Abstract
A novel one-flask synthetic method was developed in which 5-aminopyrazoles were reacted with N,N-substituted amides in the presence of PBr3. Hexamethyldisilazane was then added to perform heterocyclization to produce the corresponding pyrazolo[3,4-d]pyrimidines in suitable yields. These one-flask reactions thus involved Vilsmeier amidination, imination reactions, and the sequential intermolecular heterocyclization. To study the reaction mechanism, a series of 4-formyl-1,3-diphenyl-1H-pyrazol-5-yl-N,N-disubstituted formamidines, which were conceived as the chemical equivalent of 4-(iminomethyl)-1,3-diphenyl-1H-pyrazol-5-yl-formamidine, were prepared and successfully converted into pyrazolo[3,4-d]pyrimidines. The experiments demonstrated that the reaction intermediates were the chemical equivalents of 4-(iminomethyl)-1,3-diphenyl-1H-pyrazol-5-yl)formamidines. The rate of the reaction could be described as being proportional to the reactivity of amine reactants during intermolecular heterocyclization, especially when hexamethyldisilazane was used.
1. Introduction
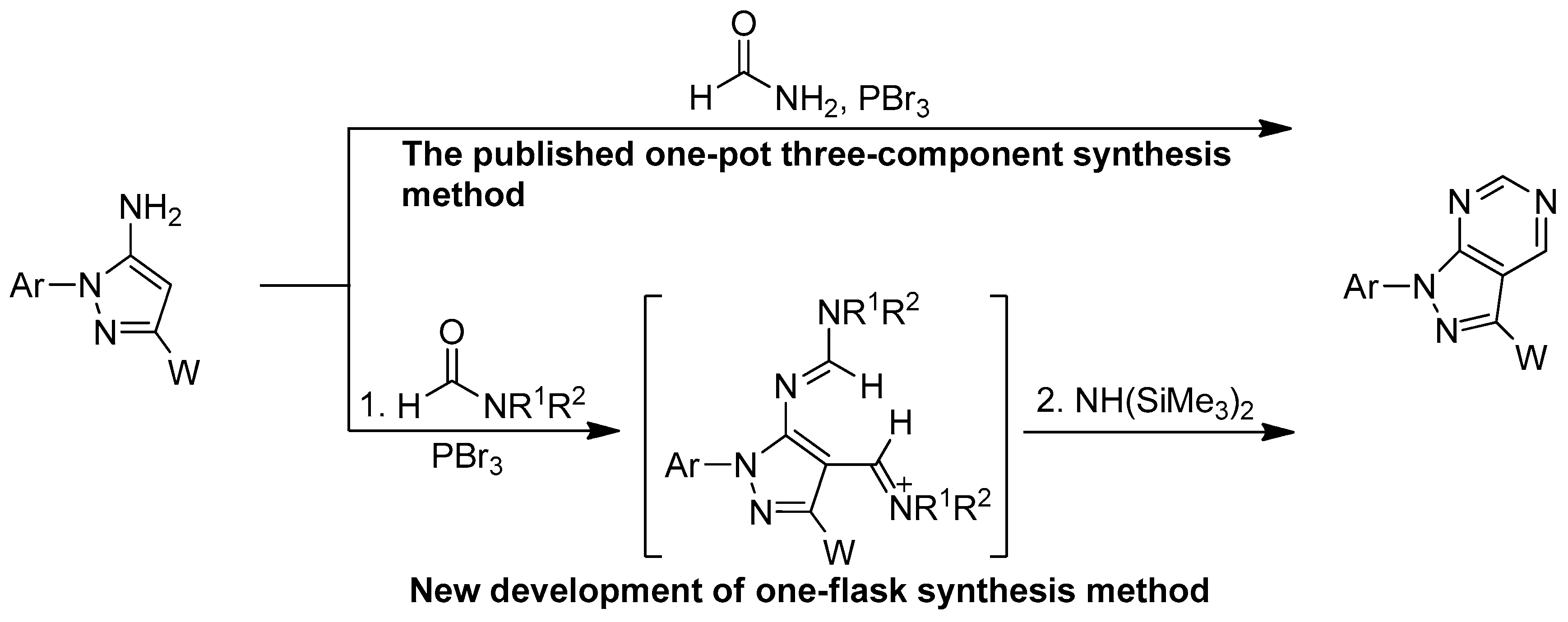
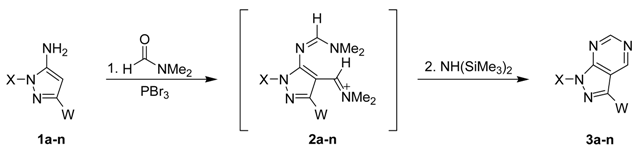
One-flask reactions possess significant advantages and have emerged as a powerful tool in synthetic organic chemistry and reaction design approaches [1,2,3]. The main advantages of using one-flask reactions in organic syntheses are their green chemistry nature and high atom economy due to the lack of workup or the isolation of intermediates involved [4,5,6,7,8,9,10,11,12]. We previously reported an efficient one-pot three-component synthesis of pyrazolo[3,4-d]pyrimidines that involved treatment of 5-aminopyrazoles with formamide using PBr3 as the coupling agent (Scheme 1) [13,14,15].
Scheme 1.
Synthesis of pyrazolo[3,4-d]pyrimidines by the different synthetic strategy via the Vilsmeier reaction and intramolecular or intermolecular heterocyclization.
Pyrazolopyrimidine derivatives are important structural moieties, found in pharmacologically active compounds such as a novel series of glucokinase activators [16], antibacterial [17], antifungal [18,19], antioxidant [20], antitumor [21,22,23,24], herbicidal [25], antivirus [26,27], anticancer compounds [28,29], and effective inhibitors of inflammatory mediators in intact cells [30,31]. The pyrazolo[3,4-d]pyrimidine core is also isomeric with the biologically significant purine system [32,33]. Numerous synthetic methods were developed for preparing pyrazolopyrimidine derivatives [34,35,36,37]. However, most of these methods are not straightforward and their purification steps are troublesome. Therefore, new and convenient routes for the synthesis of pyrazolo[3,4-d]pyrimidine systems have attracted considerable attention [13,14,15].
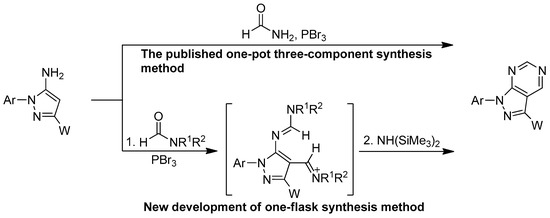
In this study, we extended our previous one-pot three-component approach for the synthesis of a series of pyrazolo[3,4-d]pyrimidine derivatives to develop a novel one-flask synthesis involving Vilsmeier amidination, imination reactions, and sequential intermolecular heterocyclization. First, 5-aminopyrazoles were treated with various Vilsmeier agents, which were generated from the corresponding amide solvents, including N,N-dimethylformamide (DMF), N,N-diethylformamide (DEF), N,N-diisopropylformamide, N,N-di-n-butylformamide, piperidine-1-carbaldehyde, and pyrrolidine-1-carbaldehyde in the presence of tribromophosphine PBr3, to produce the corresponding 4-(iminomethyl)-1,3-diphenyl-1H-pyrazol-5-yl-N,N-disubstituted formamidine intermediates (Scheme 1) [38,39]. Without isolating the intermediates, we sequentially evaluated the intermolecular heterocyclization reactivity between 4-(iminomethyl)-1,3-diphenyl-1H-pyrazol-5-yl-formamidines and amines such as hexamethyldisilazane, hexamethylenetetramine, lithium bis(trimethylsilyl)amine, and sodium bis(trimethylsilyl)amine. These experimental results revealed that commercially available N,N-dimethylformamide (DMF)/PBr3 and hexamethyldisilazane were the optimal Vilsmeier agent and the promotor, respectively. Specifically, we successfully combined the Vilsmeier amidination and imination reactions with intermolecular heterocyclization to design a high-efficiency one-flask synthesis for the preparation of a series of pyrazolo[3,4-d]pyrimidines.
2. Results and Discussion
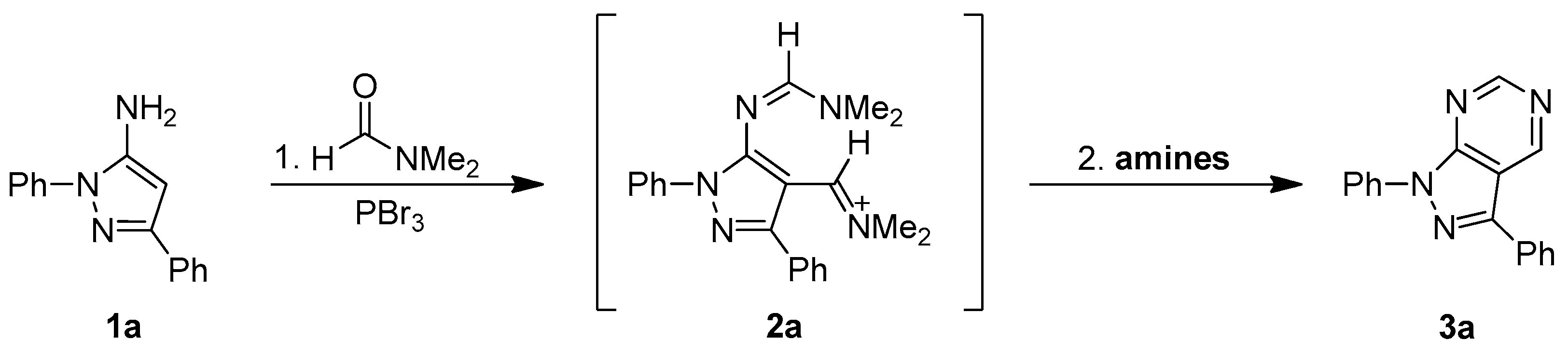
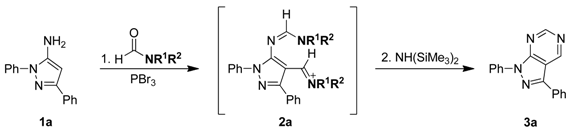
To optimize the one-flask process for the synthesis of pyrazolo[3,4-d]pyrimidine derivatives 3a–n via the sequential Vilsmeier reaction and intermolecular heterocyclization and explain the mechanism the study illustrated in Scheme 2 was performed. 5-Amino-1,3-diphenylpyrazole (1a) was prepared by our previously developed method [38,39] and used as the model starting material to improve the intermolecular heterocyclization reaction conditions. Following the reliable published procedure for the Vilsmeier reaction 5-aminopyrazole 1a was treated with 3.0 equivalent of PBr3 in N,N-dimethylformamide (DMF) solution at 60 °C for 1.0–2.0 h. The corresponding 4-(iminomethyl)-1,3-diphenyl-1H-pyrazol-5-yl-formamidine 2a was thus obtained in excellent yield (>90%).
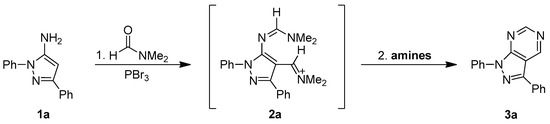
Scheme 2.
The newly developed one-flask for synthesis of pyrazolo[3,4-d]pyrimidine derivatives via Vilsmeier reaction and the sequential heterocyclization.
Without isolation of intermediate 2a, various amines, including hexamethyldisilazane (NH(SiMe3)2), hexamethylenetetramine, lithium bis(trimethylsilyl)amine (LiN(SiMe3)2), and sodium bis(trimethylsilyl)amine (NaN(SiMe3)2) were added into the reaction mixture and the solution was heated at reflux for 3–5 h to establish the best heterocyclization conditions (see Table 1). Without the amine agent, only 4-formyl-1,3-diphenyl-1H-pyrazol-5-yl-formamidine 7a, which is the chemical equivalent of 4-(iminomethyl)-1,3-diphenyl-1H-pyrazol-5-yl-formamidine intermediate 2a, was isolated after work-up and purification (see entry 1 in Table 1). Among the amines, the corresponding pyrazolo[3,4-d]pyrimidine product 3a can be produced and isolated in yields ranging from 26% to 91%. Based on the results, we found that commercially available hexamethyldisilazane (NH(SiMe3)2) provided the best result (91% yield) and the reactivity tendency of the amines was NH(SiMe3)2 > NaN(SiMe3)2 > LiN(SiMe3)2 > hexamethylenetetramine (see the Entries 2–5 in Table 1). We next tried different amounts of NH(SiMe3)2, including 1.0, 2.0, 3.0, and 4.0 equivalents. The corresponding pyrazolo[3,4-d]pyrimidine product 1a was obtained in 56–91% yield, with the best yield (91%) corresponding to 3 equivalents of (NH(SiMe3)2) (see the Entries 5 and 6–8 in Table 1). Consequently, we believe that 3.0 equivalent of NH(SiMe3)2 is the optimum amount for our reaction conditions.

Table 1.
The study of amine agents in the one-flask for synthesis of pyrazolo[3,4-d]pyrimidines.
To determine the reactivity of the different Vilsmeier agents (HC(O)NR1R2 + PBr3), we used different amide solvents, including N,N-dimethylformamide (DMF), N,N-diethylformamide (DEF), N,N-diisopropylformamide, N,N-di-n-butylformamide, piperidine-1-carbaldehyde, and pyrrolidine-1-carbaldehyde in the presence of 3.0 equivalent of PBr3 to prepare the corresponding types of Vilsmeier reagent. Compound 1a was allowed to react sequentially with these different Vilsmeier reagents at 60 °C for 1.0–2.0 h. When the starting material 1a was fully consumed, 3.0 equivalents of NH (SiMe3)2 were added to the reaction mixture which was heated at reflux for 3.0–5.0 h. After the work-up and purification, the corresponding pyrazolo[3,4-d]pyrimidine 3a was obtained in 56–91% yields (see Table 2). Based on the study, commercially available DMF was the best solvent for the preparation of the Vilsmeier reagent in this new one-flask procedure. Based on our optimized experimental results, we believe the most reliable procedure for the one-flask synthesis of pyrazole[3,4-d]pyrimidines involves the treatment of 5-aminopyrazole 1a with 3.0 equivalent of PBr3 in DMF solution at 60 °C for 1.0–2.0 h. When the Vilsmeier reaction was completed, the resulting mixture was added with 3.0 equivalents of NH(SiMe3)2 then heated at reflux at 70 °C to 80 °C for 3.0–5.0 h (monitored by TLC). After work-up and purification by chromatography, the corresponding pyrazolo[3,4-d]pyrimidine 3a was obtained in excellent yield (91%, see Table 2).

Table 2.
The study of the reactivity of the different Vilsmeier agents in the one-flask synthesis of pyrazole[3,4-d]pyrimidines.
Application of the optimized one-flask inter-heterocyclization procedure to 5-amino-1,3-disubstituted pyrazoles 1b–i bearing various N1 substituents, including o-Me-Ph, o-Cl-Ph, m-Me-Ph, m-Cl-Ph, m-NO2-Ph, p-Me-Ph, p-Cl-Ph, and p-Br-Ph, also proceeded smoothly to give the corresponding pyrazolo[3,4-d]pyrimidines 3a–i in 78–91% yields (see Table 3).

Table 3.
The results of the one-flask synthesis of pyrazolo[3,4-d]pyrimidines from 5-aminopyrazoles, DMF/PBr3 and NH(SiMe3)2.
For further investigation of the effect of the substituent on the C-3 of the pyrazole ring, the same conditions were employed with 5-amino-1-phenyl-3-substituted pyrazoles 1j–n that contained methyl, t-butyl, p-Me-Ph, p-Cl-Ph, or p-OMe-Ph groups at the C-3 position of the pyrazole ring. The reaction also proceeded smoothly gave the corresponding products 3j–n in 79–91% yields (see Table 3). All pyrazolo[3,4-d]pyrimidines 3a–n were fully characterized by spectroscopic methods and the physical properties and spectroscopic characteristics of the pyrazolo[3,4-d]pyrimidines 3a–n were consistent with our published data [13,14,15].
For further comparison of the reactivity between this new intermolecular Vilsmeier heterocyclization and the previously published intramolecular heterocyclization method [13,14,15], 5-aminopyrazoles were treated with formamide/PBr3. Based on the results of Table 3, the corresponding pyrazolopyrimidines 3a–n were obtained in 78–91% yields by the intermolecular heterocyclization route and in 87–96% yields by intramolecular heterocyclization, respectively. The data suggests that the intramolecular heterocyclization is more favorable as it provided the better isolated yields.
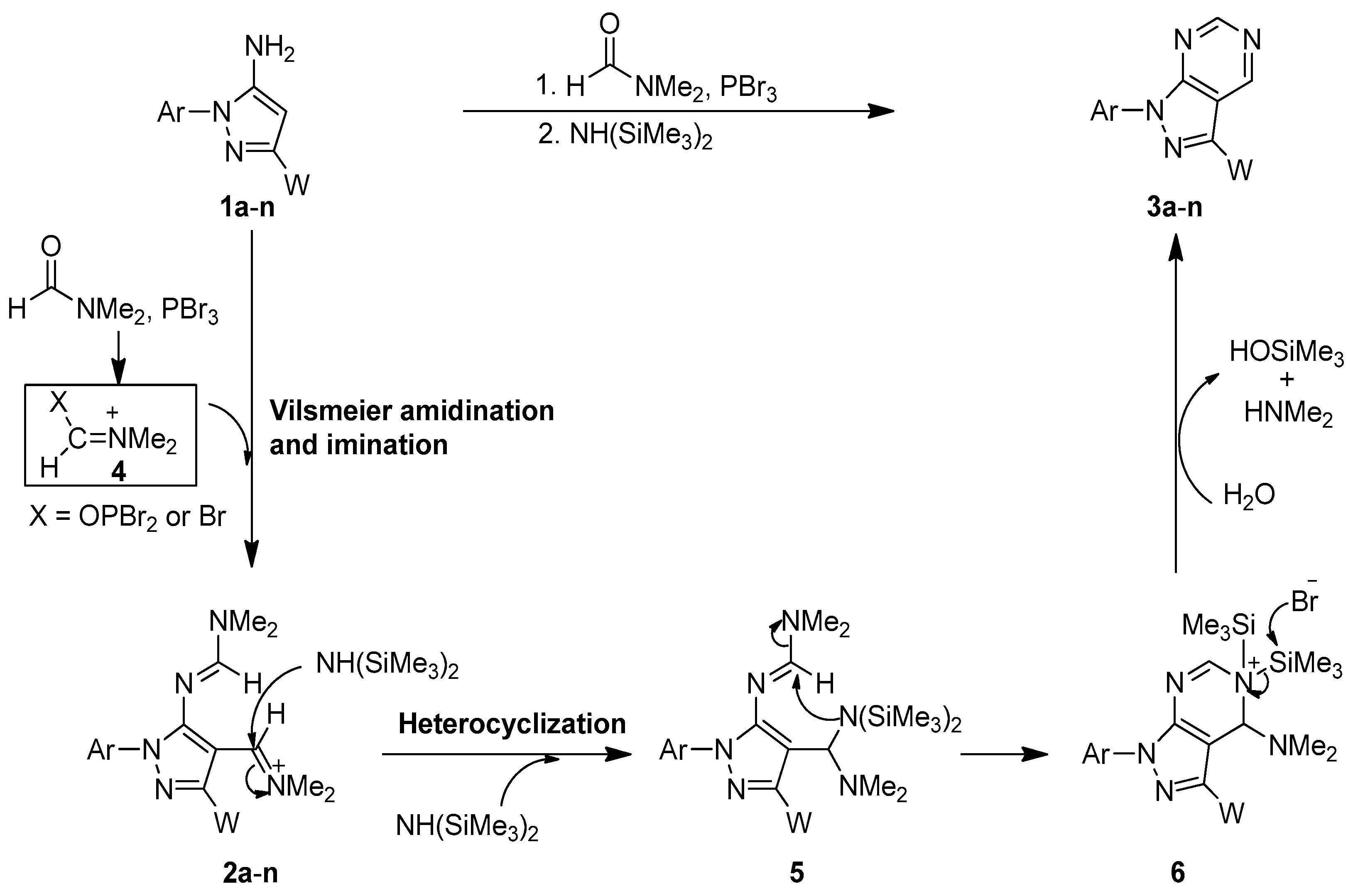
We propose a plausible mechanism for the newly developed one-flask cascade for synthesis of pyrazolo [3,4-d]pyrimidines as shown in Scheme 3. Initiallly, N,N-dimethylformamide (DMF) reacted with the coupling agent PBr3 to form the Vilsmeier reactive species 4 in situ [40,41,42,43,44]. Sequentially, 5-amino-1,3-disubstituted pyrazoles 1a–n reacted with the reactive species 4 to undergo the amidination and imination reaction to give the 1H-pyrazol-5-yl-N,N-disubstituted formamidine intermediates 2a–n (see Scheme 3). When the Vilsmeier reaction was complete (by monitoring TLC), NH(SiMe3)2 was directly added into the reaction mixture to perform the substitution reaction with the imino group to generate intermediate 5. A sequential intermolecular heterocyclization reaction then took place to produce intermediate 6. After the desilylation reaction occurred caused by bromide anion and water, the corresponding pyrazolo[3,4-d]pyrimidines 3a–n were obtained in good yields.
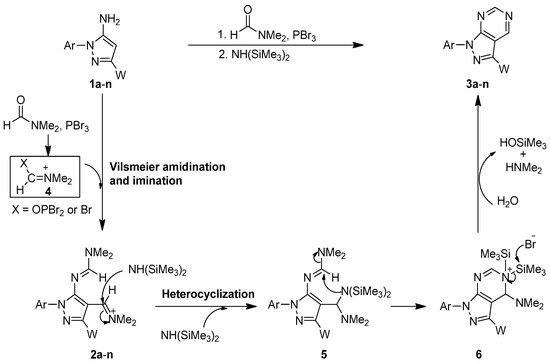
Scheme 3.
A plausible mechanism for the newly developed one-flask procedure for the synthesis of pyrazolo[3,4-d]pyrimidines.
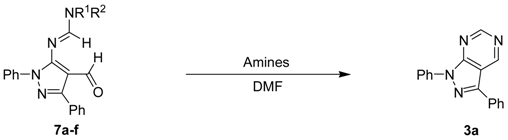
To further study the mechanism, 4-formyl-1,3-diphenyl-1H-pyrazol-5-yl-N,N-dimethyl formamidine 7a was synthesized [20] and reacted with various amines including NH(SiMe3)2, hexamethylenetetramine, LiN(SiMe3)2, and NaN(SiMe3)2, to carry out the intermolecular heterocyclization. The heterocyclization was successfully and smoothly underwent to give pyrazole[3,4-d]pyrimidine product 3a in 37–91% yields. Particularly, NH(SiMe3)2 was most efficient base for heterocyclization to afford the desired product in 91% yield (see Entry 4 in Table 4). The similar reactivity tendency of heterocyclization was observed in this study: NH(SiMe3)2 > NaN(SiMe3)2 > LiN(SiMe3)2 > hexamethylenetetramine (see Entries 1–4 in Table 4). 4-formyl-1,3-diphenyl-1H-pyrazol-5-yl-N,N-disubtituted formamidines 7b–e with grafting the different amino-substituent on amidinyl groups, such as NEt2, N(i-Pr)2, N(n-Bu)2, and piperidinyl, were then allowed to reacted with NH(SiMe3)2 in DMF solution at reflux to give the corresponding pyrazolo[3,4-d]pyrimidine 3a for the investigation of the reactivity of substrates. Based on the experimental result, among of starting substrates 7b–e displayed the good to excellent reactivity in heterocyclization, except for 7c possessing the bulky N (i-Pr)2 substituent moiety on amidinyl groups (see Entries 1 and 5–8 in Table 4). Furthermore, 4-formyl-1,3-diphenyl-1H-pyrazol-5-yl-N,N-dimethyl formamidine 7a with the NMe2 substituent on amidinyl groups was the best suitable reactant in the intermolecular heterocyclization (91%, see Table 4). The above results also gave more proof to our proposed mechanism, for example, the new one-flask reaction would take place through 4-(iminomethyl)-1H-pyrazol-5-yl-formamidine intermediates 2a–n. On the other hands, the commercial available N,N-dimethylformamide (DMF) in the presence of PBr3 and hexamethyldisilazane were the best Vilsmeier agent and the promoted cyclization base.

Table 4.
The mechanistic study for the intermolecular heterocyclization from 4-formyl-1,3-disubstituted-1H-pyrazol-5-yl-formamidines 7a–n with various amines.
3. Experimental Section
3.1. General Information
All chemicals were reagent grade and used as purchased. All reactions were carried out under nitrogen atmosphere and monitored by TLC analysis. Flash column chromatography purification of compounds was carried out by gradient elution using hexanes in ethyl acetate (EA) unless otherwise stated. Commercially available reagents were used without further purification unless otherwise noted. 1H-NMR were recorded at 200, 400, or 500 MHz and 13C-NMR recorded at 50, 100, or 125 MHz, respectively, in CDCl3, CH3OD, and DMSO-d6 as solvent (see supplementary materials). The standard abbreviations s, d, t, q, and m refer to singlet, doublet, triplet, quartet, and multiplet, respectively. Coupling constant (J), whenever discernible, have been reported in Hz. Infrared spectra (IR) were recorded as neat solutions or solids; and mass spectra were recorded using electron impact or electrospray ionization techniques. The wavenumbers reported are referenced to the polystyrene 1601 cm–1 absorption. High-resolution mass spectra were obtained by means of a JMS-HX110 mass spectrometer (JEOL, Tokyo, Japan).
3.2. Standard Procedure for the Synthesis of Pyrazolo[3,4-d]pyrimidines 3a–n
The optimized procedure involved the treatment of 5-aminopyrazoles 1a–n (1.0 equiv) with PBr3 (~3 equiv.) in various amide solutions including N,N-dimethylformamide (DMF), N,N-diethylformamide (DEF), N,N-diisopropylformamide, N,N-di-n-butylformamide, piperidine-1-carbaldehyde, or pyrrolidine-1-carbaldehyde (2 mL) at 50–60 °C for 1.0–2.0 h. When the reaction was completed (as monitored by TLC), an amine such as hexamethyldisilazane (NH(SiMe3)2), hexamethylenetetramine, lithium bis(trimethylsilyl)amine (LiN(SiMe3)2), or sodium bis(trimethylsilyl)amine (NaN(SiMe3)2) was added into the reaction mixture which was stirred at reflux for 3–5 h. When the intermolecular heterocyclization was complete, the resulting mixture was added to saturated sodium bicarbonate (15 mL) and extracted with dichloromethane (15 mL × 2). The organic extracts were dried over MgSO4, filtered, and concentrated under reduced pressure. The residue was purified by column chromatography on silica gel to give the corresponding pyrazolo[3,4-d]pyrimidines 3a–n in 69–91% yields.
1,3-Diphenyl-1H-pyrazolo[3,4-d]pyrimidine (3a) [13,14,15,45]. Light-yellow solid; m.p. 158–159 °C (hexane–EtOAc). 1H-NMR (CDCl3, 400 MHz): δ 7.34–7.38 (1H, m, ArH), 7.48–7.51 (1H, m, ArH), 7.53–7.57 (4H, m, ArH), 8.06 (2H, d, J = 8.00 Hz, ArH), 8.31 (2H, d, J = 8.00 Hz, ArH), 9.12 (1H, s), 9.51 (1H, s). 13C-NMR (CDCl3, 100 MHz,): δ 114.24, 121.46 (2 × C), 126.86, 127.39 (2 × C), 129.24 (4 × C), 129.64, 131.50, 138.50, 145.00, 152.82, 153.34, 155.61. IR (KBr): 1632, 1586, 1554, 1497, 1366, 1219 cm–1. EIMS m/z: 272 (M+, 100), 273 (18), 271 (31), 142 (11), 77 (34), 69 (24), 51 (11).
1-(2-Methylphenyl)-3-phenyl-1H-pyrazolo[3,4-d]pyrimidine (3b) [13,14,15]. Light-yellow solid; m.p. 140–141 °C (hexane–EtOAc). 1H-NMR (CDCl3, 400 MHz): δ 2.48 (3H, s, CH3), 7.18 (1H, d, J = 7.60 Hz, ArH), 7.42–7.45 (1H, m, ArH), 7.50 (1H, d, J = 8.00 Hz, ArH), 7.54–7.58 (2H, m, ArH), 8.05–8.10 (2H, m, ArH), 9.12 (1H, s), 9.50 (1H, s). 13C-NMR (CDCl3, 100 MHz): δ 21.6 (CH3), 114.18, 118.77, 122.17, 127.42 (2 × C), 127.77, 129.06, 129.23 (2 × C), 129.61, 131.55, 138.38, 139.33, 144.92, 152.80, 153.32, 155.60. IR (KBr): 1636, 1497, 1223, 1096 cm–1. EIMS m/z: 286 (M+, 100), 287 (20), 285 (19), 77 (10).
1-(2-Chlorophenyl)-3-phenyl-1H-pyrazolo[3,4-d]pyrimidine (3c) [13,14,15]. Yellow solid; m.p. 139–140 °C (hexane–EtOAc). 1H-NMR (CDCl3, 400 MHz): δ 7.48–7.51 (3H, m, ArH), 7.53–7.57 (2H, m, ArH), 7.60–7.64 (2H, m, ArH), 8.05 (2H, d, J = 8.00 Hz, ArH), 9.08 (1H, s), 9.54 (1H, s). 13C-NMR (CDCl3, 100 MHz): δ 113.01, 125.54, 127.38 (2 × C), 127.72, 128.38, 129.65 (2 × C), 130.08, 130.82, 131.40, 132.19, 134.74, 145.70, 152.87, 154.50, 155.91. IR (KBr): 3012, 1636, 1582, 1497, 1362, 1223, 1084 cm–1. EIMS m/z: 306 (M+, 96), 308 (28), 307 (M+ + 1, 15), 272 (15), 271 (100), 195 (11), 77 (42), 75 (10), 51 (11).
1-(3-Methylphenyl)-3-phenyl-1H-pyrazolo[3,4-d]pyrimidine (3d) [13,14,15]. Yellow solid; m.p. 80–81 °C (hexane–EtOAc). 1H-NMR (CDCl3, 400 MHz): δ 2.52 (3H, s, CH3), 7.18 (1H, d, J = 8.00 Hz, ArH), 7.41–7.45 (1H, m, ArH), 7.49 (1H, d, J = 7.20 Hz, ArH), 7.53–7.57 (2H, m, ArH), 8.05–8.10 (4H, m, ArH), 9.16 (1H, s), 9.53 (1H, s). 13C-NMR (CDCl3, 100 MHz): δ 21.60, 114.15, 118.70, 122.11, 127.38 (2 × C), 127.72, 129.02, 129.19 (2 × C), 129.57, 131.52, 138.36, 139.28, 144.86, 152.75, 153.27, 155.55. IR (KBr): 1632, 1613, 1585, 1493, 1420, 1366, 1265 cm–1. EIMS m/z: 286 (M+, 100), 287 (22), 285 (21), 77 (9).
1-(3-Chlorophenyl)-3-phenyl-1H-pyrazolo[3,4-d]pyrimidine (3e) [13,14,15]. White solid; m.p. 185–186 °C (hexane–EtOAc). 1H-NMR (CDCl3, 400 MHz): δ 7.31 (1H, d, J = 8.00 Hz, ArH), 7.44–7.50 (2H, m, ArH), 7.52–7.57 (2H, m, ArH), 8.32–8.42 (2H, m, ArH), 9.14 (1H, s), 9.49 (1H, s). 13C-NMR (CDCl3, 100 MHz): δ 114.51, 118.90, 121.07, 126.61, 127.40 (2 × C), 129.25 (2 × C), 129.85, 130.21, 131.17, 134.95, 139.58, 145.62, 152.88, 153.61, 155.73. IR (KBr): 1585, 1555, 1489, 1404, 1366, 1312, 1215, 1088 cm–1. EIMS m/z: 306 (M+, 100), 308 (32), 307 (M+ + 1, 26), 305 (22), 77 (16).
1-(3-Nitrophenyl)-3-phenyl-1H-pyrazolo[3,4-d]pyrimidine (3f) [13,14,15]. Yellow solid; m.p. 179–180 °C (hexane–EtOAc). 1H-NMR (CDCl3, 400 MHz): δ 7.54–7.61 (3H, m, ArH), 7.73 (1H, dd, J = 8.0, 16.4 Hz, ArH), 8.08 (2H, d, J = 8.00 Hz, ArH), 8.19 (1H, d, J = 8.00 Hz, ArH), 8.87 (1H, d, J = 8.00 Hz, ArH), 9.20 (1H, s), 9.37 (1H, s), 9.55 (1H, s). 13C-NMR (CDCl3, 100 MHz): δ 114.74, 115.70, 120.87, 126.04, 127.51 (2 × C), 129.36 (2 × C), 130.16, 130.18, 130.88, 139.64, 146.08, 148.87, 153.14, 154.00, 156.05. IR (KBr): 1636, 1528, 1489, 1346, 1003 cm–1. EIMS m/z: 317 (M+, 100), 318 (17), 77 (14).
1-(4-Methylphenyl)-3-phenyl-1H-pyrazolo[3,4-d]pyrimidine (3g) [13,14,15]. Brown solid; m.p. 133–134 °C (hexane–EtOAc). 1H-NMR (CDCl3, 400 MHz): δ 2.39 (3H, s, CH3), 7.30 (2H, d, J = 8.00 Hz, ArH), 7.45 (1H, d, J = 8.00 Hz, ArH), 7.50 (2H, dd, J = 7.2, 14.8 Hz, ArH), 8.00 (2H, d, J = 8.00 Hz, ArH), 8.11 (2H, d, J = 8.00 Hz, ArH), 9.07 (1H, s), 9.43 (1H, s). 13C-NMR (CDCl3, 100 MHz): δ 20.98, 113.92, 121.21 (2 × C), 127.18 (2 × C), 128.76 (2 × C), 129.37, 129.61 (2 × C), 131.43, 135.94, 136.56, 144.47, 152.58, 152.95, 155.33. IR (KBr): 1636, 1589, 1516, 1386, 1219, 1088 cm–1. EIMS m/z: 286 (M+, 100), 287 (22), 285 (28), 77 (10). HRMS Calcd. for C18H14N4: 286.1218; Found: 286.1216.
1-(4-Chlorophenyl)-3-phenyl-1H-pyrazolo[3,4-d]pyrimidine (3h) [13,14,15]. Yellow solid; m.p. 147–148 °C (hexane–EtOAc). 1H-NMR (CDCl3, 400 MHz): δ 7.48–7.51 (3H, m, ArH), 7.53–7.57 (2H, m, ArH), 8.03 (2H, d, J = 8.00 Hz, ArH), 8.32 (2H, d, J = 8.00 Hz, ArH), 9.11 (1H, s), 9.49 (1H, s). 13C-NMR (CDCl3, 100 MHz): δ 114.32, 122.24 (2 × C), 127.37 (2 × C), 129.23 (2 × C), 129.28 (2 × C), 129.78, 131.24, 132.11, 137.13, 145.23, 152.89, 153.36, 155.66. IR (KBr): 1632, 1555, 1497, 1215, 1054 cm–1. EIMS m/z: 306 (M+, 100), 308 (31), 307 (M+ + 1, 23), 305 (17), 77 (14).
1-(4-Bromophenyl)-3-phenyl-1H-pyrazolo[3,4-d]pyrimidine (3i) [13,14,15]. Yellow solid; m.p. 180–181 °C (hexane–EtOAc). 1H-NMR (CDCl3, 400 MHz): δ 7.51–7.59 (3H, m, ArH), 7.66 (2H, d, J = 8.40 Hz, ArH), 8.05 (2H, d, J = 8.00 Hz, ArH), 8.29 (2H, d, J = 8.00 Hz, ArH), 9.13 (1H, s), 9.51 (1H, s). 13C-NMR (CDCl3, 100 MHz): δ 114.37, 119.99, 122.53 (2 × C), 127.37 (2 × C), 129.23 (2 × C), 129.79, 131.22, 132.24 (2 × C), 137.63, 145.29, 152.88, 153.40, 155.67. IR (KBr): 1586, 1555. 1481, 1400, 1389, 1215, 1072 cm–1. EIMS m/z: 350 (M+, 100), 352 (M+ + 2, 99), 353 (15), 351 (27), 194 (14), 77 (30).
3-Methyl-1-phenyl-1H-pyrazolo[3,4-d]pyrimidine (3j) [13,14,15,46]. Brown solid; m.p. 77–78 °C (hexane–EtOAc). 1H-NMR (CDCl3, 400 MHz): δ 2.70 (3H, s, CH3), 7.32 (1H, dd, J = 7.20, 14.80 Hz, ArH), 7.50–7.79 (2H, dd, J = 7.60, 15.60 Hz, ArH), 8.19 (2H, d, J = 8.00 Hz, ArH), 9.07 (1H, s), 9.16 (1H, s). 13C-NMR (CDCl3, 100 MHz): δ 12.59, 115.79, 121.07 (2 × C), 126.49, 129.22 (2 × C), 138.50, 143.35, 151.77, 152.77, 155.70. IR (KBr): 3240, 1643, 1503, 1439, 1211 cm–1. EIMS m/z: 210 (M+, 100), 211 (16), 209 (27), 195 (13), 142 (15), 77 (37), 69 (11), 57 (16), 55 (13), 51 (13).
3-tert-Butyl-1-phenyl-1H-pyrazolo[3,4-d]pyrimidine (3k) [13,14,15]. Yellow solid; m.p. 45–46 °C (hexane–EtOAc). 1H-NMR (CDCl3, 400 MHz): δ 1.57 (9H, s, t-Bu), 7.28–7.32 (1H, m, ArH), 7.51 (2H, dd, J = 7.60, 15.60 Hz, ArH), 8.22 (2H, d, J = 8.00 Hz, ArH), 9.04 (1H, s), 9.32 (1H, s). 13C-NMR (CDCl3, 100 MHz): δ 30.05 (3 × C), 34.51, 114.02, 121.17 (2 × C), 126.33, 129.13 (2 × C), 138.66, 152.84, 153.16, 154.88, 155.04. IR (KBr): 3048, 2967, 2666, 1636, 1578, 1508, 1427, 1366, 1188, 1096 cm–1. EIMS m/z: 252 (M+, 43), 238 (18), 237 (100), 222 (12), 105(11), 77(17), 57(11).
3-(4-Methylphenyl)-1-phenyl-1H-pyrazolo[3,4-d]pyrimidine (3l) [13,14,15]. White solid; m.p. 138–139 °C (hexane–EtOAc). 1H-NMR (CDCl3, 400 MHz): δ 2.44 (3H, s, CH3), 7.36 (3H, d, J = 6.80 Hz, ArH), 7.55 (2H, dd, J = 8.00, 16.00 Hz, ArH), 7.95 (2H, d, J = 8.00 Hz, ArH), 8.30 (2H, d, J = 8.00 Hz, ArH), 9.12 (1H, s), 9.49 (1H, s). 13C-NMR (CDCl3, 100 MHz): δ 21.39, 114.24, 121.41 (2 × C), 126.74, 127.23 (2 × C), 128.64, 129.18 (2 × C), 129.87 (2 × C), 138.51, 139.77, 145.06, 152.79, 153.27, 155.52. IR (KBr): 3117, 1582, 1501, 1223, 1092 cm–1. EIMS m/z: 286 (M+, 100), 287 (21), 285 (26).
3-(4-Chlorophenyl)-1-phenyl-1H-pyrazolo[3,4-d]pyrimidine (3m) [13,14,15,45]. Light-yellow solid; m.p. 194–193 °C (hexane–EtOAc). 1H-NMR (CDCl3, 400 MHz): δ 7.37 (1H, dd, J = 7.60, 15.20 Hz, ArH), 7.51–7.57 (m, 4 H, ArH), 8.00 (2H, d, J = 8.00 Hz, ArH), 8.28 (2H, d, J = 8.00 Hz, ArH), 9.13 (1H, s), 9.47 (1H, s). 13C-NMR (CDCl3, 100 MHz): δ 114.03, 121.47 (2 × C), 127.01, 128.51 (2 × C), 129.27 (2 × C), 129.48 (2 × C), 129.98, 135.71, 138.43, 143.85, 152.60, 153.37, 155.69. IR (KBr): 1632, 1555, 1504, 1404, 1219, 1092 cm–1. EIMS m/z: 306 (M+, 100), 308 (33), 307 (M+ + 1, 26), 305 (22), 77 (10).
3-(4-Methoxylphenyl)-1-phenyl-1H-pyrazolo[3,4-d]pyrimidine (3n) [13,14,15]. Light-yellow solid; m.p. 169–170 °C (hexane–EtOAc). 1H-NMR (CDCl3, 400 MHz): δ 3.86 (3H, s, OCH3), 7.04 (2H, d, J = Hz, ArH), 7.33 (1H, dd, J = 7.60, 14.80 Hz, ArH), 7.52 (2H, dd, J = 7.60, 15.60 Hz, ArH), 7.96 (2H, d, J = 8.00 Hz, ArH), 8.28 (2H, d, J = 8.00 Hz, ArH), 9.08 (1H, s), 9.43 (1H, s). 13C-NMR (CDCl3, 100 MHz): δ 55.36 (OCH3), 114.13, 114.57 (2 × C), 121.26 (2 × C), 124.01, 126.61, 128.61 (2 × C), 129.14 (2 × C), 138.51, 144.74, 152.66, 153.18, 155.43, 160.72. IR (KBr): 3059, 1632, 1613, 1528, 1501, 1431, 1362, 1300, 1258, 1219, 1173, 1092 cm–1. EIMS m/z: 302 (M+, 100), 303 (22), 287 (23), 77 (15).
4. Conclusions
We have successfully developed the one-flask method to synthesize pyrazolo[3,4-d]pyrimidines by treating 5-amino-pyrazoles, in presence of PBr3 coupling agent and then hexamethyldisilazane. In this new one-flask reaction was contained Vilsmeier reaction and the sequential intermolecular heterocyclization two steps. Based on the improved studies of the different type of Vilsmeier agents and amines, we found the commercial available DMF/PBr3 and hexamethyldisilazane were the best Vilsmeier agent and the efficient base for this newly developed one-flask synthesis. For the mechanistic study, 4-(iminomethyl)-1,3-diphenyl-1H-pyrazol-5-yl-N,N-disubstituted formamidines were demonstrated as the reaction intermediates by using a series of 4-formyl-1,3-diphenyl-1H-pyrazol-5-yl-N,N-disubstituted formamidines successfully reacted with amines to give pyrazolo[3,4-d]pyrimidines due to they were conceived as the chemical equivalent species. On the other hands, the order of reactivity of amines in intermolecular heterocyclization was NH(SiMe3)2 > NaN(SiMe3)2 > LiN(SiMe3)2 > hexamethylenetetramine. Through the further comparison variation reactive study between intramolecular and intermolecular Vilsmeier heterocyclization reaction, we found the intramolecular heterocyclization be able to provide the better results.
Supplementary Materials
Supplementary materials are available online.
Acknowledgments
We are grateful to the Tsuzuki Institute for Traditional Medicine and the Ministry of Science and Technology of the Republic of China (MOST 105-2113-M-039-001) for financial support.
Author Contributions
Fung Fuh Wong conceived and designed the experiments; Wan-Ping Yen and Shuo-En Tsai performed the experiments; Naoto Uramaru, Hiroyuki Takayama, Shuo-En Tsai, and Wan-Ping Yen analyzed the data; Fung Fuh Wong, Hiroyuki Takayama, and Naoto Uramaru contributed reagents/materials/analysis tools; Fung Fuh Wong, Wan-Ping Yen, and Shuo-En Tsai wrote the paper.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Ugi, I.; Dömling, A.; Werner, B. Since 1995 the new chemistry of multicomponent reactions and their libraries, including their heterocyclic chemistry. J. Heterocycl. Chem. 2000, 37, 647–658. [Google Scholar] [CrossRef]
- Posner, G.H. Multicomponent one-pot annulations forming 3 to 6 bonds. Chem. Rev. 1986, 86, 831–844. [Google Scholar] [CrossRef]
- Weber, L.; Illgen, K.; Almstetter, M. Discovery of new multi component reactions with combinatorial methods. Synlett 1999, 366–374. [Google Scholar] [CrossRef]
- Fogg, D.E.; dos Santos, E.N. Tandem catalysis: A taxonomy and illustrative review. Coord. Chem. Rev. 2004, 248, 2365–2379. [Google Scholar] [CrossRef]
- Shindoh, N.; Takemoto, Y.; Takasu, K. Auto-Tandem Catalysis: A Single Catalyst Activating Mechanistically Distinct Reactions in a Single Reactor. Chem. Eur. J. 2009, 15, 12168–12179. [Google Scholar] [CrossRef] [PubMed]
- Yin, G.D.; Zhou, B.H.; Meng, X.G.; Wu, A.X.; Pan, Y.J. Efficient C−C Double-Bond Formation Reaction via a New Synthetic Strategy: A Self-Sorting Tandem Reaction. Org. Lett. 2006, 8, 2245–2248. [Google Scholar] [CrossRef] [PubMed]
- Gao, M.; Yang, Y.; Wu, Y.D.; Cong, C.; Shu, W.M.; Zhang, D.X.; Cao, L.P.; She, N.F.; Wu, A.X. An efficient synthesis of hydantoins via sustainable integration of coupled domino processes. Org. Lett. 2010, 12, 4026–4029. [Google Scholar] [CrossRef] [PubMed]
- Zhu, Y.P.; Gao, Q.H.; Lian, M.; Yuan, J.J.; Liu, M.C.; Zhao, Q.; Yang, Y.; Wu, A.X. A sustainable byproduct catalyzed domino strategy: Facile synthesis of a-formyloxy and acetoxy ketones via iodination/nucleophilic substitution/hydrolyzation/oxidation sequences. Chem. Commun. 2011, 47, 12700–12702. [Google Scholar] [CrossRef] [PubMed]
- Allenn, A.E.; MacMillan, D.W.C. Synergistic catalysis: A powerful synthetic strategy for new reaction development. Chem. Sci. 2012, 3, 633–658. [Google Scholar] [CrossRef] [PubMed]
- Harschneck, T.; Kirsch, S.F. One-pot synthesis of 1, 2-Dihydropyridines: Expanding the diverse reactivity of propargyl vinyl ethers. J. Org. Chem. 2011, 76, 2145–2156. [Google Scholar] [CrossRef] [PubMed]
- Zhu, Y.P.; Jia, F.C.; Liu, M.C.; Wu, A.X. A multipathway coupled domino strategy: Metal-free oxidative cyclization for one-pot synthesis of 2-acylbenzothiazoles from multiform substrates. Org. Lett. 2012, 14, 4414–4417. [Google Scholar] [CrossRef] [PubMed]
- Guilarte, V.; Fernández-Rodríguez, M.A.; Garcría-Garcría, P.; Hernando, E.; Sanz, R. A Practical, One-Pot Synthesis of Highly Substituted Thiophenes and Benzo[b]thiophenes from Bromoenynes and o-Alkynylbromobenzenes. Org. Lett. 2011, 13, 5100–5103. [Google Scholar] [CrossRef] [PubMed]
- Chang, C.H.; Tsai, H.J.; Huang, Y.Y.; Lin, H.Y.; Wang, L.Y.; Wu, T.S.; Wong, F.F. Selective synthesis of pyrazolo[3,4-d]pyrimidine, N-(1H-pyrazol-5-yl)formamide, or N-(1H-pyrazol-5-yl)formamidine derivatives from N-1-substituted-5-aminopyrazoles with new Vilsmeier-type reagents. Tetrahedron 2013, 69, 1378–1386. [Google Scholar] [CrossRef]
- Huang, Y.Y.; Wang, L.Y.; Chang, C.H.; Kuo, Y.H.; Kaneko, K.; Takayama, H.; Kimura, M.; Juang, S.H.; Wong, F.F. One-pot synthesis and antiproliferative evaluation of pyrazolo[3, 4-d]pyrimidine derivatives. Tetrahedron 2012, 68, 9658–9664. [Google Scholar] [CrossRef]
- Ismail, N.S.M.; Ali, E.M.H.; Ibrahim, D.A.; Serya, R.A.T.; El Ella, D.A.A. Pyrazolo[3, 4-d]pyrimidine based scaffold derivatives targeting kinases as anticancer agents. Future J. Pharm. Sci. 2016, 2, 20–30. [Google Scholar] [CrossRef]
- Peter Bonn, P.; Mikael Brink, D.; Fagerhag, J.; Jurva, U.; Robb, G.R.; Schnecke, V.; Henriksson, A.S.; Waring, M.J.; Westerlund, C. The discovery of a novel series of glucokinase activators based on a pyrazolopyrimidine scaffold. Bioorg. Med. Chem. Lett. 2012, 22, 7302–7305. [Google Scholar] [CrossRef] [PubMed]
- Bakavoli, M.; Bagherzadeh, G.; Vaseghifar, M.; Shiri, A.; Mehdi Pordel, M.; Mashreghi, M.; Pordeli, P.; Araghi, M. Molecular iodine promoted synthesis of new pyrazolo[3,4-d]pyrimidine derivatives as potential antibacterial agents. Eur. J. Med. Chem. 2010, 45, 647–650. [Google Scholar] [CrossRef] [PubMed]
- Holla, B.S.; Mahalinga, M.; Karthikeyan, M.S.; Akberali, P.M.; Shetty, N.S. Synthesis of some novel pyrazole[3,4-d]pyrimidine derivatives as potential antimicrobial agents. Bioorg. Med. Chem. 2006, 14, 2040–2047. [Google Scholar] [CrossRef] [PubMed]
- Shamroukh, A.H.; Rashad, A.E.; Sayed, H.H. Synthesis of Some Pyrazolo[3,4-d]pyrimidine Derivatives for Biological Evaluation. Phosphorous Sulfur Silicon Relat. Elem. 2005, 180, 2347–2360. [Google Scholar] [CrossRef]
- Rigueiroa, F.; Teixeiraa, S.; Salaheldinb, A.M.; Oliveira-Camposb, A.M.F.; Rodriguesb, L.M.; Peixotoc, F.; Oliveira, M.M. Evaluation of antioxidant properties of some pyrazolo[3,4-d]pyrimidines derivatives and their effects on mitochondria bioenergetics. Biochim. Biophys. Acta 2008, 1777, S10.17. [Google Scholar] [CrossRef]
- Kumar, A.; Ahmad, I.; Chhikara, B.S.; Tiwari, R.; Mandal, D.; Parang, K. Synthesis of 3-phenylpyrazolopyrimidine-1,2,3-triazole conjugates and evaluation of their Src kinase inhibitory and anticancer activities. Bioorg. Med. Chem. Lett. 2011, 21, 1342–1346. [Google Scholar] [CrossRef] [PubMed]
- Ghorab, M.M.; Ragab, F.A.; Alqasoumi, S.I.; Alafeefy, A.M.; Aboulmag, S.A. Synthesis of some new pyrazolo[3,4-d]pyrimidine derivatives of expected anticancer and radioprotective activity. Eur. J. Med. Chem. 2010, 45, 171–178. [Google Scholar] [CrossRef] [PubMed]
- Schenone, S.; Bruno, O.; Bondavalli, F.; Ranise, A.; Mosti, L.; Giulia Menozzi, G.; Paola Fossa, P.; Donnini, S.; Santoro, A.; Ziche, M.; et al. Antiproliferative activity of new 1-aryl-4-amino-1H-pyrazolo[3,4-d]pyrimidine derivatives toward the human epidermoid carcinoma A431 cell line. Eur. J. Med. Chem. 2004, 39, 939–946. [Google Scholar] [CrossRef] [PubMed]
- EI-Bendary, E.R.; Badria, F.A. Synthesis, DNA-binding, and Antiviral Activity of Certain Pyrazolo[3,4-d]pyrimidine Derivatives. Arch. Pharm. 2000, 333, 99–103. [Google Scholar] [CrossRef]
- Liu, H.; Wang, H.Q.; Liu, Z.J. Synthesis and herbicidal activity of novel pyrazolo[3,4-d]pyrimidin-4-one derivatives containing aryloxyphenoxypropionate moieties. Bioorg. Med. Chem. Lett. 2007, 17, 2203–2209. [Google Scholar] [CrossRef] [PubMed]
- Chern, J.H.; Shia, K.S.; Hsu, T.A.; Tai, C.L.; Lee, C.C.; Lee, Y.C.; Chang, C.S.; Tseng, S.N.; Shih, S.R. Design, synthesis, and structure–activity relationships of pyrazolo [3,4-d] pyrimidines: A novel class of potent enterovirus inhibitors. Bioorg. Med. Chem. Lett. 2004, 14, 2519–2525. [Google Scholar] [CrossRef] [PubMed]
- Rashad, A.E.; Hegab, M.I.; Abdel-Megeid, R.E.; Fathalla, N.; Abdel-Megeid, F.M.E. Synthesis and anti-HSV-1 evaluation of some pyrazoles and fused pyrazolopyrimidines. Eur. J. Med. Chem. 2009, 44, 3285–3291. [Google Scholar] [CrossRef] [PubMed]
- Rahmoui, A.; Souiei, S.; Belkacem, M.A.; Romdhane, A.; Bouajila, J.; Jannet, H.B. Synthesis and biological evaluation of novel pyrazolopyrimidines derivatives as anticancer and anti-5-lipoxygenase agents. Bioorg. Chem. 2016, 66, 160–168. [Google Scholar] [CrossRef] [PubMed]
- El-Kalyoubi, S.; Agili, F. A Novel Synthesis of Fused Uracils: Indenopyrimidopyridazines, Pyrimidopyridazines, and Pyrazolopyrimidines for Antimicrobial and Antitumor Evalution. Molecules 2016, 21, 1714. [Google Scholar] [CrossRef] [PubMed]
- Quintela, J.M.; Peinador, C.; Gonzalez, L.; Devesa, I.; Ferrandiz, M.L.; Alcaraz, M.J.; Riguera, R. 6-Dimethylamino 1H-pyrazolo[3,4-d]pyrimidine derivatives as new inhibitors of inflammatory mediators in intact cell. Bioorg. Med. Chem. 2003, 11, 863–868. [Google Scholar] [CrossRef]
- Abdelazeem, A.H.; Abdelatef, S.A.; El-Saadi, M.T.; Omar, H.A.; Khan, S.I.; McCurdy, C.R.; El-Moghazy, S.M. Novel pyrazolopyrimidine derivatives targeting COXs and iNOS enzymes; design, synthesis and biological evaluation as potential anti-inflammatory agents. Eur. J. Pharm. Sci. 2014, 62, 197–221. [Google Scholar] [CrossRef] [PubMed]
- Avasthi, K.; Farooq, S.M.; Raghunandan, R.; Ma, P.R. Design and synthesis of pyrazolo[3,4-d]pyrimidine core based dissymmetrical ‘Leonard linker’ compounds: 1H-NMR and crystallographic evidence for folded conformation due to arene interaction. J. Mol. Struct. 2006, 785, 106–113. [Google Scholar] [CrossRef]
- Venkatesan, G.; Paira, P.; Cheong, L.C.; Vamsikrishna, K.; Federico, S.; Klotz, K.N.; Spalluto, G.; Pastorin, G. Discovery of simplified N2-substituted pyrazolo[3,4-d]pyrimidine derivatives as novel adenosine receptor antagonists: Efficient synthetic approaches, biological evaluations and molecular docking studies. Bioorg. Med. Chem. 2014, 22, 1751–1765. [Google Scholar] [CrossRef] [PubMed]
- Molina, P.; Arques, A.; Vinader, M.V. Tandem aza-wittig reaction/electrocyclic ring-closure a facile entry to the synthesis of fused pyrimidines: Preparation of pyrazolo[3,4-d] and 1,2,3-triazolo[4,5-d]pyrimidine derivatives. Tetrahedron Lett. 1987, 28, 4451–4454. [Google Scholar] [CrossRef]
- Sureja, D.K.; Dholakia, S.P.; Vadalia, K.R. Synthesis of some novel pyrazolo[3,4-d] pyrimidin-4(5H)-one derivatives as potential antimicrobial agents. J. Pharm. Bioallied. Sci. 2016, 8, 321–326. [Google Scholar] [CrossRef] [PubMed]
- Slavish, P.J.; Price, J.E.; Hanumesh, P.; Webb, T.R. Efficient synthesis of pyrazolopyrimidine libraries. J. Comb. Chem. 2010, 12, 807–809. [Google Scholar] [CrossRef] [PubMed]
- Gilbert, A.M.; Caltabiano, S.; Koehn, F.E.; Chen, Z.J.; Francisco, G.D.; Ellingboe, J.W.; Kharode, Y.; Mangine, A.; Francis, R.; TrailSmith, M.; et al. Pyrazolopyrimidine-2,4-dione sulfonamides: Novel and selective calcitonin inducers. J. Med. Chem. 2002, 45, 2342–2345. [Google Scholar] [CrossRef] [PubMed]
- Wen, K.S.; Lin, H.Y.; Huang, Y.Y.; Kaneko, K.; Takayama, H.; Kimura, M.; Juang, S.H.; Wong, F.F. Chemoselective synthesis, antiproliferative activities, and SAR study of 1H-pyrazol-5-yl-N,N-dimethyl-formamidines and pyrazolyl-2-azadienes. Med. Chem. Res. 2012, 21, 3920–3928. [Google Scholar] [CrossRef]
- Cheng, K.M.; Huang, Y.Y.; Huang, J.J.; Kaneko, K.; Kimura, M.; Takayama, H.; Juang, S.H.; Wong, F.F. Synthesis and antiproliferative evaluation of N,N-disubstituted-N′-[1-aryl-1H-pyrazol-5-yl]-methnimidamides. Bioorg. Med. Chem. Lett. 2010, 20, 6781–6784. [Google Scholar] [CrossRef] [PubMed]
- Srivastava, V.; Negi, A.S.; Kumar, J.K.; Gupta, M.M. A simple, convenient and chemoselective formylation of sterols by Vilsmeier reagent. Steroids 2006, 71, 632–638. [Google Scholar] [CrossRef] [PubMed]
- El-Shishtawy, R.M.; Almeida, P. A new Vilsmeier-type reaction for one-pot synthesis of pH sensitive fluorescent cyanine dyes. Tetrahedron 2006, 62, 7793–7798. [Google Scholar] [CrossRef]
- Pan, W.; Dong, D.; Wang, K.; Zhang, J.; Wu, R.; Xiang, D.; Liu, Q. Efficient One-Pot Synthesis of Highly Substituted Pyridin-2(1H)-ones via the Vilsmeier−Haack Reaction of 1-Acetyl,1-Carbamoyl Cyclopropanes. Org. Lett. 2007, 9, 2421. [Google Scholar] [CrossRef] [PubMed]
- Huang, Y.Y.; Kaneko, K.; Takayama, H.; Kimura, M.; Wong, F.F. New investigation of Vilsmeier-type reaction using pyrazolones with various amides. Tetrahedron Letts. 2011, 52, 3786–3792. [Google Scholar] [CrossRef]
- Wong, F.F.; Huang, Y.Y. Novel Vilsmeier-type methylenation for synthesis of dipyrazolylmethane derivatives using formamide or N-methylformamide. Tetrahedron 2011, 67, 3863–3867. [Google Scholar] [CrossRef]
- Simay, A.; Takacs, K.; Horvath, K.; Dvortsak, P. Vilsmeier-Haack reaction of 5-amino- and 5-acylaminopyrazoles. Acta Chim. Acad. Sci. Hung. 1980, 105, 127–139. [Google Scholar]
- Jachak, M.N.; Avhale, A.B.; Medhane, V.J.; Toche, R.B. A convenient route for the synthesis of pyrazolo[3,4-d]pyrimidine, pyrazolo[3,4-b][1,6]naphthyridine and pyrazolo[3,4-b]quinoline derivatives. J. Heterocycl. Chem. 2006, 43, 1169–1175. [Google Scholar] [CrossRef]
Sample Availability: Samples of the compounds are available from the authors. |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).