Tissue Transparency In Vivo
Abstract
1. Introduction
2. Methods for Reducing Light Absorption
2.1. Chemical Decoloration of Pigments
2.2. Genetic Approach to Removing Pigments
3. Methods for Reducing Light Scattering
3.1. Chemical Reduction of Scattering In Vivo
3.2. Genetic Approach to Reducing Scattering
3.2.1. Glycosaminoglycans and Antifreeze Proteins in Fish
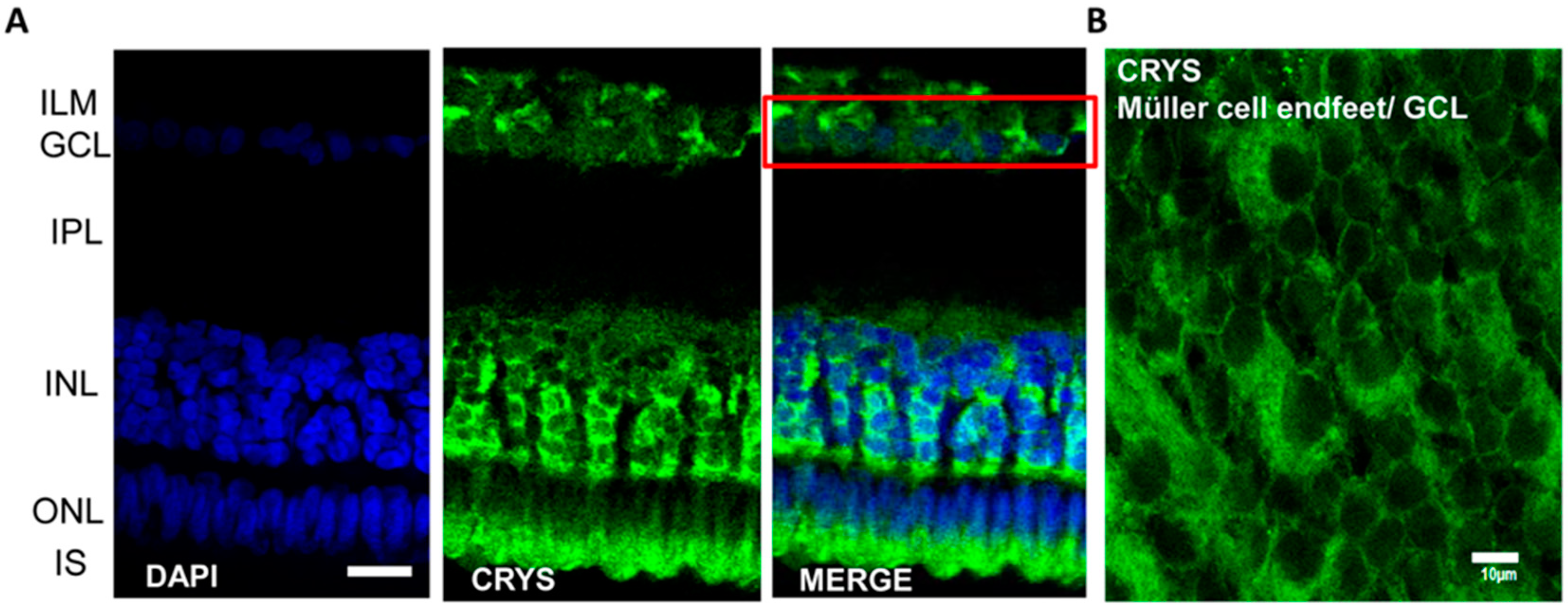
3.2.2. Crystallins
3.2.3. Possible Genetic Manipulation to Induce Transparency and Some Future Perspective
4. In Vivo Transparency of Bones and Other Mineralized Tissue
5. Other Models of Transparency
6. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Zylinski, S.; Johnsen, S. Mesopelagic cephalopods switch between transparency and pigmentation to optimize camouflage in the deep. Curr. Biol. 2011, 21, 1937–1941. [Google Scholar] [CrossRef] [PubMed]
- Zylinski, S.; Osorio, D.; Johnsen, S. Cuttlefish see shape from shading, fine-tuning coloration in response to pictorial depth cues and directional illumination. Proc. Biol. Sci. 2016, 283, 20160062. [Google Scholar] [CrossRef] [PubMed]
- Fetcho, J.R.; O’Malley, D.M. Visualization of active neural circuitry in the spinal cord of intact zebrafish. J. Neurophysiol. 1995, 73, 399–406. [Google Scholar] [CrossRef] [PubMed]
- O’Malley, D.M.; Zhou, Q.; Gahtan, E. Probing neural circuits in the zebrafish: A suite of optical techniques. Methods 2003, 30, 49–63. [Google Scholar] [CrossRef]
- White, R.M.; Sessa, A.; Burke, C.; Bowman, T.; LeBlanc, J.; Ceol, C.; Bourque, C.; Dovey, M.; Goessling, W.; Burns, C.E.; et al. Transparent adult zebrafish as a tool for in vivo transplantation analysis. Cell Stem Cell 2008, 2, 183–189. [Google Scholar] [CrossRef]
- Bin, J.M.; Lyons, D.A. Imaging Myelination In Vivo Using Transparent Animal Models. Brain Plast. 2016, 2, 3–29. [Google Scholar] [CrossRef]
- Antinucci, P.; Hindges, R. A crystal-clear zebrafish for in vivo imaging. Sci. Rep. 2016, 6, 29490. [Google Scholar] [CrossRef] [PubMed]
- Harrison, N.R.; Laroche, F.J.; Gutierrez, A.; Feng, H. Zebrafish Models of Human Leukemia: Technological Advances and Mechanistic Insights. Adv. Exp. Med. Biol. 2016, 916, 335–369. [Google Scholar] [CrossRef] [PubMed]
- Nicolson, T. The genetics of hair-cell function in zebrafish. J. Neurogenet. 2017, 31, 102–112. [Google Scholar] [CrossRef]
- Saleem, S.; Kannan, R.R. Zebrafish: An emerging real-time model system to study Alzheimer’s disease and neurospecific drug discovery. Cell Death Discov. 2018, 5, 45. [Google Scholar] [CrossRef]
- Zayas-Santiago, A.; Ríos, D.S.; Zueva, L.V.; Inyushin, M.Y. Localization of αA-Crystallin in Rat Retinal Müller Glial Cells and Photoreceptors. Microsc. Microanal. 2018, 24, 545–552. [Google Scholar] [CrossRef] [PubMed]
- Brunsting, A.; Mullaney, F. Differential light scattering from spherical mammalian cells. Biophys. J. 1974, 14, 439–453. [Google Scholar] [CrossRef]
- Tardieu, A.; Delaye, M. Eye lens proteins and transparency: From light transmission theory to solution X-ray structural analysis. Ann. Rev. Biophys. Chem. 1988, 17, 1207–1215. [Google Scholar] [CrossRef] [PubMed]
- Jacques, S.L.; Pogue, B.W. Tutorial on diffuse light transport. J. Biomed. Opt. 2008, 13, 041302. [Google Scholar] [CrossRef] [PubMed]
- Jacques, S.L. Optical properties of biological tissues: A review. Phys. Med. Biol. 2013, 58, R37–R61. [Google Scholar] [CrossRef] [PubMed]
- Perelman, L.T. Light Scattering. In Handbook of Optical Metrology: Principles and Applications; Yoshizawa, T., Ed.; CRC Press: Boca Raton, FL, USA, 2017; pp. 321–337. ISBN 9781138893634. [Google Scholar]
- Jansen, E.D.; Pickett, P.M.; Mackanos, M.A.; Virostko, J. Effect of optical tissue clearing on spatial resolution and sensitivity of bioluminescence imaging. J. Biomed. Opt. 2006, 11, 041119. [Google Scholar] [CrossRef] [PubMed]
- Zueva, L.; Golubeva, T.; Korneeva, E.; Makarov, V.; Khmelinskii, I.; Inyushin, M. Foveolar Müller Cells of the Pied Flycatcher: Morphology and Distribution of Intermediate Filaments Regarding Cell Transparency. Microsc. Microanal. 2016, 22, 379–386. [Google Scholar] [CrossRef] [PubMed]
- Xu, J.; Ruan, H.; Liu, Y.; Zhou, H.; Yang, C. Focusing light through scattering media by transmission matrix inversion. Opt. Express 2017, 25, 27234–27246. [Google Scholar] [CrossRef] [PubMed]
- Cua, M.; Zhou, H.; Yang, C. Imaging moving targets through scattering media. Opt. Express 2017, 25, 3935–3945. [Google Scholar] [CrossRef] [PubMed]
- Parvez, S.; Kang, M.; Chung, H.S.; Cho, C.; Hong, M.C.; Shin, M.K.; Bae, H. Survey and mechanism of skin depigmenting and lightening agents. Phytother. Res. 2006, 20, 921–934. [Google Scholar] [CrossRef]
- Heriniaina, R.M.; Dong, J.; Kalavagunta, P.K.; Wu, H.L.; Yan, D.S.; Shang, J. Effects of six compounds with different chemical structures on melanogenesis. Chin. J. Nat. Med. 2018, 16, 766–773. [Google Scholar] [CrossRef]
- Karlsson, J.; von Hofsten, J.; Olsson, P.E. Generating transparent zebrafish: A refined method to improve detection of gene expression during embryonic development. Mar. Biotechnol. 2001, 3, 522–527. [Google Scholar] [CrossRef] [PubMed]
- Rebelo, J.; Auerbach, G.; Bader, G.; Bracher, A.; Nar, H.; Hösl, C.; Schramek, N.; Kaiser, J.; Bacher, A.; Huber, R.; et al. Biosynthesis of Pteridines. Reaction Mechanism of GTP Cyclohydrolase I. J. Mol. Biol. 2003, 326, 503–516. [Google Scholar] [CrossRef]
- Matthews, S.J.; Ross, N.W.; Lall, S.P.; Gill, T.A. Astaxanthin binding protein in Atlantic salmon. Comp. Biochem. Physiol. B Biochem. Mol. Biol. 2006, 144, 206–214. [Google Scholar] [CrossRef] [PubMed]
- Hata, M.; Hata, M. Carotenoid pigments in goldfish (Carassius auratus L.). VI. The effect of ACTH and phenylthiourea on the discoloration and the carotenoid metabolism. Comp. Biochem. Physiol. A Comp. Physiol. 1973, 46, 95–101. [Google Scholar] [CrossRef]
- Forrest, H.S.; Glassman, E.; Mitchell, H.K. Conversion of 2-amino-4-hydroxypteridine to isoxanthopterin in D. Melanogaster. Science 1956, 124, 725–726. [Google Scholar] [CrossRef] [PubMed]
- Demski, L.S. Chromatophore systems in teleosts and cephalopods: A levels oriented analysis of convergent systems. Brain Behav. Evol. 1992, 40, 141–156. [Google Scholar] [CrossRef]
- Liu, T.H.; Chiao, C.C. Mosaic Organization of Body Pattern Control in the Optic Lobe of Squids. J. Neurosci. 2017, 37, 768–780. [Google Scholar] [CrossRef]
- Reiter, S.; Hülsdunk, P.; Woo, T.; Lauterbach, M.A.; Eberle, J.; Akay, L.A.; Longo, A.; Meier-Credo, J.; Kretschmer, F.; Langer, J.D.; et al. Elucidating the control and development of skin patterning in cuttlefish. Nature 2018, 562, 361–366. [Google Scholar] [CrossRef]
- Saenko, S.V.; Teyssier, J.; van der Marel, D.; Milinkovitch, M.C. Precise colocalization of interacting structural and pigmentary elements generates extensive color pattern variation in Phelsuma lizards. BMC Biol. 2013, 11, 105. [Google Scholar] [CrossRef]
- Teyssier, J.; Saenko, S.V.; van der Marel, D.; Milinkovitch, M.C. Photonic crystals cause active colour change in chameleons. Nat. Commun. 2015, 6, 6368. [Google Scholar] [CrossRef] [PubMed]
- Lythgoe, J.N.; Shand, J.; Foster, R.G. Visual pigment in fish iridocytes. Nature 1984, 308, 83–84. [Google Scholar] [CrossRef]
- Schweikert, L.E.; Fitak, R.R.; Johnsen, S. De novo transcriptomics reveal distinct phototransduction signaling components in the retina and skin of a color-changing vertebrate, the hogfish (Lachnolaimus maximus). J. Comp. Physiol. A Neuroethol. Sens. Neural Behav. Physiol. 2018, 204, 475–485. [Google Scholar] [CrossRef] [PubMed]
- Ziegler, I.; McDonald, T.; Hesslinger, C.; Pelletier, I.; Boyle, P. Development of the pteridine pathway in the zebrafish, Danio rerio. J. Biol. Chem. 2000, 275, 18926–18932. [Google Scholar] [CrossRef] [PubMed]
- Eskova, A.; Chauvigné, F.; Maischein, H.M.; Ammelburg, M.; Cerdà, J.; Nüsslein-Volhard, C.; Irion, U. Gain-of-function mutations in Aqp3a influence zebrafish pigment pattern formation through the tissue environment. Development 2017, 144, 2059–2069. [Google Scholar] [CrossRef] [PubMed]
- Gunia-Krzyżak, A.; Popiol, J.; Marona, H. Melanogenesis Inhibitors: Strategies for Searching for and Evaluation of Active Compounds. Curr. Med. Chem. 2016, 23, 3548–3574. [Google Scholar] [CrossRef]
- Lajis, A.F.B. A Zebrafish Embryo as an Animal Model for the Treatment of Hyperpigmentation in Cosmetic Dermatology Medicine. Medicina (Kaunas) 2018, 54, 35. [Google Scholar] [CrossRef] [PubMed]
- Ono, K.; Viet, C.T.; Ye, Y.; Dang, D.; Hitomi, S.; Toyono, T.; Inenaga, K.; Dolan, J.C.; Schmidt, B.L. Cutaneous pigmentation modulates skin sensitivity via tyrosinase-dependent dopaminergic signalling. Sci. Rep. 2017, 7, 9181. [Google Scholar] [CrossRef] [PubMed]
- Rader, K. Making Mice, Standardizing Animals for American Biomedical Research; Princeton Univ Press: Princeton, NJ, USA, 2004; 312p, ISBN 9780691016368. [Google Scholar]
- Conway, K.S.; Trudeau, J. Sunshine, fertility and racial disparities. Econ. Hum. Biol. 2018, 32, 18–39. [Google Scholar] [CrossRef] [PubMed]
- Lister, J.A.; Robertson, C.P.; Lepage, T.; Johnson, S.L.; Raible, D.W. Nacre Encodes a Zebrafish Microphthalmia-Related Protein that Regulates Neural-Crest-Derived Pigment Cell Fate. Development 1999, 126, 3757–3767. [Google Scholar]
- D’Agati, G.; Beltre, R.; Sessa, A.; Burger, A.; Zhou, Y.; Mosimann, C.; White, R.M. A defect in the mitochondrial protein Mpv17 underlies the transparent casper zebrafish. Dev. Biol. 2017, 430, 11–17. [Google Scholar] [CrossRef] [PubMed]
- Chen, Z.; Chou, S.W.; McDermott, B.M., Jr. Ribeye protein is intrinsically dynamic but is stabilized in the context of the ribbon synapse. J. Physiol. 2018, 596, 409–421. [Google Scholar] [CrossRef] [PubMed]
- Auer, F.; Vagionitis, S.; Czopka, T. Evidence for Myelin Sheath Remodeling in the CNS Revealed by In Vivo Imaging. Curr. Biol. 2018, 28, 549–559. [Google Scholar] [CrossRef] [PubMed]
- Kline, T.L.; Sussman, C.R.; Irazabal, M.V.; Mishra, P.K.; Pearson, E.A.; Torres, V.E.; Macura, S.I. Three-dimensional NMR microscopy of zebrafish specimens. NMR Biomed. 2019, 32, e4031. [Google Scholar] [CrossRef] [PubMed]
- Wakamatsu, Y.; Pristyazhnyuk, S.; Kinoshita, M.; Tanaka, M.; Ozato, K. The see-through medaka: A fish model that is transparent throughout life. Proc. Natl. Acad. Sci. USA 2001, 98, 10046–10050. [Google Scholar] [CrossRef] [PubMed]
- Chung, K.; Wallace, J.; Kim, S.Y.; Kalyanasundaram, S.; Andalman, A.S.; Davidson, T.J.; Mirzabekov, J.J.; Zalocusky, K.A.; Mattis, J.; Denisin, A.K.; et al. Structural and molecular interrogation of intact biological systems. Nature 2013, 497, 332–337. [Google Scholar] [CrossRef] [PubMed]
- Richardson, D.S.; Lichtman, J.W. Clarifying Tissue Clearing. Cell 2015, 162, 246–257. [Google Scholar] [CrossRef] [PubMed]
- Steelman, Z.A.; Eldridge, W.J.; Weintraub, J.B.; Wax, A. Is the nuclear refractive index lower than cytoplasm? Validation of phase measurements and implications for light scattering technologies. J. Biophotonics 2017, 10, 1714–1722. [Google Scholar] [CrossRef]
- Zhang, Q.; Zhong, L.; Tang, P.; Yuan, Y.; Liu, S.; Tian, J.; Lu, X. Quantitative refractive index distribution of single cell by combining phase-shifting interferometry and AFM imaging. Sci. Rep. 2017, 7, 2532. [Google Scholar] [CrossRef]
- Barer, R.; Ross, K.A. Refractometry of living cells. J. Physiol. 1952, 118, 38P–39P. [Google Scholar] [CrossRef]
- Ross, K.F.A. Measurement of the Refractive Index of Cytoplasmic Inclusions in Living Cells by the Interference Microscope. Nature 1954, 174, 836–837. [Google Scholar] [CrossRef] [PubMed]
- Tuchin, V.V. Optical Clearing of Tissues and Blood; SPIE Press: Bellingham, WA, USA, 2006; 256p, ISBN 9780819481108. [Google Scholar]
- Tuchin, V.V.; Maksimova, I.L.; Zimnyakov, D.A.; Kon, I.L.; Mavlyutov, A.H.; Mishin, A.A. Light propagation in tissues with controlled optical properties. J. Biomed. Opt. 1997, 2, 401–417. [Google Scholar] [CrossRef] [PubMed]
- Wen, X.; Mao, Z.; Han, Z.; Tuchin, V.V.; Zhu, D. In vivo skin optical clearing by glycerol solutions: Mechanism. J. Biophoton. 2010, 3, 44–52. [Google Scholar] [CrossRef] [PubMed]
- Zhu, D.; Larin, K.V.; Luo, Q.; Tuchin, V.V. Recent progress in tissue optical clearing. Laser Photon. Rev. 2013, 7, 732–757. [Google Scholar] [CrossRef]
- Shi, R.; Guo, L.; Zhang, C.; Feng, W.; Li, P.; Ding, Z.; Zhu, D. A useful way to develop effective in vivo skin optical clearing agents. J. Biophoton. 2017, 10, 887–895. [Google Scholar] [CrossRef]
- Tuan-Mahmood, T.M.; McCrudden, M.T.; Torrisi, B.M.; McAlister, E.; Garland, M.J.; Singh, T.R.; Donnelly, R.F. Microneedles for intradermal and transdermal drug delivery. Eur. J. Pharm. Sci. 2013, 50, 623–637. [Google Scholar] [CrossRef]
- Urban, B.E.; Xiao, L.; Chen, S.; Yang, H.; Dong, B.; Kozorovitskiy, Y.; Zhang, H.F. In Vivo Superresolution Imaging of Neuronal Structure in the Mouse Brain. IEEE Trans. Biomed. Eng. 2018, 65, 232–238. [Google Scholar] [CrossRef]
- Ke, M.T.; Nakai, Y.; Fujimoto, S.; Takayama, R.; Yoshida, S.; Kitajima, T.S.; Sato, M.; Imai, T. Super-Resolution Mapping of Neuronal Circuitry With an Index-Optimized Clearing Agent. Cell Rep. 2016, 14, 2718–2732. [Google Scholar] [CrossRef]
- Robison, B.H.; Reisenbichler, K.R. Macropinna microstoma and the paradox of its tubular eyes. Copeia 2008, 4, 780–784. [Google Scholar] [CrossRef]
- Pfeiler, E. Isolation and partial characterization of a novel keratan sulfate proteoglycan from metamorphosing bonefish (Albula) larvae. Fish Physiol. Biochem. 1988, 4, 175–187. [Google Scholar] [CrossRef]
- Pfeiler, E.; Toyoda, H.; Williams, M.D.; Nieman, R.A. Identification, structural analysis and function of hyaluronan in developing fish larvae (leptocephali). Comp. Biochem. Physiol. B Biochem. Mol. Biol. 2002, 132, 443–451. [Google Scholar] [CrossRef]
- Miller, M.J. Ecology of Anguilliform Leptocephali: Remarkable Transparent Fish Larvae of the Ocean Surface Layer. Aqua-BioSci. Monogr. 2009, 2, 1–94. [Google Scholar] [CrossRef]
- DeVries, A.L.; Wohlschlag, D.E. Freezing resistance in some Antarctic fishes. Science 1969, 163, 1073–1075. [Google Scholar] [CrossRef] [PubMed]
- Davies, P.L.; Baardsnes, J.; Kuiper, M.J.; Walker, V.K. Structure and function of antifreeze proteins. Philos. Trans. R. Soc. Lond. B Biol. Sci. 2002, 357, 927–935. [Google Scholar] [CrossRef] [PubMed]
- Gilbert, J.A.; Hill, P.J.; Dodd, C.E.; Laybourn-Parry, J. Demonstration of antifreeze protein activity in Antarctic lake bacteria. Microbiology 2004, 150, 171–180. [Google Scholar] [CrossRef] [PubMed]
- Krishnan, K.; Kathiresan, T.; Raman, R.; Rajini, B.; Dhople, V.M.; Aggrawal, R.K.; Sharma, Y. Ubiquitous lens alpha-, beta-, and gamma-crystallins accumulate in anuran cornea as corneal crystallins. J. Biol. Chem. 2007, 282, 18953–18959. [Google Scholar] [CrossRef]
- Jester, J.V. Corneal crystallins and the development of cellular transparency. Semin. Cell Dev. Biol. 2008, 19, 82–93. [Google Scholar] [CrossRef] [PubMed]
- Delaye, M.; Tardieu, A. Short-range order of crystallin proteins accounts for eye lens transparency. Nature 1983, 302, 415–417. [Google Scholar] [CrossRef]
- Horwitz, J.; Bova, M.P.; Ding, L.L.; Haley, D.A.; Stewart, P.L. Lens alpha-crystallin: Function and structure. Eye 1999, 13, 403–408. [Google Scholar] [CrossRef]
- Andley, U.P. Effects of alpha-crystallin on lens cell function and cataract pathology. Curr. Mol. Med. 2009, 9, 887–892. [Google Scholar] [CrossRef]
- Simirskiĭ, V.N.; Panova, I.G.; Sologub, A.A.; Aleĭnikova, K.S. Localization of crystallins in Muellerian cells in the grass frog retina. Ontogenez 2003, 34, 365–370. [Google Scholar] [PubMed]
- Deretic, D.; Aebersold, R.H.; Morrison, H.D.; Papermaster, D.S. Alpha A- and alpha B-crystallin in the retina. Association with the post-Golgi compartment of frog retinal photoreceptors. J. Biol. Chem. 1994, 269, 16853–16861. [Google Scholar] [PubMed]
- Maeda, A.; Ohguro, H.; Maeda, T.; Nakagawa, T.; Kuroki, Y. Low expression of alphaA-crystallins and rhodopsin kinase of photoreceptors in retinal dystrophy rat. Investig. Ophthalmol. Vis. Sci. 1999, 40, 2788–2794. [Google Scholar] [PubMed]
- Takemoto, L.; Boyle, D. The possible role of alpha-crystallins in human senile cataractogenesis. Int. J. Biol. Macromol. 1998, 22, 331–337. [Google Scholar] [CrossRef]
- Datiles, M.B.; Ansari, R.R.; Suh, K.I.; Vitale, S.; Reed, G.F.; Zigler, J.S.; Ferris, F.L. Clinical detection of precataractous lens protein changes using dynamic light scattering. Arch. Ophthalmol. 2008, 126, 1687–1693. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Graw, J. Genetics of crystallins: Cataract and beyond. Exp. Eye Res. 2009, 88, 173–189. [Google Scholar] [CrossRef]
- Franze, K.; Grosche, J.; Skatchkov, S.N.; Schinkinger, S.; Foja, C.; Schild, D.; Uckermann, O.; Travis, K.; Reichenbach, A.; Guck, J. Muller cells are living optical fibers in the vertebrate retina. Proc. Natl. Acad. Sci. USA 2007, 104, 8287–8292. [Google Scholar] [CrossRef]
- Reichenbach, A.; Bringmann, A. Müller Cells in the Healthy and Diseased Retina; Springer Science+Business Media: Luxemburg, 2010. [Google Scholar]
- Agte, S.; Junek, S.; Matthias, S.; Ulbricht, E.; Erdmann, I.; Wurm, A.; Schild, D.; Käs, J.A.; Reichenbach, A. Müller glial cell-provided cellular light guidance through the vital guinea-pig retina. Biophys. J. 2011, 101, 2611–2619. [Google Scholar] [CrossRef]
- Reichenbach, A.; Bringmann, A. New functions of Müller cells. Glia 2013, 61, 651–678. [Google Scholar] [CrossRef]
- Agte, S.; Savvinov, A.; Karl, A.; Zayas-Santiago, A.; Ulbricht, E.; Makarov, V.I.; Reichenbach, A.; Bringmann, A.; Skatchkov, S.N. Müller glial cells contribute to dim light vision in the spectacled caiman (Caiman crocodilus fuscus): Analysis of retinal light transmission. Exp. Eye Res. 2018, 173, 91–108. [Google Scholar] [CrossRef]
- Karl, A.; Agte, S.; Zayas-Santiago, A.; Makarov, F.N.; Rivera, Y.; Benedikt, J.; Francke, M.; Reichenbach, A.; Skatchkov, S.N.; Bringmann, A. Retinal adaptation to dim light vision in spectacled caimans (Caiman crocodilus fuscus): Analysis of retinal ultrastructure. Exp. Eye Res. 2018, 173, 160–178. [Google Scholar] [CrossRef] [PubMed]
- Lewis, G.P.; Erickson, P.A.; Kaska, D.D.; Fisher, S.K. An immunocytochemical comparison of Müller cells and astrocytes in the cat retina. Exp. Eye Res. 1988, 47, 839–853. [Google Scholar] [CrossRef]
- Ruebsam, A.; Dulle, J.E.; Myers, A.M.; Sakrikar, D.; Green, K.M.; Khan, N.W.; Schey, K.; Fort, P.E. A specific phosphorylation regulates the protective role of αA-crystallin in diabetes. JCI Insight 2018, 3, 97919. [Google Scholar] [CrossRef] [PubMed]
- Zayas-Santiago, A.; Agte, S.; Rivera, Y.; Benedikt, J.; Ulbricht, E.; Karl, A.; Dávila, J.; Savvinov, A.; Kucheryavykh, Y.; Inyushin, M.; et al. Unidirectional photoreceptor-to-Müller glia coupling and unique K+ channel expression in Caiman retina. PLoS ONE 2014, 9, e97155. [Google Scholar] [CrossRef] [PubMed]
- Woodley, C.L.; David, H.L. Effect of temperature on the rate of the transparent to opaque colony type transition in Mycobacterium avium. Antimicrob. Agents Chemother. 1976, 9, 113–119. [Google Scholar] [CrossRef] [PubMed]
- Yuan, Y.; Crane, D.D.; Simpson, R.M.; Zhu, Y.Q.; Hickey, M.J.; Sherman, D.R.; Barry, C.E., 3rd. The 16-kDa alpha-crystallin (Acr) protein of Mycobacterium tuberculosis is required for growth in macrophages. Proc. Natl. Acad. Sci. USA 1998, 95, 9578–9583. [Google Scholar] [CrossRef] [PubMed]
- Stewart, J.N.; Rivera, H.N.; Karls, R.; Quinn, F.D.; Roman, J.; Rivera-Marrero, C.A. Increased pathology in lungs of mice after infection with an alpha-crystallin mutant of Mycobacterium tuberculosis: changes in cathepsin proteases and certain cytokines. Microbiology 2006, 152, 233–244. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Cheng, A.W.; Wang, H.; Yang, H.; Shi, L.; Katz, Y.; Theunissen, T.W.; Rangarajan, S.; Shivalila, C.S.; Dadon, D.B.; Jaenisch, R. Multiplexed activation of endogenous genes by CRISPR-on, an RNA-guided transcriptional activator system. Cell Res. 2013, 23, 1163–1171. [Google Scholar] [CrossRef]
- Perez-Pinera, P.; Kocak, D.D.; Vockley, C.M.; Adler, A.F.; Kabadi, A.M.; Polstein, L.R.; Thakore, P.I.; Glass, K.A.; Ousterout, D.G.; Leong, K.W.; et al. RNA-guided gene activation by CRISPR-Cas9-based transcription factors. Nat. Methods 2013, 10, 973–976. [Google Scholar] [CrossRef]
- Woodley, D.T.; Keene, D.R.; Atha, T. Intradermal injection of lentiviral vectors corrects regenerated human dystrophic epidermolysis bullosa skin tissue in vivo. Mol. Ther. 2004, 10, 318–326. [Google Scholar] [CrossRef]
- Velasco-Hogan, A.; Deheyn, D.D.; Koch, M.; Nothdurft, B.; Arzt, E.; Meyers, M.A. On the Nature of the Transparent Teeth of the Deep-Sea Dragonfish, Aristostomias scintillans. Matter 2019, 1, 1–15. [Google Scholar] [CrossRef]
- Greenbaum, A.; Chan, K.Y.; Dobreva, T.; Brown, D.; Balani, D.H.; Boyce, R.; Kronenberg, H.M.; McBride, H.J.; Gradinaru, V. Bone CLARITY: Clearing, imaging, and computational analysis of osteoprogenitors within intact bone marrow. Sci. Transl. Med. 2017, 9, 6518. [Google Scholar] [CrossRef] [PubMed]
- Jing, D.; Yi, Y.; Luo, W.; Zhang, S.; Yuan, Q.; Wang, J.; Lachika, E.; Zhao, Z.; Zhao, H. Tissue Clearing and Its Application to Bone and Dental Tissues. J. Dent. Res. 2019, 98, 621–631. [Google Scholar] [CrossRef] [PubMed]
- Genina, E.A.; Bashkatov, A.N.; Tuchin, V.V. Optical Clearing of Cranial Bone. Adv. Opt. Technol. 2008, 2008, 267867. [Google Scholar] [CrossRef]
- Kalchenko, V.; Israeli, D.; Kuznetsov, Y.; Harmelin, A. Transcranial optical vascular imaging (TOVI) of cortical hemodynamics in mouse brain. Sci. Rep. 2014, 4, 5839. [Google Scholar] [CrossRef] [PubMed]
- Guo, Z.V.; Li, N.; Huber, D.; Ophir, E.; Gutnisky, D.; Ting, J.T.; Feng, G.; Svoboda, K. Flow of Cortical Activity Underlying a Tactile Decision in Mice. Neuron 2014, 81, 179–194. [Google Scholar] [CrossRef]
- Steinzeig, A.; Molotkov, D.; Castrén, E. Chronic imaging through “transparent skull” in mice. PLoS ONE 2017, 12, e0181788. [Google Scholar] [CrossRef]
- Quinlan, R.A.; Carte, J.M.; Sandilands, A.; Prescott, A.R. The beaded filament of the eye lens: An unexpected key to intermediate filament structure and function. Trends Cell Biol. 1996, 6, 123–126. [Google Scholar] [CrossRef]
- Zueva, L.; Makarov, V.; Zayas-Santiago, A.; Golubeva, T.; Korneeva, E.; Savvinov, A.; Eaton, M.; Skatchkov, S.; Inyushin, M. Müller cell alignment in bird fovea: possible role in vision. J. Neurosci. Neuroeng. 2014, 3, 85–91. [Google Scholar] [CrossRef]
- Makarov, V.; Zueva, L.; Golubeva, T.; Korneeva, E.; Khmelinskii, I.; Inyushin, M. Quantum mechanism of light transmission by the intermediate filaments in some specialized optically transparent cells. Neurophotonics 2017, 4, 011005. [Google Scholar] [CrossRef][Green Version]
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Inyushin, M.; Meshalkina, D.; Zueva, L.; Zayas-Santiago, A. Tissue Transparency In Vivo. Molecules 2019, 24, 2388. https://doi.org/10.3390/molecules24132388
Inyushin M, Meshalkina D, Zueva L, Zayas-Santiago A. Tissue Transparency In Vivo. Molecules. 2019; 24(13):2388. https://doi.org/10.3390/molecules24132388
Chicago/Turabian StyleInyushin, Mikhail, Daria Meshalkina, Lidia Zueva, and Astrid Zayas-Santiago. 2019. "Tissue Transparency In Vivo" Molecules 24, no. 13: 2388. https://doi.org/10.3390/molecules24132388
APA StyleInyushin, M., Meshalkina, D., Zueva, L., & Zayas-Santiago, A. (2019). Tissue Transparency In Vivo. Molecules, 24(13), 2388. https://doi.org/10.3390/molecules24132388





