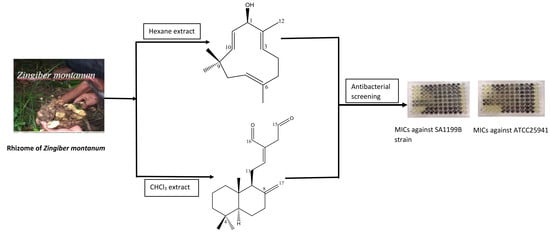
Terpenes from Zingiber montanum and Their Screening against Multi-Drug Resistant and Methicillin Resistant Staphylococcus aureus
Abstract
1. Introduction
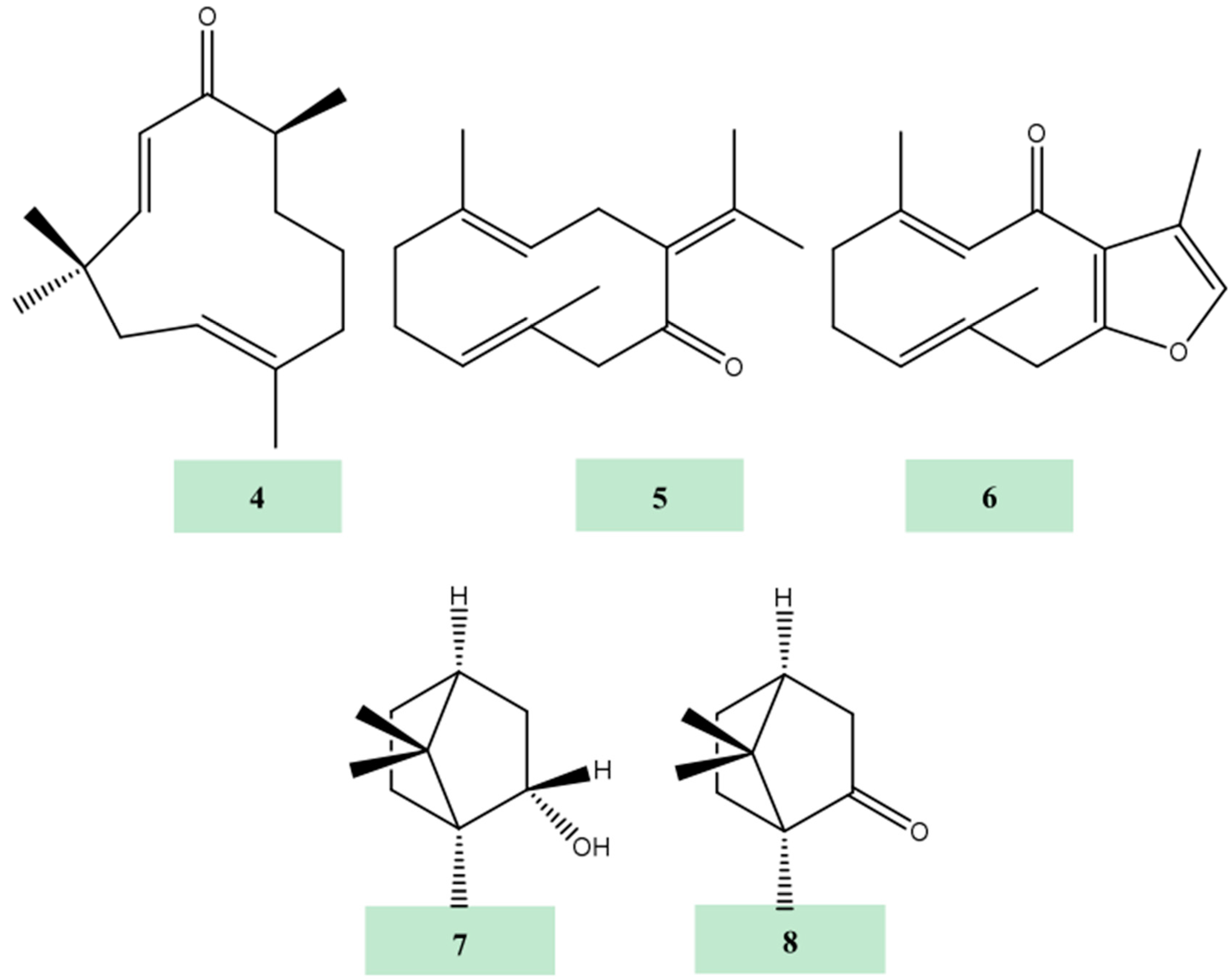
2. Results and Discussion
3. Material and Methods
3.1. General
3.2. Plant Material
3.3. Extraction and Isolation of Compounds
3.4. Antibacterial Assay against Clinical Isolates of Multi-Drug Resistant and Methicillin Resistant Staphylococcus Aureus
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- O’Neill, J. Tackling Drug-Resistant Infections Globally: Final Report and Recommendations the Review on Antimicrobial Resistance. 2016. Available online: https://amr-review.org/sites/default/files/160518_Final%20paper_with%20cover.pdf (accessed on 15 February 2018).
- World Health Organization (WHO). Antibiotic Resistance. 2018. Available online: https://www.who.int/news-room/fact-sheets/detail/antibiotic-resistance (accessed on 18 November 2018).
- Faridi, A.; Kareshk, A.T.; Fatahi-Bafghi, M.; Ziasistani, M.; Ghahraman, M.R.K.; Seyyed-Yousefi, S.Z.; Shakeri, N.; Kalantar-Neyestanaki, D. Detection of methicillin-resistant Staphylococcus aureus (MRSA) in clinical samples of patients with external ocular infection. Iran. J. Microbiol. 2018, 10, 215–219. [Google Scholar] [PubMed]
- Hollman, A. Digoxin comes from Digitalis lanata. Br. Med. J. 1996, 312, 912. [Google Scholar] [CrossRef]
- Alam, M.M.; Naeem, M.; Khan, M.M.A.; Uddin, M. Vincristine and Vinblastine Anticancer Catharanthus Alkaloids: Pharmacological Applications and Strategies for Yield Improvement. In Catharanthus Roseus; Springer: Cham, Switzerland, 2017; pp. 277–307. [Google Scholar]
- Patrick, G. The Opioid Analgesic. In An Introduction to Medicinal Chemistry, 5th ed.; Oxford University Press: Oxford, UK, 2013; p. 632. [Google Scholar]
- Butler, A.; Hensman, T. Drugs for the fever. Educ. Chem. 2000, 37, 151. [Google Scholar]
- Mahdi, J.G. Medicinal potential of willow: A chemical perspective of aspirin discovery. J. Saudi Chem. Soc. 2010, 14, 317–322. [Google Scholar] [CrossRef]
- Gautam, R.; Saklani, A.; Jachak, S.M. Indian medicinal plants as a source of antimycobacterial agents. J. Ethnopharmacol. 2007, 110, 200–234. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.; Yu, P.; Zhang, G.; Xu, L.; Wang, D.; Wang, L.; Zeng, X.; Wang, Y. Design, synthesis and antibacterial activity of novel andrographolide derivatives. Bioorg. Med. Chem. 2010, 18, 4269–4274. [Google Scholar] [CrossRef] [PubMed]
- Oluwatuyi, M.; Kaatz, G.W.; Gibbons, S. Antibacterial and resistance modifying activity of Rosmarinus officinalis. Phytochemistry 2004, 65, 3249–3254. [Google Scholar] [CrossRef] [PubMed]
- Schempp, C.M.; Pelz, K.; Wittmer, A.; Schöpf, E.; Simon, J.C. Antibacterial activity of hyperforin from St John’s wort against multi-resistant Staphylococcus aureus and Gram-positive bacteria. Lancet 1999, 353, 2129. [Google Scholar] [CrossRef]
- Shiu, W.K.P.; Rahman, M.M.; Curry, J.; Stapleton, P.D.; Zloh, M.; Malkinson, J.P.; Gibbons, S. Antibacterial acylphloroglucinols from Hypericum olympicum. J. Nat. Prod. 2012, 75, 336–343. [Google Scholar] [CrossRef] [PubMed]
- Khare, C.P. Indian Medicinal Plants: An Illustrated Dictionary; Springer: Berlin/Heidelberg, Germany, 2007; p. 733. [Google Scholar]
- Farnsworth, N.R.; Bunyapraphatsara, N. Thai Medicinal Plants: Recommended for Primary Health Care System; Medicinal Plants Information Center: Bangkok, Thailand, 1992. [Google Scholar]
- Singh, C.B.; Manglembi, N.; Swapana, N.; Chanu, S.B. Ethnobotany, phytochemistry and pharmacology of Zingiber cassumunar Roxb. (Zingiberaceae). J. Pharmacogn. Phytochem. 2015, 4, 1–6. [Google Scholar]
- Sharma, G.J.; Thokchom, D.S. Antioxidant and radioprotective properties of Zingiber montanum (J. König) A. Dietr. Planta Med. 2011, 77, 127. [Google Scholar] [CrossRef]
- Thokchom, D.S.; Sharma, T.D.; Sharma, G.J. Radioprotective effect of rhizome extract of Zingiber montanum in Rattus norvegicus. Radiat. Environ. Biophys. 2012, 51, 311–318. [Google Scholar] [CrossRef] [PubMed]
- Al-Amin, M.; Sultana, G.N.; Hossain, C.F. Antiulcer principle from Zingiber montanum. J. Ethnopharmacol. 2012, 141, 57–60. [Google Scholar] [CrossRef] [PubMed]
- Masuda, T.; Jitoe, A.; Mabry, M.J. Isolation and structure determination of cassumunarins A, B and C: New anti-inflammatory antioxidants from a tropical ginger, Zingiber cassumunar. J. Am. Oil Chem. Soc. 1995, 72, 1053–1057. [Google Scholar] [CrossRef]
- Verma, R.S.; Joshi, N.; Padalia, R.C.; Singh, V.R.; Goswami, P.; Verma, S.K.; Iqbal, H.; Chanda, D.; Verma, R.K.; Darokar, M.P.; et al. Chemical composition and antibacterial, antifungal, allelopathic and acetylcholinesterase inhibitory activities of cassumunar-ginger. J. Sci. Food Agric. 2018, 98, 321–327. [Google Scholar] [CrossRef] [PubMed]
- Sy, K.L.; Brown, D.G. Labdane diterpenoids from Alpinia chinensis. J. Nat. Prod. 1997, 60, 904–908. [Google Scholar] [CrossRef]
- Firman, K.; Kinoshita, T.; Itai, A.; Sankawa, U. Terpenoids from Curcuma Heyneana. Phytochemistry 1988, 27, 3887–3891. [Google Scholar] [CrossRef]
- Takashi, K.; Nagao, R.; Masuda, T.; Hill, R.K.; Morita, M.; Takatani, M.; Sawada, S.; Okamoto, T. The chemistry of Zerumbone IV: Asymmetric synthesis of Zerumbol. J. Mol. Catal. B Enzym. 2002, 17, 75–79. [Google Scholar]
- Nathaniel, C.; Elaine-Lee, Y.L.; Yee, B.C.; How, C.W.; Yim, H.S.; Rasadee, A.; Ng, H.S. Zerumbone-loaded nanostructured lipid carrier induces apoptosis in human colorectal adenocarcinoma (Caco-2) cell line. Nanosci. Nanotechnol. Lett. 2016, 8, 294–302. [Google Scholar] [CrossRef]
- Cai, Z.; Yongpruksa, N.; Harmata, M. Total synthesis of the terpenoid buddledone A: 11-membered ring-closing metathesis. Org. Lett. 2012, 14, 1661–1663. [Google Scholar] [CrossRef] [PubMed]
- Simova, S.D.; Bozhkova, N.V.; Orahovats, A.S. 1H and 13C NMR studies of some germacrones and isogermacrones. Org. Magn. Reson. 1984, 22, 431–433. [Google Scholar] [CrossRef]
- Brieskorn, C.H.; Noble, P. Furanosesquiterpenes from the essential oil of myrrh. Phytochemistry 1983, 22, 1207–1211. [Google Scholar] [CrossRef]
- Uchio, Y. Constituents of the essential oil of Chrysanthemum japonense. Nojigiku alcohol and its acetate. Bull. Chem. Soc. Jpn. 1978, 51, 2342–2346. [Google Scholar] [CrossRef]
- Da Silva, T.M.; Pinheiro, C.D.; Orlandi, P.P.; Pinheiro, C.C.; Sontes, G.S. Zerumbone from Zingiber zerumbet (L.) smith: A potential prophylactic and therapeutic agent against the cariogenic bacterium Streptococcus mutans. BMC Complement. Altern. Med. 2018, 18, 301. [Google Scholar]
- Gibbons, S.; Udo, E.E. The effect of reserpine, a modulator of multidrug efflux pumps, on the in vitro activity of tetracycline against clinical isolates of methicillin resistant Staphylococcus aureus (MRSA) possessing the tet(K) determinant. Phytother. Res. 2000, 14, 139–140. [Google Scholar] [CrossRef]
- Kaatz, G.W.; Seo, S.M.; Ruble, C.A. Efflux-mediated fluoroquinolone resistance in Staphylococcus aureus. Antimicrob. Agents Chemother. 1993, 37, 1086–1094. [Google Scholar] [CrossRef]
- Ross, J.L.; Farrell, A.M.; Eady, E.A.; Cove, J.H.; Cunliffe, W.J.J. Characterisation and molecular cloning of the novel macrolide-streptogramin B resistance determinant from Staphylococcus epidermidis. Antimicrob. Agents Chemother. 1989, 24, 851–862. [Google Scholar] [CrossRef]
- Richardson, J.F.; Reith, S. Characterization of a strain of methicillin-resistant Staphylococcus aureus (EMRSA-15) by conventional and molecular methods. J. Hosp. Infect. 1993, 25, 45–52. [Google Scholar] [CrossRef]
Sample Availability: Samples of the compounds 1–8 are available from the authors. |

| Extracts/Antibiotic | MICs in µg/mL | ||||
|---|---|---|---|---|---|
| SA1199B | XU212 | EMRSA15 | RN4229 | ATCC25941 | |
| n-Hexane | 128 | 128 | 256 | 256 | 128 |
| Chloroform | 64 | 128 | 128 | 128 | 256 |
| Methanol | >512 | >512 | >512 | >512 | >512 |
| Norfloxacin | 32 | 64 | 16 | 8 | 16 |
| Position | 1H | 13C | HMBC | |
|---|---|---|---|---|
| 2J | 3J | |||
| 1 | 1.06, m, 1H; 1.68, m, 1H | 39.4 | - | C-9 |
| 2 | 1.50, m, 1H; 1.57, m, 1H | 19.5 | C-3 | C-4 |
| 3 | 1.18, m, 1H; 1.41, m, 1H | 42 | - | C-18, C19 |
| 4 | - | 33.6 | - | - |
| 5 | 1.13, m, 1H | 55.8 | C6 | C3, C7, C9, C19, C20 |
| 6 | 1.34, m, 1H; 1.75, m, 1H | 24.4 | C5, C7 | C10 |
| 7 | 2.02, m, 1H; 2.42, m, 1H | 38.1 | C8 | C5, C17 |
| 8 | - | 148.4 | - | - |
| 9 | 1.90, m, 1H | 56.6 | C10, C11 | C12, C17, C20 |
| 10 | - | 39.8 | - | - |
| 11 | 2.31, m, 1H; 2.49, m, 1H | 24.8 | C9 | C8, C13 |
| 12 | 6.76, t, J = 6.6 Hz, 1H | 160.4 | - | C9, C14, C16 |
| 13 | - | 135 | - | - |
| 14 | 3.41, d, J = 16.8 Hz, 1H | 39.6 | C13 | C12, C16 |
| 3.46, d, J = 16.7 Hz, 1H | ||||
| 15 | 9.63, t, J = 14.4 Hz, 1H | 197.5 | C14 | C13 |
| 16 | 9.40, s, 1H | 193.8 | C13 | C12, C14 |
| 17 | 4.36, s, 1H; 4.86, s, 1H | 108.1 | C8 | C7, C9 |
| 18 | 0.88, s, 3H | 33.7 | - | C3, C5, C19 |
| 19 | 0.82, s, 3H | 22.1 | - | C3, C5, C18 |
| 20 | 0.72, s, 3H | 14.6 | C10 | C5, C9 |
| Position | 1H | 13C | HMBC | |
|---|---|---|---|---|
| 2J | 3J | |||
| 1 | 4.63, d, J = 7.5 Hz, 1H | 78.8 | - | C3, C10, C12 |
| 2 | - | 142.2 | - | - |
| 3 | 5.20, d, J = 7.5 Hz, 1H | 124.8 | - | C1, C12 |
| 4 | 2.20, m, 1H; 2.24, m, 1H | 24.4 | C3 | C6 |
| 5 | 2.35, m, 2H | 39.5 | - | C3, C13 |
| 6 | - | 133.2 | - | - |
| 7 | 4.82, dd, J = 10.2, 4.4 Hz, 1H | 125 | - | C5, C9 |
| 8 | 1.87, m, 1H; 2.32, m, 1H | 42.4 | C7 | C6, C10 |
| 9 | - | 37.3 | - | - |
| 10 | 5.23, d, J = 16.2 Hz, 1H | 139.5 | - | C1, C14, C15 |
| 11 | 5.56, dd, J = 16.2, 7.5 Hz, 1H | 131.2 | C1 | C9, C13 |
| 12 | 1.65, s, 3H | 12.8 | C2 | C1, C3 |
| 13 | 1.43, s, 3H | 15.3 | C6 | C5, C7 |
| 14 | 1.04, s, 3H | 24.9 | C9 | C8, C10, C15 |
| 15 | 1.06, s, 3H | 29.9 | C9 | C8, C10, C14 |
| Compound | SA1199B | XU212 | ATCC25941 | RN4220 | EMRSA15 | MRSA27819 | MRSA340702 |
|---|---|---|---|---|---|---|---|
| 1 | 0.212 | 0.424 | 0.212 | 0.212 | 0.212 | 0.424 | 0.424 |
| 2 | 0.291 | 0.582 | 0.582 | 0.582 | 0.145-0.291 | 0.582 | >0.582 |
| 3 | >0.587 | >0.587 | >0.587 | >0.587 | >0.587 | >0.587 | >0.587 |
| 4 | >0.582 | >0.582 | >0.582 | >0.582 | >0.582 | >0.582 | >0.582 |
| 5 | >0.587 | >0.587 | >0.587 | >0.587 | >0.587 | >0.587 | >0.587 |
| 6 | >0.557 | >0.557 | >0.557 | >0.557 | >0.557 | >0.557 | >0.557 |
| 7 | >0.831 | >0.831 | >0.831 | >0.831 | >0.831 | >0.831 | >0.831 |
| 8 | >0.842 | >0.842 | >0.842 | >0.842 | >0.842 | >0.842 | >0.842 |
| Norfloxacin | 0.100 | 0.200 | 0.050 | 0.025 | 0.050 | 0.100 | 0.401 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Siddique, H.; Pendry, B.; Rahman, M.M. Terpenes from Zingiber montanum and Their Screening against Multi-Drug Resistant and Methicillin Resistant Staphylococcus aureus. Molecules 2019, 24, 385. https://doi.org/10.3390/molecules24030385
Siddique H, Pendry B, Rahman MM. Terpenes from Zingiber montanum and Their Screening against Multi-Drug Resistant and Methicillin Resistant Staphylococcus aureus. Molecules. 2019; 24(3):385. https://doi.org/10.3390/molecules24030385
Chicago/Turabian StyleSiddique, Holly, Barbara Pendry, and M. Mukhlesur Rahman. 2019. "Terpenes from Zingiber montanum and Their Screening against Multi-Drug Resistant and Methicillin Resistant Staphylococcus aureus" Molecules 24, no. 3: 385. https://doi.org/10.3390/molecules24030385
APA StyleSiddique, H., Pendry, B., & Rahman, M. M. (2019). Terpenes from Zingiber montanum and Their Screening against Multi-Drug Resistant and Methicillin Resistant Staphylococcus aureus. Molecules, 24(3), 385. https://doi.org/10.3390/molecules24030385