The Keap1/Nrf2-ARE Pathway as a Pharmacological Target for Chalcones
Abstract
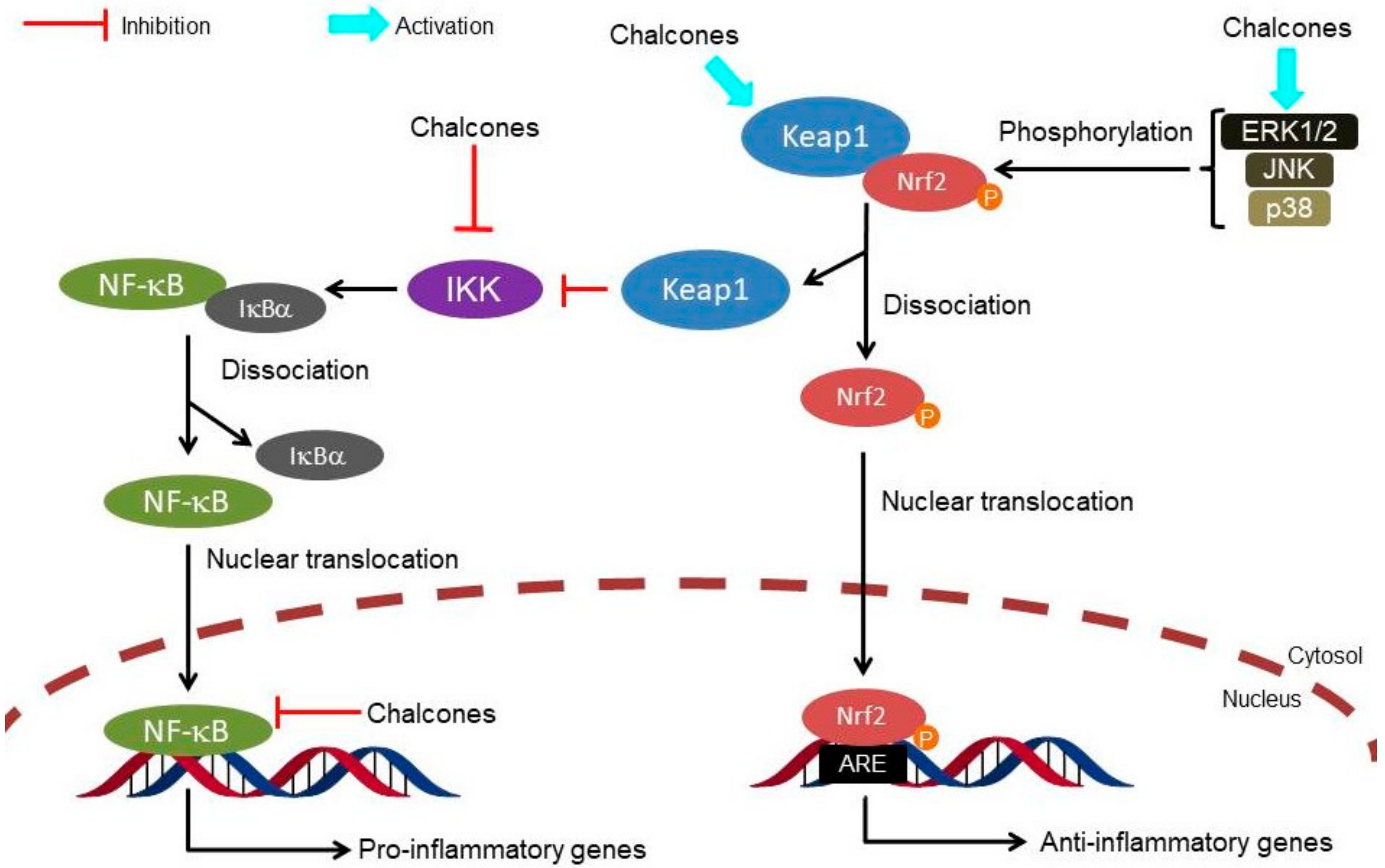
1. Introduction
2. Targeting the Nrf2 Pathway by Natural Chalcones
3. Targeting the Nrf2 Pathway by Synthetic Chalcones
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Gomes, M.N.; Muratov, E.N.; Pereira, M.; Peixoto, J.C.; Rosseto, L.P.; Cravo, P.V.L.; Andrade, C.H.; Neves, B.J. Chalcone derivatives: Promising starting points for drug design. Molecules 2017, 22, 1210. [Google Scholar] [CrossRef] [PubMed]
- Zhuang, C.; Zhang, W.; Sheng, C.; Zhang, W.; Xing, C.; Miao, Z. Chalcone: A privileged structure in medicinal chemistry. Chem. Rev. 2017, 117, 7762–7810. [Google Scholar] [CrossRef] [PubMed]
- Singh, P.; Anand, A.; Kumar, V. Recent developments in biological activities of chalcones: A mini review. Eur. J. Med. Chem. 2014, 85, 758–777. [Google Scholar] [CrossRef] [PubMed]
- Zhou, B.; Xing, C. Diverse molecular targets for chalcones with varied bioactivities. Med. Chem. (Los. Angeles). 2015, 5, 388–404. [Google Scholar] [CrossRef] [PubMed]
- Maydt, D.; De Spirt, S.; Muschelknautz, C.; Stahl, W.; Müller, T.J.J. Chemical reactivity and biological activity of chalcones and other α,β-unsaturated carbonyl compounds. Xenobiotica 2013, 43, 711–718. [Google Scholar] [CrossRef] [PubMed]
- Dinkova-Kostova, A.T.; Massiah, M.A.; Bozak, R.E.; Hicks, R.J.; Talalay, P. Potency of Michael reaction acceptors as inducers of enzymes that protect against carcinogenesis depends on their reactivity with sulfhydryl groups. Proc. Natl. Acad. Sci. USA 2001, 98, 3404–3409. [Google Scholar] [CrossRef] [PubMed]
- Yamamoto, X.M.; Kensler, T.W.; Motohashi, H. The Keap1-Nrf2 system: A thiol-based sensor-effector apparatus for maintaining redox homeostasis. Physiol. Rev. 2018, 98, 1169–1203. [Google Scholar] [CrossRef] [PubMed]
- Holland, R.; Fishbein, J.C. Chemistry of the cysteine sensors in Kelch-Like ECH-Associated Protein 1. Antioxid. Redox Signal. 2010, 13, 1749–1761. [Google Scholar] [CrossRef] [PubMed]
- Satoh, T.; Lipton, S. Recent advances in understanding NRF2 as a druggable target: Development of pro-electrophilic and non-covalent NRF2 activators to overcome systemic side effects of electrophilic drugs like dimethyl fumarate. F1000Research 2017, 6, 2138. [Google Scholar] [CrossRef] [PubMed]
- Dinkova-Kostova, A.T.; Kazantsev, A.G. Activation of Nrf2 signaling as a common treatment of neurodegenerative diseases. Neurodegener. Dis. Manag. 2017, 7, 97–100. [Google Scholar] [CrossRef] [PubMed]
- McBean, G.J.; López, M.G.; Wallner, F.K. Redox-based therapeutics in neurodegenerative disease. Br. J. Pharmacol. 2017, 174, 1750–1770. [Google Scholar] [CrossRef] [PubMed]
- Salazar, M.; Rojo, A.I.; Velasco, D.; De Sagarra, R.M.; Cuadrado, A. Glycogen synthase kinase-3β inhibits the xenobiotic and antioxidant cell response by direct phosphorylation and nuclear exclusion of the transcription factor Nrf2. J. Biol. Chem. 2006, 281, 14841–14851. [Google Scholar] [CrossRef] [PubMed]
- Wardyn, J.; Ponsford, A.; Sanderson, C. Dissecting molecular cross-talk between Nrf2 and NF-κB response pathways. Biochem. Soc. Trans. 2015, 43, 621–626. [Google Scholar] [CrossRef] [PubMed]
- Cuadrado, A.; Manda, G.; Hassan, A.; Alcaraz, M.J.; Barbas, C.; Daiber, A.; Ghezzi, P.; León, R.; López, M.G.; Oliva, B.; et al. Transcription factor Nrf2 as a therapeutic target for chronic diseases: A systems medicine approach. Pharmacol. Rev. 2018, 70, 348–383. [Google Scholar] [CrossRef] [PubMed]
- Pandey, M.K.; Sandur, S.K.; Sung, B.; Sethi, G.; Kunnumakkara, A.B.; Aggarwal, B.B. Butein, a tetrahydroxychalcone, inhibits nuclear factor (NF)-κB and NF-κB-regulated gene expression through direct inhibition of IκBα kinase β on cysteine 179 residue. J. Biol. Chem. 2007, 282, 17340–17350. [Google Scholar] [CrossRef] [PubMed]
- Lorenzo, P.; Alvarez, R.; Ortiz, M.A.; Alvarez, S.; Piedrafita, F.J.; de Lera, A.R. Inhibition of IκB kinase-β and anticancer activities of novel chalcone adamantyl arotinoids. J. Med. Chem. 2008, 51, 5431–5440. [Google Scholar] [CrossRef] [PubMed]
- Funakoshi-Tago, M.; Tanabe, S.; Tago, K.; Itoh, H.; Mashino, T.; Sonoda, Y.; Kasahara, T. Licochalcone a potently inhibits Tumor Necrosis Factor α-Induced Nuclear Factor-κB activation through the direct inhibition of IκB Kinase complex activation. Mol. Pharmacol. 2009, 76, 745–753. [Google Scholar] [CrossRef] [PubMed]
- Shen, K.-H.; Chang, J.-K.; Hsu, Y.-L.; Kuo, P.-L. Chalcone arrests cell cycle progression and induces apoptosis through induction of mitochondrial pathway and inhibition of Nuclear Factor Kappa B signalling in human bladder cancer cells. Basic Clin. Pharmacol. Toxicol. 2007, 101, 254–261. [Google Scholar] [CrossRef] [PubMed]
- Lu, M.-C.; Ji, J.-A.; Jiang, Z.-Y.; You, Q.-D. The Keap1-Nrf2-ARE pathway as a potential preventive and therapeutic target: An update. Med. Res. Rev. 2016, 36, 924–963. [Google Scholar] [CrossRef] [PubMed]
- Sun, H.; Zhu, J.; Lin, H.; Gu, K.; Feng, F. Recent progress in the development of small molecule Nrf2 modulators: A patent review (2012–2016). Expert Opin. Ther. Pat. 2017, 27, 763–785. [Google Scholar] [CrossRef] [PubMed]
- Abed, D.A.; Goldstein, M.; Albanyan, H.; Jin, H.; Hu, L. Discovery of direct inhibitors of Keap1-Nrf2 protein-protein interaction as potential therapeutic and preventive agents. Acta Pharm. Sin. B 2015, 5, 285–299. [Google Scholar] [CrossRef] [PubMed]
- Maher, J.; Yamamoto, M. The rise of antioxidant signaling-The evolution and hormetic actions of Nrf2. Toxicol. Appl. Pharmacol. 2010, 244, 4–15. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.C.; Hsieh, C.W.; Wu, C.C.; Wung, B.S. Chalcone inhibits the activation of NF-κB and STAT3 in endothelial cells via endogenous electrophile. Life Sci. 2007, 80, 1420–1430. [Google Scholar] [CrossRef] [PubMed]
- Martinez, R.M.; Pinho-Ribeiro, F.A.; Vale, D.L.; Steffen, V.S.; Vicentini, F.T.M.C.; Vignoli, J.A.; Baracat, M.M.; Georgetti, S.R.; Verri, W.A.; Casagrande, R. Trans-chalcone added in topical formulation inhibits skin inflammation and oxidative stress in a model of ultraviolet B radiation skin damage in hairless mice. J. Photochem. Photobiol. B Biol. 2017, 171, 139–146. [Google Scholar] [CrossRef] [PubMed]
- Lee, I.S.; Lim, J.; Gal, J.; Kang, J.C.; Kim, H.J.; Kang, B.Y.; Choi, H.J. Anti-inflammatory activity of xanthohumol involves heme oxygenase-1 induction via NRF2-ARE signaling in microglial BV2 cells. Neurochem. Int. 2011, 58, 153–160. [Google Scholar] [CrossRef] [PubMed]
- Yao, J.; Zhang, B.; Ge, C.; Peng, S.; Fang, J. Xanthohumol, a polyphenol chalcone present in hops, activating nrf2 enzymes to confer protection against oxidative damage in pc12 cells. J. Agric. Food Chem. 2015, 63, 1521–1531. [Google Scholar] [CrossRef] [PubMed]
- Brodziak-Jarosz, L.; Fujikawa, Y.; Pastor-Flores, D.; Kasikci, S.; Jirásek, P.; Pitzl, S.; Owen, R.W.; Klika, K.D.; Gerhäuser, C.; Amslinger, S.; et al. A click chemistry approach identifies target proteins of xanthohumol. Mol. Nutr. Food Res. 2016, 60, 737–748. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.S.; Lim, J.; Bang, Y.; Gal, J.; Lee, S.U.; Cho, Y.C.; Yoon, G.; Kang, B.Y.; Cheon, S.H.; Choi, H.J. Licochalcone E activates Nrf2/antioxidant response element signaling pathway in both neuronal and microglial cells: Therapeutic relevance to neurodegenerative disease. J. Nutr. Biochem. 2012, 23, 1314–1323. [Google Scholar] [CrossRef] [PubMed]
- Liu, S.X.; Zhang, Y.; Wang, Y.F.; Li, X.C.; Xiang, M.X.; Bian, C.; Chen, P. Upregulation of heme oxygenase-1 expression by hydroxysafflor yellow A conferring protection from anoxia/reoxygenation-induced apoptosis in H9c2 cardiomyocytes. Int. J. Cardiol. 2012, 160, 95–101. [Google Scholar] [CrossRef] [PubMed]
- Chen, M.; Wang, M.; Yang, Q.; Wang, M.; Wang, Z.; Zhu, Y.; Zhang, Y.; Wang, C.; Jia, Y.; Li, Y.; et al. Antioxidant effects of hydroxysafflor yellow A and acetyl-11-keto-boswellic acid in combination on isoproterenol-induced myocardial injury in rats. Int. J. Mol. Med. 2016, 37, 1501–1510. [Google Scholar] [CrossRef] [PubMed]
- Hu, T.; Wei, G.; Xi, M.; Yan, J.; Wu, X.; Wang, Y.; Zhu, Y.; Wang, C.; Wen, A. Synergistic cardioprotective effects of Danshensu and hydroxysafflor yellow A against myocardial ischemia-reperfusion injury are mediated through the Akt/Nrf2/HO-1 pathway. Int. J. Mol. Med. 2016, 38, 83–94. [Google Scholar] [CrossRef] [PubMed]
- Lee, D.S.; Li, B.; Im, N.K.; Kim, Y.C.; Jeong, G.S. 4,2′,5′-Trihydroxy-4′-methoxychalcone from dalbergia odorifera exhibits anti-inflammatory properties by inducing heme oxygenase-1 in murine macrophages. Int. Immunopharmacol. 2013, 16, 114–121. [Google Scholar] [CrossRef] [PubMed]
- Ajiboye, T.O.; Yakubu, M.T.; Oladiji, A.T. Electrophilic and reactive oxygen species detoxification potentials of chalcone dimers is mediated by redox transcription factor Nrf-2. J. Biochem. Mol. Toxicol. 2014, 28, 11–22. [Google Scholar] [CrossRef] [PubMed]
- Wang, R.; Zhang, C.Y.; Bai, L.P.; Pan, H.D.; Shu, L.M.; Kong, A.N.T.; Leung, E.L.H.; Liu, L.; Li, T. Flavonoids derived from liquorice suppress murine macrophage activation by up-regulating heme oxygenase-1 independent of Nrf2 activation. Int. Immunopharmacol. 2015, 28, 917–924. [Google Scholar] [CrossRef] [PubMed]
- Zeng, J.; Chen, Y.; Ding, R.; Feng, L.; Fu, Z.; Yang, S.; Deng, X.; Xie, Z.; Zheng, S. Isoliquiritigenin alleviates early brain injury after experimental intracerebral hemorrhage via suppressing ROS- and/or NF-ΚB-mediated NLRP3 inflammasome activation by promoting Nrf2 antioxidant pathway. J. Neuroinfl. 2017, 14, 1–19. [Google Scholar] [CrossRef] [PubMed]
- Han, J.Y.; Cho, S.S.; Yang, J.H.; Kim, K.M.; Jang, C.H.; Park, D.E.; Bang, J.S.; Jung, Y.S.; Ki, S.H. The chalcone compound isosalipurposide (ISPP) exerts a cytoprotective effect against oxidative injury via Nrf2 activation. Toxicol. Appl. Pharmacol. 2015, 287, 77–85. [Google Scholar] [CrossRef] [PubMed]
- Ma, Z.; Li, C.; Qiao, Y.; Lu, C.; Li, J.; Song, W.; Sun, J.; Zhai, X.; Niu, J.; Ren, Q.; et al. Safflower yellow B suppresses HepG2 cell injury induced by oxidative stress through the AKT/Nrf2 pathway. Int. J. Mol. Med. 2016, 37, 603–612. [Google Scholar] [CrossRef] [PubMed]
- Pinner, K.D.; Wales, C.T.K.; Gristock, R.A.; Vo, H.T.; So, N.; Jacobs, A.T. Flavokawains A and B from kava (Piper methysticum) activate heat shock and antioxidant responses and protect against hydrogen peroxide-induced cell death in HepG2 hepatocytes. Pharm. Biol. 2016, 54, 1503–1512. [Google Scholar] [CrossRef] [PubMed]
- Martinez, R.M.; Pinho-Ribeiro, F.A.; Steffen, V.S.; Caviglione, C.V.; Pala, D.; Baracat, M.M.; Georgetti, S.R.; Verri, W.A.; Casagrande, R. Topical formulation containing hesperidin methyl chalcone inhibits skin oxidative stress and inflammation induced by ultraviolet B irradiation. Photochem. Photobiol. Sci. 2016, 15, 554–563. [Google Scholar] [CrossRef] [PubMed]
- De Spirt, S.; Eckers, A.; Wehrend, C.; Micoogullari, M.; Sies, H.; Stahl, W.; Steinbrenner, H. Interplay between the chalcone cardamonin and selenium in the biosynthesis of Nrf2-regulated antioxidant enzymes in intestinal Caco-2 cells. Free Radic. Biol. Med. 2016, 91, 164–171. [Google Scholar] [CrossRef] [PubMed]
- Peng, S.; Hou, Y.; Yao, J.; Fang, J. Activation of Nrf2-driven antioxidant enzymes by cardamonin confers neuroprotection of PC12 cells against oxidative damage. Food Funct. 2017, 8, 997–1007. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.; Ka, S.O.; Lee, Y.; Park, B.H.; Bae, E.J. Butein induction of HO-1 by p38 MAPK/Nrf2 pathway in adipocytes attenuates high-fat diet induced adipose hypertrophy in mice. Eur. J. Pharmacol. 2017, 799, 201–210. [Google Scholar] [CrossRef] [PubMed]
- Alcaraz, M.J.; Vicente, A.M.; Araico, A.; Dominguez, J.N.; Terencio, M.C.; Ferrándiz, M.L. Role of nuclear factor-κB and heme oxygenase-1 in the mechanism of action of an anti-inflammatory chalcone derivative in RAW 264.7 cells. Br. J. Pharmacol. 2004, 142, 1191–1199. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.H.; Sohn, D.H.; Jin, X.Y.; Kim, S.W.; Choi, S.C.; Seo, G.S. 2′,4′,6′-Tris(methoxymethoxy) chalcone protects against trinitrobenzene sulfonic acid-induced colitis and blocks tumor necrosis factor-α-induced intestinal epithelial inflammation via heme oxygenase 1-dependent and independent pathways. Biochem. Pharmacol. 2007, 74, 870–880. [Google Scholar] [CrossRef] [PubMed]
- Park, P.H.; Kim, H.S.; Jin, X.Y.; Jin, F.; Hur, J.; Ko, G.; Sohn, D.H. KB-34, a newly synthesized chalcone derivative, inhibits lipopolysaccharide-stimulated nitric oxide production in RAW 264.7 macrophages via heme oxygenase-1 induction and blockade of activator protein-1. Eur. J. Pharmacol. 2009, 606, 215–224. [Google Scholar] [CrossRef] [PubMed]
- Park, P.H.; Kim, H.S.; Hur, J.; Jin, X.Y.; Jin, Y.L.; Sohn, D.H. YL-I-108, a synthetic chalcone derivative, inhibits lipopolysaccharide- stimulated nitric oxide production in RAW 264.7 murine macrophages: Involvement of heme oxygenase-1 induction and blockade of activator protein-1. Arch. Pharm. Res. 2009, 32, 79–89. [Google Scholar] [CrossRef] [PubMed]
- Jin, X.Y.; Lee, S.H.; Park, P.H.; Hur, J.; Kim, S.A.; Kim, H.S.; Sohn, D.H. 2′-Methoxy-4′6′-Bis(Methoxymethoxy)chalcone inhibits nitric oxide production in lipopolysaccharide-stimulated RAW 264.7 macrophages. Basic Clin. Pharmacol. Toxicol. 2010, 106, 454–460. [Google Scholar] [CrossRef] [PubMed]
- Shibuya, A.; Onda, K.; Kawahara, H.; Uchiyama, Y.; Nakayama, H.; Omi, T.; Nagaoka, M.; Matsui, H.; Hirano, T. Sofalcone, a gastric mucosa protective agent, increases vascular endothelial growth factor via the Nrf2-heme-oxygenase-1 dependent pathway in gastric epithelial cells. Biochem. Biophys. Res. Commun. 2010, 398, 581–584. [Google Scholar] [CrossRef] [PubMed]
- Kachadourian, R.; Pugazhenthi, S.; Velmurugan, K.; Backos, D.S.; Franklin, C.C.; McCord, J.M.; Day, B.J. 2′,5′-Dihydroxychalcone-induced glutathione is mediated by oxidative stress and kinase signaling pathways. Free Radic. Biol. Med. 2011, 51, 1146–1154. [Google Scholar] [CrossRef] [PubMed]
- Kumar, V.; Kumar, S.; Hassan, M.; Wu, H.; Thimmulappa, R.K.; Kumar, A.; Sharma, S.K.; Parmar, V.S.; Biswal, S.; Malhotra, S. V Novel chalcone derivatives as potent Nrf2 activators in mice and human lung epithelial cells. J. Med. Chem. 2011, 54, 4147–4159. [Google Scholar] [CrossRef] [PubMed]
- Sussan, T.E.; Gajghate, S.; Chatterjee, S.; Mandke, P.; McCormick, S.; Sudini, K.; Kumar, S.; Breysse, P.N.; Diette, G.B.; Sidhaye, V.K.; et al. Nrf2 reduces allergic asthma in mice through enhanced airway epithelial cytoprotective function. Am. J. Physiol. Cell. Mol. Physiol. 2015, 309, L27–L36. [Google Scholar] [CrossRef] [PubMed]
- Wei, J.; Zhu, H.; Lord, G.; Bhattachayya, M.; Jones, B.M.; Allaway, G.; Biswal, S.S.; Korman, B.; Marangoni, R.G.; Tourtellotte, W.G.; et al. Nrf2 exerts cell-autonomous antifibrotic effects: Compromised function in systemic sclerosis and therapeutic rescue with a novel heterocyclic chalcone derivative. Transl. Res. 2017, 183, 71–86.e1. [Google Scholar] [CrossRef] [PubMed]
- Wu, J.Z.; Cheng, C.C.; Shen, L.L.; Wang, Z.K.; Wu, S.B.; Li, W.L.; Chen, S.H.; Zhou, R.P.; Qiu, P.H. Synthetic chalcones with potent antioxidant ability on H2O2-induced apoptosis in PC12 cells. Int. J. Mol. Sci. 2014, 15, 18525–18539. [Google Scholar] [CrossRef] [PubMed]
- Rücker, H.; Al-Rifai, N.; Rascle, A.; Gottfried, E.; Brodziak-Jarosz, L.; Gerhäuser, C.; Dick, T.P.; Amslinger, S. Enhancing the anti-inflammatory activity of chalcones by tuning the Michael acceptor site. Org. Biomol. Chem. 2015, 13, 3040–3047. [Google Scholar] [CrossRef] [PubMed]
- Jobst, B.; Weigl, J.; Michl, C.; Vivarelli, F.; Pinz, S.; Amslinger, S.; Rascle, A. Inhibition of interleukin-3- and interferon- α-induced JAK/STAT signaling by the synthetic α-X-2′,3,4,4′-tetramethoxychalcones α-Br-TMC and α-CF3-TMC. Biol. Chem. 2016, 397, 1187–1204. [Google Scholar] [CrossRef] [PubMed]
- Lounsbury, N.; Mateo, G.; Jones, B.; Papaiahgari, S.; Thimmulappa, R.K.; Teijaro, C.; Gordon, J.; Korzekwa, K.; Ye, M.; Allaway, G.; et al. Heterocyclic chalcone activators of nuclear factor (erythroid-derived 2)-like 2 (Nrf2) with improved in vivo efficacy. Bioorg. Med. Chem. 2015, 23, 5352–5359. [Google Scholar] [CrossRef] [PubMed]
- Kaufmann, K.B.; Al-Rifai, N.; Ulbrich, F.; Schallner, N.; Rücker, H.; Enzinger, M.; Petkes, H.; Pitzl, S.; Goebel, U.; Amslinger, S. The Cytoprotective Effects of E-α-(4-Methoxyphenyl)-2′,3,4,4′-Tetramethoxychalcone (E-α-p-OMe-C6H4-TMC)—A Novel and Non-Cytotoxic HO-1 Inducer. PLoS ONE 2015, 10, e0142932. [Google Scholar] [CrossRef] [PubMed]
- Kaufmann, K.B.; Gothwal, M.; Schallner, N.; Ulbrich, F.; Rücker, H.; Amslinger, S.; Goebel, U. The anti-inflammatory effects of E-α-(p-methoxyphenyl)-2′,3,4,4′-tetramethoxychalcone are mediated via HO-1 induction. Int. Immunopharmacol. 2016, 35, 99–110. [Google Scholar] [CrossRef] [PubMed]
- Zhong, P.; Wu, L.; Qian, Y.; Fang, Q.; Liang, D.; Wang, J.; Zeng, C.; Wang, Y.; Liang, G. Blockage of ROS and NF-κB-mediated inflammation by a new chalcone L6H9 protects cardiomyocytes from hyperglycemia-induced injuries. Biochim. Biophys. Acta Mol. Basis Dis. 2015, 1852, 1230–1241. [Google Scholar] [CrossRef] [PubMed]
- Rampa, A.; Montanari, S.; Pruccoli, L.; Bartolini, M.; Falchi, F.; Feoli, A.; Cavalli, A.; Belluti, F.; Gobbi, S.; Tarozzi, A.; et al. Chalcone-based carbamates for Alzheimer’s disease treatment. Future Med. Chem. 2017, 9, 749–764. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.A.; Son, H.J.; Choi, J.W.; Kim, J.; Han, S.H.; Shin, N.; Kim, J.H.; Kim, S.J.; Heo, J.Y.; Kim, D.J.; et al. Activation of the Nrf2 signaling pathway and neuroprotection of nigral dopaminergic neurons by a novel synthetic compound KMS99220. Neurochem. Int. 2018, 112, 96–107. [Google Scholar] [CrossRef] [PubMed]
- You, S.; Qian, J.; Sun, C.; Zhang, H.; Ye, S.; Chen, T.; Xu, Z.; Wang, J.; Huang, W.; Liang, G. An Aza resveratrol–chalcone derivative 6b protects mice against diabetic cardiomyopathy by alleviating inflammation and oxidative stress. J. Cell. Mol. Med. 2018, 22, 1931–1943. [Google Scholar] [CrossRef] [PubMed]
- Chen, W.; Ge, X.; Xu, F.; Zhang, Y.; Liu, Z.; Pan, J.; Song, J.; Dai, Y.; Zhou, J.; Feng, J.; et al. Design, synthesis and biological evaluation of paralleled Aza resveratrol-chalcone compounds as potential anti-inflammatory agents for the treatment of acute lung injury. Bioorganic Med. Chem. Lett. 2015, 25, 2998–3004. [Google Scholar] [CrossRef] [PubMed]
- Sihvola, V.; Levonen, A.L. Keap1 as the redox sensor of the antioxidant response. Arch. Biochem. Biophys. 2017, 617, 94–100. [Google Scholar] [CrossRef] [PubMed]
- Schmoll, D.; Engel, C.K.; Glombik, H. The Keap1–Nrf2 protein–protein interaction: A suitable target for small molecules. Drug Discov. Today Technol. 2017, 24, 11–17. [Google Scholar] [CrossRef] [PubMed]


| Compound | Active Concentration Against Nrf2 | Target Disease | Study Model | Activity | Ref. |
|---|---|---|---|---|---|
 (E)-chalcone (trans-chalcone) | 10–50 μM | Atherogenesis | Aortic endothelial cells | Antioxidant and anti-inflammatory | [23] |
| 1% w/w a | UVB skin damage | Hairless mice | Antioxidant and anti-inflammatory | [24] | |
 (E)-3-(4-hydroxyphenyl)-1-(2,4,6-trihydroxy-3-(3-methylbut-2-en-1-yl)phenyl)prop-2-en-1-one (Xanthohumol) | 5 μg/mL | Neuroinflammation | Microglial BV2 cells | Anti-inflammatory | [25] |
| 0.5 μM | Parkinson’s disease | Neuronal PC12 cells | Antioxidant and neuroprotective | [26] | |
 (E)-3-(4-hydroxy-2-methoxy-5-(3-methylbut-3-en-2-yl)phenyl)-1-(4-hydroxyphenyl)prop-2-en-1-one (Licochalcone E) | 5 μM | Parkinson’s disease | Microglial BV2 and neuronal SH-SY5Y cells | Antioxidant and neuroprotective | [27,28] |
 3,4,5-trihydroxy-2-((E)-3-(4-hydroxyphenyl)acryloyl)-4-((2R,3R,4S,5S,6R)-3,4,5-trihydroxy-6-(hydroxymethyl)tetrahydro-2H-pyran-2-yl)-6-((2S,3R,5S,6R)-3,4,5-trihydroxy-6-(hydroxymethyl)-tetrahydro-2H-pyran-2-yl)cyclohexa-2,5-dien-1-one (Safflor yellow A) | 20 μM | Myocardial infarction | Cardiomyocyte H9c2 cells | Cytoprotective | [29] |
| 10 μM 100 mg/kg b | Myocardial infarction | Cardiomyocyte H9C2 cells Rats | Antioxidant and cytoprotective | [30] | |
| 80 μM | Myocardial infarction | Cardiomyocyte H9c2 cells | Cytoprotective | [31] | |
 (E)-1-(2,5-dihydroxy-4-methoxyphenyl)-3-(4-hydroxyphenyl)prop-2-en-1-one | 40 μM | Inflammatory disease | Peritoneal macrophages | Anti-inflammatory | [32] |
 (2S,3R)-3-(5-((E)-3-(2,4-dihydroxyphenyl)-3-oxoprop-1-en-1-yl)-2-hydroxyphenyl)-6-hydroxy-2-(4-hydroxyphenyl)chroman-4-one (Lophirones B)  (E)-3-((3S)-3-(2,4-dihydroxybenzoyl)-2-(4-hydroxyphenyl)-2,3-dihydrobenzofuran-5-yl)-1-(2,4-dihydroxyphenyl)prop-2-en-1-one (Lophirones C) | 20 mg/Kg b | Liver diseases | Rats | Antioxidant and detoxification | [33] |
 (E)-1-(2,4-dihydroxyphenyl)-3-(4-(((2S,3R,4S,5S,6R)-3,4,5-trihydroxy-6-(hydroxymethyl)tetrahydro-2H-pyran-2-yl)oxy)phenyl)prop-2-en-1-one (Isoliquiritin)  (E)-1-(2,4-dihydroxyphenyl)-3-(4-hydroxyphenyl)prop-2-en-1-one (Isoliquiritigenin) | 25–50 μM | Inflammatory disease | Macrophage RAW 264.7 and Hepatic HepG2-C8 cells | Anti-inflammatory | [34] |
| 20 mg/Kg c | Intracerebral hemorrhage | Rats | Antioxidant and anti-inflammatory | [35] | |
 (E)-1-(2,4-dihydroxy-6-(((2S,3R,5S,6R)-3,4,5-trihydroxy-6-(hydroxymethyl)tetrahydro-2H-pyran-2-yl)oxy)phenyl)-3-(4-hydroxyphenyl)prop-2-en-1-one (isosalipurposide) | 10–100 μM | Oxidative stress | Hepatic HepG2 cells | Antioxidant and cytoprotective | [36] |
 (Z)-4-(6,7-dihydroxy-5-((E)-3-(4-hydroxyphenyl)acryloyl)-4-oxo-2-((1S,2R,3R)-1,2,3,4-tetrahydroxybutyl)-7-((2R,3R,4S,5S,6R)-3,4,5-trihydroxy-6-(hydroxymethyl)tetrahydro-2H-pyran-2-yl)-2,3,4,7-tetrahydrobenzofuran-3-yl)-5,6-dihydroxy-2-((E)-1-hydroxy-3-(4-hydroxyphenyl)allylidene)-6-((2R,3R,4S,5S,6R)-3,4,5-trihydroxy-6-(hydroxymethyl)tetrahydro-2H-pyran-2-yl)cyclohex-4-ene-1,3-dione (Safflor yellow B) | 100–150 nM | Oxidative stress | Hepatic HepG2 cells | Antioxidant | [37] |
 (E)-1-(2-hydroxy-4,6-dimethoxyphenyl)-3-(4-methoxyphenyl)prop-2-en-1-one (Flavokawains A)  (E)-1-(2-hydroxy-4,6-dimethoxyphenyl)-3-phenylprop-2-en-1-one (Flavokawains B) | 20–100 μM | Oxidative stress | Hepatic HepG2 cells | Antioxidant and cytoprotective | [38] |
| 10–20 μM | |||||
 (E)-3-(3-hydroxy-4-methoxyphenyl)-1-(2-hydroxy-6-methoxy-4-(((2R,3R,4S,5S,6R)-3,4,5-trihydroxy-6-((((2R,3R,4R,5R,6S)-3,4,5-trihydroxy-6-methyltetrahydro-2H-pyran-2-yl)oxy)methyl)tetrahydro-2H-pyran-2-yl)oxy)phenyl)prop-2-en-1-one Hesperidin methyl chalcone (HMC) | 1% w/w a | UVB skin damage | Hairless mice | Antioxidant and cytoprotective | [39] |
 (E)-1-(2,4-dihydroxy-6-methoxyphenyl)-3-phenylprop-2-en-1-one (Cardamonin) | 50 μM d | Oxidative stress | Intestinal Caco-2 cells | Antioxidant | [40] |
| 10 μM | Parkinson’s disease | Neuronal PC12 cells | Neuroprotective | [41] | |
 (E)-1-(2,4-dihydroxyphenyl)-3-(3,4-dihydroxyphenyl)prop-2-en-1-one (Butein) | 30 μM | Obesity and Type 2 diabetes | Adipocyte 3T3-L1 cells | Antioxidant | [42] |
| Compound | Active Concentration Against Nrf2 | Target Disease | Study Model | Activity | Ref. |
|---|---|---|---|---|---|
 (E)-1,3-bis(3,4,5-trimethoxyphenyl)prop-2-en-1-one | 30 μM | Inflammatory disease | Macrophage RAW 264.7 cells | Anti-inflammatory | [43] |
 (E)-1-phenyl-3-(2,4,6-tris(methoxymethoxy)phenyl)prop-2-en-1-one | 20 μM | Inflammatory intestinal disease | Intestinal HT-29 cells | Anti-inflammatory | [44] |
 1-phenyl-3-(2,4,6-tris(methoxymethoxy)phenyl)prop-2-yn-1-one | 2 μM | Inflammatory disease | Macrophage RAW 264.7 cells | Anti-inflammatory | [45] |
 (E)-3-(2-methoxy-4,6-bis(methoxymethoxy)phenyl)-1-(3-nitrophenyl)prop-2-en-1-one | 2 μM | Inflammatory disease | Macrophage RAW 264.7 cells | Anti-inflammatory | [46] |
 (E)-3-(2-methoxy-4,6-bis(methoxymethoxy)phenyl)-1-phenylprop-2-en-1-one (MBMC) | 2 μM | Inflammatory disease | Macrophage RAW 264.7 cells | Anti-inflammatory | [47] |
 (E)-2-(5-((2-methylprop-1-en-1-yl)oxy)-2-(3-(4-((2-methylprop-1-en-1-yl)oxy)phenyl)acryloyl)phenoxy)-acetic acid (Sofalcone) | 20–50 μM | Gastric ulcer | Gastric epithelial RGM-1 cells | Antiulcer | [48] |
 (E)-1-(2,5-dihydroxyphenyl)-3-phenylprop-2-en-1-one | 10–20 μM | Oxidative stress | MCF-7/AREc32 | Antioxidant | [49] |
 (E)-1-(2-methoxyphenyl)-3-(2-(trifluoromethyl)phenyl)prop-2-en-1-one | 10–20 μM a 50 mg/Kg a,b | Inflammatory disease | Bronchial epithelial Beas-2B cells Mice | Anti-inflammatory | [50] |
| 400 mg/Kg b | Allergic asthma | Mice | Antioxidant and anti-inflammatory | [51] | |
| 5 μM 400 mg/Kg b | Systemic sclerosis | Fibroblasts Mice | Antioxidant | [52] | |
 (E)-2-methoxy-4-(3-(4-methoxyphenyl)-3-oxoprop-1-en-1-yl)phenyl acrylate | 10 μM a | Oxidative stress | Neuronal PC12 cells | Antioxidant | [53] |
 (E)-1-(2,4-dimethoxyphenyl)-3-(3,4-dimethoxyphenyl)-2-(trifluoromethyl)prop-2-en-1-one  (Z)-2-bromo-1-(2,4-dimethoxyphenyl)-3-(3,4-dimethoxyphenyl)prop-2-en-1-one  (Z)-2-chloro-1-(2,4-dimethoxyphenyl)-3-(3,4-dimethoxyphenyl)prop-2-en-1-one | 3.13 μM | Inflammatory disease | MCF-7/AREc32 cells | Anti-inflammatory | [54,55] |
| 6.25 μM | |||||
| 6.25 μM | |||||
 (E)-1-(2-methoxyphenyl)-3-(3-(trifluoromethyl)pyridin-2-yl)prop-2-en-1-one | 5 μM a 400 mg/Kg a,b | Oxidative stress | Bronchial epithelial Beas-2B cells Mice | Antioxidant | [56] |
 (E)-1-(2,4-dimethoxyphenyl)-3-(3,4-dimethoxyphenyl)-2-(4-methoxyphenyl)prop-2-en-1-one | 30 μM | Inflammatory disease | Macrophage RAW264.7 cells | Antioxidant, cytoprotective and anti-inflammatory | [57,58] |
 (E)-1-(2,6-difluorophenyl)-3-(4-methoxyphenyl)prop-2-en-1-one | 10 μM 20 mg/kg b | Diabetic cardiomyopathy | Cardiomyocyte H9c2 cells Mice | Anti-inflammatory and cytoprotective | [59] |
 (E)-1-(2-hydroxy-4-(3-((3-hydroxybenzyl)(methyl)amino)propoxy)phenyl)-3-(3,4,5-trimethoxyphenyl)prop-2-en-1-one | 10 μM a | Alzheimer’s disease | Neuronal SH-SY5Y cells | Neuroprotective activity | [60] |
 (E)-3-(4-(3-morpholinopropoxy)-phenyl)-1-(2-(trifluoromethyl)phenyl)-prop-2-en-1-one hydrochloride | 3 μM 30 mg/kg a | Parkinson’s disease | Dopaminergic CATH.a cells Mice | Neuroprotective | [61] |
 (E)-3-(3-(((E)-2,3-dihydroxybenzylidene)amino)-4-methoxyphenyl)-1-(3,4,5-trimethoxyphenyl)prop-2-en-1-one | 5–10 μM 5–20 mg/kg b | Diabetic cardiomyopathy | Cardiomyocyte H9c2 cells Mice | Antioxidant and anti-inflammatory | [62] |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
De Freitas Silva, M.; Pruccoli, L.; Morroni, F.; Sita, G.; Seghetti, F.; Viegas Jr, C.; Tarozzi, A. The Keap1/Nrf2-ARE Pathway as a Pharmacological Target for Chalcones. Molecules 2018, 23, 1803. https://doi.org/10.3390/molecules23071803
De Freitas Silva M, Pruccoli L, Morroni F, Sita G, Seghetti F, Viegas Jr C, Tarozzi A. The Keap1/Nrf2-ARE Pathway as a Pharmacological Target for Chalcones. Molecules. 2018; 23(7):1803. https://doi.org/10.3390/molecules23071803
Chicago/Turabian StyleDe Freitas Silva, Matheus, Letizia Pruccoli, Fabiana Morroni, Giulia Sita, Francesca Seghetti, Claudio Viegas Jr, and Andrea Tarozzi. 2018. "The Keap1/Nrf2-ARE Pathway as a Pharmacological Target for Chalcones" Molecules 23, no. 7: 1803. https://doi.org/10.3390/molecules23071803
APA StyleDe Freitas Silva, M., Pruccoli, L., Morroni, F., Sita, G., Seghetti, F., Viegas Jr, C., & Tarozzi, A. (2018). The Keap1/Nrf2-ARE Pathway as a Pharmacological Target for Chalcones. Molecules, 23(7), 1803. https://doi.org/10.3390/molecules23071803








