Polymers and Plastics Modified Electrodes for Biosensors: A Review
Abstract
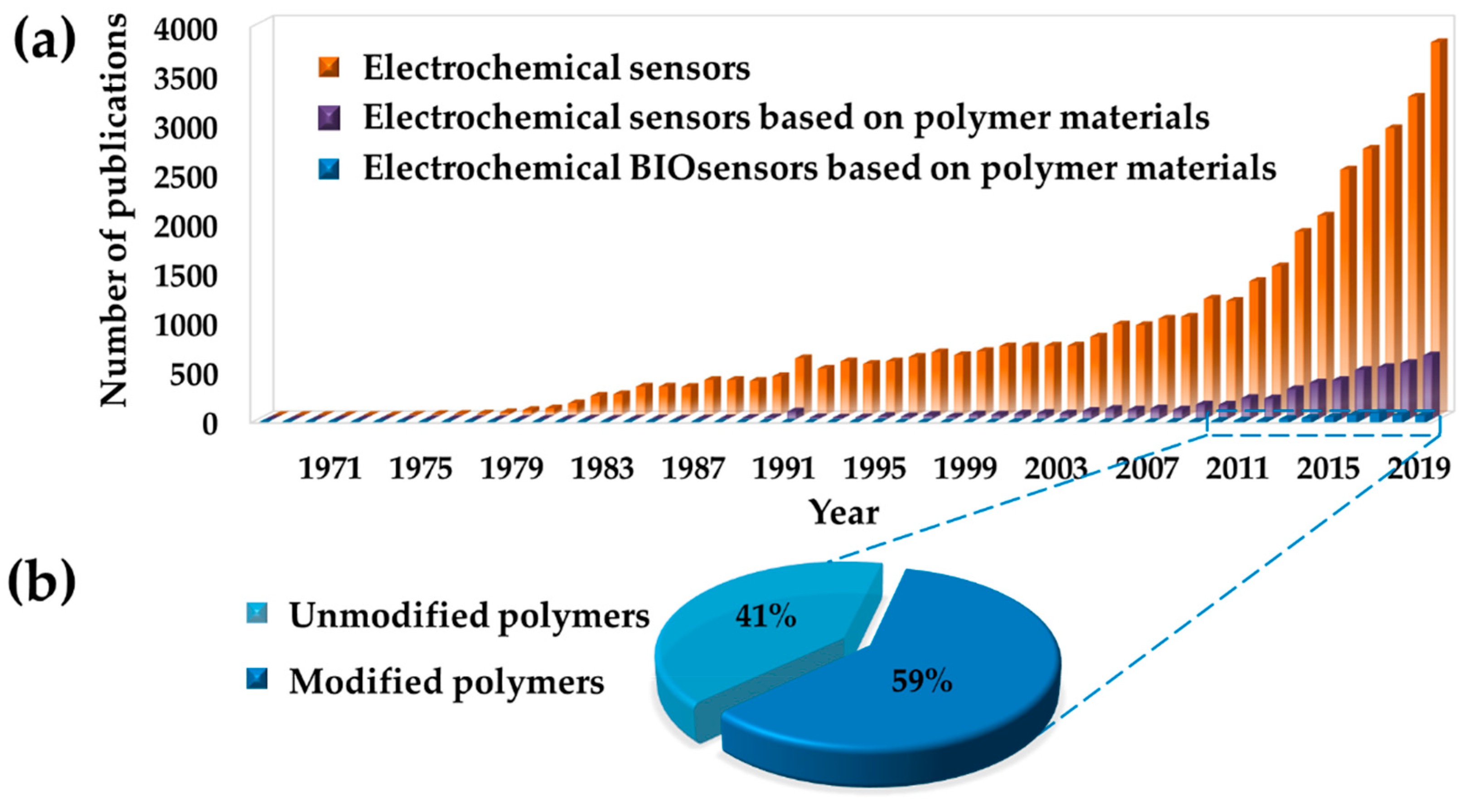
1. Introduction
2. Electro-BIOsensors Based on Modified Conducting Polymers
2.1. ICPs Modified with Metals
2.2. ICPs Modified with Carbon
2.3. ICPs Biomodified
2.4. ICPs Modified with Other Polymers
3. Electro-BIOsensors Including Non-Conducting Polymers
3.1. Bioplastics and Biodegradable Plastics
3.2. Eco/Recyclable Plastics
4. Lab on a Chip (LOC), Electro-BIOsensors Fabricated with Common Plastics
Wearable Biosensing Platforms
5. Brief Conclusions and Future Outlooks
Author Contributions
Funding
Conflicts of Interest
References
- Arduini, F.; Cinti, S.; Scognamiglio, V.; Moscone, D.; Palleschi, G. How cutting-edge technologies impact the design of electrochemical (bio)sensors for environmental analysis. A review. Anal. Chim. Acta 2017, 959, 15–42. [Google Scholar] [CrossRef] [PubMed]
- Yang, Y.; Yang, X.; Tan, Y.; Yuan, Q. Recent progress in flexible and wearable bio-electronics based on nanomaterials. Nano Res. 2017, 10, 1560–1583. [Google Scholar] [CrossRef]
- Soper, S.A.; Brown, K.; Ellington, A.; Frazier, B.; Garcia-manero, G.; Gau, V.; Gutman, S.I.; Hayes, D.F.; Korte, B.; Landers, J.L.; et al. Point-of-care biosensor systems for cancer diagnostics/prognostics. Biosens. Bioelectron. 2006, 21, 1932–1942. [Google Scholar] [CrossRef] [PubMed]
- Bruen, D.; Delaney, C.; Florea, L.; Diamond, D. Glucose Sensing for Diabetes Monitoring: Recent Developments. Sensors 2017, 17, 1866. [Google Scholar] [CrossRef] [PubMed]
- Cui, F.; Zhou, Z.; Zhou, H.S. Molecularly Imprinted Polymers and Surface Imprinted Polymers Based Electrochemical Biosensor for Infectious Diseases. Sensors 2020, 20, 996. [Google Scholar] [CrossRef]
- Jiang, L. Biomedical Diagnostics at Point-of-Care 2019–2029: Technologies, Applications, Forecasts. Available online: https://www.idtechex.com/de/research-report/biomedical-diagnostics-at-point-of-care-2019-2029-technologies-applications-forecasts/622 (accessed on 8 May 2020).
- Weltin, A.; Kieninger, J.; Urban, G.A. Microfabricated, amperometric, enzyme-based biosensors for in vivo applications. Anal. Bioanal. Chem. 2016, 408, 4503–4521. [Google Scholar] [CrossRef]
- Brosel-Oliu, S.; Abramova, N.; Uria, N.; Bratov, A. Impedimetric transducers based on interdigitated electrode arrays for bacterial detection—A review. Anal. Chim. Acta 2019, 1088, 1–19. [Google Scholar] [CrossRef]
- Leva-Bueno, J.; Peyman, S.A.; Millner, P.A. A review on impedimetric immunosensors for pathogen and biomarker detection. Med. Microbiol. Immunol. 2020. [Google Scholar] [CrossRef]
- Beitollahi, H.; Safaei, M.; Tajik, S. Voltammetric and amperometric sensors for determination of epinephrine: A short review (2013–2017). J. Electrochem. Sci. Eng. 2018, 9, 27. [Google Scholar] [CrossRef]
- Yang, X.; Cheng, H. Recent developments of flexible and stretchable electrochemical biosensors. Micromachines 2020, 11. [Google Scholar] [CrossRef]
- Bollella, P.; Gorton, L. Enzyme based amperometric biosensors Enzyme based amperometric biosensors. Curr. Opin. Electrochem. 2018. [Google Scholar] [CrossRef]
- Wang, J. Electrochemical glucose biosensors. Chem. Rev. 2008, 108, 814–825. [Google Scholar] [CrossRef] [PubMed]
- Gifford, R. Continuous glucose monitoring: 40 years, what we’ve learned and what’s next. ChemPhysChem 2013, 14, 2032–2044. [Google Scholar] [CrossRef] [PubMed]
- Dunn, A.M.; Hofmann, O.S.; Waters, B.; Witchel, E. Cloaking malware with the trusted platform module. In Proceedings of the 20th USENIX Security Symposium, San Francisco, CA, USA, 8–12 August 2011; pp. 395–410. [Google Scholar]
- Robinson, D.L.; Hermans, A.; Seipel, A.T.; Wightman, R.M. Monitoring rapid chemical communication in the brain. Chem. Rev. 2008, 108, 2554–2584. [Google Scholar] [CrossRef]
- Heien, M.L.A.V.; Khan, A.S.; Ariansen, J.L.; Cheer, J.F.; Phillips, P.E.M.; Wassum, K.M.; Wightman, R.M. Real-time measurementofdopaminefluctuationsaftercocaineinthebrainofbehaving rats. Proc. Natl. Acad. Sci. USA 2005, 102, 10023–10028. [Google Scholar] [CrossRef]
- Ewing, A.G.; Bigelow, J.C.; Wightman, R.M. Direct in vivo monitoring of dopamine released from two striatal compartments in the rat. Science 1983, 221, 169–171. [Google Scholar] [CrossRef]
- Tavakolian-Ardakani, Z.; Hosu, O.; Cristea, C.; Mazloum-Ardakani, M.; Marrazza, G. Latest trends in electrochemical sensors for neurotransmitters: A review. Sensors (Switzerland) 2019, 19, 2037. [Google Scholar] [CrossRef]
- Sokolov, A.N.; Roberts, M.E.; Bao, Z. Fabrication of low-cost electronic biosensors sensors capable of operating in biologically relevant media. Mater. Today 2009, 12, 12–20. [Google Scholar] [CrossRef]
- Sadana, A.; Sadana, N. Fabrication of Biosensors. In Handbook of Biosensors and Biosensor Kinetics; Elsevier: Amsterdam, The Netherlands, 2011; pp. 35–60. [Google Scholar]
- Tu, J.; Torrente-Rodríguez, R.M.; Wang, M.; Gao, W. The Era of Digital Health: A Review of Portable and Wearable Affinity Biosensors. Adv. Funct. Mater. 2019, 1906713. [Google Scholar] [CrossRef]
- Metkar, S.K.; Girigoswami, K. Diagnostic biosensors in medicine–A review. Biocatal. Agric. Biotechnol. 2019, 17, 271–283. [Google Scholar] [CrossRef]
- Khan, Y.; Ostfeld, A.E.; Lochner, C.M.; Pierre, A.; Arias, A.C. Monitoring of Vital Signs with Flexible and Wearable Medical Devices. Adv. Mater. 2016, 28, 4373–4395. [Google Scholar] [CrossRef] [PubMed]
- Han, S.T.; Peng, H.; Sun, Q.; Venkatesh, S.; Chung, K.S.; Lau, S.C.; Zhou, Y.; Roy, V.A.L. An Overview of the Development of Flexible Sensors. Adv. Mater. 2017, 29, 1700375. [Google Scholar] [CrossRef] [PubMed]
- Lee, K.M.; Kim, K.H.; Yoon, H.; Kim, H. Chemical design of functional polymer structures for biosensors: From nanoscale to macroscale. Polymers 2018, 10, 551. [Google Scholar] [CrossRef] [PubMed]
- Gui, R.; Guo, H.; Jin, H. Preparation and applications of electrochemical chemosensors based on carbon-nanomaterial-modified molecularly imprinted polymers. Nanoscale Adv. 2019, 1, 3325–3363. [Google Scholar] [CrossRef]
- Jin, H.; Guo, H.; Gao, X.; Gui, R. Selective and sensitive electrochemical sensing of gastrodin based on nickel foam modified with reduced graphene oxide/silver nanoparticles complex-encapsulated molecularly imprinted polymers. Sens. Actuators B Chem. 2018, 277, 14–21. [Google Scholar] [CrossRef]
- Gui, R.; Jin, H.; Guo, H.; Wang, Z. Recent advances and future prospects in molecularly imprinted polymers-based electrochemical biosensors. Biosens. Bioelectron. 2018, 100, 56–70. [Google Scholar] [CrossRef]
- Wang, G.; Morrin, A.; Li, M.; Liu, N.; Luo, X. Nanomaterial-doped conducting polymers for electrochemical sensors and biosensors. J. Mater. Chem. B 2018, 6, 4173–4190. [Google Scholar] [CrossRef]
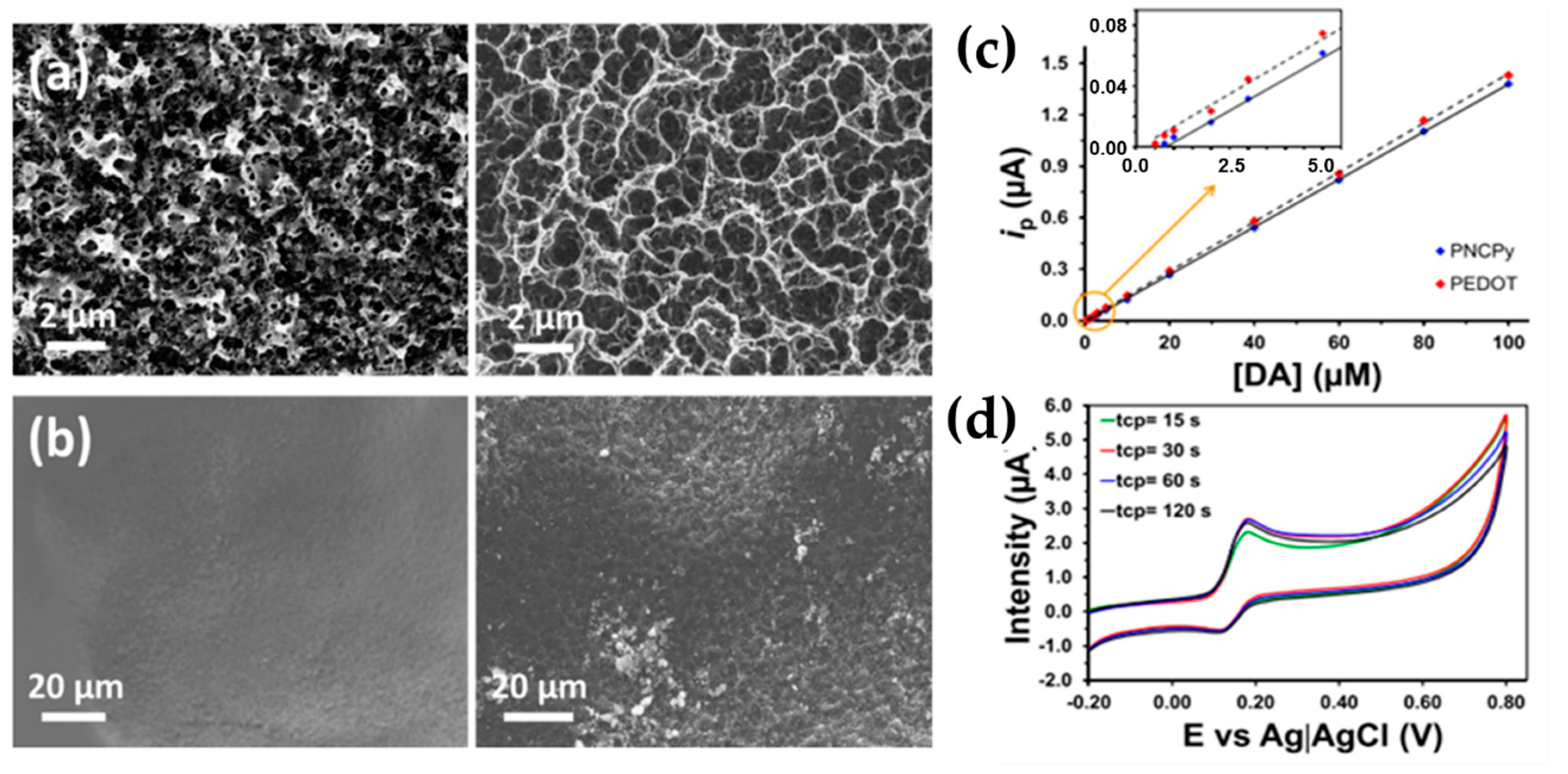
- El-Said, W.A.; Abdelshakour, M.; Choi, J.H.; Choi, J.W. Application of conducting polymer nanostructures to electrochemical biosensors. Molecules 2020, 25, 307. [Google Scholar] [CrossRef]
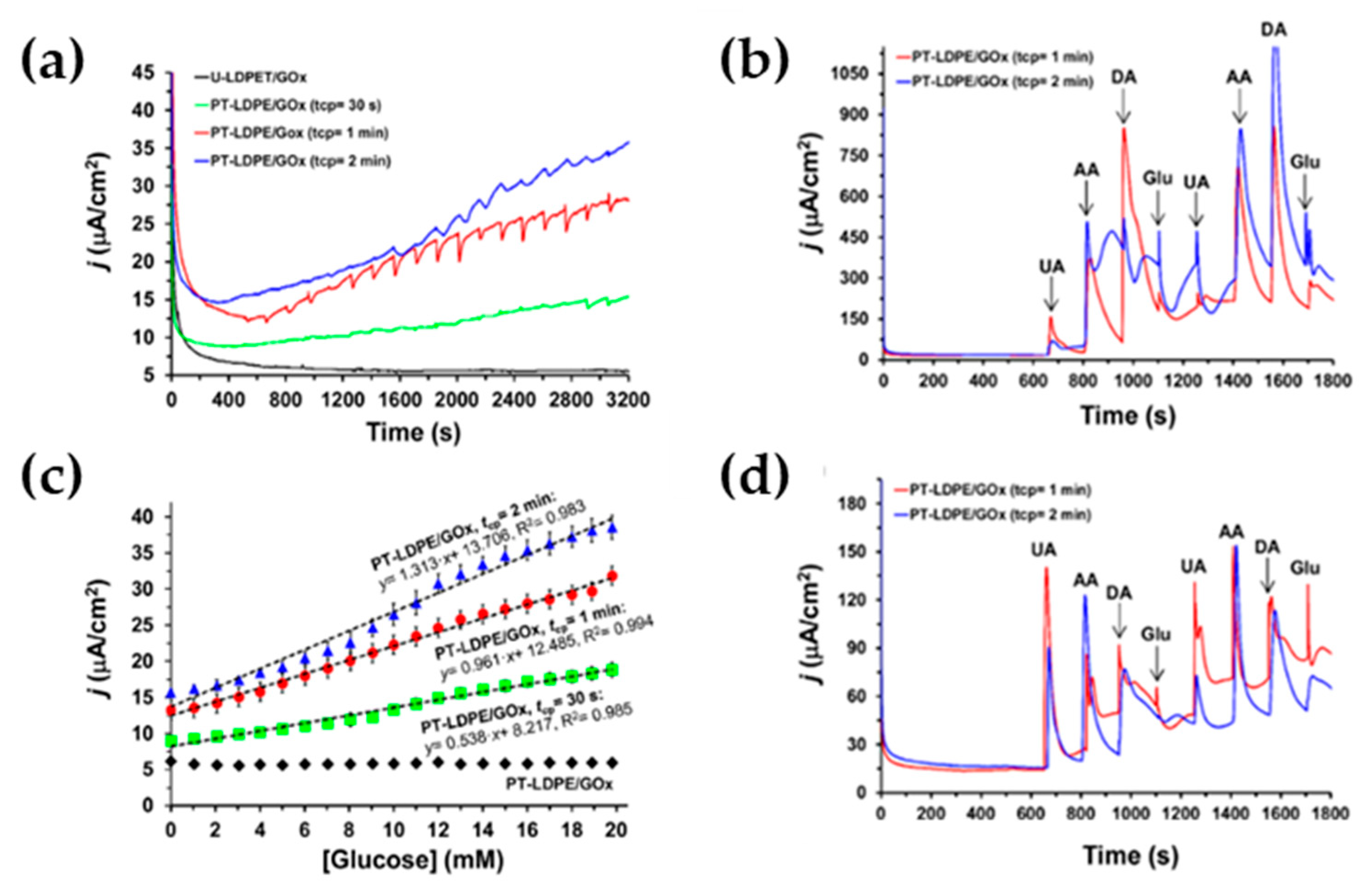
- Poletti Papi, M.A.; Caetano, F.R.; Bergamini, M.F.; Marcolino-Junior, L.H. Facile synthesis of a silver nanoparticles/polypyrrole nanocomposite for non-enzymatic glucose determination. Mater. Sci. Eng. C 2017, 75, 88–94. [Google Scholar] [CrossRef]
- Ansari, S.A.; Ahmed, A.; Ferdousi, F.K.; Salam, M.A.; Shaikh, A.A.; Barai, H.R.; Lopa, N.S.; Rahman, M.M. Conducting poly(aniline blue)-gold nanoparticles composite modified fluorine-doped tin oxide electrode for sensitive and non-enzymatic electrochemical detection of glucose. J. Electroanal. Chem. 2019, 850, 113394. [Google Scholar] [CrossRef]
- Sha, R.; Komori, K.; Badhulika, S. Graphene–Polyaniline composite based ultra-sensitive electrochemical sensor for non-enzymatic detection of urea. Electrochim. Acta 2017, 233, 44–51. [Google Scholar] [CrossRef]
- Weaver, C.L.; Li, H.; Luo, X.; Cui, X.T. A graphene oxide/conducting polymer nanocomposite for electrochemical dopamine detection: Origin of improved sensitivity and specificity. J. Mater. Chem. B 2014, 2, 5209–5219. [Google Scholar] [CrossRef] [PubMed]
- Sun, S.; Zhang, M.; Li, Y.; He, X. A molecularly imprinted polymer with incorporated Graphene oxide for electrochemical determination of quercetin. Sensors (Switzerland) 2013, 13, 5493–5506. [Google Scholar] [CrossRef] [PubMed]
- Radhakrishnan, S.; Sumathi, C.; Dharuman, V.; Wilson, J. Polypyrrole nanotubes-polyaniline composite for DNA detection using methylene blue as intercalator. Anal. Methods 2013, 5, 1010–1015. [Google Scholar] [CrossRef]
- Avelino, K.Y.P.S.; Frias, I.A.M.; Lucena-Silva, N.; Gomes, R.G.; de Melo, C.P.; Oliveira, M.D.L.; Andrade, C.A.S. Attomolar electrochemical detection of the BCR/ABL fusion gene based on an amplifying self-signal metal nanoparticle-conducting polymer hybrid composite. Colloids Surf. B 2016, 148, 576–584. [Google Scholar] [CrossRef]
- Bayram, E.; Akyilmaz, E. Development of a new microbial biosensor based on conductive polymer/multiwalled carbon nanotube and its application to paracetamol determination. Sens. Actuators B Chem. 2016, 233, 409–418. [Google Scholar] [CrossRef]
- Molina, B.G.; Cianga, L.; Bendrea, A.D.; Cianga, I.; Del Valle, L.J.; Estrany, F.; Alemán, C.; Armelin, E. Amphiphilic polypyrrole-poly(Schiff base) copolymers with poly(ethylene glycol) side chains: Synthesis, properties and applications. Polym. Chem. 2018, 9, 4218–4232. [Google Scholar] [CrossRef]
- Cui, M.; Song, Z.; Wu, Y.; Guo, B.; Fan, X.; Luo, X. A highly sensitive biosensor for tumor maker alpha fetoprotein based on poly(ethylene glycol) doped conducting polymer PEDOT. Biosens. Bioelectron. 2016, 79, 736–741. [Google Scholar] [CrossRef]
- Devnani, H.; Satsangee, S.P.; Jain, R. Nanocomposite modified electrochemical sensor for sensitive and selective determination of noradrenaline. Mater. Today Proc. 2016, 3, 1854–1863. [Google Scholar] [CrossRef]
- Xia, J.; Cao, X.; Wang, Z.; Yang, M.; Zhang, F.; Lu, B.; Li, F.; Xia, L.; Li, Y.; Xia, Y. Molecularly imprinted electrochemical biosensor based on chitosan/ionic liquid-graphene composites modified electrode for determination of bovine serum albumin. Sens. Actuators B Chem. 2016, 225, 305–311. [Google Scholar] [CrossRef]
- Ben-Yoav, H.; Winkler, T.E.; Kim, E.; Chocron, S.E.; Kelly, D.L.; Payne, G.F.; Ghodssi, R. Redox cycling-based amplifying electrochemical sensor for in situ clozapine antipsychotic treatment monitoring. Electrochim. Acta 2014, 130, 497–503. [Google Scholar] [CrossRef]
- Nordin, N.; Yusof, N.A.; Abdullah, J.; Radu, S.; Hajian, R. Characterization of polylactide-stabilized gold nanoparticles and its application in the fabrication of electrochemical DNA biosensors. J. Braz. Chem. Soc. 2016, 27, 1679–1686. [Google Scholar] [CrossRef]
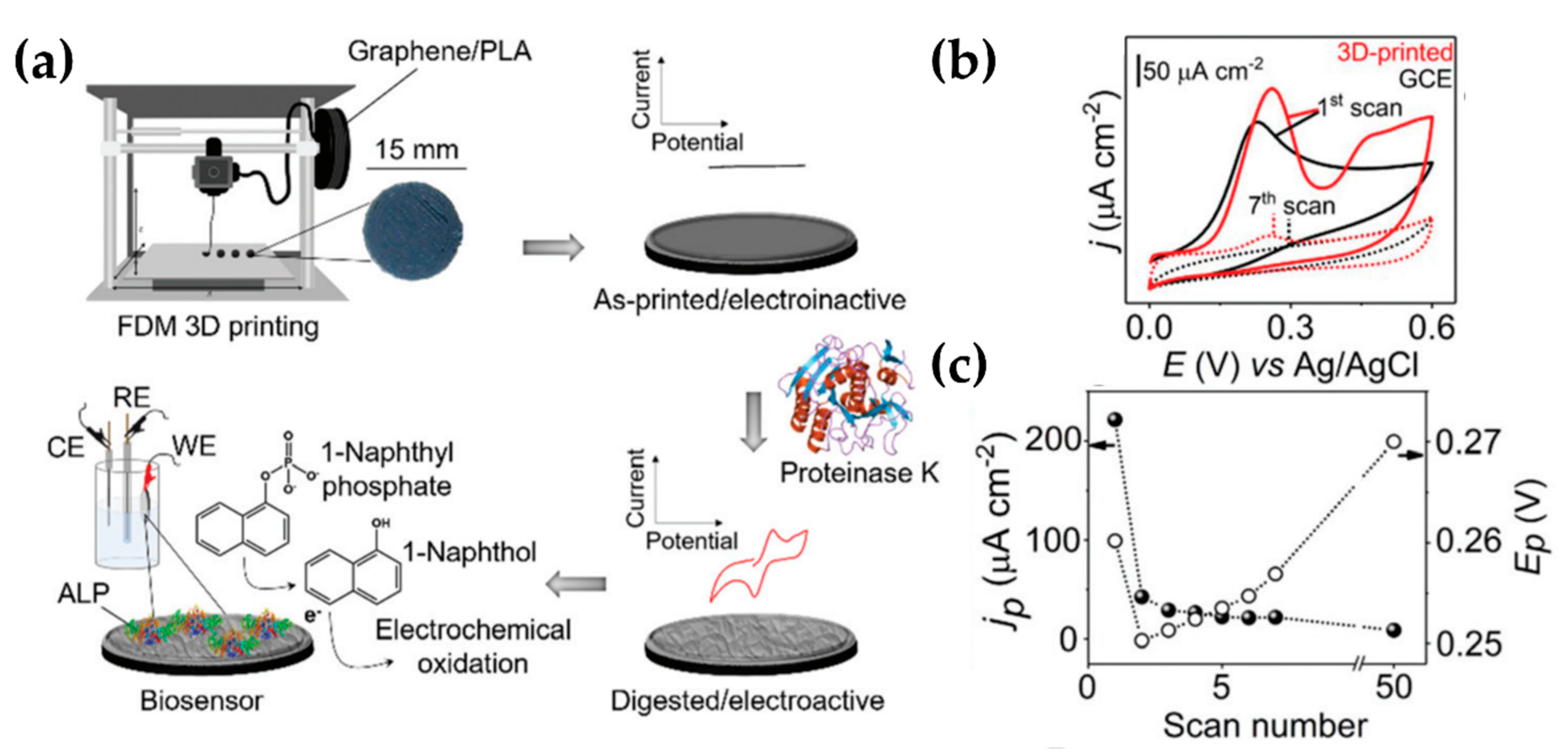
- Manzanares-Palenzuela, C.L.; Hermanova, S.; Sofer, Z.; Pumera, M. Proteinase-sculptured 3D-printed graphene/polylactic acid electrodes as potential biosensing platforms: Towards enzymatic modeling of 3D-printed structures. Nanoscale 2019, 11, 12124–12131. [Google Scholar] [CrossRef] [PubMed]
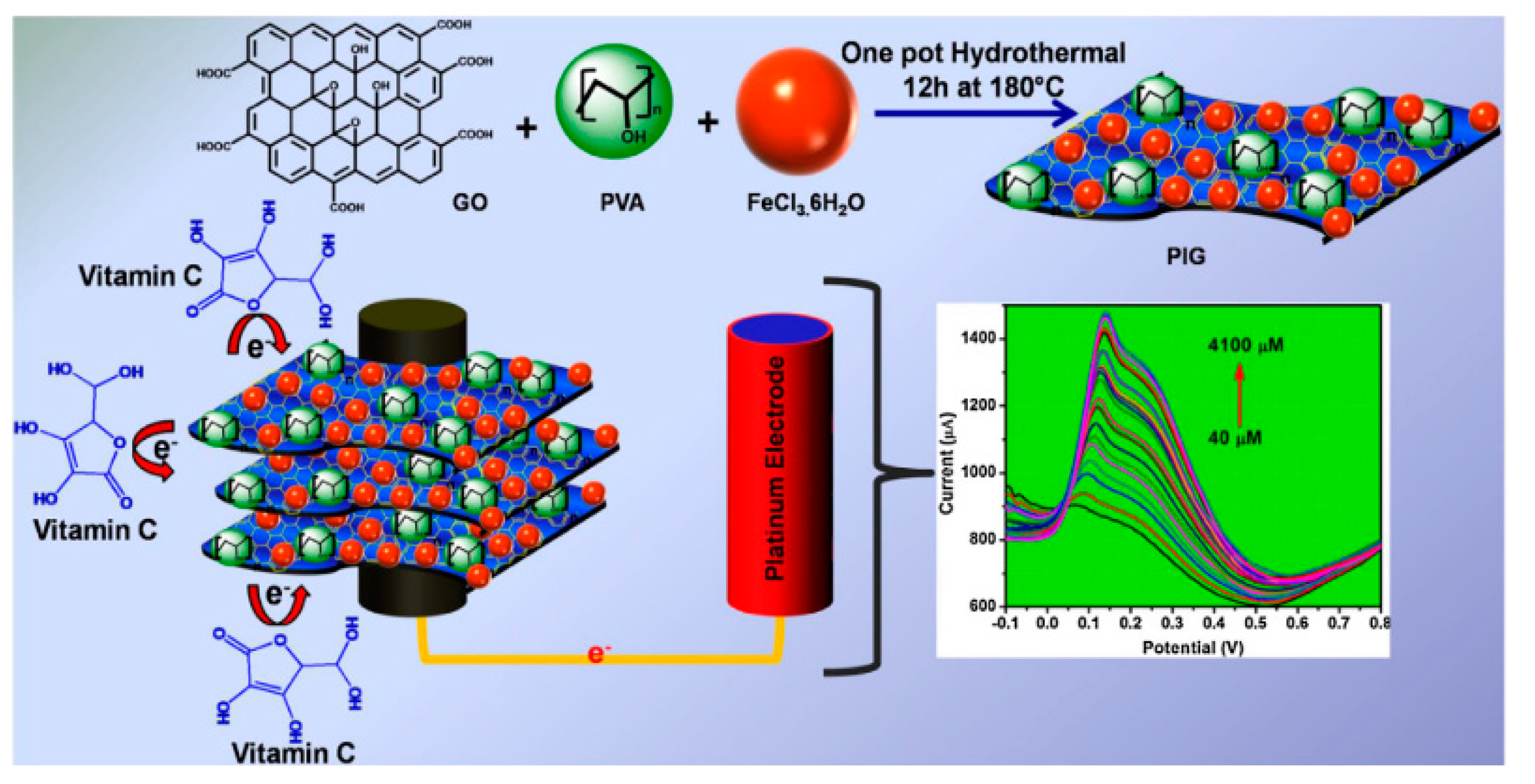
- Das, T.R.; Jena, S.K.; Madhuri, R.; Sharma, P.K. Polymeric iron oxide-graphene nanocomposite as a trace level sensor of vitamin C. Appl. Surf. Sci. 2018, 449, 304–313. [Google Scholar] [CrossRef]
- Fabregat, G.; Osorio, J.; Castedo, A.; Armelin, E.; Buendía, J.J.; Llorca, J.; Alemán, C. Plasma functionalized surface of commodity polymers for dopamine detection. Appl. Surf. Sci. 2017, 399, 638–647. [Google Scholar] [CrossRef][Green Version]
- Buendía, J.J.; Fabregat, G.; Castedo, A.; Llorca, J.; Alemán, C. Plasma-treated polyethylene as electrochemical mediator for enzymatic glucose sensors: Toward bifunctional glucose and dopamine sensors. Plasma Process. Polym. 2018, 15, 1–10. [Google Scholar] [CrossRef]
- de Oliveira, G.C.M.; de Souza Carvalho, J.H.; Brazaca, L.C.; Vieira, N.C.S.; Janegitz, B.C. Flexible platinum electrodes as electrochemical sensor and immunosensor for Parkinson’s disease biomarkers. Biosens. Bioelectron. 2020, 152, 112016. [Google Scholar] [CrossRef]
- Kalia, S. (Ed.) Polymeric Hydrogels as Smart Biomaterials; Springer Series on Polymer and Composite Materials; Springer International Publishing: Cham, Switzerland, 2016; ISBN 978-3-319-25320-6. [Google Scholar]
- Rahman, M.; Kumar, P.; Park, D.-S.; Shim, Y.-B. Electrochemical Sensors Based on Organic Conjugated Polymers. Sensors 2008, 8, 118–141. [Google Scholar] [CrossRef]
- Peng, H.; Zhang, L.; Soeller, C.; Travas-Sejdic, J. Conducting polymers for electrochemical DNA sensing. Biomaterials 2009, 30, 2132–2148. [Google Scholar] [CrossRef]
- Ghorbani Zamani, F.; Moulahoum, H.; Ak, M.; Odaci Demirkol, D.; Timur, S. Current trends in the development of conducting polymers-based biosensors. TrAC Trends Anal. Chem. 2019, 118, 264–276. [Google Scholar] [CrossRef]
- Tsakova, V.; Seeber, R. Conducting polymers in electrochemical sensing: Factors influencing the electroanalytical signal. Anal. Bioanal. Chem. 2016, 408, 7231–7241. [Google Scholar] [CrossRef] [PubMed]
- Sih, B.C.; Wolf, M.O. Metal nanoparticle-Conjugated polymer nanocomposites. Chem. Commun. 2005, 3375–3384. [Google Scholar] [CrossRef] [PubMed]
- Khan, I.; Saeed, K.; Khan, I. Nanoparticles: Properties, applications and toxicities. Arab. J. Chem. 2019, 12, 908–931. [Google Scholar] [CrossRef]
- Sarkar, S.; Guibal, E.; Quignard, F.; SenGupta, A.K. Polymer-supported metals and metal oxide nanoparticles: Synthesis, characterization, and applications. J. Nanoparticle Res. 2012, 14. [Google Scholar] [CrossRef]
- Sangamithirai, D.; Munusamy, S.; Narayanan, V.; Stephen, A. A strategy to promote the electroactive platform adopting poly(o-anisidine)-silver nanocomposites probed for the voltammetric detection of NADH and dopamine. Mater. Sci. Eng. C 2017, 80, 425–437. [Google Scholar] [CrossRef]
- Kim, D.-S.; Kang, E.-S.; Baek, S.; Choo, S.-S.; Chung, Y.-H.; Lee, D.; Min, J.; Kim, T.-H. Electrochemical detection of dopamine using periodic cylindrical gold nanoelectrode arrays. Sci. Rep. 2018, 8, 14049. [Google Scholar] [CrossRef]
- Li, W.; Sauve, A.A. NAD+ Content and Its Role in Mitochondria. In Mitochondrial Regulation; Palmeira, C., Rolo, A., Eds.; Methods in Molecular Biology (Methods and Protocols); Humana Press: New York, NY, USA, 2015; Volume 1241, pp. 39–48. [Google Scholar]
- Jia, H.; Gao, X.; Chen, Z.; Liu, G.; Zhang, X.; Yan, H.; Zhou, H.; Zheng, L. The high yield synthesis and characterization of gold nanoparticles with superior stability and their catalytic activity. CrystEngComm 2012, 14, 7600–7606. [Google Scholar] [CrossRef]
- Veith, G.M.; Lupini, A.R.; Rashkeev, S.; Pennycook, S.J.; Mullins, D.R.; Schwartz, V.; Bridges, C.A.; Dudney, N.J. Thermal stability and catalytic activity of gold nanoparticles supported on silica. J. Catal. 2009, 262, 92–101. [Google Scholar] [CrossRef]
- Signoretto, M.; Menegazzo, F.; Trevisan, V.; Pinna, F.; Manzoli, M.; Boccuzzi, F. Investigation on the stability of supported gold nanoparticles. Catalysts 2013, 3, 656–670. [Google Scholar] [CrossRef]
- Fu, S.; Fan, G.; Yang, L.; Li, F. Non-enzymatic glucose sensor based on Au nanoparticles decorated ternary Ni-Al layered double hydroxide/single-walled carbon nanotubes/graphene nanocomposite. Electrochim. Acta 2015, 152, 146–154. [Google Scholar] [CrossRef]
- Sedghi, R.; Pezeshkian, Z. Fabrication of non-enzymatic glucose sensor based on nanocomposite of MWCNTs-COOH-Poly(2-aminothiophenol)-Au NPs. Sens. Actuators B Chem. 2015, 219, 119–124. [Google Scholar] [CrossRef]
- Xianyu, Y.; Sun, J.; Li, Y.; Tian, Y.; Wang, Z.; Jiang, X. An ultrasensitive, non-enzymatic glucose assay via gold nanorod-assisted generation of silver nanoparticles. Nanoscale 2013, 5, 6303–6306. [Google Scholar] [CrossRef] [PubMed]
- Fabregat, G.; Córdova-Mateo, E.; Armelin, E.; Bertran, O.; Alemán, C. Ultrathin films of polypyrrole derivatives for dopamine detection. J. Phys. Chem. C 2011, 115, 14933–14941. [Google Scholar] [CrossRef]
- Fabregat, G.; Armelin, E.; Alemán, C. Selective detection of dopamine combining multilayers of conducting polymers with gold nanoparticles. J. Phys. Chem. B 2014, 118, 4669–4682. [Google Scholar] [CrossRef]
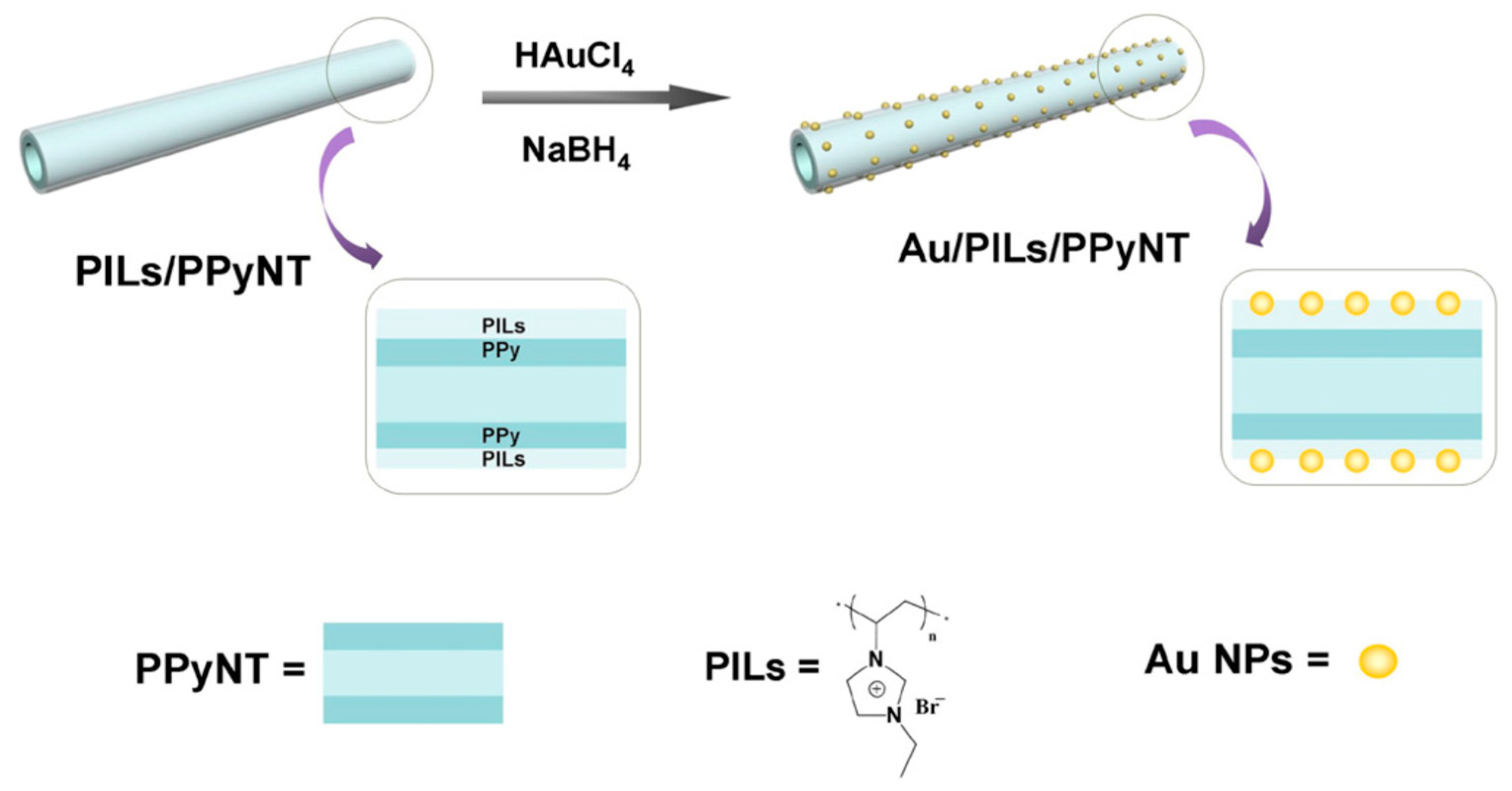
- Mao, H.; Zhang, H.; Jiang, W.; Liang, J.; Sun, Y.; Zhang, Y.; Wu, Q.; Zhang, G.; Song, X.M. Poly(ionic liquid) functionalized polypyrrole nanotubes supported gold nanoparticles: An efficient electrochemical sensor to detect epinephrine. Mater. Sci. Eng. C 2017, 75, 495–502. [Google Scholar] [CrossRef] [PubMed]
- Sacha, G.L.; Bauer, S.R.; Lat, I. Vasoactive Agent Use in Septic Shock: Beyond First-Line Recommendations. Pharmacother. J. Hum. Pharmacol. Drug Ther. 2019, 39, 369–381. [Google Scholar] [CrossRef]
- Yang, J.; Cho, M.; Pang, C.; Lee, Y. Highly sensitive non-enzymatic glucose sensor based on over-oxidized polypyrrole nanowires modified with Ni(OH)2 nanoflakes. Sens. Actuators B Chem. 2015, 211, 93–101. [Google Scholar] [CrossRef]
- Özcan, L.; Şahin, Y.; Türk, H. Non-enzymatic glucose biosensor based on overoxidized polypyrrole nanofiber electrode modified with cobalt(II) phthalocyanine tetrasulfonate. Biosens. Bioelectron. 2008, 24, 512–517. [Google Scholar] [CrossRef]
- Priyadarsini, S.; Mohanty, S.; Mukherjee, S.; Basu, S.; Mishra, M. Graphene and graphene oxide as nanomaterials for medicine and biology application. J. Nanostructure Chem. 2018, 8, 123–137. [Google Scholar] [CrossRef]
- Wang, Y.; Li, Z.; Wang, J.; Li, J.; Lin, Y. Graphene and graphene oxide: Biofunctionalization and applications in biotechnology. Trends Biotechnol. 2011, 29, 205–212. [Google Scholar] [CrossRef]
- Mittal, G.; Dhand, V.; Rhee, K.Y.; Park, S.J.; Lee, W.R. A review on carbon nanotubes and graphene as fillers in reinforced polymer nanocomposites. J. Ind. Eng. Chem. 2015, 21, 11–25. [Google Scholar] [CrossRef]
- Tang, L.; Zhao, L.; Guan, L. 7 Graphene/Polymer Composite Materials: Processing, Properties and Applications. In Advanced Composite Materials: Properties and Applications; De Gruyter Open: Warsaw, Poland, 2017. [Google Scholar]
- Papageorgiou, D.G.; Li, Z.; Liu, M.; Kinloch, I.A.; Young, R.J. Mechanisms of mechanical reinforcement by graphene and carbon nanotubes in polymer nanocomposites. Nanoscale 2020, 12, 2228–2267. [Google Scholar] [CrossRef] [PubMed]
- Novoselov, K.S. Electric Field Effect in Atomically Thin Carbon Films. Science 2004, 306, 666–669. [Google Scholar] [CrossRef] [PubMed]
- Nobel Prize Organisation The Nobel Prize in Physics. 2010. Available online: https://www.nobelprize.org/prizes/physics/2010/prize-announcement/ (accessed on 26 April 2020).
- Wang, L.; Jiang, T.; Song, Y.; Shi, W.; Cai, X. Dopamine detection using a patch-clamp system on a planar microeletrode array electrodeposited by polypyrrole/graphene nanocomposites. Sci. China Technol. Sci. 2014, 57, 288–292. [Google Scholar] [CrossRef]
- Wang, W.; Xu, G.; Cui, X.T.; Sheng, G.; Luo, X. Enhanced catalytic and dopamine sensing properties of electrochemically reduced conducting polymer nanocomposite doped with pure graphene oxide. Biosens. Bioelectron. 2014, 58, 153–156. [Google Scholar] [CrossRef] [PubMed]
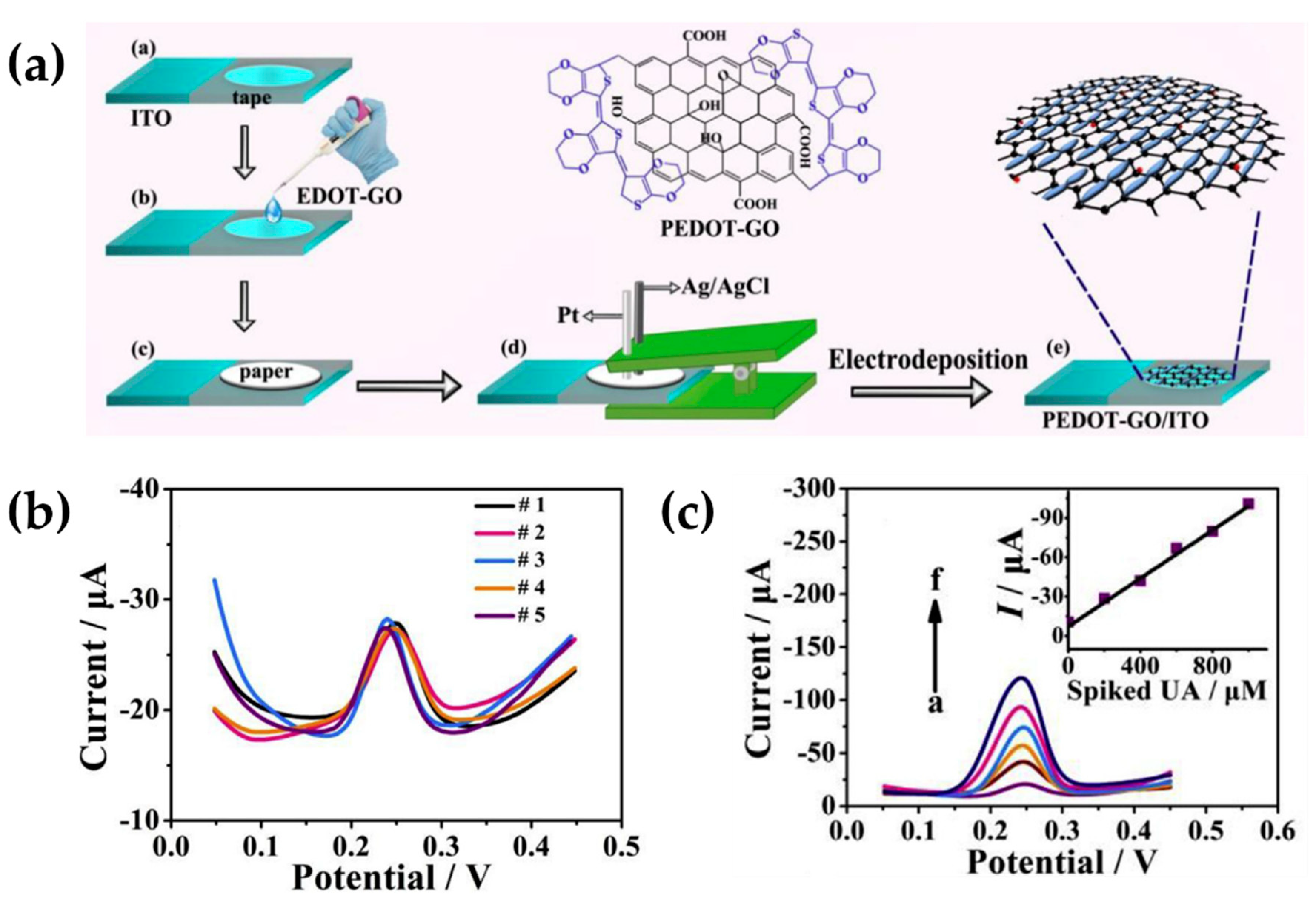
- Huang, X.; Shi, W.; Li, J.; Bao, N.; Yu, C.; Gu, H. Determination of salivary uric acid by using poly(3,4-ethylenedioxythipohene) and graphene oxide in a disposable paper-based analytical device. Anal. Chim. Acta 2020, 1103, 75–83. [Google Scholar] [CrossRef]
- Sadanandhan, N.K.; Cheriyathuchenaaramvalli, M.; Devaki, S.J.; Ravindranatha Menon, A.R. PEDOT-reduced graphene oxide-silver hybrid nanocomposite modified transducer for the detection of serotonin. J. Electroanal. Chem. 2017, 794, 244–253. [Google Scholar] [CrossRef]
- Kempe, M. CHIRAL SEPARATIONS | Molecular Imprints As Stationary Phases. In Encyclopedia of Separation Science; Elsevier: Cambridge, MA, USA, 2000; pp. 2387–2397. [Google Scholar]
- Bitas, D.; Samanidou, V. Carbon nanotubes as sorbent materials for the extraction of pharmaceutical products followed by chromatographic analysis. In Fullerens, Graphenes and Nanotubes A Pharmaceutical Approach; Elsevier: Cambridge, MA, USA, 2018; Chapter 5; pp. 135–168. [Google Scholar]
- Qian, T.; Yu, C.; Zhou, X.; Ma, P.; Wu, S.; Xu, L.; Shen, J. Ultrasensitive dopamine sensor based on novel molecularly imprinted polypyrrole coated carbon nanotubes. Biosens. Bioelectron. 2014, 58, 237–241. [Google Scholar] [CrossRef]
- Lee, C.Y.; Hsu, D.Y.; Prasannan, A.; Kalaivani, R.; Hong, P. Da Facile synthesis of hexagonal-shaped polypyrrole self-assembled particles for the electrochemical detection of dopamine. Appl. Surf. Sci. 2016, 363, 451–458. [Google Scholar] [CrossRef]
- Wei, F.; Liao, W.; Xu, Z.; Yang, Y.; Wong, D.T.; Ho, C.-M. Bio/Abiotic Interface Constructed from Nanoscale DNA Dendrimer and Conducting Polymer for Ultrasensitive Biomolecular Diagnosis. Small 2009, 5, 1784–1790. [Google Scholar] [CrossRef]
- Chauhan, N.; Chawla, S.; Pundir, C.S.; Jain, U. An electrochemical sensor for detection of neurotransmitter-acetylcholine using metal nanoparticles, 2D material and conducting polymer modified electrode. Biosens. Bioelectron. 2017, 89, 377–383. [Google Scholar] [CrossRef] [PubMed]
- Adeosun, W.A.; Asiri, A.M.; Marwani, H.M.; Rahman, M.M. Enzymeless Electrocatalytic Detection of Uric Acid Using Polydopamine/Polypyrrole Copolymeric film. ChemistrySelect 2020, 5, 156–164. [Google Scholar] [CrossRef]
- Zhang, P.; Xu, Q.; Du, J.; Wang, Y. Polydopamine-based nanoparticles with excellent biocompatibility for photothermally enhanced gene delivery. RSC Adv. 2018, 8, 34596–34602. [Google Scholar] [CrossRef]
- Zavareh, S.; Mahdi, M.; Erfanian, S.; Hashemi-Moghaddam, H. Synthesis of polydopamine as a new and biocompatible coating of magnetic nanoparticles for delivery of doxorubicin in mouse breast adenocarcinoma. Cancer Chemother. Pharmacol. 2016, 78, 1073–1084. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.; Jang, L.K.; Jang, M.; Lee, S.; Hardy, J.G.; Lee, J.Y. Electrically Conductive Polydopamine-Polypyrrole as High Performance Biomaterials for Cell Stimulation in Vitro and Electrical Signal Recording in Vivo. ACS Appl. Mater. Interfaces 2018, 10, 33032–33042. [Google Scholar] [CrossRef] [PubMed]
- Molina, B.G.; Bendrea, A.D.; Cianga, L.; Armelin, E.; Del Valle, L.J.; Cianga, I.; Alemán, C. The biocompatible polythiophene-: G-polycaprolactone copolymer as an efficient dopamine sensor platform. Polym. Chem. 2017, 8, 6112–6122. [Google Scholar] [CrossRef]
- Molina, B.G.; Cianga, L.; Bendrea, A.D.; Cianga, I.; Alemán, C.; Armelin, E. An amphiphilic, heterografted polythiophene copolymer containing biocompatible/biodegradable side chains for use as an (electro)active surface in biomedical applications. Polym. Chem. 2019, 10, 5010–5022. [Google Scholar] [CrossRef]
- Piro, B.; Dang, L.A.; Pham, M.C.; Fabiano, S.; Tran-Minh, C. A glucose biosensor based on modified-enzyme incorporated within electropolymerised poly(3,4-ethylenedioxythiophene) (PEDT) films. J. Electroanal. Chem. 2001, 512, 101–109. [Google Scholar] [CrossRef][Green Version]
- Kaur, G.; Adhikari, R.; Cass, P.; Bown, M.; Gunatillake, P. Electrically conductive polymers and composites for biomedical applications. RSC Adv. 2015, 5, 37553–37567. [Google Scholar] [CrossRef]
- Saadattalab, V.; Shakeri, A.; Gholami, H. Effect of CNTs and nano ZnO on physical and mechanical properties of polyaniline composites applicable in energy devices. Prog. Nat. Sci. Mater. Int. 2016, 26, 517–522. [Google Scholar] [CrossRef]
- Molina, B.G.; Del Valle, L.J.; Turon, P.; Armelin, E.; Alemán, C. Electrochemical Sensor for Bacterial Metabolism Based on the Detection of NADH by Polythiophene Nanoparticles. J. Phys. Chem. C 2019, 123, 22181–22190. [Google Scholar] [CrossRef]
- Jiang, L.; Wang, R.; Li, X.; Jiang, L.; Lu, G. Electrochemical oxidation behavior of nitrite on a chitosan-carboxylated multiwall carbon nanotube modified electrode. Electrochem. Commun. 2005, 7, 597–601. [Google Scholar] [CrossRef]
- Cao, X.; Luo, L.; Ding, Y.; Zou, X.; Bian, R. Electrochemical methods for simultaneous determination of dopamine and ascorbic acid using cetylpyridine bromide/chitosan composite film-modified glassy carbon electrode. Sens. Actuators B Chem. 2008, 129, 941–946. [Google Scholar] [CrossRef]
- Jia, X.; Tan, L.; Xie, Q.; Zhang, Y.; Yao, S. Quartz crystal microbalance and electrochemical cytosensing on a chitosan/multiwalled carbon nanotubes/Au electrode. Sens. Actuators B Chem. 2008, 134, 273–280. [Google Scholar] [CrossRef]
- Li, M.; Huang, S.; Zhu, P.; Kong, L.; Peng, B.; Gao, H. A novel DNA biosensor based on ssDNA/Cyt c/l-Cys/GNPs/Chits/GCE. Electrochim. Acta 2009, 54, 2284–2289. [Google Scholar] [CrossRef]
- Zhang, M.; Smith, A.; Gorski, W. Carbon nanotube-chitosan system for electrochemical sensing based on dehydrogenase enzymes. Anal. Chem. 2004, 76, 5045–5050. [Google Scholar] [CrossRef]
- Gong, J.; Wang, L.; Zhao, K.; Song, D.; Zhang, L. Bifunctional sensor of nitric oxide and oxygen based on hematite nanotubes embedded in chitosan matrix. Electrochem.Commun. 2008, 10, 1222–1225. [Google Scholar] [CrossRef]
- Da Silva, S.B.; Batista, G.L.; Santin, C.K. Chitosan for Sensors and Electrochemical Applications. In Chitin and Chitosan: Properties and Applications; van den Broek, L.A.M., Boeriu, C.G., Eds.; John Wiley & Sons Ltd.: Chichester, UK, 2019. [Google Scholar] [CrossRef]
- Kuralay, F.; Vural, T.; Bayram, C.; Denkbas, E.B.; Abaci, S. Carbon nanotube-chitosan modified disposable pencil graphite electrode for Vitamin B 12 analysis. Colloids Surfaces B Biointerfaces 2011, 87, 18–22. [Google Scholar] [CrossRef]
- Tomčik, P.; Banks, C.E.; Davies, T.J.; Compton, R.G. A Self-Catalytic Carbon Paste Electrode for the Detection of Vitamin B 12. Anal. Chem. 2004, 76, 161–165. [Google Scholar] [CrossRef]
- Lin, M.S.; Leu, H.J.; Lai, C.H. Development of Vitamin B12 based disposable sensor for dissolved oxygen. Anal. Chim. Acta 2006, 561, 164–170. [Google Scholar] [CrossRef]
- Zagal, J.H.; Aguirre, M.J.; Páez, M.A. O2 reduction kinetics on a graphite electrode modified with adsorbed vitamin B12. J. Electroanal. Chem. 1997, 437, 45–52. [Google Scholar] [CrossRef]
- Zhang, X.; Wang, S. Voltametric behavior of noradrenaline at 2-mercaptoethanol self-assembled monolayer modified gold electrode and its analytical application. Sensors 2003, 3, 61–68. [Google Scholar] [CrossRef]
- Taheri, A.R.; Mohadesi, A.; Afzali, D.; Karimi-Maleh, H.; Moghaddam, H.M.; Zamani, H.; Zad, Z.R. Simultaneous voltammetric determination of norepinephrine and folic acid at the surface of modified carbon nanotube paste electrode. Int. J. Electrochem. Sci. 2011, 6, 171–180. [Google Scholar]
- Huang, S.H.; Liao, H.H.; Chen, D.H. Simultaneous determination of norepinephrine, uric acid, and ascorbic acid at a screen printed carbon electrode modified with polyacrylic acid-coated multi-wall carbon nanotubes. Biosens. Bioelectron. 2010, 25, 2351–2355. [Google Scholar] [CrossRef]
- Yang, D. Advances in Nanocomposites-Synthesis, Characterization and Industrial Applications; Reddy, B., Ed.; InTechOPen: London, UK, 2011; ISBN 978-953-307-165-7. [Google Scholar]
- Wang, Y.; Wang, E.L.; Wu, Z.M.; Li, H.; Zhu, Z.; Zhu, X.S.; Dong, Y. Synthesis of chitosan molecularly imprinted polymers for solid-phase extraction of methandrostenolone. Carbohydr. Polym. 2014, 101, 517–523. [Google Scholar] [CrossRef]
- Liu, Y.; Dong, X.; Chen, P. Biological and chemical sensors based on graphene materials. Chem. Soc. Rev. 2012, 41, 2283–2307. [Google Scholar] [CrossRef]
- Kuila, T.; Bose, S.; Khanra, P.; Mishra, A.K.; Kim, N.H.; Lee, J.H. Recent advances in graphene-based biosensors. Biosens. Bioelectron. 2011, 26, 4637–4648. [Google Scholar] [CrossRef]
- Ashworth, M.R.F. Analytical Methods. In Chemistry of Sulphonic Acids, Esters and Their Derivatives; John Wiley & Sons, Ltd.: Hoboken, NJ, USA, 2006; pp. 323–350. [Google Scholar] [CrossRef]
- Safavi, A.; Maleki, N.; Moradlou, O.; Sorouri, M. Direct electrochemistry of hemoglobin and its electrocatalytic effect based on its direct immobilization on carbon ionic liquid electrode. Electrochem. Commun. 2008, 10, 420–423. [Google Scholar] [CrossRef]
- Zhao, L.; Zhao, F.; Zeng, B. Preparation and application of sunset yellow imprinted ionic liquid polymer - Ionic liquid functionalized graphene composite film coated glassy carbon electrodes. Electrochim. Acta 2014, 115, 247–254. [Google Scholar] [CrossRef]
- Shan, C.; Yang, H.; Han, D.; Zhang, Q.; Ivaska, A.; Niu, L. Electrochemical determination of NADH and ethanol based on ionic liquid-functionalized graphene. Biosens. Bioelectron. 2010, 25, 1504–1508. [Google Scholar] [CrossRef]
- Shu, Y.; Liu, M.; Chen, S.; Chen, X.; Wang, J. New insight into molecular interactions of imidazolium ionic liquids with bovine serum albumin. J. Phys. Chem. B 2011, 115, 12306–12314. [Google Scholar] [CrossRef] [PubMed]
- Gomez, I.J.; Arnaiz, B.; Cacioppo, M.; Arcudi, F.; Prato, M. Nitrogen-doped Carbon Nanodots for bioimaging and delivery of paclitaxel. J. Mater. Chem. B 2018, 6. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Wei, T.X. Surface plasmon resonance sensor chips for the recognition of bovine serum albumin via electropolymerized molecularly imprinted polymers. Chinese Chem. Lett. 2013, 24, 813–816. [Google Scholar] [CrossRef]
- Chen, H.J.; Zhang, Z.H.; Luo, L.J.; Yao, S.Z. Surface-imprinted chitosan-coated magnetic nanoparticles modified multi-walled carbon nanotubes biosensor for detection of bovine serum albumin. Sens. Actuators B Chem. 2012, 163, 76–83. [Google Scholar] [CrossRef]
- Pridgen, E.M.; Langer, R.; Farokhzad, O.C. Nanoparticles for Cancer Diagnosis & Therapeutics-Review Biodegradable, polymeric nanoparticle delivery systems for cancer therapy. Nanomedicine 2007, 2, 269. [Google Scholar]
- Babu, A.; Templeton, A.K.; Munshi, A.; Ramesh, R. Nanoparticle-based drug delivery for therapy of lung cancer: Progress and challenges. J. Nanomater. 2013, 2013. [Google Scholar] [CrossRef]
- Chen, H.Y.; Huang, B.H.; Lin, C.C. A highly efficient initiator for the ring-opening polymerization of lactides and ε-caprolactone: A kinetic study. Macromolecules 2005, 38, 5400–5405. [Google Scholar] [CrossRef]
- Manzanares Palenzuela, C.L.; Pumera, M. (Bio)Analytical chemistry enabled by 3D printing: Sensors and biosensors. TrAC Trends Anal. Chem. 2018, 103, 110–118. [Google Scholar] [CrossRef]
- Manzanares Palenzuela, C.L.; Novotný, F.; Krupička, P.; Sofer, Z.; Pumera, M. 3D-Printed Graphene/Polylactic Acid Electrodes Promise High Sensitivity in Electroanalysis. Anal. Chem. 2018, 90, 5753–5757. [Google Scholar] [CrossRef]
- Meyersabellek, W.; Sinha, P.; Kottgen, E. Alkaline phosphatase: Laboratory and clinical implications. J. Chromatogr. B Biomed. Sci. Appl. 1988, 429, 419–444. [Google Scholar] [CrossRef]
- Abdullah, Z.W.; Dong, Y. Biodegradable and water resistant poly(vinyl) alcohol (PVA)/starch (ST)/glycerol (GL)/halloysite nanotube (HNT) nanocomposite films for sustainable food packaging. Front. Mater. 2019, 6, 1–17. [Google Scholar] [CrossRef]
- Zuo, S.; Teng, Y.; Yuan, H.; Lan, M. Direct electrochemistry of glucose oxidase on screen-printed electrodes through one-step enzyme immobilization process with silica sol-gel/polyvinyl alcohol hybrid film. Sens. Actuators B Chem. 2008, 133, 555–560. [Google Scholar] [CrossRef]
- Ahammad, S.Z.; Gomes, J.; Sreekrishnan, T.R. Wastewater treatment forproductionofH2S-free biogas. J. Chem. Technol. Biotechnol. 2008, 83, 1163–1169. [Google Scholar] [CrossRef]
- Lad, U.; Kale, G.M.; Bryaskova, R. Glucose oxidase encapsulated polyvinyl alcohol-silica hybrid films for an electrochemical glucose sensing electrode. Anal. Chem. 2013, 85, 6349–6355. [Google Scholar] [CrossRef] [PubMed]
- Dorsey, E.R.; Elbaz, A.; Nichols, E.; Abd-Allah, F.; Abdelalim, A.; Adsuar, J.C.; Dahodwala, N. Global, regional, and national burden of Parkinson’s disease, 1990–2016: A systematic analysis for the Global Burden of Disease Study. Lancet Neurol. 2018, 17, 939–953. [Google Scholar] [CrossRef]
- Saito, Y. DJ-1 as a biomarker of Parkinson’s disease. In DJ-1/PARK7 Protein; Ariga, H., Iguchi-Ariga, S.M.M., Eds.; Advances in Experimental Medicine and Biology; Springer: Singapore, 2017; pp. 149–171. [Google Scholar]
- Kaltenbrunner, M.; Sekitani, T.; Reeder, J.; Yokota, T.; Kuribara, K.; Tokuhara, T.; Drack, M.; Schwödiauer, R.; Graz, I.; Bauer-Gogonea, S.; et al. An ultra-lightweight design for imperceptible plastic electronics. Nature 2013, 499, 458–463. [Google Scholar] [CrossRef]
- Gualandi, I.; Marzocchi, M.; Achilli, A.; Cavedale, D.; Bonfiglio, A.; Fraboni, B. Textile Organic Electrochemical Transistors as a Platform for Wearable Biosensors. Sci. Rep. 2016, 6, 33637. [Google Scholar] [CrossRef]
- Bandodkar, A.J.; Jia, W.; Wang, J. Tattoo-Based Wearable Electrochemical Devices: A Review. Electroanalysis 2015, 27, 562–572. [Google Scholar] [CrossRef]
- Mitsubayashi, K.; Arakawa, T. Cavitas Sensors: Contact Lens Type Sensors & Mouthguard Sensors. Electroanalysis 2016, 28, 1170–1187. [Google Scholar] [CrossRef]
- Pankratov, D.; González-Arribas, E.; Blum, Z.; Shleev, S. Tear Based Bioelectronics. Electroanalysis 2016, 28, 1250–1266. [Google Scholar] [CrossRef]
- Imani, S.; Bandodkar, A.J.; Mohan, A.M.V.; Kumar, R.; Yu, S.; Wang, J.; Mercier, P.P. A wearable chemical-electrophysiological hybrid biosensing system for real-time health and fitness monitoring. Nat. Commun. 2016, 7, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Green, J.M.; Pritchett, R.C.; Crews, T.R.; McLester, J.R.; Tucker, D.C. Sweat lactate response between males with high and low aerobic fitness. Eur. J. Appl. Physiol. 2004, 91, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Gao, W.; Emaminejad, S.; Nyein, H.Y.Y.; Challa, S.; Chen, K.; Peck, A.; Fahad, H.M.; Ota, H.; Shiraki, H.; Kiriya, D.; et al. Fully integrated wearable sensor arrays for multiplexed in situ perspiration analysis. Nature 2016, 529, 509–514. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.; Valdes-Ramırez, G.; Bandodkar, A.J.; Jia, W.; Martinez, A.G.; Ramırez, J.; Mercierb, P.; Wang, A. Non-invasive mouthguard biosensor for continuous salivary monitoring of metabolites. Analyst 2014, 139, 1632–1636. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.; Imani, S.; de Araujo, W.R.; Warchall, J.; Valdés-Ramírez, G.; Paixão, T.R.L.C.; Mercier, P.P.; Wang, J. Wearable salivary uric acid mouthguard biosensor with integrated wireless electronics. Physiol. Behav. 2016, 176, 139–148. [Google Scholar] [CrossRef] [PubMed]
- Liao, Y.T.; Yao, H.; Lingley, A.; Parviz, B.; Otis, B.P. A 3-μW CMOS glucose sensor for wireless contact-lens tear glucose monitoring. IEEE J. Solid-State Circuits 2012, 47, 335–344. [Google Scholar] [CrossRef]
- Thomas, N.; Lähdesmäki, I.; Parviz, B.A. A contact lens with an integrated lactate sensor. Sens. Actuators B Chem. 2012, 162, 128–134. [Google Scholar] [CrossRef]
- Yao, H.; Shum, A.J.; Cowan, M.; Lähdesmäki, I.; Parviz, B.A. A contact lens with embedded sensor for monitoring tear glucose level. Biosens. Bioelectron. 2011, 26, 3290–3296. [Google Scholar] [CrossRef]





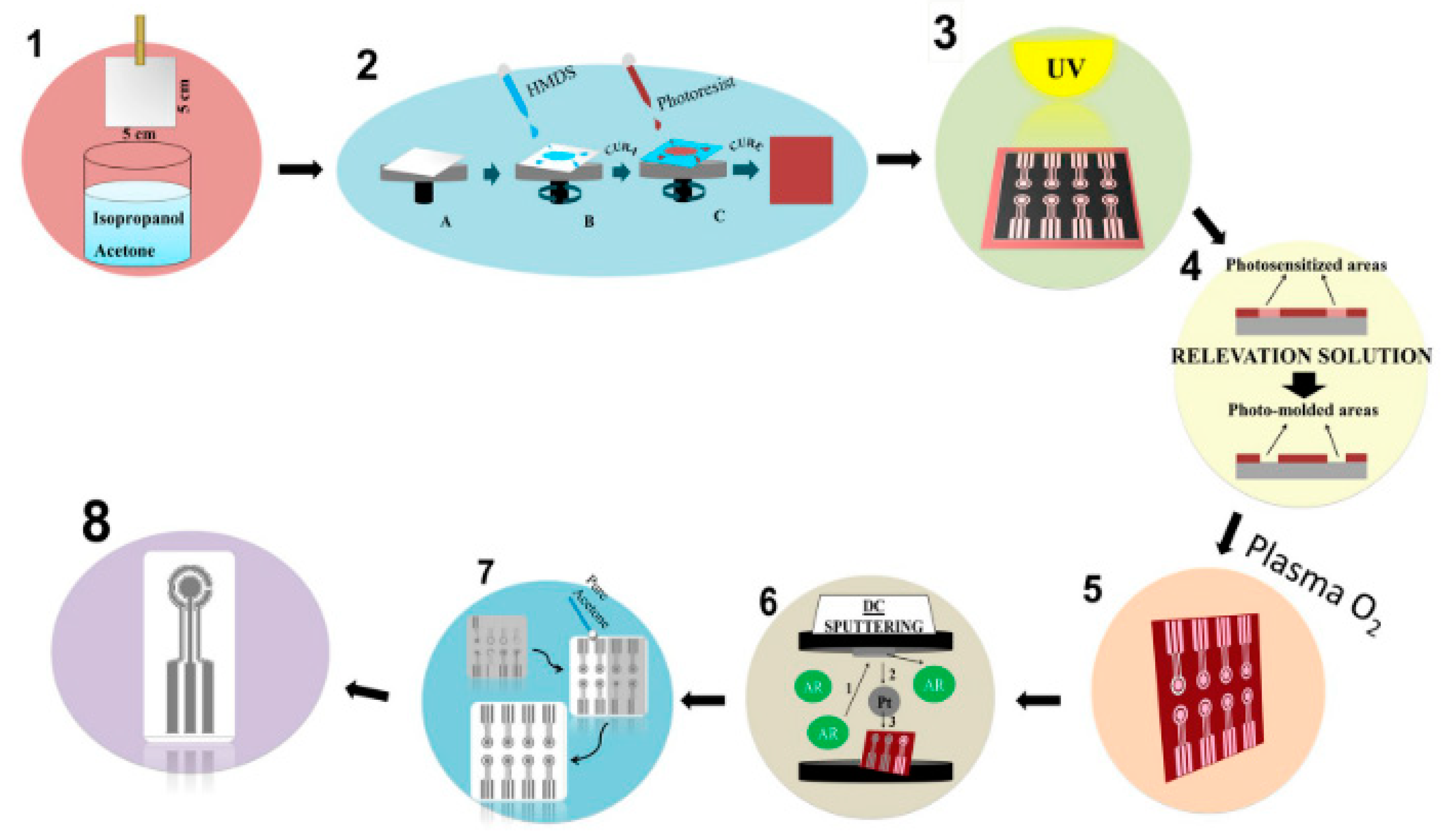
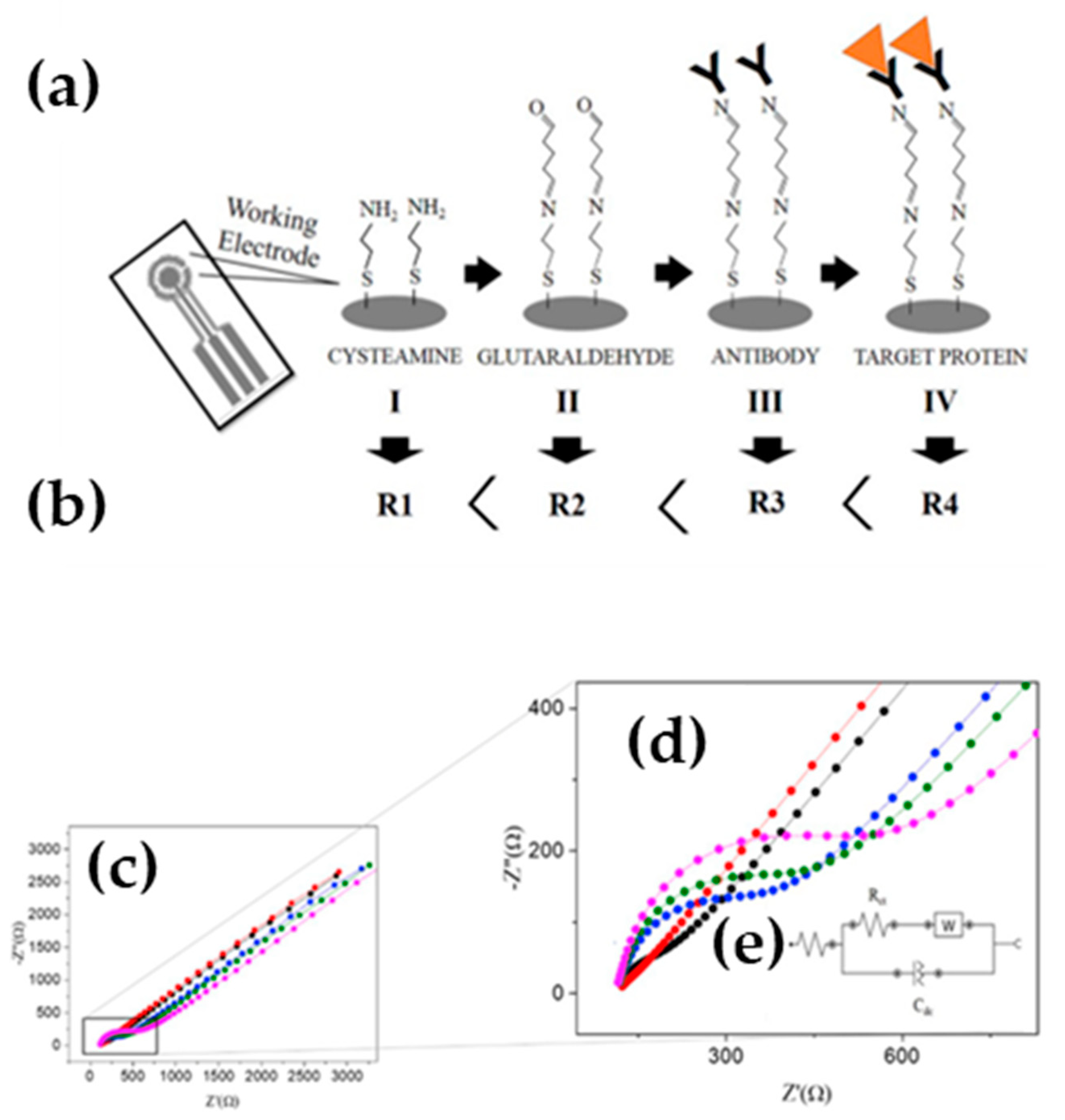
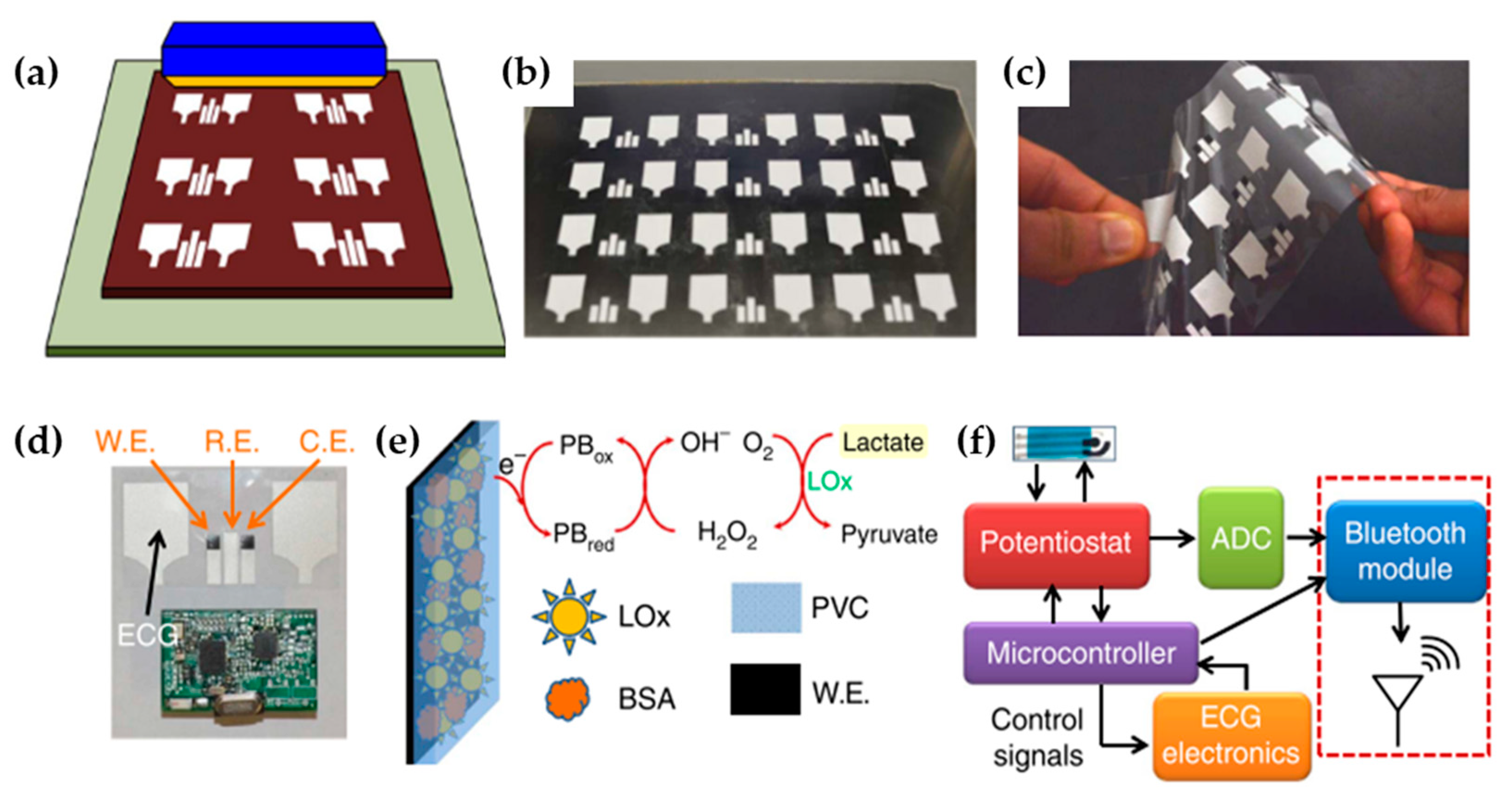
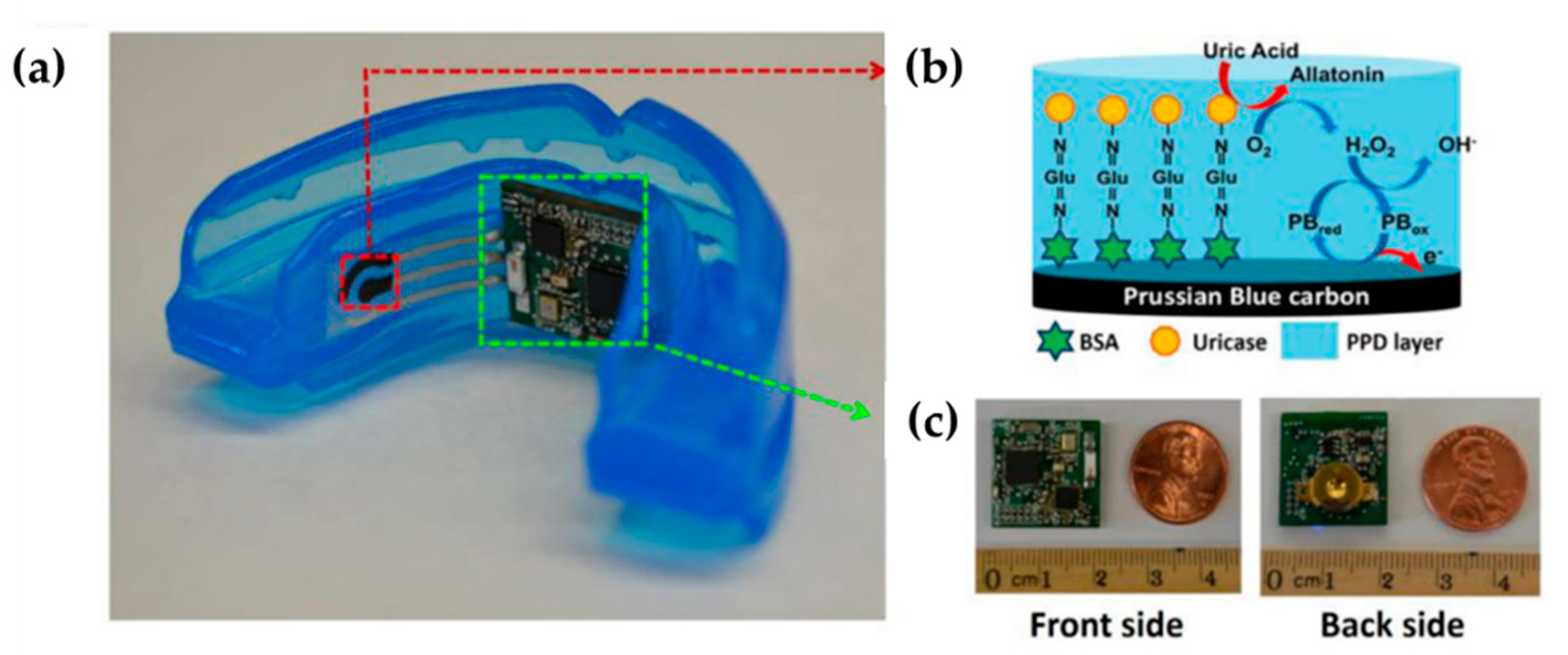
| Authors (Year) | Polymer Material 1) | Molecules Detected (EC Technique Used) 2) | Polymer Modified (Type of Modification) 3) | Limit of Detection 4) (Detection Range) | Improvements |
|---|---|---|---|---|---|
| Poletti Papi et al. [32] (2017) | PPy | Glucose, bioproduct (CA) | PPy-AgNPs (Structural, reverse microemulsion method) | 3.6 μM (25–2500 μmol L−1) | Sensitivity |
| Ansari et al. [33] (2019) | PAB | Glucose, bioproduct (CV) | PAB/AuNPs (Superficial, seed-assisted growth method) | 0.4 µM (2–250 μM) | Sensitivity |
| Sha et al. [34] (2017) | PAni | Urea, bioproduct (CA) | PAni/Gr (Superficial, electrodeposition) | 5.88 μM (10–200 mM) | Sensitivity |
| Weaver et al. [35] (2014) | PEDOT | Dopamine, neurotransmitters (CV) | PEDOT-GO (Structural, electropolymerization) | 83 nM (1–40 mM) | Sensitivity and selectivity |
| Sun et al. [36] (2013) | PPy | Quercetin, flavonoid (DPV) | MIP PPy-Gr (Structural, MIT- electropolymerization) | 4.8 × 10−8 mol/L (6.0 × 10−4–1.5 × 10−2 mM ) | Sensitivity and selectivity |
| Radhakrishnan et al. [37] (2013) | PPy/PANi | DNA, biomolecule (DPV) | PPy/PANi/GA/ssDNA (Superficial, oxidative polymerization and biologic immobilization) | 50 fM (10−6–10−10 mM) | Sensitivity and selectivity |
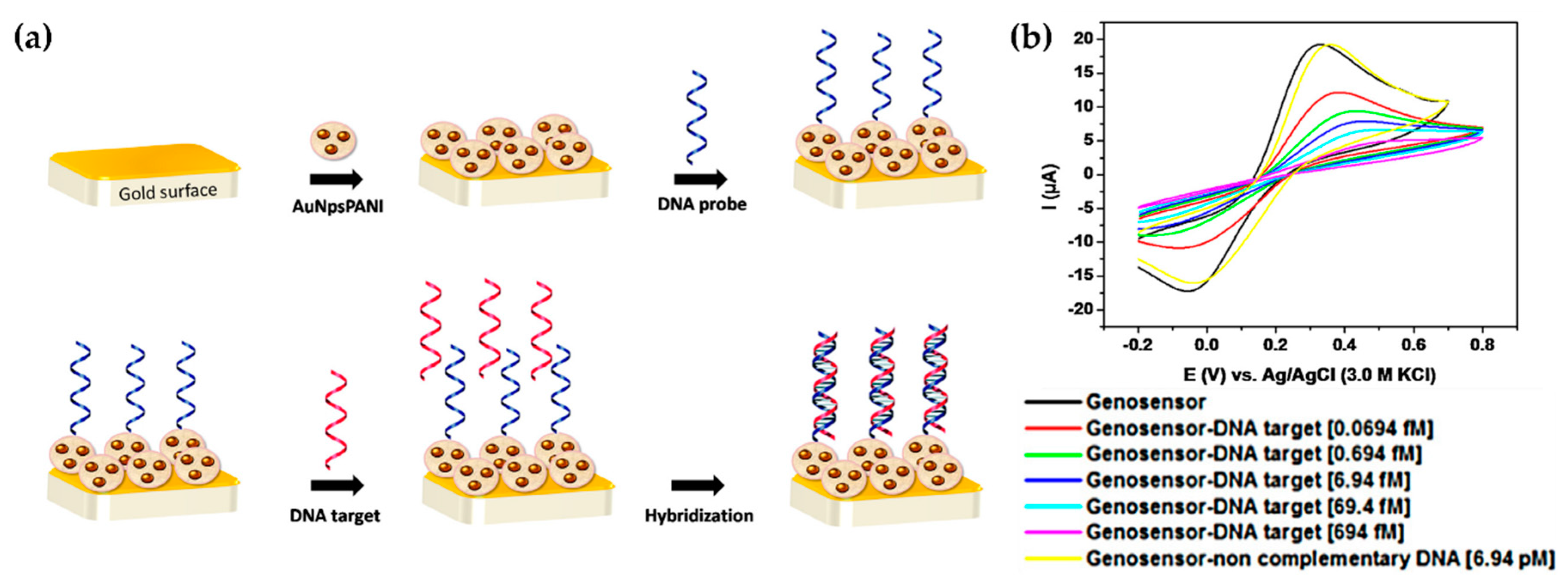
| Avelino et al. [38] (2016) | PAni | BCR/ABL (breakpoint cluster region- Abelson tyrosine kinase gene), oncogene (CV, EIS) | PAni-AuNPs/ssDNA (Structural and superficial, oxidative polymerization and biologic immobilization) | 69.4 aM (10−5–10−12 mM) | Sensitivity and selectivity |
| Bayram and Akyilmaz [39] (2016) | PAni | Paracetamol, drug (CA) | PAni-cMWCNTs/ Bacillus sp./GA (Structural and superficial, electropolymerization and dip-coated) | 2.9 μM (5–630 μM) | Sensitivity and selectivity |
| Molina et al. [40] (2018) | PPy | Serotonin, neurotransmitters (DPV) | PPy-g-PEG (Structural, “grafting through” technique) | 0.07 µM (0.5–20 µM) | Biocompatibility |
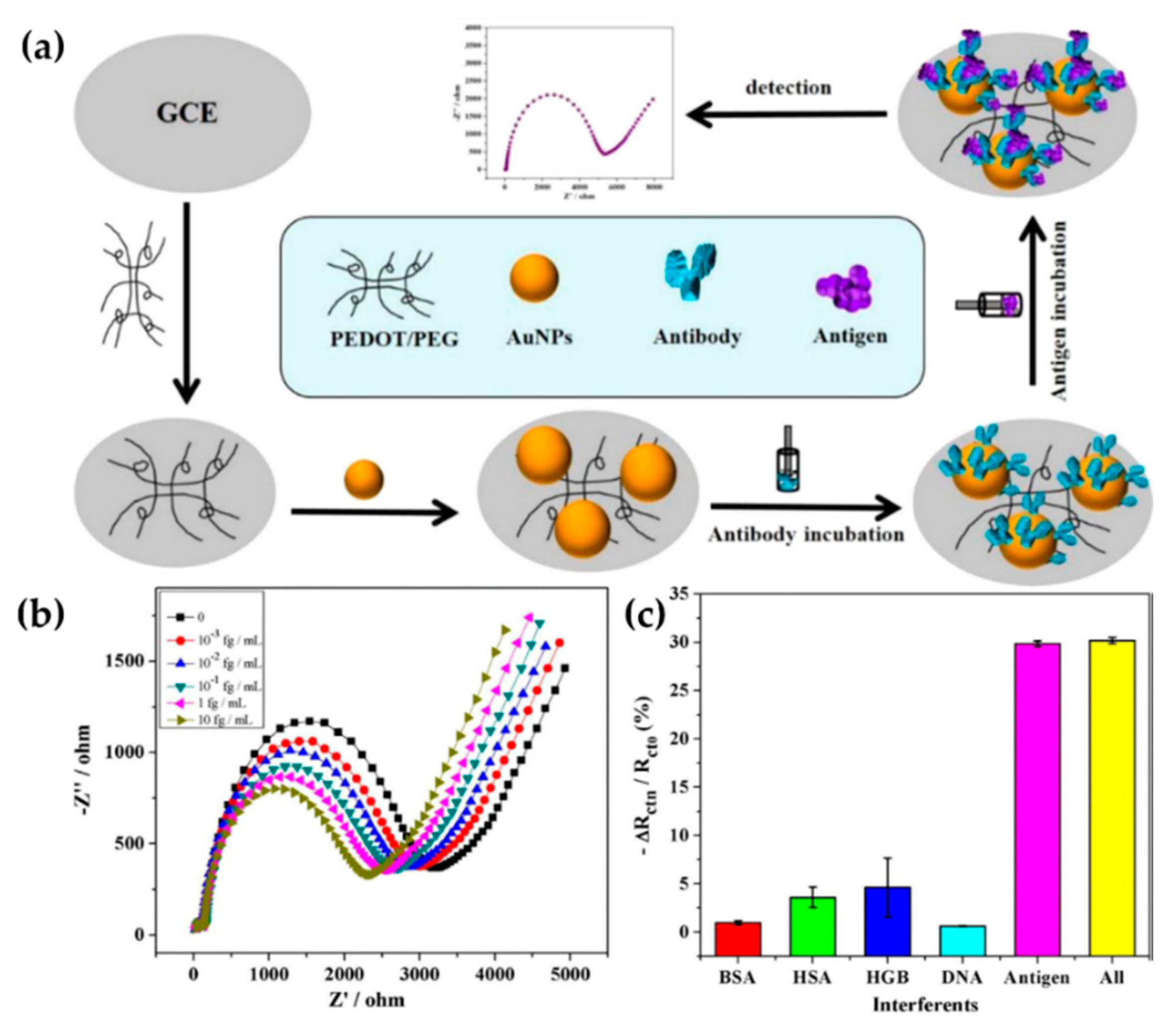
| Cui et al. [41] (2016) | PEDOT | Alpha fetoprotein, tumor marker (EIS) | PEDOT-PEG/AuNPs (Structural and superficial, electropolymerization and biologic immobilization) | 0.0003 fg/mL (0.001–10 fg/mL) | Hydrophilicity and selectivity |
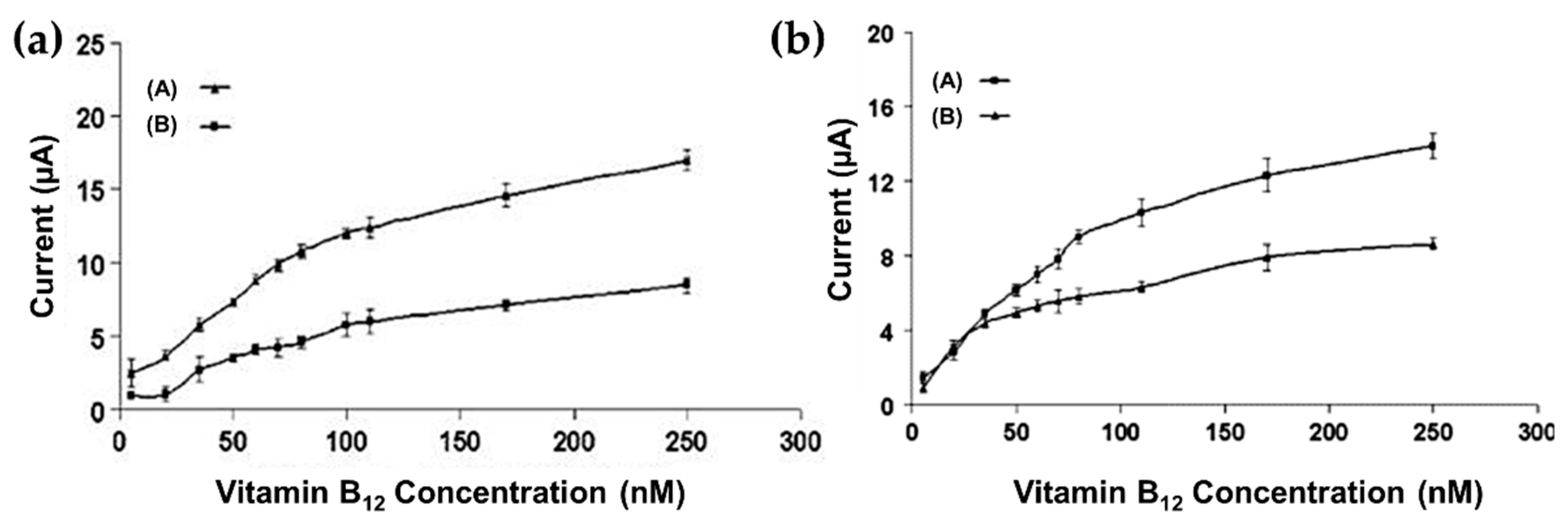
| Devnani et al. [42] (2016) | CS | Noradrenaline (CV, SWV, EIS) | Graphene-chitosan (Structural, drop casting) | 19.7 nM (200–1400 nM.) | Sensitivity and biocompatibility |
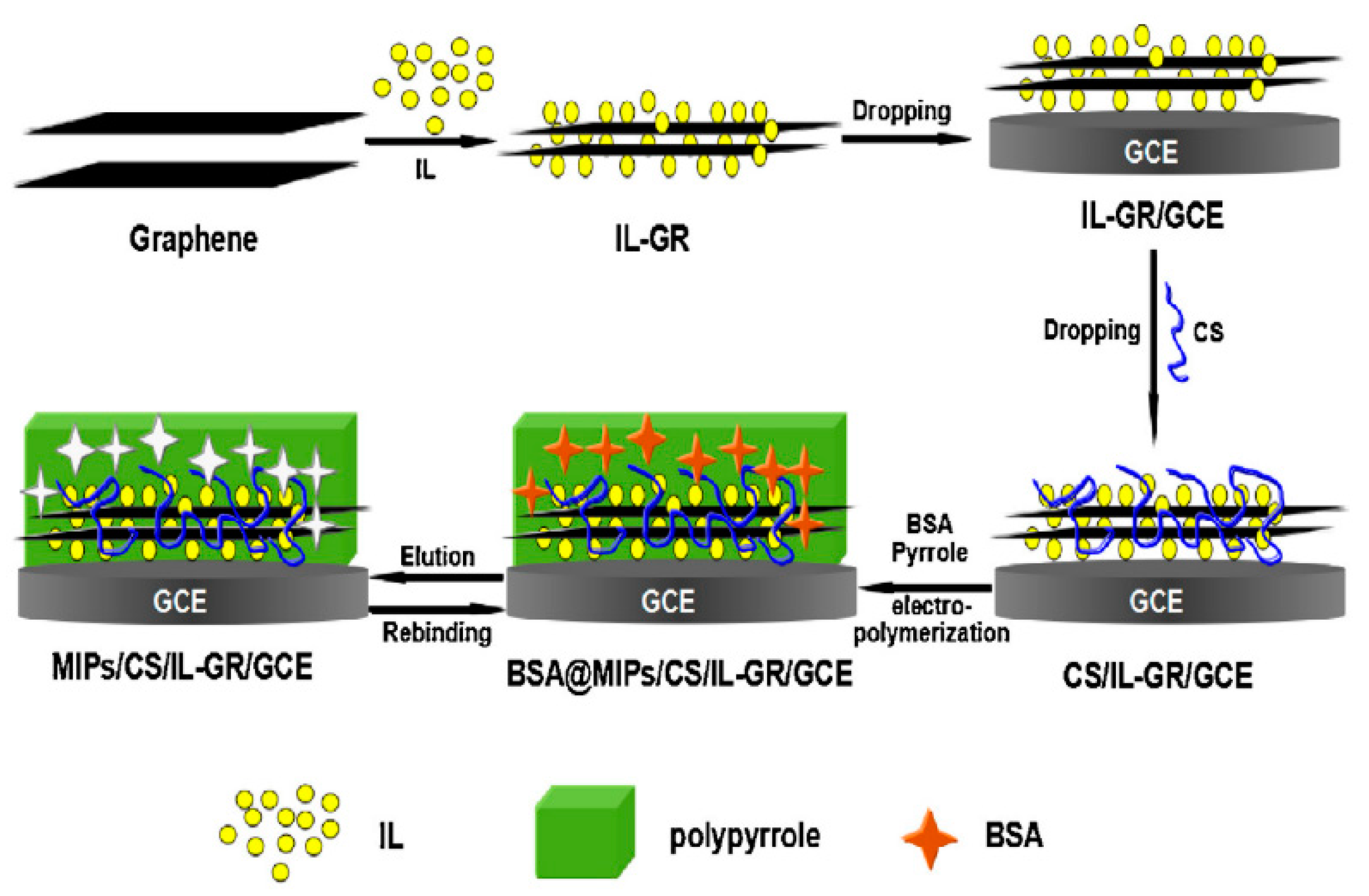
| Xia et al.[43] (2016) | CS | Bovine serum albumin (CV) | Chitosan/ionic liquid–graphene (Structural, molecular imprinting) | 2 × 10−11 g/L (1.0 × 10−10–0 × 10−4 g/L) | Selectivity, sensitivity and biocompatibility |
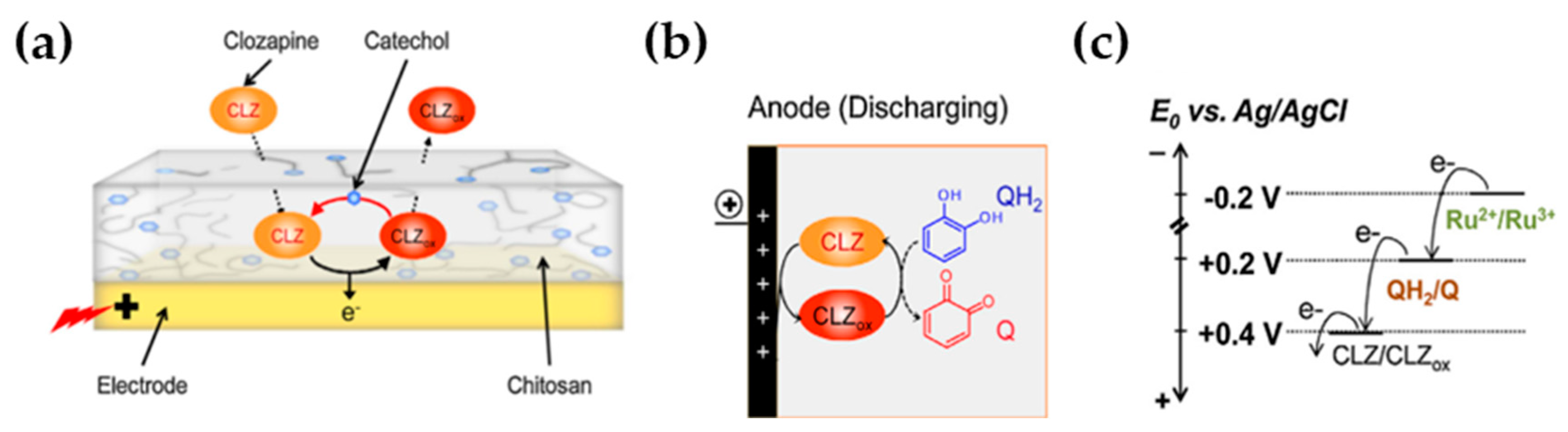
| Ben-Yoav et al. [44] (2014) | CS | Clozapine (CV, SWV) | Catechol-modified chitosan (Structural, microfabrication technology) | 0.1 μg/mL (0.1–10 μg/mL) | Sensitivity |
| Nordin et al. [45] (2016) | PLA | DNA (CV) | PLA-AuNPs (Superficial, Drop casting) | N.R. | Biocompatibility, mechanical properties, |
| Manzanares et al. [46] (2019) | PLA | Picric acid and Ascorbic acid (SWV) | Gr/PLA/proteinase K (Structural, Enzymatically sculptured 3D-printed electrode) | N.R. | Sensitivity |
| Das et al. [47] (2018) | PVA | Vitamin C, ascorbic acid (SWV) | Gr-iron oxide-polyvinyl alcohol (Structural) | 0.234 μM (40–4100 μM) | Sensitivity and stability |
| Fabregat et al. [48] (2017) | PEDOT-, PNCPy, LDPE, PP, PCL, PS, LDPE | Dopamine (CV, CA) | PEDOT-, PNCPy, LDPE, PP, PCL, PS plasma treated (Superficial, cold plasma surface functionalization) | 140 for PEDOT 750 nM for PNCPy (0.5–5 μM) | Sensitivity, selectivity, electric conductivity |
| Buendía et al. [49] (2017) | LDPE | Glucose (CA, CV) | LDPE-GOx (Superficial) | 1.7 mM for PT-LDPE/GOx plasma treated 2 min (0–20 mM) | Sensitivity, selectivity, electric conductivity |
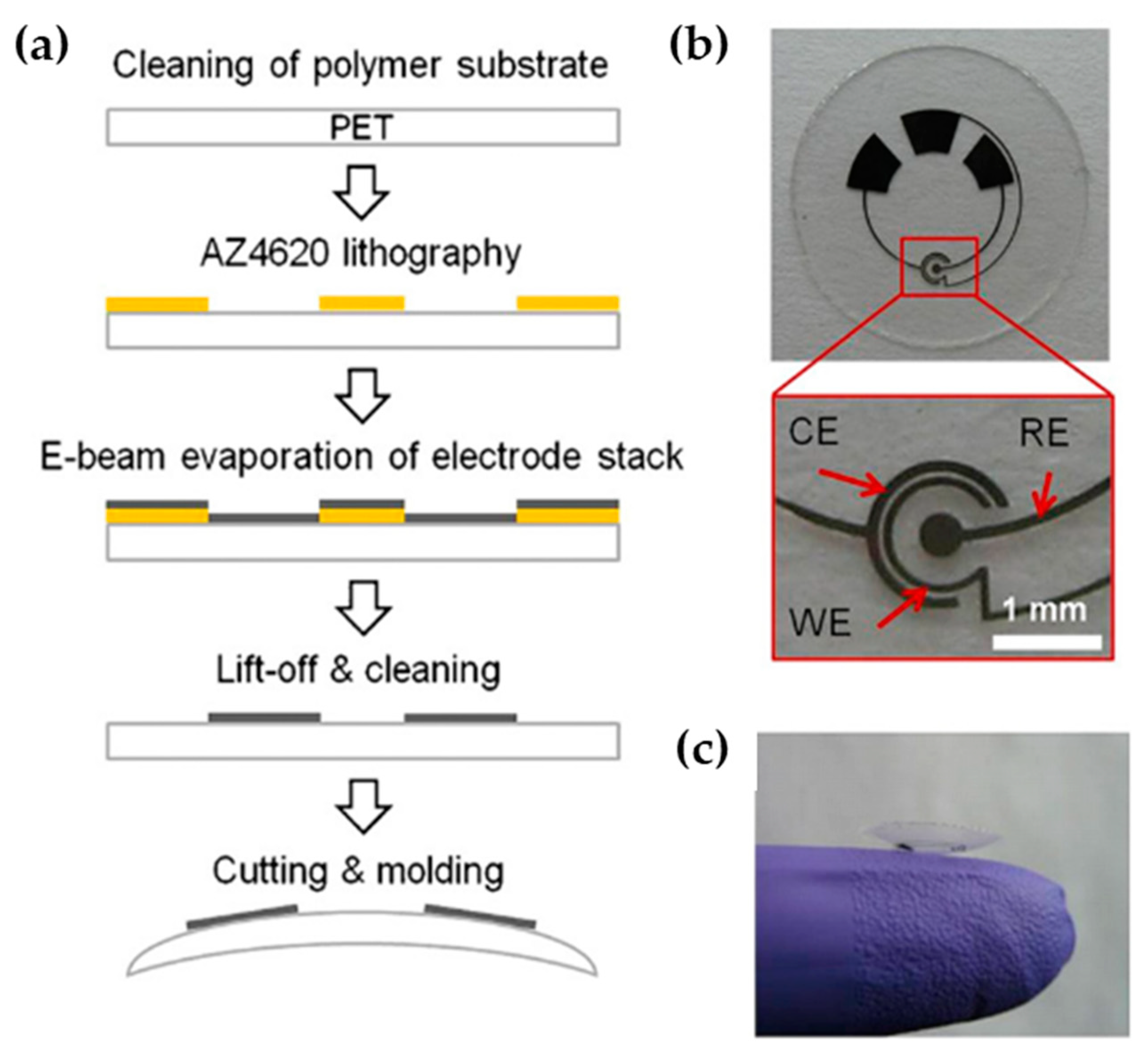
| Oliveira et al. [50] (2020) | Bio-PET sheets | Dopamine & Anti-PARK7/DJ-1 protein (SWV) | Pt electrode on Bio-PET (Superficial, microfabrication) | 5.1 × 10−3 mM (3.5 ×10−2–8.0 × 10−1 mM) | Sensitivity and selectivity |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lanzalaco, S.; Molina, B.G. Polymers and Plastics Modified Electrodes for Biosensors: A Review. Molecules 2020, 25, 2446. https://doi.org/10.3390/molecules25102446
Lanzalaco S, Molina BG. Polymers and Plastics Modified Electrodes for Biosensors: A Review. Molecules. 2020; 25(10):2446. https://doi.org/10.3390/molecules25102446
Chicago/Turabian StyleLanzalaco, Sonia, and Brenda G. Molina. 2020. "Polymers and Plastics Modified Electrodes for Biosensors: A Review" Molecules 25, no. 10: 2446. https://doi.org/10.3390/molecules25102446
APA StyleLanzalaco, S., & Molina, B. G. (2020). Polymers and Plastics Modified Electrodes for Biosensors: A Review. Molecules, 25(10), 2446. https://doi.org/10.3390/molecules25102446







