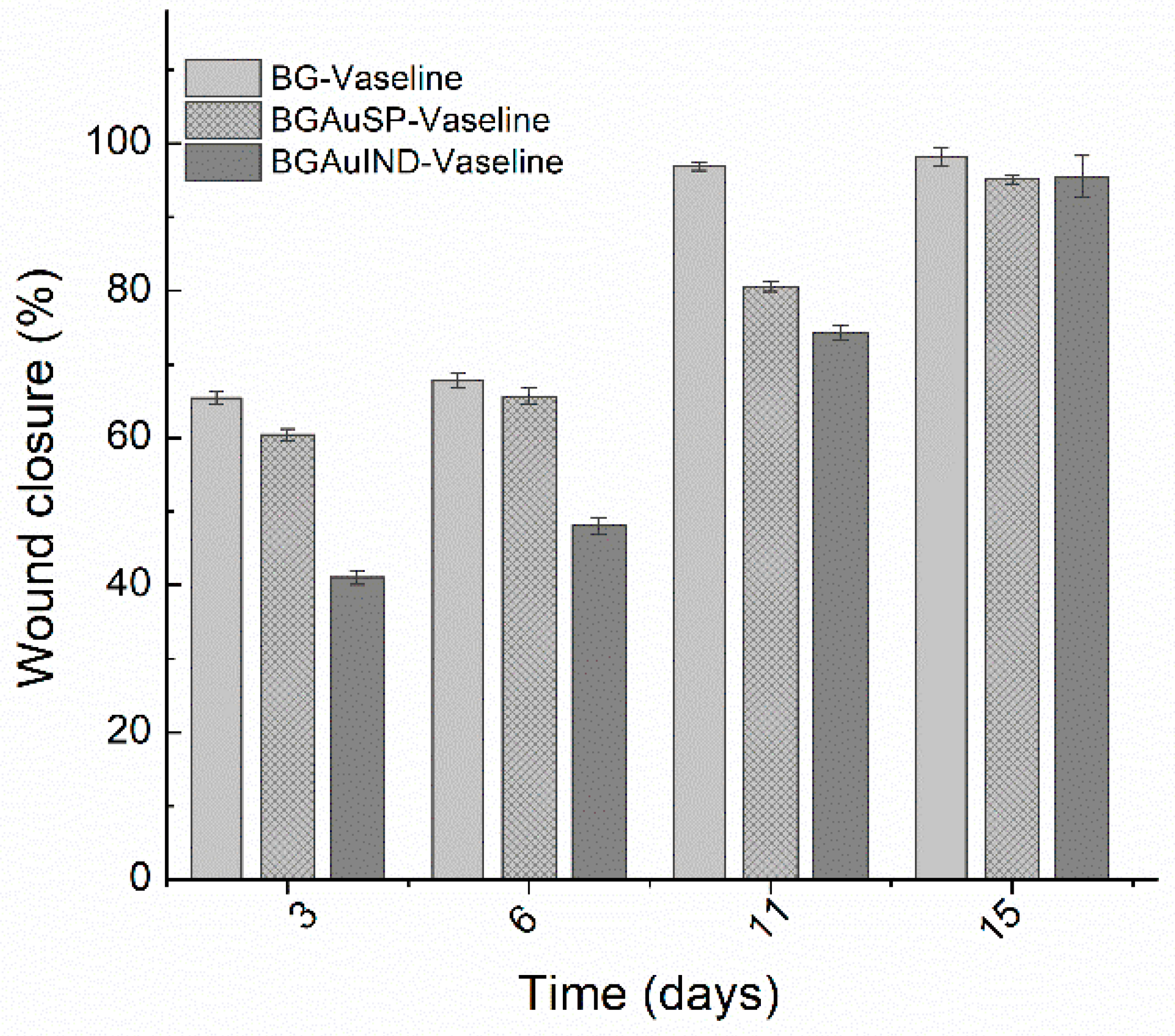
2.3. Skin Regeneration Assessment
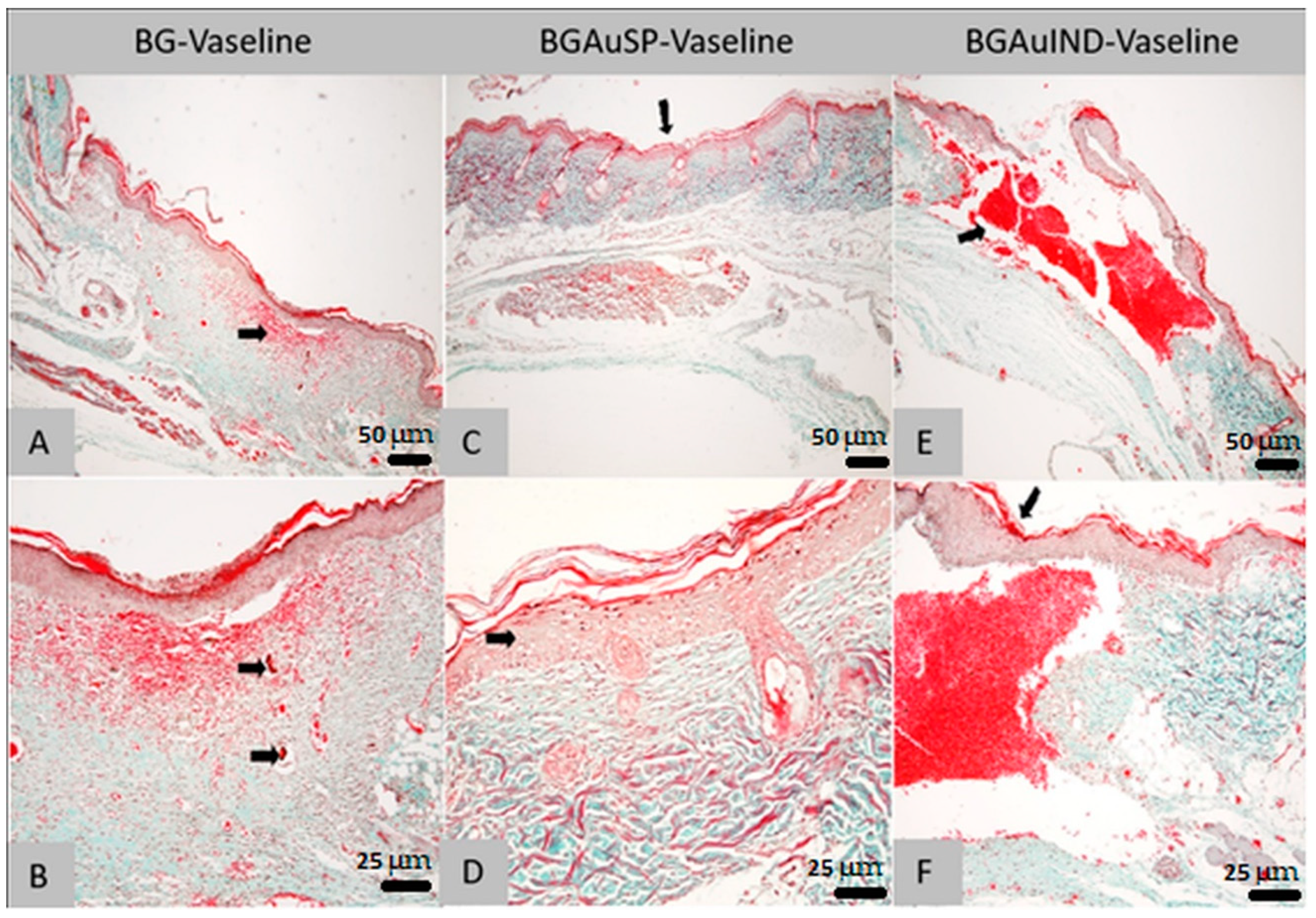
2.4. Histopathological Analysis of Skin Defects during the Wound Healing Process (Day 8)
2.4.1. Histological Analysis on Day 8 of Lot 1 Treated with BG-Vaseline
2.4.2. Histological Analysis on Day 8 of Lot 2 Treated with BGAuSP-Vaseline
2.4.3. Histological Analysis on Day 8 of Lot 3 Treated with BGAuIND-Vaseline
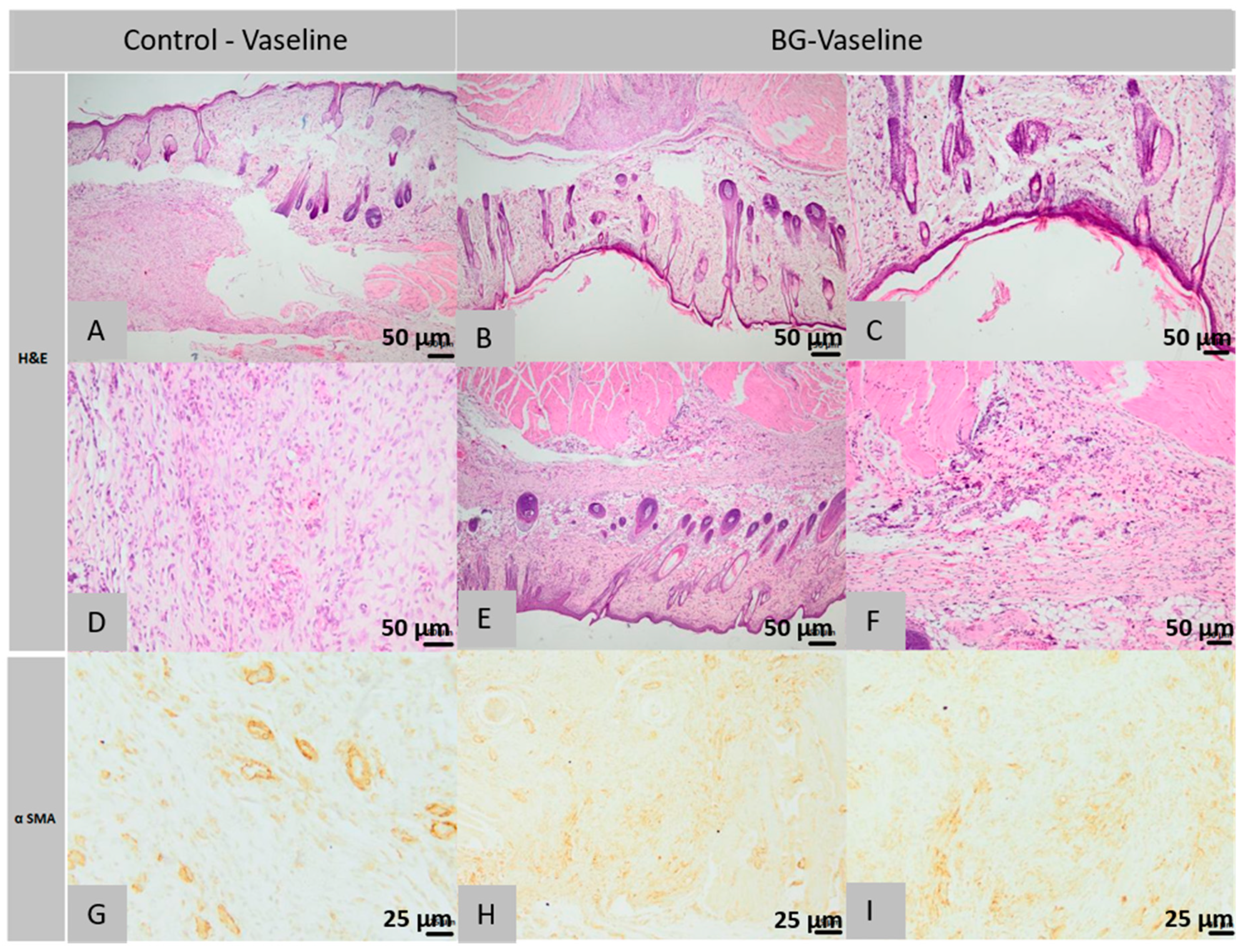
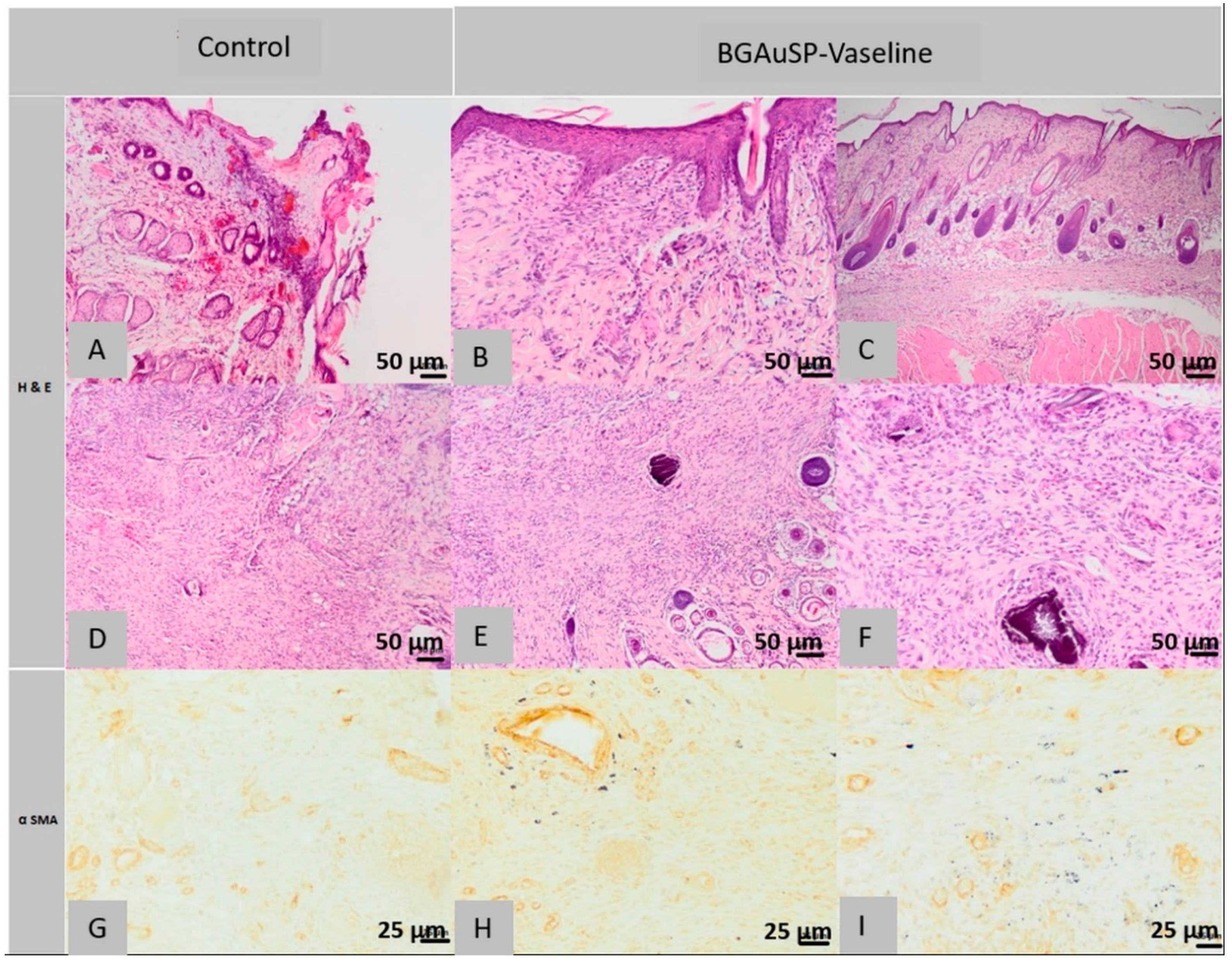
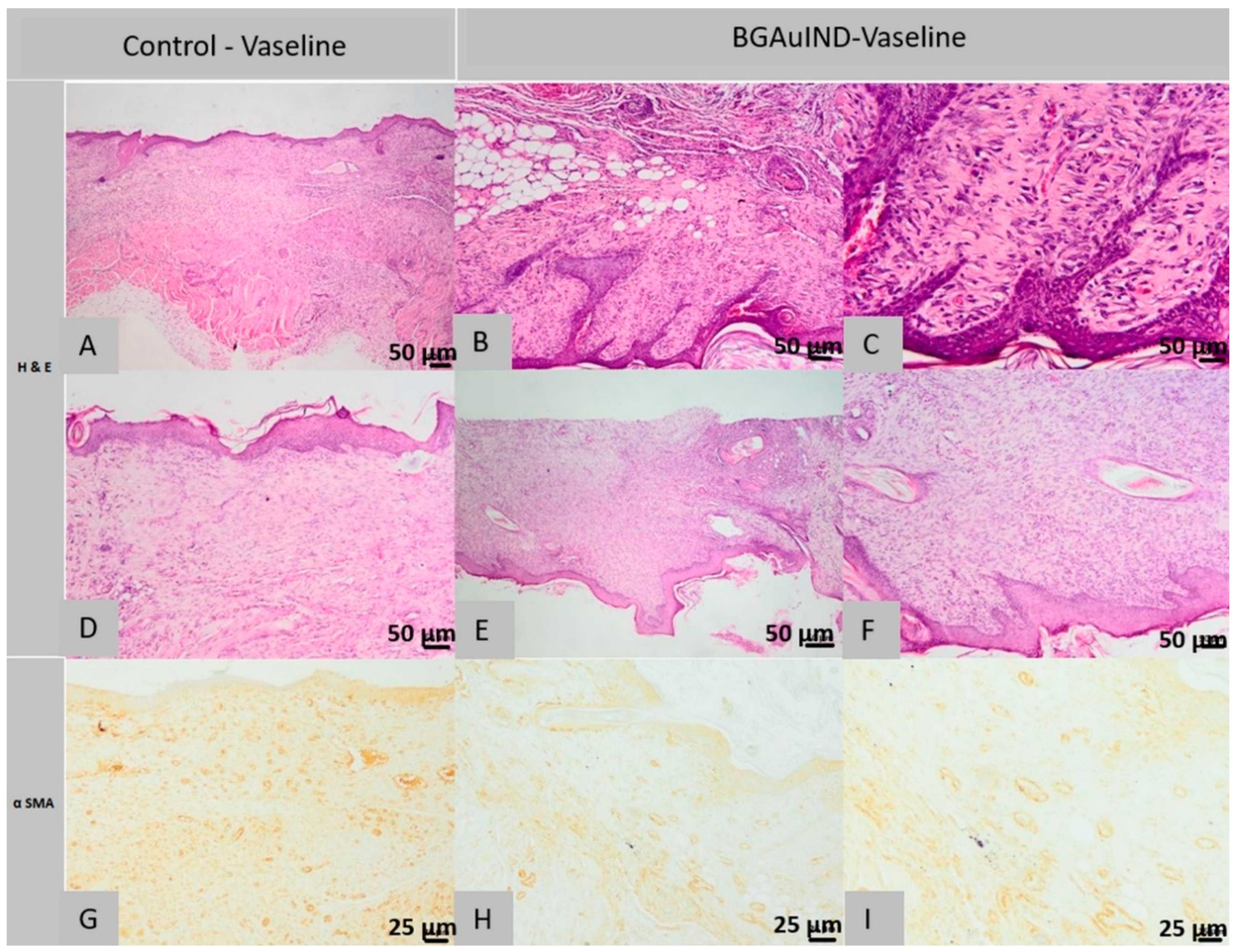
2.5. Histopathology and Immunohistochemical Analysis
2.5.1. Histological Analysis of Lot 1 Treated with BG-Vaseline
2.5.2. Histological Analysis of Lot 2 Treated with BGAuSP-Vaseline
2.5.3. Histological Analysis of Lot 3 Treated with BGAuIND-Vaseline
3. Materials and Methods
3.1. Materials
3.2. Glass Composite-Ointments Preparation and Characterization
3.2.1. The Synthesis and Characterization of Glass Composites with AuNPs
3.2.2. Glass Composite Ointments Formation
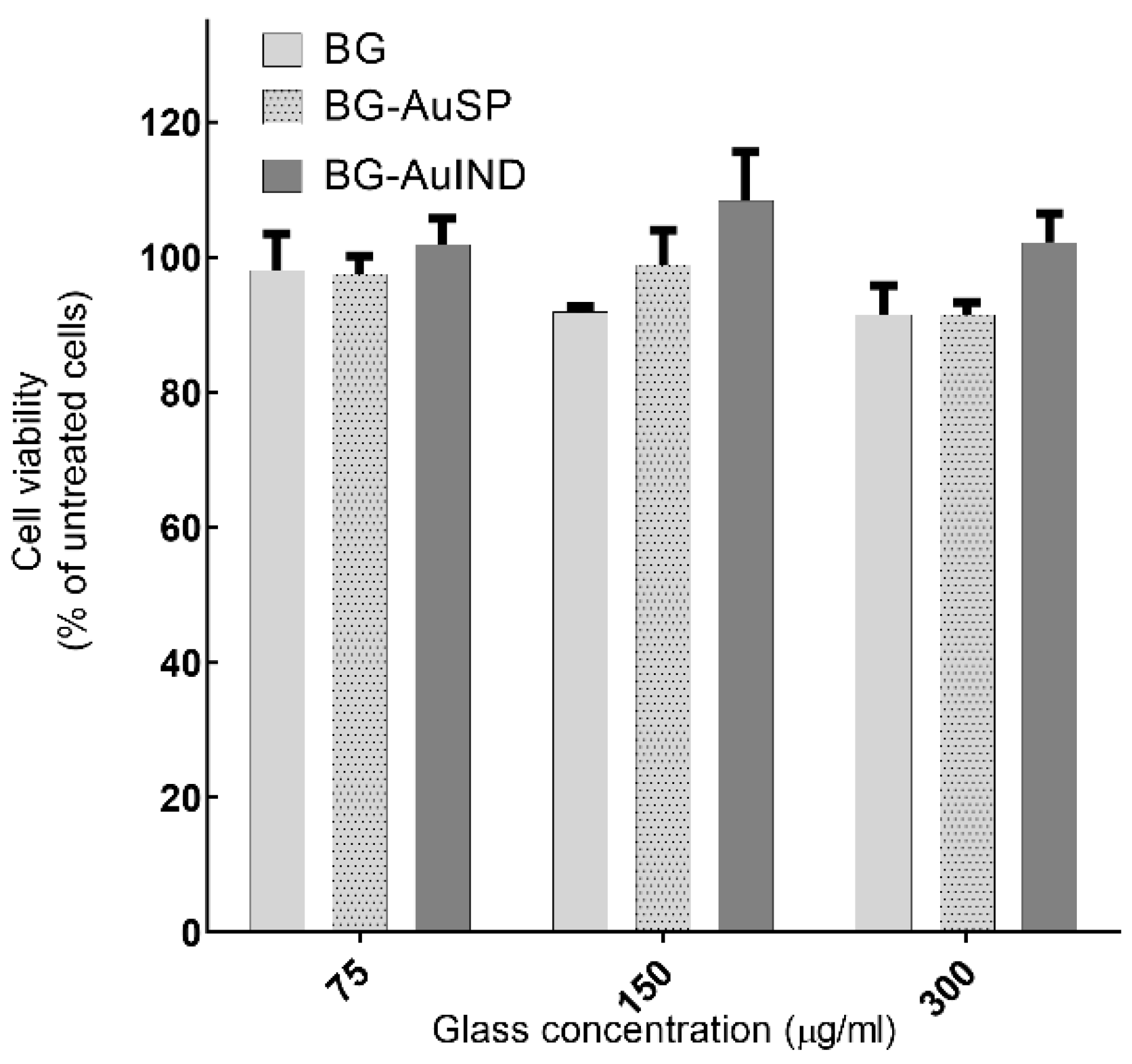
3.2.3. In Vitro Cell Viability of Glass Composites with AuNPs
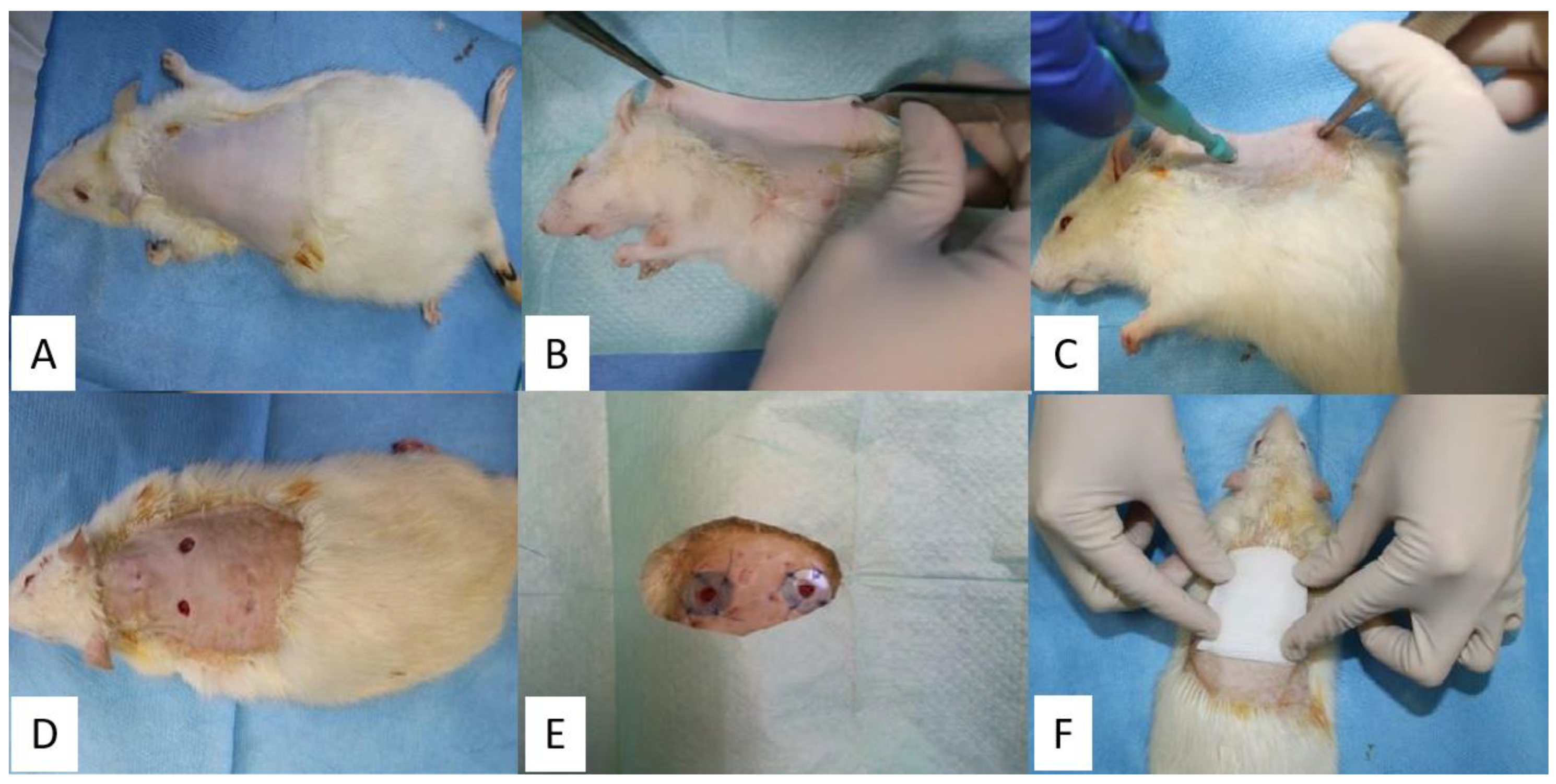
3.3. Animal Care and Use
3.4. Surgical Procedure
3.5. Immunological Assays
3.6. Bacteriological Test
3.7. Measurements of Wound Size Reduction
3.8. Histological and Immunohistological Methods
3.9. Statistical Analysis
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| BG | bioactive glasses |
| AuNPs | gold nanoparticles |
| AuSP | spherical gold nanoparticles |
| AuIND | spherical gold nanocages |
| HaCaT | human keratinocytes cells |
| BGAuSP | bioactive glasses with spherical gold nanoparticles |
| BGAuIND | bioactive glasses with spherical gold nanocages |
| DLS | dynamic light scattering |
| SMA | smooth muscle actin |
References
- Sullivan, T.P.; Eaglstein, W.H.; Davis, S.C.; Mertz, P. The pig as a model for human wound healing. Wound Repair Regen. 2001, 9, 66–76. [Google Scholar] [CrossRef] [PubMed]
- Takzare, N.; Hosseini, M.J.; Hasanzadeh, G.; Mortazavi, H.; Takzare, A.; Habibi, P. Influence of Aloe Vera gel on dermal wound healing process in rat. Toxicol. Mech. Methods 2009, 19, 73–77. [Google Scholar] [CrossRef] [PubMed]
- Shrivastav, A.; Mishra, A.K.; Ali, S.S.; Ahmad, A.; Abuzinadah, M.F.; Khan, N.A. In vivo models for assesment of wound healing potential: A systematic review. Wound Med. 2018, 20, 43–53. [Google Scholar] [CrossRef]
- Baltzis, D.; Eleftheriadou, I.; Veves, A. Pathogenesis and treatment of impaired wound healing in diabetes mellitus: New insights. Adv. Ther. 2014, 31, 817–836. [Google Scholar] [CrossRef]
- Ashtikar, M.; Wacker, M.G. Nanopharmaceuticals for wound healing—Lost in translation? Adv. Drug Deliv. Rev. 2018, 129, 194–218. [Google Scholar] [CrossRef]
- Nie, Z.; Liu, K.J.; Zhong, C.J.; Wang, L.F.; Yang, Y.; Tian, Q.; Liu, Y. Enhanced radical scavenging activity by antioxidant-functionalized gold nanoparticles: A novel inspiration for development of new artificial antioxidants. Free Radic. Biol. Med. 2007, 43, 1243–1254. [Google Scholar] [CrossRef]
- Leu, J.G.; Chen, S.A.; Chen, H.M.; Wu, W.M.; Hung, C.F.; Yao, Y.-D.; Tu, C.S.; Liang, Y.J. The effects of gold nanoparticles in wound healing with antioxidant epigallocatechin gallate and α-lipoic acid. Nanomed. Nanotechnol. Biol. Med. 2012, 8, 767–775. [Google Scholar] [CrossRef]
- Pan, Y.; Neuss, S.; Leifert, A.; Fischler, M.; Wen, F.; Simon, U.; Schmid, G.; Brandau, W.; Jahnen-Dechent, W. Size-dependent cytotoxicity of gold nanoparticles. Small 2007, 3, 1941–1949. [Google Scholar] [CrossRef]
- Dykman, L.A.; Khlebtsov, N.G. Gold nanoparticles in biology and medicine: Recent advances and prospects. Acta Nat. 2011, 3, 34–55. [Google Scholar] [CrossRef]
- Homaeigohar, S.; Boccaccini, A.R. Antibacterial biohybrid nanofibers for wound dressings. Acta Biomater. 2020, 107, 25–49. [Google Scholar] [CrossRef]
- Hernández-Sierra, J.F.; Ruiz, F.; Cruz Pena, D.C.; Martínez-Gutiérrez, F.; Martínez, A.E.; de Jesús Pozos Guillén, A.; Tapia-Pérez, H.; Martínez Castañón, G. The antimicrobial sensitivity of Streptococcus mutans to nanoparticles of silver, zinc oxide, and gold. Nanomed. Nanotechnol. Biol. Med. 2008, 4, 237–240. [Google Scholar] [CrossRef] [PubMed]
- Magyari, K.; Tóth, Z.R.; Pap, Z.; Licarete, E.; Vodnar, D.C.; Todea, M.; Gyulavári, T.; Hernadi, K.; Baia, L. Insights into the effect of gold nanospheres, nanotriangles and spherical nanocages on the structural, morphological and biological properties of bioactive glasses. J. Non-Cryst. Solids 2019, 522, 119552. [Google Scholar] [CrossRef]
- Zhang, Y.; Shareena Dasari, T.P.; Deng, H.; Yu, H. Antimicrobial activity of gold nanoparticles and ionic gold. J. Environ. Sci. Health Part C Environ. Carcinog. Ecotoxicol. Rev. 2015, 33, 286–327. [Google Scholar] [CrossRef] [PubMed]
- Paladini, F.; Pollini, M. Antimicrobial silver nanoparticles for wound healing application: Progress and future trends. Materials (Basel) 2019, 12, 2540. [Google Scholar] [CrossRef]
- Mohseni, M.; Shamloo, A.; Aghababaie, Z.; Afjoul, H.; Abdi, S.; Moravvej, H.; Vossoughi, M. A comparative study of wound dressings loaded with silver sulfadiazine and silver nanoparticles: In vitro and in vivo evaluation. Int. J. Pharm. 2019, 564, 350–358. [Google Scholar] [CrossRef]
- Tóth, Z.R.; Kovács, G.; Hernádi, K.; Baia, L.; Pap, Z. The investigation of the photocatalytic efficiency of spherical gold nanocages/TiO2and silver nanospheres/TiO2composites. Sep. Purif. Technol. 2017, 183, 216–225. [Google Scholar] [CrossRef]
- Mârza, S.M.; Magyari, K.; Bogdan, S.; Moldovan, M.; Peştean, C.; Nagy, A.; Tǎbǎran, F.; Licarete, E.; Suarasan, S.; Dreanca, A.; et al. Skin wound regeneration with bioactive glass-gold nanoparticles ointment. Biomed. Mater. 2019, 14, 025011. [Google Scholar] [CrossRef]
- Chang, R.K.; Raw, A.; Lionberger, R.; Yu, L. Generic development of topical dermatologic products: Formulation development, process development, and testing of topical dermatologic products. AAPS J. 2013, 15, 41–52. [Google Scholar] [CrossRef]
- Magyari, K.; Nagy-Simon, T.; Vulpoi, A.; Popescu, R.A.; Licarete, E.; Stefan, R.; Hernádi, K.; Papuc, I.; Baia, L. Novel bioactive glass-AuNP composites for biomedical applications. Mater. Sci. Eng. C 2017, 76, 752–759. [Google Scholar] [CrossRef]
- Yamamoto, Y.; Fukami, T.; Koide, T.; Suzuki, T.; Hiyama, Y.; Tomono, K. Pharmaceutical evaluation of steroidal ointments by ATR-IR chemical imaging: Distribution of active and inactive pharmaceutical ingredients. Int. J. Pharm. 2012, 426, 54–60. [Google Scholar] [CrossRef]
- Aguiar, H.; Serra, J.; González, P.; León, B. Structural study of sol-gel silicate glasses by IR and Raman spectroscopies. J. Non. Cryst. Solids 2009, 355, 475–480. [Google Scholar] [CrossRef]
- Min, J.H.; Patel, M.; Koh, W.G. Incorporation of conductive materials into hydrogels for tissue engineering applications. Polymers (Basel) 2018, 10, 1078. [Google Scholar] [CrossRef] [PubMed]
- Pirii, L.E.; Friedrich, A.W.; Rossen, J.W.A.; Vogels, W.; Beerthuizen, G.I.J.M.; Nieuwenhuis, M.K.; Kooistra-Smid, A.M.D.; Bathoorn, E. Extensive colonization with carbapenemase-producing microorganisms in Romanian burn patients: Infectious consequences from the Colectiv fire disaster. Eur. J. Clin. Microbiol. Infect. Dis. 2018, 37, 175–183. [Google Scholar] [CrossRef] [PubMed]
- Frens, G. Controlled nucleation for the regulation of the particle size in monodisperse gold suspensions. Nat. Phys. Sci. Vol. 1973, 241, 20–22. [Google Scholar] [CrossRef]
- Popescu, R.A.; Magyari, K.; Vulpoi, A.; Trandafir, D.L.; Licarete, E.; Todea, M.; Ştefan, R.; Voica, C.; Vodnar, D.C.; Simon, S.; et al. Bioactive and biocompatible copper containing glass-ceramics with remarkable antibacterial properties and high cell viability designed for future: In vivo trials. Biomater. Sci. 2016, 4, 1252–1265. [Google Scholar] [CrossRef]
- Brown, R.E. The rodents Il: Suborder myomorpha. In Social Odours in Mammals; Brown, R.E., MacDonald, D., Eds.; Clarendon Press Oxford: Oxford, UK, 1985; Volume 1. [Google Scholar]
- Didion, J.P.; De Villena, F.P.M. Deconstructing mus gemischus: Advances in understanding ancestry, structure, and variation in the genome of the laboratory mouse. Mamm. Genome 2013, 24, 1–20. [Google Scholar] [CrossRef]
- ISO. Biological Evaluation of Medical Devices—Part 2: Animal Welfare Requirements, 2nd ed.; ISO: Geneva, Switzerland, 2006. [Google Scholar]
- Wang, X.; Ge, J.; Tredget, E.E.; Wu, Y. The mouse excisional wound splinting model, including applications for stem cell transplantation. Nat. Protoc. 2013, 8, 302–309. [Google Scholar] [CrossRef]
- Aragón-Sánchez, J.; Quintana-Marrero, Y.; Aragón-Hernández, C.; Hernández-Herero, M.J. ImageJ: A free, easy, and reliable method to measure leg ulcers using digital pictures. Int. J. Low. Extrem. Wounds 2017, 16, 269–273. [Google Scholar] [CrossRef]


| Samples | Control (SI%) | PHA M (SI%) | ConA (SI%) | LPS (SI%) |
|---|---|---|---|---|
| BG-Vaseline | 81.13 ± 0.53 | 81.24 ± 0.42 | 81.76 ± 0.73 | 81.66 ± 0.42 |
| BGAuSP-Vaseline | 74.8 ± 2.64 | 73.32 ± 0.1 | 72.59 ± 2.53 | 74.17 ± 1.16 |
| BGAuIND-Vaseline | 81.13 ± 0.53 | 81.76 ± 0.31 | 80.5 ± 1.58 | 82.08 ± 0.42 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mârza, S.M.; Magyari, K.; Bogdan, S.; Moldovan, M.; Peștean, C.; Nagy, A.; Gal, A.F.; Tăbăran, F.; Purdoiu, R.C.; Licărete, E.; et al. The Impact of Composites with Silicate-Based Glasses and Gold Nanoparticles on Skin Wound Regeneration. Molecules 2021, 26, 620. https://doi.org/10.3390/molecules26030620
Mârza SM, Magyari K, Bogdan S, Moldovan M, Peștean C, Nagy A, Gal AF, Tăbăran F, Purdoiu RC, Licărete E, et al. The Impact of Composites with Silicate-Based Glasses and Gold Nanoparticles on Skin Wound Regeneration. Molecules. 2021; 26(3):620. https://doi.org/10.3390/molecules26030620
Chicago/Turabian StyleMârza, Sorin M., Klara Magyari, Sidonia Bogdan, Mirela Moldovan, Cosmin Peștean, Andras Nagy, Adrian Florin Gal, Flaviu Tăbăran, Robert Cristian Purdoiu, Emilia Licărete, and et al. 2021. "The Impact of Composites with Silicate-Based Glasses and Gold Nanoparticles on Skin Wound Regeneration" Molecules 26, no. 3: 620. https://doi.org/10.3390/molecules26030620
APA StyleMârza, S. M., Magyari, K., Bogdan, S., Moldovan, M., Peștean, C., Nagy, A., Gal, A. F., Tăbăran, F., Purdoiu, R. C., Licărete, E., Suarasan, S., Baia, L., & Papuc, I. (2021). The Impact of Composites with Silicate-Based Glasses and Gold Nanoparticles on Skin Wound Regeneration. Molecules, 26(3), 620. https://doi.org/10.3390/molecules26030620








