Selective Extraction of Cannabinoid Compounds from Cannabis Seed Using Pressurized Hot Water Extraction
Abstract
1. Introduction
2. Materials and Methods
2.1. Chemicals and Reagents
2.2. Plant Material
2.3. Response Surface Methodology
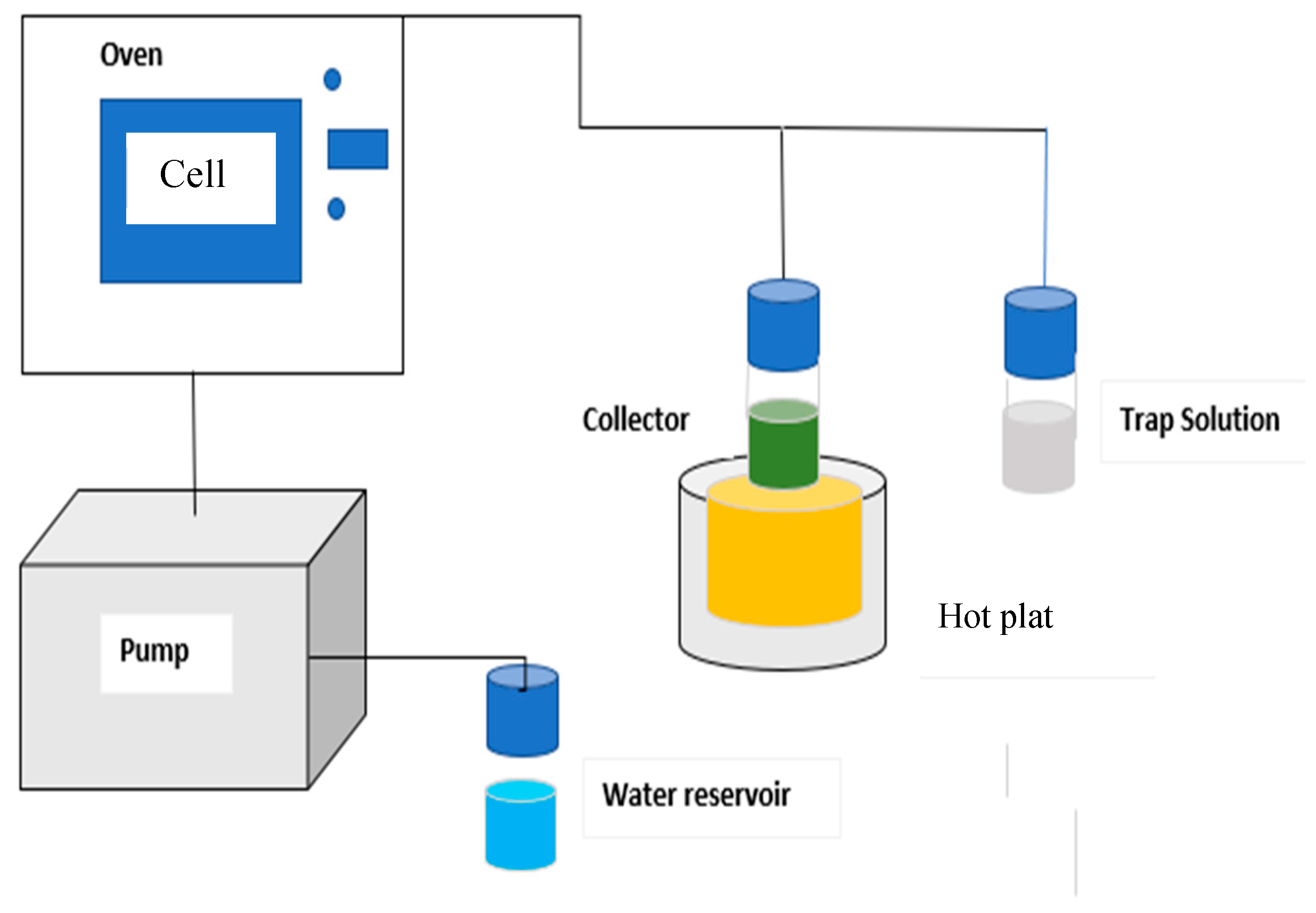
2.4. Pressurized Hot Water Extraction
2.5. GCXGC-TOFMS Method
2.6. Quality Assurance
2.7. Model Fitting and Predictive Efficiency
3. Results and Discussion
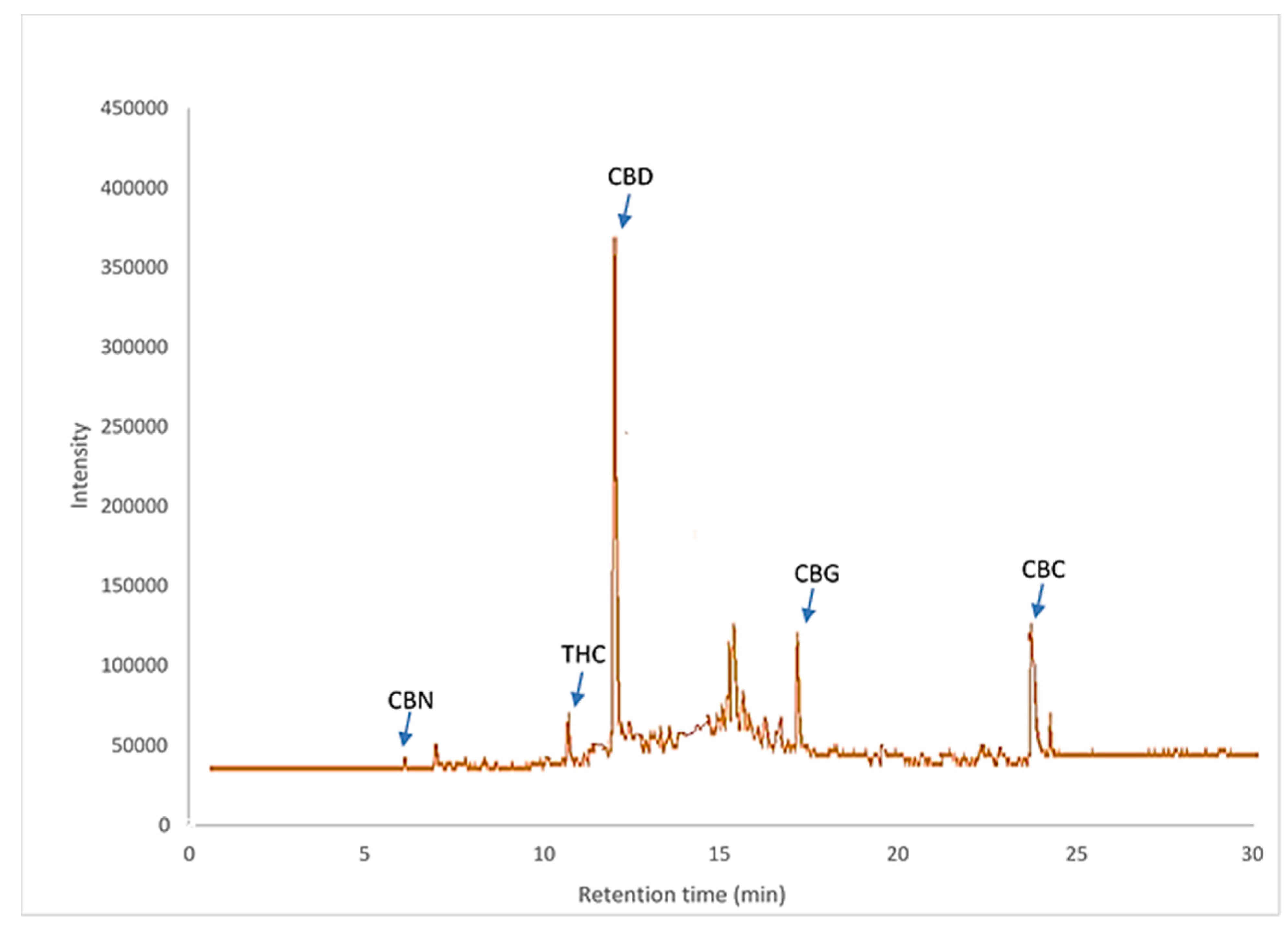
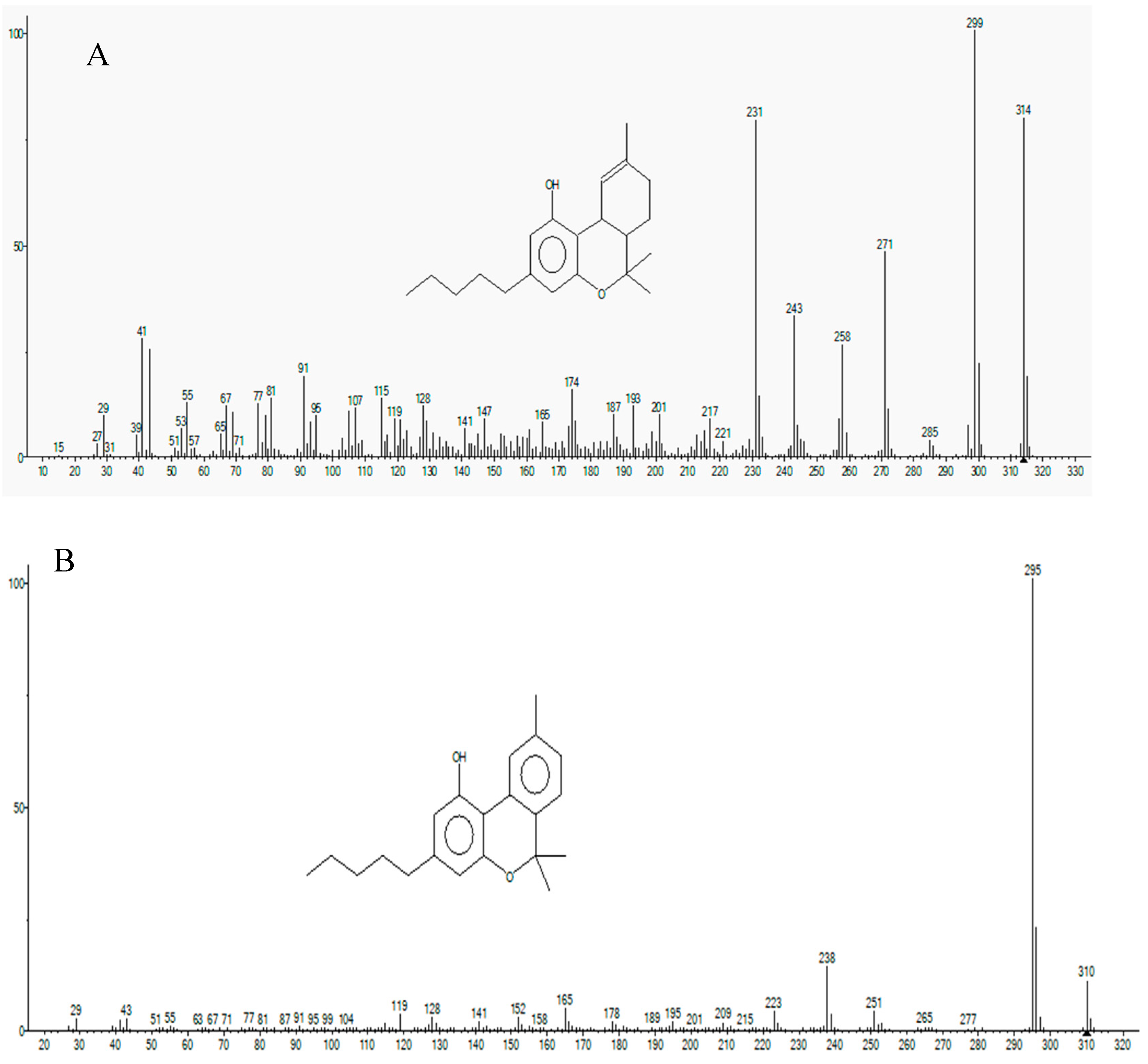
3.1. Identification of Cannabinoid Compounds
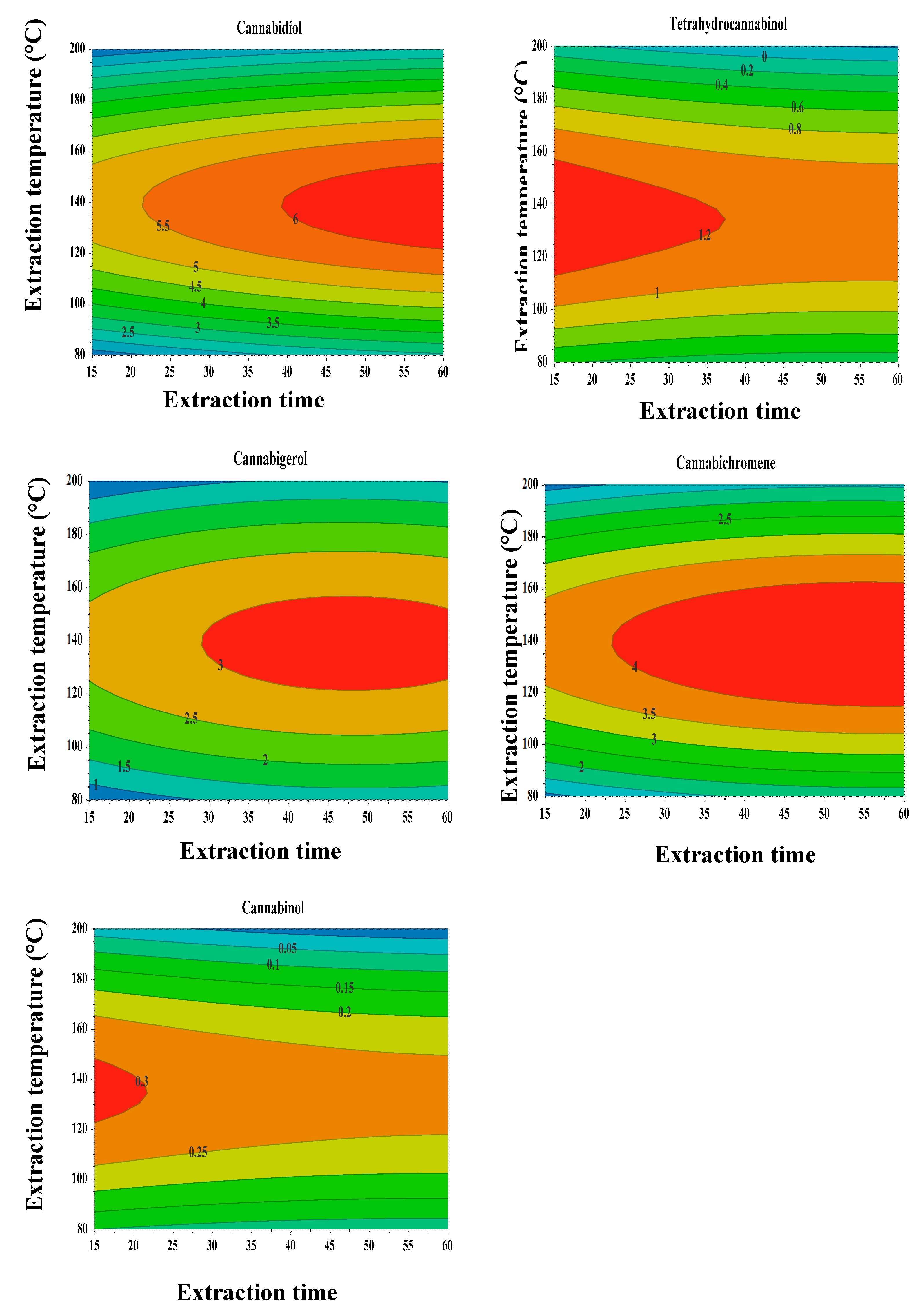
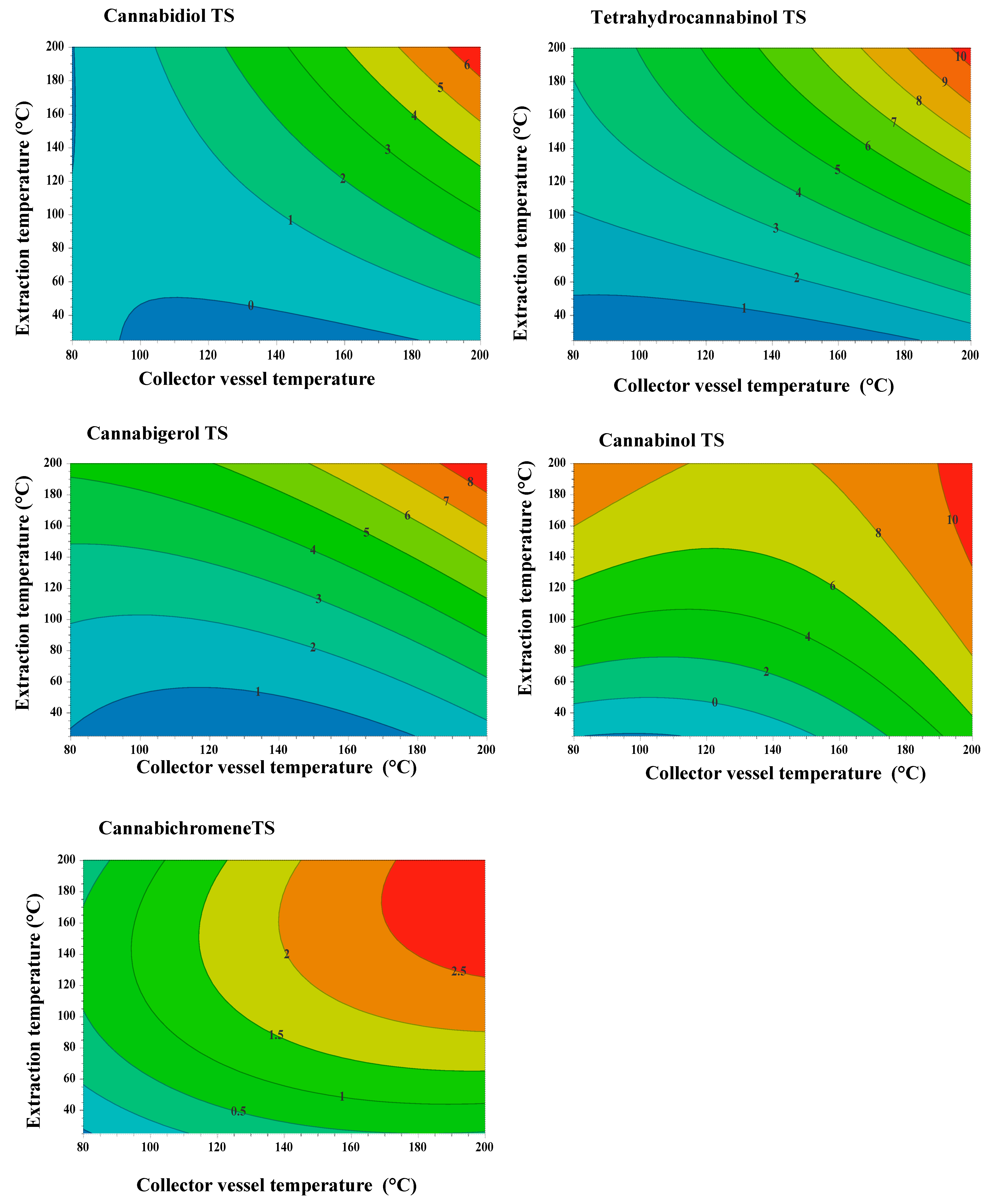
3.2. Experimental Design: Response Surface Methodology
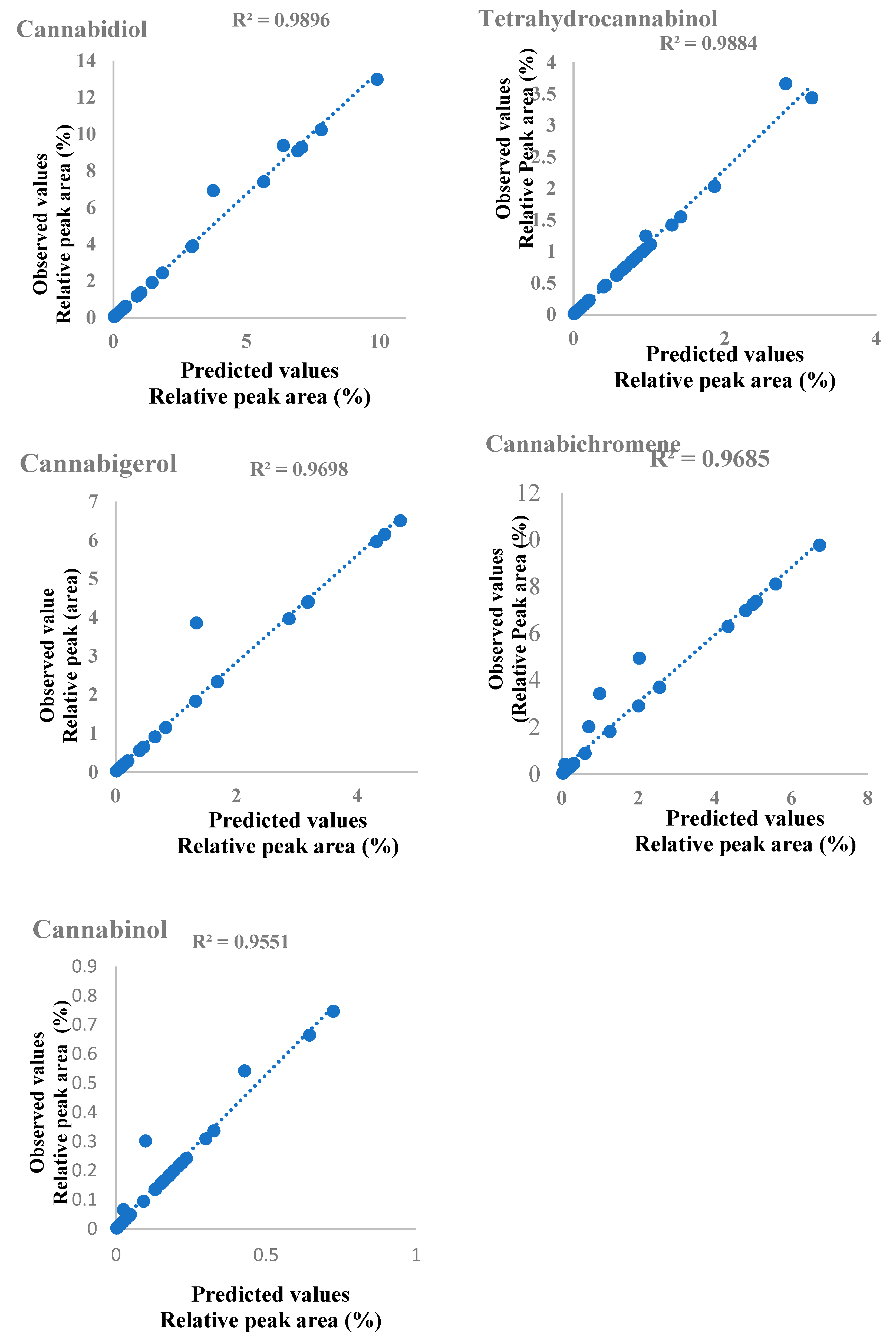
3.3. Model Fitting and Predictive Efficiency Analysis
3.4. Universal Extraction Condition of an Extract rich in CBD, CBC, and CBG
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Yoshimatsu, K.; Iida, O.; Kitazawa, T.; Sekine, T.; Kojoma, M.; Makino, Y.; Kiuchi, F. Growth Characteristics of Cannabis sativa L. Cultivated in a Phytotron and in the Field. Bull. Natl. Inst. Health Sci. 2004, 122, 16–20. [Google Scholar]
- Lafaye, G.; Karila, L.; Blecha, L.; Benyamina, A. Cannabis, cannabinoids, and health. Dialog-Clin. Neurosci. 2007, 19, 309–316. [Google Scholar]
- Li, H.-L. The origin and use of cannabis in eastern asia linguistic-cultural implications. Econ. Bot. 1974, 28, 293–301. [Google Scholar] [CrossRef]
- Cai, C.; Yu, W.; Wang, C.; Liu, L.; Li, F.; Tan, Z. Green extraction of cannabidiol from industrial hemp (Cannabis sativa L.) using deep eutectic solvents coupled with further enrichment and recovery by macroporous resin. J. Mol. Liq. 2019, 287, 110957. [Google Scholar] [CrossRef]
- Ramer, R.; Bublitz, K.; Freimuth, N.; Merkord, J.; Rohde, H.; Haustein, M.; Borchert, P.; Schmuhl, E.; Linnebacher, M.; Hinz, B. Cannabidiol inhibits lung cancer cell invasion and metastasis via intercellular adhesion molecule-1. FASEB J. 2011, 26, 1535–1548. [Google Scholar] [CrossRef] [PubMed]
- De Backer, B.; Debrus, B.; Lebrun, P.; Theunis, L.; Dubois, N.; Decock, L.; Verstraete, A.; Hubert, P.; Charlier, C. Innovative development and validation of an HPLC/DAD method for the qualitative and quantitative determination of major cannabinoids in cannabis plant material. J. Chromatogr. B 2009, 877, 4115–4124. [Google Scholar] [CrossRef] [PubMed]
- Russo, E. History of Cannabis and Its Preparations in Saga, Science, and Sobriquet. Chem. Biodivers. 2007, 4, 1614–1648. [Google Scholar] [CrossRef]
- Takeda, S.; Okajima, S.; Miyoshi, H.; Yoshida, K.; Okamoto, Y.; Okada, T.; Amamoto, T.; Watanabe, K.; Omiecinski, C.J.; Aramaki, H. Cannabidiolic acid, a major cannabinoid in fiber-type cannabis, is an inhibitor of MDA-MB-231 breast cancer cell migration. Toxicol. Lett. 2012, 214, 314–319. [Google Scholar] [CrossRef]
- Pavlovic, R.; Nenna, G.; Calvi, L.; Panseri, S.; Borgonovo, G.; Giupponi, L.; Cannazza, G.; Giorgi, A. Quality Traits of “Cannabidiol Oils”: Cannabinoids Content, Terpene Fingerprint and Oxidation Stability of European Commercially Available Preparations. Molecules 2018, 23, 1230. [Google Scholar] [CrossRef]
- Bal, V.H.; Lord, C. Replication of Standardized ADOS Domain Scores in the Simons Simplex Collection. Autism Res. 2015, 8, 583–592. [Google Scholar] [CrossRef]
- Casiraghi, A.; Roda, G.; Casagni, E.; Cristina, C.; Musazzi, U.M.; Franzè, S.; Rocco, P.; Giuliani, C.; Fico, G.; Minghetti, P.; et al. Extraction Method and Analysis of Cannabinoids in Cannabis Olive Oil Preparations. Planta Medica 2017, 84, 242–249. [Google Scholar] [CrossRef]
- Dean, J.R. Pre- and Post-Extraction Considerations. In Extraction Techniques in Analytical Sciences; Willey: Hoboken, NJ, USA, 2010; pp. 1–36. [Google Scholar]
- Rajaei, A.; Barzegar, M.; Yamini, Y. Supercritical fluid extraction of tea seed oil and its comparison with solvent extraction. Eur. Food Res. Technol. 2004, 220, 401–405. [Google Scholar] [CrossRef]
- Agarwal, C.; Máthé, K.; Hofmann, T.; Csoka, L. Ultrasound-Assisted Extraction of Cannabinoids from Cannabis Sativa L. Optimized by Response Surface Methodology. J. Food Sci. 2018, 83, 700–710. [Google Scholar] [CrossRef] [PubMed]
- Omar, J.; Olivares, M.; Alzaga, M.; Etxebarria, N. Optimisation and characterisation of marihuana extracts obtained by supercritical fluid extraction and focused ultrasound extraction and retention time locking GC-MS. J. Sep. Sci. 2013, 36, 1397–1404. [Google Scholar] [CrossRef] [PubMed]
- Devi, V.; Shabina, K. Comparative study of different extraction processes for hemp (Cannabis sativa) seed oil considering physical, chemical and industrial-scale economic aspects. J. Clean. Prod. 2019, 207, 645–657. [Google Scholar] [CrossRef]
- Chemat, F.; Vian, M.A.; Cravotto, G. Green Extraction of Natural Products: Concept and Principles. Int. J. Mol. Sci. 2012, 13, 8615–8627. [Google Scholar] [CrossRef] [PubMed]
- Pellati, F.; Brighenti, V.; Sperlea, J.; Marchetti, L.; Bertelli, D.; Benvenuti, S. New Methods for the Comprehensive Analysis of Bioactive Compounds in Cannabis sativa L. (hemp). Molecules 2018, 23, 2639. [Google Scholar] [CrossRef]
- Ramirez, C.L.; Fanovich, M.A.; Churio, M.S. Cannabinoids: Extraction Methods, Analysis, and Physicochemical Characterization. Stud. Nat. Prod. Chem. 2019, 61, 143–173. [Google Scholar]
- Romano, L.L.; Hazekamp, A. Cannabis Oil: Chemical evaluation of an upcoming cannabis-based medicin. Cannabinoids 2013, 1, 1–11. [Google Scholar]
- Bergamaschi, M.M.; Barnes, A.; Queiroz, R.H.C.; Hurd, Y.L.; Huestis, M.A. Impact of enzymatic and alkaline hydrolysis on CBD concentration in urine. Anal. Bioanal. Chem. 2013, 405, 4679–4689. [Google Scholar] [CrossRef]
- Plaza, M.; Turner, C. Pressurized hot water extraction of bioactives. TrAC Trends Anal. Chem. 2015, 71, 39–54. [Google Scholar] [CrossRef]
- Hawthorne, S.B.; Yang, Y.; Miller, D.J. Extraction of Organic Pollutants from Environmental Solids with Sub- and Supercritical Water. Anal. Chem. 1994, 66, 2912–2920. [Google Scholar] [CrossRef]
- Nuapia, Y.; Cukrowska, E.; Tutu, H.; Chimuka, L. Statistical comparison of two modeling methods on pressurized hot water extraction of vitamin C and phenolic compounds from Moringa oleifera leaves. S. Afr. J. Bot. 2018. [Google Scholar] [CrossRef]
- Teo, C.C.; Tan, S.N.; Yong, J.W.H.; Hew, C.S.; Ong, E.S. Pressurized hot water extraction (PHWE). J. Chromatogr. A 2010, 1217, 2484–2494. [Google Scholar] [CrossRef] [PubMed]
- Djande, C.H.; Piater, L.; Steenkamp, P.; Madala, N.; Dubery, I. Differential extraction of phytochemicals from the multipurpose tree, Moringa oleifera, using green extraction solvents. S. Afr. J. Bot. 2018, 115, 81–89. [Google Scholar] [CrossRef]
- Perrotin-Brunel, H.; Kroon, M.C.; Van Roosmalen, M.J.; Van Spronsen, J.; Peters, C.J.; Witkamp, G.J. Solubility of non-psychoactive cannabinoids in supercritical carbon dioxide and comparison with psychoactive cannabinoids. J. Supercrit. Fluids 2010, 55, 603–608. [Google Scholar] [CrossRef]
- Grijó, D.; Osorio, I.A.V.; Cardozo-Filho, L. Supercritical extraction strategies using CO2 and ethanol to obtain cannabinoid compounds from Cannabis hybrid flowers. J. CO2 Util. 2018, 28, 174–180. [Google Scholar] [CrossRef]
- Mariotti, K.D.C.; Marcelo, M.C.A.; Ortiz, R.S.; Borille, B.T.; Dos Reis, M.; Fett, M.S.; Ferrão, M.; Limberger, R.P.; Information, P.E.K.F.C. Seized cannabis seeds cultivated in greenhouse: A chemical study by gas chromatography–mass spectrometry and chemometric analysis. Sci. Justice 2016, 56, 35–41. [Google Scholar] [CrossRef]
Sample Availability: Cannabis samples are available from the authors. |


| Run Order | Extraction Time (min) | Extraction Temp. (°C) | Collector Temp.(°C) | THC (%) | CBD (%) | CBC (%) | CBG (%) | CBN (%) |
|---|---|---|---|---|---|---|---|---|
| 19 | 15 | 80 | 25 | 0.5700 | 0.2989 | 0.2033 | 0.1345 | 0.1308 |
| 25 | 37.5 | 80 | 25 | 0.7900 | 0.4589 | 0.3121 | 0.2065 | 0.1812 |
| 7 | 60 | 80 | 25 | 0.9584 | 0.8978 | 0.6105 | 0.4040 | 0.2204 |
| 1 | 15 | 80 | 113 | 0.3989 | 0.2890 | 0.1965 | 0.1301 | 0.0917 |
| 4 | 37.5 | 80 | 113 | 0.9088 | 0.4556 | 0.3098 | 0.2050 | 0.2091 |
| 9 | 60 | 80 | 113 | 0.4278 | 0.8926 | 0.6070 | 0.4017 | 0.0984 |
| 21 | 15 | 80 | 200 | 0.2078 | 0.2699 | 0.1835 | 0.1215 | 0.0478 |
| 5 | 37.5 | 80 | 200 | 0.1889 | 0.3174 | 0.2158 | 0.1428 | 0.0435 |
| 18 | 60 | 80 | 200 | 0.1089 | 0.3878 | 0.2637 | 0.1745 | 0.0251 |
| 12 | 15 | 140 | 25 | 3.1504 | 1.4589 | 0.9921 | 0.6565 | 0.7246 |
| 10 | 37.5 | 140 | 25 | 2.8062 | 2.9819 | 2.0277 | 1.3419 | 0.6454 |
| 29 | 60 | 140 | 25 | 1.8639 | 3.7532 | 2.5522 | 1.6889 | 0.4287 |
| 15 | 15 | 140 | 113 | 1.4193 | 6.3856 | 4.3422 | 2.8735 | 0.3264 |
| 16 | 37.5 | 140 | 113 | 1.3032 | 7.0702 | 4.8077 | 3.1816 | 0.2997 |
| 20 | 60 | 140 | 113 | 1.0187 | 9.9046 | 6.7351 | 4.4571 | 0.2343 |
| 6 | 15 | 140 | 200 | 0.9537 | 1.0347 | 0.7036 | 0.4656 | 0.2194 |
| 2 | 37.5 | 140 | 200 | 0.6547 | 1.8502 | 1.2581 | 0.8326 | 0.1506 |
| 30 | 60 | 140 | 200 | 0.5809 | 2.9506 | 2.0064 | 1.3278 | 0.1336 |
| 11 | 37.5 | 140 | 113 | 0.7637 | 6.9309 | 4.9930 | 4.3189 | 0.1757 |
| 27 | 37.5 | 140 | 113 | 0.8428 | 5.6506 | 5.0824 | 3.1928 | 0.1938 |
| 14 | 37.5 | 140 | 113 | 0.6904 | 7.8078 | 5.5893 | 4.7135 | 0.1588 |
| 17 | 15 | 200 | 25 | 0.1444 | 0.1229 | 0.0836 | 0.0553 | 0.0332 |
| 23 | 37.5 | 200 | 25 | 0.1015 | 0.3353 | 0.2280 | 0.1509 | 0.0233 |
| 22 | 60 | 200 | 25 | 0.0927 | 0.4536 | 0.3085 | 0.2041 | 0.0213 |
| 8 | 15 | 200 | 113 | 0.0702 | 0.1099 | 0.0747 | 0.0495 | 0.0162 |
| 26 | 37.5 | 200 | 113 | 0.0497 | 0.1653 | 0.1124 | 0.0744 | 0.0114 |
| 3 | 60 | 200 | 113 | 0.0312 | 0.2221 | 0.1510 | 0.0999 | 0.0072 |
| 13 | 15 | 200 | 200 | 0.0201 | 0.0384 | 0.0261 | 0.0173 | 0.0046 |
| 24 | 37.5 | 200 | 200 | 0.0148 | 0.0407 | 0.0277 | 0.0183 | 0.0034 |
| 28 | 60 | 200 | 200 | 0.0105 | 0.0751 | 0.0511 | 0.0338 | 0.0024 |
| Error | Equation |
|---|---|
| Absolute average deviation | |
| Root mean square error | RMSE |
| Mean absolute error | |
| Standard error of prediction (%) | SEP |
| Model predictive error (%) | MPE |
| Chi-square (χ2) | |
| Correlation R2 |
| Parameter | THC | CBD | CBC | CBG | CBN |
|---|---|---|---|---|---|
| AAD | 0.0067 | 0.0036 | 0.0021 | 0.0018 | 0.0014 |
| RMSE | 0.0400 | 0.0300 | 0.0600 | 0.0800 | 0.0700 |
| MAE | 0.0050 | 0.0020 | 0.0010 | 0.0020 | 0.0010 |
| SEP | 0.0120 | 0.0210 | 0.0140 | 0.0160 | 0.0190 |
| MPE | 0.0290 | 0.0160 | 0.0080 | 0.0090 | 0.0040 |
| χ2 | 0.0005 | 0.0002 | 0.0005 | 0.0003 | 0.0009 |
| Responses | Predicted Values | Experimental Value (n = 3) | Repeatability (%RSD) | Reproducibility (%RSD) |
|---|---|---|---|---|
| THC | 2.00 | 2.03 ± 0.20 | 90.23 ± 2.45 | 89.78 ± 2.34 |
| CBD | 5.40 | 5.60 ± 0.35 | 89.45 ± 2.87 | 91.34 ± 1.32 |
| CBC | 4.50 | 5.00 ± 0.60 | 92.45 ± 3.71 | 89.70 ± 5.60 |
| CBG | 3.50 | 4.10 ± 0.80 | 90.56 ± 3.56 | 92.56 ± 2.31 |
| CBN | 2.90 | 0.34 ± 0.09 | 92.61 ± 4.5 | 90.78 ± 2.19 |
| THCt */CBDt * | 0.17 | 0.18 | - | - |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Nuapia, Y.; Tutu, H.; Chimuka, L.; Cukrowska, E. Selective Extraction of Cannabinoid Compounds from Cannabis Seed Using Pressurized Hot Water Extraction. Molecules 2020, 25, 1335. https://doi.org/10.3390/molecules25061335
Nuapia Y, Tutu H, Chimuka L, Cukrowska E. Selective Extraction of Cannabinoid Compounds from Cannabis Seed Using Pressurized Hot Water Extraction. Molecules. 2020; 25(6):1335. https://doi.org/10.3390/molecules25061335
Chicago/Turabian StyleNuapia, Yannick, Hlanganani Tutu, Luke Chimuka, and Ewa Cukrowska. 2020. "Selective Extraction of Cannabinoid Compounds from Cannabis Seed Using Pressurized Hot Water Extraction" Molecules 25, no. 6: 1335. https://doi.org/10.3390/molecules25061335
APA StyleNuapia, Y., Tutu, H., Chimuka, L., & Cukrowska, E. (2020). Selective Extraction of Cannabinoid Compounds from Cannabis Seed Using Pressurized Hot Water Extraction. Molecules, 25(6), 1335. https://doi.org/10.3390/molecules25061335






