Preparation of Benzothiazolyl-Decorated Nanoliposomes
Abstract
1. Introduction
2. Results and Discussion
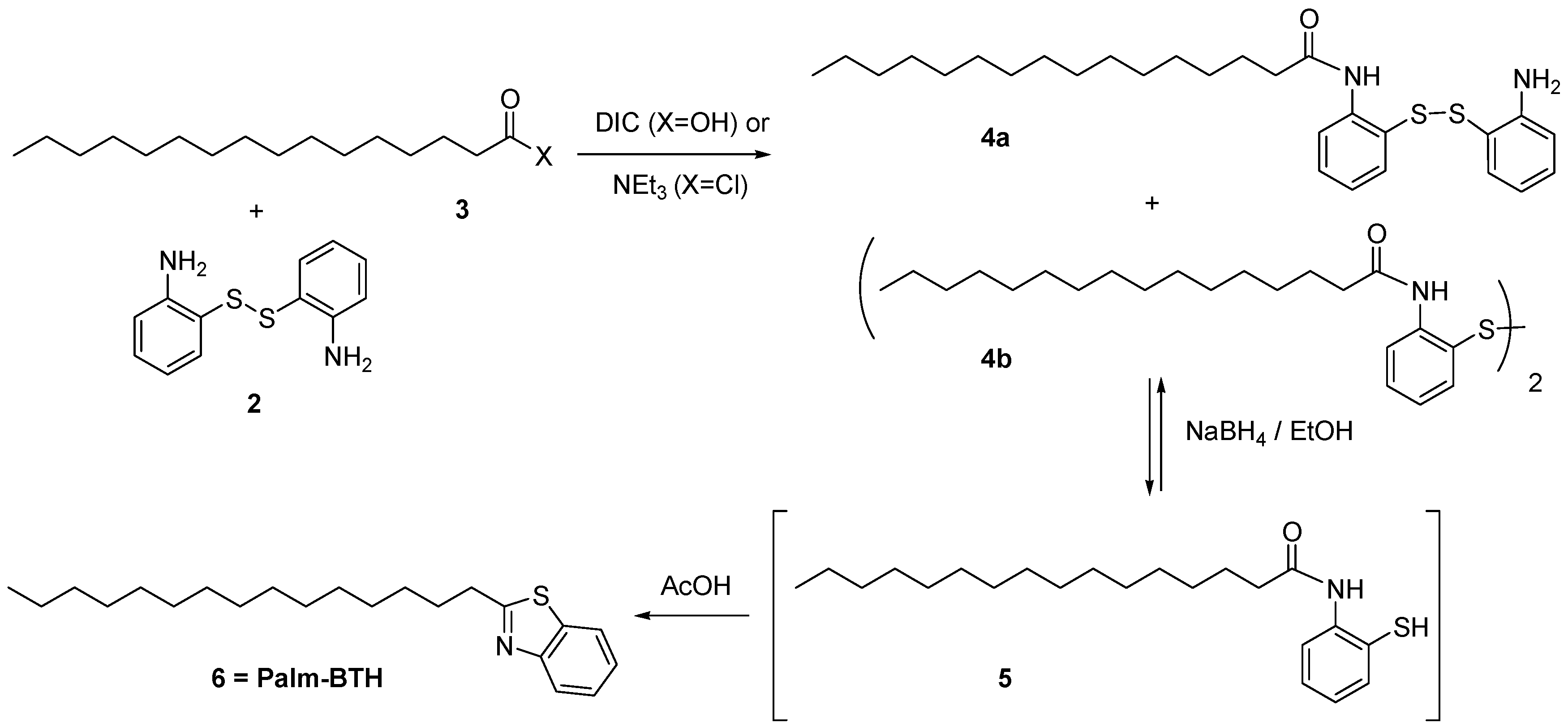
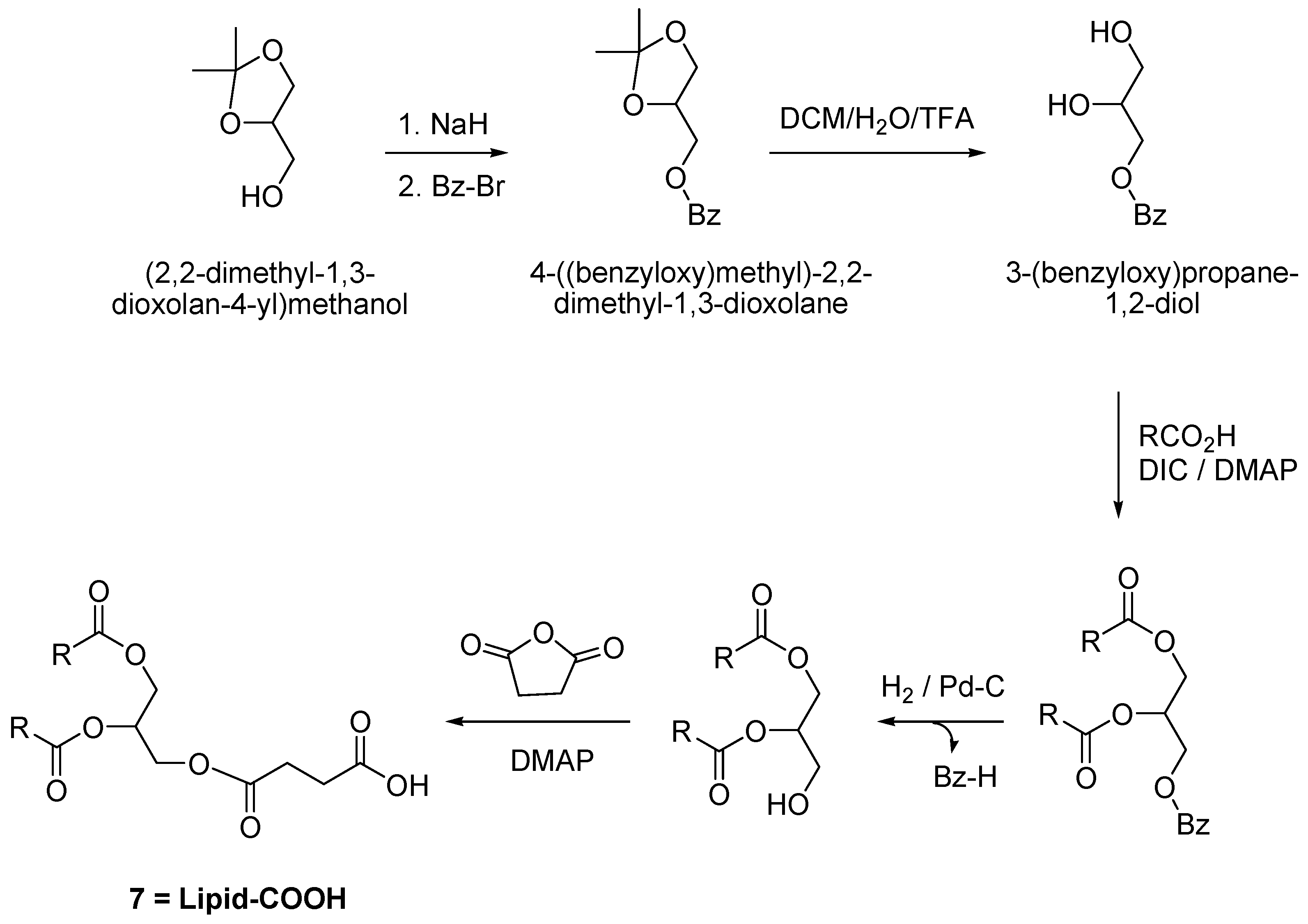
2.1. Synthesis of Compounds
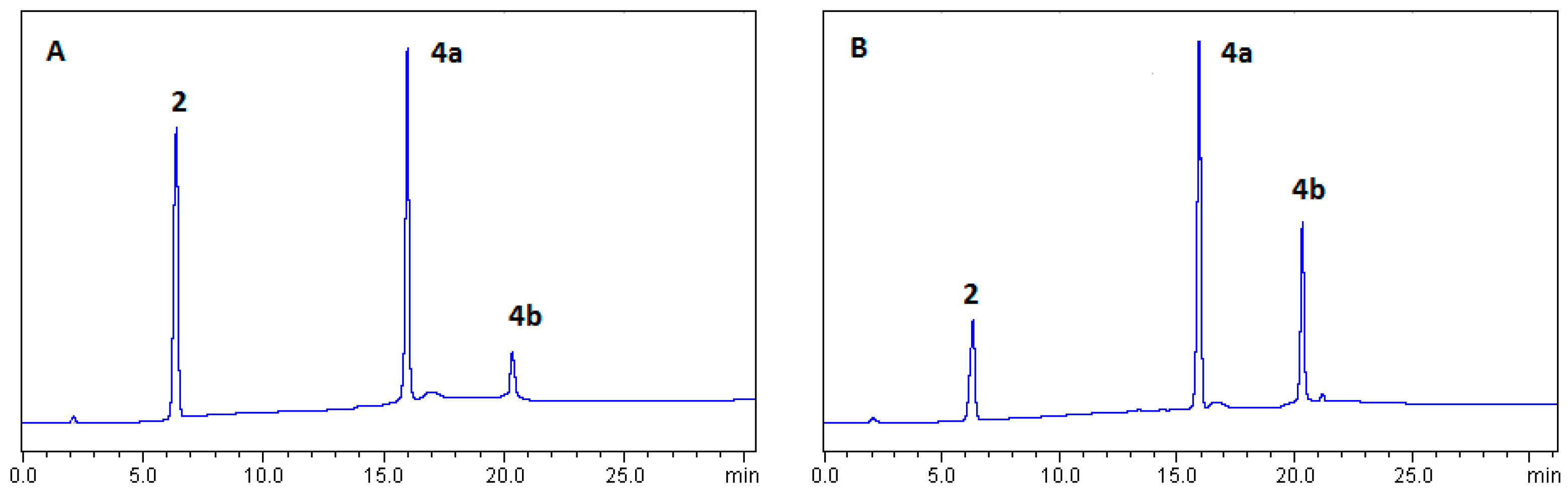
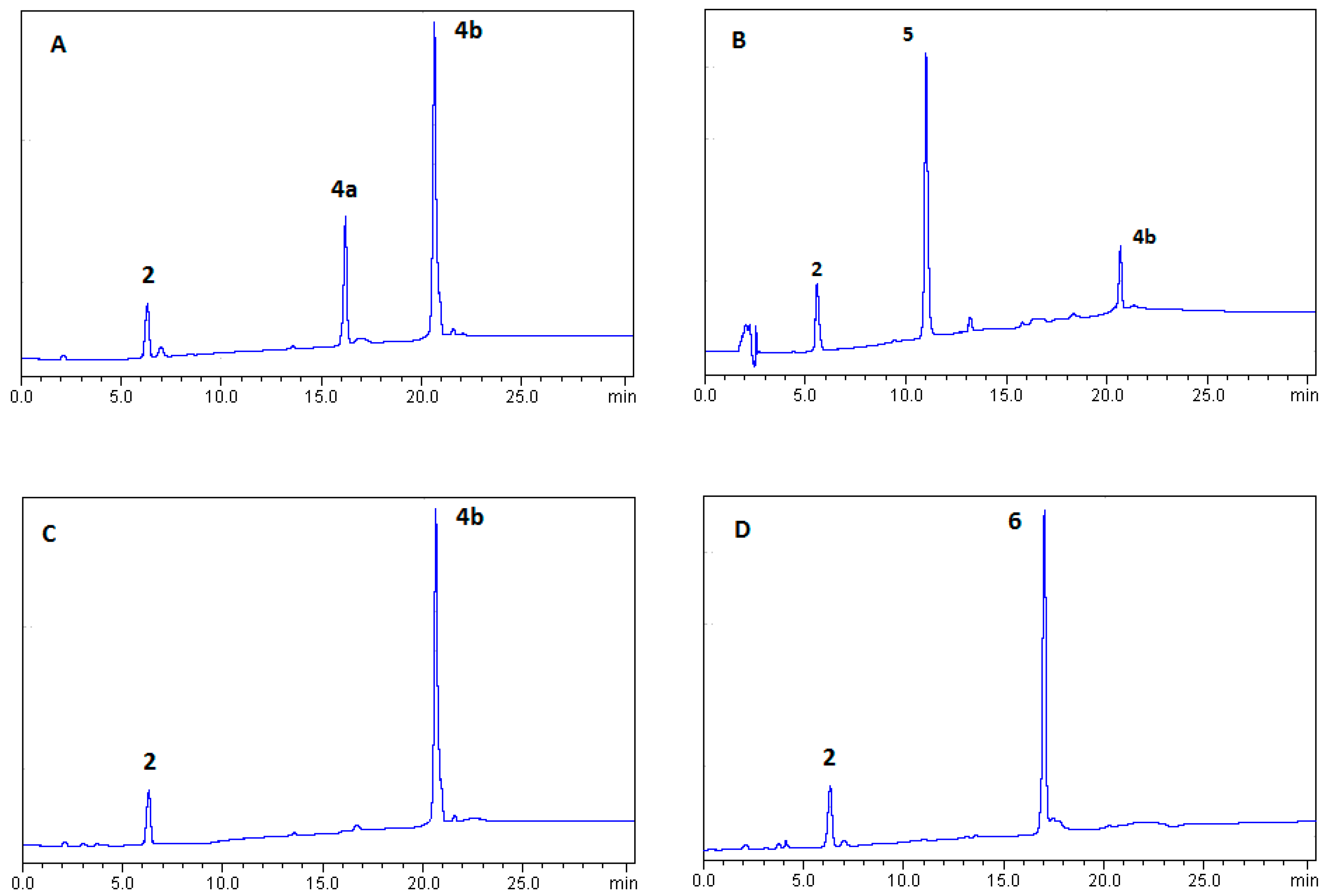
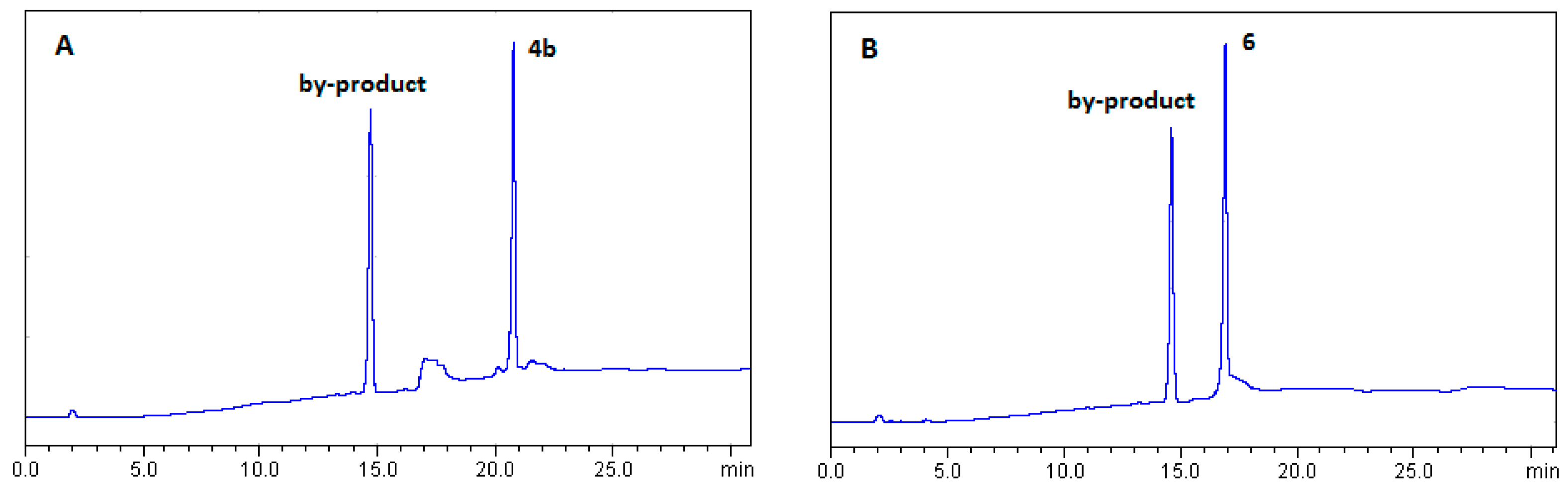
2.1.1. Optimization of BTH Formation
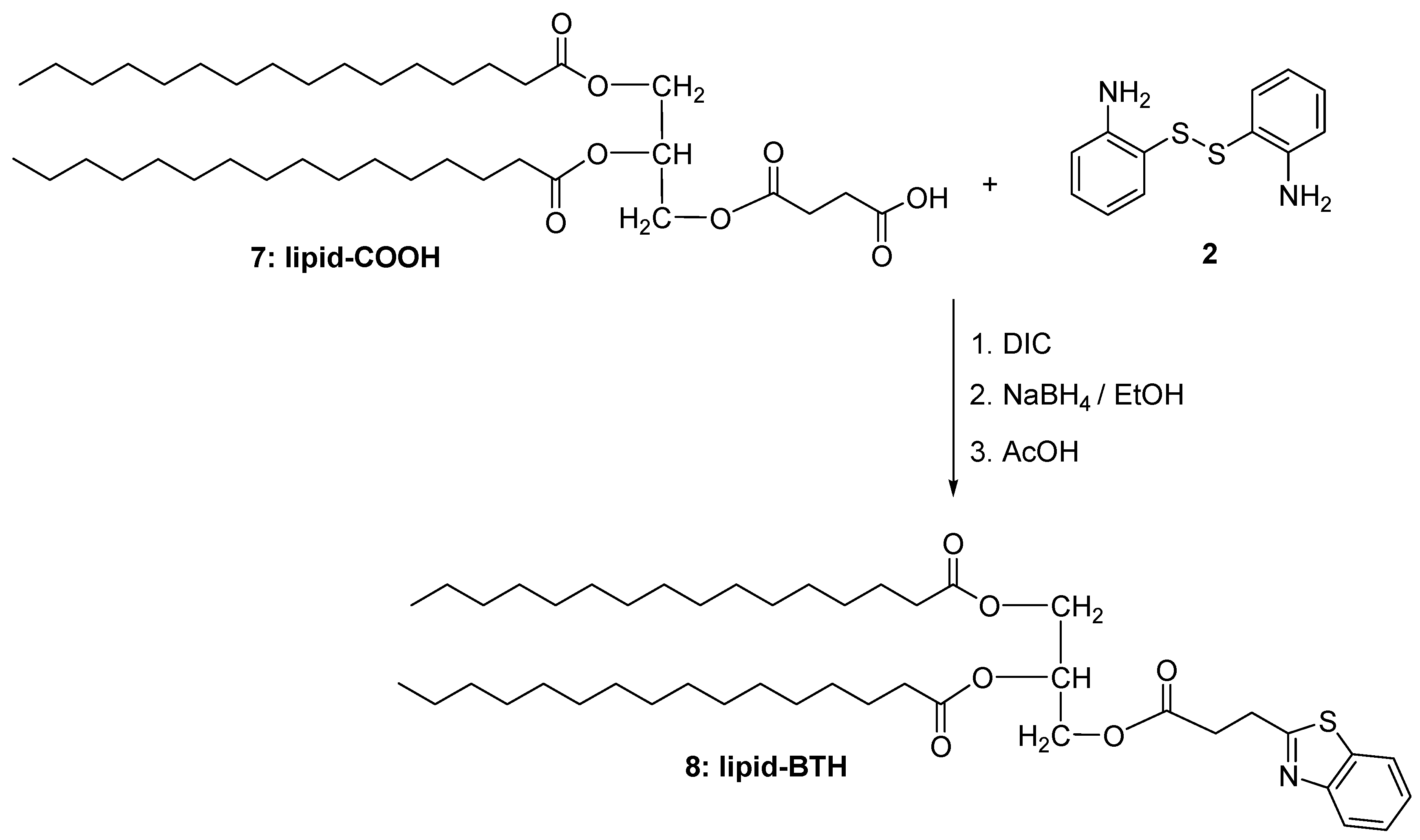
2.1.2. Synthesis of Lipid-BTH
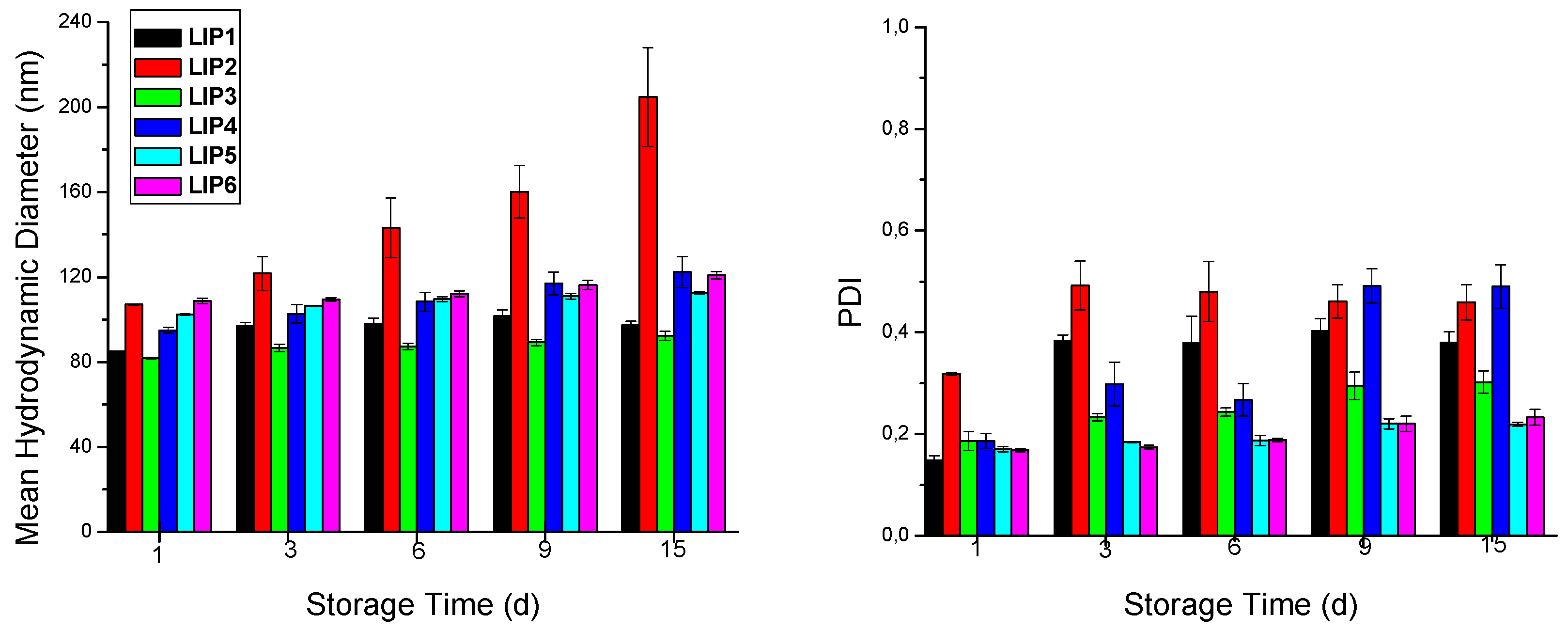
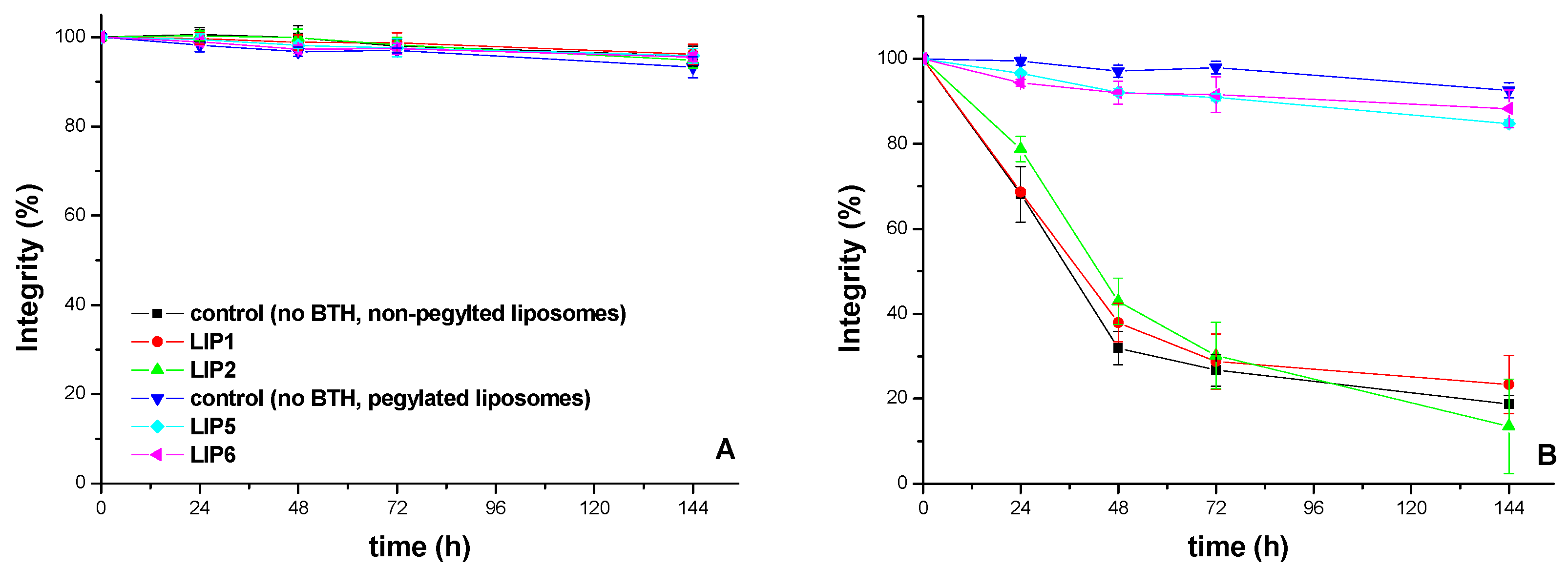
2.2. Nanoliposome Preparation
3. Materials and Methods
3.1. Materials
3.1.1. Synthesis of BTH-Derivatives (Palm-BTH and Lipid-BTH)
3.1.2. Nanoliposome Preparation
3.2. Synthetic Procedures
3.2.1. Synthesis of Bis(2-Aminophenyl) Disulfide
3.2.2. Synthesis of Lipid-COOH
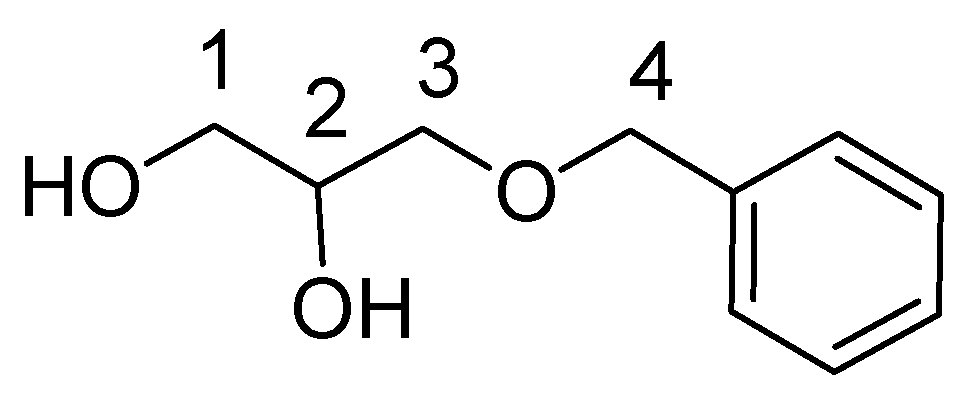

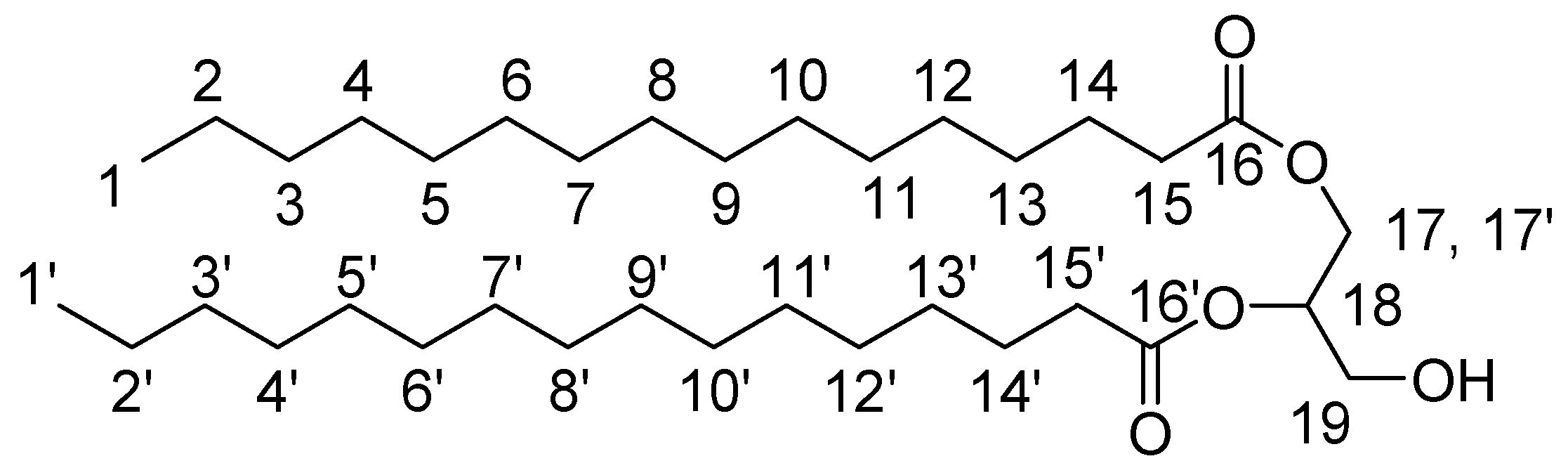
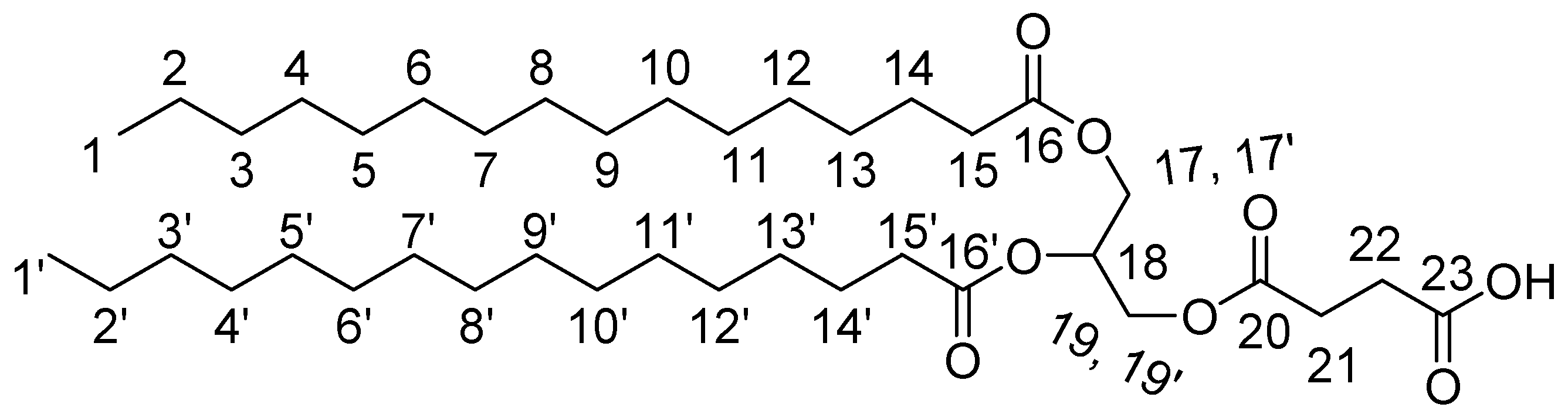
3.2.3. Lipid-BTH

3.3. Nanoliposome Preparation
3.4. Characterization of Nanoliposomes
3.4.1. Size Distribution, ζ-Potential, and Stability Studies
3.4.2. Nanoliposome Integrity Studies
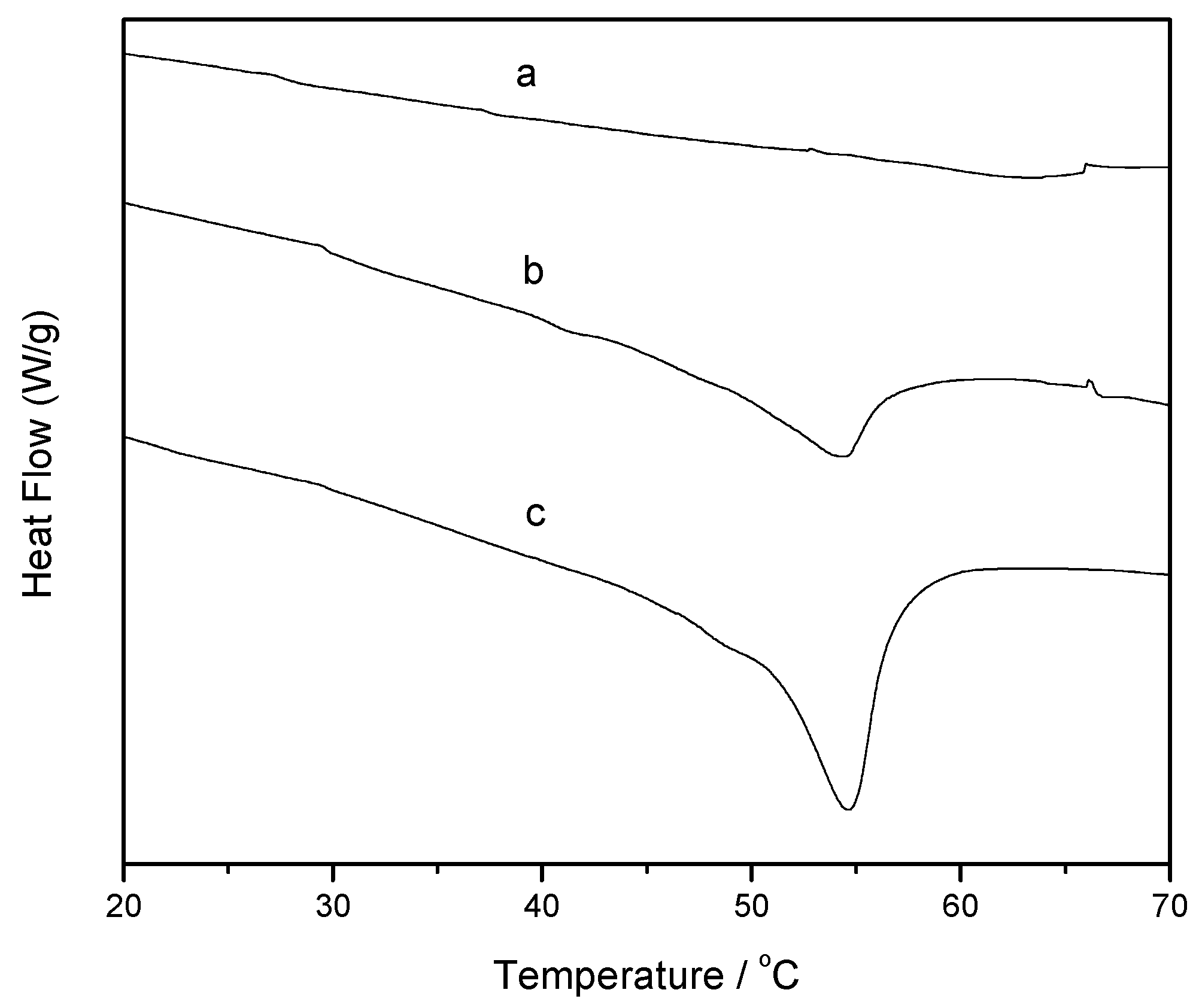
3.4.3. Nanoliposome Differential Scanning Calorimetry (DSC) Study
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Sanabria-Castro, A.; Alvarado-Echeverría, I.; Monge-Bonilla, C. Molecular Pathogenesis of Alzheimer’s Disease: An Update. Ann. Neurosci. 2017, 24, 46–54. [Google Scholar] [CrossRef] [PubMed]
- Kumar, K.; Kumar, A.; Keegan, R.M.; Deshmukh, R. Recent advances in the neurobiology and neuropharmacology of Alzheimer’s disease. Biomed. Pharmacother. 2018, 98, 297–307. [Google Scholar] [CrossRef] [PubMed]
- Citron, M. Alzheimer’s disease: Strategies for disease modification. Nat. Rev. Drug Discov. 2010, 9, 387–398. [Google Scholar] [CrossRef] [PubMed]
- Chopra, K.; Misra, S.; Kuhad, A. Current perspectives on pharmacotherapy of Alzheimer’s disease. Expert Opin. Pharmacother. 2011, 12, 335–350. [Google Scholar] [CrossRef] [PubMed]
- Folch, J.; Petrov, D.; Ettcheto, M.; Abad, S.; Sánchez-López, E.; García, M.L.; Olloquequi, J.; Beas-Zarate, C.; Auladell, C.; Camins, A. Current Research Therapeutic Strategies for Alzheimer’s Disease Treatment. Neural. Plast. 2016, 2016, 8501693. [Google Scholar] [CrossRef]
- Reiss, A.B.; Arain, H.A.; Stecker, M.M.; Siegart, N.M.; Kasselman, L.J. Amyloid toxicity in Alzheimer’s disease. Rev. Neurosci. 2018, 29, 613–627. [Google Scholar] [CrossRef]
- Takahashi, R.H.; Nagao, T.; Gouras, G.K. Plaque formation and the intraneuronal accumulation of β-amyloid in Alzheimer’s disease. Pathol. Int. 2017, 67, 185–193. [Google Scholar] [CrossRef]
- Pahnke, J.; Walker, L.C.; Scheffler, K.; Krohn, M. Alzheimer’s disease and blood-brain barrier function - Why have anti-beta-amyloid therapies failed to prevent dementia progression? Neurosci. Biobehav. Rev. 2009, 33, 1099–1108. [Google Scholar] [CrossRef]
- Dasilva, K.A.; Shaw, J.E.; McLaurin, J. Amyloid-beta fibrillogenesis: Structural insight and therapeutic intervention. Exp. Neurol. 2010, 223, 311–321. [Google Scholar] [CrossRef]
- Findeis, M.A. The role of amyloid beta peptide 42 in Alzheimer’s disease. Pharmacol. Ther. 2007, 116, 266–286. [Google Scholar] [CrossRef]
- De Felice, F.G.; Ferreira, S.T. Beta-amyloid production, aggregation, and clearance as targets for therapy in Alzheimer’s disease. Cell. Mol. Neurobiol. 2002, 22, 545–563. [Google Scholar] [CrossRef]
- Greenberg, S.M.; Grabowski, T.; Gurol, M.E.; Skehan, M.E.; Nandigam, R.N.; Becker, J.A.; Garcia-Alloza, M.; Prada, C.; Frosch, M.P.; Rosand, J.; Viswanathan, A.; Smith, E.E.; Johnson, K.A. Detection of isolated cerebrovascular beta-amyloid with Pittsburgh compound B. Ann. Neurol. 2008, 64, 587–591. [Google Scholar] [CrossRef]
- Look, G.C.; Jerecic, J.; Cherbavaz, D.B.; Pray, T.R.; Breach, J.C.; Crosier, W.J.; Igoudin, L.; Hironaka, C.M.; Lowe, R.M.; McEntee, M.; Ruslim-Litrus, L.; Wu, H.M.; Zhang, S.; Catalano, S.M.; Goure, W.F.; Summa, D.; Krafft, G.A. Discovery of ADDL--targeting small molecule drugs for Alzheimer’s disease. Curr. Alzheimer Res. 2007, 4, 562–567. [Google Scholar] [CrossRef]
- Montet, X.; Funovics, M.; Montet-Abou, K.; Weissleder, R.; Josephson, L. Multivalent effects of RGD peptides obtained by nanoparticle display. J. Med. Chem. 2006, 49, 6087–6093. [Google Scholar] [CrossRef]
- Hong, S.; Leroueil, P.R.; Majoros, I.J.; Orr, B.G.; Baker, J.R., Jr.; Banaszak Holl, M.M. The binding avidity of a nanoparticle-based multivalent targeted drug delivery platform. Chem. Biol. 2007, 14, 107–115. [Google Scholar] [CrossRef]
- Tassa, C.; Duffner, J.L.; Lewis, T.A.; Weissleder, R.; Schreiber, S.L.; Koehler, A.N.; Shaw, S.Y. Binding affinity and kinetic analysis of targeted small molecule-modified nanoparticles. Bioconjug. Chem. 2010, 21, 14–19. [Google Scholar] [CrossRef]
- Antimisiaris, S.G.; Kallinteri, P.; Fatouros, D. Liposomes and drug delivery. In Pharmaceutical Manufacturing Handbook Production and Processes; Gad, S.C., Ed.; John Wiley & Sons: Hoboken, NJ, USA, 2008; pp. 443–533. [Google Scholar]
- Gobbi, M.; Re, F.; Canovi, M.; Beeg, M.; Gregori, M.; Sesana, S.; Sonnino, S.; Brogioli, D.; Musicanti, C.; Gasco, P.; Salmona, M.; Masserini, M.E. Lipid based nanoparticles with high binding affinity for amyloid-β1-42 peptide. Biomaterials 2010, 31, 6519–6529. [Google Scholar] [CrossRef]
- Mourtas, S.; Canovi, M.; Zona, C.; Aurilia, D.; Niarakis, A.; La Ferla, B.; Salmona, M.; Nicotra, F.; Gobbi, M.; Antimisiaris, S.G. Curcumin-decorated nanoliposomes with very high affinity for amyloid-b1-42 peptide. Biomaterials 2011, 32, 1635–1645. [Google Scholar] [CrossRef]
- Canovi, M.; Markoutsa, E.; Lazar, A.N.; Pampalakis, G.; Clemente, C.; Re, F.; Sesana, S.; Masserini, M.; Salmona, M.; Duyckaerts, C.; et al. The binding affinity of anti-Aβ1-42 MAb-decorated nanoliposomes to Aβ1-42 peptides in vitro and to amyloid deposits in post-mortem tissue. Biomaterials 2011, 32, 5489–5497. [Google Scholar] [CrossRef]
- Airoldi, C.; Mourtas, S.; Cardona, F.; Zona, C.; Sironi, E.; D’Orazio, G.; Markoutsa, E.; Nicotra, F.; Antimisiaris, S.G.; La Ferla, B. Nanoliposomes presenting on surface a cis-glycofusedbenzopyran compound display binding affinity and aggregation inhibition ability towards Amyloid b1-42 peptide. Eur. J. Med. Chem. 2014, 85, 43–50. [Google Scholar] [CrossRef]
- Lazar, A.N.; Mourtas, S.; Youssef, I.; Parizot, C.; Dauphin, A.; Delatour, B.; Antimisiaris, S.G.; Duyckaerts, C. Curcumin-conjugated nanoliposomes with high affinity for Aβ deposits: Possibleapplications to Alzheimer disease. Nanomedicine 2013, 9, 712–721. [Google Scholar] [CrossRef] [PubMed]
- Mourtas, S.; Lazar, A.N.; Markoutsa, E.; Duyckaerts, C.; Antimisiaris, S.G. Multifunctional nanoliposomes with curcuminelipid derivative and brain targeting functionality with potential applications for Alzheimer disease. Eur. J. Med. Chem. 2014, 80, 175–183. [Google Scholar] [CrossRef] [PubMed]
- Wolfe, L.S.; Calabrese, M.F.; Nath, A.; Blaho, D.V.; Miranker, A.D.; Xiong, Y. Protein-induced photophysical changes to the amyloid indicator dye thioflavin T. Proc. Natl. Acad. Sci. USA 2010, 107, 16863–16868. [Google Scholar] [CrossRef] [PubMed]
- Biancalana, M.; Makabe, K.; Koide, A.; Koide, S. Molecular Mechanism of Thioflavin-T Binding to the Surface of β-Rich Peptide Self-Assemblies. J. Mol. Biol. 2009, 385, 1052–1063. [Google Scholar] [CrossRef]
- Stefansson, S.; Adams, D.L.; Tang, C.M. Common benzothiazole and benzoxazole fluorescent DNA intercalators for studying Alzheimer Aβ1-42 and prion amyloid peptides. Biotechniques 2012, 52. [Google Scholar]
- Arul Murugan, N.; Zaleśny, R.; Ågren, H. Unusual binding-site-specific photophysical properties of a benzothiazole-based optical probe in amyloid beta fibrils. Phys. Chem. Chem. Phys. 2018, 20, 20334–20339. [Google Scholar] [CrossRef] [PubMed]
- Ono, M.; Hayashi, S.; Kimura, H.; Kawashima, H.; Nakayama, M.; Saji, H. Push-pull benzothiazole derivatives as probes for detecting beta-amyloid plaques in Alzheimer’s brains. Bioorg. Med. Chem. 2009, 17, 7002–7007. [Google Scholar] [CrossRef]
- Okamato, M.; Gray, J.D.; Larson, C.S.; Kazom, S.F.; Soya, H.; McEwen, B.S.; Pereira, A.C. Riluzole reduces amyloid beta pathology, improves memory, and restores gene expression changes in a transgenic mouse model of early-onset Alzheimer’s disease. Transl. Psychiatry 2018, 8, 1–13. [Google Scholar] [CrossRef]
- Ren, S.C.; Shao, H.; Ji, W.G.; Jiang, H.H.; Xu, F.; Chen, P.Z.; Mi, Z.; Wen, B.; Zhu, G.X.; Zhu, Z.R. Riluzole prevents soluble Aβ1-42 oligomers-induced perturbation of spontaneous discharge in the hippocampal CA1 region of rats. Amyloid 2015, 22, 36–44. [Google Scholar] [CrossRef]
- Eckroat, T.J.; Mayhoub, A.S.; Garneau-Tsodikova, S. Amyloid-β probes: Review of structure–activity and brain-kinetics relationships. Beilstein J. Org. Chem. 2013, 9, 1012–1044. [Google Scholar] [CrossRef]
- Habib, L.K.; Lee, M.T.C.; Yang, J. Inhibitors of catalase-amyloid interactions protect cells from beta-amyloid-induced oxidative stress and toxicity. J. Biol. Chem. 2010, 285, 38933–38943. [Google Scholar] [CrossRef]
- Song, J.M.; DiBattista, A.M.; Sung, Y.M.; Ahn, J.M.; Turner, R.S.; Yang, J.; Pak, D.T.S.; Lee, H.K.; Hoe, H.S. A tetra(ethylene glycol) derivative of benzothiazole aniline ameliorates dendritic spine density and cognitive function in a mouse model of Alzheimer’s disease. Exp. Neurol. 2014, 252, 105–113. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Cifelli, J.L.; Chung, T.S.; Liu, H.; Prangkio, P.; Mayer, M.; Yang, J. Benzothiazole Amphiphiles Ameliorate Amyloid-Related Cell Toxicity and Oxidative Stress. ACS Chem. Neurosci. 2016, 7, 682–688. [Google Scholar] [CrossRef]
- Lin, K.S.; Debnath, M.L.; Mathis, C.A.; Klunk, W.E. Synthesis and β-amyloid binding properties of rhenium 2-phenylbenzothiazoles. Bioorg. Med. Chem. Lett. 2009, 19, 2258–2262. [Google Scholar] [CrossRef]
- Jiang, L.; Zhang, M.; Tang, L.; Weng, Q.; Shen, Y.; Hua, Y.; Sheng, R. Identification of 2-subsituted benzothiazole derivatives as triple-functional agents with potential for AD therapy. RSC Adv. 2016, 6, 17318–17327. [Google Scholar] [CrossRef]
- Wu, C.; Pike, V.W.; Wang, Y. Amyloid imaging: From benchtop to bedside. Curr. Top. Dev. Biol. 2005, 70, 171–213. [Google Scholar] [PubMed]
- Khurana, R.; Coleman, C.; Ionescu-Zanetti, C.; Carter, S.A.; Krishna, V.; Grover, R.K.; Roy, R.; Singh, S. Mechanism of thioflavin T binding to amyloid fibrils. J. Struct. Biol. 2005, 151, 229–238. [Google Scholar] [CrossRef] [PubMed]
- Klunk, W.E.; Wang, Y.; Huang, G.F.; Debnath, M.L.; Holt, D.P.; Mathis, C.A. Uncharged thioflavin-T derivatives bind to amyloid-beta protein with high affinity and readily enter the brain. Life Sci. 2001, 69, 1471–1484. [Google Scholar] [CrossRef]
- Kung, M.P.; Hou, C.; Zhuang, Z.P.; Skovronsky, D.M.; Zhang, B.; Gur, T.L.; Trojanowski, J.Q.; Lee, V.M.; Kung, H.F. Radioiodinatedstyrylbenzene derivatives as potential SPECT imaging agents for amyloid plaque detection in Alzheimer’s disease. J. Mol. Neurosci. 2002, 19, 7–10. [Google Scholar] [CrossRef]
- Mourtas, S.; Gatos, D.; Barlos, K. Solid Phase Synthesis of Benzothiazolyl Compounds. Tetrahedron Lett. 2001, 42, 2201–2204. [Google Scholar] [CrossRef]
- Dev, D.; Chandra, J.; Palakurthy, N.B.; Thalluri, K.; Kalita, T.; Mandal, B. Benzoxazole and Benzothiazole Synthesis from Carboxylic Acids in Solution and on Resin by Using Ethyl 2-Cyano-2-(2-nitrobenzenesulfonyloxyimino)acetate and para-Toluenesulfonic Acid. Asian J. Org. Chem. 2016, 5, 663–675. [Google Scholar] [CrossRef]
- Du, G.; Zhu, N.; Han, L.; Hong, H.; Suo, Q. Metal-Free Synthesis of Benzothiazoles from Disulfides of 2-Aminobenzenethiol and Carboxylic Acid via PCl3-Promoted Tandem Reaction. Heterocycles 2015, 91, 1723–1734. [Google Scholar] [CrossRef]
- Immordino, M.L.; Dosio, F.; Cattel, L. Stealth liposomes: Review of the basic science, rationale, and clinical applications, existing and potential. Int. J. Nanomed. 2006, 1, 297–315. [Google Scholar]
- Garbuzenko, O.; Barenholz, Y.; Priev, A. Effect of grafted PEG on liposome size and on compressibility and packing of lipid bilayer. Chem. Phys. Lipids 2005, 135, 117–129. [Google Scholar] [CrossRef] [PubMed]
- McMullen, T.P.; McElhaney, R.N. New aspects of the interaction of cholesterol with dipalmitoylphosphatidylcholine bilayers as revealed by high-sensitivity differential scanning calorimetry. Biochim. Biophys. Acta. 1995, 1234, 90–98. [Google Scholar] [CrossRef]
Sample Availability: Samples of the compounds 2, 6, 7 and 8 are available from the authors. |

| Nanoliposome Type | Molar Ratio | Mean Hydrodynamic Diameter (nm) | Polydispersity Index (PDI) | ζ-Potential (mV) |
|---|---|---|---|---|
| DPPC/Chol/Lipid-BTH (10%)—LIP1 | 1:1:0.1 | 84.98 ± 0.22 | 0.148 ± 0.009 | −4.85 ± 0.223 |
| DPPC/Chol/Lipid-BTH (20%)—LIP2 | 1:1:0.2 | 107.01 ± 0.38 | 0.318 ± 0.003 | −4.13 ± 0.138 |
| DPPC/DPPG/Chol/Lipid-BTH (10%)—LIP3 | 9:1:10:0.1 | 81.84 ± 0.28 | 0.186 ± 0.019 | −9.61 ± 0.156 |
| DPPC/DPPG/Chol/Lipid-BTH (20%)—LIP4 | 9:1:10:0.2 | 94.91 ± 1.30 | 0.186 ± 0.015 | −12.2 ± 0.141 |
| DPPC/Chol/DSPE-PEG2000-OMe(8%)/Lipid-BTH (10%)—LIP5 | 1:1:0.08:0.1 | 102.4 ± 0.25 | 0.170 ± 0.005 | −3.13 ± 0.235 |
| DPPC/Chol/DSPE-PEG2000-OMe(8%)/Lipid-BTH (20%)—LIP6 | 1:1:0.08:0.2 | 108.7 ± 1.22 | 0.168 ± 0.003 | −2.27 ± 0.165 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mourtas, S.; Christodoulou, P.; Klepetsanis, P.; Gatos, D.; Barlos, K.; Antimisiaris, S.G. Preparation of Benzothiazolyl-Decorated Nanoliposomes. Molecules 2019, 24, 1540. https://doi.org/10.3390/molecules24081540
Mourtas S, Christodoulou P, Klepetsanis P, Gatos D, Barlos K, Antimisiaris SG. Preparation of Benzothiazolyl-Decorated Nanoliposomes. Molecules. 2019; 24(8):1540. https://doi.org/10.3390/molecules24081540
Chicago/Turabian StyleMourtas, Spyridon, Panayiota Christodoulou, Pavlos Klepetsanis, Dimitrios Gatos, Kleomenis Barlos, and Sophia G. Antimisiaris. 2019. "Preparation of Benzothiazolyl-Decorated Nanoliposomes" Molecules 24, no. 8: 1540. https://doi.org/10.3390/molecules24081540
APA StyleMourtas, S., Christodoulou, P., Klepetsanis, P., Gatos, D., Barlos, K., & Antimisiaris, S. G. (2019). Preparation of Benzothiazolyl-Decorated Nanoliposomes. Molecules, 24(8), 1540. https://doi.org/10.3390/molecules24081540









