Abstract
This review covers the synthesis of coumarin–porphyrin, coumarin–phthalocyanine and coumarin–corrole conjugates and their potential applications. While coumarin–phthalocyanine conjugates were obtained almost exclusively by tetramerization of coumarin-functionalized phthalonitriles, coumarin–porphyrin and coumarin–corrole conjugates were prepared by complementary approaches: (a) direct synthesis of the tetrapyrrolic macrocycle using formylcoumarins and pyrrole or (b) by functionalization of the tetrapyrrolic macrocycle. In the last approach a range of reaction types were used, namely 1,3-dipolar cycloadditions, hetero-Diels–Alder, Sonogashira, alkylation or acylation reactions. This is clearly a more versatile approach, leading to a larger diversity of conjugates and allowing the access to conjugates bearing one to up to 16 coumarin units.
1. Introduction
The coumarins are an important class of heterocyclic compounds with diverse pharmacological activities [1] and outstanding optical properties. Among their varied pharmacological activities, the anti-inflammatory [2], antibacterial [3,4], antiviral [5,6], and anti-cancer [7,8,9,10] properties can be highlighted. Due to their excellent optical properties, coumarin derivatives have been used in a variety of applications such as optical brighteners [11,12], optical sensors [13], organic light emitting diodes [14,15,16,17], laser dyes [18], photonic bandgap materials [19], light harvesting materials [20,21,22], and as fluorescent labels and probes in biology and medicine [23,24,25,26,27,28,29,30].
Due to their exceptional optical properties, coumarins are the focus of an intense research effort in various scientific areas. In particular, in recent years, a large number of coumarin–tetrapyrrolic macrocycle conjugates have been synthesized and their photophysical properties evaluated. Typically, the main objective of those studies is to take advantage from the efficient energy transfer between the coumarin and the tetrapyrrole unit, aiming for light harvesting applications. In this article, we review primarily the synthetic approaches leading to coumarin–porphyrin, coumarin–phthalocyanine, and coumarin–corrole conjugates. A range of other coumarin–chromophore conjugates, such as coumarin–bodipy [18,31], coumarin–fullerene [32,33,34], coumarin–perylene [35,36,37], etc. have also been reported and exhibit highly interesting photophysical properties. However, those systems are not covered in this review.
2. Coumarin–Tetrapyrrolic Macrocycle Conjugates
2.1. Coumarin–Porphyrin Dyads
The number of porphyrin derivatives bearing coumarin units at β-pyrrolic positions are scarce. Examples of such type of compounds were reported in 2011 by Cavaleiro and co-workers [38,39]. The coumarin–porphyrin dyads 4a,b were synthesized via hetero-Diels–Alder reaction of β-vinyl-meso-tetraphenylporphyrinatozinc(II) (1) [40] with the ortho-quinone methides 3a,b generated in situ from the reaction of 4-hydroxycoumarins 2a,b with paraformaldehyde (Scheme 1). The reactions were performed in refluxing 1,4-dioxane for one hour and the resulting dyads were isolated in 88% (4a) and 95% (4b) yield, respectively. The site and regioselectivity of these reactions are in agreement with those observed in similar systems. The sensing ability of these Zn(II) conjugates towards anions was evaluated by UV-vis and fluorescence measurements. Alterations in both absorption and emission spectra, or only in the emission spectra, were detected in the presence of Cl−, CN− and CH3CO2− [39]. The sensing ability of the free-base conjugates 4c and 4d, obtained in excellent yields by demetalation of the corresponding Zn(II) complexes with a mixture of TFA/CHCl3, was also evaluated, but in the presence of several metal ions [39]. Both dyads showed a colorimetric effect (a color change from purple to yellow) and an unprecedented selectivity for Hg(II), even in an EtOH/H2O mixture. The same colorimetric effect for Hg(II) was observed when dyad 4c was incorporated in a cellulose support material. The same dyad, both in solution and in the solid support, also showed a colorimetric effect at different pH values.
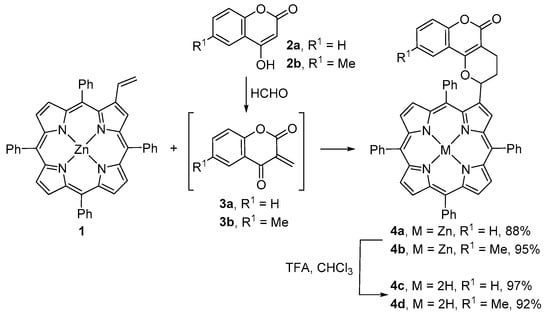
Scheme 1.
Route to coumarin–porphyrin dyads [38,39].
The coumarin–porphyrin dyad 4a showed to be a potential probe towards the alkaloids caffeine, nicotine and cotinine, with a stoichiometry of one alkaloid per ligand [41]. Additionally, it was evidenced that this probe can detect small amounts of cotinine (2.5 ± 0.3 μM) in dam water samples.
Conjugates 4a–4d, and the corresponding Cu2+ and Ni2+ complexes, were studied by electrospray mass spectrometry (ESI-MS) and tandem mass spectrometry (MS/MS) [42]. It was observed that the main fragmentation mechanism occurs via a retro hetero-Diels–Alder pathway.
The extension of the domino Knoevenagel/hetero-Diels–Alder approach to the zinc(II) complexes of chlorin e6 trimethyl ester and protoporphyrin IX dimethyl ester and the ortho-quinone methides 3a,b afforded the conjugates Zn5a,b and Zn6a,b in excellent yields (81–91%) (Figure 1) [43]. These complexes were then demetalated to the corresponding free-bases 5a,b and 6a,b. These conjugates were isolated as mixtures of diastereomers and their relative abundances were determined by advanced NMR techniques. The evaluation of their photophysical and electrochemical properties showed that the fluorescence quantum yields of the chlorin e6 derivatives are higher than those of the protoporphyrin conjugates. All conjugates were able to generate singlet oxygen but protoporphyrin IX dyads gave the highest singlet oxygen quantum yields. The free-base chlorin e6 dyads showed an unexpectedly higher ability to generate singlet oxygen when compared with the Zn(II) counterparts due to the higher tendency of the complexes to aggregate. The same group reported the synthesis of six coumarin–porphyrin dyads 8a–f from porphyrin 1 and the ortho-quinone methides 7a–f generated in situ from 4-hydroxycoumarin and suitable aromatic aldehydes.
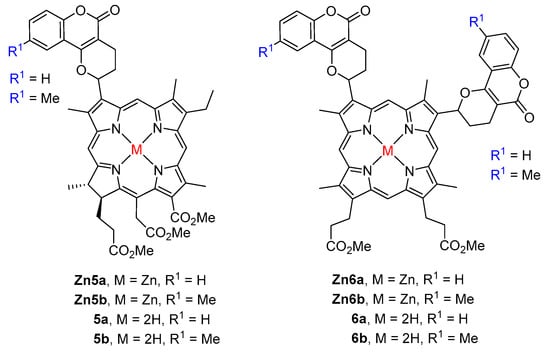
Figure 1.
Structures of coumarin conjugates obtained from natural porphyrins [43].
The reactions were carried out under three different conditions, including in water under ohmic heating (Scheme 2) [44]. Comparing the results obtained under the various experimental conditions, the authors concluded that the use of ohmic heating leads to a reduction of the reaction time, higher yields and selectivities. Moreover, the use of water as solvent facilitates the workup and product isolation over traditional organic solvents.
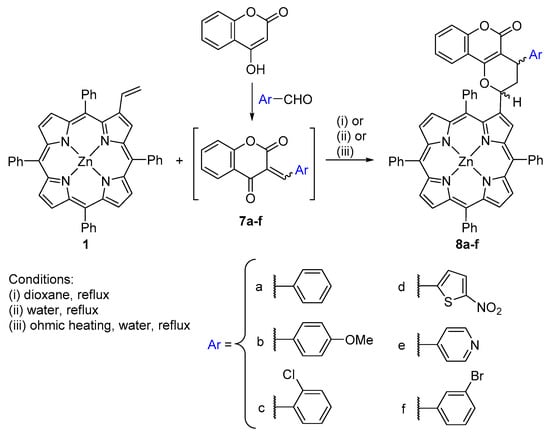
Scheme 2.
Synthesis of aryl-substituted coumarin–porphyrin dyads [44].
In 2015, Singh and Nath reported the synthesis of β-triazole bridged coumarin–porphyrin conjugates in 84–92% yield by the Cu(I)-catalyzed click reaction between 2-azido-5,10,15,20-tetraphenylporphyrinatocopper(II) (9) [45] with various alkyne-substituted coumarins 10–12 (Scheme 3) [46]. The authors also prepared the corresponding free-bases in good yield (71–80%) by demetalation of the copper derivatives. Metalation of the free-bases with zinc(II) acetate afforded the corresponding zinc(II) complexes in 91–96% yield. The photophysical characterization of these conjugates revealed, for some of them, a considerable electronic communication between both units. Additionally, in some of these dyads it was observed a significant intramolecular energy transfer between both chromophores.
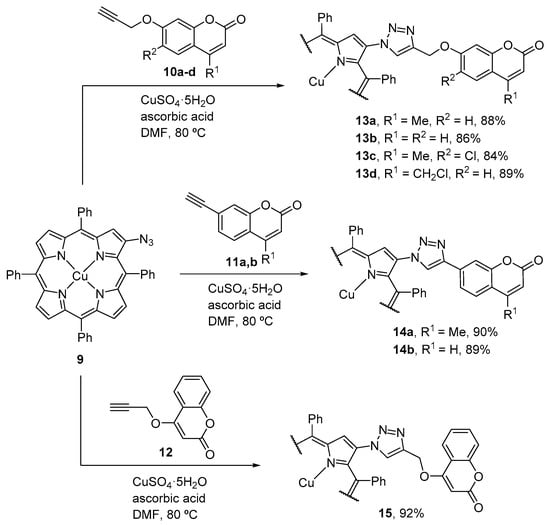
Scheme 3.
Synthesis of β-triazole bridged coumarin–Cu(II)porphyrin conjugates [46].
Using a similar strategy, the same authors also described the synthesis of zinc(II) β-triazolylmethyl-bridged coumarin–porphyrin dyads 17–19 (Scheme 4) [47]. In these conjugates the porphyrin and the triazole units are separated by a methylene spacer. Their synthesis involved the 1,3-dipolar cycloaddition between the 2-azidomethyl-5,10,15,20-tetraphenylporphyrinatozinc(II) (16) and the alkyne-substituted coumarins 10–12. The reported yields for the dyads are in the range 84–92%. Demetalation of the zinc(II) complexes afforded the corresponding dyads with free-base porphyrins in 69–79% yield. Again, the photophysical characterization of the new compounds allowed verifying an intramolecular energy transfer between both units for some of the conjugates.
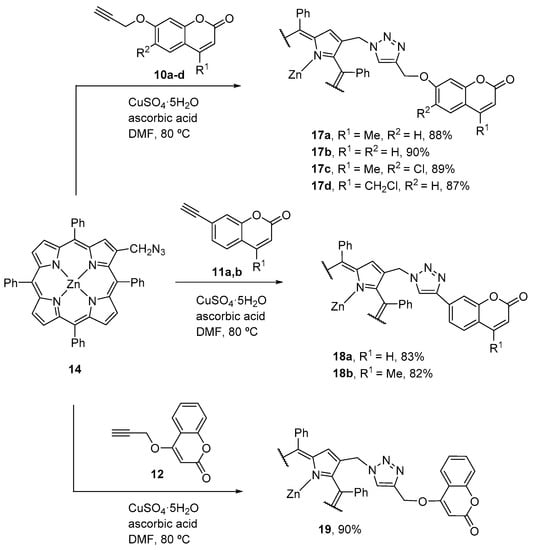
Scheme 4.
Synthesis of zinc(II) β-triazolylmethyl-bridged coumarin–porphyrin dyads [47].
The same alkyne-substituted coumarins 10–12 were used to prepare the zinc(II) meso-phenyl-triazole bridged coumarin–porphyrin dyads 21–23 through copper(I)-catalysed 1,3-dipolar cycloaddition reaction of zinc(II) 5-(4-azidophenyl)-10,15,20-triphenylporphyrin (20) (Scheme 5) [48]. The corresponding free-bases were successfully obtained after treatment with concentrated hydrochloric acid. The free-base dyads were converted into the nickel(II) complexes by treatment with nickel(II) acetate in chloroform–acetic acid. Preliminary photophysical results revealed a significant intramolecular energy transfer between both units for some of the zinc(II) conjugates.
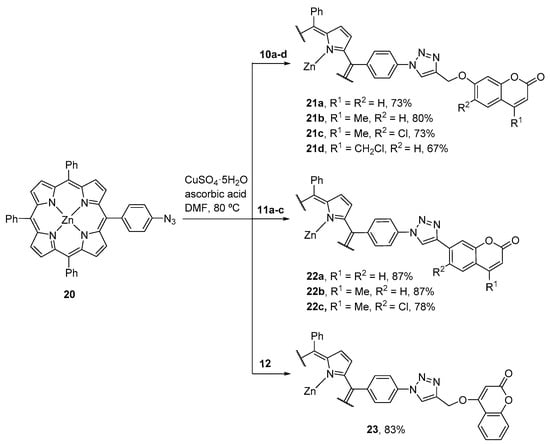
Scheme 5.
Synthesis of zinc (II) meso-phenyl-triazole bridged coumarin–porphyrin dyads [48].
Lin et al. reported the synthesis of symmetric and asymmetric porphyrins bearing coumarin units at the meso positions (Figure 2) [49]. The mono- and meso-tetrakis(coumarin-4-yl)porphyrins 25–27 were obtained in yields between ca. 5% and 13% from the reaction of pyrrole and an adequate molar ratio of benzaldehyde/coumarin-4-carbaldehydes 24a–c. Depending on the coumarin−carbaldehyde used, the porphyrins where synthesized under Adler (refluxing propionic acid) [50] or Lindsey (BF3·OEt2 or TFA, CHCl3, room temperature, then p-chloranil) [51] conditions. The symmetrical meso-tetrakis(coumarin-4-yl)porphyrins 26b and 27b, isolated in 5.3% and 2.7% yield, respectively, were obtained from the reaction between pyrrole and coumarin−carbaldehydes 24b and 24c under Lindsey conditions. The absorption and photoluminescent spectra of these dyads in dilute THF solutions and as solid films (obtained by spin-coating the derivatives in quartz plates) demonstrated that the energy transfer from the coumarin substituents to the porphyrin core is more efficient in solid film than in solution. The best results were obtained with the dyads 27a,b bearing the 7-diethylaminocoumarin moiety. This efficiency was justified by the high electron-donating ability of the amino substituent and also considering that in the solid state, the stacking porphyrins reduce the torsion angle between the porphyrin core and the coumarin substituent, forcing them to be coplanar and thus enhancing the conjugation extent of the porphyrin core and the coumarin substituent, facilitating the energy transfer from the coumarin units to the porphyrin core.
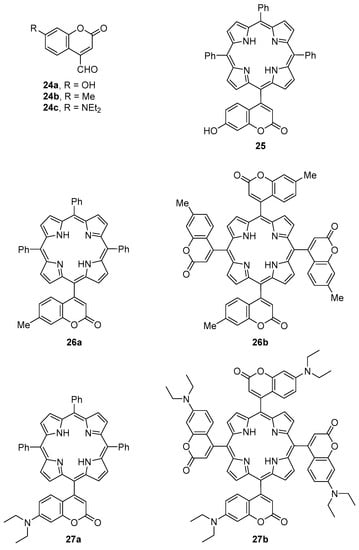
Figure 2.
meso-(Coumarin-4-yl)porphyrins reported by Lin et al. [49].
The Lindsey experimental conditions were also used for the condensation of coumarin-3-carbaldehydes 28a–d with pyrrole (Scheme 6) [52]. The resulting meso-tetrakis(4-chlorocoumarin-3-yl)porphyrins 29a–d were obtained in ca. 20% yield as mixtures of four atropisomers. The authors were able to determine the ratio of the atropisomers by high-performance thin-layer chromatography (HPTLC) (UV-detector). The metalation of the free-bases with zinc(II) acetate in CHCl3–MeOH at room temperature afforded the corresponding zinc(II) complexes Zn29a–d in 80–86% yield.
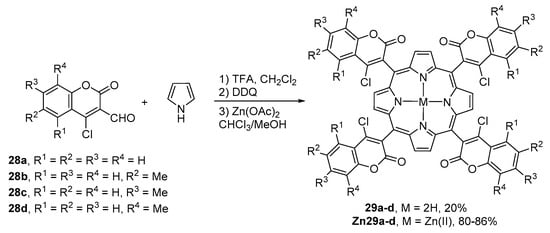
Scheme 6.
Route to meso-tetrakis(4-chlorocoumarin-3-yl)porphyrins reported by Amaravathi et al. [52].
Fréchet and co-workers reported the synthesis of branched star polymers consisting of a central porphyrin core and 16 coumarin units (Scheme 7) [53,54,55]. The compounds were prepared via esterification of meso-tetrakis(3,5-dihydroxyphenyl)porphyrin (30) with acetonide-protected 2,2-bis(hydroxymethyl)propanoic acid followed by deprotection of the diol functionalities under acidic conditions. The reaction of the resulting hexadecahydroxy-functionalized porphyrin 31 with coumarin-3-carboxylic acid chloride gave the hexadecacoumarin-functionalized porphyrin 32.
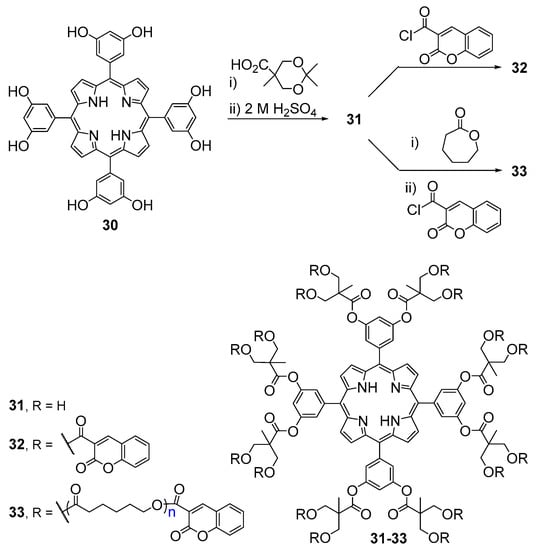
Scheme 7.
Route to coumarin–porphyrin conjugates reported by Fréchet and co-workers [53,54].
Bulk polymerization of ε-caprolactone with the initiator 31, with tin(II) 2-ethylhexanoate as the catalyst, followed by esterification of the resulting polymer with coumarin-3-carboxylic acid chloride gave 33 in quantitative yield. Polymers 33 with varying chain lengths were prepared in almost quantitative yields by adjusting the monomer-to-initiator ratio. The 16 coumarin units in compounds 32 and 33 are responsible for the large absorption in the UV region of the spectrum. It was found that the presence of coumarin donor chromophores in these systems was particularly useful to evaluate the isolation of the core functionalities by using fluorescence resonance energy transfer (FRET). When the coumarin units were excited selectively at λ = 350 nm, the emission from both the coumarin donor and the porphyrin acceptor was observed demonstrating that in these systems FRET was facile but not quantitative. It was verified that as the chain length of the polymer increases the donor emission intensity increases as the result of the reduced probability of FRET. In compound 32, due to the extremely short average donor–acceptor distance, a quantitative FRET was observed. Besides the chain length, it was verified that poor solvents seem to further increase the degree of site isolation due to a structural collapse of the polymer backbone giving rise to a more densely packed structure around the core unit and a reduced average donor–acceptor distance. This observation was supported by pulsed field gradient spin–echo (PGSE) NMR experiments that allowed the direct determination of the polymers molecular sizes in different solvents.
Mao and Song also used meso-tetrakis(3,5-dihydroxyphenyl)porphyrin (30) as a platform to synthesize three porphyrin-core dendrimers bearing eight coumarin units (Scheme 8) [56]. Dendrimer 36 was prepared in 85% yield by reacting porphyrin 30 with the coumarin derivative 34. Dendrimers 37a and 37b were synthesized in 73–75% yield by coupling porphyrin 30 with the carboxylic acid terminated coumarins 35a,b in the presence of DCC (N,N′-dicyclohexylcarbodiimide) and DPTS (4-(dimethylamino)pyridinium p-toluenesulfonate). The study of the photophysical properties of the dendrimers in CH2Cl2 solutions and in thin neat films revealed an intramolecular energy transfer from the coumarin units to the porphyrin core. The best energy-transfer efficiencies were found for the dendrimers 37a and 37b due to a better spectral overlap between the emission spectrum of the coumarin units and the absorption spectrum of the porphyrin moiety than in dendrimer 36. Comparing the optical properties of both dendrimers 37, it was found that the energy-transfer efficiency is better for 37b than 37a in both solid film and solution, probably due to the presence of the longer alkyl side-chain which improves its solubility and consequently prevents the coumarins from self-quenching. All dendrimers emit red light with higher fluorescence quantum yields than the free-base porphyrin. It was remarked that these systems could be applied as light-harvesting antenna.
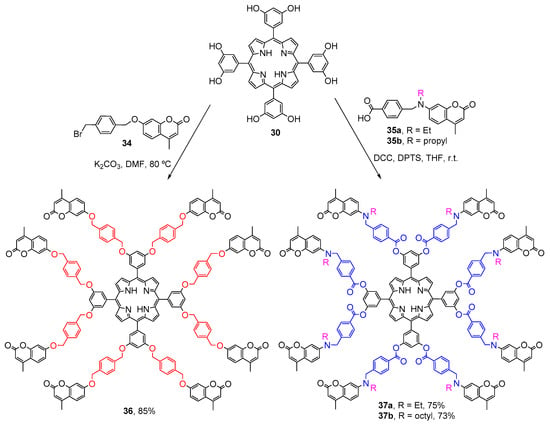
Scheme 8.
Synthesis of three porphyrin-core dendrimers with eight coumarin units [56].
Marcos and co-workers reported a new class of porphyrin-core dendrimers bearing 12 coumarin units via acylation of meso-tetrakis(4-hydroxyphenyl)porphyrin 38 with the coumarin-substituted benzoic acid derivative 39 (Scheme 9) [57]. The resulting compound 40 was obtained in 55% yield. Metalation of the free-base dendrimer 40 with zinc(II) acetate or with copper(II) acetate in CHCl3–MeOH afforded the corresponding zinc(II) or copper(II) complexes. All dendrimers formed nematic discotic mesophases and the charge mobility values of these materials are the highest ever reported for a nematic discotic phase. Excitation of the coumarin moieties (λexc = 320 nm) leaded to energy transfer to the porphyrin core. However, emission from both the coumarin units and the porphyrin acceptor was observed, thus demonstrating that FRET was facile but not quantitative in these systems [57].
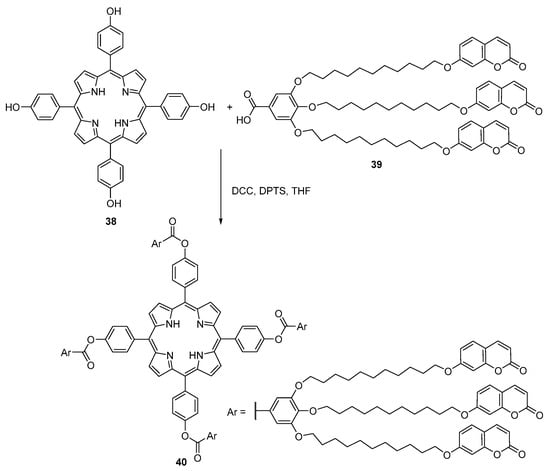
Scheme 9.
Synthesis of porphyrin-core dendrimers bearing 12 coumarin units [57].
Lin and co-workers also used meso-tetrakis(4-hydroxyphenyl)porphyrin (38) as a platform to synthesize a coumarin–porphyrin dyad (Scheme 10) [58]. This platform was alkylated with ethyl bromoacetate and the carboxylic acid 42 was obtained after hydrolysis of the ester group. The new dyad 43 was then prepared in 68% yield by coupling the amine-functionalized coumarin 41 with the carboxylic acid 42 in the presence of benzotriazol-1-yloxytris(dimethylamino)phosphonium hexafluorophosphate (BOP) and triethylamine. The dyad is highly selective and sensitive to thiols, exhibiting a remarkable change in emission colour from red to blue, being suitable for ratiometric fluorescence imaging of thiols in living cells.
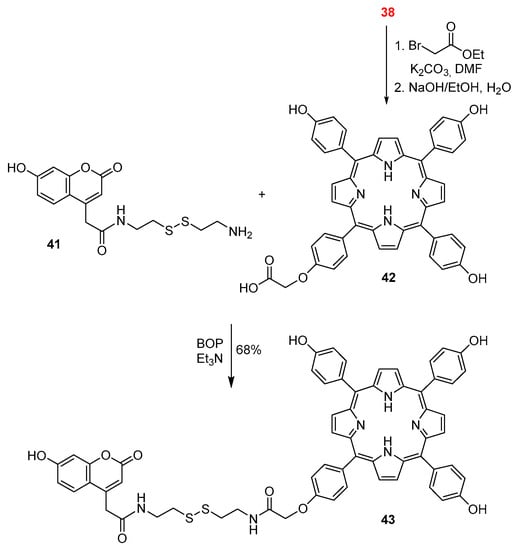
Scheme 10.
A coumarin–porphyrin dyad reported by Weiying Lin and co-workers [58].
A G3 dendrimeric coumarin-porphyrin conjugate consisting of a central Pt meso-tetrakis-(4-alkoxyphenyl)porphyrin linked to several coumarin-343 units was prepared and used as a probe for cellular two-photon oxygen imaging [59]. Knoester and co-workers reported the synthesis of a first-generation coumarin–porphyrin dendrimer where the porphyrin core is a derivative of meso-tetrakis(4-carboxyphenyl)porphyrin (Scheme 11) [60]. For the synthesis of this donor–acceptor system, meso-tetrakis(4-carboxyphenyl)porphyrin was first coupled to piperazine, using pivaloyl chloride as a coupling reagent, to form the piperazine-functionalized porphyrin 44. The coumarin units were subsequently coupled to this porphyrin using (benzotriazol-1-yloxy)tripyrrolidin-1-yl)phosphonium hexafluorophosphate (PyBOP, a peptide coupling reagent), to give compound 45. The energy transfer kinetics (from coumarin to porphyrin) was shown to be fast (transfer time ca. 500 fs) and efficient (transfer efficiency ca. 97%).
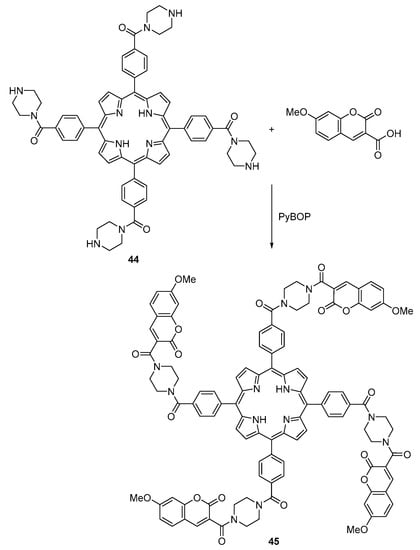
Scheme 11.
Synthesis of a donor–acceptor system [60].
Marcos and co-workers reported the formation of supramolecular system based on hydrogen-bonding between a porphyrin core and carboxylic acid dendrons functionalized with coumarin units (Scheme 12) [61]. The supramolecular porphyrin–coumarin dendrimers were prepared by mixing a dichloromethane solution of meso-tetra(4-pyridyl)porphyrin (46), or its Zn(II) complex (Zn46), and four equivalents of the carboxylic acid 47. The slow evaporation of the solvent by stirring at room temperature afforded the dendrimers 48 and Zn48. Their stoichiometry in the condensed phase is 1:4, as corroborated by NMR spectroscopy. These supramolecular complexes do not show liquid crystalline behaviour and their absorption and emission spectra are a combination of the spectra of the corresponding building blocks. The lack of energy transfer between the two chromophores is probably due to the long distance between them.
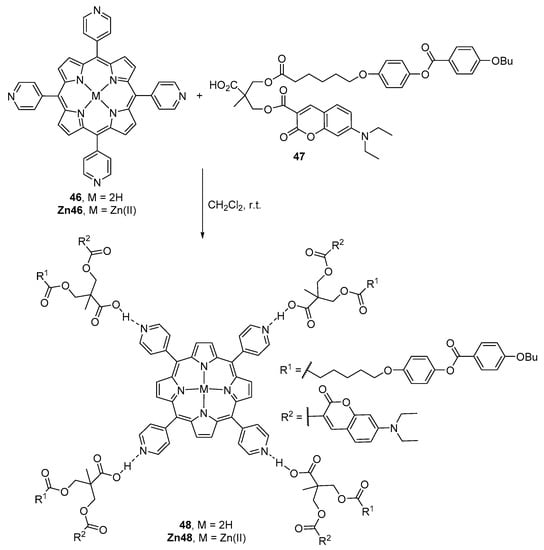
Scheme 12.
Synthesis of supramolecular porphyrin dendrimers with coumarin units [61].
Gust and co-workers reported the synthesis of a molecular hexad 50 comprising two porphyrin moieties and four coumarin antenna chromophores, all organized by a central hexaphenylbenzene core (Scheme 13) [62]. The metalated hexad self-assembles with a pyridyl-bearing fullerene moiety, through coordination with the zinc atoms of the porphyrins, to yield the artificial photosynthetic reaction center 51.
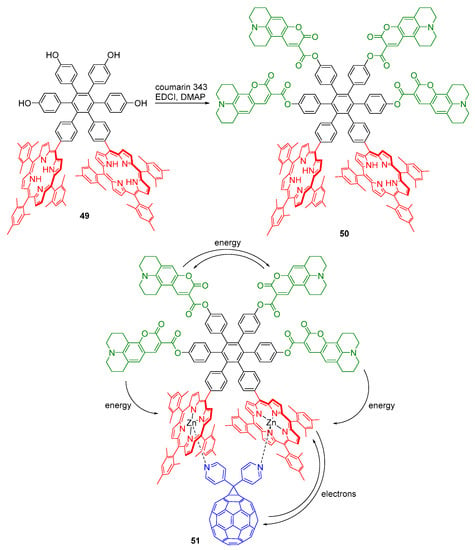
Scheme 13.
Synthesis of an artificial photosynthetic reaction center [62].
It was shown that light absorbed by any of the coumarins in hexad 50 is transferred to a porphyrin on the 1–10 ps time scale, depending on the site of initial excitation. The quantum yield of singlet energy transfer is 1.0. In heptad 51, energy transfer to the porphyrins is followed by photoinduced electron transfer to the fullerene, resulting in a charge-separated state (P•+–C60•−) with an overall quantum yield of 1.0 [62].
Compound 50 was prepared in 90% yield by coupling compound 49 with coumarin 343 using 1-ethyl-3-(3-dimethylaminopropyl)carbodiimide (EDCI) and 4-dimethylaminopyridine (DMAP). The synthesis of other compounds of type 50, with one or two coumarin units (52–54, Figure 3), was also reported [62].
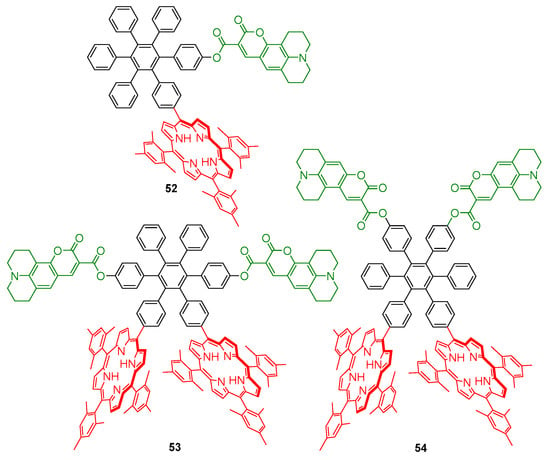
Figure 3.
Model compounds of artificial photosynthetic reaction centers [62].
These studies allowed to conclude that in spite of the relatively long, flexible ester linkages between the coumarins and the hexaphenylbenzene ring, coumarin moieties are well suited as antennas in the 400−460 nm spectral range for porphyrin based artificial photosynthetic reaction centers. The high transfer rates, and therefore efficiencies, are ensured by coumarin singlet excited state lifetimes of several ns and high fluorescence quantum yields.
2.2. Coumarin–Phthalocyanine Dyads
Several coumarin–phthalocyanine conjugates have been reported in the last years. Many of those conjugates were synthesized from substituted 7-hydroxycoumarins 55 following the synthetic route shown in Scheme 14. Typically the first step involves the formation of a coumarin-substituted phthalonitrile, usually by nucleophilic displacement of a nitro group in 3- or 4-nitrophthalonitrile, and the resulting phthalonitriles 56 are then converted into metallophthalocyanines 57 by cyclotetramerization in 2-(dimethylamino)ethanol in the presence of a metal salt. The coumarins and phthalonitriles used in many of these reactions, and the metals in the phthalocyanine complexes, are shown in Table 1 [63,64,65,66,67,68,69,70,71,72,73,74,75,76,77,78,79,80,81,82,83,84,85,86,87,88,89,90,91].
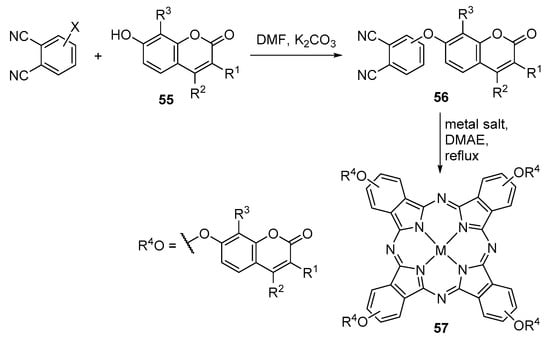
Scheme 14.
Typical route to coumarin–phthalocyanine conjugates [63,64,65,66,67,68,69,70,71,72,73,74,75,76,77,78,79,80,81,82,83,84,85,86,87,88,89,90,91].

Table 1.
Starting materials and yields of coumarin–phthalocyanine conjugates 57.
The method described above was also used for the synthesis of phthalocyanines bearing four peripheral 7-thio-coumarin units. The metal-free phthalocyanine 59a and the metallophthalocyanines 59b–d (Scheme 15) were prepared in high yields from the phthalonitrile derivative 58, respectively in 2-dimethylaminoethanol (DMAE) at 145 °C or in quinoline at 195 °C, in a sealed tube [64].
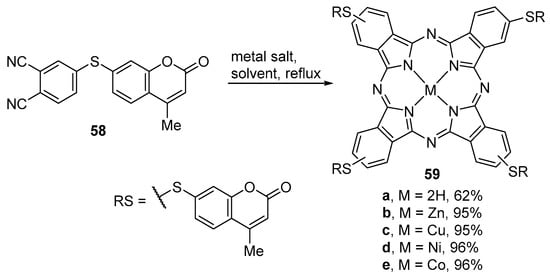
Scheme 15.
Synthesis of metal-free and metallophthalocyanines [64].
The group of Bulut reported the synthesis of several phthalonitriles 60 and their conversion into phthalocyanines 61 carrying four or eight 4-(coumarin-3-yl)phenoxy substituents (Scheme 16 and Table 2) [92,93,94,95,96,97,98,99,100,101,102,103,104].
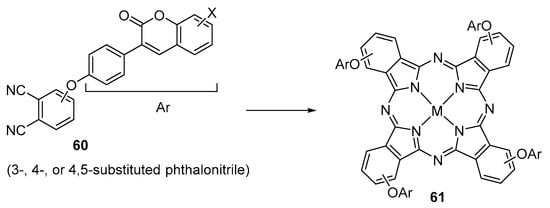
Scheme 16.
Synthesis of coumarin–phthalocyanine conjugates 61 [92,93,94,95,96,97,98,99,100,101,102,103,104].

Table 2.
Coumarin–phthalocyanine conjugates 62 and yields.
A recent study report the preparation of four coumarin–phthalocyanine conjugates bearing only one coumarin unit in which the coumarin moiety is linked to the phthalocyanine core at position 1 or 2 via a tri(ethylene glycol) spacer (63, Scheme 17) [105]. These conjugates were evaluated as potential anticancer agents with dual photodynamic and chemotherapy activity. Upon illumination at 670 nm, they show substantial cytotoxicity against HepG2 human hepatocarcinoma cells, with IC50 values in the range of 0.014–0.044 µM. The conjugate bearing the coumarin substituent at position 1 of the phthalocyanine (and R1 = H) exhibits high photocytotoxicity and significant chemocytotoxicity (IC50 = 4.43 µM). The results indicate that the coumarin and the phthalocyanine components of this conjugate work in a cooperative fashion [105].
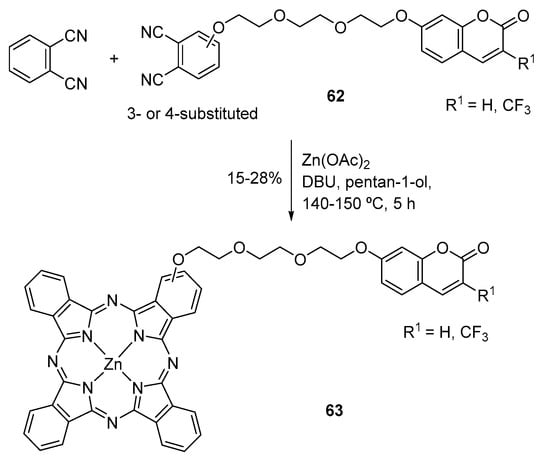
Scheme 17.
Synthesis of coumarin–phthalocyanine conjugates [105].
Bulut and co-workers reported the synthesis of the axial coumarin-substituted titanium(IV) phthalocyanines 66a,b and their characterization by IR, UV–Vis, fluorescence, 1H-NMR, and MALDI-TOF MS [106]. The new conjugates were obtained in high yields from the reaction of the ortho-dihydroxyphenylcoumarin derivatives 64a,b with the oxotitanium(IV) phthalocyanine 65 (Scheme 18). The UV–Vis spectra revealed small red-shifts of the Q-bands of the conjugates 66a,b when compared to the Q-band of the oxotitanium(IV) phthalocyanine 65. They also suggested that the conjugates have low aggregation tendency in nonpolar solvents. The fluorescence studies showed that the emission intensity is diminished by the axial coumarin substituents. Metal–insulator–semiconductor capacitors incorporating spin coated films of phthalocyanines 65 and 66a,b as the insulating layer showed a rectification behavior.
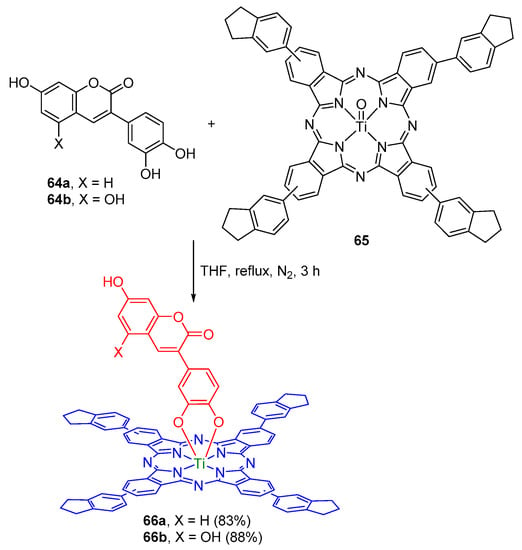
Scheme 18.
Synthesis of axial coumarin-substituted titanium(IV) phthalocyanines [106].
2.3. Coumarin–Corrole Conjugates
Corroles display highly interesting and unusual photophysical and chemical properties. The methods for their functionalization were reviewed recently [107]. Despite the importance of such tetrapyrrolic compounds, published works on the synthesis of coumarin–corrole conjugates are scarce. In fact, the first synthesis of coumarin–corrole conjugates was reported only in 2010 by Gryko and co-workers [108]. These authors used two distinct approaches to synthesize the coumarin–corrole conjugates: (a) direct synthesis of the conjugates from the condensation of a dipyrromethane with a coumarincarbaldehyde (Scheme 19) or (b) functionalization of an ethynyl-substituted corrole with a bromocoumarin derivative via a Sonogashira reaction (Scheme 20).
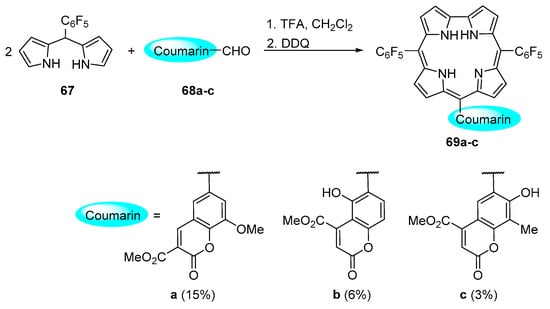
Scheme 19.
Coumarin–corrole dyads obtained by the [2 + 1] approach [108].
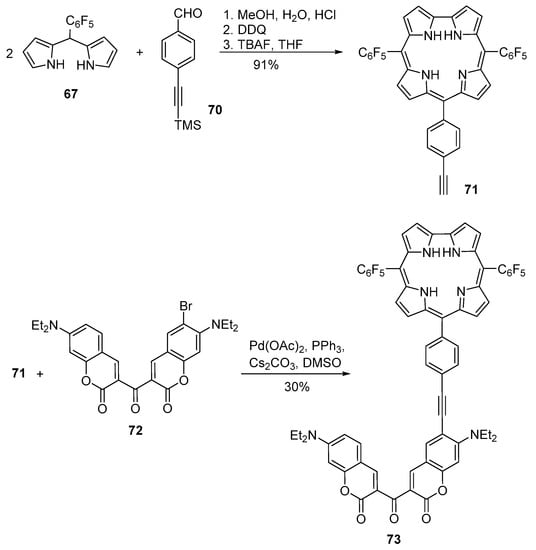
Scheme 20.
Coumarin–corrole dyad obtained under copper-free Sonogashira conditions [108].
The method used to prepare conjugates 69a–c, that bear a direct link between both chromophores, involved the condensation of 5-(pentafluorophenyl)dipyrromethane 67 [109] with the coumarincarbaldehydes 68a–c in CH2Cl2/trifluoroacetic acid (TFA) followed by oxidation with DDQ, according to established conditions [110]. The authors justified the relative low yields obtained in the [2 + 1] strategy for corroles 69a–c due to a competitive Michael addition of the dipyrromethane to the coumarin α,β-unsaturated system.
The dyad 73 was synthesized by coupling the ethynyl-corrole 71 with the adequate bromo-ketobiscoumarin 72 under copper-free Sonogashira conditions (Scheme 20) [108]. Corrole 71 was obtained from the reaction of the TMS-protected ethynylbenzaldehyde 70 [111] with dipyrromethane 67 in aqueous methanol in the presence of HCl, followed by oxidation with DDQ [112] and then removal of the protecting group with tetrabutylammonium fluoride (TBAF).
Spectroscopic studies revealed that in all dyads the electronic coupling between the components is weak. In addition, there is a quantitative and extremely fast energy transfer from the coumarin moiety to the corrole unit [108]. This behaviour was justified considering the significant Stokes shift and the resulting overlap of coumarin emission with corrole absorption as well as the short spacer between the two units. The energy transfer was ascribed to a dipole–dipole mechanism. In dyad 69b it was detected an electron-transfer from the excited corrole to the coumarin, yielding the low lying charge separated state Cum−–Corr+.
The same group extended the copper-free Sonogashira approach to the synthesis of dyads 74 and 75 (Figure 4) [113]. The coupling involved the ethynyl-corrole 71 and the adequate 6-bromocoumarins. The cross-coupling reactions were performed in DMF in the presence of Pd(AcO)2, PPh3 and Cs2CO3 and the dyads were obtained in moderate yields.
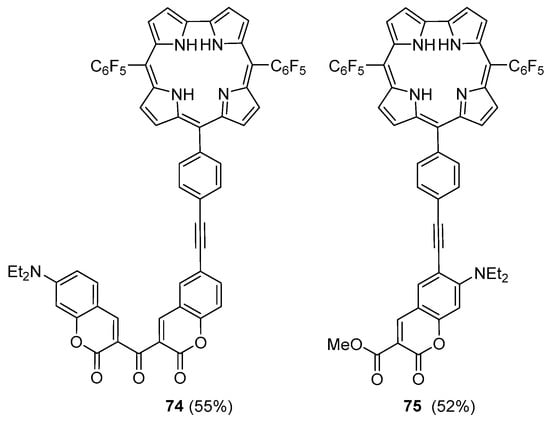
Figure 4.
Coumarin–corrole dyads obtained under copper-free Sonogashira conditions [113].
In the same article it was also reported the synthesis of a series of coumarin–corrole dyads (76–80, Figure 5) via condensation of dipyrromethane 67 with suitable formylcoumarins [113]. These condensations were performed in CH2Cl2/trifluoroacetic acid followed by oxidation with DDQ and again the competitive Michael addition of the dipyrromethane to the α,β-unsaturated coumarins was considered responsible by the low yields of the desired dyads.
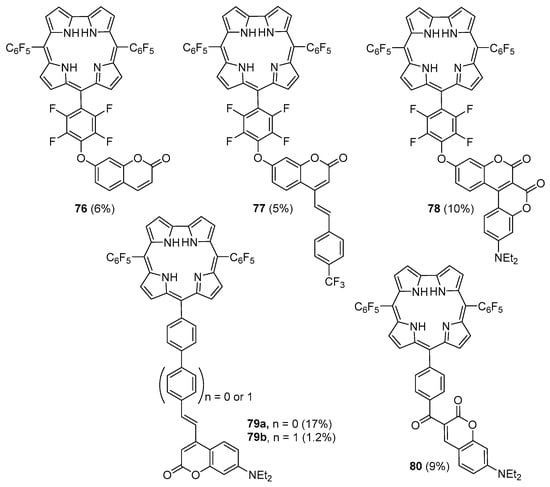
Figure 5.
Coumarin–corrole dyads obtained by the [2 + 1] strategy [113].
Gryko, Wróbel and co-workers reported the synthesis of several trans-A2B-corroles, where A is the C6F5 group and B is an aryl or hetaryl group, and their use in a comparative study of their spectroscopic features [114]. One of those compounds was the coumarin–corrole 82 that was obtained from the reaction of dipyrromethane 67 with the coumarin-4-carbaldehyde 81 (Scheme 21). The yield obtained (16%) is much higher than the ones of related systems reported in previous studies (ca. 5–8%) [108,113]. This result was attributed to the much weaker polarization of 7,8-dimetoxycoumarin-4-carbaldehyde 81 and consequently the side reactions were minimized. Spectroscopic studies (absorption and excitation emission and fluorescence life-time values) of this dye in chloroform allowed to rule out the formation of aggregates even in highly concentrated solutions. The experimental data were supported by quantum chemical calculations (TD-DFT) of the HOMO and the LUMO. The electron spin resonance (ESR) studies before and after light illumination demonstrated that an unpaired electron is localized on the corrole core but not on the substituent.
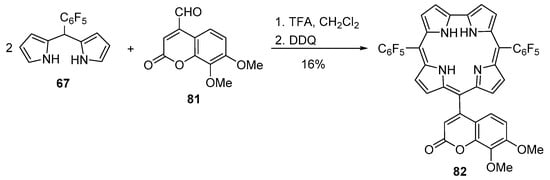
Scheme 21.
Synthesis of the dyad 82 [114].
Neves and co-workers developed a new strategy to prepare coumarin–corrole dyads where the coumarin unit is linked to a β-pyrrolic position of the corrole [39]. The new method involved a hetero-Diels–Alder reaction between the 3-vinylcorrole 83 [115] with o-quinone methides generated in situ from 4-hydroxycoumarins 2a,b and paraformaldehyde (Scheme 22). The reactions were performed in refluxing dioxane and the cycloadducts 84a,b were obtained in excellent yields.
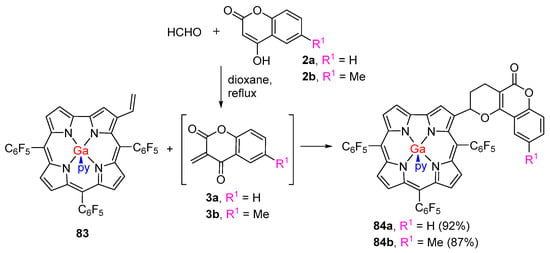
Scheme 22.
Synthesis of coumarin–corrole conjugates via hetero-Diels–Alder reactions [39].
The characterization of the photophysical properties of these dyads in various solvents (dichloromethane, DMSO, toluene, and ethanol) showed a strong solvatochromic effect for compound 84b, easily detected by naked eye (Figure 6): a bathochromic shift of the absorption bands occurs with the increase of the solvent polarity. The sensorial ability of the coumarin–corrole dyads 84a,b towards spherical (F−, Cl−), linear (CN−), and bulky (CH3COO−) anions was evaluated by UV-vis and fluorescence measurements and the more drastic alterations were detected for fluoride, cyanide and acetate.

Figure 6.
Solvatochromic effect of compound 84b. Reprinted with permission from ref. [39]. Copyright © 2013 Elsevier Ltd.
3. Final Remarks
As shown in this review, several synthetic strategies aiming the synthesis of coumarin–tetrapyrrolic macrocycle conjugates were already developed. However, it is important to develop new and versatile synthetic routes to these compounds. In particular, the access to coumarin− phthalocyanine conjugates via post-functionalization of adequately substituted phthalocyanines deserves greater attention from synthetic chemists. Considering the well-known photophysical (and biological) properties of coumarins and tetrapyrrolic macrocycles, the corresponding conjugates are expected to be found in real applications in the coming years.
Acknowledgments
Thanks are due to Fundação para a Ciência e a Tecnologia (FCT) for the financial support to project PTDC/QEQ-QOR/6160/2014 and the QOPNA research project (FCT UID/QUI/00062/2013) through national funds and where applicable co-financed by the FEDER, within the PT2020 Partnership Agreement, and also to the Portuguese NMR Network.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Gaspar, A.; Matos, M.J.; Garrido, J.; Uriarte, E.; Borges, F. Chromone: A valid scaffold in medicinal chemistry. Chem. Rev. 2014, 114, 4960–4992. [Google Scholar] [CrossRef] [PubMed]
- Kontogiorgis, C.A.; Hadjipavlou-Litina, D.J. Synthesis and antiinflammatory activity of coumarin derivatives. J. Med. Chem. 2005, 48, 6400–6408. [Google Scholar] [CrossRef] [PubMed]
- Tada, Y.; Shikishima, Y.; Takaishi, Y.; Shibata, H.; Higuti, T.; Honda, G.; Ito, M.; Takeda, Y.; Kodzhimatov, O.K.; Ashurmetov, O.; et al. Coumarins and γ-pyrone derivatives from Prangos pabularia: Antibacterial activity and inhibition of cytokine release. Phytochemistry 2002, 59, 649–654. [Google Scholar] [CrossRef]
- Matos, M.J.; Vazquez-Rodriguez, S.; Santana, L.; Uriarte, E.; Fuentes-Edfuf, C.; Santos, Y.; Muñoz-Crego, A. Synthesis and structure-activity relationships of novel amino/nitro substituted 3-arylcoumarins as antibacterial agents. Molecules 2013, 18, 1394–1404. [Google Scholar] [CrossRef] [PubMed]
- Olomola, T.O.; Mosebi, S.; Traut-Johnstone, T.; Coates, J.; Hewer, R.; Kaye, P.T. Novel furocoumarins as potential HIV-1 integrase inhibitors. Bioorg. Chem. 2014, 57, 1–4. [Google Scholar] [CrossRef] [PubMed]
- Hassan, M.Z.; Osman, H.; Ali, M.A.; Ahsan, M.J. Therapeutic potential of coumarins as antiviral agents. Eur. J. Med. Chem. 2016, 123, 236–255. [Google Scholar] [CrossRef] [PubMed]
- Musa, M.A.; Cooperwood, J.S.; Khan, M.O.F. A review of coumarin derivatives in pharmacotherapy of breast cancer. Curr. Med. Chem. 2008, 15, 2664–2679. [Google Scholar] [CrossRef] [PubMed]
- Basanagouda, M.; Jambagi, V.B.; Barigidad, N.N.; Laxmeshwar, S.S.; Narayanachar, V.D. Synthesis, structure–activity relationship of iodinated-4-aryloxymethyl-coumarins as potential anti-cancer and antimycobacterial agentes. Eur. J. Med. Chem. 2014, 74, 225–233. [Google Scholar] [CrossRef] [PubMed]
- Kaur, M.; Kohli, S.; Sandhu, S.; Bansal, Y.; Bansal, G. Coumarin: A promising scaffold for anticancer agents. Anti-Cancer Agents Med. Chem. 2015, 15, 1032–1048. [Google Scholar] [CrossRef]
- Zou, Q.; Fang, Y.; Zhao, Y.; Zhao, H.; Wang, Y.; Gu, Y.; Wu, F. Synthesis and in vitro photocytotoxicity of coumarin derivatives for one- and two-photon excited photodynamic therapy. J. Med. Chem. 2013, 56, 5288–5294. [Google Scholar] [CrossRef] [PubMed]
- Siegrist, A.E.; Hefti, H.; Meyer, H.R.; Schmidt, E. Fluorescent whitening agents 1973–1985. Rev. Prog. Coloration 1987, 17, 39–55. [Google Scholar] [CrossRef]
- Zhang, G.; Zheng, H.; Guo, M.; Du, L.; Liu, G.; Wang, P. Synthesis of polymeric fluorescent brightener based on coumarin and its performances on paper as light stabilizer, fluorescent brightener and surface sizing agent. Appl. Surface Sci. 2016, 367, 167–173. [Google Scholar] [CrossRef]
- Bazzicalupi, C.; Caltagirone, C.; Cao, Z.; Chen, Q.; Natale, C.D.; Garau, A.; Lippolis, V.; Lvova, L.; Liu, H.; Lundström, I.; et al. Multimodal use of new coumarin-based fluorescent chemosensors: Towards highly selective optical sensors for Hg2+ probing. Chem. Eur. J. 2013, 19, 14639–14653. [Google Scholar] [CrossRef] [PubMed]
- Ren, X.; Kondakova, M.E.; Giesen, D.J.; Rajeswaran, M.; Madaras, M.; Lenhart, W.C. Coumarin-based, electron-trapping iridium complexes as highly efficient and stable phosphorescent emitters for organic light-emitting diodes. Inorg. Chem. 2010, 49, 1301–1303. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.; Lee, K.J.; Kim, B.; Lee, J.; Kay, K.-Y.; Park, J. New ambipolar blue emitting materials based on amino coumarin derivatives with high efficiency for organic light emitting diodes. J. Nanosci. Nanotechnol. 2013, 13, 8020–8024. [Google Scholar] [CrossRef] [PubMed]
- Kotchapradist, P.; Prachumrak, N.; Sunonnam, T.; Tarsang, R.; Namuangruk, S.; Sudyoadsuk, T.; Keawin, T.; Jungsuttiwong, S.; Promarak, V. N-coumarin derivatives as hole-transporting emitters for high efficiency solution-processed pure green electroluminescent devices. Dyes Pigment. 2015, 112, 227–235. [Google Scholar] [CrossRef]
- Kumar, S.; Puttaraju, B.; Patil, S. A deep-blue electroluminescent device based on a coumarin derivative. ChemPlusChem 2016, 81, 384–390. [Google Scholar] [CrossRef]
- Esnal, I.; Duran-Sampedro, G.; Agarrabeitia, A.R.; Bañuelos, J.; García-Moreno, I.; Macías, M.A.; Peña-Cabrera, E.; López-Arbeloa, I.; de la Moya, S.; Ortiz, M.J. Coumarin–BODIPY hybrids by heteroatom linkage: Versatile, tunable and photostable dye lasers for UV irradiation. Phys. Chem. Chem. Phys. 2015, 17, 8239–8247. [Google Scholar] [CrossRef] [PubMed]
- Jakubiak, R.; Bunning, T.J.; Vaia, R.A.; Natarajan, L.V.; Tondiglia, V.P. Electrically switchable, one-dimensional polymeric resonators from holographic photopolymerization: A new approach for active photonic bandgap materials. Adv. Mater. 2003, 15, 241–244. [Google Scholar] [CrossRef]
- Trenor, S.R.; Shultz, A.R.; Love, B.J.; Long, T.E. Coumarins in polymers: From light harvesting to photo-cross-linkable tissue scaffolds. Chem. Rev. 2004, 104, 3059–3077. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.-S.; Hara, K.; Dan-oh, Y.; Kasada, C.; Shinpo, A.; Suga, S.; Arakawa, H.; Sugihara, H. Photophysical and (photo)electrochemical properties of a coumarin dye. J. Phys. Chem. B 2005, 109, 3907–3914. [Google Scholar] [CrossRef] [PubMed]
- Oltra, N.S.; Browne, W.R.; Roelfes, G. Hierarchical self-assembly of a biomimetic light-harvesting antenna based on DNA G-quadruplexes. Chem. Eur. J. 2013, 19, 2457–2461. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.M.; Fang, X.Z.; Yang, P.R.; Yi, J.-S.; Ko, Y.-G.; Piao, M.J.; Chung, Y.D.; Park, Y.W.; Jeon, S.-J.; Cho, B.R. Design of molecular two-photon probes for in vivo imaging. 2H-Benzo[h]chromene-2-one derivatives. Tetrahedron Lett. 2007, 48, 2791–2795. [Google Scholar] [CrossRef]
- Wagner, B.D. The use of coumarins as environmentally-sensitive fluorescent probes of heterogeneous inclusion systems. Molecules 2009, 14, 210–237. [Google Scholar] [CrossRef] [PubMed]
- Schill, H.; Nizamov, S.; Bottanelli, F.; Bierwagen, J.; Belov, V.N.; Hell, S.W. 4-Trifluoromethyl-substituted coumarins with large stokes shifts: Synthesis, bioconjugates, and their use in super-resolution fluorescence microscopy. Chem. Eur. J. 2013, 19, 16556–16565. [Google Scholar] [CrossRef] [PubMed]
- Meimetis, L.G.; Carlson, J.C.T.; Giedt, R.J.; Kohler, R.H.; Weissleder, R. Ultrafluorogenic coumarin–tetrazine probes for real-time biological imaging. Angew. Chem. Int. Ed. 2014, 53, 7531–7534. [Google Scholar] [CrossRef] [PubMed]
- Yanga, M.; Wang, H.; Huang, J.; Fang, M.; Mei, B.; Zhou, H.; Wu, J.; Tian, Y. Highly sensitive and selective colorimetric and fluorescent off–on probe for copper(II) based on unique addition reaction and its imaging in living cells. Sens. Actuators B 2014, 204, 710–715. [Google Scholar] [CrossRef]
- Rong, L.; Liu, L.-H.; Chen, S.; Cheng, H.; Chen, C.-S.; Li, Z.-Y.; Qin, S.-Y.; Zhang, X.-Z. A coumarin derivative as a fluorogenic glycoproteomic probe for biological imaging. Chem. Commun. 2014, 50, 667–669. [Google Scholar] [CrossRef] [PubMed]
- Listunov, D.; Mazères, S.; Volovenko, Y.; Joly, E.; Génisson, Y.; Maraval, V.; Chauvin, R. Fluorophore-tagged pharmacophores for antitumor cytotoxicity: Modified chiral lipidic dialkynylcarbinols for cell imaging. Bioorg. Med. Chem. Lett. 2015, 25, 4652–4656. [Google Scholar] [CrossRef] [PubMed]
- Sednev, M.V.; Belov, V.N.; Hell, S.W. Fluorescent dyes with large Stokes shifts for super-resolution optical microscopy of biological objects: A review. Method. Appl. Fluoresc. 2015, 3, 042004. [Google Scholar] [CrossRef]
- Bochkov, A.Y.; Akchurin, I.O.; Dyachenko, O.A.; Traven, V.F. NIR-fluorescent coumarin-fused BODIPY dyes with large Stokes shifts. Chem. Commun. 2013, 49, 11653–11655. [Google Scholar] [CrossRef] [PubMed]
- Brites, M.J.; Santos, C.; Nascimento, S.; Gigante, B.; Luftmann, H.; Fedorov, A.; Berberan-Santos, M.N. Synthesis and fluorescence properties of [60] and [70]fullerene–coumarin dyads: Efficient dipole–dipole resonance energy transfer from coumarin to fullerene. New J. Chem. 2006, 30, 1036–1045. [Google Scholar] [CrossRef]
- Abellán-Flos, M.; Tanç, M.; Supuran, C.T.; Vincent, S.P. Exploring carbonic anhydrase inhibition with multimeric coumarins displayed on a fullerene scaffold. Org. Biomol. Chem. 2015, 13, 7445–7451. [Google Scholar] [CrossRef] [PubMed]
- Nascimento, S.; Fedorov, A.; Brites, M.J.; Berberan-Santos, M.N. New coumarin–[60]fullerene dyads connected by an alkynyl linkage: Synthesis and fluorescence studies. Evidence for efficient singlet–singlet energy transfer. Dyes Pigment. 2015, 114, 158–165. [Google Scholar] [CrossRef]
- Hurenkamp, J.H.; Browne, W.R.; Augulis, R.; Pugzlys, A.; van Loosdrecht, P.H.M.; van Esch, J.H.; Feringa, B.L. Intramolecular energy transfer in a tetra-coumarin perylene system: Influence of solvent and bridging unit on electronic properties. Org. Biomol. Chem. 2007, 5, 3354–3362. [Google Scholar] [CrossRef] [PubMed]
- Augulis, R.; Pugzlys, A.; Hurenkamp, J.H.; Feringa, B.L.; van Esch, J.H.; van Loosdrecht, P.H.M. Optical energy transport and interactions between the excitations in a coumarin–perylene bisimide dendrimer. J. Phys. Chem. A 2007, 111, 12944–12953. [Google Scholar] [CrossRef] [PubMed]
- Aydin, E.; Nisanci, B.; Acar, M.; Dastan, A.; Bozdemir, O.A. Synthesis and use of ”clickable” bay-region tetrasubstituted perylene tetracarboxylic tetraesters and a perylene monoimide diester as energy acceptors. New J. Chem. 2015, 39, 548–554. [Google Scholar] [CrossRef]
- Menezes, J.C.J.M.D.S.; Gomes, A.T.P.C.; Silva, A.M.S.; Faustino, M.A.F.; Neves, M.G.P.M.S.; Tomé, A.C.; da Silva, F.C.; Ferreira, V.F.; Cavaleiro, J.A.S. Reaction of β-vinyl-meso-tetraphenylporphyrin with o-quinone methines. Synlett 2011, 13, 1841–1844. [Google Scholar] [CrossRef]
- Santos, C.I.M.; Oliveira, E.; Menezes, J.C.J.M.D.S.; Barata, J.F.B.; Faustino, M.A.F.; Ferreira, V.F.; Cavaleiro, J.A.S.; Neves, M.G.P.M.S.; Lodeiro, C. New coumarin–corrole and –porphyrin conjugate multifunctional probes for anionic or cationic interactions: Synthesis, spectroscopy, and solid supported studies. Tetrahedron 2014, 70, 3361–3370. [Google Scholar] [CrossRef]
- Callot, H.J. Nouvelles voies d’accès aux vinylporphyrins. Tetrahedron 1973, 29, 899–901. [Google Scholar] [CrossRef]
- Santos, C.I.M.; Oliveira, E.; Santos, H.M.; Menezes, J.C.J.M.D.S.; Faustino, M.A.F.; Cavaleiro, J.A.S.; Capelo, J.L.; Neves, M.G.P.M.S.; Lodeiro, C. Untangling interactions of a zinc(II) complex containing a coumarin–porphyrin unit with alkaloids in water solutions: A photophysical study. Photochem. Photobiol. Sci. 2015, 14, 757–764. [Google Scholar] [CrossRef] [PubMed]
- Menezes, J.C.J.M.D.S.; Neves, M.G.P.M.S.; Cavaleiro, J.A.S.; Barros, C.; Santos, S.M.; da Silva, F.C.; Ferreira, V.F.; Domingues, M.R.M. Gas phase reactions of β-substituted hetero-Diels–Alder adducts of meso-tetraphenylporphyrin using tandem mass spectrometry. Int. J. Mass Spectrom. 2013, 343–344, 1–8. [Google Scholar] [CrossRef]
- Menezes, J.C.J.M.D.S.; Faustino, M.A.F.; de Oliveira, K.T.; Uliana, M.P.; Ferreira, V.F.; Hackbarth, S.; Röder, B.; Tasso, T.T.; Furuyama, T.; Kobayashi, N.; et al. Synthesis of new chlorin e6 trimethyl and protoporphyrin IX dimethyl ester derivatives and their photophysical and electrochemical characterizations. Chem. Eur. J. 2014, 20, 13644–13655. [Google Scholar] [CrossRef] [PubMed]
- Cardoso, M.F.C.; Gomes, A.T.P.C.; Silva, V.L.M.; Silva, A.M.S.; Neves, M.G.P.M.S.; da Silva, F.C.; Ferreira, V.F.; Cavaleiro, J.A.S. Ohmic heating assisted synthesis of coumarinyl porphyrin derivatives. RSC Adv. 2015, 5, 66192–66199. [Google Scholar] [CrossRef]
- Shen, D.-M.; Liu, C.; Chen, Q.-Y. Synthesis and versatile reactions of β-azidotetraarylporphyrins. Eur. J. Org. Chem. 2007, 2007, 1419–1422. [Google Scholar] [CrossRef]
- Singh, D.K.; Nath, M. Synthesis and photophysical properties of β-triazole bridged porphyrincoumarin dyads. RSC Adv. 2015, 5, 68209–68217. [Google Scholar] [CrossRef]
- Singh, D.K.; Nath, M. Synthesis, characterization and photophysical studies of β-triazolomethyl-bridged porphyrin-benzo-α-pyrone dyads. J. Chem. Sci. 2016, 128, 545–554. [Google Scholar] [CrossRef]
- Singh, D.K.; Nath, M. meso-Phenyl-triazole bridged porphyrin-coumarin dyads: Synthesis, characterization and photophysical properties. Dyes Pigment. 2015, 121, 256–264. [Google Scholar] [CrossRef]
- Lin, W.; Long, L.; Feng, J.; Wang, B.; Guo, C. Synthesis of meso-coumarin-conjugated porphyrins and investigation of their luminescence properties. Eur. J. Org. Chem. 2007, 4301–4304. [Google Scholar] [CrossRef]
- Adler, A.D.; Longo, F.R.; Finarelli, J.D.; Goldmacher, J.; Assour, J.; Korsakoff, L. A simplified synthesis for meso-tetraphenylporphine. J. Org. Chem. 1967, 32, 476. [Google Scholar] [CrossRef]
- Lindsey, J.S.; Schreiman, I.C.; Hsu, H.C.; Kearney, P.C.; Marguerettaz, A.M. Rothemund and Adler-Longo reactions revisited: Synthesis of tetraphenylporphyrins under equilibrium conditions. J. Org. Chem. 1987, 52, 827–836. [Google Scholar] [CrossRef]
- Amaravathi, M.; Rajitha, B.; Rao, M.K.; Sitadevi, P. Synthesis of meso-tetrakis(4-chlorocoumarin-3-yl)porphyrins. J. Heterocycl. Chem. 2007, 44, 821–825. [Google Scholar] [CrossRef]
- Hecht, S.; Ihre, H.; Fréchet, J.M.J. Porphyrin core star polymers: Synthesis, modification, and implication for site isolation. J. Am. Chem. Soc. 1999, 121, 9239–9240. [Google Scholar] [CrossRef]
- Hecht, S.; Vladimirov, N.; Fréchet, J.M.J. Encapsulation of functional moieties within branched star polymers: Effect of chain length and solvent on site isolation. J. Am. Chem. Soc. 2001, 123, 18–25. [Google Scholar] [CrossRef] [PubMed]
- Dichtel, W.R.; Hecht, S.; Fréchet, J.M.J. Functionally layered dendrimers: A new building block and its application to the synthesis of multichromophoric light-harvesting systems. Org. Lett. 2005, 7, 4451–4454. [Google Scholar] [CrossRef] [PubMed]
- Mao, M.; Song, Q.-H. Non-conjugated dendrimers with a porphyrin core and coumarin chromophores as peripheral units: Synthesis and photophysical properties. Dyes Pigment. 2012, 92, 975–981. [Google Scholar] [CrossRef]
- Concellón, A.; Marcos, M.; Romero, P.; Serrano, J.L.; Termine, R.; Golemme, A. Not only columns: High hole mobility in a discotic nematic mesophase formed by metal-containing porphyrin-core dendrimers. Angew. Chem. Int. Ed. 2017, 56, 1259–1263. [Google Scholar] [CrossRef] [PubMed]
- Cao, X.; Lin, W.; Yu, Q. A ratiometric fluorescent probe for thiols based on a tetrakis(4-hydroxyphenyl)porphyrin-coumarin scaffold. J. Org. Chem. 2011, 76, 7423–7430. [Google Scholar] [CrossRef] [PubMed]
- Lebedev, A.Y.; Troxler, T.; Vinogradov, S.A. Design of metalloporphyrin-based dendritic nanoprobes for two-photon microscopy of oxygen. J. Porphyr. Phthalocyanines 2008, 12, 1261–1269. [Google Scholar] [CrossRef] [PubMed]
- Hania, P.R.; Heijs, D.J.; Bowden, T.; Pugžlys, A.; van Esch, J.; Knoester, J.; Duppen, K. Ultrafast energy transport in a first-generation coumarin-tetraphenylporphyrin dendrimer. J. Phys. Chem. B 2004, 108, 71–81. [Google Scholar] [CrossRef]
- Concellón, A.; Bucos, M.; Serrano, J.L.; Romero, P.; Marcos, M. Supramolecular liquid crystalline dendrimers with a porphyrin core and functional carboxylic acid dendrons. RSC Adv. 2016, 6, 65179–65185. [Google Scholar] [CrossRef]
- Garg, V.; Kodis, G.; Liddell, P.A.; Terazono, Y.; Moore, T.A.; Moore, A.L.; Gust, D. Artificial photosynthetic reaction center with a coumarin-based antenna system. J. Phys. Chem. B 2013, 117, 11299–11308. [Google Scholar] [CrossRef] [PubMed]
- Medyouni, R.; Mtibaa, A.C.; Mellouli, L.; Romerosa, A.; Hamdi, N. Convenient synthesis of novel unmetalled and metallophthalocyanines bearing coumarin derivatives: Synthesis, characterization, aggregation behaviors and antimicrobial activity. J. Incl. Phenom. Macrocycl. Chem. 2016, 86, 201–210. [Google Scholar] [CrossRef]
- Esenpinar, A.A.; Bulut, M. Synthesis and characterization of metal-free and metallo-phthalocyanines with four pendant coumarinthio/oxy-substituents. Dyes Pigment. 2008, 76, 249–255. [Google Scholar] [CrossRef]
- Guo, J.-J.; Wang, S.-R.; Li, X.-G.; Zhang, F.; Shao, X.-N.; Lian, X.-J. The synthesis, molecular structure and photophysical properties of 2,9,16,23-tetrakis(7-coumarinoxy-4-methyl)phthalocyanine sensitizer. J. Mol. Struct. 2014, 1060, 17–23. [Google Scholar] [CrossRef]
- Guo, J.-J.; Wang, S.-R.; Li, X.-G.; Zhang, F.; Xiao, Y.; Teng, C. The synthesis, characterisation, photophysical and thermal properties, and photovoltaic performance of 7-coumarinoxy-4-methyltetrasubstituted metallophthalocyanines. Aust. J. Chem. 2015, 68, 1025–1034. [Google Scholar] [CrossRef]
- Han, J.L.; You, J.; Yonemura, H.; Yamada, S.; Wang, S.R.; Li, X.G. Metallophthalocyanines as triplet sensitizers for highly efficient photon upconversion based on sensitized triplet–triplet annihilation. Photochem. Photobiol. Sci. 2016, 15, 1039–1045. [Google Scholar] [CrossRef] [PubMed]
- Chohan, S.; Booysen, I.N.; Mambanda, A.; Akerman, M.P. Synthesis, characterization and electrocatalytic behavior of cobalt and iron phthalocyanines bearing chromone or coumarin substituents. J. Coord. Chem. 2015, 68, 1829–1846. [Google Scholar] [CrossRef]
- Altun, S.; Özkaya, A.R.; Bulut, M. Peripheral octa-substituted metal-free, cobalt(II) and zinc(II) phthalocyanines bearing coumarin and chloro groups: Synthesis, characterization, spectral and electrochemical properties. Polyhedron 2012, 48, 31–42. [Google Scholar] [CrossRef]
- Odabas, Z.; Orman, E.B.; Durmus, M.; Dumludag, F.; Özkaya, A.R.; Bulut, M. Novel alpha-7-oxy-4-(4-methoxyphenyl)-8-methylcoumarin substituted metal-free, Co(II) and Zn(II) phthalocyanines: Photochemistry, photophysics, conductance and electrochemistry. Dyes Pigment. 2012, 95, 540–552. [Google Scholar] [CrossRef]
- Altun, S.; Odabaş, Z.; Altındal, A.; Özkaya, A.R. Coumarin-substituted manganese phthalocyanines: Synthesis, characterization, photovoltaic behaviour, spectral and electrochemical properties. Dalton Trans. 2014, 43, 7987–7997. [Google Scholar] [CrossRef] [PubMed]
- Altun, S.; Orman, E.B.; Odabaş, Z.; Altındal, A.; Özkaya, A.R. Gas sensing and electrochemical properties of tetra and octa 2H-chromen-2-one substituted iron(II) phthalocyanines. Dalton Trans. 2015, 44, 4341–4354. [Google Scholar] [CrossRef] [PubMed]
- Çalik, A.E.; Köksoy, B.; Orman, E.B.; Durmus, M.; Özkaya, A.R.; Bulut, M. 4-Carboxymethyl-8-methyl-7-oxycoumarin substituted zinc, cobalt and indium phthalocyanines: Electrochemical and photochemical properties. J. Porphyr. Phthalocyanines 2013, 17, 1046–1054. [Google Scholar] [CrossRef]
- Alemdar, A.; Özkaya, A.R.; Bulut, M. Synthesis, spectroscopy, electrochemistry and in situ spectroelectrochemistry of partly halogenated coumarin phthalonitrile and corresponding metal-free, cobalt and zinc phthalocyanines. Polyhedron 2009, 28, 3788–3796. [Google Scholar] [CrossRef]
- Alemdar, A.; Özkaya, A.R.; Bulut, M. Preparation, characterization, electrochemistry and in situ spectroelectrochemistry of novel α-tetra[7-oxo-3-(2-chloro-4-fluorophenyl)coumarin]-substituted metal-free, cobalt and zinc phthalocyanines. Synth. Met. 2010, 160, 1556–1565. [Google Scholar] [CrossRef]
- Piskin, M.; Durmus, M.; Bulut, M. Highly soluble 7-oxy-3-(4-methoxyphenyl)coumarin bearing zinc phthalocyanines: Synthesis and investigation of photophysical and photochemical properties. J. Photochem. Photobiol. A 2011, 223, 37–49. [Google Scholar] [CrossRef]
- Odabas, Z.; Kara, H.; Ozkaya, A.R.; Bulut, M. Synthesis, characterization and electrochemical properties of novel β 7-oxy-4-(4-methoxyphenyl)-8-methylcoumarin substituted metal-free, Zn(II) and Co(II) phthalocyanines. Polyhedron 2012, 39, 38–47. [Google Scholar] [CrossRef]
- Kaya, E.N.; Durmus, M.; Bulut, M. The effects of substituents’ positions and variety of axial groups on the photochemical properties of coumarin substituted indium(III) phthalocyanines. J. Organomet. Chem. 2014, 774, 94–100. [Google Scholar] [CrossRef]
- Kaya, E.N.; Durmus, M.; Bulut, M. Novel 7-oxy-3-(3′,4′,5′-trimethoxyphenyl)coumarin substituted zinc(II) phthalocyanines: Synthesis, characterization, photophysical and photochemical properties. J. Porphyr. Phthalocyanines 2015, 19, 1114–1122. [Google Scholar] [CrossRef]
- Tastemel, A.; Karaca, B.Y.; Durmus, M.; Bulut, M. Photophysical and photochemical properties of novel metallophthalocyanines bearing 7-oxy-3-(m-methoxyphenyl)coumarin groups. J. Lumin. 2015, 168, 163–171. [Google Scholar] [CrossRef]
- Erdogan, T.; Bulut, M.; Çamur, M. Novel phthalocyanines bearing 7-oxy-3-(3,5-difluorophenyl)coumarin moieties: Synthesis, characterization, photophysical and photochemical properties. J. Photochem. Photobiol. A 2015, 300, 6–14. [Google Scholar] [CrossRef]
- Gok, A.; Orman, E.B.; Salan, U.; Özkaya, A.R.; Bulut, M. Synthesis, characterization and electrochemical properties of tetra 7-oxy-3-biphenylcoumarin substituted metal-free, zinc(II), cobalt(II) and indium(III) phthalocyanines. Dyes Pigment. 2016, 133, 311–323. [Google Scholar] [CrossRef]
- Esenpinar, A.A.; Durmaz, E.; Karaca, F.; Bulut, M. Synthesis and characterization of metallo phthalocyanines bearing 7-oxy-3-(4-pyridyl)coumarin substituents and their supramolecular structures with vanadyl bis(acetylacetonate). Polyhedron 2012, 38, 267–274. [Google Scholar] [CrossRef]
- Çamur, M.; Esenpinar, A.A.; Özkaya, A.R.; Bulut, M. Synthesis, characterization, spectroscopic and electrochemical properties of phthalocyanines substituted with four 3-ferrocenyl-7-oxycoumarin moieties. J. Organomet. Chem. 2011, 696, 1868–1873. [Google Scholar] [CrossRef]
- Piskin, M.; Durmus, M.; Bulut, M. Synthesis, characterization, photophysical and photochemical properties of 7-oxy-3-methyl-4-phenylcoumarin-substituted indium phthalocyanines. Inorg. Chim. Acta 2011, 373, 107–116. [Google Scholar] [CrossRef]
- Piskin, M.; Durmus, M.; Bulut, M. Synthesis and investigation on photophysical and photochemical properties of 7-oxy-3-methyl-4-phenylcoumarin bearing zinc phthalocyanines. Spectrochim. Acta A Mol. Biomol. Spectrosc. 2012, 97, 502–511. [Google Scholar] [CrossRef] [PubMed]
- Dur, E.; Bulut, M. Derivatizable novel β-tetra 7-oxycoumarin-3-carboxylate substituted metallophthalocyanines: Synthesis and characterization. Polyhedron 2010, 29, 2689–2695. [Google Scholar] [CrossRef]
- Dur, E.; Ozkaya, A.R.; Bulut, M. Synthesis, characterisation and electrochemistry of derivatisable novel α-tetra 7-oxycoumarin-3-carboxylate-substituted metallophthalocyanines. Supramol. Chem. 2011, 23, 379–388. [Google Scholar] [CrossRef]
- Cakici, H.; Esenpinar, A.A.; Bulut, M. Synthesis and characterization of novel phthalocyanines bearing quaternizable coumarin. Polyhedron 2008, 27, 3625–3630. [Google Scholar] [CrossRef]
- Esenpınar, A.A.; Durmus, M.; Bulut, M. Tetra-3-[(2-diethylamino)ethyl]-7-oxo-4-methylcoumarin- substituted zinc phthalocyanines: Synthesis, characterization and aggregation effects on photophysical/photochemical properties. J. Photochem. Photobiol. A 2010, 213, 171–179. [Google Scholar] [CrossRef]
- Esenpinar, A.A.; Durmus, M.; Bulut, M. Photophysical, photochemical and BSA binding/BQ quenching properties of quaternizable coumarin containing water soluble zinc phthalocyanine complexes. Spectrochim. Acta A Mol. Biomol. Spectrosc. 2011, 79, 608–617. [Google Scholar] [CrossRef] [PubMed]
- Kartaloglu, N.; Esenpinar, A.A.; Bulut, M. Synthesis, characterization, and photophysical and photochemical properties of 3-(4-phenyloxy)coumarin containing metallo- and metal-free phthalocyanines. Turk. J. Chem. 2014, 38, 1102–1117. [Google Scholar] [CrossRef]
- Çamur, M.; Bulut, M.; Kandaz, M.; Guney, O. Effects of coumarin substituents on the photophysical properties of newly synthesized phthalocyanine derivatives. Supramol. Chem. 2009, 21, 624–631. [Google Scholar] [CrossRef]
- Çamur, M.; Bulut, M. The synthesis and characterization of novel soluble phthalocyanines substituted with 7-octyloxy-3-(4-oxyphenyl)coumarin moieties. Dyes Pigment. 2008, 77, 165–170. [Google Scholar] [CrossRef]
- Camur, M.; Bulut, M.; Kandaz, M.; Guney, O. Synthesis, characterization and fluorescence behavior of new fluorescent probe phthalocyanines bearing coumarin substituents. Polyhedron 2009, 28, 233–238. [Google Scholar] [CrossRef]
- Esenpinar, A.A.; Bulut, M. Synthesis and characterization of novel α- or β-tetra[6,7-dihexyloxy-3-(4-oxyphenyl)coumarin]-substituted metal-free and metallo phthalocyanines. Polyhedron 2009, 28, 3129–3137. [Google Scholar] [CrossRef]
- Çamur, M.; Özkaya, A.R.; Bulut, M. Synthesis, characterization and spectroscopic properties of new fluorescent 7,8-dihexyloxy-3-(4-oxyphenyl)coumarin substituted phthalocyanines. J. Porphyr. Phthalocyanines 2009, 13, 691–701. [Google Scholar] [CrossRef]
- Çamur, M.; Durmus, M.; Bulut, M. Highly singlet oxygen generative water-soluble coumarin substituted zinc(II) phthalocyanine photosensitizers for photodynamic therapy. Polyhedron 2012, 41, 92–103. [Google Scholar] [CrossRef]
- Esenpinar, A.A.; Durmus, M.; Bulut, M. Synthesis and properties of crown ether functionalized coumarin substituted zinc phthalocyanine. Polyhedron 2011, 30, 2067–2074. [Google Scholar] [CrossRef]
- Esenpinar, A.A.; Özkaya, A.R.; Bulut, M. Synthesis and electrochemical properties of crown ether functionalized coumarin substituted cobalt and copper phthalocyanines. J. Organomet. Chem. 2011, 696, 3873–3881. [Google Scholar] [CrossRef]
- Camur, M.; Durmus, M.; Ozkaya, A.R.; Bulut, M. Synthesis, photophysical, photochemical and electrochemical properties of crown ether bearing coumarin substituted phthalocyanines. Inorg. Chim. Acta 2012, 383, 287–299. [Google Scholar] [CrossRef]
- Çamur, M.; Durmus, M.; Bulut, M. Coumarino-12-crown-4 bearing phthalocyanine photosensitizers for singlet oxygen production. J. Photochem. Photobiol. A 2011, 222, 266–275. [Google Scholar] [CrossRef]
- Koksoy, M.A.; Koksoy, B.; Durmus, M.; Bulut, M. Preparation, characterization and photophysicochemical properties of novel tetra 7-(diethyl 2-methylmalonatoxy)-3-(p-oxyphenyl)coumarin-substituted zinc(II) and indium(III)chloride phthalocyanines. J. Organomet. Chem. 2016, 822, 125–134. [Google Scholar] [CrossRef]
- Can, O.S.; Kaya, E.N.; Durmus, M.; Bulut, M. High photosensitized singlet oxygen generating zinc(II) and indium(III) acetate phthalocyanines containing 6,8-di-tert-butyl-3-(p-oxyphenyl)coumarin groups. J. Photochem. Photobiol. A 2016, 317, 56–67. [Google Scholar] [CrossRef]
- Zhou, X.-Q.; Meng, L.-B.; Huang, Q.; Li, J.; Zheng, K.; Zhang, F.-L.; Liu, J.-Y.; Xue, J.-P. Synthesis and in vitro anticancer activity of zinc(II) phthalocyanines conjugated with coumarin derivatives for dual photodynamic and chemotherapy. Chem. Med. Chem. 2015, 10, 304–311. [Google Scholar] [CrossRef] [PubMed]
- Altun, S.; Altindal, A.; Bulut, M. Synthesis, characterization and dielectric properties of novel axial coumarin-substituted titanium(IV) phthalocyanines. Polyhedron 2013, 49, 41–49. [Google Scholar] [CrossRef]
- Barata, J.F.B.; Neves, M.G.P.M.S.; Faustino, M.A.F.; Tomé, A.C.; Cavaleiro, J.A.S. Strategies for corrole functionalization. Chem. Rev. 2017, 117, 3192–3253. [Google Scholar] [CrossRef] [PubMed]
- Tasior, M.; Gryko, D.T.; Pielacińska, D.J.; Zanelli, A.; Flamigni, L. Trans-A2B-corroles bearing a coumarin moiety—From synthesis to photophysics. Chem. Asian J. 2010, 5, 130–140. [Google Scholar] [CrossRef] [PubMed]
- Laha, J.K.; Dhanalekshmi, S.; Taniguchi, M.; Ambroise, A.; Lindsey, J.S. A scalable synthesis of meso-substituted dipyrromethanes. Org. Process Res. Dev. 2003, 7, 799–812. [Google Scholar] [CrossRef]
- Gryko, D.T.; Tasior, M.; Peterle, T.; Bröring, M. Meso-substituted corroles bearing peripheral donor sites. J. Porphyr. Phthalocyanines 2006, 10, 1360–1370. [Google Scholar] [CrossRef]
- Rao, P.D.; Dhanalekshmi, S.; Littler, B.J.; Lindsey, J.S. Rational syntheses of porphyrins bearing up to four different meso substituents. J. Org. Chem. 2000, 65, 7323–7344. [Google Scholar] [CrossRef]
- Koszarna, B.; Gryko, D.T. Efficient synthesis of meso-substituted corroles in a H2O–MeOH mixture. J. Org. Chem. 2006, 71, 3707–3717. [Google Scholar] [CrossRef]
- Tasior, M.; Voloshchuk, R.; Poronik, Y.M.; Rowicki, T.; Gryko, D.T. Corroles bearing diverse coumarin units—Synthesis and optical properties. J. Porphyr. Phthalocyanines 2011, 15, 1011–1023. [Google Scholar] [CrossRef]
- Bursa, B.; Barszcz, B.; Bednarski, W.; Lewtak, J.P.; Koszelewski, D.; Vakuliuk, O.; Gryko, D.T.; Wróbel, D. New meso-substituted corroles possessing pentafluorophenyl groups—Synthesis and spectroscopic characterization. Phys. Chem. Chem. Phys. 2015, 17, 7411–7423. [Google Scholar] [CrossRef]
- Santos, C.I.M.; Oliveira, E.; Barata, J.F.B.; Faustino, M.A.F.; Cavaleiro, J.A.S.; Neves, M.G.P.M.S.; Lodeiro, C. Corroles as anion chemosensors: Exploiting their fluorescence behaviour from solution to solid-supported devices. J. Mater. Chem. 2012, 22, 13811–13819. [Google Scholar] [CrossRef]
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).