Evaluation of 1,2-Benzothiazine 1,1-Dioxide Derivatives In Vitro Activity towards Clinical-Relevant Microorganisms and Fibroblasts
Abstract
1. Introduction
2. Results
2.1. Chemistry
2.2. Biological Tests
3. Discussion
4. Materials and Methods
4.1. Chemistry
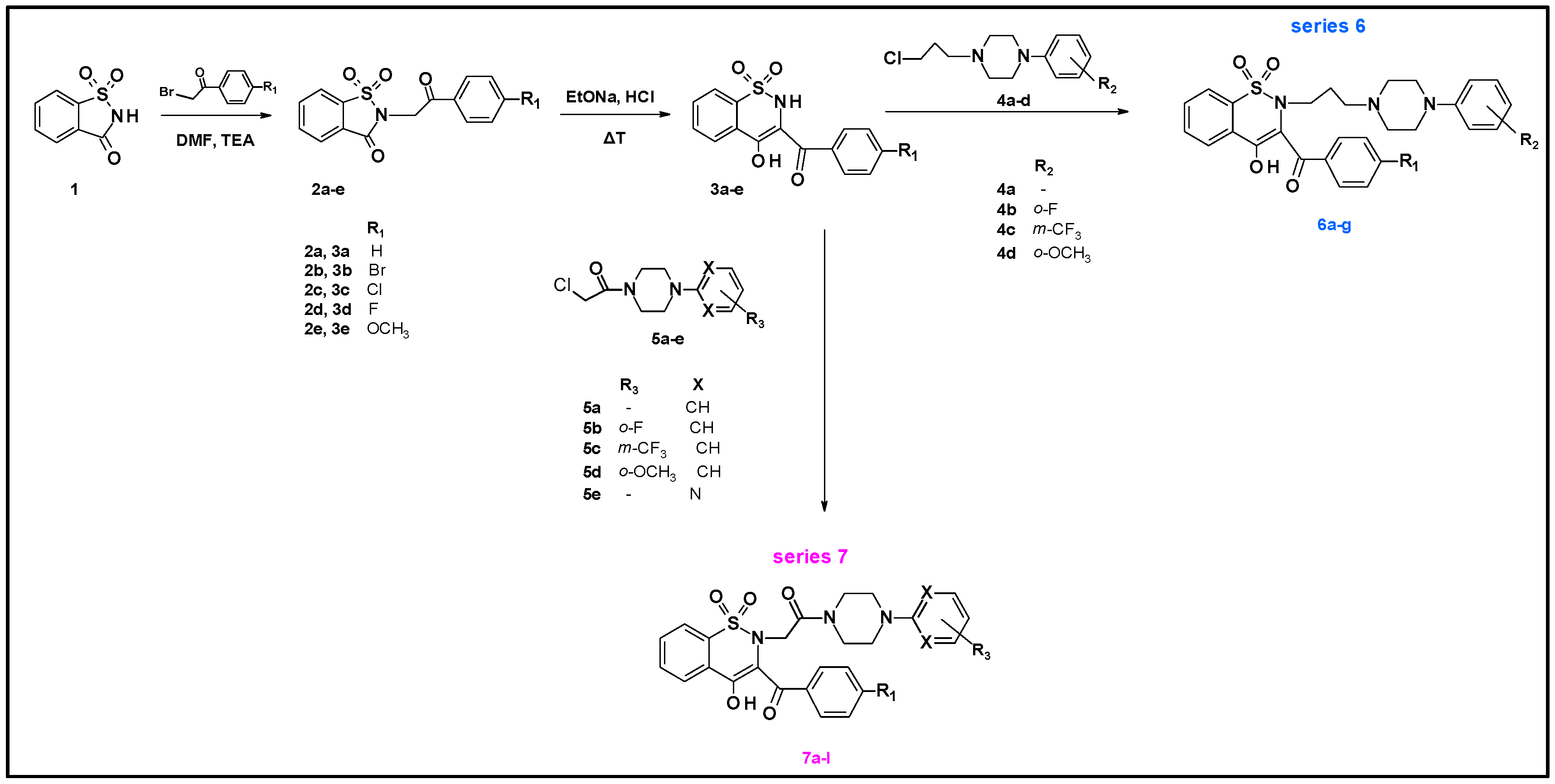
4.2. General Procedure for the Preparation of Series 6 and Series 7 Compounds
Synthesis of Series 7 Compounds (7a–7l):
4.3. Biological Tests
- (a)
- the five following bacterial strains and one fungal strain from the American Tissue and Cell Culture Collection (ATCC) were applied in this study: Staphylococcus aureus 6538; Pseudomonas aeruginosa 15442, Enterococcus faecalis 29212, Klebsiella pneumoniae 70063, Escherichia coli 2592, Candida albicans 10231.
- (b)
- fibroblasts L929 (ATCC® CCL-1) were used to evaluate cytotoxic potential of the tested compounds.
- (c)
- all microbial strains and fibroblast line are part of Strain and Line Collection of Pharmaceutical Microbiology and Parasitology Department of Medical University of Wroclaw
- (d)
- 1,2-benzothiazine 1,1-dioxide derivatives were synthesized in the Department of Chemistry of Drugs of Medical University of Wroclaw. All tested compounds were divided to three series. Series 6 includes seven compounds, series 7 includes 12 compounds and series 3 is represented by one compound. All tested compounds are presented in the Figure 6, Table 7 and Table 8 below.
4.3.1. Evaluation of the Antimicrobial Activity of 1,2-Benzothiazine 1,1-Dioxide Derivatives Using Disc Diffusion Method
4.3.2. Evaluation of the Minimum Inhibitory Concentration (MIC) of 1,2-Benzothiazine 1,1-Dioxide Derivatives vs. Povidone-Iodine (PVP-I) and Polihexanidine (PHMB) Antiseptics Using Serial Microdilution Method
4.3.3. Cytotoxicity Assay on L929 Fibroblast Cells
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Lee Ventola, C. The Antibiotic Resistance Crisis. Part 1: Causes and Threats. Pharm. Ther. 2015, 40, 277–283. [Google Scholar]
- Mazer-Amirshahi, M.; Pourmand, A.; May, L. Newly approved antibiotics and antibiotics reserved for resistant infections: Implications for emergency medicine. Am. J. Emerg. Med. 2017, 35, 154–158. [Google Scholar] [CrossRef] [PubMed]
- Harbarth, S.; Tuan Soh, S.; Horner, C.; Wilcox, M.H. Is reduced susceptibility to disinfectants and antiseptics a risk in healthcare settings? A point/counterpoint review. J. Hosp. Infect. 2014, 87, 194–202. [Google Scholar] [CrossRef] [PubMed]
- Hardy, K.; Sunnucks, K.; Gil, H.; Shabir, S.; Trampari, E.; Hawkey, P.; Webber, M. Increased Usage of Antiseptics Is Associated with Reduced Susceptibility in Clinical Isolates of Staphylococcus aureus. MBio 2018, 9, e00894-18. [Google Scholar] [CrossRef] [PubMed]
- Lal Badshah, S.; Naeem, A. Bioactive Thiazine and Benzothiazine Derivatives: Green Synthesis Methods and Their Medicinal Importance. Molecules 2016, 21, 1054. [Google Scholar] [CrossRef]
- Ahmad, M.; Rizvi, S.; Siddiqui, H.; Ahmad, S.; Parvez, M.; Suliman, R. Antioxidant and antimicrobial studies of novel N0-(substituted-2-chloroquinolin-3-yl)methylidene-4-hydroxy-2H-1,2-benzothiazine-3-carbo-hydrazides 1,1-dioxides. Med. Chem. Res. 2012, 21, 2340–2348. [Google Scholar] [CrossRef]
- Bihovsky, R.; Tao, M.; Mallamo, J.; Wells, G. 1,2-benzothiazine 1,1-dioxide-ketoamide analogues as potent calpain I inhibitors. Bioorg. Med. Chem. Lett. 2004, 14, 1035–1038. [Google Scholar] [CrossRef]
- Tasaki, Y.; Yamamoto, J.; Omura, T.; Noda, T.; Kamiyama, N.; Yoshida, K.; Satomi, M.; Sakaguchi, T.; Asari, M.; Ohkubo, T.; et al. Oxicam structure in non-steroidal anti-inflammatory drugs is essential to exhibit Akt-mediated neuroprotection against 1-methyl-4-phenylpyridinium-induced cytotoxicity. Eur. J. Pharmacol. 2012, 676, 57–63. [Google Scholar] [CrossRef]
- Rouzer, C.A.; Marnett, L.J. Oxicams, a class of nonsteroidal anti-inflammatory drugs and beyond. IUBMB Life 2014, 66, 803–811. [Google Scholar] [CrossRef]
- Szczęśniak-Sięga, B.M.; Mogilski, S.; Wiglusz, R.J.; Janczak, J.; Maniewska, J.; Malinka, W.; Filipek, B. Synthesis and pharmacological evaluation of novel arylpiperazine oxicams derivatives as potent analgesics without ulcerogenicity. Bioorg. Med. Chem. 2019, 27, 1619–1628. [Google Scholar] [CrossRef]
- Patel, C.; Bassin, J.P.M.; Flye, J.; Hunter, A.P.; Martin, L.; Goyal, M. Synthesis and Antimicrobial Activity of 1,2-Benzothiazine Derivatives. Molecules 2016, 21, 861. [Google Scholar] [CrossRef] [PubMed]
- Shan, Y.; Zhang, C.M.; Tang, L.Q.; Liu, Z.P.; Bearss, N.; Sarver, J.; Luniwal, A.; Erhardt, P. Syntheses of 2,3-Diarylated 2H-Benzo[e][1,2]Thiazine 1,1-Dioxides and their 3,4-Dihydro Derivatives, and Assessment of their Inhibitory Activity Against MCF-7 Breast Cancer Cells. Med. Chem. 2011, 7, 561–571. [Google Scholar] [CrossRef] [PubMed]
- Szczęśniak-Sięga, B.; Gębczak, K.; Gębarowski, T.; Maniewska, J. Synthesis, COX-1/2 inhibition and antioxidant activities of new oxicam analogues designed as potential chemopreventive agents. Acta Biochim. Pol. 2018, 65, 199–207. [Google Scholar] [CrossRef] [PubMed]
- Soheili, V.; Bazzaz, B.S.F.; Abdollahpour, N.; Hadizadeh, F. Investigation of Pseudomonas aeruginosa quorum-sensing signaling system for identifying multiple inhibitors using molecular docking and structural analysis methodology. Microb. Pathog. 2015, 89, 73–78. [Google Scholar] [CrossRef] [PubMed]
- Krzyżak, E.; Szczęśniak-Sięga, B.; Malinka, W. Synthesis and thermal behaviour of new benzo-1,2-thiazine long-chain aryl-piperazine derivatives. J. Therm. Anal. Calorim. 2014, 115, 793–802. [Google Scholar] [CrossRef][Green Version]
- Szczęśniak-Sięga, B.; Maniewska, J.; Poła, A.; Środa-Pomianek, K.; Malinka, W.; Michalak, K. Synthesis of new Piroxicam derivatives and their influence on lipid bilayers. Acta Pol. Pharm. Drug Res. 2014, 71, 1045–1050. [Google Scholar] [CrossRef]
- Maniewska, J.; Szczęśniak-Sięga, B.; Poła, A.; Środa-Pomianek, K.; Malinka, W.; Michalak, K. The interaction of new piroxicam derivatives with lipid bilayers—A calorimetric and fluorescence spectroscopic study. Acta Biochim. Pol. 2018, 65, 185–191. [Google Scholar] [CrossRef]
- Yoshimura, A.; Koski, S.R.; Fuchs, J.M.; Saito, A.; Nemykin, V.N.; Zhdankin, V.V. Saccharin-based μ-oxo imidoiodane: A readily available and highly reactive reagent for electrophilic amination. Chem. Eur. J. 2015, 21, 5328–5331. [Google Scholar] [CrossRef]
- Lachapelle, J.; Castel, O.; Casado, F.; Leroy, B.; Micali, G.; Tennstedt, D.; Lambert, J. Antiseptics in the era of bacterial resistance: A focus on povidone iodine. Future Med. 2013, 10, 579–592. [Google Scholar] [CrossRef]
- Koburger, T.N.O.; Braun, M.; Siebert, J.; Kramer, A. Standardized comparison of antiseptic efficacy of triclosan, PVP–iodine, octenidine dihydrochloride, polyhexanide and chlorhexidine digluconate. J. Antimicrob. Chemother. 2010, 65, 1712–1719. [Google Scholar] [CrossRef]
- Liu, W.; Wang, W.D.; Wang, W.; Bai, S.; Dybowski, C. Influence of structure on the spectroscopic properties of the polymorphs of piroxicam. J. Phys. Chem. B 2010, 114, 16641–16649. [Google Scholar] [CrossRef] [PubMed]
- Politzer, P.; Laurence, P.R.; Jayasuriya, K. Molecular electrostatic potentials: An effective tool for the elucidation of biochemical phenomena. Environ. Health Perspect. 1985, 61, 191–202. [Google Scholar] [CrossRef] [PubMed]
- Politzer, P.; Thular, D.G. (Eds.) Chemical Applications of Atomic and Molecular Electrostatic Potentials; Plenum Press: New York, NY, USA, 1981. [Google Scholar]
- Murray, J.S.; Politzer, P. The electrostatic potential: An overview. Wiley Interdiscip. Rev. Comput. Mol. Sci. 2011, 1, 153–163. [Google Scholar] [CrossRef]
- Vincent, J.L.; Rello, J.; Marshall, J.; Silva, E.; Anzueto, A.C.D.; Moreno, R.; Lipman, J.; Gomersall, C.; Sakr, Y.; EPIC II Group of Investigators; et al. International Study of the Prevalence and Outcomes of Infection in Intensive Care Units. JAMA 2009, 302, 2323–2329. [Google Scholar] [CrossRef] [PubMed]
- Sommer, L.M.; Krauss, J.; Hultén, K.G.; Dunn, J.; Kaplan, S.L.; McNeil, J.A. The Prevalence of Antiseptic Tolerance Genes Among Staphylococci and Enterococci in a Pediatric Population. Infect. Control Hosp. Epidemiol. 2019, 40, 333–340. [Google Scholar] [CrossRef] [PubMed]
- Ahmad, N.; Zia-Ur-Rehman, M.; Siddiqui, H.L.; Ullah, M.F.; Parvez, M. Microwave assisted synthesis and structure-activity relationship of 4-hydroxy-N′-[1-phenylethylidene]-2H/2-methyl-1.2-benzothiazine-3- carbohydrazide 1.1-dioxides as anti-microbial agents. Eur. J. Med. Chem. 2011, 46, 2368–2377. [Google Scholar] [CrossRef]
- Balouiri, M.; Sadiki, M.; Ibnsouda, S.K. Methods for in vitro evaluating antimicrobial activity: A review. J. Pharm. Anal. 2016, 6, 71–79. [Google Scholar] [CrossRef]
- Reller, B.L.; Weinstein, M.; Jorgensen, J.H.; Ferraro, M.J. Antimicrobial Susceptibility Testing: A Review of General Principles and Contemporary Practices. Clin. Infect. Dis. 2009, 49, 1749–1755. [Google Scholar] [CrossRef]
- Bonev, B.; Hooper, J.; Parisot, J. Principles of assessing bacterial susceptibility to antibiotics using the agar diffusion method. J. Antimicrob. Chemother. 2008, 61, 1295–1301. [Google Scholar] [CrossRef]
- Valgas, C.; de Souza, S.M.; Smânia, E.F.A.; Smânia, A., Jr. Screening methods to determine antibacterial activity of natural products. Braz. J. Microbiol. 2007, 38, 369–380. [Google Scholar] [CrossRef]
- Zia-ur-Rehman, M.; Choudary, J.A.; Elsegood, M.R.J.; Siddiqui, H.L.; Khan, K.M. A Facile Synthesis of Novel Biologically Active 4-hydroxy-N’-(benzylidene)-2H-benzo[e][1,2]thiazine-3-carbohydrazide 1,1-dioxides. Eur. J. Med. Chem. 2009, 44, 1311–1316. [Google Scholar] [CrossRef] [PubMed]
- Ahmad, M.; Siddiqui, H.L.; Zia-ur-Rehman, M.; Parvez, M. Anti-oxidant and anti-bacterial activities of novel N′-arylmethylidene-2-(3,4-dimethyl-5,5-dioxidopyrazolo[4,3-c][1,2]benzothiazin-2(4H)-yl) acetohydrazides. Eur. J. Med. Chem. 2010, 45, 698–704. [Google Scholar] [CrossRef] [PubMed]
- Pollard, C.; Lauter, W.; Nuessle, N. Derivatives of piperazine. XXXIV. Some reactions of trimethylene chlorobromide with 1-arylpiperazines. J. Org. Chem. 1959, 24, 764–767. [Google Scholar] [CrossRef]
- Brown, D.A.; Kharkar, P.S.; Parrington, I.; Reith, M.E.A.; Dutta, A.K. Structurally Constrained Hybrid Derivatives Containing Octahydrobenzo[g or f]quinolone Moieties for Dopamine D2 and D3 Receptors: Binding Characterization at D2/D3 Receptors and Elucidation of a Pharmacophore Model. J. Med. Chem. 2008, 51, 7806–7819. [Google Scholar] [CrossRef]
- Muszalska, I.; Ciemniejewski, M.P.; Lesniewska, M.A.; Szkatuła, D.; Malinka, W. Forced degradation and photodegradation studies of pyrrolo[3,4-c]pyridine-1,3-dione derivatives as analgesic active compounds using HPLC, UV and IR spectrometry and HPLC/MS methods. J. AOAC Int. 2015, 98, 1248–1259. [Google Scholar] [CrossRef] [PubMed]
- Colabufo, N.A.; Berardi, F.; Perrone, R.; Rapposelli, S.; Digiacomo, M.; Vanni, M.; Balsamo, A. 2-[(3-Methoxyphenylethyl) phenoxy]-Based ABCB1 Inhibitors: Effect of Different Basic Side-Chains on Their Biological Properties. J. Med. Chem. 2008, 51, 7602–7613. [Google Scholar] [CrossRef] [PubMed]
- Malinka, W.; Sieklucka-Dziuba, M.; Rajtar, G.; Rubaj, A.; Kleinrok, Z. Synthesis and pharmacological screening of some N-(4-substituted-piperazin-1-ylalkyl)-3,4-pyrroledicarboximides). Il Farm. 1999, 54, 390–401. [Google Scholar] [CrossRef]
- CrysAlis CCD and CrysAlis Red, Version 1.171.33.42; Oxford Diffraction Ltd.: Wrocław, Poland, 2009.
- Sheldrick, G.M. SHELXT—Integrated space-group and crystal-structure determination. Acta Crystallogr. Sect. A Found. Adv. 2015, 71, 3–8. [Google Scholar] [CrossRef]
- Sheldrick, G.M. Crystal structure refinement with SHELXL. Acta Crystallogr. Sect. C Cryst. Struct. Commun. 2015, 71, 3–8. [Google Scholar] [CrossRef]
- Brandenburg, K.; Putz, H. DIAMOND Version 3.0; Crystal Impact GbR: Bonn, Germany, 2006. [Google Scholar]
- Frisch, M.J.; Trucks, G.W.; Schlegel, H.B.; Scuseria, G.E.; Robb, M.A.; Cheeseman, J.R.; Scalmani, G.; Barone, V.; Mennucci, B.; Petersson, G.A.; et al. Gaussian09, Revision, D.01, Programme; Gaussian Inc.: Wallingford, CT, USA, 2013; Available online: https://gaussian.com/g09citation/ (accessed on 31 July 2020).
- Becke, A.D. Density-functional thermochemistry. IV. A new dynamical correlation functional and implications for exact-exchange mixing. J. Chem. Phys. 1996, 104, 1040–1046. [Google Scholar] [CrossRef]
- Lee, C.; Yang, W.; Parr, R.G. Development of the Colic-Salvetti correlation-energy formula into a functional of the electron density. Phys. Rev. B 1988, 37, 785–789. [Google Scholar] [CrossRef] [PubMed]
- Parr, B.G.; Yang, W. Density-Functional Theory of Atoms and Molecules; Oxford University Press: New York, NY, USA, 1989. [Google Scholar]
- Bai, R.; Pei, X.; Boye, O.; Getanunt, Z.; Grover, S.; Nekisz, J.; Nguyen, N.; Brossi, A.; Hamel, E.J. Identification of cysteine 354 of beta-tubulin as part of the binding site for the A ring of colchicine. J. Biol. Chem. 1996, 272, 12639–12645. [Google Scholar] [CrossRef]
Sample Availability: Samples of the compounds are not available from the authors. |

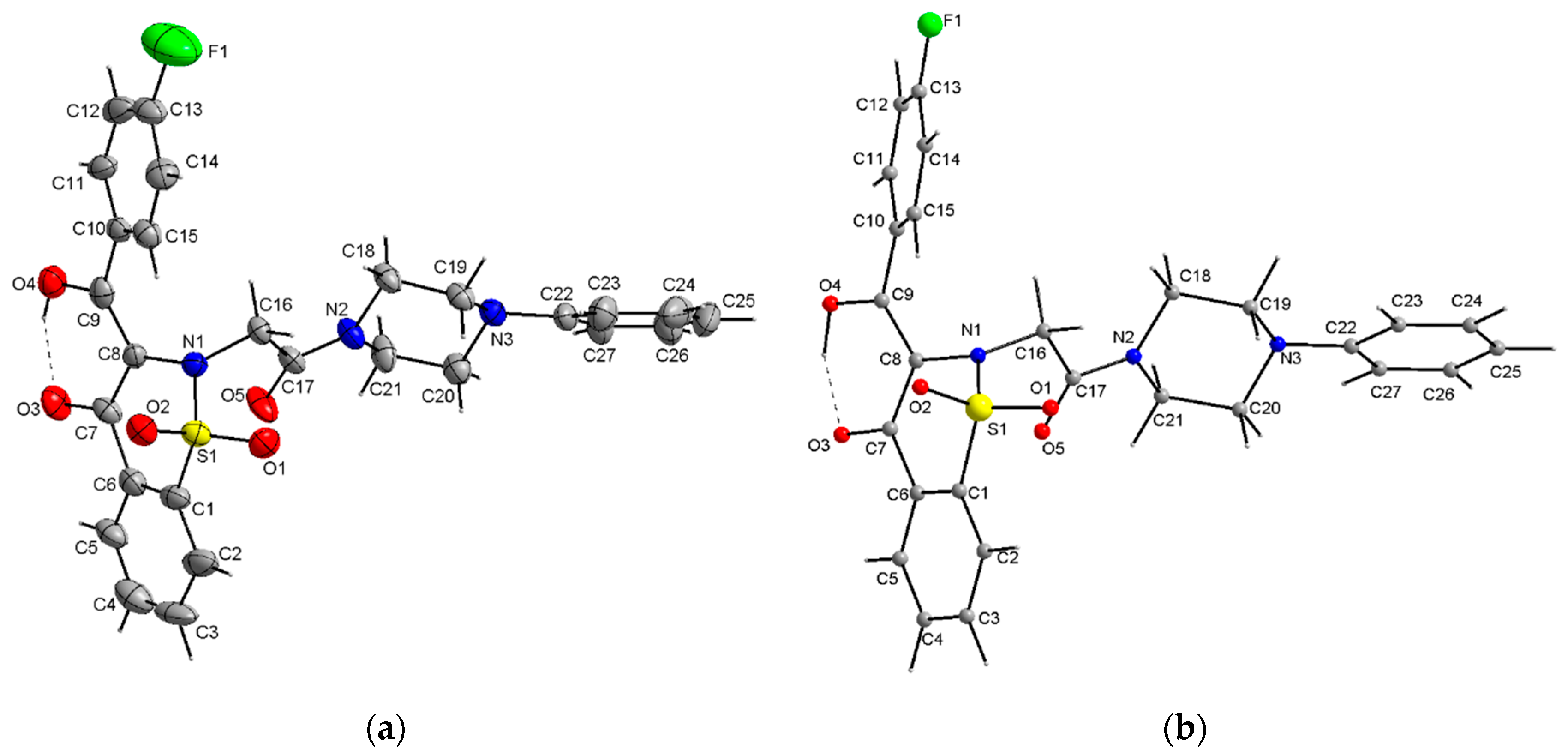





| X-ray | DFT | |
|---|---|---|
| S1—O1 | 1.420 (3) | 1.468 |
| S1—O2 | 1.420 (4) | 1.467 |
| S1—N1 | 1.618 (4) | 1.684 |
| S1—C1 | 1.742 (6) | 1.790 |
| C7—O3 | 1.275 (6) | 1.254 |
| C8—N1 | 1.429 (6) | 1.433 |
| C9—O4 | 1.297 (5) | 1.331 |
| N1—C16 | 1.477 (6) | 1.462 |
| C17—O5 | 1.219 (6) | 1.229 |
| C17—N2 | 1.318 (6) | 1.368 |
| N3—C22 | 1.427 (6) | 1.420 |
| O2—S1—O1 | 119.3 (2) | 120.18 |
| N1—S1—C1 | 102.4 (3) | 101.34 |
| C5—C6—C7—C8 | −170.0 (6) | −169.97 |
| C8—C9—C10—C15 | −30.0 (8) | −39.05 |
| C8—N1—C16—C17 | −69.0 (5) | −62.23 |
| S1—N1—C16—C17 | 85.8 (5) | 92.72 |
| N1—C16—C17—N2 | 178.1 (4) | 178.14 |
| C19—N3—C22—C23 | 7.3 (5) | 10.27 |
| 7i | 7a | |
|---|---|---|
| C1—S1—N1—C16 | −106.8 (4) | −107.7 (4) |
| S1—N1—C16—C17 | 85.7 (4) | 82.7 (4) |
| N1—C16—C17—O5 | 4.0 (6) | 0.3 (7) |
| N1—C16—C17—N2 | 178.1 (4) | −178.9 (5) |
| C16—C17—N2—C18 | −1.4 (7) | −7.1 (7) |
| C17—N2—C18—c19 | 129.8 (5) | 139.7 (7) |
| N2—C18—C19—N3 | 58.5 (6) | 57.7 (6) |
| C18—C19—N3—C22 | 166.5 (5) | 165.0 (5) |
| C19—N3—C22—C23 | 7.3 (7) | 15.5 (7) |
| N3—C22—C23—C24 | −174.9 (5) | −176.6 (5) |
| D-H···A | D-H | H···A | D···A | D-H···A | |
|---|---|---|---|---|---|
| O4-H4A···O3 | 0.82 | 1.69 | 2.433 (6) | 150 | (in crystal) |
| O4-H4A···O3 | 1.011 | 1.599 | 2.519 | 148.90 | (DFT) |
| Zones of S. aureus Growth Inhibition [mm] | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 4 mg | 2 mg | 1 mg | 0.5 mg | 0.25 mg | 0.2 mg | 0.125 mg | 0.1 mg | 0.0625 mg | 0.05 mg | 0.0312 mg | 0.025 mg | |
| 6d | 0 | 0 | 8 (±0) | 8 (±0) | 8 (±0) | N/A | 9 (±0) | N/A | 9 (±0) | N/A | 10 (±0) | N/A |
| 6g | N/A | N/A | N/A | N/A | N/A | 6 (±0) | N/A | 6 (±0) | N/A | 8 (±0) | N/A | 9 (±0) |
| 7a | 0 | 0 | 9.33 (±0.58) | 9.66 (±1.15) | 8.33 (±0.58) | N/A | 9 (±1) | N/A | 0 | N/A | N/A | N/A |
| 7b | 10 (±0) | 10 (±0) | 10.33 (±0.58) | 10 (±0) | 9.33 (±0.58) | N/A | 7.33 (±1.15) | N/A | 0 | N/A | N/A | N/A |
| 7e | 0 | 0 | 0 | 9 (±0) | 9 (±0) | N/A | 8 (±0) | N/A | 6 (±0) | N/A | N/A | N/A |
| 7f | 0 | 0 | 0 | 9 (±0) | 8 (±0) | N/A | 9 (±0) | N/A | 10 (±0) | N/A | 10 (±0) | N/A |
| 7h | 0 | 10 (±0) | 8 (±0) | 9 (±0) | 9 (±0) | N/A | 0 | N/A | 0 | N/A | N/A | N/A |
| 7l | 0 | 0 | 9 (±0) | 9 (±0) | 8 (±0) | N/A | 9 (±0) | N/A | 0 | N/A | N/A | N/A |
| Zones of E. faecalis Growth Inhibition [mm] | ||||||||||||
| 6d | 0 | 0 | 0 | 10 (±0) | 10 (±0) | N/A | 11 (±0) | N/A | 10 (±0) | N/A | 10.33 (±0.58) | N/A |
| 7b | 0 | 0 | 0 | 9 (±0) | 8 (±0) | N/A | 8 (±0) | N/A | 0 | N/A | N/A | N/A |
| 7d | 0 | 0 | 0 | 8 (±0) | 7 (±0) | N/A | 7 (±0) | N/A | N/A | N/A | N/A | N/A |
| 7f | 0 | 0 | 0 | 6 (±0) | 8 (±0) | N/A | 10 (±0) | N/A | 11 (±0) | N/A | 11.33 (±0.58) | N/A |
| 7g | 0 | 0 | 0 | 0 | 9 (±0) | N/A | 10 (±0) | N/A | 8 (±0) | N/A | N/A | N/A |
| 7j | 0 | 0 | 0 | 9 (±0) | 9 (±0) | N/A | 9 (±0) | N/A | 6 (±0) | N/A | N/A | N/A |
| MIC [mg/mL] | |||||
|---|---|---|---|---|---|
| S. aureus | E. faecalis | P. aeruginosa | E. coli | K. pneumoniae | |
| 3e | 1.04 | 2.08 | 1.04 | 1.04 | 1.04 |
| Series 6 | |||||
| 6a | 0.078 | 0.078 | 2.5 | 1.25 | 1.25 |
| 6b | 0.024 | 0.024 | 3.125 | 3.125 | 3.125 |
| 6c | 0.065 | 0.065 | 2.07 | 2.07 | 2.07 |
| 6d | 0.024 | 0.024 | 3.125 | 3.125 | 3.125 |
| 6e | 0.024 | 0.024 | 3.125 | 3.125 | 3.125 |
| 6f | 0.195 | 0.195 | 3.125 | 3.125 | 3.125 |
| 6g | 0.00975 | 0.00975 | 1.25 | 1.25 | 1.25 |
| Series 7 | |||||
| 7a | 0.26 | 0.065 | 4.16 | 4.16 | 4.16 |
| 7b | 0.195 | 0.39 | 6.25 | 6.25 | 6.25 |
| 7c | 0.13 | 0.13 | 2.07 | 2.07 | 2.07 |
| 7d | 0.024 | 0.024 | 3.125 | 3.125 | 3.125 |
| 7e | 0.0655 | 0.0655 | 0.525 | 0.525 | 0.525 |
| 7f | 0.024 | 0.024 | 3.125 | 3.125 | 3.125 |
| 7g | 0.024 | 0.024 | 3.125 | 3.125 | 3.125 |
| 7h | 0.78 | 0.78 | 3.125 | 3.125 | 3.125 |
| 7i | 0.78 | 0.78 | 1.56 | 1.56 | 1.56 |
| 7j | 0.195 | 0.39 | 3.125 | 3.125 | 3.125 |
| 7k | 0.156 | 0.156 | 0.625 | 0.625 | 0.625 |
| 7l | 0.195 | 0.39 | 1.56 | 3.125 | 3.125 |
| Antiseptics | |||||
| PHMB | 0.024 | 0.097 | 3.125 | 0.097 | 0.39 |
| PVP-I | 1.56 | 6.25 | 12.5 | 6.25 | 3.125 |
| MIC [mg/mL] | Cytotoxicity [%] | |
|---|---|---|
| 3e | 1.04 *,** 2.08 # | 59.55 (±12.39) 66.32 (±5.44) |
| Series 6 | ||
| 6a | 0.078 *,# | −1.51 (±5.30) |
| 6b | 0.024 *,# | 15.11 (±1.19) |
| 6c | 0.065 *,# | 15.61 (±7.80) |
| 6d | 0.024 *,# | 79.76 (±0.62) |
| 6e | 0.024 *,# | 23.61 (±5.21) |
| 6f | 0.195 *,# | 15.10 (±2.69) |
| 6g | 0.00975 *,# | 21.25 (±6.26) |
| Series 7 | ||
| 7a | 0.26 * 0.065 # | 2.40 (±14.82) 20.97 (±6.99) |
| 7b | 0.195 * 0.39 # | −10.83 (±5.19) −3.795 (±6.94) |
| 7c | 0.13 *,# | −2.32 (1.43) |
| 7d | 0.024 *,# | −11.09 (±1.96) |
| 7e | 0.0655 *,# | 2.68 (±4.20) |
| 7f | 0.024 *,# | 2.92 (±13.42) |
| 7g | 0.024 *,# | 9.68 (±4.09) |
| 7h | 0.78 *,# | 38.26 (±10.86) |
| 7i | 0.78 *,# 1.56 ** | 65.60 (±1.55) 61.64 (±4.98) |
| 7j | 0.195 * 0.39 # | 4.30 (±6.19) 32.53 (±0.67) |
| 7k | 0.156 *,# | 17.27 (±13.97) |
| 7l | 0.195 * 0.39 # | 5.90 (±3.49) 3.11 (±2.30) |
 Series 6 | ||
|---|---|---|
| Compound | R1 | R2 |
| 6a | H | o-OCH3 |
| 6b | Cl | o-F |
| 6c | F | o-OCH3 |
| 6d | F | m-CF3 |
| 6e | F | - |
| 6f | OCH3 | - |
| 6g | OCH3 | m-CF3 |
 Series 7 | |||
|---|---|---|---|
| Compound | R1 | R3 | X |
| 7a | H | - | CH |
| 7b | H | - | N |
| 7c | H | o-OCH3 | CH |
| 7d | H | o-F | CH |
| 7e | Br | - | N |
| 7f | Cl | o-F | CH |
| 7g | F | o-F | CH |
| 7h | F | m-CF3 | CH |
| 7i | F | - | CH |
| 7j | OCH3 | - | CH |
| 7k | OCH3 | - | N |
| 7l | OCH3 | o-OCH3 | CH |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Dudek-Wicher, R.K.; Szczęśniak-Sięga, B.M.; Wiglusz, R.J.; Janczak, J.; Bartoszewicz, M.; Junka, A.F. Evaluation of 1,2-Benzothiazine 1,1-Dioxide Derivatives In Vitro Activity towards Clinical-Relevant Microorganisms and Fibroblasts. Molecules 2020, 25, 3503. https://doi.org/10.3390/molecules25153503
Dudek-Wicher RK, Szczęśniak-Sięga BM, Wiglusz RJ, Janczak J, Bartoszewicz M, Junka AF. Evaluation of 1,2-Benzothiazine 1,1-Dioxide Derivatives In Vitro Activity towards Clinical-Relevant Microorganisms and Fibroblasts. Molecules. 2020; 25(15):3503. https://doi.org/10.3390/molecules25153503
Chicago/Turabian StyleDudek-Wicher, Ruth K., Berenika M. Szczęśniak-Sięga, Rafał J. Wiglusz, Jan Janczak, Marzenna Bartoszewicz, and Adam F. Junka. 2020. "Evaluation of 1,2-Benzothiazine 1,1-Dioxide Derivatives In Vitro Activity towards Clinical-Relevant Microorganisms and Fibroblasts" Molecules 25, no. 15: 3503. https://doi.org/10.3390/molecules25153503
APA StyleDudek-Wicher, R. K., Szczęśniak-Sięga, B. M., Wiglusz, R. J., Janczak, J., Bartoszewicz, M., & Junka, A. F. (2020). Evaluation of 1,2-Benzothiazine 1,1-Dioxide Derivatives In Vitro Activity towards Clinical-Relevant Microorganisms and Fibroblasts. Molecules, 25(15), 3503. https://doi.org/10.3390/molecules25153503









