Abstract
An efficient and convenient method to synthesize highly functionalized 3,7′-bisindole derivatives has been developed via a Michael addition and cyclic condensation reaction of heterocyclic ketene aminals (HKAs) with 2-(1H-indol-3-yl)cyclohexa-2,5-diene-1,4-dione derivatives in ethanol-based solvents at room temperature. This strategy provides an efficient, environmentally friendly approach for easy access to various novel 3,7′-bisindole derivatives in moderate to good yields.
1. Introduction
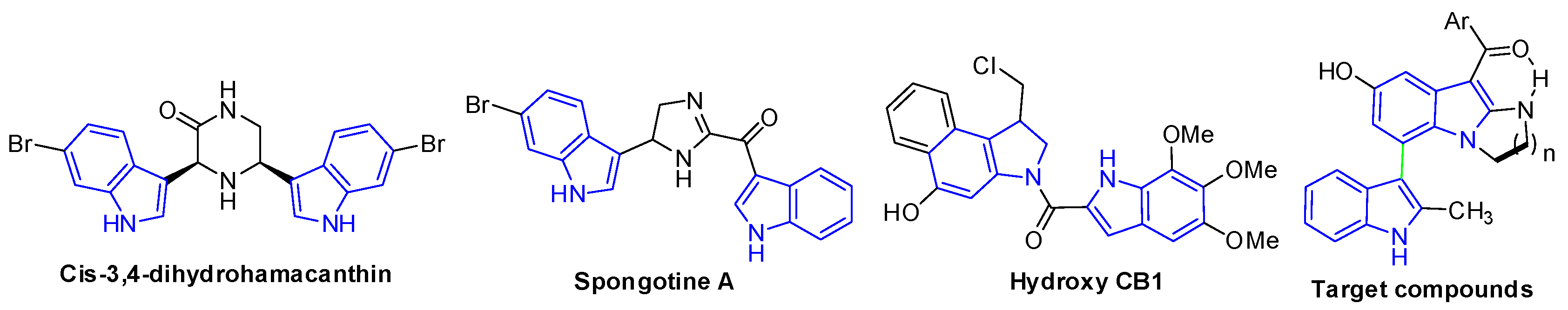
Bisindole-containing systems are prevalent molecular architectures that are widely found in natural products [1,2,3,4]. Furthermore, bisindole derivatives are especially important [5,6,7,8] due to their potent biological activities, including methicillin-resistant S. aureus (MRSA) pyruvate kinase inhibitors (cis-3,4-dihydrohamacanthin and spongotine A, Figure 1) [9,10,11], antitumor agents (Hydroxy CB1, Figure 1) [12,13,14], antihistamines and antimicrobials [15], anti-inflammatories [16], antibacterials and so on [17,18]. Because of their unique biological activities, more and more synthetic strategies to generate bisindole skeletons have been developed.
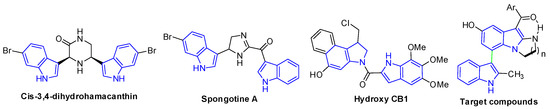
Figure 1.
Biologically active bisindole derivatives.
Generally, Lewis acids as well as Brønsted acids are employed as catalysts to form bisindole derivatives starting from indoles reacted with carbonyl compounds and their synthetic equivalents [19,20,21,22,23,24,25,26,27,28,29]. However, the synthetic pathways of highly functionalized bisindole derivatives usually suffer from common limitations, including harsh reaction conditions, multistep reactions, use of toxic solvents, and costly catalysts or enzymes [30]. Consequently, the development of more straightforward, eco-friendly and efficient strategies is highly desirable for the synthesis of bisindoles.
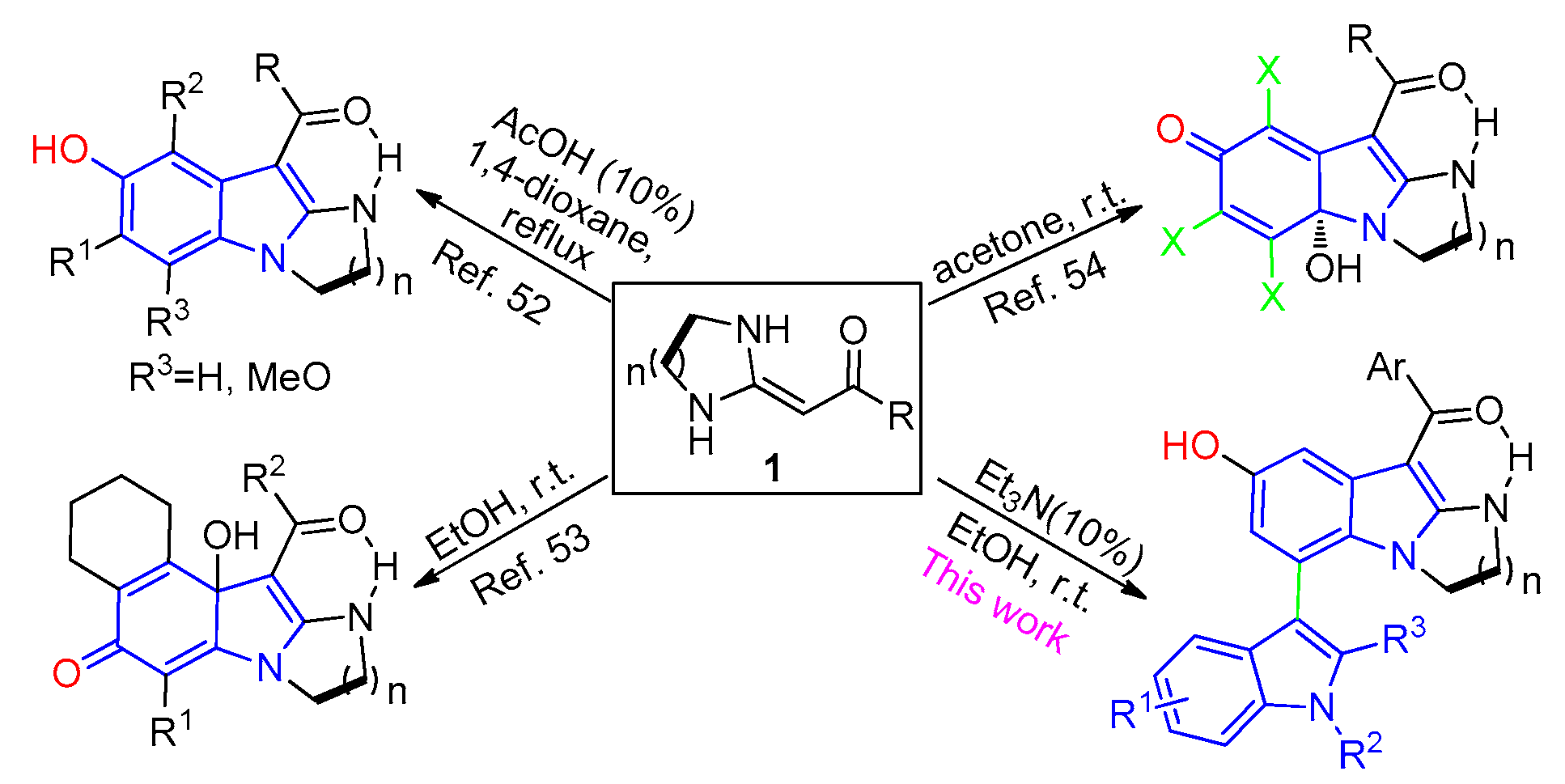
Heterocyclic ketene aminals (HKAs) are versatile building blocks used to construct a variety of fused heterocyclic compounds [31,32,33], such as quinolones [34,35], pyridines [36,37,38,39,40,41,42], pyrroles [43,44,45,46,47], spirooxindoles [48,49], etc. In recent years, we have developed some protocols to synthesize different substituted indole derivatives based on HKA building blocks [50,51,52] (Figure 2). Herein, we report an efficient and concise process to construct highly functionalized 3,7′-bisindole derivatives via an environmentally friendly and highly selective one-pot protocol.
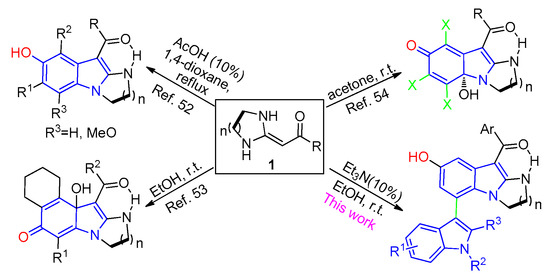
Figure 2.
Synthesis of 3,7′-bisindole derivatives based on the HKAs.
2. Results and Discussion
Initially, the model reaction of 1a and 2a with different catalysts, solvents and temperatures was studied, and the results are summarized in Table 1. Results showed that alkaline catalysts are better than acid catalysts. Furthermore, different bases have remarkable effects on the reactions. The use of triethylamine as the catalyst in ethanol at room temperature made the reaction proceed smoothly and afforded the target product 3a in good yield (Table 1, entry 3). Carbonate catalysts, such as Na2CO3 and K2CO3, also gave product 3a with moderate yield (Table 1, entries 4–5). However, NaOEt provided product 3a with poor yield, which may be due to its strong basicity (Table 1, entry 6). Notably, trace product was detected when 1,8-diazabicyclo[5,4,0]undec-7ene (DBU) was employed as the catalyst (Table 1, entry 7). Next, solvent effects were examined. Most solvents had little influence and could facilitate good yield of the products, except H2O (Table 1, entries 8–13). Ultimately, EtOH was proved to be the best solvent (Table 1, entry 3). To gain further insight into the effects of reaction temperature, we examined 40 °C and reflux temperature. The results revealed that high temperature was adverse to the reaction (Table 1, entries 14–15). Therefore, it could be concluded that the optimum conditions for the synthesis of 3a were EtOH as the solvent and triethylamine as the catalyst at room temperature for 12 h (Table 1, entry 3).

Table 1.
Optimization of reaction conditions a. 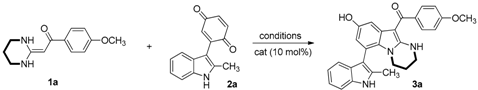

With the optimized conditions in hand, the substrate scope was investigated (Table 2). The results showed that the reaction was tolerant to a variety of HKAs bearing an electron-donating or an electron-withdrawing group. Furthermore, the ring size of HKA 1 has a slight effect on the reaction yield. Six- and seven-membered HKAs as substrates usually afforded superior yields to that of the five-membered HKAs. Additionally, aryl-substituted substrate 2 (R4 = Ph) was also tolerant to the reaction. Notably, substrate 2, with or without a substituent group at N1 (R3 = Me, H), reacted smoothly with HKA 1 to provide the corresponding products 3 in moderate to good yields. Substrate 2 with a methoxy at C5 and no substituent at C2 reacted cleanly with HKA 1 to provide the corresponding product 3 in good yield. All new compounds were fully characterized using IR, HR-MS, 1H-NMR, 13C-NMR (Please see the Supplementary Materials).

Table 2.
Preparation of the of 3,7′-bisindole derivatives a. 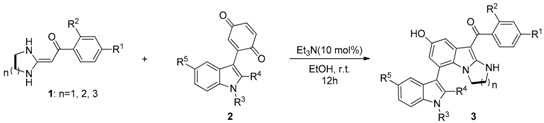

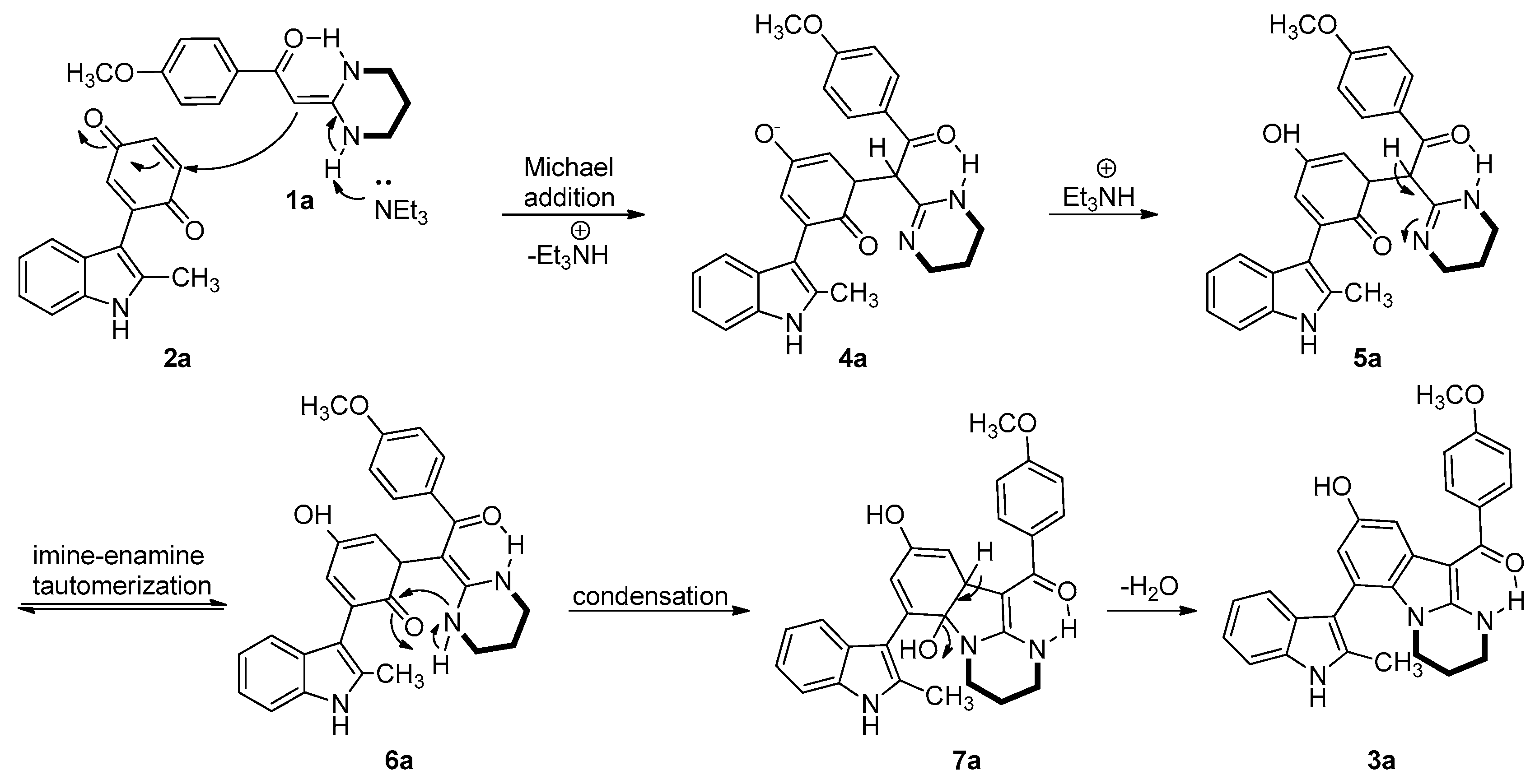
A proposed mechanism of the base-catalyzed cyclocondensation of 1a with 2a is depicted in Scheme 1. Initially, HKA 1a reacted with 2a in the presence of Et3N to form intermediate 4a by a Michael addition reaction. Intermediate 4a was subsequently protonated to form compound 5a. Imine-enamine tautomerization of compound 5a then generates 6a, which cyclizes to 7a by intramolecular attack of the NH on the 2,4-cyclohexadienone. Loss of H2O from intermediate 7a then provides the final product 3a.
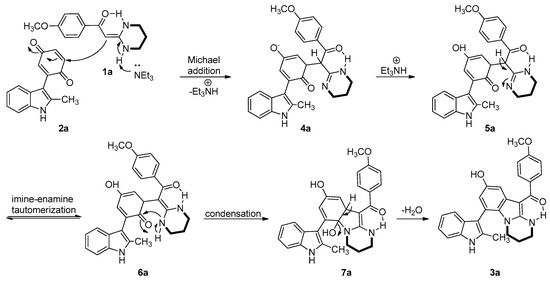
Scheme 1.
A plausible mechanism for the synthesis of 3a.
3. Experimental Section
3.1. General Information and Materials
All compounds were fully characterized by spectroscopic data. The NMR spectra were recorded on a Bruker DRX400 and DRX500 (Fällanden, Zürich, Switzerland). Chemical shifts (δ) are expressed in ppm, J values are given in Hz, and deuterated DMSO-d6 and CDCl3 were used as solvent. IR spectra were recorded on a FT-IR Thermo Nicolet Avatar 360 (Boston, MA, USA) using a KBr pellet. The reactions were monitored by thin layer chromatography (TLC) using silica gel GF254. The melting points were determined on a XT-4A melting point apparatus and are uncorrected. HRMs were performed on an Agilent LC/Msd TOF instrument (Palo Alto, CA, USA). All chemicals and solvents were used as received without further purification unless otherwise stated. Raw materials 1 and 2 were prepared according to the literature [53,54,55,56].
3.2. General Procedure for for the Preparation of the 3,7′-Bisindole Derivatives 3a–3w
Et3N (0.1 equiv) was added to a mixture of HKAs 1 (0.1 mmol) and compound 2 (0.11 mmol) in ethanol, and the mixture was stirred at room temperature until the HKAs 1 were completely consumed. Then, the solution was concentrated under reduced pressure and purified by flash column chromatography (petroleum ether/EtOAc = 6/1) to afford the corresponding products 3a–3w with 65%–91% yield. The products were further identified by FT-IR, NMR, and HRMS, and were in good agreement with the assigned structures.
(8-Hydroxy-6-(2-methyl-1H-indol-3-yl)-1,2,3,4-tetrahydro-pyrimido[1,2-a]indol-10-yl)(4-methoxyphenyl)methanone (3a). Yellow solid, yield 91%; Mp 221.5–222.5 °C; 1H-NMR (DMSO-d6, 500 MHz) δ 10.92 (br, 1H, NH), 8.51 (br, 1H, NH), 8.23 (s, 1H, ArH), 7.58 (d, J = 8.0 Hz, 2H, ArH), 7.28–7.30 (m, 2H, ArH), 7.06 (d, J = 8.0 Hz, 2H, ArH), 6.97–7.03 (m, 1H, ArH), 6.91–6.94 (m, 2H, ArH), 6.50 (br, 1H, OH), 3.91–3.95 (m, 2H, NCH2), 3.85 (s, 3H, OCH3), 3.46–3.50 (m, 2H, CH2N), 2.30 (s, 3H, CH3), 2.07–2.11 (m, 2H, CH2); 13C-NMR (DMSO-d6, 125 MHz) δ 186.8, 160.7, 153.1, 150.3, 135.6, 135.5, 133.1, 129.4, 129.4, 128.9, 128.8, 125.3, 120.2, 118.9, 118.7, 114.7, 113.9, 113.9, 113.9, 110.6, 110.5, 105.4, 94.9, 55.6, 39.3, 38.1, 20.6, 13.0; IR (KBr) 3439, 3230, 2904, 2586, 1722, 1599, 1514, 1333, 1223, 752 cm−1; HRMS (ESI-TOF): m/z calcd for C28H26N3O3 [M + H]+, 452.1969; found, 452.1947.
(8-Hydroxy-6-(2-methyl-1H-indol-3-yl)-1,2,3,4-tetrahydro-pyrimido[1,2-a]indol-10-yl)(p-tolyl)methanone (3b). Yellow solid, yield 88%; Mp 228–230 °C; 1H-NMR (DMSO-d6, 500 MHz) δ 10.82 (br, 1H, NH), 8.44 (br, 1H, NH), 8.19 (s, 1H, ArH), 7.41 (d, J = 7.5 Hz, 2H, ArH), 7.23–7.32 (m, 3H, ArH), 7.21 (d, J = 7.5 Hz, 1H, ArH), 6.98 (t, J = 7.5 Hz, 1H, ArH), 6.85–6.92 (m, 2H, ArH), 6.32 (br, 1H, OH), 3.82–3.89 (m, 2H, NCH2), 3.42–3.46 (m, 2H, CH2N), 2.37 (s, 3H, CH3), 2.24 (s, 3H, ArCH3), 2.04–2.08 (m, 2H, CH2); 13C-NMR (DMSO-d6, 125 MHz) δ 187.6, 153.3, 150.1, 139.8, 139.7, 135.5, 133.3, 129.3, 129.3, 129.0, 128.7, 127.3, 127.3, 125.1, 120.4, 118.9, 118.7, 115.1, 110.8, 110.5, 110.2, 105.4, 95.2, 39.5, 38.0, 21.4, 20.3, 12.7; IR (KBr) 3394, 3053, 2928, 2316, 1728, 1591, 1443, 1335, 1171, 750 cm−1; HRMS (ESI-TOF): m/z calcd for C28H26N3O2 [M + H]+, 436.2020; found, 436.2005.
(8-Hydroxy-6-(2-methyl-1H-indol-3-yl)-1,2,3,4-tetrahydro-pyrimido[1,2-a]indol-10-yl)(phenyl)methanone (3c). Yellow solid, yield 82%; Mp 317–318.5 °C; 1H-NMR (DMSO-d6, 500 MHz) δ 10.95 (br, 1H, NH), 8.60 (br, 1H, NH), 8.19 (s, 1H, ArH), 7.54–7.58 (m, 5H, ArH), 7.30 (t, J = 7.5 Hz, 2H, ArH), 6.98–7.04 (m, 1H, ArH), 6.90–6.98 (m, 2H, ArH), 6.34 (br, 1H, OH), 3.92–3.96 (m, 2H, NCH2), 3.47–3.51 (m, 2H, CH2N), 2.30 (s, 3H, CH3), 2.08–2.12 (m, 2H, CH2); 13C-NMR (DMSO-d6, 125 MHz) δ 187.2, 153.2, 150.3, 143.2, 135.7, 133.2, 129.8, 128.9, 128.7, 128.7, 128.7, 127.3, 127.3, 125.2, 120.2, 118.9, 118.7, 114.9, 110.7, 110.6, 110.5, 105.4, 95.0, 39.3, 38.1, 20.6, 13.0; IR (KBr) 3323, 3055, 2972, 2866, 2314, 1726, 1614, 1529, 1319, 1174, 746 cm−1; HRMS (ESI-TOF): m/z calcd for C27H24N3O2 [M + H]+, 422.1863; found, 422.1871.
(4-Chlorophenyl)(8-hydroxy-6-(2-methyl-1H-indol-3-yl)-1,2,3,4-tetrahydropyrimido[1,2-a]indol-10-yl)methanone (3d). Yellow solid, yield 89%; Mp 199.0–201.5 °C; 1H-NMR (DMSO-d6, 500 MHz) δ 10.91 (br, 1H, NH), 8.55 (br, 1H, NH), 8.27–8.32 (m, 1H, ArH), 7.55–7.59 (m, 4H, ArH), 7.23–7.31 (m, 2H, ArH), 6.96–7.02 (m, 1H, ArH), 6.87–6.96 (m, 2H, ArH), 6.32 (br, 1H, OH), 3.91–3.95 (m, 2H, NCH2), 3.42–3.46 (m, 2H, CH2N), 2.27 (s, 3H, CH3), 2.08–2.12 (m, 2H, CH2); 13C-NMR (DMSO-d6, 125 MHz) δ 185.6, 153.2, 150.5, 141.8, 135.6, 134.3, 133.1, 129.3, 129.3, 129.3, 128.9, 128.9, 128.9, 128.9, 124.9, 120.2, 118.9, 118.7, 115.0, 110.7, 110.5, 105.1, 95.0, 39.4, 38.1, 20.5, 13.0; IR (KBr) 3400, 3063, 2951, 2866, 2349, 1680, 1616, 1527, 1331, 750 cm−1; HRMS (ESI-TOF): m/z calcd for C27H23ClN3O2 [M + H]+, 456.1473; found, 456.1459.
(2-Chlorophenyl)(8-hydroxy-6-(2-methyl-1H-indol-3-yl)-1,2,3,4-tetrahydropyrimido[1,2-a]indol-10-yl)methanone (3e). Yellow solid, yield 86%; Mp 301–303 °C; 1H-NMR (DMSO-d6, 500 MHz) δ 10.94 (br, 1H, NH), 8.52 (br, 1H, NH), 8.07 (s, 1H, ArH), 7.61 (d, J = 1.0 Hz, 1H, ArH), 7.47–7.61 (m, 2H, ArH), 7.34–7.39 (m, 1H, ArH), 7.30 (d, J = 8.0 Hz, 1H, ArH), 7.26 (d, J = 7.5 Hz, 1H, ArH), 6.96–7.03 (m, 1H, ArH), 6.88–6.95 (m, 2H, ArH), 5.71 (br, 1H, OH), 3.90–3.94 (m, 2H, NCH2), 3.48–3.52 (m, 2H, CH2N), 2.26 (s, 3H, CH3), 2.08–2.12 (m, 2H, CH2); 13C-NMR (DMSO-d6, 125 MHz) δ 183.7, 152.8, 150.5, 142.3, 135.7, 133.2, 130.4, 130.1, 129.6, 129.0, 128.9, 128.2, 128.1, 125.0, 120.3, 118.8, 118.7, 115.1, 110.7, 110.7, 110.4, 104.7, 95.7, 39.3, 38.1, 20.4, 13.0; IR KBr) 3342, 3061, 2966, 2868, 1726, 1618, 1531, 1429, 1329, 1176, 748 cm−1; HRMS (ESI-TOF): m/z calcd for C27H23ClN3O2 [M + H]+, 456.1473; found, 456.1462.
(4-Fluorophenyl)(8-hydroxy-6-(2-methyl-1H-indol-3-yl)-1,2,3,4-tetrahydropyrimido[1,2-a]indol-10-yl)methanone (3f). Yellow solid, yield 89%; Mp 247–248.5 °C; 1H-NMR (DMSO-d6, 500 MHz) δ 10.93 (br, 1H, NH), 8.54 (br, 1H, NH), 8.27 (s, 1H, ArH), 7.58–7.65 (m, 2H, ArH), 7.34 (t, J = 9.0 Hz, 2H, ArH), 7.28 (t, J = 9.0 Hz, 2H, ArH), 7.00 (t, J = 7.5 Hz, 1H, ArH), 6.87–6.97 (m, 2H, ArH), 6.31 (br, 1H, OH), 3.90–3.97 (m, 2H, NCH2), 3.47–3.51 (m, 2H, CH2N), 2.28 (s, 3H, CH3), 2.08–2.12 (m, 2H, CH2); 13C-NMR (DMSO-d6, 125 MHz) δ 185.9, 164.0, 162.1, 153.1, 150.4, 139.6, 135.6, 133.1, 129.7, 129.7, 128.9, 125.1, 120.2, 118.9, 118.7, 115.7, 115.5, 114.9, 110.7, 110.7, 110.5, 105.1, 95.0, 39.4, 38.1, 20.5, 13.0; IR (KBr) 3394, 3053, 2928, 2860, 2316, 1720, 1591, 1441, 1335, 1170, 750 cm−1; HRMS (ESI-TOF): m/z calcd for C27H23FN3O2 [M + H]+, 440.1769; found, 440.1775.
(9-Hydroxy-7-(2-methyl-1H-indol-3-yl)-2,3,4,5-tetrahydro-1H-[1,3]diazepino[1,2-a]indol-11-yl)(4-methoxyphenyl)methan-one (3g). Yellow solid, yield 87%; Mp 179.5–182 °C; 1H-NMR (DMSO-d6, 500 MHz) δ 10.93 (br, 1H, NH), 8.96 (br, 1H, NH), 8.32 (s, 1H, ArH), 7.59 (d, J = 7.6 Hz, 2H, ArH), 7.29 (d, J = 7.5 Hz, 1H, ArH), 7.26 (d, J = 7.4 Hz, 1H, ArH), 7.12 (s, 1H, ArH), 7.07 (d, J = 7.7 Hz, 2H, ArH), 7.00 (m, 1H, ArH), 6.91 (m, 1H, ArH), 6.45 (br, 1H, OH), 4.03–4.07 (m, 2H, NCH2), 3.86 (s, 3H, OCH3), 3.41–3.45 (m, 2H, CH2N), 2.29 (s, 3H, CH3), 1.87–1.97 (m, 4H, CH2CH2); 13C-NMR (DMSO-d6, 125 MHz) δ 188.3, 161.0, 160.0, 150.5, 135.6, 135.0, 133.2, 129.7, 129.7, 129.5, 128.9, 125.7, 120.2, 118.9, 118.7, 115.7, 113.9, 113.9, 112.2, 110.6, 105.3, 97.7, 55.6, 45.5, 45.1, 29.3, 27.0, 13.0; IR (KBr) 3396, 3063, 2928, 2850, 2351, 1726, 1593, 1444, 1313, 1250, 1167, 1022, 744 cm−1; HRMS (ESI-TOF): m/z calcd for C29H28N3O3 [M + H]+, 466.2125; found, 466.2145.
(9-Hydroxy-7-(2-methyl-1H-indol-3-yl)-2,3,4,5-tetrahydro-1H-[1,3]diazepino[1,2-a]indol-11-yl)(p-tolyl)methanone (3h). Yellow solid, yield 84%; Mp 242–244 °C; 1H-NMR (DMSO-d6, 500 MHz) δ 10.92 (br, 1H, NH), 9.05 (br, 1H, NH), 8.23 (s, 1H, ArH), 7.49 (d, J = 7.7 Hz, 2H, ArH), 7.33 (d, J = 7.6 Hz, 2H, ArH), 7.29 (d, J = 7.9 Hz, 1H, ArH), 7.25 (d, J = 7.8 Hz, 1H, ArH), 7.11 (s, 1H, ArH), 6.96–7.03 (m, 1H, ArH), 6.91 (t, J = 7.3 Hz, 1H, ArH), 6.34 (br, 1H, OH), 4.04–4.11 (m, 2H, NCH2), 3.39–3.43 (m, 2H, CH2N), 2.43 (s, 3H, ArCH3), 2.28 (s, 3H, CH3), 1.88–1.97 (m, 4H, CH2CH2); 13C-NMR (DMSO-d6, 125 MHz) δ 188.8, 160.0, 150.5, 139.9, 139.8, 135.6, 133.2, 129.7, 129.2, 129.2, 128.9, 127.7, 127.7, 125.6, 120.2, 118.9, 118.7, 115.8, 112.2, 110.6, 110.5, 105.4, 97.6, 45.5, 45.0, 29.2, 26.9, 21.5, 13.0; IR (KBr) 3394, 3053, 2926, 2858, 2314, 1726, 1593, 1446, 1335, 1169, 748 cm−1; HRMS (ESI-TOF): m/z calcd for C29H28N3O2 [M + H]+, 450.2176; found, 450.2184.
(9-Hydroxy-7-(2-methyl-1H-indol-3-yl)-2,3,4,5-tetrahydro-1H-[1,3]diazepino[1,2-a]indol-11-yl)(phenyl)methanone (3i). Yellow solid, yield 83%; Mp 289–290 °C; 1H-NMR (DMSO-d6, 500 MHz) δ 10.92 (br, 1H, NH), 9.10 (br, 1H, NH), 8.21 (s, 1H, ArH), 7.53–7.57 (m, 5H, ArH), 7.29 (d, J = 8.0 Hz, 1H, ArH), 7.24 (d, J = 7.5 Hz, 1H, ArH), 7.11 (s, 1H, ArH), 6.96–7.03 (m, 1H, ArH), 6.87–6.94 (m, 1H, ArH), 6.23 (br, 1H, OH), 4.04–4.08 (m, 2H, NCH2), 3.45–3.49 (m, 2H, CH2N), 2.27 (s, 3H, CH3), 1.95–1.99 (m, 2H, CH2), 1.89–1.93 (m, 2H, CH2); 13C-NMR (DMSO-d6, 125 MHz) δ 188.8, 160.1, 150.5, 142.8, 135.6, 133.2, 130.1, 129.7, 128.9, 128.8, 128.8, 127.4, 127.4, 125.6, 120.2, 118.9, 118.7, 115.9, 112.2, 110.7, 110.4, 105.4, 97.5, 45.5, 44.9, 29.1, 26.9, 12.9; IR (KBr) 3356, 3057, 2941, 2858, 2351, 1714, 1593, 1539, 1419, 1323, 1171, 748 cm−1; HRMS (ESI-TOF): m/z calcd for C28H26N3O2 [M + H]+, 436.2020; found, 436.2034.
(4-Chlorophenyl)(9-hydroxy-7-(2-methyl-1H-indol-3-yl)-2,3,4,5-tetrahydro-1H-[1,3]diazepino[1,2-a]indol-11-yl)methan-one (3j). Yellow solid, yield 87%; Mp 191–192.5 °C; 1H-NMR (DMSO-d6, 500 MHz) δ 10.92 (br, 1H, NH), 9.09 (br, 1H, NH), 8.37 (s, 1H, ArH), 7.56–7.60 (m, 4H, ArH), 7.28 (d, J = 8.0 Hz, 1H, ArH), 7.24 (d, J = 7.5 Hz, 1H, ArH), 7.11 (s, 1H, ArH), 6.99 (t, J = 7.5 Hz, 1H, ArH), 6.87–6.94 (m, 1H, ArH), 6.25 (br, 1H, OH), 4.05–4.09 (m, 2H, NCH2), 3.44–3.48 (m, 2H, CH2N), 2.27 (s, 3H, CH3), 1.95–1.99 (m, 2H, CH2), 1.88–1.92 (m, 2H, CH2); 13C-NMR (DMSO-d6, 125 MHz) δ 187.1, 160.1, 150.7, 141.4, 135.6, 134.7, 133.2, 129.7, 129.4, 129.4, 128.9, 128.9, 128.9, 125.3, 120.2, 118.9, 118.7, 115.9, 112.3, 110.6, 110.5, 105.1, 97.4, 45.5, 44.9, 29.0, 26.8, 13.0; IR (KBr) 3394, 3057, 2926, 2854, 2353, 1687, 1599, 1539, 1417, 1169, 1092, 746 cm−1; HRMS (ESI-TOF): m/z calcd for C28H25ClN3O2 [M + H]+, 470.1630; found, 470.1637.
(2-Chlorophenyl)(9-hydroxy-7-(2-methyl-1H-indol-3-yl)-2,3,4,5-tetrahydro-1H-[1,3]diazepino[1,2-a]indol-11-yl)methan-one (3k). Yellow solid, yield 81%; Mp 240.5–241.5 °C; 1H-NMR (DMSO-d6, 500 MHz) δ 10.94 (br, 1H, NH), 9.21 (br, 1H, NH), 8.15 (s, 1H, ArH), 7.61 (d, J = 7.5 Hz, 1H, ArH), 7.46–7.56 (m, 2H, ArH), 7.35–7.41 (m, 1H, ArH), 7.30 (d, J = 8.0 Hz, 1H, ArH), 7.24 (d, J = 7.5 Hz, 1H, ArH), 7.09 (s, 1H, ArH), 7.00 (t, J = 7.5, 1H, ArH), 6.91 (t, J = 7.5 Hz, 1H, ArH), 5.69 (br, 1H, OH), 4.01–4.08 (m, 2H, NCH2), 3.51–3.55 (m, 2H, CH2N), 2.27 (s, 3H, CH3), 1.98–2.02 (m, 2H, CH2), 1.89–1.94 (m, 2H, CH2); 13C-NMR (DMSO-d6, 125 MHz) δ 185.0, 159.8, 150.8, 142.0, 135.6, 133.3, 130.6, 130.1, 130.0, 129.5, 128.9, 128.1, 128.1, 125.4, 120.3, 118.9, 118.8, 116.0, 112.3, 110.7, 110.3, 104.6, 97.8, 45.5, 44.7, 28.8, 26.8, 13.0; IR (KBr) 3398, 3063, 2937, 2347, 1726, 1597, 1439, 1336, 1176, 750 cm−1; HRMS (ESI-TOF): m/z calcd for C28H25ClN3O2 [M + H]+, 470.1630; found, 470.1621.
(4-Fluorophenyl)(9-hydroxy-7-(2-methyl-1H-indol-3-yl)-2,3,4,5-tetrahydro-1H-[1,3]diazepino[1,2-a]indol-11-yl)methan-one (3l). Yellow solid, yield 83%; Mp 259–261 °C; 1H-NMR (DMSO-d6, 500 MHz) δ 10.91 (br, 1H, NH), 9.06 (br, 1H, NH), 8.32 (s, 1H, ArH), 7.59–7.65 (m, 2H, ArH), 7.35 (t, J = 8.5 Hz, 2H, ArH), 7.29 (d, J = 7.5 Hz, 1H, ArH), 7.24 (d, J = 7.5 Hz, 1H, ArH), 7.11 (s, 1H, ArH), 6.96–7.03 (m, 1H, ArH), 6.86–6.94 (m, 1H, ArH), 6.24 (br, 1H, OH), 4.04–4.08 (m, 2H, NCH2), 3.44–3.48 (m, 2H, CH2N), 2.28 (s, 3H, CH3), 1.95–1.99 (m, 2H, CH2), 1.89–1.93 (m, 2H, CH2); 13C-NMR (DMSO-d6, 125 MHz) δ 187.4, 164.2, 162.3, 160.0, 150.7, 139.2, 135.6, 133.2, 129.9, 129.7, 129.7, 128.9, 125.5, 120.2, 118.9, 118.7, 115.8, 115.6, 112.3, 110.6, 110.5, 105.1, 97.5, 45.5, 44.9, 29.1, 26.9, 13.0; IR (KBr) 3390, 3064, 2929, 2343, 1720,1595, 1535, 1428, 1222, 1167, 749 cm−1; HRMS (ESI-TOF): m/z calcd for C28H25FN3O2 [M + H]+, 454.1925; found, 454.1936.
(7-Hydroxy-5-(2-methyl-1H-indol-3-yl)-2,3-dihydro-1H-imidazo[1,2-a]indol-9-yl)(4-methoxyphenyl)methanone (3m). Yellow solid, yield 82%; Mp 269–271 °C; 1H-NMR (DMSO-d6, 500 MHz) δ 10.93 (br, 1H, NH), 8.40 (br, 1H, NH), 7.63–7.67 (m, 2H, ArH), 7.28–7.32 (m, 2H, ArH), 7.04–7.08 (m, 5H, ArH), 6.90–6.94 (m, 2H, ArH), 3.98–4.10 (m, 4H, CH2CH2), 3.85 (s, 3H, OCH3), 2.32 (s, 3H, CH3); 13C-NMR (DMSO-d6, 125 MHz) δ 186.5, 161.2, 159.5, 150.4, 135.6, 134.8, 133.1, 130.7, 129.7, 129.7, 128.9, 126.0, 120.2, 118.9, 118.7, 115.0, 114.0, 114.0, 111.1, 110.7, 106.8, 93.4, 55.6, 49.5, 42.5, 13.0; IR (KBr) 3390, 3059, 2966, 2843, 2353, 1726, 1597, 1473, 1325, 1248, 1163, 744 cm−1; HRMS (ESI-TOF): m/z calcd for C27H24N3O3 [M + H]+, 438.1812; found, 438.1786.
(7-Hydroxy-5-(2-methyl-1H-indol-3-yl)-2,3-dihydro-1H-imidazo[1,2-a]indol-9-yl)(p-tolyl)methanone (3n). Yellow solid, yield 75%; Mp 298.5–300 °C; 1H-NMR (DMSO-d6, 300 MHz) δ 10.91 (br, 1H, NH), 8.38 (br, 1H, NH), 7.53 (d, J = 6.5 Hz, 2H, ArH), 7.25–7.34 (m, 4H, ArH), 6.88–7.02 (m, 5H, ArH), 3.97–4.09 (m, 4H, CH2CH2), 2.40 (s, 3H, ArCH3), 2.29 (s, 3H, CH3); 13C-NMR (DMSO-d6, 125 MHz) δ 187.2, 159.7, 150.4, 140.1, 139.6, 135.6, 133.1, 130.5, 129.3, 129.3, 128.9, 127.6, 127.6, 126.0, 120.3, 118.9, 118.7, 115.2, 111.1, 110.7, 110.5, 106.9, 93.5, 49.5, 42.5, 21.5, 13.0; IR (KBr) 3408, 2899, 2584, 2345, 1726, 1597, 1475, 1327, 1167, 752 cm−1; HRMS (ESI-TOF): m/z calcd for C27H24N3O2 [M + H]+, 422.1863; found, 422.1837.
(7-Hydroxy-5-(2-methyl-1H-indol-3-yl)-2,3-dihydro-1H-imidazo[1,2-a]indol-9-yl)(phenyl)methanone (3o). Yellow solid, yield 73%; Mp 289.5–290.5 °C; 1H-NMR (DMSO-d6, 500 MHz) δ 10.92 (br, 1H, NH), 8.33 (br, 1H, NH), 7.61–7.65 (m, 2H, ArH), 7.52–7.56 (m, 3H, ArH), 7.28–7.32 (m, 2H, ArH), 6.91–7.02 (m, 5H, ArH), 3.99–4.11 (m, 4H, CH2CH2), 2.31 (s, 3H, CH3); 13C-NMR (DMSO-d6, 125 MHz) δ 187.1, 159.7, 150.4, 142.6, 135.6, 133.1, 130.8, 130.4, 130.3, 128.8, 128.8, 127.5, 127.5, 126.1, 120.2, 118.9, 118.7, 115.2, 111.1, 110.7, 110.5, 106.8, 93.5, 49.5, 42.5, 13.0; IR (KBr): 3419, 3059, 2970, 2316, 1730, 1603, 1510, 1335, 1227, 744 cm−1; HRMS (ESI-TOF): m/z calcd for C26H22N3O2 [M + H]+, 408.1707; found, 408.1713.
(4-Chlorophenyl)(7-hydroxy-5-(2-methyl-1H-indol-3-yl)-2,3-dihydro-1H-imidazo[1,2-a]indol-9-yl)methanone (3p). Yellow solid, yield 75%; Mp 204–206 °C; 1H-NMR (DMSO-d6, 500 MHz) δ 10.95 (br, 1H, NH), 8.48 (br, 1H, NH), 7.60–7.66 (m, 2H, ArH), 7.54–7.60 (m, 2H, ArH), 7.25–7.32 (m, 3H, ArH), 7.00 (t, J = 7.0 Hz, 1H, ArH), 6.89–6.95 (m, 3H, ArH), 4.05–4.11 (m, 2H, NCH2), 3.98–4.03 (m, 2H, CH2N), 2.30 (s, 3H, CH3); 13C-NMR (DMSO-d6, 125 MHz) δ 185.6, 159.7, 150.5, 141.2, 135.6, 134.9, 133.1, 130.3, 129.5, 129.5, 128.9, 128.9, 128.9, 126.0, 120.2, 118.9, 118.7, 115.3, 111.2, 110.7, 110.5, 106.7, 93.3, 49.5, 42.5, 13.0; IR (KBr) 3435, 3072, 2902, 2347, 1724, 1600, 1510, 1402, 1330, 1223, 752 cm−1; HRMS (ESI-TOF): m/z calcd for C26H21ClN3O2 [M + H]+, 442.1317; found, 442.1309.
(2-Chlorophenyl)(7-hydroxy-5-(2-methyl-1H-indol-3-yl)-2,3-dihydro-1H-imidazo[1,2-a]indol-9-yl)methanone (3q). Yellow solid, yield 72%; mp 339–341 °C; 1H-NMR (DMSO-d6, 500 MHz) δ 10.92 (br, 1H, NH), 7.87–8.07 (m, 1H, ArH), 7.10–7.60 (m, 8H, ArH), 6.87–7.02 (m, 3H, ArH), 4.08– 4.12(m, 4H, CH2CH2), 2.26 (s, 3H, CH3); 13C-NMR (DMSO-d6, 125 MHz) δ 184.0, 160.1, 150.1, 142.1, 135.6, 133.2, 130.6, 130.2, 129.5, 128.8, 128.1, 128.1, 128.1, 126.1, 120.3, 118.8, 118.8, 115.4, 111.2, 110.7, 110.3, 105.7, 94.1, 49.6, 42.4, 12.9; IR (KBr) 3429, 3346, 3059, 2918, 2580, 2318, 1728, 1520, 1464, 1327, 1225, 748 cm−1; HRMS (ESI-TOF): m/z calcd for C26H21ClN3O2 [M + H]+, 442.1317; found, 442.1302.
(4-Fluorophenyl)(7-hydroxy-5-(2-methyl-1H-indol-3-yl)-2,3-dihydro-1H-imidazo[1,2-a]indol-9-yl)methanone (3r). Yellow solid, yield 77%; Mp 268–270 °C; 1H-NMR (DMSO-d6, 500 MHz) δ 10.92 (br, 1H, NH), 8.39 (br, 1H, NH), 7.65–7.72 (m, 2H, ArH), 7.27–7.37 (m, 4H, ArH), 6.89–7.04 (m, 5H, ArH), 4.06–4.13 (m, 2H, NCH2), 3.98–4.04 (m, 2H, CH2N), 2.31 (s, 3H, CH3); 13C-NMR (DMSO-d6, 125 MHz) δ 185.8, 164.4, 162.4, 159.7, 150.5, 139.0, 135.6, 133.1, 130.4, 130.0, 130.0, 128.9, 126.0, 120.2, 118.9, 118.7, 115.8, 115.6, 115.2, 111.2, 110.7, 106.7, 93.3, 49.5, 42.5, 13.0; IR (KBr) 3429, 3072, 2902, 2582, 2347, 1724, 1601, 1510, 1402, 1331, 1223, 1157, 752 cm−1; HRMS (ESI-TOF): m/z calcd for C26H21FN3O2 [M + H]+, 426.1612; found, 426.1622.
(6-(1,2-Dimethyl-1H-indol-3-yl)-8-hydroxy-1,2,3,4-tetrahydropyrimido[1,2-a]indol-10-yl)(4-methoxyphenyl)methanone (3s). Yellow solid, yield 87%; Mp 261–263 °C; 1H-NMR (CDCl3, 400 MHz) δ 8.61 (br, 1H, NH), 7.69 (d, J = 8.8 Hz, 2H, ArH), 7.39 (d, J = 8.0 Hz, 1H, ArH), 7.34 (d, J = 8.0 Hz, 1H, ArH), 7.20–7.22 (m, 1H, ArH), 7.07–7.11 (m, 1H, ArH), 6.98 (d, J = 8.4 Hz, 2H, ArH), 6.86 (s, 1H, ArH), 6.71 (s, 1H, ArH), 4.93 (br, 1H, OH), 3.90–3.93 (m, 2H, CH2N), 3.86 (s, 3H, NCH3), 3.75 (s, 3H, OCH3), 3.53–3.57 (m, 2H, CH2), 2.34 (s, 3H, CH3), 2.21–2.24 (m, 2H, CH2N); 13C-NMR (CDCl3, 100 MHz) δ 188.8, 161.0, 153.5, 149.3, 137.0, 135.4, 134.9, 129.4, 129.4, 129.3, 127.5, 126.6, 121.5, 119.9, 118.9, 113.6, 113.6, 113.2, 109.5, 108.9, 107.7, 104.7, 95.8, 55.3, 39.3, 38.1, 29.9, 20.8, 11.1; IR (KBr) 3439, 2926, 2853, 2347, 1728, 1616, 1510, 1471, 1350, 1324, 1168, 740 cm−1; HRMS (ESI-TOF): m/z calcd for C29H28N3O3 [M + H]+, 466.2125; found, 466.2120.
(6-(1,2-Dimethyl-1H-indol-3-yl)-8-hydroxy-1,2,3,4-tetrahydropyrimido[1,2-a]indol-10-yl)(4-methoxyphenyl)methanone(6-(1,2-dimethyl-1H-indol-3-yl)-8-hydroxy-1,2,3,4-tetrahydropyrimido[1,2-a]indol-10-yl)(4-fluorophenyl)methanone (3t). Yellow solid, yield 70%; Mp 173–175 °C; 1H-NMR (CDCl3, 400 MHz) δ 8.62 (br, 1H, NH), 7.67–7.71 (m, 2H, ArH), 7.34–7.39 (m, 2H, ArH), 7.11–7.26 (m, 3H, ArH), 7.08–7.11 (m, 1H, ArH), 6.87 (s, 1H, ArH), 6.53 (s, 1H, ArH), 4.88 (br, 1H, OH), 3.94 (t, J = 6.0 Hz, 2H, CH2N), 3.77 (s, 3H, NCH3), 3.57–3.63 (m, 2H, CH2), 2.35 (s, 3H, CH3), 2.23–2.29 (m, 2H, CH2N); 13C-NMR (CDCl3, 100 MHz) δ 188.0, 162.5, 153.6, 149.4, 138.5, 137.0, 135.4, 129.6, 129.5, 129.4, 127.4, 126.4, 121.6, 120.0, 118.8, 115.5, 115.3, 113.4, 109.6, 108.9, 107.6, 104.5, 95.9, 39.3, 38.1, 29.9, 20.7, 11.1; IR (KBr) 3437, 2925, 2582, 1721, 1617, 1534, 1470, 1325, 1221, 1173, 775 cm−1; HRMS (ESI-TOF): m/z calcd for C28H25N3O2 [M + H]+, 454.1925; found, 454.1931.
(8-Hydroxy-6-(2-phenyl-1H-indol-3-yl)-1,2,3,4-tetrahydropyrimido[1,2-a]indol-10-yl)(4-methoxyphenyl)methanone (3u). Yellow solid, yield 85%; Mp 179–181 °C; 1H-NMR (CDCl3, 400 MHz) δ 8.92 (br, 1H, NH), 8.59 (br, 1H, NH), 7.69 (d, J = 8.8 Hz, 2H, ArH), 7.45–7.40 (m, 4H, ArH), 7.28–7.22 (m, 4H, ArH), 7.13–7.10 (m, 1H, ArH), 6.96 (d, J = 8.8 Hz, 2H, ArH), 6.85 (s, 1H, ArH), 6.72 (s, 1H, ArH), 4.94 (br, 1H, OH), 3.82–3.86 (m, 5H, CH2N, OCH3), 3.47–3.51 (m, 2H, NCH2), 2.19–2.16 (m, 2H, CH2); 13C-NMR (CDCl3, 100 MHz) δ 188.9, 161.1, 153.6, 149.3, 136.2, 135.2, 134.8, 131.9, 129.7, 129.5, 129.4, 129.4, 128.9, 128.9, 127.9, 127.0, 127.0, 123.1, 120.6, 119.8, 113.6, 113.6, 113.1, 113.0, 111.1, 109.5, 108.8, 105.2, 95.9, 55.3, 39.2, 38.0, 20.7; IR (KBr) 3438, 2925, 2854, 1728, 1616, 1577, 1532, 1445, 1326, 1253, 1168, 747 cm−1; HRMS (ESI-TOF): m/z calcd for C33H28N3O3 [M + H]+, 514.2125; found, 514.2121.
(4-Fluorophenyl)(8-hydroxy-6-(2-phenyl-1H-indol-3-yl)-1,2,3,4-tetrahydropyrimido[1,2-a]indol-10-yl)methanone (3v). Yellow solid; yield 75%; Mp 264–266 °C; 1H-NMR (CDCl3, 400 MHz): δ 8.75 (br, 1H, NH), 8.61 (br, 1H, NH), 7.71–7.67 (m, 2H, ArH), 7.44–7.40 (m, 4H, ArH), 7.31–7.26 (m, 4H, ArH), 7.16–7.11 (m, 3H, ArH), 6.85 (s, 1H, ArH), 6.54 (s, 1H, ArH), 4.92 (br, 1H, OH), 3.86–3.83 (m, 2H, CH2N), 3.52–3.56 (m, 2H, NCH2), 2.22–2.19 (m, 2H, CH2); 13C-NMR (CDCl3, 100 MHz): δ 187.9, 165.0, 162.5, 153.7, 149.4, 138.4, 138.4, 136.1, 135.2, 131.9, 129.7, 129.6, 129.5, 128.9, 128.0, 126.9, 126.7, 123.2, 120.7, 119.8, 115.5, 115.3, 113.2, 111.1, 109.7, 108.7, 105.0, 104.9, 95.9, 39.2, 38.0, 20.4; IR (KBr) 3426, 2924, 1721, 1617, 1535, 1478, 1325, 1221, 1176, 774 cm−1; HRMS (ESI-TOF): m/z calcd for C32H25FN3O2 [M + H]+, 502.1925; found, 502.1933.
(8-Hydroxy-6-(5-methoxy-1H-indol-3-yl)-1,2,3,4-tetrahydropyrimido[1,2-a]indol-10-yl)(4-methoxyphenyl)methanone (3w). Yellow solid; yield 65%; Mp 184–186 °C; 1H-NMR (CDCl3, 400 MHz) δ 8.60 (br, 1H, NH), 8.42 (br, 1H, NH), 7.70–7.68 (m, 1H, ArH), 7.64–7.61 (m, 2H, ArH), 7.35–7.33 (m, 1H, ArH), 7.01–6.94 (m, 5H, ArH), 6.70 (s, 1H, ArH), 5.08 (br, 1H, OH), 3.98–3.94 (m, 2H, CH2N), 3.88 (s, 3H, OCH3), 3.80 (s, 3H, OCH3), 3.58–3.56 (m, 2H, NCH2), 2.27–2.23 (m, 2H, CH2); 13C-NMR (CDCl3, 100 MHz) δ 188.8, 161.0, 154.8, 153.6, 148.9, 137.8, 134.7, 131.5, 129.4, 129.4, 126.5, 123.9, 118.8, 116.3, 113.6, 113.5, 112.3, 108.7, 107.6, 105.3, 105.1, 101.1, 95.8, 55.9, 55.3, 39.2, 38.0, 20.7; IR (KBr) 3430, 2921, 2852, 1724, 1612, 1557, 1528, 1450, 1340, 1250, 1170, 750 cm−1; HRMS (ESI-TOF): m/z calcd for C28H26N3O4 [M + H]+, 468.1918; found, 468.1922.
4. Conclusions
In summary, we have successfully developed a facile, economical, and environmentally friendly method for the construction of highly functionalized 3,7′-bisindole derivatives via a Michael addition/cyclocondensation reaction. This allowed for the rapid construction of a novel library of highly substituted 3,7′-bisindole derivatives through the simple and easy raw material HKAs 1 and 2-(1H-indol-3-yl)cyclohexa-2,5-diene-1,4-dione derivatives 2. Our further investigations into the in vitro biological activities of compounds 3 are currently ongoing.
Supplementary Materials
Supplementary materials can be accessed at: http://www.mdpi.com/1420-3049/21/5/638/s1.
Acknowledgments
This work was supported by the Program for Changjiang Scholars and Innovative Research Team in University (IRT13095), the Program for the National Natural Science Foundation of China (Nos. U1202221, 21362042, 21262042, 21162037 and 21402070), the Talent Found in Yunnan Province (2012HB001), Excellent Young Talents, Yunnan University (XT412003), High-Level Talents Introduction Plan of Yunnan Province (CB143001), and Yang Fan Innovative & Entepreneurial Research Team Project (No. 201312S09).
Author Contributions
The list authors contributed to this work as follows: Sheng-Jiao Yan and Jun Lin conceived and designed the study. Teng Liu, Hong-You Zhu and Da-Yun Luo performed the experiments. Teng Liu wrote the paper. Sheng-Jiao Yan and Jun Lin edited and revised the manuscript.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Fang, E.; Potts, B.C.M.; Faulkner, D.J. 6-Bromotryptamine Derivatives from the Gulf of California Tunicate Didemnum Candidum. J. Nat. Prod. 1991, 54, 564–569. [Google Scholar]
- Ells, R.; Carmeli, H.; Vibrindole, A. A Metabolite of the Marine Bacterium, Vzbrzo Parahaemolytzcus, Isolated from the Mucus of the Boxfish Ostraczon Cubzcus. J. Nat. Prod. 1994, 57, 1587–1590. [Google Scholar]
- Bifulco, G.; Bruno, I.; Riccio, R. Further Brominated Bis- and Tris-Indole Alkaloids from the Deep-Water New Caledonian Marine Sponge Orzna Sp. J. Nat. Prod. 1995, 58, 1254–1260. [Google Scholar] [CrossRef] [PubMed]
- Garbe, T.R.; Kobayashi, M.; Shimizu, N.; Takesue, N.; Ozawa, M.; Yukawa, H. Indolyl Carboxylic Acids by Condensation of Indoles with α-Keto Acids. J. Nat. Prod. 2000, 63, 596–598. [Google Scholar] [CrossRef] [PubMed]
- Ozkazanc, H. Characterization and Charge Transfer Mechanism of PIN–CdSe Nanocomposites. Polym. Compos. 2015. [Google Scholar] [CrossRef]
- Sazou, D. The dynamical behavior of the electrochemical polymerization of indole on Fe in acetonitrile–water mixtures. Synth. Met. 2002, 130, 45–54. [Google Scholar] [CrossRef]
- Ismail, A.A.; Sanad, S.H.; El-Meligi, A.A. Inhibiting effect of indole and some of its derivatives on corrosion of C-steel in HCl. J. Mater. Sci. Technol. 2000, 16, 397–400. [Google Scholar]
- Dudukcu, M.; Yazici, B.; Erbil, M. The effect of indole on the corrosion behaviour of stainless steel. Mater. Chem Phys. 2004, 87, 138–141. [Google Scholar] [CrossRef]
- Zoraghi, R.; Worrall, L.; See, R.H.; Strangman, W.; Popplewell, W.L.; Gong, H.; Samaai, T.; Swayze, R.D.; Kaur, S.; Vuckovic, M.; et al. Methicillin-resistant Staphylococcus aureus (MRSA) pyruvate kinase as a target for bis-indole alkaloids with antibacterial activities. J. Biol. Chem. 2011, 286, 44716–44725. [Google Scholar] [CrossRef] [PubMed]
- Veale, C.G.L.; Zoraghi, R.; Young, R.M.; Morrison, J.P.; Pretheeban, M.; Lobb, K.A.; Reiner, N.E.; Andersen, R.J.; Davies-Coleman, M.T. Synthetic Analogues of the Marine Bisindole Deoxytopsentin: Potent Selective Inhibitors of MRSA Pyruvate Kinase. J. Nat. Prod. 2015, 78, 355–362. [Google Scholar] [CrossRef] [PubMed]
- Miyake, F.Y.; Yakushijin, K.; Horne, D.A. Synthesis of Marine Sponge Bisindole Alkaloids Dihydrohamacanthins. Org. Lett. 2002, 4, 941–943. [Google Scholar] [CrossRef] [PubMed]
- Stevenson, R.J.; Denny, W.A.; Tercel, M.; Pruijn, F.B.; Ashoorzadeh, A. Nitro seco Analogues of the Duocarmycins Containing Sulfonate Leaving Groups as Hypoxia-Activated Prodrugs for Cancer Therapy. J. Med. Chem. 2012, 55, 2780–2802. [Google Scholar] [CrossRef] [PubMed]
- Gan, C.Y.; Etoh, T.; Hayashi, M.; Komiyama, K.; Kam, T.S. Leucoridines A−D, Cytotoxic Strychnos−Strychnos Bisindole Alkaloids from Leuconotis. J. Nat. Prod. 2010, 73, 1107–1111. [Google Scholar] [CrossRef] [PubMed]
- Bao, B.; Sun, Q.; Yao, X.; Hong, J.; Lee, C.O.; Sim, C.J.; Im, K.S.; Jung, J.H. Cytotoxic Bisindole Alkaloids from a Marine Sponge Spongosorites sp. J. Nat. Prod. 2005, 68, 711–715. [Google Scholar] [CrossRef] [PubMed]
- Chakraborty, D.P.; Roy, S. Carbazole Alkaloids III. Prog. Chem. Org. Nat. Prod. 1991, 57, 71–152. [Google Scholar]
- Qu, J.; Fang, L.; Ren, X.D.; Lin, Y.B.; Yu, S.S.; Li, L.; Bao, X.Q.; Zhang, D.; Li, Y.; Ma, S.G. Bisindole Alkaloids with Neural Anti-inflammatory Activity from Gelsemium elegans. J. Nat. Prod. 2013, 76, 2203–2209. [Google Scholar] [CrossRef] [PubMed]
- Mcarthur, K.A.; Mitchell, S.S.; Tsueng, G.; Rheingold, A.; White, D.J.; Grodberg, J.; Lam, K.S.; Potts, B.C.M. Lynamicins A−E, Chlorinated Bisindole Pyrrole Antibiotics from a Novel Marine Actinomycete. J. Nat. Prod. 2008, 71, 1732–1737. [Google Scholar] [CrossRef] [PubMed]
- Hirasawa, Y.; Hara, M.; Nugroho, A.E.; Sugai, M.; Zaima, K.; Kawahara, N.; Goda, Y.; Awang, K.; Hadi, A.H.A.; Litaudon, M.; et al. Bisnicalaterines B and C, Atropisomeric Bisindole Alkaloids from Hunteria zeylanica, Showing Vasorelaxant Activity. J. Org. Chem. 2010, 75, 4218–4223. [Google Scholar] [CrossRef] [PubMed]
- Yadav, J.S.B.; Reddy, V.S.; Sunitha, S. Efficient and Eco-Friendly Process for the Synthesis of Bis(1H-indol-3-yl)methanes using Ionic Liquids. Adv. Synth. Catal. 2003, 345, 349–352. [Google Scholar] [CrossRef]
- Gibbs, T.J.K.; Tomkinson, N.C.O. Aminocatalytic preparation of bisindolylalkanes. Org. Biomol. Chem. 2005, 3, 4043–4045. [Google Scholar] [CrossRef] [PubMed]
- Miyake, F.Y.; Yakushijin, K.; Horne, D.A. A Facile Synthesis of Dragmacidin B and 2,5-Bis(6’-bromo-3’-indolyl)piperazine. Org. Lett. 2000, 2, 3185–3187. [Google Scholar] [CrossRef] [PubMed]
- Chakrabarty, M.; Basak, R.; Ghosh, N.; Harigaya, Y. Michael reaction of indoles with 3-(2’-nitrovinyl)indole under solvent-free conditions and in solution. An efficient synthesis of 2,2-bis(indolyl)nitroethanes and studies on their reduction. Tetrahedron 2004, 60, 1941–1949. [Google Scholar] [CrossRef]
- Ma, S.; Yu, S. Sc(OTf)3-Catalyzed Indolylation of 1,2-Allenic Ketones: Controlled Highly Selective Synthesis of β-Indolyl-α,β-unsaturetated (E)-Enones and β,β-Bisindolyl Ketones. Org. Lett. 2005, 7, 5063–5065. [Google Scholar] [CrossRef] [PubMed]
- Nair, V.; Abhilash, K.G.; Vidya, N. Practical Synthesis of Triaryl- and Triheteroarylmethanes by Reaction of Aldehydes and Activated Arenes Promoted by Gold(III) Chloride. Org. Lett. 2005, 7, 5857–5859. [Google Scholar] [CrossRef] [PubMed]
- Li, H.; Wang, Y.Q.; Deng, L. Enantioselective Friedel-Crafts Reaction of Indoles with Carbonyl Compounds Catalyzed by Bifunctional Cinchona Alkaloids. Org. Lett. 2006, 8, 4063–4065. [Google Scholar] [CrossRef] [PubMed]
- Shirakawa, S.; Kobayashi, S. Carboxylic Acid Catalyzed Three-Component Aza-Friedel-Crafts Reactions in Water for the Synthesis of 3-Substituted Indoles. Org. Lett. 2006, 8, 4939–4942. [Google Scholar] [CrossRef] [PubMed]
- Lucarini, S.; Mari, M.; Piersanti, G.; Spadoni, G. Organocatalyzed coupling of indoles with dehydroalanine esters: Synthesis of bis(indolyl)propanoates and indolacrylates. RSC Adv. 2013, 3, 19135–19143. [Google Scholar] [CrossRef]
- Xu, H.Y.; Zi, Y.; Xu, X.P.; Wang, S.Y.; Ji, S.J. TFA-catalyzed CeN bond activation of enamides with indoles: Efficient synthesis of 3,3-bisindolylpropanoates and other bisindolylalkanes. Tetrahedron 2013, 69, 1600–1605. [Google Scholar] [CrossRef]
- Mari, M.; Tassoni, A.; Lucarini, S.; Fanelli, M.; Piersanti, G.; Spadoni, G. Brønsted Acid Catalyzed Bisindolization of α-Amido Acetals: Synthesis and Anticancer Activity of Bis(indolyl)ethanamino Derivatives. Eur. J. Org. Chem. 2014, 2014, 3822–3830. [Google Scholar] [CrossRef]
- Xie, Z.B.; Sun, D.Z.; Jiang, G.F.; Le, Z.G. Facile Synthesis of Bis(indolyl)methanes Catalyzed by α-Chymotrypsin. Molecules 2014, 19, 19665–19677. [Google Scholar] [CrossRef] [PubMed]
- Huang, Z.T.; Wang, M.X. Heterocyclic Ketene Aminals. Heterocycles 1994, 37, 1233–1262. [Google Scholar] [CrossRef]
- Wang, K.M.; Yan, S.J.; Lin, J. Heterocyclic Ketene Aminals: Scaffolds for Heterocycle Molecular Diversity. Eur. J. Org. Chem. 2014, 6, 1129–1145. [Google Scholar] [CrossRef]
- Yang, L.F. Heterocyclic Ketene Aminals. Synlett 2014, 25, 2964–2965. [Google Scholar] [CrossRef] [Green Version]
- Yu, F.; Yan, S.; Hu, L.; Wang, Y.; Lin, J. Cascade Reaction of Isatins with Heterocyclic Ketene Aminals: Synthesis of Imidazopyrroloquinoline Derivatives. Org. Lett. 2011, 13, 4782–4785. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.C.; Liu, Z.C.; Yang, R.; Zhang, J.H.; Yan, S.J.; Lin, J. Regioselective construction of 1,3-diazaheterocycle fused [1,2-a][1,8]naphthyridine derivatives via cascade reaction of quinolines with heterocyclic ketene aminals: A joint experimental-computational approach. Org. Biomol. Chem. 2013, 11, 7276–7288. [Google Scholar] [CrossRef] [PubMed]
- Li, M.; Shao, P.; Wang, S.W.; Kong, W.; Wen, L.R. Four-Component Cascade Heteroannulation of Heterocyclic Ketene Aminals: Synthesis of Functionalized Tetrahydroimidazo[1,2-a]pyridine Derivatives. J. Org. Chem. 2012, 77, 8956–8967. [Google Scholar] [CrossRef] [PubMed]
- Wen, L.R.; Sun, Q.C.; Zhang, H.L.; Li, M. A new rapid multicomponent domino heteroannulation of heterocyclic ketene aminals: Solvent-free regioselective synthesis of functionalized benzo[g]imidazo[1,2-a]quinolinediones. Org. Biomol. Chem. 2013, 11, 781–786. [Google Scholar] [CrossRef] [PubMed]
- Yan, S.; Chen, Y.; Liu, L.; He, N.; Lin, J. Three-component solvent-free synthesis of highly substituted bicyclic pyridines containing a ring-junction nitrogen. Green Chem. 2010, 12, 2043–2052. [Google Scholar] [CrossRef]
- Yaqub, M.; Arif, N.; Perveen, R.; Batool1, J.; Riaz, M.T.; Yaseen, M. One-Pot Three Component Cascade Synthesis of Fused Ring Quinazoline-2,4-dione Derivatives Employing Heterocyclic Ketene Aminals as a Versatile Synthone. Asian J. Chem. 2015, 27, 1013–1018. [Google Scholar] [CrossRef]
- Xu, W.Y.; Jia, Y.M.; Yang, J.K.; Huang, Z.T.; Yu, C.Y. Reactions of heterocyclic ketene aminals with 2-[3-oxoisobenzofuran-1(3H)-ylidene]malononitrile: Synthesis of novel polyfunctionalized 1,4-dihydropyridine-fused 1,3-diazaheterocycles. Synlett 2010, 11, 1682–1684. [Google Scholar] [CrossRef]
- Ma, Y.L.; Wang, K.M.; Lin, X.R.; Yan, S.J.; Lin, J. Three-component cascade reaction synthesis of polycyclic 1,4-dihydropyridine derivatives in water. Tetrahedron 2014, 70, 6578–6584. [Google Scholar] [CrossRef]
- Zhu, D.D.; Chen, X.B.; Huang, R.; Yan, S.J.; Lin, J. Three-component solvent-free synthesis of fluorine substituted bicyclic pyridines. Tetrahedron 2015, 71, 2363–2368. [Google Scholar] [CrossRef]
- Chen, N.; Meng, X.; Zhu, F.; Cheng, J.; Shao, X.; Li, Z. Tetrahydroindeno[1′,2′:4,5]pyrrolo[1,2-a] imidazol-5(1H)-ones as Novel Neonicotinoid Insecticides: Reaction Selectivity and Substituent Effects on the Activity Level. J. Agric. Food Chem. 2015, 63, 1360–1369. [Google Scholar] [CrossRef] [PubMed]
- Chen, X.B.; Liu, Z.C.; Yang, L.F.; Yan, S.J.; Lin, J. A Three-Component Catalyst-Free Approach to Regioselective Synthesis of Dual Highly Functionalized Fused Pyrrole Derivatives in Water-Ethanol Media: Thermodynamics versus Kinetics. ACS Sustain. Chem. Eng. 2014, 2, 1155–1163. [Google Scholar] [CrossRef]
- Shao, X.; Fu, H.; Xu, X.; Xu, X.; Liu, Z.; Li, Z.; Qian, X. Divalent and Oxabridged Neonicotinoids Constructed by Dialdehydes and Nitromethylene Analogues of Imidacloprid: Design, Synthesis, Crystal Structure, and Insecticidal Activities. J. Agric. Food Chem. 2010, 58, 2696–2702. [Google Scholar] [CrossRef] [PubMed]
- Chen, X.B.; Liu, Z.C.; Lin, X.R.; Huang, R.; Yan, S.J.; Lin, J. Highly Diastereoselective Convergent Synthesis of Polycyclic Pyrroles with Consecutive Quaternary Stereocenters: Cascade Construction of Multiple C–C and C–Hetero Bonds. ACS Sustain. Chem. Eng. 2014, 2, 2391–2398. [Google Scholar] [CrossRef]
- Chen, X.B.; Wang, X.Y.; Zhu, D.D.; Yan, S.J.; Lin, J. Three-component domino reaction synthesis of highly functionalized bicyclic pyrrole derivatives. Tetrahedron 2014, 70, 1047–1054. [Google Scholar]
- Yu, F.C.; Huang, R.; Ni, X.C.; Fan, J.; Yan, S.J.; Lin, J. Three-component stereoselective synthesis of spirooxindole derivatives. Green Chem. 2013, 15, 453–462. [Google Scholar] [CrossRef]
- Chen, X.B.; Liu, X.M.; Huang, R.; Yan, S.J.; Lin, J. Three-Component Synthesis of Indanone-Fused Spirooxindole Derivatives. Eur. J. Org. Chem. 2013, 2013, 4607–4613. [Google Scholar] [CrossRef]
- Yang, L.J.; Yan, S.J.; Chen, W.; Lin, J. A Facile Route to 1,3-Diazaheterocycle-Fused [1,2-a]Indole Derivatives via Acetic Acid Catalyzed Cyclocondensation Reactions. Synthesis 2010, 20, 3536–3544. [Google Scholar] [CrossRef]
- Zhou, B.; Liu, Z.C.; Qu, W.W.; Yang, R.; Lin, X.R.; Yan, S.J.; Lin, J. An environmentally benign, mild, and catalyst-free reaction of quinones with heterocyclic ketene aminals in ethanol: Site-selective synthesis of rarely fused [1,2-a]indolone derivatives via an unexpected anti-Nenitzescu strategy. Green Chem. 2014, 16, 4359–4370. [Google Scholar] [CrossRef]
- Yu, F.C.; Hao, X.P.; Lin, X.R.; Yan, S.J.; Lin, J. Synthesis of fused polyhalogeno-7a-hydroxy-[1,2-a]indol -5-one derivatives. Tetrahedron 2015, 71, 4084–4089. [Google Scholar] [CrossRef]
- Huang, Z.T.; Wang, M.X. A New Route to 3H-1,5-Benzodiazepines and Heterocylic Ketene Aminals from Benzoyl Substituted Ketene Dithioacetals and Diamines. Synthesis 1992, 12, 1273–1276. [Google Scholar] [CrossRef]
- Li, Z.J.; Charles, D. The Synthesis of fluoroheterocyclic ketene aminals. Synth. Commun. 2001, 31, 527–533. [Google Scholar] [CrossRef]
- Zhang, H.B.; Liu, L.; Chen, Y.J.; Wang, D.; Li, C.J. “On Water”-Promoted Direct Coupling of Indoles with 1,4-Benzoquinones without Catalyst. Eur. J. Org. Chem. 2006, 2006, 869–873. [Google Scholar] [CrossRef]
- Niu, F.; Liu, C.C.; Cui, Z.M.; Zhai, J.; Jiang, L.; Song, W.G. Promotion of organic reactions by interfacial hydrogen bonds on hydroxyl group rich nano-solids. Chem. Commun. 2008, 24, 2803–2805. [Google Scholar] [CrossRef] [PubMed]
- Sample Availability: Samples of the compounds 3a–3w are available from the authors.
© 2016 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license ( http://creativecommons.org/licenses/by/4.0/).